| Taxon treatments | Taxon names |
|
Turn highlighting On/Off |







(C) 2012 Yves Bousquet. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
All scientific names of Trachypachidae, Rhysodidae, and Carabidae (including cicindelines) recorded from America north of Mexico are catalogued. Available species-group names are listed in their original combinations with the author(s), year of publication, page citation, type locality, location of the name-bearing type, and etymology for many patronymic names. In addition, the reference in which a given species-group name is first synonymized is recorded for invalid taxa. Genus-group names are listed with the author(s), year of publication, page citation, type species with way of fixation, and etymology for most. The reference in which a given genus-group name is first synonymized is recorded for many invalid taxa. Family-group names are listed with the author(s), year of publication, page citation, and type genus. The geographical distribution of all species-group taxa is briefly summarized and their state and province records are indicated.
One new genus-group taxon, Randallius new subgenus (type species: Chlaenius purpuricollis Randall, 1838), one new replacement name, Pterostichus amadeus new name for Pterostichus vexatus Bousquet, 1985, and three changes in precedence, Ellipsoptera rubicunda (Harris, 1911) for Ellipsoptera marutha (Dow, 1911), Badister micans LeConte, 1844 for Badister ocularis Casey, 1920, and Agonum deplanatum Ménétriés, 1843 for Agonum fallianum (Leng, 1919), are proposed. Five new genus-group synonymies and 65 new species-group synonymies, one new species-group status, and 12 new combinations (see Appendix 5) are established.
The work also includes a discussion of the notable private North American carabid collections, a synopsis of all extant world geadephagan tribes and subfamilies, a brief faunistic assessment of the fauna, a list of valid species-group taxa, a list of North American fossil Geadephaga (Appendix 1), a list of North American Geadephaga larvae described or illustrated (Appendix 2), a list of Geadephaga species described from specimens mislabeled as from North America (Appendix 3), a list of unavailable Geadephaga names listed from North America (Appendix 4), a list of nomenclatural acts included in this catalogue (Appendix 5), a complete bibliography with indication of the dates of publication in addition to the year, and indices of personal names, supraspecific names, and species-group names.
Ground beetles, Trachypachidae, Rhysodidae, Carabidae, North America
The Adephaga, a name coined by the Swiss entomologist and botanist Joseph Philippe de Clairville [1742-1830] in 1806, represents the second largest suborder of Coleoptera with an estimated 39, 300 species described to 2005. The group is undisputedly natural, based on the presence of several synapomorphies in the adult and immature stages (Beutel and Ribera 2005: 53; Beutel et al. 2008; Lawrence et al. 2011). The term Adephaga comes from the Greek word adephagos meaning gluttonous, greedy, in reference to the predaceous habits of adults and larvae of the vast majority of the species. Conventionally the Adephaga are divided into two groups, the Geadephaga for the terrestrial families and the Hydradephaga for the aquatic families.
The extant hydradephagan families include the Gyrinidae (about 875 species), Haliplidae (about 220 species), Noteridae (about 250 species), Amphizoidae (five species), Hygrobiidae (six species), Dytiscidae (about 3, 700 species), Aspidytidae (two species), and Meruidae (one species). Some studies, based on structural features of the adult (Burmeister 1976; Baehr 1979) and larva (Ruhnau 1986) as well as molecular data (Shull et al. 2001; Ribera et al. 2002; Hunt et al. 2007), suggest that the Hydradephaga is monophyletic. Other studies, including recent DNA sequence analyses (Maddison et al. 2009), indicate a polyphyletic origin for the complex.
The extant geadephagan groups include the trachypachids (six species), rhysodids (about 355 species), cicindelids (about 2, 415 species), and carabids (about 31, 490 species). The monophyletic origin of the Geadephaga was supported in some structural and molecular studies but rejected in others (see Maddison et al. 2009 for an overview). While the taxonomic concept of the hydradephagan families is stable, that of the geadephagan families is not. Several authors consider either the trachypachids, rhysodids, or cicindelids as Carabidae.
This work catalogues all geadephagan taxa of America, north of Mexico. The last catalogue covering the Geadephaga of the region is that of Bousquet and Larochelle in 1993. Since then relatively few taxonomic studies have been published on the North American fauna. The increased interest toward the inadequately known but amazingly rich Neotropical Region is probably one of the reasons behind the situation. So, is there a need for this catalogue? For one, it is more informative than the previous one. It includes, besides the usual information on nomenclature, the type locality of each available species, locations of the primary type specimens, references to the original synonymies of invalid names, and a short description of the geographical distribution of each species. Furthermore, a number of errors were discovered in the previous catalogue and needed to be corrected.
Brief historyThe first checklist / catalogue covering the North American Geadephaga was the checklist of beetles of the United States by Friedrich Ernst Melsheimer [1784-1873] published in July 1853. The interest for this work originated with the establishment in 1842 of the first entomological society in America, The Entomological Society of Pennsylvania. The compilation of this list was one of the main objects of the Society (Sorensen 1995: 17) and it prevailed upon Melsheimer, the first and only President of the Society, to complete the task. The manuscript was delivered in 1848 to the Smithsonian Institution in Washington. Its secretary, Joseph Henry, asked Samuel S. Haldeman and John L. LeConte to advise on its publication. The two gentlemen volunteered to update the manuscript, which delayed its release considerably. The work was a straightforward list of valid species, with abridged references and synonyms but without distributional data, arranged under the valid generic names. Although limited to the United States, it included more than 90% of the species known from North America at the time. Melsheimer, a physician by profession, was the son of Frederick Valentine Melsheimer [1749-1814] who in 1806 published the first book on American entomology, a 60-page booklet entitled “Catalogue of insects of Pennsylvania. Part first.” It enumerates 111 genera and 1, 363 species of Coleoptera (Meisel 1929: 367), though almost all of them are nomina nuda.
In April 1866, John Lawrence LeConte [1825-1883] published the first part of a checklist of the Coleoptera of North America (north of Mexico) for the Smithsonian Institution. It covered the Adephaga and a large section of the Polyphaga. The first 49 pages, which included the Adephaga, were reprinted with minor modifications from a list already issued in March 1863. The list included synonyms but no geographical information. The second part of the checklist, covering the Chrysomeloidea and Curculionoidea, was never published. Two additional checklists of North American beetles would be published in the United States during the xix Century, both straightforward lists without geographical data. The first one, issued in 1874, was authored by George Robert Crotch [1842-1874], a British coleopterist who at the time was assistant to Hermann Hagen at the Museum of Comparative Zoology. A supplement to Crotch’s checklist was authored in 1880 by Edward Payson Austin, an amateur coleopterist and member of the Cambridge Entomological Club in its early years. The second checklist was published in 1885 by Samuel Henshaw [1852-1941], then assistant to Professor Hyatt at Lowell Technological Institute. Three supplements, in 1887, 1889, and 1895, were later issued by Henshaw.
In Europe, the German Max Gemminger [1820-1887] and Freiherr Edgar von Harold [1830-1886] published, between 1868 and 1876, a checklist of beetles of the world in 12 volumes, compiling 77, 008 species over 3, 800 pages. The Geadephaga were included in the first (Carabidae including cicindelids and trachypachids), second (paussids on pages 700-706), and third volumes (rhysodids on pages 867-868), all issued in 1868. Along with each specific name the authors listed the publication year as well as the original reference and region(s) of capture. This work spurred a large number of additions and corrections by many coleopterists. It stood alone in its class until the publication of the Coleopterorum Catalogus under the editorship of Walther Junk and Sigmund Schenkling. Published between 1909 and 1940, this catalogue was issued in 170 parts forming 30 volumes and involved the participation of more than 60 entomologists. A list by parts and another by families can be found in Blackwelder (1957: 1022-1034). The Geadephaga were covered in parts 1 (Rhysodidae by Raffaello Gestro in 1910), 5 (Paussinae by R. Gestro in 1910), 86 (Cicindelinae by Walther Horn in 1926), 91, 92, 97, 98, 104, 112, 115, 121, 124, 126, and 127 (Carabidae, including trachypachids, by Ernst Csiki between 1927 and 1933). Second editions of the Rhysodidae, by Walter D. Hincks in 1950, and Paussinae, by Emile Janssens in 1953, were issued much later.
While the Coleopterorum Catalogus was being published in Berlin, Charles William Leng [1859-1941], then director of the museum at the Staten Island Institute of Arts and Sciences, released in 1920 his catalogue of the Coleoptera of America, north of Mexico, still known as the “Leng catalogue.” His goal was “to enumerate systematically all the species of Coleoptera described prior to January 1, 1919 ... with consecutive numbers, synonyms, citation of original description, and an indication of distribution.” Leng and Andrew J. Mutchler in 1927 (covering the years 1919-1924) and 1933 (for 1925-1932), Richard E. Blackwelder in 1939 (for 1933-1938), and Blackwelder and his wife, Ruth M. Blackwelder, in 1948 (for 1939-1947) published supplements to Leng’s catalogue.
In 1972, Ross H. Arnett, Jr. [1919-1999], the catalyst behind the birth of the Coleopterist’s Society and its journal The Coleopterists Bulletin, initiated the “North American beetle fauna project” (NABF) with the help of a small group of coleopterists. The main goal of this cooperative adventure was to “produce a series of manuals for the identification of the species of beetles of the United States and adjacent Canada, and adjacent Mexico.” Although no such book was ever published, a preliminary checklist of North American beetles, known as the “Red Version, ” was compiled by 1976 by Richard E. Blackwelder and Arnett. This version was used as a “working copy” for the next one, the “Yellow Version” defined as the “definitive checklist and the one which will be kept up-to-date.” Of this version, only two families would be compiled and published (July 1977), the Cupedidae by Arnett and the Carabidae (including trachypachids but excluding cicindelids) by Terry L. Erwin, Donald R. Whitehead, and George E. Ball. The “Red Version” was reissued with modifications in 1983 under the editorship of Arnett.
In November 1978, the Science and Educational Administration, USDA, released its first fascicle, covering the family Heteroceridae, of “A catalogue of the Coleoptera of America north of Mexico.” The goal was to “supplant the Leng catalogue and supply additional essential information.” A total of 34 fascicles, treating various family-group taxa, would be published up to February 1997. Among the fascicles, one only, the Rhysodidae by Ross T. Bell in 1985, deals with Geadephaga.
In 1993, Bousquet and Larochelle published the first catalogue specifically devoted to the geadephagan beetles of North America. They listed, for the first time, the original combination of every available species-group taxon and provided a general idea of the distribution of each species by listing state and province records. One of the goals behind their work was to stimulate interest toward publication of distributional records as done regularly in Europe.
In 1998, Wolfgang Lorenz issued the first edition of his “Systematic list of extant ground beetles of the world” compiling 32, 567 species (in 1861 genera) of Geadephaga. Despite being limited to scientific names with their authors and publication years, the list soon became a useful tool to those interested in carabids. A second edition was released in 2005, compiling the same information for 34, 281 extant species, placed in 1929 genera.
The first catalogue of the world Coleoptera published is that of Schönherr issued in four parts, 1806, 1808, 1817 and 1826. The Carabidae were grouped in the following genera: Scarites (23 species), Cychrus (seven species), Manticora (two species), Carabus (340 species), Calosoma (12 species), Galerita (nine species), Brachinus (16 species), Anthia (27 species), Agra (three species), Collyris (four species), Odocantha [sic!] (seven species), Drypta (four species), Cicindela (67 species), Elaphrus (11 species), Scolytes [sic!] (three species), all included in the first volume (1806), and Paussus (ten species) and Cerapterus (two species) included in the third volume (1817). Overall 547 species of Geadephaga were listed along with references and synonyms. By comparison, the number of Carabidae (including Cicindelinae) listed in the four catalogue editions of the Dejean collection amounted to 104 (first edition, 1802), 908 (second edition, 1821), 2494 (third edition, 1833), and 2791 (fourth edition, 1836).
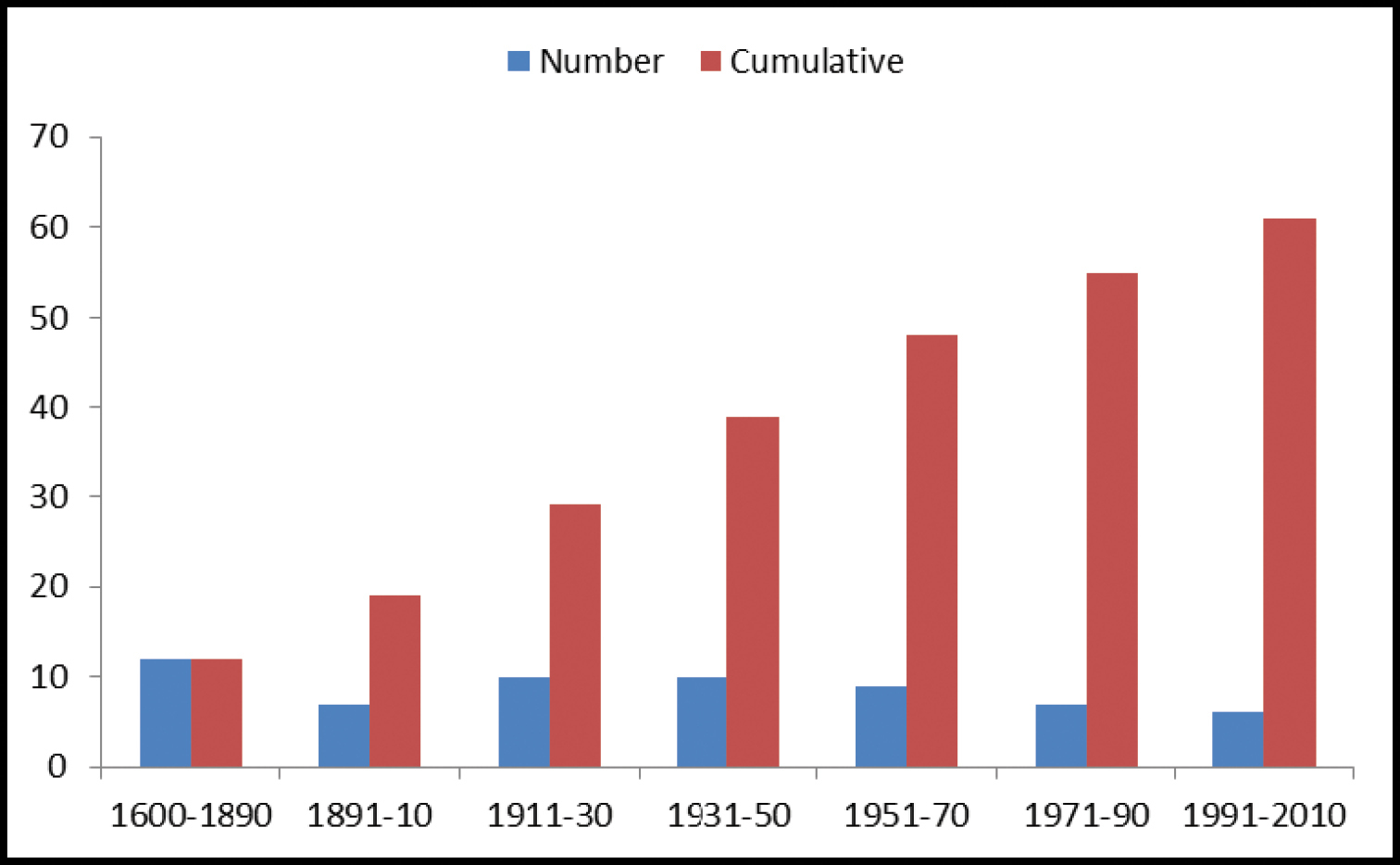
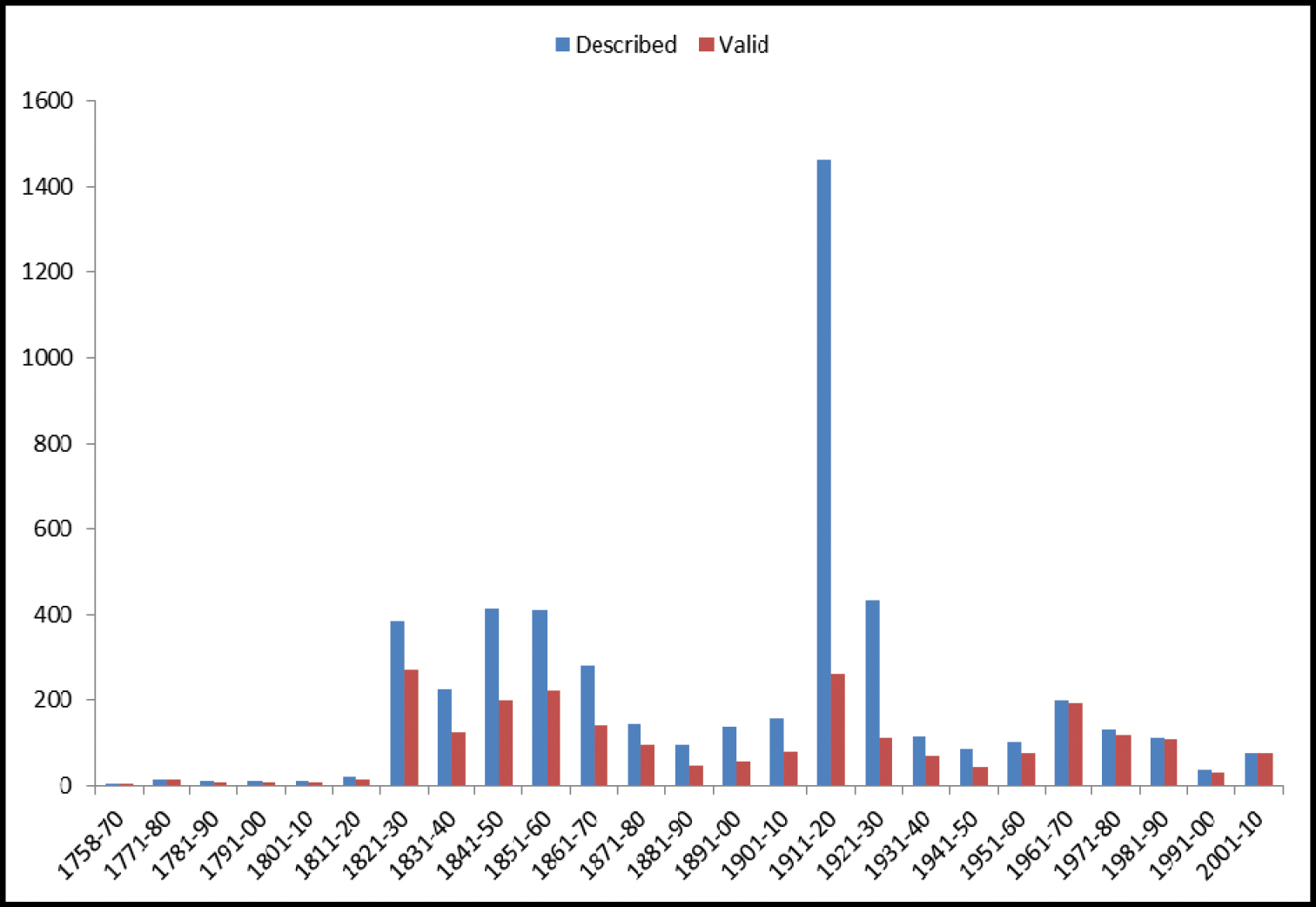
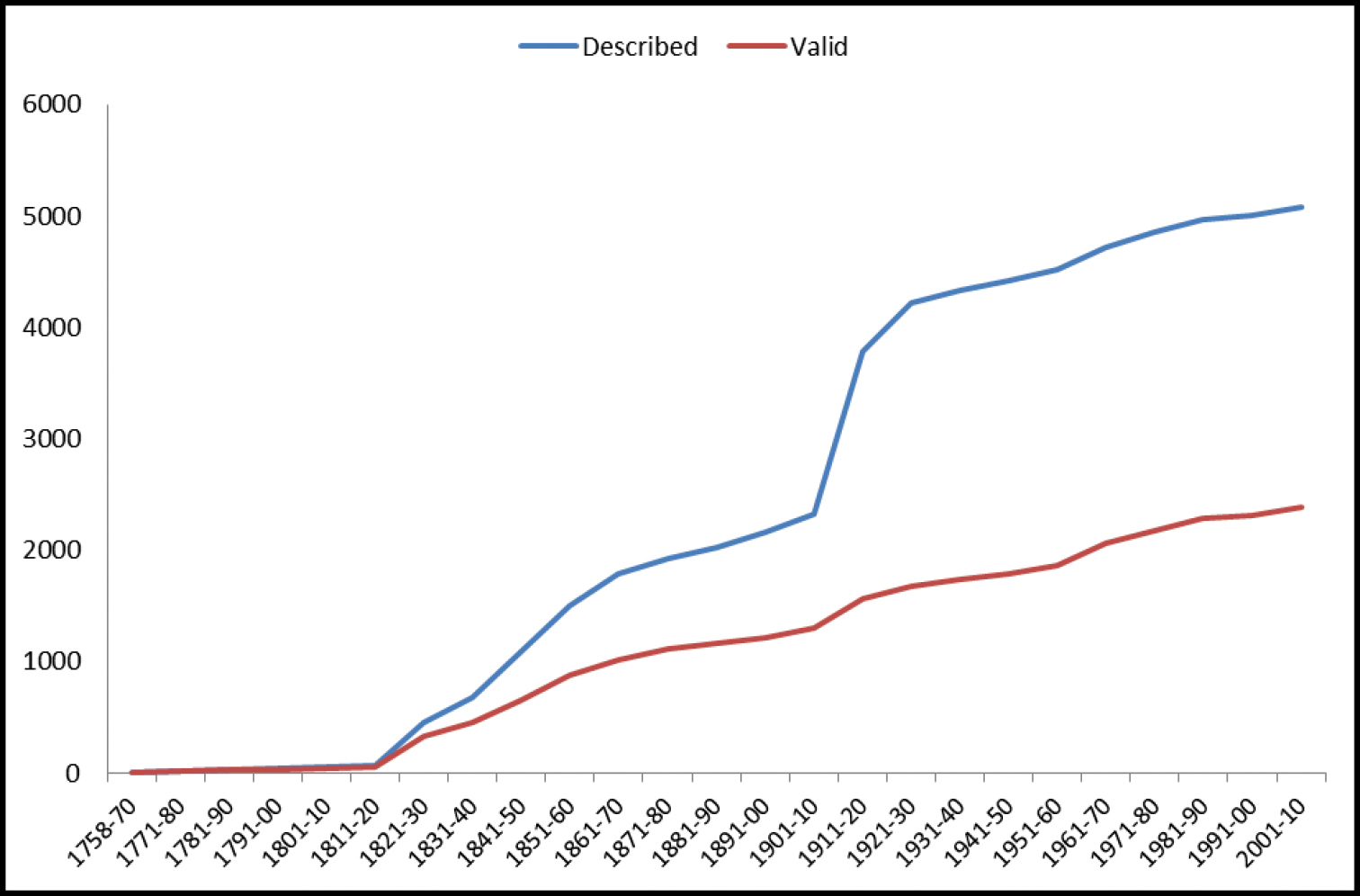
A comparison of the number of valid species and genera between this and previous checklists / catalogues is presented in Table 1.
North American Geadephaga species/genera counts in checklists.
| Publications | Trachyp | Rhysod | Cicindel | Carabid | Total |
|---|---|---|---|---|---|
| Melsheimer 1853 | 0 | 3/1 | 64/4 | 935/112 | 1002/117 |
| LeConte 1866 | 2/1 | 2/2 | 65/4 | 1090/107 | 1159/117 |
| Gemminger & Harold 1868 | 2/1 | 2/1 | 61/5 | 1167/124 | 1232/131 |
| Crotch 1874a | 2/1 | 2/2 | 67/4 | 1097/118 | 1168/125 |
| Henshaw 1885 | 2/1 | 4/2 | 70/4 | 1179/114 | 1255/121 |
| Leng 1920 | 2/1 | 4/2 | 114/4 | 2207/207 | 2327/214 |
| Coleopterorum Catalogus 1926-33 | 6/1 | 4/2 | 70/4 | 2916/144 | 2996/151 |
| Erwin et al. 1977 | 3/1 | 9/21 | 109/42 | 2308/169 | 2429/176 |
| Bousquet & Larochelle 1993 | 3/1 | 8/2 | 107/4 | 2230/183 | 2348/190 |
| Present catalogue | 3/1 | 8/2 | 112/12 | 2316/193 | 2439/208 |
1 Species count from Bell (1985b)
2 Species count from Boyd (1982)
The information on species-group taxa comprises a nomenclatural and a distributional component. The nomenclatural component consists of the scientific name with its author, date and page of publication, the type locality (see section Type locality under “Nomenclature” below), and the repository of the name-bearing type of each valid and invalid taxon. In addition, the reference in which a given scientific name is first synonymized is listed. Such references were difficult to find for several names, simply because they were never compiled before. Taxa listed as varieties subsequently to their original descriptions were not considered as listed in synonymy but those listed as aberrations or as “simple varieties” were. Codens used for collection repositories are given in the next section. When available, the accession numbers of name-bearing types for each institution are recorded.
This catalogue deals with extant available taxa. Fossil taxa are listed in Appendix 1. Unavailable names found in the literature are listed in Appendix 4 without comment. Listings of valid species-group names are alphabetic but listings of invalid names are chronologic. Synonyms of adventive and Holarctic species found in North America are selective. Misidentifications by subsequent authors are not listed. All species-group names are given in their original combinations.
The distributional component consists of a list of state and province records, using the same two-letter postal service style abbreviations used in the 1993 catalogue (Table 2), and a short description of the distribution, usually referring to the northeasternmost, northwesternmost, southwesternmost, and southeasternmost states or provinces. In addition, records for Cape Breton Island, the Queen Charlotte Islands, Vancouver Island, and the Channel Islands are indicated in parentheses after their respective provinces or states. Western Hemisphere countries are listed for species found south of the area covered. States and provinces placed in quotation marks in the descriptive section indicate that only the state or province was given without further precision in the reference cited. The starting point for the distributional records used in this work is Bousquet and Larochelle’s (1993) catalogue. However, many of their records were undocumented or came from old lists and were not always reliable. State and province records undocumented or considered doubtful are shown in square brackets following the accepted records. Except for the Amara records which come from identifications generally made by Fritz Hieke, almost all records from CMNH specimens are based on identifications made by Robert L. Davidson, those from LSAM specimens on identifications made by Igor Sokolov, and those from CNC, MCZ, and USNM specimens from identifications or confirmations made by myself. The records provided by Ken Karns and Brian Raber are based on identifications made by Robert L. Davidson.
Two-letter abbreviations for political regions covered by this catalogue.
| AB | Alberta | MA | Massachusetts | OH | Ohio |
| AK | Alaska | MB | Manitoba | OK | Oklahoma |
| AL | Alabama | MD | Maryland | ON | Ontario |
| AR | Arkansas | ME | Maine | OR | Oregon |
| AZ | Arizona | MI | Michigan | PA | Pennsylvania |
| BC | British Columbia | MN | Minnesota | PE | Prince Edward Island |
| CA | California | MO | Missouri | PM | St.Pierre and Miquelon |
| CO | Colorado | MS | Mississippi | QC | Quebec |
| CT | Connecticut | MT | Montana | RI | Rhode Island |
| DC | District of Columbia | NB | New Brunswick | SC | South Carolina |
| DE | Delaware | NC | North Carolina | SD | South Dakota |
| FL | Florida | ND | North Dakota | SK | Saskatchewan |
| GA | Georgia | NE | Nebraska | TN | Tennessee |
| GL | Greenland | NF | Newfoundland | TX | Texas |
| IA | Iowa | NH | New Hampshire | UT | Utah |
| ID | Idaho | NJ | New Jersey | VA | Virginia |
| IL | Illinois | NM | New Mexico | VT | Vermont |
| IN | Indiana | NS | Nova Scotia | WA | Washington |
| KS | Kansas | NT | Northwest Territories | WI | Wisconsin |
| KY | Kentucky | NU | Nunavut | WV | West Virginia |
| LA | Louisiana | NV | Nevada | WY | Wyoming |
| LB | Labrador | NY | New York | YT | Yukon Territory |
The information on supraspecific taxa consists of the scientific name with its author and date and page of publication. Type species of genus-group taxa are also given, in their original combinations, followed by the valid names in parentheses when applicable, and type genera are listed for family-group taxa. Etymology is given for all valid generic names and for some of the invalid names; the works of Brown (1956) and Cailleux and Komorn (1981) have been particularly useful.
The listing of valid supraspecific taxa is “phylogenetic, ” starting with taxa putatively branching off early along the evolutionary path of the group. Synonyms of supraspecific taxa are listed chronologically. If readily available, the first reference in which a given genus-group name is synonymized is included.
In the references section, titles of journals are cited in full. Titles of papers and books using alphabets other than Latin have been translated into English and the original language listed in square brackets after the title. An improvised title is given in square brackets, in the language used by the author(s), to papers without formal title. Unless otherwise noted, all references listed were seen. Except when only the year was found, the date of publication [DP] is given in square brackets at the end of each citation.
Institution / collection acronyms and abbreviationsCollections cited in the catalogue are referred to by the abbreviations listed below.
ALM Alabama Museum of Natural History, Tuscaloosa, Alabama, USA
AMNH American Museum of Natural History, New York, New York, USA
ANSP Academy of Natural Sciences, Philadelphia, Pennsylvania, USA
BMNH The Natural History Museum, London, United Kingdom
BYUC Brigham Young University, Provo, Utah, USA
CAS California Academy of Sciences, San Francisco, California, USA
CMC Cincinnati Museum of Natural History, Cincinnati, Ohio, USA
CMN Canadian Museum of Nature, Gatineau, Quebec, Canada
CMNH Carnegie Museum of Natural History, Pittsburgh, Pennsylvania, USA
CNC Canadian National Collection of Insects, Arachnids and Nematodes, Ottawa, Ontario, Canada
CUIC Cornell University Insect Collection, Cornell University, Ithaca, New York, USA
DAPC Darren A. Pollock collection, Eastern New Mexico University, Portales, New Mexico, USA
DEI Institute für Pfanzenschutzforschung (formerly Deutsches Entomologisches Institut), Kleinmachnow, Eberswalde, Germany
EMEC Essig Museum of Entomology Collection, University of California, Berkeley, California, USA
ETHZ Entomologisches Institut, Eidgenössische Technische Hochschule, Zürich, Switzerland
FFPC Foster Forbes Purrington collection, The Ohio State University, Columbus, Ohio, USA
FMNH Field Museum of Natural History, Chicago, Illinois, USA
FSCA Florida State Collection of Arthropods, Gainesville, Florida, USA
GNM Göteborgs Naturhistoriska Museum, Göteborg, Sweden
HMUG Hunterian Museum, University of Glasgow, Glasgow, United Kingdom
INHS Illinois Natural History Survey, Champaign (Urbana), Illinois, USA
IRSN Institut Royal des Sciences Naturelles, Brussels, Belgium
IZWP Museum and Institute of Zoology of the Polish Academy of Sciences, Warszawa, Poland
KSUC Kansas State University, Manhattan, Kansas, USA
LACM Los Angeles County Museum of Natural History, Los Angeles, California, USA
LMMC Lyman Entomological Museum, McGill University, Macdonald Campus, Sainte-Anne-de-Bellevue, Quebec, Canada
LSAM Louisiana State Arthropod Museum, Baton Rouge, Louisiana, USA
LSL Linnean Society, London, United Kingdom
MCZ Museum of Comparative Zoology, Harvard University, Cambridge, Massachusetts, USA
MHNG Muséum d’Histoire Naturelle, Geneva, Switzerland
MHNP Muséum National d’Histoire Naturelle, Paris, France
MSB Museum of Southwestern Biology, University of New Mexico, Albuquerque, New Mexico, USA
MSNG Museo Civico di Storia Naturale, Genoa, Italy
MSNM Museo Civico di Storia Naturale, Milano, Italy
MSNT Museo Civico di Storia Naturale, Trieste, Italy
MSUE Michigan State University, East Lansing, Michigan, USA
MVM Museum Victoria, Melbourne, Australia
NCSU North Carolina State University, Raleigh, North Carolina, USA
NHMW Naturhistorisches Museum Wien, Wien, Austria
NIAS National Institute for Agro-environmental Sciences, Tsukuba, Japan [formerly National Institute of Agricultural Sciences, Tokyo]
NMNS National Museum of Nature and Science, Tokyo, Japan
NMP National Museum, Prague, Czech Republic
NRSS Naturhistoriska Riksmuseet, Stockholm, Sweden
NSNH Nova Scotia Museum of Natural History, Halifax, Nova Scotia, Canada
ODAC Oregon Department of Agriculture, Plant Division, Salem, Oregon, USA
ORUM Collection Ouellet-Robert, Université de Montréal, Montreal, Quebec, Canada
OSAC Oregon State Arthropod Collection, Oregon State University, Corvallis, Oregon, USA
OSUO Ohio State University, Columbus, Ohio, USA
PMNH Peabody Museum of Natural History, Yale University, New Haven, Connecticut, USA
PURC Purdue State University, West Lafayette, Indiana, USA
SIM Staten Island Museum, Staten Island, New York, USA
SMEK Snow Museum of Entomology, University of Kansas, Lawrence, Kansas, USA
SMTD Staatliches Museum für Tierkunde, Dresden, Germany
TAMU Texas A&M University, College Station, Texas, USA
TMB Magyar Természettudományi Múzeum, Budapest, Hungary
TME Texas Museum of Entomology, Pipe Creek, Texas, USA
UAIC University of Arkansas, Fayetteville, Arkansas, USA
UASM Strickland Museum, University of Alberta, Edmonton, Alberta, Canada
UBC Spencer Entomological Museum, University of British Columbia, Vancouver, British Columbia, Canada
UCD University of California, Davis, California, USA
UCM University of Colorado Museum, Boulder, Colorado, USA
UICU University of Illinois, Urbana, Illinois, USA
UMAA University of Michigan, Ann Arbor, Michigan, USA
UMM Philipps-Universität Marburg, Zoologische Sammlung, Marburg, Germany
UMO The University Museum, University of Oxford, Oxford, United Kingdom
UMSP University of Minnesota, Saint Paul, Minnesota, USA
USMT Ueno Science Museum, Tokyo, Japan
USNM National Museum of Natural History, Smithsonian Institute, Washington, DC, USA
USS University of Sydney, Sydney, Australia
UZIU Uppsala Universitet, Zoologiska Museum, Uppsala, Sweden
VMNH Virginia Museum of Natural History, Martinsville, Virginia, USA
WSU Washington State University, Pullman, Washington, USA
ZILR Zoological Institute, Academy of Sciences, Saint Petersburg, Russia
ZMH Zoologiska Museum, University of Helsinki, Helsinki, Finland
ZMHB Zoologisches Museum, Humboldt Universität, Berlin, Germany
ZMLS Zoological Museum, Lund University, Lund, Sweden
ZMMU Zoological Museum, Moscow University, Moscow, Russia
ZMUA Zoologisch Museum, Universiteit van Amsterdam, Amsterdam, The Netherlands
ZMUC Zoologisk Museum, Universitets Copenhagen, Copenhagen, Denmark
ZMUO Zoological Museum, University of Oslo, Oslo, Norway
ZMUT Zoological Museum, University of Turku, Turku (= Åbo), Finland
Besides those used for provinces and states (see Table 2), the following abbreviations are used in the text:
B.P. Before Present
CAN Canada
CBI Cape Breton Island
CHI Channel Islands (Santa Barbara Islands)
DEN Denmark
DP Date of publication
FRA France
ICZN International Commission on Zoological Nomenclature
QCI Queen Charlotte Islands
USA United States of America
VCI Vancouver Island
In addition, the International Commission on Zoological Nomenclature is sometimes abridged to “Commission” and United States of America to “United States.”
Geographical termsFor simplicity, North America, north of Mexico, is referred to simply as North America in the text. Middle America refers to Mexico and the republics of Central America taken collectively. The West Indies refers to the Greater and Lesser Antilles and the Bahamas. The North American continent proper is referred to as North and Middle America.
For practical reasons, the zoogeographical regions of the world are defined following national boundaries as much as possible. The Nearctic Region corresponds to Canada, the continental United States, Saint-Pierre and Miquelon, and Greenland. Although the region extends into Mexico, its southern limit is difficult to define and often varies depending on the group under study. This concept implies that North America and the Nearctic Region are equivalent in this work. The Neotropical Region comprises Middle America and South America. The Afrotropical Region consists of Africa, including Madagascar and a number of smaller islands of the Indian Ocean, such as the Comoros, the Mascarene Islands, and the Seychelles, and of the Atlantic Ocean, such as Cape Verde Islands and São Tomé, but excludes the northern countries of Morocco (including Western Sahara), Algeria, Tunisia, Libya, and Egypt west of the Suez Canal, and the Canary and Madeira Islands. The limits of the Palaearctic Region are similar to those used in the Catalogue of Palaearctic Coleoptera (Löbl and Smetana 2003: 8). The region thus comprises Europe, Africa north of the Sahara, and Asia as far south as the Arabian Peninsula, Pakistan, Jammu and Kashmir, Himachal Pradesh, Uttar Pradesh, Nepal, Sikkim, Bhutan, Arunachal Pradesh, China, and Taiwan. The Oriental Region is Asia south of the regions used to define the southern limit of the Palaearctic Region. It includes all the Malay Archipelago (except New Guinea). The Australian Region comprises Australia, New Zealand, New Guinea, and some smaller islands of the Pacific, such as Fiji, New Britain, New Caledonia, and Solomon Islands.
The New World consists of the Nearctic, Neotropical, and Australian Regions combined and the Old World of the Oriental, Palaearctic, and Afrotropical Regions grouped. The Northern Hemisphere is the Nearctic and Palaearctic Regions combined and the Southern Hemisphere is the Afrotropical, Oriental, Australian, and Neotropical Regions united. The Western Hemisphere consists of the Nearctic and Neotropical Regions and the Eastern Hemisphere of the Palaearctic, Afrotropical, Oriental, and Australian Regions. Far East used in reference to the Palaearctic Region includes the Russian Far Eastern Region, the Korean Peninsula, Japan, Taiwan, and China excluding the Autonomous Regions of Inner Mongolia, Sinkian Uighur, and Tibet. Middle East is used for the southwestern Asian countries, including Egypt, Turkey, Syria, Lebanon, Israel, Jordan, Saudi Arabia, Yemen, Oman, Iraq, Iran, Afghanistan, and Pakistan.
The adjective “Holarctic” is used to denote a taxon that occurs naturally in both the Nearctic and Palaearctic Regions. The adjective “Australian” (as in “Australian species”) refers to the zoogeographical region, not to the country itself. The adjective “worldwide” is used to denote a genus-group or family-group taxon represented by at least one native species in all six zoogeographical regions as defined above including both the European and Asian parts of the Palaearctic Region. The adjective “endemic” indicates that the taxon is found only in the region listed.
Names of geographical places are given in their current English forms based on Merriam-Webster’s Geographical Dictionary, third edition (1997).
NomenclatureThe rules outlined in the fourth edition of the International Code of Zoological Nomenclatural, published in 1999, have been followed throughout this catalogue. The following are comments about some nomenclatural issues.
Principle of priority. Priority for identical taxa made available the same year, whether under the same name or not, is determined by the date, other than the year, of publication. If not specified in the work itself, the publication date is the earliest day or month on which the work is demonstrated to be in existence (ICZN 1999: Article 21.3). When both works are published or assumed to be published the same day, precedence is determined by the First Reviser (Article 24.2). Unless listed in the work itself, dates of publication besides the year can be demonstrated only for some works. Those without specific dates are listed as published the last day of the year (Article 21.3.2) and priority goes to the work with a “demonstrated” date of publication. However, the situation is subject to change with new bibliographic discoveries, which could challenge the validity of synonyms (as well as relative precedence of homonyms and validity of nomenclatural acts) and bring nomenclatural instability. In this catalogue, priority was given to the publication “in prevailing usage” when the dates of publication were determined from external sources.
New taxa. In the xviii and first half of the xix Century it was common practice for authors not to indicate the attribution of the new species-group taxa. Instead, some authors added the word mihi after the specific name, usually to indicate a taxon that the author, himself, was describing. Several collectors provided names for their specimens, even for undescribed ones, and these specimens often circulated among European coleopterists through exchange, gift, or sale. Many undescribed species were subsequently described or illustrated under the collector’s names by different authors. For these, citations are provided in this catalogue only to the first description or illustration of each species unless the term “new species” or an equivalent expression (such as an asterisk preceding the specific epithet as in Say 1823a
New taxa first published as synonyms. The International Commission on Zoological Nomenclature admits the availability of taxa first published in an available work as junior synonyms and adopted before 1961 as valid taxa or treated as senior homonyms (ICZN 1999: Article 11.6.1). In such cases the taxa date from their first publication as synonyms. Even though this ruling has existed since the publication of the ICZN first edition in 1960, it has rarely been enforced in the carabid literature. A few cases were found during the preparation of this catalogue. For example, Notiophilus sylvaticus has been credited in the past to Eschscholtz (1833: 24) but the name was first proposed as a junior synonym of Notiophilus biguttatus Fabricius by Dejean (1831: 589). The name is credited to Dejean (1831) in this catalogue. It is possible that other cases like this one will eventually be found.
Lectotype. Prior to 2000, a lectotype could be selected by using the term “the type” instead of “lectotype” (ICZN 1999: Article 74.5). The words “type” and “holotype” are also acceptable if the author unambiguously selects a particular syntype to act as the unique name-bearing type of the taxon. This is the case for almost all designations using the word “type” or “holotype” relating to North American Carabidae published after 1950, in particular by George E. Ball and his students. In this catalogue the expression “lectotype [as type]” or “lectotype [as holotype]” applies to such cases. Unfortunately the Commission does not mandate the addition of “lectotype” labels to selected specimens, which often creates ambiguity when authors fail to do so.
Type locality. According to the ICZN (1999: Article 76.1), the type locality is the geographical place of capture of the primary type (holotype or lectotype). In the absence of a primary type, the type locality encompasses the localities of all the syntypes (Article 73.2.3). This information can be obtained from labels attached to primary types or to syntypes or from the original publication (referred to as “original citation” in the text) whichever is more inclusive, or inferred from the title of the publication or even from the name of the species. When a neotype is designated, its place of capture becomes the type locality (Article 76.3) even if the specimen was collected outside the original area. In this catalogue, type localities taken from labels or from original publications are listed as indicated although the order of the elements is sometimes changed; any additional information is placed in square brackets. Many species described in the xviii and xix Centuries had but little informative place of origin, such as a country, state, province, or large geographical area (e.g., Rocky Mountains or Lake Superior). Lindroth (1961-1969) restricted the type locality of several of these North American species by selecting a specific locality or a county within the original region specified. This practice is followed in this catalogue and specific type localities are selected for several species-group taxa. Of course, only localities where a given species was actually collected can be selected.
Notable private carabid collectionsMany North American species of carabids described in the xix and beginning of the xx Centuries were from specimens held in private collections. The whereabouts of these collections are important to taxonomists. Some of the more significant ones are discussed.
Pierre François Marie Auguste Dejean (1780-1845) CollectionDejean, a French military officer by profession, certainly held the largest private beetle collection of his time, which he built through exchanges, purchases, gifts, and his own collecting in various parts of Europe. He described a total of 289 new carabid species-group taxa from North America, of which 182 (63%) had not been described earlier according to the present catalogue. At the sale of his collection in 1840, the carabid section (which also included the agyrtid genus Pteroloma) was the most significant, not only because it contained 3, 014 species and 17, 914 specimens, but because it was the only one to include name-bearing types. Dejean did not describe a new species-group taxon during his lifetime that he did not consider a carabid. Dejean’s carabid collection (including tiger beetles) was purchased for 7, 000 francs by Marquis F. Thibault de LaFerté-Sénectère who sold it, along with his own carabids, to Baron Maximilien de Chaudoir [q.v.] in 1859. Dejean’s carabid specimens are at MHNP today. Lindroth (1955b) discussed the name-bearing types and status of almost all North American species described by Dejean.
Thomas Say (1787-1834) CollectionSay was the first naturalist born in North America to describe new species of beetles from this continent. In the course of 17 years (1817-1834), he described 164 carabid species from North American material which he believed were new to science. Based on their current status, 142 (87%) had effectively not been previously described. Say left his collection by verbal bequest through his wife to the Academy of Natural Sciences in Philadelphia in 1834 (Weiss 1936: 277). After his death, which occurred in October of the same year, the collection was shipped from New Harmony, Indiana, to Philadelphia through New Orleans. In 1836, Charles Pickering sent Say’s insects to Thaddeus W. Harris in Cambridge, Massachusetts, in order to “put them in good order, and return them in a condition to be preserved” (Harris to D.H. Storer, 2 November 1836). In the same letter Harris reported “They [Say’s specimens] arrived about the middle of July; but on examination were found to be in a deplorable condition, most of the pins having become loose, the labels detached, and the insects themselves without heads, antennae and legs, or devoured by destructive larvae, and ground to powder by the perilous shakings which they had received in their transportation from New Harmony.” In a letter to C.J. Ward, dated 8 March 1837, Harris wrote “I assure you that Mr. Say’s cabinet does not contain one half of the species which he has described; of the insects in it, many are without names, and all more or less mutilated, and so badly preserved that most of them are now absolutely worthless.” On July 16, 1838, Harris indicated in a letter to S.G. Morton (see Fox 1902: 11) that he had “been obliged to bake a considerable part of the insects lately belonging to Mr. Say twice, and some of them three times, in order to destroy the vermin with which they are infested.” Say’s collection was returned to the Academy of Natural Sciences in Philadelphia in March 1842 “in such a state of ruin and dilapidation as to be almost useless” (Ruschenberger 1852: 25).
During his life, Say sent some of his specimens abroad including many to Dejean in Paris (see Dejean 1826: vi). Fortunately Dejean’s carabid collection has remained intact and in good condition to this day. In their attempt to bring taxonomic stability to Say’s names, Lindroth and Freitag (1969) selected lectotypes for eight carabid species described by Say for which Say’s authentic specimens could be located in Dejean’s collection. They also designated neotypes from the MCZ material for 131 of the remaining 156 of Say’s species leaving the tiger beetles (14 species) and a few taxa, all currently considered invalid, without type specimens. Say’s species were interpreted by Lindroth and Freitag from LeConte’s concept according to his collection. LeConte never saw Say’s collection and his interpretation of Say’s species came exclusively from the original descriptions which he considered adequate: “The entire destruction of his [Say’s] original specimens would be the subject of much greater regret, were it not for the fact that his descriptions are so clear as to leave scarcely a doubt regarding the object designated. I am thus enabled to assign to nearly all of his Coleoptera their proper place in the modern system” (LeConte 1859d: vi).
Thaddeus William Harris (1795-1856) CollectionHarris, well known for his work in economic entomology (his profile having appeared on every cover of the Journal of Economic Entomology for more than 35 years), described 28 new carabid species from North America. Ten (36%) are considered valid in this catalogue. To his defence, several of his species were made available by the posthumous publication of some of his letters several decades after they were written. At Say’s suggestion, Harris sent his entire collection to Thomas Say in Philadelphia, in 1825, who labeled the specimens as well as he could. Harris’ collection, which included “4, 838 specimens in 2, 241 species of Coleoptera, ” contained “many typical specimens described by Harris, Say, and others” (Scudder 1860: 72). It was bought by friends in 1858 and presented to the Boston Society of Natural History. Harris’ collection was transferred to the Museum of Comparative Zoology at Cambridge in April 1941 (Darlington 1941b: 273) where it stands separately from the general collection in two standard 25 drawer cabinets.
Gustav Graf von Mannerheim (1797-1854) CollectionMannerheim, a Finnish noble by birth and wealthy by inheritance, described 72 new North American carabid species, all from Alaska and California. Of these, 23 (32%) had not been described previously. Mannerheim never visited the New World and his descriptions were based on specimens brought back chiefly by Russian collectors such as Johann F. Eschscholtz, Eduard L. Blaschke, Egor L. Tschernikh, and Il’ia G. Vosnesensky. His library and personal collection, which consisted, at the end, of 18, 000 species and nearly 100, 000 specimens, were sold for the sum of 8, 000 silver rubles by his widow, Countess Eva Mannerheim, in 1855 to the University of Helsinki. The money used to buy the collection came from a loan made by the Emperor to the University with the understanding that the University will pay back annually the sum of 500 rubles to the Imperial Bank of Finland which will use it for poor- and workhouses in the country (Rein 1857). Mannerheim’s collection is kept separately at the University of Helsinki (Silfverberg 1995: 43).
Jules Antoine Adolphe Henri Putzeys (1809-1882) CollectionPutzeys described 38 new North American species of carabids; 15 (39%) are listed as valid in this catalogue. He worked in close collaboration with Chaudoir, the leading carabidologist of the time, and described several new species from specimens in Chaudoir’s collection. These specimens are now in MHNP. He also gave many of his own types to Chaudoir. His personal collection was bequeathed in 1885 to the Société Royale Belge d’Entomologie under the care of the Musée Royal d’Histoire Naturelle in Brussels. Putzeys’ collection consisted of 26, 429 specimens of carabids (including cicindelids) and 6, 123 species (Preudhomme de Borre 1885: clx) as well as many other beetles and various insects.
Victor de Motschulsky (1810-1871) CollectionMotschulsky, a Russian Imperial Army Colonel, described 121 new geadephagan species from North America; 27 (22%) were undescribed at the time based on current practice. A large part of this material came from a 10-month trip he made in 1853-54 to the United States and Panama. He collected at several locations including New York, Niagara Falls, Cleveland, Cincinnati, Cawington, Lexington, the Mammoth Cave, Nashville, Louisville, New Orleans, Mobile, Montgomery, Atlanta, Washington, D.C., and Philadelphia. In the last city, he visited LeConte, Haldeman, Melsheimer, and Zeigler. The first three gentlemen gave Motschulsky several specimens from their collections including “types” (Motschulsky 1856: 16). LeConte also identified part of the beetles Motschulsky collected in Louisiana, Alabama, Georgia, and Carolina. Motschulsky’s main collection, which included almost 60, 000 specimens and about 4, 000 types of beetles, was bequeathed to the Société Impériale des Naturalistes de Moscou. It was stored in poor condition and suffered considerable damage before it was acquired in 1911 by the Zoological Museum, Moscow Lomonosov State University (Antonova 1991: 72). Keleinikova (1976) catalogued the carabid syntypes of Motschulsky’s collection at ZMMU.
Samuel Stehman Haldeman (1812-1880) CollectionHaldeman described 45 new carabid species from North America; 22 (49%) had not been described previously. In 1869 Haldeman, who had purchased Hentz’s collection, sold his collection of beetles to Simon Snyder Rathvon of Lancaster, Pennsylvania, “for about what the cases cost” (Rathvon in Geist 1881: 125). Rathvon’s collection and library were purchased for $1, 000 by Henry Bobb of East Greenville, Pennsylvania, and presented to the Franklin and Marshall College in Lancaster, Pennsylvania, as a memorial of his son (Dubbs 1903: 369). In a letter dated April 1875 and addressed to Alexander Agassiz (see below), John L. LeConte stated that he owned “all the unique types” of Haldeman. This leads one to speculate that Haldeman, a close friend of LeConte, gave his name-bearing specimens to LeConte prior to selling his collection to Rathvon.
Maximilien Stanislavovitch Baron de Chaudoir (1816-1881) CollectionRussian aristocrat of French origin, Chaudoir was not the typical insect collector. He made a single extensive collecting trip in his life, a 40 day-journey to the Caucasus in company of M.H. Hochhuth in 1845. His collection was mostly built through purchases and gifts. The single most significant purchase was LaFerté-Sénectère’s carabid collection in 1859 which included Dejean’s original specimens. In January 1874 Chaudoir gave his tiger beetle specimens, representing 713 species, to MHNP. After his death in May 1881 his collection passed into the hands of René Oberthür in Rennes as agreed upon between Chaudoir and the Oberthür brothers. Over nearly five decades, Chaudoir described 126 new carabid species based on specimens collected in North America; 58 (46%) had not been described earlier based on this catalogue.
René Oberthür died in April 1944 and his collection, certainly one of the two largest private beetle collections ever built, was classified as “monument historique” in January 1948 by the French government. The collection, which included at least five million specimens, was acquired for the sum of 32 million francs by the Muséum d’Histoire Naturelle in Paris (MHNP) in 1951 (Cambefort 2006: 249).
Henry Ulke (1821-1910) CollectionAlthough Ulke described only two North American carabids in his life, Bembidion nevadense in 1875 and Pterostichus johnsoni in 1889, his collection, which he sold in 1900 to the Carnegie Museum in Pittsburgh, was used extensively by LeConte and Horn and contains numerous syntypes of new species described by the two coleopterists. However, recognition of many of these syntypes can be difficult. Sometimes all syntypes were retained by LeConte and Horn while on other occasions all or some of them were returned to Ulke. Furthermore, syntypes returned to Ulke were often reincorporated in his collection with others of the same species from the same place. Usually these were marked with a number or colored square, but since many syntypes were left unmarked at the time, it is sometimes impossible to recognize them at the Carnegie Museum (Robert L. Davidson pers. comm. 2008).
John Lawrence LeConte (1825-1883) CollectionLeConte is without doubt the most outstanding North American coleopterist of the xix Century, not only because he described 514 new genus-group and about 4, 730 new species-group taxa of beetles (Henshaw 1882: 270), but because he was the first to work seriously on the classification of the North American fauna. During his scientific activity, which lasted almost 40 years, he described 724 new species-group taxa of Geadephaga from North America, 439 (61%) of which were not previously described. LeConte built his collection through his own collecting but also from gifts he received and identifications he provided to many persons from whom he usually retained all or some of the specimens. There is also little doubt that his father, Major John Eatton LeConte
In April 1875, LeConte wrote to his friend Alexander Agassiz, director of the Museum of Comparative Zoology in Cambridge, and expressed the wish that his collection be deposited at the museum after his death
LeConte used small colored paper disks to indicate the provenance of his specimens. The color system used is as follows:
Pale blue Lake Superior, Canada
Pink Middle states, i.e., Maryland, Delaware, New York, New Jersey, Pennsylvania, and possibly also Connecticut and Rhode Island
Pale pink Vermont, New Hampshire, Massachusetts
White Northern and eastern states, Canada, and possibly also Alaska
Orange (brick red) Southern and Gulf states, i.e., Virginia, North Carolina, South Carolina, Georgia, Florida, Alabama, Mississippi, Louisiana, and possibly also eastern Tennessee and Arkansas
Dark red Texas
Yellow Ohio, Illinois, Indiana, Missouri, western Tennessee, Kentucky, and possibly Iowa and the southern edge of the Great Lakes
Pale green Nebraska, Kansas, North Dakota, South Dakota, Oklahoma, Colorado, Wyoming, Montana
Dark green New Mexico
Black Utah
Silver Arizona and Valley of Gila (so including also southwestern New Mexico)
Silver with edge cut Baja California, Mexico
Gold California
Dark blue Oregon, Washington
Brown Russian America, i.e., probably the region around Colony Ross, a farming community about 75 miles north of San Francisco along the coast in California, and Alaska
George Henry Horn (1840-1897) CollectionA physician by profession, Horn authored or coauthored more than 250 papers, in which he described 154 new genera and more than 1, 600 new species of beetles, including 103 North American Geadephaga. Based on the current classification, 75 (73%) of his new geadephagan species had not been described previously. His collection and library were bequeathed to the American Entomological Society, which deposited them at the Academy of Natural Sciences in Philadelphia. In October 1974, the Horn and William G. Dietz collections were delivered to the Museum of Comparative Zoology in return for the Scudder and Morse orthopteroid insects of the MCZ (Philip D. Perkins pers. comm. 2004; see Lawrence 1973: 151). Horn’s collection is preserved along with that of LeConte apart from the general collection.
Thomas Lincoln Casey (1857-1925) CollectionFrom 1884 to the end of his life, Casey described 1, 864 new species-group taxa of North American Geadephaga; only 307 (16%) had not been described previously based on current concepts. Still many of his remaining “valid species” have not been subsequently studied, particularly those belonging to small species of the tribe Harpalini, and a substantial proportion will certainly end up in synonymy. Furthermore, several of Casey’s species are valid simply by chance as he did not recognize or study the proper characters (such as the male genitalia) that distinguished them from their closely related taxa known at the time. His collection, consisting of almost 117, 000 specimens, including name-bearing types for more than 9, 200 species-group taxa (Buchanan 1935: 7; Blackwelder 1950: 65), was built through Casey’s own collecting and by purchases. It was bequeathed to the United States National Museum in Washington, D.C. Casey (1918: 291) stated that “about a dozen” of his types “disappeared from ... [his] collection while temporarily at the Cambridge Museum.” The syntypes of some of these species (e.g., Bembidion militare, Tachys occultator, Amara pallida, Amara ferruginea, and Amara marylandica among Carabidae) are at the MCZ. Casey did not designate holotypes as such and therefore, unless he expressly indicated in the original description that he had but a single specimen or that a lectotype had been designated, all type specimens in his collection are syntypes.
Willis Stanley Blatchley (1859-1940) CollectionBlatchley described 12 new North American carabid species; only two (17%) are considered valid in this work. His library and large insect collection, which included 470 name-bearing specimens, were given to Purdue University. Blatchley did not select type specimens in his publications but subsequently designated lectotypes [as types] for all the new species he had described (Blatchley 1930: 33-50).
Charles Frederic August Schaeffer (1860-1934) CollectionSchaeffer, curator of the insect collection at the Museum of the Brooklyn Institute of Arts and Sciences, described 30 new carabid species; 22 (73%) are still valid today. In 1929, the Brooklyn Museum transferred 37, 100 insect specimens, including many of Schaeffer’s carabid types, to the USNM (Debbie Feher pers. comm. 2008). Currently the type material of 25 (possibly 26) of Schaeffer’s species-group taxa are in the USNM. It is clear in his 1910 paper that Schaeffer was selecting one of the specimens from his series as “the type.” However he may not have labeled them as such because lectotypes have been designated for several of his new species by various authors.
Henry Clinton Fall (1862-1939) CollectionA teacher by profession, Fall owned one of the largest private collections of North American beetles toward the end of his life, with an estimated 250, 000 specimens (including those of Charles Liebeck which came to Fall in the 1930s) representing between 14, 000 and 15, 000 species or about 90% of the fauna of the time (Darlington 1940a: 46) if one excludes the “species” described by Casey. Over a period of about 40 years, Fall described 47 new North American carabid species-group taxa; 31 (66%) are still considered valid today. He left his collection, together with his correspondence, notebooks, and reprints, to the Museum of Comparative Zoology at Harvard University where his specimens are kept separately at the end of each genus in the general collection. In one of his 1910 papers, Fall designated holotypes (as “the type”) for the first time. From this publication, “type” specimens labeled as such in his collection are considered holotypes. All original specimens of his new species described prior to 1910 should be considered syntypes. Type labels on some of these specimens were probably added after the publication of the original descriptions.
Roland Hayward (1865-1906) CollectionHayward, a member of the Boston Stock Exchange and of the Boston Society of Natural History, described 42 new species of carabids from North America, all in the tribe Bembidiini and the genus Amara. Currently 32 (76%) are considered valid. His collection, which he built through purchases, gifts, exchanges, and his own collecting in New England as well as in Colorado, Manitoba, and New Brunswick, was bequeathed to the Museum of Comparative Zoology in Cambridge. Hayward did not designate type specimens for his new species.
Edwin Cooper Van Dyke (1869-1952) CollectionProfessor Van Dyke described 73 new carabid and one new trachypachid species from North America; 54 (73%) of which had not been described previously based on their current status. His collection, consisting of about 200, 000 specimens (Essig 1953: 88), was presented to the California Academy of Sciences in 1924 where the holotypes of all but three of his 74 new species of Geadephaga are currently stored.
Howard Notman (1881-1966) CollectionNotman described 38 new carabid species from North America between 1919 and 1929; 21 (55%) had not been described previously based on their current status. In 1948 he donated his entire collection to the Staten Island Institute of Arts and Sciences, where it is still today (Smetana and Herman 2001: 118). Based on Hennessey’s (1990) type catalogue of that institution, type specimens of all new species Notman collected himself, most from the Adirondacks where he owned a summer home, are in his collection in SIM (18 in total). He also described several new species from material owned by institutions, such as the USNM. Notman did not designate type specimens in his papers of 1919 and 1920 but did so after.
Classification of GeadephagaUnfortunately, there is no consensus among coleopterists concerning the classification of Geadephaga even at the family level. Some authors rank the cicindelids, rhysodids, and trachypachids as Carabidae while others consider one, two, or all three groups as distinct families. Even the paussines are sometimes raised to family level by modern authors. At this time, I prefer to classify the Geadephaga into three families, i.e., Trachypachidae, Rhysodidae, and Carabidae.
Following Jeannel’s (1941b-1942) classification of the carabids of France, a number of authors, mostly French and Spanish taxonomists, still recognized several families of “ground beetles.” Such an approach does not add anything to the understanding of carabid evolution. It simply adds another level to the Linnaean classification. If Jeannel’s approach is followed, it could and should have an impact on the classification of the other adephagan groups, particularly the dytiscids. Since I have been under the influence of Lindroth’s work on the carabids of Canada and Alaska, Jeannel’s approach seems to me unjustified.
Following is a discussion of the family-group taxa of Geadephaga.
Family Trachypachidae. Monophyly of this family is well supported by larval and adult apomorphies (Arndt and Beutel 1995; Beutel 1994; Beutel 1998). The systematic position of this group, however, is contentious. Bell (1966b, 1967), Bils (1976), Evans (1977a, 1985), Hammond (1979), Ward (1979), Burmeister (1980), Roughley (1981), Nichols (1985c), Beutel and Belkaceme (1986), Ruhnau (1986), Beutel and Roughley (1988), Acorn and Ball (1991), Arndt (1993), Deuve (1993), Arndt and Beutel (1995), Arndt (1998), and Beutel (1998) provided or discussed elements suggesting that trachypachids are more closely related to hydradephagans or part of Hydradephaga (i.e., Dytiscoidea) than to carabids. While most authors have regarded the Hydradephaga and Carabidae as distinct phyletic lineages, Bils (1976) and Nichols (1985c) argued that the hydradephagan-trachypachid lineage may have arisen within the Carabidae. Kavanaugh (1986) reevaluated the evidence supporting relationships of Trachypachidae with Hydradephaga. He concluded that trachypachids could be the sister-group of carabids and ranked the group as a subfamily within the Carabidae. Ponomarenko (1977) also postulated, from fossil evidence, that trachypachids and carabids are sister-groups that evolved from a common eodromeid ancestor. Beutel and Haas (1996), Kavanaugh (1998: 337), Fedorenko (2009), Dressler and Beutel (2010), and Martínez-Navarro et al. (2011) found support for monophyly of a clade including trachypachids and carabids. Recent molecular studies also suggested that trachypachids are more closely related to Geadephaga than to Hydradephaga (Shull et al. 2001; Maddison et al. 2009). In addition, pygidial gland compounds in trachypachids are more similar to those known from Carabidae than from Hydradephaga (Attygalle et al. 2004: 586). In this catalogue, trachypachids are included in the Geadephaga and given family rank.
The Trachypachidae includes two extant genera: Systolosoma Solier with two species in Chile and Argentina and Trachypachus Motschulsky with four species, one in Eurasia and three in western North America.
Many putative trachypachid fossils were found in Mesozoic deposits of Asia. Ponomarenko (1977), who studied the material, included all seven genera of trachypachid fossils in a distinct subfamily, Eodromeinae. Beutel (1998: 83) pointed out that the affinities between trachypachids and eodromeines are unclear because there are no apparent synapomorphic character states between the two groups.
Family Rhysodidae. Traditionally ranked as a distinct family, rhysodids (also known as wrinkled bark beetles) have been included within the family Carabidae in recent years by several authors following evidence or discussion provided by Bell and Bell (1962), Bell (1970), Forsyth (1972), Reichardt (1977), Baehr (1979), Beutel (1990, 1992c), Yahiro (1996), Bell (1998), Liebherr and Will (1998), and others. Some authors have treated the group as a tribe related to Scaritini or Clivinini. Reichardt (1977: 393) stated that rhysodids were “closest” to salcediines and Bell (1998: 268) even suggested that the genus Solenogenys Westwood, traditionally included within the Salcediini, is the sister-group to rhysodids. Erwin (1991a: 10) on the other hand included rhysodids within his subfamily Psydrinae along with gehringiines, psydrines, moriomorphines, patrobines, trechines, zolines, pogonines, and bembidiines. Molecular data published by Maddison et al. (1999: 125) suggest that rhysodids could be the sister-group to cicindelids and that both could be closely related to the subfamily Harpalinae. Others taxonomists, however, have continued to treat the rhysodids as a distinct family. Regenfuss (1975) and Nagel (1979) suggested that the Rhysodidae could be the sister-group of the remaining Geadephaga; Deuve (1993: 100) the sister-group to the other Adephaga (with the possible exception of Gehringiinae); Beutel and Roughley (1988) the sister-group of the remaining Adephaga excluding Gyrinidae; Beutel (1992a, 1993, 1998) the sister-group to Carabidae (without trachypachids). Recently Makarov (2008) found no evidence from the larval morphology suggesting that rhysodids are specialized Carabidae. Instead rhysodid larvae share several features with those of the suborder Archostemata. At this time, I prefer to rank rhysodids as a distinct family based on tradition but also on the fact that there is no solid morphological or molecular evidence presented to date pointing out that the Carabidae (with or without trachypachids) are paraphyletic in regard to rhysodids.
About 355 species of rhysodids are currently known and are placed into seven family-group taxa, namely Leoglymmiini, Medisorini, Rhysodini, Dhysorini, Sloanoglymmiini, Omoglymmiini, and Clinidiini. These taxa are usually ranked as subtribes when rhysodids are included in the carabids. I have followed Bousquet and Larochelle (1993) in listing them as tribes. Only the last two-mentioned tribes are represented in North America.
Tribe Leoglymmiini. This tribe contains a single species, Leoglymmius lignarius (Olliff), from Australia. Contrary to other rhysodids, the minor setae on antennomeres 5-10 are arranged in broad bands encircling the distal third of the segment and the mentum is separated from the ventral lobe of the gena by a distinct suture in its anterior half.
Tribe Medisorini. A single species, Medisores abditus Bell and Bell, belongs to this tribe. The few known specimens have been found in Cape Province in the Republic of South Africa.
Tribe Rhysodini. This tribe is confined to the Eastern Hemisphere and includes about 25 species in three genera: Rhysodes Germar (two Palaearctic species), Kupeus Bell and Bell (one New Zealand species), and Kaveinga Bell and Bell (23 Australian species).
Tribe Dhysorini. This tribe includes ten species placed in three genera, Dhysores Grouvelle in Africa, Tangarona Bell and Bell in New Zealand, and Neodhysores Bell and Bell in South America.
Tribe Sloanoglymmiini. This tribe has been proposed for one species, Sloanoglymmius planatus (Lea), endemic to southeastern Australia. The genus is taxonomically isolated and its relationship to other rhysodid genera is obscure.
Tribe Omoglymmiini. This tribe includes 180 species placed in eight genera. The group is represented in all zoogeographical regions but less so in Australia, Africa, and South America (Bell and Bell 1978: 66). The two North American species belong to the subgenus Boreoglymmius Bell and Bell, of the genus Omoglymmius Ganglbauer, along with one Japanese species. According to Bell and Bell (1983: 141), the two North American species are probably more closely related to each other than either is to the Japanese species.
Tribe Clinidiini. This tribe contains about 135 species placed in the genera Clinidium Kirby, Rhyzodiastes Fairmaire, and Grouvellina Bell and Bell. The species are found in all zoogeographical regions, including Madagascar, but are absent from the African continent. The North American fauna has only six species, five in the east and one in the west, included in the subgenus Arctoclinidium Bell of the genus Clinidium. This subgenus also contains three Palaearctic species, one in Japan and two in Europe. According to Bell and Bell (1985: 77), the North American species and the Japanese one form a clade and the European species another clade. These authors also placed the Japanese species, Clinidium veneficum Lewis, as the sister-group to Clinidium valentinei Bell of eastern North America.
Family Carabidae. Monophyly of the Carabidae, as defined here, is not evident. The layout of the prehypopharyngeal setae in the larvae (Beutel 1993) and the development of antennal pubescence in the adults (Beutel 1995) have been suggested as synapomorphies for the family. However, Arndt et al. (2005: 138) considered these character states not very convincing given the variation involved in the structures. Recent molecular sequence analyses conducted by Maddison et al. (2009) found little support for monophyly of the group no matter if the trachypachids, rhysodids, and/or cicindelids were included or excluded unless the Carabidae was considered equivalent to the Geadephaga. Therefore, the Carabidae, as defined here, could be paraphyletic in regard to rhysodids, trachypachids, and possibly even to Hydradephaga.
Carabids are found on all continents, except Antarctica, and on most islands. They range from well above the arctic circle to Tierra del Fuego and South Georgia in the Southern Hemisphere. Based on Lorenz’s (2005) checklist, 33, 920 valid species are recognized.
The current classification of the Carabidae is based mainly on morphological data of adults although molecular sequence data have been used recently to discuss various aspects of carabid phylogeny. Despite several attempts there is no consensus on the classification of several subfamilies or tribes. This is particularly evident among ‘basal grade’ carabids.
Fossils belonging to the family Carabidae are known from the early Jurassic (Ponomarenko 1977) which suggests that the family emergence dates back to the beginning of the Jurassic or the end of the Triassic (Kryzhanovskij 1983). Ponomarenko (1977) proposed two family-group taxa of Carabidae among Mesozoic fossils, the subfamily Protorabinae for five genera and the tribe Conjunctiini for two genera.
The world classification of family-group taxa, which has been adopted for the North American fauna in this catalogue, is outlined in Table 3.
Classification of world family-group taxa of Carabidae. Taxa represented in North America are followed by a dot.
| Subfamily Nebriinae | |
| Tribe Pelophilini • | |
| Tribe Opisthiini • | |
| Tribe Nebriini • | |
| Tribe Notiokasiini | |
| Tribe Notiophilini • | |
| Subfamily Cicindinae | |
| Tribe Cicindini | |
| Subfamily Carabinae | |
| Tribe Cychrini • | |
| Tribe Pamborini | |
| Tribe Ceroglossini | |
| Tribe Carabini • | |
| Subfamily Cicindelinae | |
| Tribe Amblycheilini • | |
| Tribe Manticorini | |
| Tribe Megacephalini • | |
| Tribe Cicindelini • | |
| Tribe Ctenostomatini | |
| Tribe Collyridini | |
| Subfamily Loricerinae | |
| Tribe Loricerini • | |
| Subfamily Elaphrinae | |
| Tribe Elaphrini • | |
| Subfamily Omophroninae | |
| Tribe Omophronini • | |
| Subfamily Migadopinae | |
| Tribe Amarotypini | |
| Tribe Migadopini | |
| Subfamily Hiletinae | |
| Tribe Hiletini | |
| Subfamily Scaritinae | |
| Tribe Pasimachini • | |
| Tribe Carenini | |
| Tribe Scaritini • | |
| Tribe Clivinini • | |
| Tribe Salcediini | |
| Tribe Dyschiriini • | |
| Tribe Promecognathini • | |
| Tribe Dalyatini | |
| Subfamily Broscinae | |
| Tribe Broscini • | |
| Subfamily Apotominae | |
| Tribe Apotomini | |
| Subfamily Siagoninae | |
| Tribe Enceladini | |
| Tribe Siagonini | |
| Tribe Lupercini | |
| Subfamily Melaeninae | |
| Tribe Melaenini | |
| Subfamily Gehringiinae | |
| Tribe Gehringiini • | |
| Subfamily Trechinae | |
| Tribe Trechini • | |
| Tribe Zolini | |
| Tribe Bembidiini • | |
| Tribe Pogonini • | |
| Subfamily Patrobinae | |
| Tribe Lissopogonini | |
| Tribe Patrobini • | |
| Subfamily Psydrinae | |
| Tribe Psydrini • | |
| Subfamily Moriomorphinae | |
| Tribe Moriomorphini | |
| Tribe Amblytelini | |
| Subfamily Nototylinae | |
| Tribe Nototylini | |
| Subfamily Paussinae | |
| Tribe Metriini • | |
| Tribe Mystopomini | |
| Tribe Ozaenini • | |
| Tribe Protopaussini | |
| Tribe Paussini | |
| Subfamily Brachininae | |
| Tribe Crepidogastrini | |
| Tribe Brachinini • | |
| Subfamily Harpalinae | |
| Supertribe Pterostichitae | |
| Tribe Morionini • | |
| Tribe Cnemalobini | |
| Tribe Microcheilini | |
| Tribe Chaetodactylini | |
| Tribe Cratocerini | |
| Tribe Abacetini • | |
| Tribe Pterostichini • | |
| Tribe Zabrini • | |
| Tribe Metiini | |
| Tribe Drimostomatini | |
| Tribe Chaetogenyini | |
| Tribe Dercylini | |
| Tribe Melanchitonini | |
| Tribe Oodini • | |
| Tribe Peleciini | |
| Tribe Brachygnathini | |
| Tribe Bascanini | |
| Tribe Panagaeini • | |
| Tribe Chlaeniini • | |
| Tribe Cuneipectini | |
| Tribe Orthogoniini | |
| Tribe Idiomorphini | |
| Tribe Glyptini | |
| Tribe Amorphomerini | |
| Supertribe Harpalitae | |
| Tribe Licinini • | |
| Tribe Harpalini • | |
| Tribe Geobaenini | |
| Tribe Omphreini | |
| Tribe Sphodrini • | |
| Tribe Platynini • | |
| Tribe Perigonini • | |
| Tribe Ginemini | |
| Tribe Enoicini | |
| Tribe Atranini • | |
| Tribe Catapieseini | |
| Tribe Lachnophorini • | |
| Tribe Pentagonicini • | |
| Tribe Odacanthini • | |
| Tribe Calophaenini | |
| Tribe Ctenodactylini • | |
| Tribe Hexagoniini | |
| Tribe Cyclosomini • | |
| Tribe Somoplatini | |
| Tribe Masoreini | |
| Tribe Corsyrini | |
| Tribe Sarothrocrepidini | |
| Tribe Graphipterini | |
| Tribe Lebiini • | |
| Tribe Dryptini | |
| Tribe Galeritini • | |
| Tribe Zuphiini • | |
| Tribe Physocrotaphini | |
| Tribe Anthiini | |
| Tribe Helluonini • | |
| Tribe Xenaroswellianini | |
| Tribe Pseudomorphini • | |
Subfamily Nebriinae. This subfamily includes the tribes Nebriini, Notiokasiini, Notiophilini, Opisthiini, and Pelophilini. All but notiokasiines are Northern Hemisphere elements and represented in North America. Evidence supporting monophyly of Nebriinae is not overwhelming. The only known synapomorphy in the adult stage is the asetose parameres (Kavanaugh and Nègre 1983), a character state found in other, clearly unrelated carabid lineages. Arndt (1993: 21) listed three putative synapomorphies upon examination of the larval morphology. The molecular data analyses by Maddison et al. (1999: 125) provided only moderate support for monophyly of the subfamily and Kavanaugh’s (1998) phylogenetic analysis suggested that this subfamily represents a grade rather than a clade.
The subfamilies Nebriinae and Carabinae could be closely related as pointed out by Jeannel (1940: 7), Bell (1967: 105), Beutel (1992c: 57), and Su et al. (2004: 49). Both groups have open procoxal cavities, contrary to the remaining carabids. In addition, the external lamella of the metepimeron is completely covered and functionally replaced by an extension of the hind margin of the anepisternum (Beutel 1992c: 57). Some authors (e.g., Lorenz 2005: 125) also include the cicindines within the subfamily suggesting a close relationship between these groups. Based on similarities in the genitalia, Deuve (1993: 125) raised the possibility that the Hydradephaga, trachypachids, omophronines, and nebriines form a clade.
Tribe Pelophilini. This tribe includes a single genus, Pelophila Dejean, which has been retained in the tribe Nebriini until recently. Two species are known, both living in the boreal and subarctic regions: one is circumpolar, the other restricted to Canada and Alaska. Kavanaugh (1996: 34) suggested that the genus represents the sister-group to the remaining Nebriinae. One of Kavanaugh’s (1998: Fig. 2) cladograms suggested that Pelophila is more closely related to the tribe Nebriini than are the Opisthiini, Notiophilini, and Notiokasiini.
Tribe Opisthiini. This tribe includes two genera with five species and is doubtless monophyletic. Kavanaugh and Nègre (1983: 564) argued that opisthiines could be the sister-group to the remaining Nebriinae. On the other hand, Kavanaugh’s (1996: Fig. 1A) most parsimonious tree suggested that this tribe is the sister-group to Notiophilini and that these two tribes, along with Notiokasiini, form a clade which represents the sister-group to Nebriini.
Tribe Nebriini. This tribe contains about 600 species in the Palaearctic, Nearctic, and northern parts of the Oriental Regions. However, the group is clearly more diverse in the Palaearctic. The main genera of the tribe are Leistus Frölich, Archastes Jedlička, and particularly Nebria Latreille with more than 60% of the species. The limits of the genus Nebria are not quite settled. Kavanaugh (1995, 1996) regarded Nippononebria Uéno (including Vancouveria Kavanaugh) as the sister-group to Leistus while Ledoux and Roux (2005) listed Nippononebria and Vancouveria as subgenera of Nebria and suggested they form the sister-group to Eonebria Semenov and Znojko and Sadonebria Ledoux and Roux, a complex of 60 Palaearctic species.
Tribe Notiokasiini. This tribe contains a single species, Notiokasis chaudoiri Kavanaugh and Nègre, found in South America. Although the relationships of the tribe are obscure (Kavanaugh and Nègre 1983), Kavanaugh (1996: 33) found 12 synapomorphies supporting monophyly of a clade including notiokasiines, notiophilines, and opisthiines.
Tribe Notiophilini. The tribe includes a single genus, Notiophilus Duméril, very characteristic in the adult stage. The larvae, however, are similar in most structural features to those of Nebriini as pointed out by van Emden (1942). Jeannel (1941b: 175) included Notiophilini, Nebriini (with Pelophila), and Opisthiini in his family Nebriidae, suggesting implicitly a close relationship between the three groups. Kavanaugh’s (1996: Fig. 1A) most parsimonious cladogram suggested a sister-group relationship between Notiophilini and Opisthiini based on adult and larval morphological data. Based on confluent procoxal cavities, Nichols (1985c: Fig. 5) considered the tribe to be the sister-group to {Omophronini + Trachypachini + Hydradephaga}. Erwin (1991a: 11) noted that notiophilines, along with omophronines, hiletines, and trachypachids, have the first mesotarsomere slightly dilated and with squamate setae underneath. However, it remains to ascertain whether this character state is synapomorphic or convergent. Based on female reproductive tracts, Liebherr and Will (1998: 146) suggested that the tribe Notiophilini represents the sister-group to {Opisthiini + Nebriini (with Pelophila) + Omophronini}.
Notiophilines, with about 55 species described to date, live in the Nearctic and Palaearctic Regions and at higher altitudes in the northern parts of the Neotropical and Oriental Regions. They are more speciose in Asia than anywhere else. The phylogenetic relationships of the species have not been studied yet.
Subfamily Cicindinae. This subfamily includes two species, Archaeocindis johnbeckeri (Bänninger) from the Persian Gulf (Kuwait and Iran) and Cicindis horni Bruch from the Córdoba Province of Argentina. Very little can be said at this time about the relationships of the subfamily except that it represents a basal grade carabid taxon. Kryzhanovskij (1976a: 87) associated cicindines with paussines (excluding metriines) and nototylines; Nagel (1979, 1987) and Roig-Juñent et al. (2011) viewed them as the sister-group to paussines. Ball (1979: 100), however, doubted such proposed affinities between cicindines and paussines. Erwin (1985, 1991a), followed by Lorenz (2005: 125), included the Cicindini in the Nebriitae. Kavanaugh and Erwin (1991) studied the structural features and reviewed the relationships of the group. They concluded that cicindines are best placed in a distinct supertribe near the Nebriitae and Elaphritae (sensu Kryzhanovskij 1976a: 88). Kavanaugh’s (1998: Fig. 3) phylogenetic analysis using 153 characters of adult external and male genitalic structures suggested that cicindines may be closely related to omophronines, carabines, cychrines, and cicindelines. Aspects of the behaviour and life history of the Argentine species have been published recently (Erwin and Aschero 2004).
Subfamily Carabinae. This subfamily contains about 1, 300 species (Lorenz 2005: [i]) placed in four tribes: Cychrini, Pamborini, Ceroglossini, and Carabini. Most authors agree that the subfamily is monophyletic. According to Deuve (2004: 32), adults of this group are characterized by two significant autopomorphies: the presence of two strip-like apodemes flanking the basal orifice of the median lobe of the aedeagus and the presence of an alveolus on the epipleurite of the abdominal segment IX at the opening of the defensive gland. Arndt (1998: 179) noted several autopomorphies in larvae of Carabinae: an extensive decrease of number of setae on the tergites and sternites with an increase in the number of pores, size reduction of the sensorial appendage on antennomere III, and a markedly sclerotized body.
Tribe Cychrini. This well-defined and likely monophyletic group of about 200 species is restricted to the Northern Hemisphere. Osawa et al. (2004: 31) and Su et al. (2004: 49), based on molecular data, argued that the tribe is probably the sister-group to the remaining clades of the subfamily. Moore (1966b), Prüser and Mossakowski (1998: 316), and Arndt (1998: 180), based on morphological data, suggested that pamborines are the closest relatives to cychrines. Jeannel (1941b: 167) indicated that cychrines are more closely related to pamborines and ceroglossines than to carabines (sensu stricto) and calosomatines based on the shape of the parameres.
Relationships among the four genera have not been investigated. Whether or not Sphaeroderus Dejean and Scaphinotus Dejean, the two endemic North American genera, are sister-groups, as suggested by Erwin (2007a: 139), remains to be investigated. For example, Prüser and Mossakowski (1998: 316) listed several putative synapomorphies suggesting that Cychrus Fabricius and Sphaeroderus are sister-groups (Cychropsis Boileau was not included in their analysis). The phylogenetic tree by Osawa et al. (2004: Fig. 5.2) based on molecular sequence data suggested that Scaphinotus is the sister-group to the remaining Cychrini and Sphaeroderus the sister-group to Cychropsis.
Tribe Pamborini. The 13 species currently included in this tribe are placed in two genera: Pamborus Latreille from Australia and the monospecific Maoripamborus Brookes from New Zealand. Jeannel (1941b: 94) stated that pamborines are more closely related to ceroglossines than to any other Carabinae.
Tribe Ceroglossini. This tribe comprises only the genus Ceroglossus Solier (eight species and 46 subspecies) which is restricted to Chile and western Argentina. The genus has traditionally been included within the Carabini but recent analyses based on molecular sequence data suggest that ceroglossines are more closely related to pamborines than to carabines (Prüser and Mossakowski 1998: 297; Su et al. 2004: 50) or form the sister-group to the remaining members of Carabinae (Osawa et al. 2004: Fig. 5.2). Arndt (1998: 179) found evidence from the larval morphology to support the latter hypothesis.
Tribe Carabini. Carabines rank among the most popular groups for beetle collectors. Adults of many species are elegant, colorful, and large (often exceeding 15 mm). Such interest has generated a market for these beetles, particularly in Europe, and unfortunately also a race to describe new varieties, morphs, and aberrations. More than 1, 080 species of Carabini are recognized today worldwide. They inhabit all zoogeographical regions but are much more diverse in the Palaearctic Region than anywhere else.
The supraspecific classification of Carabini is debated. Some authors recognize only two genera, Carabus Linnaeus and Calosoma Weber, while others admit many, more or less clearly defined genera which are often grouped in two subtribes: Carabina and Calosomatina. I have followed the first approach and list all North American species in the genera Carabus (15 species) and Calosoma (41 species). The main difference between the two genera is the regression (or complete disappearance) of the ostial ligula of the aedeagus in members of Carabus (Deuve 2004: 33).
Based on morphological (larvae and endophallus of adults) and molecular sequence data, Deuve (2004) recognized eight major lineages within the genus Carabus: Spinulati, Digitulati, Lipastrimorphi, Archicarabomorphi, Tachypogenici, Metacarabi, Arcifera, and Neocarabi. The North American species are arrayed in nine subgenera: Carabus s.str. belongs to the Digitulati, Archicarabus Seidlitz to Archicarabomorphi, Tachypus Weber to Tachypogenici, Megodontus Solier to Neocarabi, and Diocarabus Reitter, Homoeocarabus Reitter, Aulonocarabus Reitter, Hemicarabus Géhin, and Tanaocarabus Reitter to Metacarabi.
Jeannel (1940: 68) recognized two major lineages within the calosomatines: the Calosomes lobés with a membranous ligula at the proximal opening of the male median lobe and the Calosomes ongulés with a chitinized ligula. The first lineage is represented in the Australian and Palaearctic Regions, and by six species belonging to the genus-group taxa Calosoma s.str. and Calodrepa Motschulsky in the Nearctic Region, the West Indies, and Mexico. The second lineage was divided by Jeannel (1940: 69-71) into three clades, the phyletic series of Castrida-Caminara represented in North America by a single species belonging to the genus-group taxon Castrida Motschulsky, the phyletic series of Callisthenes represented in the Nearctic Region by 23 species of the taxa Chrysostigma Kirby and Callistenia Lapouge, and the phyletic series of Callitropa represented in North America by 11 species of Blaptosoma Géhin, Carabosoma Géhin, Camegonia Lapouge, and Callitropa Motschulsky.
The systematic position of Aplothorax burchelli Waterhouse, a species endemic to the island of Saint Helena off the west coast of Africa, is controversial. Jeannel (1940) included the taxon within his Calosomes ongulés but Basilewsky (1972: 18-22) was convinced that the species is a relict of an old clade that evolved before the splitting of the Carabus and Calosoma lineages. He advocated placing the species in a distinct tribe which, in his opinion, was as justified as those of Pamborini and Ceroglossini.
Subfamily Cicindelinae. This group, referred to as the tiger beetles, has been regarded traditionally as a distinct family, but more and more coleopterists include it within the carabids. There is little doubt, based on characters of adults and larvae, that cicindelines form a monophyletic lineage. Relationships of the group, however, remain uncertain. It has been regarded as the sister-group to the remaining Carabidae by Nichols (1985c) and as the sister-group to Carabidae (minus paussines) by Regenfuss (1975). A close affinity between this subfamily and the Carabinae has been suggested by Erwin and Sims (1984: 366), Deuve (1993: 160; 2004: 32), Kavanaugh (1998: 338), and Liebherr and Will (1998: 151), although Liebherr and Will also emphasized that the Cicindelinae could instead be closely related to Promecognathini and Amarotypini. Maddison et al. (1999) indicated that most of their phylogenetic analyses of 18 rDNA place the Cicindelinae and Rhysodidae as sister-groups, near the Harpalinae. They also pointed out that the alternative placement of the cicindelines outside the Carabidae was more parsimonious than placing them among the basal-grade carabids. Deuve (2004: 32) noted two exceptional and primitive character states shared between cicindelines and carabines: presence of the abdominal tergite X in the male and presence of a phallobase on the aedeagus. He also pointed out numerous similarities between the two groups: the ectodermal genital ducts of the females are almost identical with a vagina differentiated in a bursa copulatrix, the presence of a sclerotized ligular apophysis, the presence of a filiform spermatheca and absence of an accessory gland, the presence of rod-shaped apophyses on the female abdominal epipleurites VIII allowing the formation of a telescopic egg-laying tube, the parameres of the aedeagus are glabrous and symmetrical, the endophallus often shows comparable dentiform sclerites, the digestion is extra-oral, and the ventral surface of the adult body often shows metallic coloration, an exceptional character state in the Adephaga.
This subfamily currently includes more than 2, 500 species distributed worldwide, except Tasmania, Antarctica, and remote oceanic islands, with the greatest diversity in the tropics (Pearson 1988). Tiger beetles are classified by most authors, following Horn (1926), into two major lineages, Alocosternales with a very narrow and deeply longitudinally-furrowed metepisternum in the adult and Platysternales with a wider metepisternum that has either no longitudinal furrow or a horseshoe-shaped furrow posteriorly. These groups are often listed as supertribes Collyriditae and Cicindelitae respectively. However, based on a combined analysis of molecular and morphological data, Vogler and Barraclough (1998: 254) noted that collyridites nested within the cicindelites, rendering the latter paraphyletic. Arndt (1998: 178) also noted that, based on larval character states, Cicindelitae and the tribe Megacephalini are not monophyletic. Based on the above information, the species of Cicindelidae are simply placed here in six tribes without further grouping.
Tribe Amblycheilini. This tribe includes the genera Omus Eschscholtz and Amblycheila Say represented in North America and Mexico, and the genus Pycnochila Motschulsky with one species in the Strait of Magellan. Amblycheilines have been classified in the past within the megacephalines but larval characters (Arndt and Putchkov 1997) and mitochondrial and nuclear RNA gene sequences (Vogler and Barraclough 1998: 251) suggest a basal position for amblycheilines, well removed from the true megacephalines. The tribe, however, may well be a grade rather than a clade. For example, Arndt (1998: 178) placed Omus as the sister-group to the remaining Cicindelinae based on larval characters.
Tribe Manticorini. This tribe includes 14 species, arrayed in the genera Mantica Kolbe (one rarely collected species from southern Namibia) and Manticora Fabricius (13 Afrotropical species). Contrary to other tiger beetles, males of this tribe have asymmetric mandibles and unexpanded protarsomeres (Werner 2000: 22).
Tribe Megacephalini. This tribe includes about 200 species arrayed in 11 genera (see Naviaux 2007: 15). Even without the amblycheilines, monophyly of the tribe is doubtful. For example, the genus Oxycheila Dejean, traditionally considered a member of Megacephalini, nested with rather strong support within the basal groups of Cicindelini in Vogler and Barraclough’s (1998: 254) cladistic analysis based on molecular and morphological data.
Tribe Cicindelini. This tribe is by far the most diversified clade of tiger beetles. The number of recognized genera varies to a great extent among taxonomists. In this work the 98 North American species (202 species-group taxa) are placed in nine genera. All but two (Cylindera and Cicindela) of these genera are New World endemics. Arndt (1998: 178) stated that Cicindelini forms the sister-group to {Ctenostomatini + Collyridini}.
Tribe Ctenostomatini. This tribe includes two genera: Ctenostoma Klug with about 115 Neotropical species, and Pogonostoma Klug with about 110 Madagascan species. Members of this tribe are synapomorphic in lacking the articulated hook at the extremity of the inner lobe of the maxilla (Jeannel 1946: 104).
Tribe Collyridini. This tribe contains about 335 species in Asia, of which one extends into the Australian Region. The species are arrayed in two subtribes: Tricondylina for the genera Derocrania Chaudoir (16 species) and Tricondyla Latreille (about 45 species), and Collyridina for the genera Protocollyris Mandl (16 species), Neocollyris Horn (about 250 species), and Collyris Fabricius (ten species). Naviaux (1994: 149) indicated the structural differences between the two subtribes.
Subfamily Loricerinae. This subfamily contains a single genus, Loricera Latreille, although some authors have treated Elliptosoma Wollaston, with one species from Madeira, as a distinct genus. The group is restricted to the Nearctic and Palaearctic Regions with some taxa found on mountains in the northern parts of the Neotropical and Oriental Regions. Loricerini is a basal grade taxon with obscure affinity. Jeannel (1941b: 80) associated loricerines with the Carabinae, Nebriinae, Cicindelinae, Elaphrinae, Omophroninae, and Siagoninae (including Promecognathus) in his Caraboidea Simplicia, characterized by the absence of metepimeric lobes (Jeannel 1941b: 93). Bell (1967: 105) included loricerines within his Anisochaeta Isopleuri along with elaphrines, scaritines, and cicindelines. Arndt (1993: 22) found several common derived larval features in larvae of Loricerinae and Cicindelinae to suggest a sister-group relationship between the two taxa. Maddison et al. (1999: 126) pointed out that placement of Loricera within {Migadopini + Amarotypini} received relatively strong support in their 18S rDNA analyses. These three taxa were also recovered as a monophyletic unit in analyses of the same gene by Ribera et al. (2005: 290). Vigna Taglianti and Rossi (1998: 515) indicated that loricerines could be closely related to elaphrines based on the presence of the same parasitic laboulbeniales species found on these groups. Erwin (2007a: 69) listed Elaphrini as the sister-group to Loricerini.
Recently Sciaky and Facchini (1999) described a new subgenus (Plesioloricera) for the new Chinese species Loricera balli which has eight, instead of 12 or more, striae. This species could possibly be the most basal taxon of the genus.
Klausnitzer (2003) described a new species, Loricera electrica, based on a larva found in Baltic amber. He believes the species probably belongs to the pilicornis group as defined by Ball and Erwin (1969).
Subfamily Elaphrinae. This subfamily includes a single tribe with obscure relationships. Bell (1967) listed the Elaphrini within his Isopleuri along with Loricerini, Scaritini, and Cicindelini. Following Jeannel’s (1941b: 214) intuition, both Kryzhanovskij (1976a: 88) and Erwin (1985: 469) considered the elaphrines as the sister-group to Migadopini (with and without Amarotypus respectively) and this hypothesis was supported by Deuve’s (1993: 160) study of the female genitalia and Roig-Juñent’s (1998: Figs 9-10) parsimony analysis using 33 adult and larval characters. On the other hand, Goulet (1983: 445) regarded Melaenini and the subtribe Broscina as the taxa most closely related to Elaphrini. This possibility was found most parsimonious by Maddison et al. (1999) based on their molecular sequence analysis which did not, however, include melaenine exemplars. In addition, Yahiro (1990: 42) reported a similar type of alimentary canal in elaphrines and broscines. Roig-Juñent’s (1998) parsimony analysis shows that elaphrines are not related to broscines. Parsimony analysis based on the female reproductive tract characters placed Elaphrini as the sister-group to {Opisthiini + Nebriini + Notiophilini + Omophronini} or near the Promecognathini and Amarotypini (Liebherr and Will 1998: 146).
The tribe is represented only in the Northern Hemisphere and includes three genera, all represented in the Nearctic Region.
Subfamily Omophroninae. This subfamily includes a single genus, Omophron Latreille. Some authors have suggested that Omophronini, Trachypachidae, and the Hydradephaga form a monophyletic group. Putative synapomorphies proposed for the complex include the presence, in the larvae, of an undivided cardo and a dorsal insertion of the femoro-tibial extensor (Ruhnau 1986) and, in the adults, the atrophy of intertergal muscle M24 (Bils 1976), partial housing of the procoxae by the mesosternum, and the prominent prosternal process contacting the metasternum (Nichols 1985c). Deuve (1993: 160) noted the presence of a peculiar sclerotized structure (named “sclerite helminthoïde”) in the female genitalia of omophronines, nebriines (sensu lato), trachypachids, and the Hydradephaga which raises the possibility of close phylogenetic affinities between these groups. Jeannel (1941b: 219) suggested that cicindelines were most closely related to omophronines. Bell (1967: 106) indicated that Omophronini might be aberrant Hemipleuri, a group including nebriines (sensu lato) and carabines (sensu lato). Kavanaugh (1998: 338), based on a parsimonious analysis of adult characters, suggested a close affinity between Omophroninae, Carabinae, and Cicindelinae. Liebherr and Will (1998: 156) listed several “potentially synapomorphic characters” supporting placement of omophronines with nebriines (sensu lato) and their preferred cladogram, based on 20 characters of female ovipositors and reproductive tracts, placed them as the sister-group to notiophilines. Many authors, however, have treated the Omophronini as a basal-grade taxon with unclear affinity. Based on larval character states, Landry and Bousquet (1984) found no evidence to indicate a sister-group to Omophronini. Such conclusions were also reached by Beutel (1991) from his study of the larval head and adult thoracic structures. Erwin (2007a: 63) indicated the possibility that cicindines were closely related to omophronines.
The genus Omophron includes about 70 species and is represented in all zoogeographical regions except the Australian one. There is no evidence yet known to suggest that the Nearctic or the Western Hemisphere species form a clade within the genus.
Subfamily Migadopinae. This subfamily currently consists of two tribes: Amarotypini and Migadopini. One of the main character states of the group is the presence of a long scutellar stria reaching the apical declivity of the elytra (Jeannel 1938b: 4) as in members of Pelophila. This characteristic, however, is absent in members of the genus Aquilex Moret, which have a short scutellar stria (Moret 2005: 30). Jeannel (1938b) revised the species of migadopines (as Migadopidae) and classified them into two groups: Monolobinae for the Chilean genus Monolobus Solier (two species) and Migadopinae for the remaining genera which are represented in South America and in the Australian Region. He also postulated that elaphrines were without doubt the group most closely related to migadopines in the Northern Hemisphere (Jeannel 1938b: 10). Moore et al. (1987: 65) included the migadopines from Australia within the supertribe Elaphritae.
Tribe Amarotypini. This tribe includes a single species, Amarotypus edwardsii Bates, from New Zealand. Until recently the species was placed in the tribe Migadopini but it differs by having a setiform unguitractor plate between the tarsal claws which is missing in migadopines. Erwin (1985: 469) postulated that Amarotypini could be the sister-group to {Migadopini + Elaphrini}. In Liebherr and Will’s (1998: Fig. 57) preferred cladogram, based on 20 characters of the female ovipositors and reproductive tract, amarotypines were positioned as the sister-group to promecognathines. In Roig-Juñent (2004: Fig. 1) phylogenetic analysis, based on 57 characters of the adult morphology, the genus Amarotypus Bates nested inside the remaining migadopine genera, as the sister-group to {Calathosoma + Stichonotus}.
Tribe Migadopini. This group of about 30 species in 15 genera is restricted to the temperate areas of the Neotropical and Australian Regions. Moret (2005: 30) recently proposed the subtribe Aquilicina for the genera Aquilex Moret (one species in Ecuador) and Rhytidognathus Chaudoir (one species in Uruguay). He also pointed out the close relationship between the South American genus Lissopterus Waterhouse (two species from the Tierra del Fuego Archipelago and Falkland Islands) and the New Zealand genera Loxomerus Chaudoir (five species) and Calathosoma Jeannel (one species). The phylogenetic analysis performed by Roig-Juñent (2004) do not support Moret’s conclusions although Aquilex was recovered as the sister-group to the other migadopine genera. Liebherr and Will (1998: 147) alluded to the possibility that the tribe is not monophyletic.
Subfamily Hiletinae. This subfamily includes two genera, Hiletus Schiødte (= Camaragnathus Bocandé) with six species in tropical Africa and Eucamaragnathus Jeannel with 15 species in the Afrotropical (six species), Oriental (five species), and Neotropical (four species) Regions. Jeannel (1941b: 80; 1946: 209) postulated that hiletines were closely related to scaritines (sensu lato) based mainly on the fact that these two taxa were the only disjuncti (i.e., with disjunct mesocoxae) with the metepimera lobed as in the conjuncti. Erwin and Stork (1985: 445) believed that hiletines were related to cnemalobines (as Cnemacanthini), elaphrines, migadopines, promecognathines, pseudomorphines, scaritines, and siagonines based on some tarsal character states and suggested that this complex forms the sister-group to the paussine-brachinine clade based on the presence of distinct epimera, brushy non-styliform parameres, long empodial unguitractor plates, and non-conjunct mesocoxae. They also concluded that hiletines represent the sister-group to {scaritines + cnemalobines (as Cnemacanthini) + pseudomorphines} and that this clade was characterized by having a single long guard seta at the apex of the fifth tarsomere which projects between the two tarsal claws.
Subfamily Scaritinae. This subfamily is inadequately defined and possibly polyphyletic. The species possess a mesothoracic peduncle which frees the prothorax from the elytra and allows greater mobility (Basilewsky 1973: 9). It includes about 1, 870 species worldwide which are grouped in this work into eight tribes: Pasimachini, Carenini, Scaritini, Clivinini, Dyschiriini, Salcediini, Promecognathini, and Dalyatini. Until the subfamily is better defined, it is difficult to comment on its relationships. Jeannel (1938a: 206) underlined a number of morphological features in the adults suggesting that scaritines and hiletines shared a common ancestor. Lindroth (1969b: xxiii) hypothesized that the similarities in “habitus and general organization” between scaritines (including promecognathines, clivinines, and dyschiriines) and broscines are probably an indication of close affinity.
Tribe Pasimachini. This tribe is represented by the genera Pasimachus Bonelli, with 32 species ranging collectively from southern Canada to Panama, and Mouhotia Laporte, with three species in eastern Asia. Monophyly of this group is doubtful. Bänninger (1950: 484) noted that if pasimachines and carenines are retained as distinct subtribes, Mouhotia cannot be placed in either of them and a separate subtribe would need to be established. To avoid proposing a new family-group name, Mouhotia is included here in the Pasimachini. Lorenz (2005: 132) included it with the carenines.
Relationships of the tribe have been little discussed. Sloane (1905b: 103) retained pasimachines and carenines under one family-group name implying a close relationship between the two groups. Nichols (1988a: 214) argued that Pasimachini is the sister-group to the tribe Carenini.
Tribe Carenini. This clade, which is endemic to Australia, includes about 195 species placed in ten genera. The genus Scaraphites Westwood (seven Australian species), usually listed as a member of this tribe (e.g., Lorenz 2005: 133), has been removed from it and placed in the tribe Scaritini by Moore and Lawrence (1994: 512). According to Moore and Lawrence (1994: 503), carenines represent the sister-group to the remaining Scaritini (sensu lato, i.e., pasimachines, scaritines, clivinines, dyschiriines).
Tribe Scaritini. This tribe, with about 495 species in 42 genera, is represented in all zoogeographical regions but is predominantly tropical. Four subtribes are currently recognized: Acanthoscelitina (one Afrotropical species in Acanthoscelis Dejean), Oxylobina (about 30 Oriental species in Oxylobus Chaudoir), Scapterina (about 25 Australian-Oriental-Afrotropical species in Passalidius Chaudoir, Scapterus Dejean, Parathlibops Basilewsky, Thlibops Putzeys, and Steganomma Macleay), and Scaritina (including the Madagascan storthodontines and dyscherines and the Afrotropical ochyropines [one species] and corintascarines [one species]), with the bulk of the species. Jeannel (1946: 220) assigned scapterines to his Clivinitae.
The North American fauna is represented by seven species of Scarites Fabricius.
Tribe Clivinini. This tribe is the most diversified group of the subfamily with about 60 genera. The inclusion of the clivinines within the Scaritinae has not been challenged often but parsimony analysis based on the female reproductive tract by Liebherr and Will (1998) suggests that clivinines could be more closely related to rhysodids than to scaritines. They also emphasized that the defensive secretions of the pygidial glands differ drastically between the two groups: clivinines use ketones or quinones while scaritines eject aliphatic acids.
At least three putative clades are recognized within the tribe and are usually ranked as subtribes. The forcipatorines, exclusively Neotropical, include the genera Camptidius Putzeys (one species), Camptodontus Dejean (14 species), Forcipator Maindron (four species), Kultianella Perrault (two species), Obadius Burmeister (two species), and Stratiotes Putzeys (two species). According to Perrault (1994: 686), members of this clade differ from those of remaining clivinines in having the ligula truncate and glabrous instead of prolonged and with at least one apical seta, the gula either vanishing posteriorly or very narrow instead of wide, the first antennomere asetose instead of having an apical seta (except in a few genera), the penultimate labial palpomere glabrous (except in one genus) instead of having two setae (except in some genera), and the clypeus glabrous (except in two species) instead of having a seta on each side. Another clade, the ardistomines, is restricted to the Western Hemisphere. Bousquet (2006c) restricted the group to the genera Ardistomis Putzeys, Semiardistomis Kult, and Aspidoglossa Putzeys whose members have a projection on pleurite VII. Kult (1950b) also included the genus Neoreicheia Kult. Whitehead (in Reichardt 1977: 386, 391) remarked that Oxydrepanus Putzeys was “doubtless related to Neoreicheia” and probably belonged to the ardistomine radiation along with such Old World genera as Reicheia Saulcy, Syleter Andrewes, and allies. Basilewsky (1973: 276) indicated that ardistomines are relatively closely related to dyschiriines. The third clade, the reicheiines, is represented only in the Old World and contains many genera including Reicheia Saulcy, Trilophus Andrewes, Typhloreicheia Holdhaus, Trilophidius Jeannel, and Leleuporella Basilewsky.
Iablokoff-Khnzorian (1960: 93) described a new genus, Dyschiriomimus, from Baltic amber which he viewed as an intermediate taxon between Dyschirius and Clivina. However, Fedorenko (1996: 37) believed the taxon is a typical clivinine more closely related to Trilophus and Oxydrepanus than to Clivina.
Tribe Salcediini. This tribe includes four genera placed in three subtribes following Bell (1998): Salcedia Fairmaire (nine Indo-African species) in Salcediina, Holoprizus Putzeys (one Amazonian species) and Solenogenys Westwood (three Brazilian species) in Solenogenyina, and Androzelma Dostal (one Vietnamese species) in Androzelmina. According to Bell (1998: 264), salcediines, forcipatorines, and clivinines form a well-defined clade supported by the following synapomorphies: the metepimeron is lobate and overlaps the anterior corners of abdominal sternum 2; the elytron possesses a ventral carina in form of a projected lobe which engages the dorsal angles of abdominal sternum VII; the labial pits on the mentum each have a posterior duct opening into the submental suture contrary to other scaritines in which the ducts open anteriad the suture and distant from it. Bell (1998: 265) also indicated that rhysodids shared three synapomorphies with salcediines (excluding Androzelma): a kind of coating on the exoskeleton, minute and retractile palpi, and a distinct lobe on which the eye is located. Furthermore among salcediines, members of Solenogenys share two synapomorphies with rhysodids: the medial margins of the ventral groove of the head are oblique, nearly straight, and meet near the “neck” condyle and the mandible has a dorsolateral lobe. Based on the above evidence, Bell (1998: 269) concluded that Solenogenys is the sister-group to rhysodids.
Tribe Dyschiriini. Relationships of this tribe within the Scaritinae are not documented. Fedorenko (1996: 37) suggested that dyschiriines share a common ancestor with clivinines but failed to disclose any characteristics that would support this claim. Jeannel (1946: 214) combined the ardistomines and dyschiriines in his Dyschiriitae.
Tribe Promecognathini. This small and well-defined tribe includes one genus with two species in western North America and four genera with six species in Cape Province in South Africa. Jeannel (1941b: 244; 1946: 206) postulated that promecognathines were closely related to siagonines without, however, offering any evidence. Lindroth (1961a: 125) and Kryzhanovskij (1976a: 88) associated promecognathines with scaritines (sensu lato) implying a close relationship between the two groups. Several apomorphic features, including details of the chaetotaxy, structure of the mouthparts and thorax, and marked similarity in their specialized way of attacking millipedes suggest that promecognathines could be closely related to peleciines. However, Straneo and Ball (1989) regarded the similarities between the two groups as evolutionary convergence.
McKay (1991) described a fossil from Cretaceous crater lake deposits at Orapa, Botswana, under the name Palaeoaxinidium orapensis, which he believed represents the sister-group to the Promecognathini.
Tribe Dalyatini. Molecular (Ribera et al. 2005) and morphological (Mateu and Bellés 2004) data suggest that the single, highly modified cave species of this tribe, Dalyat mirabilis Mateu from southeastern Spain, could be the sister-group to promecognathines.
Subfamily Broscinae. This subfamily includes a single tribe with about 290 species in 34 genera, arrayed in five subtribes (see Roig-Juñent 2000): Axonyina (five species), Broscina (about 75 species), Nothobroscina (about 90 species), Barypodina (about 25 species), and Creobiina (about 95 species). Broscines are represented in all major regions of the world, except the Afrotropical Region, but are more diverse in the Australian Region than anywhere else. They live almost exclusively in temperate areas, with only a few groups extending to the edges of the tropics. Several authors have suggested explicitly or implicitly that broscines are closely related to apotomines but Liebherr and Will’s (1998) morphological data on the female reproductive tract and the molecular sequence data provided by Maddison et al. (1999) did not support such an association. Erwin (1991a: 10) included broscines, apotomines, melaenines, and cymbionotines in his subfamily Broscinae.
Roig-Juñent’s (2000) parsimony analysis based on morphological characters of adults suggested that the three native North American genera (Broscosoma, Zacotus, and Miscodera) form a clade.
Subfamily Apotominae. This subfamily includes a single genus, Apotomus Illiger, with about 15 species in warm temperate and tropical regions of the Eastern Hemisphere and one species in Brazil. Kryzhanovskij (1976a: 88) and Moore et al. (1987: 122) associated apotomines with broscines and in several classifications these two groups are placed sequentially in the text. However, Roig-Juñent’s (1998: Fig. 10) parsimony analysis using 33 characters placed apotomines as the sister-group to melaenines (no Cymbionotum exemplars were included). Liebherr and Will (1998: 150) noted that apotomines do not have conjunct mesocoxae as in broscines and the other members of Jeannel’s Stylifera and that the placement of apotomines within the Stylifera should be rejected. They placed apotomines as a basal grade with clivinines and rhysodids but noted they could be closely related to scaritines and hiletines. Molecular data analyzed by Maddison et al. (1999: 128) did not provide support for a close relationship between apotomines and broscines.
Subfamily Siagoninae. This subfamily includes three genera, each arrayed in its own tribe: Enceladus Bonelli, Luperca Laporte, and Siagona Latreille. Relationships of the subfamily are obscure. Jeannel (1941b: 244; 1946: 206) associated siagonines with promecognathines. Erwin (1985: 467; 1991a: 9-10) listed siagonines with amarotypines, migadopines, elaphrines, promecognathines, hiletines, pseudomorphines, and scaritines (including cnemalobines, as Cnemacanthini) in his subfamily Scaritinae. The preferred cladogram of Liebherr and Will (1998: Fig. 57), based on the female reproductive tract, placed siagonines as the sister-group to the subfamily Carabinae. Some of the analyses on 18 rDNA performed by Maddison et al. (1999: 127) suggested that Siagona could be closely related to {Gehringia + Cymbionotum}. Based on a morphological study of larvae of Siagona and Enceladus, Grebennikov (1999: 9) did not find evidence to suggest a close relationship for siagonines.
Tribe Enceladini. Only the genus Enceladus belongs to this tribe, with one species found in the Guyana-Venezuelan area, possibly also in Amazonia (Reichardt 1977: 384).
Tribe Siagonini. This tribe contains only the genus Siagona with about 80 species in the Old World. Erwin (1978: 105) listed several apomorphic states shared by Siagona and Cymbionotum and stated that the two were undoubtedly closely related.
Tribe Lupercini. Two species of the genus Luperca are included in this tribe, one is found in tropical Africa, the other on the Indian subcontinent. Erwin (1978: 105) combined the genus with Siagona and Cymbionotum in his tribe Siagonini which he included in his subfamily Siagoninae along with the tribe Enceladini.
Subfamily Melaeninae. This subfamily includes one tribe with two genera: Melaenus Dejean with two species confined to the Oriental Region, Egypt, and the Afrotropical Region, excluding the Congo Basin and southern parts (Ball and Shpeley 2005: 37), and Cymbionotum Baudi di Selve with 20 species arrayed in two subgenera, Procoscinia Ball and Shpeley with two species in northern South America and Cymbionotum s.str. with 18 species confined to the warmer parts of the Old World. According to Ball and Shpeley (2005: 22), monophyly of this subfamily is indicated by the very long diverticulum of the spermathecal gland. Prior to these authors, the two genera had been variously classified. Several authors placed them in separate tribes though suggesting implicitly or explicitly that they were closely related (e.g., Erwin 1985: 469; Liebherr and Will 1998: 137). Others have separated the two rather widely. For example van Emden (1936a: 46) listed Melaenus in his Harpalinae piliferae from which the cymbionotines were excluded. Jeannel (1941b: 291-292) placed the genus Melaenus in his Psydritae along with psydrines, melisoderines (= moriomorphines), and meonines (= moriomorphines) and included Cymbionotum in a family-group taxon of its own which he considered closely related to siagonines (Jeannel 1946: 206).
Relationships of the subfamily are unclear. Liebherr and Will (1998: 150) suggested that Melaeninae could be closely related to Clivinini. Roig-Juñent’s (1998) parsimony analysis using 33 characters showed Melaenus to be the sister-group to apotomines; Cymbionotum exemplars were not included in his analysis.
This group has been reported in publications of the xix and early xx Centuries under the name Granigerini, because Graniger algirinus Motschulsky, the sole species included by Motschulsky in his new genus Graniger, was listed in synonymy with Coscinia semelederi Chaudoir (Chaudoir 1876d: 63). Because Coscinia Dejean was a junior homonym of Coscinia Hübner, Graniger Motschulsky became the valid name for this genus. However, Andrewes (1933: 3) showed that Motschulsky’s species was in fact identical with the ditomine Carterophonus femoralis Coquerel. Cymbionotum Baudi di Selve was the next available name for the species of Coscinia Dejean.
Subfamily Gehringiinae. This subfamily includes three genera placed in two subtribes: Gehringiina Darlington with a single western North American species, Gehringia olympica Darlington, and Helenaeina Deuve with four rarely collected species from Egypt, Turkey, Yemen, and Namibia placed in the genera Helenaea Schatzmayr and Koch and Afrogehringia Baehr, Schüle and Lorenz. The taxonomic position of the group is debated. Jeannel (1941b, 1946: 46) combined gehringiines with trachypachids and paussines (as Caraboidea Isochaeta) and both Lindroth (1969b) and Kryzhanovskij (1976a: 87) associated gehringiines with trachypachids. Bell (1967: 106) indicated that the form of the palpi and the anterior tibia suggest that gehringiines could be derived from the genus Tachys but he also raised the possibility that gehringiines could belong to his Hemipleuri, a group comprising the nebriines and carabines. In Nagel’s (1987: Fig. 2) cladistic analysis, gehringiines were positioned as the sister-group to {cicindines + paussines}. Beutel (1992c) indicated that the isochaetous protibia of gehringiines suggests that the group could be “an early offshoot of the metriine-paussine lineage.” In listing the tribe in his supertribe Psydritae, Erwin (1985: 468) suggested implicitly that gehringiines could be closely related to psydrines and patrobines and Lorenz (2005: 243) placed the tribe Gehringiini within the Psydrinae. Deuve (2005) made a detailed analysis of the morphology of gehringiine adults and concluded that several character states suggest “a very basal position in the phylogeny of adephagan Coleoptera” for gehringiines. However he also noted that the peculiar “abdominal type” found in the group is similar to that of the genus Cymbionotum. A close affiliation between Gehringia and Cymbionotum received support from the molecular analysis of Maddison et al. (1999). Arndt et al. (2005: 140) pointed out that the condition of the protibial spurs in gehringiines could not be unambiguously assigned to either of the two types found in other carabids. They noted that if the protibial spurs of gehringiines are considered to be of the isochaete type, then gehringiines could be the sister-group to Paussinae and if considered to be of the anisochaete type, they could be the sister-group to Nebriinae.
Subfamily Trechinae. Several authors agree that the tribes Trechini, Zolini, Bembidiini, and Pogonini are closely related and probably constitute a clade. Monophyly is supported by characteristic features of the adult morphology (Roig-Juñent and Cicchino 2001), larval morphology (Grebennikov and Maddison 2000: 226; 2005: 44), and 18S ribosomal sequence data (Maddison et al. 1999). In addition, males of Trechinae studied lack chiasmata in meiosis (Serrano 1981) contrary to most other Carabidae, a notable synapomorphy (Maddison and Ober 2011: 243). Jeannel (1941b: 299) also included mecyclothoracines in the subfamily (as Trechidae) but most recent authors place them within the Moriomorphinae. Deuve (1993: 156) included patrobines within the Trechinae.
As discussed under Patrobinae, this subfamily is probably the sister-group to patrobines.
Tribe Trechini. A relatively well-defined and very diverse group with more than 2, 500 species currently arranged in 170 genera or so. Although represented in all zoogeographical regions, the tribe is more diverse in the temperate zones than in the tropics. Many species are endogean or troglodytic and flightless. Casale and Laneyrie (1982: 7) classified the Trechini into six groups placed in two major complexes, one including cnidines, trechodines, and plocamotrechines characterized by the median lobe of the aedeagus being wide open dorsally, the basal orifice lying between two symmetric lobes, and one comprising the perileptines, aepines, and trechines with the median lobe partly closed dorsally, the basal orifice opening on the ventral surface of the basal bulb. However, this classification has been challenged in recent times. Uéno (1989: 12-13) presented arguments to combine cnidines with perileptines and Grebennikov and Maddison (2005: 46-47) found evidences in the larval characters that perileptines were closely related to trechodines. In this work, the Trechini are grouped into two subtribes: Trechodina (including perileptines, cnidines, and plocamotrechines) and Trechina (including aepines).
Based on larval character states, Grebennikov and Maddison (2005) suggested that Trechini is the sister-group to {Zolini + Bembidiini + Pogonini}. Arndt’s (1993: 33) analysis of larval characters suggested that trechines are closely related to tachyines.
The North American fauna comprises about 225 species grouped into nine genera, all belonging to the subtribe Trechina. Barr (1985a: 351) recognized four series among the North American genera, the Trechus series with Trechus, the Trechoblemus series with Trechoblemus, Blemus, Pseudanophthalmus, Neaphaenops, and Nelsonites, the Darlingtonea series with Darlingtonea and Ameroduvalius, and the Aphaenops series with Xenotrechus. The genus Pseudanophthalmus, whose members are cave inhabitants except for a few rare occurrences in forest floor humus and in abandoned coal mines, is closely related to Duvaliopsis Jeannel which includes six edaphic species in the Carpathian and Transylvanian Alps of eastern Europe. In fact, Barr (2004: 7) listed Duvaliopsis as a junior synonym of Pseudanophthalmus since both genera are not readily separable on purely morphological grounds. Xenotrechus, with two species in southeastern Missouri caves, is apparently the sister-group to the monospecific genus Chaetoduvalius Jeannel (Barr 2004: 10) of the Apuseni Mountains in the western Carpathians, Romania.
Tribe Zolini. The 57 species of this tribe are currently arrayed in ten genera and three subtribes: Zolina with 50 species in South America (genus Merizodus Solier) and the Australian Region (genera Oopterus Guérin-Méneville, Zolus Sharp, Synteratus Broun, Percodermus Sloane, Idacarabus Lea, Sloaneana Csiki, and Pterocyrtus Sloane), Sinozolina for the genus Sinozolus Deuve (six Chinese species), and Chalteniina for Chaltenia patagonica Roig-Juñent and Cicchino of Argentina. Jeannel (1962) recognized two lineages within Zolina based on structural features of the male genitalia.
Tribe Bembidiini. This relatively well-defined tribe is represented in all zoogeographical regions of the world. Adults possess characteristic subulate apical palpomeres (except in Horologion), a condition found otherwise only in gehringiines and a few trechines. Bembidiines are grouped into six subtribes: Bembidiina, with about 1, 350 species, is distributed worldwide but is more diverse in the temperate regions than in the tropics; Xystosomina is represented in the New World and tropical Australia (Erwin 1994: 560) by about 125 species with only one (Mioptachys flavicauda) found in North America; Tachyina (including lymnastines) with nearly 800 species is also worldwide but, contrary to Bembidiina, is more diverse in the tropics; Anillina with about 375 minute, apterous, and blind species is distributed in all zoogeographical regions; Horologionina with a single cave-inhabiting species, known only from the holotype collected in West Virginia; and Lovriciina represented by four cavernicolous species, placed in three genera (see Giachino et al. 2011), found in the Balkans. Erwin (1982b: 459) postulated that anillines and horologionines represent a grade of several lineages derived from Paratachys Casey and allies, a hypothesis refuted by Maddison and Ober (2011: 249). Arndt (1993: 33) found a number of putative synapomorphies in larvae of Tachyina and Trechini and suggested that the two taxa are sister-groups. Grebennikov (2002) and Grebennikov and Maddison (2005), working with larvae, found evidence suggesting that Anillina is the sister-group to {Tachyina + Xystosomina} and that xystosomines are probably nested within the tachyines. Van Emden (1936a) suggested that Horologion Valentine was closely related to psydrines and Jeannel (1949b: 93) believed it could be related to patrobines.
Tribe Pogonini. This tribe is found in all zoogeographical regions of the world but is more diverse, both in terms of species and lineages, in the Palaearctic Region. All 83 species currently recognized are more or less halobiont and live along sea coasts or near salt lakes. Jeannel (1941b: 552) stated that this group is related to mecyclothoracines (currently placed in the subfamily Moriomorphinae) of the Hawaiian islands and the Australian Region. Müller (1975) postulated that Pogonini is the sister-group to Bembidiini. Based on karyotypic data, Serrano and Galián (1998: 196) suggested that pogonines are closely related to Bembidiina. Arndt (1993: 33), working on larval characters, suggested a close relationship between pogonines and Bembidiini (excluding tachyines).
Subfamily Patrobinae. This subfamily, which includes the tribes Lissopogonini and Patrobini, is considered to be the sister-taxon to Trechinae by several authors based on male tarsal structure (Müller 1975), larval characteristics (Arndt 1993: 32), and similar abdominal morphology (Deuve 1993). This association is also supported by molecular sequence data (Maddison et al. 1999: 128; Maddison and Ober 2011: 243). Erwin (1985: 469) and Baehr (1998: 363) suggested that patrobines may be closely related to Moriomorphinae. Jeannel (1941b: 80-81) placed patrobines in his Limbata Stylifera along with apotomines, broscines, psydrines, moriomorphines, melaenines, trechines, bembidiines, pogonines, and zolines but indicated that some character states, particularly of the larvae, suggest that they may belong to the Limbata Conchifera.
This subfamily is found in the Northern Hemisphere and Oriental Region. Only the tribe Patrobini is represented in North America.
Tribe Lissopogonini. This tribe includes a single genus, Lissopogonus Andrewes, with eight species in Asia. The genus was originally described in the tribe Pogonini and subsequently transferred to the tribe Patrobini by Zamotajlov and Sciaky (1996: 40). Bousquet and Grebennikov (1999: 11) alluded to the possibility that Lissopogonus could be a highly derived taxon related to Patrobus and Platypatrobus based on the shared apomorphic condition of the median sulcus of the pronotum being wide and deep in the basal fifth and reaching the basal edge. Deuve and Tian (2002: 30) suggested that the genus could belong at the base of the Trechinae and Patrobinae lineages (their Trechidae).
Tribe Patrobini. The 215 species or so listed in this tribe are currently arrayed in four subtribes: Deltomerina with the genus Deltomerus Motschulsky only, Deltomerodina with the genus Deltomerodes Deuve, Patrobina with 18 genera, and Platidiolina with Platidiolus Chaudoir. In a cladistic analysis conducted by Roig-Juñent and Cicchino (2001: Fig. 1), this tribe is positioned as the sister-group to Amblytelini (currently included in the Moriomorphinae).
Subfamily Psydrinae. Following Maddison and Ober (2011: 237), this subfamily is restricted to the tribe Psydrini and includes only six species. Two (Laccocenus ambiguus Sloane and Laccocenus vicinus Moore) lives in southeastern Australia, another one (Psydrus piceus LeConte) ranges widely across the northern parts of North America, extending southwards to the mountains of northern California, Arizona, and New Mexico, and the other three, all members of the genus Nomius Laporte, are restricted to central Africa and Madagascar (two species) or to the Northern Hemisphere although apparently extinct in Asia (Nomius pygmaeus Dejean). Baehr’s (1998: Fig. 1) preliminary cladistic analysis using 19 characters of adults suggested that Psydrini could be the sister-group to {Patrobinae + the remaining Psydrinae [= Moriomorphinae]}. Relationships among the three genera of Psydrini have not been investigated.
Subfamily Moriomorphinae. Members of this subfamily were traditionally included in the Psydrinae but recent morphological (Baehr 1998) and molecular data (Maddison and Ober 2011: 237) studies suggest that the Moriomorphinae form a clade and that the group is not closely related to the true Psydrinae. Baehr (1998: 363) argued that Patrobinae could be the sister-group to Moriomorphinae. Many moriomorphines are similar to pterostichines in body form but the presence of a scrobal seta and setose parameres in almost all moriomorphines, unlike pterostichines, suggest that they are probably not closely related. Ober’s (2002) phylogenetic analysis based on molecular sequence data suggested that the subfamily Moriomorphinae, termed “austral psydrines, ” could be the sister-group to {Brachininae + Harpalinae}.
This subfamily, which includes about 470 species, is represented only in the Southern Hemisphere and is particularly diverse in the Australian Region. Five tribes were traditionally recognized (see Baehr 2004): Mecyclothoracini with about 285 species placed in the genera Neonomius Moore and Mecyclothorax Sharp; Meonini with about 20 species in the genera Raphetis Moore, Meonis Laporte, Selenochilus Chaudoir, and Meonochilus Liebherr and Marris; Moriomorphini with six species in five genera, all endemic to southeastern Australia; Tropopterini with about 50 species in seven genera; and Amblytelini with six genera and about 95 species endemic to Australia, including Tasmania. Recently, Liebherr (2011) proposed an entirely new classification, dividing the moriomorphines into two groups based on characters of the parameres. His classification is adopted here.
The genus Bembidiomorphum Champion (two species in Chile), included in this group since van Emden (1936a: 51), belongs to the Broscini (Roig-Juñent et al. 2008: 212).
Tribe Moriomorphini. This group includes about 55 species, all endemic to the Australian Region, placed in 13 genera: Celanida Laporte (one species), Melisodera Westwood (three species), Molopsida White (28 species), Moriodema Laporte (two species), Moriomorpha Laporte (one species), Neonomius Moore (three species), Pterogmus Sloane (one species), Rhaebolestes Sloane (two species), Rossjoycea Liebherr (one species), Sitaphe Moore (eight species), Teraphis Laporte (six species), Theprisa Moore (three species), and Trephisa Moore (one species). These species are characterized by having elongate, parallel-sided parameres that are glabrous or sparsely clothed with very short setae.
Tribe Amblytelini. This tribe contains about 415 species in 12 genera: Amblytelus Erichson (43 species), Dystrichothorax Blackburn (48 species), Epelyx Blackburn (five species), Mecyclothorax Sharp (about 280 species), Meonis Laporte (16 species), Meonochilus Liebherr and Marris (six species), Paratrichothorax Baehr (one species), Pseudamblytelus Baehr (one species), Raphetis Moore (three species), Selenochilus Chaudoir (six species), Trichamblytelus Baehr (one species), and Tropopterus Solier (four species). These species are restricted to Australia and New Zealand except those of Mecyclothorax which occur also in New Guinea, Borneo, Java, and the Polynesian islands in the Pacific Ocean and Tropopterus which are found in Chile and Peru. Amblytelines differ from members of Moriomorphini by having more setose parameres that are either shorter, basally broader and narrowly rounded apically or elongate with whiplike apex.
Subfamily Nototylinae. This subfamily includes a single species, Nototylus fryi (Schaum), known only from the female holotype collected in the state of Espírito Santo, Brazil, in the xix Century. The species is aberrant structurally: it lacks the grooming structures of the protibiae present in all other Geadephaga except Paussini and lacks the pubescence on antennomeres 5-10 which is present in other Geadephaga except Trachypachidae, Rhysodidae, and Gehringiinae (Deuve 1994b: 141). Bänninger (1927) suggested that Nototylus Gemminger and Harold was related to Ozaenini, Kryzhanovskij (1976a: 87) associated it with paussines (excluding metriines) and cicindines, and Erwin (1979: 591) postulated that the species was an independently adapted myrmecophile from an ozaenine stock. However, Ball (1979: 100) doubted the possibility of a close affinity between nototylines and paussines as suggested by the above-mentioned authors. Deuve (1994b) published a detailed description of the structural character states of the species and suggested, but with some doubt, a sister-group relationship between nototylines and paussines. He noted several synapomorphies between the two groups including the compressed protibia, the tergite IX which is differentiated into a thin transverse arch, the reduced and lateral position of the laterotergite IX, and the diffuse dorsal pubescence.
Subfamily Paussinae. There is little doubt that this subfamily constitutes a monophyletic lineage. The known larvae share a unique transformation of the abdomen in which the epipleurites of the 9th segment are greatly enlarged and fused with the tergum of the 8th segment to form a plate, displacing the urogomphi and the 10th segment in a vertical plane (Bousquet 1986). The relationship of the subfamily is highly debated but it could be closely related to brachinines. Adults of both groups possess a two-chambered pygidial gland which produces a quinonoid secretion by mixing hydroquinones and hydrogen peroxide from the inner chamber with enzymes produced in the outer chamber (Schildknecht and Holoubek 1961); the secretion is discharged at temperatures of 55-100°C (Aneshansley et al. 1969; Aneshansley et al. 1983). The structure of the pygidial glands and the chemistry of the secretions are unique among beetles. However, based on structural dissimilarities, several authors, including Ball and McCleve (1990), Beutel (1992b), and Geiselhardt et al. (2007), believed that the similarities in the pygidial gland structures and secretions between the two groups are convergent.
Erwin and Stork (1985: 445) concluded that paussines and brachinines are closely related and form the sister-group to a large clade comprising {Elaphrini + Migadopini + Siagonini + Promecognathini + Hiletini + Pseudomorphini + Cnemacanthini (= Cnemalobini) + Scaritini} based on a suite of character states associated with tarsal claws. Deuve (1988), working on the structures of the last abdominal segments of adults, supported the view of a close relationship between paussines and brachinines. However, alternate placements of the paussines have been proposed. Jeannel (1941b: 89) placed trachypachids, gehringiines, and paussines in his Isochaeta based on the apical position of both protibial spurs. Kryzhanovskij (1976a: 87), followed by Lawrence and Newton (1995), included the Cicindini and Nototylini within the Paussinae, implying a close relationship between these three elements. Beutel (1995) suggested a close affinity between paussines and gehringiines. Liebherr and Will’s (1998) preferred cladogram based on 20 characters of the female ovipositors and reproductive tract placed paussines as the sister-group to the remaining Geadephaga (excluding trachypachids). An interesting observation is that of Vigna Taglianti and Rossi (1998: 516) who noted the similarity between the laboulbeniale parasitic species found on the brachinine Pheropsophus Solier and paussine Pachyteles Perty. They added that paussines and brachinines “might be more closely related than suggested by morphological data, thus supporting the result of recent biochemical studies on explosive secretions of members of these groups.”
Members of this subfamily are currently arrayed in five family-group taxa which have been ranked differently during the past few decades. In this catalogue, they are ranked as tribes. All five are probably monophyletic except for the Ozaenini which is likely paraphyletic. The phylogenetic relationships among extinct and extant genera have been expressed in a cladogram based on adult and larval characters by Geiselhardt et al. (2007: Fig. 1).
Tribe Metriini. This group includes two genera: Metrius Eschscholtz, with two species in western North America, and Sinometrius Wrase and Schmidt with a single species recently found in Hubei province in China. This tribe is usually listed as the sister-group to the remaining paussines because of the lack of the apico-lateral fold on each elytron (flange of Coanda of Stork 1985) characteristic of the remaining paussines. This fold, located at the opening of the defence gland, is apparently used to deflect discharges of secretions from the defence glands as showed by Eisner and Aneshansley (1982) for the Neotropical genus Goniotropis Gray. However, Vigna Taglianti et al. (1998: 292), based on a set of 20 larval characters, considered {Metriini + Ozaenini} as the sister-group to Paussini, suggesting that the elytral fold was secondarily lost in metriines or that the fold evolved twice in the subfamily.
Tribe Mystropomini. This tribe includes only the genus Mystropomus Chaudoir, with two Australian species. It is probably the most primitive extant genus of the subfamily excluding metriines. Adult ozaenines, protopaussines, and paussines (sensu stricto) are synapomorphic in having the elytral fold short, the pterothorax and abdomen parallel-sided and the epimera and anepisterna largely covered by the elytral epipleura (Beutel 1992c: 56). In adults of Mystropomus the elytral fold is markedly long and extends over the apical half of the elytron (Jeannel 1946: 47).
Tribe Ozaenini. This group of about 160 species is mainly represented in the tropics; only a few species enter the southern parts of the Northern Hemisphere in Japan, China, Taiwan, and southern United States. Ozaenines differ from protopaussines and paussines by having the mouthparts not modified, and from paussines also in having all 11 antennomeres normally developed. Several authors (e.g., Ball and McCleve 1990; Nagel 1997: 356; Di Giulio and Moore 2004) believed that ozaenines are paraphyletic in regard to the remaining Paussinae (excluding mystropomines and metriines). Beutel (1992b; 1995) and Di Giulio et al. (2003) proposed that the ozaenine genus Physea Brullé is the sister-group to {protopaussines + paussines} while Ball (in Nagel 1997: 356) regarded Ozaena Olivier as the best candidate based on the enlarged first antennomere and the reduced antennal cleaner of the protibia.
Tribe Protopaussini. This tribe includes eight extant Asian species placed in the genus Protopaussus Gestro. Some authors (e.g., Basilewsky 1953a: 23, 1962a: 6-9; Nagel 1987: 27) associated protopaussines with ozaenines based on the presence of 11 antennomeres in both groups but most have associated them with paussines. Nagel (1997: 348, 356) did not find any derived character states shared between protopaussines and ozaenines but noted that the small lacinia lacking the dense brushlike pilosity, typical of other carabids, is a putative synapomorphy for protopaussines and paussines (sensu stricto). From a zoogeographic point of view, it is interesting to note that a Tertiary fossil species of Protopaussus has been described from Dominican amber (Nagel 1997).
Tribe Paussini. This group, also known under the vernacular name “ant nest beetles, ” currently includes about 565 myrmecophilous species arrayed in this work in seven subtribes: Carabidomemnina for the genera Eohomopterus Wasmann (two Neotropical species) and Carabidomemnus Kolbe (27 African species); Arthropterina for the Australian genera Megalopaussus Lea (one species) and Arthropterus Macleay (about 65 species); Cerapterina for the genera Mesarthropterus Wasmann (one species in Ethiopia) and Cerapterus Swederus (32 species in the Afrotropical and Oriental Regions with two species extending into the Himalayas); Pentaplatarthrina for the genera Hexaplatarthrus Jeannel (one Madagascan species) and Pentaplatarthrus Westwood (eight Afrotropical species); Homopterina for the genus Homopterus Westwood (12 Neotropical species); Heteropaussina for the genus Heteropaussus Thomson (about 25 species in the Afrotropical and Oriental Regions); and Paussina for the remaining 12 genera (about 385 species). Luna de Carvalho (1989: 361) used a different approach and recognized three tribes among his extant Paussinae (Paussini in this work): Cerapterini (including carabidomemnines, homopterines, heteropaussines, and arthropterines), Pentaplatarthrini, and Paussini. Within his Paussini, he included the following subtribes: Platyrhopalina for the Asian genera Platyrhopalopsis Desneux (three species), Platyrhopalus Westwood (14 species), Stenorhopalus Wasmann (two species), Lebioderus Westwood (seven species), and Euplatyrhopalus Desneux (six species); Ceratoderina for the genera Paussomorphus Raffray (three Afrotropical species), Melanospilus Westwood (three Oriental species with one species extending into the Himalayas), and Ceratoderus Westwood (seven Asian species); Leleupaussina for the genus Leleupaussus Luna de Carvalho (one Afrotropical species); Hylotorina for the Afrotropical genera Granulopaussus Kolbe (four species), Hylopaussus Luna de Carvalho (two species), and Hylotorus Dalman (six species); and Paussina for numerous genera that some authors sink into one large genus, Paussus Linnaeus (about 330 species in the Old World of which only two, Paussus favieri Fairmaire and Paussus turcicus Frivaldszk von Frivald, reach Europe). Nagel (1987, 1997, as Carabidomemnitae) viewed the Carabidomemnina as the sister-group of the remaining Paussini.
Subfamily Brachininae. There is little doubt that this group, known under the vernacular name “bombardier beetles, ” constitutes a monophyletic lineage. The adults have seven (females) or eight (males) exposed abdominal sterna instead of six as in other carabids. Such modification provides a greater abdominal mobility, allowing a more efficient alignment of the defence spray. However, brachinines do not appear monophyletic in terms of their 18S rDNA (Maddison et al. 1999: 129). The group has a worldwide distribution but is clearly more diverse in the Southern Hemisphere. Most authors recognize two main lineages, ranked here as tribes, among brachinines: Brachinini, represented in most regions of the world including North America, and Crepidogastrini, restricted to southern India and Africa.
For a long time brachinines have been associated with the “Truncatipennes, ” an informal name use to group several tribes whose adults have more or less truncate elytra at the apex. Jeannel (1942, 1949a) included brachinines and pseudomorphines in his Balteifera, implicitly suggesting a close affinity between the two groups. Liebherr and Will (1998: 152-153) placed brachinines with the {Harpalinae + Trechinae + Moriomorphinae}in their study of the female reproductive tract. These authors also alluded to the possibility of a close relationship between brachinines and clivinines. Analysis of molecular data presented by Ribera et al. (2005: 289) indicated a close relationship between brachinines and the subfamily Harpalinae, not with the Paussinae. Maddison et al. (1999: 129) suggested, from 18S r-DNA sequence analyses, an intriguing possibility, that the paussines and brachinines are closely related and that both in turn are related to Harpalinae. In my opinion their hypothesis is credible.
Tribe Crepidogastrini. This tribe is mostly represented in the Afrotropical Region but a few species are found in the Indian subcontinent. It contains the genera Brachynillus Reitter (three species), Crepidogaster Boheman (about 100 species), Crepidogastrillus Basilewsky (one species), Crepidogastrinus Basilewsky (one species), Crepidolomus Basilewsky (two species), and Crepidonellus Basilewsky (five species).
According to Erwin (1970a: 27), adults of crepidogastrines differ from those of brachinines in having the mesepimeron absent or almost so (instead of broad), the adhesive setae on the male protarsi of the “spongy” type (instead of the “seriate” type), the terminal palpomeres swollen and usually securiform (instead of subcylindrical or wedge-shaped), and the gular suture convergent behind (instead of divergent).
Tribe Brachinini. This tribe includes about 540 species of which 50, all belonging to the genus Brachinus Weber, occur in North America. Erwin’s (1970a: 175) study suggested that all New World species of Brachinus, along with a relict species found in the Himalayas, form a clade for which he proposed the subgeneric name Neobrachinus. He also postulated that the subgenus Cnecostolus Reitter, endemic to the Palaearctic Region, was the sister-group to Neobrachinus. Erwin (1970a: 28) arrayed the brachinine genera into four subtribes: Aptinina, Brachinina, Mastacina, and Pheropsophina. In his cladistic analysis (Erwin 1970a: Fig. 451), masticines were positioned as the sister-group to pheropsophines and the two form the sister-group to {aptinines + brachinines}.
Unlike most carabid larvae, those of brachinines are ectoparasites and feed on carabid and water beetle pupae.
Subfamily Harpalinae. Harpalinae is the largest subfamily of Carabidae and the one usually placed at the end of the carabid classification. Molecular data analyses (Maddison et al. 1999; Ober 2002; Ribera et al. 2005) suggest that the subfamily is monophyletic.
In this catalogue, members of Harpalinae are arrayed conveniently in two supertribes: Pterostichitae and Harpalitae. Adults of the vast majority of Pterostichitae, which includes the tribes Morionini, Abacetini, Pterostichini, Zabrini, Oodini, Panagaeini, and Chlaeniini in North America, have crossed epipleura and most secrete something else than formic acid as major constituent of the pygidial glands. Adults of Harpalitae have non-crossed epipleura and, except in the sole species of Pentagonicini studied, secrete formic acid as major constituent of their pygidial glands as far as known. The absence of a crossed epipleuron could be an evolutionary feature providing greater flexibility to aim the powerful formic acid secretion of the pygidial glands. The presence of a transverse membranous band on the stipes of larvae prompted Arndt (1998: 184) to suggest that the tribes Licinini and Harpalini, herein included in the Harpalitae, were closely related to members of Pterostichitae.
Tribe Morionini. This relatively well-defined, likely monophyletic tribe is represented in all zoogeographical regions of the world but is more diverse in the tropics than in temperate areas. Its relationships have been debated. Indeed, some larval character states suggest that morionines could be related to scaritines while some adult character states suggest they may be related to pterostichines. Bousquet (2001) discussed the larval character states of morionines in detail and concluded that they do not yield evidence to favor one hypothesis over the other. However, when features of the adults are also taken into account, there is little doubt that morionines are more closely related to pterostichines than to scaritines. Recently Will (2004: 218), following Liebherr and Will (1998: 156), found three “unambiguously optimized and unreversed synapomorphies” suggesting that cnemalobines and morionines are sister-groups. A review and cladistic analysis of the morionine genus-group taxa have been published recently (Will 2004).
Moore (1965: 5) included the Australian genus Catadromus Macleay (seven species) in the tribe Morionini but his view has not been retained by subsequent authors.
Tribe Cnemalobini. This tribe includes only the Neotropical genus Cnemalobus Guérin-Méneville (32 species in Chile, Argentina, and Uruguay). Jeannel (1941b: 286) stated that the genus should be placed near the perigonines and Reichardt (1977: 416) followed his suggestion. Erwin (1985: 467) associated cnemalobines (as Cnemacanthini) with scaritines and clivinines. Arndt (1993: 40) suggested that the tribes Cnemalobini and Harpalini form a clade based on larval characteristics. Roig-Juñent (1993: 12) suggested, from a preliminary analysis, that cnemalobines and zabrines are sister-groups and the two groups are closely related to morionines. Other cladistic analyses (Liebherr and Will 1998: 156; Will 2004: 217) placed morionines as the sister-group to cnemalobines. Molecular data (18S rDNA) analyses (Maddison et al. 1999: 129) did not endorse placement of cnemalobines with Scaritinae but supported an association with the subfamily Harpalinae.
Tribe Microcheilini. This tribe includes a single genus, Microcheila Brullé, with two Madagascan species. Besides their relatively aberrant facies, adults of this group possess a number of character states unusual for pterostichines. The penultimate labial palpomere has more than two setae, each sternum possesses a transverse row of setae, the protibia has a latero-apical dentiform protuberance, all tarsomeres are densely pubescent beneath, and the first four protarsomeres of the male have adhesive setae (Jeannel 1948a: 616). The elytral plica is well developed as in members of Pterostichini. The group was included, along with morionines, chaetodactylines, and pterostichines (including sphodrines and platynines), in Jeannel’s (1948a: 380) family Pterostichidae.
Tribe Chaetodactylini. This group includes a single genus, Chaetodactyla Tschitschérine with 20 species endemic to Madagascar. The species superficially resemble several pterostichine taxa but the male protarsomeres are not expanded and have no adhesive setae (Jeannel 1948a: 619). The group was associated with morionines, metiines, zabrines, microcheilines, and pterostichines (including sphodrines and platynines) in Jeannel’s (1942: 734-735) family Pterostichidae.
Alluaud (1935: 28) reported that one of his colleagues rearing pupae of various insect groups for parasitic Hymenoptera discovered 14 adults of Chaetodactyla emerging from pupal chambers of two cetonid species. Jeannel (1948a: 620) postulated that Chaetodactyla females probably lay their eggs on the cetoniid larvae and that the carabid larvae remain inside the cetonid pupae, eventually feeding upon them.
Tribe Cratocerini. This tribe includes the genera Cratocerus Dejean with two Neotropical species and Brachidius Chaudoir with one australo-oriental species. Chaudoir (1873a) also listed Basoleia Westwood (= Catapiesis Solier) in this tribe and Lorenz (2005: 248) also included the genus Oxyglychus Straneo, with one Japanese species, previously included within the caelostomines (= drimostomatines). Cratocerines have been little studied and their taxonomic position is not well established. They are usually placed within the Pterostichini (e.g., Reichardt 1977: 407). Lorenz (2005: 248-252) combined cratocerines with catapieseines and drimostomatines in his subfamily Pterostichinae.
Tribe Abacetini. This tribe is proposed here to include the abacetines proper, the loxandrines, and the celioscheseines based on a preliminary cladistic analysis conducted by Will (2000) suggesting that these three groups are closely related. Van Emden (1949) and Arndt (1988) had already drawn attention to the fact that some putative apomorphic character states were shared by abacetines (with more than 95% of the species endemic to the Old World) and loxandrines (with more than 95% of the species restricted to the New World). As defined here, this tribe, as well as all three groups included in it, is inadequately characterized except for some abacetine genera which have an asymmetrical insertion of the second antennomere in the adults, and some loxandrine genera which have the first three protarsomeres of the males obliquely expanded. Monophyly of this tribe has not yet been demonstrated.
Tribe Pterostichini. This highly diverse tribe is represented in all continents, except Antarctica, and the species are found from the arctic regions to the tropics. There are no structural features yet discovered to suggest that the tribe, as currently conceived, forms a clade and there is little doubt, as suggested by Ball (1979: 102), that it represents a grade.
A number of putative clades have been recognized within the pterostichines and some of them have received formal scientific names. These include, among others, the euchroines with the genera Bothynoproctus Tschitschérine (one Neotropical species), Euchroa Brullé (38 Neotropical species), Lobobrachus Sharp (two Neotropical species), Setalis Laporte (three Australian species) and, according to Will (2000: 64), Microcephalus Dejean (15 Neotropical species); the Northern Hemisphere myadines with the genus-group taxa Aristochroa Tschitschérine (18 Asian species), Myas Sturm (with about 30 species in North America and Asia placed in the subgenus Trigonognatha Motschulsky and one European species), Steropanus Fairmaire (11 Asian species, some of them endemic to the Oriental Region), and Xenion Tschitschérine (one European species) to which Stereocerus Kirby (two Holarctic species) is probably closely related (Bousquet 1999: 85); the trigonotomines (including deliniines) with the genera Delinius Westwood (three Australian species), Leiradira Laporte (12 Australian species), Lesticus Dejean (about 100 Asio-Australian species), Trigonotoma Dejean (about 55 Asian species), and Euryaptus Bates (six Asian species), Pareuryaptus Dubault, Lassalle and Roux (17 Asian species); the Australian darodiliines (including cratogastrines) with the genera Loxogenius Sloane (one species), Liopasa Tschitschérine (one species), Cratogaster Blanchard (five species), and Darodilia Laporte (ten species); the New Caledonian abacomorphines with the genera Abacoleptus Fauvel (three species), Abacomorphus Chaudoir (two species), Platysmodes Fauvel (one species), and Setalidius Chaudoir (two species); the molopines with the North American genus Cyclotrachelus Chaudoir (45 species) and the western Palaearctic genera Abax Bonelli (18 species), Henrotiochoromus Busulini (one species), Molopidius Jeannel (one species), Molops Bonelli (40 species), Oscadytes Lagar Mascaro (one species), Percus Bonelli (19 species), Speomolops Patrizi (one species), Stenochoromus Miller (one species), Styracoderus Chaudoir (three species), Tanythrix Schaum (three species), Typhlochoromus Moczarski (two species), and Zariquieya Jeannel (one species) to which Jeannel (1948a: 450-451) added several Madagascan genera (Abacodes Jeannel, Eucamptognathus Chaudoir, Eudromus Klug, Eurypercus Jeannel, and Molopinus Jeannel); and the poecilines as defined by Jeannel (1942: 738) with the genera Stomis Clairville, Pedius Motschulsky, Argutor Dejean, Orthomus Chaudoir, Poecilus Bonelli, Phonias des Gozis, Bothriopterus Chaudoir, and Melanius Bonelli. Some of these groups, such as the poecilines, are probably polyphyletic.
Tribe Zabrini. Zabrines are most diversified in the Palaearctic and Nearctic Regions but are also represented in the mountains of the northern Neotropical, northern Oriental, and eastern Afrotropical Regions. Some authors have recognized several, more or less clearly defined genera in this tribe, others only two, Amara Bonelli and Zabrus Clairville, each with many subgenera. Adults of zabrines are structurally most similar to members of Pterostichini and probably represent a clade within the Pterostichini as presently conceived.
Tribe Metiini. This tribe includes about 75 species restricted to the southern part of South America, predominantly in Chile and extending north to Peru and east to southern Brazil, Uruguay, and Argentina. These species are arrayed in the following genera: Kuschelinus Straneo (one species), Metius Curtis (about 60 species), Abropus Waterhouse (one species), Antarctiola Straneo (four species) and, according to Will (2000: 60), Feroniola Tschitschérine (nine species). Metiines are often included within the Pterostichini.
This tribe has been known in the past under the name Antarctiini. However, because its type genus Antarctia Dejean is a junior homonym, the family-group name Antarctiini is permanently invalid (ICZN 1999: Article 39).
Tribe Drimostomatini (including cyrtolaines). The association of the Eastern Hemisphere drimostomatines (also known under the name caelostomines) with the Western Hemisphere cyrtolaines (Cyrtolaus Bates with 11 Middle American species and Barylaus Liebherr with two species in the West Indies) was proposed by Liebherr (1986) and supported by Will’s (2000) preliminary cladistic analysis. The main characteristic of this group is the inverted aedeagus. However, this modification is absent in some groups (e.g., Diceromerus Chaudoir) traditionally placed within the drimostomatines and consequently monophyly of this tribe is uncertain. The drimostomatines include about 290 species arrayed in 29 genera (Lorenz 2005: 248-252, as Drimostomatina). The most speciose genera are Caelostomus Macleay (about 160 species, of which one is adventive in the West Indies), Trichillinus Straneo (21 species), Platyxythrius Lorenz (20 species), and Strigomerus Chaudoir (18 species).
The name Caelostomini, proposed by Burgeon (1935: 194), is often used for this tribe but Drimostomatini, established by Chaudoir (1872c: 283), is older and has priority. Drimostoma Dejean is usually treated as a junior synonym of Caelostomus Macleay but the family-group name Caelostomini was not proposed because of the synonymy of the type genus. Therefore, Article 40.2 of the ICZN (1999) does not apply in this case.
Tribe Chaetogenyini. This South American tribe includes five species of the genus Chaetogenys van Emden arrayed in two subgenera: Chaetogenys s.str. and Camptotoma Reiche. The group has been ranked as a subtribe of Pterostichini by some authors, including van Emden (1958), Straneo (1977), and Reichardt (1977: 408). However, the adhesive setae on the male protarsi are of the “spongy” type (Reichardt 1977: 408), not of the “seriate” type as in other pterostichines. Erwin (1985: 468) associated chaetogenyines with cuneipectines, chlaeniines, oodines, and licinines.
Tribe Dercylini. The 35 species of this exclusively Neotropical tribe are currently arrayed in one genus (Dercylus Laporte) with four subgenera (Moret and Bousquet 1995: 759): Asporina Laporte (two species), Dercylus s.str., with Dercylodes Chaudoir and Pterodercylus Kuntzen as synonyms (12 species), Eurydercylus Moret and Bousquet (seven species), and Licinodercylus Kuntzen, with Physomerus Chaudoir (a junior homonym) as synonym (14 species). Chaudoir (1883), Reichardt (1977), and Ball (1979: 102) suggested that dercylines were closely related to oodines. Moret and Bousquet (1995: 759) stated that the character states of the adult and of the putative larva studied indicate that dercylines are more closely related to oodines and chlaeniines than to pterostichines. Bousquet (1996a: 449) commented that dercylines were closely related to {oodines + panagaeines + chlaeniines} but that the nature of the relationship remained to be ascertained. Jeannel (1948a: 626) related dercylines to melanchitonines and Kryzhanovskij (1976a: 89) to pterostichines, microcheilines, chaetodactylines, platynines, zabrines, and cuneipectines without mentioning any character state that would justify such grouping. The adhesive setae on the male protarsi are of the “spongy” type as in chaetogenyines, oodines, and chlaeniines.
Jeannel (1948a: 627) indicated that the genus Dercylinus (one North American species), of which he had seen no specimen, probably belongs to dercylines and Lorenz (2005: 327) listed the genus, along with Evolenes (one North American species), in the subtribe Dercylina. However, these two genera are typical oodines (see Bousquet 1996a).
Tribe Melanchitonini. This tribe currently includes three genera, Melanchiton Andrewes (a replacement name for Melanodes Chaudoir), Melanchrous Andrewes (a replacement name for Patellus Chaudoir), and Dicaelindus Macleay. The lineage contains about 70 Old World species. As for many other groups, relationships of melanchitonines are unclear. Chaudoir (1883) included Melanchiton and Melanchrous within the tribe Oodini, likely because of the similar adhesive setae on the male protarsi. Subsequently, the two genera have been placed by some authors within the Pterostichini. Jeannel (1948a: 626) included them with dercylines in his family Dercylidae but offered no pertinent evidence to suggest that the group is monophyletic.
Straneo (1950: 65) first included the genus Dicaelindus, previously placed in the Pterostichini, in this tribe. Adults of Dicaelindus are rather similar phenetically to those of Melanchiton, but the male protarsi are not dilated and lack adhesive setae. Monophyly of this tribe has not yet been demonstrated.
Tribe Oodini. Members of Oodini sensu stricto share several apomorphic character states in the adult stage (Bousquet 1996a: 448) suggesting the tribe is monophyletic. Several authors have included or associated oodines with chlaeniines but the pygidial gland components suggest rather that panagaeines and chlaeniines are more closely related to each other than to oodines (Bousquet 1987b). Oodines, panagaeines, and chlaeniines possibly constitute a clade since the adults (except in some chlaeniines) have the metepisterna coadunate with the elytral epipleura, a synapomorphic condition that has probably been secondarily lost in some chlaeniine lineages.
Some groups, such as dercylines, melanchitonines, and geobaenines, are sometimes included within the Oodini as distinct subtribes. However, there is little evidence that they are indeed closely related to oodines and in my opinion they should be treated as distinct tribes.
This tribe is represented in all zoogeographical regions of the world and includes about 295 species in 32 genera. Jeannel (1949a: 829) recognized three family-group taxa within the oodines: sphoerodines represented in the Afrotropical Region, oodines (sensu stricto) represented in all zoogeographical regions, and thryptocerines represented in the Afrotropical Region.
Tribe Peleciini. Relationships of peleciines are unclear. The group has been associated with panagaeines by Kryzhanovskij (1976a: 89), Ball (1979), and Erwin (1985: 468) and included in the superfamily Odacanthomorphi, along with odacanthines, perigonines, lachnophorines, and ctenodactylines, by Jeannel (1948a: 376). Many apomorphic features, including some details of the chaetotaxy, structure of the mouthparts and thorax, and marked similarity in their specialized way of attacking millipedes, suggest that peleciines could be closely related to promecognathines. However, Straneo and Ball (1989) regarded these similarities as evolutionary convergence, not phylogenetic affinity. Larvae of Eripus oaxacanus Straneo and Ball, the only peleciine species known in its larval stage, are similar in some structural features to larvae of Brachinini and Pseudomorphini but Liebherr and Ball (1990) concluded that these similarities were an example of convergence due to a similar parasitic lifestyle. Arndt (1993: 36), based on larval features, suggested that peleciines, panagaeines, licinines, oodines, and chlaeniines form a clade. Liebherr and Will (1998: 156-157) noted from their analysis of the female reproductive tract that placement of peleciines as a basal group of pterostichine stock was firmly supported.
Peleciines are restricted to the Southern Hemisphere. Straneo and Ball (1989) recognized two subtribes: Agonicina for the genera Pseudagonica Moore and Agonica Sloane of Tasmania and adjacent southeastern Australia, and Peleciina (including disphericines) for the remaining genera which are represented in the Afrotropical, Oriental, and Neotropical Regions. Vigna Taglianti and Rossi (1998: 515) noted that the laboulbeniale parasitic species found in Agonica and in the moriomorphine genera Pterogmus Sloane, Theprisa Moore, and Sitaphe Moore were very similar and alluded to the possibility of a close relationship between agonicines and moriomorphines.
Tribe Brachygnathini. This tribe contains only the Neotropical genus Brachygnathus Perty (seven species). Relationships of the genus are uncertain. Jeannel (1949a: 849) associated it with the genus Microcephalus Dejean (as Tichonia Semenov), under the subfamily name Tichoniitae, and placed it in his family Panagaeidae. Reichardt (1977: 404) noted that inclusion of Brachygnathus in the tribe Panagaeini was doubtful and that the adults show some similarities to those of peleciines.
Tribe Bascanini. This tribe contains a single genus, Bascanus Péringuey (including Bascanidius Péringuey), with a few species in eastern and southern Africa. Van Emden (1936a), Basilewsky (1953a: 164-165), and Erwin (1979) suggested that bascanines are closely related to panagaeines. Csiki (1933a: 1651) associated the genus with Melaenus Dejean.
Tribe Panagaeini. This moderately diverse group occurs in all continents except Antarctica but is much more diverse in the tropics than in temperate regions. Panagaeines, at least those that have been analysed, secrete phenol through their pygidial glands (see Schildknecht et al. 1968; Kanehisa and Murase 1977; Moore 1979). This compound is also found, as far as known, only in some chlaeniines, which suggests that panagaeines are probably most closely related to chlaeniines. On the other hand, several authors, including Kryzhanovskij (1976a: 89), consider peleciines as the group most closely related to panagaeines.
Jeannel (1949a: 849) associated the genus Microcephala Dejean (as Tichonia Semenov) with Panagaeini but most authors, including Reichardt (1977: 407), regard it as a member of Pterostichini.
Tribe Chlaeniini. Chlaeniines are found in all zoogeographical regions of the world but are more diverse, both in terms of lineages and species, in the Afrotropical and Oriental Regions than anywhere else. Jeannel (1949a: 776) recognized six tribes among chlaeniines and Basilewsky and Grundmann (1955) ten tribes and two subfamilies. However, following Ball (1960b) and Lindroth (1969a), all the species are grouped in a single tribe in this catalogue. Several authors have suggested a close relationship between chlaeniines, panagaeines, and oodines.
Two major groups among Chlaenius species could be distinguished based on defensive secretions of the pygidial glands (see Schildknecht et al. 1968; Kanehisa and Murase 1977; Moore 1979; Balestrazzi et al. 1985): one secretes phenol, like panagaeines; the other one quinone. In the first group, the secretory lobes of the pygidial glands are elongate, in the second one they are shorter and thicker (Kanehisa and Shiraga 1978). I believe these two groups should be recognized either as genera or subtribes. However, owing to the lack of information on the pygidial glands and their secretions for many chlaeniine lineages, such action is futile at this time.
This tribe includes almost a thousand species worldwide arranged in 18 genera and two subtribes. The 51 North American species are assigned to the genus Chlaenius Bonelli and arrayed in ten subgenera of which five, Pseudanomoglossus Bell (one species), Anomoglossus Chaudoir (three species), Callistometus Grundmann (one species), Brachylobus Chaudoir (one species), and Randallius n.subg. (one species), are North American endemics.
Tribe Cuneipectini. This tribe includes one genus, Cuneipectus Sloane, with two flightless species in western Australia. Members of this group have rarely been collected and very little is known about their way of life. Kryzhanovskij (1976a: 89) listed cuneipectines in his supertribe Pterostichitae along with dercylines, zabrines, platynines, chaetodactylines, microcheilines, and pterostichines. Erwin (1985: 468) associated them with chaetogenyines, chlaeniines, oodines, and licinines in his supertribe Callistitae (= Chlaeniitae). Moore et al. (1987: 215) included them with morionines, pterostichines, abacetines, geobaenines, drimostomatines, and platynines in their Pterostichitae.
Tribe Orthogoniini. This group includes six genera represented in Asia and Africa only: Orthogonius Macleay (about 240 species), Neoorthogonius Tian and Deuve (one species), Hexachaetus Chaudoir (nine species), Actenoncus Chaudoir (four species), Anoncopeucus Chaudoir (two species), and Nepalorthogonius Habu (one species). Relationships of the tribe remain unresolved and problematic. Jeannel (1948a: 377) indicated that orthogoniines and licinines are closely related based on the shape of the frontale on the cephalic capsule of the larvae. Basilewsky (1953a: 180) associated them with glyptines, Kryzhanovskij (1976a: 90) with lebiines, anthiines, helluonines, physocrotaphines, zuphiines, galeritines, and dryptines, and Erwin (1985: 468) with idiomorphines, catapieseines, and amorphomerines. Ober and Maddison (2008: 18) found strong support in their phylogenetic analyses based on molecular data sequences for a clade comprising orthogoniines, graphipterines, and pseudomorphines. The genus Glyptus Brullé has been included by some authors within the tribe Orthogoniini, but both Jeannel (1948a) and Erwin (1985) believe that Glyptus and Orthogoniini are not closely related. Members of this tribe are termitophilous.
Tribe Idiomorphini. This tribe currently includes the genera Idiomorphus Chaudoir (three Indian species), Perochnoristhus Basilewsky (one species in Namibia), Rathymus Dejean (three Afrotropical species), and Strigia Brullé (three Oriental species) arrayed in two subtribes, Perochnoristhina for the genus Perochnoristhus and Idiomorphina for the remaining genera (Lorenz 2005: 391). Erwin (1984b: 378) also included the genus Glyptus in this tribe. Crowson (1980) stated that the genus Perochnoristhus could be closely related to broscines and apotomines.
Tribe Glyptini. Glyptini consists of two Afrotropical genera: Neoglyptus Basilewsky with six species and Glyptus Brullé with two species. Few authors agree on the systematic position of the group. Jeannel (1948a: 377) associated them with chlaeniines, Basilewsky (1953a: 180) with orthogoniines, and Erwin (1984b: 378) with idiomorphines. Both Chaudoir (1850a) and Lacordaire (1854) stated that glyptines were closely related to the genus Idiomorphus Chaudoir.
Tribe Amorphomerini. This group includes a single genus, Amorphomerus Sloane, represented by a few species in eastern Africa and Madagascar. Jeannel (1948a: 376) associated amorphomerines with pterostichines (sensu lato, including platynines), dercylines, and harpalines in his superfamily Harpalomorphi, characterized by having the mesotibiae spinose and the median lobes of the aedeagi more or less bent, with the basal bulbs well developed. He also stated (Jeannel 1948a: 731) that the tribe was more closely related to harpalines than to any other Conchifera groups. Kryzhanovskij (1976a: 89) associated amorphomerines with harpalines, cnemalobines (as Cnemacanthini), and agonicines (currently included in the Peleciini) and Erwin (1985: 468) associated them with idiomorphines, orthogoniines, and catapieseines. The tribe was listed as part of the tribe Lebiini by Erwin (1979).
Tribe Licinini. A clearly defined, likely monophyletic group with representatives in all zoogeographical regions of the world. Jeannel (1948a: 377) associated licinines with pentagonicines, orthogoniines, panagaeines, chlaeniines (including oodines), and glyptines, Kryzhanovskij (1976a: 89) with oodines and chlaeniines, and Erwin (1991a: 10) with oodines, chaetogenyines, chlaeniines, and cuneipectines. Ball (1992a) considered the tribe to be the sister-group to {Oodini + Chlaeniini + Panagaeini} and Ball and Bousquet (2000: 100) noted that members of the four tribes show similarities in structure of the male protarsi, genitalia, and larvae. Beutel (1992d) reported several putative synapomorphies in larval head structures between Licinini and Panagaeini, and Arndt (1993: 37) noted several synapomorphies in larvae of licinines, panagaeines, and peleciines. However, contrary to the oodine-chlaeniine-panagaeine complex, licinines have simple (i.e., non-crossed) epipleura and secrete formic acid as the major constituent of their defensive glands like harpalines and Truncatipennes members. Also Ober and Maddison (2008: 19) found no close relationship between licinines and the chlaeniine-oodine-panagaeine complex based on their analyses derived from molecular data sequences. Recently Liebherr and Will (1998: 144) suggested that licinines, orthogonines, panagaeines, melanchitonines, graphipterines, and loxandrines form a clade based on the presence of a villous canal extended forward on the common oviduct.
The 235 or so species are arrayed in 23 genera distributed among four subtribes following Ball (1992a).
Tribe Harpalini. This is one of the largest and most diversified carabid tribes. Although its limits are fairly stable, there is as yet no strong evidence to substantiate that the tribe is monophyletic. Based on a study of the world fauna, Noonan (1976) recognized four subtribes among harpalines: Anisodactylina, Pelmatellina, Stenolophina (including polpochilines and pachytrachelines), and Harpalina which he divided into eight genus-groups, namely Harpali, Selenophori, Bradybaeni, Acinopi, Bleusei, Dapti, Amblystomi, and Ditomi. Based on a parsimony analysis of molecular sequence data, Martínez-Navarro et al. (2005) concluded that the subtribe Harpalina was polyphyletic, that daptines were related to stenolophines, not to Harpalina, that the Selenophori group was polyphyletic and not related to Harpalina but perhaps to anisodactylines, that the Amblystomi group may be related to stenolophines instead of Harpalina, and that the subtribe Pelmatellina was related to stenolophines (see also Martínez-Navarro et al. 2003) and that the latter could be paraphyletic in regard to the former. They also advocated raising selenophorines, ditomines, and amblystomines to subtribe level.
Relationships of harpalines to other carabid groups are not well established. In the course of his work on the French fauna, Jeannel (1942: 575) associated harpalines with perigonines, anchonoderines, lachnophorines, omphreines, pterostichines (including platynines), zabrines, chaetodactylines, morionines, and metiines in his superfamily Harpalomorphi. Later, working on the Madagascan fauna, Jeannel (1948a: 376) united the harpalines with amorphomerines, dercylines, melanchitonines, pterostichines (including platynines), morionines, microcheilines, and chaetodactylines. Kryzhanovskij (1976a: 89) listed harpalines with amorphomerines, cnemalobines, and agonicines (currently included in Peleciini) in his supertribe Harpalitae. Based on the presence of a membranous transverse band on the stipes lateroventrally in larvae, Arndt (1998: 184) associated harpalines with morionines, pterostichines, zabrines, panagaeines, peleciines, chlaeniines, oodines, licinines, and cnemalobines. In a cladistic analysis conducted by Roig-Juñent and Cicchino (2001: Fig. 1), this tribe was positioned as the sister-group to {Platynini + Sphodrini}. Ruiz et al. (2008) indicated that, based on their molecular data sequence analyses, the tribe Harpalini was the sister-group to {Sphodrini + Platynini + Pterostichini + Zabrini}.
Tribe Geobaenini. The Geobaenini includes a single genus, Geobaenus Dejean, with four flightless species: three occur in South Africa, one in Australia. The group was first included within the tribe Harpalini and associated subsequently with pterostichines. Basilewsky (1949), because of similarity in the adhesive setae on the male protarsi, suggested that geobaenines could be closely related to melanchitonines, although later (1950, 1953, 1985) he associated the genus with platynines (as Anchomeninae or Platyninae). Liebherr and Will (1998: 144) in their study of the female reproductive tract found an “uncontested synapomorphy” uniting geobaenines with lachnophorines, odacanthines (including pentagonicines), and pseudomorphines. In these taxa, the spermathecal duct is joined to the common oviduct by an elongate sclerite.
Tribe Omphreini. This tribe includes a single genus, Omphreus Dejean (18 species), which is endemic to the Balkan Peninsula and Asia Minor. Omphreines have been included within the tribe Platynini by most authors but Jeannel (1942: 577), followed by Kryzhanovskij (1976a: 89), associated them with perigonines, anchonoderines (including atranines), and lachnophorines.
Tribe Sphodrini. Members of this group have been traditionally included within the Platynini. However, in recent decades numerous taxonomists dealing with the Palaearctic fauna, where this group is by far more diversified than anywhere else, rank this complex as a distinct tribe. Based on morphological characters, there seems to be little doubt that the two groups are closely related. However, from molecular data sequence analyses conducted by Ruiz et al. (2008), this relationship did not receive “the expected strong support, though it can not be completely dismissed.” The Sphodrini include about 825 species, arranged in about 40 genera, and are grouped into the following six subtribes: Atranopsina (about 100 species), Calathina (about 185 species), Dolichina (17 species), Pristosiina (about 65 species), Synuchina (almost 100 species), and Sphodrina (about 360 species). Based on Casale’s (1988: 130) cladogram, Dolichina and Synuchina are sister-groups, and Sphodrina, Calathina, and Pristosiina form a clade with Pristosiina the sister-group to the other two; the position of Atranopsina is ambiguous. From the molecular data sequence analyses conducted by Ruiz et al. (2008), only the position of the subtribe Atranopsina as the sister-group to all other subtribes was well supported.
Tribe Platynini. This is a large, complex, and worldwide group which is more diverse in the tropics than in temperate regions. There are no synapomorphies, in either adult or larval structures, yet discovered to suggest that the tribe represents a monophyletic lineage. Platynines are combined by various authors with pterostichines based on phenetic similarity between the two groups. I believe the two groups are not closely related because of the differences in elytral epipleuron configurations and pygidial gland structures and secretions. Basilewsky (1985, as Platyninae) gave an excellent introduction to the systematics of the group.
Relationships among the North American genus-group taxa have been addressed but are still inadequately understood. According to Liebherr (1991b: 5), Tetraleucus, Anchomenus, Sericoda, and Elliptoleus form a clade characterized by the synapomorphic condition of the female spermatheca having a basal reservoir and a long apical filament. Within this clade Tetraleucus is the sister-group to the remaining taxa. Liebherr and Schmidt’s (2004: 168) parsimony-based cladistic analysis led to the recognition of four subgenera within the genus Agonum forming two clades, {Platynomicrus Casey + Europhilus Chaudoir} and {Agonum s.str. + Agonothorax Motschulsky (= Olisares Motschulsky)}. Liebherr and Schmidt (2004: 153) suggested a sister-group relationship between the genus Agonum and the African taxa described in combination with Agonidium Jeannel and Neobatenus Jeannel as well as several others described under Megalonychus Chaudoir.
Tribe Perigonini. This small tribe is represented by about 115 species arranged in five genera. The place of the tribe within the carabids is unsettled. LeConte and Horn (1883: 35) and Sloane (1923: 248) included it as a separate group within the Platynini; Jeannel (1942: 577) as a distinct subfamily within his family Perigonidae along with anchonoderines, omphreines, and lachnophorines and later (Jeannel 1948a: 376) as a distinct family within his superfamily Odacanthomorphi along with lachnophorines, odacanthines, ctenodactylines, and peleciines. Kryzhanovskij (1976a: 89) followed Jeannel (1942) and combined the tribes Perigonini, Lachnophorini (including anchonoderines), and Omphreini in his supertribe Perigonitae. Erwin (1984b: 375) placed this tribe in his supertribe Lebiitae along with amorphomerines, catapieseines, graphipterines, tetragonoderines, masoreines, pentagonicines, odacanthines, and lebiines. Later (Erwin 1991a: 10) the amorphomerines and catapieseines were excluded from the Lebiitae.
The North American fauna includes two species of the genus Perigona Laporte which contains about 100 species worldwide. One of our species is adventive and the second one is endemic to the eastern part of the continent.
Tribe Ginemini. This tribe includes a single species, Ginema thomasi Ball and Shpeley, known from a single female specimen collected in the departament of Santa Cruz in Bolivia. Ball and Shpeley (2002a: 96) noted some marked similarities between this genus and members of Cyclosomini but still postulated a rather isolated position in the rank of the more derived Harpalinae lineages.
Tribe Enoicini. This tribe includes two South African genera: Enoicus Péringuey with one species and Abacetodes Straneo (= Phimus Péringuey, a preoccupied name) with four species. Basilewsky (1985: 15-16) associated enoicines with platynines, geobaenines, and sphodrines while earlier (Basilewsky 1953a: 61) he included them within the platynines (as Anchomenini).
Tribe Atranini. This tribe contains only two species, both included in the genus Atranus LeConte: one lives in Europe and the Caucasus, the other one in eastern North America. The systematic position of the genus has been debated. Dejean (1828: 122) described the North American species in the genus Anchomenus Bonelli, currently placed within the Platynini. LeConte (1847: 438; 1861a: 28), Seidlitz (1887: 10), and Sloane (1923: 250) associated the genus with chlaeniines, LeConte and Horn (1883: 37), Fauvel (1888: 15), and Jeannel (1942: 582) with anchonoderines, and Ball (1960b: 136), Lindroth (1966: 648), Liebherr (1986: 20), Kryzhanovskij et al. (1995: 118), and several others with platynines. Adults and larvae of Atranus possess several structural features not exhibited in other Platynini. In my opinion the morphological evidence relating Atranus to platynines is weak and for that reason the genus is retained here in its own tribe. Analysis of the pygidial secretions could be useful to indicate if the genus is more closely related to chlaeniines or to the platynine-anchonoderine lineage. Basilewsky (1962b: 155) believed the genus was more closely related to platynines than to any other group suggested to date, but because the adhesive setae on the male protarsi are of the “spongy” type rather than the “seriate” type, he advocated placing it in a distinct subfamily. Phylogenetic relationships as inferred from 28S ribosomal DNA and the wingless gene conducted by Ober and Maddison (2008) placed Atranus as the sister-group to the Platynini.
Tribe Catapieseini. This small Neotropical tribe includes two genera, Catapiesis Brullé with eight species and Homalomorpha Brullé with one species, ranging collectively from southern Mexico to northeastern Argentina. Catapieseines have been placed by some authors (e.g., Reichardt 1977) in the vicinity of the Morionini and Pterostichini. Lorenz (2005: 248) included them within the tribe Cratocerini, in his subfamily Pterostichinae, along with drimostomatines. Ober and Maddison (2008: 16), following Erwin (1984b: 375), placed them in their lebiomorph assemblage pointing out that catapieseines have truncate elytra and specialized eighth abdominal tergite turrets like the remaining lebiomorph taxa. Horn (1881: 163) believed that Catapiesis (as Basoleia) has a close relationship with the Helluonini. Erwin (1985: 468) listed the group within his supertribe Orthogoniitae along with idiomorphines, amorphomerines, and orthogoniines.
Tribe Lachnophorini (including anchonoderines). This small tribe includes about 120 species in ten genera. There is no solid structural or molecular evidence that would suggest this group is monophyletic. Its relationships have been discussed by several authors. Jeannel (1942: 578), followed by Kryzhanovskij (1976a: 89), associated lachnophorines with perigonines, anchonoderines (including Atranus), and omphreines. Liebherr (1988) suggested that lachnophorines derived from a platynine-like ancestor. He also included calophaenines within lachnophorines but Ball and Bousquet (2000: 107), following Erwin (1991b: 44), placed them with ctenodactylines.
The Lachnophorini are represented in the New World, and by one Indo-African species, Selina westermanni Motschulsky. Jeannel (1948a: 744) also considered the genus Amoebea Péringuey (one Afrotropical species) as lachnophorine. However, the name is a junior synonym of Smeringocera Chaudoir (six species) which belongs to the tribe Odacanthini (see Lorenz 2005: 444).
In Liebherr’s (1988) parsimony-based cladistic analysis of the West Indies lachnophorines, the taxa with setose body and setose maxillary palpomeres, represented by the genera Euphorticus, Calybe, and Lachnophorus, constituted a clade.
Tribe Pentagonicini. Few characteristics hold the pentagonicines together and this tribe is possibly polyphyletic. Jeannel (1949a: 767) included pentagonicines in his superfamily Callistomorphi, along with licinines, orthogoniines, panagaeines, chlaeniines, and glyptines, and indicated that pentagonicines are best placed near licinines. Moore (1966a: 162) wrote that larval characters of the pentagonicine Scopodes “suggest a rather close affinity with the Odacanthinae.” Liebherr (1988) included pentagonicines within the Odacanthini based mainly on the structure of the spermatheca, and Ober and Maddison (2008: 17) found support from their molecular data for such a relationship. Erwin (1984b: 375) placed this tribe in his supertribe Lebiitae along with amorphomerines, perigonines, catapieseines, graphipterines, tetragonoderines, masoreines, odacanthines, and lebiines. The author later adopted a similar arrangement (Erwin 1985: 468), with the exclusion of amorphomerines and catapieseines and the inclusion of lachnophorines. It is of interest to note that the sole species of pentagonicines studied (Scopodes boops Erichson) produces a saturated acid as major component (Moore 1979) of the pygidial glands, while members of the so-called Truncatipennes, with which this tribe is usually associated, produce formic acid (see Schildknecht et al. 1968; Moore and Wallbank 1968; Kanehisa and Murase 1977; Moore 1979).
This tribe includes close to 170 species in the world. Only six, all in the genus Pentagonica Schmidt-Göbel (86 species worldwide), are found in North America.
Tribe Odacanthini. The Odacanthini, with about 300 species in 30 genera, constitutes a moderately diverse group represented in all zoogeographical regions. They are more diverse in the tropics than in temperate regions. Several authors believed the group to be closely related to ctenodactylines, and both groups have been combined in a single tribe by some (Csiki 1932b, Liebke 1938, van Emden 1942). Jeannel (1948a: 376) associated odacanthines with perigonines, lachnophorines, ctenodactylines, hexagoniines, and peleciines, and Basilewsky (1962b: 154) with lachnophorines. Liebherr (1988) concluded that odacanthines (including pentagonicines) have a sister-group relationship with lachnophorines (including calophaenines) based on the presence of a bipartite spermatheca. However, phylogenetic analyses based on molecular data sequences presented by Ober and Maddison (2008: 5) did not support odacanthines and lachnophorines as a clade but did support a close relationship between pentagonicines and odacanthines and between calophaenines and lachnophorines. Erwin (1985: 468) placed the tribe Odacanthini within his Lebiitae along with perigonines, lachnophorines, graphipterines, tetragonoderines, cyclosomines (as Masoreini), pentagonicines, and lebiines.
The tribe is represented in North America by six species belonging to the New World genus Colliuris DeGeer, which currently includes about 80 species.
Tribe Calophaenini. This tribe includes two Neotropical genera: Calophaena Klug (48 species) and Calophaenoidea Liebke (one species). Calophaenines have been placed in the tribe Odacanthini (Horn 1881: 147; Bates 1883a: 163; Reichardt 1977: 435; Lorenz 2005: 439), Lachnophorini (Liebherr 1988: 18), or Ctenodactylini (Erwin 1991b: 44; Ball and Bousquet 2000: 107). Liebherr’s (1988) assumption of a relationship with lachnophorines received support from most molecular analyses conducted by Ober and Maddison (2008: 17). The association of calophaenines with ctenodactylines is based on similarity of the adhesive setae under the tarsomeres (Stork in Ball and Bousquet 2000: 107). Until the relationship of calophaenines is better established I prefer to place them in a distinct tribe.
Tribe Ctenodactylini. This tribe is represented only in the New World and includes about 115 species in 18 genera. Most recent authors agree that this group is closely related to the tribe Hexagoniini of the Eastern Hemisphere. Only three species are found north of Mexico and they belong to the genus Leptotrachelus Latreille along with about 30 more species in the tropics.
The main characteristic of the Ctenodactylini and Hexagoniini is the inverted median lobe of the aedeagus as in the drimostomatines.
Tribe Hexagoniini. This tribe contains 65 species in three genera: Hexagonia Kirby (47 species in Asia, Africa, New Guinea [one species], and Australia [one undescribed species, cf. Darlington 1968: 202]), Dinopelma Bates (13 species in the Oriental Region), and Omphreoides Fairmaire (five Madagascan species). Vigna Taglianti and Rossi (1998: 515) indicated that hexagoniines could be related to odacanthines based on similar parasitic laboulbeniales.
Tribe Cyclosomini. This tribe is used here in a restricted sense (see Ball and Bousquet 2000: 109). It includes about 120 species, predominantly tropical, placed in four genera: Mnuphorus Chaudoir with 11 species in the Palaearctic Region; Cyclosomus Latreille with 13 species in the Afrotropical and Oriental Regions; Cyclicus Jeannel with 22 species in the Afrotropical and Oriental Regions; and Tetragonoderus Dejean represented by about 80 species in the Afrotropical, Oriental, Neotropical, and Nearctic Regions, and on the southern fringe of the Palaearctic Region. Several authors, including Jeannel (1949a: 860) and Basilewsky (1984: 527), have considered the New World genus Nemotarsus LeConte as related to cyclosomines, but following Ball (1960b: 157) and Lindroth (1969a: 1014) the genus is listed here in the tribe Lebiini.
Cyclosomines are associated with somoplatines, graphipterines, corsyrines, masoreines, and sarothrocrepidines by most authors based on the presence of long tibial spurs in adults. However, Ball and Bousquet (2000: 109) remarked that the complex as a whole is probably not monophyletic. Molecular analyses published by Ober and Maddison (2008: 17) did not support a close relationships between cyclosomines (sensu lato, i.e., including somoplatines, corsyrines, masoreines, and sarothrocrepidines) and graphipterines but suggested they may be associated with members of dromiine and / or cymindidine lebiines.
Tribe Somoplatini. Ball and Bousquet (2000: 109) restricted this tribe to the genera Somoplatus Dejean (14 Indo-African species), Somoplatodes Basilewsky (two Afrotropical species), and Lophidius Dejean (one Afrotropical species), with Paralophidius Basilewsky recently placed in synonymy with Somoplatus (Schüle 2009: 461). Basilewsky (1986) listed these genera as part of his tribe Masoreini.
Tribe Masoreini. This tribe, as restricted by Ball and Bousquet (2000: 109), comprises the genera Masoreus Dejean (seven Palaearctic species), Atlantomasoreus Mateu (two Moroccan species), Anaulacus Macleay (38 species), and Leuropus Andrewes (one Oriental species). Odontomasoreus Darlington (one species from New Guinea), listed as a distinct genus by Lorenz (2005: 451), has been considered a subgenus of Anaulacus by Ball and Shpeley (2002b: 279). Jeannel (1949a: 860) associated masoreines with cyclosomines and nemotarsines.
Tribe Corsyrini. This tribe comprises the Palaearctic Asian genera Corsyra Dejean (one species) and Discoptera Semenov (five species). Jeannel (1949a: 860) included them with masoreines and Ball and Bousquet (2000: 109) with graphipterines. These authors did not offer evidence to support their groupings.
Tribe Sarothrocrepidini. This tribe is represented by a single genus, Sarothrocrepis Chaudoir, with 26 Indo-Australian species. Jeannel (1949a: 860) associated the genus with graphipterines, masoreines, cyclosomines, and nemotarsines.
Tribe Graphipterini. This tribe, represented in Africa and the Middle East, includes the genera Graphipterus Latreille (about 145 species), Piezia Brullé (18 species), and Trichopiezia Nègre (one species). Jeannel (1949a: 860) associated graphipterines with sarothrocrepidines, masoreines, cyclosomines, and nemotarsines and Kryzhanovskij (1976a: 90) with the same groups with the exception of the nemotarsines. Ober and Maddison (2008: 17) found no support from their molecular analyses for a close relationship between graphipterines and cyclosomines (sensu lato). Instead they found graphipterines to be closely related to pseudomorphines and orthogoniines.
Tribe Lebiini. A markedly complex, worldwide tribe undoubtedly more diverse both in terms of species and lineages in the tropics than in temperate regions. No synapomorphy is known to suggest that this tribe constitutes a monophyletic lineage and Ober and Maddison (2008: 18) did not recover a monophyletic Lebiini in their analyses based on molecular data sequences. The supraspecific classification is not established clearly since most modern studies on lebiines have focussed on regional faunas. The systematic position of some groups within the Lebiini is still debated. For example, the genus Celaenephes Schmidt-Göbel has been considered the most ancestral group of extant lebiines by Shpeley et al. (1985) but as a platynine by Basilewsky (1984). In phylogenetic analyses derived from molecular sequence data by Ober and Maddison (2008: 18), Celaenephes was not clearly associated with members of Lebiini.
Relationships of the tribe are not clearly understood. Jeannel (1948a: 378) included lebiines with anthiines, helluonines, dryptines, galeritines, physocrotaphines, zuphiines, and calophaenines in his superfamily Lebiomorphi. Kryzhanovskij (1976a: 90) used a similar grouping with the exception that he also included orthogoniines, a group that Jeannel (1948a: 377) and Basilewsky (1984: 528) included within the Callistomorphi (i.e., pentagonicines, licinines, panagaeines, chlaeniines, and glyptines). Erwin and Sims (1984: 357) and Erwin (1985: 468) associated lebiines with perigonines, lachnophorines, graphipterines, cyclosomines, masoreines, pentagonicines, and odacanthines.
The 220 genera currently recognized within this tribe are arrayed in the following 16 subtribes (see Ball and Bousquet 2000: 110): Celaenephina, Pericalina (including coptoderines and eucheilines), Sugimotoina, Actenonycina, Apenina, Cymindidina, Dromiusina, Lebiina, Physoderina, Metallicina, Agrina, Calleidina, Gallerucidiina, Peliocypadina, Demetriadina, and Nemotarsina. In the phylogenetic analyses from molecular data published by Ober and Maddison (2008), a small number of clades within the Lebiini were supported but none of these corresponded to the current subtribes, and the subtribes were not recovered as monophyletic.
Tribe Dryptini. Dryptines have been included within the galeritines by several authors (including Darlington 1971: 198). The two lineages are now placed in different tribes but most authors agree that they are closely related. Dryptines and galeritines share some character states with zuphiines and these three groups, referred to as supertribe Zuphiitae by Erwin and Sims (1984: 356) and Erwin (1985: 468), probably constitute a clade. Basilewsky (1960) recognized six dryptine genera in the world, only one of them, the monobasic Amazonian Neodrypta Basilewsky, is found in the New World. Most species are tropically-adapted in the Afrotropical, Australian, and Oriental Regions, with a few species in the southern parts of the Palaearctic Region.
Tribe Galeritini. This tribe is represented in all zoogeographical regions but is more diverse in the tropics than in the temperate zones. Basilewsky (1963b: 7) and Ball (1985) have recognized two lineages within galeritines, treated as subtribes by Ball (1985): Planetina for the genus Planetes Macleay (27 species in the Eastern Hemisphere) and Galeritina for the remaining genera. Lorenz (2005: 507), however, included planetines in zuphiines and Ober and Maddison (2008: Fig.5) found support for such a grouping in some of their analyses and reported that the tribe Galeritini was not monophyletic, based on their molecular data analyses. As indicated previously, this tribe is probably closely related to the Dryptini and Zuphiini.
Tribe Zuphiini. This group is represented in all zoogeographical regions of the world but is more diverse in the tropics than in temperate regions. Zuphiines are grouped by some authors (e.g., Basilewsky 1962a: 100-101) into three subtribes: Leleupidiina, Patriziina, and Zuphiina. Baehr (1985) briefly discussed the status of each of these subtribes. On the other hand, Lorenz (2005: 505-507) included patriziines within the subtribe Zuphiina. Most authors agree that Zuphiini are closely related to Galeritini. For example, LeConte and Horn (1883: 41) and Erwin (1991a: 10) combined zuphiines with galeritines and dryptines. Habu (1967) included zuphiines and galeritines in a single tribe and recognized three subtribes: Zuphiina, Galeritina, and Planetina. Moore (1998: 369) suggested a close relationship between zuphiines and physocrotaphines based on characters of adults and Jeannel (1949a: 1047) enlisted zuphiines, galeritines, dryptines, and physocrotaphines in his family Dryptidae. Ober and Maddison (2008: 18) found a well-supported “Zuphiitae” clade including zuphiines, anthiines, dryptines, galeritines, helluonines, and physocrotaphines in their phylogenetic analyses. However, the tribe Zuphiini was not found to be monophyletic.
The hypogean and monospecific genus Ildobates Español from the Iberian Mountain range was originally described in the Dryptini and was subsequently transferred to the Galeritini by Jeanne (1972) and finally to the Zuphiini by Ortuño et al. (2005).
Tribe Physocrotaphini. The Physocrotaphini includes the following genera: Helluodes Westwood (three species in southwestern India and Sri Lanka), Physocrotaphus Parry (one species from Sri Lanka), Pogonoglossus Chaudoir (35 species), and Schuelea Baehr (three species in New Guinea). The monobasic genus Holoponerus Fairmaire from New Britain was originally described as a lebiine but Darlington (1968) and Moore (1998: 370) agreed that the genus probably belongs to the physocrotaphines. Unfortunately the sole known specimen of Holoponerus godeffroyi Fairmaire was destroyed in 1943 during the bombing of Hamburg in World War II (Moore 1998: 370). All but two species of physocrotaphines are very rarely collected and little is known about their way of life (Sabu et al. 2008: 30). Members of Helluodes are probably termitophilous and those of Pogonoglossus are predominantly subcorticolous though some species could be litter-dwelling forms (Sabu et al. 2008: 41-42).
Jeannel (1949a: 1047) associated physocrotaphines with zuphiines, galeritines, and dryptines and most authors currently agree that these lineages are probably closely related. Jeannel (1949a: 1047) claimed that the genus Pogonoglossus belongs to the Zuphiini near the genus Eunostus Laporte but both Darlington (1968) and Moore (1998: 375) retained the genus within the Physocrotaphini. In his work on the French fauna, Jeannel (1942: 1017) associated physocrotaphines with anthiines and helluonines.
Tribe Anthiini. Anthiines are large, apterous beetles which live in the steppes and subdesert areas of Africa and southwestern Asia. The species are classified into eight or nine genera. The group is clearly defined and probably closely related to the tribe Helluonini (van Emden 1937; Jeannel 1949a: 1040; Bousquet 1987c: 928; Arndt 1993: 44). Basilewsky (1962a: 93) even suggested that anthiines could be derived from a helluonine genus close to Triaenogenius Chaudoir of the Afrotropical Region. Based on larval character states, Bousquet (1987c: 928) suggested that pseudomorphines and/or galeritines could be closely related to the anthiine-helluonine lineage. Erwin and Sims (1984: 356) and Erwin (1985: 468) combined the anthiines with the helluonines and physocrotaphines in their supertribe Anthiitae.
Tribe Helluonini. This is a moderately diverse group with representatives in all continents except Europe and Antarctica. Despite the fact that the group appears homogeneous from the morphology of the adults, Ober and Maddison (2008: 18) did not recover it as monophyletic in their analyses derived from molecular data sequences. Several larval features suggest that helluonines are closely related to anthiines (Bousquet 1987c; Arndt 1998: 186). However, Reichardt (1974, 1977) suggested, based on the structures of the adult mouthparts, that helluonines could be closely related to eucheilines (currently placed within the Lebiini). Liebherr and Will (1998: 145) concluded that Helluonini and Galeritini might be sister-groups based on their possession of a secondary spermathecal gland.
Sloane (1914) and Reichardt (1974) recognized two subtribes among helluonines: Helluonina with representatives in Australia and New Guinea and Helluomorphina (= Omphrina) with representatives in Asia, Africa (including Madagascar), Australia, and the Western Hemisphere. About 165 species, placed in 25 genera, are known worldwide but only eight, all belonging to the New World genus Helluomorphoides Ball, are found in North America.
Tribe Xenaroswellianini. Erwin (2007b: 567) suggested that this recently described tribe, which includes a single species known only from the holotype collected in the Brazilian state of Goiás, could have “a possible relationship with the enigmatic Pseudomorphini.”
Tribe Pseudomorphini. Members of Pseudomorphini are structurally aberrant possibly in response to the group’s evolution into myrmecophily. Nevertheless placement of the group within the subfamily Harpalinae has been confirmed in almost all recent analyses (Arndt et al. 2005: 141). Relationships of pseudomorphines to other harpaline tribes are unsettled as stated by Deuve (1993: 98). Erwin (1981a: 66) remarked that the male genitalia, tarsi, and adult chemical defences suggest that pseudomorphines could be “related somehow to a basal Pterostichitae stock” and that the “paramere vestiture also suggests a connection with Psydritae.” Liebherr and Will (1998: 144) indicated that pseudomorphines, geobaenines, lachnophorines, and odacanthines (including pentagonicines) may be closely related based on the presence of an elongate sclerite joining the spermathecal duct to the common oviduct. Erwin and Stork (1985: 445) concluded that {pseudomorphines + cnemacanthines (= cnemalobines) + scaritines} form a clade that could be the sister-group to Hiletini. Jeannel (1942: 1102; 1949a: 1079) associated pseudomorphines with brachinines under the name Balteifera but almost all authors now agree that the two lineages are not closely related. Ober and Maddison (2008: 18-19) reported strong molecular support for a clade including pseudomorphines, orthogoniines, and graphipterines. They indicated that while morphology does not support such a close relationship, all or some of the members of each tribe have obligate relationships with social insects.
Brief faunistic assessmentThe North American fauna currently consists of 2, 676 valid species-group taxa (2, 439 species) of Geadephaga. Of this number, 64 are adventive on this continent, leaving 2, 612 (97.6%) native species-group taxa (2, 375 species).
Adventive species. Table 4 lists the adventive species found on this continent. All but two are accidental introductions (i.e., immigrants). Several carabids were intentionally introduced in New England during the first half of the XX Century for gypsy moth (Lymantria dispar Linnaeus) control, including Calosoma chinense Kirby, Calosoma inquisitor Linnaeus, Calosoma reticulatum Fabricius, Carabus arvensis Herbst, Carabus violaceus Linnaeus, Carabus glabratus Paykull, and Carabus coriaceus Linnaeus (see Smith 1959), but only Calosoma sycophanta and Carabus auratus auratus have become established.
Among the 62 species accidentally introduced, Laemostenus complanatus, Laemostenus terricola terricola, Somotrichus unifasciatus, Plochionus pallens, and Perigona nigriceps are commonly found in cellars or associated with stored products and are now considered subcosmopolitan (Hinton 1945: 21, 27-34). These species have probably been introduced in food and household goods in North America and many other places in the world.
Most of the remaining 57 adventive species found on this continent prior to the XX Century have probably been introduced with bulk rock, bricks, mortar and no doubt soil taken aboard sailing vessels as ballast and dump ashore on this continent (Brown 1940a; Lindroth 1957c). In the XX Century, species have been introduced into North America likely through shipments of nursery stock, within the soil bagged around the roots, as suggested by Kavanaugh and Erwin (1985), Spence and Spence (1988: 166), and Bousquet (1992a). Despite that quarantine regulations were established in the 1960s for the importation of nursery stock, additional species still continue to land and become established on this continent (Fig. 1). The most recent one is Nebria brevicollis in the Willamette Valley in northwestern Oregon (Kavanaugh and LaBonte 2008).
All but four of the adventive species, other than those introduced with stored food products, occur naturally in Europe and their North American populations likely originated from that continent and particularly from southwestern England (Lindroth 1957c: 172). Clivina vespertina and Tetragonoderus laevigatus are native to South America (Nichols 1985b: 380; Shpeley and Ball 2008) while Metacolpodes buchanani (see Habu 1978: 125) and Mochtherus tetraspilotus (see Habu 1982: 87) are native to Asia.
As far as known, none of the adventive species of carabids found in North America are invasive in that they threaten ecosystems, habitats, or even native species.
Figure 1 illustrates the arrival of adventive species in North America through time based on the collecting date of the first inventoried specimen. The rate of arrival has been steady in the XX Century.
List of species-group taxa adventive and established in North America.
| Species | Introduced in | YFC | ||
|---|---|---|---|---|
| East | West | South | ||
| Nebriini | ||||
| Leistus ferrugineus (Linnaeus) | • | 1977 | ||
| Nebria brevicollis (Fabricius) | • | 2007 | ||
| Notiophilini | ||||
| Notiophilus biguttatus (Fabricius) | • | • | 1923 | |
| Notiophilus palustris (Duftschmid) | • | 1968 | ||
| Carabini | ||||
| Calosoma sycophanta (Linnaeus)* | • | N/A | ||
| Carabus granulatus granulatus Linnaeus | • | • | 1890 | |
| Carabus nemoralis nemoralis O.F. Müller | • | • | 1890 | |
| Carabus auratus auratus Linnaeus* | • | N/A | ||
| Clivinini | ||||
| Clivina vespertina Putzeys | • | 1948 | ||
| Clivina collaris (Herbst) | • | • | <1838 | |
| Clivina fossor fossor (Linnaeus) | • | • | 1915 | |
| Dyschiriini | ||||
| Dyschirius globosus Herbst | • | 1978 | ||
| Broscini | ||||
| Broscus cephalotes (Linnaeus) | • | 1987 | ||
| Trechini | ||||
| Blemus discus discus (Fabricius) | • | 1933 | ||
| Trechus obtusus Erichson | • | 1927 | ||
| Trechus quadristriatus (Schrank) | • | 1965 | ||
| Trechus rubens (Fabricius) | • | <1863 | ||
| Bembidiini | ||||
| Asaphidion curtum curtum (Heyden) | • | 1930 | ||
| Bembidion lampros (Herbst) | • | • | 1947 | |
| Bembidion properans (Stephens) | • | 1942 | ||
| Bembidion obtusum Audinet-Serville | • | 1956 | ||
| Bembidion nigropiceum (Marsham) | • | <1897 | ||
| Bembidion stephensii Crotch | • | 1891 | ||
| Bembidion bruxellense Wesmael | • | 1907 | ||
| Bembidion femoratum femoratum Sturm | • | 1967 | ||
| Bembidion tetracolum tetracolum Say | • | • | <1823 | |
| Elaphropus parvulus (Dejean) | • | 1940 | ||
| Porotachys bisulcatus (Nicolai) | • | • | <1900 | |
| Pterostichini | ||||
| Stomis pumicatus (Panzer) | • | 1984 | ||
| Pterostichus vernalis (Panzer) | • | 1997 | ||
| Pterostichus strenuus (Panzer) | • | • | 1937 | |
| Pterostichus melanarius melanarius (Illiger) | • | • | 1926 | |
| Abax parallelepipedus (Piller & Mitterpacher) | • | 1965 | ||
| Zabrini | ||||
| Amara aulica (Panzer) | • | 1929 | ||
| Amara apricaria (Paykull) | • | ? | <1865 | |
| Amara fulva (O.F. Müller) | • | 1905 | ||
| Amara bifrons (Gyllenhal) | • | 1929 | ||
| Amara ovata (Fabricius) | • | • | 1925 | |
| Amara aenea (DeGeer) | • | • | 1904 | |
| Amara anthobia Villa & Villa | • | • | 1929 | |
| Amara communis (Panzer) | • | 1988 | ||
| Amara eurynota (Panzer) | • | 1971 | ||
| Amara familiaris (Duftschmid) | • | • | 1901 | |
| Harpalini | ||||
| Anisodactylus binotatus (Fabricius) | • | 1911 | ||
| Bradycellus harpalinus (Audinet-Serville) | • | 1951 | ||
| Acupalpus meridianus (Linnaeus) | • | • | 1931 | |
| Ophonus puncticeps Stephens | • | 1954 | ||
| Ophonus rufibarbis (Fabricius) | • | 1953 | ||
| Harpalus rufipes (DeGeer) | • | 1937 | ||
| Harpalus affinis (Schrank) | • | • | <1798 | |
| Harpalus rubripes (Duftschmid) | • | 1981 | ||
| Platynini | ||||
| Calathus fuscipes (Goeze) | • | 1928 | ||
| Laemostenus complanatus (Dejean) | • | <1874 | ||
| Laemostenus terricola terricola (Herbst) | • | ? | <1894 | |
| Paranchus albipes (Fabricius) | • | <1835 | ||
| Agonum muelleri (Herbst) | • | • | 1840 | |
| Metacolpodes buchanani (Hope) | • | 1931 | ||
| Perigonini | ||||
| Perigona nigriceps (Dejean) | • | • | <1853 | |
| Cyclosomini | ||||
| Tetragonoderus laevigatus Chaudoir | • | 2007 | ||
| Lebiini | ||||
| Mochtherus tetraspilotus (Macleay) | • | 1992 | ||
| Somotrichus unifasciatus (Dejean) | • | ? | ||
| Dromius fenestratus (Fabricius) | • | 1952 | ||
| Philorhizus melanocephalus (Dejean) | • | 1996 | ||
| Plochionus pallens (Fabricius) | • | ? | • | <1833 |
YFC: Year of first collection
Native species. The known North American fauna contains 2, 612 native species-group taxa (2, 375 species), of which 2, 071 are endemic (79.3%). Among the 541 non-endemic species-group taxa, 98 (3.7% of the entire native fauna) are Holarctic and 446 (17.1%) extend south of North America as defined in this catalogue.
The number of 2, 612 native species-group taxa obviously does not give an accurate account of the size of the North American carabid fauna. Several factors are involved. First, 65 species-group taxa described by Casey have not been treated subsequently. Considering that only 13.1% of Casey’s North American species-group taxa subsequently treated are currently valid, I estimate that about 50 of the remaining Casey taxa will eventually be synonymized. This would bring the number of valid Nearctic species-group taxa to about 2, 560 (2, 325 species). Second, several genus-group taxa have not been revised in “modern times” and obviously the number of valid species-group taxa will change. The following generic taxa are those that need revisions: Clivina Latreille (except Antroforceps), Paraclivina Kult, Elaphropus Motschulsky, Tachys Dejean, Poecilus Bonelli, Hypherpes Chaudoir, Stenocellus Casey, Selenophorus Dejean, Discoderus LeConte, Olisthopus Dejean, Pinacodera Schaum, Apristus Chaudoir, Axinopalpus LeConte, Zuphium Latreille, Pseudaptinus Laporte, and Pseudomorpha Kirby. Third, many species-group taxa are known but still undescribed. For example, Barr (2004: 1) reported that he was aware of 80 undescribed species of Pseudanophthalmus. Fourth, through modern techniques, such as DNA sequences, several “species” will probably be found to be a complex of two or more species. For example, Maddison (2008) found out that Lindroth’s (1963b) concept of Bembidion chalceum and Bembidion honestum was a complex of seven species. Finally, there is no doubt that many species remain to be discovered in North America, especially in peculiar habitats, such as caves, burrows, and deep litter.
Figure 2 illustrates through decades the number of North American species-group taxa described and the number of those regarded as valid today based on the present catalogue. It shows that the periods between 1821-1880, 1911-1930, and 1961-1990 have been the most prolific. The first one, 1821-1880, can be associated with the works of John L. LeConte (724 geadephagan species-group taxa described), Count Dejean (289 taxa), Thomas Say (164 taxa), Baron de Chaudoir (126 taxa), and Victor Motschulsky (121 taxa). The second period, 1911-1930, relates of course to the work of Colonel Casey. By the time of his last publication in 1924, Casey had described 1, 864 species-group taxa of carabids based on North American specimens while the number of taxa proposed by all authors combined since Linnaeus amounted to 2, 288. The third one, 1961-1990, is connected to the magnificent work of Carl H. Lindroth (76 taxa) on the Carabidae of Canada and Alaska, the work of Thomas Barr (147 taxa) on the cave fauna, and the leadership of George Ball who, besides his own research, directed many students.
Figure 3 shows the growth, through time, of the number of species-group taxa described from North American specimens and those still considered valid. Taking into consideration the trend of description of species-group taxa still valid, the number of native species-group taxa should be slightly over 3, 000 by the year 2060.
Number of adventive species of Geadephaga established in North America through time.
Number of North American species-group taxa of Geadephaga described and currently valid, by decade.
Cumulative number of species-group taxa of Geadephaga described and currently valid, by decade.
Political regions. Table 5 gives statistics regarding the number of geadephagan species-group taxa for each political region covered in this catalogue.
Texas (693 native species-group taxa), California (646 taxa), North Carolina (536 taxa), New York (528 taxa), and Virginia (520 taxa) have the highest number of native taxa recorded and Greenland (4 native taxa), Nunavut (34 taxa), Saint Pierre and Miquelon (50 taxa), Labrador (91 taxa), and Prince Edward Island (144 taxa) the lowest. Are these numbers representative of the relative sizes of the regions? Only to some extent. Some regions, for example Nunavut and Labrador, have been much less surveyed than others like Newfoundland proper. The geadephagan fauna of most states are still inadequately known. In the past 50 years, only Maine (Majka et al. 2011; Bousquet 2010a), New Hampshire and Vermont (Bousquet 2010a), Wisconsin (Messer 2010), Rhode Island (Sikes 2004), Connecticut (Krinsky and Oliver 2001), South Carolina (Ciegler 2000), Florida (Peck and Thomas 1998), South Dakota (Kirk and Balsbaugh 1975), and Alaska (Lindroth 1961-1969) have been the subject of faunistic assessments (see Table 3). Several states never had a checklist of their geadephagan fauna published.
Species-group taxon statistics for political regions.
| Political regions | NbNT | NbAd | %NF | Rank |
|---|---|---|---|---|
| Alabama | 439 | 4 | 16.8 | 22 |
| Alaska | 248 | 2 | 09.5 | 56 |
| Alberta | 405 | 11 | 15.5 | 32 |
| Arizona | 502 | 3 | 19.2 | 9 |
| Arkansas | 403 | 4 | 15.5 | 33 |
| British Columbia | 479 | 28 | 18.3 | 12 |
| California | 646 | 12 | 24.7 | 2 |
| Colorado | 450 | 3 | 17.2 | 20 |
| Connecticut | 383 | 23 | 14.7 | 39 |
| Delaware | 201 | 5 | 07.7 | 59 |
| District of Columbia | 337 | 6 | 12.9 | 48 |
| Florida | 383 | 8 | 14.7 | 39 |
| Georgia | 478 | 3 | 18.3 | 14 |
| Greenland | 4 | 0 | 0.2 | 66 |
| Idaho | 338 | 10 | 12.9 | 47 |
| Illinois | 504 | 9 | 19.3 | 8 |
| Indiana | 479 | 9 | 18.3 | 12 |
| Iowa | 431 | 9 | 16.5 | 25 |
| Kansas | 408 | 4 | 15.6 | 30 |
| Kentucky | 371 | 2 | 14.2 | 42 |
| Labrador | 91 | 4 | 03.5 | 63 |
| Louisiana | 399 | 4 | 15.3 | 34 |
| Maine | 383 | 27 | 14.7 | 39 |
| Manitoba | 370 | 6 | 14.1 | 43 |
| Maryland | 408 | 9 | 15.5 | 30 |
| Massachusetts | 426 | 27 | 16.3 | 27 |
| Michigan | 474 | 15 | 18.2 | 16 |
| Minnesota | 419 | 10 | 16.0 | 28 |
| Mississippi | 439 | 5 | 16.8 | 22 |
| Missouri | 396 | 5 | 15.1 | 36 |
| Montana | 358 | 6 | 13.6 | 45 |
| Nebraska | 358 | 4 | 13.7 | 45 |
| Nevada | 239 | 3 | 09.2 | 57 |
| New Brunswick | 302 | 30 | 11.6 | 52 |
| Newfoundland | 151 | 27 | 05.8 | 61 |
| New Hampshire | 429 | 25 | 16.4 | 26 |
| New Jersey | 456 | 10 | 17.5 | 18 |
| New Mexico | 454 | 0 | 17.3 | 19 |
| New York | 528 | 21 | 20.2 | 4 |
| North Carolina | 536 | 4 | 20.4 | 3 |
| North Dakota | 275 | 4 | 10.5 | 54 |
| Northwest Territories | 218 | 1 | 08.3 | 58 |
| Nova Scotia | 258 | 35 | 09.8 | 55 |
| Nunavut | 34 | 0 | 01.3 | 65 |
| Ohio | 499 | 16 | 19.1 | 11 |
| Oklahoma | 387 | 2 | 14.8 | 37 |
| Ontario | 510 | 21 | 19.5 | 6 |
| Oregon | 478 | 22 | 18.2 | 14 |
| Pennsylvania | 500 | 19 | 19.1 | 10 |
| Prince Edward Island | 144 | 29 | 05.5 | 62 |
| Quebec | 446 | 32 | 17.1 | 21 |
| Rhode Island | 300 | 17 | 11.5 | 53 |
| Saint Pierre and Miquelon | 50 | 15 | 01.9 | 64 |
| Saskatchewan | 336 | 6 | 12.9 | 49 |
| South Carolina | 414 | 4 | 15.8 | 29 |
| South Dakota | 384 | 4 | 14.7 | 38 |
| Tennessee | 506 | 2 | 19.3 | 7 |
| Texas | 693 | 1 | 26.5 | 1 |
| Utah | 323 | 5 | 12.4 | 50 |
| Vermont | 397 | 21 | 15.2 | 35 |
| Virginia | 520 | 11 | 19.9 | 5 |
| Washington | 440 | 24 | 16.8 | 22 |
| West Virginia | 367 | 7 | 14.1 | 44 |
| Wisconsin | 463 | 16 | 17.7 | 17 |
| Wyoming | 309 | 4 | 11.8 | 51 |
| Yukon Territory | 201 | 0 | 07.7 | 59 |
NbNT = Number of native species-group taxa
NbAd = Number of adventive species-group taxa
%NF = % of the native North American fauna
Checklists and taxonomic treatments on Geadephaga for North American political regions.
| Political regions | References |
|---|---|
| Alabama | Löding (1945) |
| Alaska | Hamilton (1894a); Lindroth (1961-1969); Bousquet (1991b) |
| Alberta | Lindroth (1961-1969); Bousquet (1991b) |
| British Columbia | Hatch (1953); Lindroth (1961-1969); Bousquet (1991b) |
| Colorado | Wickham (1902) |
| Connecticut | Britton (1920); Krinsky and Oliver (2001) |
| District of Columbia | Ulke (1902) |
| Florida | Schwarz (1878); Leng (1915); Choate (1990); Peck and Thomas (1998) |
| Georgia | Fattig (1949) |
| Greenland | Henriksen (1939); Böcher (1988) |
| Idaho | Hatch (1953) |
| Indiana | Blatchley (1910) |
| Iowa | Wickham (1911b); Jaques and Redlinger (1946) |
| Kansas | Popenoe (1877) |
| Labrador | Sherman (1910); Lindroth (1961-1969); Bousquet (1991b); Bousquet (2010a) |
| Maine | Bousquet (2010a); Majka et al. (2011) |
| Manitoba | Lindroth (1961-1969); Bousquet (1991b) |
| Massachusetts | Harris (1833); Harris (1835) |
| New Brunswick | Lindroth (1961-1969); Bousquet (1991b); Majka et al. (2007); Bousquet (2010a) |
| Newfoundland | Lindroth (1955a); Lindroth (1961-1969); Bousquet (1991b); Bousquet (2010a) |
| New Hampshire | Bousquet (2010a) |
| New Jersey | Smith (1890); Smith (1900); Smith (1910) |
| New Mexico | Fall and Cockerell (1907) |
| New York | Notman (1928) |
| North Carolina | Brimley (1938) |
| Northwest Territories | Lindroth (1961-1969); Bousquet (1991b) |
| Nova Scotia | Lindroth (1961-1969); Lindroth (1954c); Bousquet (1991b); Majka et al. (2007); Bousquet (2010a) |
| Ontario | Lindroth (1961-1969); Bousquet (1991b) |
| Oregon | Hatch (1953) |
| Prince Edward Island | Lindroth (1961-1969); Bousquet (1991b); Majka et al. (2007); Majka et al. (2008); Bousquet (2010a) |
| Quebec | Chagnon (1917); Lindroth (1961-1969); Larochelle (1975); Larochelle (1976); Larochelle (1979); Bousquet (1991b); Laplante et al. (1991); Bousquet (2010a) |
| Rhode Island | Davis (1904); Sikes (2004) |
| Saint Pierre and Miquelon | Lindroth (1955a); Roux (1984); Bousquet (2010a) |
| Saskatchewan | Lindroth (1961-1969); Bousquet (1991b) |
| South Carolina | Kirk (1969 and 1970); Ciegler (2000) |
| South Dakota | Kirk and Balsbaugh (1975) |
| Vermont | Bousquet (2010a) |
| Washington | Hatch (1939b); Hatch (1953) |
| Wisconsin | Messer (2010) |
| Yukon Territory | Lindroth (1961-1969); Bousquet (1991b); Ball and Currie (1997) |
Trachypachus gibbsii LeConte, 1861
Trachypachus inermis Motschulsky, 1850
Trachypachus slevini Van Dyke, 1925
ClinidiiniClinidium (Arctoclinidium) apertum allegheniense Bell & Bell, 1975
Clinidium (Arctoclinidium) apertum apertum Reitter, 1880
Clinidium (Arctoclinidium) baldufi Bell, 1970
Clinidium (Arctoclinidium) calcaratum LeConte, 1875
Clinidium (Arctoclinidium) rosenbergi Bell, 1970
Clinidium (Arctoclinidium) sculptile (Newman, 1838)
Clinidium (Arctoclinidium) valentinei Bell, 1970
OmoglymmiiniOmoglymmius (Boreoglymmius) americanus (Laporte, 1836)
Omoglymmius (Boreoglymmius) hamatus (LeConte, 1875)
PelophiliniPelophila borealis (Paykull, 1790)‡
Pelophila rudis (LeConte, 1863)
OpisthiiniOpisthius richardsoni Kirby, 1837
NebriiniLeistus (Leistus) ferrugineus (Linnaeus, 1758)†
Leistus (Neoleistus) ferruginosus Mannerheim, 1843
Leistus (Neoleistus) longipennis Casey, 1920
Leistus (Neoleistus) madmeridianus Erwin, 1970
Nippononebria (Vancouveria) altisierrae (Kavanaugh, 1984)
Nippononebria (Vancouveria) campbelli (Kavanaugh, 1984)
Nippononebria (Vancouveria) virescens (Horn, 1870)
Nebria (Boreonebria) bellorum Kavanaugh, 1979
Nebria (Boreonebria) crassicornis crassicornis Van Dyke, 1925
Nebria (Boreonebria) crassicornis intermedia Van Dyke, 1949
Nebria (Boreonebria) frigida Sahlberg, 1844‡
Nebria (Boreonebria) gouleti Kavanaugh, 1979
Nebria (Boreonebria) gyllenhali castanipes (Kirby, 1837)
Nebria (Boreonebria) gyllenhali lassenensis Kavanaugh, 1979
Nebria (Boreonebria) gyllenhali lindrothi Kavanaugh, 1979
Nebria (Boreonebria) hudsonica LeConte, 1863
Nebria (Boreonebria) lacustris Casey, 1913
Nebria (Boreonebria) nivalis gaspesiana Kavanaugh, 1979
Nebria (Boreonebria) nivalis nivalis (Paykull, 1790)‡
Nebria (Nakanebria) paradisi Darlington, 1931
Nebria (Nakanebria) turmaduodecima Kavanaugh, 1981
Nebria (Reductonebria) acuta acuta Lindroth, 1961
Nebria (Reductonebria) acuta quileute Kavanaugh, 1979
Nebria (Reductonebria) acuta sonorae Kavanaugh, 1981
Nebria (Reductonebria) appalachia Darlington, 1932
Nebria (Reductonebria) arkansana arkansana Casey, 1913
Nebria (Reductonebria) arkansana edwardsi Kavanaugh, 1979
Nebria (Reductonebria) arkansana fragilis Casey, 1924
Nebria (Reductonebria) arkansana oowah Kavanaugh, 1979
Nebria (Reductonebria) charlottae Lindroth, 1961
Nebria (Reductonebria) danmanni Kavanaugh, 1981
Nebria (Reductonebria) darlingtoni Kavanaugh, 1979
Nebria (Reductonebria) desolata Kavanaugh, 1971
Nebria (Reductonebria) diversa LeConte, 1863
Nebria (Reductonebria) eschscholtzii Ménétriés, 1843
Nebria (Reductonebria) georgei Kavanaugh, 2008
Nebria (Reductonebria) gregaria Fischer von Waldheim, 1820
Nebria (Reductonebria) haida Kavanaugh, 1984
Nebria (Reductonebria) jeffreyi Kavanaugh, 1984
Nebria (Reductonebria) lituyae Kavanaugh, 1979
Nebria (Reductonebria) louiseae Kavanaugh, 1984
Nebria (Reductonebria) lyelli Van Dyke, 1925
Nebria (Reductonebria) mannerheimii Fischer von Waldheim, 1828
Nebria (Reductonebria) navajo Kavanaugh, 1979
Nebria (Reductonebria) obliqua chuskae Kavanaugh, 1979
Nebria (Reductonebria) obliqua obliqua LeConte, 1867
Nebria (Reductonebria) pallipes Say, 1823
Nebria (Reductonebria) sahlbergii modoc Kavanaugh, 1979
Nebria (Reductonebria) sahlbergii sahlbergii Fischer von Waldheim, 1828
Nebria (Reductonebria) sahlbergii triad Kavanaugh, 1979
Nebria (Reductonebria) suturalis LeConte, 1850
Nebria (Reductonebria) wallowae Kavanaugh, 1984
Nebria (Reductonebria) zioni oasis Kavanaugh, 1979
Nebria (Reductonebria) zioni zioni Van Dyke, 1943
Nebria (Catonebria) calva Kavanaugh, 1984
Nebria (Catonebria) carri Kavanaugh, 1979
Nebria (Catonebria) catenata Casey, 1913
Nebria (Catonebria) coloradensis Van Dyke, 1943
Nebria (Catonebria) gebleri albimontis Kavanaugh, 1984
Nebria (Catonebria) gebleri cascadensis Kavanaugh, 1979
Nebria (Catonebria) gebleri fragariae Kavanaugh, 1979
Nebria (Catonebria) gebleri gebleri Dejean, 1831
Nebria (Catonebria) gebleri rathvoni LeConte, 1853
Nebria (Catonebria) gebleri siskiyouensis Kavanaugh, 1979
Nebria (Catonebria) ingens ingens Horn, 1870
Nebria (Catonebria) ingens riversi Van Dyke, 1925
Nebria (Catonebria) kincaidi balli Kavanaugh, 1979
Nebria (Catonebria) kincaidi kincaidi Schwarz, 1900
Nebria (Catonebria) labontei Kavanaugh, 1984
Nebria (Catonebria) meanyi giulianii Kavanaugh, 1981
Nebria (Catonebria) meanyi lamarckensis Kavanaugh, 1979
Nebria (Catonebria) meanyi meanyi Van Dyke, 1925
Nebria (Catonebria) meanyi sylvatica Kavanaugh, 1979
Nebria (Catonebria) metallica Fischer von Waldheim, 1820
Nebria (Catonebria) ovipennis LeConte, 1878
Nebria (Catonebria) piperi Van Dyke, 1925
Nebria (Catonebria) piute piute Erwin & Ball, 1972
Nebria (Catonebria) piute sevieri Kavanaugh, 1984
Nebria (Catonebria) piute utahensis Kavanaugh, 1979
Nebria (Catonebria) praedicta Kavanaugh & Schoville, 2009
Nebria (Catonebria) purpurata LeConte, 1878
Nebria (Catonebria) schwarzi beverlianna Kavanaugh, 1979
Nebria (Catonebria) schwarzi schwarzi Van Dyke, 1925
Nebria (Catonebria) sierrablancae Kavanaugh, 1984
Nebria (Catonebria) spatulata sierrae Kavanaugh, 1979
Nebria (Catonebria) spatulata spatulata Van Dyke, 1925
Nebria (Catonebria) steensensis Kavanaugh, 1984
Nebria (Catonebria) trifaria pasquineli Kavanaugh, 1984
Nebria (Catonebria) trifaria trifaria LeConte, 1878
Nebria (Catonebria) vandykei vandykei Bänninger, 1928
Nebria (Catonebria) vandykei wyeast Kavanaugh, 1979
Nebria (Nebria) brevicollis (Fabricius, 1792)†
NotiophiliniNotiophilus aeneus (Herbst, 1806)
Notiophilus aquaticus (Linnaeus, 1758)‡
Notiophilus biguttatus (Fabricius, 1779)†
Notiophilus borealis Harris, 1869‡
Notiophilus directus Casey, 1920
Notiophilus intermedius Lindroth, 1955
Notiophilus nemoralis Fall, 1906
Notiophilus nitens LeConte, 1857
Notiophilus novemstriatus LeConte, 1847
Notiophilus palustris (Duftschmid, 1812)†
Notiophilus semiopacus Eschscholtz, 1833
Notiophilus semistriatus Say, 1823‡
Notiophilus sierranus Casey, 1920
Notiophilus simulator Fall, 1906
Notiophilus sylvaticus Dejean, 1831
CychriniSphaeroderus bicarinatus (LeConte, 1853)
Sphaeroderus canadensis canadensis Chaudoir, 1861
Sphaeroderus canadensis lengi Darlington, 1933
Sphaeroderus indianae (Blatchley, 1910)
Sphaeroderus nitidicollis Guérin-Méneville, 1829
Sphaeroderus schaumii Chaudoir, 1861
Sphaeroderus stenostomus lecontei Dejean, 1826
Sphaeroderus stenostomus stenostomus (Weber, 1801)
Cychrus hemphillii hemphillii Horn, 1879
Cychrus hemphillii rickseckeri LeConte, 1884
Cychrus tuberculatus Harris, 1839
Scaphinotus (Scaphinotus) elevatus coloradensis Van Dyke, 1907
Scaphinotus (Scaphinotus) elevatus elevatus (Fabricius, 1787)
Scaphinotus (Scaphinotus) elevatus flammeus Haldeman, 1844
Scaphinotus (Scaphinotus) elevatus lengi Van Dyke, 1938
Scaphinotus (Scaphinotus) elevatus neomexicanus Van Dyke, 1924
Scaphinotus (Scaphinotus) elevatus tenebricosus Roeschke, 1907
Scaphinotus (Scaphinotus) kelloggi (Dury, 1912)
Scaphinotus (Scaphinotus) parisiana Allen & Carlton, 1988
Scaphinotus (Scaphinotus) petersi biedermani Roeschke, 1907
Scaphinotus (Scaphinotus) petersi catalinae Van Dyke, 1924
Scaphinotus (Scaphinotus) petersi corvus (Fall, 1910)
Scaphinotus (Scaphinotus) petersi grahami Van Dyke, 1938
Scaphinotus (Scaphinotus) petersi kathleenae Ball, 1966
Scaphinotus (Scaphinotus) petersi petersi Roeschke, 1907
Scaphinotus (Scaphinotus) snowi roeschkei Van Dyke, 1907
Scaphinotus (Scaphinotus) snowi snowi (LeConte, 1881)
Scaphinotus (Scaphinotus) unicolor (Fabricius, 1787)
Scaphinotus (Scaphinotus) vandykei Roeschke, 1907
Scaphinotus (Irichroa) irregularis (Beutenmüller, 1903)
Scaphinotus (Irichroa) viduus (Dejean, 1826)
Scaphinotus (Irichroa) webbi Bell, 1959
Scaphinotus (Nomaretus) bilobus (Say, 1823)
Scaphinotus (Nomaretus) cavicollis (LeConte, 1859)
Scaphinotus (Nomaretus) fissicollis (LeConte, 1853)
Scaphinotus (Nomaretus) infletus Allen & Carlton, 1988
Scaphinotus (Nomaretus) liebecki Van Dyke, 1936
Scaphinotus (Maronetus) debilis alpinus (Beutenmüller, 1903)
Scaphinotus (Maronetus) debilis debilis (LeConte, 1853)
Scaphinotus (Maronetus) hoffmani (Barr, 2009)
Scaphinotus (Maronetus) hubbardi (Schwarz, 1895)
Scaphinotus (Maronetus) imperfectus (Horn, 1861)
Scaphinotus (Maronetus) incompletus (Schwarz, 1895)
Scaphinotus (Maronetus) reichlei (Barr, 2009)
Scaphinotus (Maronetus) schwarzi (Beutenmüller, 1913)
Scaphinotus (Maronetus) tenuis (Casey, 1914)
Scaphinotus (Maronetus) unistriatus (Darlington, 1932)
Scaphinotus (Steniridia) aeneicollis (Beutenmüller, 1903)
Scaphinotus (Steniridia) andrewsii amplicollis (Casey, 1920)
Scaphinotus (Steniridia) andrewsii andrewsii (Harris, 1839)
Scaphinotus (Steniridia) andrewsii darlingtoni (Valentine, 1935)
Scaphinotus (Steniridia) andrewsii germari (Chaudoir, 1861)
Scaphinotus (Steniridia) andrewsii mutabilis (Casey, 1920)
Scaphinotus (Steniridia) andrewsii parvitarsalis (Valentine, 1935)
Scaphinotus (Steniridia) andrewsii waldensius (Valentine, 1935)
Scaphinotus (Steniridia) guyotii (LeConte, 1863)
Scaphinotus (Steniridia) lodingi lodingi (Valentine, 1935)
Scaphinotus (Steniridia) lodingi obscurus (Valentine, 1935)
Scaphinotus (Steniridia) ridingsii monongahelae Leng, 1917
Scaphinotus (Steniridia) ridingsii ridingsii (Bland, 1863)
Scaphinotus (Steniridia) tricarinatus (Casey, 1914)
Scaphinotus (Steniridia) violaceus (LeConte, 1863)
Scaphinotus (Pseudonomaretus) mannii Wickham, 1919
Scaphinotus (Pseudonomaretus) merkelii (Horn, 1890)
Scaphinotus (Pseudonomaretus) regularis (LeConte, 1884)
Scaphinotus (Pseudonomaretus) relictus (Horn, 1881)
Scaphinotus (Stenocantharus) angusticollis (Mannerheim, 1823)
Scaphinotus (Stenocantharus) hatchi Beer, 1971
Scaphinotus (Stenocantharus) johnsoni Van Dyke, 1924
Scaphinotus (Stenocantharus) velutinus (Ménétriés, 1843)
Scaphinotus (Brennus) bullatus Van Dyke, 1924
Scaphinotus (Brennus) cordatus (LeConte, 1853)
Scaphinotus (Brennus) crenatus (Motschulsky, 1859)
Scaphinotus (Brennus) cristatus (Harris, 1839)
Scaphinotus (Brennus) interruptus (Ménétriés, 1843)
Scaphinotus (Brennus) marginatus (Fischer von Waldheim, 1820)
Scaphinotus (Brennus) obliquus (LeConte, 1868)
Scaphinotus (Brennus) oreophilus (Rivers, 1890)
Scaphinotus (Brennus) punctatus (LeConte, 1859)
Scaphinotus (Brennus) riversi (Roeschke, 1907)
Scaphinotus (Brennus) rugiceps incipiens (Casey, 1897)
Scaphinotus (Brennus) rugiceps rugiceps (Horn, 1872)
Scaphinotus (Brennus) striatopunctatus (Chaudoir, 1844)
Scaphinotus (Brennus) subtilis (Schaum, 1863)
Scaphinotus (Brennus) ventricosus (Dejean, 1831)
Scaphinotus (Neocychrus) angulatus (Harris, 1839)
Scaphinotus (Neocychrus) behrensi (Roeschke, 1907)
Scaphinotus (Neocychrus) longiceps Van Dyke, 1924
CarabiniCalosoma (Castrida) sayi Dejean, 1826
Calosoma (Calosoma) frigidum Kirby, 1837
Calosoma (Calosoma) sycophanta (Linnaeus, 1758)†
Calosoma (Calodrepa) aurocinctum Chaudoir, 1850
Calosoma (Calodrepa) scrutator (Fabricius, 1775)
Calosoma (Calodrepa) splendidum Dejean, 1831
Calosoma (Calodrepa) wilcoxi LeConte, 1847
Calosoma (Camegonia) marginale Casey, 1897
Calosoma (Camegonia) parvicolle Fall, 1910
Calosoma (Camegonia) prominens LeConte, 1853
Calosoma (Carabosoma) angulatum Chevrolat, 1834
Calosoma (Carabosoma) eremicola Fall, 1910
Calosoma (Carabosoma) peregrinator Guérin-Méneville, 1844
Calosoma (Carabosoma) sponsa Casey, 1897
Calosoma (Callitropa) externum (Say, 1823)
Calosoma (Callitropa) macrum LeConte, 1853
Calosoma (Callitropa) protractum LeConte, 1862
Calosoma (Blaptosoma) haydeni haydeni Horn, 1870
Calosoma (Blaptosoma) haydeni punctulicolle Bates, 1891
Calosoma (Chrysostigma) affine Chaudoir, 1843
Calosoma (Chrysostigma) calidum (Fabricius, 1775)
Calosoma (Chrysostigma) cancellatum Eschscholtz, 1833
Calosoma (Chrysostigma) lepidum LeConte, 1845
Calosoma (Chrysostigma) morrisonii Horn, 1885
Calosoma (Chrysostigma) obsoletum Say, 1823
Calosoma (Chrysostigma) semilaeve LeConte, 1852
Calosoma (Chrysostigma) simplex LeConte, 1878
Calosoma (Chrysostigma) tepidum LeConte, 1852
Calosoma (Callistenia) dawsoni (Dajoz, 1997)
Calosoma (Callistenia) dietzii Schaeffer, 1904
Calosoma (Callistenia) discors LeConte, 1857
Calosoma (Callistenia) lariversi Van Dyke, 1943
Calosoma (Callistenia) latipenne Horn, 1870
Calosoma (Callistenia) luxatum Say, 1823
Calosoma (Callistenia) moniliatum (LeConte, 1852)
Calosoma (Callistenia) monticola Casey, 1897
Calosoma (Callistenia) oregonum (Gidaspow, 1959)
Calosoma (Callistenia) placerum (Gidaspow, 1959)
Calosoma (Callistenia) schaefferi Breuning, 1928
Calosoma (Callistenia) subaeneum Chaudoir, 1869
Calosoma (Callistenia) subasperatum Schaeffer, 1915
Calosoma (Callistenia) wilkesii (LeConte, 1852)
Carabus (Carabus) goryi Dejean, 1831
Carabus (Carabus) granulatus granulatus Linnaeus, 1758†
Carabus (Carabus) vinctus (Weber, 1801)
Carabus (Diocarabus) chamissonis Fischer von Waldheim, 1820
Carabus (Aulonocarabus) truncaticollis truncaticollis Eschscholtz, 1833‡
Carabus (Homoeocarabus) maeander maeander Fischer von Waldheim, 1820‡
Carabus (Hemicarabus) serratus Say, 1823
Carabus (Archicarabus) nemoralis nemoralis Müller, 1764†
Carabus (Tachypus) auratus auratus Linnaeus, 1760†
Carabus (Tanaocarabus) finitimus Haldeman, 1852
Carabus (Tanaocarabus) forreri forreri Bates, 1882
Carabus (Tanaocarabus) sylvosus Say, 1823
Carabus (Tanaocarabus) taedatus agassii LeConte, 1850
Carabus (Tanaocarabus) taedatus bicanaliceps Casey, 1920
Carabus (Tanaocarabus) taedatus rainieri Van Dyke, 1945
Carabus (Tanaocarabus) taedatus taedatus Fabricius, 1787
Carabus (Megodontus) vietinghoffii vietinghoffii Adams, 1812‡
AmblycheiliniAmblycheila baroni Rivers, 1890
Amblycheila cylindriformis (Say, 1823)
Amblycheila hoversoni Gage, 1991
Amblycheila picolominii Reiche, 1840
Amblycheila schwarzi Horn, 1904
Omus audouini Reiche, 1838
Omus californicus angustocylindricus Horn, 1913
Omus californicus californicus Eschscholtz, 1829
Omus californicus intermedius Leng, 1902
Omus californicus subcylindricus Nunenmacher, 1940
Omus cazieri van den Berghe, 1994
Omus dejeanii Reiche, 1838
Omus submetallicus Horn, 1869
MegacephaliniTetracha (Tetracha) carolina carolina (Linnaeus, 1763)
Tetracha (Tetracha) floridana Leng & Mutchler, 1916
Tetracha (Tetracha) virginica (Linnaeus, 1767)
Tetracha (Neotetracha) impressa (Chevrolat, 1841)
CicindeliniCylindera (Cylindera) celeripes (LeConte, 1846)
Cylindera (Cylindera) cursitans (LeConte, 1856)
Cylindera (Cylindera) debilis (Bates, 1890)
Cylindera (Cylindera) lemniscata lemniscata (LeConte, 1854)
Cylindera (Cylindera) lemniscata rebaptisata (Vaurie, 1951)
Cylindera (Cylindera) lunalonga (Schaupp, 1884)
Cylindera (Cylindera) terricola cinctipennis (LeConte, 1846)
Cylindera (Cylindera) terricola continua (Knaus, 1923)
Cylindera (Cylindera) terricola imperfecta (LeConte, 1851)
Cylindera (Cylindera) terricola kaibabensis (Johnson, 1990)
Cylindera (Cylindera) terricola susanagreae (Kippenhan, 2007)
Cylindera (Cylindera) terricola terricola (Say, 1824)
Cylindera (Cylindera) unipunctata (Fabricius, 1775)
Cylindera (Cylindera) viridisticta arizonensis (Bates, 1884)
Ellipsoptera blanda (Dejean, 1831)
Ellipsoptera cuprascens (LeConte, 1852)
Ellipsoptera gratiosa (Guérin-Méneville, 1840)
Ellipsoptera hamata lacerata (Chaudoir, 1854)
Ellipsoptera hamata monti (Vaurie, 1951)
Ellipsoptera hirtilabris (LeConte, 1875)
Ellipsoptera lepida (Dejean, 1831)
Ellipsoptera macra ampliata (Vaurie, 1951)
Ellipsoptera macra fluviatilis (Vaurie, 1951)
Ellipsoptera macra macra (LeConte, 1856)
Ellipsoptera marginata (Fabricius, 1775)
Ellipsoptera nevadica citata (Rumpp, 1977)
Ellipsoptera nevadica knausii (Leng, 1902)
Ellipsoptera nevadica lincolniana (Casey, 1916)
Ellipsoptera nevadica makosika (Spomer, 2004)
Ellipsoptera nevadica nevadica (LeConte, 1875)
Ellipsoptera nevadica olmosa (Vaurie, 1951)
Ellipsoptera nevadica tubensis (Cazier, 1939)
Ellipsoptera puritana (Horn, 1871)
Ellipsoptera rubicunda (Harris, 1911)
Ellipsoptera sperata inquisitor (Casey, 1897)
Ellipsoptera sperata sperata (LeConte, 1856)
Ellipsoptera wapleri (LeConte, 1875)
Microthylax olivaceus (Chaudoir, 1854)
Opilidia chlorocephala smythi (Harris, 1913)
Brasiella wickhami (Horn, 1903)
Dromochorus belfragei Sallé, 1877
Dromochorus pilatei Guérin-Méneville, 1849
Dromochorus pruininus Casey, 1897
Dromochorus velutinigrens Johnson, 1992
Habroscelimorpha californica mojavi (Cazier, 1937)
Habroscelimorpha californica pseudoerronea (Rumpp, 1958)
Habroscelimorpha circumpicta circumpicta (LaFerté-Sénectère, 1841)
Habroscelimorpha circumpicta johnsonii (Fitch, 1857)
Habroscelimorpha circumpicta pembina (Johnson, 1993)
Habroscelimorpha dorsalis dorsalis (Say, 1817)
Habroscelimorpha dorsalis media (LeConte, 1856)
Habroscelimorpha dorsalis saulcyi (Guérin-Méneville, 1840)
Habroscelimorpha dorsalis venusta (LaFerté-Sénectère, 1841)
Habroscelimorpha fulgoris albilata (Acciavatti, 1981)
Habroscelimorpha fulgoris erronea (Vaurie, 1951)
Habroscelimorpha fulgoris fulgoris (Casey, 1913)
Habroscelimorpha gabbii (Horn, 1867)
Habroscelimorpha pamphila (LeConte, 1873)
Habroscelimorpha praetextata pallidofemora (Acciavatti, 1981)
Habroscelimorpha praetextata praetextata (LeConte, 1854)
Habroscelimorpha severa (LaFerté-Sénectère, 1841)
Habroscelimorpha striga (LeConte, 1875)
Eunota togata fascinans (Casey, 1914)
Eunota togata globicollis (Casey, 1913)
Eunota togata togata (LaFerté-Sénectère, 1841)
Cicindela (Cicindelidia) abdominalis Fabricius, 1801
Cicindela (Cicindelidia) amargosae amargosae Dahl, 1939
Cicindela (Cicindelidia) amargosae nyensis Rumpp, 1956
Cicindela (Cicindelidia) cazieri Vogt, 1949
Cicindela (Cicindelidia) floridana Cartwright, 1939
Cicindela (Cicindelidia) hemorrhagica arizonae Wickham, 1899
Cicindela (Cicindelidia) hemorrhagica hemorrhagica LeConte, 1851
Cicindela (Cicindelidia) highlandensis Choate, 1984
Cicindela (Cicindelidia) hornii hornii Schaupp, 1883
Cicindela (Cicindelidia) marginipennis Dejean, 1831
Cicindela (Cicindelidia) nigrocoerulea bowditchi Leng, 1902
Cicindela (Cicindelidia) nigrocoerulea nigrocoerulea LeConte, 1846
Cicindela (Cicindelidia) nigrocoerulea subtropica Vogt, 1949
Cicindela (Cicindelidia) obsoleta neojuvenilis Vogt, 1949
Cicindela (Cicindelidia) obsoleta obsoleta Say, 1823
Cicindela (Cicindelidia) obsoleta santaclarae Bates, 1890
Cicindela (Cicindelidia) obsoleta vulturina LeConte, 1853
Cicindela (Cicindelidia) ocellata ocellata Klug, 1834
Cicindela (Cicindelidia) ocellata rectilatera Chaudoir, 1843
Cicindela (Cicindelidia) politula barbaraannae Sumlin, 1976
Cicindela (Cicindelidia) politula petrophila Sumlin, 1985
Cicindela (Cicindelidia) politula politula LeConte, 1875
Cicindela (Cicindelidia) politula viridimonticola Gage, 1988
Cicindela (Cicindelidia) punctulata chihuahuae Bates, 1890
Cicindela (Cicindelidia) punctulata punctulata Olivier, 1790
Cicindela (Cicindelidia) roseiventris tascosaensis Davis, 1918
Cicindela (Cicindelidia) rufiventris cumatilis LeConte, 1851
Cicindela (Cicindelidia) rufiventris hentzii Dejean, 1831
Cicindela (Cicindelidia) rufiventris rufiventris Dejean, 1825
Cicindela (Cicindelidia) scabrosa Schaupp, 1884
Cicindela (Cicindelidia) schauppii Horn, 1876
Cicindela (Cicindelidia) sedecimpunctata sedecimpunctata Klug, 1834
Cicindela (Cicindelidia) senilis Horn, 1867
Cicindela (Cicindelidia) tenuisignata LeConte, 1851
Cicindela (Cicindelidia) trifasciata ascendens LeConte, 1851
Cicindela (Cicindelidia) trifasciata sigmoidea LeConte, 1851
Cicindela (Cicindelidia) willistoni echo Casey, 1897
Cicindela (Cicindelidia) willistoni estancia Rumpp, 1962
Cicindela (Cicindelidia) willistoni funaroi Rotger, 1972
Cicindela (Cicindelidia) willistoni hirtifrons Willis, 1967
Cicindela (Cicindelidia) willistoni praedicta Rumpp, 1956
Cicindela (Cicindelidia) willistoni pseudosenilis Horn, 1900
Cicindela (Cicindelidia) willistoni sulfontis Rumpp, 1977
Cicindela (Cicindelidia) willistoni willistoni LeConte, 1879
Cicindela (Cicindela) albissima Rumpp, 1962
Cicindela (Cicindela) ancocisconensis Harris, 1852
Cicindela (Cicindela) arenicola Rumpp, 1967
Cicindela (Cicindela) arida Davis, 1928
Cicindela (Cicindela) bellissima bellissima Leng, 1902
Cicindela (Cicindela) bellissima frechini Leffler, 1979
Cicindela (Cicindela) columbica Hatch, 1938
Cicindela (Cicindela) decemnotata bonnevillensis Knisley & Kippenhan, 2012
Cicindela (Cicindela) decemnotata decemnotata Say, 1817
Cicindela (Cicindela) decemnotata meriwetheri Knisley & Kippenhan, 2012
Cicindela (Cicindela) decemnotata montevolans Knisley & Kippenhan, 2012
Cicindela (Cicindela) denikei Brown, 1934
Cicindela (Cicindela) denverensis Casey, 1897
Cicindela (Cicindela) depressula depressula Casey, 1897
Cicindela (Cicindela) depressula eureka Fall, 1901
Cicindela (Cicindela) duodecimguttata Dejean, 1825
Cicindela (Cicindela) formosa formosa Say, 1817
Cicindela (Cicindela) formosa generosa Dejean, 1831
Cicindela (Cicindela) formosa gibsoni Brown, 1940
Cicindela (Cicindela) formosa pigmentosignata Horn, 1930
Cicindela (Cicindela) formosa rutilovirescens Rumpp, 1986
Cicindela (Cicindela) fulgida fulgida Say, 1823
Cicindela (Cicindela) fulgida pseudowillistoni Horn, 1938
Cicindela (Cicindela) fulgida westbournei Calder, 1922
Cicindela (Cicindela) hirticollis abrupta Casey, 1913
Cicindela (Cicindela) hirticollis athabascensis Graves, 1988
Cicindela (Cicindela) hirticollis coloradula Graves, 1988
Cicindela (Cicindela) hirticollis corpuscula Rumpp, 1962
Cicindela (Cicindela) hirticollis couleensis Graves, 1988
Cicindela (Cicindela) hirticollis gravida LeConte, 1851
Cicindela (Cicindela) hirticollis hirticollis Say, 1817
Cicindela (Cicindela) hirticollis rhodensis Calder, 1916
Cicindela (Cicindela) hirticollis shelfordi Graves, 1988
Cicindela (Cicindela) hirticollis siuslawensis Graves, 1988
Cicindela (Cicindela) latesignata latesignata LeConte, 1851
Cicindela (Cicindela) lengi jordai Rotger, 1974
Cicindela (Cicindela) lengi lengi Horn, 1908
Cicindela (Cicindela) lengi versuta Casey, 1913
Cicindela (Cicindela) limbalis Klug, 1834
Cicindela (Cicindela) limbata hyperborea LeConte, 1863
Cicindela (Cicindela) limbata labradorensis Johnson, 1991
Cicindela (Cicindela) limbata limbata Say, 1823
Cicindela (Cicindela) limbata nogahabarensis Knisley, 2008
Cicindela (Cicindela) limbata nympha Casey, 1913
Cicindela (Cicindela) longilabris laurentii Schaupp, 1884
Cicindela (Cicindela) longilabris longilabris Say, 1824
Cicindela (Cicindela) longilabris perviridis Schaupp, 1884
Cicindela (Cicindela) nebraskana Casey, 1909
Cicindela (Cicindela) nigrior Schaupp, 1884
Cicindela (Cicindela) ohlone Freitag & Kavanaugh, 1993
Cicindela (Cicindela) oregona guttifera LeConte, 1856
Cicindela (Cicindela) oregona maricopa Leng, 1902
Cicindela (Cicindela) oregona navajoensis Van Dyke, 1947
Cicindela (Cicindela) oregona oregona LeConte, 1856
Cicindela (Cicindela) parowana parowana Wickham, 1905
Cicindela (Cicindela) parowana platti Cazier, 1937
Cicindela (Cicindela) parowana wallisi Calder, 1922
Cicindela (Cicindela) patruela consentanea Dejean, 1825
Cicindela (Cicindela) patruela patruela Dejean, 1825
Cicindela (Cicindela) pimeriana LeConte, 1867
Cicindela (Cicindela) plutonica Casey, 1897
Cicindela (Cicindela) pugetana Casey, 1914
Cicindela (Cicindela) pulchra dorothea Rumpp, 1977
Cicindela (Cicindela) pulchra pulchra Say, 1823
Cicindela (Cicindela) purpurea audubonii LeConte, 1845
Cicindela (Cicindela) purpurea cimarrona LeConte, 1868
Cicindela (Cicindela) purpurea hatchi Leffler, 1980
Cicindela (Cicindela) purpurea lauta Casey, 1897
Cicindela (Cicindela) purpurea purpurea Olivier, 1790
Cicindela (Cicindela) repanda novascotiae Vaurie, 1951
Cicindela (Cicindela) repanda repanda Dejean, 1825
Cicindela (Cicindela) repanda tanneri Knaus, 1929
Cicindela (Cicindela) scutellaris flavoviridis Vaurie, 1950
Cicindela (Cicindela) scutellaris lecontei Haldeman, 1853
Cicindela (Cicindela) scutellaris rugata Vaurie, 1950
Cicindela (Cicindela) scutellaris rugifrons Dejean, 1825
Cicindela (Cicindela) scutellaris scutellaris Say, 1823
Cicindela (Cicindela) scutellaris unicolor Dejean, 1825
Cicindela (Cicindela) scutellaris yampae Rumpp, 1986
Cicindela (Cicindela) sexguttata Fabricius, 1775
Cicindela (Cicindela) splendida Hentz, 1830
Cicindela (Cicindela) tenuicincta Schaupp, 1884
Cicindela (Cicindela) theatina Rotger, 1944
Cicindela (Cicindela) tranquebarica cibecuei Duncan, 1958
Cicindela (Cicindela) tranquebarica diffracta Casey, 1909
Cicindela (Cicindela) tranquebarica joaquinensis Knisley & Haines, 2007
Cicindela (Cicindela) tranquebarica kirbyi LeConte, 1867
Cicindela (Cicindela) tranquebarica parallelonota Casey, 1914
Cicindela (Cicindela) tranquebarica sierra Leng, 1902
Cicindela (Cicindela) tranquebarica tranquebarica Herbst, 1806
Cicindela (Cicindela) tranquebarica vibex Horn, 1867
Cicindela (Cicindela) tranquebarica viridissima Fall, 1910
Cicindela (Cicindela) waynei Leffler, 2001
LoriceriniLoricera (Loricera) decempunctata Eschscholtz, 1833
Loricera (Loricera) foveata LeConte, 1851
Loricera (Loricera) pilicornis congesta Mannerheim, 1853 ‡
Loricera (Loricera) pilicornis pilicornis (Fabricius, 1775)‡
ElaphriniDiacheila arctica amoena (Faldermann, 1835)‡
Diacheila polita (Faldermann, 1835)‡
Blethisa catenaria Brown, 1944‡
Blethisa hudsonica Casey, 1924
Blethisa julii LeConte, 1863
Blethisa multipunctata aurata Fischer von Waldheim, 1828‡
Blethisa oregonensis LeConte, 1853
Blethisa quadricollis Haldeman, 1847
Elaphrus (Arctelaphrus) lapponicus lapponicus Gyllenhal, 1810‡
Elaphrus (Arctelaphrus) lapponicus obliteratus Mannerheim, 1853
Elaphrus (Neoelaphrus) cicatricosus LeConte, 1847
Elaphrus (Neoelaphrus) clairvillei Kirby, 1837
Elaphrus (Neoelaphrus) fuliginosus Say, 1830
Elaphrus (Neoelaphrus) laevigatus LeConte, 1852
Elaphrus (Neoelaphrus) lindrothi Goulet, 1983
Elaphrus (Neoelaphrus) olivaceus LeConte, 1863
Elaphrus (Elaphrus) americanus americanus Dejean, 1831
Elaphrus (Elaphrus) americanus sylvanus Goulet, 1982
Elaphrus (Elaphrus) californicus Mannerheim, 1843
Elaphrus (Elaphrus) finitimus Casey, 1920
Elaphrus (Elaphrus) lecontei Crotch, 1876
Elaphrus (Elaphrus) marginicollis Goulet, 1983
Elaphrus (Elaphrus) mimus Goulet, 1983
Elaphrus (Elaphrus) ruscarius Say, 1830
Elaphrus (Elaphrus) trossulus Semenov, 1904‡
Elaphrus (Elaphrus) tuberculatus Mäklin, 1878‡
Elaphrus (Elaphrus) viridis Horn, 1878
Elaphrus (Elaphroterus) angusticollis angusticollis Sahlberg, 1844‡
Elaphrus (Elaphroterus) purpurans Hausen, 1891
OmophroniniOmophron (Omophron) americanum Dejean, 1831
Omophron (Omophron) dentatum LeConte, 1852
Omophron (Omophron) gilae LeConte, 1852
Omophron (Omophron) grossum Casey, 1909
Omophron (Omophron) labiatum (Fabricius, 1801)
Omophron (Omophron) nitidum LeConte, 1847
Omophron (Omophron) obliteratum Horn, 1870
Omophron (Omophron) ovale Horn, 1870
Omophron (Omophron) robustum Horn, 1870
Omophron (Omophron) solidum Casey, 1897
Omophron (Omophron) tessellatum Say, 1823
PasimachiniPasimachus (Pasimachus) californicus Chaudoir, 1850
Pasimachus (Pasimachus) depressus (Fabricius, 1787)
Pasimachus (Pasimachus) duplicatus LeConte, 1853
Pasimachus (Pasimachus) elongatus LeConte, 1846
Pasimachus (Pasimachus) marginatus (Fabricius, 1787)
Pasimachus (Pasimachus) obsoletus LeConte, 1846
Pasimachus (Pasimachus) punctulatus Haldeman, 1843
Pasimachus (Pasimachus) strenuus LeConte, 1874
Pasimachus (Pasimachus) sublaevis (Palisot de Beauvois, 1811)
Pasimachus (Pasimachus) subsulcatus Say, 1823
Pasimachus (Pasimachus) viridans LeConte, 1858
ScaritiniScarites (Scarites) lissopterus Chaudoir, 1881
Scarites (Scarites) marinus Nichols, 1986
Scarites (Scarites) ocalensis Nichols, 1986
Scarites (Scarites) quadriceps Chaudoir, 1843
Scarites (Scarites) stenops Bousquet & Skelley, 2010
Scarites (Scarites) subterraneus Fabricius, 1775
Scarites (Scarites) vicinus Chaudoir, 1843
ClivininiClivina (Semiclivina) dentipes Dejean, 1825
Clivina (Semiclivina) vespertina Putzeys, 1867†
Clivina (Clivina) choatei Bousquet & Skelley, 2012
Clivina (Clivina) collaris (Herbst, 1784)†
Clivina (Clivina) fossor fossor (Linnaeus, 1758)†
Clivina (Clivina) impressefrons LeConte, 1844
Clivina (Clivina) myops Bousquet, 1997
Clivina (Clivina) oregona Fall, 1922
Clivina (Clivina) pallida Say, 1823
Clivina (Clivina) planicollis LeConte, 1857
Clivina (Clivina) punctigera LeConte, 1857
Clivina (Clivina) punctulata LeConte, 1852
Clivina (Antroforceps) alabama Bousquet, 2012
Clivina (Antroforceps) rubicunda LeConte, 1857
Clivina (Antroforceps) sasajii Ball, 2001
Clivina (Leucocara) acuducta Haldeman, 1843
Clivina (Leucocara) americana Dejean, 1831
Clivina (Leucocara) californica Van Dyke, 1925
Clivina (Leucocara) morio Dejean, 1831
Clivina (Leucocara) rufa LeConte, 1857
Paraclivina bipustulata (Fabricius, 1798)
Paraclivina convexa (LeConte, 1844)
Paraclivina fasciata (Putzeys, 1846)
Paraclivina ferrea (LeConte, 1857)
Paraclivina marginipennis (Putzeys, 1846)
Paraclivina postica (LeConte, 1846)
Paraclivina stigmula (Putzeys, 1846)
Paraclivina striatopunctata (Dejean, 1831)
Paraclivina sulcipennis (Putzeys, 1867)
Schizogenius (Genioschizus) crenulatus crenulatus LeConte, 1852
Schizogenius (Schizogenius) amphibius (Haldeman, 1843)
Schizogenius (Schizogenius) auripennis Bates, 1881
Schizogenius (Schizogenius) brevisetosus Whitehead, 1972
Schizogenius (Schizogenius) chiricahuanus Whitehead, 1972
Schizogenius (Schizogenius) depressus LeConte, 1852
Schizogenius (Schizogenius) falli Whitehead, 1972
Schizogenius (Schizogenius) ferrugineus Putzeys, 1846
Schizogenius (Schizogenius) lindrothi Whitehead, 1972
Schizogenius (Schizogenius) lineolatus (Say, 1823)
Schizogenius (Schizogenius) litigiosus Fall, 1901
Schizogenius (Schizogenius) longipennis Putzeys, 1867
Schizogenius (Schizogenius) neovalidus Whitehead, 1972
Schizogenius (Schizogenius) ochthocephalus Whitehead, 1972
Schizogenius (Schizogenius) ozarkensis Whitehead, 1972
Schizogenius (Schizogenius) planulatus LeConte, 1863
Schizogenius (Schizogenius) planuloides Whitehead, 1972
Schizogenius (Schizogenius) pluripunctatus LeConte, 1852
Schizogenius (Schizogenius) pygmaeus Van Dyke, 1925
Schizogenius (Schizogenius) sallei Putzeys, 1867
Schizogenius (Schizogenius) scopaeus Whitehead, 1972
Schizogenius (Schizogenius) seticollis seticollis Fall, 1901
Schizogenius (Schizogenius) sulcifrons Putzeys, 1846
Schizogenius (Schizogenius) tibialis Whitehead, 1972
Halocoryza arenaria (Darlington, 1939)
Oxydrepanus rufus (Putzeys, 1846)
Ardistomis obliquata Putzeys, 1846
Ardistomis schaumii LeConte, 1857
Semiardistomis puncticollis (Dejean, 1831)
Semiardistomis viridis (Say, 1823)
Aspidoglossa subangulata (Chaudoir, 1843)
DyschiriiniAkephorus marinus LeConte, 1852
Akephorus obesus (LeConte, 1863)
Dyschirius abbreviatus Putzeys, 1846
Dyschirius aeneolus LeConte, 1850
Dyschirius affinis Fall, 1901
Dyschirius alticola Lindroth, 1961
Dyschirius analis LeConte, 1852
Dyschirius aratus LeConte, 1852
Dyschirius brevispinus LeConte, 1878
Dyschirius campicola Lindroth, 1961
Dyschirius carrorum Bousquet, 1997
Dyschirius cerberus Larson, 1968
Dyschirius chiricahuae (Dajoz, 2004)
Dyschirius comatus Bousquet, 1988
Dyschirius compactus Lindroth, 1961
Dyschirius consobrinus LeConte, 1852
Dyschirius criddlei Fall, 1925
Dyschirius curvispinus Putzeys, 1846
Dyschirius dejeanii Putzeys, 1846
Dyschirius edentulus Putzeys, 1846
Dyschirius erythrocerus LeConte, 1857
Dyschirius exochus Whitehead, 1970
Dyschirius ferrugineus Bousquet, 1988
Dyschirius gibbipennis LeConte, 1857
Dyschirius globosus (Herbst, 1784)†
Dyschirius globulosus (Say, 1823)
Dyschirius haemorrhoidalis (Dejean, 1831)
Dyschirius hiemalis Bousquet, 1987
Dyschirius interior Fall, 1922
Dyschirius laevifasciatus Horn, 1878
Dyschirius larochellei Bousquet, 1988
Dyschirius longulus LeConte, 1850
Dyschirius melancholicus Putzeys, 1867‡
Dyschirius montanus LeConte, 1879
Dyschirius owen (Dajoz, 2004)
Dyschirius pacificus Lindroth, 1961
Dyschirius pallipennis (Say, 1823)
Dyschirius patruelis LeConte, 1852
Dyschirius perversus Fall, 1922
Dyschirius pilosus LeConte, 1857
Dyschirius planatus Lindroth, 1961
Dyschirius politus politus (Dejean, 1825)‡
Dyschirius pumilus (Dejean, 1825)
Dyschirius quadrimaculatus Lindroth, 1961
Dyschirius salivagans LeConte, 1875
Dyschirius sculptus Bousquet, 1988
Dyschirius sellatus LeConte, 1857
Dyschirius setosus LeConte, 1857
Dyschirius sextoni Bousquet, 1987
Dyschirius soda (Dajoz, 2004)
Dyschirius sphaericollis (Say, 1823)
Dyschirius subarcticus subarcticus Lindroth, 1961
Dyschirius sublaevis Putzeys, 1846
Dyschirius tenuispinus Lindroth, 1961
Dyschirius terminatus LeConte, 1846
Dyschirius timidus Lindroth, 1961
Dyschirius tridentatus LeConte, 1852
Dyschirius truncatus LeConte, 1857
Dyschirius unipunctatus Fall, 1901
Dyschirius varidens Fall, 1910
Dyschirius wayah (Dajoz, 2005)
PromecognathiniPromecognathus crassus LeConte, 1868
Promecognathus laevissimus (Dejean, 1829)
BrosciniMiscodera arctica (Paykull, 1798)‡
Broscodera (Broscodera) insignis (Mannerheim, 1852)
Zacotus matthewsii LeConte, 1869
Broscus cephalotes (Linnaeus, 1758)†
GehringiiniGehringia olympica Darlington, 1933
TrechiniTrechoblemus westcotti Barr, 1972
Pseudanophthalmus abditus Krekeler, 1973
Pseudanophthalmus acherontis Barr, 1959
Pseudanophthalmus alabamae Valentine, 1932
Pseudanophthalmus aladdini Valentine, 1945
Pseudanophthalmus assimilis Barr, 1981
Pseudanophthalmus audax (Horn, 1883)
Pseudanophthalmus avernus Valentine, 1945
Pseudanophthalmus barberi Jeannel, 1928
Pseudanophthalmus barri Krekeler, 1973
Pseudanophthalmus beakleyi Valentine, 1937
Pseudanophthalmus bendermani Barr, 1959
Pseudanophthalmus caecus Krekeler, 1973
Pseudanophthalmus calcareus Barr, 1981
Pseudanophthalmus catherinae Barr, 1959
Pseudanophthalmus catoryctos Krekeler, 1973
Pseudanophthalmus cerberus cerberus Barr, 1985
Pseudanophthalmus cerberus completus Barr, 1985
Pseudanophthalmus chthonius Krekeler, 1973
Pseudanophthalmus ciliaris ciliaris Valentine, 1937
Pseudanophthalmus ciliaris orlindae Barr, 1959
Pseudanophthalmus cnephosus Krekeler, 1973
Pseudanophthalmus colemanensis Barr, 1959
Pseudanophthalmus conditus Krekeler, 1973
Pseudanophthalmus cordicollis Barr, 1981
Pseudanophthalmus cumberlandus Valentine, 1937
Pseudanophthalmus darlingtoni darlingtoni Barr, 1985
Pseudanophthalmus darlingtoni persimilis Barr, 1985
Pseudanophthalmus deceptivus Barr, 1981
Pseudanophthalmus delicatus Valentine, 1932
Pseudanophthalmus desertus Krekeler, 1973
Pseudanophthalmus digitus Valentine, 1932
Pseudanophthalmus distinguens Valentine, 1948
Pseudanophthalmus egberti Barr, 1965
Pseudanophthalmus elongatus Krekeler, 1973
Pseudanophthalmus emersoni Krekeler, 1958
Pseudanophthalmus engelhardti (Barber, 1928)
Pseudanophthalmus eremita (Horn, 1871)
Pseudanophthalmus exiguus Krekeler, 1973
Pseudanophthalmus exoticus Krekeler, 1973
Pseudanophthalmus farrelli Barr, 1959
Pseudanophthalmus fastigatus Barr, 1981
Pseudanophthalmus fluviatilis Valentine, 1948
Pseudanophthalmus fowlerae Barr, 1980
Pseudanophthalmus frigidus Barr, 1981
Pseudanophthalmus fulleri Valentine, 1932
Pseudanophthalmus fuscus constrictus Valentine, 1932
Pseudanophthalmus fuscus fuscus Valentine, 1931
Pseudanophthalmus georgiae Barr, 1981
Pseudanophthalmus globiceps Barr, 1985
Pseudanophthalmus gracilis Valentine, 1931
Pseudanophthalmus grandis elevatus Valentine, 1932
Pseudanophthalmus grandis grandis Valentine, 1931
Pseudanophthalmus hadenoecus Barr, 1965
Pseudanophthalmus henroti Jeannel, 1949
Pseudanophthalmus hesperus Barr, 1959
Pseudanophthalmus higginbothami Valentine, 1931
Pseudanophthalmus hirsutus Valentine, 1931
Pseudanophthalmus hoffmani Barr, 1965
Pseudanophthalmus holsingeri Barr, 1965
Pseudanophthalmus horni (Garman, 1892)
Pseudanophthalmus hortulanus Barr, 1965
Pseudanophthalmus hubbardi (Barber, 1928)
Pseudanophthalmus hubrichti Valentine, 1948
Pseudanophthalmus humeralis Valentine, 1931
Pseudanophthalmus hypertrichosis Valentine, 1932
Pseudanophthalmus hypolithos Barr, 1981
Pseudanophthalmus illinoisensis Barr & Peck, 1966
Pseudanophthalmus inexpectatus Barr, 1959
Pseudanophthalmus inquisitor Barr, 1980
Pseudanophthalmus insularis Barr, 1959
Pseudanophthalmus intermedius (Valentine, 1931)
Pseudanophthalmus intersectus Barr, 1965
Pseudanophthalmus jonesi Valentine, 1945
Pseudanophthalmus krameri Krekeler, 1973
Pseudanophthalmus krekeleri Barr, 1965
Pseudanophthalmus lallemanti Jeannel, 1949
Pseudanophthalmus leonae Barr, 1960
Pseudanophthalmus limicola Jeannel, 1931
Pseudanophthalmus lodingi Valentine, 1931
Pseudanophthalmus loganensis Barr, 1959
Pseudanophthalmus longiceps Barr, 1981
Pseudanophthalmus macradyi Valentine, 1948
Pseudanophthalmus major Krekeler, 1973
Pseudanophthalmus menetriesii campestris Barr, 1985
Pseudanophthalmus menetriesii menetriesii (Motschulsky, 1862)
Pseudanophthalmus meridionalis Valentine, 1945
Pseudanophthalmus montanus Barr, 1965
Pseudanophthalmus nelsoni Barr, 1965
Pseudanophthalmus nickajackensis Barr, 1981
Pseudanophthalmus nortoni Barr, 1981
Pseudanophthalmus occidentalis Barr, 1959
Pseudanophthalmus ohioensis Krekeler, 1973
Pseudanophthalmus orientalis Krekeler, 1973
Pseudanophthalmus orthosulcatus Valentine, 1932
Pseudanophthalmus packardi Barr, 1959
Pseudanophthalmus pallidus Barr, 1981
Pseudanophthalmus paradoxus Barr, 1981
Pseudanophthalmus parvicollis Jeannel, 1931
Pseudanophthalmus parvus Krekeler, 1973
Pseudanophthalmus paulus Barr, 1981
Pseudanophthalmus paynei Barr, 1981
Pseudanophthalmus petrunkevitchi Valentine, 1945
Pseudanophthalmus pholeter Krekeler, 1973
Pseudanophthalmus pilosus Barr, 1985
Pseudanophthalmus pontis Barr, 1965
Pseudanophthalmus potomaca Valentine, 1932
Pseudanophthalmus praetermissus Barr, 1981
Pseudanophthalmus princeps Barr, 1979
Pseudanophthalmus productus Barr, 1980
Pseudanophthalmus profundus Valentine, 1945
Pseudanophthalmus pubescens intrepidus Barr, 1985
Pseudanophthalmus pubescens pubescens (Horn, 1869)
Pseudanophthalmus punctatus Valentine, 1931
Pseudanophthalmus pusillus Barr, 1981
Pseudanophthalmus pusio (Horn, 1869)
Pseudanophthalmus puteanus Krekeler, 1973
Pseudanophthalmus quadratus Barr, 1965
Pseudanophthalmus rittmani Krekeler, 1973
Pseudanophthalmus robustus Valentine, 1931
Pseudanophthalmus rogersae Barr, 1981
Pseudanophthalmus rotundatus Valentine, 1932
Pseudanophthalmus sanctipauli Barr, 1981
Pseudanophthalmus scholasticus Barr, 1981
Pseudanophthalmus scutilus Barr, 1981
Pseudanophthalmus seclusus Barr, 1981
Pseudanophthalmus senecae Valentine, 1932
Pseudanophthalmus sequoyah Barr, 1981
Pseudanophthalmus sericus Barr, 1981
Pseudanophthalmus shilohensis mayfieldensis Krekeler, 1958
Pseudanophthalmus shilohensis shilohensis Krekeler, 1958
Pseudanophthalmus sidus Barr, 1965
Pseudanophthalmus simplex Barr, 1980
Pseudanophthalmus simulans Barr, 1985
Pseudanophthalmus solivagus Krekeler, 1973
Pseudanophthalmus steevesi Barr, 1981
Pseudanophthalmus striatus (Motschulsky, 1862)
Pseudanophthalmus stricticollis Jeannel, 1931
Pseudanophthalmus sylvaticus Barr, 1967
Pseudanophthalmus templetoni Valentine, 1948
Pseudanophthalmus tenebrosus Krekeler, 1973
Pseudanophthalmus tennesseensis Valentine, 1937
Pseudanophthalmus tenuis (Horn, 1871)
Pseudanophthalmus thomasi Barr, 1981
Pseudanophthalmus tiresias Barr, 1959
Pseudanophthalmus transfluvialis Barr, 1985
Pseudanophthalmus troglodytes Krekeler, 1973
Pseudanophthalmus tullahoma Barr, 1959
Pseudanophthalmus umbratilis Krekeler, 1973
Pseudanophthalmus unionis Barr, 1981
Pseudanophthalmus valentinei Jeannel, 1949
Pseudanophthalmus vanburenensis Barr, 1959
Pseudanophthalmus ventus Barr, 1981
Pseudanophthalmus vicarius Barr, 1965
Pseudanophthalmus virginicus (Barr, 1960)
Pseudanophthalmus wallacei Barr, 1981
Pseudanophthalmus youngi Krekeler, 1958
Nelsonites jonesei Valentine, 1952
Nelsonites walteri Valentine, 1952
Neaphaenops tellkampfii henroti Jeannel, 1949
Neaphaenops tellkampfii meridionalis Barr, 1959
Neaphaenops tellkampfii tellkampfii (Erichson, 1844)
Neaphaenops tellkampfii viator Barr, 1979
Blemus discus discus (Fabricius, 1792)†
Xenotrechus condei Barr & Krekeler, 1967
Xenotrechus denticollis Barr & Krekeler, 1967
Darlingtonea kentuckensis Valentine, 1952
Ameroduvalius jeanneli jeanneli Valentine, 1952
Ameroduvalius jeanneli rockcastlei Valentine, 1952
Trechus (Trechus) alinae Dajoz, 1990
Trechus (Trechus) apache Dajoz, 1990
Trechus (Trechus) apicalis Motschulsky, 1845‡
Trechus (Trechus) arizonae Casey, 1918
Trechus (Trechus) caliginis Barr, 1985
Trechus (Trechus) carolinae Schaeffer, 1901
Trechus (Trechus) chalybeus Dejean, 1831
Trechus (Trechus) coloradensis Schaeffer, 1915
Trechus (Trechus) conformis Jeannel, 1927
Trechus (Trechus) crassiscapus Lindroth, 1955
Trechus (Trechus) cumberlandus Barr, 1962
Trechus (Trechus) humboldti Van Dyke, 1945
Trechus (Trechus) hydropicus avus Barr, 1962
Trechus (Trechus) hydropicus beutenmuelleri Jeannel, 1931
Trechus (Trechus) hydropicus canus Barr, 1962
Trechus (Trechus) hydropicus hydropicus Horn, 1883
Trechus (Trechus) mitchellensis Barr, 1962
Trechus (Trechus) obtusus Erichson, 1837†
Trechus (Trechus) oregonensis Hatch, 1951
Trechus (Trechus) ovipennis Motschulsky, 1845
Trechus (Trechus) pomonae Fall, 1901
Trechus (Trechus) quadristriatus (Schrank, 1781)†
Trechus (Trechus) roanicus Barr, 1962
Trechus (Trechus) rubens (Fabricius, 1792)†
Trechus (Trechus) schwarzi saludae Barr, 1979
Trechus (Trechus) schwarzi schwarzi Jeannel, 1931
Trechus (Trechus) schwarzi scopulosus Barr, 1979
Trechus (Trechus) tenuiscapus Lindroth, 1961
Trechus (Trechus) yvesbousqueti Donabauer, 2010
Trechus (Microtrechus) aduncus Barr, 1962
Trechus (Microtrechus) balsamensis Barr, 1962
Trechus (Microtrechus) barberi (Jeannel, 1931)
Trechus (Microtrechus) bowlingi Barr, 1962
Trechus (Microtrechus) cheoahensis Donabauer, 2005
Trechus (Microtrechus) clingmanensis Donabauer, 2005
Trechus (Microtrechus) coweensis Barr, 1979
Trechus (Microtrechus) haoe Barr, 1979
Trechus (Microtrechus) haoeleadensis Donabauer, 2005
Trechus (Microtrechus) howellae Barr, 1979
Trechus (Microtrechus) inexpectatus Barr, 1985
Trechus (Microtrechus) luculentus cheoahbaldensis Donabauer, 2005
Trechus (Microtrechus) luculentus joannabaldensis Donabauer, 2005
Trechus (Microtrechus) luculentus luculentus Barr, 1962
Trechus (Microtrechus) luculentus wayahensis Barr, 1979
Trechus (Microtrechus) nantahalae Barr, 1979
Trechus (Microtrechus) nebulosus Barr, 1962
Trechus (Microtrechus) novaculosus Barr, 1962
Trechus (Microtrechus) plottbalsamensis Donabauer, 2005
Trechus (Microtrechus) pseudobarberi Donabauer, 2009
Trechus (Microtrechus) pseudonovaculosus Donabauer, 2005
Trechus (Microtrechus) pseudosubtilis Donabauer, 2009
Trechus (Microtrechus) ramseyensis Donabauer, 2005
Trechus (Microtrechus) rivulis Dajoz, 2005
Trechus (Microtrechus) rosenbergi Barr, 1962
Trechus (Microtrechus) satanicus Barr, 1962
Trechus (Microtrechus) snowbirdensis Donabauer, 2005
Trechus (Microtrechus) stefanschoedli Donabauer, 2005
Trechus (Microtrechus) stupkai Barr, 1979
Trechus (Microtrechus) subtilis Barr, 1962
Trechus (Microtrechus) talequah Barr, 1962
Trechus (Microtrechus) tennesseensis tauricus Barr, 1962
Trechus (Microtrechus) tennesseensis tennesseensis Barr, 1962
Trechus (Microtrechus) thomasbarri Donabauer, 2005
Trechus (Microtrechus) thunderheadensis Donabauer, 2005
Trechus (Microtrechus) tobiasi Donabauer, 2005
Trechus (Microtrechus) tonitru Barr, 1962
Trechus (Microtrechus) toxawayi Barr, 1979
Trechus (Microtrechus) tuckaleechee Barr, 1962
Trechus (Microtrechus) tusquitee Barr, 1979
Trechus (Microtrechus) tusquitensis Donabauer, 2005
Trechus (Microtrechus) uncifer Barr, 1962
Trechus (Microtrechus) unicoi Barr, 1979
Trechus (Microtrechus) valentinei Barr, 1979
Trechus (Microtrechus) vandykei pisgahensis Barr, 1979
Trechus (Microtrechus) vandykei vandykei (Jeannel, 1927)
Trechus (Microtrechus) verus Barr, 1962
Trechus (Microtrechus) wayahbaldensis Donabauer, 2005
BembidiiniAmerizus (Amerizus) oblonguloides (Lindroth, 1963)
Amerizus (Amerizus) oblongulus (Mannerheim, 1852)
Amerizus (Amerizus) spectabilis (Mannerheim, 1852)
Amerizus (Amerizus) utahensis (Van Dyke, 1926)
Amerizus (Amerizus) wingatei (Bland, 1864)
Lionepha casta (Casey, 1918)
Lionepha chintimini (Erwin & Kavanaugh, 1981)
Lionepha disjuncta (Lindroth, 1963)
Lionepha erasa (LeConte, 1859)
Lionepha lindrothellus (Erwin & Kavanaugh, 1981)
Lionepha lummi (Erwin & Kavanaugh, 1981)
Lionepha osculans (Casey, 1918)
Lionepha pseudoerasa (Lindroth, 1963)
Lionepha sequoiae (Lindroth, 1963)
Asaphidion alaskanum Wickham, 1919
Asaphidion curtum curtum (Heyden, 1870)†
Asaphidion yukonense Wickham, 1919
Bembidion (Hirmoplataphus) alpineanum Casey, 1924
Bembidion (Hirmoplataphus) avidum Casey, 1918
Bembidion (Hirmoplataphus) concolor (Kirby, 1837)
Bembidion (Hirmoplataphus) humboldtense Blaisdell, 1902
Bembidion (Hirmoplataphus) nigrum Say, 1823
Bembidion (Hirmoplataphus) quadrulum LeConte, 1861
Bembidion (Hirmoplataphus) recticolle LeConte, 1863
Bembidion (Hirmoplataphus) salebratum (LeConte, 1847)
Bembidion (Hirmoplataphus) subaerarium Casey, 1924
Bembidion (Hydriomicrus) brevistriatum Hayward, 1897
Bembidion (Hydriomicrus) californicum Hayward, 1897
Bembidion (Hydriomicrus) innocuum Casey, 1918
Bembidion (Hydriomicrus) quadratulum Notman, 1920
Bembidion (Hydriomicrus) semistriatum (Haldeman, 1843)
Bembidion (Odontium) aenulum Hayward, 1901
Bembidion (Odontium) bowditchii LeConte, 1878
Bembidion (Odontium) carinatum (LeConte, 1852)
Bembidion (Odontium) confusum Hayward, 1897
Bembidion (Odontium) coxendix Say, 1823
Bembidion (Odontium) durangoense Bates, 1891
Bembidion (Odontium) gilae Lindroth, 1963
Bembidion (Odontium) paraenulum Maddison, 2009
Bembidion (Odontium) robusticolle Hayward, 1897
Bembidion (Odontium) sculpturatum (Motschulsky, 1859)
Bembidion (Bracteon) alaskense Lindroth, 1962‡
Bembidion (Bracteon) balli Lindroth, 1962
Bembidion (Bracteon) carinula Chaudoir, 1868
Bembidion (Bracteon) foveum Motschulsky, 1844‡
Bembidion (Bracteon) hesperium Fall, 1910
Bembidion (Bracteon) inaequale Say, 1823
Bembidion (Bracteon) lapponicum Zetterstedt, 1828‡
Bembidion (Bracteon) levettei carrianum Casey, 1924
Bembidion (Bracteon) levettei levettei Casey, 1918
Bembidion (Bracteon) lorquinii Chaudoir, 1868
Bembidion (Bracteon) punctatostriatum Say, 1823
Bembidion (Bracteon) zephyrum Fall, 1910
Bembidion (Ochthedromus) americanum Dejean, 1831
Bembidion (Ochthedromus) bifossulatum (LeConte, 1852)
Bembidion (Ochthedromus) cheyennense Casey, 1918
Bembidion (Pseudoperyphus) antiquum Dejean, 1831
Bembidion (Pseudoperyphus) arenobile Maddison, 2008
Bembidion (Pseudoperyphus) bellorum Maddison, 2008
Bembidion (Pseudoperyphus) chalceum Dejean, 1831
Bembidion (Pseudoperyphus) honestum Say, 1823
Bembidion (Pseudoperyphus) integrum Casey, 1918
Bembidion (Pseudoperyphus) louisella Maddison, 2008
Bembidion (Pseudoperyphus) rothfelsi Maddison, 2008
Bembidion (Pseudoperyphus) rufotinctum Chaudoir, 1868
Bembidion (Cillenus) palosverdes Kavanaugh & Erwin, 1992
Bembidion (Actedium) lachnophoroides Darlington, 1926
Bembidion (Ocydromus) scopulinum (Kirby, 1837)‡
Bembidion (Peryphus) actuosum Casey, 1918
Bembidion (Peryphus) bruxellense Wesmael, 1835†
Bembidion (Peryphus) consanguineum Hayward, 1897
Bembidion (Peryphus) dauricum (Motschulsky, 1844)‡
Bembidion (Peryphus) femoratum femoratum Sturm, 1825†
Bembidion (Peryphus) lugubre LeConte, 1857
Bembidion (Peryphus) mexicanum Dejean, 1831
Bembidion (Peryphus) nevadense Ulke, 1875
Bembidion (Peryphus) obscurellum obscurellum (Motschulsky, 1845)‡
Bembidion (Peryphus) pernotum Casey, 1918
Bembidion (Peryphus) perspicuum (LeConte, 1848)
Bembidion (Peryphus) petrosum attuense Lindroth, 1963
Bembidion (Peryphus) petrosum petrosum Gebler, 1833‡
Bembidion (Peryphus) plagiatum (Zimmermann, 1869)
Bembidion (Peryphus) poppii schalleri Lindroth, 1963
Bembidion (Peryphus) rupicola (Kirby, 1837)
Bembidion (Peryphus) sarpedon Casey, 1918
Bembidion (Peryphus) satelles Casey, 1918
Bembidion (Peryphus) sejunctum sejunctum Casey, 1918
Bembidion (Peryphus) sejunctum semiaureum Fall, 1922
Bembidion (Peryphus) striola (LeConte, 1852)
Bembidion (Peryphus) tetracolum tetracolum Say, 1823†
Bembidion (Peryphus) transversale Dejean, 1831
Bembidion (Terminophanes) mckinleyi carneum Lindroth, 1963
Bembidion (Terminophanes) mckinleyi mckinleyi Fall, 1926
Bembidion (Asioperyphus) bimaculatum (Kirby, 1837)
Bembidion (Asioperyphus) lenae Csiki, 1928‡
Bembidion (Asioperyphus) postremum Say, 1830
Bembidion (Asioperyphus) renoanum Casey, 1918
Bembidion (Asioperyphus) sordidum (Kirby, 1837)
Bembidion (Asioperyphus) umiatense Lindroth, 1963‡
Bembidion (Peryphanes) grapii Gyllenhal, 1827‡
Bembidion (Peryphanes) lacunarium (Zimmermann, 1869)
Bembidion (Peryphanes) platynoides Hayward, 1897
Bembidion (Peryphanes) stephensii Crotch, 1866†
Bembidion (Peryphanes) subangustatum Hayward, 1897
Bembidion (Peryphanes) texanum Chaudoir, 1868
Bembidion (Peryphanes) yukonum Fall, 1926‡
Bembidion (Testediolum) commotum Casey, 1918
Bembidion (Testediolum) modocianum Casey, 1924
Bembidion (Testediolum) nebraskense LeConte, 1863
Bembidion (Testediolum) obscuripenne Blaisdell, 1902
Bembidion (Testediolum) perbrevicolle Casey, 1924
Bembidion (Testediolum) ulkei Lindroth, 1963
Bembidion (Leuchydrium) tigrinum LeConte, 1879
Bembidion (Bembidion) adductum Casey, 1918
Bembidion (Bembidion) mutatum Gemminger & Harold, 1868
Bembidion (Bembidion) oregonense Hatch, 1953
Bembidion (Bembidion) pedicellatum LeConte, 1857
Bembidion (Bembidion) praecinctum LeConte, 1879
Bembidion (Bembidion) quadrimaculatum dubitans (LeConte, 1852)
Bembidion (Bembidion) quadrimaculatum oppositum Say, 1823
Bembidion (Cyclolopha) jucundum Horn, 1895
Bembidion (Cyclolopha) poculare Bates, 1884
Bembidion (Cyclolopha) sphaeroderum Bates, 1882
Bembidion (Furcacampa) affine Say, 1823
Bembidion (Furcacampa) egens Casey, 1918
Bembidion (Furcacampa) fuchsii Blaisdell, 1902
Bembidion (Furcacampa) impotens Casey, 1918
Bembidion (Furcacampa) mimus Hayward, 1897
Bembidion (Furcacampa) nogalesium Casey, 1924
Bembidion (Furcacampa) timidum (LeConte, 1847)
Bembidion (Furcacampa) triviale Casey, 1918
Bembidion (Furcacampa) versicolor (LeConte, 1847)
Bembidion (Neobembidion) constricticolle Hayward, 1897
Bembidion (Neobembidion) nitidicolle Bousquet, 2006
Bembidion (Neobembidion) nudipenne Lindroth, 1963
Bembidion (Neobembidion) tencenti Hatch, 1951
Bembidion (Diplocampa) transparens transparens (Gebler, 1830)‡
Bembidion (Semicampa) convexulum Hayward, 1897
Bembidion (Semicampa) morulum LeConte, 1863
Bembidion (Semicampa) muscicola Hayward, 1897
Bembidion (Semicampa) nigrivestis Bousquet, 2006
Bembidion (Semicampa) praticola Lindroth, 1963
Bembidion (Semicampa) roosevelti Pic, 1902
Bembidion (Semicampa) rubiginosum LeConte, 1879
Bembidion (Semicampa) semicinctum Notman, 1919
Bembidion (Notaphus) acticola Casey, 1884
Bembidion (Notaphus) aeneicolle (LeConte, 1847)
Bembidion (Notaphus) approximatum (LeConte, 1852)
Bembidion (Notaphus) aratum (LeConte, 1852)
Bembidion (Notaphus) auxiliator Casey, 1924
Bembidion (Notaphus) callens Casey, 1918
Bembidion (Notaphus) castor Lindroth, 1963
Bembidion (Notaphus) coloradense Hayward, 1897
Bembidion (Notaphus) consimile Hayward, 1897
Bembidion (Notaphus) conspersum Chaudoir, 1868
Bembidion (Notaphus) constrictum (Leconte, 1847)
Bembidion (Notaphus) consuetum Casey, 1918
Bembidion (Notaphus) contractum Say, 1823
Bembidion (Notaphus) cordatum (LeConte, 1847)
Bembidion (Notaphus) debiliceps Casey, 1918
Bembidion (Notaphus) dejectum Casey, 1884
Bembidion (Notaphus) dorsale Say, 1823
Bembidion (Notaphus) evidens Casey, 1918
Bembidion (Notaphus) flohri Bates, 1878
Bembidion (Notaphus) graphicum Casey, 1918
Bembidion (Notaphus) hageni Hayward, 1897
Bembidion (Notaphus) idoneum Casey, 1918
Bembidion (Notaphus) indistinctum Dejean, 1831
Bembidion (Notaphus) insulatum (LeConte, 1852)
Bembidion (Notaphus) intermedium (Kirby, 1837)
Bembidion (Notaphus) jacobianum Casey, 1918
Bembidion (Notaphus) latebricola Casey, 1918
Bembidion (Notaphus) lecontei Csiki, 1928
Bembidion (Notaphus) luculentum Casey, 1918
Bembidion (Notaphus) mormon Hayward, 1897
Bembidion (Notaphus) nigripes (Kirby, 1837)‡
Bembidion (Notaphus) nubiculosum Chaudoir, 1868
Bembidion (Notaphus) oberthueri Hayward, 1901
Bembidion (Notaphus) obscuromaculatum (Motschulsky, 1859)
Bembidion (Notaphus) obtusangulum LeConte, 1863
Bembidion (Notaphus) obtusidens Fall, 1922
Bembidion (Notaphus) operosum Casey, 1918
Bembidion (Notaphus) patruele Dejean, 1831
Bembidion (Notaphus) pilatei Chaudoir, 1868
Bembidion (Notaphus) pimanum Casey, 1918
Bembidion (Notaphus) rapidum (LeConte, 1847)
Bembidion (Notaphus) scintillans Bates, 1882
Bembidion (Notaphus) scudderi LeConte, 1878
Bembidion (Notaphus) semiopacum Casey, 1924
Bembidion (Notaphus) semipunctatum (Donovan, 1806)‡
Bembidion (Notaphus) umbratum (LeConte, 1847)
Bembidion (Notaphus) versutum LeConte, 1878
Bembidion (Notaphus) viridicolle (LaFerté-Sénectère, 1841)
Bembidion (Notaphus) vividum Casey, 1884
Bembidion (Notaphus) vulpecula Casey, 1918
Bembidion (Trepanedoris) acutifrons LeConte, 1879
Bembidion (Trepanedoris) ampliceps Casey, 1918
Bembidion (Trepanedoris) anguliferum (LeConte, 1852)
Bembidion (Trepanedoris) canadianum Casey, 1924
Bembidion (Trepanedoris) clemens Casey, 1918
Bembidion (Trepanedoris) concretum Casey, 1918
Bembidion (Trepanedoris) connivens (LeConte, 1852)
Bembidion (Trepanedoris) elizabethae Hatch, 1950
Bembidion (Trepanedoris) fortestriatum (Motschulsky, 1845)
Bembidion (Trepanedoris) frontale (LeConte, 1847)
Bembidion (Trepanedoris) pseudocautum Lindroth, 1963
Bembidion (Trepanedoris) scenicum Casey, 1918
Bembidion (Trepanedoris) siticum Casey, 1918
Bembidion (Peryphodes) ephippigerum (LeConte, 1852)
Bembidion (Peryphodes) salinarium Casey, 1918
Bembidion (Emphanes) diligens Casey, 1918
Bembidion (Emphanes) vile (LeConte, 1852)
Bembidion (Blepharoplataphus) hastii Sahlberg, 1827‡
Bembidion (Plataphus) arcticum Lindroth, 1963‡
Bembidion (Plataphus) basicorne Notman, 1920
Bembidion (Plataphus) brachythorax Lindroth, 1963‡
Bembidion (Plataphus) breve (Motschulsky, 1845)
Bembidion (Plataphus) carolinense Casey, 1924
Bembidion (Plataphus) complanulum (Mannerheim, 1853)
Bembidion (Plataphus) compressum Lindroth, 1963‡
Bembidion (Plataphus) curtulatum Casey, 1918
Bembidion (Plataphus) falsum Blaisdell, 1902
Bembidion (Plataphus) farrarae Hatch, 1950
Bembidion (Plataphus) gebleri turbatum Casey, 1918
Bembidion (Plataphus) gordoni Lindroth, 1963
Bembidion (Plataphus) gratiosum Casey, 1918
Bembidion (Plataphus) haruspex Casey, 1918
Bembidion (Plataphus) hyperboraeorum Munster, 1923‡
Bembidion (Plataphus) improvidens Casey, 1924
Bembidion (Plataphus) kalumae Lindroth, 1963
Bembidion (Plataphus) kuprianovii Mannerheim, 1843
Bembidion (Plataphus) laxatum Casey, 1918
Bembidion (Plataphus) manningense Lindroth, 1969
Bembidion (Plataphus) neocoerulescens Bousquet, 1993
Bembidion (Plataphus) nigrocoeruleum Hayward, 1897
Bembidion (Plataphus) occultator Notman, 1920
Bembidion (Plataphus) oppressum Casey, 1918
Bembidion (Plataphus) placeranum Casey, 1924
Bembidion (Plataphus) planiusculum Mannerheim, 1843
Bembidion (Plataphus) quadrifoveolatum Mannerheim, 1843
Bembidion (Plataphus) rosslandicum Lindroth, 1963
Bembidion (Plataphus) rufinum Lindroth, 1963
Bembidion (Plataphus) rusticum lenensoides Lindroth, 1963
Bembidion (Plataphus) rusticum rusticum Casey, 1918
Bembidion (Plataphus) sierricola Casey, 1924
Bembidion (Plataphus) simplex Hayward, 1897
Bembidion (Plataphus) stillaguamish Hatch, 1950
Bembidion (Plataphus) sulcipenne hyperboroides Lindroth, 1963
Bembidion (Plataphus) sulcipenne prasinoides Lindroth, 1963
Bembidion (Plataphus) vandykei Blaisdell, 1902
Bembidion (Plataphus) viator Casey, 1918
Bembidion (Hydrium) interventor Lindroth, 1963
Bembidion (Hydrium) levigatum Say, 1823
Bembidion (Hydrium) nitidum (Kirby, 1837)
Bembidion (Hydrium) obliquulum LeConte, 1859
Bembidion (Metallina) dyschirinum LeConte, 1861
Bembidion (Metallina) lampros (Herbst, 1784)†
Bembidion (Metallina) properans (Stephens, 1828)†
Bembidion (Lindrochthus) wickhami Hayward, 1897
Bembidion (Eupetedromus) graciliforme Hayward, 1897
Bembidion (Eupetedromus) immaturum Lindroth, 1954
Bembidion (Eupetedromus) incrematum LeConte, 1860‡
Bembidion (Eupetedromus) iridipenne Bousquet & Webster, 2006
Bembidion (Eupetedromus) variegatum Say, 1823
Bembidion (Trechonepha) iridescens (LeConte, 1852)
Bembidion (Trechonepha) trechiforme (LeConte, 1852)
Bembidion (Liocosmius) festivum Casey, 1918
Bembidion (Liocosmius) horni Hayward, 1897
Bembidion (Liocosmius) mundum (LeConte, 1852)
Bembidion (Melomalus) planatum (LeConte, 1847)
Bembidion (Trichoplataphus) fugax (LeConte, 1848)
Bembidion (Trichoplataphus) grandiceps Hayward, 1897
Bembidion (Trichoplataphus) ozarkense Maddison & Hildebrandt, 2011
Bembidion (Trichoplataphus) planum (Haldeman, 1843)
Bembidion (Trichoplataphus) rolandi Fall, 1922
Bembidion (Phyla) obtusum Audinet-Serville, 1821†
Bembidion (Lymnaeum) laticeps (LeConte, 1858)
Bembidion (Lymnaeum) nigropiceum (Marsham, 1802)†
Phrypeus rickseckeri (Hayward, 1897)
Mioptachys flavicauda (Say, 1823)
Tachyta (Tachyta) angulata Casey, 1918
Tachyta (Tachyta) falli (Hayward, 1900)
Tachyta (Tachyta) inornata (Say, 1823)
Tachyta (Tachyta) kirbyi Casey, 1918
Tachyta (Tachyta) parvicornis Notman, 1922
Elaphropus (Barytachys) anceps (LeConte, 1848)
Elaphropus (Barytachys) anthrax (LeConte, 1852)
Elaphropus (Barytachys) brevis (Casey, 1918)
Elaphropus (Barytachys) brunnicollis (Motschulsky, 1862)
Elaphropus (Barytachys) capax (LeConte, 1863)
Elaphropus (Barytachys) cockerelli (Fall, 1907)
Elaphropus (Barytachys) congener (Casey, 1918)
Elaphropus (Barytachys) conjugens (Notman, 1919)
Elaphropus (Barytachys) dolosus (LeConte, 1848)
Elaphropus (Barytachys) fatuus (Casey, 1918)
Elaphropus (Barytachys) ferrugineus (Dejean, 1831)
Elaphropus (Barytachys) fuscicornis (Chaudoir, 1868)
Elaphropus (Barytachys) granarius (Dejean, 1831)
Elaphropus (Barytachys) incurvus (Say, 1830)
Elaphropus (Barytachys) liebecki (Hayward, 1900)
Elaphropus (Barytachys) monticola (Casey, 1918)
Elaphropus (Barytachys) nebulosus (Chaudoir, 1868)
Elaphropus (Barytachys) obesulus (LeConte, 1852)
Elaphropus (Barytachys) rapax (LeConte, 1852)
Elaphropus (Barytachys) renoicus (Casey, 1918)
Elaphropus (Barytachys) rubricauda (Casey, 1918)
Elaphropus (Barytachys) saturatus (Casey, 1918)
Elaphropus (Barytachys) sectator (Casey, 1918)
Elaphropus (Barytachys) sedulus (Casey, 1918)
Elaphropus (Barytachys) tahoensis (Casey, 1918)
Elaphropus (Barytachys) tripunctatus (Say, 1830)
Elaphropus (Barytachys) vernicatus (Casey, 1918)
Elaphropus (Barytachys) vivax (LeConte, 1848)
Elaphropus (Barytachys) xanthopus (Dejean, 1831)
Elaphropus (Tachyura) parvulus (Dejean, 1831)†
Micratopus aenescens (LeConte, 1848)
Pericompsus (Pericompsus) ephippiatus (Say, 1830)
Pericompsus (Pericompsus) laetulus LeConte, 1852
Pericompsus (Pericompsus) sellatus LeConte, 1852
Porotachys bisulcatus (Nicolai, 1822)†
Polyderis diaphana (Casey, 1918)
Polyderis laeva (Say, 1823)
Polyderis rufotestacea (Hayward, 1900)
Tachys (Tachys) bradycellinus Hayward, 1900
Tachys (Tachys) bryanti Lindroth, 1966
Tachys (Tachys) corax LeConte, 1852
Tachys (Tachys) halophilus Lindroth, 1966
Tachys (Tachys) litoralis Casey, 1884
Tachys (Tachys) misellus LaFerté-Sénectère, 1841
Tachys (Tachys) mordax LeConte, 1852
Tachys (Tachys) pallidus Chaudoir, 1868
Tachys (Tachys) pulchellus LaFerté-Sénectère, 1841
Tachys (Tachys) translucens Darlington, 1937
Tachys (Tachys) virgo LeConte, 1852
Tachys (Tachys) vittiger LeConte, 1852
Tachys (Paratachys) aeneipennis Motschulsky, 1862
Tachys (Paratachys) albipes LeConte, 1863
Tachys (Paratachys) austinicus (Casey, 1918)
Tachys (Paratachys) columbiensis Hayward, 1900
Tachys (Paratachys) edax LeConte, 1852
Tachys (Paratachys) hyalinus Casey, 1918
Tachys (Paratachys) oblitus Casey, 1918
Tachys (Paratachys) potomaca (Erwin, 1981)
Tachys (Paratachys) proximus (Say, 1823)
Tachys (Paratachys) pumilus (Dejean, 1831)
Tachys (Paratachys) rectangulus Notman, 1919
Tachys (Paratachys) rhodeanus Casey, 1918
Tachys (Paratachys) sagax Casey, 1918
Tachys (Paratachys) scitulus LeConte, 1848
Tachys (Paratachys) sequax LeConte, 1848
Tachys (Paratachys) spadix Casey, 1918
Tachys (Paratachys) umbripennis Chaudoir, 1868
Tachys (Paratachys) ventricosus LeConte, 1863
Tachys (Paratachys) vernilis Casey, 1918
Tachys (Paratachys) vorax LeConte, 1852
Anillodes debilis (LeConte, 1853)
Anillodes minutus Jeannel, 1963
Anillodes walkeri Jeannel, 1963
Anillinus affabilis (Brues, 1902)
Anillinus aleyae Sokolov & Watrous, 2008
Anillinus balli Sokolov & Carlton, 2004
Anillinus barberi Jeannel, 1963
Anillinus barri Sokolov & Carlton, 2004
Anillinus campbelli Giachino, 2011
Anillinus carltoni Sokolov, 2011
Anillinus chandleri Sokolov, 2011
Anillinus cherokee Sokolov & Carlton, 2008
Anillinus chilhowee Sokolov, 2011
Anillinus cieglerae Sokolov & Carlton, 2007
Anillinus cornelli Sokolov & Carlton, 2004
Anillinus daggyi Sokolov & Carlton, 2004
Anillinus depressus (Jeannel, 1963)
Anillinus docwatsoni Sokolov & Carlton, 2004
Anillinus dohrni (Ehlers, 1884)
Anillinus elongatus Jeannel, 1963
Anillinus erwini Sokolov & Carlton, 2004
Anillinus folkertsi Sokolov & Carlton, 2004
Anillinus fortis (Horn, 1869)
Anillinus gimmeli Sokolov & Carlton, 2010
Anillinus indianae Jeannel, 1963
Anillinus juliae Sokolov & Carlton, 2010
Anillinus kovariki Sokolov & Carlton, 2004
Anillinus langdoni Sokolov & Carlton, 2004
Anillinus lescheni Sokolov & Carlton, 2004
Anillinus longiceps Jeannel, 1963
Anillinus loweae Sokolov & Carlton, 2004
Anillinus magazinensis Sokolov & Carlton, 2004
Anillinus merritti Sokolov & Carlton, 2010
Anillinus moseleyae Sokolov & Carlton, 2004
Anillinus murrayae Sokolov & Carlton, 2004
Anillinus nantahala Dajoz, 2005
Anillinus pecki Giachino, 2011
Anillinus pusillus Sokolov & Carlton, 2007
Anillinus robisoni Sokolov & Carlton, 2004
Anillinus sinuaticollis Jeannel, 1963
Anillinus sinuatus Jeannel, 1963
Anillinus smokiensis Sokolov, 2011
Anillinus steevesi Barr, 1995
Anillinus stephani Sokolov & Carlton, 2004
Anillinus tishechkini Sokolov & Carlton, 2004
Anillinus turneri Jeannel, 1963
Anillinus unicoi Sokolov, 2011
Anillinus valentinei (Jeannel, 1963)
Anillinus virginiae Jeannel, 1963
Serranillus dunavani (Jeannel, 1963)
Serranillus jeanneli Barr, 1995
Serranillus septentrionis Sokolov & Carlton, 2008
Anillaspis caseyi Jeannel, 1963
Anillaspis explanata (Horn, 1888)
Horologion speokoites Valentine, 1932
PogoniniThalassotrechus barbarae (Horn, 1892)
Diplochaetus emaciatus (Bates, 1891)
Diplochaetus megacephalus Bousquet & Laplante, 1997
Diplochaetus planatus (Horn, 1876)
Diplochaetus rutilus (Chevrolat, 1863)
Pogonus texanus Chaudoir, 1868
PatrobiniDiplous (Platidius) aterrimus (Dejean, 1828)
Diplous (Platidius) californicus (Motschulsky, 1844)
Diplous (Platidius) filicornis (Casey, 1918)
Diplous (Platidius) rugicollis (Randall, 1838)
Patrobus cinctus Motschulsky, 1860‡
Patrobus fossifrons (Eschscholtz, 1823)
Patrobus foveocollis (Eschscholtz, 1823)‡
Patrobus lecontei Chaudoir, 1872
Patrobus longicornis (Say, 1823)
Patrobus septentrionis septentrionis Dejean, 1828‡
Patrobus stygicus Chaudoir, 1872‡
Platypatrobus lacustris Darlington, 1938
Platidiolus vandykei Kurnakov, 1960
PsydriniNomius pygmaeus (Dejean, 1831)
Psydrus piceus LeConte, 1846
MetriiniMetrius contractus contractus Eschscholtz, 1829
Metrius contractus planatus Van Dyke, 1925
Metrius contractus sericeus Rivers, 1900
Metrius explodens Bousquet & Goulet, 1990
PaussiniPachyteles gyllenhalii (Dejean, 1825)
Goniotropis kuntzeni kuntzeni Bänninger, 1927
Goniotropis parca (LeConte, 1884)
Physea hirta LeConte, 1853
Ozaena lemoulti Bänninger, 1932
BrachininiBrachinus (Neobrachinus) aabaaba Erwin, 1970
Brachinus (Neobrachinus) adustipennis Erwin, 1969
Brachinus (Neobrachinus) aeger Chaudoir, 1876
Brachinus (Neobrachinus) alexiguus Erwin, 1970
Brachinus (Neobrachinus) alternans Dejean, 1825
Brachinus (Neobrachinus) americanus (LeConte, 1844)
Brachinus (Neobrachinus) azureipennis Chaudoir, 1876
Brachinus (Neobrachinus) capnicus Erwin, 1970
Brachinus (Neobrachinus) cibolensis Erwin, 1970
Brachinus (Neobrachinus) conformis Dejean, 1831
Brachinus (Neobrachinus) cordicollis Dejean, 1826
Brachinus (Neobrachinus) costipennis Motschulsky, 1859
Brachinus (Neobrachinus) cyanipennis Say, 1823
Brachinus (Neobrachinus) cyanochroaticus Erwin, 1969
Brachinus (Neobrachinus) elongatulus Chaudoir, 1876
Brachinus (Neobrachinus) explosus Erwin, 1970
Brachinus (Neobrachinus) favicollis Erwin, 1965
Brachinus (Neobrachinus) fulminatus Erwin, 1969
Brachinus (Neobrachinus) fumans (Fabricius, 1781)
Brachinus (Neobrachinus) gebhardis Erwin, 1965
Brachinus (Neobrachinus) geniculatus Dejean, 1831
Brachinus (Neobrachinus) hirsutus Bates, 1884
Brachinus (Neobrachinus) ichabodopsis Erwin, 1970
Brachinus (Neobrachinus) imperialensis Erwin, 1965
Brachinus (Neobrachinus) imporcitis Erwin, 1970
Brachinus (Neobrachinus) janthinipennis (Dejean, 1831)
Brachinus (Neobrachinus) javalinopsis Erwin, 1970
Brachinus (Neobrachinus) kansanus LeConte, 1863
Brachinus (Neobrachinus) kavanaughi Erwin, 1969
Brachinus (Neobrachinus) lateralis Dejean, 1831
Brachinus (Neobrachinus) medius Harris, 1828
Brachinus (Neobrachinus) mexicanus Dejean, 1831
Brachinus (Neobrachinus) microamericanus Erwin, 1969
Brachinus (Neobrachinus) mobilis Erwin, 1970
Brachinus (Neobrachinus) neglectus LeConte, 1844
Brachinus (Neobrachinus) ovipennis LeConte, 1863
Brachinus (Neobrachinus) oxygonus Chaudoir, 1843
Brachinus (Neobrachinus) pallidus Erwin, 1965
Brachinus (Neobrachinus) patruelis LeConte, 1844
Brachinus (Neobrachinus) perplexus Dejean, 1831
Brachinus (Neobrachinus) phaeocerus Chaudoir, 1868
Brachinus (Neobrachinus) puberulus Chaudoir, 1868
Brachinus (Neobrachinus) quadripennis Dejean, 1825
Brachinus (Neobrachinus) rugipennis Chaudoir, 1868
Brachinus (Neobrachinus) sublaevis Chaudoir, 1868
Brachinus (Neobrachinus) tenuicollis LeConte, 1844
Brachinus (Neobrachinus) texanus Chaudoir, 1868
Brachinus (Neobrachinus) velutinus Erwin, 1965
Brachinus (Neobrachinus) viridipennis Dejean, 1831
Brachinus (Neobrachinus) vulcanoides Erwin, 1969
MorioniniMorion aridus Allen, 1969
Morion monilicornis (Latreille, 1805)
AbacetiniLoxandrus accelerans Casey, 1918
Loxandrus agilis (Dejean, 1828)
Loxandrus algidus Allen, 1972
Loxandrus brevicollis (LeConte, 1846)
Loxandrus celer (Dejean, 1828)
Loxandrus cervicalis Casey, 1918
Loxandrus cincinnati Casey, 1924
Loxandrus circulus Allen, 1972
Loxandrus collucens Casey, 1918
Loxandrus crenatus LeConte, 1853
Loxandrus duryi Wright, 1939
Loxandrus erraticus (Dejean, 1828)
Loxandrus extendus Allen, 1972
Loxandrus floridanus LeConte, 1878
Loxandrus gibbus Allen, 1972
Loxandrus icarus Will & Liebherr, 1998
Loxandrus infimus Bates, 1882
Loxandrus lucens Chaudoir, 1868
Loxandrus micans Chaudoir, 1868
Loxandrus minor (Chaudoir, 1843)
Loxandrus nitidulus (LeConte, 1846)
Loxandrus pactinullus Allen, 1972
Loxandrus parallelus Casey, 1918
Loxandrus parvulus Chaudoir, 1868
Loxandrus piceolus Chaudoir, 1868
Loxandrus piciventris (LeConte, 1846)
Loxandrus pravitubus Allen, 1972
Loxandrus proximus Chaudoir, 1868
Loxandrus pusillus LeConte, 1853
Loxandrus rectangulus LeConte, 1878
Loxandrus rectus (Say, 1823)
Loxandrus robustus Allen, 1972
Loxandrus rossi Allen, 1972
Loxandrus saccisecundaris Allen, 1972
Loxandrus saphyrinus (Chaudoir, 1843)
Loxandrus sculptilis Bates, 1884
Loxandrus spinilunatus Allen, 1972
Loxandrus straneoi Will & Liebherr, 1998
Loxandrus taeniatus LeConte, 1853
Loxandrus uniformis Allen, 1972
Loxandrus unilobus Allen, 1972
Loxandrus velocipes Casey, 1918
Loxandrus velox (Dejean, 1828)
Loxandrus vulneratus Casey, 1918
Stolonis intercepta Chaudoir, 1874
PterostichiniAbaris (Abaridius) splendidula (LeConte, 1863)
Hybothecus flohri (Bates, 1882)
Poecilus (Poecilus) chalcites (Say, 1823)
Poecilus (Poecilus) coloradensis (Csiki, 1930)
Poecilus (Poecilus) corvus (LeConte, 1873)
Poecilus (Poecilus) cursitor LeConte, 1853
Poecilus (Poecilus) cyanicolor Chaudoir, 1876
Poecilus (Poecilus) diplophryus Chaudoir, 1876
Poecilus (Poecilus) laetulus (LeConte, 1863)
Poecilus (Poecilus) lucublandus (Say, 1823)
Poecilus (Poecilus) mexicanus Chaudoir, 1876
Poecilus (Poecilus) occidentalis (Dejean, 1828)
Poecilus (Poecilus) scitulus LeConte, 1846
Poecilus (Poecilus) texanus (LeConte, 1863)
Poecilus (Derus) nearcticus (Lindroth, 1966)‡
Lophoglossus gravis LeConte, 1873
Lophoglossus haldemanni (LeConte, 1846)
Lophoglossus scrutator (LeConte, 1846)
Lophoglossus substrenuus (Csiki, 1930)
Lophoglossus tartaricus (Say, 1823)
Lophoglossus vernix Casey, 1913
Piesmus submarginatus (Say, 1823)
Gastrellarius blanchardi (Horn, 1891)
Gastrellarius honestus (Say, 1823)
Gastrellarius unicarum (Darlington, 1932)
Stomis (Neostomis) termitiformis (Van Dyke, 1926)
Stomis (Stomis) pumicatus (Panzer, 1795)†
Stereocerus haematopus (Dejean, 1831)‡
Stereocerus rubripes (Motschulsky, 1860)‡
Myas (Trigonognatha) coracinus (Say, 1823)
Myas (Trigonognatha) cyanescens Dejean, 1828
Pterostichus (Argutor) commutabilis (Motschulsky, 1866)
Pterostichus (Argutor) praetermissus (Chaudoir, 1868)
Pterostichus (Argutor) vernalis (Panzer, 1795)†
Pterostichus (Phonias) corrusculus LeConte, 1873
Pterostichus (Phonias) femoralis (Kirby, 1837)
Pterostichus (Phonias) patruelis (Dejean, 1831)
Pterostichus (Phonias) strenuus (Panzer, 1796)†
Pterostichus (Bothriopterus) adstrictus Eschscholtz, 1823‡
Pterostichus (Bothriopterus) lustrans LeConte, 1851
Pterostichus (Bothriopterus) mutus (Say, 1823)
Pterostichus (Bothriopterus) oregonus LeConte, 1861
Pterostichus (Bothriopterus) pensylvanicus LeConte, 1873
Pterostichus (Bothriopterus) trinarius (Casey, 1918)
Pterostichus (Melanius) castor Goulet & Bousquet, 1983
Pterostichus (Melanius) corvinus (Dejean, 1828)
Pterostichus (Melanius) ebeninus (Dejean, 1828)
Pterostichus (Pseudomaseus) luctuosus (Dejean, 1828)
Pterostichus (Pseudomaseus) tenuis (Casey, 1924)
Pterostichus (Feronina) barri Bousquet, 2006
Pterostichus (Feronina) palmi Schaeffer, 1910
Pterostichus (Paraferonia) lubricus LeConte, 1853
Pterostichus (Pseudoferonina) amadeus Bousquet, nomen novum
Pterostichus (Pseudoferonina) bousqueti Bergdahl, 2011
Pterostichus (Pseudoferonina) campbelli Bousquet, 1985
Pterostichus (Pseudoferonina) humidulus (Van Dyke, 1943)
Pterostichus (Pseudoferonina) lanei Van Dyke, 1926
Pterostichus (Pseudoferonina) lolo Bergdahl, 2011
Pterostichus (Pseudoferonina) shulli (Hatch, 1949)
Pterostichus (Pseudoferonina) smetanai Bousquet, 1985
Pterostichus (Pseudoferonina) spathifer Bousquet, 1992
Pterostichus (Gastrosticta) enodis Bousquet, 1992
Pterostichus (Gastrosticta) mutoides Bousquet, 1992
Pterostichus (Gastrosticta) obesulus LeConte, 1873
Pterostichus (Gastrosticta) ophryoderus (Chaudoir, 1878)
Pterostichus (Gastrosticta) punctiventris (Chaudoir, 1878)
Pterostichus (Gastrosticta) putus Casey, 1913
Pterostichus (Gastrosticta) sayanus Csiki, 1930
Pterostichus (Gastrosticta) subacutus (Casey, 1918)
Pterostichus (Gastrosticta) tumescens LeConte, 1863
Pterostichus (Gastrosticta) ventralis (Say, 1823)
Pterostichus (Morphnosoma) melanarius melanarius (Illiger, 1798)†
Pterostichus (Euferonia) coracinus (Newman, 1838)
Pterostichus (Euferonia) ingens (Casey, 1918)
Pterostichus (Euferonia) lachrymosus (Newman, 1838)
Pterostichus (Euferonia) novus Straneo, 1944
Pterostichus (Euferonia) relictus (Newman, 1838)
Pterostichus (Euferonia) stygicus (Say, 1823)
Pterostichus (Lenapterus) agonus Horn, 1880‡
Pterostichus (Lenapterus) costatus (Ménétriés, 1851)‡
Pterostichus (Lenapterus) punctatissimus (Randall, 1838)
Pterostichus (Lenapterus) vermiculosus (Ménétriés, 1851)‡
Pterostichus (Metallophilus) sublaevis (Sahlberg, 1880)‡
Pterostichus (Abacidus) atratus (Newman, 1838)
Pterostichus (Abacidus) fallax (Dejean, 1828)
Pterostichus (Abacidus) hamiltoni Horn, 1880
Pterostichus (Abacidus) permundus (Say, 1830)
Pterostichus (Abacidus) sculptus LeConte, 1853
Pterostichus (Orsonjohnsonus) johnsoni Ulke, 1889
Pterostichus (Lamenius) caudicalis (Say, 1823)
Pterostichus (Eosteropus) circulosus Lindroth, 1966
Pterostichus (Eosteropus) moestus (Say, 1823)
Pterostichus (Eosteropus) superciliosus (Say, 1823)
Pterostichus (Monoferonia) carolinus carolinus Darlington, 1932
Pterostichus (Monoferonia) carolinus fumorum Darlington, 1932
Pterostichus (Monoferonia) diligendus (Chaudoir, 1868)
Pterostichus (Monoferonia) mancus (LeConte, 1853)
Pterostichus (Monoferonia) primus Darlington, 1932
Pterostichus (Cylindrocharis) acutipes acutipes Barr, 1971
Pterostichus (Cylindrocharis) acutipes kentuckensis Barr, 1971
Pterostichus (Cylindrocharis) hypogeus Barr, 1971
Pterostichus (Cylindrocharis) rostratus (Newman, 1838)
Pterostichus (Leptoferonia) angustus (Dejean, 1828)
Pterostichus (Leptoferonia) beyeri Van Dyke, 1926
Pterostichus (Leptoferonia) blodgettensis Will, 2007
Pterostichus (Leptoferonia) caligans Horn, 1891
Pterostichus (Leptoferonia) cochlearis Hacker, 1968
Pterostichus (Leptoferonia) deino Will, 2007
Pterostichus (Leptoferonia) enyo Will, 2007
Pterostichus (Leptoferonia) falli Van Dyke, 1926
Pterostichus (Leptoferonia) fenyesi fenderi Hacker, 1968
Pterostichus (Leptoferonia) fenyesi fenyesi Csiki, 1930
Pterostichus (Leptoferonia) fuchsi Schaeffer, 1910
Pterostichus (Leptoferonia) hatchi Hacker, 1968
Pterostichus (Leptoferonia) humilis Casey, 1913
Pterostichus (Leptoferonia) idahoae Csiki, 1930
Pterostichus (Leptoferonia) inanis Horn, 1891
Pterostichus (Leptoferonia) infernalis Hatch, 1936
Pterostichus (Leptoferonia) inopinus (Casey, 1918)
Pterostichus (Leptoferonia) lobatus Hacker, 1968
Pterostichus (Leptoferonia) marinensis Hacker, 1968
Pterostichus (Leptoferonia) mattolensis Hacker, 1968
Pterostichus (Leptoferonia) pemphredo Will, 2007
Pterostichus (Leptoferonia) pumilus pumilus Casey, 1913
Pterostichus (Leptoferonia) pumilus willamettensis Hacker, 1968
Pterostichus (Leptoferonia) rothi (Hatch, 1951)
Pterostichus (Leptoferonia) sphodrinus LeConte, 1863
Pterostichus (Leptoferonia) stapedius Hacker, 1968
Pterostichus (Leptoferonia) trinitensis Hacker, 1968
Pterostichus (Leptoferonia) yosemitensis Hacker, 1968
Pterostichus (Anilloferonia) lanei (Hatch, 1935)
Pterostichus (Anilloferonia) malkini (Hatch, 1953)
Pterostichus (Anilloferonia) testaceus (Van Dyke, 1926)
Pterostichus (Hypherpes) adoxus (Say, 1823)
Pterostichus (Hypherpes) algidus LeConte, 1853
Pterostichus (Hypherpes) amethystinus Mannerheim, 1843
Pterostichus (Hypherpes) annosus Casey, 1913
Pterostichus (Hypherpes) arcanus Casey, 1913
Pterostichus (Hypherpes) baldwini (Casey, 1924)
Pterostichus (Hypherpes) barbarinus Casey, 1913
Pterostichus (Hypherpes) brachylobus Kavanaugh & LaBonte, 2006
Pterostichus (Hypherpes) californicus (Dejean, 1828)
Pterostichus (Hypherpes) canallatus Casey, 1913
Pterostichus (Hypherpes) castaneus (Dejean, 1828)
Pterostichus (Hypherpes) castanipes (Ménétriés, 1843)
Pterostichus (Hypherpes) congestus (Ménétriés, 1843)
Pterostichus (Hypherpes) craterensis (Hatch, 1949)
Pterostichus (Hypherpes) crenicollis LeConte, 1873
Pterostichus (Hypherpes) ecarinatus Hatch, 1936
Pterostichus (Hypherpes) esuriens Casey, 1913
Pterostichus (Hypherpes) gliscans Casey, 1913
Pterostichus (Hypherpes) gracilior LeConte, 1873
Pterostichus (Hypherpes) herculaneus Mannerheim, 1843
Pterostichus (Hypherpes) hornii LeConte, 1873
Pterostichus (Hypherpes) illustris LeConte, 1851
Pterostichus (Hypherpes) inermis Fall, 1901
Pterostichus (Hypherpes) isabellae LeConte, 1851
Pterostichus (Hypherpes) jacobinus Casey, 1913
Pterostichus (Hypherpes) laborans Casey, 1913
Pterostichus (Hypherpes) lacertus Casey, 1913
Pterostichus (Hypherpes) lama (Ménétriés, 1843)
Pterostichus (Hypherpes) lassulus (Casey, 1920)
Pterostichus (Hypherpes) lattini LaBonte, 2006
Pterostichus (Hypherpes) luscus (Casey, 1918)
Pterostichus (Hypherpes) menetriesii LeConte, 1873
Pterostichus (Hypherpes) mercedianus (Casey, 1918)
Pterostichus (Hypherpes) miscellus Casey, 1913
Pterostichus (Hypherpes) morionides (Chaudoir, 1868)
Pterostichus (Hypherpes) neobrunneus Lindroth, 1966
Pterostichus (Hypherpes) nigrocaeruleus Van Dyke, 1926
Pterostichus (Hypherpes) obsidianus Casey, 1913
Pterostichus (Hypherpes) occultus Casey, 1913
Pterostichus (Hypherpes) ordinarius Casey, 1913
Pterostichus (Hypherpes) ovalipennis Casey, 1913
Pterostichus (Hypherpes) panticulatus Casey, 1913
Pterostichus (Hypherpes) pergracilis (Casey, 1920)
Pterostichus (Hypherpes) planctus LeConte, 1853
Pterostichus (Hypherpes) protensiformis (Casey, 1924)
Pterostichus (Hypherpes) protractus LeConte, 1860
Pterostichus (Hypherpes) restrictus (Casey, 1918)
Pterostichus (Hypherpes) scutellaris LeConte, 1873
Pterostichus (Hypherpes) serripes (LeConte, 1875)
Pterostichus (Hypherpes) setosus Hatch, 1951
Pterostichus (Hypherpes) sierranus Casey, 1913
Pterostichus (Hypherpes) sponsor Casey, 1913
Pterostichus (Hypherpes) spraguei LeConte, 1873
Pterostichus (Hypherpes) suffusus Casey, 1913
Pterostichus (Hypherpes) tarsalis LeConte, 1873
Pterostichus (Hypherpes) tristis (Dejean, 1828)
Pterostichus (Hypherpes) tuberculofemoratus Hatch, 1936
Pterostichus (Hypherpes) vandykei Schaeffer, 1910
Pterostichus (Hypherpes) vicinus Mannerheim, 1843
Pterostichus (Hypherpes) ybousqueti Berlov, 1999
Pterostichus (Cryobius) arcticola (Chaudoir, 1868)
Pterostichus (Cryobius) auriga Ball, 1962
Pterostichus (Cryobius) barryorum Ball, 1962
Pterostichus (Cryobius) brevicornis brevicornis (Kirby, 1837)‡
Pterostichus (Cryobius) bryanti biocryus Ball, 1962
Pterostichus (Cryobius) bryanti bryanti (Van Dyke, 1951)
Pterostichus (Cryobius) bryanti bryantoides Ball, 1962
Pterostichus (Cryobius) bryanti cacumenis Ball, 1966
Pterostichus (Cryobius) bryanti stantonensis Ball, 1966
Pterostichus (Cryobius) bryanti tiliaceoradix Ball, 1962
Pterostichus (Cryobius) caribou Ball, 1962
Pterostichus (Cryobius) chipewyan Ball, 1962
Pterostichus (Cryobius) empetricola (Dejean, 1828)‡
Pterostichus (Cryobius) gerstlensis Ball, 1962
Pterostichus (Cryobius) hudsonicus LeConte, 1863
Pterostichus (Cryobius) kotzebuei Ball, 1962
Pterostichus (Cryobius) mandibularoides Ball, 1966
Pterostichus (Cryobius) nivalis (Sahlberg, 1844)‡
Pterostichus (Cryobius) parasimilis Ball, 1962‡
Pterostichus (Cryobius) pinguedineus (Eschscholtz, 1823)‡
Pterostichus (Cryobius) planus (Sahlberg, 1885)
Pterostichus (Cryobius) riparius (Dejean, 1828)
Pterostichus (Cryobius) similis Mannerheim, 1852‡
Pterostichus (Cryobius) soperi Ball, 1966
Pterostichus (Cryobius) surgens LeConte, 1878
Pterostichus (Cryobius) tareumiut Ball, 1962‡
Pterostichus (Cryobius) ventricosus ventricosus (Eschscholtz, 1823)‡
Pterostichus (Cryobius) woodi Ball & Currie, 1997
Cyclotrachelus (Cyclotrachelus) alabamensis (Casey, 1920)
Cyclotrachelus (Cyclotrachelus) approximatus (LeConte, 1846)
Cyclotrachelus (Cyclotrachelus) brevoorti (LeConte, 1846)
Cyclotrachelus (Cyclotrachelus) dejeanellus (Csiki, 1930)
Cyclotrachelus (Cyclotrachelus) faber (Germar, 1824)
Cyclotrachelus (Cyclotrachelus) freitagi Bousquet, 1993
Cyclotrachelus (Cyclotrachelus) fucatus (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) hernandensis (Van Dyke, 1943)
Cyclotrachelus (Cyclotrachelus) iuvenis (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) laevipennis (LeConte, 1846)
Cyclotrachelus (Cyclotrachelus) levifaber (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) macrovulum (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) ovulum (Chaudoir, 1868)
Cyclotrachelus (Cyclotrachelus) parafaber (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) spoliatus (Newman, 1838)
Cyclotrachelus (Cyclotrachelus) texensis (Freitag, 1969)
Cyclotrachelus (Cyclotrachelus) unicolor (Say, 1823)
Cyclotrachelus (Cyclotrachelus) vinctus (LeConte, 1853)
Cyclotrachelus (Evarthrus) alabamae (Van Dyke, 1926)
Cyclotrachelus (Evarthrus) alternans (Casey, 1920)
Cyclotrachelus (Evarthrus) blatchleyi (Casey, 1918)
Cyclotrachelus (Evarthrus) constrictus (Say, 1823)
Cyclotrachelus (Evarthrus) convivus (LeConte, 1853)
Cyclotrachelus (Evarthrus) deceptus (Casey, 1918)
Cyclotrachelus (Evarthrus) engelmani (LeConte, 1853)
Cyclotrachelus (Evarthrus) floridensis (Freitag, 1969)
Cyclotrachelus (Evarthrus) furtivus (LeConte, 1853)
Cyclotrachelus (Evarthrus) gigas (Casey, 1918)
Cyclotrachelus (Evarthrus) gravesi (Freitag, 1969)
Cyclotrachelus (Evarthrus) gravidus (Haldeman, 1853)
Cyclotrachelus (Evarthrus) heros (Say, 1823)
Cyclotrachelus (Evarthrus) hypherpiformis (Freitag, 1969)
Cyclotrachelus (Evarthrus) incisus (LeConte, 1846)
Cyclotrachelus (Evarthrus) iowensis (Freitag, 1969)
Cyclotrachelus (Evarthrus) lodingi (Van Dyke, 1926)
Cyclotrachelus (Evarthrus) nonnitens (LeConte, 1873)
Cyclotrachelus (Evarthrus) parasodalis (Freitag, 1969)
Cyclotrachelus (Evarthrus) sallei (LeConte, 1873)
Cyclotrachelus (Evarthrus) seximpressus (LeConte, 1846)
Cyclotrachelus (Evarthrus) sigillatus (Say, 1823)
Cyclotrachelus (Evarthrus) sinus (Freitag, 1969)
Cyclotrachelus (Evarthrus) sodalis colossus (LeConte, 1846)
Cyclotrachelus (Evarthrus) sodalis sodalis (LeConte, 1846)
Cyclotrachelus (Evarthrus) substriatus (LeConte, 1846)
Cyclotrachelus (Evarthrus) torvus (LeConte, 1863)
Cyclotrachelus (Evarthrus) whitcombi (Freitag, 1969)
Abax (Abax) parallelepipedus (Piller & Mitterpacher, 1783)†
ZabriniAmara (Curtonotus) alpina (Paykull, l790)‡
Amara (Curtonotus) aulica (Panzer, 1796)†
Amara (Curtonotus) blanchardi Hayward, 1908
Amara (Curtonotus) bokori Csiki, 1929‡
Amara (Curtonotus) carinata (LeConte, 1847)
Amara (Curtonotus) daurica (Motschulsky, 1844)‡
Amara (Curtonotus) deparca (Say, 1830)
Amara (Curtonotus) hyperborea Dejean, 1831‡
Amara (Curtonotus) jacobina LeConte, 1855
Amara (Curtonotus) kurnakowi Hieke, 1994‡
Amara (Curtonotus) lacustris LeConte, 1855
Amara (Curtonotus) pennsylvanica Hayward, 1908
Amara (Curtonotus) pterostichina Hayward, 1908
Amara (Curtonotus) thoracica Hayward, 1908
Amara (Curtonotus) torrida (Panzer, 1796)‡
Amara (Bradytus) apricaria (Paykull, 1790)†
Amara (Bradytus) avida (Say, 1823)
Amara (Bradytus) browni Lindroth, 1968
Amara (Bradytus) exarata Dejean, 1828
Amara (Bradytus) fulva (Müller, 1776)†
Amara (Bradytus) glacialis (Mannerheim, 1853)‡
Amara (Bradytus) insignis Dejean, 1831
Amara (Bradytus) insularis Horn, 1875
Amara (Bradytus) latior (Kirby, 1837)
Amara (Bradytus) lindrothi Hieke, 1990
Amara (Bradytus) neomexicana (Casey, 1924)
Amara (Bradytus) schwarzi Hayward, 1908
Amara (Neopercosia) fortis LeConte, 1880
Amara (Percosia) obesa (Say, 1823)
Amara (Xenocelia) apachensis Casey, 1884
Amara (Xenocelia) bradytonota Hieke, 2001
Amara (Xenocelia) chalcea Dejean, 1828
Amara (Xenocelia) discors Kirby, 1837
Amara (Xenocelia) gibba (LeConte, 1847)
Amara (Xenocelia) harpalonota Hieke, 2001
Amara (Xenocelia) hicksi Lindroth, 1968‡
Amara (Xenocelia) lugubris (Casey, 1918)
Amara (Xenocelia) merula (Casey, 1918)
Amara (Xenocelia) rectangula ciudadensis (Bates, 1891)
Amara (Xenocelia) rectangula rectangula LeConte, 1855
Amara (Xenocelia) spuria Lindroth, 1968
Amara (Reductocelia) colvillensis Lindroth, 1968‡
Amara (Celia) bifrons (Gyllenhal, 1810)†
Amara (Celia) brunnea (Gyllenhal, 1810)‡
Amara (Celia) californica californica Dejean, 1828
Amara (Celia) exlineae Minsk & Hatch, 1939
Amara (Celia) harpalina LeConte, 1855
Amara (Celia) idahoana (Casey, 1924)
Amara (Celia) musculis (Say, 1823)
Amara (Celia) pseudobrunnea Lindroth, 1968
Amara (Celia) rubrica Haldeman, 1843
Amara (Celia) sinuosa (Casey, 1918)
Amara (Celia) texana (Putzeys, 1866)
Amara (Celia) volatilis (Casey, 1918)
Amara (Amarocelia) ellipsis (Casey, 1918)
Amara (Amarocelia) erratica (Duftschmid, 1812)‡
Amara (Amarocelia) farcta LeConte, 1855
Amara (Amarocelia) interstitialis Dejean, 1828‡
Amara (Amarocelia) laevipennis Kirby, 1837
Amara (Amarocelia) lugens Zimmermann, 1832
Amara (Amarocelia) nexa (Casey, 1918)
Amara (Amarocelia) patruelis Dejean, 1831
Amara (Amarocelia) rugulifera Hieke, 2002
Amara (Amarocelia) sodalicia Casey, 1924
Amara (Amarocelia) tenebrionella (Bates, 1882)
Amara (Amarocelia) transberingiensis Hieke, 2002‡
Amara (Amara) aenea (DeGeer, 1774)†
Amara (Amara) aeneopolita Casey, 1918
Amara (Amara) anthobia Villa & Villa, 1833†
Amara (Amara) aurata Dejean, 1828
Amara (Amara) basillaris (Say, 1823)
Amara (Amara) coelebs Hayward, 1908
Amara (Amara) communis (Panzer, 1797)†
Amara (Amara) conflata LeConte, 1855
Amara (Amara) confusa LeConte, 1847
Amara (Amara) convexa LeConte, 1847
Amara (Amara) crassispina LeConte, 1855
Amara (Amara) cupreolata Putzeys, 1866
Amara (Amara) emancipata Lindroth, 1968
Amara (Amara) eurynota (Panzer, 1796)†
Amara (Amara) externefoveata Hieke, 2002
Amara (Amara) familiaris (Duftschmid, 1812)†
Amara (Amara) haywardi Csiki, 1929
Amara (Amara) impuncticollis (Say, 1823)
Amara (Amara) littoralis Dejean, 1828‡
Amara (Amara) lunicollis Schiødte, 1837‡
Amara (Amara) neoscotica Casey, 1924
Amara (Amara) occidentalis Hieke, 2002
Amara (Amara) otiosa Casey, 1918
Amara (Amara) ovata (Fabricius, 1792)†
Amara (Amara) pomona Casey, 1918
Amara (Amara) sanjuanensis Hatch, 1949
Amara (Amara) sera Say, 1830
Amara (Amara) tenax Casey, 1918
Amara (Amara) turbata Casey, 1918
Amara (Paracelia) quenseli quenseli (Schönherr, 1806)‡
Amara (Zezea) angustata (Say, 1823)
Amara (Zezea) angustatoides Hieke, 2000
Amara (Zezea) belfragei Horn, 1892
Amara (Zezea) flebilis (Casey, 1918)
Amara (Zezea) inexspectata Hieke, 1990
Amara (Zezea) kavanaughi Hieke, 1990
Amara (Zezea) longula LeConte, 1855
Amara (Zezea) pallipes Kirby, 1837
Amara (Zezea) scitula Zimmermann, 1832
OodiniDercylinus impressus (LeConte, 1853)
Evolenes exarata (Dejean, 1831)
Anatrichis minuta (Dejean, 1831)
Anatrichis oblonga Horn, 1891
Oodinus alutaceus (Bates, 1882)
Oodinus pseudopiceus Bousquet, 1996
Lachnocrepis parallela (Say, 1830)
Oodes amaroides Dejean, 1831
Oodes americanus Dejean, 1826
Oodes brevis Lindroth, 1957
Oodes fluvialis LeConte, 1863
Stenocrepis (Stenocrepis) insulana (Jacquelin du Val, 1857)
Stenocrepis (Stenous) cuprea (Chaudoir, 1843)
Stenocrepis (Stenous) duodecimstriata (Chevrolat, 1836)
Stenocrepis (Stenous) elegans (LeConte, 1851)
Stenocrepis (Stenous) mexicana (Chevrolat, 1835)
Stenocrepis (Stenous) tibialis (Chevrolat, 1834)
PanagaeiniPanagaeus (Hologaeus) cruciger Say, 1823
Panagaeus (Hologaeus) fasciatus Say, 1823
Panagaeus (Hologaeus) sallei Chaudoir, 1862
Micrixys distincta (Haldeman, 1852)
ChlaeniiniChlaenius (Pseudanomoglossus) maxillosus Horn, 1876
Chlaenius (Eurydactylus) pimalicus Casey, 1914
Chlaenius (Eurydactylus) tomentosus (Say, 1823)
Chlaenius (Anomoglossus) amoenus Dejean, 1831
Chlaenius (Anomoglossus) emarginatus Say, 1823
Chlaenius (Anomoglossus) pusillus Say, 1823
Chlaenius (Chlaenius) aestivus Say, 1823
Chlaenius (Chlaenius) augustus Newman, 1838
Chlaenius (Chlaenius) azurescens Chaudoir, 1876
Chlaenius (Chlaenius) chaudoiri Horn, 1876
Chlaenius (Chlaenius) cumatilis LeConte, 1851
Chlaenius (Chlaenius) erythropus Germar, 1824
Chlaenius (Chlaenius) fuscicornis Dejean, 1831
Chlaenius (Chlaenius) laticollis Say, 1823
Chlaenius (Chlaenius) orbus Horn, 1871
Chlaenius (Chlaenius) patruelis LeConte, 1844
Chlaenius (Chlaenius) platyderus Chaudoir, 1856
Chlaenius (Chlaenius) sericeus (Forster, 1771)
Chlaenius (Chlaenius) sparsus LeConte, 1863
Chlaenius (Chlaenius) viduus Horn, 1871
Chlaenius (Lithochlaenius) cordicollis Kirby, 1837
Chlaenius (Lithochlaenius) leucoscelis monachus LeConte, 1851
Chlaenius (Lithochlaenius) leucoscelis sanantonialis Casey, 1914
Chlaenius (Lithochlaenius) leucoscelis sonomae Casey, 1920
Chlaenius (Lithochlaenius) prasinus Dejean, 1826
Chlaenius (Lithochlaenius) purpureus Chaudoir, 1876
Chlaenius (Lithochlaenius) solitarius Say, 1823
Chlaenius (Chlaeniellus) brevilabris LeConte, 1847
Chlaenius (Chlaeniellus) circumcinctus Say, 1830
Chlaenius (Chlaeniellus) flaccidus Horn, 1876
Chlaenius (Chlaeniellus) floridanus Horn, 1876
Chlaenius (Chlaeniellus) glaucus LeConte, 1856
Chlaenius (Chlaeniellus) impunctifrons Say, 1823
Chlaenius (Chlaeniellus) nebraskensis LeConte, 1856
Chlaenius (Chlaeniellus) nemoralis Say, 1823
Chlaenius (Chlaeniellus) obsoletus LeConte, 1851
Chlaenius (Chlaeniellus) oxygonus Chaudoir, 1843
Chlaenius (Chlaeniellus) pennsylvanicus blanditus Casey, 1920
Chlaenius (Chlaeniellus) pennsylvanicus pennsylvanicus Say, 1823
Chlaenius (Chlaeniellus) pertinax Casey, 1920
Chlaenius (Chlaeniellus) simillimus Chaudoir, 1856
Chlaenius (Chlaeniellus) texanus Horn, 1876
Chlaenius (Chlaeniellus) tricolor tricolor Dejean, 1826
Chlaenius (Chlaeniellus) tricolor vigilans Say, 1830
Chlaenius (Chlaeniellus) vafer LeConte, 1852
Chlaenius (Chlaeniellus) variabilipes Eschscholtz, 1833
Chlaenius (Callistometus) ruficauda Chaudoir, 1856
Chlaenius (Brachylobus) caurinus (Horn, 1885)
Chlaenius (Brachylobus) lithophilus Say, 1823
Chlaenius (Agostenus) alternatus Horn, 1871
Chlaenius (Agostenus) caeruleicollis Chaudoir, 1876
Chlaenius (Agostenus) harpalinus Eschscholtz, 1833
Chlaenius (Agostenus) interruptus Horn, 1876
Chlaenius (Agostenus) niger Randall, 1838
Chlaenius (Randallius) purpuricollis Randall, 1838
LicininiDiplocheila (Isorembus) assimilis (LeConte, 1844)
Diplocheila (Isorembus) crossi Will, 1998
Diplocheila (Isorembus) impressicollis (Dejean, 1831)
Diplocheila (Isorembus) major major (LeConte, 1847)
Diplocheila (Isorembus) major melissisa Ball, 1959
Diplocheila (Isorembus) nupera Casey, 1897
Diplocheila (Isorembus) obtusa (LeConte, 1847)
Diplocheila (Isorembus) oregona (Hatch, 1951)
Diplocheila (Isorembus) striatopunctata (LeConte, 1844)
Diplocheila (Isorembus) undulata Carr, 1920
Dicaelus (Paradicaelus) ambiguus LaFerté-Sénectère, 1841
Dicaelus (Paradicaelus) dilatatus dilatatus Say, 1823
Dicaelus (Paradicaelus) dilatatus sinuatus Ball, 1959
Dicaelus (Paradicaelus) elongatus Bonelli, 1813
Dicaelus (Paradicaelus) furvus carinatus Dejean, 1831
Dicaelus (Paradicaelus) furvus furvus Dejean, 1826
Dicaelus (Paradicaelus) politus Dejean, 1826
Dicaelus (Paradicaelus) sculptilis intricatus LeConte, 1873
Dicaelus (Paradicaelus) sculptilis sculptilis Say, 1823
Dicaelus (Paradicaelus) sculptilis upioides Ball, 1959
Dicaelus (Paradicaelus) teter Bonelli, 1813
Dicaelus (Dicaelus) alternans Dejean, 1826
Dicaelus (Dicaelus) costatus LeConte, 1853
Dicaelus (Dicaelus) crenatus LeConte, 1853
Dicaelus (Dicaelus) purpuratus purpuratus Bonelli, 1813
Dicaelus (Dicaelus) purpuratus splendidus Say, 1823
Dicaelus (Dicaelus) quadratus LeConte, 1847
Dicaelus (Dicaelus) subtropicus Casey, 1913
Dicaelus (Liodicaelus) chermocki Ball, 1959
Dicaelus (Liodicaelus) laevipennis laevipennis LeConte, 1847
Dicaelus (Liodicaelus) suffusus (Casey, 1913)
Badister (Badister) elegans LeConte, 1880
Badister (Badister) ferrugineus Dejean, 1831
Badister (Badister) flavipes flavipes LeConte, 1853
Badister (Badister) maculatus LeConte, 1853
Badister (Badister) neopulchellus Lindroth, 1954
Badister (Badister) notatus Haldeman, 1843
Badister (Badister) obtusus LeConte, 1878
Badister (Badister) pulchellus LeConte, 1847
Badister (Baudia) grandiceps Casey, 1920
Badister (Baudia) micans LeConte, 1844
Badister (Baudia) parviceps Ball, 1959
Badister (Baudia) reflexus LeConte, 1880
Badister (Baudia) submarinus Motschulsky, 1859
Badister (Baudia) transversus Casey, 1920
HarpaliniNotiobia (Anisotarsus) brevicollis (Chaudoir, 1837)
Notiobia (Anisotarsus) cephala (Casey, 1914)
Notiobia (Anisotarsus) maculicornis (Chaudoir, 1843)
Notiobia (Anisotarsus) mexicana (Dejean, 1829)
Notiobia (Anisotarsus) nitidipennis (LeConte, 1847)
Notiobia (Anisotarsus) purpurascens (Bates, 1882)
Notiobia (Anisotarsus) sayi (Blatchley, 1910)
Notiobia (Anisotarsus) terminata (Say, 1823)
Xestonotus lugubris (Dejean, 1829)
Anisodactylus (Anisodactylus) agricola (Say, 1823)
Anisodactylus (Anisodactylus) binotatus (Fabricius, 1787)†
Anisodactylus (Anisodactylus) californicus Dejean, 1829
Anisodactylus (Anisodactylus) carbonarius (Say, 1823)
Anisodactylus (Anisodactylus) consobrinus LeConte, 1851
Anisodactylus (Anisodactylus) furvus LeConte, 1863
Anisodactylus (Anisodactylus) harrisii LeConte, 1863
Anisodactylus (Anisodactylus) kirbyi Lindroth, 1953
Anisodactylus (Anisodactylus) lodingi Schaeffer, 1911
Anisodactylus (Anisodactylus) melanopus (Haldeman, 1843)
Anisodactylus (Anisodactylus) nigerrimus (Dejean, 1831)
Anisodactylus (Anisodactylus) nigrita Dejean, 1829
Anisodactylus (Anisodactylus) pseudagricola Noonan, 1996
Anisodactylus (Anisodactylus) similis LeConte, 1851
Anisodactylus (Gynandrotarsus) anthracinus (Dejean, 1829)
Anisodactylus (Gynandrotarsus) dulcicollis (LaFerté-Sénectère, 1841)
Anisodactylus (Gynandrotarsus) haplomus Chaudoir, 1868
Anisodactylus (Gynandrotarsus) harpaloides (LaFerté-Sénectère, 1841)
Anisodactylus (Gynandrotarsus) merula (Germar, 1824)
Anisodactylus (Gynandrotarsus) opaculus (LeConte, 1863)
Anisodactylus (Gynandrotarsus) ovularis (Casey, 1914)
Anisodactylus (Gynandrotarsus) rusticus (Say, 1823)
Anisodactylus (Gynandrotarsus) texanus Schaeffer, 1910
Anisodactylus (Anadaptus) alternans (Motschulsky, 1845)
Anisodactylus (Anadaptus) discoideus Dejean, 1831
Anisodactylus (Anadaptus) pitychrous LeConte, 1861
Anisodactylus (Anadaptus) porosus (Motschulsky, 1845)
Anisodactylus (Anadaptus) rudis LeConte, 1863
Anisodactylus (Anadaptus) sanctaecrucis (Fabricius, 1798)
Anisodactylus (Spongopus) verticalis (LeConte, 1847)
Anisodactylus (Aplocentrus) amaroides LeConte, 1851
Anisodactylus (Aplocentrus) caenus (Say, 1823)
Anisodactylus (Pseudaplocentrus) laetus Dejean, 1829
Geopinus incrassatus (Dejean, 1829)
Amphasia (Pseudamphasia) sericea (Harris, 1828)
Amphasia (Amphasia) interstitialis (Say, 1823)
Dicheirus brunneus (Dejean, 1829)
Dicheirus dilatatus angulatus Casey, 1914
Dicheirus dilatatus dilatatus (Dejean, 1829)
Dicheirus obtusus LeConte, 1852
Dicheirus piceus (Ménétriés, 1843)
Dicheirus strenuus (Horn, 1869)
Pelmatellus (Pelmatellus) obtusus Bates, 1882
Pelmatellus (Pelmatellus) stenolophoides parallelus Goulet, 1974
Stenolophus (Stenolophus) anceps LeConte, 1857
Stenolophus (Stenolophus) carbo Bousquet, 1993
Stenolophus (Stenolophus) cincticollis LeConte, 1858
Stenolophus (Stenolophus) dissimilis Dejean, 1829
Stenolophus (Stenolophus) flavipes LeConte, 1858
Stenolophus (Stenolophus) fuliginosus Dejean, 1829
Stenolophus (Stenolophus) fuscatus Dejean, 1829
Stenolophus (Stenolophus) humidus Hamilton, 1893
Stenolophus (Stenolophus) incultus Casey, 1914
Stenolophus (Stenolophus) limbalis LeConte, 1857
Stenolophus (Stenolophus) megacephalus Lindroth, 1968
Stenolophus (Stenolophus) ochropezus (Say, 1823)
Stenolophus (Stenolophus) plebejus Dejean, 1829
Stenolophus (Stenolophus) splendidulus Motschulsky, 1864
Stenolophus (Stenolophus) spretus Dejean, 1831
Stenolophus (Agonoderus) binotatus (Casey, 1914)
Stenolophus (Agonoderus) comma (Fabricius, 1775)
Stenolophus (Agonoderus) infuscatus (Dejean, 1829)
Stenolophus (Agonoderus) lecontei (Chaudoir, 1868)
Stenolophus (Agonoderus) lineola (Fabricius, 1775)
Stenolophus (Agonoderus) maculatus (LeConte, 1869)
Stenolophus (Agonoderus) rugicollis (LeConte, 1859)
Agonoleptus conjunctus (Say, 1823)
Agonoleptus dolosus (Casey, 1914)
Agonoleptus parviceps Casey, 1914
Agonoleptus rotundatus (LeConte, 1863)
Agonoleptus rotundicollis (Haldeman, 1843)
Agonoleptus thoracicus (Casey, 1914)
Agonoleptus unicolor (Dejean, 1829)
Bradycellus (Liocellus) curticollis (Casey, 1924)
Bradycellus (Liocellus) intermedius (Fall, 1905)
Bradycellus (Liocellus) laticollis (Casey, 1924)
Bradycellus (Liocellus) nitidus (Dejean, 1829)
Bradycellus (Liocellus) obtusus (Fall, 1905)
Bradycellus (Liocellus) politus (Fall, 1905)
Bradycellus (Liocellus) tahoensis (Casey, 1924)
Bradycellus (Bradycellus) fenderi Hatch, 1951
Bradycellus (Bradycellus) harpalinus (Audinet-Serville, 1821)†
Bradycellus (Catharellus) lecontei Csiki, 1932
Bradycellus (Stenocellus) ardelio (Casey, 1914)
Bradycellus (Stenocellus) aridus (Casey, 1914)
Bradycellus (Stenocellus) californicus (LeConte, 1857)
Bradycellus (Stenocellus) carolinensis (Casey, 1924)
Bradycellus (Stenocellus) congener (LeConte, 1847)
Bradycellus (Stenocellus) decorus (Casey, 1914)
Bradycellus (Stenocellus) discipulus (Casey, 1914)
Bradycellus (Stenocellus) exstans (Casey, 1914)
Bradycellus (Stenocellus) festinans (Casey, 1914)
Bradycellus (Stenocellus) humboldtianus (Casey, 1924)
Bradycellus (Stenocellus) insulsus (Casey, 1914)
Bradycellus (Stenocellus) larvatus (Casey, 1914)
Bradycellus (Stenocellus) lineatus (Casey, 1914)
Bradycellus (Stenocellus) lustrellus (Casey, 1914)
Bradycellus (Stenocellus) montanus (Casey, 1914)
Bradycellus (Stenocellus) nebulosus LeConte, 1853
Bradycellus (Stenocellus) neglectus (LeConte, 1847)
Bradycellus (Stenocellus) nigerrimus Lindroth, 1968
Bradycellus (Stenocellus) nigriceps LeConte, 1869
Bradycellus (Stenocellus) nubifer LeConte, 1858
Bradycellus (Stenocellus) picipes (Casey, 1914)
Bradycellus (Stenocellus) provoensis (Casey, 1914)
Bradycellus (Stenocellus) puncticollis (Casey, 1914)
Bradycellus (Stenocellus) purgatus (Casey, 1914)
Bradycellus (Stenocellus) rivalis LeConte, 1858
Bradycellus (Stenocellus) rupestris (Say, 1823)
Bradycellus (Stenocellus) sejunctus (Casey, 1914)
Bradycellus (Stenocellus) suavis (Casey, 1914)
Bradycellus (Stenocellus) subcordatus Chaudoir, 1868
Bradycellus (Stenocellus) supplex (Casey, 1914)
Bradycellus (Stenocellus) symetricus (Motschulsky, 1850)
Bradycellus (Stenocellus) tantillus (Dejean, 1829)
Bradycellus (Stenocellus) veronianus (Casey, 1924)
Bradycellus (Lipalocellus) nigrinus (Dejean, 1829)
Bradycellus (Lipalocellus) semipubescens Lindroth, 1968
Bradycellus (Triliarthrus) atrimedeus (Say, 1823)
Bradycellus (Triliarthrus) badipennis (Haldeman, 1843)
Bradycellus (Triliarthrus) conformis (Fall, 1905)
Bradycellus (Triliarthrus) georgei Lindroth, 1968
Bradycellus (Triliarthrus) kirbyi (Horn, 1883)
Bradycellus (Triliarthrus) lugubris (LeConte, 1847)
Amerinus linearis (LeConte, 1863)
Dicheirotrichus (Oreoxenus) mannerheimii mannerheimii (Sahlberg, 1844)‡
Dicheirotrichus (Trichocellus) cognatus (Gyllenhal, 1827)‡
Acupalpus (Acupalpus) canadensis Casey, 1924
Acupalpus (Acupalpus) carus (LeConte, 1863)
Acupalpus (Acupalpus) hydropicus (LeConte, 1863)
Acupalpus (Acupalpus) meridianus (Linnaeus, 1760)†
Acupalpus (Acupalpus) nanellus Casey, 1914
Acupalpus (Acupalpus) pumilus Lindroth, 1968
Acupalpus (Tachistodes) indistinctus Dejean, 1831
Acupalpus (Tachistodes) partiarius (Say, 1823)
Acupalpus (Tachistodes) pauperculus Dejean, 1829
Acupalpus (Tachistodes) testaceus Dejean, 1829
Acupalpus (Anthracus) punctulatus Hatch, 1953
Acupalpus (Anthracus) tener (LeConte, 1857)
Philodes (Philodes) alternans (LeConte, 1853)
Philodes (Goniolophus) flavilimbus (LeConte, 1869)
Philodes (Goniolophus) longulus (Dejean, 1829)
Philodes (Goniolophus) rectangulus (Chaudoir, 1868)
Pogonodaptus mexicanus (Bates, 1878)
Polpochila (Phymatocephalus) capitata (Chaudoir, 1852)
Polpochila (Phymatocephalus) erro (LeConte, 1854)
Polpochila (Polpochila) rotundicollis Bates, 1882
Piosoma setosum LeConte, 1847
Euryderus grossus (Say, 1830)
Ophonus (Metophonus) puncticeps Stephens, 1828 †
Ophonus (Metophonus) rufibarbis (Fabricius, 1792)†
Harpalus (Pseudoophonus) actiosus Casey, 1914
Harpalus (Pseudoophonus) compar LeConte, 1847
Harpalus (Pseudoophonus) erythropus Dejean, 1829
Harpalus (Pseudoophonus) faunus Say, 1823
Harpalus (Pseudoophonus) hatchi Ball & Anderson, 1962
Harpalus (Pseudoophonus) liobasis Chaudoir, 1868
Harpalus (Pseudoophonus) paratus Casey, 1924
Harpalus (Pseudoophonus) pensylvanicus (DeGeer, 1774)
Harpalus (Pseudoophonus) poncei Will, 2002
Harpalus (Pseudoophonus) protractus Casey, 1914
Harpalus (Pseudoophonus) rufipes (DeGeer, 1774)†
Harpalus (Pseudoophonus) texanus Casey, 1914
Harpalus (Pseudoophonus) vagans LeConte, 1865
Harpalus (Megapangus) caliginosus (Fabricius, 1775)
Harpalus (Megapangus) katiae Battoni, 1985
Harpalus (Plectralidus) erraticus Say, 1823
Harpalus (Plectralidus) retractus LeConte, 1863
Harpalus (Opadius) animosus Casey, 1924
Harpalus (Opadius) apache Kataev, 2010
Harpalus (Opadius) cordatus (LeConte, 1853)
Harpalus (Opadius) cordifer Notman, 1919
Harpalus (Opadius) desertus LeConte, 1859
Harpalus (Opadius) fraternus LeConte, 1852
Harpalus (Opadius) fulvilabris Mannerheim, 1853
Harpalus (Opadius) gravis LeConte, 1858
Harpalus (Opadius) indianus Csiki, 1932
Harpalus (Opadius) indigens Casey, 1924
Harpalus (Opadius) laevipes Zetterstedt, 1828‡
Harpalus (Opadius) laticeps LeConte, 1850
Harpalus (Opadius) lewisii LeConte, 1865
Harpalus (Opadius) megacephalus LeConte, 1847
Harpalus (Opadius) nigritarsis Sahlberg, 1827‡
Harpalus (Opadius) providens Casey, 1914
Harpalus (Opadius) reversus Casey, 1924
Harpalus (Opadius) spadiceus Dejean, 1829
Harpalus (Opadius) ventralis LeConte, 1847
Harpalus (Harpalus) affinis (Schrank, 1781)†
Harpalus (Harpalus) amputatus amputatus Say, 1830
Harpalus (Harpalus) atrichatus Hatch, 1949
Harpalus (Harpalus) balli Noonan, 1991
Harpalus (Harpalus) cautus Dejean, 1829
Harpalus (Harpalus) ellipsis LeConte, 1847
Harpalus (Harpalus) herbivagus Say, 1823
Harpalus (Harpalus) innocuus LeConte, 1863
Harpalus (Harpalus) martini Van Dyke, 1926
Harpalus (Harpalus) obnixus Casey, 1924
Harpalus (Harpalus) ochropus Kirby, 1837
Harpalus (Harpalus) opacipennis (Haldeman, 1843)
Harpalus (Harpalus) plenalis Casey, 1914
Harpalus (Harpalus) rubripes (Duftschmid, 1812)†
Harpalus (Harpalus) solitaris Dejean, 1829‡
Harpalus (Harpalus) somnulentus Dejean, 1829
Harpalus (Harpalus) vittatus alaskensis Lindroth, 1968‡
Harpalus (Glanodes) cohni Ball, 1972
Harpalus (Glanodes) corpulentus (Casey, 1914)
Harpalus (Glanodes) huachuca Ball, 1972
Harpalus (Glanodes) obliquus Horn, 1880
Harpalus (Glanodes) puncticeps (Casey, 1914)
Harpalus (Glanodes) stephani Ball, 1972
Harpalus (Harpalobius) fuscipalpis Sturm, 1818‡
Harpalobrachys leiroides (Motschulsky, 1844)‡
Hartonymus alternatus (LeConte, 1863)
Hartonymus hoodi Casey, 1914
Amblygnathus evansi Ball & Maddison, 1987
Amblygnathus iripennis (Say, 1823)
Amblygnathus mexicanus Bates, 1882
Amblygnathus subtinctus (LeConte, 1867)
Athrostictus punctatulus (Putzeys, 1878)
Selenophorus (Celiamorphus) adjunctus (Casey, 1914)
Selenophorus (Celiamorphus) contractus (Casey, 1914)
Selenophorus (Celiamorphus) discopunctatus Dejean, 1829
Selenophorus (Celiamorphus) ellipticus Dejean, 1829
Selenophorus (Celiamorphus) fossulatus Dejean, 1829
Selenophorus (Celiamorphus) granarius Dejean, 1829
Selenophorus (Celiamorphus) municeps (Casey, 1924)
Selenophorus (Celiamorphus) nanulus (Casey, 1924)
Selenophorus (Celiamorphus) subtropicus (Casey, 1924)
Selenophorus (Selenophorus) aeneopiceus Casey, 1884
Selenophorus (Selenophorus) blanchardi Manee, 1915
Selenophorus (Selenophorus) chaparralus Purrington, 2000
Selenophorus (Selenophorus) concinnus Schaeffer, 1910
Selenophorus (Selenophorus) cupreolus Casey, 1914
Selenophorus (Selenophorus) discoderoides Schaeffer, 1910
Selenophorus (Selenophorus) elongatus (LeConte, 1847)
Selenophorus (Selenophorus) famulus Casey, 1914
Selenophorus (Selenophorus) fatuus LeConte, 1863
Selenophorus (Selenophorus) gagatinus Dejean, 1829
Selenophorus (Selenophorus) houstoni Casey, 1914
Selenophorus (Selenophorus) hylacis (Say, 1823)
Selenophorus (Selenophorus) implicans Casey, 1914
Selenophorus (Selenophorus) integer (Fabricius, 1798)
Selenophorus (Selenophorus) laesus (LeConte, 1858)
Selenophorus (Selenophorus) maritimus Casey, 1914
Selenophorus (Selenophorus) opalinus (LeConte, 1863)
Selenophorus (Selenophorus) otiosus Casey, 1914
Selenophorus (Selenophorus) palliatus (Fabricius, 1798)
Selenophorus (Selenophorus) parumpunctatus Dejean, 1829
Selenophorus (Selenophorus) pedicularius Dejean, 1829
Selenophorus (Selenophorus) planipennis LeConte, 1847
Selenophorus (Selenophorus) riparius Casey, 1914
Selenophorus (Selenophorus) schaefferi Csiki, 1932
Selenophorus (Selenophorus) scolopaceus Casey, 1914
Selenophorus (Selenophorus) sinuaticollis Notman, 1922
Selenophorus (Selenophorus) striatopunctatus Putzeys, 1878
Selenophorus (Selenophorus) trepidus (Casey, 1924)
Selenophorus breviusculus Horn, 1880
Discoderus aequalis Casey, 1914
Discoderus amoenus LeConte, 1863
Discoderus congruens Casey, 1914
Discoderus cordicollis Horn, 1891
Discoderus crassicollis Horn, 1891
Discoderus dallasensis Casey, 1924
Discoderus impotens (LeConte, 1858)
Discoderus longicollis Casey, 1914
Discoderus obsidianus Casey, 1914
Discoderus papagonis Casey, 1924
Discoderus parallelus (Haldeman, 1843)
Discoderus parilis (Casey, 1914)
Discoderus peregrinus Casey, 1924
Discoderus pinguis Casey, 1884
Discoderus robustus piceus Casey, 1914
Discoderus robustus robustus Horn, 1883
Discoderus subviolaceus Casey, 1914
Discoderus symbolicus Casey, 1914
Discoderus tenebrosus (LeConte, 1847)
Discoderus texanus Casey, 1924
Stenomorphus californicus californicus (Ménétriés, 1843)
Stenomorphus californicus rufipes LeConte, 1858
Stenomorphus convexior Notman, 1922
Stenomorphus sinaloae Darlington, 1936
Trichotichnus (Trichotichnus) dichrous (Dejean, 1829)
Trichotichnus (Trichotichnus) vulpeculus (Say, 1823)
Trichotichnus (Iridessus) autumnalis (Say, 1823)
Trichotichnus (Iridessus) fulgens (Csiki, 1932)
Aztecarpalus schaefferi Ball, 1970
Cratacanthus dubius (Palisot de Beauvois, 1811)
SphodriniPseudamara arenaria (LeConte, 1847)
Calathus (Calathus) fuscipes (Goeze, 1777)†
Calathus (Neocalathus) calceus Ball & Nègre, 1972
Calathus (Neocalathus) gregarius (Say, 1823)
Calathus (Neocalathus) ingratus Dejean, 1828
Calathus (Neocalathus) opaculus LeConte, 1854
Calathus (Neocalathus) peropacus Casey, 1920
Calathus (Neocalathus) ruficollis grandicollis Casey, 1920
Calathus (Neocalathus) ruficollis ignicollis Casey, 1920
Calathus (Neocalathus) ruficollis ruficollis Dejean, 1828
Calathus (Acalathus) advena (LeConte, 1846)
Synuchus dubius (LeConte, 1854)
Synuchus impunctatus (Say, 1823)
Laemostenus (Laemostenus) complanatus (Dejean, 1828)†
Laemostenus (Pristonychus) terricola terricola (Herbst, 1784)†
PlatyniniOlisthopus brevicornis Casey, 1913
Olisthopus filicornis Casey, 1913
Olisthopus innuens Casey, 1913
Olisthopus iterans Casey, 1913
Olisthopus micans LeConte, 1846
Olisthopus parmatus (Say, 1823)
Olisthopus pusio Casey, 1913
Elliptoleus acutesculptus Bates, 1882
Sericoda bembidioides Kirby, 1837
Sericoda bogemannii (Gyllenhal, 1813)‡
Sericoda obsoleta (Say, 1823)
Sericoda quadripunctata (DeGeer, 1774)‡
Tetraleucus picticornis (Newman, 1844)
Anchomenus (Anchomenus) aeneolus (LeConte, 1854)
Anchomenus (Anchomenus) funebris (LeConte, 1854)
Anchomenus (Anchomenus) quadratus (LeConte, 1854)
Rhadine albamontana Dajoz, 1998
Rhadine anthicoides Casey, 1913
Rhadine austinica Barr, 1974
Rhadine babcocki (Barr, 1960)
Rhadine balesi (Gray, 1937)
Rhadine bullis Reddell & Cokendolpher, 2004
Rhadine caudata (LeConte, 1863)
Rhadine constricta Casey, 1913
Rhadine dissecta (LeConte, 1863)
Rhadine exilis (Barr & Lawrence, 1960)
Rhadine grubbsi Reddell & Dupérré, 2009
Rhadine howdeni (Barr & Lawrence, 1960)
Rhadine infernalis ewersi (Barr, 1960)
Rhadine infernalis infernalis (Barr & Lawrence, 1960)
Rhadine insolita Barr, 1974
Rhadine ivyi Reddell & Cokendolpher, 2004
Rhadine jejuna (LeConte, 1878)
Rhadine koepkei koepkei (Barr, 1960)
Rhadine koepkei privata Barr, 1974
Rhadine lanei (Gray, 1937)
Rhadine larvalis LeConte, 1846
Rhadine lindrothi Barr, 1965
Rhadine longiceps Van Dyke, 1949
Rhadine longicollis Benedict, 1927
Rhadine longipes Casey, 1913
Rhadine myrmecodes (Horn, 1892)
Rhadine nivalis (Horn, 1881)
Rhadine noctivaga Barr, 1974
Rhadine ozarkensis Sanderson & Miller, 1941
Rhadine perlevis Casey, 1913
Rhadine persephone Barr, 1974
Rhadine pertenuis Casey, 1920
Rhadine reyesi Reddell & Cokendolpher, 2001
Rhadine rossi Van Dyke, 1949
Rhadine rubra (Barr, 1960)
Rhadine russelli Barr, 1974
Rhadine specum crinicollis Barr, 1974
Rhadine specum gentilis Barr, 1974
Rhadine specum specum (Barr, 1960)
Rhadine sprousei Reddell & Cokendolpher, 2004
Rhadine sublustris Casey, 1913
Rhadine subterranea mitchelli Barr, 1974
Rhadine subterranea subterranea (Van Dyke, 1919)
Rhadine tenebrosa mckenziei Barr, 1974
Rhadine tenebrosa tenebrosa (Barr, 1960)
Rhadine testacea Casey, 1920
Rhadine umbra Casey, 1913
Mexisphodrus valverdensis Barr, 1982
Tanystoma cuyama Liebherr, 1985
Tanystoma maculicolle (Dejean, 1828)
Tanystoma striatum (Dejean, 1828)
Tanystoma sulcatum (Dejean, 1828)
Paranchus albipes (Fabricius, 1794)†
Oxypselaphus pusillus (LeConte, 1854)
Agonum (Platynomicrus) ferruginosum (Dejean, 1828)
Agonum (Platynomicrus) nigriceps LeConte, 1846‡
Agonum (Europhilus) anchomenoides Randall, 1838
Agonum (Europhilus) canadense Goulet, 1969
Agonum (Europhilus) consimile (Gyllenhal, 1810)‡
Agonum (Europhilus) darlingtoni Lindroth, 1954
Agonum (Europhilus) exaratum (Mannerheim, 1853)‡
Agonum (Europhilus) galvestonicum (Casey, 1920)
Agonum (Europhilus) gratiosum (Mannerheim, 1853)‡
Agonum (Europhilus) limbatum Motschulsky, 1845
Agonum (Europhilus) lutulentum (LeConte, 1854)
Agonum (Europhilus) palustre Goulet, 1969
Agonum (Europhilus) picicornoides Lindroth, 1966
Agonum (Europhilus) retractum LeConte, 1846
Agonum (Europhilus) simile Kirby, 1837
Agonum (Europhilus) sordens Kirby, 1837
Agonum (Europhilus) superioris Lindroth, 1966
Agonum (Europhilus) thoreyi Dejean, 1828‡
Agonum (Agonum) bicolor (Dejean, 1828)‡
Agonum (Agonum) muelleri (Herbst, 1784)†
Agonum (Agonum) piceolum (LeConte, 1879)
Agonum (Agonum) placidum (Say, 1823)
Agonum (Olisares) aeruginosum Dejean, 1828
Agonum (Olisares) affine Kirby, 1837
Agonum (Olisares) albicrus Dejean, 1828
Agonum (Olisares) anthracinum Dejean, 1831
Agonum (Olisares) basale LeConte, 1846
Agonum (Olisares) belleri (Hatch, 1933)
Agonum (Olisares) brevicolle Dejean, 1828
Agonum (Olisares) collare (Say, 1830)
Agonum (Olisares) corvus (LeConte, 1860)
Agonum (Olisares) crenistriatum (LeConte, 1863)
Agonum (Olisares) crenulatum (LeConte, 1854)
Agonum (Olisares) cupreum Dejean, 1831
Agonum (Olisares) cupripenne (Say, 1823)
Agonum (Olisares) cyanopis (Bates, 1882)
Agonum (Olisares) cyclifer (Bates, 1884)
Agonum (Olisares) deceptivum (LeConte, 1879)
Agonum (Olisares) decorum (Say, 1823)
Agonum (Olisares) deplanatum Ménétriés, 1843
Agonum (Olisares) elongatulum (Dejean, 1828)
Agonum (Olisares) errans (Say, 1823)
Agonum (Olisares) excavatum Dejean, 1828
Agonum (Olisares) extensicolle (Say, 1823)
Agonum (Olisares) extimum Liebherr, 1986
Agonum (Olisares) ferreum Haldeman, 1843
Agonum (Olisares) fidele Casey, 1920
Agonum (Olisares) fossiger Dejean, 1828
Agonum (Olisares) harrisii LeConte, 1846
Agonum (Olisares) imitans (Notman, 1919)
Agonum (Olisares) melanarium Dejean, 1828
Agonum (Olisares) metallescens (LeConte, 1854)
Agonum (Olisares) moerens Dejean, 1828
Agonum (Olisares) muiri Liebherr, 1984
Agonum (Olisares) mutatum (Gemminger & Harold, 1868)
Agonum (Olisares) nutans (Say, 1823)
Agonum (Olisares) octopunctatum (Fabricius, 1798)
Agonum (Olisares) pacificum Casey, 1920
Agonum (Olisares) pallipes (Fabricius, 1787)
Agonum (Olisares) parextimum Liebherr, 1986
Agonum (Olisares) propinquum (Gemminger & Harold, 1868)
Agonum (Olisares) punctiforme (Say, 1823)
Agonum (Olisares) quadrimaculatum (Horn, 1885)
Agonum (Olisares) quinquepunctatum Motschulsky, 1844‡
Agonum (Olisares) rigidulum (Casey, 1920)
Agonum (Olisares) rufipes Dejean, 1828
Agonum (Olisares) striatopunctatum Dejean, 1828
Agonum (Olisares) sulcipenne (Horn, 1881)
Agonum (Olisares) suturale Say, 1830
Agonum (Olisares) tenue (LeConte, 1854)
Agonum (Olisares) texanum (LeConte, 1878)
Agonum (Olisares) trigeminum Lindroth, 1954
Platynus (Microplatynus) agilis LeConte, 1863
Platynus (Microplatynus) pecki Barr, 1982
Platynus (Platynus) brunneomarginatus (Mannerheim, 1843)
Platynus (Platynus) daviesi Bousquet, 2012
Platynus (Platynus) decentis (Say, 1823)
Platynus (Platynus) indecentis Liebherr & Will, 1996
Platynus (Platynus) opaculus LeConte, 1863
Platynus (Platynus) ovipennis (Mannerheim, 1843)
Platynus (Platynus) parmarginatus Hamilton, 1893
Platynus (Platynus) tenuicollis (LeConte, 1846)
Platynus (Platynus) trifoveolatus Beutenmüller, 1903
Platynus (Batenus) angustatus Dejean, 1828
Platynus (Batenus) cincticollis (Say, 1823)
Platynus (Batenus) hypolithos (Say, 1823)
Platynus (Batenus) mannerheimii (Dejean, 1828)‡
Platynus (Batenus) prognathus Van Dyke, 1926
Platynus (Glyptolenopsis) ovatulus (Bates, 1884)
Platynus (Trapezodera) cohni Liebherr & Will, 1996
Platynus (Dyscolus) cazieri Liebherr & Will, 1996
Platynus (Dyscolus) falli (Darlington, 1936)
Platynus (Dyscolus) lyratus (Chaudoir, 1879)
Platynus (Dyscolus) megalops (Bates, 1882)
Platynus (Dyscolus) rufiventris (Van Dyke, 1926)
Metacolpodes buchanani (Hope, 1831)†
PerigoniniPerigona (Trechicus) nigriceps (Dejean, 1831)†
Perigona (Trechicus) pallipennis (LeConte, 1853)
AtraniniAtranus pubescens (Dejean, 1828)
LachnophoriniAnchonoderus quadrinotatus Horn, 1878
Anchonoderus schaefferi Liebke, 1928
Lachnophorus elegantulus Mannerheim, 1843
Euphorticus occidentalis Horn, 1891
Euphorticus pubescens (Dejean, 1831)
Calybe (Ega) laetula (LeConte, 1851)
Calybe (Ega) sallei (Chevrolat, 1839)
Eucaerus (Eucaerus) varicornis LeConte, 1853
PentagoniciniPentagonica bicolor (LeConte, 1863)
Pentagonica felix Bell, 1987
Pentagonica flavipes flavipes (LeConte, 1853)
Pentagonica marshalli Mateu, 1995
Pentagonica nigricornis Darlington, 1934
Pentagonica picticornis Bates, 1883
OdacanthiniColliuris (Mimocasnonia) pilatei (Chaudoir, 1848)
Colliuris (Cosnania) lengi (Schaeffer, 1910)
Colliuris (Cosnania) pensylvanica (Linnaeus, 1758)
Colliuris (Calocolliuris) caymanensis Darlington, 1947
Colliuris (Calocolliuris) lioptera (Bates, 1891)
Colliuris (Calocolliuris) ludoviciana (Sallé, 1849)
CtenodactyliniLeptotrachelus depressus Blatchley, 1923
Leptotrachelus dorsalis (Fabricius, 1801)
Leptotrachelus pallidulus Motschulsky, 1864
CyclosominiTetragonoderus (Crossonychus) fasciatus (Haldeman, 1843)
Tetragonoderus (Crossonychus) intersectus (Germar, 1824)
Tetragonoderus (Crossonychus) laevigatus Chaudoir, 1876†
Tetragonoderus (Crossonychus) latipennis LeConte, 1874
Tetragonoderus (Crossonychus) pallidus Horn, 1869
LebiiniMochtherus tetraspilotus (Macleay, 1825)†
Phloeoxena (Oenaphelox) signata (Dejean, 1825)
Eucheila (Inna) boyeri (Solier, 1835)
Somotrichus unifasciatus (Dejean, 1831)†
Coptodera (Coptodera) aerata Dejean, 1825
Coptodera (Coptodera) brunnea Shpeley & Ball, 1994
Coptodera (Coptodera) festiva Dejean, 1825
Coptodera (Coptodera) nitidula (Buquet, 1835)
Coptodera (Coptodera) picea Dejean, 1826
Cymindis (Tarulus) americana Dejean, 1826
Cymindis (Tarulus) arizonensis Schaeffer, 1910
Cymindis (Tarulus) borealis LeConte, 1863
Cymindis (Tarulus) californica Horn, 1895
Cymindis (Tarulus) cribricollis Dejean, 1831
Cymindis (Tarulus) elegans LeConte, 1846
Cymindis (Tarulus) evanescens Casey, 1913
Cymindis (Tarulus) interior Lindroth, 1969
Cymindis (Tarulus) laticollis Say, 1830
Cymindis (Tarulus) neglecta Haldeman, 1843
Cymindis (Tarulus) pilosa Say, 1823
Cymindis (Tarulus) planipennis LeConte, 1863
Cymindis (Tarulus) seriata Hatch, 1953
Cymindis (Tarulus) unicolor Kirby, 1837
Cymindis (Tarulus) uniseriata Bates, 1884
Cymindis (Tarulus) vaporariorum (Linnaeus, 1758)‡
Cymindis (Pinacodera) abbreviata (Casey, 1920)
Cymindis (Pinacodera) ampliata (Casey, 1920)
Cymindis (Pinacodera) atripennis (Casey, 1920)
Cymindis (Pinacodera) blanda Casey, 1913
Cymindis (Pinacodera) complanata Dejean, 1826
Cymindis (Pinacodera) limbata Dejean, 1831
Cymindis (Pinacodera) obscura (Casey, 1920)
Cymindis (Pinacodera) platicollis (Say, 1823)
Cymindis (Pinacodera) punctifera (LeConte, 1884)
Cymindis (Pinacodera) punctigera LeConte, 1851
Cymindis (Pinacodera) subcarinata (Casey, 1920)
Apenes (Apenes) angustata Schwarz, 1878
Apenes (Apenes) coriacea (Chevrolat, 1863)
Apenes (Apenes) hilariola Bates, 1891
Apenes (Apenes) lucidula lucidula (Dejean, 1831)
Apenes (Apenes) nebulosa LeConte, 1867
Apenes (Apenes) opaca LeConte, 1851
Apenes (Apenes) pallidipes (Chevrolat, 1836)
Apenes (Apenes) parallela parallela (Dejean, 1825)
Apenes (Apenes) sinuata (Say, 1823)
Dromius (Dromius) fenestratus (Fabricius, 1794)†
Dromius (Dromius) piceus Dejean, 1831
Philorhizus atriceps (LeConte, 1880)
Philorhizus melanocephalus (Dejean, 1825)†
Microlestes brevilobus brevilobus Lindroth, 1969
Microlestes curtipennis (Casey, 1920)
Microlestes lindrothi Mateu, 1995
Microlestes linearis (LeConte, 1851)
Microlestes lucidus lucidus (LeConte, 1851)
Microlestes major Lindroth, 1969
Microlestes nigrinus (Mannerheim, 1843)
Microlestes pusio (LeConte, 1863)
Apristus actuosus Casey, 1920
Apristus agitatus Casey, 1920
Apristus cephalus Casey, 1920
Apristus constrictus Casey, 1920
Apristus latens (LeConte, 1846)
Apristus laticollis LeConte, 1851
Apristus liratus Casey, 1920
Apristus nevadensis Casey, 1920
Apristus pugetanus Casey, 1920
Apristus subdeletus Casey, 1920
Apristus subsulcatus (Dejean, 1826)
Apristus thoracicus Casey, 1920
Apristus tuckeri Casey, 1920
Syntomus americanus (Dejean, 1831)
Axinopalpus biplagiatus (Dejean, 1825)
Axinopalpus denticulatus Hatch, 1949
Axinopalpus fusciceps LeConte, 1851
Axinopalpus illectus Casey, 1920
Axinopalpus pratti Hatch, 1949
Axinopalpus utahensis Tanner, 1928
Axinopalpus vittatus Hatch, 1949
Lebia (Loxopeza) atriceps LeConte, 1863
Lebia (Loxopeza) atriventris Say, 1823
Lebia (Loxopeza) deceptrix Madge, 1967
Lebia (Loxopeza) grandis Hentz, 1830
Lebia (Loxopeza) pimalis (Casey, 1920)
Lebia (Loxopeza) subdola Madge, 1967
Lebia (Loxopeza) subgrandis Madge, 1967
Lebia (Loxopeza) tricolor Say, 1823
Lebia (Polycheloma) lecontei Madge, 1967
Lebia (Lamprias) divisa LeConte, 1850
Lebia (Lebia) abdita Madge, 1967
Lebia (Lebia) abdominalis Chaudoir, 1843
Lebia (Lebia) analis Dejean, 1825
Lebia (Lebia) arizonica Schaeffer, 1910
Lebia (Lebia) bilineata Motschulsky, 1859
Lebia (Lebia) bitaeniata Chevrolat, 1834
Lebia (Lebia) bivittata (Fabricius, 1798)
Lebia (Lebia) bumeliae Schaeffer, 1910
Lebia (Lebia) calliope Bates, 1883
Lebia (Lebia) collaris Dejean, 1826
Lebia (Lebia) cyanipennis Dejean, 1831
Lebia (Lebia) esurialis Casey, 1920
Lebia (Lebia) fuscata Dejean, 1825
Lebia (Lebia) guttula LeConte, 1851
Lebia (Lebia) histrionica Bates, 1883
Lebia (Lebia) insulata Madge, 1967
Lebia (Lebia) lecta Horn, 1885
Lebia (Lebia) lobulata LeConte, 1863
Lebia (Lebia) marginicollis Dejean, 1825
Lebia (Lebia) miranda (Horn, 1872)
Lebia (Lebia) moesta LeConte, 1850
Lebia (Lebia) nigricapitata Madge, 1967
Lebia (Lebia) ornata Say, 1823
Lebia (Lebia) pectita Horn, 1885
Lebia (Lebia) perita Casey, 1920
Lebia (Lebia) perpallida Madge, 1967
Lebia (Lebia) pleuritica LeConte, 1846
Lebia (Lebia) pulchella Dejean, 1826
Lebia (Lebia) pumila Dejean, 1831
Lebia (Lebia) rufopleura Schaeffer, 1910
Lebia (Lebia) scalpta Bates, 1883
Lebia (Lebia) scapula Horn, 1885
Lebia (Lebia) solea Hentz, 1830
Lebia (Lebia) subrugosa Chaudoir, 1871
Lebia (Lebia) tuckeri (Casey, 1920)
Lebia (Lebia) viridipennis Dejean, 1826
Lebia (Lebia) viridis Say, 1823
Lebia (Lebia) vittata (Fabricius, 1777)
Hyboptera auxiliadora Erwin, 2004
Plochionus (Menidius) amandus Newman, 1840
Plochionus (Menidius) bicolor Notman, 1919
Plochionus (Menidius) discoideus LeConte, 1880
Plochionus (Menidius) timidus Haldeman, 1843
Plochionus (Plochionus) pallens (Fabricius, 1775)†
Tecnophilus croceicollis croceicollis (Ménétriés, 1843)
Tecnophilus croceicollis peigani Larson, 1969
Tecnophilus pilatei Chaudoir, 1877
Calleida (Calleida) circumcincta Bates, 1883
Calleida (Calleida) decora (Fabricius, 1801)
Calleida (Calleida) fimbriata Bates, 1883
Calleida (Calleida) fulgida Dejean, 1831
Calleida (Calleida) obrieni Mateu, 1995
Calleida (Calleida) planulata LeConte, 1858
Calleida (Calleida) platynoides Horn, 1882
Calleida (Calleida) punctata LeConte, 1846
Calleida (Calleida) punctulata Chaudoir, 1848
Calleida (Calleida) purpurea (Say, 1823)
Calleida (Calleida) viridipennis (Say, 1823)
Philophuga caerulea Casey, 1913
Philophuga viridicollis (LeConte, 1846)
Philophuga viridis amoena (LeConte, 1846)
Philophuga viridis horni Chaudoir, 1877
Philophuga viridis klamathea Larson, 1969
Philophuga viridis viridis (Dejean, 1831)
Infernophilus castaneus (Horn, 1882)
Onota angulicollis (Reiche, 1842)
Onota floridana Horn, 1881
Cylindronotum aeneum Putzeys, 1845
Agra oblongopunctata oblongopunctata Chevrolat, 1836
Euproctinus (Neoeuproctus) abjectus (Bates, 1883)
Euproctinus (Neoeuproctus) balli Shpeley, 1986
Euproctinus (Neoeuproctus) trivittatus (LeConte, 1878)
Nemotarsus elegans LeConte, 1853
Nemotarsus rhombifer Bates, 1883
ZuphiiniZuphium americanum Dejean, 1831
Zuphium delectum Liebke, 1933
Zuphium longicolle LeConte, 1879
Zuphium magnum Schaeffer, 1910
Zuphium mexicanum Chaudoir, 1863
Zuphium pseudamericanum Mateu, 1981
Pseudaptinus (Pseudaptinus) lecontei (Dejean, 1831)
Pseudaptinus (Pseudaptinus) oviceps Van Dyke, 1926
Pseudaptinus (Pseudaptinus) tenuicollis (LeConte, 1851)
Pseudaptinus (Thalpius) cubanus (Chaudoir, 1877)
Pseudaptinus (Thalpius) deceptor Darlington, 1934
Pseudaptinus (Thalpius) dorsalis (Brullé, 1834)
Pseudaptinus (Thalpius) hoegei (Bates, 1883)
Pseudaptinus (Thalpius) horni (Chaudoir, 1872)
Pseudaptinus (Thalpius) microcephalus (Van Dyke, 1926)
Pseudaptinus (Thalpius) nobilis Liebke, 1934
Pseudaptinus (Thalpius) pygmaeus (Dejean, 1826)
Pseudaptinus (Thalpius) rufulus (LeConte, 1851)
GaleritiniGalerita (Progaleritina) atripes LeConte, 1858
Galerita (Progaleritina) bicolor (Drury, 1773)
Galerita (Progaleritina) forreri Bates, 1883
Galerita (Progaleritina) janus (Fabricius, 1792)
Galerita (Progaleritina) lecontei lecontei Dejean, 1831
Galerita (Progaleritina) mexicana Chaudoir, 1872
Galerita (Progaleritina) reichardti Ball & Nimmo, 1983
Galerita (Galerita) aequinoctialis Chaudoir, 1852
HelluoniniHelluomorphoides clairvillei (Dejean, 1831)
Helluomorphoides ferrugineus (LeConte, 1853)
Helluomorphoides latitarsis (Casey, 1913)
Helluomorphoides nigripennis (Dejean, 1831)
Helluomorphoides papago (Casey, 1913)
Helluomorphoides praeustus bicolor (Harris, 1828)
Helluomorphoides praeustus floridanus Ball, 1956
Helluomorphoides praeustus praeustus (Dejean, 1825)
Helluomorphoides texanus (LeConte, 1853)
PseudomorphiniPseudomorpha (Pseudomorpha) alleni Van Dyke, 1953
Pseudomorpha (Pseudomorpha) alutacea Notman, 1925
Pseudomorpha (Pseudomorpha) augustata Horn, 1883
Pseudomorpha (Pseudomorpha) behrensi Horn, 1870
Pseudomorpha (Pseudomorpha) castanea Casey, 1909
Pseudomorpha (Pseudomorpha) champlaini Notman, 1925
Pseudomorpha (Pseudomorpha) consanguinea Notman, 1925
Pseudomorpha (Pseudomorpha) cronkhitei Horn, 1867
Pseudomorpha (Pseudomorpha) cylindrica Casey, 1889
Pseudomorpha (Pseudomorpha) excrucians Kirby, 1823
Pseudomorpha (Pseudomorpha) falli Notman, 1925
Pseudomorpha (Pseudomorpha) hubbardi Notman, 1925
Pseudomorpha (Pseudomorpha) parallela Van Dyke, 1943
Pseudomorpha (Pseudomorpha) schwarzi Notman, 1925
Pseudomorpha (Pseudomorpha) tenebroides Notman, 1925
Pseudomorpha (Pseudomorpha) vandykei Notman, 1925
Pseudomorpha (Pseudomorpha) vicina Notman, 1925
Pseudomorpha (Pseudomorpha) vindicata Notman, 1925
Six species in western North America (three species), South America (two species), and northern Eurasia (one species). The species are arrayed in two genera: Systolosoma Solier (two South American species) and Trachypachus (four species).
Northern Hemisphere, with four species in the Nearctic (three species) and Palaearctic (one species) Regions.
Lindroth (1961a: 1-4) reviewed the North American species and discussed the structural differences between the three taxa.
This species ranges from southern British Columbia (Lindroth 1961a: 4) to northwestern Montana (Russell 1968: 42; Edwards 1975: 48), south to the southern part of the Sierra Nevada (Lindroth 1961a: 4) and to the Coast Ranges (Fall 1901a: 39) in California.
CAN: BC USA: CA, ID, MT, OR, WA
Trachypachus gibbsii LeConte. This species is a typical western element as are the other two North American trachypachids. Although superficially similar to some large Bembidion or small Amara and having the same ecological preferences, we now believe that these beetles, along with members of the related genus Systolosoma of South America, are not closely related to any groups of carabids.
The range of this species extends from the Kenai Peninsula in Alaska (Lindroth 1961a: 2) to northwestern Saskatchewan (Hooper 1980: 65), south to southern Colorado (Wickham 1902: 230; LeConte 1878a: 464; Bell 1971: 58), southern Utah (Garfield County, Foster F. Purrington pers. comm. 2011), and the Sierra Nevada and Coast Ranges in California (Fall 1901a: 39). One old specimen, simply labeled “Ks” is known (MCZ, collection LeConte).
CAN: AB, BC (VCI), NT, SK, YT USA: AK, CA, CO, ID, MT, NV, OR, UT, WA, WY [KS]
This species is known only from the western regions of Washington and Oregon (Lindroth 1961a: 4).
USA: OR, WA
Worldwide, with about 355 species arrayed in six tribes: Clinidiini (about 135 species), Dhysorini (ten species), Leoglymmiini (one species), Medisorini (one species), Omoglymmiini (about 180 species), and Rhysodini (about 25 species). Over 90% of the species are found in the Southern Hemisphere.
Bell (1970) revised the North American, Middle American, and West Indies species and provided keys for their identification.
Worldwide, with about 135 species arrayed in three genera: Clinidium (about 75 species), Grouvellina Bell and Bell (17 Madagascan species), and Rhyzodiastes Fairmaire (about 45 species). The vast majority of species are found in the Southern Hemisphere, with only 11 species (about 8% of the world fauna) occurring in the Northern Hemisphere.
About 75 species in the Nearctic (six species), Neotropical (about 65 species), and Palaearctic (three species) Regions arrayed in four subgenera: Arctoclinidium (nine species), Clinidium s.str. (about 50 Neotropical species), Mexiclinidium Bell and Bell (11 Middle American species), and Tainoa Bell and Bell (four West Indian species).
Bell and Bell (1985) revised the species of the world and provided keys for their identification.
Northern Hemisphere, with nine species in North America (six species), Japan (one species), Caucasian region (one species), and southern Europe (one species).
This subspecies is known only from southwestern Pennsylvania and the Black Mountains in western North Carolina (Bell and Bell 1985: 91). The record from “Ohio” (Bousquet and Larochelle 1993: 42) needs confirmation.
USA: NC, PA [OH]
This subspecies is known only from the type locality in northern Georgia (Bell and Bell 1985: 90).
USA: GA
This species ranges from New Jersey to central Iowa, including southwestern Wisconsin (Messer 2010: 33), south to southern Mississippi (Bell and Bell 1985: 89) and northern Florida (Bell 1970: 313). Old specimens simply labeled from Nebraska, Kansas, Missouri, and Texas are known (Bell 1970: 313). The records from “Arkansas” and “Louisiana” (Bousquet and Larochelle 1993: 43) need confirmation.
USA: AL, FL, GA, IA, IL, IN, KY, MD, MS, NC, NJ, OH, PA, SC, TN, VA, WI, WV [AR, KS, LA, MO, NE, TX]
This species ranges from southern British Columbia, including Vancouver Island, south to Mendocino County in the Coast Ranges of California and Tuolumne County in the Sierra Nevada (Bell and Bell 1985: 84).
CAN: BC (VCI) USA: CA, OR, WA
This species ranges from northwestern Pennsylvania to eastern Missouri, south to east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009) and northern Alabama (Madison County, CMNH).
USA: AL, IL, IN, KY, LA, MO, NC, OH, PA, TN, VA
The range of this species extends from the Catskills in southern New York to west-central Indiana (Bell and Bell 1985: 92), south to east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), northern Alabama (Bell and Bell 1985: 92), and the Florida Panhandle (Peck and Thomas 1998: 15). Specimens simply labeled from Texas are known (Bell and Bell 1985: 92). The record from central Illinois (Wolcott 1896: 235) needs confirmation.
USA: AL, DC, DE, FL, GA, IN, KY, LA, MD, MO, NC, NJ, NY, OH, PA, SC, TN, VA, WV [IL, TX]
This species seems to be confined to the Appalachian Mountains from southwestern Pennsylvania to north-central Alabama and northeastern Georgia (Bell and Bell 1985: 85).
USA: AL, GA, NC, PA, SC, TN
Worldwide, with about 180 species arrayed in eight genera. The tribe is much more diverse, both in term of species and lineages, in Asia than anywhere else.
About 150 species (Lorenz 2005: 158-159) in the Nearctic (two species), Australian, Oriental, Palaearctic (ten species, only one of them present in Europe), and Afrotropical (one species) Regions arrayed in 11 subgenera. More than 90% of the species are found in Asia.
Bell and Bell (1983) revised the species of the world and provided keys for their identification.
Three species in North American (two species) and Japan (one species).
This species ranges from central New York to eastern Minnesota, south to east-central Texas (Bell and Bell 1983: 145) and northern Florida (Peck and Thomas 1998: 15).
CAN: ON USA: AL, AR, DE, FL, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI
Omoglymmius americanus (Laporte). This species is one of the seven rhysodid species-group taxa found in eastern North America. These species live in decaying wood, such as logs, stumps or roots, where they feed on slime molds and fungi. The carabids, on the other hand, are carnivorous, herbivorous, or omnivorous feeding on both animal and plant matters.
This species ranges from northern Idaho and southeastern Washington south to southern California, including the Sierra Nevada, and southeastern Arizona (Bell and Bell 1983: 143-144). Old specimens without specific localities from British Columbia and Texas (Bell and Bell 1983: 144) are known.
USA: AZ, CA, ID, NV, OR, WA [BC, TX]
About 665 species in the Nearctic (about 75 species), Neotropical (one South American species), and Palaearctic and northern parts of the Oriental (about 595 species) Regions. The species are arrayed in five tribes: Nebriini (about 600 species), Notiokasiini (one South American species), Notiophilini (about 55 species), Opisthiini (five species), and Pelophilini (two species).
This tribe contains a single genus.
Northern Hemisphere, with one Holarctic species and one species endemic to northern North America.
Lindroth (1961a: 57-60) covered both species and discussed the structural differences between them.
This species is found from northern Europe to the Bering Sea coast (Farkač 2003: 98) and from Alaska, including the Aleutian and Kodiak Islands, to Newfoundland, south to the Abitibi region in western Quebec (Larochelle 1975: 98) [see Lindroth 1963a: Fig. 59]. Fossil remnants of this species, dated between about 14, 000 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96; Schwert 1992: 76) and southern Ontario (Morgan and Morgan 1981: 1107).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, SK, YT USA: AK – Holarctic
Pelophila borealis (Paykull). This pelophiline is one of the 97 species-group taxa which are Holarctic and found naturally in both the Palaearctic and Nearctic Regions. Most of these species are confined to the arctic, subarctic or boreal regions but a few are found mainly in the temperate regions such as Dyschirius politus. Most biogeographers agree that these species have spread between the two continents through Beringia during the Quaternary period.
This species is known from scattered localities from western Newfoundland (Lindroth 1955a: 39) to the Fairbanks area in Alaska (David H. Kavanaugh pers. comm. 2009), south to central British Columbia (Lindroth 1961a: 59) [see Lindroth 1963a: Fig. 65]. Fossil remnants, dated between 14, 000 and 15, 500 years B.P., have been unearthed in central Iowa (Schwert 1992: 76).
CAN: AB, BC, MB, NF, NT, ON, SK, YT USA: AK
Northern Hemisphere, with five species in North America (one species) and the Himalayas and China, including Taiwan (four species). The species are arrayed in two genera: Opisthius (one species) and Paropisthius Casey (four species).
Bousquet and Smetana (1996) reviewed the species and provided a key for their identification.
One North American species.
The species was treated in Lindroth’s (1961a: 88-90) monograph on the Carabidae of Canada and Alaska.
The range of this species extends from central Saskatchewan to the Arctic Circle in central Alaska (Lindroth 1961a: 89-90), south to Tuolumne County in the Sierra Nevada of California (Dajoz 2007: 17) and north-central New Mexico (Taos County, CNC). The record from “Iowa” (Jaques and Redlinger 1946: 295) is probably based on a mislabeled specimen or a stray. Fossil remnants of this species from the late Wisconsinan age have been found in northeastern Illinois, north-central Iowa, south-central Minnesota, and northwestern Ontario (see Ashworth and Schwert 1991: 511); others from a Plio-Pleistocene sequence have been found in northwestern Greenland and Meighen Island (Böcher 1995: 18).
CAN: AB, BC (VCI), NT, SK, YT USA: AK, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY
About 600 species (Lorenz 2005: 116-124) in the Nearctic (about 60 species), Palaearctic, and northern parts of the Oriental Regions. The species are arrayed in four genera: Archastes Jedlička (27 Chinese species), Leistus (about 180 species), Nebria (including Oreonebria Daniel) (about 380 species), and Nippononebria (seven species).
Northern Hemisphere, with about 180 species (Lorenz 2005: 116-118) in the Nearctic (four species, one of them adventive) and Palaearctic (about 180 species) Regions. The species are arrayed in six subgenera: Evanoleistus Jedlička (about 95 Asian species), Leistus s.str. (about 40 species), Nebrileistus Bänninger (two species on Madeira and Canary Islands), Neoleistus (three species), Sardoleistus Perrault (one Mediterranean species), and Pogonophorus Latreille (about 35 Palaearctic species).
About 40 Palaearctic species of which one is adventive in North America.
Larson (1978: 307-308) discussed the structural differences between the adventive species in North America and the three native species of the subgenus Neoleistus.
This European species is adventive in North America where it is known only from near Saint John’s, Newfoundland (Larson 1978: 307). The first inventoried specimen collected on this continent was caught in 1977.
CAN: NF – Adventive
Three western North American species.
Erwin (1970b) revised the species and provided a key for their identification.
Perrault (1991a) added three species from the Far East (Leistus angulicollis Fairmaire, Leistus niger Gebler, and Leistus shenseensis Perrault) to this subgenus but Shilenkov (1999: 76) rejected this association and the Asian species are listed in the nominotypical subgenus by Farkač and Janata (2003: 81-82).
This species ranges from the Gulf of Alaska coast south to west-central Oregon, east to western Montana (Russell 1968: 44) [see Erwin 1970b: Fig. 7]. At least one specimen simply labeled from California is known (Erwin 1970b: 115).
CAN: AB, BC (QCI, VCI) USA: AK, MT, OR, WA [CA]
The range of this species is restricted to the extreme northwestern tip of Humboldt County in northern California (Erwin 1970b: 117) and southwestern Oregon (Curry County, James R. LaBonte pers. comm. 1992).
USA: CA, OR
This species is known only from a few localities along the Pacific Coast in northern California [see Erwin 1970b: Fig. 7].
USA: CA
Seven species in western North America (three species), Japan (three species), and Jilin Province in China (one species) arrayed in two subgenera: Nippononebria s.str. for the Japanese and Chinese species and Vancouveria for the Nearctic ones.
Kavanaugh (1995, 1996) regarded Nippononebria as the sister-group to Leistus while Ledoux and Roux (2005) listed Nippononebria and Vancouveria as subgenera of Nebria and suggested they form the sister-group to {Eonebria Semenov and Znojko + Sadonebria Ledoux and Roux}, a complex of 60 Palaearctic species. Because the North American students are used to Kavanaugh’s approach, the taxon is retained here as a distinct genus.
Three species in western North America.
Ledoux and Roux (2005: 712) reviewed the species and provided a key for their identification.
This species occurs at high elevations in the Sierra Nevada of California, from El Dorado County south to Sequoia National Park [see Kavanaugh 1984: Fig. 31].
USA: CA
This species ranges from the Three Brothers Mountain in southern British Columbia south to Mount Baker in Washington [see Kavanaugh 1984: Fig. 31].
CAN: BC USA: WA
This species ranges from southwestern British Columbia, including Vancouver Island, south to the northern Sierra Nevada of California in Plumas County, east to the western edge of the Rockies in west-central Idaho (Kavanaugh 1978: 349).
CAN: BC (VCI) USA: CA, ID, OR, WA
About 380 species (535 species-group taxa) in the arctic, subarctic, boreal, and temperate areas of the Nearctic and Palaearctic (including northern Africa and the Canary Islands) Regions arrayed in 25 subgenera (Ledoux and Roux 2005: 76 excluding Nippononebria and Vancouveria). The North American fauna has 52 species (82 species-group taxa) placed in four subgenera.
Ledoux and Roux (2005) reviewed the species of the world and provided keys for the identification of the species. Lindroth’s (1961a) key included all North American species then known but many species-group taxa have been described subsequently by Kavanaugh (1979a, 1981b, 1984, 2008).
The species of Nebria (including Nippononebria) have been segregated in two main lineages by Ledoux and Roux (2005: 71-75), one (named Vetanebri) represented in the Palaearctic Region by 90 species and in the Nearctic Region by the three species of Vancouveria, the other one (Notanebri) containing about 290 species, 52 in the Nearctic and almost 240 in the Palaearctic.
Thirty-one species (Ledoux and Roux 2005: 82) in North America (seven species) and Eurasia (26 species). Two species are Holarctic (Nebria frigida and Nebria nivalis).
[gyllenhali group]
This subspecies is confined to a small area of the Coast Ranges and Cascade Range in southwestern British Columbia and western Washington [see Kavanaugh 1988: Fig. 15].
CAN: BC USA: WA
This subspecies ranges from northern British Columbia south to northeastern Oregon and southern Utah, east to northwestern Wyoming and central Utah [see Kavanaugh 1988: Fig. 15].
CAN: AB, BC USA: ID, MT, OR, UT, WA, WY
This Holarctic species is found in eastern Siberia (Farkač and Janata 2003: 88) and from the Alaskan Coast Range to the Anderson River Delta in northern Northwest Territories, south to northern British Columbia (Kavanaugh 1978: 714-715).
CAN: BC, NT, YT USA: AK – Holarctic
This species is placed in its own group by Ledoux and Roux (2005: 82).
This subspecies ranges from Greenland (Böcher 1988: 5) to Alaska (Lindroth 1961a: 79), south to northeastern Oregon, northeastern Nevada, western South Dakota, and northeastern New York (Kavanaugh 1978: 731-741).
DEN: GL CAN: AB, BC (VCI), LB, MB, NF, NT, ON, QC, SK, YT USA: AK, ID, ME, MI, MN, MT, NH, NV, NY, OR, SD, WA, WI, WY
The name Nebria rufescens (Strøm, 1768) is used by several authors instead of Nebria gyllenhali (Schönherr, 1806). I concur with Kavanaugh (1979a: 111) and Ledoux and Roux (2005: 107) that Carabus rufescens Strøm should be considered a nomen dubium.
This subspecies is restricted to mountains in the southern part of the Cascade Range, south of the lower Columbia River valley, and the northern part of the Sierra Nevada, south to Sonora Pass [see Kavanaugh 1979a: Fig. 54].
USA: CA, OR
This subspecies is found in the southern Rocky Mountains from the Medicine Bow Mountains and Sierra Madre of southern Wyoming south to northern New Mexico, west to the Uinta Mountains in eastern Utah and the Chuska Mountains in northeastern Arizona (David H. Kavanaugh pers. comm. 2008) [see Kavanaugh 1979a: Fig. 54].
USA: AZ, CO, NM, UT, WY
This subspecies is known from western Newfoundland, coastal Labrador (Lindroth 1961a: 82), and the east edge of the Ungava Bay in northern Quebec; isolated on high mountains in the Gaspé Peninsula in Quebec and on Mount Katahdin in Maine [see Kavanaugh 1979a: Fig. 53].
CAN: LB, NF, QC USA: ME
This Holarctic subspecies is known from northern Europe to the Bering Sea Coast (Farkač and Janata 2003: 88), and from the Arctic Plains in Alaska to Baffin Island and the western edge of the Ungava Bay in northern Quebec (Kavanaugh 1978: 779-782).
CAN: BC, NT, QC, YT USA: AK – Holarctic
Ledoux and Roux (2005: 112) retained Nebria bifaria Mannerheim, 1853 as a valid subspecies of Nebria nivalis.
[hudsonica group]
This species is restricted to the Great Smoky Mountains National Park and adjacent mountain ranges in the southern Appalachians [see Kavanaugh 1979a: Fig. 52]. Two specimens labeled from Jefferson County in Colorado and Saint Tammany Parish in Louisiana seen by Kavanaugh (1979a: 96) are likely mislabeled.
USA: NC, TN
This taxon, originally described as a subspecies of Nebria lacustris Casey, has been raised to species status by Kavanaugh et al. (2011).
This species is restricted to Washington, northern Oregon, and Idaho [see Kavanaugh 1979a: Fig. 51]. Seven specimens labeled from Longview (Highwood River) in southwestern Alberta seen by Kavanaugh (1979a: 95) are listed as doubtful by him; two specimens simply labeled from California are likely mislabeled.
USA: ID, OR, WA [AB]
This species ranges from the north shore of Lake Superior in western Ontario to southern Yukon Territory and southeastern Alaska (Skagway, David H. Kavanaugh pers. comm. 2008), south to the Columbia River drainage in northern Oregon and to north-central Utah and central Colorado along the Rocky Mountains (Kavanaugh 1978: 745-753). The records from New Mexico (Snow 1885: 66; Fall and Cockerell 1907: 156) are probably in error.
CAN: AB, BC, MB, NT, ON, SK, YT USA: AK, CO, ID, MT, OR, UT, WA, WY
This species is found from northern New Brunswick (Restigouche County, CNC) to northern Minnesota, north to southeastern Manitoba, south to east-central Iowa, southern Indiana, and western North Carolina along the Appalachians [see Kavanaugh 1979a: Fig. 52]. The record from “Texas” (Casey 1913: 56) is in error (Kavanaugh 1979a: 112).
CAN: MB, NB, ON, QC USA: CT, DC, IA, IL, IN, KY, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, TN, VA, VT, WI, WV
Six species in western North America (two species) and the Far East (four species).
According to Ledoux and Roux (2005: 75), Nakanebria is the sister-group to Reductonebria and the two form the sister-group to Catonebria.
This species ranges in the Cascade Range from northwestern Washington to northwestern Oregon [see Kavanaugh 1988: Fig. 19].
USA: OR, WA
This species is endemic to the Trinity Alps in northwestern California [see Kavanaugh 1981b: Fig. 21].
USA: CA
Twenty-eight species in North America (24 species) and Siberia and the Far East (four species).
Nebria carbonaria Eschscholtz was reported from Alaska by Horn (1870b: 104) and LeConte (1878a: 479) and there is a specimen of this species in the LeConte collection labeled “Ins. S. Pauli, ” one of the Pribilof Islands in Alaska. Lindroth (1961a: 74) believes the specimen is probably mislabeled since no other specimens of the species have been found on the island despite extensive search. The species was described from specimens collected “in Kamtschatka, bei St. Peter und Paul” which, according to Lindroth (1961a: 74), refers to the town of Petropavlovsk in Kamtschatka. Until recent specimens are collected on this continent, the species is not listed as a North American resident.
[gregaria group]
This subspecies is found in southern Wyoming, Colorado, northern New Mexico, and southeastern Utah (Kavanaugh 1978: 675-678). One specimen labeled from Nez Perce County in western Idaho is considered doubtful by Kavanaugh (1978: 678).
USA: CO, NM, UT, WY [ID]
This species is placed with the species of the lyelli group by Ledoux and Roux (2005: 195).
This subspecies is widely distributed in the Rocky Mountain region from southern Yukon Territory south to northeastern Nevada, southern Idaho, and northern Wyoming [see Kavanaugh 1979a: Fig. 58].
CAN: AB, BC, YT USA: ID, MT, NV, OR, WA, WY
This subspecies is restricted to mountains in western Wyoming and northern and central Utah (Kavanaugh 1979a: Figs 58–59).
USA: UT, WY
This subspecies is endemic to the La Sal Mountains in eastern Utah [see Kavanaugh 1979a: Fig. 58].
USA: UT
This species is restricted to the Queen Charlotte Archipelago (Kavanaugh 1992: 55).
CAN: BC (QCI)
This species is endemic to the Aleutian Islands [see Kavanaugh 1981a: Fig. 8].
USA: AK
Nebria macrocephala Motschulsky was described from specimens originating «probablement de Sitka ou d’Ounalachka [Alaska]» (Motschulsky 1844: 128). The name is listed as a junior synonym of Nebria gregaria Fischer von Waldheim, 1820 in Ledoux and Roux (2005: 222) but according to Bänninger (1923: 131) only the specimens reported subsequently by Motschulsky (1860: 97; 1866: 273) as Nebria macrocephala belong to Nebria gregaria. The syntypes are conspecific with specimens of Nebria stigmula Dejean, 1826 (= Nebria hellwigii Panzer, 1803) as noted by Mannerheim (1853: 111). The provenance given by Motschulsky is probably incorrect.
This species is endemic to high elevations in the Queen Charlotte Islands [see Kavanaugh 1984: Fig. 31] and adjacent mainland on Mount McNeil (Kavanaugh 1992: 56) in British Columbia.
CAN: BC (QCI)
Clarke et al. (2001: 1416) concluded that this taxon may be more appropriately treated as a subspecies of Nebria charlottae given the minor differences in morphology and biology.
This species is known only from the Steens Mountains in south-central Oregon [see Kavanaugh 1984: Fig. 31].
USA: OR
This species is placed with the species of the lyelli group by Ledoux and Roux (2005: 195).
This species is known only from a small area in the Alexander Archipelago and northwestern British Columbia [see Kavanaugh 1988: Fig. 24].
CAN: BC USA: AK
This species is known from several islands in the Queen Charlotte Archipelago, British Columbia (Kavanaugh 1992: 55).
CAN: BC (QCI)
Based on DNA sequence analyses, Clarke et al. (2001: 1416) concluded that this taxon may not represent a distinct taxonomic unit but rather a variant of Nebria charlottae.
This subspecies is known only from the Warner Mountains in northeastern California [see Kavanaugh 1979a: Fig. 56].
USA: CA
This subspecies ranges from the Aleutian Islands in Alaska (Lindroth 1961a: 70) to southwestern Northwest Territories (Tungsten, David H. Kavanaugh pers. comm. 2008), south to northwestern Montana (Edwards 1975: 50) and southern Oregon [see Kavanaugh 1988: Fig. 20]. The records from New Mexico (Snow 1885: 66; Fall and Cockerell 1907: 156), Colorado (Wickham 1902: 232; Armin 1963: 94), and Wyoming (Lavigne 1977: 46) are probably in error.
CAN: AB, BC (QCI, VCI), NT, YT USA: AK, MT, OR, WA
This species is placed in the lyelli group by Ledoux and Roux (2005: 195).
This subspecies is yet recorded only from the Klamath Mountains system in northwestern California [see Kavanaugh 1979a: Fig. 56].
USA: CA
This subspecies is restricted to the Pine Valley Mountains in southwestern Utah [see Kavanaugh 1979a: Fig. 60].
USA: UT
This subspecies is known only from mountains in southwestern Utah [see Kavanaugh 1979a: Fig. 60].
USA: UT
This species is placed in the lyelli group by Ledoux and Roux (2005: 195).
[lyelli group]
This subspecies ranges from the Kenai Peninsula in Alaska (Kavanaugh 1978: 671) and southwestern Yukon Territory (Kluane Lake, Sydney G. Cannings pers. comm. 2009) south to the southern part of the Cascade Range and the Sierra Nevada in central California (Kavanaugh 1978: 671).
CAN: BC, YT USA: AK, CA, OR, WA
This subspecies is endemic to the Olympic Peninsula in Washington [see Kavanaugh 1979a: Fig. 55].
USA: WA
As far as known, this subspecies is restricted to the Sierra Nevada, between Sonora Pass and Buckeye Pass, in California [see Kavanaugh 1981b: Fig. 21].
USA: CA
This species is endemic to high elevations in the Olympic Mountains in northwestern Washington [see Kavanaugh 1984: Fig. 21].
USA: WA
This species is known only from Mount Lyell at the edge of the Yosemite National Park in the Sierra Nevada, California (Kavanaugh 1978: 766).
USA: CA
This species is known only from the Wallowa Mountains in northeastern Oregon [see Kavanaugh 1984: Fig. 31].
USA: OR
[mannerheimii group]
This species is known only from the canyon of the South Fork of the American River in the Sierra Nevada, California [see Kavanaugh 1979a: Fig. 62].
USA: CA
This species is known only from the type locality in south-central Utah.
USA: UT
This species ranges along the Pacific Coast from the Queen Charlotte Islands to northern California (Kavanaugh 1992: 57).
CAN: BC (QCI, VCI) USA: CA, OR, WA
This species is placed in a separate group (pallipes group) along with Nebria appalachia and Nebria pallipes by Ledoux and Roux (2005: 195).
The range of this species extends from northwestern Washington to northeastern Idaho, south to southern California [see Kavanaugh 1979b: Fig. 2]. The records from Colorado (Wickham 1902: 232; Armin 1963: 89) and Montana (Hatch 1933a: 7) must be in error.
USA: CA, ID, NV, OR, WA
This species is known only from the original two specimens collected 141 kilometers apart in the Grand Canyon National Park (Kavanaugh 2008: 5).
USA: AZ
This species ranges from the Kenai Peninsula in southern Alaska (Lindroth 1961a: 75) south to western Idaho and central Oregon (Kavanaugh 1978: 766-772). According to Kavanaugh (1978: 772), a number of specimens labeled from “California, ” Nevada, and Yukon Territory are doubtful. The records from the Absaroka Range in south-central Montana (Hatch 1933a: 7) and western San Juan Mountains in Colorado (Wickham 1902: 232) are also doubtful.
CAN: BC (QCI, VCI) USA: AK, ID, OR, WA [CA, CO, MT, NV, YT]
This species is known only from the vicinity of the type locality in northeastern Arizona [see Kavanaugh 1979a: Fig. 63].
USA: AZ
[obliqua group]
This species is known from the southern Appalachian Mountains in western North Carolina and eastern Tennessee (Kavanaugh 1978: 674).
USA: NC, TN
This species is placed in a distinct group (pallipes group) along with Nebria diversa and Nebria pallipes by Ledoux and Roux (2005: 195).
This subspecies, as far as known, is endemic to the Chuska Mountains in northeastern Arizona [see Kavanaugh 1979a: Fig. 61].
USA: AZ
This subspecies ranges from the west edge of the Wrangell-St. Elias National Park in southeastern Alaska to the Great Slake Lake in Northwest Territories, south to northern New Mexico, northern Arizona, central Nevada, and the lower eastern slope of the Sierra Nevada in California, east to western South Dakota and western Nebraska [see Kavanaugh 1979b: Fig. 4].
CAN: AB, BC, NT, SK, YT USA: AK, AZ, CA, CO, ID, MT, NE, NM, NV, OR, SD, UT, WA, WY
This species is found east of the Mississippi River from Nova Scotia (Lindroth 1961a: 77) to north-central Illinois (Kavanaugh 1978: 800), south to northern Alabama (Löding 1945: 12), northeastern Georgia (Leng 1910: 73; Fattig 1949: 12), and east-central South Carolina (Ciegler 2000: 20) along the Appalachian Mountains. The record from east-central Missouri (Summers 1873: 133) needs confirmation; that from Colorado (Elias 1987: 632) is likely based on a mislabeled specimen; that from northern Wisconsin along Lake Superior (Wickham 1896c: 131) probably refers to Nebria lacustris.
CAN: NB, NS, ON, PE, QC USA: AL, CT, DC, DE, GA, IL, IN, KY, MA, MD, ME, MI, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WV [MO]
This species is placed in a special group (pallipes group) along with Nebria appalachia and Nebria diversa by Ledoux and Roux (2005: 195).
This species ranges from the coast of Labrador and the Ungava Bay region in northern Quebec to the Rocky Mountains in western Alberta, south to central Colorado, the Adirondack Mountains in northeastern New York, and New England [see Kavanaugh 1979b: Fig. 3].
CAN: AB, LB, ON, QC USA: CO, ME, NH, NY, VT, WY
Thirty-one species (Ledoux and Roux 2005: 240) in North America (20 species) and Siberia and the Far East (11 species).
[metallica group]
As far as known, this species is endemic to the Wallowa Mountains in northeastern Oregon [see Kavanaugh 1984: Fig. 31].
USA: OR
This subspecies is known only from the western slope of the White Mountains in eastern California [see Kavanaugh 1984: Fig. 21].
USA: CA
This subspecies has been found only on the eastern slope of the southern Sierra Nevada in California [see Kavanaugh 1979a: Fig. 67].
USA: CA
This subspecies is known from the Skagway area in southeastern Alaska and northwestern British Columbia south along the Cascade Range to Mount Shasta in north-central California [see Kavanaugh 1979a: Fig. 67]. The record from “Whitehorse Pass, ” Yukon Territory (Kavanaugh 1978: 773), refers to the Skagway area in Alaska (Sydney G. Cannings pers. comm. 2009).
CAN: BC USA: AK, CA, OR, WA
This subspecies in known from Vancouver Island and the Olympic Peninsula in northwestern Washington [see Kavanaugh 1979a: Fig. 67].
CAN: BC (VCI) USA: WA
This species ranges from the Aleutian Islands south to western Montana (Hatch 1939a: 118) and southern Washington (Kavanaugh 1978: 775-778). A few specimens labeled from Arizona, California, and Oregon are known (Kavanaugh 1978: 778) but considered doubtful. The record from Colorado (Elias 1987: 632) is in error (David H. Kavanaugh pers. comm. 2012).
CAN: AB, BC (VCI) USA: AK, ID, MT, WA [AZ, CA, OR]
[ovipennis group]
This species is found in the mountains of south-central and western Idaho [see Kavanaugh 1979a: Fig. 65] from the Seven Devils Mountains in the north to the Sawtooth Range in the south.
USA: ID
This subspecies is known only from the type locality in the White Mountains of eastern California.
USA: CA
This subspecies is found from southern British Columbia, including southern Vancouver Island, south along the Cascade Range to central Oregon [see Kavanaugh 1979a: Fig. 64]. One specimen labeled from Leavenworth Valley in Colorado seen by Kavanaugh (1979a: 106) is likely mislabeled.
CAN: BC (VCI) USA: OR, WA
This subspecies is yet recorded only from the type locality in the Strawberry Mountains of eastern Oregon [see Kavanaugh 1979a: Fig. 64].
USA: OR
This subspecies occurs along the Cordilleras from the Alexander Archipelago to southernmost Yukon Territory (Lindroth 1961a: 84), south to southwestern Montana, south-central Idaho, and northeastern Oregon [see Kavanaugh 1979a: Fig. 64]. The record from northern Colorado (Armin 1963: 89) is probably in error.
CAN: AB, BC, YT USA: AK, ID, MT, OR, WA
This species is placed in its own group by Ledoux and Roux (2005: 240).
This subspecies is endemic to the Sierra Nevada and adjacent mountains in western Nevada [see Kavanaugh 1979a: Fig. 64].
USA: CA, NV
This subspecies is restricted to the Klamath Mountains system of the Coast Ranges in southwestern Oregon and northwestern California [see Kavanaugh 1979a: Fig. 64].
USA: CA, OR
This subspecies is restricted to the Cascade Range of central Washington and northern Oregon [see Kavanaugh 1979a: Fig. 65].
USA: OR, WA
This subspecies ranges along the mountains of the Pacific Coast from the Alexander Archipelago to northern Washington [see Kavanaugh 1979a: Fig. 65].
CAN: BC (VCI) USA: AK, WA
This species is known from the Sierra Nevada in California (Erwin and Ball 1972: 81) and adjacent mountains in Washoe County, western Nevada (Kavanaugh 1978: 798).
USA: CA, NV
This subspecies is restricted to the main chain and Eastern Divide of the Sierra Nevada of California [see Kavanaugh 1979a: Fig. 66; David H. Kavanaugh pers. comm. 2012].
USA: CA
This subspecies is endemic to the Western Divide of the southern Sierra Nevada of California [see Kavanaugh 1979a: Fig. 66; David H. Kavanaugh pers. comm. 2012].
USA: CA
[trifaria group]
This species is yet known only from the type locality in northeastern Arizona [see Kavanaugh 1984: Fig. 32].
USA: AZ
This species is restricted to the San Juan Mountains in southwestern Colorado and the Abajo Mountains in southeastern Utah (Erwin and Ball 1972: 97).
USA: CO, UT
This species is found in the Rocky Mountains in southern Wyoming and Colorado (Erwin and Ball 1972: 97).
USA: CO, WY
This taxon has been considered a synonym of Nebria catenata by Lindroth (1961a: 84), a subspecies of Nebria trifaria by Erwin and Ball (1972: 96) and Ledoux and Roux (2005: 285), and a distinct species by Kavanaugh (1985: 421, 423).
This subspecies is restricted to the Sierra Nevada in Fresno, Inyo, and Tulare Counties, California (Kavanaugh 1978: 753-754).
USA: CA
This subspecies is known only from Mono and Tuolumne Counties in the Sierra Nevada of California (Kavanaugh 1978: 754; David H. Kavanaugh pers. comm. 2012).
USA: CA
This species ranges along the mountains paralleling the Pacific Coast from southeastern Alaska along the British Columbia border (Lindroth 1961a: 86) and Kluane National Park in southwestern Yukon Territory (David H. Kavanaugh pers. comm. 2010) to west-central Oregon (Kavanaugh 1978: 811).
CAN: BC, YT USA: AK, OR, WA
This species is placed in the metallica group by Ledoux and Roux (2005: 240).
This subspecies is known only from Beaver and Piute Counties in southern Utah (Kavanaugh 1978: 813).
USA: UT
This subspecies is known only from southern Utah [see Kavanaugh 1984: Fig. 32]. One specimen labeled from Navajo County in Arizona seen by Kavanaugh (1984: 165) is likely mislabeled.
USA: UT
This subspecies is restricted to the Henry Mountains in south-central Utah [see Kavanaugh 1979a: Fig. 70].
USA: UT
This species is endemic to the Trinity Alps of northwestern California.
USA: CA
This species is found along the Rocky Mountains in Colorado and New Mexico (Kavanaugh 1978: 813-815). The record from Idaho (LeConte 1879d: 500) is probably in error.
USA: CO, NM
This beautiful subspecies is recorded only from the type locality in Gros Ventre Mountains, western Wyoming [see Kavanaugh 1979a: Fig. 69].
USA: WY
This subspecies is restricted to the Rocky Mountains in western Alberta and southeastern British Columbia [see Kavanaugh 1979a: Fig. 69].
CAN: AB, BC
This species is placed in the metallica group by Ledoux and Roux (2005: 240).
This species is known only from the Sierra Blanca and Capitan Mountains in central New Mexico [see Kavanaugh 1984: Fig. 32].
USA: NM
This species is endemic to the Steens Mountains in south-central Oregon [see Kavanaugh 1984: Fig. 32].
USA: OR
This subspecies ranges from Medicine Bow and Sierra Madre Ranges in southern Wyoming south to the Rampart Range in central Colorado [see Kavanaugh 1984: Fig. 32].
USA: CO, WY
This subspecies ranges along the Rocky Mountains from southern Montana and southeastern Idaho south to northeastern Nevada, southern Utah, and west-central Wyoming [see Erwin and Ball 1972: Fig. 42)].
USA: ID, MT, NV, UT, WY
The form tetonensis is retained as a valid subspecies by Ledoux and Roux (2005: 285).
This subspecies is known only from the Olympic Mountains and the Cascade Range of Washington [see Kavanaugh and Schoville 2009: Fig. 13].
USA: WA
This subspecies is restricted to the Cascade Range in Oregon from Mount Hood south to the Three Sisters area [see Kavanaugh and Schoville 2009: Fig. 13].
USA: OR
Sixty-five species in the Palaearctic Region, one of them adventive in North America.
This Palaearctic species is adventive in North America where it is known from the Willamette Valley in northwestern Oregon and southwestern Washington [see LaBonte 2011: Fig. 4]. Since over 3, 000 specimens have been collected, the species is clearly established in the region. The first inventoried specimen found in the area was caught in late 2007 (Kavanaugh and LaBonte 2008: 482). The species has also been collected twice in eastern North America, in Quebec in 1930 and Saint Pierre and Miquelon in 1937 (Lindroth 1961a: 78) but it did not become established in this part of the continent.
USA: OR, WA – Adventive
This tribe includes a single genus.
About 55 species in the arctic, subarctic, boreal, and temperate areas of the Nearctic (15 species, of which two are adventive), Neotropical (two species in mountains of Middle America), and Palaearctic (about 40 species) Regions, including northern Africa (see Barševskis 2007). Two species, Notiophilus aquaticus and Notiophilus borealis, are Holarctic.
Lindroth (1961a) reviewed the North American species and provided a key for their identification. One adventive species (Notiophilus palustris) has been discovered subsequently in eastern Canada.
Notiophilus rufipes Curtis is known in North America from one specimen collected in Ware County, Georgia (Barševskis 2004). In my opinion, the specimen could be mislabeled and therefore the species is not considered as a North American entity.
This species ranges from Nova Scotia (Lindroth 1961a: 94) to southeastern Minnesota (Donald P. Schwert pers. comm. 1989), south at least to southeastern Nebraska (Richardson County, Foster F. Purrington pers. comm. 2009), central Missouri (Boone County, CMNH), northeastern Georgia (Leng 1910: 73; Fattig 1949: 11), and southwestern South Carolina (Ciegler 2000: 20). Two specimens labeled from the Santa Catalina Mountains in Arizona (MCZ, collection Fall) are known and so the species probably ranges farther west in southern United States.
CAN: NB, NS, ON, PE, QC USA: AZ, CT, DC, DE, GA, IA, IL, IN, MA, MD, ME, MI, MN, MO, NC, NE, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV
The range of this circumpolar species extends from Iceland to the Bering Sea Coast (Bousquet and Barševskis 2003: 96) and from Alaska (Lindroth 1961a: 95) to Newfoundland (Lindroth 1955a: 34), south to Pennsylvania (Capogreco 1989b: 4) and to New Mexico and Arizona (Fall 1906: 85; Lindroth 1961a: 95) along the Rocky Mountains. Fossil remnants from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 18).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), ON, QC, SK, YT USA: AK, AZ, CO, ID, IL, MA, ME, MI, MN, MT, NH, NM, NY, OH, PA, UT, VT, WI, WY – Holarctic
Notiophilus aquaticus (Linnaeus). This species is an example of a circumboreal taxon; it is found in the Northern Hemisphere without major gaps north of latitude 45. Linnaeus gave this species the epithet aquaticus in the 10th edition of his Systema Naturae, published in 1758, on the assumption that the species lived close to water. We know today that this is not the case and the species is found in relatively dry, open habitats. Notiophilus larvae and adults prey on collembolans. The adults are challenging to catch by hand because of their small size and swiftness.
This Palaearctic species is adventive in North America where it is known from Newfoundland and Saint Pierre and Miquelon (Lindroth 1955a: 37) to the Gaspé Peninsula in Quebec (LeSage 1996: 23), south to Connecticut (Krinsky and Oliver 2004: 396) and “Rhode Island” (Sikes 2003: 7), and from southwestern British Columbia (Lindroth 1961a: 101). The first inventoried specimen found on the east side of this continent was caught in Newfoundland in 1923 (Brown 1950b: 197) and on the west coast in Vancouver in 1957 (Lindroth 1961a: 101). Dejean’s (1831: 589) record of this species from «Amérique septentrionale» almost certainly refers to Notiophilus novemstriatus LeConte.
FRA: PM CAN: BC, NB, NF, NS, PE, QC USA: CT, ME, NH, RI – Adventive
This species is known from the Far East (Bousquet and Barševskis 2003: 96) and from Alaska (Lindroth 1961a: 97) to Newfoundland (Lindroth 1955a: 36); isolated on some mountains of New England (Lindroth 1961a: 96-97) and New York (Essex County, CNC, MCZ), and also known from northern Wyoming (Johnson County, CMNH). Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, SK, YT USA: AK, ME, NH, NY, VT, WY – Holarctic
This species occurs in the western mountain ranges, from southwestern Alberta and southern British Columbia, south to northeastern California (Lindroth 1961a: 98), northeastern Arizona, and northern New Mexico [see Morgan and Morgan 1979: Fig. 5]. Fossil remnants, dated about 11, 800 years B.P., have been unearthed in northeastern Wisconsin (Morgan and Morgan 1979: 232).
CAN: AB, BC USA: AZ, CA, CO, ID, MT, NM, OR, UT, WA, WY
This species is known from scattered localities from Newfoundland (Lindroth 1955a: 37) to the Gulf of Alaska coast (Lindroth 1961a: 98); also known from northwestern Minnesota (Clearwater County, CNC).
CAN: BC, LB, MB, NF, QC USA: AK, MN
This species is restricted to some mountains in New England (Lindroth 1961a: 100) and the Adirondacks in northeastern New York (Notman 1928: 211).
USA: MA, ME, NH, NY, VT
This species is found from Vancouver Island (Lindroth 1961a: 99) to northwestern Montana (Russell 1968: 46), south to southern Oregon (Harney County, CMNH). The record from Texas (Fall 1906: 90) is probably in error.
CAN: BC (VCI) USA: ID, MT, OR, WA
The range of this species extends from western Maine (Oxford County, André Larochelle pers. comm. 1990) to “South Dakota” (Kirk and Balsbaugh 1975: 15), south to east-central Texas (Riley 2011) and northern Florida (Peck and Thomas 1998: 15), west along the southwest to “Arizona” (Lindroth 1961a: 101) including northwestern Colorado (Barševskis 2009: 138). The record from Cape Breton Island in Nova Scotia (McCorquodale 2000: 339) is based on a misidentified specimen of Notiophilus palustris.
USA: AL, AR, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT
This Palaearctic species is adventive in North America where it is known only from Nova Scotia, including Cape Breton Island (McCorquodale 2000: 339, as Notiophilus novemstriatus), and Prince Edward Island (Larochelle and Larivière 1990b: 211). The first inventoried specimen found on this continent was caught in Halifax, Nova Scotia in 1968 (CNC).
CAN: NS (CBI), PE – Adventive
This species is known from California (Fall 1906: 91; Lindroth 1961a: 99), as far north as the San Francisco Bay area (Alameda County, MCZ), Arizona (Wickham 1898: 300; Lindroth 1961a: 99), and the states of Sonora and Chihuahua in Mexico (Erwin 2007a: 59). The record from “Oregon” (Fall 1906: 91) needs confirmation.
USA: AZ, CA [OR] – Mexico
This species is found from Labrador to central Alaska, south to northern British Columbia (Lindroth 1961a: 94-95), New Mexico (Fall 1906: 84; Lindroth 1961a: 94) along the Rocky Mountains, Oklahoma (Latimer County, UASM), southwestern Arkansas (Columbia County, INHS), east-central Alabama (Lee County, CNC), central Georgia (Fattig 1949: 11), and southern South Carolina (Ciegler 2000: 20); also recorded from Sakhalin Island (Barševskis 2001: 38).
CAN: AB, BC, LB, MB, NB, NS (CBI), ON, QC, YT USA: AK, AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, MI, MN, MO, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, SC, SD, TN, VA, WI, WV – Holarctic
As far as known, this species is restricted to the Sierra Nevada in California (Fall 1906: 90; Lindroth 1961a: 99).
USA: CA
This species is found in the western mountain ranges from western Alberta and British Columbia (Lindroth 1961a: 97) south at least to central Oregon (Crook County, CNC) and southwestern Colorado (Elias 1987: 632). The record from Houston, Texas (Fall 1906: 87) needs confirmation; that from Alaska (Fall 1926a: 129) refers to Notiophilus intermedius (Lindroth 1961a: 97).
CAN: AB, BC USA: CO, ID, MT, OR, UT, WA, WY [TX]
This species ranges from southern Alaska (Lindroth 1961a: 100) to northern California, at least as far south as Mendocino County (Fall 1906: 89), east to northern Idaho (Hatch 1953: 61). Fossil remnants, dated from the Late Pleistocene, have been unearthed in northeastern Siberia (Kiselev 1981: 12).
CAN: BC (QCI, VCI) USA: AK, CA, ID, OR, WA
Worldwide, with about 1, 300 species (Lorenz 2005: [i]) arrayed in four tribes: Carabini (about 1, 080 species), Ceroglossini (eight South American species), Cychrini (about 200 species), and Pamborini (13 species).
Northern Hemisphere and mountains in Mexico, with about 200 species arrayed by most authors in four genera: Cychropsis Boileau (about 25 Asian species), Cychrus (about 115 species), Scaphinotus (55 species), and Sphaeroderus (six species).
Six species restricted to the boreal and temperate regions of eastern North America.
Lindroth’s (1961a) key covers four of the species currently recognized; Sphaeroderus indianae was not included and Sphaeroderus schaumii was considered a junior synonym of Sphaeroderus nitidicollis. A taxonomic revision of the genus is needed.
This species is found along the southern parts of the Appalachians from North Carolina and Tennessee (Darlington 1932: 152) to northern Alabama (Löding 1945: 11) and northeastern Georgia (LeConte 1853c: 399; Leng 1910: 73).
USA: AL, GA, NC, SC, TN
Roeschke (1907a: 263) and Lindroth (1961a: 29) regarded this form as a subspecies of Sphaeroderus stenostomus (Weber).
The range of this subspecies extends from Cape Breton Island to southern Ontario (Lindroth 1961a: 28), south to the Black Mountains in North Carolina (Darlington 1933a: 64). The record from “South Carolina” (Bousquet and Larochelle 1993: 78) refers to Sphaeroderus canadensis lengi; that from “Michigan” (Bousquet and Larochelle 1993: 78) needs confirmation.
CAN: NB, NS (CBI), ON, QC USA: CT, KY, MA, MD, ME, NC, NH, NY, OH, PA, TN, VA, VT, WV [MI]
This subspecies occurs in the mountains south of the French Broad River (Barr 1969: 76) from southwestern North Carolina and adjacent parts of Tennessee (Darlington 1933a: 64) to northeastern Georgia (Fattig 1949: 9) and northwestern South Carolina (Ciegler 2000: 30).
USA: GA, NC, SC, TN
This species ranges from northwestern Ohio (Barr 1974a) to central Indiana (Blatchley 1910: 42), south to north-central Mississippi (Grenada County, Drew A. Hildebrandt pers. comm. 2009) and southwestern Virginia (Barr 1974a).
USA: IN, KY, MS, OH, TN, VA, WV
This species ranges from Newfoundland (Lindroth 1955a: 23) to east-central Saskatchewan (Hooper 1978: 19), south to northeastern Minnesota (Gandhi et al. 2005: 923), northern Wisconsin along Lake Superior (Wickham 1896c: 131), the Adirondack Mountains in northeastern New York (Notman 1928: 208), and New England [see Lindroth 1963a: Fig. 68]. The record from southwestern Ohio (Dury 1906: 257) is probably in error. Roeschke (1907a: 268) statement that this species is found “Küstengebiet von Maine bis Washington D.C.” is in error. Fossil remnants, dated between 22, 200 and 25, 200 years B.P., have been unearthed in central Illinois (Schwert 1992: 76).
CAN: MB, NB, NF, NS (CBI), ON, QC, SK USA: ME, MN, NH, NY, VT, WI
Lindroth (1961a: 29) retained brevoorti as “a weak subspecies, at most.” I studied several specimens from Newfoundland, the Maritimes, and Quebec, including Anticosti Island, and found no structural differences between populations of the forms nitidicollis and brevoorti except for the fact that adults of the nitidicollis form are on average slightly larger than those of the brevoorti form.
This species is known from a small area from Michigan to northern Virginia (Barr 1974a) and western Maryland (Bailey et al. 1994: 320). The record from “Illinois” (Bousquet and Larochelle 1993: 78) is probably in error.
USA: DC, MD, MI, OH, VA, WV
Roeschke (1907a: 266), Lindroth (1961a: 29), Lorenz (2005: 64), and Erwin (2007a: 170) listed this taxon as a subspecies of Sphaeroderus nitidicollis Guérin-Méneville. Thomas C. Barr, Jr. (pers. comm. 1977) believes it represents a distinct species, though closely related to Sphaeroderus nitidicollis.
This subspecies is found from Newfoundland (Lindroth 1955a: 21) to southeastern Manitoba (Lindroth 1961a: 29), south to east-central Iowa (Wickham 1888: 81, as Cychrus stenostomus; Lindroth 1961a: 29), northeastern Mississippi (Tishomingo County, CNC), northern Alabama (Löding 1945: 11; Madison County, CNC), northern Georgia (Fattig 1949: 9), and southern South Carolina (Ciegler 2000: 30). The records from east-central Missouri (Summers 1873: 133) and southeastern Louisiana (Summers 1874a: 79) need confirmation; that from “Saskatchewan” (Bousquet and Larochelle 1993: 78) is in error.
CAN: MB, NB, NF, NS (CBI), ON, PE, QC USA: AL, CT, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, MS, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [LA, MO]
This mainly coastal subspecies ranges from southeastern New York (Barr 1974a) and southeastern Pennsylvania (York County, CMNH) south to Wake County in east-central North Carolina (Brimley 1938: 115). The record from northern Georgia (Fattig 1949: 9) probably refers to Sphaeroderus stenostomus lecontei.
USA: DC, DE, MD, NC, NJ, NY, PA, VA, WV
Northern Hemisphere, with about 115 species (Lorenz 2005: 64-66) in the Nearctic (two western species) and Palaearctic Regions.
Gidaspow (1973) revised the North American species. Lindroth (1961a: 25-26) covered the species in his monograph of the Canadian and Alaskan Carabidae.
This subspecies is known from southwestern Idaho, western Wyoming, and northern Utah [see Gidaspow 1973: Fig. 7].
USA: ID, UT, WY
This subspecies is found from southeastern British Columbia south to northeastern Oregon (LaBonte 1988: 264), northern Idaho, and the Bitter Root Mountains in western Montana (Roeschke 1907a: 227) [see Gidaspow 1973: Fig. 7]. One specimen labeled from Logan Canyon in northern Utah is also known (Gidaspow 1973: 98).
CAN: BC USA: ID, MT, OR, WA [UT]
Lindroth (1961a: 26) treated this form as a valid species but both Roeschke (1907a: 227) and Gidaspow (1973: 96) regarded it as a subspecies of Cychrus hemphillii Horn.
This species ranges from British Columbia, including the Queen Charlotte Islands and Vancouver Island, south at least to southern Oregon [see Gidaspow 1973: Fig. 7], possibly even Mendocino County in northern California (Roeschke 1907a: 229). The record from “Alaska” (Roeschke 1907a: 229) is likely in error.
CAN: BC (QCI, VCI) USA: OR, WA [CA]
Cychrus tuberculatus (Harris). This species belongs to a genus that contains numerous species in the Palaearctic Region but only two in North America, both west coastal elements. The two species are morphologically very similar and likely sister-species, suggesting that a single ancestral stock crossed Beringia. Stomis and Trechoblemus are other carabid genera well represented in the Palaearctic with a single species each on the West Coast of North America.
Fifty-five species in the boreal and temperate regions of North America and the Sierra Madre Occidental in Mexico.
Roeschke (1907a) reviewed the species known at the time.
Nine species in North America (seven species) and the Sierra Madre Occidental of Mexico (two species).
Van Dyke (1938) reviewed the species and provided a key for their identification. One new species was described subsequently by Allen and Carlton in 1988.
[elevatus group]
The range of this subspecies extends from southern Manitoba south to “Iowa” (Lindroth 1961a: 18; Esau and Peters 1975: 510), northwestern Nebraska, and north-central New Mexico (Van Dyke 1938: 123-124). The record from Wisconsin (Rauterberg 1885: 12, as Cychrus elevatus) needs confirmation.
CAN: MB USA: CO, IA, MN, ND, NE, NM, SD [WI]
This subspecies ranges from southern New Hampshire (Merrimack and Strafford Counties, Donald S. Chandler pers. comm. 2008) south to the Florida Panhandle (Peck and Thomas 1998: 16), west to eastern Texas (Lamar and Fort Bend Counties, Brian Raber pers. comm. 2010; Riley 2011). The records from Maine (Bousquet and Larochelle 1993: 79) and Lawrence in Kansas (Van Dyke 1938: 120) need confirmation.
USA: AL, CT, DC, DE, FL, LA, MA, MD, MS, NC, NH, NJ, NY, PA, RI, SC, TX [KS, ME]
This subspecies ranges from Nebraska to Ohio, south to Tennessee, Arkansas (Van Dyke 1938: 121), and southwestern Oklahoma (Kondratieff et al. 2005: 173). The record from “Louisiana” (Roeschke 1907a: 142) needs confirmation.
USA: AR, IA, IL, IN, KS, MO, NE, OH, OK, TN [LA]
Van Dyke (1938: 121) noted that “the subspecies grades gradually into typical elevatus along its eastern boundary and into coloradensis along its western boundary.”
This subspecies is known so far only from the holotype collected in southeastern Virginia.
USA: VA
This subspecies is known so far only from the holotype and several specimens collected at the type locality (Eric van den Berghe and Robert L. Davidson pers. comm.) in southern New Mexico.
USA: NM
According to Roeschke (1907a: 141), this subspecies is found along the coast from New Jersey to South Carolina. Van Dyke (1938: 122) saw specimens from Virginia and North Carolina and noted that “it is apparently confined to the more maritime or coastal portion of the South Atlantic states, replacing almost entirely the [nomino]typical phase in its area of distribution.”
USA: NJ, NC, SC, VA
This species is known only from mountains in southwestern New Mexico in Grant County and other places in the neighborhood of Silver City (Van Dyke 1938: 104).
USA: NM
This subspecies is confined to the Rincon and Huachuca Mountains of southern Arizona (Ball 1966c: 711-712).
USA: AZ
This subspecies is known from the Santa Catalina Mountains in Pima County (Ball 1966c: 711) and Aravaipa Canyon in Pinal County (McCleve 1979: 452), southern Arizona.
USA: AZ
This subspecies is confined to the Chiricahua Mountains in Cochise County, southeastern Arizona (Ball 1966c: 714).
USA: AZ
This subspecies is found in the Pinaleño and White Mountains of southeastern Arizona (Ball 1966c: 713-714).
USA: AZ
This subspecies is known only from Mount Wrightson in the Santa Rita Mountains, southern Arizona (Ball 1966c: 715).
USA: AZ
This subspecies is confined to the Pinal and Sierra Ancha Mountains in Gila County, eastern Arizona (Ball 1966c: 709).
USA: AZ
This subspecies is restricted to high mountains in Coconino County, northern Arizona (Van Dyke 1938: 103).
USA: AZ
Van Dyke (1938: 102) treated this form as a species but Ball (1966c: 693) retained it as a subspecies of Scaphinotus snowi.
This subspecies is found in western New Mexico, eastern Arizona, southwestern Colorado, and southeastern Utah (Van Dyke 1938: 100).
USA: AZ, CO, NM, UT
This species is confined to southern Coconino, Yavapai, and Gila Counties in Arizona (Van Dyke 1938: 106).
USA: AZ
Van Dyke (1938: 106) retained Scaphinotus fuchsi Roeschke as a valid subspecies of Scaphinotus vandykei Roeschke.
[unicolor group]
This species is known only from the type locality in western Arkansas.
USA: AR
This species ranges from New Jersey (Smith 1910: 199) and Maryland (Van Dyke 1938: 128) to southwestern Illinois (Jackson County, Robert L. Davidson pers. comm. 2012), south to east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), northeastern Mississippi (Snodgrass and Cross 1983: 15), and the Florida Panhandle (Peck and Thomas 1998: 16).
USA: AL, AR, DC, FL, GA, IL, IN, KY, LA, MD, MO, MS, NC, NJ, OH, SC, TN, VA
Three eastern North American species.
There is no published key for the identification of these species.
This species ranges along the Appalachian Mountains from southwestern Virginia (Hoffman et al. 2006: 18) to northern Georgia (Towns County, CMNH).
USA: GA, NC, TN, VA
This taxon has been treated as an aberration of Scaphinotus viduus (Dejean) by Roeschke (1907a: 144) but according to Barr (1969: 73) and Hoffman et al. (2006: 18) it represents a distinct species.
This species ranges from Nova Scotia (CNC) to northern Indiana (Barr 1969: 74), south to northeastern Georgia (Fattig 1949: 8) along the Appalachian Mountains. The records from northern Florida (Harris 1839: 192) and “Louisiana” (Roeschke 1907a: 145) are probably in error.
CAN: NB, NS, QC USA: CT, DC, DE, GA, IN, MA, ME, MD, NC, NH, NJ, NY, OH, PA, VA, VT, WV
This species is known from a few localities in Virginia (Bell 1959: 11), northeastern West Virginia (Hampshire and Randolph Counties, CMNH), the Allegheny Plateau in southwestern Pennsylvania (Allegheny and Fayette Counties, Robert L. Davidson pers. comm. 2008), and the Big Black Mountain in southeastern Kentucky (Davis and Barbour 1978: 139).
USA: KY, PA, VA, WV
Hoffman et al. (2006: 18) reported that distinction between this taxon and Scaphinotus viduus is controversial at best and that adults of the two may be conspecific.
Five eastern North American species.
Van Dyke (1936) reviewed the species. Gidaspow (1973: 78-89) revised the species and provided a key for their identification. One new species was subsequently described by Allen and Carlton in 1988.
This species ranges from Cape Breton Island to southeastern Manitoba, south to eastern Kansas (Popenoe 1877: 22), southeastern Missouri, and Massachusetts [see Gidaspow 1973: Fig. 6]. The record from southeastern Pennsylvania (Rathvon 1869: 524) is likely in error.
CAN: MB, NB, NS (CBI), ON, QC USA: IA, IL, KS, MA, ME, MI, MN, MO, NE, NH, NY, OH, VT, WI
This species is known from eastern Kansas, western Missouri, western Arkansas (Allen and Thompson 1977: 32; Allen and Carlton 1988: 136), and central Oklahoma [see Gidaspow 1973: Fig. 6]. The records from “Iowa” and “Texas” (Leng and Beutenmüller 1894: 138) are likely in error; that from Buffalo, New York (Leng and Beutenmüller 1894: 138), is in error.
USA: AR, KS, MO, OK
This species ranges from southeastern Minnesota (Gandhi et al. 2005: 923) south to northern Arkansas (Allen and Thompson 1977: 32; Allen and Carlton 1988: 136), west to eastern Kansas [see Gidaspow 1973: Fig. 6]. The record from “Texas” (Schwarz 1895: 270) is probably in error.
USA: AR, IA, IL, KS, MN, MO
This species is known only from Newton County in northwestern Arkansas.
USA: AR
This species, as far as known, is restricted to Arkansas (Stone County, CNC), western Louisiana, and eastern Texas [see Gidaspow 1973: Fig. 6].
USA: AR, LA, TX
This subgenus includes ten species-group taxa found along the Appalachians. Several undescribed taxa are known (Thomas C. Barr, Jr. pers. comm. 2009).
There is no recent systematic treatment of the species of Maronetus and such work is much needed. A key to the species of the debilis group has been published (Barr 2009).
[debilis group]
This subspecies has been recorded so far only from the Black Mountains in southwestern North Carolina. Roeschke (1907a: 160) reported that this form occurs above 5000 feet of altitude.
USA: NC
This subspecies is known along the Appalachian Mountains from western North Carolina (Leng and Beutenmüller 1894: 138), northeastern Georgia (Fattig 1949: 9), and northwestern South Carolina (Ciegler 2000: 32). It is found from 1800 to 5000 feet (Roeschke 1907a: 160).
USA: GA, NC, SC
This species is known only from the holotype collected in southwestern Virginia.
USA: VA
This species is known from the Cumberland Plateau in southeastern Kentucky and the Appalachian Valley in southwestern Virginia (Barr 1969: 76; Barr 2009: 314).
USA: KY, VA
This species is known from two specimens collected in Cumberland and Anderson Counties, eastern Tennessee (Barr 2009: 315).
USA: TN
[hubbardi group]
This species is known along the Appalachian Mountains in eastern Tennessee and western North Carolina (Barr 1969: 76).
USA: NC, TN
This species is known from southwestern Virginia (Hoffman et al. 2006: 18) and the Black Mountains in western North Carolina (Beutenmüller, 1913: 139).
USA: NC, VA
[imperfectus group]
This species ranges from southeastern Ohio and Pennsylvania (Barr 1969: 76) south to western North Carolina (Thomas C. Barr, Jr. pers. comm. 2009; Leng and Beutenmüller 1894: 138).
USA: MD, NC, OH, PA, VA, WV
This species is endemic, as far as known, to the Black Mountains (Barr 1969: 75) in southwestern North Carolina.
USA: NC
Barr (1969: 75) listed this taxon as a subspecies of Scaphinotus imperfectus.
This species is found in the Blue Ridge Mountains (Great Balsams and Highlands Plateau) and the Nantahala Mountains (Barr 1969: 75) in southwestern North Carolina and northeastern Georgia.
USA: GA, NC
Seven species restricted to the Appalachian region.
Valentine (1935) reviewed the species and provided a key for their identification.
This species is endemic to the Black Mountains in western North Carolina (Barr 1969: 74). The record from the mountains of Tennessee (Roeschke 1907a: 147) needs confirmation.
USA: NC [TN]
This subspecies is found in the mountains of southwestern Virginia, western North Carolina (Valentine 1936: 227), and northeastern Tennessee (Carter County, CMNH).
USA: NC, TN, VA
This subspecies is known only from Orange and Guilford Counties in northern North Carolina (Valentine 1936: 226). The record from “Tennessee” (Roeschke 1907a: 147) needs confirmation.
USA: NC [TN]
This subspecies ranges along the Appalachian Mountains from the Great Smokies in eastern Tennessee and western North Carolina eastwards to the Saluda Mountains (Barr 1970: 4) in northwestern South Carolina.
USA: NC, SC, TN
This subspecies is found along the Appalachians in southern West Virginia (Fayette and Webster Counties, CMNH), eastern Kentucky, southwestern Virginia, and northeastern and southeastern Tennessee (Valentine 1936: 231; Davis and Barbour 1978: 139). The records from Pennsylvania (Roeschke 1907a: 148), southwestern Ohio (Wright and Whitehouse 1941: 70), and southern Indiana (Blatchley 1910: 43) probably refer to other subspecies of the species.
USA: KY, TN, VA, WV
This subspecies ranges from southwestern Pennsylvania and southern Ohio to north-central Kentucky (Valentine 1936: 232) and northwestern Maryland (Glaser 1996: 4).
USA: KY, MD, OH, PA, WV
This subspecies is known in the Appalachians from the Nantahala Mountains, adjacent Blue Ridge, and the Unicoi Mountains in southwestern North Carolina, southeastern Tennessee, and northeastern Georgia (Valentine 1936: 227; Barr 1970: 4).
USA: GA, NC, TN
This subspecies is known only from southeastern Kentucky (Pulaski County, CMNH) and the Walden Ridge in southeastern Tennessee (Valentine 1935: 357).
USA: KY, TN
This species is found along the Appalachians from south-central West Virginia (Fayette County, CMNH) and Virginia (Valentine 1935: 363) to northern Georgia (Rabun, Towns, and Union Counties, CMNH) and northwestern South Carolina (Ciegler 2000: 31).
USA: GA, NC, SC, TN, VA, WV
This subspecies is found from central Tennessee (Barr 1969: 74) to west-central Alabama (Löding 1945: 11).
USA: AL, TN
This subspecies is known only from eastern Alabama (Löding 1945: 11).
USA: AL
This subspecies is known from the Appalachian Plateau in southwestern Pennsylvania and the Allegheny Mountains in West Virginia (Barr 1969: 75), western Virginia (CMNH), and northwestern Maryland (Bailey et al. 1994: 320).
USA: MD, PA, VA, WV
This subspecies is known from northwestern West Virginia and a few counties in northern and western Virginia (Valentine 1935: 367). The record from “Tennessee” (Roeschke 1907a: 150) needs confirmation.
USA: VA, WV [TN]
This species is found at high altitudes in the Great Balsam, Plott Balsam, Great Smoky (Barr 1969: 74), and Nantahala Mountains (Barr 1970: 5) in eastern Tennessee, western North Carolina, and northeastern South Carolina.
USA: NC, SC, TN
This species has been reported from the Appalachians in western North Carolina, northern Georgia (Valentine 1935: 358), and northwestern South Carolina (Ciegler 2000: 32).
USA: GA, NC, SC
Four western North American species.
Gidaspow (1973: 73-78) revised the species and provided a key for their identification.
This species is restricted to southeastern Washington (Gidaspow 1973: Fig. 4) and northeastern Oregon (Hatch 1953: 46; Westcott et al. 2006: 9).
USA: OR, WA
This species is known from western Montana (Russell 1968: 42), northern Idaho, Whitman County in southeastern Washington (CMNH), and the Creston area (Lindroth 1961a: 20) in southeastern British Columbia [see Gidaspow 1973: Fig. 4].
CAN: BC USA: ID, MT, WA
This species is known from southeastern British Columbia, Idaho, and eastern Washington [see Gidaspow 1973: Fig. 5].
CAN: BC USA: ID, WA
This form was listed as a synonym of Scaphinotus relictus (Horn) by Lindroth (1961a: 20), regarded as a variety of Scaphinotus relictus by Roeschke (1907a: 163) and Hatch (1953: 45), and treated as a valid species by Gidaspow (1973: 77).
This species is found in southern British Columbia, western Alberta, western Montana (Russell 1968: 42), northern Idaho, eastern Washington, and from one isolated locality in southwestern Oregon [see Gidaspow 1973: Fig. 5]. Old specimens simply labeled from California are also known (Gidaspow 1973: 77).
CAN: AB, BC USA: ID, MT, OR, WA [CA]
Four species restricted to the Pacific coastal and western montane regions of North America.
Van Dyke (1944) and Gidaspow (1973) revised the species then placed in this subgenus. Gidaspow (1973) provided a key for their identification.
This species ranges from Kodiak Island in the Gulf of Alaska to northwestern California, east at least to eastern British Columbia [see Gidaspow 1973: Fig. 2]. The record from Torrington, Alberta (Gidaspow 1973: 68) is probably based on a mislabeled specimen.
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is known only from Waldo Lake area in western Oregon (Gidaspow 1973: 66; CNC, CMNH).
USA: OR
This rarely collected species is restricted to the southern part of Vancouver Island (Lindroth 1961a: 22) and the Olympic Mountains in northwestern Washington (Van Dyke 1924b: 3).
CAN: BC (VCI) USA: WA
This species has been placed in the subgenus Brennus by Lindroth (1961a: 22) and Gidaspow (1968: 155) but van den Berghe and Davidson (Robert L. Davidson pers. comm. 2008) agreed that this small species is a member of the subgenus Stenocantharus.
This species ranges from northwestern Oregon to the San Francisco Bay area in California [see Gidaspow 1973: Fig. 3]. The record from Vancouver Island (LeConte 1869c: 370) is probably in error.
USA: CA, OR
Fifteen western North American species of which two extend into the Baja California Peninsula.
Gidaspow (1968) revised the species and provided a key for their identification.
This species is found within the Sierra Nevada in central California, from El Dorado County to Sequoia National Park [see Gidaspow 1968: Fig. 9].
USA: CA
This species is restricted to central California [see Gidaspow 1968: Fig. 7].
USA: CA
This species ranges from Sonoma County in California to the Pacific Coast of Baja California Norte, east to the San Bernardino Mountains [see Gidaspow 1968: Fig. 10]. One specimen from Hidalgo state in Mexico is probably mislabeled, as pointed out by Gidaspow (1968: 176).
USA: CA – Mexico
This species ranges from southwestern Oregon south to the Los Angeles region in southern California, east to the northern part of the Sierra Nevada [see Gidaspow 1968: Fig. 2].
USA: CA, OR
The range of this species extends over much of California, including the Coast Ranges and the Sierra Nevada, as far south as Riverside County [see Gidaspow 1968: Fig. 6]. Some specimens simply labeled from Oregon are known.
USA: CA [OR]
The range of this species extends from the Aleutians Islands and the Gulf Coast of Alaska south to northern California, northern Arizona (Coconino County, Eric van den Berghe pers. comm. 2009), and northwestern Wyoming [see Gidaspow 1968: Fig. 4]. Gidaspow (1968: 149) considered the possibility that the species ranges further east toward the Hudson Bay because of the presence in collections of several specimens labeled from “Hudson Bay Territory.”
CAN: AB, BC (QCI, VCI) USA: AK, AZ, CA, ID, MT, OR, WA, WY
This species is known from the northern part of California, as far south as Madera County [see Gidaspow 1968: Fig. 5], and from Washoe County in northwestern Nevada (La Rivers 1947: 133, as Scaphinotus obliquus convergens). Some specimens simply labeled from Oregon are known.
USA: CA, NV [OR]
This species is known from central California [see Gidaspow 1968: Fig. 3] and from Grand County in eastern Utah (Gidaspow 1968: 165). The record from “Nevada” (Bousquet and Larochelle 1993: 84) needs confirmation.
USA: CA, UT [NV]
This species is found in the southern third of California, including Santa Catalina Island, and in “Baja California” [see Gidaspow 1968: Fig. 7].
USA: CA (CHI) – Mexico
This species is found in the southern half of California, in Tulare (Sequoia National Park), Fresno, Kern, and Los Angeles Counties [see Gidaspow 1968: Fig. 5].
USA: CA
This subspecies is found in southwestern Oregon (Westcott et al. 2006: 9) and northwestern California as far south as Colusa County [see Gidaspow 1968: Fig. 3].
USA: CA, OR
This subspecies is known for sure only from western Oregon as far south as the border with California [see Gidaspow 1968: Fig. 3]. The records from “California” (Casey 1897: 328, 332, as Brennus porcatus and Brennus compositus) need confirmation.
USA: OR [CA]
This species is found along western California from Mendocino County south to Los Angeles County [see Gidaspow 1968: Fig. 9].
USA: CA
This species is known only from central California, mostly along the west side of the Sierra Nevada from Calaveras County to Tulare and Kern Counties [see Gidaspow 1968: Fig. 8].
USA: CA
This species ranges from southern Oregon to San Luis Obispo County along the coast and to Yosemite National Park along the Sierra Nevada; also known from the Santa Catalina Island [see Gidaspow 1968: Fig. 10].
USA: CA, OR
Three species restricted to the Pacific coastal region of North America.
Van Dyke (1944) first reviewed the species. Subsequently, Gidaspow (1973) revised them and provided a key for their identification.
This species ranges from southwestern British Columbia, including Vancouver Island, to southwestern Oregon [see Gidaspow 1973: Fig. 1].
CAN: BC (VCI) USA: OR, WA
The range of this species extends near the Pacific Coast from Lincoln County in Oregon (Gidaspow 1973: 61) to Humboldt County in northern California [see Gidaspow 1973: Fig. 1].
USA: CA, OR
This species is known only from a few specimens collected in Humboldt and Mendocino Counties (Weber and Kavanaugh 1992: 394), northern California.
USA: CA
Worldwide, with about 1, 080 species arrayed in three genera: Aplothorax Waterhouse (one species on the island of Saint Helena in South Atlantic Ocean), Calosoma (about 170 species), and Carabus (about 910 species).
Worldwide, with about 170 species in the Nearctic (41 species, of which one is adventive), Neotropical (about 55 species, many shared with North America), Australian (three species), Oriental (six species), Palaearctic (about 45 species), and Afrotropical (about 35 species) Regions. The species are arrayed in about 25 genus-group taxa.
Gidaspow (1959) revised the North American species and provided a key for their identification. Subsequently Lindroth (1961a: 50, 55) listed in synonymy some of the species that were considered valid by Gidaspow (e.g., Calosoma concretum, Calosoma pimelioides, Calosoma zimmermani) and Dajoz (1997a) described a new species (Calosoma dawsoni).
The genus Calosoma is retained here in its wide sense following several authors, including Culot (1988). Others used different arrangements. For example, Lorenz (2005) and Erwin (2007a) listed Callisthenes Fischer von Waldheim as a valid genus with Chrysostigma Kirby and Callistenia Lapouge as subgenera.
Burgess and Collins (1917: 86) reported that Calosoma palmeri Horn “occurs in California and Mexico.” The record for California is doubtful since the species, as far as known, is endemic to Guadalupe Island (Gidaspow 1959: 276).
Western Hemisphere, with 14 species in the Nearctic (one species) and Neotropical (14 species) Regions.
This species ranges from Long Island, New York (Notman 1928: 209) to “Iowa” (Burgess and Collins 1917: 62), south to Guatemala (Gidaspow 1963: 301) and southern Florida except for the Keys (Peck and Thomas 1998: 15), west along southern United States to “California” (Burgess and Collins 1917: 62); also known from the Greater Antilles as far south as Puerto Rico (Gidaspow 1963: 301). The records from “Minnesota, ” “North Dakota, ” and “Wisconsin” (Bousquet and Larochelle 1993: 70) are probably in error or based on strays.
USA: AL, AR, AZ, CA, DC, FL, GA, IA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, SC, TN, TX, VA – Cuba, Dominican Republic, Guatemala, Haiti, Jamaica, Mexico, Puerto Rico
Northern Hemisphere, with six species in the Nearctic (two species, one of them adventive) and Palaearctic (five species) Regions.
This subgenus is retained in its narrow sense and excludes members of Australodrepa Lapouge and Calodrepa Motschulsky.
This species occurs from Cape Breton Island to the Skeena River drainage in west-central British Columbia (Lindroth 1961a: 47), south to northeastern Nevada (La Rivers 1947: 134), central Utah (La Rivers 1947: 135), southeastern Texas in the Galveston area (Snow 1906a: 140; Gidaspow 1959: 245), and northern Georgia (Fattig 1949: 11).
CAN: AB, BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CO, CT, GA, IA, IL, IN, LA, MA, MD, ME, MI, MN, MO, NC, ND, NE, NH, NJ, NV, NY, OH, OK, PA, RI, SD, TN, TX, UT, VA, VT, WI, WV
This European species was introduced as early as 1906 in many places in Canada and United States for the biological control of two introduced lymantriids: the gypsy moth, Lymantria dispar (Linnaeus), and the browntail moth, Euproctis chrysorrhoea (Linnaeus). Based on the extensive survey of Schaefer et al. (1999), the species is now established in eastern United States from southern Maine to Maryland and West Virginia, west to western Pennsylvania. The record from “Michigan” (Bousquet and Larochelle 1993: 71) must be in error. There is no confirmation that the species is established on the west coast and the record from “Washington” (Bousquet and Larochelle 1993) should be deleted.
USA: CT, DE, MA, MD, ME, NH, NJ, NY, PA, RI, VA, VT, WV – Adventive
Calosoma sycophanta (Linnaeus). This species is one of only two intentionally introduced carabids in North America that became established on this continent, the other one being Carabus auratus. These species were introduced for the biological control of the gypsy moth, Lymantria dispar, and the browntail moth, Euproctis chrysorrhoea. Since its release in the vicinity of Boston in 1906-1907, Calosoma sycophanta has spread to southern Maine and to West Virginia.
Western Hemisphere, with four species in the Nearctic (four species, one of them endemic) and Neotropical (three species) Regions.
This subgenus is listed in synonymy with the nominotypical subgenus by some authors (e.g., Breuning 1927: 155; Lorenz 2005: 68).
This species is found from the Rio Grande in southeastern Texas (Wickham 1897: 102; Burgess and Collins 1917: 122) south to Nicaragua (Gidaspow 1963: 283) [see Gidaspow 1959: Fig. 2].
USA: TX – Mexico, Nicaragua
This widely distributed species ranges from western Maine (Majka et al. 2011: 45) to northeastern North Dakota (Tinerella 2003: 635), including southern Quebec (only as strays) and Ontario (Lindroth 1961a: 46), south to Venezuela (Gidaspow 1963: 283) and southern Florida (Peck and Thomas 1998: 15), west along the southwest to “California” (Gidaspow 1959: 250) and Baja California (Leng 1915: 565). Two specimens, likely strays, have been collected in New Brunswick (Webster and Bousquet 2008: 16) and on Sable Island, Nova Scotia (Majka et al. 2007: 6). The species is not known from the West Indies.
CAN: NB, NS, ON, QC USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV – Guatemala, Mexico, Venezuela
This species is known from northeastern Georgia and southern Florida but only from a few century-old specimens (Gidaspow 1959: 248), and from the Bahamas (Turnbow and Thomas 2008: 12), Cuba, and Hispaniola (Gidaspow 1963: 283). Erwin (2007a: 104) listed this species also from Yucatán, Mexico.
USA: FL, GA – Bahamas, Cuba, Haiti, Dominican Republic, Mexico
This species ranges from southeastern New Hampshire (Rockingham County [probably only as strays], Donald S. Chandler pers. comm. 1992) to southeastern Minnesota (Donald P. Schwert pers. comm. 1989), including southern Quebec (only as strays) and southern Ontario (Lindroth 1961a: 47), south to southeastern Texas (Brazoria County, Brian Raber pers. comm. 2010), southeastern Louisiana (Saint John the Baptist and Saint Tammany Parishes, Igor M. Sokolov pers. comm. 2009), and central Georgia (Fattig 1949: 11). The record from “California” (Burgess and Collins 1917: 38) is likely in error or based on a stray.
CAN: ON, QC USA: AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI, WV
Three North American species, all of them extending into Mexico.
This subgenus is listed in synonymy with Carabosoma Géhin by some authors (e.g., Lorenz 2005: 70).
This species ranges from “Iowa” (Jaques and Redlinger 1946: 295, as Calosoma lugubre) to southeastern Colorado (Michels et al. 2008), south to southern Mexico (Gidaspow 1959: 254-255) and eastern Arkansas (Arkansas County, Ken Karns pers. comm. 2009). Gidaspow (1963: 282) reported the presence of two specimens from Costa Rica. The records from “Illinois” (Bousquet and Larochelle 1993: 71) and northern Arizona (Wickham 1896a: 156) need confirmation. The specimen labeled from Duparquet in Quebec (Lindroth 1961a: 49) is almost certainly mislabeled.
USA: AR, CO, IA, KS, MO, NE, NM, OK, TX [AZ, IL] – Costa Rica, Mexico
This species is found from central California to eastern Utah, south to southwestern New Mexico, Sonora in Mexico, and Baja California (Gidaspow 1959: 256).
USA: AZ, CA, NM, NV, UT – Mexico
This species is found from Inyo County in eastern California (Riley 1893: 239; Fall 1901a: 40) to central New Mexico, south to Sonora and the Baja California Peninsula (Gidaspow 1959: 255).
USA: AZ, CA, NM – Mexico
Western Hemisphere, with five species in the Nearctic (four species, one of them endemic) and Neotropical (four species, only one, Calosoma glabratum, endemic) Regions.
[angulatum group]
This species is found in Mexico south at least to Colombia and Venezuela (Erwin 1991a: 26). It is also occasionally found in southwestern United States from California to Texas (Gidaspow 1959: 253).
USA: AZ, CA, NM, TX – Colombia, Costa Rica, Guatemala, Honduras, Mexico, Nicaragua, Venezuela
[peregrinator group]
This species is found in southern California and northern Baja California (Gidaspow 1959: 259); it is also known from one locality in southwestern New Mexico (Gidaspow 1959: 259), from Montezuma County in Colorado (FFPC), and has been reported from Nevada by Erwin (2007a: 91).
USA: CA (CHI), CO, NM, NV – Mexico
The range of this species extends from Sacramento County in central California (Clark 1999: 202) to western Kansas (Popenoe 1877: 22), south to the Federal District in Mexico and Baja California (Gidaspow 1959: 258).
USA: AZ, CA, CO, KS, NM, OK, TX, UT – Mexico
This species is known from western Utah (Millard County, Ken Karns pers. comm. 2009), western Nevada, southern California, and “Arizona” (Gidaspow 1959: 260).
USA: AZ, CA, NV, UT
Three North American species, two of them extending into Mexico.
This species ranges from “Vermont” (Ross T. Bell pers. comm. 1989; probably only as strays) to “Nebraska, ” including southernmost Ontario (only as strays), south to “Texas” (Gidaspow 1959: 274) and southern Georgia (Fattig 1949: 10).
CAN: ON USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, TN, TX, VA, VT, WI
This species is known from “Arkansas” (Jeannel 1940: 214) to “New Mexico” (Erwin 2007a: 98), south to northeastern Mexico (Gidaspow 1959: 274) and “Louisiana” (Burgess and Collins 1917: 25).
USA: AR, LA, NM, OK, TX – Mexico
This species is known from “Colorado” (Gidaspow 1959: 275) and southeastern Arizona (Snow 1906b: 161) south at least to the state of Guerrero in Mexico (Gidaspow 1959: 275). The records from southern Kansas (Snow 1903: 192; Knaus 1905a: 218; Burgess and Collins 1917: 26) are doubtful (see Gidaspow 1959: 275).
USA: AZ, CO – Mexico
Seven Mexican species, one of them extending into North America.
This subspecies is known from southern Colorado (Wickham 1902: 231), northwestern Arizona, “New Mexico, ” Brewster County in western Texas (Gidaspow 1959: 281), and Chihuahua in Mexico (Erwin 2007a: 96).
USA: AZ, CO, NM, TX – Mexico
This subspecies is known from Mexico as far south as the Federal District and from Brewster County in western Texas (Gidaspow 1959: 281).
USA: TX – Mexico
Western Hemisphere, with ten species in North America (nine species) and Middle America (four species, only one, Chrysostigma ampliator Bates, being endemic).
This species ranges from southern Minnesota (Gandhi et al. 2005: 922) to the Pacific Coast in southern California, south to the state of Oaxaca in Mexico (Gidaspow 1959: 269). The records from Missouri (probably based on the type locality of Calosoma triste) and “Arkansas” (Burgess and Collins 1917: 87), possibly based on a stray, need confirmation.
USA: AZ, CA, CO, KS, MN, NE, NM, NV, OK, TX, UT [AR, MO] – Mexico
The range of this species extends from Newfoundland (Larson and Langor 1982: 592) and Saint Pierre and Miquelon (Lindroth 1955a: 28) to eastern British Columbia (Lindroth 1961a: 51), south to “Oregon” (Hatch 1953: 52), northeastern New Mexico (Casey 1897: 344), “Kansas” (Horn 1872c: 384), east-central Missouri (Summers 1873: 133), and northern Georgia (Fattig 1949: 11); several specimens have also been caught along the Slave and Mackenzie Rivers, up to 65°30'N, in Northwest Territories (White 1851: 357; Lindroth 1961a: 51). The records from southeastern Louisiana (Summers 1874a: 79), “Alabama, ” “Arkansas, ” and “Mississippi” (Bousquet and Larochelle 1993: 72) are probably in error.
FRA: PM CAN: AB, BC, MB, NB, NS (CBI), NT, ON, QC, SK USA: CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SD, TN, UT, VA, VT, WA, WI, WV, WY
Gidaspow (1959: 266) treated Calosoma concretum Casey as a valid species. However, both Lindroth (1961a: 50) and Obydov (2003: 534) retained the name as synonym of Calosoma calidum (Fabricius).
This species ranges from south-central British Columbia (Lindroth 1961a: 53) to north-central North Dakota (McHenry County, Foster F. Purrington pers. comm. 2009), south to “Arizona” (Gidaspow 1959: 263) and southern California along the coast (Casey 1913: 64-65, as Calosoma esuriens and Calosoma transversa). The record from “Indian Territory” (= Oklahoma) (Burgess and Collins 1917: 111) is probably in error.
CAN: BC USA: AZ, CA, ID, MT, ND, NV, OR, UT, WA
This species inhabits the Great Plains ranging from the southern parts of the Prairie Provinces south to Montana (Gidaspow 1959: 265; Lindroth 1961a: 51) and north-central South Dakota (Walworth County, CNC). The record from “Wyoming” (Bousquet and Larochelle 1993: 73) needs confirmation.
CAN: AB, MB, SK USA: MT, ND, SD [WY]
This species is known from southern California to “Colorado, ” south to Durango in western Mexico (Gidaspow 1959: 270).
USA: CA, CO, NM, NV – Mexico
This species ranges from Saskatchewan (Lindroth 1961a: 50) to eastern Oregon, south to northeastern Nevada (La Rivers 1947: 136), northwestern New Mexico (Casey 1897: 345, as Calosoma obsoleta microsticta; McKinley County, UASM) and southwestern Oklahoma (Kondratieff et al. 2005: 171), east to eastern Minnesota (Gandhi et al. 2005: 922). The record from “Texas” (Burgess and Collins 1917: 87) needs confirmation.
CAN: AB, MB, SK USA: CO, IA, ID, KS, MN, MT, ND, NE, NM, NV, OK, OR, SD, UT, WY [TX]
This species ranges from eastern Oregon and “Idaho” (Gidaspow 1959: 271) south to southern Arizona (Snow 1907: 141) and southern California along the coast (Fall 1901a: 40; Moore 1937: 4); also found on Guadalupe Island, Mexico (Gidaspow 1959: 271). The record from northeastern Kansas (Popenoe 1877: 22) is likely in error.
USA: AZ, CA (CHI), ID, OR, UT – Mexico
This species is confined to California where it is known from Yolo County to Riverside (Gidaspow 1959: 272) and San Diego Counties (Moore 1937: 4). The records from “Arizona, ” “Colorado, ” “Texas, ” and “Mexico” (Burgess and Collins 1917: 93) are probably in error.
USA: CA
This species is found from Vancouver Island (Lindroth 1961a: 52) to “Nebraska, ” south to “Colorado” (Gidaspow 1959: 264) and southern California (La Rivers 1947: 136). The records from “North Dakota” (Bousquet and Larochelle 1993: 73) and “Alberta” (Burgess and Collins 1917: 107) need confirmation.
CAN: BC (VCI) USA: AZ, CA, CO, ID, MT, NE, NV, OR, UT, WA, WY [AB, ND]
Obydov (2003: 536), based on a study of two specimens only, concluded that Calosoma cogitans Casey represents a valid subspecies of Calosoma tepidum LeConte. I agree with Gidaspow (1959: 263) and Lindroth (1961a: 52) that the name does not apply to a distinct taxon.
Fourteen North American species in the western half of the continent.
This species is known only from the original 16 specimens collected at the type locality in the Sierra Nevada.
USA: CA
This species has been recorded so far from the Sierra Nevada in Tulare County and “South Fork” in Humboldt County, California (Gidaspow 1959: 308).
USA: CA
This species is found in California from El Dorado and Sacramento Counties to Santa Cruz and Madera Counties (Gidaspow 1959: 308); it was cited also from Washington (Burgess and Collins 1917: 119) but the record is probably in error or based on a stray.
USA: CA
According to La Rivers (1947: 137), this species is “apparently the predominant Calosoma of eastern Nevada, and a marked montane isolate.”
USA: NV
This species is found in California from Sacramento County to Los Angeles and San Bernardino Counties (Gidaspow 1959: 307); according to Burgess and Collins (1917: 122), it was also collected in Reno, western Nevada.
USA: CA, NV
This species ranges from southern Saskatchewan to the Okanagan Valley in south-central British Columbia (Lindroth 1961a: 56), south to southeastern California, northwestern New Mexico (Casey, 1913: 75, as Callisthenes diffractus), and northern Kansas [see Gidaspow 1959: Fig. 11, as Callisthenes luxatus, Callisthenes pimelioides, and Callisthenes zimmermanni]. At least one specimen simply labeled from New Mexico is known (Gidaspow 1959: 311). The records from “Oklahoma” and “Texas” (Burgess and Collins 1917: 121, as Callisthenes zimmermani) need confirmation.
CAN: AB, BC, SK USA: AZ, CA, CO, ID, KS, MT, NE, NM, NV, OR, SD, UT, WA, WY [OK, TX]
Gidaspow (1959) considered Callisthenes pimeloides Walker [synonyms: tegulatus Casey, viator Casey, pustulosus Casey, reflexus Casey, and parowanus Casey] and Carabus zimmermanni LeConte [synonyms: opacus Géhin, exaratus Casey, debilis Casey, and tahoensis Van Dyke] as distinct species but Lindroth (1961a: 55) argued that the structural differences noted between these forms are no more than intraspecific variation.
This species ranges from Saskatchewan to southeastern British Columbia (Lindroth 1961a: 53-54), south to “California, ” “Arizona, ” and “Nebraska” (Gidaspow 1959: 305). The records from “Vancouver Island” (Gidaspow 1959: 305) and “Northwest Territory, Canada” (Burgess and Collins 1917: 114) are probably in error.
CAN: AB, BC, SK USA: AZ, CA, ID, MT, NE, OR, WA
The range of this species extends from central Washington south at least to El Dorado County in California and western Nevada (Gidaspow 1959: 315); also recorded from “Utah” (Erwin 2007a: 79). One specimen simply labeled from Wyoming is known (Gidaspow 1959: 315). Notwithstanding Gidaspow (1959: 315), Hatch (1953: 54) did not record this species from Washington, Idaho, Oregon, and British Columbia and so the records from “Oregon” and “Idaho” (Bousquet and Larochelle 1993: 74) need confirmation.
USA: CA, NV, UT, WA [ID, OR, WY]
This species is known from four specimens without locality data from “Oregon” (Gidaspow 1959: 317).
USA: OR
This species is confined to California where it is found as far south as San Diego County along the coast and Tuolumne County in the Sierra Nevada (Gidaspow 1959: 310).
USA: CA
This species ranges from western Oregon (Hatch 1953: 54, as Calosoma striatus) to Santa Cruz County in California (Gidaspow 1959: 309) along the Coast Ranges.
USA: CA, OR
This species is known from “Washington, ” “Idaho, ” and “California” (Burgess and Collins 1917: 114; Gidaspow 1959: 305). The records from British Columbia (Hatch 1953: 53; Gidaspow 1959: 305) probably refer to Calosoma cancellatum Eschscholtz as pointed out by Lindroth (1961a: 53).
USA: CA, ID, WA
This rarely collected species is known from Klamath and Harney Counties in southern Oregon and Siskiyou County in northern California (Gidaspow 1959: 316); also recorded from “Nevada” (Erwin 2007a: 82).
USA: CA, NV, OR
This species is found west of the Rocky Mountains from south-central British Columbia south to “California” (Lindroth 1961a: 54).
CAN: BC USA: CA, ID, OR, WA
About 910 species (Lorenz 2005: 72-114) in North America (14 species), Mexico (two species), and the Palaearctic (about 895 species) and Oriental (about five species) Regions.
Van Dyke (1944) reviewed the North American species and provided a key for the identification of all species except Carabus auratus. Lindroth (1961a: 30-42) covered all but two species (Carabus finitimus and Carabus forreri).
Twenty-six species (Deuve 2004: 109-119) in the Nearctic (three species, one of them adventive) and Palaearctic (24 species) Regions.
Members of Archaeocarabus Semenov (38 Chinese species) are included in this subgenus by some authors (e.g., Lorenz 2005: 72-73).
[granulatus group]
This European subspecies is adventive in North America where it is known from Newfoundland (Larson and Langor 1982: 592) to southeastern Manitoba (Roughley et al. 2010: 230; CMNH), and from east-central Minnesota (Gandhi et al. 2011: 673), Massachusetts (Van Dyke 1945a: 129), and Connecticut (Middlesex County, William L. Krinsky pers. comm. 2012) in the east, and from western British Columbia (Lindroth 1961a: 37), including the Queen Charlotte Islands (Kavanaugh 2010: 385), western Washington (Hatch 1953: 51), and Edmonton, Alberta (UASM), in the west. The first inventoried specimen collected in the east was found in New Brunswick in 1890 (Lindroth 1961a: 37) and in the west in Seattle, Washington, in 1924 (Hatch 1933c: 117).
FRA: PM CAN: AB, BC (QCI, VCI), MB, NB, NF, NS, ON, PE, QC USA: CT, MA, MN, WA – Adventive
[vinctus group]
The range of this species extends from southern Maine (Dearborn and Donahue 1993: 2) to southeastern Minnesota (Donald P. Schwert pers. comm. 1989), including southernmost Ontario (Lindroth 1961a: 35), south to northeastern Mississippi (Snodgrass and Cross 1983: 14), northern Alabama (Löding 1945: 11), and northern Georgia (Fattig 1949: 10; CMNH). The record from Idaho (LeConte 1878a: 471; Hatch 1953: 51) is in error (Lindroth 1961a: 35); that from southeastern Louisiana (Summers 1874a: 79) needs confirmation.
CAN: ON USA: AL, CT, DC, DE, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, MS, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [LA]
This species ranges from Massachusetts (Harris 1833: 567; Frost 1910: 86) to southeastern Minnesota (Gandhi et al. 2005: 923), including southern Ontario (Lindroth 1961a: 34), south to southeastern Mississippi (Stone County, UASM), southwestern Alabama (Van Dyke 1945a: 126), and the Florida Panhandle (Peck and Thomas 1998: 16). The record from “Louisiana” (Summers 1874a: 79) is probably in error.
CAN: ON USA: AL, CT, DC, DE, FL, GA, IA, IL, IN, MA, MD, MI, MN, MS, NC, NJ, NY, OH, PA, RI, SC, TN, VA, WV
Eleven species in North America (one northern species) and Asia (ten species, one of them extending into northern European Russia).
Deuve (2004: 186) included members of this taxon in the subgenus Tomocarabus Reitter, 1896.
This species occurs from the Labrador coast to the Seward Peninsula in Alaska (Lindroth 1961a: 33-34), including the Aleutians and Kodiak Island, south to northern British Columbia (CNC); isolated on high mountains in Gaspé Peninsula (Quebec), New Hampshire (Lindroth 1961a: 34), and Maine (Mount Katahdin, CNC). Fossil remnants of this species, dated between about 16, 700 and 21, 500 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96) and northeastern Illinois (Garry et al. 1990: 394).
CAN: AB, BC, LB, MB, NT, NU, ON, QC, SK, YT USA: AK, ME, NH
Thirteen species (Deuve 2004: 197-203) in North America (one Holarctic species) and Asia (13 species, one of them extending into northern European Russia).
This Holarctic subspecies is known from a few old specimens collected on the Kamchatka Peninsula (Shilenkov in Kryzhanovskij et al. 1995: 43) and from Alaska, including the Pribilof Islands, to the Mackenzie River delta in northern Northwest Territories (Lindroth 1961a: 40).
CAN: NT, YT USA: AK – Holarctic
1. This species is listed in the subgenus Aulonocarabus Reitter by Kryzhanovskij et al. (1995: 43), Bousquet et al. (2003: 134), and Deuve (2004: 197) and in the subgenus Diocarabus by Casale and Kryzhanovskij (2003: 93) and Lorenz (2005: 85). 2. Two other subspecies of Carabus truncaticollis occur in Asia (Deuve 2004: 197-198).
One Holarctic species.
This Holarctic subspecies ranges from western Siberia (Bousquet et al. 2003: 154) to the Bering Sea Coast and from Alaska (Lindroth 1961a: 36) to Newfoundland (Lindroth 1955a: 23), south at least to northeastern New York (Powell 1977: 148), northern Iowa (Larsen et al. 2003: 292), and northern New Mexico (Brantley et al. 2003: 382; Sandoval County, UASM). The record from “Pennsylvania” (Bousquet and Larochelle 1993: 76) needs confirmation.
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, CO, IA, IL, IN, ME, MI, MN, MT, ND, NM, NY, OH, SD, VT, WI [PA] – Holarctic
Two other subspecies of this species, Carabus maeander chejuensis Deuve from South Korea and Carabus maeander paludis Géhin from the Far East, Japan, and China, are found in Asia. Deuve (2004: 183) also retained Carabus lapilayi Laporte as a valid subspecies.
Four species in North America (one species), Asia (two species), and Europe (one species).
This species ranges from Newfoundland (Lindroth 1955a: 26) to the Okanagan Valley in south-central British Columbia (Lindroth 1961a: 41), south to eastern Oregon (Hatch 1953: 50), central New Mexico (Fall and Cockerell 1907: 156), eastern Kansas (Popenoe 1877: 22; Horn 1872c: 384), and northwestern South Carolina (Ciegler 2000: 29).
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SC, SD, VA, VT, WA, WI, WV, WY
Ten species in Europe and the Middle East (Deuve 2004: 167-173), one of them adventive in North America.
This European subspecies is adventive in North America where it is found from Newfoundland (Lindroth 1955a: 27) to central Minnesota (Crow Wing County, CNC), south to northeastern Virginia (Falls Church, UASM) in the east, and from the Queen Charlotte Islands (Kavanaugh 1992: 51) to central Alberta (Lindroth 1961a: 37), south to southeastern Wyoming (Burne 1989: 290), northern Utah (Salt Lake County, CMNH), and central California (Lindroth 1961a: 37); seemingly isolated in the Saskatoon area, Saskatchewan (Ronald R. Hooper pers. comm. 1990). The first inventoried specimens collected on this continent was found in New Brunswick in 1890 (Horn 1892d: 61) and on the west coast around 1909 in Seattle, Washington (Hatch 1933c: 117). The date of 1870 listed by Lindroth (1961a: 37) for the first occurrence of this species on the continent is probably a lapsus for 1890. The species was also intentionally introduced in New England as predators of gypsy moths prior to 1911 (Smith 1959: 9).
CAN: AB, BC (QCI, VCI), NB, NF, NS, ON, PE, QC, SK USA: CA, CT, DE, ID, IL, IN, MA, ME, MI, MN, MT, NH, NJ, NV, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WY – Adventive
Carabus nemoralis Müller. This European species was first discovered on this continent in New Brunswick around 1890 and was recovered on the west side of Lake Ontario in 1919 and in southeastern Wisconsin by 1934. Unless the species went undetected for a long period of time, these dates would suggest that the species spread westwards for an average of 38 km per year. This is highly improbable for a wingless species and therefore its spread on this continent was undoubtedly enhanced by human transport unless separate introductions occurred.
Four European species, one of them extending into Siberia. One species is adventive in eastern North America.
The European Carabus cancellatus Illiger has been recorded from North Carolina (Horn 1883b: 270), Georgia (Fattig 1949: 10), and Wisconsin (Leng 1920: 44) but no specimens have been collected in recent decades. The species is probably not established on this continent.
This European subspecies was intentionally introduced into Massachusetts in 1908 and 1910 (Smith 1959: 7) for gypsy moth control. It is now established in New England (Lindroth 1961a: 37). The first inventoried specimen collected subsequently to its release was found in June 1920 in Winchester, Massachusetts (Smith 1959: 7).
USA: CT, MA, ME, NH, VT – Adventive
In a cladistic analysis conducted by Arndt et al. (2003: 312, Fig. 7.16), this species turned out as the sister-group to all remaining species of Carabus.
Five species in North America (four species) and Mexico (two species). One of Mexican species, Carabus hendrichsi Bolivar, Rotger, and Coronado, is endemic to the Sierra Madre Oriental.
This species is known from Kansas (Chautauqua County, CMNH; Erwin 2007a: 110), Wichita National Forest in Oklahoma, and eastern Texas (Van Dyke 1945a: 117).
USA: KS, OK, TX
This species is known from southeastern Arizona and the state of Durango in Mexico (Van Dyke 1945a: 118-119).
USA: AZ – Mexico
Carabus forreri willi Deuve is known from Chihuahua in Mexico.
This species ranges from “Maine” (Larochelle and Larivière 1990a: 27) to northern Minnesota (Gandhi et al. 2005: 923), north to north-central Ontario (Spires 1985: 79), south to “Texas” (Van Dyke 1945a: 116, as Carabus sylvosus lherminieri) and central Florida (Peck and Thomas 1998: 16). The record from “Utah” (Bousquet and Larochelle 1993: 77) is in error.
CAN: ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV
This subspecies ranges from Newfoundland (Lindroth 1955a: 25) to Yukon Territory, south to the Sierra Nevada in east-central California, southern Arizona, southern New Mexico, and southwestern South Dakota (Purrington et al. 2002: 202) [see Edelbrock 1986: Fig. 69].
CAN: AB, BC (QCI), MB, NF, NT, ON, QC, SK, YT USA: AZ, CA, CO, ID, MT, ND, NM, NV, OR, SD, UT, WA, WY
This subspecies is known from Vancouver Island and the Puget Sound area in Washington south to central Oregon between the coast and the Cascade Range (Edelbrock 1986: 49, Fig. 69).
CAN: BC (VCI) USA: OR, WA
This subspecies is known from high altitude along the northern Cascades and from coastal mountains of Oregon (Edelbrock 1986: 49).
CAN: BC USA: OR, WA
This subspecies is restricted to southeastern Alaska including the Aleutian Islands (Edelbrock 1986: 49, Fig. 69).
USA: AK
1. Edelbrock (1986) studied the geographical variation in Carabus taedatus and recognized four species, and one of them (Carabus agassii) was segregated into two subspecies (agassii and franciscanus). I have followed Deuve (1994a: 144) in listing Edelbrock’s species as subspecies. Furthermore I do not recognize the form Carabus agassii franciscanus as subspecifically distinct. 2. This species is placed in the subgenus Oreocarabus Géhin by some authors (e.g., Breuning 1933b: 719; Lorenz 2005: 91).
Twenty-two species (Deuve 2004: 363-375) in North America (one Holarctic species) and Eurasia (22 species).
This subspecies is found in eastern Siberia and in North America from the Seward Peninsula in Alaska to Bathurst Inlet on the arctic coast of Nunavut (Lindroth 1961a: 42).
CAN: NT, NU, YT USA: AK – Holarctic
Two other subspecies of this species occur in eastern Asia.
Worldwide, with more than 2, 500 species (Lorenz 2005: 22-62) arrayed in five tribes: Amblycheilini (13 species), Cicindelini (more than 1, 710 species), Collyridini (about 335 Asian species), Ctenostomatini (about 225 species in the Neotropical and Afrotropical Regions), Manticorini (14 Afrotropical species), and Megacephalini (about 200 species).
Western Hemisphere, with 13 species in North America (ten species), Mexico (two species), and South America (one species) arrayed in three genera: Amblycheila (seven species), Omus (five species), and Picnochile Motschulsky (one species in Chile and Argentina).
Seven species (Pearson et al. 2006: 48) in western North America (five species) and northern Mexico (two species: Amblycheila nyx Sumlin from west-central Coahuila and Amblycheila halffteri Mateu from San Luis Potosí).
Vaurie (1955) reviewed the species and offered a key for their identification. Subsequently one new North American species, Amblycheila hoversoni Gage, was described in 1991. Pearson et al. (2006: 22) field guide includes a key to the North American species.
The record of Amblycheila halffteri from “Texas” (Bousquet and Larochelle 1993: 52) is in error. The species is known so far only from San Luis Potosí in Mexico.
This species, also known as the “Montane Giant Tiger Beetle
USA: AZ, TX
This species, also known as the “Great Plains Giant Tiger Beetle”, ranges from southwestern South Dakota (Howden 1970: 8) and eastern Wyoming (Huber 1978: 75) south to western Texas [see Pearson et al. 2006: Map 3]. The record from “Arizona” (Horn 1910a: 123) is in error; those from “Arkansas” (Leng 1902: 97; Erwin and Pearson 2008: 20) are probably based on a misinterpretation of the type locality.
USA: CO, KS, NE, NM, OK, SD, TX, WY
This species, also known as the “South Texas Giant Tiger Beetle”, is found only in south and west-central Texas (Pearson et al. 2006: 51).
USA: TX
This species, also known as the “Plateau Giant Tiger Beetle”, occurs from the northern half of Arizona to western Texas, north to southwestern Colorado (Kippenhan 1994: 21) and southeastern Utah (Krell and Brookhart 2012: 110). The record from “Oklahoma” (Bousquet and Larochelle 1993: 52) is in error.
USA: AZ, CO, NM, TX, UT
This species, also known as the “Mojave Giant Tiger Beetle”, is found from northwestern Arizona and southwestern Utah west to the desert mountains of southeastern California east of the Sierra Nevada [see Vaurie 1955: Fig. 1 and Pearson et al. 2006: Map 2].
USA: AZ, CA, NV, UT
Five species in western North America inhabiting the Pacific coastal lowlands and the mountain slopes, including those of the Cascade and Sierra Nevada ranges.
Casey, between 1897 and 1924, described so many forms in this genus (90 species-group taxa) that at the time of his death in 1925 it was virtually impossible to identify members of Omus. Cazier (1942) wrote a detailed monographic revision of the genus and recognized three species (Anisodactylus californicus, Anisodactylus dejeanii, and Anisodactylus submetallicus) with four subspecies for Anisodactylus californicus. He also provided keys for the identification of the taxa. Unfortunately his thesis was not published. In his thesis on the cicindelids of the Pacific Northwest, Leffler (1979a) followed Cazier’s (1942) conclusions concerning the taxonomy of Omus except that he recognized a fourth species, according specific rank to Omus audouini considered a synonym of Omus californicus californicus by Cazier. Leffler (1979a) provided a key for the separation of the species but his work also remained unpublished. Subsequently, a new species was described by van den Berghe in 1994. The field guide of Pearson et al. (2006: 23) includes a key to all five species.
The range of this species, also known as the “Audouin’s Night-stalking Tiger Beetle”, extends from southwestern British Columbia, including southern Vancouver Island, south to northwestern California [see Pearson et al. 2006: Map 9].
CAN: BC (VCI) USA: CA, OR, WA
This subspecies, also known as the “Narrow Night-stalking Tiger Beetle”, is found above 1500 m of elevation in Plumas and Lassen Counties in northeastern California (Leffler 1979a: 218).
USA: CA
This subspecies, also known as the “California Night-stalking Tiger Beetle”, ranges from southwestern Oregon to southern California along the coast and through the Sierra Nevada, at elevation below 900 m near the range of Omus californicus intermedius (Leffler 1979a: 218; Fig. 18).
USA: CA, OR
This subspecies, also known as the “Intermediate Night-stalking Tiger Beetle”, is found above 900 meters in the Sierra Nevada between El Dorado and Tulare Counties, California (Leffler 1979a: 222).
USA: CA
This subspecies, also known as the “Subcylindrical Night-stalking Tiger Beetle”, is restricted to San Martin, Santa Clara County, in western California (Leffler 1979a: 217).
USA: CA
This taxon, also known as the “Mount Ashland Night-stalking Tiger Beetle”, is known only from Jackson County in southwestern Oregon.
USA: OR
This species, also known as the “Greater Night-stalking Tiger Beetle”, is found from southwestern British Columbia, including Vancouver Island, to southwestern Oregon [see Pearson et al. 2006: Map 6]. The record from northern California (Leng 1902: 105) needs confirmation; that from “Montana” (Leng 1902: 105) is in error.
CAN: BC (VCI) USA: OR, WA [CA]
Omus dejeanii Reiche. The Greater Night-stalking Tiger Beetle is one of the few species-group taxa currently recognized in the genus Omus although over one hundred taxa have been described, particularly by Thomas Casey. Even today, taxonomists do not agree on the number of valid taxa that should be recognized in Omus. The difficulty of defining the species-group taxa is not unusual among old, apterous carabid lineages because they tend to form small, local populations.
This species, also known as the “Lustrous Night-stalking Tiger Beetle”, is restricted to a small area east of Warthan Canyon in western Fresno County, central California (Knisley and Haines 2010: 245).
USA: CA
About 200 species in the Nearctic (four species), Neotropical (about 165 species), Australian (25 species), Palaearctic (one species), and Afrotropical (12 species) Regions. The species are arrayed in 11 genera (see Naviaux 2007: 10-11): Aniara Hope (one Neotropical species), Australicapitona Sumlin (eight Australian species), Cheiloxia Guérin-Méneville (two Neotropical species), Grammognatha Motschulsky (one Mediterranean species), Megacephala Latreille (12 Afrotropical species), Metriocheila Thomson (one Neotropical species), Oxycheila Dejean (about 30 Neotropical species), Phaeoxantha Chaudoir (12 Neotropical species), Pseudotetracha Fleutiaux (17 Australian species), Pseudoxycheila Guérin-Méneville (about 20 Neotropical species), and Tetracha (about 95 species).
Western Hemisphere, with about 95 species (110 species-group taxa) (Naviaux 2007: 3) in the Nearctic (four species, only one endemic) and Neotropical (about 95 species) Regions. The species are arrayed in seven subgenera: Apterotetracha Naviaux (three Brazilian species), Microtetracha Naviaux (one Argentine species), Neotetracha (about 55 species), Oblongotetracha Naviaux (one Venezuelan species), Paratetracha Naviaux (five South American species), Prototetracha Naviaux (one Mexican species), and Tetracha s.str. (29 species).
Naviaux (2007) recently revised the species and provided keys for the subgenera and the species groups but not for all species. Pearson et al. (2006: 24) field guide to the tiger beetles of North America included a key to all North American species (Tetracha impressa under the name Tetracha affinis) except Tetracha floridana. Naviaux (2007: 42-43) redescribed Tetracha floridana and pointed out the structural differences between the species and Tetracha carolina.
In their phylogenetic analysis based on the nuclear 18S and the mitochondrial 16S and cytochrome oxidase III genes, Zerm et al. (2007) found out that the genus Tetracha was paraphyletic in regard to the monospecific genus Aniara Hope and that the two genera and Phaeoxantha form a clade.
From a zoogeographical point of view it is of interest to note that a specimen identical to present day Tetracha carolina carolina has been found in amber from the east coast of Germany (Horn 1906).
Twenty-nine species in North America (three species, one of them endemic) and the Neotropical Region (28 species), including the West Indies.
[carolina group]
The range of this subspecies, also known as the “Pan-American Big-headed Tiger Beetle” (Pearson et al. 2006: 57) or the “Carolina Metallic Tiger Beetle” (Erwin and Pearson 2008: 70), extends from Maryland to southeastern California, south to Nicaragua (Naviaux 2010: 70) and the Florida Keys [see Pearson et al. 1997: Fig. 3]; two specimens from San Diego County in California are also known (Moore 1937: 3). The records from the Bahamas, Cuba, Cayman Islands, and Jamaica refer to Tetracha carolina occidentalis. The records from “Colorado” (Wickham 1902: 228), “Connecticut, ” “Indiana, ” “New Jersey, ” and “New York” (see Bousquet and Larochelle 1993: 54) are probably in error or based on strays; that from Nebraska (Bruner 1901: 97) needs confirmation (see Spomer et al. 2008a: 11).
USA: AL, AR, AZ, CA, DC, FL, GA, IA, IL, KS, KY, LA, MD, MO, MS, NC, NM, NV, OK, SC, TN, TX, VA [NE] – Guatemala, Mexico, Nicaragua
Three more subspecies are recognized by Naviaux (2007: 40): Tetracha carolina chevrolatii Chaudoir from the Yucatán Peninsula and Belize, Tetracha carolina moraveci Naviaux from the state of Mexico, and Tetracha carolina occidentalis (Klug) from the West Indies.
This species, also known as the “Florida Metallic Tiger Beetle”, is found only in southern Florida, from Dixie County to the Keys (Choate 2003: 63).
USA: FL
[virginica group]
The range of this species, also known as the “Virginia Big-headed Tiger Beetle” (Pearson et al. 2006: 59) or the “Virginia Metallic Tiger Beetle” (Erwin and Pearson 2008: 100), extends from southwestern Connecticut (Dunn 1985b: 21) to southwestern Nebraska (Spomer et al. 2008a: 54), south to southwestern Texas, northeastern Mexico (Pearson et al. 2006: 60), and the Florida Keys [see Pearson et al. 2006: Map 13]; apparently isolated in southeastern Arizona (Dajoz 2004: 116) and south-central Wisconsin (Lawton 1971: 57). The records from “Colorado” (Boyd 1982: 5) and “Tabasco” (Erwin and Pearson 2008: 100) need confirmation.
USA: AL, AR, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI, WV [CO] – Mexico
About 55 Neotropical species, one of them extending into southeastern Texas.
This species, also known as the “Upland Metallic Tiger Beetle”, ranges from southeastern Texas (Darlington 1935b: 161, as Tetracha angustata) to Veracruz, Mexico (Naviaux 2007: 144).
USA: TX – Mexico
This species has passed under the name Tetracha angustata (Chevrolat, 1841) or Tetracha affinis angustata in the North American literature until recently.
Worldwide, with more than 1, 710 species arrayed in five subtribes: Apteroessina (one species from south India known only from three partial specimens collected in the early xix Century), Cicindelina (more than 1, 090 species), Iresina (about 45 species, most in the Neotropical and Australian Regions), Dromicina [= Prothymina] (about 475 species in the Neotropical, Australian, Oriental, Asian Palaearctic, and Afrotropical Regions), and Theratina (about 100 Asian species in the genus Therates Latreille).
Worldwide, with about 1, 090 species. The North American fauna is represented by 98 species (about 9% of the world fauna). The number of genera recognized varies greatly depending on the approach used (lumper versus splitter).
Willis (1968) published a simplified key based on external characters for 91 North American forms, representing all the current recognized species except for the following 11: Cicindela albissima, Cicindela arida, Cicindela cazieri, Cicindela highlandensis, Cicindela nigrior, Cicindela ohlone, Cicindela scabrosa, Cicindela waynei, Habroscelimorpha fulgoris, Cylindera lunalonga, and Dromochorus velutinigrens. Pearson et al. (2006: 24-42) field guide to the North American tiger beetles included a key to all currently recognized species, except Cylindera lunalonga which has been raised to species level very recently, and a few subspecies. The key is based on external characters and distribution ranges.
About 210 species in all zoogeographical regions. The North American fauna includes eight species (about 4% of the world fauna) placed in the nominotypical subgenus.
Lorenz (2005: 54-58) listed the following taxa as subgenera of Cylindera: Apterodela Rivalier (four species), Conidera Rivalier (two species), Eriodera Rivalier (one species), Eugrapha Rivalier (32 species), Gaymara Freitag and Barnes (five Neotropical species), Glomera Acciavatti and Pearson (two species), Ifasina Jeannel (67 species), Leptinomera Rivalier (25 species), Oligoma Rivalier (two species), Plectographa Rivalier (19 species), Setinteridenta Acciavatti (one species), and Verticina Rivalier (six species). Cicindelina Jeannel (one Madagascan species) was transferred from the genus Cicindela to Cylindera by Moravec (2010: 198).
About 45 species (Lorenz 2005: 55-56) in the Nearctic (eight species, 14 species-group taxa), Neotropical (about 15 species), Oriental (four species), Palaearctic (14 species), and Afrotropical (six species) Regions.
This species, also known as the “Swift Tiger Beetle”, has been recorded from Nebraska and western Iowa south to north-central Texas and north-central Arkansas [see MacRae and Brown 2011b: Fig. 8]. The record from Indiana (Montgomery and Montgomery 1931: 359) was based on misidentified Cylindera cursitans (Knisley et al. 1990: 279); those from “Illinois, ” and “Dakota” (Leng 1902: 117) are in error or based on strays. According to MacRae and Brown (2011b: 230), once abundant in Nebraska, western Iowa, and eastern Kansas, the species has declined below detectable levels in much of this area during the past century.
USA: AR, IA, KS, MO, NE, OK, TX
The range of this species, also known as the “Ant-like Tiger Beetle”, extends from North Dakota and west-central Minnesota (Tinerella and Rider 2000: 367) to the Ohio River Valley in West Virginia (Kirchner and Kondratieff 1999: 84), south to western Alabama (Löding 1945: 10), Louisiana, and northern Kansas (Popenoe 1877: 22) [see Pearson et al. 2006: Map 90]; also recorded from “Montana” (Erwin and Pearson 2008: 207).
USA: AL, AR, IA, IL, IN, KS, KY, LA, MN, MO, MS, ND, NE, OH, SD, TN, WV [MT]
This species, also known as the “Grass-runner Tiger Beetle”, ranges from southeastern Arizona to southwestern Texas [see Pearson et al. 2006: Map 87], south to Durango (Cazier 1954: 287).
USA: AZ, NM, TX – Mexico
This subspecies, also known as the “White-striped Tiger Beetle”, ranges from southern California to southwestern New Mexico, as far north as southwestern Utah (Tanner 1929a: 86), south to Nayarit (Cazier 1960: 12) [see Shook 1989: Fig. 1].
USA: AZ, CA, NM, NV, UT – Mexico
This subspecies, the “Rouged Tiger Beetle”, is known from northeastern New Mexico south to southern Durango and Tamaulipas [see Shook 1989: Fig. 1].
USA: NM, TX – Mexico
Intergrades between the two subspecies of Cylindera lemniscata occur over a small area in south-central New Mexico and northern Chihuahua [see Shook 1989: Fig. 1]. On the Baja California Peninsula, this species is represented by Cylindera lemniscata bajacalifornica (Shook).
This species, also known as the “Meadow Tiger Beetle”, is restricted to the western and northern slopes of the Sierra Nevada and adjacent eastern portion of the San Joaquin Valley of California (Woodcock et al. 2006: 869). Specimens labeled from Riverside in California, Prescott in Arizona (see Cazier 1939: 28), and Olmito and San Benito in Texas are doubtful or in error (Woodcock et al. 2006: 869); the records from San Pedro Mártir Mountains in Baja California (Cazier 1948: 18) and “Nevada” (Boyd 1982: 16) need confirmation. Extensive surveys in the past 20 years have produce a sole extant population, near Westwood in Lassen County leading Woodcock et al. (2006: 875) to suggest that the species should be considered as a candidate for listing as a Threatened and Endangered species by the U.S. Fish and Wildlife Service.
USA: CA [NV]
This form has been considered a subspecies of Cylindera terricola (Say) or a full species depending on the author until mitochondrial DNA analysis indicates that it is a distinct species (Woodcock et al. 2006).
The range of the “Belted-winged Tiger Beetle” is disputed. According to Johnson (1990b: Fig. 1), it ranges from west-central Alberta to southeastern Saskatchewan, south to central New Mexico and central Arizona; isolated in southwestern Utah and southwestern Yukon Territory. According to Pearson et al. (2006: 153), it is found in “lower elevations of Colorado, New Mexico, and Arizona.” Erwin and Pearson (2008: 222) recorded the subspecies from the same states as Pearson et al. (2006: 153) and added Kansas (also cited by Leng 1902: 155).
CAN: AB, SK, YT USA: AZ, CO, MT, ND, NE, NM, SD, UT, WY
This subspecies, the “Interior Tiger Beetle”, is known from Nye County in western Nevada and from the Traverse Mountain Range in western Ventura County eastwards to the San Bernardino Mountains in San Bernardino County and the southern tip of the Sierra Nevada in Kern County, California (Kippenhan 2007: 14, Fig. 8). The record from “NE” (Erwin and Pearson 2008: 222) is probably an error for “NV.”
USA: CA, NV
Freitag (1999: 87) listed this taxon as a junior synonym of Cylindera terricola imperfecta (LeConte) but Pearson et al. (2006: 153) and Kippenhan (2007: 7) retained it as a valid subspecies.
This subspecies, the “Imperfect Tiger Beetle”, ranges from westernmost Alberta and British Columbia, as far north as Fort Saint John along the Peace River (Catling 2007: 19), south to southern Utah, southern Nevada, and Mono County (Kippenhan 2007: 13) in west-central California [see Johnson 1990b: Fig. 1]; also recorded from “Arizona” and “Wyoming” by Erwin and Pearson (2008: 222). The record from New Mexico (Fall and Cockerell 1907: 155) must be in error.
CAN: AB, BC USA: CA, ID, MT, NV, OR, UT, WA [AZ, WY]
This subspecies, the “Kaibab Tiger Beetle”, is known only from the Kaibab Plateau of northern Arizona (Johnson 1990b: 1).
USA: AZ
This subspecies, the “Susan’s Tiger Beetle”, is restricted to the valleys east of the Sierra Nevada Mountains in Mono and Inyo Counties, California [see Kippenhan 2007: Fig. 8].
USA: CA
This subspecies, also known as the “Variable Tiger Beetle”, ranges from northwestern Ontario (Lawton 2008: 73) to southeastern Saskatchewan, south to northwestern Nebraska [see Johnson 1990b: Fig. 1]. The record from Minnesota (Horn 1928: 12) needs confirmation.
CAN: MB, ON, SK USA: ND, NE, SD [MN]
The range of this species, also known as the “One-spotted Tiger Beetle”, extends from northeastern New York to central Georgia and southern Mississippi, west to western Missouri [see Pearson et al. 1997: Fig. 19]. The record from “Texas” (Horn 1915: 389) is probably in error or based on a stray.
USA: AL, AR, DC, GA, IA, IL, IN, KY, MD, MN, MO, MS, NC, NJ, NY, OH, PA, SC, TN, VA, WV
This subspecies, the “Pygmy Tiger Beetle”, ranges from central Arizona (Pearson et al. 2006: 156) south to Durango (Cazier 1954: 285).
USA: AZ – Mexico
The subspecies Cylindera viridisticta viridisticta (Bates) and Cylindera viridisticta interjecta (Horn) are Mexican endemics.
Thirteen North American species (27 species-group taxa), of which five extend into Mexico, as far south as the state of Yucatan, and one into the Bahamas and Cuba.
[marginata group]
This species, also known as the “Sandbar Tiger Beetle”, is endemic to the Coastal Plain ranging from southeastern North Carolina to the Florida Panhandle west to southeastern Mississippi and southwestern Louisiana [see Pearson et al. 2006: Map 97].
USA: AL, FL, GA, LA, MS, NC, SC
This species, also known as the “Coppery Tiger Beetle”, ranges from southern Ohio to southwestern Manitoba and central Montana, south to east-central New Mexico, eastern Texas, and southern Alabama [see Pearson et al. 2006: Map 100]; also known from one locality in northern Georgia. The record from “Wisconsin” (Bousquet and Larochelle 1993: 68) is likely in error (Peter W. Messer pers. comm. 2007).
CAN: MB USA: AL, AR, CO, GA, IA, IL, IN, KS, KY, LA, MN, MO, MS, MT, ND, NE, NM, OH, OK, SD, TN, TX, WV, WY
This species, also known as the “Whitish Tiger Beetle”, inhabits the Coastal Plain ranging from Virginia (Hoffman et al. 2006: 17) to the Florida Panhandle, west to southwestern Alabama (Löding 1945: 10) [see Pearson et al. 2006: Map 106]; also recorded from “Mississippi” (Erwin and Pearson 2008: 229). The record from Louisiana (Boyd 1982: 17) is probably in error or based on a stray.
USA: AL, FL, GA, NC, SC, VA [MS]
This subspecies, the “Gulf Beach Tiger Beetle”, is found along the Gulf coast of Florida, including the Keys (Choate 2003: Map 61), and Alabama (Löding 1945: 10). The record from Horn Island, Mississippi (Richmond 1968: 234), is in error for Ellipsoptera hamata monti (Graves and Pearson 1973: 187); those from “North Carolina, ” “South Carolina, ” “Georgia, ” and “Louisiana” (Erwin and Pearson 2008: 229) are probably in error or based on strays.
USA: AL, FL
The nominotypical subspecies is known from the states of Tabasco and Veracruz and Ellipsoptera hamata pallifera (Chaudoir) from the states of Quintana Roo and Yucatán (Erwin and Pearson 2008: 229-230).
This subspecies, the “Coastal Tiger Beetle”, is found along the Gulf Coast from Mississippi (Graves and Pearson 1973: 187) to the state of Veracruz in Mexico (Erwin and Pearson 2008: 230).
USA: LA, MS, TX – Mexico
This species, also known as the “Moustached Tiger Beetle”, ranges from east-central Georgia (Beaton 2008: 42) to southern Florida (Choate 2003: Map 63; Pearson et al. 2006: 174). The record from “North Carolina” (Erwin and Pearson 2008: 230) is probably in error or based on a stray.
USA: FL, GA
This species, also known as the “Ghost Tiger Beetle”, ranges from southern Quebec to southeastern Alberta (Hilchie 1985: 333), south to Chihuahua (Cazier 1954: 297), central Texas, southern Louisiana, and eastern North Carolina, west to western Arizona and eastern Nevada [see Pearson et al. 1997: Fig. 17]. The record from South Carolina, based on a specimen in CMNH, is probably in error (Knisley and Schultz 1997: 113). According to Erwin and Pearson (2008: 231), the Ghost Tiger Beetle has been extirpated over much of its former range due to habitat loss.
CAN: AB, MB, ON, QC, SK USA: AL, AR, AZ, CO, CT, DE, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, MT, NC, ND, NE, NJ, NM, NV, NY, OH, OK, PA, SD, TN, TX, UT, VA, WI, WY – Mexico
This subspecies, the “Denton Tiger Beetle”, is known from Dallas, Denton, and Kaufman Counties in northern Texas (Vaurie 1951: 10) [see Willis 1967: Fig. 137].
USA: TX
This subspecies, the “Panhandle Tiger Beetle”, is found in Oklahoma, northern Texas, and northeastern New Mexico [see Pearson et al. 2006: Map 101]. The records from “Arkansas” and “Kansas” (Boyd 1982: 17) need confirmation.
USA: NM, OK, TX [AR, KS]
Willis (1967) indicated the presence of narrow zones of intergradation between this subspecies and the other two.
This subspecies, also known as the “Sandy Stream Tiger Beetle”, ranges from Beaver Islands in northern Michigan (Dunn 1987: 11) to southern Wyoming, south to north-central Colorado (Kippenhan 1990: 314), Arkansas, and Tennessee [see Pearson et al. 2006: Map 101]. The record from “Ohio” (Willis 1967: 269) needs confirmation (see Graves and Brzoska 1991: 28); that from “Texas” (Freitag 1999: 94) probably refers to the fluviatilis form; those from central and southeastern New Mexico (Fall and Cockerell 1907: 155) are probably in error.
USA: AR, CO, IA, IL, IN, KS, KY, LA, MI, MN, MO, NE, OK, SD, TN, WI, WY [OH]
Intergrades between this and the fluviatilis forms occur in southern Kansas and eastern Oklahoma (Pearson et al. 2006: 169).
This species, also known as the “Margined Tiger Beetle”, is found along the Atlantic Coast from Kings County in Nova Scotia (Neil and Majka 2008: 4) to the Florida Keys and along the Gulf Coast in Florida [see Pearson et al. 2006: Map 96]; it is also recorded from the Bahamas and Cuba (Peck 2005: 27). According to Dunn (1983: 4), the species has declined significantly along the New Hampshire coast.
CAN: NS USA: CT, DC, DE, FL, GA, MA, MD, ME, NC, NH, NJ, NY, RI, SC, VA – Bahamas, Cuba
Cicindela marginata Fabricius. This species is a typical coastal species inhabiting mudflats and salt marshes from New Brunswick to southern Florida and along the Florida Gulf coast; it is also known from the Bahamas and the coast of Cuba. The breeding populations in New Brunswick were discovered only recently. Whether the species had been in the area for some time but went unnoticed, or extended its range in recent years, is the subject of speculation.
This species, also known as the “Puritan Tiger Beetle”, is now restricted to two areas, one along the Connecticut River in southern Massachusetts and Connecticut, the other one along the Chesapeake Bay in Maryland (Pearson et al. 2006: 170, 193). It formerly occurred along the Connecticut River as far north as central New Hampshire.
USA: CT, MA, MD, NH, NJ, NY, VA, VT
This species has been listed as threatened by the U.S. Fish and Wildlife Service in August 1990.
This species, also known as the “White-sand Tiger Beetle”, is found along a small area within the Coastal Plain from south-central Georgia to eastern Louisiana [see Pearson et al. 2006: Map 98]. Beaton (2008: 42) did not find this species at any historical sites in Georgia during his survey of the tiger beetles of the state.
USA: AL, FL, GA, LA, MS
[sperata group]
This subspecies, the “Chiracahua Tiger Beetle”, is known from southeastern Arizona and Sonora, Mexico (Spomer 2004: 409); also recorded from “New Mexico” (Erwin and Pearson 2008: 233).
USA: AZ [NM] – Mexico
This subspecies, the “Knaus’s Tiger Beetle”, ranges from southern Manitoba to southeastern Alberta, south to central New Mexico and central Texas [see Willis 1967: Fig. 143].
CAN: AB, MB, SK USA: CO, KS, MT, ND, NE, NM, OK, SD, TX, WY
This subspecies, also known as the “Salt Creek Nevada Tiger Beetle”, has been reported yet only from around the type locality in eastern Nebraska [see Willis 1967: Fig. 143]. Population estimates vary yearly from a few hundred to under a thousand specimens (Spomer et al. 2008a: 43).
USA: NE
This species has been listed as endangered by the U.S. Fish and Wildlife Service in October 2005.
This subspecies, the “Indian Creek Tiger Beetle”, is known only from the type locality in the South Dakota Badlands (Spomer 2004: 410).
USA: SD
This subspecies, also known as the “Nevada Tiger Beetle”, is found in the Great Basin region of Nevada and California south to northern Sonora [see Willis 1967: Fig. 143].
USA: CA, NV – Mexico
Another subspecies, Ellipsoptera nevadica metallica (Sumlin), is known from the state of Coahuila.
This subspecies, the “Olmos Creek Tiger Beetle”, ranges from southeastern Arizona to southeastern Texas, including northern Coahuila [see Willis 1967: Fig. 143].
USA: AZ, NM, TX – Mexico
This subspecies, the “Tube Tiger Beetle”, ranges from northeastern Utah to central Arizona and northwestern New Mexico [see Willis 1967: Fig. 143].
USA: AZ, CO, NM, UT
This species, also known as the “Aridland Tiger Beetle”, is found from northern Utah to southern Colorado, south to Chihuahua (Cazier 1954: 297) [see Pearson et al. 1997: Fig. 29]. The record from “Arkansas” (Boyd 1982: 17) is in error or based on a stray.
USA: AZ, CO, NM, TX, UT – Mexico
Rumpp (1977: 176) recognized marutha and rubicunda as distinct subspecies under the specific name Ellipsoptera marutha. I have followed Freitag (1999: 95) in considering the two taxa as synonyms.
This subspecies, the “Inquisitor Tiger Beetle”, is found in central Texas (Pearson et al. 2006: 171).
USA: TX
This subspecies, also known as the “Rio Grande Tiger Beetle”, ranges from northern Utah to northeastern New Mexico [see Pearson et al. 2006: Map 103], south to Durango and Tamaulipas (Cazier 1954: 296). The records from western Kansas (Popenoe 1877: 22; Snow 1878: 63) and “Oklahoma” (Boyd 1982: 17) need confirmation; that from Yuma, California (Leng 1902: 167) is likely in error.
USA: AZ, CO, NM, TX, UT [KS, OK] – Mexico
Another subspecies, Ellipsoptera sperata vauriei (Cazier), is known from the state of Sonora in Mexico.
Three species (five species-group taxa) in United States and Cuba (one species) and Mexico (two species), including Baja California.
This species, also known as the “Olive Tiger Beetle”, is found in Cuba and southern Florida, including the Keys [see Woodruff and Graves 1963: Fig. 3]. Apparently the species has not been sighted in Florida since the 1980s (Pearson et al. 2006: 149).
USA: FL – Cuba
Six Neotropical species, of which one is represented by a distinct subspecies in southeastern Texas.
This subspecies, the “Smyth’s Beach Tiger Beetle”, is known only from the type locality in southeastern Texas. No specimens have been collected since the original ones (over 80 specimens) in June 1912.
USA: TX
The nominotypical subspecies is found along the Gulf Coast of Mexico, as far south as Veracruz, and from Honduras (Erwin and Pearson 2008: 290).
Thirty-eight Neotropical species (Lorenz 2005: 58), of which one extends into southwestern North America.
The Cuban Brasiella viridicollis (Dejean) is known in North America from a single specimen, probably a stray, collected in the Florida Keys in 1983 (Schiefer 2005: 551; Pearson et al. 2006: 151). The species is not considered here as a North American entity.
This species, also known as the “Sonoran Tiger Beetle”, ranges from southern Arizona south to Sinaloa; it is also found in southern Baja California Peninsula (Cazier 1954: 286). The record from “California” (Erwin and Pearson 2008: 118) needs confirmation.
USA: AZ [CA] – Mexico
Four North American species, one of them extending into northern Mexico.
This species, also known as the “Loamy-ground Dromo Tiger Beetle”, ranges from central and coastal Texas (Pearson et al. 2006: 160) south to Tamaulipas (Cazier 1954: 297); it was also recorded from southeastern Colorado (Michels et al. 2008).
USA: CO, TX – Mexico
This species, also known as the “Cajun Dromo Tiger Beetle”, is found along and near the Gulf Coast in southern Louisiana (Schaupp 1884a: 85) and northeastern Texas [see Pearson et al. 2006: Map 92].
USA: LA, TX
This species, also known as the “Frosted Dromo Tiger Beetle”, ranges from northern Kansas and central Missouri (MacRae and Brown 2011a) south to Nueces and Webb Counties in southern Texas and west into the Texas Panhandle (Pearson et al. 2006: 160). There is also one record from east-central Nebraska (Pearson et al. 2006: 160).
USA: KS, MO, OK, TX [NE]
This species, also known as the “Velvet Dromo Tiger Beetle”, is found only in southern Texas, primarily along the coast [see Pearson et al. 2006: Map 94].
USA: TX
Western Hemisphere, with 18 species (33 species-group taxa) in the Nearctic (nine species, 18 species-group taxa) and Neotropical (14 species) Regions.
This subspecies, the “California Tiger Beetle”, ranges from the Mojave Desert of southern California (Pearson et al. 2006: 138) south to northeastern Baja California Peninsula and northwestern Sonora (Cazier 1954: 289).
USA: CA – Mexico
This form intergrades with the pseudoerronea form at Soda Lake in northeastern San Bernardino County, California (Pearson et al. 2006: 139).
This subspecies, the “Inland Tiger Beetle”, is found only in the Death Valley in Inyo and San Bernardino Counties, California (Rumpp 1958: 151).
USA: CA
The nominotypical subspecies is found in the Baja California Peninsula and Habroscelimorpha californica brevihamata (Horn) occurs in the states of Sinaloa and Sonora in Mexico.
This subspecies, also known as the “Cream-edged Tiger Beetle”, is found from southeastern Oklahoma to northeastern Mexico [see Johnson 1993b: Fig. 1].
USA: OK, TX – Mexico
This form intergrades with the johnsonii form along the Oklahoma-Texas border (Pearson et al. 2006: 140).
This subspecies, the “Johnson’s Tiger Beetle”, ranges from central Missouri to southeastern Colorado, north to west-central Nebraska (Spomer et al. 2008a: 58), south to New Mexico and southwestern Texas [see Johnson 1993b: Fig. 1]. The taxon is also found in Coahuila in northern Mexico (Murray 1979: 55). The records from “North Dakota” (Boyd 1982: 14; Freitag 1999: 77) probably refer to the pembina form.
USA: CO, KS, MO, NE, NM, OK, TX – Mexico
This subspecies, the “Pembina Tiger Beetle”, is known only from a small area in northeastern North Dakota [see Johnson 1993b: Fig. 1].
USA: ND
This subspecies, also known as the “Eastern Beach Tiger Beetle”, once occurred along the Atlantic Coast from Cape Cod, Massachusetts to the Chesapeake Bay. It is now found at two isolated sites on the coast of Massachusetts and along both shores of the Chesapeake Bay in Maryland and Virginia (Boyd and Rust 1982: 234; Pearson et al. 2006: 144). The subspecies was successfully reintroduced at Sandy Hook, New Jersey (Pearson et al. 2006: 192). The records from “Delaware” (Bousquet and Larochelle 1993: 65) and from near Lancaster, Pennsylvania (Cresson 1861: 12) are probably based on strays.
USA: CT, MA, MD, NJ, NY, RI, VA
This subspecies is listed as threatened under the Endangered Species Act by the U.S. Fish and Wildlife in 1990 (Pearson et al. 2006: 191).
This subspecies, the “Eastern Beach Tiger Beetle”, is found along the Atlantic Coast from Ocean County in New Jersey to southern Florida (Boyd and Rust 1982: 234).
USA: DE, FL, GA, MD, NC, NJ, SC, VA
According to Knisley and Schultz (1997: 103), the ranges of this form and of the dorsalis form are contiguous in southern New Jersey, the southern tip of coastal Virginia, and near the mouth of the Chesapeake Bay on the Virginia side and little intergradation can be observed on those sites.
This subspecies, the “Saulcy’s Beach Tiger Beetle”, ranges from the Gulf Coast of Florida to the Mississippi River delta in Louisiana (Pearson et al. 2006: 144); it is also known from the Isla Arcas in Campeche (Bates 1884: 260). The records from Cuba (Leng and Mutchler 1916: 697, as Cicindela dorsalis var. venusta; Cazier 1954: 294; Erwin and Pearson 2008: 247) are based on mislabeled specimens (Valdés 1999: 13).
USA: AL, FL, LA, MS – Mexico
This subspecies, the “Gulf Beach Tiger Beetle”, is found from coastal Mississippi (Lago et al. 2002: 201) south along the Gulf to the state of Tamaulipas (Cazier 1954: 294). The record from east-central Colorado (Snow 1877: 16) is probably in error.
USA: LA, MS, TX – Mexico
This subspecies, the “Pale Tiger Beetle”, is known from the Salt Basin of western Texas and adjacent New Mexico and from a single site in Dawson County, Texas (Acciavatti 1981: 239).
USA: NM, TX
This subspecies, the “Willcox Tiger Beetle”, is known only from the type locality in southeastern Arizona (Acciavatti 1981: 238).
USA: AZ
This subspecies, also known as the “Glittering Tiger Beetle”, is found from east-central Arizona to north-central New Mexico, south to the Rio Grande area in westernmost Texas and southeastern New Mexico [see Acciavatti 1981: Fig. 1]; also recorded from Chihuahua in Mexico (Murray 1979: 55).
USA: AZ, NM, TX – Mexico
This subspecies intergrades with the albilata form in southeastern New Mexico (Pearson et al. 2006: 143).
This species, also known as the “Western Tidal Flat Tiger Beetle”, occurs along the Pacific Coast from southern California to central Baja California Peninsula, and along the Gulf of California Coast from northern Sonora to Sinaloa (Cazier 1954: 291) [see Pearson et al. 1997: Fig. 28]. According to Pearson et al. (2006: 139), this species is now found in the United States only in three or four protected areas in Ventura, Orange, and San Diego Counties.
USA: CA – Mexico
This species, also known as the “Gulfshore Tiger Beetle”, is found along the Gulf Coast from eastern Mississippi (Grammer 2009) to northern Tamaulipas in Mexico (Pearson et al. 2006: 145).
USA: LA, MS, TX – Mexico
This subspecies, the “Virgin River Tiger Beetle”, is found only along the Virgin River in southwestern Utah and southeastern Nevada [see Acciavatti 1981: Fig. 1].
USA: NV, UT
This subspecies, also known as the “Riparian Tiger Beetle”, is found from the Gila River Basin in eastern Arizona westwards to the Salton Sea Basin in California, north to southern Nevada [see Pearson et al. 2006: Map 74]. The record from New Mexico (Fall and Cockerell 1907: 155) needs confirmation. According to Erwin and Pearson (2008: 251), the species has been extirpated from many of its historic sites.
USA: AZ, CA, NV [NM]
This subspecies, also known as the “Saltmarsh Tiger Beetle”, is found along the Gulf Coast from the Florida Keys to Tamaulipas in Mexico (Cazier 1954: 261) [see Pearson et al. 1997: Fig. 4]; also recorded from Yucatán (Horn 1897a: 354).
USA: AL, FL, LA, MS, TX – Mexico
Habroscelimorpha yucatana (Horn) from Yucatán is considered a subspecies of Habroscelimorpha severa by some authors, including Erwin and Pearson (2008: 253).
This species, also known as the “Elusive Tiger Beetle”, is found along the Atlantic Coast from southern South Carolina (Cartwright 1935: 73; Ciegler 1997: 191) to central Florida, and along the Gulf Coast of Florida [see Pearson et al. 1997: Fig. 10].
USA: FL, GA, SC
One North American species which extends into northern Mexico.
This subspecies, the “Salt Flat Tiger Beetle”, is known only from Torrance and Guadalupe Counties in central New Mexico and Hudspeth County in western Texas (Pearson et al. 2006: 148).
USA: NM, TX
This subspecies, the “Alkali Tiger Beetle”, ranges from eastern Nebraska (Carter 1989: 15) and central Colorado (Kippenhan 1990: 312) south to northern Texas (Gaumer and Murray 1971: 10) and southeastern New Mexico (Acciavatti et al. 1980: 31) [see Pearson et al. 2006: Map 80]. The record from north-central Utah (Tanner 1929a: 87) is probably in error.
USA: CO, KS, NE, NM, OK, TX
This subspecies, also known as the “White-cloaked Tiger Beetle”, is known from scattered localities from southern South Carolina (Cartwright 1935: 75) to northeastern Texas, south to northern Florida (Choate 2003: Map 48) and Tamaulipas (Cazier 1954: 297) [see Pearson et al. 2006: Map 80]. Ciegler (1997: 191) reported that the last specimen seen from South Carolina was collected in 1935 and that the species may be extinct in the state.
USA: AL, FL, LA, MS, SC, TX – Mexico
Worldwide, with about 340 species described by 2005 assigned to 24 subgenera (Lorenz 2005: 41-51). The North American fauna is represented by 60 species (about 18% of the world fauna) placed in two subgenera.
The genus is employed here in a restricted sense as used by most taxonomists working on the Palaearctic and African faunas and recently by Erwin and Pearson (2008) for the North American fauna.
Western Hemisphere, with about 65 species (Lorenz 2005: 48-49) in the Nearctic (21 species, 42 species-group taxa) and Neotropical (about 55 species).
1. Cicindela fera Chevrolat is known north of Mexico from a single specimen collected in the 1950s at the southern border of Arizona and New Mexico (Pearson et al. 2006: 125). The specimen was probably a stray and the species is not listed here as a North American entity. 2. Cicindela sommeri Mannerheim is known from the Sierra Madre Occidental of western Mexico and from several specimens labeled from San Diego County, California (Leng 1902: 181; Pearson et al. 2006: 136). However, there is serious doubt about the origin of the California specimens and the species is not included here as a North American entity.
This species, also known as the “Eastern Pinebarrens Tiger Beetle”, is found mainly along the Coastal Plain and Piedmont Plateau from Long Island in southeastern New York (Leng 1928: 206) to central Florida, west to southeastern Louisiana [see Pearson et al. 1997: Fig. 11].
USA: AL, DE, FL, GA, LA, MD, MS, NC, NJ, NY, PA, SC, VA
This subspecies, also known as the “Great Basin Tiger Beetle”, is found in the Death Valley area in eastern California (Leffler 1987: 8).
USA: CA
1. Rumpp (1956: 141) reported the presence of intergrade populations between this subspecies and the nyensis form at places located between the type localities of amargosae and nyensis. 2. Cicindela amargosae has been regarded as a subspecies of Cicindela senilis LeConte by some authors (e.g., Willis 1968) or Cicindela willistoni LeConte (e.g., Kippenhan 1996b: 56) but treated as a closely related but distinct species by Leffler (1987: 8) and Pearson et al. (2006: 117).
This subspecies, also known as the “Nye Tiger Beetle”, is found in southeastern Oregon (Leffler 1979a: Fig. 60) and western Nevada (Rumpp 1956: 140).
USA: NV, OR
Kippenhan (2005) indicated from an analysis of populations that the variation in the dorsal coloration in Cicindela amargosae did not coincide with the accepted subspecific criteria. Probably the form nyensis should not be recognized as a valid entity. Kippenhan (2005: Fig. 1) provided a detailed map of the known populations of Cicindela amargosae.
This species, also known as the “Cazier’s Tiger Beetle”, is found along a small area in Jim Hogg and Starr Counties, southeastern Texas [see Pearson et al. 2006: Map 63], and in Tamaulipas, Mexico (Erwin and Pearson 2008: 127).
USA: TX – Mexico
This taxon is listed as a subspecies of Cicindela politula LeConte by some authors (e.g., Murray and Acciavatti 1976).
This species is known only from a few sites in the Richmond Heights area of Miami (Brzoska et al. 2011: 5).
USA: FL
This subspecies, also known as the “Grand Canyon Tiger Beetle”, is restricted to the Colorado River at the bottom of the Grand Canyon in northern Arizona and along the Virgin River in adjacent Utah and Nevada (Pearson et al. 2006: 135).
USA: AZ, NV, UT
The range of this subspecies, also known as the “Wetsalts Tiger Beetle”, extends from central Washington to northwestern Wyoming, south to western Texas and along the Pacific Coast to the northern parts of the Baja California Peninsula (Cazier 1948: 11) [see Pearson et al. 1997: Fig. 35].
USA: AZ, CA (CHI), CO, ID, NM, NV, OR, TX, UT, WA, WY – Mexico
1. Some authors (e.g., Nagano 1982: 39) have treated Cicindela pacifica Schaupp as a valid subspecies of Cicindela hemorrhagica LeConte. 2. Freitag (1999: 62), Pearson et al. (2006: 135), and Erwin and Pearson (2008: 141) considered Cicindela woodgatei Casey as a valid subspecies of Cicindela hemorrhagica LeConte despite the fact that there seem to be no consistent characters to separate the adults from those of the nominate form. 3. Another subspecies, Cicindela hemorrhagica hentziana Leng, is found in Baja California; its record from “Utah” (Leng 1920: 42) is in error.
This species, also known as the “Highlands Tiger Beetle”, is found in Highlands and Polk Counties, central Florida (Choate 2003: 84; Pearson et al. 2006: 126).
USA: FL
This species, also known as the “Horn’s Tiger Beetle”, ranges from southern Arizona to southwestern Texas, south to Durango (Cazier 1954: 248) [see Pearson et al. 1997: Fig. 25].
USA: AZ, NM, TX – Mexico
The subspecies Cicindela hornii scotina Bates is known from the states of Chihuahua, Durango, and Zacatecas in Mexico (Erwin and Pearson 2008: 146).
The range of this species, also known as the “Cobblestone Tiger Beetle”, is disjunct: one population is known from New Brunswick (Sabine 2005: 53) south to central New Jersey (Boyd 1978: Fig. 28), northwestern West Virginia (Allen and Acciavatti 2002: 26), southeastern Kentucky (Laudermilk et al. 2010: 28), and southeastern Indiana; the second is found in northeastern Mississippi and western Alabama [see Pearson et al. 2006: Map 67]. The record from “South Carolina” (Choate 2003: Map 30) needs confirmation.
CAN: NB USA: AL, IN, KY, ME, MS, NH, NJ, NY, OH, PA, VT, WV [SC]
This species is listed on the IUCN Red List of Threatened Species (IUCN 2007) and has been extirpated from many historical sites (Erwin and Pearson 2008: 155).
This subspecies, the “Bowditch’s Tiger Beetle”, is known from southwestern Colorado (Kippenhan 1994: 65) and northwestern New Mexico (Rumpp 1962: 172). The record from “Arizona” (Boyd 1982: 11) is in error or based on a stray.
USA: CO, NM
This subspecies intergrades with the nominate form in north-central New Mexico (Pearson et al. 2006: 119).
This subspecies, also known as the “Black Sky Tiger Beetle”, occurs from Salton Sea in southern California (LaRue 1991: 49) and southernmost Nevada (Kippenhan 2002: 381) to western Texas (Gaumer and Murray 1971: 10), north to southwestern Utah (Tanner 1929a: 85) and northeastern Colorado (Kippenhan 1990: 311), south to Aguascalientes and San Luis Potosí in Mexico (Cazier 1960: 8).
USA: AZ, CA, CO, KS, NM, NV, OK, TX, UT – Mexico
This subspecies, the “Subtropic Tiger Beetle”, is confined to Hidalgo and Cameron Counties in southern Texas (Pearson et al. 2006: 119).
USA: TX
This subspecies, the “Rio Grande Grassland Tiger Beetle”, is known from the lower Rio Grande Valley in southern Texas from Maverick County to Hidalgo County (Pearson et al. 2006: 122), north to Kimble County in central Texas (Mawdsley 2009: 9).
USA: TX
This subspecies, also known as the “Large Grassland Tiger Beetle”, ranges from western Kansas to central Arizona, north to northern Colorado (Kippenhan 1994: 66), south to southern New Mexico and southwestern Texas; also known from one locality in eastern Kansas [see Mawdsley 2009: Fig. 18]. The record from “Utah” (Boyd 1982: 11) is likely in error.
USA: AZ, CO, KS, NM, OK, TX
This subspecies intergrades with the santaclarae form in New Mexico and western Texas (Mawdsley 2009: 6). Two other subspecies of this species are found in Mexico, Cicindela obsoleta juvenilis Horn from the states of Jalisco, Nayarit, Sonora, and Sinaloa and Cicindela obsoleta latemaculata Becker from the state of Durango (Erwin and Pearson 2008: 159, 160).
This subspecies, the “Santa Clara Grassland Tiger Beetle”, ranges from southern Colorado (Kippenhan 1994: 67) south to northern Durango (Cazier 1954: 251), including southwestern Texas and western Arizona [see Mawdsley 2009: Fig. 18].
USA: AZ, CO, NM, TX – Mexico
This subspecies, the “Prairie Tiger Beetle”, ranges from southern Missouri and north-central Arkansas to north-central Texas, south to southeastern Texas [see Mawdsley 2009: Fig. 18] and Coahuila in Mexico (Cazier 1954: 250). The record from central New Mexico (Fall and Cockerell 1907: 154) is suspect (Mawdsley 2009: 8); that from “Colorado” (Wickham 1902: 228) is probably in error.
USA: AR, LA, MO, OK, TX [NM] – Mexico
This subspecies, also known as the “Ocellated Tiger Beetle”, occurs from southeastern Arizona and adjacent New Mexico (Pearson et al. 2006: 133) south to Costa Rica (Blackwelder 1944: 18); also recorded from “Texas” (Erwin and Pearson 2008: 162).
USA: AZ, NM [TX] – Belize, Costa Rica, El Salvador, Guatemala, Honduras, Mexico, Nicaragua
This subspecies, the “Dark-abdomened Tiger Beetle”, ranges from western Louisiana (Graves and Pearson 1973: 180) to northern New Mexico [see Pearson et al. 2006: Map 66], including southern Oklahoma (Schmidt 2004: 5), south to southern Tamaulipas (Cazier 1954: 278).
USA: LA, NM, OK, TX – Mexico
This subspecies, the “Barbaraann’s Tiger Beetle”, occurs in the Hueco, Sierra Diablo, and Apache mountains in western Texas (Gage 1988: 146-147) and in the Sacramento Mountains of southern New Mexico (Pearson et al. 2006: 129) where it is found above 1500 m.
USA: NM, TX
This subspecies, the “Rock-loving Tiger Beetle”, is known only from above 1670 m in the Guadalupe Mountains in western Texas and southeastern New Mexico (Gage 1988: 146).
USA: NM, TX
This subspecies, also known as the “Limestone Tiger Beetle”, occurs from Carter and Murray Counties in southern Oklahoma (Pearson et al. 2006: 128) south to Coahuila and Nuevo León (Sumlin 1985: Fig. 9).
USA: OK, TX – Mexico
Another subspecies, Cicindela politula laetipennis Horn, is known from the state of Coahuila in Mexico.
This subspecies, the “Green Mountain Tiger Beetle”, is known only from the type locality in southeastern New Mexico.
USA: NM
This subspecies, the “Chihuahua Tiger Beetle”, is known from northeastern Colorado to west-central Nevada [see Pearson et al. 2006: Map 56], south to Chihuahua (Cazier 1954: 253). The records from Oklahoma (Drew and Van Cleave 1962: 113), “Nebraska, ” “Kansas, ” and “Texas” (Freitag 1999: 68) apparently refer to intergrades and these records are registered under the nominotypical subspecies.
USA: AZ, CO, NM, NV, UT – Mexico
Bertholf (1983: 21) listed this form in synonymy with the nominotypical subspecies.
This subspecies, also known as the “Punctured Tiger Beetle”, ranges from New Brunswick to southern Alberta, south to southern Texas and southern Florida [see Pearson et al. 2006: Map 56].
CAN: AB, MB, NB, ON, QC, SK USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV, WY
The two subspecies of Cicindela punctulata intergrade over a large area in southwestern United States (Pearson et al. 2006: 123) and northern Mexico (Murray 1979: 51). Another subspecies, Cicindela punctulata catharinae Chevrolat, is endemic to Mexico.
This subspecies, also known as the “Tascoa Tiger Beetle”, has been recorded from Oldham County in northwestern Texas (Davis 1918: 34), Alajuela province in Costa Rica (Horn 1905: 22), and Panama (Erwin and Pearson 2008: 178).
USA: TX – Costa Rica, Panama
1. No specimens of this subspecies have been collected in United States since the original ones in 1917 and the subspecies has never been found in Mexico. Cazier (1954: 279) believed that the US specimens of Cicindela tascosaensis were probably mislabeled. However, Davis (1922: 130) wrote to the collector of the Texan specimens, Miss Mildred McGill, who replied on December 1920 that she remembered well collecting the tiger beetles “on the sandy, grassy spots of the ground, and on the wide floors of white sand rocks” about “a mile or a little more” of the house she lived in. 2. Two other subspecies, Cicindela roseiventris mexicana Klug and Cicindela roseiventris roseiventris Chevrolat, are found in Mexico and in Central America.
This subspecies, the “Mexican Red-bellied Tiger Beetle”, ranges from Louisiana (Pearson et al. 2006: 131) south through Texas to Queretaro and Veracruz (Murray 1979: 53). The records from “Georgia” (Boyd 1982: 12) and western Alabama (Löding 1945: 9) probably refer to the nominotypical subspecies; those from “Mississippi, ” “Arkansas” (Boyd 1982: 12), and Oklahoma (Drew and Van Cleave 1962: 114) are probably based on intergrades and these records are listed under the nominotypical subspecies.
USA: LA, TX – Mexico
This subspecies, the “Hentz’s Tiger Beetle”, is found only along eastern Massachusetts [see Leonard and Bell 1999: Fig. 111]. The record from “Rhode Island” (Bousquet and Larochelle 1993: 64) is in error or based on a stray.
USA: MA
This subspecies, also known as the “Eastern Red-bellied Tiger Beetle”, ranges from southwestern Vermont (Leonard and Bell 1999: 104) to southwestern Missouri, south to east-central Texas and the Florida Panhandle [see Pearson et al. 2006: Map 64].
USA: AL, AR, CT, DC, DE, FL, GA, IL, IN, KY, LA, MA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WV
This subspecies intergrades with the cumatilis form through southern Missouri, Arkansas, and Louisiana (Pearson et al. 2006: 131). Another subspecies, Cicindela rufiventris reducens Horn, is known from the states of Jalisco and Colima in Mexico.
This species, also known as the “Scabrous Tiger Beetle”, is found from southeastern Georgia to southern Florida [see Pearson et al. 2006: Map 61].
USA: FL, GA
This species, also known as “Schaupp’s Tiger Beetle”, occurs from southeastern Kansas and northwestern Arkansas [see Pearson et al. 2006: Map 69] south to Nuevo León (Cazier 1954: 287). The record from “Missouri” (Erwin and Pearson 2008: 181) needs confirmation.
USA: AR, KS, OK, TX [MO] – Mexico
This subspecies, also known as the “Western Red-bellied Tiger Beetle”, ranges from northeastern New Mexico to central Arizona [see Pearson et al. 2006: Map 65], south to Guanajuato in Mexico (Cazier 1954: 271).
USA: AZ, NM, TX – Mexico
Besides the nominotypical subspecies, four other subspecies are known, ranging collectively from Mexico to Costa Rica (see Pearson et al. 2006: 132; Erwin and Pearson 2008: 185-186).
This species, also known as the “Senile Tiger Beetle”, is found along western California, as far north as Sonoma and Lake Counties, and the northern part of the Baja California Peninsula. According to Pearson et al. (2006: 116), it is now known in the United States only from a few protected coastal populations and two interior populations, one near Lake Elsinore in western Riverside County and one near Jacumba in San Diego County.
USA: CA (CHI) – Mexico
This species, also known as the “Thin-lined Tiger Beetle”, ranges from western Nebraska (Brust 2007: 9) to southern California [see Pearson et al. 1997: Fig. 38], south to northern Sinaloa and southern Tamaulipas (Cazier 1954: 257).
USA: AZ, CA, CO, KS, NE, NM, NV, OK, TX, UT – Mexico
This highly vagile subspecies, also known as the “Ascendant Tiger Beetle”, is found primarily along the Atlantic and Gulf Coasts from Virginia to southern Florida, west to eastern Texas [see Pearson et al. 2006: Map 70], south to Panama (Erwin and Pearson 2008: 197). This form has also been found inland as far north as north-central Kansas (Charlton and Kopper 2000: 266) and along the Atlantic Coast as far north as Massachusetts (Comboni and Schultz 1989: 151); however there is no known established populations inland anywhere in North America (Pearson and Vogler 2001: 105). The record from the “West Indies” (LeConte 1856a: 51) needs confirmation.
USA: AL, AR, FL, GA, KS, LA, MA, MD, MO, MS, NC, NJ, OK, SC, TN, TX, VA – Belize, Costa Rica, El Salvador, Guatemala, Honduras, Mexico, Nicaragua, Panama.
Besides the two subspecies found in North America, six other subspecies are recognized among this polymorphic species in Middle and South America and the West Indies.
This subspecies, the “Sigmoid Tiger Beetle”, is found along the Pacific Coast from southern California (Nagano 1982: 38) to the Baja California Peninsula (Cazier 1954: 291); it is occasionally found inland, as far as the Salton Sea in Imperial County (see LaRue 1991). The record from “Arizona” (Freitag 1999: 74) needs confirmation.
USA: CA (CHI) [AZ] – Mexico
This subspecies, the “Echo Tiger Beetle”, is found mainly within the Great Basin from Wyoming to southern Oregon, south to east-central California, southern Nevada, and southern Utah (Pearson et al. 2006: 114).
USA: CA, ID, NV, OR, UT, WY
This subspecies, the “Torrance Tiger Beetle”, is known only from Torrance County in central New Mexico (Pearson et al. 2006: 114).
USA: NM
This subspecies, the “Funaro’s Tiger Beetle”, is known only from the type locality in northwestern New Mexico.
USA: NM
This subspecies, the “Hairy-fronted Tiger Beetle”, is found in central Kansas, western Oklahoma, west-central Texas, and east-central New Mexico (Willis 1967: 302); also recorded from “Arizona” (Erwin and Pearson 2008: 202).
USA: KS, NM, OK, TX [AZ]
This subspecies, the “Augured Tiger Beetle”, is known from Inyo County in eastern California and Nye County in Nevada (Rumpp 1956: 135).
USA: CA, NV
This subspecies, the “Owens Lake Tiger Beetle”, is restricted to Owens and adjacent Panamint Valley of east-central California (Pearson et al. 2006: 115).
USA: CA
This subspecies, the “Sulphur Valley Tiger Beetle”, is endemic to the Sulphur Springs Valley in southeastern Arizona.
USA: AZ
This subspecies, also known as the “Williston’s Tiger Beetle”, is endemic to the Laramie Plain of Wyoming (Rumpp 1962: 168).
USA: WY
This subspecies intergrades with the echo form to the west (Pearson et al. 2006: 114). In a cladistic analysis based on molecular data by Vogler and Welsh (1997), Cicindela willistoni clearly embedded within the subgenus Cicindela while morphological characters suggest that it belongs to the subgenus Cicindelidia.
Northern Hemisphere, with about 75 species (Lorenz 2005: 43-48) in the Nearctic (38 species, of which four extend into northern Mexico; 92 species-group taxa) and Palaearctic (36 species) Regions.
[decemnotata group]
This eastern species, also known as the “Appalachian Tiger Beetle”, ranges from southwestern New Brunswick (Webster and Bousquet 2008: 16) south to northeastern Georgia, west at least to eastern Kentucky (Laudermilk et al. 2010: 28) [see Pearson et al. 1997: Fig. 15]. Old records from Indiana, northern Illinois, and western Missouri (see Wilson and Larochelle 1980: 37-38) suggest that the species was more widely distributed at one time. Beaton (2008: 40) did not find the species in Georgia during his intensive survey of tiger beetles in the state.
CAN: NB, QC USA: GA, KY, MA, MD, ME, NC, NH, NJ, NY, OH, PA, TN, VA, VT, WV [IL, IN, MO]
This species, the “Death Valley Tiger Beetle”, is known from the Death Valley region (Kritsky and Horner 1998: 17) in Inyo County, California, and Nye County, Nevada.
USA: CA, NV
This form has been listed as a subspecies of Cicindela tranquebarica Herbst by most authors, including Erwin and Pearson (2008: 191), but Kritsky and Horner (1998: 17) found enough structural differences to substantiate that it represents a distinct species.
This subspecies is restricted to the area of ancient Lake Bonneville in north-central Utah [see Knisley et al. 2012: Fig. 23].
USA: UT
This subspecies, also known as the “Badlands Tiger Beetle”, ranges in patchy colonies from eastern Alaska south through the Rocky Mountains to northeastern New Mexico and southern Utah, east to western North Dakota and western Nebraska [see Knisley et al. 2012: Figs 23, 24]. The records from “Kansas” (Leng 1902: 134) and “Manitoba” (Knisley et al. 2012: 14) need confirmation.
CAN: AB, SK, YT USA: AK, CO, ID, MT, ND, NE, NM, UT, WY [KS, MB]
This subspecies ranges from south-central British Columbia to southeastern Washington [see Knisley et al. 2012: Fig. 23].
CAN: BC USA: WA
This subspecies is restricted to high elevations of the Bear River Mountains of southeastern Idaho and northeastern Utah [see Knisley et al. 2012: Fig. 23].
USA: ID, UT
This species, also known as the “Green Claybank Tiger Beetle”, inhabits the Great Plains from eastern Montana and North Dakota south to northern Louisiana, northern Texas, and northeastern New Mexico [see Schincariol and Freitag 1991: Fig. 13].
USA: AR, CO, KS, LA, MT, ND, NE, NM, OK, SD, TX, WY
According to Pearson et al. (2006: 92), individuals with green elytra and blue head and thorax from northwestern Louisiana and southwestern Arkansas (originally described under the name ludoviciana) may be either an isolated population of this species or a local green morph of Cicindela splendida. They also added that based on the ecology, behavior, and distribution, the greenish population is more likely a local variant of Cicindela splendida.
This subspecies, also known as the “Crimson Saltflat Tiger Beetle”, ranges from Minnesota to southern Alberta (Hilchie 1985: 330), south to northeastern Arizona and northern Texas [see Pearson et al. 1997: Map 11]. The record from “Saskatchewan” (Freitag 1999: 27) needs confirmation.
CAN: AB USA: AZ, CO, KS, MN, MT, ND, NE, NM, OK, SD, TX, UT, WY [SK]
Pearson et al. (2006: 99), followed by Erwin and Pearson (2008: 138), considered Cicindela fulgida williamlarsi Knudsen and Cicindela fulgida winonae Knudsen as synonyms of Cicindela fulgida pseudowillistoni Horn. They also listed Cicindela fulgida rumppi Knudsen as a valid subspecies restricted to the Laguna del Perro area in Torrance County, central New Mexico.
This subspecies, also known as the “Alkaline Tiger Beetle”, is found in southern Wyoming and northwestern Colorado (Kippenhan 1996a: 42).
USA: CO, WY
Pearson et al. (2006: 99) recorded this subspecies from a much larger area, throughout the western Great Plains and intermontane southern Rocky Mountains.
This subspecies, also known as the “Westbourne’s Tiger Beetle”, is found in southern Manitoba, southern Saskatchewan (Wallis 1961: 51), north-central North Dakota, and northwestern Minnesota (Knudsen 1985: 186); also recorded from “Montana” (Erwin and Pearson 2008: 139).
CAN: MB, SK USA: MN, ND, UT [MT]
This subspecies, also known as the “Western Beach Tiger Beetle”, is found along the Pacific Coast from southern California to the central parts of the Baja California Peninsula (Cazier 1948: 14) and also along the Gulf of California coast in northern Sonora [see Pearson et al. 1997: Fig. 27]. According to Pearson et al. (2006: 195), this taxon is now gone from most of its former sites in southern California.
USA: CA – Mexico
Some authors (e.g., Nagano 1982: 37) consider Cicindela obliviosa Casey as a valid subspecies. Cicindela latesignata parkeri Cazier is found in Sonora and Baja California in Mexico.
This subspecies, the “Jorda’s Tiger Beetle”, is known from northeastern Arizona (Bertholf 1983: 12) and northern New Mexico (Acciavatti et al. 1980: 30). Based on Pearson et al. (2006: Map 47), it is also found in southern Utah and southwestern Colorado. The record from “Wyoming” (Boyd 1982: 9) probably refers to the nominotypical subspecies.
USA: AZ, CO, NM, UT
This subspecies, also known as the “Blowout Tiger Beetle”, ranges from western South Dakota (Spomer et al. 2008a: 57) and southern Wyoming, south to northern New Mexico, extreme northwestern Texas, and southern Oklahoma [see Pearson et al. 2006: Map 47]. The records from “Montana” (Horn 1915: 372) and “Iowa” (Boyd 1982: 9) need confirmation.
USA: CO, KS, MO, NE, NM, OK, SD, TX, WY [IA, MT]
This subspecies intergrades with the jordai form in the southwestern part of its range and with the versuta form in the northern part of its range.
This subspecies, the “Adroit Tiger Beetle”, ranges from southern Manitoba to northern Alberta (Wallis 1961: 56), south to Wyoming and northwestern South Dakota (Spomer et al. 2008a: 34).
CAN: AB, MB, SK USA: MT, ND, SD, WY
This species, also known as the “Common Claybank Tiger Beetle”, ranges from Newfoundland to eastern British Columbia, north to northern Yukon Territory (Eagle River, Sydney G. Cannings pers. comm. 2009), south to eastern Utah, northern New Mexico, central Missouri, southern Pennsylvania, and New Jersey [see Schincariol and Freitag 1991: Fig. 13; Pearson et al. 1997: Fig. 3]. According to Knisley and Schultz (1997: 114), the literature records from Virginia, western North Carolina, and northwestern Georgia could refer instead to Cicindela splendida. The record from the District of Columbia (Boyd 1982: 8) needs confirmation.
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: CO, CT, IA, IL, IN, KS, KY, MA, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NM, NY, OH, PA, RI, SD, UT, VT, WI, WY [DC, GA, NC, VA]
Spomer et al. (2008a: 25) noted that this species intergrades occasionally with Cicindela denverensis, rarely with Cicindela splendida. Based on results from a limited mitochondrial DNA analysis, Woodcock and Knisley (2010) concluded that Cicindela limbalis, Cicindela splendida, and Cicindela denverensis may represent a single species.
This species, also known as the “Autumn Tiger Beetle”, is confined to the Coastal Plain and Piedmont Plateau ranging from “North Carolina” (Knisley and Schultz 1997: 116) to the Florida Panhandle (Choate 2003: Map 13), west to southeastern Mississippi (George County, Drew A. Hildebrandt pers. comm. 2009).
USA: AL, FL, GA, MS, NC, SC
This species, the “Ohlone Tiger Beetle”, is known only from remnant stands of native grassland on coastal terraces in Santa Cruz County, California (Freitag et al. 1993: 117). According to Pearson et al. (2006: 193), it is known from only nine sites where populations range from less than 100 to several hundreds.
USA: CA
This species has been listed as endangered under the Endangered Species Act by the U.S. Fish and Wildlife Service in October 2001. Collection of specimens is illegal (Pearson et al. 2006: 88).
This subspecies, also known as the “Dark Saltflat Tiger Beetle”, occurs from southeastern Oregon and southwestern Idaho to southwestern Utah (Leffler 1987: 7). The record from “Washington” (Freitag 1999: 39) probably refers to the wallisi form.
USA: ID, NV, OR, UT
Some authors, including Freitag (1999: 39), consider Cicindela remittens Casey as a valid subspecies of Cicindela parowana Wickham. According to Pearson et al. (2006: 100), all three subspecies of Cicindela parowana intergrade in a narrow zone in southeastern Oregon.
This subspecies, the “Platt Tiger Beetle”, occurs in southeastern Oregon, east-central California, and western Nevada [see Pearson et al. 2006: Map 42].
USA: CA, NV, OR
This subspecies, the “Wallis’ Tiger Beetle”, ranges from south-central British Columbia south to southeastern Oregon (Leffler 1987: 7), including southwestern Idaho (Shook 1984: 159).
CAN: BC USA: OR, WA
This species, also known as the “Cochise Tiger Beetle”, is restricted to southeastern Arizona, southwestern New Mexico [see Pearson et al. 1997: Fig. 31] and adjacent regions in Sonora, Mexico (LeConte 1856a: 62, as Cicindela viatica Chevrolat).
USA: AZ, NM – Mexico
This rare species, also known as the “Alpine Tiger Beetle”, ranges from southern Idaho to south-central Oregon, south to east-central California and west-central Utah [see Pearson et al. 2006: Map 31]. The record from one locality in northern Montana (see Pearson et al. 2006: Map 31) is possibly based on a stray.
USA: CA, ID, NV, OR, UT [MT]
This species, also known as the “Sagebrush Tiger Beetle”, is found from southern British Columbia south to north-central Oregon [see Pearson et al. 1997: Fig. 32].
CAN: BC USA: OR, WA
According to Pearson et al. (2006: 89), further studies may show that this taxon is rather a subspecies of Cicindela plutonica Casey.
This subspecies, the “Audubon’s Tiger Beetle”, ranges from southern Manitoba to central British Columbia, south to east-central California, Arizona, and northern Texas [see Pearson et al. 2006: Map 28]. The records from “Wisconsin, ” “Illinois, ” “Kentucky, ” “Tennessee” and “Arkansas” (Boyd 1982: 7) apparently refer to the nominate form.
CAN: AB, BC, MB, SK USA: AZ, CA, CO, IA, ID, KS, MN, MT, ND, NE, NM, NV, OK, OR, SD, TX, UT, WA, WY
This subspecies, the “Cimarron Tiger Beetle”, is known from north and central Colorado (Kippenhan 1994: 44-45), much of New Mexico (Acciavatti et al. 1980: 30), and southeastern Arizona (Bertholf 1983: 22) [see Pearson et al. 2006: Map 28]. The records from northern (Tanner 1929a: 79) and southwestern (Horn 1926: 266) Utah need confirmation.
USA: AZ, CO, NM [UT]
This subspecies, also known as the “Hatch’s Tiger Beetle”, ranges from Vancouver Island to the central region of the Sierra Nevada in California (Leffler 1987: 4).
CAN: BC (VCI) USA: CA, OR, WA
This subspecies intergrades with the lauta form in the Willamette Valley of Oregon south to Shasta and Modoc Counties in northwestern California (Pearson et al. 2006: 87).
This subspecies, the “Elegant Tiger Beetle”, ranges from the Columbia River in southern Washington to northwestern California (Leffler 1987: 4).
USA: CA, OR, WA
This subspecies, also known as the “Cow Path Tiger Beetle”, ranges from southern Quebec to Minnesota, south to central Arkansas and northern Georgia [see Pearson et al. 2006: Map 28]. Beaton (2008: 39) indicated that despite extensive search he was unable to find any population of this species in Georgia, including at all known historical sites, and Ciegler (1997: 189) noted that it has not been collected in South Carolina since 1936. The record from “Nova Scotia” (Bousquet and Larochelle 1993: 59) was based on a misidentified specimen (see Majka et al. 2007: 6); that from “New Brunswick” (Erwin and Pearson 2008: 175) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, VA, VT, WI, WV [NB]
This subspecies intergrades with the audubonii form over a wide area from North Dakota to Oklahoma (Pearson et al. 2006: 86).
This subspecies, the “Chartreuse Tiger Beetle”, is known only from north-central Texas (Pearson et al. 2006: 102).
USA: TX
This subspecies, the “LeConte’s Tiger Beetle”, ranges from southern Quebec to southern Manitoba (Wallis 1961: 35-36), south to Kansas, northern Mississippi, West Virginia, and Connecticut [see Pearson et al. 2006: Map 43). The records from “Saskatchewan” and “Alberta” (Bousquet and Larochelle 1993: 60) refer to the nominotypical subspecies.
CAN: MB, ON, QC USA: CT, IA, IL, IN, KS, MA, ME, MI, MN, MO, MS, ND, NE, NH, NY, OH, PA, SD, TN, VT, WI, WV
This subspecies intergrades with the nominate form over a broad area in the Great Plains and with the unicolor form in northern Missouri and Tennessee (Pearson et al. 2006: 102, 103).
This subspecies, the “Rugate Tiger Beetle”, is known from southwestern Arkansas (Ward 1972: 70), western Louisiana (Graves and Pearson 1973: 175), and eastern Texas (Vaurie 1950: 4). The record from “Oklahoma” (Erwin and Pearson 2008: 183) needs confirmation.
USA: AR, LA, TX [OK]
This subspecies intergrades with the nominate form in the northwestern part of its range and with the lecontei form in the northeastern part of its range (Pearson et al. 2006: 103).
This subspecies, also known as the “Wrinkle-fronted Tiger Beetle”, ranges east of the Appalachians from Massachusetts (Leonard and Bell 1999: 133) to North Carolina (Harris 1911: 28, as Cicindela scutellaris var. carolina). The records from South Carolina (Cartwright 1935: 72) and “Georgia” (J.E. LeConte 1849: 25) probably refer to the unicolor form.
USA: CT, DC, DE, MA, MD, NC, NJ, NY, PA, RI, VA
This form intergrades with the lecontei form in Massachusetts and Connecticut and with the unicolor form in North Carolina (Pearson et al. 2006: 103).
This subspecies, also known as the “Festive Tiger Beetle”, ranges from southwestern Saskatchewan and southeastern Alberta (Wallis 1961: 35) south to east-central New Mexico (Acciavatti et al. 1980: 30), northern Texas (Gaumer and Murray 1971: 10), Arkansas (Graves and Pearson 1973: 175), and northwestern Mississippi (Bolivar County, Drew A. Hildebrandt pers. comm. 2009). The record from “Iowa” (Freitag 1999: 49) probably refers to the lecontei form.
CAN: AB, SK USA: AR, CO, KS, MS, MT, ND, NE, NM, OK, SD, TX, WY
According to Pearson et al. (2006: 102), this subspecies intergrades with the lecontei form over a broad zone in eastern South Dakota, Nebraska, and Kansas.
This subspecies, the “Unicolored Tiger Beetle”, is known from eastern Tennessee and North Carolina (Pearson et al. 2006: 103) south to central Florida (Peck and Thomas 1998: 16; Choate 2003: Map 74), southern Alabama (Löding 1945: 9), and southern Mississippi (Graves and Pearson 1973: 174); also recorded from “Virginia” (Erwin and Pearson 2008: 184). The records from Texas (Tucker 1906: 85), “Louisiana, ” “Colorado” (Leng 1902: 125), New Mexico (Fall and Cockerell 1907: 155), and Oklahoma (Drew and Van Cleave 1962: 115) could be based on strays, on intergrades, or be in error.
USA: AL, FL, GA, MS, NC, SC, TN [VA]
According to Pearson et al. (2006: 103), this form intergrades with the rugifrons form in North Carolina and with the lecontei form in northern Missouri and Tennessee.
This subspecies, the “Yampa Tiger Beetle”, is known only from the Maybell Sand Hills area in northwestern Colorado (Kippenhan 1994: 55).
USA: CO
This species, also known as the “Splendid Tiger Beetle”, occurs from southern Pennsylvania to eastern Wyoming, north to southern Wisconsin, south to central Texas, northeastern Georgia, and northern South Carolina [see Schincariol and Freitag 1991: Fig. 13; Pearson et al. 1997: Fig. 20]; The records from “New York” (Schaupp 1884a: 90), “Minnesota” (Horn 1928: 10), and “New Mexico” (Freitag 1999: 50, 51) need confirmation.
USA: AL, AR, CO, DC, GA, IA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NE, OH, OK, PA, SC, SD, TN, TX, VA, WI, WV, WY [MN, NM, NY]
1. The name Cicindela discus, credited to Klug (1834: 23), is often listed as a synonym of Cicindela splendida Hentz but Klug did not apply the name to a new species. 2. According to Pearson et al. (2006: 91), recent DNA studies on this species, Cicindela denverensis and Cicindela limbalis suggest that members of all three could be conspecific. Spomer et al. (2008a: 23) noted that this species hybridizes with Cicindela denverensis in a narrow zone in central Nebraska and possibly also with Cicindela limbalis.
This species, also known as the “Short-legged Tiger Beetle”, is found from southern Oregon to east-central California, east to southeastern Utah [see Pearson et al. 2006: Map 48]. The records from “Arizona” and “New Mexico” (Bousquet and Larochelle 1993: 61) are in error or based on strays.
USA: CA, NV, OR, UT
This subspecies is known only from east-central Arizona [see Kritsky and Horner 1998: Fig. 5].
USA: AZ
Bertholf (1983: 26) considered this form as a synonym of the lassenica form (= Cicindela tranquebarica parallelonota Casey).
This subspecies, the “Diffracted Tiger Beetle”, is found from southern Wyoming to New Mexico and eastern Arizona (Kritsky and Horner 1998: 25, Fig. 5). According to Pearson et al. (2006: 107), it occurs in southern Nevada, northern Arizona, and New Mexico; also recorded from Utah (Tanner 1929a: 80) and “Nebraska” (Erwin and Pearson 2008: 192).
USA: AZ, CO, NM, NV, WY [NE, UT]
This subspecies, the “Joaquin Tiger Beetle”, is known only from the San Joaquin Valley of California.
USA: CA
According to Knisley and Haines (2007: 109), this subspecies intergrades with the vibex form along the margins of the San Joaquin Valley.
This subspecies, the “Kirby’s Tiger Beetle”, ranges from Manitoba to Alberta, north to southern Northwest Territories; it southern limit is debated. Kritsky and Horner (1998: Fig. 5) placed it at southern Wyoming and northern Nebraska while Pearson et al. (2006: Map 45) placed it at southeastern New Mexico and northern Texas. I have accepted the latest range for the records. The subspecies is also recorded from “British Columbia, ” “Ontario, ” “Arkansas, ” “Iowa, ” “Minnesota, ” “Missouri, ” “Utah, ” and “Washington” by Erwin and Pearson (2008: 193).
CAN: AB, MB, NT, SK USA: CO, KS, MT, ND, NE, NM, OK, SD, TX, WY [AR, BC, IA, MN, MO, ON, UT, WA]
This subspecies intergrades with the nominotypical form over a large area in the Midwest (Pearson et al. 2006: 106).
This subspecies, also known as the “Opal Tiger Beetle”, occurs in Utah, Nevada, and eastern California (Kritsky and Horner 1998: 25).
USA: CA, NV, UT
Some authors, including Pearson et al. (2006: 107, 108), consider the forms inyo and moapana as distinct subspecies: the first one is confined to the Owens Valley of interior central California and adjacent Nevada and the second one is found in east-central Nevada and adjacent Utah. Freitag (1999: 57) and Erwin and Pearson (2008: 194) listed the lassenica form as a distinct subspecies and recorded it from “California, ” “Nevada, ” “Utah, ” and “Arizona.”
This subspecies, the “Sierra Tiger Beetle”, is restricted to the Sierra Nevada in eastern California [see Kritsky and Horner 1998: Fig. 5].
USA: CA
This subspecies, also known as the “Oblique-lined Tiger Beetle”, ranges over much of eastern North America, from Newfoundland to Nebraska, south to northern Texas and northern Florida (Choate 2003: Map 26) [see Kritsky and Horner 1998: Fig. 5; Pearson et al. 2006: Map 45]. Several state and province records (e.g., AB, CA, ID, MB, MT, ND, NT, NV, OR, SK, UT, WY) in Boyd (1982: 9), Bousquet and Larochelle (1993: 61), and Freitag (1999: 57) apparently refer to other subspecies of Cicindela tranquebarica Herbst. Choate (2003: 81) reported that there are no recent collection records of this species in Florida.
CAN: LB, NB, NF, NS (CBI), ON, PE, QC USA: AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
Cicindela tranquebarica is extremely variable in color and maculation which lead to the recognition of several subspecies. However, it appears that the species varies seasonally. As remarked by Sumlin (1976a: 103) “if one were to sample this population [at Owen’s Lake, Inyo County, California] from early April to the middle of June one would have approximately 5 subspecific phenotypes represented; i.e., inyo Fall (green in color with narrow to thick lunules), kirbyi LeConte (brown or black in color with very wide lunules), cibecuei Duncan (blue in color with wide lunules), borealis E.D. Harris (brown or black in color with very narrow lunules), and parallelonota Casey (light to dark green in color with thickened lunules).” A thorough study of the variation in this species would probably lead to a reduction in the number of subspecies.
This subspecies, the “Wealed Tiger Beetle”, ranges from British Columbia to western Montana, south to northern Utah and northern California [see Kritsky and Horner 1998: Fig. 5]. The record from “Northwest Territories” (Boyd 1982: 10) is probably in error.
CAN: BC USA: CA, ID, MT, NV, OR, UT, WA, WY
Freitag (1999: 55) and Pearson et al. (2006: 107) considered the form borealis as a valid subspecies although Pearson et al. stated that “perhaps the entire population represents a zone of intergradation and not a distinct subspecies.” Erwin and Pearson (2008: 195) also retained the roguensis form as a distinct subspecies listing it from several western states (i.e., ID, OR, MT, NV, WA, WY).
This taxon, also known as the “Santa Ana Tiger Beetle”, is found in southern California, primarily in Orange and western San Bernardino Counties (Pearson et al. 2006: 108).
USA: CA
[formosa group]
This species, also known as the “Laurentian Tiger Beetle”, is restricted to a small area in southeastern Manitoba, northwestern (Lawton 2008: 73) and southwestern Ontario, and adjacent parts of Minnesota (Kaulbars and Freitag 1993a: 307; Pearson et al. 2006: 95); isolated at Manitoulin Island, Ontario (Bouchard et al. 2006: 21).
CAN: MB, ON USA: MN
This subspecies, also known as the “Big Sand Tiger Beetle”, ranges from southern Saskatchewan and southern Alberta south to New Mexico and southern Texas (Gaumer 1977: 188-189); also recorded from “Manitoba” and “Minnesota” (Erwin and Pearson 2008: 135).
CAN: AB, SK USA: CO, KS, MT, ND, NE, NM, OK, SD, TX, WY [MB, MN]
Gaumer (1977: 194-195) reported the presence of intergrade populations between this subspecies and the generosa form in central United States and with the pigmentosignata form in north-central and central Texas.
This subspecies, also known as the “Eastern Sand Tiger Beetle”, ranges from Kings County in Nova Scotia (Neil and Majka 2008: 4) to southern Manitoba, south to northeastern South Dakota (Spomer et al. 2008a: 21), central Louisiana, southern Mississippi, and southern Virginia (Gaumer 1977: 202-203). The records from “Saskatchewan, ” “Montana, ” “Nebraska” (Erwin and Pearson 2008: 136), “Colorado” (Leng 1902: 136), and “Alabama” (Freitag 1999: 25) need confirmation.
CAN: MB, NS, ON, QC USA: AR, CT, DE, IA, IL, IN, KY, LA, MA, MD, ME, MI, MN, MO, MS, ND, NH, NJ, NY, OH, PA, RI, SD, TN, VA, VT, WI, WV [AL, CO, MT, NE, SK]
This subspecies, also known as the “Gibson’s Sand Tiger Beetle”, is known from southwestern Saskatchewan (Wallis 1961: 38; Gaumer 1977: 216) and northwestern Colorado (Kippenhan 1994: 41). The record from “Alberta” (Bousquet and Larochelle 1993: 55) is in error; that from “North Dakota” (Freitag 1999: 26) needs confirmation; that from “Utah” (Erwin and Pearson 2008: 136) is probably based on intergrades found along the Green River (see Pearson et al. 2006: 84).
CAN: SK USA: CO [SD, UT]
In his unpublished thesis, Gaumer (1977: 219) treated the Colorado population of this subspecies as a distinct subspecies of Cicindela formosa. This subspecies intergrades narrowly with the formosa form on all sides of its small range in southern Saskatchewan and along the Green River in northeastern Utah (Pearson et al. 2006: 84).
This subspecies, also known as the “Reddish-green Sand Tiger Beetle”, is found in southwestern Arkansas, northern Louisiana, and eastern Texas (Gaumer 1977: 210).
USA: AR, LA, TX
This subspecies, also known as the “Mescalero Sand Tiger Beetle”, is found in Terry and Yoakum Counties in northwestern Texas and in Chaves, Eddy, and Roosevelt Counties in eastern New Mexico (Rumpp 1986: 143).
USA: NM, TX
Rumpp (1986: 144) reported the presence of intergrade populations between this subspecies and the nominotypical form in Bailey and Lamb Counties, northwestern Texas, and Quay County, eastern New Mexico.
This subspecies, the “Laurent’s Long-lipped Tiger Beetle”, ranges from north-central Montana to western South Dakota, south to central New Mexico, central Arizona, and southern Nevada [see Spanton 1988: Fig. 39].
USA: AZ, CO, ID, MT, NM, NV, SD, UT, WY
The range of this subspecies, also known as the “Boreal Long-lipped Tiger Beetle”, extends from Newfoundland to eastern Alaska, south to southern Alberta, central Minnesota, central Wisconsin, and southern New York [see Spanton 1988: Fig. 39]. Intergrade populations between the three subspecies of Cicindela longilabris are found in southeastern British Columbia, southwestern Alberta, western Montana, Idaho, southeastern Washington, and northeastern Oregon [see Spanton 1988: Fig. 39]; these records are listed under this subspecies.
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, ID, ME, MI, MN, MT, NH, NY, OR, VT, WA, WI
The range of this subspecies, the “Green Long-lipped Tiger Beetle”, extends along the Cascade Range and Sierra Nevada from southwestern British Columbia to east-central California (Spanton 1988: 129, Fig. 39). The records from Colorado (Wickham 1902: 228), “Idaho, ” and “Montana” (Boyd 1982: 6) probably refer to the laurentii form; that from “Nevada” (Bousquet and Larochelle 1993: 58) needs confirmation.
CAN: BC USA: CA, OR, WA [NV]
This species, also known as the “Prairie Long-lipped Tiger Beetle”, ranges from the Fraser River in British Columbia to northwestern Ontario (Lawton 2008: 72), south to west-central Nebraska (Spomer et al. 2008a: 54), northern Colorado, southern Utah, and east-central California [see Spanton 1988: Fig. 40]. The records from “New Mexico” (Freitag 1999: 35) and Minnesota (Horn 1928: 11) need confirmation.
CAN: AB, BC, MB, ON, SK USA: CA, CO, ID, MT, ND, NE, NV, OR, SD, UT, WA, WY [MN, NM]
This subspecies, also known as the “Consenta’s Tiger Beetle”, was once found in Long Island and New Jersey but is now restricted to the Pine Barrens region of New Jersey (Mawdsley 2007: 17). Single specimens, possibly strays, are known from Delaware, “Maryland, ” and “Pennsylvania” (Mawdsley 2007: 17). The taxon is also recorded from “North Carolina” and “Virginia” by Erwin and Pearson (2008: 166).
USA: NJ, NY [DE, MD, NC, PA, VA]
This subspecies, also known as the “Northern Barrens Tiger Beetle”, ranges from New Hampshire (Leonard and Bell 1999: 47) to Minnesota, south to northern Alabama (Löding 1945: 9) northeastern Georgia, and northwestern South Carolina [see Kaulbars and Freitag 1993a: Fig. 40]. The species has been collected also at two sites in the Outaouais region in western Quebec and eastern Ontario (Leonard and Bell 1999: 47). The record from New Jersey (Smith 1910: 197) is questionable (see Boyd 1978: 215). Beaton (2008: 39) indicated that he was unable to find any extant populations of this species in Georgia despite intensive searching at historical sites and other areas of suitable habitat.
CAN: ON, QC USA: AL, CT, DC, DE, GA, IN, KY, MA, MD, MI, MN, NC, NH, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [NJ]
This subspecies, the “Dorothy’s Tiger Beetle”, in known from southeastern Arizona (Bertholf 1983: 21), New Mexico (Acciavatti et al. 1980: 30), and a small area in western Texas [see Pearson et al. 2006: Map 39].
USA: AZ, NM, TX
This subspecies, also known as the “Beautiful Tiger Beetle”, ranges from western South Dakota (Larsen and Willis 2008: 480; Brust 2010: 1) and northeastern Wyoming (Brust 2011: 78) south to northern Texas, New Mexico, and Arizona (Bertholf 1983: 20) [see Pearson et al. 2006: Map 39]; also recorded from Nuevo León (Erwin and Pearson 2008: 172).
USA: AZ, CO, KS, NE, NM, OK, SD, TX, WY
This species, also known as the “Six-spotted Tiger Beetle”, ranges from Nova Scotia to the Black Hills in southwestern South Dakota, south to east-central Texas and northern Florida [see Kaulbars and Freitag 1993a: Fig. 39; Pearson et al. 2006: Map 36]. The record from “North Dakota” (Freitag 1999: 50) needs confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [ND]
[hirticollis group]
This species, also known as the “Coral Pink Sand Dune Tiger Beetle”, is confined to a small area of less than 400 hectares in the Coral Pink Sand Dunes formation in southwestern Utah [see Johnson 1991: Fig. 22] and is considered an endangered species.
USA: UT
This taxon was listed as a subspecies of Cicindela limbata Say by Johnson (1991) and Freitag (1999: 32). Molecular studies have shown that it represents a distinct species (Morgan et al. 2000).
This species, also known as the “St. Anthony Dune Tiger Beetle”, is found in the Snake River Valley of southeastern Idaho [see Shook and Clark 1988: Fig. 1; Pearson et al. 1997: Fig. 22] and in southwestern Montana (Winton et al. 2010: 43).
USA: ID, MT
This subspecies, also known as the “Pacific Coast Tiger Beetle”, ranges along the seacoast from Grays Harbor County in northern Washington to Del Norte County in northern California [see Leffler 1979a: Fig. 30; Pearson et al. 2006: Map 26].
USA: CA, OR, WA
This subspecies, also known as the “Frechin’s Tiger Beetle”, is known only from a small area around Neah Bay in the extreme northwestern Olympic Peninsula, Washington (Pearson et al. 2006: 82).
USA: WA
This species, also known as the “Columbia River Tiger Beetle”, was once found along the Columbia, Salmon, and Snake Rivers in north-central Oregon, south-central Washington, and western Idaho [see Leffler 1979a: Fig. 29; Pearson et al. 1997: Fig. 27]. According to Pearson et al. (2006: 81), it is now known only from a few sites along the Salmon River in Idaho.
USA: ID, OR, WA
This species is listed on the IUCN Red List of Threatened Species (IUCN 2007).
This montane subspecies, also known as the “Dispirited Tiger Beetle”, ranges from southern Alaska south to the Sierra Nevada in east-central California and western Nevada; also found in the Rocky Mountains in southeastern British Columbia, northern Idaho, and western Montana [see Freitag 1965: Fig. 33; Pearson et al. 2006: Map 19].
CAN: BC USA: AK, CA, ID, MT, NV, OR, WA
Intergrade populations are known between the two subspecies of Cicindela depressula in western Washington at intermediate altitudes (Pearson et al. 2006: 70).
This subspecies, also known as the “Eureka Tiger Beetle”, inhabits a narrow area along or near the Pacific Coast from northern Washington to northern California [see Freitag 1965: Fig. 33].
USA: CA, OR, WA
This species, also known as the “Twelve-spotted Tiger Beetle”, is found from Northwest Territories and the eastern front of the Rocky Mountains in Alberta to Newfoundland (Lindroth 1955a: 16), south to Alabama, central Texas, and Colorado (Kippenhan 1990: 309) [see Freitag 1965: Fig.17]. The record from Vancouver, British Columbia (Wallis 1961: 22) is possibly based on a mislabeled specimen.
FRA: PM CAN: AB, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV, WY
This subspecies, also known as the “Sacramento Valley Hairy-necked Tiger Beetle”, is endemic to a small area within the Sacramento Valley of California (Graves et al. 1988: 660). An extensive survey in 2001-2004 within the known distributional range of the subspecies yield no specimens and Knisley and Fenster (2006) concluded that the subspecies has been extirpated in the late 1980s to early 1990s possibly from the construction of the Oroville Dam on the Feather River in the 1960s (see also Fenster and Knisley 2006).
USA: CA
This subspecies, the “Athabascan Tiger Beetle”, is known only from the Lake Athabasca Sand Dunes of northern Alberta and Saskatchewan (Graves et al. 1988: 667). This is the northernmost and most isolated known population of Cicindela hirticollis. The record from “Northwest Territories” (Erwin and Pearson 2008: 142) needs confirmation.
CAN: AB, SK [NT]
This subspecies, also known as the “Colorado River Tiger Beetle”, is restricted to the valley of the Little Colorado River in Navajo County, northeastern Arizona (Graves et al. 1988: 669).
USA: AZ
This subspecies, the “Southwest Hairy-necked Tiger Beetle”, ranges from western Colorado and western New Mexico westwards to southeastern California and the northern parts of the Baja California Peninsula [see Graves et al. 1988: Fig. 6]. The record from “Wyoming” (Erwin and Pearson 2008: 143) needs confirmation. According to Pearson et al. (2006: 73), the subspecies is now probably extirpated from many former sites along the Gila River in central and western Arizona.
USA: AZ, CA, CO, NM, NV, UT [WY] – Mexico
This subspecies, the “Coulee Tiger Beetle”, inhabits mainly the Columbia-Snake river system (Graves et al. 1988: 671) ranging from southern British Columbia to southern Oregon and Idaho. Its western limit is in Cowlitz County, within 100 km of the coast (Pearson et al. 2006: 73). The record from “Montana” (Erwin and Pearson 2008: 143) needs confirmation.
CAN: BC USA: ID, OR, WA [MT]
This subspecies, The “Pacific Hairy-necked Tiger Beetle”, is restricted to the Pacific Coast of northern Baja California and southern California (Graves et al. 1988: 659), as far north as Santa Cruz County (Pearson et al. 2006: 73). According to Erwin and Pearson (2008: 143), this taxon is threatened and has been eliminated over most of its former range.
USA: CA (CHI) – Mexico
This subspecies, also known as the “Hairy-necked Tiger Beetle”, ranges east of the Mississippi River from New Jersey to southern Florida (Peck and Thomas 1998: 16), west to the Mississippi River Valley (Graves et al. 1988: 658, Fig. 6) then north to the southern region of the Great Lakes. According to Allen and Acciavatti (2002: 12), this subspecies is considered rare and even endangered throughout the Ohio River drainage and other regions. The records from Oklahoma (Drew and Van Cleave 1962: 110), “Kansas, ” and “Nebraska” (Erwin and Pearson 2008: 144) are based on intergrades and are recorded here under the shelfordi form; that from “Utah” (Erwin and Pearson 2008: 144) is probably in error or based on a stray.
USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KY, LA, MD, MI, MN, MO, MS, NC, NJ, NY, OH, PA, SC, TN, TX, VA, WI, WV
This subspecies intergrades with the shelfordi form along the Mississippi River and with the rhodensis form in the southern Great Lakes region and at Long Island, New York (Graves et al. 1988: 658, 661, 663).
This subspecies, the “Rhode’s Tiger Beetle”, ranges from Newfoundland to the Lake Superior region in Ontario, south to southeastern Wisconsin (Messer 2010: 33), Michigan, Connecticut, and Rhode Island [see Graves et al. 1988: Fig. 6]. Numerous state and province records (e.g., MB, IA, IL, IN, MN, ND, NE, SD) listed by Erwin and Pearson (2008: 145) are reported here under other subspecies of Cicindela hirticollis.
FRA: PM CAN: LB, NB, NF, NS (CBI), ON, PE, QC USA: CT, MA, ME, MI, NH, NY, RI, VT
This subspecies, the “Shelford’s Tiger Beetle”, inhabits the Great Plains from southern Manitoba to southern Alberta, south to New Mexico and eastern Texas (Graves et al. 1988: 665, Fig. 6). Numerous state records (e.g., AR, AZ, LA, MN, UT) listed by Erwin and Pearson (2008: 145) are reported here under other subspecies of Cicindela hirticollis.
CAN: AB, MB, SK USA: CO, IA, KS, MN, MO, MT, ND, NE, NM, OK, SD, TX, WY
This subspecies intergrades with the nominate form over a large area west of the Mississippi river, from southern Manitoba to Louisiana (Graves et al. 1988: Fig. 6), and with the corpuscula form along the Green River in eastern Utah (Graves et al. 1988: 666).
This subspecies, the “Northwest Hairy-necked Tiger Beetle”, is found along the Pacific Coast from northern Washington to northern California (Graves et al. 1988: 672). According to Pearson et al. (2006: 74), it is now extirpated from most historic sites.
USA: CA, OR, WA
This subspecies, the “Hyperboreal Tiger Beetle”, ranges from southern Northwest Territories to east-central Alberta, central Saskatchewan, west-central Manitoba [see Johnson 1991: Fig. 22] and northeastern Manitoba (Woodcock et al. 2011: 118).
CAN: AB, MB, NT, SK
This subspecies intergrades with the nympha form along a narrow zone in central Alberta (Pearson et al. 2006: 76).
This subspecies, the “Labrador Tiger Beetle”, is known only from southern Labrador (Johnson 1991: Fig. 22; Brzoska and Stamatov 2008: 50-51).
CAN: LB
This subspecies, also known as the “Sandy Tiger Beetle”, is restricted to a small area enclosing southeastern Wyoming, southern South Dakota, Nebraska, and northeastern Colorado [see Johnson 1991: Fig. 22]. The record from “Kansas” (Boyd 1982: 7) needs confirmation.
USA: CO, NE, SD, WY [KS]
This subspecies, the “Nogahabar Tiger Beetle”, is known only from the type locality.
USA: AK
This subspecies, also known as the “Nymphal Tiger Beetle”, ranges from central Alberta to northwestern Minnesota, south to northern South Dakota (Spomer et al. 2008a: 19) and northern Montana [see Johnson 1991: Fig. 22; Pearson et al. 2006: Map 21].
CAN: AB, MB, SK USA: MN, MT, ND, SD
This subspecies, the “Dappled Tiger Beetle”, ranges from eastern Alaska to western Northwest Territories, north to above the Arctic Circle (Brzoska 2008: 65), south to the Queen Charlotte Islands and, along the Rocky Mountains, to central New Mexico [see Freitag 1965: Fig. 18]; also recorded from “Arizona” (Erwin and Pearson 2008: 163) and “Idaho” (Boyd 1982: 7).
CAN: AB, BC (QCI), NT, YT USA: AK, CO, KS, MT, NM, UT, WY [AZ, ID]
This subspecies intergrades with the nominate form in southern British Columbia south, along the crest of the Rocky Mountains, to central Utah, with the navajoensis form in western Colorado and New Mexico, and with the maricopa form in western Utah (Pearson et al. 2006: 68).
This subspecies, the “Maricopa Tiger Beetle”, ranges from southern California to south-central New Mexico, north to southern Nevada [see Freitag 1965: Fig. 18]. According to Pearson et al. (2006: 68), distinct individuals of this form are confined to southeastern and central Arizona. The record from “Texas” (Bousquet and Larochelle 1993: 58) is in error; that from “Utah” is apparently based on intergrades.
USA: AZ, CA, NM, NV
1. According to Freitag (1965: 111), the type series of Cicindela provensis Casey, Cicindela mormonella Casey, and Cicindela nephiana Casey are hybrid specimens of Cicindela oregona guttifera x Cicindela oregona maricopa. Boyd (1982: 7) and Freitag (1999: 37) listed these names as synonyms of Cicindela oregona maricopa Leng and I am following them. 2. Intergrade populations between this subspecies and the navajoensis form are known in southwestern New Mexico (Pearson et al. 2006: 69).
This subspecies, the “Navajo Tiger Beetle”, inhabits a small area in the southern parts of the Rocky Mountains in Utah, southwestern Colorado, northern Arizona, and New Mexico [see Freitag 1965: Fig. 18; Pearson et al. 2006: Map 18].
USA: AZ, CO, NM, UT
This subspecies, also known as the “Western Tiger Beetle”, ranges from Vancouver Island to western Alberta, south to central Utah and southern California along the Mexican border [see Freitag 1965: Fig. 18]; also recorded from Baja California (Murray 1979: 50) and northern Sonora (Cazier 1954: 242). The records from New Mexico (Fall and Cockerell 1907: 155), “Arizona, ” and “New Mexico” (Boyd 1982: 6) probably refer to the guttifera form.
CAN: AB, BC (VCI) USA: CA (CHI), ID, MT, NV, OR, UT, WA, WY – Mexico
This subspecies, the “Nova Scotia Tiger Beetle”, is known from Nova Scotia, including Cape Breton Island, Prince Edward Island, and the Magdalen Islands in Quebec (Leonard and Bell 1999: 101). The record from “New Brunswick” (Boyd 1982: 6) needs confirmation.
CAN: NS (CBI), PE, QC [NB]
This subspecies, also known as the “Bronze Tiger Beetle”, ranges from Newfoundland to southwestern British Columbia, south to southern Oregon, northeastern Arizona, central Texas, and northern Florida (Choate 2003: Map 18) [see Pearson et al. 2006: Map 16]. The record from “Northwest Territories” (Boyd 1982: 6) needs confirmation.
CAN: AB, BC, LB, MB, NB, NF, NS, ON, PE, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY [NT]
Intergrade populations between this subspecies and the tanneri form are known from northwestern Colorado (Kippenhan 1994: 29).
This subspecies, the “Tanner’s Tiger Beetle”, is known only from the Green River Valley in eastern Utah (Pearson et al. 2006: 65).
USA: UT
This species, also known as the “Colorado Dune Tiger Beetle”, is restricted to the Great Sand Dunes National Monument and adjacent areas in Alamosa, Costilla, and Saguache Counties in south-central Colorado (Kippenhan 1990: 310; Pearson et al. 2006: 79). The record from “New Mexico” (Boyd 1982: 7) is in error.
USA: CO
This species, also known as the “Bruneau Dune Tiger Beetle”, has yet been found only at the type locality in southwestern Idaho [see Leffler 2001: Fig. 1].
USA: ID
This subfamily contains a single tribe.
This tribe includes a single genus.
Thirteen species in the Nearctic (three species, one of them Holarctic), Neotropical (two species in mountains of Middle America), and Palaearctic (nine species) Regions arrayed in three subgenera: Elliptosoma Wollaston (one species from Madeira), Loricera s.str. (11 species), and Plesioloricera Sciaky and Facchini (one species from Szechwan, China).
Ball and Erwin (1969: 883) published a key to all species then known, including the three found in North America. Lindroth (1961a: 121-125) covered the Nearctic species.
Eleven species in North America (three species), Middle America (two species), Asia (seven species), and Europe (one species which is also found in Asia and North America).
This species ranges from Kodiak Island and the Kenai Peninsula in southern Alaska south to northern California (Lindroth 1961a: 122; Humboldt County, MCZ).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is found along the coastal region from Washington (Hatch 1953: 64) to at least the San Francisco Bay area (LeConte 1851: 180).
USA: CA, OR, WA
This Holarctic subspecies is found on the Kuril Islands and on the Aleutian Islands and Kenai Peninsula in Alaska (Lindroth 1961a: 125).
USA: AK – Holarctic
Loricera pilicornis (Fabricius). This is a widely distributed Holarctic species, occurring in Europe as far south as northern Spain and continental Italy, over Asia as far south as Kazakhstan and Sichuan, and in North America as far south as the San Bernardino Mountains in southern California and northern West Virginia. The specific name derives from the Latin pili (hairs) and cornus (horn) in reference to the presence of conspicuous stiff setae on the basal antennomeres of the adults. These setae, oriented in three main directions, act as a trap for the capture of collembolan prey.
This Holarctic subspecies ranges over most of Europe and a large part of Asia (see Bousquet 2003a: 98) and from the west coast of Alaska above the arctic circle (Lindroth 1961a: 125) to Newfoundland (Lindroth 1955a: 33), south to northern West Virginia (Tucker and Preston Counties, CMNH), western Nebraska (Kimball County, USNM), northern New Mexico (Sandoval and San Miguel Counties, UASM), southern Arizona (Greenlee County, UASM), and the San Bernardino Mountains in southern California (Fall 1901a: 40, as Loricera californica).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, IA, ID, IN, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NM, NY, OH, OR, PA, SD, UT, VT, WA, WI, WV, WY – Holarctic
This subfamily includes a single tribe.
Northern Hemisphere, with about 50 species arrayed in three genera, all represented in North America.
Three species in the arctic and subarctic areas of the Nearctic (two Holarctic species) and Palaearctic (three species) Regions.
Lindroth (1954a) reviewed the species and provided a key for their identification. The two species found in North America were also covered in his monograph of the Carabidae of Canada and Alaska (Lindroth 1961a: 102-104).
Goulet (1983: 447) regarded Diacheila as the sister-group to {Blethisa + Elaphrus}.
This Holarctic subspecies is known in Asia from Kazakhstan, Mongolia, and eastern Siberia (Goulet 2003: 206) and in the Nearctic Region from a few localities in Alaska, Northwest Territories (Lindroth 1961a: 102), northern Alberta (Bourassa and Wood 2011: 144; Fort McMurray area, Gerald J. Hilchie pers. comm. 2009), and Labrador (Lindroth 1961a: 102). Fossil remnants, dated between 10, 400 and 17, 000 years B.P., have been unearthed in Cape Breton Island, Nova Scotia (Miller 1997: 250) and Iowa (Schwert 1992: 76).
CAN: AB, LB, NT USA: AK – Holarctic
The nominotypical subspecies is found in northern Europe and eastern Siberia.
Diacheila arctica amoena (Faldermann). This subspecies is widely distributed over the arctic and subarctic regions of the Northern Hemisphere, ranging from Labrador to Kazakhstan, but populations seem to be highly localized. The German coleopterist Franz Faldermann found the adults of this taxon “pleasant” hence his scientific name amoena. The nominotypical subspecies ranges from the Nordic regions of Scandinavia to the Komi Republic in northern European Russia.
This Holarctic species is known from scattered localities in Norway, European Russia, Siberia, Mongolia (Goulet 2003: 206), and northeastern China (Li 1992: 30, as Nebria xiaoxinganensis) in the Palaearctic Region, and from Alaska to northwestern Northwest Territories [see Morgan and Morgan 1981: map 3] in the Nearctic Region. Fossil remnants of this species, believed to be 2.0-2.5 million years old, have been found in Greenland (Bennike and Böcher 1990: 336; Böcher 1995: 20); others, dated between about 12, 000 and 21, 500 years B.P., have been unearthed in Iowa and north-central Illinois (Baker et al. 1986: 96; Garry et al. 1990: 394; Schwert 1992: 76; Woodman et al. 1996: 17), northeastern Pennsylvania (Barnosky et al. 1988: 178), southern Ontario and southern Quebec (Morgan and Morgan 1981: 1107).
CAN: NT, YT USA: AK – Holarctic
Eight species in the arctic, subarctic, boreal, and temperate areas of North America (six species) and Eurasia (four species). Two species-group taxa are Holarctic (Blethisa catenaria and Blethisa multipunctata aurata).
Lindroth (1954a) reviewed the species and subsequently treated all the North American species in his monograph of the Carabidae of Canada and Alaska (Lindroth 1961a: 104-108). Goulet and Smetana (1983) provided a key to all known species.
Blethisa eschscholtzii Zoubkoff has been recorded from North American on the basis of a single specimen in USNM labeled “5 mi. E Sanderson, Texas” (Lindroth 1954b: 157). Obviously the specimen is mislabeled. The species is restricted to the region north of the Caspian Sea and the area around Lake Balkhash in southeastern Kazakhstan.
This species is found in northern European Russia and eastern Siberia in the Palaearctic Region (Goulet 2003: 206) and from western Alaska to the western shore of the Hudson Bay in northern Manitoba [see Nielsen et al. 1987: Fig. 17b; Morgan et al. 1986: Fig. 1]. Fossil remnants from the Pliocene or early Pleistocene have been unearthed in northwestern Greenland, Meighen Island, Ellesmere Island, and eastern Siberia (Böcher 1995: 20).
CAN: MB, NT, NU, YT USA: AK – Holarctic
This Nearctic species ranges from Newfoundland (Lindroth 1955a: 31) to westernmost Yukon Territory (Goulet et al. 2009: 33), south to southern British Columbia (Lindroth 1961a: 106), northwestern Montana (Russell 1968: 46; LaBonte and Johnson 1989: 170), northern Illinois (Blatchley 1910: 50), and Massachusetts (Middlesex County, MCZ, USNM) [see Morgan et al. 1986: Fig. 4]. The record from “Pennsylvania” (Bousquet and Larochelle 1993: 86, as Blethisa multipunctata aurata), based on several specimens labeled from Erie County in CMNH, is doubtful (Robert L. Davidson pers. comm. 2008); that from Circle in Alaska (Lindroth 1961a: 106) needs confirmation.
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: IL, MA, ME, MI, MT, ND, NH, NY, VT, WI [AK, PA]
This species has passed under the name Blethisa multipunctata (Linnaeus, 1758) or Blethisa multipunctata aurata Fischer von Waldheim, 1828 in the North American literature.
The range of this species extends from Newfoundland (Lindroth 1955a: 30) to the foothills of the Rocky Mountains in Alberta (Lindroth 1961a: 107-108), south to northern New York and New England (Lindroth 1961a: 107) [see Morgan et al. 1986: Fig. 3]. The record from Michigan (Bousquet and Larochelle 1993: 86), based on a specimen labeled from Lake Superior in CMNH, needs confirmation.
CAN: AB, MB, NB, NF, NS (CBI), NT, ON, QC, SK USA: ME, NH, NY, VT [MI]
This Holarctic subspecies is known from Hokkaidō in Japan and the Russian Far Eastern Region, including Sakhalin and Kamchatka, in the Palaearctic Region and from Alaska, as far south as Anchorage, and northwestern Northwest Territories in the Nearctic Region (Goulet et al. 2009: 33).
CAN: NT USA: AK – Holarctic
The nominotypical subspecies ranges from the Atlantic Coast in Europe to the Lake Baikal region in Siberia (Goulet et al. 2009: 33).
This species is found mainly west of the Cascade Range from southwestern British Columbia, including Vancouver Island, to south-central Oregon (LaBonte and Johnson 1989: 171).
CAN: BC (VCI) USA: OR, WA
The range of this species extends from Newfoundland (Lindroth 1955a: 31-32) to southern Alaska, south to central British Columbia (Lindroth 1961a: 106), northwestern Montana (LaBonte and Johnson 1989: 170), northern Indiana (Blatchley 1910: 50), and New Jersey (Smith 1890: 73) [see Morgan et al. 1986: Fig. 2; Ball and Currie 1997: Fig. 3].
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, IL, IN, MA, ME, MI, MN, MT, NH, NJ, NY, OH, PA, RI, VT, WI
Thirty-nine species in the arctic, subarctic, boreal, and temperate areas of North America (19 species) and Eurasia (24 species). These species are arrayed in five subgenera, all but Sinoelaphrus Shi and Liang (one species in northern China) represented in North America. Four species are Holarctic.
Goulet (1983) revised the species then known and provided a key for their identification.
One species in the subarctic regions of North America, Asia, and Europe.
Goulet (1983) concluded from his phylogenetic analysis using adult and larval characters that this subgenus was the most basal lineage of Elaphrus.
This Holarctic subspecies is widely distributed in the subarctic areas. In the Palaearctic Region, it ranges from the British Isles to the Kamchatka Peninsula, and in the Nearctic Region from Alaska to Labrador [see Goulet 1983: Fig. 157]. Fossil remnants of this species, dated between 10, 100 and 20, 700 years B.P., have been unearthed in southern Quebec (Mott et al. 1981: 146), southern Ontario (Morgan and Morgan 1981: 1107), Illinois (Schwert 1992: 76), and southeastern Iowa (Baker et al. 1986: 96); others, believed to be 2.0-2.5 million years old, have also been found in Greenland and Meighen Island (Bennike and Böcher 1990: 336; Böcher 1995: 22).
CAN: AB, BC, LB, MB, NT, NU, ON, QC, SK, YT USA: AK – Holarctic
This subspecies is known only from a few localities on Kodiak Island, Alaska (Goulet 1983: 244).
USA: AK
Fourteen species in the Nearctic (six species) and Palaearctic (eight species) Regions.
Goulet’s (1983) phylogenetic analysis based on adult and larval characters suggests that Neoelaphrus is the sister-group to {Elaphrus s.str. + Elaphroterus}.
Fossil remnants of Elaphrus sibiricus Motschulsky, probably about 2.0-2.5 million years B.P., have been unearthed from northwestern Yukon Territory (Elias and Matthews 2002: 914) and northwestern Greenland (Böcher 1995: 22). The species is currently found in Siberia, Japan, Mongolia, and Inner Mongolia in China (Goulet 2003: 207).
[clairvillei group]
This species extends throughout the temperate and boreal regions of North America from Newfoundland to Alaska, south to northern California along the west coast, to the White Mountains in eastern Arizona and to central New Mexico along the Rocky Mountains, to west-central Nebraska (Arthur County, Foster F. Purrington pers. comm. 2010), and to New Jersey along the east coast [see Goulet 1983: Fig. 160]. The record from east-central Missouri (Summers 1873: 132) needs confirmation.
CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, ID, MA, ME, MI, MN, MT, NE, NH, NJ, NM, NV, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WY [MO]
This species is known from western Nevada and California, from the Oregon border to the Los Angeles area [see Goulet 1983: Fig. 161].
USA: CA, NV
This species ranges from the west coast of Newfoundland to southern British Columbia, south to southeastern Utah, central Colorado, central Illinois, and New Jersey [see Goulet 1983: Fig. 161]. The species is known from only one locality west of the Rocky Mountains, in southern British Columbia.
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: CO, CT, IL, IN, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NY, RI, SD, UT, VT, WI, WY
[fuliginosus group]
This species ranges from Maine, southern Quebec, and Michigan south to northern Mississippi (Pontotoc County, Drew A. Hildebrandt pers. comm. 2009) and North Carolina (Northampton County, CNC) [see Goulet 1983: Fig. 159]. The record from Georgia (Fattig 1949: 11) needs confirmation; those from eastern Iowa (Wickham 1911b: 5; King 1914: 320) are probably in error.
CAN: QC USA: CT, DE, MA, MD, ME, MI, MS, NC, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WV [GA]
This species ranges from Quebec to southern Manitoba, south to Nebraska and Maryland [see Goulet 1983: Fig. 158]. It is also known from one unexpected locality in central Alberta (Bousquet 1987a: 112), which suggests that the species is more widely distributed at least in the northern part of its range.
CAN: AB, MB, ON, QC USA: CT, IA, IL, IN, MA, MD, ME, MI, MN, ND, NE, NH, NJ, NY, PA, RI, SD, VT, WI
Elaphrus fuliginosus Say. Why the great naturalist Thomas Say gave this eastern species the name fuliginosus (sooty) is not evident. It may refer to the mirrors on the elytra which gives the impression that the animals are dirty. Members of Elaphrus have the ability to produce stridulating chirps by rubbing rows of bristles on the dorsal surface of the abdomen against two areas of parallel ridges on the ventral surface of the elytra. The sound is produced when the beetle is under stress.
This species is known from two areas, Maryland and southeastern Virginia (Surry County, CNC), and southern Indiana, southern Illinois, and southern Tennessee (Marion County, CMNH) [see Goulet 1983: Fig. 159]. The apparent gap is probably due to inadequate samplings.
USA: IL, IN, MD, TN, VA
Eighteen species in North America (ten species) and Eurasia (ten species). Two species (Elaphrus trossulus and Elaphrus tuberculatus) are Holarctic.
This subspecies ranges from Newfoundland to southwestern Alaska, south to central British Columbia, southeastern Alberta, northern Minnesota, north-central Ohio (Purrington and Stanton 1996: 44), northern New York, and Maine [see Goulet and Baum 1982: Fig. 1]. The records from “California, ” “Wyoming, ” “South Dakota, ” “Iowa” (see Bousquet and Larochelle 1993: 87), and Missouri (Anonymous 2007) are likely based on mislabeled specimens or are in error.
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, ME, MI, MN, NH, NY, OH, WI
This subspecies ranges from the Queen Charlotte Islands (Kavanaugh 1992: 58) to southwestern Alberta, south to central Colorado, central Idaho, and southern Oregon (Goulet and Baum 1982: 2272; Fig. 1).
CAN: AB, BC (QCI, VCI) USA: CO, ID, OR, WA
This species is widely distributed from central Alaska to Nova Scotia, south to northern Virginia, central Missouri, northern New Mexico, and southern California near the Mexican border [see Goulet 1983: Fig. 172]. The records from “North Carolina, ” northeastern Florida, “Louisiana, ” and “Texas” [see Goulet 1983: Fig. 172] need confirmation.
CAN: AB, BC (VCI), MB, NB, NS, NT, ON, PE, QC, SK USA: AK, CA, CO, CT, DC, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NM, NV, NY, OH, OR, PA, SD, UT, VA, VT, WA, WI, WV, WY [FL, LA, NC, TX]
This species extends from western Montana to southern Oregon, south to southernmost California, central Arizona, and southern Colorado [see Goulet 1983: Fig. 170].
USA: AZ, CA, CO, ID, MT, NV, OR, UT
This species ranges from northwestern Quebec along the James Bay (Chisasibi, Serge Laplante pers. comm. 2011) to central British Columbia, north to the Great Slave Lake in Northwest Territories, south to southern California, southern Arizona, and central Kansas [see Goulet 1983: Fig. 169]. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 23).
CAN: AB, BC, MB, NT, ON, QC, SK USA: AZ, CA, CO, ID, KS, MN, MT, ND, NE, NV, OR, SD, UT, WA, WY
This species is known from a few localities in southeastern Washington, northern California, southeastern Wyoming, and Colorado [see Goulet 1983: Fig. 168].
USA: CA, CO, WA, WY
This species is known only from the original two specimens collected at the type locality.
USA: CA
This species ranges from southern Quebec to northern Minnesota, south to eastern Texas (Casey, 1924: 17, as Elaphrus texanus), east-central Louisiana (Allen 1965: 61), southwestern Alabama (Löding 1945: 11), and northern Florida (Peck and Thomas 1998: 16) [see Goulet 1983: Fig. 168].
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV
This species is found in the montane regions of northern Mongolia and neighbouring Russia west and south of Lake Baikal, in northeastern Siberia, and in the arctic regions from the Commander Islands in the Bering Sea to the eastern shore of James Bay in Quebec (Morgan and Pilny 1997: 146) [see Goulet 1983: Fig. 171]. Fossil remnants of this species, dated between 10, 100 and 11, 050 years old, have been found in southern Quebec (Mott et al. 1981: 146); others, older than 33, 000 years B.P., has been unearthed in southwestern Ontario (Warner et al. 1988: 37).
CAN: MB, NT, NU, ON, QC, YT USA: AK – Holarctic
According to Shilenkov (in Kryzhanovskij et al. 1995: 61), the type material of Elaphrus trossulus Semenov is conspecific with members of Elaphrus tuberculatus Mäklin. However, Goulet and Smetana (1997: 203) concluded that adults of Elaphrus trossulus and Elaphrus parviceps Van Dyke “cannot be separated.” Lindroth (1961a: 116) regarded Elaphrus parviceps as a junior synonym of Elaphrus riparius (Linnaeus), a species restricted to the Palaearctic Region according to Goulet (1983: 313).
This Holarctic species ranges from northern Scandinavia to eastern Siberia, and from Alaska to the Mackenzie River in the Northwest Territories [see Goulet 1983: Fig. 171]. Fossil remnants of this species, believed to be 2.0-2.5 million years old, have been found in Greenland (Bennike and Böcher 1990: 336; Böcher 1995: 23).
CAN: NT, YT USA: AK – Holarctic
This species is known from a small area in Solano County, California [see Goulet 1983: Fig. 168].
USA: CA
This species, also known under the vernacular name “Delta Green Ground Beetle”, is considered an endangered species by the World Wildlife Fund and listed on the IUCN Red List of Threatened Species.
Five species in northern North America (two species) and Eurasia (four species). One species (Elaphroterus angusticollis) is Holarctic.
This Holarctic subspecies ranges from the Lena River in eastern Siberia to the Bering Sea Coast, and from Alaska to the Mackenzie River in Northwest Territories [see Goulet 1983: Fig. 173]. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 23).
CAN: NT, YT USA: AK – Holarctic
The subspecies Elaphrus angusticollis longicollis Sahlberg occurs in the Palaearctic Region.
This species ranges from central Alaska to the Mackenzie River in Northwest Territories, south to southeastern Alberta, central Idaho, and central California along the west coast [see Goulet 1983: Fig. 173].
CAN: AB, BC, NT, YT USA: AK, CA, ID, MT, OR, WA
This subfamily includes a single tribe.
This tribe includes a single genus.
About 70 species in the Nearctic (11 species), Neotropical (six species in Middle America), Oriental (16 species), Palaearctic (16 species), and Afrotropical (20 species) Regions. The species are arrayed in two subgenera: Omophron s.str. (about 60 species) and Phrator Semenov (eight species in the Mediterranean region and Africa).
About 60 species in the Nearctic (11 species), Neotropical (six species in Middle America), Oriental (16 species), Palaearctic (15 species), and Afrotropical (13 species) Regions.
Benschoter and Cook (1956) revised the North American species and provided a key for their identification. Lindroth’s (1961a: 10) key covered seven species.
This species ranges from Newfoundland (Lindroth 1955a: 150) to the foothills of the Rocky Mountains in Alberta (Lindroth 1961a: 12), south to northeastern Arizona, the state of Coahuila in Mexico (Benschoter and Cook 1956: 427), and the Florida Panhandle (Peck and Thomas 1998: 15). The record from “Vera Cruz” (Benschoter and Cook 1956: 428) needs confirmation.
CAN: AB, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV, WY – Mexico
This species seems to be confined to the southern half of California (Benschoter and Cook 1956: 422) and the Baja California Peninsula (Erwin 2007a: 64). Old specimens simply labeled from Arizona and Texas are known (Benschoter and Cook 1956: 422) but are probably mislabeled.
USA: CA (CHI) – Mexico
This species ranges from southern California to western Colorado (Benschoter and Cook 1956: 416) and northern New Mexico (Taos County, UASM), south to Sonora, Mexico (Erwin 2007a: 65). Old specimens simply labeled from Texas are known (Benschoter and Cook 1956: 416).
USA: AZ, CA, CO, NM, UT [TX] – Mexico
This species ranges from western Wisconsin (Messer 2010: 34) to southern Nebraska (Adams County, Foster F. Purrington pers. comm. 2010), south at least to northeastern Texas (Benschoter and Cook 1956: 425), east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), and southwestern Mississippi (Lago and Zucarro 1984: 118; Wilkinson County, UASM).
USA: AR, IA, KS, LA, MO, MS, NE, OK, TX, WI
This species is found along the Atlantic and Gulf of Mexico coasts, from Sable Island off the coast of Nova Scotia (Lindroth 1969a: 1108) to southern Florida (Peck and Thomas 1998: 15), west to southeastern Texas (Benschoter and Cook 1956: 420).
CAN: NS USA: AL, CT, DC, FL, GA, LA, MA, MD, ME, MS, NC, NJ, NY, PA, SC, TX, VA
This species ranges from northern Nebraska to northwestern Indiana, north to the Minneapolis region in western Minnesota, south to Alabama and southern Texas (Benschoter and Cook 1956: 420, 422). The records from Wisconsin (Rauterberg 1885: 11) and Charity Island in Michigan (Andrews 1916: 72) are probably based on misidentified Omophron americanum.
USA: AL, AR, IA, IL, IN, KS, LA, MN, MO, MS, NE, OK, TN, TX
This species ranges from southern California to western Texas, north to southern Utah (Benschoter and Cook 1956: 416), south to Zacatecas in Mexico (Erwin 2007a: 67). The record from “Montana” (Bousquet and Larochelle 1993: 90) is likely in error.
USA: AZ, CA, NM, TX, UT – Mexico
The range of this species extends from southwestern Saskatchewan (Ronald R. Hooper pers. comm. 1990) to Vancouver Island (Lindroth 1961a: 13), south to northern California and southern Wyoming (Benschoter and Cook 1956: 426).
CAN: AB, BC (VCI), SK USA: CA, ID, MT, NV, OR, UT, WA, WY
This species ranges from the southern part of the Ontario Peninsula to south-central North Dakota (Benschoter and Cook 1956: 418), south to northwestern Texas (Hutchinson County, Robert L. Davidson pers. comm. 2012) and northwestern Tennessee (Lake County, CMNH); seemingly isolated in southeastern Alberta (Lindroth 1961a: 11).
CAN: AB, ON USA: IA, IL, IN, KS, MI, MN, ND, NE, OH, OK, SD, TN, TX, WI
This species is known from southwestern Oregon and northern California (Benschoter and Cook 1956: 425).
USA: CA, OR
This species ranges from Cape Breton Island to southern Alberta, south to southwestern Arizona, northwestern Oklahoma (Cimarron County, CNC), southwestern Arkansas (Hempstead County, MCZ), and Virginia (Benschoter and Cook 1956: 424). One specimen labeled from Alameda County in western California, seen by Benschoter and Cook (1956: 424), is possibly mislabeled. The record from “Texas” (Bousquet and Larochelle 1993: 90) needs confirmation.
CAN: AB, MB, NB, NS (CBI), ON, PE, QC, SK USA: AR, AZ, CO, CT, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SD, VA, VT, WI [CA, TX]
Worldwide, with about 1, 870 species arrayed in eight tribes: Carenini (about 195 species), Clivinini (about 820 species), Dalyatini (one species), Dyschiriini (about 300 species), Pasimachini (35 species), Promecognathini (eight species), Salcediini (about 15 species), and Scaritini (about 495 species).
Thirty-five species arrayed in two genera: Mouhotia Laporte (three Oriental species) and Pasimachus.
Western Hemisphere, with 32 species arrayed in two subgenera: Emydopterus Lacordaire (13 Middle American species) and Pasimachus s.str. (19 species).
Bänninger (1950) reviewed all the species and provided a key for their identification. Purrington and Drake (2005: 254-255) published a key to the North American species. A modern taxonomic revision of the genus is needed as challenging problems remain to be resolved (Ball and Bousquet 2000: 76).
Nineteen species in the temperate, subtropical, and tropical areas of North America (11 species) and Middle America (13 species).
Snow (1907: 141) recorded Pasimachus mexicanus Gray from Pima County in Arizona. Because the presence of this species in United States has not been confirmed subsequently, Pasimachus mexicanus is not retained as a North American entity in this catalogue.
[depressus group]
This species ranges from “Utah” (LeConte 1874a: 273) to southeastern Nebraska (Pawnee County, Peter W. Messer pers. comm. 2008), south at least to eastern Arkansas (Cook and Holt 2006: 2313) and Michoacán in Mexico (Ball and Shpeley 1992a: 46).
USA: AR, AZ, CO, KS, NE, NM, OK, TX, UT – Mexico
This species ranges from southeastern New York (Notman 1928: 211) and New Jersey (Bänninger 1950: 491) to Wisconsin (Messer 2010: 34), south to southeastern Texas (Galveston County, MCZ; Bänninger 1950: 491) and central Florida (Lake County, MCZ).
USA: AL, AR, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI, WV
The range of this species extends from northern Arizona to “Missouri” (Bänninger 1950: 492), north to northern Nebraska (Cherry and Sheridan Counties, Peter W. Messer pers. comm. 2004), south to southern Texas (Johnson 1978: 67) and northeastern Mexico (Bänninger 1950: 492).
USA: AZ, CO, KS, MO, NE, NM, OK, TX – Mexico
Bänninger (1950: 510) retained Pasimachus costifer LeConte as a questionable subspecies of Pasimachus duplicatus.
This species ranges from the southern part of the Prairie Provinces (Lindroth 1961a: 131) south to northern Sonora (Bates 1884: 264), western and northern Texas (Wheeler, Grayson, Presidio, and Brewster Counties, MCZ, CMNH; Bänninger 1950: 490), and southeastern Louisiana (Summers 1874a: 79), east to Indiana (Blatchley 1910: 55; Bänninger 1950: 490) and southwestern Michigan (Dunn 1982a: 206). The record from southwestern Virginia (Horn 1869a: 123) is probably in error.
CAN: AB, MB, SK USA: AR, AZ, CO, IA, ID, IL, IN, KS, LA, MI, MN, MO, MT, ND, NE, NM, OH, OK, SD, TX, WI, WY – Mexico
Pasimachus elongatus LeConte. Because of its large size and robust shape, this species is one of the most conspicuous carabid elements of the grassland regions of North America. It belongs to a genus which is endemic to North and Middle America. Although not firmly established, the sister taxon to Pasimachus could be Mouhotia, a genus of massive and handsome species endemic to southeast Asia.
This species is found from “Iowa” (Bänninger 1950: 492) to southeastern Wyoming (Laramie County, CMNH), south to southeastern Arizona (Casey 1913: 89, as Pasimachus atronitens) and Chihuahua in northern Mexico (Bänninger 1950: 492).
USA: AZ, CO, IA, KS, NE, NM, OK, SD, TX, WY – Mexico
Bänninger (1950: 510) retained Pasimachus atronitens and Pasimachus acuminatus as questionable subspecies of Pasimachus obsoletus. Erwin (2011b: 51) treated Pasimachus atronitens as a valid species.
This species ranges from New Jersey (Smith 1890: 74; Smith 1910: 201) and north-central Virginia (Carrington 2002: 107) to west-central Kansas (Snow 1878: 63; Knaus 1907: 233), south to central Texas (LeConte 1846a: 146; Lee County, MCZ) and the Florida Panhandle (Peck and Thomas 1998: 16); also recorded from Durango in Mexico (García 2004: 289, as Pasimachus punctatus Haldeman). The record from Cochise County, Arizona (Snow 1906b: 161) is probably in error.
USA: AL, AR, FL, IL, IN, KS, KY, LA, MO, MS, NC, NJ, OH, OK, PA, SC, TN, TX, VA, WV – Mexico
This species ranges from southern Arizona (Schaeffer 1905: 142) south at least to Durango in Mexico (Bänninger 1950: 493).
USA: AZ – Mexico
Bänninger (1950: 510) retained Pasimachus ignicinctus Bates as a valid subspecies of Pasimachus viridans.
[marginatus group]
This species ranges from southern Maryland (Peter W. Messer pers. comm. 2010) to the Florida Keys (Nichols 1988b: Fig. 5-4; Peck and Thomas 1998: 16), west to “Texas” (Leng 1915: 565; Bänninger 1950: 488), north along the Mississippi River drainage to southwestern Kentucky (Mammoth Cave National Park, CMNH).
USA: AL, FL, GA, KY, LA, MD, MS, NC, SC, TN, TX
Bänninger (1950: 509) retained Pasimachus crassus Casey as a questionable subspecies of Pasimachus marginatus.
This species is probably restricted to the Coastal Plain ranging from South Carolina (Kirk 1970: 9; Ciegler 2000: 38) to southern Florida including the Keys (Peck and Thomas 1998: 17), west to southeastern Louisiana (Allen 1965: 62).
USA: AL, FL, GA, LA, SC
Bänninger (1950: 510) retained Pasimachus floridanus Casey and Pasimachus subnitens Casey as questionable subspecies of Pasimachus subsulcatus. Nichols (1988a: 217) retained Pasimachus floridanus as a valid species but added that “further study is needed to determine ... whether it is a taxon worthy of distinction from Pasimachus subsulcatus Say.”
[strenuus group]
This species is known from southeastern Georgia (Fattig 1949: 12), the Florida Peninsula and Panhandle (Peck and Thomas 1998: 17), and southwestern Alabama (Löding 1945: 12).
USA: AL, FL, GA
The range of this species extends from Massachusetts (Miliotis 1974: 114) to central Iowa (Purrington and Drake 2005: 256), south to southeastern Mississippi (Casey 1913: 76, as Pasimachus brevitarsis) and southern Florida including the Keys (Nichols 1988b: Fig. 5-4; Peck and Thomas 1998: 17). The records from South Dakota (Kirk and Balsbaugh 1975: 15, as Scarites substriatus), “Kansas” (Bousquet and Larochelle 1993: 96), and southeastern Louisiana (Summers 1874a: 79) need confirmation.
USA: AL, DC, FL, GA, IA, IL, IN, MA, MS, NC, NJ, NY, OH, SC, TN, VA [KS, LA, SD]
Bänninger (1950: 510) retained Pasimachus substriatus Haldeman as a questionable subspecies of Pasimachus sublaevis. Nichols (1988a: 224-225) treated it as a junior synonym of Pasimachus sublaevis.
Worldwide, with about 495 species (Lorenz 2005: 135-141 and the genus Scaraphites Westwood) placed in 42 genera arrayed in four subtribes: Acanthoscelitina (one species on the southwest coast of Africa), Oxylobina (29 Asian species in the genus Oxylobus Chaudoir), Scapterina (22 species in the Eastern Hemisphere except Europe), and Scaritina (about 445 species).
Worldwide, with about 445 species arrayed in 37 genera. The tribe is much more diversified in term of species (about 51% of the world fauna) in the Afrotropical (particularly on Madagascar) than anywhere else. The Western Hemisphere has only four endemic genus-group taxa: Antilliscaris Bänninger (four West Indian species), Baenningeria Reichardt (two species on the Galápagos), Glyptogrus Bates (seven Neotropical species), and Taeniolobus Chaudoir (about 40 Neotropical species). The Nearctic (with seven species) and Australian (with 12 species) Regions are underrepresented.
About 190 species (Lorenz 2005: 137-140) arrayed in four subgenera: Orientolobus Dostal (eight Indo-African species), Parallelomorphus Motschulsky (15 Old World species), Scarites s.str. (about 130 species), and Taeniolobus Chaudoir (about 40 Neotropical species). The genus is more diversified in term of species in the tropics of the Old World (about 60.5% of the world fauna) than anywhere else.
Bänninger (1938) reviewed the species and provided keys for their identification. Three new North American species were subsequently described. Bousquet and Skelley (2010) published a key to all Nearctic species except Scarites lissopterus Chaudoir.
About 130 species in the Nearctic (seven species), Neotropical (about 25 species), Oriental (about 45 species), Palaearctic (about 25 species, most of them in Asia), and Afrotropical (about 45 species) Regions. The bulk of the species (about 77% of the world fauna) are found in the tropics of the Old World.
The taxonomy of the species of the quadriceps group is based on Stephen W. Nichols’ unpublished manuscript entitled “A provisional key to the North American species of the Scarites subterraneus group” as well as his thesis (Nichols 1988a).
[quadriceps group]
This species inhabits the Great Plains from south-central Kansas (Sedgwick County, MCZ) to the Rio Grande in Texas (Hidalgo and Cameron Counties, MCZ, USNM), west to western Texas (El Paso County, USNM), including southeastern and central New Mexico (Chaves County, CMNH; Ellis et al. 2001: 16), east to northeastern Louisiana (Franklin and Tensas Parishes, Igor M. Sokolov pers. comm. 2009). The records from “Wisconsin” and “Iowa” (Bousquet and Larochelle 1993: 96) are likely in error.
USA: KS, LA, NM, OK, TX
Bänninger (1938: 152) regarded this taxon as a subspecies of Scarites subterraneus Fabricius.
This species ranges along the Coastal Plain from New Jersey (Smith 1890: 74, as Scarites subterraneus var. substriatus; CNC) to southern Florida (Nichols 1988b: Fig. 5-7; Peck and Thomas 1998: 17), west to southeastern Texas (Jefferson County, USNM). Several records (i.e., AR, IA, IL, IN, KS, KY, MI, MN, MO, OH, OK, ON, PA, SD, TN, WI) listed in Bousquet and Larochelle (1993: 97) refer to other species, particularly Scarites vicinus Chaudoir.
USA: AL, FL, GA, LA, MD, MS, NC, NJ, SC, TX
This species ranges from southern Ontario (CNC) to eastern North Dakota (Tinerella 2003: 635 as Scarites quadriceps), south to northeastern Texas (Cass County, USNM) and northern Alabama (Madison County, USNM).
CAN: ON USA: AL, AR, IA, IL, IN, KS, KY, LA, MN, MO, MS, ND, NE, OH, OK, TN, TX, WI
[subterraneus group]
This species is known from coastal Florida, including the Keys (Peck and Thomas 1998: 17), the Bahamas, Cuba, and the Yucatán Peninsula in southern Mexico [see Nichols 1986: Fig. 9]. One old specimen simply labeled from Louisiana is known (Nichols 1986: 261).
USA: FL [LA] – Bahamas, Cuba, Mexico
This species is endemic to the Florida Peninsula north of Lake Okeechobee [see Nichols 1986: Fig. 10].
USA: FL
This species is known only from the holotype.
USA: FL
This species is found from southeastern New Hampshire (Rockingham and Strafford Counties, Donald S. Chandler pers. comm. 2008) to eastern North Dakota (Cass County, Donald P. Schwert pers. comm. 1989), including southern Ontario (Lindroth 1961a: 129), south to the Yucatán Peninsula (Nichols 1988a: 52), the Florida Keys (Peck and Thomas 1998: 17), and Cuba (Darlington 1934: 67; Nichols 1988b: Fig. 5-8), west along the southwest to southwestern California (LeConte, 1852a: 198, as Scarites californicus) and Baja California (Horn 1894: 307).
CAN: ON USA: AL, AR, AZ, CA, CT, CO, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Cuba, Mexico
Bänninger (1938: 151) retained Scarites patruelis LeConte, Scarites californicus LeConte, and Scarites texanus Chaudoir as valid subspecies of Scarites subterraneus. All three names are listed in synonymy with Scarites subterraneus by Nichols (1988a: 49).
Worldwide, with about 820 species (Lorenz 2005: 141-150) arrayed provisionally in three subtribes, Forcipatorina (25 Neotropical species), Ardistomina (about 90 species), and Clivinina (about 705 species). The last two subtribes are represented in North America. The Nearctic fauna includes 57 species (about 7% of the world fauna).
Worldwide, with about 705 species. The Northern Hemisphere is represented by about 170 species (roughly 24% of the world fauna) and North America by 52 species (about 7% of the world fauna).
Worldwide, with about 375 species described (Lorenz 2005: 141-145) arrayed in nine subgenera: Antroforceps, Clivina s.str., Cliviniella Kult (four Afrotropical species), Dacca Putzeys (one Oriental species), Eoclivina Kult (eight Indo-African species), Physoclivina Kult (one Afrotropical species), Reichardtula Whitehead, Leucocara, and Semiclivina. Seventeen species, three of them adventive, are found in the boreal (marginal), temperate, and subtropical areas of North America.
Western Hemisphere, with at least 30 species in the Nearctic (two species, one of them adventive) and Neotropical (at least 30 species) Regions. One species, possibly adventive, is known from the suburbs of Sydney, Australia (Baehr 2008: 23-25).
Nichols (1985b: 380) discussed the structural differences between the two species found in North America.
This subgenus as defined by Kult (1947: 31) includes the species of groups 19 (mistakenly reported as group 29 by Kult) and 24 of Putzeys (1867b: 145, 166-178). Recently Dostal (2011) listed this taxon as a distinct genus and described a new subgenus, Uroclivina Dostal, for the species of Semiclivina with a peculiar tubercle behind the posterior edge of the eye.
The range of this species extends from Connecticut (Krinsky and Oliver 2001: 44) to eastern South Dakota (Kirk and Balsbaugh 1975: 16), including southernmost Ontario (Bousquet 1987a: 119), south to southern Texas (Zapeta, Kleberg, and Gonzales Counties, CMNH; Leng 1915: 570) and southern Florida (Peck and Thomas 1998: 17), west along the south to the Colorado River drainage in San Bernardino County, California (Fall 1901a: 41); also recorded from Cuba (Dejean 1825: 415; Jacquelin du Val 1857: 15), Jamaica (Nichols 1988b: Fig. 5-14), and Mexico as far south as Oaxaca (Erwin 2011b: 169). One old specimen labeled “Mass” is known (MCZ).
CAN: ON USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, VA, WI, WV [MA] – Cuba, Jamaica, Mexico
This species is native to South America and is adventive in North America where it is known from southeastern United States (Nichols 1985b: 380). The first inventoried specimen collected on this continent was found in Mobile, Alabama in 1948 (Nichols 1985b: 380).
USA: AL, LA, MS – Adventive
Worldwide, with over 250 species described. The number of species cannot be assessed at this time since many species included by Lorenz (2005: 141-145) in this subgenus belong to Semiclivina, Reichardtula, and Leucocara. The Nearctic Region has only ten described species and two of them are adventive.
Bousquet (1997c: 347-348) published a key to all North American species and two unnamed ones. One species (Clivina choatei) was described subsequently.
This species is known from Levy and Gilchrist Counties in northern Florida.
USA: FL
This European species is adventive in North America where is it known from southern Quebec (Larochelle 1975: 78) to southwestern Ohio (Dury 1879: 162) and Connecticut (Krinsky and Oliver 2001: 44), from southern Manitoba (Pollock 1991b: 298), from British Columbia (Lindroth 1961a: 162), and from Washington (Hatch 1953: 66). The first inventoried specimen collected on this continent was found prior to 1838 (Randall 1838b: 34, as Clivina elongata) in Massachusetts. The record from northeastern Kansas (Popenoe 1877: 22, as Clivina elongata) is probably in error.
CAN: BC, MB, ON, QC USA: CT, MA, ME, NH, OH, WA – Adventive
This Palaearctic subspecies is adventive in North America where it is found in the east from Newfoundland (Lindroth 1955a: 45) to northern Minnesota (Petrice et al. 2002: 9), south to northern Pennsylvania (Bradford County, CMNH) and in the west from southwestern British Columbia (Bousquet 1987a: 119) to south-central Saskatchewan (Ronald R. Hooper pers. comm. 2002), south to northwestern Wyoming (Teton County, Foster F. Purrington pers. comm. 2010) and southern Oregon (Nelson and Reynolds 1987: 12). The first inventoried specimen collected in the east was found in 1915 in the Montreal region (Lindroth 1961a: 161) and in the west in 1937 in western Washington (Hatch 1949b: 118). The records from Georgia (Fattig 1949: 14) and Alabama (Löding 1945: 12) are probably in error; those from “Ohio” (Hamilton 1889b: 93), “Illinois, ” “Indiana, ” and “Idaho” (Bousquet and Larochelle 1993: 102) need confirmation.
FRA: PM CAN: AB, BC, NB, NF, NS (CBI), ON, PE, QC, SK USA: CT, MA, ME, MI, MN, NH, NY, OR, PA, VT, WA, WI, WY [ID, IL, IN, OH] – Adventive
The subspecies Clivina fossor sachalinica Nakane is found in the Far East and Japan.
Clivina fossor (Linnaeus). This Clivina is one of the 62 carabid species accidentally introduced and established in North America and one of 18 that have been introduced independently on the Atlantic and Pacific Coasts. Many of the adventive species reached our borders in ballast used by Europeans to improve the stability of their trade ships on their way to America. Ballast consisted of stones, broken slates, mortar, bricks, rubbish, soil, and sand that were usually dumped along the shore at ports of entry and certainly constituted a good environment for the survival of carabids during ocean voyages.
The range of this species, also known under the vernacular name “slender seed-corn Beetle”, extends from the Saint Lawrence Plain in southern Quebec (Lindroth 1961a: 164) to northern Utah (Knowlton and Wood 1947: 94; Davis and Utah Counties, USNM), south to southern Texas (Hlavac 1967: 31; Johnson 1978: 67) and northern Georgia (Fattig 1949: 14; Hlavac 1967: 30). At least one specimen simply labeled from Florida is known (Hlavac 1967: 30). The record from Idaho (Anonymous 1960: 642) needs confirmation.
CAN: ON, QC USA: AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV [FL, ID]
This species is known from the holotype collected in east-central North Carolina and six old specimens labeled “D C, ” “Md, ” and “Ill.” (CMNH).
USA: DC, IL, MD, NC
This species occurs from southern British Columbia (Lindroth 1961a: 163) and northern Idaho (Hatch 1953: 66) south to central Oregon (Fall 1922b: 164; Benton County, MCZ).
CAN: BC USA: ID, OR, WA
Hlavac (1967: 27) believed this species was morphologically distinct enough to propose a new subgenus, Betaclivina, for it. His thesis being unpublished, the name is unavailable.
This species ranges from southern Maine (Nelson 1995: 71) to “Illinois” (Hlavac 1967: 23), including southeastern Michigan (Saint Clair and Wayne Counties, CMNH) and west-central Indiana (Montgomery County, R. Michael Brattain collection), south to north-central Texas (Knaus 1905b: 348), southeastern Louisiana (Tangipahoa Parish, USNM) and the Florida Panhandle (Peck and Thomas 1998: 17).
USA: AL, AR, DC, FL, GA, IL, IN, LA, MD, ME, MI, MS, NC, NJ, NY, OH, PA, SC, TN, TX, VA
This species ranges from South Carolina (LeConte 1857b: 82; Ciegler 2000: 42) and northern Georgia (Fattig 1949: 14; Floyd County, MCZ) west to southwestern Texas (Dajoz 2004: 117; El Paso County, MCZ), north along the Mississippi River drainage to west-central Indiana (Tippecanoe County, CMNH) and east-central Kansas (Dickinson and Douglas Counties, MCZ); also recorded from Jalisco, Nayarit, Tamaulipas (Erwin 2011b: 183) and Veracruz in Mexico (Bates 1881: 32). The record from the District of Columbia (Ulke 1902: 6) needs confirmation.
USA: AL, AR, GA, IN, KS, LA, MS, OK, SC, TX [DC] – Mexico
Hlavac (1967: 19) believed this species was morphologically distinct enough to propose a new subgenus, Alphaclivina, for it. His thesis being unpublished, the name is unavailable.
This species ranges from southern Missouri (Hlavac 1967: 32; Ripley County, CMNH) to central Virginia (Nelson County, USNM) and the District of Columbia (Ulke 1902: 37), including southern Ohio (Washington County, UASM; Wright and Whitehouse 1941: 70), south to the Florida Panhandle (Jackson County, CNC), southwestern Mississippi (Copiah County, MCZ), and southern Texas (Zapata, San Patricio and Dimmit Counties, CMNH, UASM).
USA: AL, AR, DC, IL, IN, FL, GA, LA, MO, MS, OH, OK, SC, TN, TX, VA, WV
This species is found in California from Siskiyou County (CAS) to San Diego County (Moore 1937: 5).
USA: CA
Four species in the temperate regions of eastern North America (three species) and caves in the Sierra de Guatemala mountains in northeastern Mexico (one species, Clivina bolivari Barr).
Ball (2001) revised the species and provided a key for their identification. One species (Clivina alabama) was described subsequently.
In a cladistic analysis of the Western Hemisphere subgenera proposed by Ball (2001: Fig. 9), this taxon turn out as the sister-group to {Semiclivina + Leucocara (under the name Reichardtula)}.
This species is known only from two specimens collected in Blount County, north-central Alabama.
USA: AL
This species is confined to eastern United States ranging from Connecticut (Krinsky and Oliver 2001: 46) to southeastern Kansas (Knaus 1885: 57), including eastern Iowa (Ball 2001: 147), south to “Louisiana” (LeConte 1857b: 81) and central Florida (Peck and Thomas 1998: 17).
USA: AL, CT, DC, FL, GA, IA, IL, IN, KS, LA, MD, MI, MS, NJ, NY, OH, PA, SC, VA
This species is known only from eastern Oklahoma (Ball 2001: 147).
USA: OK
This subgenus includes 72 species in the Nearctic (5 species), Palaearctic (6 species), Oriental (18 species) and Afrotropical (43 species) Regions (see Bousquet 2009: Table 2).
There is no key for the identification of the North American species of this subgenus. Nichols (1988a: 147-153) covered two species (Clivina acuducta and Clivina americana) in his thesis. A revision of the group is needed.
Until recently, the species of this subgenus were included in the subgenus Reichardtula Whitehead, a replacement name for Eupalamus Schmidt-Göbel.
This species ranges from Massachusetts (Norfolk County, CMNH) to southwestern Wisconsin (Grant County, CMNH), south to eastern Texas (Riley 2011; San Augustine County, CMNH) and southern Florida (Nichols 1988a: 150).
USA: AL, AR, FL, GA, IL, IN, KY, LA, MA, MD, MO, MS, NC, NJ, NY, OH, PA, SC, TN, TX, VA, WI, WV
Clivina acuducta has been listed in synonymy with Clivina americana in most catalogues (Leng 1920: 48; Erwin et al. 1977: 4.12; Bousquet and Larochelle 1993: 103) but Nichols (1988a: 148) treated it as a valid species.
This species ranges from Nova Scotia (Lindroth 1954c: 301) to southeastern North Dakota (Tinerella 2003: 635), south to western (Dajoz 2007: 23) and southern (Gonzales County, MCZ) Texas, southern Louisiana (Hine 1906: 76, as Clivina analis; Calcasieu and Orleans Parishes, MCZ), and southern Florida including the Keys (Peck and Thomas 1998: 17); also recorded from the Bahamas (Nichols 1988a: 153).
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Bahamas
This species is known only from the type locality.
USA: CA
This species is known from Harrison County in southeastern Mississippi (Drew A. Hildebrandt pers. comm. 2010), Saint Tammany Parish in southeastern Louisiana, Trinity County in eastern Texas (Bousquet 2009: 44), and LeFlore County in eastern Oklahoma (Matthew Gimmel collection). The records from southeastern New York (Leng and Beutenmüller 1893: 135), Lancaster County in Pennsylvania (Rathvon 1869: 524), northwestern Georgia (Fattig 1949: 15), and southwestern Florida (Leng 1915: 571) are in error (see Bousquet 2006c: 3).
USA: LA, MS, OK, TX
This species ranges from southeastern New York (Notman 1928: 213) to eastern South Dakota (Kirk and Balsbaugh 1975: 17), south to eastern Oklahoma (Latimer County, UASM), “Louisiana” (LeConte 1879a: 34), and southern Florida (Peck and Thomas 1998: 17). The record from the lower peninsula of Michigan (Hubbard and Schwarz 1878: 644) needs confirmation.
USA: AL, AR, DC, DE, FL, GA, IA, IL, IN, KS, LA, NC, NJ, NY, OK, SD [MI]
At least 33 species (see Bousquet 2009: table 1) in temperate, subtropical, and tropical areas of the Nearctic (nine species) and Neotropical (29 species) Regions. Kult’s (1947: 31) statement that Paraclivina is represented by two species in Australia is apparently erroneous (see Baehr 2008: 23).
There is no revision of the North American species of this genus. The last key published, that of LeConte (1879a), included seven of the eight species recorded at the time (Paraclivina sulcipennis was omitted). Since then, one tropical species (Paraclivina fasciata) has been recovered in Florida but it is unclear if it is established in the area. Putzeys (1846 and 1867b) included descriptions of all the North American species currently recognized. In his 1867 work, these species were listed in groups 21 (most species) and 22 (Paraclivina fasciata and Paraclivina ferrea) of the genus Clivina. A revision of the genus is needed.
This taxon is very likely monophyletic, characterized by the synapomorphic condition of the lateral bead of the pronotum uninterrupted and removed from the base. The genus Clivina is markedly speciose and inadequately understood both taxonomically and phylogenetically. In these circumstances, I believe it is more convenient to isolate this taxon as a distinct genus even if eventually it may prove to be nested within Clivina. In a cladistic analysis of the five Western Hemisphere subgenera of Clivina (sensu auctorum) conducted by Ball (2001: Fig. 9), this taxon turn out as the sister-group to the remaining subgenera.
The range of this species extends from Massachusetts (Purrington 1997: 96) to eastern South Dakota (Kirk and Balsbaugh 1975: 17; French et al. 2004: 557), south to southeastern Texas (Snow 1906a: 141; Cameron County, MCZ) and southern Florida including the Keys (Peck and Thomas 1998: 17), west along the southwest to southern Arizona (Dajoz 2004: 116; Cochise and Pima Counties, UASM); also recorded from several islands of the West Indies, Mexico, Honduras, and Nicaragua (Erwin 2011b: 162). The record from “New Hampshire” (Bousquet and Larochelle 1993: 103) needs confirmation.
CAN: ON USA: AL, AR, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV [NH] – Cayman Islands, Cuba, Dominican Republic, Haiti, Jamaica, Mexico
This species is known from Long Island, New York (Cooper 1935: 144) to east-central South Carolina (Ciegler 2000: 41) and “Georgia” (LeConte 1844: 50) and from “Louisiana” (LeConte 1879a: 34) and Cuba (Chevrolat 1863: 193, as Clivina bisignata). The records from “Arkansas” (Bousquet and Larochelle 1993: 103) and “Texas” (Leng and Beutenmüller 1893: 96) need confirmation.
USA: GA, LA, NC, NJ, NY, SC, VA [AR, TX] – Cuba
This species has been reported from several islands of the West Indies and several countries from southern Mexico to South America (Erwin 2011b: 171). It is also known from one specimen, the holotype of Clivina dissimilis Blatchley, collected in Pinellas County, central Florida. The species is apparently adventive in the Philippines and the Marianas (Darlington 1970: 12).
USA: FL – Bahamas, Brazil, Cayman Islands, Colombia, Costa Rica, Cuba, Dominica, Dominican Republic, Grenada, Guadeloupe, Haiti, Honduras, Jamaica, Mexico, Panama, Puerto Rico, Saint Croix, Saint Thomas, Venezuela, Virgin Islands
This species occurs from southeastern New York (LeConte 1857b: 82) to southeastern South Dakota (Kirk and Balsbaugh 1975: 17), south to southern Texas (Wickham 1897: 103; Johnson 1978: 67) and northern Florida (Leon County, USNM), west along southern United States to southern California (Imperial, Riverside, and Kern Counties, CAS, USNM) and the Baja California Peninsula (Horn 1894: 307).
USA: AL, AR, AZ, CA, CO, DC, FL, GA, IA, IL, IN, KS, LA, MD, MO, MS, NC, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, VA, WI – Mexico
This species has been recorded by mistake as Clivina ferruginea LeConte by Horn (1872c: 384).
This species is known from northeastern Mississippi (Tishomingo County, Drew A. Hildebrandt pers. comm. 2009), southeastern Louisiana (Putzeys 1867b: 149), and some islands of the West Indies (Nichols 1988a: 143; Peck 2009b: 5).
USA: LA, MS – Dominica, Dominican Republic, Guadeloupe, Puerto Rico, Saint Lucia
This species ranges from southwestern Ohio (Dury 1882: 218) to southeastern South Dakota (Kirk and Balsbaugh 1975: 17), including southern Wisconsin (Messer 2010: 35), south to east-central Texas (Riley 2011), southeastern Louisiana (Jefferson Parish, MCZ; Allen 1965: 62; LeConte 1879a: 34), southern Mississippi (Hancock County, Drew A. Hildebrandt pers. comm. 2008), central Alabama (Shelby County, CMNH), and southwestern South Carolina (Ciegler 2000: 42).
USA: AR, AL, GA, IA, IL, IN, KS, LA, MO, MS, NE, OH, OK, SC, SD, TN, TX, WI
This species is known for sure only from “Texas” (Putzeys 1846: 104; MCZ). The record from “Kansas” (Bousquet and Larochelle 1993: 104) needs confirmation.
USA: TX [KS]
This species may be restricted to the Coastal Plain and Piedmont Plateau. It is known from Staten Island, New York (Leng 1915: 569) and New Jersey (Hamilton 1889a: 30; Leng and Beutenmüller 1893: 96) south to southern Florida (Peck and Thomas 1998: 17), west to south-central Texas (Bexar County, CMNH, USNM). The record from eastern Iowa (Wickham 1911b: 5) is probably in error. One specimen simply labeled from Tennessee (CMNH) is also known.
USA: AL, DE, FL, GA, LA, MS, NJ, NY, SC, TX [TN]
This species is known only from the Atlantic shore of Virginia (Hoffman et al. 2006: 18), southern North Carolina (Brunswick County, Ken Karns pers. comm. 2009), southeastern South Carolina (Charleston County, USNM), Florida as far south as Collier County (USNM), and southwestern Alabama (Van Dyke 1925: 125; Baldwin County, MCZ).
USA: AL, FL, NC, SC, VA
Western Hemisphere, with 75 species in temperate, subtropical, and tropical areas of the Nearctic (24 species) and Neotropical (about 60 species) Regions arrayed in three subgenera: Genioschizus (10 species), Listropus Putzeys (eight Neotropical species), and Schizogenius s.str. (57 species). One species has been described by Baehr (1983) from the Fiji Islands in the Central Pacific Ocean based upon a single specimen. In my opinion, the specimen could be mislabeled and confirmation is needed.
Whitehead (1972) revised all the North American and some of the Neotropical species.
Ten species in North America (one species) and Middle and South America (ten species).
This subspecies is found in southeastern California and Arizona south to southern Sinaloa and northern Nayarit [see Whitehead 1972: Fig. 74].
USA: AZ, CA – Mexico
The subspecies Schizogenius crenulatus chiapatecus Whitehead is found in Mexico and Honduras.
Fifty-seven species in the Nearctic (23 species) and Neotropical (45 species) Regions.
[brevisetosus group]
This species is found along the Colorado River in eastern New Mexico to the Rio Grande drainage system in central Texas and Coahuila [see Whitehead 1972: Fig. 146]; it is also recorded from southwestern Oklahoma (Kondratieff et al. 2005: 173).
USA: NM, OK, TX – Mexico
[depressus group]
This species ranges from the Okanagan Valley in southern British Columbia to northwestern South Dakota, south to the state of Mexico and southern California [see Whitehead 1972: Fig. 241].
CAN: BC USA: AZ, CA (CHI), CO, ID, MT, NM, OR, SD, TX, UT, WA, WY – Mexico
The known range of this species extends from southern Illinois to southern California, south to Baja California Sur and Colima in Mexico [see Whitehead 1972: Fig. 240].
USA: AR, AZ, CA, CO, IL, MO, NE, NM, OK, TX, UT – Mexico
This species ranges west of the Rocky Mountains from Vancouver Island to western Idaho, south to southern California [see Whitehead 1972: Fig. 237].
CAN: BC (VCI) USA: CA, ID, NV, OR, WA
This species is known only from a few localities in northern and central California [see Whitehead 1972: Fig. 240] and from San Bernardino County in the southeast (Dajoz 2007: 20).
USA: CA
This widely distributed species ranges from northern California south through Arizona and western New Mexico to Colombia, including Baja California [see Whitehead 1972: Fig. 239].
USA: AZ, CA, NM – Colombia, Costa Rica, El Salvador, Guatemala, Honduras, Mexico, Nicaragua, Panama
This species ranges from southern Missouri to southeastern Colorado south to Nuevo León and Tamaulipas [see Whitehead 1972: Fig. 238].
USA: AR, CO, MO, OK, TX – Mexico
This species is widely distributed east of the Mississippi River from New Brunswick (Lindroth 1961a: 167) to eastern Illinois, south to east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), southern Mississippi (Clairborne, Covington, Pearl River, Stone, and Wilkinson Counties, Drew A. Hildebrandt pers. comm. 2008), and western North Carolina (Whitehead 1972: 267, Fig. 236). Old specimens simply labeled from “Georgia, ” “South Carolina, ” and “Wisconsin” are known (Whitehead 1972: 267). The record from “Alabama” (Bousquet and Larochelle 1993: 106) needs confirmation.
CAN: NB, ON, QC USA: DC, IL, IN, KY, LA, MA, MD, ME, MS, NC, NH, NJ, NY, OH, PA, TN, VA, VT, WV [AL, GA, SC, WI]
[ferrugineus group]
This species ranges from southern Arizona south to Costa Rica in Pacific drainage areas [see Whitehead 1972: Fig. 101].
USA: AZ – Belize, Costa Rica, Guatemala, Honduras, Mexico
This species ranges from southern Quebec (Larochelle 1975: 106) to South Dakota (Kirk and Balsbaugh 1975: 16), north to southwestern Saskatchewan (CNC), south to central Texas and northern Florida, west along southern United States to southeastern Arizona [see Whitehead 1972: Fig. 100]. Also recorded from the Bahamas (Erwin 2011b: 215).
CAN: ON, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, ME, MD, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, SC, SD, TX, VA, VT, WI – Bahamas
[lindrothi group]
This species is currently known only from a few specimens collected in Florida, Guatemala, and Costa Rica (Whitehead 1972: 201). Also recorded from Honduras and Panama (Erwin 2011b: 218).
USA: FL – Costa Rica, Guatemala, Honduras, Panama
Whitehead (1972: 201) stated that he was uncertain if the two Central American specimens he saw were conclusively conspecific with those from Florida.
[lineolatus group]
This species is distributed from New Brunswick (Webster and Bousquet 2008: 16) to northeastern Montana, south to the Rio Grande Valley in south-central Texas, Tamaulipas in Mexico, and central Alabama [see Whitehead 1972: Fig. 206]. One old specimen simply labeled from “South Carolina” is known (Whitehead 1972: 251). Except for four apparently mislabeled “Florida” specimens, the species is unknown south of New Jersey along the Atlantic Coast.
CAN: NB, ON, QC USA: AL, AR, CT, DC, IA, IL, IN, KS, KY, MA, MD, ME, MN, MO, MS, MT, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SD, TN, TX, VA, VT, WI, WV, WY [SC] – Mexico
[longipennis group]
This species is known only from a few localities in southern Arizona [see Whitehead 1972: Fig. 209].
USA: AZ
The range of this species extends from southern Arizona to Tamaulipas, south to Costa Rica [see Whitehead 1972: Fig. 208].
USA: AZ – Belize, Costa Rica, Guatemala, Honduras, Mexico
This species is known from eastern Arizona and southwestern New Mexico [see Whitehead 1972: Fig. 207].
USA: AZ, NM
[pluripunctatus group]
This species ranges from Arizona and western New Mexico, south to Nayarit [see Whitehead 1972: Fig. 147].
USA: AZ, NM – Mexico
This subspecies is restricted to central and western California, from Shasta County in the north to San Diego County in the south [see Whitehead 1972: Fig. 147].
USA: CA
The subspecies Schizogenius seticollis vandykei Whitehead is known only from a few localities in Baja California Sur (Whitehead 1972: 213).
[sallei group]
This species ranges from Kansas south to the Rio Grande Valley in southern Texas [see Whitehead 1972: Fig. 185]. One specimen simply labeled from “Ohio” (Whitehead 1972: 229) could be mislabeled. The record from south-central Colorado (Wickham 1902: 232) is probably in error.
USA: KS, OK, TX [OH]
[tristriatus group]
This species ranges from Maine, southern Quebec, and Michigan south to Tennessee and North Carolina [see Whitehead 1972: Fig. 190]; also seen from eastern Oklahoma (Le Flore County, FFPC). The records from eastern Iowa (Wickham 1911b: 6) and Missouri (Summers 1873: 133) need confirmation; that from southwestern Colorado (Wickham 1902: 232) must be in error.
CAN: QC USA: CT, DC, IL, IN, KY, MA, MD, ME, MI, NC, NH, NJ, NY, OH, OK, PA, RI, TN, VA, VT, WV [IA, MO, TX]
Whitehead (1972: 237) noted that four specimens from Texas, without definite locality data, may represent an isolated form of this species.
Schizogenius amphibius (Haldeman). This small clivinine belongs to a genus endemic to the Western Hemisphere and is closely related to Halocoryza which is represented, besides North America, along the east and west coast of Africa and on many islands in the eastern parts of the Indian Ocean. The adults usually live along river banks near the water and this ecological preference was hinted at by Samuel Haldeman through the species’ epithet.
This species is known only from a few specimens collected in or near the Ozark Mountains in southern Missouri, western Arkansas, and eastern Oklahoma [see Whitehead 1972: Fig. 189].
USA: AR, MO, OK
This species is known for sure only from a few specimens collected in New York, West Virginia, and Kentucky [see Whitehead 1972: Fig. 189]; it was also reported from southwestern Ohio (Dury 1910: 66) and northeastern Georgia (Fattig 1949: 15).
USA: KY, NY, WV [GA, OH]
This species is known from a few localities throughout much of Texas [see Whitehead 1972: Fig. 189].
USA: TX
This species occurs from southern Texas south to southeastern Oaxaca and northern Chiapas [see Whitehead 1972: Fig. 188].
USA: TX – Mexico
Four species are placed in this genus: one (Halocoryza acapulcana Whitehead) is found on the Pacific Coast of Mexico and on the Galápagos Islands, one (Halocoryza arenaria) in southeastern North America, the West Indies, Brazil, and the west coast of Africa, another one (Halocoryza maindroni Alluaud) on the east coast of Africa and several islands along the western part of the Indian Ocean, including Madagascar, and the southern part of the Red Sea, and the last one (Halocoryza whiteheadiana Erwin) on Baja California Sur.
Whitehead (1967) and Erwin (2011a) reviewed the species and provided keys for their identification.
This species is known from southern Florida, the Yucatán Peninsula, Jamaica (Nichols 1988b: Fig. 5-12), Puerto Rico, the Dominican Republic (Whitehead 1969: 36), the Bahamas, Cuba, Cayman Islands (Peck 2005: 29) and several islands of the Lesser Antilles (Peck 2009a: 12). The species has been recorded also from the Gulf of Biafra in Africa (Bruneau de Miré 1979) and the state of Pernambuco, Brazil (Nichols 1988b: 89).
USA: FL – Bahamas, Barbados, Brazil, Cayman Islands, Cuba, Dominica, Dominican Republic, Jamaica, Martinique, Mexico, Puerto Rico, Saint Lucia, Saint Vincent, Virgin Islands
Thirteen species in the subtropical and tropical regions of the Western Hemisphere, including the West Indies, with one species reaching southeastern North America.
Nichols (1988a: 172-182) covered the four species found in the West Indies in his thesis and provided a key for their identification.
This species has been reported from southeastern Louisiana (Colby 2002: 37), southern Florida (Darlington 1935a: 161), the Bahamas (Peck and Thomas 1998: 18), Cuba (Darlington 1934: 70), Cayman Islands and Puerto Rico (Peck 2005: 29), Dominican Republic (Erwin 2011b: 204), Jamaica, and eastern Mexico in Veracruz and Campeche (Nichols 1988a: 181).
USA: FL, LA – Bahamas, Cayman Islands, Cuba, Dominican Republic, Jamaica, Mexico, Puerto Rico
Western Hemisphere, with about 90 species, arrayed in three genera. All but five species are found in the Neotropical Region. One of the North American species has been collected in Japan.
Bousquet (2006c) revised the North American species and provided a key for their identification.
This genus contains 44 species (Valdés 2009: 70) restricted to the temperate, subtropical, and tropical regions of the Nearctic (two species) and Neotropical (42 species) Regions, including the West Indies.
This species ranges along the Coastal Plain and Piedmont Plateau from New Jersey (Smith 1890: 76; Smith 1910: 202) and southeastern Pennsylvania (Rathvon 1969: 524) to southern Florida, west to central Louisiana [see Bousquet 2006c: Fig. 35].
USA: AL, DC, FL, GA, LA, MS, NC, NJ, NY, PA, SC, TN, VA
This species is found from South Carolina (Ciegler 2000: 43) to southern Florida, west to the Rio Grande in south-central Texas, north along the Mississippi River drainage to southern Illinois [see Bousquet 2006c: Fig. 34].
USA: AL, AR, FL, GA, IL, LA, MS, OK, SC, TN, TX, VA
Twenty species (Valdés 2012) restricted to the temperate, subtropical, and tropical regions of the Western Hemisphere, including the West Indies.
This species ranges from southeastern Iowa (Wickham 1911b: 6; King 1914: 321) to western Kentucky, south to the Rio Grande in southeastern Texas, east to southern Florida, north along the coast to southern Virginia [see Bousquet 2006c: Fig. 33]. The record from southeastern Pennsylvania (Rathvon 1869: 524) is probably in error.
USA: AL, AR, FL, GA, IA, IL, IN, KS, KY, LA, MO, MS, NC, OK, SC, TX, VA
The range of this species extends from Long Island, New York to southwestern Wisconsin (Messer 2010: 35), south to the Rio Grande in south-central Texas and southern Florida [see Bousquet 2006c: Fig. 32]; also recorded from the Bahamas (Turnbow and Thomas 2008: 15).
USA: AL, AR, CT, DC, DE, FL, GA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI, WV – Bahamas
Twenty-six species (Lorenz 2005: 146-147) in the temperate, subtropical, and tropical regions of the Western Hemisphere, including the West Indies. One species only is found in North America.
This species ranges over eastern United States from Washington D.C. to northeastern Kansas, including southeastern Iowa (Wickham 1911b: 6; King 1914: 323), south to the big bend along the Rio Grande in Texas and northeastern Mexico (Bousquet 2006c: 9) and southern Florida; also known from southeastern Arizona [see Bousquet 2006c: Fig. 31]. Two specimens labeled from Klamath County, Oregon (AMNH) and Gallatin County, Montana (CAS) are known (Bousquet 2006c: 9). The species has been recorded also from Japan (Habu 1963: 19).
USA: AL, AR, AZ, DC, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NC, NM, OH, OK, PA, SC, TN, TX, VA [MT, OR] – Mexico
Worldwide, with about 300 species arrayed in nine genera: Akephorus (two species), Clivinopsis Bedel (three species in northern Africa, Kazakhstan, and Turkmenistan), Cribrodyschirius Bruneau de Miré (seven species in Africa, Madagascar, and Asia), Dyschirius (about 255 species), Neodyschirius Kult (one Afrotropical species), Reicheiodes Ganglbauer (16 Palaearctic species, including the Himalayas), Setodyschirius Fedorenko (13 species from Australia), Torretassoa Schatzmayr (one species in Egypt and Yemen), and Caledyschirius Bulirsch (five species in New Caledonia). The Northern Hemisphere is well represented with about 220 species (73.5% of the world fauna).
Two species restricted to the seashore of the Pacific in North America.
The two species can be differentiated using Lindroth’s (1961a) and Bousquet’s (1988a) keys to Dyschirius.
This taxon has been considered a subgenus of Dyschirius by Lindroth (1961a) and a distinct genus by Fedorenko (1996). The phylogenetic analysis of Fedorenko (1996) did not yield clear evidence as to the position of the group. Furthermore there are little structural characteristics for members of Akephorus. The presence of conspicuous microsculpture on the body is also found in a few Dyschirius groups, such as the exochus group, phenetically similar to the remaining Dyschirius. The body shape, however, is distinctive. With the current phylogenetic knowledge of the tribe, I see no harm in retaining this taxon as a distinct genus.
This species is confined to the seashore of the Pacific Coast of California, as far north as San Mateo County (CAS), and of the Baja California Peninsula (CNC).
USA: CA (CHI) – Mexico
This species ranges along the seashore of the Pacific from Kunghit Island in the Queen Charlotte Islands (James C. Bergdahl pers. comm. 2009) south at least to central California (LeConte 1867b: 363; San Mateo County, CAS).
CAN: BC (QCI, VCI) USA: CA, OR, WA
About 245 species (Lorenz 2005: 151-154) in the Nearctic (about 60 species, one of them adventive), Neotropical (about 20 species, five shared with North America), Oriental (about 25 species), Palaearctic (about 140 species, many shared with the Oriental and Afrotropical Regions), and Afrotropical (about 30 species) Regions.
The North American species have never been revised. Bousquet (1988a) published a key to all species found in the area and briefly discussed each species-group. Subsequently five new species have been described by Bousquet (1997a) and Dajoz (2004), an adventive species has been detected (Dyschirius globosus) in North America, two species have been treated as junior synonyms (Dyschirius filiformis with Dyschirius sublaevis and Dyschirius integer with Dyschirius dejeanii), and two species had their names changed (Dyschirius nigricornis for Dyschirius melancholicus and Dyschirius integer for Dyschirius dejeanii).
Fedorenko (1996) recognized two genera within Dyschirius (minus Akephorus): Dyschirius with about 20 Palaearctic species and Dyschiriodes for the remaining species. However, there seems to be no consistent character state that separates the two “genera” except possibly for the basal sclerite of the endophallus which is “strongly sclerotized” in Dyschirius and “poorly sclerotized” in Dyschiriodes. In addition there is no solid evidence presented suggesting that the genus Dyschirius of authors is polyphyletic or that Dyschirius sensu Fedorenko and Dyschiriodes are sister-taxa. The genus Dyschirius sensu auctorum (prior to Fedorenko 1996) is a well-defined taxon and I see no reason to recognize Dyschiriodes as generically distinct. A similar approach was adopted by Balkenohl (2003: 223-230) in the Catalogue of Palaearctic Coleoptera, Lorenz (2005: 151-154), and Erwin (2011b: 97-128).
Fedorenko (1996) recognized five subgenera within “Dyschiriodes:” Antidyschirius Fedorenko, 1996 [type species: Dyschirius laevifasciatus Horn, 1878], Eudyschirius Fedorenko, 1996 [type species: Dyschirius lafertei Putzeys, 1846], Chiridysus Fedorenko, 1996 [type species: Dyschirius strumosus Erichson, 1837], Dyschiriodes Jeannel, 1941 [type species: Clivina punctata Dejean sensu Jeannel, 1941 (= Clivina minuta Dejean, 1825)], and Paradyschirius Fedorenko, 1996 [type species: Dyschirius parallelus Motschulsky, 1844]. In my opinion there are several other species groups actually recognized within Dyschirius that deserve a subgeneric name. For that reason I have listed the North American species under species-group names and did not retain Fedorenko’s subgenera until further studies are done on the North American fauna. For information, the species of the laevifasciatus group belong to Antidyschirius; those of the tridentatus, ferrugineus, and brevispinus groups, as well as Dyschirius globosus (classified here in the dejeanii group), Dyschirius comatus (placed here in the pilosus group), Dyschirius criddlei and Dyschirius edentulus (both included here in the politus group) belong to Eudyschirius; those of the analis and quadrimaculatus groups to Paradyschirius; and those of all remaining North American groups to Dyschiriodes sensu stricto.
[analis group]
This species ranges from Maine (Larochelle and Larivière 1990a: 28; Androscoggin County, CNC) to southeastern Montana (Herman 1986: 62), south to westernmost (Fall 1901a: 209, as Dyschirius duplicatus) and southern Texas (Cameron, San Patricio, and Zapata Counties, CMNH, MCZ) and Maryland (Erwin 1981b: 140). The record from “Arizona” (Bousquet and Larochelle 1993: 98) needs confirmation.
CAN: ON, QC USA: AR, CO, CT, DC, IA, IL, IN, KS, MA, MD, ME, MT, ND, NE, NH, NM, NY, OH, OK, PA, TN, TX, VA, VT, WI, WY [AZ]
The range of this species extends from eastern Washington (Hatch 1953: 67) to southern California (Fall 1901a: 41; Moore 1937: 5), east to southwestern Louisiana (Cameron Parish, LSAM), south to Veracruz in Mexico (Kult 1950a: 138, as Dyschirius hintoni). The records from southeastern British Columbia (Hatch 1953: 67) and “Idaho” (Bousquet and Larochelle 1993: 98) need confirmation; that from “Nebraska” (Bousquet and Larochelle 1993: 98) is probably in error.
USA: AZ, CA, LA, NM, NV, OR, TX, WA [BC, ID] – Mexico
This species is found from southern New Hampshire (Hillsborough County, Ross T. Bell pers. comm. 2008) to “North Dakota” (Donald P. Schwert pers. comm. 1989), south to eastern Texas (San Augustine and Wood Counties, CMNH; Bousquet 1988a: 373; Riley 2011) and southern Florida (Peck and Thomas 1998: 17). The record from “New Mexico” (Bousquet and Larochelle 1993: 99) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV [NM]
This species ranges from Massachusetts (Hampshire County, CNC) to northeastern Kansas (Douglas County, MCZ), including central Wisconsin (Messer 2010: 34) and southeastern Iowa (Des Moines County, MCZ), south to San Luis Potosí in Mexico (Bousquet 1988a: 373) and the Florida Panhandle (Peck and Thomas 1998: 17), west along southern United States to southeastern California (Imperial County, CAS; Dajoz 2007: 16). One old specimen simply labeled “Neb” is known (MCZ).
USA: AL, AR, AZ, CA, CT, DE, FL, IA, IL, IN, KS, LA, MA, MD, MI, MO, MS, NC, NJ, NM, NY, OH, OK, PA, SC, TN, TX, VA, WI [NE] – Mexico
[brevispinus group]
This species is known from scattered localities from the Saint Lawrence Plain in southern Quebec (Larochelle 1975: 81; Bousquet 1987a: 112) to southeastern Michigan (LeConte 1878c: 593), south to southeastern Pennsylvania (Dauphin County, MCZ; Lindroth 1961a: 139); also known from northeastern Nebraska (Clopton 1991: 61).
CAN: ON, QC USA: MI, NE, OH, PA, VT
This species is known only from a few specimens collected in southern Alberta (Lindroth 1961a: 139; Bousquet 1987a: 113), northeastern Colorado (Bell 1971: 56; Lavigne 1978: 104), and northeastern Arizona (Apache County, UASM).
CAN: AB USA: AZ, CO
[carrorum group]
This species is known only from the holotype collected in south-central Manitoba.
CAN: MB
[exochus group]
This species is known from the type locality in southeastern Texas, from Jackson County in southeastern Mississippi (CMNH), and from two localities in the Florida Panhandle (Frank 1985: 481; Herman 1986: 63).
USA: FL, MS, TX
This species is known only from the holotype collected along the Gulf Coast of Florida.
USA: FL
[ferrugineus group]
This species is known only from a few localities along the Gulf Coast in southern Texas (Bousquet 1988a: 371; FMNH).
USA: TX
[globulosus group]
This species ranges from southern Labrador to the Kenai Peninsula in Alaska (Lindroth 1961a: 152, as Dyschirius frigidus), south to northern Idaho (Hatch 1953: 67), northeastern Minnesota (Gandhi et al. 2005: 923), northeastern Illinois (Peter W. Messer pers. comm. 2008), and the upper and lower peninsulas of Michigan (Hubbard and Schwarz 1878: 627, 644). The records from Colorado (LeConte 1879d: 500; Wickham 1902: 232), Washington and southwestern Oregon (Herman 1986: 60, as Dyschirius frigidus; Hatch 1953: 67), and “California” (Leng and Beutenmüller 1894: 184) need confirmation; that from Iowa (Wickham 1911b: 5) is probably in error.
CAN: AB, BC, LB, NT, ON, QC, SK, YT USA: AK, ID, IL, MI, MN, WI [CA, CO, OR, WA]
This species is known from a few montane locations in south-central British Columbia (Lindroth 1961a: 152; Bousquet 1987a: 116), Idaho (Bear Lake County, CNC), and the Sierra Nevada of California (Papp 1978: 165; Mono and Placer Counties, USNM). The record from “Wyoming” (Bousquet and Larochelle 1993: 101) needs confirmation.
CAN: BC USA: CA, ID [WY]
This species is known only from two specimens collected at the type locality in southeastern Arizona.
USA: AZ
This species is known from a few montane locations from southwestern Alberta to south-central British Columbia (Lindroth 1961a: 152; Bousquet 1987a: 116).
CAN: AB, BC
This species ranges from southwestern Washington (Herman 1986: 60) to Riverside County (CAS) in southeastern California and “the middle Sierras” (Fall 1901a: 41). The records from southeastern British Columbia and northern Idaho (Hatch 1953: 67) are probably in error.
USA: CA, OR, WA
This species ranges from Newfoundland (Lindroth 1955a: 42, as Dyschirius nigripes) to east-central Alaska (Lindroth 1961a: 150), south to southern California (Fall 1901a: 41, as Dyschirius basalis), central New Mexico (Socorro County, CNC), Kansas (Popenoe 1877: 22; Horn 1872c: 384, as Dyschirius sulcatus), central Pennsylvania (Northumberland County, CMNH), and northeastern New Jersey (Smith 1910: 201, as Dyschirius nigripes; Bergen County, MCZ).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NM, NV, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WY
This species ranges from southwestern Oregon (Herman 1986: 60) to southern California (Fall 1901a: 41; San Diego County, CAS). The record from “Arizona” (Bousquet and Larochelle 1993: 101) needs confirmation.
USA: CA (CHI), OR [AZ]
This European species is adventive in North America where it is known only from the Vancouver area in southwestern British Columbia (Bousquet 1997a: 94, as Dyschirius glomerosus). The first inventoried specimen collected on this continent was found in 1978.
CAN: BC – Adventive
The range of this species extends from Newfoundland to central Alaska, south to southern British Columbia (Lindroth 1961a: 155), northern Arizona (Coconino County, CMNH), southern New Mexico (Otero County, CNC), east-central Texas (Riley 2011), southeastern Louisiana (Summers 1874a: 79), northern Mississippi (Drew A. Hildebrandt pers. comm. 2007), and southern Florida (Peck and Thomas 1998: 17).
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK, YT USA: AK, AL, AZ, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TX, VA, VT, WI, WV, WY
This species ranges from Labrador and the Ungava Bay area to Alaska, south to south-central British Columbia; isolated on the Shickshock Mountains in Gaspé Peninsula, Quebec [see Bousquet 1987a: map 1]. Fossil remnants, dated between 10, 400 and 12, 600 years B.P., have been unearthed in Cape Breton Island, Nova Scotia (Miller 1997: 250).
CAN: AB, BC, LB, MB, NT, ON, QC, SK, YT USA: AK
This taxon has been treated as a subspecies of Dyschirius melancholicus Putzeys by Fedorenko (1996: 149) but retained as a distinct species by Bousquet (1987a: 116) and Balkenohl and Lompe (2003: 99).
This species is found from Newfoundland (Lindroth 1955a: 43) to south-central British Columbia (Lindroth 1961a: 156), south to northwestern Minnesota (Gandhi et al. 2005: 924). The records from South Dakota (Kirk and Balsbaugh 1975: 16), Indiana (Blatchley 1910: 57), the upper and lower peninsulas of Michigan (Hubbard and Schwarz 1878: 627, 644; Hatch 1925: 548; Silvey 1936: 657), Ohio (Leng and Beutenmüller 1893: 91; Dury 1902: 110), and “Pennsylvania” (Bousquet and Larochelle 1993: 101) are in error.
CAN: AB, BC, MB, NF, QC, SK USA: MN
This Holarctic species ranges from Scandinavia (Balkenohl 2003: 226) to the Hudson Bay in northeastern Manitoba (Garry 1993: 94, as Dyschirius nigricornis). Fossil remnants, dated between 10, 800 and 20, 530 years B.P., have been unearthed in central North Dakota (Ashworth and Schwert 1992: 260), northeastern Iowa (Schwert 1992: 76; Woodman et al. 1996: 17), and southeastern Iowa (Baker et al. 1986: 96).
CAN: MB, NT, NU, YT USA: AK – Holarctic
This species has long been known under the name Dyschirius nigricornis Motschulsky, 1844 in the North American literature.
This species is known from the southern part of the Prairie Provinces (Lindroth 1961a: 155; Bousquet 1987a: 118) and from northwestern Montana (Teton County, CNC). The records from British Columbia (Jarrett and Scudder 2001: 382), “Wyoming, ” and “Minnesota” (Bousquet and Larochelle 1993: 101) need confirmation.
CAN: AB, MB, SK USA: MT [BC, MN, WY]
This subspecies is found from Alaska to the Great Slave Lake area in Northwest Territories (Lindroth 1961a: 151).
CAN: NT, YT USA: AK
The subspecies Dyschirius subarcticus altaicus Fedorenko occurs in the eastern part of the Palaearctic Region.
This species is known only from southern Manitoba (Lindroth 1961a: 154) and Alberta (Langor et al. 2006: 13).
CAN: AB, MB
This species is known only from the type locality in southwestern North Carolina.
USA: NC
[laevifasciatus group]
This species is found from the foothills of the Rocky Mountains in southern Alberta to south-central British Columbia (Lindroth 1961a: 140), south at least to central Oregon (Benton and Lincoln Counties, CNC, MCZ, USNM). One old specimen labeled “Cal” is known (MCZ).
CAN: AB, BC USA: OR, WA [CA]
[pilosus group]
This species is confined to the Coastal Plain ranging from North Carolina to central Florida, west to southeastern Louisiana (Tangipahoa and Saint Tammany Parishes, LSAM) [see Bousquet 1988a: Fig. 38]. One specimen simply labeled from Texas (CMNH) is known.
USA: AL, FL, LA, MS, NC, SC [TX]
This species ranges from southern New Brunswick (Webster and Bousquet 2008: 16) to southern Manitoba, south to eastern Texas, southeastern Louisiana (LeConte 1857b: 80; East Baton Rouge Parish, LSAM), Mississippi (Bolivar and Issaquena Counties, Drew A. Hildebrandt pers. comm. 2009), and northeastern North Carolina [see Bousquet 1988a: Fig. 38]. The records from southwestern Georgia (Fattig 1949: 13) and southwestern Alabama (Löding 1945: 12) probably refer to Dyschirius comatus; that from “Montana” (Bousquet and Larochelle 1993: 100) is probably in error.
CAN: MB, NB, ON, QC USA: CT, DC, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, TN, TX, VA, VT, WI, WV
This species is found from Prince Edward Island (Larochelle and Larivière 1990a: 27) to south-central British Columbia (Lindroth 1961a: 157), south to eastern Washington (Hatch 1953: 68, as Dyschirius alternatus), northern Utah (Utah County, USNM), Oklahoma (Custer and Latimer Counties, CMNH, UASM), and Long Island in New York (MCZ). Seemingly isolated at Fairbanks, Alaska (Lindroth 1961a: 157). The records from “New Jersey, ” “Maryland” (Bousquet and Larochelle 1993: 100), and “Colorado” (Wickham 1902: 232) need confirmation.
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: AK, IA, ID, IL, MA, ME, MI, MN, MT, ND, NE, NH, NY, OK, PA, RI, SD, UT, VT, WA, WI, WY [CO, MD, NJ]
[politus group]
This species is known only from the holotype collected in northeastern Kansas.
USA: KS
This species is known from southern Manitoba (Lindroth 1961a: 148), North Dakota (Grand Forks County, CNC, UASM), southeastern Nebraska (Foster F. Purrington pers. comm. 2010), Kansas (Stafford County, CNC, UASM), New Mexico (Chaves County, CNC, UASM), northwestern Texas (Hutchinson County, Darren A. Pollock pers. comm. 2011), and southern Florida (Monroe County, FFPC, UASM).
CAN: MB USA: FL, KS, ND, NE, NM, TX
This species is known only from north-central Oklahoma (Herman 1986: 61), southeastern Texas (Putzeys 1846: 52; Larson 1968: 1110, as Dyschirius colossus; Cameron and Aransas Counties, MCZ, UASM), and Florida (Dixie County, CMNH; Monroe County, FFPC). The record from the lower peninsula of Michigan (Hubbard and Schwarz 1878: 644) is in error.
USA: FL, OK, TX
This species ranges from southern New Brunswick (Webster and Bousquet 2008: 16) to southeastern South Dakota (Kirk and Balsbaugh 1975: 16), south to southwestern Oklahoma (Kondratieff et al. 2005: 172), southern Louisiana (Saint Martin Parish, LSAM), northern Mississippi (Bolivar, Marshall, and Warren Counties, Peter W. Messer pers. comm. 2009), and Delaware [see Bousquet 1988a: Fig. 37]. The records from the Bahamas (Turnbow and Thomas 2008: 12) and Horn Island, Mississippi (Richmond 1968: 233) refer to Dyschirius larochellei Bousquet; that from Colorado (Wickham 1902: 232) is in error.
CAN: NB, ON, QC USA: AR, CT, DE, IA, IL, IN, KS, KY, LA, MA, ME, MI, MN, MS, NE, NH, NJ, NY, OH, OK, PA, SD, TN, VT, WI
Bulirsch (2009: 19) reported that Dyschirius weyrauchi Kult, described from one specimen collected in Peru in 1900, is a junior synonym of Dyschirius erythrocerus. If this synonymy is correct, then the specimen from Peru is probably mislabeled.
This species is found along the Atlantic Coast from western Newfoundland (Lindroth 1955a: 45, as Dyschirius erythrocerus) to southern Florida, including the Keys (Peck and Thomas 1998: 17), the Bahamas, Cuba, and Hispaniola (Nichols 1988b: Fig. 5-6, as D. nr. erythrocerus), west along the Gulf Coast to southern Texas; also known from Yucatán in southern Mexico (Whitehead 1970: 181, as Dyschirius erythrocerus) [see Bousquet 1988a: Fig. 37].
CAN: NB, NF, NS USA: CT, FL, LA, MA, MS, NH, NJ, NY, SC, VA, TX – Bahamas, Cuba, Hispaniola, Mexico
This species is confined to the Pacific Coast ranging from the Queen Charlotte Islands (Kavanaugh 1992: 59) south at least to northern California (Mendocino County, CNC).
CAN: BC (QCI, VCI) USA: CA, OR, WA
This rarely collected species is known from scattered localities in the southern parts of the Prairie Provinces (Lindroth 1961a: 147; Bousquet 1988a: 373), southeastern Oregon (Harney County, CNC), northwestern (Pershing County, MCZ) and west-central (Bechtel et al. 1983: 474) Nevada, north-central Utah (Utah County, USNM), and eastern California (Fall 1925: 310, as Dyschirius desertus; Inyo and Plumas Counties, CAS).
CAN: AB, MB USA: CA, NV, OR, UT
This Holarctic subspecies is known from the British Islands to eastern Siberia, as far south as Italy, Bulgaria, Iran, Turkmenistan, Kyrgyzstan, and Mongolia (Balkenohl 2003: 226), and in the Nearctic Region from Alaska (Lindroth 1961a: 146) to Newfoundland (Lindroth 1955a: 36), south to southwestern Pennsylvania (Allegheny County, CMNH), eastern South Dakota (Kirk and Balsbaugh 1975: 16), northern New Mexico (Rio Arriba County, USNM), northeastern Nevada (Elko County, MCZ), and southwestern California (Los Angeles County, CAS).
CAN: AB, BC, MB, NB, NF, NT, ON, QC, SK, YT USA: AK, CA, CO, ID, MA, ME, MI, MT, NH, NM, NV, NY, OH, OR, PA, SD, UT, VT, WA, WI, WY – Holarctic
The subspecies Dyschirius politus chamunensis Fedorenko and Dyschirius politus meridianus Fedorenko occur in Asia.
The range of this species extends from Newfoundland (Lindroth 1955a: 44) to south-central British Columbia, south to eastern Oregon (Baker County, MCZ), southeastern Arizona (Greenlee and Graham Counties, CMNH), central New Mexico (Socorro County, CNC), southern Texas (Herman 1986: 61; Dajoz 2004: 117), eastern Tennessee (Knox County, MCZ), and North Carolina (Herman 1986: 63). The records from “Georgia” (J.E. LeConte 1849: 25), Florida (Leng 1915: 568), and San Bernardino County in southwestern California (Riley 1893: 239) need confirmation.
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AR, AZ, CO, CT, DC, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY [CA, FL, GA]
This species ranges from southern Manitoba to south-central British Columbia, north to east-central Alaska (Lindroth 1961a: 147), south to southern California (Fall 1901a: 41), central Arizona (Griffith 1900: 565), and southern Colorado (LeConte 1879d: 500; Wickham 1902: 232; Douglas County, CNC); also recorded from Baja California Sur (Horn 1895: 225). One old specimen labeled “Pittsburg VI Pa” (CMNH) is known.
CAN: AB, BC, MB, NT, SK USA: AK, AZ, CA, CO, IA, ID, IL, IN, MO, ND, NE, NV, SD, UT, WA, WI [PA] – Mexico
[pumilus group]
This species inhabits the Coastal Plain ranging from the coast of southern North Carolina (Brunswick County, Ken Karns pers. comm. 2009) to southern Florida (Nichols 1988b: Fig. 5-5; Peck and Thomas 1998: 17), west to southeastern Texas (Putzeys 1867b: 51).
USA: AL, FL, GA, LA, MS, NC, TX
According to Whitehead (1970: 185), members of Dyschirius darlingtoni Kult, 1950 from Mexico are probably conspecific with those of Dyschirius abbreviatus.
This species ranges from west-central Wisconsin (Messer 2010: 34) and southern Manitoba (Lindroth 1961a: 142, as Dyschirius dentiger) to the Okanagan Valley in south-central British Columbia (Bousquet 1987a: 115), south to southeastern California (Whitehead 1970: 186), Sonora (CNC), Chihuahua (CNC), southeastern Texas (Galveston County, CNC), and southeastern Mississippi (Jackson County, Drew A. Hildebrandt pers. comm. 2007).
CAN: AB, BC, MB USA: AZ, CA, CO, ID, KS, MS, MT, NE, NM, NV, OK, OR, TX, UT, WA, WI, WY – Mexico
This species is known from southern Maine (Kennebec County, Robert E. Nelson pers. comm. 1989), Connecticut (Litchfield County, CMNH) and “Rhode Island” (Sikes 2003: 7) in the northeast and from the Florida Panhandle (Peter W. Messer pers. comm. 2008) to southeastern Texas (Putzeys 1846: 42; Whitehead 1970: 186; Herman 1986: 61) in the southeast.
USA: CT, FL, LA, ME, MS, RI, TX
This species ranges from southern Quebec (Larochelle 1975: 82) to south-central British Columbia (Lindroth 1961a: 141), south to the Sierra Nevada in California (Dajoz 2004: 119), southeastern Arizona (Graham County, CNC), south-central Colorado (Wickham 1902: 232), Nebraska (Clopton 1991: 61), Wisconsin (Messer 2010: 34), and “Michigan” (Garry A. Dunn pers. comm. 1986). One old specimen simply labeled from New Mexico is known (Fall and Cockerell 1907: 156).
CAN: AB, BC, MB, ON, QC, SK USA: AZ, CA, CO, MI, MN, NE, NV, OR, SD, WA, WI, WY [NM]
This species is known only from the two specimens collected at the type locality in eastern California.
USA: CA
This species is known along the Coastal Plain and Piedmont Plateau from southeastern New York (Notman 1928: 212) to southern Florida including the Keys (Peck and Thomas 1998: 17), west to southern Texas (Aransas and Brooks Counties, CNC), and also from Minnesota (Gandhi et al. 2005: 924) and west-central Wisconsin (Messer 2010: 34). The record from southern Arizona (Wickham 1898: 300) is probably in error (Nichols 1988a: 209).
USA: AL, DC, FL, GA, LA, MD, MN, MS, NC, NJ, NY, PA, SC, TX, VA, WI
This species is known only from the type locality in southern Ontario, from Monroe and Sheboygan Counties in Wisconsin (Purrington and Maxwell 1998: 190; Messer 2010: 35), and from Highlands County in central Florida (Vince Golia collection).
CAN: ON USA: FL, WI
This species is known only from eight specimens collected at the type locality in southeastern California.
USA: CA
This species is found along the Atlantic and Gulf of Mexico coasts from Maine (Larochelle and Larivière 1990a: 27, as Dyschirius filiformis; York County, CNC) and New Hampshire (Rockingham County, CNC) to southern Florida including the Keys (Nichols 1988b: Fig. 5-6; Peck and Thomas 1998: 17), west to southeastern Texas (Putzeys 1846: 43; Wickham 1897: 103); also recorded from the Bahamas (Turnbow and Thomas 2008: 12), Cuba (Nichols 1988b: Fig. 5-4), Cayman Islands (Erwin 2011b: 124), and Yucatán, Mexico (Peck 2005: 28). The record from northeastern Kansas (Popenoe 1878: 78, as Dyschirius filiformis) needs confirmation; one old specimen labeled “Milwaukee WIS” is also known (MCZ).
USA: AL, CT, DE, FL, GA, LA, MA, MD, ME, MS, NC, NH, NJ, NY, RI, TX, VA [KS, WI] – Bahamas, Cayman Islands, Cuba, Mexico
[quadrimaculatus group]
This species is known from the Prairie Provinces (Lindroth 1961a: 148), “Montana” (Bousquet 1988a: 372), Wyoming (Park County, CNC), and “North Dakota” (Bousquet 1988a: 372). The record from “Idaho” (Bousquet and Larochelle 1993: 98) needs confirmation.
CAN: AB, MB, SK USA: MT, ND, WY [ID]
[sellatus group]
This species ranges from the southern part of the Prairie Provinces (Lindroth 1961a: 143; Bousquet 1987a: 115) to southern Arizona (Bousquet 1987a: 115; Dajoz 2007: 21), southern Texas, and central Arkansas, east to northeastern Ohio [see Davidson and Lee 1990: Fig. 1)].
CAN: AB, MB USA: AR, AZ, CO, ID, IL, KS, ND, NE, NM, OH, OK, SD, TX, UT, WY
The range of this species extends from southern Quebec (Larochelle 1975: 82) to southern Alberta (Bousquet 1987a: 115), south to northern Texas (Bowie and Winkler Counties, CMNH, USNM), southeastern Louisiana (West Baton Rouge Parish, LSAM), and southern Florida (Herman 1986: 63).
CAN: AB, ON, QC, SK USA: AR, FL, GA, IL, KS, LA, MI, MO, MS, MT, ND, NE, NJ, NY, OK, PA, SD, TX, VA
This species has been reported from southern Oregon (Herman 1986: 60), western Nevada (Bousquet 1988a: 376), and Utah (Knowlton 1939: 2; Lindroth 1961a: 144; Herman 1986: 61). The records from South Dakota (Kirk and Balsbaugh 1975: 16) and Albuquerque in New Mexico (Fall and Cockerell 1907: 156) need confirmation.
USA: NV, OR, UT [NM, SD]
This species ranges from Newfoundland (Lindroth 1955a: 45) to central Florida (Peck and Thomas 1998: 17), west to northeastern Texas (Lindroth 1961a: 143), including southeastern Mississippi (George and Greene Counties, Drew A. Hildebrandt pers. comm. 2008), and north to southern North Dakota (Burleigh County, Donald P. Schwert pers. comm. 1989) and eastern Minnesota (Gandhi et al. 2005: 924). The record from “Pennsylvania” (Bousquet and Larochelle 1993: 100) needs confirmation.
CAN: NB, NF, NS, PE, QC USA: FL, GA, IL, LA, MD, MN, MO, MS, NC, ND, NE, NJ, NY, OK, SD, TX [PA]
[tridentatus group]
This species ranges from the southern part of the Prairie Provinces (Lindroth 1961a: 139) south to east-central California (Dajoz 2004: 119), northern Arizona (Van Dyke 1943: 22, as Dyschirius arizonicus; Mojave and Navajo Counties, MCZ, USNM), and western Texas (Randall and Ward Counties, CMNH, USNM); also known from southwestern Oregon (Curry County, DAPC).
CAN: AB, MB, SK USA: AZ, CA, CO, ID, NE, NM, NV, OR, TX, UT, WY
This species is found along the Pacific Coast in southern California (LeConte 1852a: 196). The record from “Oregon” (Leng 1920: 47) needs confirmation; that from southwestern British Columbia (Hatch 1953: 67) is in error.
USA: CA [OR]
This species is found from northern Washington (Lindroth 1961a: 138) to western Montana (Russell 1968: 47), south to southwestern New Mexico (Sierra County, CMNH) and southwestern California (LeConte 1852a: 195; Moore 1937: 5). The records from “Nebraska” (Bousquet and Larochelle 1993: 98) and northwestern British Columbia (Hatch 1953: 67) are probably in error. One old specimen simply labeled “Van” is known (MCZ).
USA: AZ, CA, ID, MT, NM, NV, OR, WA [BC]
This species is found in southern California (Fall 1901a: 207).
USA: CA
This species ranges from “Washington” (Hatch 1953: 67) to western Montana (Jefferson County, CNC), south to northeastern Nevada (Elko County, CNC) and southern California (Fall 1910: 93; San Diego County, CAS, UASM). The record from “Wyoming” (Bousquet and Larochelle 1993: 98) needs confirmation.
USA: CA, ID, MT, NV, OR, WA [WY]
Eight species in North America (two species) and South Africa (six species) arrayed in five genera: Axinidium Sturm (two species), Holaxinidium Basilewsky (one species), Metaxinidium Basilewsky (two species), Paraxinidium Basilewsky (one species), and Promecognathus (two species).
Two species restricted to western North America.
Lindroth (1961a: 125-128) commented on the structural differences between the two species.
The status of the two forms as distinct species is questionable in my opinion. Van Dyke (1925: 123) considered the two forms as conspecific.
This species is found along the Pacific Coast from southwestern British Columbia, including Vancouver Island (Lindroth 1961a: 128), to central California (Casey 1913: 94, as Promecognathus corpulentus).
CAN: BC (VCI) USA: CA, OR, WA
Promecognathus crassus LeConte. This species, along with its close relative Promecognathus laevissimus, are western coastal elements that feed on polydesmid millipedes. The adults straddle their prey, moving quickly toward the head to pierce the neck and sever the ventral nerve cord with their long mandibles, thereby preventing the millipedes from using their cyanide defense spray. Promecognathines represent a very old, relic lineage, likely originating from Pangea, represented today only in western North America and the Cape Province in South Africa.
This species is known from “Oregon” south at least to central California (Lindroth 1961a: 127; Fresno County, CAS), east to Washoe County in northwestern Nevada (La Rivers 1947: 139; Lindroth 1961a: 127).
USA: CA, NV, OR
One tribe is included in this subfamily.
About 290 species (Häckel et al. 2010) in the Nearctic (four species, one of them adventive), Neotropical (about 30 species, one in Mexico, the other ones in South America), Australian (about 180 species), Oriental (five species), and Palaearctic (about 70 species) Regions placed in 34 genera. The genera are arrayed in five subtribes (Roig-Juñent 2000): Broscina (about 75 species), Creobiina (about 95 species in South America and the Australian Region), Anoxyina (five species in Mexico, Iran, and the Oriental Region), Baripodina (about 25 South American species), and Nothobroscina (about 90 species in the Australian Region and one in Chile). The group is better represented in the New World with about 215 species (74% of the world fauna) than in the Old World.
About 75 species in the Nearctic (four species), Oriental (four species), and Palaearctic (about 65 species) Regions. One species (Miscodera arctica) is Holarctic and one is adventive (Broscus cephalotes) in North America.
One Holarctic species in the subarctic and boreal regions.
Lindroth (1961a: 170-171) covered the species.
Cladistic analysis of broscine genera performed by Roig-Juñent (2000) placed this genus as the sister-group of the Holarctic genus Broscodera Lindroth.
This circumpolar species ranges from Ireland to the Bering Sea coast (Bousquet 2003b: 237) and from Alaska (Lindroth 1961a: 171) to Newfoundland (Lindroth 1955a: 133), south to northeastern New York (Notman 1928: 241), the upper peninsula of Michigan (Chippewa County, MCZ), northern Wisconsin (Bayfield County, MCZ), northern Colorado (Dajoz 1989: 337; Gilpin County, CMNH), southern Montana, and northern Washington (Hatch 1933b: 7). Fossil remnants of this species, believed to be 2.0-2.5 million years old, have been found in Greenland (Bennike and Böcher 1990: 336; Böcher 1995: 23); others, about 20, 530 years B.P., have been unearthen in northeastern Iowa (Woodman et al. 1996: 17).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: AK, CO, ME, MI, MT, NH, NY, VT, WA, WI, WY – Holarctic
Northern Hemisphere, with four species in the Nearctic (one species) and Palaearctic (three species) Regions arrayed in two subgenera: Broscodera s.str. (one species) and Sinobrosculus Deuve (three species in Nepal, Gansu, and Sichuan).
Cladistic analysis of broscine genera performed by Roig-Juñent (2000) placed this genus as the sister-group of the Holarctic genus Miscodera Eschscholtz.
One species in the temperate regions of western North America.
Lindroth (1961a: 171-172) treated the species.
This species is confined to the Pacific Coast and adjacent Coast Ranges, ranging from the Alexander Archipelago in southeastern Alaska to western Oregon (Lindroth 1961a: 172). The record from southeastern Wyoming (Lavigne 1977: 45) must be based on a mislabeled specimen.
CAN: BC (QCI) USA: AK, OR, WA
One species in temperate western North America.
The species is covered in Lindroth’s (1961a: 172-173) monograph.
Cladistic analysis of broscine genera performed by Roig-Juñent (2000) placed this genus as the sister-group of the Asian genus Eobroscus Kryzhanovskij.
This species ranges from the southern part of the Alexander Archipelago to northwestern California, east to the Bitter Root Mountains in southwestern Montana (Ball 1956b: 34, Fig. 1).
CAN: BC (QCI, VCI) USA: AK, CA, ID, MT, OR, WA
Based on variation in color, lustre, and surface sculpture in adults, Ball (1956b) recognized a western and eastern “races” for this species.
Twenty-three species in the Palaearctic Region, one of them adventive in eastern North America.
Larochelle and Larivière (1989a) provided a description of the external structures as well as the male and female genitalia of the species found in North America.
Cladistic analysis of broscine genera performed by Roig-Juñent (2000) placed this genus as the sister-group of the temperate Asian genus Craspedonotus Schaum.
This Palaearctic species is adventive in North America where it is known from Cape Breton Island and eastern Prince Edward Island (Larochelle and Larivière 1989a: Fig. 4). The first inventoried specimen found on this continent was caught in 1987.
CAN: NS (CBI), PE – Adventive
Broscus cephalotes (Linnaeus). This relatively large Palaearctic carabid is one of the most recently established species in North America. The first recorded specimen was found in the late 1980s and, considering the species’ size, it is doubtful that it would have escaped notice for a long period. On this continent, the species has been found only on bare, fine sand along coastal beaches in Prince Edward Island and Cape Breton in company with amphipods of the genus Gammarus which probably make up part of their diet. The pathway of introduction of this broscine on American soil is uncertain.
Five species placed in two subtribes: Gehringiina (one species) and Helenaeina (three rarely collected species in the genus Helenaea Schatzmayr and Koch from Egypt, Turkey, and Yemen and one species in the genus Afrogehringia Baehr, Schüle and Lorenz from Namibia).
Baehr et al. (2009: 106) provided a key to all species of this subfamily.
Three genera and five species are placed in this tribe.
One North American species belongs to this subtribe.
One species in western North America.
The species was covered in Lindroth’s (1961a: 4-5) monograph on the carabids of Canada and Alaska.
This minute species ranges from central Alaska (66.79376°N, 150.73164°W, Derek S. Sikes pers. comm. 2008) to southern Northwest Territories (CNC), south to northwestern Montana (Edwards 1975: 48), southeastern Idaho (Caribou County, CNC), and northeastern Oregon (Lindroth 1961a: 5).
CAN: AB, BC, NT, YT USA: AK, ID, MT, OR, WA
Worldwide, with about 5, 410 species arrayed in four tribes: Bembidiini (about 2, 630 species), Pogonini (about 80 species), Trechini (about 2, 650 species), and Zolini (about 50 species). The North American fauna is represented by about 615 species (roughly 11.3% of the world fauna).
Worldwide, with about 2, 650 species arrayed in two subtribes: Trechina (about 2, 470 species) and Trechodina (about 180 species). Only the subtribe Trechina is represented in North America.
Worldwide, with about 2, 470 species (Lorenz 2005: 168-200). The North American fauna is represented by about 225 species (roughly 9% of the world fauna) arrayed in nine genera.
Northern Hemisphere, with six species in temperate areas of the Nearctic (one western species) and Palaearctic (five species) Regions.
Barr (1972) provided a description of the external structures and male genitalia of the North American species.
This species is known only from a few localities in the Willamette Valley in northwestern Oregon.
USA: OR
About 145 described species, though more than 220 are known (Barr 2004: 1), restricted to eastern North America south of the last glaciation.
Barr (2004: 11-16) provided a key for the identification of the 26 species groups currently recognized. Barr (1959) revised the species of the robustus group (four species). Krekeler (1973) revised the species of the barri (two species), horni (13 species), inexpectatus (six species), and rittmani (three species) groups and provided keys for the identification of the species of the horni and inexpectatus groups. Barr (1981) revised the species of the alabamae (two species), hirsutus (seven species), hubrichti (six species), hypolithos (five species), jonesi (eight species), and tennesseensis (four species) groups but did not provide keys for their identification. He also treated the engelhardti group but many species were added subsequently.
Duvaliopsis Jeannel, with six species in Carpathian and Transylvanian Alps of eastern Europe, is listed as a synonym of Pseudanophthalmus by Barr (2004: 7). Most other trechine students treat Duvaliopsis as a distinct genus.
Tennessarius Valentine (1952: 15), listed as a junior synonym of Pseudanophthalmus Jeannel by Barr (1962b: 111), is an unavailable name since the original description was not accompanied by the fixation of a type species (ICZN 1999: Article 13.3). Barr (2004: 7) provided a type species for Tennessarius along with a bibliographic reference to a previously published description but since he listed Tennessarius in synonymy with Pseudanophthalmus, the name is still unavailable (ICZN 1999: Article 11.6).
[alabamae group]
This species is known from a number of caves in DeKalb County, northeastern Alabama (Barr 2004: 40).
USA: AL
This species is known only from a few caves in Chattooga and Walker Counties, northwestern Georgia (Barr 2004: 40).
USA: GA
[audax group]
This species is known only from two caves in Hart and Edmonson Counties, Kentucky (Barr 2004: 24).
USA: KY
This species is known only from two caves in Lawrence County, southern Indiana (Barr 2004: 24).
USA: IN
This species is known from several caves in Carter and Elliott Counties, northeastern Kentucky (Barr 2004: 24).
USA: KY
[barri group]
This species is known from several caves in southern Clark County, southern Indiana (Barr 2004: 24).
USA: IN
This species is known only from two nearby caves in Jefferson County, north-central Kentucky (Barr 2004: 24).
USA: KY
[cumberlandus group]
This species is known from several caves in Rutherford and Wilson Counties, Tennessee (Barr 2004: 33).
USA: TN
This species is known only from the type-locality cave located two miles west of Southport in west-central Tennessee (Barr 2004: 34).
USA: TN
This species is known only from the type-locality cave in south-central Tennessee (Barr 2004: 33).
USA: TN
This species is known from a few caves in Smith and Macon Counties, northern Tennessee (Barr 1980: 91; Barr 2004: 33).
USA: TN
This species is known only from the type-locality cave in northern Tennessee (Barr 2004: 34).
USA: TN
This species is known only from the type-locality cave in north-central Tennessee (Barr 2004: 33).
USA: TN
This species is known from two nearby caves in Lewis and Hickman Counties, western Tennessee (Barr 1980: 93; Barr 2004: 33).
USA: TN
This species is known from several caves in Smith, Putnam, and Jackson Counties, northern Tennessee (Barr 1980: 92; Barr 2004: 33).
USA: TN
This species is still known only from two nearby caves in central Tennessee (Barr 1980: 92; Barr 2004: 33).
USA: TN
This species is known only from Carroll and Riley Creek caves, Coffee County, south-central Tennessee (Barr 1980: 93; Barr 2004: 33).
USA: TN
[engelhardti group]
This species is known from a few caves in Madison County, northern Alabama (Barr 2004: 35).
USA: AL
This species is known only from the type-locality cave in southwestern Virginia (Barr 2004: 34).
USA: VA
This species is known from a few caves in Morgan County, northern Alabama (Barr 2004: 36).
USA: AL
This species is known only from the type-locality cave in northeastern Tennessee (Barr 2004: 34).
USA: TN
This species is known only from the type-locality cave in northwestern Georgia (Barr 2004: 35).
USA: GA
This species is known from several caves in Marshall and Morgan Counties, northern Alabama (Barr 2004: 36).
USA: AL
This species is known only from a number of caves in Hamilton County, southeastern Tennessee, and Dade County, northwestern Georgia (Barr 1981: 49; Barr 2004: 35).
USA: GA, TN
This species is known from a few caves in Perry and Wayne Counties, Tennessee (Barr 2004: 36).
USA: TN
This species has been found yet only at the type-locality cave in southwestern Virginia (Barr 2004: 35).
USA: VA
This species is known from a few caves in Grundy and Franklin Counties, southern Tennessee (Barr 2004: 35).
USA: TN
This species is known from several caves in Madison County, northern Alabama (Barr 2004: 35).
USA: AL
This species is known from a few caves in northern Alabama (Barr 2004: 36).
USA: AL
This species is known only from the type-locality cave in southern Tennessee (Barr 2004: 35).
USA: TN
This species is known only from the type-locality cave in east-central Tennessee (Barr 2004: 35).
USA: TN
This species is known from a few caves in northern Alabama (Barr 2004: 35).
USA: AL
This species has been reported from a number of caves in Claiborne and Hancock Counties in northeastern Tennessee and Lee County in southwestern Virginia (Barr 2004: 35).
USA: TN, VA
This species is known only from the type-locality cave in northeastern Alabama (Barr 2004: 35).
USA: AL
This species is known only from the type-locality cave in northern Tennessee (Barr 2004: 35).
USA: TN
This species is known from a few caves in Blount County, north-central Alabama (Barr 2004: 35).
USA: AL
This species is known only from the type-locality cave in eastern Tennessee (Barr 2004: 34).
USA: TN
[eremita group]
This species is known only from two caves in central Kentucky (Barr 2004: 27).
USA: KY
This species is known from two caves in Crawford and Harrison Counties, southern Indiana (Barr 2004: 27).
USA: IN
[gracilis group]
This species is known from a few caves in Giles and Craig Counties, western Virginia (Barr 2004: 21).
USA: VA
This species is known only from the type-locality cave, near Onego, in eastern West Virginia (Barr 2004: 21).
USA: WV
[grandis group]
This subspecies is known only from a few caves in Greenbrier and Monroe Counties, southeastern West Virginia (Barr 2004: 17).
USA: WV
This subspecies is known from several caves in Greenbrier County, southeastern West Virginia (Barr 2004: 17).
USA: WV
This subspecies is known only from a few caves in Greenbrier and Monroe Counties, southeastern West Virginia (Barr 2004: 17).
USA: WV
This subspecies is known from several caves in Greenbrier County, southeastern West Virginia (Barr 2004: 17).
USA: WV
This species is known from a few caves in southern Greenbrier County, West Virginia (Barr 2004: 17).
USA: WV
This species is known only from a few caves in southern Pocahontas County, east-central West Virginia (Barr 2004: 17).
USA: WV
This species is known only from a few specimens collected at the type-locality cave in northeastern West Virginia (Barr 2004: 18).
USA: WV
This species is known only from two caves in Tucker County, northeastern West Virginia (Barr 2004: 18).
USA: WV
This species is known only from a few caves in Monroe County, southeastern West Virginia (Barr 2004: 17).
USA: WV
This species is known only from two nearby places in the Yew Mountains, east-central West Virginia (Barr 2004: 17).
USA: WV
This species is still known only from the type-locality cave in southwestern Virginia (Barr 2004: 18).
USA: VA
[hirsutus group]
This species is known only from two caves in DeKalb County, northeastern Alabama (Barr 2004: 37).
USA: AL
This species is known from several caves in Lee County, southwestern Virginia (Barr 1981: 60; Barr 2004: 37).
USA: VA
This species is known from caves in Hamilton County, Tennessee, and Dade County, Georgia (Barr 1981: 64; Barr 2004: 37).
USA: GA, TN
This species is known from two nearby caves in Cumberland Gap National Park, Lee County, southwestern Virginia (Barr 1981: 60; Barr 2004: 37).
USA: VA
This species is known only from the type-locality cave in southeastern Tennessee (Barr 2004: 37).
USA: TN
This species is known only from the type-locality cave in southwestern Virginia (Barr 2004: 37).
USA: VA
This species is known only from the type-locality cave in southern Tennessee (Barr 2004: 37).
USA: TN
[horni group]
This species is known from a few caves in Woodford and Jessamine Counties, Kentucky (Barr 2004: 22).
USA: KY
This species is known only from the type-locality cave in east-central Kentucky (Barr 2004: 22).
USA: KY
This species is known from a few caves in Jefferson, Jennings, and Clark Counties, southern Indiana (Krekeler 1973: 51; Barr 2004: 23).
USA: IN
This species is known from a few widely scattered caves in Clark, Henry, Scott, and Owen Counties, Kentucky (Krekeler 1973: 49; Barr 2004: 22).
USA: KY
According to Barr (2004: 22), the Henry and Owen County populations possibly represent a distinct species.
This species is known from a few caves in Mercer and Garrard Counties, central Kentucky (Barr 2004: 22).
USA: KY
This species is known only from the holotype collected in Lee County, not Estill County as reported by Krekeler (1973: 53), in eastern Kentucky (Barr 2004: 23).
USA: KY
This species is known from several caves in Fayette County, north-central Kentucky (Barr 2004: 22).
USA: KY
This species is known only from the type-locality cave in southern Ohio (Barr 2004: 23).
USA: OH
This species is known only from the type-locality cave, 35 miles northeast of Lexington, northern Kentucky (Barr 2004: 22).
USA: KY
This species is known only from the type-locality cave in southern Ohio (Barr 2004: 23).
USA: OH
This species is known only from the type-locality cave in east-central Kentucky (Barr 2004: 23).
USA: KY
This species is known from a few caves in Woodford and Jessamine Counties, Kentucky (Barr 2004: 22).
USA: KY
This species is known only from the type-locality cave in northern Kentucky (Barr 2004: 22).
USA: KY
[hubbardi group]
This species is known only from the type-locality cave in west-central Virginia (Barr 2004: 18).
USA: VA
This species is known only from the type-locality cave in northern Virginia (Barr 2004: 18).
USA: VA
This species is known only from the type-locality cave in western Virginia (Barr 2004: 18).
USA: VA
This species is known from a few nearby caves in Shenandoah County, northern Virginia (Barr 2004: 18).
USA: VA
This species is known only from the type-locality cave in northern Virginia (Barr 2004: 18).
USA: VA
This species is known from two nearby caves in Pendleton County, eastern West Virginia, and Highland County, western Virginia (Barr 2004: 18).
USA: VA, WV
This species is known from a few nearby caves in eastern West Virginia (Barr 2004: 19).
USA: WV
[hubrichti group]
This species is known from two caves in Giles County, western Virginia (Barr 1981: 69; Barr 2004: 38).
USA: VA
This species is known only from the type-locality cave in southwestern Virginia (Barr 2004: 38).
USA: VA
This species is known only from the type-locality cave in northeastern Tennessee (Barr 2004: 38).
USA: TN
This species is known only from two nearby caves near Eggleston, western Virginia (Barr 2004: 38).
USA: VA
This species is known only from two caves in Russell and Scott Counties, southwestern Virginia (Barr 2004: 38).
USA: VA
This species is known from a number of caves in Tazewell County, southwestern Virginia (Barr 1981: 70; Barr 2004: 38).
USA: VA
[hypolithos group]
This species is known only from the type-locality cave in Whitley County, southeastern Kentucky (Barr 2004: 40).
USA: KY
This species is known only from the type-locality cave in southeastern Kentucky (Barr 2004: 40).
USA: KY
This species is known from the type-locality cave in Pike County and two abandoned coal mines in Floyd County, eastern Kentucky (Barr 2004: 40).
USA: KY
This species is known only from the type-locality cave in southwestern Virginia (Barr 2004: 40).
USA: VA
This species is known only from the type-locality cave in southeastern Kentucky (Barr 2004: 40).
USA: KY
[inexpectatus group]
This species is known only from two caves in Larue and Nelson Counties, central Kentucky (Barr 2004: 21).
USA: KY
This species is known from a few caves located in Mammoth Cave National Park, Kentucky (Barr 2004: 21).
USA: KY
This species is known from several caves in Green, Hart, and Taylor Counties, central Kentucky (Barr 2004: 21).
USA: KY
This species is known only from the type-locality cave in central Kentucky (Barr 2004: 21).
USA: KY
This species is known only from two nearby caves in Mercer and Boyle Counties, Kentucky (Barr 2004: 21).
USA: KY
This species is known from several caves in Garrard, Fayette, Woodford, and Owen Counties, Kentucky (Barr 2004: 21).
USA: KY
[intermedius group]
This species is known from several caves in Grundy and Franklin Counties, southern Tennessee (Barr 2004: 29).
USA: TN
This species is known only from several caves in Warren and Grundy Counties, Tennessee (Barr 2004: 29).
USA: TN
This species is known from several caves in Warren and Grundy Counties, Tennessee (Barr 2004: 29).
USA: TN
This species is known only from caves in Van Buren County, central Tennessee (Barr 2004: 29).
USA: TN
[jonesi group]
This species is known only from a few caves in Wise County, southwestern Virginia (Barr 2004: 39).
USA: VA
This species is known only from three caves in Grassy Cove, Cumberland County, in east-central Tennessee (Barr 1981: 73; Barr 2004: 39).
USA: TN
This species is known from two caves in Lee County, southwestern Virginia, and Hancock County, northeastern Tennessee (Barr 1981: 80; Barr 2004: 39).
USA: TN, VA
This species is known from a few caves in Claiborne County, northeastern Tennessee (Barr 2004: 39). The record from “Virginia” (Bousquet and Larochelle 1993: 114) is in error (see Hoffman et al. 2006: 19).
USA: TN
This species is known only from the type-locality cave in southeastern Kentucky (Barr 2004: 39). The record from “Virginia” (Bousquet and Larochelle 1993: 115) is in error (see Hoffman et al. 2006: 19).
USA: KY
This species is known only from the type-locality cave in northern Tennessee (Barr 2004: 39).
USA: TN
This species is known from several caves in the Rye Cove karst near Clinchport in southwestern Virginia (Barr 1981: 77).
USA: VA
This species is known only from two caves in Scott County, southwestern Virginia (Barr 2004: 39).
USA: VA
[leonae group]
This species is known only from the type-locality cave, located two miles southwest of Springville, southern Indiana (Barr 2004: 27).
USA: IN
[menetriesii group]
This subspecies is known from several caves in Monroe, Barren, Metcalfe, Adair, Cumberland, and Clay Counties in southern Kentucky and Jackson County in northern Tennessee (Barr 1985b: 124).
USA: KY, TN
This subspecies is known from a number of caves in central Barren County, southern Kentucky (Barr 2004: 32).
USA: KY
According to Barr (2004: 32), intergrades between this subspecies and the nominotypical subspecies occur in Bowles Branch Cave in Barren County.
This subspecies is known from several caves in northeastern Metcalfe, northern Adair, and southern Green Counties, southern Kentucky (Barr 1985b: 126; Barr 2004: 32).
USA: KY
This subspecies is known from several caves in central Green and eastern Hart Counties, Kentucky (Barr 2004: 32).
USA: KY
This species is known only from the type-locality cave in central Kentucky (Barr 2004: 31).
USA: KY
This subspecies is found in several caves in Barren and Warren Counties, southern Kentucky (Barr 2004: 31).
USA: KY
According to Barr (2004: 31), intergrades between this subspecies and the nominotypical subspecies occur in caves in Warren County.
This subspecies is known from several caves in Hart, Edmonson, Barren, and Warren Counties in Kentucky (Barr 2004: 31).
USA: KY
This species is known from caves in Hardin and Hart Counties, central Kentucky (Barr 2004: 31).
USA: KY
This species is known only from the type-locality cave in central Kentucky (Barr 2004: 31).
USA: KY
This species is known from several caves in Hart, Edmonson, Metcalf, and Warren Counties, southern Kentucky (Barr 2004: 32).
USA: KY
The MCZ holds a specimen [# 7397], incorrectly labeled lectotype, of Promecognathus interstitialis Hubbard from Cave City, Kentucky.
This species is known from a few caves in Warren and Logan Counties, southern Kentucky (Barr 1985b: 123; Barr 2004: 31).
USA: KY
[petrunkevitchi group]
This species is known from a few caves in Smyth and Bland Counties, Virginia (Barr 2004: 19).
USA: VA
This species is known only from the type-locality cave in southwestern Virginia (Barr 2004: 19).
USA: VA
This species, the only one of the genus Pseudanophthalmus with pigmented eyespots, is known from a few specimens collected at two nearby caves in Warren and Pages Counties, northern Virginia (Barr 2004: 19).
USA: VA
[pubescens group]
This subspecies occurs in several caves in Montgomery, Cheatham, and Robertson Counties in northern Tennessee and Christian and Logan Counties in southern Kentucky (Barr 2004: 30).
USA: KY, TN
This subspecies is known from a few caves in Logan and Simpson Counties, southern Kentucky, and Robertson County, northern Tennessee (Barr 2004: 30).
USA: KY, TN
According to Barr (2004: 30), intergrades between this subspecies and the nominotypical subspecies occur in one cave in Robertson County, Tennessee.
This species is known from several caves in Montgomery County, northern Tennessee (Barr 2004: 31).
USA: TN
This species is known from several caves in Warren, Simpson, and Logan Counties, southern Kentucky, and Robertson and Sumner Counties, northern Tennessee (Barr 2004: 30).
USA: KY, TN
This species is known from a few caves in Simpson and Warren Counties in southern Kentucky and Sumner County in northern Tennessee (Barr 2004: 30).
USA: KY, TN
This subspecies is known two caves in Allen and Barren Counties, southern Kentucky (Barr 1985b: 128).
USA: KY
This subspecies is known from several caves from Hart and Metcalfe Counties westwards to eastern Warren County, Kentucky (Barr 2004: 30).
USA: KY
According to Barr (2004: 30), intergrades between the two subspecies of Promecognathus pubescens are found in Beckton Cave, Barren County.
[pusio group]
This species is known only from a few caves in Greenbrier and Pocahontas Counties, eastern West Virginia (Jeannel 1949b: 75; Barr 2004: 20).
USA: WV
This species is known only from the type-locality cave in southeastern West Virginia (Barr 2004: 20).
USA: WV
This species is known only from two caves in Allegheny County, western Virginia (Barr 2004: 20).
USA: VA
This species is still known only from the type-locality cave in northern Virginia (Barr 2004: 20).
USA: VA
This species is known from a few caves in Giles County, western Virginia (Barr 2004: 20).
USA: VA
This species is found in several caves in Montgomery and Roanoke Counties, western Virginia (Barr 2004: 20).
USA: VA
[rittmani group]
This species is known only from the type-locality cave in east-central Kentucky (Barr 2004: 23).
USA: KY
This species is known from several caves in Estill, Powell, and Lee Counties, eastern Kentucky (Barr 2004: 23).
USA: KY
This species is known from several caves in Estill and Powell Counties, eastern Kentucky (Barr 2004: 23).
USA: KY
[robustus group]
This species is known from several caves in Fentress, Overton, and Pickett Counties, northern Tennessee, and in McCreary and Wayne Counties, southern Kentucky (Barr 2004: 28).
USA: KY, TN
This species occurs in several caves in DeKalb and Smith Counties, Tennessee (Barr 2004: 28).
USA: TN
This species is known from a several caves in DeKalb, Overtone, Putnam, Warren, White, Van Buren, and Grundy Counties, Tennessee (Barr 2004: 28).
USA: TN
This species is found in caves in Putnam and Overton Counties, northern Tennessee (Barr 2004: 28).
USA: TN
[simplex group]
This species is known only from the type-locality cave in northern Tennessee (Barr 2004: 32).
USA: TN
This species is known from a few caves in Jackson County, northern Tennessee (Barr 2004: 32).
USA: TN
[tennesseensis group]
This species is known only from a few caves in Anderson County, eastern Tennessee (Barr 2004: 37).
USA: TN
This species is known only from the type-locality cave in eastern Tennessee (Barr 2004: 36).
USA: TN
This species is known from a few caves in Knox and Roane Counties, eastern Tennessee (Barr 1981: 55; Barr 2004: 36).
USA: TN
This species is known from two caves in Union County, northeastern Tennessee (Barr 2004: 37).
USA: TN
[tenuis group]
This species is known from several caves in Meade, Breckinridge, Hardin, Hart, and Larue Counties, Kentucky (Barr 2004: 26).
USA: KY
This species is known only from the type-locality cave (Barr 2004: 26).
USA: IL
This subspecies is known from several caves in Monroe, Lawrence, and Owen Counties, southern Indiana (Barr 2004: 26).
USA: IN
Barr (2004: 26) noted that intergrades between this subspecies and Promecognathus shilohensis shilohensis occur in some caves in Lawrence County.
This subspecies is known from a few caves in central Lawrence County, southern Indiana (Barr 2004: 26).
USA: IN
This species is known from several caves in Crawford, Washington, Orange, Lawrence, and Monroe Counties, southern Indiana (Barr 2004: 25).
USA: IN
This species is known from several caves in Crawford, Harrison, and Washington Counties, southern Indiana (Barr 2004: 25).
USA: IN
This species is known from several caves in Crawford, Orange, Lawrence, and Washington Counties, southern Indiana (Barr 2004: 26).
USA: IN
Two cave-inhabiting Appalachian species.
Valentine (1952) discussed the structural differences between the two species.
This species is known from a few caves in Pulaski County, southeastern Kentucky.
USA: KY
This species is known from two nearby caves in Putnam and Van Buren Counties, Tennessee.
USA: TN
One polymorphic cave-inhabiting species in the Appalachians.
Barr (1979a) revised the species and provided a key for the identification of its subspecies.
This subspecies is known from several caves in Breckinridge, Hardin, Hart, and Meade Counties in northwest and central Kentucky (Barr 1979a: 8).
USA: KY
This subspecies has been found in several caves in Allen, Simpson, Warren, and Logan Counties in southern Kentucky (Barr 1979a: 9).
USA: KY
This subspecies has been collected in several caves in Allen, Barren, Edmonson, Hart, and Warren Counties in central and southern Kentucky (Barr 1979a: 6).
USA: KY
Intergrades between this subspecies and the meridionalis form occur in a narrow zone in Warren and Allen Counties (Barr 1979a: 10) and between this subspecies and the viator form in eastern Hart County (Barr 1979a: 8).
This subspecies is known from several caves in Green, Hart, and Metcalfe Counties in central and southern Kentucky (Barr 1979a: 8).
USA: KY
Two Palaearctic species, one of them adventive in North America.
The species found in North America was covered in Lindroth’s (1961a: 194) monograph.
This Palaearctic subspecies is adventive in North America where it is known from Nova Scotia (NSMC) to Wisconsin (Messer 2010: 35), south to southern Pennsylvania (Bradford and Allegheny Counties, CMNH). The first inventoried specimen collected on this continent was found in the Montreal area in 1933 (Brown 1940a: 69).
CAN: NB, NS, ON, PE, QC USA: CT, MA, ME, MI, NH, NY, OH, PA, VT, WI – Adventive
The subspecies Blemus discus orientalis Jeannel is known from Yunnan, China.
Two troglobitic species restricted to southeastern Missouri.
Barr and Krekeler (1967) provided a description of the external structures and male genitalia of both species.
This species is known from two nearby caves, Friedman’s and Pleasant Valley Caves, in eastern Missouri.
USA: MO
This species is known from two nearby caves, Kohm’s and Sims Caves, in eastern Missouri.
USA: MO
One cave-inhabiting species in Kentucky.
Valentine (1952) lengthy described, neatly illustrated, and discussed the structural differences between members of Darlingtonea and those of the other troglobitic genera of the Appalachian region.
This species is known from several caves in Kentucky located as far north as Estill County and as far south as Fentress County [see Marsh 1969: Fig. 5].
USA: KY
One cave-inhabiting species in Kentucky.
Valentine (1952: 24-29) described, illustrated, and discussed the status of the species.
This subspecies is known from several caves in the Somerset area, southeastern Kentucky.
USA: KY
This subspecies is known only from the type-locality cave in southeastern Kentucky.
USA: KY
About 870 species arrayed in eight subgenera: Arabotrechus Mateu (one Yemenite species), Atlantotrechus Lompe (one species in the Madeira Islands), Elgonophyes Jeannel (one Afrotropical species), Elgonotrechus Jeannel (14 Afrotropical species), Meruitrechus Jeannel (two Afrotropical species), Microtrechus (41 species), Minitrechus Vigna Taglianti and Magrini (one species in Ethiopia), and Trechus s.str. (about 775 species). More than 92% of the species are found in the Palaearctic Region.
About 775 species in North America (23 species, of which three are adventive), mountains in Mexico (about five species), Oriental (two species in the Philippines), Palaearctic (about 725 species), and Afrotropical (about 25 species on Mont Elgon, Mont Meru, and Ethiopia) Regions. More than 95% of the species inhabit the Northern Hemisphere.
There is no published key for the identification of the species of this subgenus.
[chalybeus group]
This species ranges from Newfoundland (Lindroth 1955a: 78-79, as Trechus apicalis micans) to Alaska, south to southeastern British Columbia (Lindroth 1963b: 202), southern Colorado (Elias 1987: 632; Mineral County, UASM) along the Rocky Mountains, and northeastern West Virginia (Tucker County, CMNH). Also found in the Far East and on Hokkaidō, Japan (Moravec et al. 2003: 326).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, CT, IN, MA, ME, MI, MN, MT, NH, NJ, NY, OH, PA, RI, VT, WI, WV – Holarctic
Trechus apicalis Motschulsky. This trechine species is “wing dimorphic” with the vast majority of individuals being micropterous (with short wing vestiges) and a few macropterous (with long wings). Usually macropterous individuals of dimorphic species are able to fly but this is not always the case as flight muscles could be atrophied. Carl Lindroth argued that in stable periods, when the species’ habitat is not subject to drastic changes, the brachypterous form normally predominates but in unstable periods, the situation is reversed.
This species ranges from the Aleutian Islands in Alaska (Lindroth 1961a: 198) south to the Sierra Nevada in eastern California (Inyo and Tulare Counties, CAS; Casey 1918: 407, as Trechus tahoensis; Dajoz 1990: 158) and to New Mexico along the Rocky Mountains (Snow 1885: 67; Fall and Cockerell 1907: 158).
CAN: AB, BC (QCI, VCI) USA: AK, CA, CO, ID, MT, NM, NV, OR, WA, WY
This species ranges from northwestern Washington (Hatch 1951: 113, as Trechus pugetensis) and northern Idaho (Lindroth 1963b: 201) south to northern New Mexico (Dajoz 1990: 158; Santa Fe County, CMNH), northeastern Arizona (Donabauer 2010a: 41), and northwestern California (Humboldt County, James R. LaBonte pers. comm. 2008).
USA: AZ, CA, CO, ID, NM, UT, WA
This species ranges from Newfoundland and the coast of Labrador to northeastern Minnesota (Gandhi et al. 2005: 924), south to mountains in northern New York and New England [see Lindroth 1963a: Fig. 69]. The record from “Massachusetts” (Bousquet and Larochelle 1993: 120) is in error.
CAN: LB, NB, NF, NS (CBI), ON, QC USA: ME, MN, NH, NY, VT, WI
This species inhabits the North American Cordilleras ranging from British Columbia and southwestern Alberta (Lindroth 1963b: 201) south to western Montana (Russell 1968: 48) and the Sierra Nevada in California (Lindroth 1963b: 201). The record from “Colorado” (Bousquet and Larochelle 1993: 121) needs confirmation.
CAN: AB, BC USA: CA, ID, MT, OR, WA [CO]
This species ranges from southern Yukon Territory (Lindroth 1961a: 200) south to mountains in northwestern Montana (Edwards 1975: 51), west-central Idaho (Boise County, CMNH), and western Oregon (Lane County, Foster F. Purrington pers. comm. 2009). The record from “Northwestern Territories” (Bousquet and Larochelle 1993: 121) needs confirmation.
CAN: AB, BC (VCI), YT USA: ID, MT, OR, WA [NT]
This species is known only from Apache County (Donabauer 2010a: 41) in northeastern Arizona.
USA: AZ
[hydropicus group]
This species is known only from the type locality in the Bald Mountains between Greeneville, Tennessee and Asheville, North Carolina (Barr 1985b: 128).
USA: TN/NC
This species is known only from the type locality, at the summit of Mount Mitchell where it is found in deep spruce and fir needle duff.
USA: NC
This species is found in caves in the Cumberland Plateau, from Rockcastle County in southeastern Kentucky southwest to Grundy County in southern Tennessee (Barr 1979b: 37).
USA: KY, TN
This subspecies is known from Grandfather Mountain in Avery County and Three Top Mountain in Ashe County in western North Carolina (Barr 1979b: 43).
USA: NC
This subspecies intergrades with the beutenmuelleri form on Beech Mountain, Avery County, North Carolina (Barr 1979b: 43).
This subspecies ranges southwestwards from the Roan Mountain in Tennessee to the Black and Great Craggy Mountains at the edge of the Blue Ridge in western North Carolina (Barr 1979b: 44).
USA: NC, TN
This subspecies is known from Grayson, Washington, and Lee Counties, southwestern Virginia, and Harlan and Letcher Counties, southeastern Kentucky (Barr 1979b: 36).
USA: KY, VA
This subspecies is known from western Maryland, Virginia, and eastern West Virginia (Barr 1979b: 36). The record from Sassafras Mountain in northwestern South Carolina (Ciegler 2000: 44) is apparently in error (see Ciegler 2003: [1]) as well as the state record of “South Carolina” by Bousquet and Larochelle (1993: 121).
USA: MD, VA, WV
This species is found in the Black Mountains in Yancey, Buncombe, and McDowell Counties, North Carolina (Barr 1979b: 37).
USA: NC
This species is known from Carter County in northeastern Tennessee and from Mitchell County in western North Carolina (Barr 1979b: 37).
USA: NC, TN
This subspecies is known from several specimens collected at the type locality in southwestern North Carolina.
USA: NC
This subspecies is restricted to the Pishah Ledge, which is the eastern arm of the Great Balsam Mountains, in western North Carolina (Barr 1979b: 45).
USA: NC
This subspecies is known from the Black and Great Craggy mountains and adjacent Blue Ridge, in Buncombe, McDowell, and Yancey Counties, western North Carolina (Barr 1979b: 46).
USA: NC
[ovipennis group]
This species is known only from the San Jacinto Mountains in southeastern California.
USA: CA
This species is known only from the holotype.
USA: AZ
This species is endemic to the Pinaleno Mountains in Graham County, southeastern Arizona (Donabauer 2010a: 39).
USA: AZ
This species is known only from the holotype.
USA: CA
This species is known from northwestern California (Van Dyke 1945b: 101) and southern Oregon (Lane County, CMNH).
USA: CA, OR
This species ranges along the Pacific Coast from southeastern Alaska (Lindroth 1961a: 196) to at least Marin County, central California (Kavanaugh and Erwin 1985: 177).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is known only from southwestern California (Jeannel 1927: 189; Donabauer 2010a: 39).
USA: CA
[quadristriatus group]
This European species is adventive in North America where it is known from the Queen Charlotte Islands (Kavanaugh 1992: 61) to north-central Idaho (LaBonte 1989: 17; Hatten et al. 2007: 359), south to northern Utah (Davis and Salt Lake Counties, CMNH) and west-central California [see Kavanaugh and Erwin 1985: Fig. 1]. The first inventoried specimen collected on this continent was found in North Creek, King County, Washington in 1925 (Kavanaugh and Erwin 1985: 171). The species is also adventive in Hawaii since 1998 (Liebherr and Takumi 2003).
CAN: BC (QCI, VCI) USA: CA, ID, OR, UT, WA – Adventive
This European species is adventive in North America where it is known from Nova Scotia (Majka et al. 2006: 603) to northern Wisconsin (Iron County, CMNH; Messer 2010: 35), as far north as the Abitibi region in Quebec (Paquin and Dupérré 2002: 87), south to northeastern West Virginia (Hampshire County, CMNH) and eastern Maryland (Queen Annes County, Foster F. Purrington pers. comm. 2009). The first inventoried specimen collected on this continent was found at Port Credit, southern Ontario, in 1965 (Bousquet et al. 1984: 215).
CAN: NS, ON, QC USA: MD, MI, NY, PA, WI, WV – Adventive
[rubens group]
This European species is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 82; Larson and Langor 1982: 593) to western Quebec (Larochelle 1975: 112), and from the New England area (Bousquet and Larochelle 1993: 121). The first inventoried specimen collected on this continent was found prior to 1863 (LeConte 1863b: 14) probably in Nova Scotia as recorded by Horn (1875: 131). The record from eastern Ontario (Hamilton 1889b: 94) is probably in error.
FRA: PM CAN: NB, NF, NS (CBI), PE, QC USA: ME, NH, VT – Adventive
Forty-one species (46 species-group taxa) restricted to the Appalachian Mountains in North Carolina, Tennessee, South Carolina, and Georgia and currently placed in three species groups.
Barr (1979b) revised and provided a key to the species of this subgenus (22 species, 29 species-group taxa). Subsequently, 15 new species and two subspecies have been described by Barr (1985b), Dajoz (2005), and Donabauer (2005a, b).
[nebulosus group]
This species is known only from the Plott Balsam Mountains, western North Carolina.
USA: NC
This species is known only from the type locality.
USA: NC
This species is known only from the type locality.
USA: NC/TN
This species is known only from the type locality.
USA: NC/TN
This subspecies is known only from the type locality.
USA: NC
This subspecies is known only from the type locality.
USA: NC/TN
This subspecies is known from the central Great Smoky Mountains in North Carolina and Tennessee (Barr 1979b: 40).
USA: NC, TN
This subspecies is endemic to the Nantahala Mountains in southwestern North Carolina (Donabauer 2005b: 88).
USA: NC
This species is known only from the type locality in the northern Nantahala Mountains, southwestern North Carolina.
USA: NC
This species is found in central and eastern Great Smoky Mountains in North Carolina and Tennessee (Barr 1979b: 40).
USA: NC, TN
This species is found in central Great Smoky Mountains in North Carolina and Tennessee (Barr 1979b: 40).
USA: NC, TN
This species is known only from the type locality.
USA: NC/TN
This species is known only from the type locality.
USA: TN
This species is known from the Plott Balsam Mountains and Great Balsam Mountains in western North Carolina (Barr 1979b: 40).
USA: NC
This species is known only from the type locality.
USA: NC
This species is known only from the type locality.
USA: NC/TN
This species is known only from the holotype collected in eastern Tennessee.
USA: TN
This subspecies is known only from Bull Cave in the Cades Cove Mountains, Great Smoky Mountains, in eastern Tennessee (Barr 1979b: 40).
USA: TN
This subspecies is known only from the original 28 specimens collected in a cave in the Appalachian Valley, 8 miles south of Kingston and ¼ mile west of the Tennessee River on the southeast side of a valley east of Huckleberry Ridge (Barr 1962a: 87).
USA: TN
This species is known only from the holotype.
USA: NC/TN
This species is known only from the type locality in the Nantahala Mountains.
USA: NC
This species is known only from the original specimens collected in a large stream cavern at the north side of the Great Smoky Mountains.
USA: TN
This species is endemic to the Unicoi Mountains in southeastern Tennessee and western North Carolina (Barr 1979b: 40).
USA: NC, TN
This taxon was first described as a subspecies but raised to species by Donabauer (2005b: 87).
This species is found in the central Great Smoky Mountains in Tennessee and North Carolina (Barr 1979b: 40).
USA: NC, TN
This species is known only from the conifer forests in the eastern end of the Great Smoky Mountains in Haywood County, western North Carolina, and Cocke County, eastern Tennessee.
USA: NC, TN
This species is known only from the type locality.
USA: NC
[uncifer group]
This species occurs in the Great Balsam Mountains in western North Carolina (Barr 1979b: 39).
USA: NC
This species is known only from the Cowee Mountains in southwestern North Carolina (Donabauer 2005a: 56).
USA: NC
This species is known only from Big Butt in the Nantahala Mountains, southwestern North Carolina (Donabauer 2005a: 57).
USA: NC
This species is known only from the type locality.
USA: TN/NC
This species is known only from the type locality in the Plott Balsam Mountains, southwestern North Carolina.
USA: NC
This species is endemic to the western Pisgah Ridge (Donabauer 2005a: 57) in the Great Balsam Mountains.
USA: NC
This species has been found yet only in the Unicoi Mountains, between 4800-5000 feet, in North Carolina and Tennessee (Barr 1979b: 40).
USA: NC, TN
This species is known only from Thunderhead Mountain at the junction of Swain County in North Carolina and Blount County in Tennessee.
USA: NC/TN
This species is known only from the Toxaway Mountain in southwestern North Carolina.
USA: NC
This species is known only from the type locality in southwestern North Carolina.
USA: NC
This species is known from the central Great Smoky Mountains east to Plott Balsam Mountains in Sevier and Cocke Counties, eastern Tennessee, and in Haywood and Jackson Counties, western North Carolina (Barr 1979b: 38).
USA: NC, TN
[vandykei group]
The range of this common species extends from the vicinity of Asheville, western North Carolina, to the mountains of northeast Georgia (Barr 1979b: 38) and northwestern South Carolina (Ciegler 2000: 44).
USA: GA, NC, SC, TN
This species is found in the spruce-fir forests, between 3000-6500 feet, in the Great Smoky Mountains of North Carolina and Tennessee (Barr 1979b: 38; Donabauer 2009: 137).
USA: NC, TN
This species is known only from the Unicoi Mountains, along the Tennessee-North Carolina border (Donabauer 2009: 138).
USA: NC
This species is known only from two localities in western North Carolina (Donabauer 2009: 136).
USA: NC
This species is known only from the type locality.
USA: NC/TN
This species is known only from the type locality in southwestern North Carolina.
USA: NC
This species has been found at several locations in the Great Smoky Mountains and the Plott Balsams in western North Carolina (Barr 1979b: 38; Donabauer 2009: 131).
USA: NC
This species is known only from the type locality in the western Great Smoky Mountains on the border between Blount County, Tennessee, and Swain County, North Carolina.
USA: TN
This species is known at present from small mountain ranges in southwestern North Carolina (Barr 1979b: 37).
USA: NC
This subspecies is found in the Great Balsam and Cowee Mountains in western North Carolina (Barr 1979b: 37).
USA: NC
This subspecies is found in the Bald and Unaka Mountains of Tennessee and in the Black and Great Craggy Mountains of North Carolina (Barr 1979b: 37; Donabauer 2009: 138). Populations very similar to those of Trechus vandykei have been reported by Donabauer (2009: 138) in the Great Smoky Mountains.
USA: NC, TN
Worldwide, with about 2, 630 species arrayed in six subtribes: Anillina (about 375 species), Bembidiina (about 1, 340 species), Horologionina (one species), Lovriciina (four species), Tachyina (about 790 species), and Xystosomina (about 125 species). The Northern Hemisphere contains roughly 55.5% of the world fauna and North America alone 14.8% (about 390 species).
Worldwide, with about 1, 350 species. The number of genera admitted varies greatly depending on the authors. In this work, the species are arrayed in nine genera following Maddison (2012: 570): Amerizus Chaudoir, Asaphidion des Gozis, Bembidion Latreille, Caecidium Uéno, Lionepha Casey, Ocys Gistel, Orzolina Machado, Sakagutia Uéno, and Sinechostictus Motschulsky (including Pseudolimnaeum Kraatz). The majority of the species are found in the Northern Hemisphere (roughly 75.5% of the world fauna).
About 50 species in North America (five species) and Asia (about 45 species) placed in two subgenera: Tiruka Andrewes for most Asian species and Amerizus for the Nearctic species and one Asian species.
Toledano (2011) noted that Amerizus is closely related to the genus Caecidium Uéno (two species in Japan). Lindroth (1980: 203) pointed out that Bembidion (Gnatholymnaeum) blackburni (Sharp) from Hawaii is structurally similar to the species of this genus and indeed listed the species within the subgenus Amerizus. However, Liebherr (2008: 36) argued against such association. Both Caecidium and Bembidion blackburni were not sequenced by Maddison (2012).
Six species in North America (five species) and the Altai Mountains in Russia (Amerizus teles Belousov and Dudko, 2010).
Lindroth (1963b: 403-406) treated four of the five North American species, leaving Amerizus utahensis.
This species is restricted to the Queen Charlotte Islands and adjacent mainland (Kavanaugh 1992: 69).
CAN: BC (QCI)
This species ranges from the Kenai Peninsula in Alaska (Lindroth 1963b: 405) south to “California” (Hayward 1897: 131) and to the Sangre de Cristo Mountains in northeastern New Mexico (Ball 1966b: 30). The record from “Mexico” (Hayward 1897: 131) is probably in error.
CAN: BC (QCI) USA: AK, CA, ID, NM, OR, WA
The range of this species extends from the Alexander Archipelago (Mannerheim 1852: 298; Lindroth 1963b: 404) south at least to the Santa Cruz Mountains of the Coast Ranges (Casey 1918: 166, as Amerizus longicornis) and the Sierra Nevada in California (Kavanaugh 1992: 68).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is known only from Utah.
USA: UT
This species was originally placed in the subgenus Lymneops Casey but subsequently transferred to Amerizus Chaudoir by Van Dyke (1949b: 56).
This eastern species occurs from Newfoundland (Lindroth 1955a: 78, as Bembidium oblongulum) to northern Minnesota (Petrice et al. 2002: 9; Gandhi et al. 2005: 925), south to Tennessee (Carter County, CMNH) and North Carolina (Lindroth 1963b: 406; Mitchell and Yancey Counties, CMNH) along the Appalachian Mountains.
FRA: PM CAN: LB, NB, NF, NS (CBI), ON, PE, QC USA: IL, IN, KY, MA, ME, MI, MN, NC, NH, NY, OH, PA, TN, VA, VT, WI, WV
This species has long been confused with Amerizus oblongulus Mannerheim, under that name, following Horn (1875: 131).
Nine species in western North America.
Erwin and Kavanaugh (1981) revised the species and provided a key for their identification.
[casta group]
This species ranges from the southern part of the Alexander Archipelago, Alaska (Lindroth 1963b: 261) and west-central British Columbia south to central California along the coast mountain systems [see Erwin and Kavanaugh 1981: Fig. 23].
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
[erasa group]
This species is known from several mountains in the Coast Ranges of Oregon (David R. Maddison pers. comm. 2012).
USA: OR
This species is known from four localities, the northernmost in southern British Columbia and the southernmost in the mid-Sierra Nevada [see Erwin and Kavanaugh 1981: Fig. 24].
CAN: BC USA: CA, OR
This species ranges from southwestern Alberta and southeastern British Columbia south to the San Jacinto Mountains in southern California, including the Sierra Nevada and Cascade Range, and northern Colorado along the Rocky Mountains [see Erwin and Kavanaugh 1981: Fig. 25].
CAN: AB, BC USA: CA, CO, ID, MT, NV, OR, WA, WY
This species is known from a few localities along the Pacific Coast from southern Alaska near the Yukon Territory border to northern Washington [see Erwin and Kavanaugh 1981: Fig. 26].
CAN: BC (VCI) USA: AK, WA
This species corresponds to Bembidion brumale Casey sensu Lindroth (1963b: 262).
This species is known from two localities, one on the eastern edge of the Central Plateau in eastern British Columbia, the other on an island on Puget Sound, Washington [see Erwin and Kavanaugh 1981: Fig. 27].
CAN: BC USA: WA
[osculans group]
This species ranges from the lower Columbia River drainage in northwestern Idaho and southern Washington south to the Sierra Nevada and Coast Ranges in central California [see Erwin and Kavanaugh 1981: Fig. 20].
USA: CA, ID, OR, WA
This species is endemic to the Sierra Nevada in California from Nevada County to Sequoia National Park [see Erwin and Kavanaugh 1981: Fig. 21].
USA: CA
This species ranges along the Cascade Range and Sierra Nevada from southern British Columbia to central California [see Erwin and Kavanaugh 1981: Fig. 22].
CAN: BC USA: CA, OR, WA
Northern Hemisphere, with 39 species in the arctic, subarctic, boreal, and temperate areas of the Nearctic (three species, one of them adventive) and Palaearctic (37 species) Regions.
Lindroth (1963b: 203-206) covered all three species found in North America, the adventive Asaphidion curtum under the name Asaphidion flavipes (Linnaeus).
This species is found from Alaska to northwestern Northwest Territories (Lindroth 1963b: 204). Fossil remnants of this species, believed to be 2.0-2.5 million years old, have been found in Greenland and Meighen Island (Bennike and Böcher 1990: 336; Böcher 1995: 23).
CAN: NT, YT USA: AK
This European and North African subspecies is adventive in North America where it is known from Maine (Larochelle and Larivière 1990a: 28, 33, as Asaphidion flavipes) and southeastern New Hampshire (Bell 1989b: 204, as Asaphidion flavipes) to Long Island, New York (Davidson and Langworthy 1981: 280, as Asaphidion flavipes). The first inventoried specimens collected on this continent were found in the late 1920s in Long Island (Cooper 1930: 21, as Asaphidion flavipes), the next ones were found in 1976 also on Long Island (Davidson and Langworthy 1981: 280).
USA: CT, MA, ME, NH, NY, RI – Adventive
Two other subspecies, one from Morocco (Asaphidion curtum moroccanum Antoine) and the other from the Canary Islands (Asaphidion curtum delatorrei Uyttenboogaart), are recognized.
Asaphidion curtum curtum (Heyden). This adventive bembidiine was reported in the North American literature under the name Asaphidion flavipes (Linnaeus) until a study of the male genitalia showed that the specimens were in fact conspecific with the morphologically similar Asaphidion curtum. Asaphidion species are odd-looking bembidiines and Linnaeus originally associated them with tiger beetles probably because of their large eyes and absence of elytral striae.
This species is known from Alaska, Yukon Territory, northeastern British Columbia, and west-central Alberta [see Morgan and Morgan 1979: Fig. 4; Morgan and Morgan 1981: map 4]. Fossil remnants of this species from the Pleistocene and early Holocene have been found in west-central Illinois, northeastern Wisconsin, Vermont, southern Ontario, and northwestern Ontario (see Ashworth and Schwert 1991: 512; Bajc et al. 1997: 691).
CAN: AB, BC, YT USA: AK
Worldwide, with about 1, 250 species (Lorenz 2005: 215-236, as Bembidiina excluding Phrypeus, Amerizus, Lionepha, and Asaphidion) arrayed in about 105 subgenera. The Northern Hemisphere includes roughly 75% of the species and the Westhern Hemisphere approximately 30% of the species. The North American fauna alone is represented by 253 species (about 20% of the world fauna) placed in 37 subgenera. Eight of the North American species are adventive and 20 species are Holarctic.
Hayward (1897) reviewed the North American species but his work is now outdated. Lindroth (1963b) covered 191 (six of them in the key alone) of the 253 species found in North America (about 75% of the fauna).
Maddison (2012: 570) presented an entirely new classification of this genus based on molecular data analyses. He found support for monophyly of Bembidion exclusive of the subgenus Phyla; however, he retained the taxon in the genus pending further study. He recognized three series, the Odontium, Ocydromus, and Bembidion Series with the following subgenera represented in North America left unplaced: Blepharoplataphus, Plataphus (including Plataphodes), Hydrium (including Eurytrachelus), Metallina, Lindrochthus, Eupetedromus, Trechonepha, Liocosmius, Melomalus, Trichoplataphus, Phyla, and Lymnaeum. The North American taxa in the Odontium Series were placed in two complexes, the Hydriomicrus (Hirmoplataphus and Hydriomicrus) and Odontium (Odontium, Bracteon, Ochthedromus, and Pseudoperyphus) Complexes. The North American taxa in the Ocydromus Series were placed in two complexes, the Princidium (Cillenus and Actedium) and Ocydromus (Ocydromus, Peryphus, Terminophanes, Asioperyphus, Peryphanes, Testediolum, and Leuchydrium) Complexes. The North American taxa in the Bembidion Series were placed in three complexes, the Bembidion (Bembidion s.str. and Cyclolopha), Furcacampa (Furcacampa and Neobembidion) and Diplocampa (Diplocampa and Semicampa) Complexes with Notaphus, Trepanedoris, Peryphodes, and Emphanes left unplaced.
Edwards (1975: 53) recorded a “Bembidion nigricornis Hayward” from Glacier National Park in Montana. I was unable to find any Bembidion of that name or with a similar name described by Hayward or any other North American authors.
Northern Hemisphere, with 11 species in the Nearctic (nine species) and Palaearctic (two species: Bembidium friebi Netolitzky and Bembidium hirmocaelum Chaudoir) Regions.
There is no taxonomic revision of the North American species and such study is needed. Lindroth (1963b: 301-305) covered all but three (Bembidium alpineanum, Bembidium avidum, and Bembidium subaerarium) species described by Casey.
This species is known only from the holotype collected in eastern California.
USA: CA
According to Erwin (1984a: 167), this species “is found in the Basin and Range Province of the western United States.”
USA: NV
This species ranges from Newfoundland (Lindroth 1955a: 56, as Bembidion longulum) to eastern Alaska (Lindroth 1963b: 304), south to northern Oregon (Gilliam and Morrow Counties, CMNH), central Nevada (Eureka County, Ken Karns pers. comm. 2009), northern Arizona (Coconino County, Ken Karns pers. comm. 2009), central New Mexico (Fall and Cockerell 1907: 157; Casey 1918: 54, as Bembidion longulum), eastern South Dakota (Kirk and Balsbaugh 1975: 17), the upper peninsula of Michigan (LeConte 1847: 456, as Bembidion longulum), and northeastern New York (Casey 1918: 51). The record from “California” (Lindroth 1955a: 56) needs confirmation (see Lindroth 1963b: 304); those from “New Hampshire” and “Maine” (Bousquet and Larochelle 1993: 133) are probably in error (Ross T. Bell pers. comm. 2008).
CAN: AB, BC (VCI), MB, NB, NF, NS, NT, ON, QC, SK, YT USA: AK, AZ, CO, ID, MI, MT, ND, NM, NV, NY, OR, SD, UT, WA, WI, WY [CA]
As far as known, this species is confined to the west coast of southern Oregon (Hatch 1953: 88, as Bembidion chetcoens) and northern California (Blaisdell 1902: 74; Casey 1918: 55). The record from the central Sierra Nevada (Papp 1978: 164) needs confirmation.
USA: CA, OR
The range of this eastern species extends from Cape Breton Island (Lindroth 1954c: 302) to eastern South Dakota (Kirk and Balsbaugh 1975: 17), south to east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), southwestern Mississippi (Drew A. Hildebrandt pers. comm. 2007), and northern Georgia (Fattig 1949: 17). The record from “Kansas” (Bousquet and Larochelle 1993: 134) needs confirmation.
CAN: NB, NS (CBI), ON, PE, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, PA, SD, TN, VA, VT, WI, WV [KS]
The range of this western species extends from southern Yukon Territory (Lindroth 1963b: 304) south to the Sierra Nevada in California (Casey 1918: 50, as Bembidion callidum), northern Arizona (Navajo County, CNC), and northern New Mexico (Casey 1918: 55, as Bembidion porrectum) along the Rocky Mountains.
CAN: AB, BC (VCI), YT USA: AZ, CA, CO, ID, MT, NM, OR, UT, WA, WY
This western species is found from south-central Alberta to Vancouver Island, north to the Skeena River drainage in central British Columbia (Lindroth 1963b: 305), south at least to central California in the Coast Ranges (Casey 1918: 53, as Bembidion tetragonoderum) and “New Mexico” (Hayward 1897: 61). The record from southeastern Wisconsin (Messer 2010: 36) needs confirmation since the specimen could be mislabeled.
CAN: AB, BC (VCI) USA: AZ, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY [WI]
This species ranges from Newfoundland (Lindroth 1955a: 56) to the foothills of the Rockies in Alberta and northeastern British Columbia (Lindroth 1963b: 302), south to southeastern Wyoming (Hayward 1897: 60, as Bembidion concolor), central Minnesota (Crow Wing and Sherburne Counties, CNC), Vermont (Addison County, CNC), and Maine (Aroostook County, CNC). The record from southern South Dakota (Kirk and Balsbaugh 1975: 17) needs confirmation; those from “Idaho, ” “Iowa, ” and “Pennsylvania” (Bousquet and Larochelle 1993: 134) are probably in error. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 25).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, QC, SK USA: ME, MI, MN, MT, ND, NH, NY, VT, WI, WY [SD]
This species has passed under the name Bembidion concolor (Kirby, 1837) until Lindroth (1963b).
This species is known only from the type locality in the Sierra Nevada in California.
USA: CA
Five North American species, two in the east and three in the west.
Lindroth’s (1963b: 212-229) key to Bembidion included all but one species (Bembidion innocuum); the two species found in Canada were treated in detail (Lindroth 1963b: 306).
Based on molecular data analyses, Maddison (2012: 568) concluded that this subgenus is closely related to Hirmoplataphus.
This species is known from southwestern Oregon (Hatch 1953: 88) to southern California “in the higher mountain cañons or valleys between the ranges” (Fall 1901a: 42).
USA: CA, OR
This species is known from southwestern Oregon (Josephine County, James R. LaBonte pers. comm. 1992) to the southern part of California where it is “widely distributed” (Fall 1901a: 42). The record from northwestern Montana (Edwards 1975: 51) needs confirmation.
USA: CA, OR [MT]
This species is known only from Del Norte (Maddison 2012: Supplementary content Table S1), Marin (Casey 1924: 29, as Bembidion marinianum) and Humboldt (Casey 1918: 63) Counties in coastal California.
USA: CA
This species ranges from Newfoundland (Lindroth 1955a: 50) to the Laurentides region in Quebec (Larochelle 1975: 60), south to southwestern (Hardin County, CMNH) and eastern Tennessee (Johnson, Morgan, Roane, and Union Counties, CMNH).
CAN: NB, NF, NS (CBI), QC USA: CT, ME, NH, NY, TN, VT
The range of this eastern species extends from Nova Scotia (Lindroth 1954c: 302) to west-central Indiana (Blatchley 1910: 79), including southeastern Michigan (Wayne County, CMNH), south to northwestern Arkansas (Newton County, CNC, UASM), northeastern Mississippi (Tishomingo County, Drew A. Hildebrandt pers. comm. 2010), and southern Georgia (Torres and Ruberson 2006: 31).
CAN: NB, NS, ON, QC USA: AR, CT, DC, GA, IN, KY, MA, MD, ME, MI, MS, NC, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WV
Twenty-three species in the Nearctic (ten species), Neotropical (one species also present in southern Arizona), Palaearctic (12 species), and Oriental (one species, Bembidion subfusum Darlington, in the Philippines) Regions.
Lindroth (1963b: 241-246, as bowditchi and coxendix groups) treated all the species found in North America, Bembidion durangoense under the name Bembidion arizonae. One species, Bembidion paraenulum, was subsequently described.
This species is known from eastern South Dakota to southwestern Nebraska, east to eastern Iowa [see Maddison and Arnold 2009: Fig. 9]. Two specimens simply labeled “Missouri” and “Wisconsin” (Maddison and Arnold 2009: 60) are known.
USA: IA, NE, SD [MO, WI]
This species is known from a few localities in southern British Columbia (Lindroth 1963b: 242), northwestern Washington (Maddison 2012: Supplementary content Table S1), “Idaho” (Maddison 1993: 161), western Montana (Russell 1968: 49), and southwestern Wyoming (Hayward 1897: 51).
CAN: BC USA: ID, MT, WA, WY
This species is known from a few localities in southeastern California and southern Arizona (Lindroth 1963b: 245; Dajoz 2007: 19).
USA: AZ, CA
This species is found east of the Rocky Mountains from New Brunswick (Queens County, Reginald P. Webster pers. comm. 2008) to southeastern Alberta (Lindroth 1963b: 245), south to southeastern Colorado (Maddison 1985: 111), northwestern (Jones County, CNC) and east-central (Riley 2011) Texas, and central Florida (Peck and Thomas 1998: 18).
CAN: AB, MB, NB, ON, QC, SK USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI
The range of this species extends from southwestern and north-central Pennsylvania (Westmoreland and Lycoming Counties, Robert L. Davidson pers. comm. 2008) to Alberta (Lindroth 1963b: 244), south to central New Mexico (Fall and Cockerell 1907: 157; Socorro County, UASM), southern Texas (Snow 1906a: 141; Casey 1918: 12, as Bembidion unicum; Cameron and Victoria Counties, MCZ, UASM), northeastern Louisiana (Franklin Parish, Igor M. Sokolov pers. comm. 2009), northwestern Mississippi (Bolivar County, Foster F. Purrington pers. comm. 2012), and southwestern Georgia (Fattig 1949: 16). The records from “New Hampshire, ” “Vermont, ” “Idaho, ” and “Northwest Territories” (Bousquet and Larochelle 1993: 126) are in error; that from Connecticut (Krinsky and Oliver 2001: 70) was based on misidentified specimens of Bembidion confusum (William L. Krinsky pers. comm. 2009).
CAN: AB, MB, SK USA: AR, CO, DC, GA, IA, IL, IN, KS, LA, MI, MN, MO, MS, MT, NC, ND, NE, NM, OH, OK, PA, SC, SD, TN, TX, WI, WY
This species is known from southern Arizona (Lindroth 1963b: 246; Dajoz 2007: 21) and northern Mexico (Bates 1891a: 263).
USA: AZ – Mexico
This species is known from the Gila River drainage in southern Arizona.
USA: AZ
This taxon has been listed in synonymy with Bembidion durangoense Bates by Erwin (1984a: 171) but according to David R. Maddison (pers. comm. 2007) it represents a distinct species.
This species ranges from southern New Hampshire south to northern Florida [see Maddison and Arnold 2009: Fig. 9], west to eastern Texas (Hardin County, Robert L. Davidson pers. comm. 2012), including eastern Louisiana (East Feliciana Parish, Igor M. Sokolov pers. comm. 2009).
USA: AL, FL, GA, LA, MS, NC, NH, TX, VA
This species is known from a few scattered localities from Connecticut (Lindroth 1963b: 243; Krinsky and Oliver 2001: 82) and northern Vermont (Lamoille County, Ross T. Bell pers. comm. 2008) to western North Dakota (McKenzie County, Donald P. Schwert pers. comm. 1989), south at least to “Kansas” (Hayward 1897: 50) and “Kentucky” (Lindroth 1963b: 243).
USA: CT, IA, IL, KS, KY, MI, MO, ND, VT, WI
This species is known from eastern Washington (Hatch 1953: 80) to central California (Lindroth 1963b: 245).
USA: CA, OR, WA
Northern Hemisphere, with 17 species in the Nearctic (11 species) and Palaearctic (nine species) Regions. Three species are Holarctic.
Maddison (1993) revised the species of this subgenus and provided a key for their identification.
This Holarctic species is known from the region of Lake Baikal in Siberia and Alaska, from the Brooks Range south to the Alaska Peninsula [see Maddison 1993: Fig. 264]. Fossil remnants from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 24).
USA: AK – Holarctic
This species is known only from west-central Saskatchewan and eastern Alberta [see Maddison 1993: Fig. 261]. The record from Churchill, Manitoba (Elias 1984: 142) is based on misidentified Bembidion carinula (Maddison 1993: 174). Fossil remnants, dated about 13, 200 years B.P., have been unearthed in southern Ontario (Karrow et al. 2007: 290).
CAN: AB, SK
A common species ranging from Newfoundland to southwestern British Columbia, north to central Yukon Territory and the Ungava Bay in Quebec, south to northern Iowa and southwestern Pennsylvania [see Maddison 1993: Fig. 265]. The records from “Georgia, ” “Arkansas, ” “Oregon” and Vancouver Island (Hayward 1897: 46) are probably in error; that from “Kentucky” (Maddison 1993: Fig. 265) needs confirmation. Lindroth’s (1963b: 237) record from Washington is based on a misidentified Bembidion lapponicum (Maddison 1993: 182). Three specimens labeled “Or., ” “Tex, ” and from Pena Blanca in Arizona seen by Maddison (1993: 182) are probably mislabeled.
CAN: AB, BC, LB, MB, NB, NF, NT, NU, ON, QC, SK, YT USA: CT, IA, IL, IN, MA, ME, MI, MN, NH, NJ, NY, OH, PA, VT, WI [KY]
This Holarctic species ranges in the Palaearctic Region from northeastern Europe to Kamchatka, and in the Nearctic Region, from Alaska east to Churchill, Manitoba, and south to central Saskatchewan [see Maddison 1993: Fig. 262a].
CAN: BC, MB, NT, SK, YT USA: AK – Holarctic
Bembidion glabriusculum Motschulsky, 1844, listed as a questionable synonym of this species by Lindroth (1962: 16), is a junior synonym of Bembidion argenteolum Ahrens (Maddison 1993: 176).
This western species ranges from southeastern British Columbia to Vancouver Island, south to east-central California [see Maddison 1993: Fig. 269].
CAN: BC (VCI) USA: CA, OR, WA
This species ranges from Cape Breton Island to western Alaska, south to northwestern California, south-central Colorado, northern Kansas, southeastern Louisiana (East Baton Rouge Parish, Igor M. Sokolov pers. comm. 2009), northern Alabama, and northern Georgia [see Maddison 1993: Fig. 273]. The records from “Florida, ” “Arkansas, ” and “Texas” (Hayward 1897: 44) are probably in error.
CAN: AB, BC (QCI, VCI), MB, NB, NS (CBI), ON, PE, QC, SK, YT USA: AK, AL, CA, CO, CT, DC, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NV, NY, OH, OR, PA, SD, TN, UT, VA, VT, WA, WI, WV, WY
This Holarctic species ranges in the Palaearctic Region from northernmost Scandinavia to Kamchatka, south to Mongolian Peoples’ Republic. In North America, it occurs commonly in the northwest from Alaska to the Anderson River; it is also found at scattered localities in Washington, northern Oregon, Idaho, and Wyoming, and in western Canada east to central Saskatchewan [see Maddison 1993: Fig. 267a].
CAN: AB, BC, NT, SK, YT USA: AK, ID, OR, WA, WY – Holarctic
This transamerican subspecies ranges from western Newfoundland (Lindroth 1955a: 47) and Labrador to western Alaska, south to west-central British Columbia, southeastern New Mexico, southern Wisconsin, southwestern Michigan, central New York, and southeastern New Brunswick [see Maddison 1993: Fig. 272].
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, NU, ON, QC, SK, YT USA: AK, CO, ME, MI, MN, ND, NH, NM, NY, VT, WI
The range of this subspecies extends from central Washington to southwestern Alberta, south to northern New Mexico [see Maddison 1993: Fig. 272]. One specimen labeled “Cal.” is known (Maddison 1993: 191). Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland and Banks Island (Böcher 1995: 23).
CAN: AB, BC USA: CO, ID, MT, NM, WA, WY
This species has long been known in the North American literature under the name Bembidion lit[t]orale (Olivier).
This western species is found from southwestern British Columbia, including Vancouver Island, to western Idaho, south to southwestern California [see Maddison 1993: Fig. 270]. The records from Wyoming (LeConte 1878a: 465), Colorado (Wickham 1902: 232; Lindroth 1963b: 236), and “Montana” (Hayward 1897: 46) are probably in error.
CAN: BC (VCI) USA: CA, ID, NV, OR, WA
This species shows a disjunct distribution. In the east, it ranges from Nova Scotia (Majka et al. 2007: 8), Maine, and southern Quebec south to the District of Columbia and Kentucky; in the west it occurs from western North Dakota and Saskatchewan to southwestern British Columbia, north to northern Northwest Territories and southwestern Alaska (Elias 1988: 41) [see Maddison 1993: Fig. 268]. I have also seen one specimen from northwestern Colorado (Eagle County, CMNH). The records from “Missouri, ” “Arkansas” (Hayward 1897: 47), southwestern Iowa (Wickham 1911b: 6), “Washington” and “Oregon” (Hatch 1953: 81) need confirmation; that from “California” (Hayward 1897: 47) is probably in error.
CAN: AB, BC, NB, NS, NT, ON, QC, SK, YT USA: AK, CO, CT, DC, IN, KY, MA, ME, MI, MT, ND, NH, NJ, NY, OH, PA, VT, WV [AR, IA, MO, OR, WA]
This species ranges from the Queen Charlotte Islands (Kavanaugh 1992: 62) south to San Francisco, California; it is mainly found on the Pacific Coast though a few inland localities are also known [see Maddison 1993: Fig. 271]. The record from Mono County in eastern California (Dajoz 2007: 17) needs confirmation.
CAN: BC (QCI, VCI) USA: CA, OR, WA
Three North American species.
Lindroth (1963b: 249-250) recognized two species, Bembidium americanum and Bembidium bifossulatum, in this subgenus and treated both in his monograph. Maddison (1993: 160) showed that one of the species, Bembidium bifossulatum, consisted of two distinct forms which he treated as subspecies. Subsequently (Maddison 2012: 541) he listed them as distinct species.
This species ranges from southern New Brunswick (Webster and Bousquet 2008: 16) to southwestern Wisconsin (Richland County, Peter W. Messer pers. comm. 2008), south to eastern Texas (Sabine County, Brian Raber pers. comm. 2010; Riley 2011) and central Florida (Peck and Thomas 1998: 18). The record from “Minnesota” (Bousquet and Larochelle 1993: 126) needs confirmation.
CAN: NB, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV [MN]
This species ranges from southern British Columbia, including Vancouver Island (Lindroth 1963b: 250), south to southern California (LeConte 1852a: 186) and southwestern Utah (Casey 1918: 14, as Bembidion ferreum). The previous records from other states and provinces refer to Bembidion cheyennense.
CAN: BC (VCI) USA: CA, ID, NV, OR, UT, WA
If the lectotypes currently associated with the labels for Bembidion regestum and Bembidion sufflatum are the original ones, then both type localities are in error. However, both lectotypes have been dissected and remounted in the last decade and possibly the specimens were not properly reassociated with the labels.
The range of this species extends from Nova Scotia (Majka et al. 2007: 7) to the Rocky Mountains in Alberta (Lindroth 1963b: 250, as Bembidion bifossulatum), south to southern Arizona (Pima and Graham Counties, CMNH), Durango in Mexico (CNC), southeastern Missouri (Wayne County, CNC), northern Ohio (Lee 1994: 58, as Bembidion bifossulatum), and northwestern Pennsylvania (Erie County, CMNH). The record from “Arkansas” (Bousquet and Larochelle 1993: 127, as Bembidion bifossulatum) needs confirmation.
CAN: AB, MB, NS, ON, QC, SK USA: AZ, CO, IA, KS, MI, MN, MO, MT, ND, NE, NH, NM, OH, OK, PA, SD, TX, VT, WI, WY [AR] – Mexico
Nine North American species, one of them extending into northern Mexico.
Maddison (2008) revised the species and provided a key for their identification. Some of the species are very difficult to recognize on external characters and the male genitalia must be examined.
[chalceum group]
This species ranges from Nova Scotia to northwestern Wisconsin, south to northern Arkansas, central Mississippi (Madison County, Drew A. Hildebrandt pers. comm. 2008), northern Alabama, and central North Carolina [see Maddison 2008: Fig. 21].
CAN: NB, NS, ON, QC USA: AL, AR, DC, IA, IL, IN, KY, MA, MD, ME, MO, MS, NC, NH, NJ, NY, OH, PA, TN, VA, VT, WI, WV
This species ranges from New England and northeastern New York south to northeastern Kentucky [see Maddison 2008: Fig. 20A].
USA: KY, ME, NH, NY, OH, PA, VT, WV
The range of this species extends from Newfoundland to southwestern Northwest Territories, south to northeastern California, northern Colorado, north-central Arkansas, and South Carolina [see Maddison 2008: Fig. 18]. The record from “Mississippi” (Bousquet and Larochelle 1993: 126) probably refers to another species of the subgenus.
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, QC, SK USA: AR, CA, CO, CT, DC, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, NE, NH, NJ, NY, OH, OK, OR, PA, RI, SC, SD, VA, VT, WA, WI, WV, WY
This species ranges from Newfoundland to southeastern Quebec, south to central New Hampshire [see Maddison 2008: Fig. 22]
CAN: NB, NF, NS (CBI), QC USA: ME, NH, VT
This species ranges from Cape Breton Island to east-central Missouri, south to southwestern Mississippi and southern North Carolina along the Appalachian Mountains [see Maddison 2008: Fig. 19].
CAN: NB, NS (CBI), ON, QC USA: CT, DE, IL, MD, ME, MO, MS, NC, NH, NY, OH, PA, TN, VA, VT, WV
[honestum group]
This species ranges from the Roanoke River drainage in Virginia south to southeastern Mississippi in the Piedmont, east to eastern North Carolina [see Maddison 2008: Fig. 23]
USA: AL, MS, NC, SC, VA
This species ranges from Cape Breton Island to eastern Illinois, south to North Carolina along the Appalachian Mountains [see Maddison 2008: Fig. 23]. One old specimen simply labeled from Wisconsin is known (Messer 2010: 35). The records from “Iowa, ” “Kansas” (Hayward 1897: 56), and “Michigan” (Bousquet and Larochelle 1993: 126) need confirmation; that from Churchill in northern Manitoba (Elias 1984: 142) is in error (Maddison 2008: 181); those from “Wyoming, ” “Texas” (Hayward 1897: 56), “Arkansas, ” and “Mississippi” (Bousquet and Larochelle 1993: 126) are probably in error.
CAN: NB (CBI), NS, ON, QC USA: CT, DC, DE, IL, IN, KY, MA, MD, ME, NC, NH, NJ, NY, OH, PA, VA, VT, WV [IA, KS, MI, WI]
This species ranges from west-central Wisconsin to south-central Montana, south to north-central New Mexico, southern Texas, northwestern Tamaulipas in Mexico, eastern Louisiana (East Feliciana Parish, Igor M. Sokolov pers. comm. 2009), and southern Mississippi, east to west-central Indiana [see Maddison 2008: Fig. 24].
USA: AR, CO, IA, IL, IN, KS, LA, MN, MO, MS, MT, NE, NM, OK, SD, TX, WI – Mexico
This species ranges from New Brunswick (Webster and DeMerchant 2012: 5) and southern Quebec (André Larochelle and Reginald P. Webster pers. comm. 1997, 2009) south to northern Georgia (Choate and Choate 1995: 371) and northwestern South Carolina (Ciegler 2003: [1]) along the Appalachian Mountains, east to northeastern Massachusetts [see Maddison 2008: Fig. 20B).
CAN: NB, QC USA: GA, KY, MA, NC, NH, NY, PA, SC, TN, VA, VT
Two species, one along the Pacific Coast of North America, the other (Bembidion laterale Samouelle) along the coasts of Europe and northern Africa. The three other species listed in this subgenus by Lorenz (2005: 216) belong to the subgenus Desarmatocillenus Netolitzky (see Lindroth 1980).
Kavanaugh and Erwin (1992: 317) emended Lindroth’s (1963b) key to Bembidion to accommodate the new species.
This taxon is ranked as a distinct genus by many authors, with Armatocillenus Dupuis (three species in Taiwan and New Guinea), Desarmatocillenus Netolitzky (14 species in southeastern Asia and the Australian Region), Chinocillenus Netolitzky (one Chinese species), Corallicillenus Uéno (two Japanese species), and Novicillenus Uéno and Habu (two Japanese species) listed as subgenera (see Lorenz 2005: 216). Recently Sasakawa (2007) synonymized Novicillenus and Corallicillenus with Desarmatocillenus. Recent molecular data analyses conducted by Maddison and Ober (2011: 251) and Maddison (2012: 568) strongly support Cillenus as a member of Bembidion.
This species is known only from two localities on the Palos Verdes Peninsula along the coast of southern California (Kavanaugh and Erwin 1992: 315).
USA: CA
Northern Hemisphere, with five species in the Nearctic (one species) and Palaearctic (four European and northern African species, one of them endemic to the Canary Islands) Regions.
Lindroth (1963b: 265) treated the North American species.
This species is known from the Whitefox River drainage in east-central Saskatchewan and several localities in Alberta, as far north as the Fort Vermilion area (Bousquet 1987a: 120; CNC).
CAN: AB, SK
About 40 species in the Nearctic (one Holarctic species), Palaearctic (about 30 species), and Afrotropical (nine species) Regions.
Lindroth (1963b: 342-343) covered the species found in North America.
This Holarctic species is found in Kazakhstan, Siberia, Mongolia, Liaoning in China, the Korean Peninsula, and Japan (Marggi et al. 2003: 255), and from mainland Alaska (Lindroth 1963b: 343) to Newfoundland (Lindroth 1955a: 58-59), south to Connecticut (Litchfield County, William L. Krinsky pers. comm. 2008; Lindroth 1963b: 343), north-central Ohio (Lee 1994: 59), southeastern South Dakota (Kirk and Balsbaugh 1975: 18), and northern New Mexico (Casey 1918: 71, as Bembidion scopulinum bellulum; Taos County, UASM) along the Rocky Mountains. The record from northeastern Georgia (Fattig 1949: 17) is probably in error; that from “Pennsylvania” (Bousquet and Larochelle 1993: 138) needs confirmation.
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, CT, IL, IN, MA, ME, MI, MN, MT, ND, NH, NM, NY, OH, RI, SD, VT, WI, WY – Holarctic
About 65 species in the Nearctic (21 species, three of them adventive), Neotropical (four species in Middle America shared with North America), Palaearctic (about 50 species), and Afrotropical (one species in Ethiopia, Bembidion scottustulatum Netolitzky) Regions. Three species (Bembidion dauricum, Bembidion obscurellum and Bembidion petrosum) are Holarctic.
Lindroth (1963b: 330-342, as tetracolum, transversale, striola, and in part grapei groups) covered all but six (Bembidion lugubre, Bembidion pernotum, Bembidion mexicanum, Bembidion perspicuum, Bembidion sarpedon, and Bembidion femoratum) of the species found in North America. Maddison and Swanson (2010: 26-29) briefly discussed the status of each species of the transversale group but did not include a key to separate all species.
[dauricum group]
This Holarctic species ranges from Scandinavia to eastern Siberia and Mongolia (Marggi et al. 2003: 261) in the Palaearctic Region and from the Chukchi coast in Alaska to the Hudson Bay coast in northern Manitoba (Lindroth 1963b: 325); it also occurs quite isolated in the southern parts of the Rocky Mountains in Colorado and New Mexico (Lindroth 1969a: 1114). Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96); others, older than 33, 000 years B.P., in southwestern Ontario (Warner et al. 1988: 35).
CAN: MB, NT, YT USA: AK, CO, NM – Holarctic
[striola group]
This species inhabits the North American Cordilleras from southwestern Alberta and southern British Columbia (Lindroth 1963b: 318, as Bembidion debilicolle) south at least to Lake County in the Coast Ranges of California (Casey 1924: 34, as Bembidion debilicolle), northern Utah (Salt Lake and Utah Counties, CMNH), and north-central Colorado (Casey 1918: 65; Armin 1963: 169, as Bembidion debilicolle) along the Rocky Mountains.
CAN: AB, BC USA: CA, CO, ID, MT, NV, OR, UT, WY
This species is known from southeastern Wyoming (Lavigne 1977: 44), south-central Colorado (Wickham 1902: 233; Lindroth 1963b: 316), and “New Mexico” (Casey 1918: 74) west to Guadaloupe Island off the Pacific Coast of Baja California (Hayward 1897: 76). The record from “Montana” (Bousquet and Larochelle 1993: 135) needs confirmation; that from “Oregon” (Notman 1919b: 227) is in error according to Lindroth (1963b: 316).
USA: AZ, CA, CO, NM, NV, UT, WY [MT] – Mexico
This species is widely distributed in the North American Cordilleras from southern British Columbia south to central California along the Sierra Nevada (Lindroth 1963b: 317), southeastern Arizona (Cochise County, UASM, Ken Karns pers. comm. 2009), and “New Mexico” (Casey 1918: 65, as Bembidion viaticum) along the Rocky Mountains.
CAN: BC USA: AZ, CA, ID, MT, NM, NV, OR, UT, WA, WY
This species, the only one of the group occurring in eastern North America, is known from southernmost Ontario (Lindroth 1963b: 319) to “New Jersey, ” south to south-central North Carolina (Hoffman 1982: 146), northeastern Georgia (Horn and Ulyshen 2009: 121), eastern Alabama (Tallapoosa County, UASM, Drew A. Hildebrandt pers. comm. 2009), and east-central Mississippi (Lowndes County, Drew A. Hildebrandt pers. comm. 2008).
CAN: ON USA: AL, GA, KY, MD, MS, NC, NJ, OH, PA, TN, VA, WV
This species is known from a few localities in the Sierra Nevada north of Yosemite National Park (Lindroth 1963b: 318; Maddison 1985: 113).
USA: CA
This species ranges from Vancouver Island (Lindroth 1963b: 315) south to southern California (LeConte 1852a: 190) where it is “widely distributed” (Fall 1901a: 42). The records from Colorado (Wickham 1902: 233), New Mexico (Fall and Cockerell 1907: 157), central Arizona (Griffith 1900: 565), Utah (Knowlton 1939: 2), northern Idaho (Hatch 1953: 90), and northeastern Kansas (Popenoe 1878: 79) are probably in error.
CAN: BC (VCI) USA: CA (CHI), OR, WA
[tetracolum group]
This European species is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 61, as Bembidion rupestre) to western Quebec (Larochelle 1975: 53), and from Maine (Larochelle and Larivière 1990a: 28). The record from “New Hampshire” (Bousquet and Larochelle 1993: 137) needs confirmation. The first inventoried specimen collected on this continent was found in Newfoundland in 1907 (Lindroth 1957c: 151).
FRA: PM CAN: NB, NF, NS (CBI), PE, QC USA: ME [NH] – Adventive
This European subspecies is adventive in North America where it is known from New Brunswick and Nova Scotia (Bousquet 1992a: 504; Majka 2005: 536) and from one specimen discovered in 2003 in Grant County, Washington (Robert L. Davidson pers. comm. 2008). The first inventoried specimen collected on this continent was found near Halifax, Nova Scotia in 1967 (Bousquet 1992a: 504).
CAN: NB, NS (CBI) USA: WA – Adventive
This Holarctic subspecies ranges from Scandinavia to northeastern Siberia (Motschulsky 1845a: 27) and Mongolia (Marggi et al. 2003: 262) and in North America from Alaska (Lindroth 1963b: 339) to Newfoundland (Bousquet 1987a: 120), south to Virginia (Hoffman et al. 2006: 20), east-central Iowa (Iowa County, MCZ), southern New Mexico (Fall and Cockerell 1907: 157), southwestern Nevada (Esmeralda County, CMNH), and “California” (Cooper 1976: 163).
CAN: AB, BC, MB, NB, NF, NS, NT, ON, PE, QC, SK, YT USA: AK, CA, CO, IA, ID, IL, IN, ME, MI, MN, MT, ND, NH, NM, NV, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WY – Holarctic
Five more subspecies are recognized within the Palaearctic fauna.
This subspecies is known only from numerous specimens collected at the type locality.
USA: AK
This Holarctic subspecies is known from Scandinavia to eastern Siberia (Marggiet al. 2003: 262) and from Alaska (Lindroth 1963b: 334) to Newfoundland (Lindroth 1955a: 60-61), south to “Rhode Island” (Sikes 2003: 7), southwestern Pennsylvania (Allegheny County, CMNH), “Nebraska” (Hayward 1897: 81, as Bembidion lucidum), northeastern New Mexico (Casey, 1918: 75, as Bembidion castalium), southern Arizona (Wickham 1898: 300, as Bembidion lucidum), and southeastern California (San Bernardino County, MCZ).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, ID, IL, MA, ME, MI, MN, MT, ND, NE, NH, NM, NV, NY, OH, OR, PA, RI, UT, VT, WA, WI, WY – Holarctic
This subspecies is known only from a few localities on mainland Alaska.
USA: AK
Besides the nominotypical subspecies found in eastern Siberia, two more subspecies have been described from Japan and Szechwan, China.
The range of this species extends from the Ontario Peninsula (Bousquet 1987a: 120) to southern British Columbia, north to western Alaska (Lindroth 1963b: 337-338), south to northern Oregon (Gilliam and Morrow Counties, CMNH; Hatch 1953: 90; Lindroth 1963b: 337), southern Arizona (Dajoz 2007: 21), western Texas (Jeff Davis County, MCZ), and northwestern Pennsylvania (Warren and Forest Counties, CMNH). The species has been reported also from “Mexico” (Casey 1924: 35, as Bembidion aversans). The record from “California” (Bousquet and Larochelle 1993: 138) was based on a misidentified specimen of Bembidion petrosum in MCZ.
CAN: AB, BC, MB, NT, ON, SK USA: AK, AZ, CO, IA, ID, MI, MN, MT, ND, NE, NM, NV, OH, OR, PA, SD, TX, UT, WA, WI, WY – Mexico
The range of this subspecies is disjunct. It occurs in the east along the coast of Newfoundland, Saint Pierre and Miquelon (Lindroth 1955a: 63), the Maritimes, Magdalen Islands (Lindroth 1963b: 340), and the Saint Lawrence Estuary (Larochelle 1975: Fig. 169) and in the west from the Mackenzie River in central Northwest Territories south to northern New Mexico (Taos County, UASM), including southwestern North Dakota (Tinerella 2003: 635) [see Lindroth 1963a: Fig. 66].
FRA: PM CAN: AB, LB, NB, NF, NS (CBI), NT, PE, QC, SK USA: CO, ND, NM, UT, WY
This subspecies is confined to the west coast ranging from the Queen Charlotte Islands (Kavanaugh 1992: 66) south at least to northern California (Fall 1922c: 171; Hatch 1953: 91) [see Lindroth 1963a: Fig. 66].
CAN: BC (QCI, VCI) USA: CA, OR, WA
This Palaearctic subspecies is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 62, as Bembidion ustulatum) to western Montana (Russell 1968: 54), including southern Quebec and the Ontario Peninsula (Lindroth 1963b: 332), south to northern Utah (Salt Lake County, CMNH), west-central Nebraska (Keith County, Foster F. Purrington pers. comm. 2010), and central Virginia (Davidson 1995: 16); also in the Pacific Northwest from southwestern British Columbia (Lindroth 1963b: 332) to northern California (Notman 1929b: 222; Tehama County, Foster F. Purrington pers. comm. 2010). The records from Colorado (Wickham 1902: 233, as Bembidion ustulatum), “Tennessee, ” and “North Carolina” (Bousquet and Larochelle 1993: 138) need confirmation; that from “Texas” (Lindroth 1955a: 62) could be based on a mislabeled specimen. The first inventoried specimen collected on this continent was found prior to 1823, likely along the east coast. The species has been unearthed from a xvii Century colonial site in Boston, Massachusetts (Bain 1998: 39), suggesting that the species was established on this continent long before Thomas Say collected his specimens. The species is also adventive in southeastern Australia (Moore et al. 1987: 136).
FRA: PM CAN: BC, NB, NF, NS, ON, PE, QC USA: CA, CT, DC, IA, IL, IN, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI [CO, NC, TN, TX] – Adventive
This species has been known for a long time under the name Bembidion ustulatum (Linnaeus, 1758). Lindroth (1957b: 334-335) suggested to drop the name to promote stability. Four other subspecies of Bembidion tetracolum are found in the Palaearctic Region.
[transversale group]
This species ranges from “California” to “Texas, ” south at least to Oaxaca, Mexico (Maddison and Swanson 2010: 29).
USA: AZ, CA, NM, TX, UT – Mexico
This species ranges from South Dakota (Maddison and Swanson 2010: 28) and eastern Wyoming (Lavigne 1977: 44, as Bembidion lugubre) south to Costa Rica (Erwin 1982b: 469, Fig. 65).
USA: AZ, CO, NE, NM, SD, WY – Belize, Costa Rica, Guatemala, Mexico
This species is known from southern Colorado and northern New Mexico (Maddison and Swanson 2010: 29).
USA: CO, NM
This species ranges from “Oregon” (Maddison and Swanson 2010: 27) to western Nebraska (Keith County, Robert L. Davidson pers. comm. 2012), south to western Texas (Jeff Davis and Presidio Counties, CNC) and southern California (Maddison and Swanson 2010: 27).
USA: AZ, CA, CO, KS, NE, NM, NV, OK, TX, UT
This species ranges from “Wyoming” south to northern New Mexico and northeastern Arizona (Maddison and Swanson 2010: 28).
USA: AZ, CO, NM, UT, WY
This species ranges from Newfoundland (Lindroth 1955a: 59) to Alaska (Maddison and Swanson 2010: 27), south to southern California (LeConte 1852a: 190; Maddison 1985: 114), “Utah, ” “Colorado” (Maddison and Swanson 2010: 27), northern Michigan (Hubbard and Schwarz 1878: 629; Dunn 1985a: 10; CNC), and central New York (Notman 1928: 215). The record from South Dakota (Kirk and Balsbaugh 1975: 17) refers to Bembidion mexicanum (USNM); thatfrom “Illinois” (Bousquet and Larochelle 1993: 138) needs confirmation.
CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: AK, CA, CO, ID, MI, MN, MT, ND, NV, NY, OR, UT, WA, WI, WY [IL]
Maddison and Swanson (2010: 27) stated that specimens of this species from western British Columbia, western Oregon, and California represent another form, with no consistent differences in COI or 28S from the typical form but with clearly different mentum shape. They added that Bembidion transversale as currently understood could be a complex of multiple species.
About 15 species in the Nearctic (one northwestern species), Oriental (one species, Bembidion vitalisi Andrewes from Laos), and Palaearctic (14 species) Regions. Some of the species included by Lorenz (2005: 229) in the subgenus Ocydromus belong to this subgenus according to Toledano (2000: 66).
Lindroth (1963b: 307-310) covered the species found in North America.
This subspecies is known only from the Rocky Mountains and foothills in British Columbia and Alberta, as far north as northern British Columbia near the Yukon Territory border (Lindroth 1963b: 309). The record from southwestern Alaska (Elias 1988: 41) possibly refers to the nominotypical subspecies and needs confirmation.
CAN: AB, BC [AK]
This subspecies is known only from southwestern Yukon Territory and Alaska from the Gulf Coast to beyond the arctic circle (Lindroth 1963b: 308). Fossil remnants of this species, dated between about 10, 000 and 11, 000 years old, have been found in southern Quebec (Mott et al. 1981: 146) and southern Ontario (Pilny et al. 1987: 620).
CAN: YT USA: AK
Another subspecies, Bembidion mckinleyi scandicum Lindroth, is found along the northern parts of the Palaearctic Region from Sweden to the Far East and Mongolia (Marggi et al. 2003: 268).
Northern Hemisphere, with about 30 species in the Nearctic (six species) and Palaearctic (27 species) Regions. Two species (Bembidion umiatense and Bembidion lenae) are Holarctic.
Lindroth (1963b: 326-330, as bimaculatum and lenae groups) covered all but one (Bembidion renoanum) of the species found in North America.
This species ranges from north-central Ontario (CNC) to the Arctic Plains of Alaska (Lindroth 1963b: 326), south to San Bernardino County in southeastern California (Dajoz 2007: 20), northeastern Arizona (Apache County, UASM; Wickham 1896a: 157), and southern New Mexico (Fall and Cockerell 1907: 157). The record from “Kansas” (Hamilton 1894a: 7) is probably in error.
CAN: AB, BC (VCI), MB, NT, ON, SK, YT USA: AK, AZ, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY
This Holarctic species is found from eastern Siberia (Marggi et al. 2003: 243) to the Anderson River delta in northern Northwest Territories (Lindroth 1963b: 311).
CAN: NT, YT USA: AK – Holarctic
This eastern species is found from “New Brunswick” (Larochelle and Larivière 1990a: 28) to northern Minnesota (Gandhi et al. 2005: 925), south to northeastern Iowa (Purrington and Larsen 1997: 50), southwestern Ohio (Dury 1910: 66), and southwestern Pennsylvania (Hayward 1897: 80; Lindroth and Freitag 1969: 336). The record from northeastern Georgia (Fattig 1949: 17) is probably in error.
CAN: NB, QC USA: IA, IL, IN, MA, ME, MN, NH, NY, OH, PA, VT, WI
This species is known only from the type locality in western Nevada.
USA: NV
The range of this species extends from the Ungava Bay area (Larochelle 1975: Fig. 173) in northern Quebec to the Arctic Plains of Alaska (Lindroth 1963b: 329), south to east-central Utah (Grand County, UASM), southwestern Colorado (Elias 1987: 632), southern South Dakota (Kirk and Balsbaugh 1975: 18), and the Saint Lawrence River drainage in southern Quebec (Larochelle 1975: Fig. 173). The record from “Michigan” (Bousquet and Larochelle 1993: 137) needs confirmation. Fossil remnants, dated from about 10, 400 and 21, 500 years B.P., have been unearthed in eastern Minnesota, northeastern Wisconsin, Iowa, northern Illinois (Schwert 1992: 77), northeastern Pennsylvania (Barnosky et al. 1988: 178), and Cape Breton Island in Nova Scotia (Miller 1997: 250).
CAN: AB, BC, MB, NT, ON, QC, SK, YT USA: AK, CO, MT, ND, SD, UT, WY [MI]
This Holarctic species is found from the northern regions of European Russia to eastern Siberia (Marggi et al. 2003: 244) and in North America from the Arctic Plains in Alaska (Lindroth 1963b: 330) to Contwoyto Lake in western Nunavut (Shpeley and Pilny 1995: 233).
CAN: NU, YT USA: AK – Holarctic
Northern Hemisphere, with about 35 species in the Nearctic (seven species, one of them adventive) and Palaearctic (about 30 species) Regions. Two species (Bembidion grapii and Bembidion yukonum), placed in this subgenus for convenience, are Holarctic.
Lindroth (1963b) covered all but one (Bembidion subangustatum) of the species found in North America.
This Holarctic species ranges in North America from the Seward Peninsula in Alaska (Lindroth 1963b: 320) to Newfoundland (Lindroth 1955a: 57) and Greenland (Böcher 1988: 12), including the Aleutian and Kodiak Islands, south to mountains in New England (Lindroth 1963b: 320), southeastern upper peninsula of Michigan (Mackinac County, UMAA), northern Wisconsin (Bayfield County, MCZ), northeastern Minnesota (Kamal J.K. Gandhi pers. comm. 2008), southern New Mexico (Fall and Cockerell 1907: 157), and southeastern Arizona (Dajoz 2007: 21; Lindroth 1969a: 1114). The records from “Massachusetts” and “Pennsylvania” (Bousquet and Larochelle 1993: 136) are in error.
DEN: GL FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: AK, AZ, CO, ME, MI, MN, MT, NH, NM, NV, NY, UT, VT, WI, WY – Holarctic
This species is listed as incertae sedis by Marggi et al. (2003: 271) and Maddison (2012: 542). It belongs to the Ocydromus Complex. For convenience, the species is listed in the subgenus Peryphanes where Lindroth (1963b: 319, as grapei group) placed it.
This eastern species is found from New Brunswick (Webster and Bousquet 2008: 17) to “North Dakota” (Donald P. Schwert pers. comm. 1989), south to Big Bend National Park in western Texas (Dajoz 2007: 23) and Tennessee (Cannon, Clay, Fayette, Macon, Madison, Overton, Smith, Wayne, and Wilson Counties, CMNH), including northeastern New Mexico (Fall and Cockerell 1907: 157, as Bembidion picipes). The record from British Columbia (Jarrett and Scudder 2001: 381) was based on misidentified specimens of Bembidion platynoides Hayward (UBC).
CAN: NB, ON, QC USA: CT, IA, IN, KS, KY, MA, MD, ME, MI, MN, MO, ND, NH, NJ, NM, NY, OH, PA, SD, TN, TX, VA, VT, WI, WV
This species has been known for a long time under the name Bembidion picipes (Kirby). Lindroth (1953b: 176) showed that Kirby’s syntypes of Bembidion picipes were conspecific with those of Bembidion grapii Gyllenhal.
The range of this western species extends from Vancouver Island (Lindroth 1963b: 323) to western Montana (Russell 1968: 53; Edwards 1975: 53), north to central British Columbia (Lindroth 1963b: 323), south to southern California (Hayward 1897: 79; Fall 1901a: 42; Moore 1937: 6). The record from “New Mexico” (Bousquet and Larochelle 1993: 136) is probably in error.
CAN: BC (VCI) USA: AZ, CA (CHI), ID, MT, OR, WA
This Palaearctic species is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 58) to northeastern Wisconsin (Forest County, CMNH; Messer 2010: 36), south to northern Ohio (Lee 1994: 58) and Massachusetts (Lindroth 1955a: 58). The records from “Rhode Island” and “Virginia” (Bousquet and Larochelle 1993: 136) need confirmation. The first inventoried specimen collected on this continent was found in Ottawa, Ontario in 1891 (Lindroth 1963b: 323).
FRA: PM CAN: NB, NF, NS (CBI), ON, PE, QC USA: MA, ME, NH, NY, OH, VT, WI [RI, VA] – Adventive
This species is known from western Texas (Jeff Davis County, Ken Karns pers. comm. 2009), southern New Mexico (Fall and Cockerell 1907: 157), southern Arizona (Casey 1918: 67), southeastern California (Dajoz 2007: 20), and “Mexico” (Casey 1918: 67).
USA: AZ, CA, NM, TX – Mexico
This species ranges from northwestern Ohio (Holeski and Graves 1982: 216) to southern Manitoba (Lindroth 1963b: 326), south to southeastern Arizona (Maddison 1985: 114; Dajoz 2007: 21), northwestern New Mexico (Casey 1918: 67 as Bembidion inquietum), west-central and central Texas (Blanco, Crockett, Edwards, Travis, and Williamson Counties, CMNH), southeastern Louisiana (Summers 1874a: 81), and northern Mississippi (Grenada and Pontotoc Counties, Drew A. Hildebrandt pers. comm. 2010).
CAN: MB USA: AR, AZ, CO, IA, IL, IN, KS, KY, LA, MN, MO, MS, NE, NM, OH, OK, SD, TN, TX, WI
This Holarctic species is found from Scandinavia to eastern Siberia (Marggi et al. 2003: 272) and in North America from the Arctic Plains in northern Alaska (Lindroth 1963b: 321) to the north shore of the Saint Lawrence Estuary (Larochelle 1975: Fig. 183), south to east-central British Columbia (Lindroth 1963b: 321).
CAN: BC, MB, NT, QC, YT USA: AK – Holarctic
This species is listed as incertae sedis by Marggi et al. (2003: 272). It was not sequenced by Maddison (2012). For convenience, the species is listed in the subgenus Peryphanes where Lindroth (1963b: 321, as grapei group) placed it.
Northern Hemisphere, with 24 species in the Nearctic (6 species) and Palaearctic (18 species) Regions arrayed in three species groups, two of these groups being exclusively Eurasian. One species from India, Bembidion braminum Andrewes, is usually also included in this subgenus (see Lorenz 2005: 234) but its taxonomic position should be reevaluated.
Lindroth (1963b: 312-314, as nebraskense group) covered all but two species (Bembidion modocianum and Bembidion perbrevicolle) described by Casey.
This species is found in the North American Cordilleras from southeastern Alberta and southern British Columbia south to the Sierra Nevada in California (Lindroth 1963b: 313) and to central Colorado (Wickham 1902: 233, as Bembidion breve) along the Rocky Mountains.
CAN: AB, BC USA: CA, CO, ID, MT, NV, OR, WA, WY
This species has been known until the 1960s under the name Bembidion breve (Motschulsky).
This species is known so far only from the type series collected in northeastern California.
USA: CA
This western species is known from southwestern Alberta and southern British Columbia (Lindroth 1963b: 312) south at least to northeastern California (Modoc County, CNC; Hayward 1897: 69), Nevada (White Pine County, CMNH), northern Utah (Rich County, UASM), and northern New Mexico (Taos County, UASM; Hayward 1897: 69; Maddison 2012: Supplementary content Table S1). The record from “Nebraska” (Hayward 1897: 69) probably originated from the original statement about the type locality.
CAN: AB, BC USA: CA, CO, ID, MT, NM, NV, OR, UT, WA, WY
This species is found along the North American Cordilleras from northern Washington (Hatch 1950: 101, as Bembidion immaculosum) to Sequoia National Park in southeastern California along the Sierra Nevada (Fall 1910: 96, as Bembidion whitneyi; Dajoz 2007: 16). The records from southern Idaho (Horning and Barr 1970: 24; Stafford et al. 1986: 288, as Bembidion immaculosum) and British Columbia (Hatch 1953: 89, as Bembidion nevadense sensu Hatch) need confirmation.
USA: CA, OR, WA [BC, ID]
According to Erwin (1984a: 178), this species occurs in the foothills of the Sierra Nevada of California.
USA: CA
This species is known only from the original six specimens collected in “Nevada.”
USA: NV
One North American species along the Pacific Coast.
The species is included in Lindroth’s (1963b: 224) key to the Canadian Bembidion.
This species ranges along the Pacific Coast from the southern tip of Vancouver Island (Bousquet 1987a: 121) to southern California (LeConte 1879d: 509; Fall 1901a: 9).
CAN: BC USA: CA, OR, WA
Northern Hemisphere, with 11 species in the Nearctic (six species) and Palaearctic (six species) Regions. One species (Bembidion quadrimaculatum) is Holarctic but with different subspecies in the Nearctic and Palaearctic Regions.
Lindroth (1963b: 382-387) covered four of the North American species in his monograph. A revision of the Nearctic species is needed.
This species is known from the Sierra Nevada (Casey 1918: 153, as Bembidion relictum; Dajoz 2007: 17) and the Coast Ranges in west-central California. The record from eastern Oregon (Hatch 1953: 98, as Bembidion relictum) needs confirmation.
USA: CA [OR]
The range of this species extends from Newfoundland (Lindroth 1955a: 73) to east-central Alaska (Lindroth 1963b: 387), south to Nechako River in central British Columbia, northern New Mexico (Fall and Cockerell 1907: 157), west-central Minnesota (Gandhi et al. 2005: 924), the Adirondack Mountains in northeastern New York (Notman 1928: 218), and New England (Lindroth 1963b: 387). The record from “Washington” (Bousquet and Larochelle 1993: 146) is probably in error.
CAN: AB, BC, MB, NB, NF, NS, NT, ON, QC, SK, YT USA: AK, CO, ID, ME, MI, MN, NH, NM, NY, UT, VT, WI, WY
As far as known, this species has been recorded from the type locality and Frenchglen, Harney County, in Oregon (Hatch 1953: 98).
USA: OR
This species ranges from southeastern Pennsylvania (Delaware and Lebanon Counties, Robert L. Davidson pers. comm. 2008) to southeastern Minnesota (Gandhi et al. 2005: 925), south to central Texas (Bosque County, CMNH) and eastern Tennessee (Sevier County, CNC). The records from “New Jersey” and “North Carolina” (Bousquet and Larochelle 1993: 146) need confirmation.
USA: DC, DE, IA, IL, IN, KY, KS, MD, MI, MN, MO, NE, OH, PA, TN, TX, VA, WI, WV [NC, NJ]
This species is found from southern Manitoba (Lindroth 1963b: 386) to southeastern Washington (Hatch 1953: 98), including southern Saskatchewan and Alberta (Lindroth 1963b: 386), south at least to Santa Barbara County in southwestern California (Maddison 1985: 113), southern Arizona (Dajoz 2007: 21), and southern Colorado (LeConte 1879d: 509; Wickham 1902: 234).
CAN: AB, MB, SK USA: AZ, CA, CO, ID, MT, NV, OR, UT, WA, WY
This subspecies ranges from the southwestern edge of Lac Winnipeg in southern Manitoba to Vancouver Island (Hayward 1897: 119), north to the Brook Range in Alaska (Lindroth 1963b: 385), south to the Baja California Peninsula (Horn 1894: 308) and southern New Mexico (Fall and Cockerell 1907: 157). The records from Kansas (Snow 1903: 193; Knaus 1905a: 218) need confirmation.
CAN: AB, BC (VCI), MB, NT, SK, YT USA: AK, AZ, CA, CO, ID, MT, NM, NV, OR, WA, WY [KS] – Mexico
Three other subspecies, including the nominotypical one, are known from the Palaearctic Region (Toledano 1999: 205-210).
This subspecies ranges from Newfoundland (Lindroth 1955a: 72) to the Rocky Mountains in Alberta (Lindroth 1963b: 384), south to central Colorado (Wickham 1902: 235), “Texas” (Lindroth 1963b: 384), northern Georgia (Fattig 1949: 18), and east-central South Carolina (Ciegler 2000: 48).
CAN: AB, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CO, CT, DC, GA, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
Six Neotropical species of which three extend into southwestern North America.
Perrault (1982a) revised the species except Bembidion jucundum Horn.
This species is known from the Baja California Peninsula and “Arizona” (Hayward 1897: 121).
USA: AZ – Mexico
This species ranges from southeastern Arizona and southwestern New Mexico (Hidalgo County, CMNH) south to the Isthmus of Tehuantepec in southern Mexico [see Perrault 1982a: Fig. 64].
USA: AZ, NM – Mexico
This species is found from Arizona and New Mexico south to Oaxaca in Mexico [see Perrault 1982a: Fig. 64].
USA: AZ, NM – Mexico
Western Hemisphere, with nine species in North America (nine species) and northern Neotropical (three species in Cuba and Mexico, all shared with North America) Regions.
Lindroth (1963b: 376-381, as affine and versicolor groups) covered six of the nine species, leaving three species described by Casey. A taxonomic revision of the subgenus is needed.
[affine group]
This species ranges from southwestern Quebec (CNC) to “North Dakota” (Donald P. Schwert pers. comm. 1989), south to east-central Texas (Casey 1918: 128; Riley 2011) and the Florida Panhandle (Peck and Thomas 1998: 18); also recorded from Cuba (Jacquelin du Val 1857: 23). The records from Arizona (Wickham 1896a: 157; Hayward 1897: 122) and “Colorado” (Wickham 1902: 235) need confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MO, MS, NC, ND, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [AZ, CO] – Cuba
[versicolor group]
This species is known from central Wyoming (Natrona County, CMNH) south to northwestern New Mexico (Casey 1918: 132) and southern Arizona (Casey 1918: 133, as Bembidion demissum; Cochise and Pima Counties, CMNH). The record from northeastern California (Notman 1929b: 223, as Bembidion demissum) needs confirmation.
USA: AZ, CO, NM, WY [CA]
This species is known from eastern Washington (Hatch 1953: 97) to central Wyoming (Natrona County, CMNH), south at least to the Sierra Nevada in east-central California (Blaisdell 1902: 77).
USA: CA, ID, OR, WA, WY
This species occurs from southwestern New Brunswick (Larochelle and Larivière 1990a: 29, 34) to southern British Columbia (Lindroth 1963b: 381), south to southeastern California (Casey 1918: 130, as Bembidion frugale; Andrews et al. 1979: 28), the Federal District in Mexico (Casey 1918: 133, as Bembidion indigens), and southern Florida (Peck and Thomas 1998: 18).
CAN: BC, MB, NB, ON, QC, SK USA: AR, AZ, CA, CO, CT, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, NE, NJ, NM, NV, OH, OK, OR, PA, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
The range of this species extends from southern Newfoundland (Lindroth 1955a: 72) to southern Saskatchewan (Ronald R. Hooper pers. comm. 1990), south to northern Nebraska (Cherry County, Foster F. Purrington pers. comm. 2010), “Illinois” (Lindroth 1955a: 72), and northeastern West Virginia (Tucker County, CMNH). The record from the Atlantic Coast in South Carolina (Kirk 1969: 10; Ciegler 2000: 48) needs confirmation.
CAN: MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CT, IA, IL, MA, ME, MI, MN, NE, NH, NJ, NY, OH, PA, RI, SD, VT, WI, WV [SC]
This species has been collected in western Texas (Brewster County, CMNH), southern Arizona (Cochise, Pima, and Pinal Counties, CMNH; Casey, 1924: 42), and in Baja California Sur (CMNH).
USA: AZ, TX – Mexico
This species is found from western Ontario to Vancouver Island (Lindroth 1963b: 379), north to northern Yukon Territory (Maddison 1985: 114) and southern Northwest Territories (Lindroth 1963b: 379), south to southern California (Fall 1901a: 42; Andrews et al. 1979: 28), southern Arizona (Wickham 1898: 300; Griffith 1900: 565; Snow 1907: 142), central New Mexico (Ellis et al. 2001: 16), southern South Dakota (Kirk and Balsbaugh 1975: 19), and northwestern Minnesota (Polk County, CMNH; Kittson and Roseau Counties, Peter W. Messer pers. comm. 2009). The record from “Michigan” (Bousquet and Larochelle 1993: 145) needs confirmation.
CAN: AB, BC (VCI), MB, NT, ON, SK, YT USA: AZ, CA, CO, ID, MN, MT, ND, NM, NV, OR, SD, UT, WA, WY [MI]
This species is known only from Lake County in the California Coast Ranges.
USA: CA
This species is found from Newfoundland (Lindroth 1955a: 70-71) to central Alaska (Lindroth 1963b: 378), south to northwestern Oregon (Westcott et al. 2006: 7), southern Colorado (LeConte 1879d: 501; Wickham 1902: 234; Elias 1987: 632), southwestern Nebraska (Keith County, CMNH), and southern South Carolina (Ciegler 2000: 49). The records from “Kansas” (Popenoe 1878: 79) and Arkansas (Wickham 1897: 104) need confirmation; those from Texas (Wickham 1897: 104), New Mexico (Fall and Cockerell 1907: 157) and Arizona (Wickham 1897: 104; Griffith 1900: 565) possibly refer to Bembidion impotens.
FRA: PM CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, CO, CT, DE, IA, ID, IL, IN, KY, MA, ME, MI, MN, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, VA, VT, WA, WI, WV, WY [AR, KS]
Four North American species.
Bousquet and Webster (2006) provided a key for the identification of three species. Subsequently, a species described by Casey and incorrectly placed in another subgenus, as a synonym, was found to belong to this subgenus (see below under Bembidion tencenti).
This species is found from southern Alberta (Lindroth 1969a: 1115) to southeastern Arizona (Cochise County, CMNH), southern New Mexico (Hidalgo County, CMNH; Fall and Cockerell 1907: 157), and western Texas (Brewster and Culberson Counties, CMNH, MCZ).
CAN: AB USA: AZ, CO, NM, TX, UT, WY
This species is known from southern Saskatchewan, central South Dakota (Bousquet and Webster 2006: 32), and southeastern Colorado (Pueblo County, Foster F. Purrington pers. comm. 2009).
CAN: SK USA: CO, SD
Members of this species are probably conspecific with Bembidion tencenti Hatch (see “Note” section under Bembidion tencenti).
This species ranges from southern Manitoba to southern Alberta (Lindroth 1963b: 347), south to southern Texas (Kenedy County, CNC).
CAN: AB, MB, SK USA: CO, ND, TX
This species is known only from the holotype collected in southeastern Oregon.
USA: OR
This taxon has been listed as a junior synonym of Bembidion dejectum Casey by Lindroth (1963b: 356) but the holotype is not conspecific with members of Bembidion dejectum. The specimen belongs to the subgenus Neobembidion and is externally very similar and probably conspecific with members of Bembidion nitidicolle Bousquet.
Northern Hemisphere, with eight species in the Nearctic (one Holarctic species) and Palaearctic (eight species) Regions.
Lindroth (1963b: 393-395) covered the North American species in his monograph of the Canadian and Alaskan Carabidae.
One species (Bembidion longipenne Putzeys) from Venezuela is currently included in this subgenus but is probably not consubgeneric with members of Diplocampa. Bembidion hesperus Crotch, described from the Azores and listed in this subgenus by Lorenz (2005: 223), is a junior synonym of Bembidion ambiguum Dejean (Lindroth 1960b: 6), a species of the subgenus Neja Motschulsky.
This Holarctic subspecies is found from northern Europe (Marggi et al. 2003: 248) to eastern Siberia (Kryzhanovskij et al. 1995: 82) and from the Chukchi Sea coast in Alaska (Lindroth 1963b: 394) to Newfoundland (Lindroth 1955a: 75-76), south to northern Pennsylvania (Bradford County, CMNH), northeastern Illinois (Lake County, CNC), east-central South Dakota (Kirk and Balsbaugh 1975: 20), and “Oregon” (Hatch 1953: 99).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, IA, ID, IL, IN, MA, ME, MI, MN, MT, NH, NY, OH, OR, PA, SD, VT, WA, WI – Holarctic
The subspecies Bembidion transparens prostratum (Motschulsky) occurs in the eastern part of Asia.
Northern Hemisphere, with 26 species in the Nearctic (eight species) and Palaearctic (18 species) Regions.
Bousquet and Webster (2006) provided a key for the identification of all North American species except Bembidion rubiginosum. Lindroth (1963b: 387-393) covered six of the eight North American species.
This species inhabits the North American Cordilleras from the arctic circle in eastern Alaska and west-central Yukon Territory (Lindroth 1963b: 392) south to San Bernardino County in southeastern California (Dajoz 2007: 20, as Bembidion convexiuscula), northern Idaho (Hatch 1953: 94), northwestern Wyoming (Teton County, UASM), and east-central Montana (Russell 1968: 57).
CAN: AB, BC, YT USA: AK, CA, ID, MT, OR, WA, WY
This species is known from Newfoundland to central Alaska, south to northeastern British Columbia and southern Alberta in the Rocky Mountains (Lindroth 1963b: 389). Fossil remnants of this species, dated between about 10, 400 and 28, 000 years B.P., have been unearthed in eastern Minnesota, northeastern Wisconsin, Illinois (Schwert 1992: 77), Iowa (Baker et al. 1986: 96; Schwert 1992: 77), central North Dakota (Ashworth and Schwert 1992: 260), and Cape Breton Island in Nova Scotia (Miller 1997: 250).
CAN: AB, BC, MB, NF, NT, ON, SK, YT USA: AK
This eastern species ranges from Cape Breton Island to southern Saskatchewan (Lindroth 1963b: 388), south to eastern South Dakota (Larsen and Purrington 2010: 571), northern Illinois (Hayward 1897: 123), and northeastern West Virginia (Tucker County, CMNH).
CAN: MB, NB, NS (CBI), ON, PE, QC, SK USA: IL, MA, ME, MI, MN, NH, NJ, NY, PA, RI, SD, VT, WI, WV
This species ranges from New Brunswick and Maine west to eastern Minnesota and southeastern Manitoba (Bousquet and Webster 2006: 24).
CAN: MB, NB, ON, QC USA: MA, ME, MI, MN, NH, WI
This species is found from New Brunswick (Bousquet 1987a: 121) to the Osoyoos Valley in southern British Columbia, south to central Washington (Lindroth 1963b: 389), northern Iowa (Purrington et al. 2002: 201), northeastern Illinois (Lake County, CNC), and New York (Liebherr and Song 2002: 132; Saint Lawrence County, CMNH). The species seems to be local.
CAN: BC, NB, ON, QC, SK USA: IA, IL, ME, MI, MN, NH, NY, WA, WI
This species is found from the southern part of the Prairie Provinces (Lindroth 1963b: 393) to eastern Oregon (Baker County, CNC), south to Riverside County in southern California (Dajoz 2007: 19), central Nevada (Lander County, CNC), central Utah (Sevier County, CNC), southern Wyoming (Lindroth 1963b: 393), and southwestern North Dakota (Bowman County, CNC).
CAN: AB, MB, SK USA: CA, ID, MT, ND, NV, OR, UT, WY
This species is known from Saskatchewan and Alberta (David R. Maddison pers. comm. 2012), southwestern Idaho (Owyhee County, CNC), southeastern Wyoming (Lavigne 1977: 44), and Costilla County in south-central Colorado (LeConte 1879d: 508; Wickham 1902: 234).
CAN: AB, SK USA: CO, ID, WY
This eastern species ranges from Cape Breton Island (CNC) to “Michigan” (Garry A. Dunn pers. comm. 1986), south to northeastern West Virginia (Randolph County, CMNH).
CAN: NB, NS (CBI), ON, QC USA: CT, MA, ME, MI, NH, NJ, NY, PA, RI, VT, WV
About 90 species in the Nearctic (50 species), Neotropical (about 35 species), and Palaearctic (seven species) Regions. One species from the Philippines (Bembidion igorot Darlington) and one from Myanmar (Bembidion serpentinum Landin) are also listed in this subgenus by Lorenz (2005: 220). Two species (Bembidion nigripes and Bembidion semipunctatum) are Holarctic. One South American species (Bembidion brullei Gemminger and Harold) has been introduced in Australia and New Zealand (Lindroth 1976a: 197-198). The North American species are arrayed here in six species groups for convenience.
Lindroth (1963b: 357-375, as scudderi, obtusangulum, dorsale, contractum, and oberthueri groups) covered 28 of the 50 North American species. A taxonomic revision of the North American species is needed.
[contractum group]
This species is found along the Atlantic Coast from Long Island (Casey 1924: 41, as Bembidion assensum) to Maryland (Erwin 1984a: 166).
USA: DE, MD, NJ, NY, RI
This species is found along the Atlantic and Gulf of Mexico coasts from the Maritime Provinces (Lindroth 1963b: 373; Bousquet 1987a: 121) to southern Florida including the Keys (Leng 1915: 572), west to southeastern Texas (Wickham 1897: 104; Casey 1918: 124, as Bembidion constrictum civile). The records from Ohio (Everly 1938: 141), Indiana (Wolcott and Montgomery 1933: 125), South Dakota (Kirk and Balsbaugh 1975: 19), “Nebraska” (Hayward 1897: 108), Kansas (Knaus 1887: 87; Snow 1903: 193), and “New Mexico” (Hayward 1897: 108) probably refer to Bembidion viridicolle LaFerté-Sénectère.
CAN: NB, NS (CBI), PE USA: AL, CT, DC, DE, FL, GA, LA, MA, MD, ME, MS, NC, NH, NJ, NY, RI, VA, TX
This species is found along the Atlantic Coast from Newfoundland (Lindroth 1955a: 70), the Maritime Provinces (Lindroth 1963b: 373), and the Magdalen Islands (Larochelle 1975: 55) to southern Florida (Peck and Thomas 1998: 18) and along the Gulf Coast to southern Louisiana (Summers 1874a: 81; Hine 1906: 76; Allen 1965: 65). The records from “Ohio, ” “Tennessee” (Hayward 1897: 109), eastern Kansas (Popenoe 1877: 24), and “Pennsylvania” (Bousquet and Larochelle 1993: 144) need confirmation.
FRA: PM CAN: NB, NF, NS (CBI), PE, QC USA: AL, CT, DC, DE, FL, GA, LA, MA, MD, ME, NH, NJ, NY, RI, SC [KS, OH, PA, TN]
This species is known so far only from the Florida Peninsula (Peck and Thomas 1998: 18).
USA: FL
This species is known from central Texas (Lee County, CMNH), southwestern Louisiana (Cameron Parish, Igor M. Sokolov pers. comm. 2009), and southwestern Florida (Monroe County, CMNH).
USA: FL, LA, TX
Hayward (1901: 158) listed Bembidion pilatei Chaudoir as a synonym of Bembidion constrictum LeConte but Lindroth (1963b: 373) regarded the two forms as distinct species.
This species ranges from Connecticut (Krinsky and Oliver 2001: 88) to southeastern Alberta (Lindroth 1963b: 375), including southernmost Ontario (Bousquet 1987a: 121), north to Fort Smith in southern Northwest Territories (Lindroth 1963b: 374-375), south to northeastern Arizona (Apache County, CMNH), west-central New Mexico (Cibola County, CMNH), southern Texas (Johnson 1978: 67), and southern Florida, including the Keys (Peck and Thomas 1998: 18). The species is also known from the Bahamas (Darlington 1953: 5, as Bembidion chevrolati), several islands of the West Indies (Peck and Thomas 1998: 18), and “Mexico” (Erwin et al. 1977: 4.21).
CAN: AB, MB, NT, ON, SK USA: AZ, CO, CT, FL, GA, IA, IN, IL, KS, LA, MS, ND, NE, NJ, NM, OH, OK, PA, SC, SD, TX, VA, WI, WY – Bahamas, Cuba, Dominica Republic, Haiti, Jamaica, Mexico, Puerto Rico
This species is known only from the type locality, from Atlantic County in New Jersey (CMNH), and from one specimen simply labeled “Md” (CMNH).
USA: MD, NJ
This form was listed in synonymy with Bembidion contractum Say by Horn (1885b: 108) but considered a valid species by Erwin (1984a: 185).
[dorsale group]
This species is found from southeastern Manitoba to the foothills of the Rocky Mountains in southern Alberta, north to northeastern British Columbia (Lindroth 1963b: 359), south to northeastern Wyoming (Crook County, CNC), Kansas (Popenoe 1877: 24; Snow 1903: 193), Missouri (Summers 1873: 147; Lindroth and Freitag 1969: 337), southern Illinois (Hayward 1897: 101), and southwestern Ohio (Dury 1902: 111). The record from “Colorado” (Bousquet and Larochelle 1993: 141) needs confirmation.
CAN: AB, BC, MB, SK USA: IA, IL, IN, KS, MI, MN, MO, MT, ND, NE, OH, SD, WI, WY [CO]
The range of this species extends from New Brunswick to the foothills of the Rocky Mountains in Alberta (Lindroth 1963b: 375), south to south-central Montana (Carbon County, UASM), “Nebraska, ” “Illinois” (Hayward 1897: 103, as Bembidion viridicolle), and southeastern West Virginia (Greenbrier County, David R. Maddison pers. comm. 2011). The record from San Bernardino County, California (Cooper 1976: 163) is probably based on a mislabeled specimen; that from “Indiana” (Bousquet and Larochelle 1993: 144) needs confirmation. One specimen from Kamloops, British Columbia, was regarded as possibly mislabeled by Lindroth (1963b: 375).
CAN: AB, MB, NB, ON, QC, SK USA: CT, IA, IL, MA, ME, MI, MN, MT, ND, NE, NH, NY, OH, PA, SD, VT, WI, WV [BC, IN]
This species is found from Saskatchewan to western British Columbia, north to southern Northwest Territories (Lindroth 1963b: 361), south to central California along the coast (Motschulsky, 1859a: 131, as Bembidion variolosum) and southern Colorado (LeConte 1879d: 501; Wickham 1902: 234). The record from “Alaska” (Hayward 1897: 102, as Bembidion variolosum) needs confirmation.
CAN: AB, BC, NT, SK USA: CA, CO, ID, MT, NV, OR, WA, WY [AK]
This species ranges from New Brunswick (Larochelle and Larivière 1990a: 29) to northwestern Wisconsin (Hayward 1897: 100; Messer 2010: 36; Barron County, CMNH), south to northeastern Ohio (Lake and Trumbull Counties, CMNH, Harry J. Lee pers. comm. 2008) and northeastern West Virginia (Hampshire and Tucker Counties, CMNH). The record from southwestern Iowa (Wickham 1911b: 6) is probably in error.
CAN: NB, ON, QC USA: CT, MA, ME, MI, NH, NJ, NY, OH, PA, RI, VT, WI, WV
[obtusangulum group]
This species has been recorded yet only from the type locality in southern Arizona.
USA: AZ
The range of this species extends from southern Manitoba (Lindroth 1963b: 357) to eastern Washington (Hatch 1953: 96, as Bembidion aberti), including southern Alberta (Lindroth 1963b: 357), south to Inyo County in eastern California (UASM), southeastern Arizona (Snow 1906b: 161; Casey 1884c: 67), east-central Colorado (Wickham 1902: 234), and west-central Kansas (Knaus 1905a: 218).
CAN: AB, MB, SK USA: AZ, CA, CO, KS, ND, NE, NV, OR, SD, UT, WA, WY
This rarely collected species is known only from the Colorado River valley in southeastern California to western New Mexico (Cibola County, CMNH).
USA: AZ, CA, NM
This species is known from southwestern California (Fall 1901a: 43; Moore 1937: 8).
USA: CA
This species ranges from the Okanagan Valley in southern British Columbia (Lindroth 1963b: 356) south to Inyo County in the Sierra Nevada of California (CMNH, MCZ, UASM) and southern Colorado (Elias 1987: 632). The record from southeastern South Dakota (Kirk and Balsbaugh 1975: 19) needs confirmation.
CAN: BC USA: CA, CO, ID, NV, OR, UT, WA, WY [SD]
The range of this species extends from southwestern Manitoba to the Okanagan Valley in British Columbia (Lindroth 1963b: 356), south to central California along the Sierra Nevada (Dajoz 2007: 18), central Colorado (LeConte 1878a: 465; Wickham 1902: 234) along the Rocky Mountains, and northwestern Nebraska (Sheridan County, CNC).
CAN: AB, BC, MB, SK USA: CA, CO, ID, MT, ND, NE, OR, UT, WA, WY
[patruele group]
This species is known from southeastern Manitoba to southwestern British Columbia (Lindroth 1963b: 365), south to eastern Washington (Hatch 1953: 96), northwestern Utah (Toole County, CMNH), south-central Colorado (Wickham 1902: 234; LeConte 1879d: 501; Lindroth 1963b: 365; Armin 1963: 165), central Nebraska (Blaine County, Foster F. Purrington pers. comm. 2010), and southeastern Wisconsin (Milwaukee County, Peter W. Messer pers. comm. 2009). The record from “Northwest Territories” (Bousquet and Larochelle 1993: 141) was based on four old specimens in the CNC labeled “N.W.T.” which does not refer to present day Northwest Territories or Nunavut.
CAN: AB, BC, MB, SK USA: CO, ID, MT, ND, NE, SD, UT, WA, WI, WY
This species is found from Skamania County in southern Washington (Hatch 1953: 93, as Bembidion stevensoni) to the San Diego area in southwestern California (LeConte 1852a: 187; Moore 1937: 7). The records from Nevada (Bechtel et al. 1983: 474), “British Columbia, ” “Arizona, ” “Dacota” (Hayward 1897: 99), and northern Idaho (Hatch 1953: 93) are likely based on misidentified specimens (see Lindroth 1963b: 359).
USA: CA, OR, WA
This species is known only from the type series collected in central California.
USA: CA
The range of this species extends from western Newfoundland to the Rocky Mountains in Alberta (Lindroth 1963b: 367), south to north-central Colorado (Motz and Morgan 2001: Fig. 4d), northern Nebraska (Cherry County, Foster F. Purrington pers. comm. 2010), central Iowa (Story County, CNC), western West Virginia (Cabell County, CMNH), and Maryland (Erwin 1981b: 147). The records from northern Oklahoma (French et al. 2001: 228; Elliott et al. 2006: 125) need confirmation.
CAN: AB, MB, NB, NF, NS, ON, QC, SK USA: CO, CT, IA, MA, MD, ME, MI, MT, ND, NE, NH, NY, OH, PA, SD, VA, VT, WI, WV [OK]
This species ranges from southern Manitoba to Vancouver Island (Lindroth 1963b: 358), north to the Great Slave Lake area in Northwest Territories (Bousquet 1987a: 121), south at least to Sierra County in the Sierra Nevada of California (MCZ; Lindroth 1963b: 358), the Grand Canyon in northern Arizona (CMNH), the Sacramento Mountains in southern New Mexico (CMNH), west-central Minnesota (Gandhi et al. 2005: 924), and central Wisconsin (Messer 2010: 36).
CAN: AB, BC (VCI), MB, NT, SK USA: AZ, CA, CO, ID, MN, MT, ND, NM, NV, OR, UT, WA, WI
According to Erwin (1984a: 169), this species is found throughout California.
USA: CA
This species, as far as known, is restricted to the San Francisco Bay area in California (Erwin 1984a: 170).
USA: CA
This species is found from western New Hampshire (Cooper 1976: 164) to eastern North Dakota (Grand Forks County, CNC), north to southern Manitoba (Lindroth 1963b: 362) and southern Saskatchewan (Ronald R. Hooper pers. comm. 2002), south to southern Arizona (Snow 1906b: 161; Coconino County, CMNH), including north-central Utah (Cache County, CMNH), the Big Bend National Park in western Texas (Dajoz 2007: 23), west-central Arkansas (Garland County, Robert L. Davidson pers. comm. 2012), northern Tennessee (Montgomery County, Foster F. Purrington pers. comm. 2010), and west-central West Virginia (Roane County, CMNH).
CAN: MB, ON, SK USA: AR, AZ, CO, IA, IL, IN, KS, MI, MO, ND, NE, NH, NM, NY, OH, OK, PA, SD, TN, TX, UT, VA, VT, WI, WV, WY
This species is found along the Pacific Coast from southwestern British Columbia, including Vancouver Island (Lindroth 1963b: 369), south at least to west-central Oregon (Casey 1918: 104).
CAN: BC (VCI) USA: OR, WA
This species is known only from the type locality in southwestern Utah.
USA: UT
The range of this species extends from western Ontario (Lindroth 1963b: 369) to central Yukon Territory (Dawson, CNC), south to south-central Oregon (Lake County, UASM), southern Arizona (Casey 1918: 118, as Bembidion lassulum), southern Colorado (Elias 1987: 632), and northeastern Wisconsin (Casey 1918: 108). The records from “Nebraska” and “South Dakota” (Bousquet and Larochelle 1993: 142) need confirmation.
CAN: AB, BC, MB, NT, ON, SK, YT USA: AZ, CO, MN, MT, OR, UT, WA, WI, WY [NE, SD]
According to Erwin (1984a: 174), the range of this species “extends throughout the western United States in the mountainous regions;” the only documented records are those of the type localities in California and that from Valley County in central Idaho (Maddison 2012: Supplementary content Table S1). David R. Maddison (pers. comm. 2012) caught the species in Steens Mountains, southeastern Oregon.
USA: CA, ID, OR
This species is found from Kupreanof Island in the Alexander Archipelago to southern California (Moore 1937: 7; Kavanaugh 1992: 67).
CAN: BC (QCI, VCI) USA: AK, CA (CHI), OR, WA
This species ranges from southern Manitoba to southern British Columbia though not reaching the coast, north to Fort Smith in southern Northwest Territories (Lindroth 1963b: 363), south to northern Baja California (CNC), southern Arizona (Casey 1918: 119, as Bembidion caliginosum; Maddison 2012: Supplementary content Table S1), southwestern New Mexico (Hidalgo County, CNC), and southeastern South Dakota (Kirk and Balsbaugh 1975: 19). The record from northwestern Texas (Merickel and Wangberg 1981: 157) needs confirmation.
CAN: AB, BC, MB, NT, SK USA: AZ, CA, MT, ND, NM, NV, OR, SD, UT, WA, WY [TX] – Mexico
This species occurs from the Saint Lawrence Plain in southern Quebec (Larochelle 1975: 57) to central British Columbia, north to east-central Alaska (Lindroth 1963b: 365), south to southern Colorado (Wickham 1902: 234; Bell 1971: 28), southern South Dakota (Kirk and Balsbaugh 1975: 18), southern Wisconsin (Messer 2010: 36), and southwestern Pennsylvania (Allegheny County, Robert L. Davidson pers. comm. 2008). The records from Missouri (Casey 1918: 109), southern Oklahoma (Hatch and Ortenburger (1930: 11), Texas (Wickham 1897: 104), Kansas (Popenoe 1877: 24; Snow 1878: 63), New Mexico (Fall and Cockerell 1907: 157), southern Arizona (Wickham 1897: 104), and “Delaware” (Bousquet and Larochelle 1993: 142) are probably in error.
CAN: AB, BC, MB, ON, QC, SK USA: AK, CO, MI, MN, MT, NY, OH, PA, SD, VT, WI, WY
This species is known only from the type locality in southwestern California.
USA: CA
This species is known only from the type series.
USA: AZ
This species ranges from Newfoundland (Lindroth 1955a: 67) to Vancouver Island, north to central Alaska and the Mackenzie River delta in northern Northwest Territories (Lindroth 1963b: 370-371), south to El Dorado County in eastern California (Maddison 1985: 113), “Utah” (Hayward 1897: 105), northern New Mexico (Taos County, UASM), eastern North Dakota (Tinerella 2003: 635), northeastern Wisconsin (Messer 2010: 36), and the upper peninsula of Michigan (Casey 1924: 35, as Bembidion concitatum); also found in eastern Siberia (Lindroth 1963b: 371) and Japan (Marggi et al. 2003: 254). The records from western Maine (Harvey and Knight (1897: 79), northeastern New York (Notman 1928: 216), “Pennsylvania” (Leng and Beutenmüller 1893: 136), “Illinois” (Bousquet and Larochelle 1993: 142), southern South Dakota (Kirk and Balsbaugh 1975: 18), and southern Oklahoma (Elliott et al. 2006: 125) are likely in error. Fossil remnants, dated between 15, 100 and 15, 300 years B.P., have been unearthed in central Iowa (Schwert 1992: 77).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS, NT, ON, PE, QC, SK, YT USA: AK, CA, CO, ID, MI, MN, MT, ND, NM, NV, OR, UT, WA, WI, WY – Holarctic
This species is yet known only from the lectotype.
USA: CA
This species is distinct from all other North American Bembidion (see Bousquet 1997b: 332) but only the original specimen is known. It is possible that the lectotype is mislabeled. Motschulsky (1859a: 130) added at the end of this species description “Je crois que cette espèce se rencontre aussi dans les possessions russes, notamment sur l’île Kenoi [Alaska]”.
This species is yet known only from the type locality and Santa Barbara County (Maddison 1985: 113) in western California.
USA: CA
This widely distributed species ranges from Newfoundland (Lindroth 1955a: 68) to western British Columbia, north to southern Northwest Territories (Lindroth 1963b: 372), south to Oregon (Hatch 1953: 94), southern Colorado (Elias 1987: 632), southern Texas (Kenedy County, CNC), northwestern Florida (Holmes County, UASM), and southern South Carolina (Ciegler 2000: 48).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NH, NJ, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV, WY
This species is known only from the type locality in southeastern Arizona.
USA: AZ
This species is known only from the California central valley (Erwin 1984a: 182).
USA: CA
This Holarctic species is found from Great Britain to eastern Siberia and Japan (Marggi et al. 2003: 254), and from the Arctic Plains in northern Alaska to the Hudson Bay shore in northwestern Ontario, south to southeastern Minnesota (Olmsted Country, CMNH), north-central Utah (Casey 1918: 105, as Bembidion provoanum), and northeastern California (Notman 1929b: 223, as Bembidion provoanum).
CAN: AB, BC, MB, NT, ON, SK, YT USA: AK, CA, MN, MT, OR, UT, WA – Holarctic
Elaphrus sturmii, credited to Duftschmid (1812: 203), is usually listed as junior synonym of this species (e.g., Marggi et al. 2003: 254). However, it is quite evident that Duftschmid (1812: 203) referred to Carabus sturmii Panzer listed on the preceding page (page 202).
This species is known from southern Texas (Johnson 1978: 67) and “Mexico” (Erwin 1984a: 185).
USA: TX – Mexico
[rapidum group]
This species is known from the Gila River drainage in southwestern New Mexico (David R. Maddison pers. comm. 2007) and southern Arizona (Casey 1918: 116, as Bembidion definitum; Cochise and Santa Cruz Counties, CMNH), south to San Luis Potosí in the Sierra Madre Oriental (Ball and Shpeley 1992a: 47, as Bembidion definitum). The record from Inyo County in eastern California (Riley 1893: 239) needs confirmation.
USA: AZ, NM [CA] – Mexico
This species is found from the Baja California Peninsula (Horn 1894: 308) and southeastern California (Casey 1918: 120; Andrews et al. 1979: 28) to southeastern Texas (Snow 1906a: 141; Casey, 1918: 121, as Bembidion amnicum) and northern Mexico (Erwin 1984a: 178). The record from “Utah” (Hayward 1897: 96) needs confirmation.
USA: AZ, CA, NM, TX [UT] – Mexico
The range of this species extends from Nova Scotia (Majka et al. 2007: 8) to the Rocky Mountains in Alberta (Lindroth 1963b: 364), south to southeastern Arizona (Dajoz 2007: 21; Cochise County, UASM), southern Texas (Johnson 1978: 67), northern Alabama (Madison County, CMNH), and eastern South Carolina (Ciegler 2000: 48), west along the southwest to southern California (Lindroth 1963b: 364; Los Angeles County, CMNH).
CAN: AB, MB, NB, NS, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV, WY
This species is known from the Gila River drainage in southwestern New Mexico (David R. Maddison pers. comm. 2007) and southeastern Arizona (Graham and Greenlee Counties, CMNH; Casey 1884b: 15, as Bembidion vinnulum), and from southeastern Mexico (Bates 1882a: 150).
USA: AZ, NM – Mexico
This taxon has been listed in synonymy with Bembidion aratum (LeConte) by Erwin (1982b: 479) but according to Maddison (2012: 535) it represents a distinct species.
[scudderi group]
This species is found from southern Manitoba to southern Alberta (Lindroth 1963b: 353), south to Mono County in east-central California (Dajoz 2007: 17), west-central Nevada (Bechtel et al. 1983: 474), central Colorado, and Nebraska (Hayward 1897: 88).
CAN: AB, MB, SK USA: CA, CO, ND, NE, NV, SD, UT, WY
This species ranges from southern Manitoba to central British Columbia (Lindroth 1963b: 354, as Bembidion henshawi), south to southern California (Hayward 1897: 88) and to the “Mexican transverse volcanic belt” (Erwin 1984a: 172).
CAN: AB, BC, MB, SK USA: CA, CO, MT, ND, NV, OR, UT, WA, WY – Mexico
This species occurs from northwestern Ontario (CNC) to central Alberta (Lindroth 1963b: 355), south to central Utah (Fall 1922c: 171), southeastern Wyoming (Albany County, MCZ), and east-central South Dakota (Kirk and Balsbaugh 1975: 20). The record from “Colorado” (Bousquet and Larochelle 1993: 140) needs confirmation.
CAN: AB, MB, ON, SK USA: MT, ND, SD, UT, WY [CO]
This species is found from southern Manitoba to the Okanagan Valley in British Columbia (Lindroth 1963b: 352), south to Kern County in southern California (Fall 1901a: 43), northern Utah (LeConte 1878a: 451; Casey, 1924: 40, as Bembidion spissicorne; Box Elder and Utah Counties, MCZ), and Yellowstone National Park in Wyoming (MCZ). The record from “Colorado” (Bousquet and Larochelle 1993: 140) needs confirmation.
CAN: AB, BC, MB, SK USA: CA, MT, ND, NV, OR, UT, WA, WY [CO]
Northern Hemisphere, with 16 species in the Nearctic (13 species) and Palaearctic (three species) Regions. The North American species are placed in two groups following Maddison (2012: 544).
Lindroth (1963b: 395-402) covered all North American species except for three (Bembidion scenicum, Bembidion clemens and Bembidion elizabethae).
[connivens group]
The range of this species extends from southern Manitoba to Vancouver Island, north to east-central Alaska (Lindroth 1963b: 400), south to western Oregon (Hatch 1953: 100, as Bembidion microreticulatum), southern Colorado (LeConte 1879d: 509; Wickham 1902: 235), and east-central South Dakota (Kirk and Balsbaugh 1975: 20).
CAN: AB, BC (VCI), MB, NT, SK USA: AK, CO, MT, ND, OR, SD, UT, WA, WY
This species is known from east-central Oregon (Union County, CMNH; Lindroth 1963b: 401) to southeastern California (Dajoz 2007: 20). The records from southwestern British Columbia and eastern Washington (Hatch 1953: 100) are probably in error (see Lindroth 1963b: 401).
USA: CA, OR
This species is found from the Saint Lawrence Estuary in Quebec (Larochelle 1975: 53) to south-central British Columbia (Jarrett and Scudder 2001: 381) and central Washington (Grant County, Foster F. Purrington pers. comm. 2011), north to south-central Northwest Territories (near Hay River, CNC), south to southern Utah (Garfield County, FFPC).
CAN: AB, BC, MB, NT, ON, QC, SK USA: MT, UT, WA
This species is known from western California (Casey 1918: 160, 161, 163 as Bembidion vapidum, Bembidion disparile, and Bembidion remotum), north-central Utah (Casey 1918: 159), central Arizona (Maddison 2012: Supplementary content Table S1), and northwestern New Mexico (Casey 1918: 162, as Bembidion invidiosum).
USA: AZ, CA, NM, UT
This species is found from Vancouver Island (Lindroth 1963b: 401) to northwestern Montana (Russell 1968: 58), south to central New Mexico (Fall and Cockerell 1907: 157) and southwestern California (Moore 1937: 8).
CAN: BC (VCI) USA: CA, CO, ID, MT, NM, OR, WA
This species is known from the type locality and Lincoln County in western Oregon (Maddison 2012: Supplementary content Table S1).
USA: OR, WA
This name has been listed in synonymy with Bembidion connivens (LeConte) by Lindroth (1963b: 401) but according to Maddison (2012: 535) it applies to a valid species.
This species is found from Cape Breton Island (Bousquet 1987d: 105) to south-central British Columbia, south to northern Idaho (Kootenai County, CNC), Missouri (Summers 1873: 147), and West Virginia (Pocahontas and Randolph Counties, CMNH). The records from “Washington, ” “Oregon, ” and “California” (Hayward 1897: 127, as Bembidion assimile) probably refer to Bembidion siticum; those from Colorado (Wickham 1902: 235) and New Mexico (Fall and Cockerell 1907: 157, as Bembidion assimile) need confirmation as they could also refer to Bembidion siticum.
CAN: BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, ND, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WI, WV [CO, NM]
This species has passed under the name Bembidion assimile Gyllenhal, 1810 until the 1950s.
This species is known only from the type locality in the Sierra Nevada.
USA: CA
This species is known from Vancouver Island (Lindroth 1963b: 402) to northwestern Montana (Russell 1968: 58), south to “Nevada” (Lindroth 1963b: 402) and Mendocino County in western California (Casey 1918: 157).
CAN: BC (VCI) USA: CA, ID, MT, NV, OR, WA
[fortestriatum group]
According to Lindroth (1963b: 396), this species is “probably restricted to C[entral] Calif[ornia].” It has been recorded so far from Placer (Casey 1918: 162, as Bembidion tersum), Mono (Dajoz 2007: 17), Nevada (Lindroth 1963b: 396), and San Francisco Counties (LeConte 1852a: 188).
USA: CA
This species ranges from Newfoundland (Lindroth 1955a: 77, as Bembidion anguliferum) to east-central Alaska, south to eastern Oregon (Harney County, UASM), southern South Dakota (Kirk and Balsbaugh 1975: 20), “Indiana” (Schrock 1985: 346), and western Virginia (Richard L. Hoffman pers. comm. 1992).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, IL, IN, MA, MD, ME, MI, MN, MT, ND, NH, NY, OH, OR, PA, RI, SD, VA, VT, WA, WI, WV
This species has passed under the name Bembidion anguliferum (LeConte, 1852) until the 1960s.
This species occurs from Newfoundland (Lindroth 1963b: 397) to central Alaska (Lindroth 1963b: 397), south to northeastern Oregon (Umatilla County, MCZ), western Montana (Russell 1968: 57), western Nebraska (Keith County, CMNH), and northeastern West Virginia (Tucker County, CMNH). The records from central New Mexico (Fall and Cockerell 1907: 157, as Bembidion cautum) and south-central Colorado (Wickham 1902: 235, as Bembidion cautum) probably refer to Bembidion connivens.
CAN: AB, BC (QCI, VCI), MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CT, ID, IL, IN, MA, ME, MI, MN, MT, NE, NH, NY, OR, PA, RI, VT, WA, WI, WV
This species is found from Nova Scotia (Maddison 2012: Supplementary content Table S1) to the Okanagan Valley in British Columbia (Lindroth 1963b: 399), north to southern Northwest Territories (CNC), south to northern Illinois (Purrington et al. 2002: 200; McHenry County, MCZ), southwestern Pennsylvania (Allegheny County, CMNH), and Connecticut (Lindroth 1963b: 398).
CAN: AB, BC, MB, NB, NS, NT, ON, QC, SK USA: CT, IL, MA, ME, MI, MN, NH, NY, OH, PA, VT, WI
Two North American species in the temperate regions.
Lindroth (1963b: 345-346) treated both species.
This species is confined to the west coast ranging from Vancouver Island (Lindroth 1963b: 345) to southern California (LeConte 1852a: 188; Fall 1901a: 42). The records from “Montana, ” “Wyoming, ” “Utah, ” “Arizona” (Hayward 1897: 111), and “Colorado” (Csiki 1928: 118) almost certainly refer to Bembidion salinarium Casey.
CAN: BC (VCI) USA: CA (CHI), OR, WA
The range of this species extends from southern Manitoba (Lindroth 1963b: 346) to the Okanagan Valley in south-central British Columbia (Bousquet 1987a: 121), south at least to Inyo County in California (MCZ), “Arizona” (Lindroth 1963b: 346), southern Colorado (Alamosa County, CNC), and northeastern North Dakota (Ramsay County, MCZ).
CAN: AB, BC, MB, SK USA: AZ, CA, CO, MT, ND, NV, OR, UT, WA, WY
Northern Hemisphere, with 21 species in the Nearctic (two species) and Palaearctic (19 species) Regions.
Lindroth (1963b: 381-382) treated both species found in North America.
This species ranges from central British Columbia to southwestern Manitoba (Lindroth 1963b: 382), south to northern Nebraska (Cherry County, Robert L. Davidson pers. comm. 2012), southwestern Colorado (Elias 1987: 632), and Mono County in east-central California (Dajoz 2007: 17).
CAN: AB, BC, MB, SK USA: CA, CO, MT, NE, NV, OR, SD, UT, WY
This species is confined to the Pacific Coast from southern Vancouver Island (Lindroth 1963b: 382) to southern California (LeConte 1852a: 189; Fall 1901a: 43). The record from northern Idaho (Hatch 1953: 95) is probably in error.
CAN: BC (VCI) USA: CA, OR, WA
Northern Hemisphere, with five species in the Nearctic (one Holarctic species) and Palaearctic (five species) Regions.
Lindroth (1963b: 297-298) covered the species found in North America.
This taxon is listed as a junior synonym of Trichoplataphus Netolitzky by some authors (e.g., Kryzhanovskij et al. 1995: 84). Toledano (2000: 36-37) discussed the taxonomic status of both taxa. According to Maddison (2012: 569), Blepharoplataphus is closely related to the subgenus Plataphus.
The range of this Holarctic species extends from Norway to the Far East (Marggi et al. 2003: 246) in the Palaearctic Region and from the Seward Peninsula in Alaska east to northern Labrador (Lindroth 1963b: 298).
CAN: BC, LB, MB, NT, ON, QC, YT USA: AK – Holarctic
Northern Hemisphere, with about 75 species in the Nearctic (36 species) and Palaearctic (45 species) Regions. Only two species (Bembidion hyperboraeorum and Bembidion prasinum Duftschmid) occur in Europe. Seven species are Holarctic, some of them represented by different subspecies in the Nearctic and Palaearctic Regions.
Lindroth (1963b: 268-297, as simplex, kuprianovii, incertum, and planiusculum groups) covered all the species found in North America except Bembidion falsum, Bembidion oppressum, Bembidion vandykei, and Bembidion placeranum. Subsequently to Lindroth’s (1963b) work, the names of five species were changed because of homonymy or synonymy: Bembidion flebile for Bembidion curtulatum, Bembidion lenense for Bembidion sulcipenne, Bembidion coerulescens for Bembidion neocoerulescens, Bembidion incertum for Bembidion breve, and Bembidion ochropus for Bembidion manningense. Species identifications are difficult and examination of the male genitalia is usually required for confirmation.
[planiusculum group]
The range of this species extends from “Nova Scotia” (Larochelle and Larivière 1990a: 28) to northeastern Ohio (Lee 1994: 58), south to western North Carolina (Swain County, CMNH) and south-central Tennessee (Grundy County, CMNH) along the Appalachian Mountains.
CAN: NS, QC USA: MA, ME, NC, NH, NY, OH, PA, TN, VA, VT, WV
This species ranges from Newfoundland (Lindroth 1955a: 55) to southern Quebec (Larochelle 1975: 54), south to northeastern Tennessee (Greene and Unicoi Counties, CMNH) and northeastern Georgia (Fattig 1949: 18) along the Appalachian Mountains.
FRA: PM CAN: NB, NF, NS (CBI), QC USA: GA, MA, ME, NC, NH, NY, TN, VA, VT
The range of this species extends from central Alaska to west-central Northwest Territories (Maddison 1985: 111, as Bembidion flebile), south at least to west-central California along the Coast Ranges (Casey 1918: 41, as Bembidion flebile) and to southwestern Colorado (Hinsdale County, CMNH; Casey 1918: 41, as Bembidion decrepitum) along the Rocky Mountains.
CAN: AB, BC (VCI), NT, YT USA: AK, CA, CO, ID, MT, OR, WA
Maddison (2012: 535) noted that there at least two species within the current concept of this species.
This species is found along the Cascade Range and Coastal Ranges (Erwin 1984a: 172) from northwestern Washington to central California.
USA: CA, OR, WA
This subspecies ranges from southern Yukon Territory (Lindroth 1963b: 291) to northern Colorado along the Rocky Mountains (Casey 1918: 32) and to “California” (Lindroth 1963b: 291).
CAN: AB, BC (VCI), YT USA: CA, CO, ID, MT, UT, WA, WY
Three other subspecies of Bembidion gebleri Gebler live in eastern Asia.
This mountain species is known from southern British Columbia south at least to central Oregon along the Cascade Range (Lindroth 1963b: 286), west to Gallatin County in southern Montana (Maddison 2012: Supplementary content Table S1).
CAN: BC USA: MT, OR, WA
The range of this species extends from the Alaska Peninsula to southern Yukon Territory (Lindroth 1963b: 290), south at least to Ouray County in southwestern Colorado (CNC) along the Rocky Mountains (Lindroth 1963b: 289) and to the Sierra Nevada in eastern California (Dajoz 2007: 16).
CAN: AB, BC, YT USA: AK, CA, CO, MT, OR, UT, WA
This Holarctic species ranges from Scandinavia to eastern Siberia and the Far East (Marggi et al. 2003: 265) and from the Seward Peninsula in Alaska (Lindroth 1963b: 295) to the Hudson Bay in northeastern Manitoba (Garry 1993: 95).
CAN: MB, NT, NU, YT USA: AK – Holarctic
This species is known only from a few localities in central and west-central British Columbia (Lindroth 1963b: 286).
CAN: BC
This species is known from the Coast Ranges from Humboldt County (Notman 1929b: 222) to central California (Erwin 1984a: 178).
USA: CA
This species is known from the Kenai Peninsula in Alaska (Lindroth 1963b: 288) south to northern Washington (Glesne et al. 2000: 89) and central Alberta (Maddison 1985: 113). All records prior to Lindroth (1963b) listed by Bousquet and Larochelle (1993: 132) need confirmation.
CAN: AB, BC (QCI, VCI) USA: AK, WA [CO, MT, NM, OR, UT, WY]
The range of this species extends from the Haines district in southeastern Alaska to southeastern Yukon Territory (Lindroth 1963b: 287), south to western Montana (Russell 1968: 51) along the Rocky Mountains and foothills and to “California” (Lindroth 1963b: 287). The record from “Idaho” (Bousquet and Larochelle 1993: 132) needs confirmation.
CAN: AB, BC, YT USA: AK, CA, MT [ID]
This subspecies occurs from northern Alaska to southeastern Yukon Territory (Lindroth 1963b: 293), south to northwestern Montana (Edwards 1975: 54, as Bembidion rusticum) and central British Columbia (Lindroth 1963b: 293).
CAN: AB, BC, YT USA: AK, MT
This subspecies is found from Newfoundland (Lindroth 1955a: 54) to the Lake Superior area in western Ontario (Lindroth 1963b: 293), south to north-central Pennsylvania (Lycoming County, Robert L. Davidson pers. comm. 2008; Lindroth 1955a: 54).
FRA: PM CAN: LB, NB, NF, NS (CBI), ON, QC USA: MA, ME, NH, NY, PA, VT
This subspecies is reported from eastern Siberia and the Far East (Marggi et al. 2003: 266) but the specimens probably belong to a distinct (still undescribed) subspecies as pointed out by Lindroth (1963b: 293).
This species is known from a few mountain locations in the Similkameen district in southern British Columbia (Lindroth 1963b: 286) and from the Sierra Nevada in California (Casey 1924: 28; Maddison 1985: 114).
CAN: BC USA: CA
The range of this species extends from western Maine (Oxford County, MCZ; Larochelle and Larivière 1990a: 28) to southeastern Wisconsin (Rauterberg 1885: 22; Messer 2010: 36), including the Appalachian region in southern Quebec (Larochelle 1975: 62), south to Tennessee (Sevier County, CMNH) and North Carolina (Hayward 1897: 64; Brimley 1938: 117; Swain County, CMNH) along the Appalachian Mountains. The records from “Labrador, ” “Hudson Bay Territory, ” and “Missouri” (Hayward 1897: 64) refer to other species.
CAN: QC USA: CT, MA, ME, MI, NC, NH, NY, OH, PA, TN, VA, VT, WI, WV
Lindroth (1963b: 268) included this species in its own group. However, I believe the species is closely related to some of the species included by Lindroth (1963b) in his planiusculum group.
This species ranges from southwestern British Columbia, including Vancouver Island (Lindroth 1963b: 290), to southwestern Oregon (Hatch 1950: 98).
CAN: BC (VCI) USA: OR, WA
This subspecies is known from Alaska, including the arctic zone and Kodiak Island, southeastern Yukon Territory, and adjacent northern British Columbia (Lindroth 1963b: 294).
CAN: BC, YT USA: AK
This subspecies ranges from Newfoundland (Lindroth 1955a: 52, as Bembidion lenense) to southwestern Alaska (Elias 1988: 41), south to southern British Columbia (Lindroth 1963b: 294) and the Saint Lawrence Estuary near Quebec City (Larochelle 1975: Fig. 175).
CAN: AB, BC, LB, MB, NB, NF, NS, QC USA: AK
This species is known from the California west coast region as far south as the San Francisco area (Casey 1918: 40, as Bembidion vespertinum).
USA: CA
[complanulum group]
This Holarctic species ranges from western Siberia (Marggi et al. 2003: 265) to the Dempster Highway in Yukon Territory (Maddison 1985: 111; UASM), including the Aleutian Islands and Alaska Peninsula (Lindroth 1963b: 275), south to Saint Elias Mountains in northwestern British Columbia (Jarrett and Scudder 2001: 380).
CAN: BC, YT USA: AK – Holarctic
This species is found from eastern Siberia (Marggi et al. 2003: 265) to the Hudson Bay coast in northern Manitoba (Holliday 1982: 116), including the Great Slave Lake area in Northwest Territories (Lindroth 1963b: 279).
CAN: MB, NT, YT USA: AK – Holarctic
The range of this species extends from the Aleutian Islands and the Gulf of Alaska coast to southwestern Yukon Territory (Lindroth 1963b: 273, as Bembidion incertum), south to north-central New Mexico (Fall and Cockerell 1907: 157; Hayward 1897: 68, as Bembidion incertum) along the Rocky Mountains and to the Sierra Nevada in eastern California (Casey 1918: 30, as Bembidion testatum).
CAN: AB, BC (QCI), YT USA: AK, CA, CO, ID, MT, NM, NV, OR, WA, WY
The range of this species extends from the Aleutian Islands and the Alaska Peninsula (Lindroth 1963b: 283) south along the Rocky Mountains to northwestern Montana (Russell 1968: 51; Edwards 1975: 51) and to the central Sierra Nevada in California (Papp 1978: 164). The records from “Northwest Territories” (Bousquet and Larochelle 1993: 130) and from the San Juan River drainage in southwestern Colorado (Wickham 1902: 232) are probably in error.
CAN: AB, BC (QCI) USA: AK, CA, MT, OR, WA
This Holarctic species is found from eastern Siberia (Marggi et al. 2003: 265) to the Hudson Bay in northeastern Manitoba (Garry 1993: 95), including northern Alberta (Birch Mountains, Gerald J. Hilchie pers. comm. 2009). Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96).
CAN: AB, MB, NT, NU, YT USA: AK – Holarctic
This species is known from the Kenai Peninsula in Alaska (Lindroth 1963b: 278) south to “Oregon” (Hatch 1953: 84) and from north-central Colorado (Lindroth 1963b: 278).
CAN: BC (QCI) USA: AK, CO, OR, WA
This species ranges from British Columbia, as far north as the Prince Rupert area (Lindroth 1963b: 274), to Mendocino County in western California (Maddison 1985: 112) and to southern Colorado along the Rocky Mountains (Elias 1987: 632).
CAN: BC (VCI) USA: CA, CO, ID, MT, OR, WA
This species is known from Mount Rainier in Washington (Lindroth 1963b: 283) and the Sierra Nevada in California (Casey 1924: 25; Papp 1978: 164).
USA: CA, WA
This species ranges from central Alaska including the Alaska Peninsula and Kodiak Island (Lindroth 1963b: 270) to western Northwest Territories (Tungsten, UASM), south at least to northern Utah (LeConte 1878a: 465, as Bembidion maeklini), including western Montana (Russell 1968: 50; Edwards 1975: 52) and north-central Wyoming (Big Horn County, Ken Karns pers. comm. 2009; Hamilton 1894a: 7, as Bembidion funerum [sic]), and the Sierra Nevada in California (Casey 1918: 33, as Bembidion adultum and Bembidion dilutum). The record from southwestern Colorado (Wickham 1902: 233, as Bembidion maeklini) needs confirmation.
CAN: AB, BC, NT, YT USA: AK, CA, ID, MT, NV, OR, UT, WA, WY [CO]
This species is known from Mount Rainier in Washington and the Sierra Nevada in California (Lindroth 1963b: 274). The record from British Columbia (Jarrett and Scudder 2001: 381) needs confirmation.
USA: CA, WA [BC]
This species ranges from the Yukon River in western Alaska to the Mackenzie River delta in northwestern Northwest Territories (Lindroth 1963b: 278-279, as Bembidion ochropus), south through the Rocky Mountains to western Montana (Russell 1968: 51) and mountains in southern British Columbia (Lindroth 1963b: 279, as Bembidion ochropus).
CAN: AB, BC, NT, YT USA: AK, MT
This species is known yet only from the San Francisco Bay area in California.
USA: CA
This species is known from southeastern British Columbia (Syringa Provincial Park, James C. Bergdahl pers. comm. 2008) south at least to northern California (Casey 1924: 26, as Bembidion expansipenne) and northwestern Montana (Edwards 1975: 53).
CAN: BC USA: CA, MT, OR, WA
The range of this species extends from Newfoundland (Lindroth 1955a: 51) to southeastern Alberta (Lindroth 1963b: 282), south to east-central Minnesota (Aitkin County, CMNH) and mountains in New York and New England (Lindroth 1963b: 282). The four specimens labeled from the Anchorage area in southern Alaska (Lindroth 1963b: 282) could be mislabeled in my opinion. The record from “Pennsylvania” (Bousquet and Larochelle 1993: 131) needs confirmation.
CAN: AB, LB, NB, NF, NS (CBI), ON, QC, SK USA: ME, MI, MN, NH, NY, VT [AK, PA]
According to Erwin (1984a: 179), this species is found in the foothills of the Sierra Nevada in California.
USA: CA
This species ranges from the Aleutian Islands and Alaska Peninsula (Lindroth 1963b: 276) to south-central Yukon Territory, south to the Sierra Nevada in California (Papp 1978: 164; Dajoz 2007: 16) and at least to northwestern Montana along the Rocky Mountains (Russell 1968: 50; Edwards 1975: 53).
CAN: AB, BC (QCI), YT USA: AK, CA, MT, OR, WA
This species is known from a few localities in northwestern Montana (Russell 1968: 51), southwestern Alberta, and southeastern British Columbia (Lindroth 1963b: 277).
CAN: AB, BC USA: MT
This species is known from the Queen Charlotte Archipelago and adjacent mainland in British Columbia (Kavanaugh 1992: 65) and from northwestern Washington (Skagit County, James C. Bergdahl pers. comm. 2008).
CAN: BC (QCI) USA: WA
Northern Hemisphere, with ten species in the Nearctic (four species) and Palaearctic (six species) Regions.
Lindroth (1963b: 250-254, as nitidum and levigatum groups) covered all four North American species.
Habu and Uéno (1955: 45) described the subgenus Aptenidium (one Japanese species) in the genus Hydrium implying (though not discussed by the authors) a close relationship between the two genus-group taxa. Based on the original description, Aptenidium is likely not closely related to Hydrium.
[levigatum group]
This widely distributed species ranges from east-central Maine (Majka et al. 2011: 45) to “Montana” (Hayward 1897: 40) and southeastern Alberta (CNC), south to eastern Utah (Grand County, CNC), the Rio Grande in western Texas (Brewster County, CNC) and northern Chihuahua (Bates 1891a: 263), and the Florida Panhandle (Peck and Thomas 1998: 18).
CAN: AB USA: AL, AR, CO, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NM, NY, OH, OK, PA, SC, SD, TN, TX, UT, VA, WI, WV – Mexico
[nitidum group]
This species occurs from the Laurentides region in western Quebec (Larochelle 1975: 57) to central Alaska (Lindroth 1963b: 253), south at least to northern Oregon (Morrow County, CMNH), northern Colorado (Eagle County, CMNH) along the Rocky Mountains, and southwestern North Dakota (Morton County, UASM).
CAN: AB, BC, MB, NT, ON, QC, SK, YT USA: AK, CO, ID, MT, ND, OR, WA, WY
The range of this species extends from Prince Edward Island (Majka et al. 2008: 130) and New Brunswick (Webster and Bousquet 2008: 16) to eastern Alaska (Lindroth 1969a: 1114), south to eastern Idaho (Clark County, UASM), southern Colorado (Wickham 1896c: 132; Maddison 1985: 95) along the Rocky Mountains, “Kansas” (Wickham 1896c: 132), Missouri (Summers 1873: 147), northwestern Indiana (Blatchley 1910: 69), and New York (Notman 1928: 214). One old specimen simply labeled “N.M.” is known (MCZ). The record from “New Jersey” (Wickham 1896c: 132) needs confirmation.
CAN: AB, BC, MB, NB, NT, ON, PE, QC, SK, YT USA: AK, CO, IA, ID, IN, KS, MA, ME, MI, MN, MO, MT, ND, NE, NH, NY, OH, SD, VT, WI, WY [NJ, NM]
This species is known from scattered localities from northern Idaho (Bonner County, MCZ) to west-central Washington (Grays Harbor County, CMNH), south to southern California (Ventura County, CMNH). The record from British Columbia (Jarrett and Scudder 2001: 381) needs confirmation.
USA: CA, ID, OR, WA [BC]
Northern Hemisphere, with seven species in the Nearctic (three species, of which two are adventive) and Palaearctic (six species) Regions.
Lindroth (1963b: 254-258, as lampros group) covered all three species found in North America.
All recent authors seen (e.g., Marggi et al. 2003: 251; Lorenz 2005: 218; Ortuño and Toribio 2005: 278; Maddison 2012: 545) used Metallina as the valid name for this subgenus despite that Leja is the oldest available name and is not a junior homonym of any genus-group names. I am following them but the case needs to be submitted to the Commission for a ruling.
According to Maddison (2012: 569), this group is closely related to subgenus Hydrium.
This species ranges from southeastern Alaska to southern Yukon Territory (Lindroth 1963b: 256), south along the Rocky Mountains to central Colorado (Wickham 1902: 234; Hayward 1897: 115; Casey 1918: 115, as Bembidion perturbatum) and to the Sierra Nevada in California (Dajoz 2007: 17). Fossil remnants from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 24).
CAN: AB, BC (QCI, VCI), YT USA: AK, CA, CO, ID, MT, OR, UT, WA, WY
This Palaearctic species is adventive in North America where it is known from southern British Columbia (Hatch 1953: 83; Smith et al. 2004: 96) to northwestern Oregon (Westcott et al. 2006: 7) in the west and from Newfoundland (Lindroth 1955a: 49; Larson and Langor 1982: 592) in the east. The first inventoried specimen collected in the eastern part of this continent was found in 1949 (see Lindroth 1955a: 49) and in the western part in 1947 (Hatch 1949c: 145). Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 24).
CAN: BC, NF USA: OR, WA – Adventive
Carabus pygmaeus attributed to Paykull (1798: 148) is often listed as a junior synonym of this species. However, Paykull did not describe a new species under that name since he referred to Carabus pygmaeus Fabricius, 1792.
This Palaearctic species is adventive in North America where it is known from Newfoundland (Larson and Langor 1982: 592) to the coastal area of New Brunswick (Bousquet 1987a: 120), including the Magdalen Islands in the Gulf of Saint Lawrence (Larochelle 1975: 60), and from southeastern Maine (Larochelle and Larivière 1990a: 28, 33). One specimen has been collected recently in a campground in Kittitas County, central Washington (Robert L. Davidson pers. comm. 2008). The first inventoried specimen collected on this continent was found in Nova Scotia in 1942 (Majka et al. 2007: 7).
CAN: NB, NF, NS (CBI), PE, QC USA: ME, WA – Adventive
One species in western North America.
The species is described in detail, under the name Bembidion carlhi, by Erwin and Kavanaugh (1981: 37, 39).
This species is known from the Cascade Range in southwestern Oregon south to central California along the Coast Ranges (Casey, 1918: 44, as Bembidion delectum), including the Klamath Mountains, and along the Sierra Nevada [see Erwin and Kavanaugh 1981: Fig. 18, as Bembidion carlhi]; also recorded from “Nevada” (Hayward 1897: 112).
USA: CA, NV, OR
Eleven species in the Nearctic (five species) and Palaearctic (seven species) Regions. One species (Bembidion incrematum) is Holarctic.
Bousquet and Webster (2006) provided a key for the identification of all North American species. Lindroth (1963b: 348-351, 360) covered four species, three he placed in the incrematum group, the other (Bembidion variegatum) in the dorsale group.
The range of this species extends from Nova Scotia (CNC) to eastern Minnesota (Epstein and Kulman 1990: 214), south to east-central Iowa (Johnson County, USNM), west-central Indiana (Blatchley 1910: 76), and southeastern West Virginia (Greenbrier County, MCZ). The record from southern South Dakota (Kirk and Balsbaugh 1975: 19) needs confirmation.
CAN: NB, NS, ON, QC USA: CT, IA, IL, IN, MA, ME, MD, MI, MN, NH, NJ, NY, OH, PA, RI, VA, VT, WI, WV [SD]
This species occurs from Newfoundland (Lindroth 1955a: 66) to southeastern Ontario (Ottawa, CNC), south to New England and the Adirondack Mountains in northeastern New York (Lindroth 1963b: 349). The record from eastern Iowa (Cooper 1976: 163) needs confirmation.
CAN: NB, NF, NS (CBI), ON, QC USA: ME, NH, NY, VT [IA]
This species ranges from Newfoundland (Lindroth 1955a: 64) to east-central Alaska (Lindroth 1963b: 349), south at least to Sonoma County in western California (Casey, 1918: 96, as Bembidion nubiferum), northeastern New Mexico (Mora County, Foster F. Purrington pers. comm. 2012), east-central Ohio (Usis and MacLean 1998: 67), the Adirondack Mountains in New York (Notman 1928: 217, as Bembidion arcuatum), and southeastern Maine (Washington County, CNC); also known from eastern Siberia (Marggi et al. 2003: 250). The records from “Massachusetts” (Bousquet and Larochelle 1993: 139), “Pennsylvania, ” and “West Virginia” (Hamilton 1894a: 8, as Bembidion dentellum) need confirmation.
FRA: PM CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CA, CO, ID, ME, MI, MT, ND, NH, NY, OH, OR, SD, VT, WA, WI [MA, PA, WV] – Holarctic
This species has passed for a long time under the name Bembidion dentellum (Thunberg, 1787).
This species ranges from New Brunswick to southwestern Quebec, south to Virginia (Bousquet and Webster 2006: 29).
CAN: NB, QC USA: NH, PA, VA, VT
The range of this species extends from New Brunswick (CNC) to eastern South Dakota (Kirk and Balsbaugh 1975: 19), south to western (Dajoz 2007: 23) and east-central (Tucker 1906: 85) Texas, southwestern Alabama (Baldwin County, CMNH), northeastern Georgia (Leng 1910: 73; Fattig 1949: 17), and eastern South Carolina (Ciegler 2000: 49). The record from Colorado (Wickham 1902: 234) needs confirmation; that from British Columbia (Jarrett and Scudder 2001: 382) was based on misidentified specimens of Bembidion patruele, Bembidion graphicum, and Bembidion nigripes (UBC).
CAN: NB, ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [CO]
This species has been listed in the subgenus Notaphus by Lindroth (1963b: 360) but placed in the subgenus Eupetedromus by Bousquet and Webster (2006: 30).
Western Nearctic Region, with two species.
Both species are included in Lindroth’s (1963b: 266-267) monograph.
This species ranges from central Alberta (Lindroth 1963b: 267) to the Queen Charlotte Islands (Kavanaugh 1992: 64), south to southern California (Fall 1901a: 10; Moore 1937: 6) and northern Utah (Knowlton 1939: 2; Davis and Salt Lake Counties, CMNH).
CAN: AB, BC (QCI, VCI) USA: CA (CHI), ID, MT, NV, OR, UT, WA
This species is known from southern California (Fall 1901a: 43; Moore 1937: 6; Maddison 2012: Supplementary content Table S1). The record from “Washington” (Hayward 1897: 128) was based on a misidentified Bembidion iridescens (Lindroth 1963b: 267).
USA: CA
Three western North American species.
Lindroth (1963b: 343-345) treated two species (Bembidion mundum and Bembidion horni) and listed the other one, based on the original description alone, as synonym of Bembidion mundum. Later he rectified the synonymy (Lindroth 1969a: 1115).
This species has been recorded from Benton and Jackson Counties in western Oregon (Westcott et al. 2006: 7), from Humboldt and Yolo Counties in northern California (Maddison 2012: Supplementary content Table S1), and from southwestern California (Casey 1918: 45; Moore 1937: 6).
USA: CA, OR
This species is known from southern California (Fall 1901a: 43; Moore 1937: 6), northern Arizona (Navajo and Apache Counties, UASM; Hayward 1897: 117), southern Utah (San Juan and Kane Counties, MCZ, UASM), and western New Mexico (Maddison 2012: Supplementary content Table S1). The records from “Washington” and “Oregon” (Bousquet and Larochelle 1993: 139) are likely in error.
USA: AZ, CA, NM, UT
This species is found from southwestern British Columbia, including Vancouver Island (Lindroth 1963b: 344), south to southern California (Fall 1901a: 43, as Bembidion bifasciatum) and northern Arizona (Snow 1906b: 161; Coconino County, CMNH), including southwestern (Tanner 1928: 269, as Bembidion bifasciatum) and eastern Utah (Grand County, Foster F. Purrington pers. comm. 2010).
CAN: BC (VCI) USA: AZ, CA, NV, OR, UT, WA
Northern Hemisphere, with two species in the Nearctic (Bembidion planatum) and Palaearctic (Bembidion altaicum Gebler in Asia) Regions.
The North American species is treated in Lindroth’s (1963b: 284) monograph.
This subgenus was placed in synonymy with Plataphus Motschulsky by Netolitzky (1943b: 61) and Lindroth (1963b: 283) but considered a valid taxon by Toledano (2008: 12). According to Maddison (2012: 569), Melomalus is not closely related to the subgenus Plataphus.
This species ranges from Newfoundland (Lindroth 1955a: 52) to the Norton Sound area in Alaska (Lindroth 1963b: 284), south to central California along the Coast Ranges, southern Colorado along the Rocky Mountains, Isle Royale in northernmost Michigan (Hubbard and Schwarz 1878: 629), and Cape Breton Island [see Lindroth 1963a: Fig. 63]. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 24).
CAN: AB, BC (VCI), NB, NF, NS (CBI), NT, ON, QC, YT USA: AK, CA, CO, ID, MI, MT, NV, OR, UT, WA, WY
Northern Hemisphere, with 22 species in the Nearctic (five eastern species) and Palaearctic (17 Asian species) Regions (see Toledano and Schmidt 2010).
Four of the North American species are included in Lindroth’s (1963b: 212-229) key to species of Bembidion; three of these were treated in detail (Lindroth 1963b: 298-300). One new species was described subsequently by Hildebrandt and Maddison (2011) who provided a modified version of Lindroth’s key to include the new species.
This eastern species ranges from “Vermont” (Hayward 1897: 72) and southwestern Massachusetts (Hampden County, MCZ) to “Illinois” (Hayward 1897: 72), south to northern and eastern Tennessee (Cheatham, Fentress, and Sevier Counties, CMNH, MCZ). The record from southern Wisconsin (Rauterberg 1885: 22) needs confirmation; those from Missouri (Summers 1873: 147) and the Rocky Mountains in northern Colorado (Wickham 1902: 232) must be in error.
USA: DC, IL, IN, MA, MD, MI, NJ, NY, OH, PA, TN, VA, VT [WI]
This species is known from central Iowa (Maddison 2012: Supplementary content Table S1), northeastern Kansas (Pottawatomie County, CMNH; Lindroth 1963b: 218; Knaus 1903: 188), eastern Oklahoma (Latimer County, UASM) and central Texas (Blanco County, UASM; Hayward 1897: 70). According to Hildebrandt and Maddison (2011: 273), the records from Massachusetts, “New Jersey, ” “Pennsylvania, ” the District of Columbia (Hayward 1897: 70), and New York (Notman 1928: 216) are likely incorrect and may be based on Bembidion fugax.
USA: IA, KS, OK, TX
This species is known from the Ozark Plateau of Missouri and Arkansas [see Hildebrandt and Maddison 2011: Fig. 6].
USA: AR, MO
The range of this eastern species extends from Nova Scotia (Lindroth 1954c: 302) to “Minnesota” (Hildebrandt and Maddison 2011: 274), south to eastern Oklahoma (Le Flore County, FFPC ), northern Arkansas (Lindroth 1969a: 1114; Pope, Searcy, and Washington Counties, CMNH), northern Mississippi (Drew A. Hildebrandt pers. comm. 2010), central Alabama (Jefferson County, Drew A. Hildebrandt pers. comm. 2009), northern Georgia (Fattig 1949: 17), and northwestern South Carolina (Ciegler 2000: 48).
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, VA, VT, WI, WV
This eastern species ranges from Nova Scotia (Larochelle and Larivière 1990a: 28; Maddison 2012: Supplementary content Table S1) to the Ontario Peninsula (Lindroth 1963b: 300) and northern Ohio (Ashtabula, Cuyahoga, Erie, and Lorain Counties, Harry J. Lee, Jr. pers. comm. 2008), south to west-central Tennessee (Hickman County, CMNH) and northeastern Virginia (Richard L. Hoffman pers. comm. 1992).
CAN: NB, NS, ON, QC USA: DC, KY, MA, MD, ME, NJ, NY, OH, PA, TN, VA, VT, WV
West Palaearctic Region, with nine species of which one is adventive in eastern North America.
The species found in North America was covered in Lindroth’s (1963b: 258) monograph.
Maddison (2012: 561) noted that this taxon may be the sister-group of the remaining Bembidion.
This Palaearctic species is adventive in North America where it is known from Prince Edward Island (Majka et al. 2008: 130), and from western Quebec, as far north as the Abitibi region (Paquin and Dupérré 2002: 86), to the Algoma District in northern Ontario (Pearce et al. 2003), south to southern Michigan (Purrington et al. 2002: 200), northeastern Ohio, and Long Island, New York [see Hoebeke et al. 1991: Fig. 1]. The first inventoried specimen collected on this continent was found in the Great Lake region in Ontario in 1956 (Lindroth 1963b: 258).
CAN: ON, PE, QC USA: OH, MI, NY, PA, VT – Adventive
Northern Hemisphere, with four coastal species in North America (two species, one endemic along the Pacific and one adventive species along the Atlantic) and Europe (three species: Bembidion abeillei Bedel, Bembidion eichleri Marggi, Wrase and Huber, and Bembidion nigropiceum Marsham).
Both species found in North America are included in Lindroth’s (1963b: 213) key to the Canadian and Alaskan Bembidion.
This species occurs on the seashore along the coast of California.
USA: CA
This adventive species was known on this continent until recently only from the four specimens upon which Hayward (1897: 129) described his Bembidion puritanum. Erwin and Kavanaugh (1980: 241) stated that the species was probably “accidentally introduced into Massachusetts from England through shipping in the late 1800s, that a population was sustained for a short time, but that it is not likely to have persisted to the present.” However, more than 80 specimens were collected between 2007 and 2010 in Boston Harbor, Suffolk County, Massachusetts (Davidson and Rykken 2011: 491).
USA: MA – Adventive
One species in western North America.
Lindroth (1963b: 407-408) covered the species in his monograph of the Canadian and Alaskan Carabidae.
Molecular data analyses presented by Maddison and Ober (2011) and Maddison (2012) strongly suggest that this genus is not closely related to the other members of Bembidiina. However, its relationship is not established. Because there are no family-group name proposed for Phrypeus, the genus is left for lack of anything better in the subtribe Bembidiina.
This species ranges from Vancouver Island (Lindroth 1963b: 408) to western Montana (Russell 1968: 58), south to Nevada County in the Sierra Nevada (Dajoz 2007: 16) and to Sonoma County along the Pacific Coast (Lindroth 1963b: 408).
CAN: BC (VCI) USA: CA, MT, OR, WA
Phrypeus rickseckeri (Hayward). Thomas Casey is well known to North American coleopterists for the numerous species he proposed that are considered invalid today. His extremely rigid species concept allowed almost no morphological variation. He was, however, more successful at the generic level and many of his taxa are still recognized as valid today. One of these is Phrypeus which he proposed for a small, peculiar bembidiine species living along river banks in the coastal region of western North America. Associated with the subtribe Bembidiina for a long time, recent molecular analyses have revealed that the species is not closely related to that group.
New World, with about 125 species arrayed in seven genera: Erwiniana Paulsen and Smith (about 55 Neotropical species), Geballusa Erwin (five Neotropical species), Gouleta Erwin (four Neotropical species), Inpa Erwin (one South American species), Mioptachys (13 species), Philipis Erwin (38 Australian species), and Xystosomus Schaum (nine Neotropical species). The North American fauna is represented by a single species.
Western Hemisphere, with 13 species in temperate, subtropical, and tropical areas of the Nearctic (one species) and Neotropical (12 species) Regions.
The North American species is treated in Lindroth’s (1966: 441) monograph under the genus Tachys.
This species ranges from the Nova Scotia Peninsula (Christopher G. Majka pers. comm. 2007) to western Washington (Hatch 1953: 104), north to southern British Columbia (Lindroth 1966: 441) and southern Alberta (CNC), south to southwestern California (Los Angeles County, CAS), southeastern Arizona (Dajoz 2007: 21), south-central Texas (Bastrop County, CNC), and southern Florida (Peck and Thomas 1998: 18).
CAN: AB, BC, NB, NS, ON, QC USA: AL, AR, AZ, CA, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV
Mioptachys flavicauda (Say). This small species is a characteristic element of the fauna that lives under the bark of dead deciduous trees. Despite its size, it is easily recognized in the field by its conspicuous yellow apical third of the elytra. Thomas Say, the first entomologist born on this continent worthy of the name, was struck by the coloration and for that reason proposed the name flavicauda, derived from the Latin flavus (yellow) and cauda (tail), for the species.
Worldwide, with about 790 species (Lorenz 2005: 207-215). The Northern Hemisphere is represented by about 230 species (roughly 29% of the world fauna) and North America by 73 species (9.2% of the world fauna). Two species are adventive (Porotachys bisulcatus and Elaphropus parvulus) in the Nearctic Region. No species are Holarctic.
The genitive form of tachys is tachéos (see Alonso-Zarazaga 2007) and the family-group name based on the genus Tachys should be spelled Tacheina. However the spelling Tachyina being in prevailing usage, it is maintained (ICZN 1999: Article 29.5).
Worldwide, with 24 species in boreal, temperate, subtropical, and tropical areas. The species are arrayed in two subgenera: Paratachyta Erwin (five species in the Eastern Hemisphere) and Tachyta s.str. (19 species).
Erwin (1975) revised the species and provided a key for their identification.
Worldwide, with 19 species in the Nearctic (five species), Neotropical (three species, only one of them endemic), Australian (five species), Oriental (four species), Palaearctic (two species), and Afrotropical (four species) Regions.
[falli group]
This species ranges from Cape Breton Island (Lindroth 1954c: 303) to west-central Yukon Territory, south to Veracruz in Mexico, northeastern Mississippi (Oktibbeha County, Drew A. Hildebrandt pers. comm. 2009), and northern Georgia [see Erwin 1975: Fig. 121]. The record from “Oregon” (Bousquet and Larochelle 1993: 149) needs confirmation.
CAN: AB, BC, MB, NB, NS (CBI), ON, QC, SK, YT USA: AZ, CT, DC, GA, IN, MA, MD, ME, MI, MN, MO, MS, MT, NC, NH, NJ, NM, NY, PA, RI, SC, VA, VT, WI, WV [OR] – Mexico
This western species ranges from southwestern British Columbia, including Vancouver Island, to western Idaho, south to Fresno County in California along the Sierra Nevada and to the San Francisco Bay area along the Coast Ranges [see Erwin 1975: Fig. 111].
CAN: BC (VCI) USA: CA, ID, OR, WA
[inornata group]
This species ranges from Maine and southern Quebec to south-central British Columbia, south to southwestern California, Belize and Guatemala, and southern Florida, including the Keys; also known from Cuba [see Erwin 1975: Fig. 135].
CAN: BC, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, MT, NC, ND, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV – Belize, Cuba, Guatemala, Mexico
The range of this species extends from the Gaspé Peninsula in eastern Quebec to west-central British Columbia, south to northwestern Oregon, south-central New Mexico through the Rocky Mountains, the Black Hills in southwestern South Dakota, east-central Minnesota, western New York near the Great Lakes, and Massachusetts (Erwin 1975: 65) along the east coast [see Erwin 1975: Fig. 135]. The record of Tachys rivularis from “Sitka” in the Alexander Archipelago (Motschulsky 1850a: 8) probably refers to this species.
CAN: AB, BC, ON, QC, SK USA: CO, ID, MA, ME, MN, MT, ND, NH, NM, NY, OR, SD, VT, WA, WI [AK]
This species ranges from Massachusetts to the Black Hills in southwestern South Dakota, south to southern Arizona, southern Texas, southern Mississippi (Harrison and Lamar Counties, Drew A. Hildebrandt pers. comm. 2008), and southern Florida, including the Keys [see Erwin 1975: Fig. 156].
USA: AL, AR, AZ, CO, DC, FL, GA, IN, LA, MA, MD, MS, NC, NJ, NM, NY, PA, RI, SC, SD, TN, TX, VA, WI, WV
Worldwide, with about 340 species (Lorenz 2005: 207-211, as Elaphropus and Nototachys) arrayed in seven subgenera following Sciaky and Vigna Taglianti (2003, as phyletic line of Elaphropus): Amaurotachys Jeannel (Old World), Barytachys, Elaphropus s.str. (about 40 species in the Eastern Hemisphere), Sphaerotachys Müller (Eastern Hemisphere) with Nototachys Alluaud, Tachyphanes Jeannel, and Trepanotachys Alluaud as synonyms, Tachylopha Motschulsky (40 species in the Eastern Hemisphere), Tachyura, and Tachyuropsis Shilenkov (one species in the Far East).
Several authors have discussed the taxonomic status of Elaphropus and related taxa and most have different opinions as to the statuses they should be assigned. In view of this, I prefer to include all taxa of the phyletic line of Elaphropus (sensu Sciaky and Vigna Taglianti 2003) into a single genus, as done by Erwin (1974a) and Shilenkov (2002), and recognize several subgenera.
Western Hemisphere, with 37 species in North America (28 species), Middle America (ten species), and the West Indies (three species, two of them endemic).
Hayward (1900: 201-212, as incurvus group) reviewed the North American species then known (15 species). Lindroth (1966: 414-424, as incurvus group) treated 12 species (one in the key only) found in North America. A taxonomic revision of the subgenus is needed.
Sciaky and Vigna Taglianti (2003) considered Elaphropus s.str. and Barytachys as two distinct genera, arguing that “Barytachys could possibly be considered a subgenus of Elaphropus, but from a biogeographical point of view, this classification seems highly implausible.” However, there seem to be no significant character states to differentiate the two taxa. In their key, members of Elaphropus are characterized by their “body almost always depigmented, nearly uniform in colour, almost round and convex” and those of Barytachys by their “body generally blackish, sometimes with lighter markings, oval and relatively flat.” Elaphropus is said to be represented only in the Eastern Hemisphere while Barytachys is represented in the Western Hemisphere. Based on the evidence presented I am not convince that these two taxa should be considered distinct. However, for the time being, Barytachys is retained as a valid subgenus pending further study.
The range of this species extends from Cape Breton Island to southeastern Alberta, north to southern Northwest Territories (Lindroth 1966: 418-419), south to southeastern Arizona (Dajoz 2007: 21), northwestern Texas (Nolan County, CMNH), northwestern Louisiana (Caddo Parish, Igor M. Sokolov pers. comm. 2009), and Maryland (Clark et al. 2006: 1306). The records from “British Columbia, ” “Washington, ” “Oregon, ” “Idaho, ” “Utah” (Bousquet and Larochelle 1993: 150), based on specimens identified under this name in USNM, need confirmation.
CAN: AB, MB, NB, NS (CBI), NT, ON, QC, SK USA: AR, AZ, CO, CT, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MT, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SD, TN, TX, VA, VT, WI, WV, WY [BC, ID, OR, UT, WA]
Lindroth (1966: 417) considered Elaphropus nebulosus (Chaudoir), Elaphropus vernicatus (Casey), and Elaphropus congener (Casey) as synonyms of this species. Erwin (1974a) treated Elaphropus nebulosus and Elaphropus congener as valid species and Elaphropus vernicatus as a synonym of Elaphropus unionis (Csiki).
This species ranges from southern Vancouver Island (CNC) to northern Idaho (Hatch 1953: 103), south to southern California (LeConte 1852a: 192; Fall 1901a: 44).
CAN: BC (VCI) USA: CA, ID, OR, WA
This species is known only from the type locality in southeastern Virginia.
USA: VA
This form was considered a synonym of Elaphropus granarius (Dejean) by Lindroth (1966: 414) but listed as a valid species by Erwin (1974a: 133).
This species is currently known only from the Coastal Plain ranging from New Jersey (Casey 1884c: 71, as Barytachys gemellus) to at least central Florida (Blatchley 1924: 164, as Tachys subpunctatus), west to southwestern Alabama (Motschulsky 1862b: 28).
USA: AL, FL, GA, NJ, SC, VA
The range of this species extends from western Vermont (Addison and Chittenden Counties, CMNH) to eastern Iowa (Hayward 1900: 209), including southernmost Ontario (Bousquet 1987a: 122), south to southeastern Louisiana (East Baton Rouge and Saint Tammany Parishes, Igor M. Sokolov pers. comm. 2009), southern Mississippi (Peter W. Messer pers. comm. 2008), and southern Florida (Peck and Thomas 1998: 18).
CAN: ON USA: DC, FL, GA, IA, IL, IN, LA, MA, MD, MO, MS, NJ, NY, OH, PA, TN, VA, VT
This form was regarded as a valid species by Hayward (1900: 209), Casey (1918: 187), Lindroth (1966: 422), and Bousquet (1987a: 122) but listed as a synonymy of Elaphropus vivax (LeConte) by Erwin (1974a: 137).
This species is known from northeastern New Mexico (Fall 1907: 218) and southeastern Arizona (Cochise County, USNM).
USA: AZ, NM
This species has been reported from localities in South Dakota (Kirk and Balsbaugh 1975: 21), west-central Mississippi, and eastern and central Texas (Casey 1918: 181).
USA: MS, SD, TX
This form was considered a synonym of Elaphropus anceps (LeConte) by Lindroth (1966: 417) but listed as a valid species by Erwin (1974a: 133).
This species is known only from southern Arizona.
USA: AZ
This species ranges from southern New Hampshire (Hillsborough County, CMNH) to south-central South Dakota (Kirk and Balsbaugh 1975: 21), including southern Ontario (Lindroth 1966: 419), south to southern Texas (Casey 1918: 177, as Tachyura audax; Hayward 1900: 205), southwestern Alabama (Baldwin County, CMNH), and the District of Columbia (Beutenmüller 1897: 40), west along southern United States to southern California (Fall 1901a: 44, as Tachys audax) and the Baja California Peninsula (Horn 1894: 308, as Tachys audax). The records from “Alberta, ” “Idaho, ” “Montana, ” “Wyoming” (Bousquet and Larochelle 1993: 151) and northern California (Notman 1929b: 223) are probably in error.
CAN: ON USA: AL, AR, AZ, CA, CO, DC, IA, IL, IN, KS, MA, MO, NE, NH, NM, NY, OH, OK, PA, SD, TN, TX, VT, WI, WV – Mexico
This species is known only from the type locality in southern Florida.
USA: FL
This species ranges from “Massachusetts” (Hayward 1900: 212) and Connecticut (Krinsky and Oliver 2001: 90) to northeastern Kansas (Popenoe 1878: 79), including eastern Iowa (Lindroth 1966: 423), south to northwestern and central Texas (Barr 1964: 3), southern Alabama (Conecuh County, CMN), and the Florida Panhandle (Washington County, CNC). The records from “Colorado” (Beutenmüller 1897: 39) and “New Hampshire” (Bousquet and Larochelle 1993: 151) need confirmation.
USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, MA, MD, MO, NC, NJ, NY, OH, OK, PA, SC, TN, TX [CO, NH]
Under this name, LeConte (1863b: 15) listed “truncorum Hald.” as synonym. I have not found any species described by that name in Haldeman’s publications.
This species seems restricted to the Coastal Plain ranging from New Jersey (Smith 1890: 79; Smith 1910: 203) to southern Florida (Leng 1915: 573), west to “Louisiana” (Chaudoir 1868b: 214) including southwestern Alabama (Löding 1945: 14).
USA: AL, FL, GA, LA, NJ
The range of this species extends from Nova Scotia (Lindroth 1966: 416) to “Nebraska” (Hayward 1900: 204), south to northern Oklahoma (French et al. 2001: 228), southeastern Louisiana (East Baton Rouge, Tangipahoa, and Saint Tammany Parishes, Igor M. Sokolov pers. comm. 2009), and northern Florida (Peck and Thomas 1998: 18). The records from southern Colorado (Elias 1987: 632) and “Minnesota” (Bousquet and Larochelle 1993: 151) need confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, VA, VT, WI [CO, MN]
This species ranges from Cape Breton Island (Bousquet 1987a: 121) to the Okanagan Valley in south-central British Columbia (Lindroth 1966: 416), south to “Oregon” (Hayward 1900: 205; Hatch 1953: 104), central New Mexico (Fall and Cockerell 1907: 157), and southeastern Tennessee (Sequatchie and Bledsoe Counties, Robert L. Davidson pers. comm. 2009). Several state records listed in Bousquet and Larochelle (1993: 151) need confirmation.
CAN: BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CO, CT, DC, DE, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, MT, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SD, TN, VT, WA, WI, WV [AR, FL, LA, NC, SC, TX]
This species is known only from the type series.
USA: TX
This species is known only from the type locality in northwestern New Mexico.
USA: NM
This species is known from North Carolina (Brimley 1938: 118) to “Utah” (Hayward 1900: 207), south to southeastern Arizona (Cochise County, USNM), southeastern Texas (Casey 1918: 178, as Tachuyra solita), southwestern Mississippi (Casey 1918: 179), and northern Georgia (Fattig 1949: 18); also recorded from the state of Veracruz in Mexico and Guatemala (Bates 1882a: 141). The records from eastern Washington, eastern Oregon, southwestern Idaho (Hatch 1953: 103), “Nevada” (Bousquet and Larochelle 1993: 151), and Pennsylvania (Bates 1882a: 141) are probably in error.
USA: AZ, CO, GA, KS, LA, MS, NC, NM, SC, TX, UT – Guatemala, Mexico
This form was listed as a synonym of Elaphropus anceps (LeConte) by Lindroth (1966: 417) but regarded as a valid species by Hayward (1900: 207), Casey (1918: 179), and Erwin (1974a: 135).
This species is known from Sonora in Mexico (USNM), southern Arizona (Pima and Santa Cruz Counties, CMNH, USNM), and “California” (Hayward 1900: 211). The record from southwestern Utah (Tanner 1928: 270) needs confirmation.
USA: AZ, CA [UT] – Mexico
This species is known from southeastern Washington (Walla Walla County, MCZ) and northern Idaho (Hatch 1953: 103) south to southern California (Fall 1901a: 43) and central Arizona (Griffith 1900: 565; Hayward 1900: 206; Casey 1918: 177), east along the south to western Texas (Casey 1918: 177; El Paso County, MCZ).
USA: AZ, CA, ID, NM, NV, OR, TX, WA
This form has been listed in synonymy with Elaphropus dolosus (LeConte) by Erwin (1974a: 134) but treated as a valid species by Hayward (1900: 205), Casey (1918: 177), and Lindroth (1966: 419). Upon examination of the type specimens of both taxa, I agree with the last-mentioned authors.
This species is known from east-central Oregon (Grant County, James R. LaBonte pers. comm. 1992), Humboldt County in northwestern California, and Washoe County in northwestern Nevada (Casey 1918: 183).
USA: CA, NV, OR
This species is known from “Indiana” to southeastern Iowa (Casey 1918: 186), south to the Rio Grande in southern Texas (Casey 1918: 188, as Tachyura gaudens), central Louisiana, and west-central Mississippi (Casey 1918: 186).
USA: IA, IN, LA, MO, MS, TX
The range of this species extends from Nova Scotia (Lindroth 1966: 422) to southwestern Wisconsin (Messer 2010: 37), south to central Louisiana (Grant Parish, CMNH), southwestern Alabama (Clarke County, CMNH), and northern Georgia (Fattig 1949: 19). The records from “Minnesota” and “Kansas” (Bousquet and Larochelle 1993: 152) need confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, GA, IA, LA, MD, ME, NC, NH, NY, OH, PA, TN, VA, VT, WI, WV [KS, MN]
This species is known from “Arizona” and northern Utah (Casey 1918: 180).
USA: AZ, UT
This species has been reported from northwestern Nevada (Casey 1918: 185, as Tachyura profuga), Santa Cruz County in western California (Casey 1918: 184-185; Notman 1929b: 223; Dajoz 2007: 19), and southwestern Oregon (Westcott et al. 2006: 8).
USA: CA, NV, OR
This species is known only from the type locality in the Sierra Nevada.
USA: CA
This species ranges from Cape Breton Island (Lindroth 1966: 423) to “Iowa” (Hayward 1900: 210), south to southern Texas (Casey 1918: 189, as Tachyura laredoana) and northern Georgia (Fattig 1949: 19). The records from “South Dakota” and “Wyoming” (Bousquet and Larochelle 1993: 152) are probably in error.
CAN: NB, NS (CBI), ON, QC USA: AL, AR, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, NC, NH, NJ, NY, OH, OK, PA, TN, TX, VA, VT, WV
This species is known from Nova Scotia (Majka et al. 2007: 8) to southern Saskatchewan (Ronald R. Hooper pers. comm. 2008), south to Arkansas (Franklin County, CNC) and North Carolina (Casey 1918: 183, as Tachyura laetifica).
CAN: MB, NB, NS, ON, QC, SK USA: AR, CT, DC, IA, IL, IN, KY, MA, MD, ME, MO, NC, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WV
The range of this species extends from Nova Scotia (Majka and Vickery 2008) to “Wisconsin” (Rauterberg 1885: 23), including southern Quebec and southern Ontario (Lindroth 1966: 423), south to northwestern Texas (Nolan County, CMNH), southwestern Alabama (Clarke County, CMNH), and northern Florida (Suwanee County, CMNH). The record from southern Arizona (Snow 1907: 142) is probably in error.
CAN: NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, SC, TN, TX, VA, VT, WI, WV
This species ranges from Nova Scotia (Lindroth 1966: 422) to western Kansas (Trego County, CNC), south to southeastern Texas (Wickham 1897: 104; Cameron County, CNC) and central Florida (Peck and Thomas 1998: 18). The species was also recorded from the Bahamas (Darlington 1953: 5) and Cuba (Darlington 1934: 78).
CAN: NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV – Bahamas, Cuba
According to Sciaky and Vigna Taglianti (2003: 90), members of Tachyura occur in the Old World though a few species extends into the Australian Region in New Guinea. The number of species is difficult to assess. Kopecký (2003: 278-280) listed 47 species for the Palaearctic Region. One species is adventive in North America.
The species was included in Lindroth’s (1966: 410) key to the species of Tachys.
This European species is adventive in North America where it is known from southwestern British Columbia, Washington, Oregon, and Idaho [see LaBonte and Nelson 1998: Fig. 1]. The first inventoried specimen reported from this continent was collected in Seattle in 1940 (Hatch 1950: 105).
CAN: BC USA: ID, OR, WA – Adventive
Western Hemisphere, with five species in the Nearctic (one species) and Neotropical (four species) Regions, including the West Indies. Erwin (1974a: 125) stated that he was aware of 35 undescribed species in this genus.
Barr (1971b) redescribed the North American species and illustrated its male genitalia.
This taxon is possibly the sister-group to Lymnastis Motschulsky (Erwin 1974a: 125), a genus of about 40 species in the Eastern Hemisphere with one endemic species, Lymnastis americanus Darlington, in Cuba. According to Robert L. Davidson (pers. comm. 2008), there is at least two species of Micratopus, besides Micratopus aenescens, that occur in southern United States. One of them, found in Florida, is probably the Cuban species.
This species occurs from Connecticut (Krinsky and Oliver 2001: 96) to “Indiana” (Schrock 1985: 351), south to southeastern Texas (San Patricio County, UASM) and southern Florida including the Keys (Peck and Thomas 1998: 18), west along the southwest to southwestern California (Riverside County, CAS); one specimen, possibly mislabeled, was seen from western Oregon (Benton County, CNC). The record from “Illinois” (Bousquet and Larochelle 1993: 152) needs confirmation.
USA: AL, AR, AZ, CA, CT, FL, GA, IN, KY, LA, MD, MI, MS, NC, NM, OH, OK, PA, SC, TN, TX, VA [IL, OR]
New World, with about 70 species arrayed in three subgenera: Eidocompsus Erwin (13 Neotropical species), Pericompsus s.str. (about 45 species), and Upocompsus Erwin (nine Australian species).
Erwin (1974b) revised the species and provided a key for their identification.
Western Hemisphere, with about 45 species in the Nearctic (three species) and Neotropical (about 45 species) Regions.
[ephippiatus group]
This species ranges from southwestern New Hampshire (Choate 1977: 115) to northern Kansas, south to Honduras and central Florida [see Erwin 1974b: Fig. 73]. The record from southern Wisconsin (Rauterberg 1885: 23) needs confirmation.
USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MO, MS, NC, NH, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WV [WI] – Honduras, Mexico
This species is known from central Oklahoma (Grady County, CMNH), southwestern New Mexico, and southeastern Arizona, south to Durango in Mexico. The species is apparently absent from the desert region of northern Mexico [see Erwin 1974b: Fig. 75].
USA: AZ, NM, OK – Mexico
[sellatus group]
This species is known only from “California” and southern Arizona [see Erwin 1974b: Fig. 134].
USA: AZ, CA
Five species in the Palaearctic (three species) and Oriental (two species) Regions. Among the Palaearctic species, one is endemic to Japan (Porotachys recurvicollis Andrewes) and one to Turkey (Porotachys ottomanus Schweiger). The three species from the Sinai (Porotachys efflatouani, Porotachys shahinei, and Porotachys zulficari, all described by Schatzmayr and Koch) included in this genus by Kopecký (2003: 274-275) belong to Sphaerotachys (see Sciaky and Vigna Taglianti 2003: 93). One species is adventive in North America.
The species found in North America was covered in Lindroth’s (1966: 424) monograph.
This Palaearctic species is adventive in North America where it ranges from Nova Scotia (Majka et al. 2006: 605) to eastern North Dakota (Cass County, Donald P. Schwert pers. comm. 1989), south along the east coast to New Jersey (Smith 1910: 203, as Tachys frontalis); also recorded from Missouri (Anonymous 2007). The species is also known from the Vancouver area in southwestern British Columbia (CNC), western Washington (Nelson 1987: 394), and northern Oregon (Westcott et al. 2006: 8). The first inventoried specimen collected on this continent was found in the east prior to 1900 (Hayward 1900: 212, as Tachys frontalis).
CAN: BC, NB, NS, ON, QC USA: CT, MA, ME, MI, MN, MO, ND, NH, NJ, NY, OH, OR, PA, RI, VT, WA, WI – Adventive
Worldwide, with about 40 species in the Nearctic (three species), Neotropical (ten species), Australian (nine species), Oriental (eight species), Palaearctic (nine species), and Afrotropical (ten species) Regions. One species is also known from the Hawaiian Islands.
Lindroth (1966: 424-427) treated two (Polyderis laeva and Polyderis rufotestacea) of the three species found in North America.
This taxon is considered a subgenus of Tachys by some authors (e.g., Lorenz 2005: 211).
This species is known only from the type locality in central Texas.
USA: TX
This species ranges from Nova Scotia (Bousquet 1987a: 123) to southwestern North Dakota (Smith et al. 1979: 92), south to southeastern Texas (Casey 1918: 213) and southern Florida including the Keys (Peck and Thomas 1998: 18).
CAN: NB, NS, ON, QC USA: AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI
This species is known from central Idaho (Custer County, Ken Karns pers. comm. 2009), southeastern Oregon (Malheur County, James R. LaBonte pers. comm. 1992), California, from Lake County (CAS) to Riverside County (CAS) east to Mono and Inyo Counties (CAS; Dajoz 2007: 18), southern Arizona (Cochise and Maricopa Counties, CMNH; Hayward 1900: 217), southern Texas (Zapata County, CMNH; Casey 1918: 211), northwestern Arkansas (Newton County, Peter W. Messer pers. comm. 2008), and eastern South Dakota (Kirk and Balsbaugh 1975: 21; French et al. 2004: 557). The record from Aklavik in northern Northwest Territories (Lindroth 1966: 427) is quite obviously based on a mislabeled specimen.
USA: AR, AZ, CA, ID, OR, SD, TX
Worldwide, with about 180 species (Lorenz 2005: 212-214, as Paratachys and Tachys) arrayed in two subgenera, both represented in North America.
The first use of the name Tachys is by Schönherr (1806: 221) who included 23 species-group taxa under it. Ádám (1996: 11) selected Carabus quadriguttatus Fabricius, 1775 (= Cicindela quadrimaculata Linnaeus, 1760), one of the species originally included, as type species of Tachys Schönherr, 1806. This action implies that Tachys Dejean, 1821 is a junior homonym of Tachys Schönherr, 1806 which is a junior objective synonym of Bembidion Latreille, 1802. Obviously, to preserve nomenclatural stability, the name Tachys Schönherr, 1806 should be rejected.
Shilenkov (2002) treated Tachys and Paratachys as distinct genera, pointing out four major structural differences between the two taxa. Sciaky and Vigna Taglianti (2003) took the same approach. In view of the large concept used here for the genus Elaphropus, I believe it is more appropriate to retain the two taxa as subgenera of Tachys, following in this sense Kopecký (2003: 275-277) and Lorenz (2005: 212-214). Sciaky and Vigna Taglianti (2003: 82) suggested that Polyderis is closely related to Tachys and Paratachys based on the presence of labial pits.
About 35 species in the Nearctic (11 species), Neotropical (seven species), Australian (one non-endemic species), Oriental (five species), Palaearctic (12 species, none in the Far East), and Afrotropical (seven species, three shared with Eurasia) Regions. One species, Tachys oahuensis Blackburn, is endemic to the Hawaiian Islands.
Lindroth (1966: 427-431) treated five (one of them, Tachys corax, in the key only) of the species found in North America and Hayward (1900: 218-224) eight species. A taxonomic revision of the species of this subgenus is needed.
This species has been reported from a few islands in the West Indies (Peck and Thomas 1998: 18; Steiner 2008: 131), “Louisiana” (Hayward 1900: 224), and southeastern Texas (Casey 1918: 208); it is also known from southern Mississippi (Hancock and Jackson Counties, Drew A. Hildebrandt pers. comm. 2009) and Grady County in central Oklahoma (Robert L. Davidson pers. comm. 2012). The record from “Florida” (Bousquet and Larochelle 1993: 153) needs confirmation.
USA: LA, MS, OK, TX [FL] – Cuba, Hispaniola, Jamaica, Navassa
This species is still known only from the holotype.
CAN: AB
This species ranges from eastern Washington (Hatch 1953: 104) to “Montana” (Hayward 1900: 223), south to western Texas (Dajoz 2007: 23), southeastern New Mexico (Chaves County, CNC), and the Baja California Peninsula (Horn 1894: 308). The records from “Nebraska” and “Texas” (Hayward 1900: 223) probably refer to Tachys halophilus Lindroth.
USA: AZ, CA, CO, MT, NM, NV, OR, TX, UT, WA – Mexico
This species is known from the southern parts of the Prairie Provinces (Lindroth 1966: 429; Bousquet 1987a: 123) and eastern North Dakota (Grand Forks County, UASM, USNM). The records of Tachys corax from “Nebraska” and “Texas” (Hayward 1900: 223) probably refer to this species.
CAN: AB, MB, SK USA: ND [NE, TX]
This species is found along the Coastal Plain from Long Island, New York (Cooper 1935: 144) to southern Florida, including the Keys (Peck and Thomas 1998: 18), west to southeastern Texas (Casey 1918: 207, as Tachys luridicollis); also known from some islands in the West Indies (Darlington 1934: 78; Darlington 1941a: 11, as Tachys occultator Casey).
USA: AL, DE, FL, GA, LA, MS, NJ, NY, SC, TX, VA – Bahamas, Cuba, Jamaica
This species has been reported from southwestern Alabama (Löding 1945: 14), “Texas” (LaFerté-Sénectère 1841a: 48), and Guatemala (Bates 1882a: 139, as Tachys lymnaeoides). The record from “Arizona” (Bousquet and Larochelle 1993: 154) needs confirmation.
USA: AL, TX [AZ] – Guatemala
This species has been recorded from “California, ” “Arizona, ” and Salt Lake Valley in Utah (Hayward 1900: 220). The record from “Colorado” (LeConte 1858a: 28) probably refers to the Colorado River.
USA: AZ, CA, UT
This species is known from New Jersey (Beutenmüller 1897: 37; Hayward 1900: 221; Smith 1910: 203), the Florida Peninsula (Leng 1915: 574), Big Pine Key, one of the Florida Keys (Foster F. Purrington pers. comm. 2012), Claiborne County in southwestern Mississippi (Robert L. Davidson pers. comm. 2012), Cameron Parish in southwestern Louisiana (Hine 1906: 76), “several localities” in Texas (Hayward 1900: 221), and Chaves County in southeastern New Mexico (Robert L. Davidson pers. comm. 2012); it has been recorded also from Guana Island (Valentine and Ivie 2005: 275) in the British Virgin Islands.
USA: FL, LA, MS, NJ, NM, TX – Guana Island
This species is known from southeastern Georgia (Glynn County, CMNH) and the Florida Peninsula (Monroe and Pinellas Counties, CMNH; Hayward 1900: 221, as Tachys occultator Casey sensu Hayward) west to southeastern New Mexico (Chaves County, CNC). The records from the District of Columbia (Ulke 1902: 6), Missouri (Summers 1873: 147), and “Yucatan” (Chaudoir 1868: 212) need confirmation.
USA: AL, FL, GA, LA, MS, NM, TX [DC, MO]
This species is known from Cuba and from Big Pine Key, Monroe County, in southern Florida (Foster F. Purrington and Robert L. Davidson pers. comm. 2009). This is a new record for America north of Mexico.
USA: FL – Cuba
This species is found along the seacoast from Santa Barbara County (Fall 1901a: 43) to San Diego County (LeConte 1852a: 194; Moore 1937: 8) in southwestern California.
USA: CA
This species is known from Vancouver Island (Lindroth 1966: 428, listed as “doubtful”), along the seacoast in southern California, including Santa Catalina Island (LeConte 1852a: 193; Fall 1901a: 43), Baja California Norte (Moore and Legner 1974: 289), Guatemala (Bates 1882a: 139), the Galápagos Islands (Mutchler 1925: 223, as Tachys beebei), Antigua (Putzeys 1874: 119, as Tachys picturatus), Montserrat (Ivie et al. 2008: 238, as Tachys ensenada), Guana Island (Valentine and Ivie 2005: 275, as Tachys ensenada), and Puerto Rico (Mutchler 1934: 3, as Tachys ensenadae). The records from Colorado (Wickham 1902: 235; LeConte 1858a: 28, as Tachys marginellus) must be in error.
CAN: BC (VCI) USA: CA (CHI) – Antigua and Barbuda, British Virgin Islands, Dominican Republic, Ecuador, Guana Island, Guatemala, Mexico, Montserrat, Puerto Rico
Worldwide, with about 145 species in the Nearctic (20 species), Neotropical (about 35 species), Australian (about 15 species), Oriental, Palaearctic (about 30 species), and Afrotropical Regions. Erwin (1974a: 126) stated that the number of New World species known to him is over 300.
Lindroth (1966: 431-436) treated six and Hayward (1900: 224-232) 11 (ten in the key) of the 20 species found in North America. A revision of the group is needed.
This species has been reported from southern Alabama (Motschulsky 1862b: 29) and western Mississippi (Casey 1918: 201).
USA: AL, MS
This species is known from the Coastal Plain ranging from North Carolina (Brimley 1938: 118) to southern Florida (Peck and Thomas 1998: 18), west to “Louisiana” (LeConte 1863c: 20), and from Cuba (Darlington 1934: 80) and Guadeloupe (Fleutiaux and Sallé, 1890: 368, as Tachys putzeysi) in the West Indies.
USA: FL, GA, LA, NC, SC – Cuba, Guadeloupe
This species is known from southwestern Pennsylvania (Allegheny County, CMNH), Virginia (Hoffman et al. 2006: 21), South Carolina (Ciegler 2000: 54), east-central Georgia (Emanuel County, Robert L. Davidson pers. comm. 2012), central Florida (Pinellas County, CMNH), southwestern Alabama (Baldwin County, Robert L. Davidson pers. comm. 2012), northeastern Mississippi (Oktibbeha County, Drew A. Hildebrandt pers. comm. 2009), Texas (Cameron, Nolan, Travis, and Zapata Counties, CMNH; Riley 2011), and central Arkansas (Pulaski County, CMNH). The record from South Dakota (Kirk and Balsbaugh 1975: 21) needs confirmation.
USA: AL, AR, FL, GA, MS, PA, SC, TX, VA [SD]
This species seems confined to the Coastal Plain and Piedmont Plateau ranging from southeastern Pennsylvania (Lebanon and Dauphin Counties, CMNH) to southern Florida, including the Keys (Peck and Thomas 1998: 18), west to Arkansas (Pulaski and Garland Counties, CMNH) and eastern Texas (Sabine County, Robert L. Davidson pers. comm. 2012), including Alabama (Löding 1945: 14) and Winston County in east-central Mississippi (Drew A. Hildebrandt pers. comm. 2009).
USA: AL, AR, FL, GA, MS, NC, PA, SC, TX, VA
This species ranges from south-central British Columbia (Lindroth 1966: 436) south to “Utah” (Hayward 1900: 231) and at least Santa Clara in west-central California (Erwin 1974a: 140).
CAN: BC USA: CA, ID, NV, OR, UT, WA
This species has been reported only from central Texas (Casey 1918: 200) and southeastern New York (Casey 1918: 200, as Tachys temporalis).
USA: NY, TX
This species ranges from southern Quebec (Larochelle 1975: 111, as Tachys obliquus) to eastern Iowa (Casey 1918: 195, as Tachys iowensis), south to eastern Texas (Polk County, CMNH), central Louisiana (Grant Parish, CMNH), northern Alabama (De Kalb and Madison Counties, CMNH), and central Georgia (Butts County, CMNH). The record from central Florida (Frost 1975: 37, as Tachys obliquus) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, GA, IA, IL, IN, KY, LA, MA, MD, NC, NH, NJ, NY, OH, PA, RI, SC, TN, TX, VA, VT, WV [FL]
This species is known from south-central Ohio (Fairfield County, CMNH) to “Massachusetts, ” south to “North Carolina” (Erwin 1981b: 152).
USA: MA, MD, NC, OH, PA, VA
This species ranges from southeastern Maine (Majka et al. 2011: 46) and the Saint Lawrence Plain in southern Quebec (Larochelle 1975: 111) to eastern South Dakota (Kirk and Balsbaugh 1975: 21), south to southeastern New Mexico, northern Coahuila in Mexico (Barr and Reddell 1967: 269), and central Florida (Seminole County, CMNH). The record from Jamaica (Darlington 1941a: 11) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WI, WV – Mexico
The range of this species extends from southwestern Quebec (LeSage 1996: 23) to southern Wisconsin (Messer 2010: 37), south to southeastern Texas (Kleberg County, CMNH; Hayward 1900: 230) and southern Florida (Peck and Thomas 1998: 18); also recorded from the Bahamas (Darlington 1953: 6, as Tachys corruscus), Cuba (Darlington 1934: 83), Jamaica (Darlington 1941a: 11), Puerto Rico (Wolcott 1941: 81), Mexico, and Guatemala (Bates 1882a: 139). The records from Yuma, California (Hayward 1900: 230), Sacramento Mountains in New Mexico (Fall and Cockerell 1907: 158), and “Colorado” (Leng 1920: 54) probably refer to other species.
CAN: QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI – Bahamas, Cuba, Guatemala, Jamaica, Mexico, Puerto Rico
This species is known only from the holotype.
None.
This species is known from scattered localities from New Brunswick (Webster and Bousquet 2008: 17) to northern Ohio (Cuyahoga County, Harry J. Lee, Jr. pers. comm. 2008), south to southern South Carolina (Ciegler 2003: [2]) and southwestern Alabama (Baldwin County, CMNH).
CAN: NB, ON, QC USA: AL, CT, DC, MA, NH, OH, PA, RI, SC, VA, VT, WV
Besides the type locality, this species is known from several counties in Virginia (Hoffman et al. 2006: 21), Maryland (Lepping 2009: 65), Montgomery County in northern Tennessee (Foster F. Purrington pers. comm. 2011), Oktibbeha County in northeastern Mississippi (Drew A. Hildebrandt pers. comm. 2009), and Desha County in southeastern Arkansas (Foster F. Purrington pers. comm. 2010).
USA: AR, IL, MD, MS, TN, VA
This species ranges from New Brunswick (Bousquet 1987a: 124) to central South Dakota (Kirk and Balsbaugh 1975: 21), south to central Texas (Blanco and Travis Counties, CMNH; Hayward 1900: 229; Riley 2011) and southern Florida (Peck and Thomas 1998: 18); also recorded from Cuba (Darlington 1934: 81) and Jamaica (Darlington 1941a: 12).
CAN: NB, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WI, WV – Cuba, Jamaica
This species is known only from the lectotype. The record from “Colorado” (Leng 1920: 54) needs confirmation.
USA: [CO]
This species is known only from east-central and westernmost Texas (Casey 1918: 202).
USA: TX
This species has been reported from “Arkansas” (Casey 1918: 193), “Louisiana” (Chaudoir 1868b: 213), and southeastern Texas along the Gulf Coast (Casey 1918: 193).
USA: AR, LA, TX
This form has been listed as a synonym of Tachys pumilus (Dejean) by Hayward (1900: 235) but regarded as a valid species by Casey (1918: 193) and Erwin (1974a: 143).
This species is known along the Coastal Plain from southwestern Georgia (Fattig 1949: 19) to southern Florida (Peck and Thomas 1998: 18), west to southeastern Texas (Casey 1918: 197).
USA: AL, FL, GA, LA, TX
This species is known only from the type locality in southeastern Texas.
USA: TX
This species is found from southeastern Texas (Wickham 1897: 104) to southwestern California (Fall 1901a: 43; Moore 1937: 8) and the Baja California Peninsula (Horn 1894: 308), north to southwestern Utah (Tanner 1928: 270). The records from the Bahamas (Darlington 1953: 6), Cuba (Darlington 1934: 82; Mateu 1977: 378), Jamaica (Darlington 1941a: 11), and Puerto Rico (Wolcott 1936: 188) need confirmation; that from eastern Washington (Hatch 1953: 105) is probably in error; that from “Colorado” (LeConte 1858a: 28) probably refers to the Colorado River.
USA: AZ, CA, NM, TX, UT – Mexico
Worldwide, with about 375 species in the Nearctic (50 species), Neotropical (24 species), Australian (about 45 species), Oriental (about 22 species), Palaearctic (about 175 species), and Afrotropical (about 60 species) Regions. There is no genus-group taxa shared between the Western Hemisphere and the Eastern Hemisphere.
The genus contains three species found in California.
Jeannel (1963a: 54-58) revised the species. Soon after, Jeannel (1963b) published a supplement to his monograph and pointed out that his Anillodes debilis consisted of two very similar species, one of which being undescribed (Anillodes minutus). A revision of the genus is needed.
A study of the type specimens of Anillodes affabilis (Brues), Anillodes sinuatus Jeannel, and Anillodes debilis (LeConte) showed that the first two species, described from Texas, are morphologically more similar to members of Anillinus Casey than to Anillodes debilis, the type species of Anillodes described from California. In fact, Anillodes affabilis and Anillodes sinuatus are externally indistinguishable from many Anillinus species and examination of the male genitalia is required for positive identifications. Based on this fact, the Texas species of Anillodes are transferred to Anillinus and the California species are retained in the genus Anillodes.
This species is known from a few specimens collected in Tulare and Santa Clara Counties in California (Jeannel 1963a: 56).
USA: CA
This species is known only from the two original specimens collected at the type locality in south-central California.
USA: CA
This species is known from a few specimens collected near Springville in south-central California.
USA: CA
Western Hemisphere, with 44 species in eastern North America (42 species), most of them in the Appalachian region, and the state of Santa Catarina in Brazil (two species).
Sokolov et al. (2004) reviewed the North American species and provided a key for the identification of all but four species (Anillinus daggyi, Anillinus dohrni, Anillinus longiceps, and Anillinus valentinei). Subsequently 16 new species were described by Dajoz (2005), Sokolov et al. (2007), Sokolov and Watrous (2008), Sokolov and Carlton (2008, 2010), Sokolov (2011), and Giachino (2011) and three species are transferred to the genus in this work.
This species is known only from the three original specimens collected at the type locality in central Texas.
USA: TX
This species is known only from Taney and Barry Counties in southwestern Missouri (Sokolov and Watrous 2008: 541).
USA: MO
This species is known from the original two specimens collected at the type locality in southeastern Kentucky (Sokolov et al. 2004: 208).
USA: KY
This species is known from a few specimens collected at the type locality on the Potomac River in Maryland and northern Virginia (Sokolov et al. 2004: 198).
USA: MD, VA
This species is known from Monroe County (Sokolov et al. 2004: 209) and Polk County (Giachino 2011: 113) in southeastern Tennessee.
USA: TN
This species is known only from the type locality in southwestern North Carolina.
USA: NC
This species is known only from the type locality along the state line between Tennessee and North Carolina.
USA: TN
This species is known only from the holotype collected in western South Carolina near the Georgia border.
USA: SC
This species is known from the southeastern parts of Blount County in Tennessee and adjacent parts of Nantahala National Forest in Graham County, North Carolina (Sokolov and Carlton 2008: 42).
USA: NC, TN
This species is known only from the holotype.
USA: TN
This species is known only from central and northeastern Blount County in Tennessee and adjacent parts of Swain County in North Carolina (Sokolov et al. 2007: 7).
USA: NC, TN
This species is known only from two nearby localities in Gaston County, North Carolina, and York County, South Carolina (Sokolov et al. 2004: 210).
USA: NC, SC
This species is known from two nearby localities in Burke County, western North Carolina (Sokolov et al. 2004: 211).
USA: NC
This species is known only from the holotype collected in central Texas.
USA: TX
This species is known only from the type locality in southwestern North Carolina (Sokolov et al. 2004: 212).
USA: NC
This species is known only from the holotype.
USA: FL
Jeannel (1963a: 76-77) reported that “Horn (1888: 27)” doubted the state provenance of the holotype and that the specimens in the USNM from Clayton, Georgia belong to Anillinus dohrni. I have not found any statement of that sort made by Horn. On the other hand, Schwarz (1891: 24) had “some doubt regarding the correctness of the [type] locality.”
This species is known from several localities in Orange, Mecklenburg, and Cabarrus Counties in North Carolina (Sokolov et al. 2004: 200).
USA: NC
This species is known from southwestern Virginia and North Carolina, north of the French Broad River (Sokolov et al. 2004: 213).
USA: NC, VA
This species is known only from the type locality in southwestern Alabama (Sokolov et al. 2004: 214).
USA: AL
This species is known from North Carolina, north of the French Broad River (Sokolov et al. 2004: 201), and presumably eastern Tennessee (Horn 1869a: 127). The records from the District of Columbia (Ulke 1902: 6), Virginia (Schwarz 1891: 24), South Carolina (Ciegler 2000: 55), and northeastern Georgia (Leng 1910: 73; Fattig 1949: 18) are probably in error.
USA: NC, TN
This species is known only from the eastern part of Rich Mountain ridge, Blount County, eastern Tennessee (Sokolov and Carlton 2010: 12).
USA: TN
This species is known only from the original four specimens collected in Lawrence and Crawford Counties in southern Indiana (Sokolov et al. 2004: 202).
USA: IN
This species is known only from the type locality in southeastern Tennessee.
USA: TN
This species is known only from the type locality in northern Florida (Sokolov et al. 2004: 215).
USA: FL
This species is known only from the Great Smoky Mountains in Cocke, Monroe, and Sevier Counties, Tennessee (Sokolov et al. 2004: 216; Giachino 2011: 111).
USA: TN
This species is known only from Latimer County in eastern Oklahoma (Sokolov et al. 2004: 217).
USA: OK
This species is known only from the holotype collected in southern Tennessee.
USA: TN
This species is known in the Appalachians from western North Carolina, eastern Tennessee, and northwestern South Carolina (Sokolov et al. 2004: 219; Giachino 2011: 110).
USA: NC, SC, TN
This species is known only from Mount Magazine in western Arkansas (Sokolov et al. 2004: 221).
USA: AR
This species is known from two localities in Swain and Macon Counties in western North Carolina (Sokolov and Carlton 2010: 11).
USA: NC
This species is known only from high elevations in central Great Smoky Mountains National Park in eastern Tennessee and western North Carolina (Sokolov et al. 2004: 222).
USA: NC, TN
This species is known only from Swain and Jackson Counties in North Carolina (Sokolov et al. 2004: 223).
USA: NC
This species is known only from the type locality in southwestern North Carolina.
USA: NC
This species is known from Avery and Wilkes Counties along the Blue Ridge Parkway in western North Carolina (Giachino 2011: 115).
USA: NC
This species is known from southwestern Blount County in Tennessee and Swain County in North Carolina in the Great Smoky Mountains National Park (Sokolov et al. 2007: 10).
USA: NC, TN
This species is known only from the southern parts of the Ouachita National Forest in western Arkansas (Sokolov et al. 2004: 224).
USA: AR
This species is known only from the holotype collected in eastern Tennessee (Sokolov et al. 2004: 204).
USA: TN
This species is known only from the holotype collected in south-central Texas.
USA: TX
This species is known only from eight specimens collected at the type-locality cave.
USA: TN
This species is known from Tishomingo County in northeastern Mississippi, northern Alabama (Sokolov and Carlton 2010: 13; Giachino 2011: 111), Blount County in eastern Tennessee, Swain County in western North Carolina, and Dade County in northwestern Georgia (Sokolov et al. 2004: 205).
USA: AL, GA, MS, NC, TN
This species is known only from Latimer County in eastern Oklahoma (Sokolov et al. 2004: 225).
USA: OK
This species is known only from the type locality in central Arkansas (Sokolov et al. 2004: 226).
USA: AR
This species is known for sure only from the two original specimens collected in central Georgia. The records from York and Lancaster Counties in northern South Carolina (Ciegler 2000: 55) need confirmation.
USA: GA [SC]
This species is known only from the holotype collected in the central part of the Unicoi Mountains in North Carolina.
USA: NC
This true troglobite species is confined to caves in northeastern Alabama east of the Wills Creek anticline (Barr 1995: 243).
USA: AL
This species is known for sure only from the type locality in northern Virginia (Sokolov et al. 2004: 207). The record from “West Virginia” (Bousquet and Larochelle 1993: 157) needs confirmation.
USA: VA [WV]
Three species in eastern North America.
Sokolov and Carlton (2008: 40) provided a key for the identification of all species.
This species is known only from the holotype collected in northwestern South Carolina. The records from North Carolina and northeastern Georgia (Barr 1995: 245) refer to Anillinus loweae Sokolov and Carlton (Sokolov et al. 2004: 189).
USA: SC
This species is known only from the type locality in southwestern North Carolina.
USA: NC
This species is known from Giles and Botetourt Counties in Virginia (Sokolov and Carlton 2008: 40).
USA: VA
Two species in the Sierra Nevada of California.
Jeannel (1963a: 79) provided a key for the identification of the two species.
This species is known only from the holotype collected in eastern California.
USA: CA
This species is known only from the holotype collected in eastern California.
USA: CA
One troglobitic species in eastern North America.
One species in eastern United States.
Valentine (1932b) published a detailed description of the species enhanced by several accurate drawings.
This species is known only from the holotype collected in a cave three miles north of Lewisburg, near Maxwelton, in southeastern West Virginia. The cave has two rather small rooms connected by a narrow, descending, and tortuous passage. The specimen was found in the lower room, which was wet, muddy, and quite dark (Valentine 1932b: 1-2).
USA: WV
About 85 species (Lorenz 2005: 237-238) in the Nearctic (six species), Neotropical (five species), Australian (about 15 species), Palaearctic (about 50 species), and Afrotropical (five species) Regions arrayed in 12 genera: Bedeliolus Semenov (three Palaearctic species), Cardiaderus Dejean (one Palaearctic species), Diodercarus Lutshnik (one Palaearctic species), Diplochaetus (four species), Ochtozetus Chaudoir (two South American species), Olegius Komarov (one species from Turkmenistan), Pogonistes Chaudoir (eight Palaearctic species), Pogonopsis Bedel (one north African species), Pogonus (about 55 species), Sirdenus Dejean (five Palaearctic species, one of them extending into the Afrotropical Region), Syrdenoidius Baehr and Hudson (one species from south Australia), and Thalassotrechus (one species).
Bousquet and Laplante (1997) revised the Western Hemisphere species and provided a key for their identification.
One species confined to the Pacific Coast of California and Baja California.
This species is found along the Pacific Coast from northern California to southern Baja California [see Bousquet and Laplante 1997: map 1].
USA: CA – Mexico
Western Hemisphere, with four species in the Nearctic (four species, two of them endemic) and Neotropical (two species) Regions, including the West Indies.
This species is known from a few localities around Salton Sea in southern California, from the northern part of the Baja California Peninsula, and from the state of Sinaloa along the Pacific Coast in Mexico [see Bousquet and Laplante 1997: map 2].
USA: CA – Mexico
This species is presently known from numerous specimens collected in two localities in Chaves County, New Mexico [see Bousquet and Laplante 1997: map 2].
USA: NM
This species ranges from south-central Oregon to south-central Kansas, south to western Texas near the Rio Grande, southeastern Arizona [see Bousquet and Laplante 1997: map 3], and southwestern California (LeConte 1874b: 44). One specimen without specific locality is known from Nebraska (Bousquet and Laplante 1997: 714).
USA: AZ, CA, KS, NM, NV, OK, OR, TX, UT [NE]
This species is known from New Jersey, southern South Carolina (Ciegler 2000: 56), the Florida Peninsula including the Keys, some islands in the Greater Antilles, southeastern Texas, Chiapas in Mexico, southern New Mexico (Otero County, CMNH), and southeastern Arizona [see Bousquet and Laplante 1997: map 2]. The species is also found in Colombia (Bousquet and Laplante 1997: 710) and Venezuela (Chaudoir 1872a: 36).
USA: AZ, FL, NJ, NM, SC, TX – Colombia, Cuba, Cayman Islands, Dominican Republic, Mexico, Venezuela
About 55 species in two subgenera: Pogonoidius Carret (six Palaearctic species) and Pogonus s.str. (about 50 species).
About 50 species in the Nearctic (one species), Australian (17 species), Palaearctic (about 30 species), and Afrotropical (four species) Regions.
This species is known only from a few localities in southwestern Louisiana and eastern Texas south to the Rio Grande [see Bousquet and Laplante 1997: map 1]; it was also collected at least twice at Atlantic City, New Jersey (G.E. Horn in Smith 1890: 79) in the xix Century.
USA: LA, NJ, TX
About 220 species in the Nearctic, Oriental, and Palaearctic Regions arrayed in two tribes: Lissopogonini (six species in the Oriental and Palaearctic Regions) and Patrobini (about 215 species).
Northern Hemisphere, with about 215 species arrayed in four subtribes following Zamotajlov (2002): Deltomerina (about 115 species), Deltomerodina (13 species in the genus Deltomerodes Deuve), Patrobina (about 80 species), and Platidiolina (five species). The North American fauna is represented by 13 species (roughly 6.2 % of the world fauna).
Darlington (1938) and Lindroth (1961a) reviewed the North American species and provided keys for their identification. Subsequently to Lindroth’s work, one species (Patrobus septentrionis) was shown to consist of two distinct forms by Zamotajlov (2003c).
Northern Hemisphere, with about 115 species in the Nearctic (four species) and Palaearctic (about 105 species) Regions arrayed in nine genera: Deltomerus (about 70 species in eastern Europe, northern Africa, and the Middle East), Diplous (23 species), Himalopenetretus Zamotajlov (one species in Uttar Pradesh), Ledouxius Zamotajlov (seven species in Kashmir and Pakistan), Minipenetetrus Zamotajlov (one Chinese species), Naxipenetetrus Zamotajlov (two Chinese species), Patanitretus Zamotajlov (one species in Pakistan), Penetretus Motschulsky (five south European species), and Qiangopatrobus Zamotajlov (three Chinese species).
Twenty-three species (Lorenz 2005: 241-242) in the Nearctic (four species) and Palaearctic (19 species in Asia only) Regions arrayed in two subgenera: Diplous s.str. (17 species) and Platidius (six species).
Six species in the Nearctic (four species) and Palaearctic (Diplous depressus Gebler and Diplous dolini Zamotajlov in eastern Asia) Regions.
Marek and Kavanaugh (2005) revised the North American species and provided a key for their identification.
The range of this species extends from northern Northwest Territories and northern Yukon Territory south to central Oregon (Darlington 1938: 150), north-central Utah, and southern Colorado [see Marek and Kavanaugh 2005: Fig. 10].
CAN: AB, BC (QCI, VCI), NT, YT USA: AK, CO, ID, MT, OR, UT, WA, WY
This species ranges from western Montana (Russell 1968: 47) to Vancouver Island (Lindroth 1961a: 188), south to the northern part of the Coast Ranges in California and the Sierra Nevada (Darlington 1938: 149; Marek and Kavanaugh 2005: 160).
CAN: BC (VCI) USA: CA, ID, MT, NV, OR, WA
This species ranges from the Skeena River drainage in central British Columbia (Lindroth 1961a: 189) south to the Coast Ranges and the Sierra Nevada in central California (Darlington 1938: 151; Marek and Kavanaugh 2005: 164).
CAN: BC USA: CA, OR, WA
This eastern species is found from Cape Breton Island (Lindroth 1961a: 189) to the Saint Lawrence Valley in southern Quebec (Larochelle 1975: 81), south to northeastern West Virginia (Randolph County, CMNH). Fossil remnants from the Early Wisconsinan have been unearthed in southern Ontario (Morgan and Morgan 1981: 1107).
CAN: NB, NS (CBI), QC USA: MA, ME, NH, NY, PA, VT, WV
Northern Hemisphere, with about 80 species in the Nearctic (eight species) and Palaearctic (about 75 species, only six of them occurring in Europe) Regions arrayed in ten genera: Apatrobus Habu and Baba (28 species), Archipatrobus Zamotajlov (three species), Chaetapatrobus Lafer (one species), Chinapenetetrus Kurnakov (11 species), Dimorphopatrobus Casale and Sciaky (one species), Minypatrobus Uéno (four species), Parapenetretus Kurnakov (14 species), Patrobus (16 species), Platypatrobus (one species), and Quasipenetretus Zamotajlov (one species).
Sixteen species in the arctic, subarctic, boreal, and temperate areas of the Nearctic (seven species) and Palaearctic (13 species) Regions. Four species are Holarctic.
[fossifrons group]
This western species ranges from southeastern Alberta to Vancouver Island, north to the Gulf Coast of Alaska, including the Aleutian Islands (Lindroth 1961a: 181), south to Mono County in the Sierra Nevada (CAS) and mountains in southwestern Colorado (Darlington 1938: 162-163). The record from Kamchatka (Eschscholtz 1823: 104) is probably in error since the species is not listed from the Palaearctic Region by Zamotajlov (2003b: 284-285).
CAN: AB, BC (VCI) USA: AK, CA, CO, ID, MT, OR, UT, WA, WY
Pohl (1998) reported that members of this species can be segregated into a “coastal” and “inland” morphs and that hybridization occurs between the coastal form of Patrobus fossifrons and Patrobus stygicus.
This species is found from Newfoundland (Lindroth 1955a: 83) to the Okanagan River in south-central British Columbia, north to the Peace River in northeastern British Columbia (Lindroth 1961a: 181), south to eastern South Dakota (Kirk and Balsbaugh 1975: 21), northern Minnesota (Lindroth 1963a: Fig. 61), and the Pontiac region in southwestern Quebec (Lindroth 1961a: 181); isolated in high mountains in Colorado (Darlington 1938: 160).
FRA: PM CAN: AB, BC, MB, NB, NF, QC, SK USA: CO, MN, ND, SD
This species ranges from Newfoundland (Lindroth 1955a: 83) to the Chukchi Sea coast in northwestern Alaska (Lindroth 1961a: 183), south to north-central Washington (Pohl 1998: 690), northern Minnesota (Clearwater and Lake Counties, CNC), the upper peninsula of Michigan (Escanaba, MCZ), and north-central Maine (Larochelle and Larivière 1990a: 28; Piscataquis County, CNC). The species is also known from the Far East (Zamotajlov 2003b: 285). Fossil remnants, dated between 10, 400 and 15, 400 years B.P., have been unearthed in Cape Breton Island in Nova Scotia (Miller 1997: 250) and central Iowa (Schwert 1992: 76); others from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 23).
CAN: AB, BC, LB, MB, NF, NT, ON, QC, SK, YT USA: AK, ME, MI, MN, WA – Holarctic
[foveocollis group]
This Holarctic species is found in easternmost Siberia (Zamotajlov 2003b: 285) and on this continent from Alaska, south of the Arctic Circle and including the Aleutian and Kodiak Islands (Lindroth 1961a: 186), to Newfoundland (Lindroth 1955a: 85), south to Mont Katahdin in Maine, the White Mountains in New Hampshire (Darlington 1938: 169), the Adirondack Mountains in northeastern New York (Notman 1928: 221, as Patrobus septentrionis var. tenuis), the upper peninsula of Michigan along Lake Superior (Hubbard and Schwarz 1878: 629, as Patrobus tenuis), northeastern Minnesota (Gandhi et al. 2005: 925), northern Wyoming (Johnson County, CMNH), and northern British Columbia (Lindroth 1961a: 186); isolated in high mountains in Colorado (Darlington 1938: 169; Armin 1963: 191).
CAN: AB, BC, LB, MB, NB, NF, NT, ON, QC, SK, YT USA: AK, CO, ME, MI, MN, NH, NY, VT, WY – Holarctic
[longicornis group]
This widely distributed species ranges from Newfoundland (Lindroth 1955a: 82) to at least the Okanagan Valley in British Columbia, north to southern Northwest Territories (Lindroth 1961a: 180), south to southern Arizona (Darlington 1938: 158), the Sacramento Mountains in New Mexico (Fall and Cockerell 1907: 158), northern Oklahoma (Tulsa and Cimarron Counties, CNC), and northeastern Florida (Darlington 1938: 158). The record from “Texas” (Wickham 1896c: 132) needs confirmation.
FRA: PM CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AL, AR, AZ, CO, CT, DC, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, UT, VA, VT, WA, WI, WV, WY [TX]
[septentrionis group]
The range of this species remains largely undocumented. It was reported by Zamotajlov (2003c: 242) from eastern Siberia, including the Kurils and Commander Islands, southern Alaska, including the Aleutian Islands, the upper peninsula of Michigan along Lake Superior (Casey 1920: 186, as Patrobus tritus), southern Quebec, and “Newfoundland.”
CAN: NF, QC USA: AK, MI – Holarctic
This circumpolar species ranges from Greenland (Böcher 1988: 8) west to Iceland (Zamotajlov 2003c: 241), south on this continent to northern Maine (Darlington 1938: 166; Dearborn and Donahue 1993: 5), northern New Hampshire (Coos County, Ross T. Bell pers. comm. 2008; Leng and Beutenmüller 1895: 75), northern Michigan (Lindroth 1961a: 184), northern Minnesota (Petrice et al. 2002: 9; Gandhi et al. 2005: 926), and southern Montana (Hatch 1933a: 7); isolated on high mountains in Colorado (Wickham 1902: 235; Zamotajlov 2003c: 241). Three specimens labeled “W[ashington]T[erritory]” are known (MCZ). Some of the records listed here may in fact refer to Patrobus cinctus.
DEN: GL CAN: AB, BC, LB, MB, NB, NF, NT, ON, QC, SK, YT USA: AK, CO, ME, MI, MN, MT, NH, WA – Holarctic
According to Zamotajlov and Isaev (2006), this species is represented by four subspecies in the Palaearctic Region: the nominotypical subspecies, Patrobus septentrionis australis Sahlberg, Patrobus septentrionis volgensis Zamotajlov and Isaev, and Patrobus septentrionis sajanus Zamotajlov.
One species in boreal and northern temperate regions of North America.
Bousquet and Grebennikov (1999) redescribed the species and illustrated the male and female genitalia.
With the exception of accessory setae on the pronotum, elytra, and mesosternum, this species is structurally very similar to those of Patrobus and possibly simply represents an offspring of Patrobus. The larva does not differ significantly from those of Patrobus (Bousquet and Grebennikov 1999).
This species ranges from New Brunswick to Great Slave Lake in Northwest Territories, south to the foothills of the Rocky Mountains in southern Alberta, northeastern Minnesota (Gandhi et al. 2005: 926), northeastern Ohio (Ashtabula and Trumbull Counties, Harry J. Lee, Jr. pers. comm. 2008), northeastern Pennsylvania (Pike County, CMNH), and Connecticut (Krinsky and Oliver 2004: 396) [see Bousquet 1987a: map 2].
CAN: AB, MB, NB, NT, ON, QC, SK USA: CT, ME, MN, NH, OH, PA, VT
Platypatrobus lacustris Darlington. Described in 1938, this species remained a great rarity for more than 20 years. The few specimens known had all been captured at light. Henri Goulet, then a summer student working at the Biosystematics Research Centre in Ottawa (now part of the Eastern Cereal and Oilseed Research Centre), found out that a mite of the genus Protodinychus, members of which occurred commonly in beaver houses, had been found on one of the specimens of Patrobus lacustris. Armed with this information, Goulet visited an abandoned beaver house in Gatineau Park in southwestern Quebec and found more than 50 specimens of Platypatrobus. The habitat requirements of the species were discovered: Platypatrobus lacustris lives in the walls of active or recently deserted beaver houses.
Five species placed in one genus.
Five species in Siberia (four species) and western North America (one species).
This rarely collected species is known from Yukon Territory (CNC), northeastern Alaska (Lindroth 1961a: 191), and the Kodiak Island (Dave H. Kavanaugh pers. comm. 2009) south to northwestern Oregon (Westcott et al. 2006: 8) and northwestern Montana (Edwards 1975: 50).
CAN: AB, BC, YT USA: AK, MT, OR, WA
This subfamily includes a single tribe.
Six species arrayed in three genera: Laccocenus Sloane (two species in southeastern Australia), Nomius (three species), and Psydrus (one species).
Three species, two in tropical Africa and one in North America, Mexico, southern Europe, and the Middle East.
The species found in North America was treated in Lindroth’s (1961a: 175) monograph.
The range of this species, also known as the “stinking Beetle”, extends from southern Quebec, as far north as Mistassini (Larochelle 1975: 95), to the Queen Charlotte and Vancouver Islands (Lindroth 1961a: 175), south at least to central California (Sequoia National Park, CAS), southern Arizona (Ober and Maddison 2008: 24; Cochise County, CNC), “Oklahoma” (David R. Maddison pers. comm. 2008), Alabama (LeConte 1846b: 209), northern Georgia (Fattig 1949: 16), and southern South Carolina (Ciegler 2000: 56); one specimen was collected in the highlands of Chiapas in Mexico (Reichardt 1977: 394). In the Old World, this species has been found in southern Europe, Cyprus (Fauvel 1889b: 96), Morocco, and Iran (Talysh) (Hůrka 2003: 346).
CAN: AB, BC (QCI, VCI), MB, ON, QC, SK USA: AL, AR, AZ, CA, CO, DE, GA, ID, IL, IN, ME, MI, MN, MT, NC, NJ, NY, OH, OK, OR, PA, SC, TN, UT, WA, WI, WV – Mexico
Nomius pygmaeus (Dejean). This species is known under the vernacular name “stinking beetle” because of the strong fetid smell that the adults produce. They are attracted to lights and sometimes find their way into houses. It was reported in the literature that at one occasion an entire village had to be evacuated because of the odor produced by these small beetles. The species was often listed as very common at light in the xix Century but is rare today. The species has an unusual range being found in North America and Europe and there is no evidence that it was transported by man from one continent to the other.
One North American species.
The species was treated in Lindroth’s (1961a: 175-176) monograph.
This species is known from scattered localities from the Lac Saint-Jean area in Quebec (Larochelle 1975: 99) to Vancouver Island, north to the Skeena River in northwestern British Columbia (Lindroth 1961a: 176), south to the San Gabriel and San Bernardino Mountains in southern California (Noonan 1967: 92), southern Arizona (McCleve 1975: 176), southwestern New Mexico (Grant County, USNM), and the upper peninsula of Michigan along Lake Superior (LeConte 1846a: 154). The record from “Illinois” and “South Dakota” (Bousquet and Larochelle 1993: 158) need confirmation.
CAN: AB, BC (VCI), MB, ON, QC, SK USA: AZ, CA, CO, ID, MI, MT, NM, NV, OR, WA [IL, SD]
Psydrus piceus LeConte. This species is widely distributed west of the Rocky Mountains where it is usually found under the bark of relatively large coniferous trees. It is also found within the boreal regions east of the Rockies but is very rarely collected there. This species belongs to the same tribe as Nomius pygmaeus but unlike that species, the adults do not emit an unpleasant smell.
Worldwide, with about 735 species in 47 genera. These genera are placed in five tribes: Metriini (three species), Mystropomini (two species in the genus Mystropomus), Ozaenini (about 180 species), Paussini (about 560 species), and Protopaussini (eight species in the genus Protopaussus Gestro). The Northern Hemisphere fauna is represented by about 70 species (roughly 9.5% of the world fauna) and North America by only seven species (less than 1% of the world fauna).
Three species in the genera Metrius (two species) and Sinometrius Wrase and Schmidt (one Chinese species).
Two species in western North America.
Bousquet and Goulet (1990) commented on the structural differences between the two species and on the three subspecies of Metrius contractus.
This subspecies ranges from southwestern British Columbia (Lindroth 1961a: 8) to San Luis Obispo County in California along the Coast Ranges and, usually at low elevation, to Tulare County in the Sierra Nevada (Bousquet and Goulet 1990: 17, Fig. 6).
CAN: BC USA: CA, OR, WA
This subspecies is found in the Sierra Nevada near Lake Tahoe and in Yosemite National Park (Bousquet and Goulet 1990: 17, Fig. 6).
USA: CA
This subspecies occurs in the southern part of the Sierra Nevada from Kings Canyon and Sequoia National Park to northern Kern County (Bousquet and Goulet 1990: 17, Fig. 6).
USA: CA
This species is known from three localities in Idaho County in north-central Idaho (James C. Bergdahl pers. comm. 2008) and in Ravalli County in western Montana (Moore 2008: 81).
USA: ID, MT
About 180 species arrayed in 21 genera in the Neotropical Region (about 120 species of which five extend into southwestern United States), Asia (about 40 species in five endemic genera, most species in the Oriental Region, a few in New Guinea, Japan, Taiwan, and Nepal), and Afrotropical Region (about 20 species in two endemic genera).
All North American species have been covered in the revision of the ozaenine species of southwestern United States and of selected ones from Mexico by Ball and McCleve (1990). The work includes a key for the identification of ten species.
About 50 species ranging from southwestern North America to Chile and Uruguay.
Recent molecular data analyses (Moore 2008) suggest that Pachyteles s.str., Goniotropis, and Tropopsis Solier (two South American species), which have been treated as subgenera of Pachyteles (Ball and McCleve 1990: 90), are not so closely related as suspected from the morphological data. These groups are recognized here as distinct genera.
This species ranges from southeastern California (San Bernardino County, CMNH) to southern Texas (Cameron County, CMNH), south to Brazil; it is also known from Cuba and from the Tres Marias Islands off the Pacific Coast of Mexico (Ball and McCleve 1990: 95).
USA: AZ, CA, TX – Brazil, Colombia, Costa Rica, Cuba, Mexico, Surinam
About 40 species extending collectively from southwestern United States to Brazil and Paraguay.
This subspecies is known from Arizona and the states of Durango and Sonora in northwestern Mexico (Ball and McCleve 1990: 92).
USA: AZ – Mexico
Pachyteles kuntzeni maracayensis (Deuve) is known from Venezuela.
This species is known from southern Arizona, western Durango, northern Sonora, and Baja California (Ball and McCleve 1990: 91).
USA: AZ – Mexico
Six species in the Neotropical Region, of which one extends into southern United States.
This species ranges from southeastern Texas (Ball and McCleve 1990: 87) to Guatemala (Bates 1881: 27).
USA: TX – Guatemala, Mexico
Twelve Neotropical species, of which one extends into southwestern United States.
This species ranges from southern Arizona to northern Argentina, including the Cayman Islands in the West Indies (Ball and Shpeley 1990: 814-815).
USA: AZ – Argentina, Belize, Bolivia, Brazil, Costa Rica, Cayman Islands, Colombia, Ecuador, Guatemala, Guyana, Mexico, Paraguay, Panama, Venezuela.
About 650 species arrayed in two tribes: Brachinini (about 535 species) and Crepidogastrini (about 115 species, most from the Afrotropical Region, a few from the Oriental Region). The Northern Hemisphere is represented by about 230 species (roughly 35% of the world fauna) and North America by 48 species (roughly 7.3% of the world fauna).
About 535 species arrayed in nine genera. The genera are classified in four subtribes following Erwin (1970a: 27-38): Aptinina (about 55 Old World species in three genera), Brachinina (about 305 species), Mastacina (about 50 Old World species in the genus Mastax Fischer von Waldheim), and Pheropsophina Jeannel (about 125 species in the Eastern Hemisphere genus Pheropsophus Dejean and the Western Hemisphere genus Pheropsophidius Hubenthal).
About 305 species arrayed in three genera: Aptinoderus Hubenthal (five species in southern Africa), Brachinus (about 300 species), and Brachinulus Basilewsky (one species on Príncipe Island in the Gulf of Guinea).
About 300 species (Lorenz 2005: 17-20) in the Nearctic (50 species), Neotropical (about 50 species, several shared with North America), Australian (one species in New Guinea), Oriental, Palaearctic (about 120 species), and Afrotropical Regions. These species are arrayed in nine subgenera: Brachinus s.str. (ten species), Brachynolomus Reitter (43 species), Metabrachinus Jeannel (38 Afrotropical species), Aploa Hope (three species), Brachinoaptinus Lutshnik (17 species), Aptinomimus Alluaud (seven Madagascan species), Dysbrachinus Schuler (two species), Cnecostolus Reitter (11 species), and Neobrachinus (about 85 species), with 89 species not assigned to subgenera.
Erwin (1970a) revised the North and Middle American species. No new species from the region have been described subsequently.
Eighty-three species in the temperate, subtropical, and tropical regions of the Western Hemisphere and one species (Brachinus dryas Andrewes) in the Himalayas. Forty-eight species inhabit North America.
In a cladistic analysis based on adult characters carried out by Erwin (1970a: 170), Neobrachinus was positioned as the sister-group to Cnecostolus whose members inhabit Eurasia.
The records of Brachinus cinctipennis Chevrolat from Albuquerque in New Mexico and Nogales in Arizona (Schaeffer 1910: 401) are probably in error since the species is recorded only from Mexico, as far north as San Luis Potosí, by Erwin (1970a: 98).
[aabaaba group]
This species is found from northeastern Kansas south to southeastern New Mexico and San Luis Potosí in Mexico [see Erwin 1970a: Fig. 450].
USA: KS, NM, TX – Mexico
[alternans group]
This species extends from Connecticut and “Rhode Island” (Sikes 2003: 8) to southern Minnesota and northeastern Nebraska, south to the Rio Grande in south-central New Mexico and southeastern Texas, and the Florida Keys [see Erwin 1970a: Fig. 201].
USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, RI, SC, TN, TX, VA, WV
This species ranges from Massachusetts to western Wyoming and western Colorado, south to central New Mexico, southeastern Texas, southeastern Mississippi (George County, Drew A. Hildebrandt pers. comm. 2008), and the Florida Keys [see Erwin 1970a: Fig. 200]. The populations in the northeast are isolated from the remaining ones but this is probably the result of inadequate collecting.
USA: AL, AR, CO, FL, GA, KS, MA, MS, NE, NJ, NM, OK, PA, TN, TX, VA, WY
This species is found only in the southern parts of the United States from southern South Carolina to southern Florida, west to southern Arkansas and eastern Texas [see Erwin 1970a: Fig. 199]. The record from New Jersey (Smith 1910: 212) needs confirmation.
USA: AL, AR, FL, GA, SC, TX [NJ]
[americanus group]
This species is known from east-central Texas, Latimer County in Oklahoma [see Erwin 1970a: Fig. 89], and from southwestern Mississippi (Adams County, Drew A. Hildebrandt pers. comm. 2008).
USA: MS, OK, TX
This species ranges from western New York to southeastern Minnesota, south to southeastern Texas, east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), and northern Florida [see Erwin 1970a: Fig. 88].
USA: AL, AR, FL, GA, IA, IL, IN, KS, KY, LA, MI, MN, MO, MS, NC, NY, OH, OK, PA, TN, TX, WI, WV
This species is known only from the holotype.
USA: NC
This species is known only from a few specimens collected in eastern Michigan, Missouri, northwestern Louisiana (Bossier Parish, Igor M. Sokolov pers. comm. 2009), and northwestern Mississippi [see Erwin 1970a: Fig. 90].
USA: LA, MI, MO, MS
[cordicollis group]
The range of this species extends from New Brunswick to east-central South Dakota (Kirk and Balsbaugh 1975: 40), south to northwestern Arkansas, northwestern Tennessee (Cheatham County, CMNH), and Virginia. The species is also known from a number of localities in the southern part of the Rocky Mountains, in Colorado and New Mexico [see Erwin 1970a: Fig. 391]. The records from Georgia (J.E. LeConte 1849: 25; Fattig 1949: 42), southwestern Alabama (Löding 1945: 23), and “Utah” (Horn 1872c: 384; Erwin 1970a: 147) need confirmation.
CAN: NB, ON, QC USA: AR, CO, CT, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NE, NH, NJ, NM, NY, OH, PA, RI, SD, TN, VA, VT, WI, WV [AL, GA, UT]
This species ranges from southwestern New Brunswick (Webster and Bousquet 2008: 23) to south-central British Columbia, south to southwestern Idaho, central Colorado, Missouri, and New Jersey along the Atlantic Coast [see Erwin 1970a: Fig. 392].
CAN: BC, MB, NB, ON, QC, SK USA: CO, CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NY, OH, PA, SD, VT, WI, WY
This species is known from western New York and southern New Hampshire (Hillsborough, Rockingham, and Strafford Counties, Ross T. Bell pers. comm. 2008) south to North Carolina, from several localities in southern Indiana [see Erwin 1970a: Fig. 414], and from Waukesha County in southeastern Wisconsin (Messer 2010: 34).
USA: CT, DE, IN, MA, MD, NC, NH, NJ, NY, PA, RI, VA, WI
This species is known only from the original two specimens, one collected at the type locality and the other at an unspecified locality in Florida (Erwin 1970a: 150).
USA: FL
The range of this species extends from Maine (Larochelle and Larivière 1990a: 33) and southern Quebec to western South Dakota, south to central Texas and central Georgia (Butts County, CMNH; Horn and Ulyshen 2009: 121) [see Erwin 1970a: Fig. 417].
CAN: ON, QC USA: CO, CT, GA, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, MS, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SD, TX, VA, VT, WI
This species is known only from the original five specimens collected at the type locality in southwestern Alabama.
USA: AL
This species ranges from North Carolina to the Florida Keys, west to southern Alabama [see Erwin 1970a: Fig. 416]; also known from one locality in central Missouri (Erwin 1970a: 153).
USA: AL, FL, GA, MO, NC, SC
This species is known from southeastern Michigan southeastwards to southern Florida, including the Keys, and southwestwards to east-central and southwestern Texas [see Erwin 1970a: Fig. 393]. The records from southeastern South Dakota (Kirk and Balsbaugh 1975: 39) and southwestern Ohio (Wright and Whitehouse 1941: 70, as Brachinus pulchellus) need confirmation.
USA: AL, AR, FL, GA, IN, KY, LA, MI, MO, MS, OK, SC, TN, TX, VA [OH, SD]
This species is known from a small area along the Atlantic Coast from New Hampshire to New Jersey [see Erwin 1970a: Fig. 415]. One specimen labeled from Crescent City, Florida is probably mislabeled (Erwin 1970a: 155).
USA: CT, MA, NH, NJ, NY, RI
[costipennis group]
This species ranges from northern California to eastern Utah, south to Guatemala (Erwin 1973b: 82) and northern Baja California [see Erwin 1970a: Fig. 197]. Specimens labeled from “Arkansas” and “Kansas” are known but could be mislabeled (Erwin 1970a: 87).
USA: AZ, CA, NM, TX, UT – Guatemala, Mexico
[explosus group]
This species is known from the type locality and “Arizona” (Erwin 2011b: 285).
USA: AZ – Mexico
[fumans group]
This species ranges from southeastern Arizona south to Guerrero, Mexico [see Erwin 1970a: Fig. 276].
USA: AZ – Mexico
This species ranges from northern Arizona and central New Mexico south to Durango City, Mexico [see Erwin 1970a: Fig. 219].
USA: AZ, NM – Mexico
This species is confined to northern and central Florida [see Erwin 1970a: Fig. 302].
USA: FL
This species ranges from Cumberland County in Nova Scotia (Majka and Gilhen 2008: 1) to southern Manitoba, south to eastern Texas, northwestern Louisiana (Natchitoches Parish, Igor M. Sokolov pers. comm. 2009), southern Alabama, northwestern South Carolina (Ciegler 2000: 35), and Virginia along the east coast [see Erwin 1970a: Fig. 330]. The species is also recorded from an unspecified locality in New Mexico (Erwin 1970a: 128). The records from “Georgia” (J.E. LeConte 1849: 25, as Brachinus cephalotes) and Enterprise in Florida (Castle and Laurent 1896: 303) need confirmation.
CAN: MB, NB, NS, ON, QC USA: AL, AR, CT, DC, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI [FL, GA, NM]
The range of this species extends from eastern Arizona to the Pacific Coast in southern California and Baja California Norte [see Erwin 1970a: Fig. 365].
USA: AZ, CA – Mexico
This widely distributed species ranges from southwestern New Brunswick (Webster and Bousquet 2008: 23) to south-central British Columbia, south to southwestern California, the state of Tabasco in Mexico, and central Florida [see Erwin 1970a: Fig. 369].
CAN: AB, BC, MB, NB, ON, QC USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
This species ranges from northern California to southern Baja California Sur and southeastern Arizona [see Erwin 1970a: Fig. 332].
USA: AZ, CA – Mexico
The range of this species extends from the Pacific Coast in southern California to northeastern Texas, north to northeastern Colorado, south to San Luis Potosí in central Mexico [see Erwin 1970a: Fig. 364].
USA: AZ, CA, CO, NM, NV, TX – Mexico
This species is known only from Arizona [see Erwin 1970a: Fig. 278].
USA: AZ
This species ranges from western Arizona to the Texas Panhandle and southeastern Texas [see Erwin 1970a: Fig. 254]; also known from northwestern Mississippi (Coahoma County, Drew A. Hildebrandt pers. comm. 2008).
USA: AZ, MS, NM, TX
This species is found west of the Appalachian Mountains from western New York to eastern Wyoming, including southwestern Wisconsin (Messer 2010: 34) and est-central Minnesota (Gandhi et al. 2011: 673), south to northeastern Mexico [see Erwin 1970a: Fig. 253].
USA: CO, IL, KS, MN, MO, NE, NY, OH, OK, SD, TX, WI, WY – Mexico
This species ranges from the Atlantic Coast in New England to south-central British Columbia, south to southern California, northeastern Mexico, and central Florida [see Erwin 1970a: Fig. 331].
CAN: BC, ON, QC USA: AL, AR, CA, CO, CT, DC, FL, GA, IA, ID, IL, IN, KS, LA, MA, ME, MI, MN, MO, MS, MT, ND, NE, NH, NJ, NY, OH, OK, OR, PA, RI, SC, SD, TX, UT, VT, WA, WI – Mexico
This species occurs from eastern Washington to northern Illinois, south to Guatemala and the southern part of the Baja California Peninsula [see Erwin 1970a: Fig. 252].
USA: AR, AZ, CA, IL, NM, NV, OR, SD, TX, UT, WA – Guatemala, Mexico
This species is restricted to the Coastal Plain where it ranges from North Carolina to central Florida, west to “Mississippi” (Drew A. Hildebrandt pers. comm. 2007) [see Erwin 1970a: Fig. 255].
USA: AL, FL, GA, MS, NC, SC
This species ranges from southeastern Maine (Majka et al. 2011: 47) and southern Quebec to southwestern South Dakota, south to northern Texas, “Mississippi” (Drew A. Hildebrandt pers. comm. 2007) and, east of the Appalachian Mountains, to Long Island, New York [see Erwin 1970a: Fig. 303].
CAN: ON, QC USA: CT, IA, IL, IN, KS, MA, ME, MI, MN, MO, MS, NY, OH, OK, PA, RI, SD, TX, VT, WI
This species ranges from southwestern Maine (Majka et al. 2011: 48) to Wisconsin (Messer 2010: 34), including northeastern Ohio (Ashtabula County, Foster F. Purrington pers. comm. 2009), south to Maryland (Kent County, CMNH) [see Erwin 1970a: Fig. 301]. The records from “Georgia” (J.E. LeConte 1849: 25) and Missouri (Summers 1873: 133) are likely in error.
USA: CT, IL, MA, MD, ME, MI, NH, NJ, NY, OH, RI, WI
This species ranges from southeastern Virginia (Norfolk, Robert L. Davidson pers. comm. 2012) to eastern Oklahoma, south to southeastern Texas and southwestern Florida [see Erwin 1970a: Fig. 366]. The records from New York (LeConte 1846b: 203), New Jersey (Smith 1910: 212), southeastern Iowa (King 1914: 322; Hendrickson 1930: 94), and Missouri (Summers 1873: 133) need confirmation.
USA: AL, AR, FL, GA, LA, MS, NC, OK, SC, TN, TX, VA [IA, MO, NJ, NY]
This species is restricted to the Great Plains and ranges from northeastern Iowa (Black Hawk County, Doug A. Veal pers. comm. 2009) to north-central Colorado, south to Chihuahua in northern Mexico and southeastern Texas [see Erwin 1970a: Fig. 279]. The specimens collected near Ithaca, New York (Erwin 1970a: 114), are possibly strays or mislabeled.
USA: AZ, CO, IA, KS, NE, NM, OK, TX [NY] – Mexico
This species is known from a few specimens collected in the Gulf Plain in eastern Texas [see Erwin 1970a: Fig. 368].
USA: TX
The range of this species extends from Massachusetts to south-central British Columbia, south to southern California along the Pacific Coast, north-central Utah, central Kansas, southeastern Texas, southern Mississippi (George and Wilkinson Counties, Drew A. Hildebrandt pers. comm. 2008), and southern Florida [see Erwin 1970a: Fig. 251]. At least one specimen simply labeled from Arizona is known (Erwin 1970a: 101); the record from southwestern New Mexico (Fall and Cockerell 1907: 160, as Brachinus tschernikhii) needs confirmation.
CAN: AB, BC, MB, ON, SK USA: AL, AR, CA, FL, GA, IA, ID, IL, IN, KS, LA, MA, MI, MN, MO, MS, MT, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, TN, TX, UT, WA, WI [AZ, NM]
This species ranges from southern Quebec to southeastern Wyoming (Lavigne 1977: 44), south to southwestern Texas and northern Florida [see Erwin 1970a: Fig. 329].
CAN: ON, QC USA: AR, CO, CT, DC, DE, FL, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NE, NH, NJ, NM, NY, OH, OK, SC, TN, TX, VT, WI, WY
This species is known so far only from the Central Valley in California between the Sierra Nevada and the Coast Ranges [see Erwin 1970a: Fig. 367].
USA: CA
[hirsutus group]
This species ranges from southern Utah to the Rio Grande in southwestern Texas, south to the Mexican High Plateau in Jalisco [see Erwin 1970a: Fig. 220].
USA: AZ, NM, TX, UT – Mexico
This species ranges from northeastern Washington to southern California [see Erwin 1970a: Fig. 218].
USA: CA, OR, WA
[kansanus group]
This species is mainly restricted to the Great Plains and ranges from southwestern Ohio to western Kansas south to southern Oklahoma and western Arkansas [see Erwin 1970a: Fig. 198].
USA: AR, IA, IL, KS, MN, MO, NE, OH, OK
[lateralis group]
This widely distributed species ranges from southeastern New York to southern Nebraska (Franklin County, Foster F. Purrington pers. comm. 2010), north to southwestern Wisconsin (Messer 2010: 34) and Michigan, south to Panama, southern Florida, and Cuba, west to the coast of the Gulf of California [see Erwin 1970a: Fig. 166].
USA: AL, AR, FL, GA, IA, IL, IN, KS, KY, LA, MA, MI, MO, MS, NE, NM, NY, OK, SC, TN, TX, VA, WI – Costa Rica, Cuba, Mexico, Panama
This species ranges from southern Sonora in Mexico to Colombia [see Erwin 1970: Fig. 168]. It was also recorded from “Texas” (Erwin 2011b: 277).
USA: TX – Belize, Colombia, Costa Rica, Guatemala, Guyana, Honduras, Mexico, Panama, Venezuela
This species extends from northern Arizona, southern Nevada, and southern California south to the southern part of the Baja California Peninsula and Chiapas in Mexico [see Erwin 1970a: Fig. 169]; also recorded from Nicaragua (Erwin 2011b: 292). The record from Cuba (Jacquelin du Val 1857: 12) must be in error.
USA: AZ, CA, NV – Mexico, Nicaragua
[texanus group]
This species extends along the Pacific Coast from central Oregon to the southern extremity of the Baja California Peninsula, and from western Arizona to southwestern Oklahoma (Kondratieff et al. 2005: 171), south to Oaxaca, Mexico [see Erwin 1970a: Fig. 109].
USA: AZ, CA, NM, OK, OR, TX – Mexico
This species ranges from east-central Texas south at least to northern Colombia (Dejean 1831: 428) [see Erwin 1970a: Fig. 110].
USA: TX – Belize, Colombia, Costa Rica, El Salvador, Guatemala, Honduras, Mexico, Nicaragua, Panama.
This species ranges from Massachusetts to “Minnesota, ” south to the Rio Grande River in southern Texas and west-central Florida [see Erwin 1970a: Fig. 108]. The species is quite common west of the Mississippi Basin and south of latitude 40°, but known only from a few isolated localities outside this area. The record from southeastern Alberta (Erwin 1970a: 62) is possibly based on a mislabeled specimen or a stray; that from Riverside County, California (Dajoz 2007: 19) is probably in error.
USA: AL, AR, FL, KS, LA, MA, MI, MN, MO, MS, ND, NE, NY, OK, TN, TX, VA, WI [AB]
Worldwide, with about 19, 600 species arrayed here in two supertribes for convenience: Pterostichitae and Harpalitae.
Worldwide, with about 6, 400 species arrayed in the following 24 tribes: Abacetini (about 810 species), Amorphomerini (two species in the genus Amorphomerus Sloane), Bascanini (ten species in the genus Bascanus Péringuey), Brachygnathini (seven species in the genus Brachygnathus Perty), Chaetodactylini (about 20 species in the genus Chaetodactyla Tschitschérine), Chaetogenyini (five species), Chlaeniini (about 980 species), Cnemalobini (about 30 species), Cratocerini (four species), Cuneipectini (two species in the genus Cuneipectus Sloane), Dercylini (35 species in the genus Dercylus Laporte), Drimostomatini [= Caelostomini] (about 290 species), Glyptini (nine species), Idiomorphini (ten species), Melanchitonini (about 70 species), Metiini (about 75 species), Microcheilini (two species in the genus Microcheila Brullé), Morionini (about 85 species), Oodini (about 300 species), Orthogoniini (about 120 species), Panagaeini (about 270 species), Peleciini (about 75 species), Pterostichini (about 2, 500 species), and Zabrini (about 700 species).
About 85 species (Lorenz 2005: 247-248) in the Nearctic (two species, none endemic), Neotropical (11 species), Australian (16 species), Oriental (11 species), Palaearctic (six species), and Afrotropical (40 species) Regions. These species are arrayed in nine genera: Buderes Murray (one Afrotropical species), Hyperectenus Alluaud (two Afrotropical species), Hyperion Laporte (one Australian species), Megamorio Chaudoir (six Afrotropical species), Morion (about 40 species), Morionidius Chaudoir (five Asian species), Moriosomus Motschulsky (two Neotropical species), Platynodes Westwood (one Afrotropical species), and Stereostoma Kirby (28 Afrotropical species).
About 40 species (Lorenz 2005: 247) in temperate, subtropical, and tropical areas of the Nearctic (two species), Neotropical (9 species), Australian (15 species), Oriental (eight species), Palaearctic (four species), and Afrotropical (three species) Regions.
Allen (1969) reviewed the Western Hemisphere species.
This species is known from southern Arizona, the Baja California Peninsula (Allen 1969: 146), and the state of Sonora (CAS).
USA: AZ – Mexico
This species ranges from east-central South Dakota to central New York, south to southern Florida and southeastern Texas (Allen 1969: 147). The record from Cuba (Jacquelin du Val 1857: 18) needs confirmation.
USA: AL, AR, FL, GA, IA, IL, KY, LA, MD, MO, MS, NC, NY, OH, OK, SC, SD, TX, VA
Worldwide, with approximately 810 species in about 45 genera. The tribe is underrepresented in the Northern Hemisphere with around 75 species (less than 10% of the world fauna). Only five species, all in the genus Abacetus, are known from Europe and northern Africa. The Western Hemisphere is represented by about 235 species (29% of the world fauna) in the genera Adrimus Bates (22 Neotropical species), Loxandrus (about 190 species), Metoncidus Bates (one Neotropical species), Oxycrepis Reiche (four Neotropical species), and Stolonis (19 species). All these genera belong to the loxandrine complex.
Members of this tribe are often included in the tribe Pterostichini.
About 210 species in the Nearctic (44 species), Neotropical (about 150 species), and Australian (about 20 species) Regions.
Allen (1972) revised the North American species though he left out more than 20 species because their type series were not located or included only females. These species were studied by Bousquet (2006a) leading to several new synonymies.
[agilis group]
This species is known only from the type locality in southeastern Texas.
USA: TX
This species has been recorded from southeastern South Carolina (Ciegler 2003: [2]), northern Georgia (J.E. LeConte 1849: 26; Fattig 1949: 28), all over Florida except the Keys (Peck and Thomas 1998: 19), southwestern Alabama (Löding 1945: 17), Mississippi (Snodgrass and Cross 1983: 18), one locality (probably Mittie in Cherokee County) in eastern Texas (see Allen 1972: 94), and the Bahamas (Peck and Thomas 1998: 19). The record from “Arkansas” (Wickham 1896b: 43) needs confirmation; those from the District of Columbia (Ulke 1902: 7) and southwestern Ohio (Wright and Whitehouse 1941: 70) are probably in error.
USA: AL, FL, GA, MS, SC, TX [AR] – Bahamas
This species is known from a few specimens collected in Maryland (Allen 1972: 110; CNC), eastern Alabama, and southeastern Oklahoma (Bousquet 2006a: 151-152).
USA: AL, MD, OK
This species is known from several localities in northern and central Florida and one locality in central Arkansas [see Allen 1972: Fig. 176]. The records from South Carolina (Kirk 1969: 11) and Georgia (Fattig 1949: 28, also as Loxandrus mundus) need confirmation.
USA: AR, FL [GA, SC]
This species is known from Dubois and Vigo Counties in western Indiana, from an unspecified locality in Arkansas (Allen 1972: 84), and from Noxubee County in eastern Mississippi (CMNH).
USA: AR, IN, MS
This species is known from the Bahamas and along the Coastal Plain from southern Florida west to eastern Texas, north to northeastern Louisiana [see Allen 1972: Fig. 171]. The record from southern Georgia (Fattig 1949: 27, as Loxandrus comptus) needs confirmation.
USA: AL, FL, LA, MS, TX [GA] – Bahamas
This species is known only from the original 13 specimens collected at the type locality.
USA: VA
This species is known from northeastern Arkansas south to southeastern Louisiana, east to eastern Mississippi (Noxubee County, CMNH) and west to east-central Texas [see Allen 1972: Fig. 170]; also recorded from South Carolina (Kirk 1969: 11; Ciegler 2000: 59). One specimen simply labeled from Kentucky is known (Allen 1972: 98)
USA: AR, LA, MS, SC, TX [KY]
This species is known from a few localities in central Florida and southern Alabama [see Allen 1972: Fig. 174, as Loxandrus aduncus]. It probably occurs also farther north as implied by the type locality given by Chaudoir (1868b: 342).
USA: AL, FL
This species is known only from the holotype (Bousquet 2006a: 149).
USA: TX
This species is known only from the type series.
USA: GA
This species is known from southeastern Georgia to central Florida including the Panhandle (Peck and Thomas 1998: 19) [see Allen 1972: Fig. 175, as Loxandrus minutus].
USA: FL, GA
This species is known from a few localities in central Georgia, northern Florida (Peck and Thomas 1998: 19), northeastern Alabama (Allen 1972: 108), and northeastern Arkansas [see Allen 1972: Fig. 174].
USA: AL, AR, FL, GA
This species is known only from the holotype collected in Georgia from an unspecified locality.
USA: GA
This species is known from two unspecified localities in Arkansas and Louisiana (Allen 1972: 102).
USA: AR, LA
This species is known along the Coastal Plain from North Carolina and South Carolina, west to southern Louisiana and eastern Arkansas [see Allen 1972: Fig. 175]. Specimens simply labeled from Georgia are known (Allen 1972: 87). The records from Missouri (Summers 1873: 134), central Kansas (Knaus 1885: 58), and southeastern Texas (Casey 1918: 384) need confirmation.
USA: AL, AR, LA, MS, NC, SC [GA, KS, MO, TX]
This species is found along the Coastal Plain from northeastern Florida to central Louisiana and east-central Arkansas [see Allen 1972: Fig. 172]. At least one specimen simply labeled from Texas is known (Allen 1972: 78).
USA: AR, FL, LA [TX]
This species is known for sure only from the four original specimens. The records from “Pennsylvania” (LeConte 1853a: 252), South Carolina (Kirk 1969: 11), Georgia (J.E. LeConte 1849: 26; Leng 1910: 73; Fattig 1949: 28), and southwestern Alabama (Löding 1945: 17) need confirmation.
[AL, GA, PA, SC]
Dejean’s name was listed as synonym of Loxandrus rectus (Say) by LeConte (1846b: 338) but later considered a valid species (e.g., LeConte 1853a: 252; LeConte 1878b: 376).
[celer group]
This species ranges from Virginia (Virginia Beach County, VMNH) to southern Florida, west to southeastern Texas (Casey 1918: 389, as Loxandrus parvicollis) and central Oklahoma (Grady County, Robert L. Davidson pers. comm. 2012), south to Chiapas in southern Mexico; also known from the Bahamas, Cuba, Cayman Islands, and Puerto Rico (Peck 2005: 31, as Loxandrus celeris) [see Allen 1972: Fig. 147]. The records from “Pennsylvania” (LeConte 1853a: 252) and southwestern Ohio (Wright and Whitehouse 1941: 70) need confirmation.
USA: AL, FL, GA, LA, MS, OK, SC, TX, VA [OH, PA] – Bahamas, Cayman Islands, Cuba, Mexico, Puerto Rico
[crenatus group]
This species inhabits the Coastal Plain from southeastern Virginia (Norfolk, Robert L. Davidson pers. comm. 2008) to central Florida, west to southeastern Texas (Brazoria County, Brian Raber pers. comm. 2010) [see Allen 1972: Fig. 162], including western Arkansas (Polk and Garland Counties, Robert L. Davidson pers. comm. 2012); also recorded from Cuba (Darlington 1934: 91).
USA: AL, AR, FL, GA, LA, MS, NC, SC, TX, VA – Cuba
This species is at present known only from the holotype which Chaudoir (1868b: 344) believed had been collected in Texas.
USA: TX
[erraticus group]
This species is found from southeastern Pennsylvania to Iowa (Wickham 1888: 82; King 1914: 323; Jaques and Redlinger 1946: 297), south to southern Louisiana and central Florida including the Panhandle (Peck and Thomas 1998: 19) [see Allen 1972: Fig. 160]. Old specimens simply labeled from Massachusetts and Oklahoma are known (Allen 1972: 144).
USA: AL, AR, DC, DE, FL, GA, IA, IL, IN, LA, MO, MS, NC, PA, SC, TN, VA [MA, OK]
This species is known from southwestern Ohio and northeastern Kentucky to western Illinois, south to north-central Texas, southern Alabama, and the Florida Panhandle (Peck and Thomas 1998: 19) [see Allen 1972: Fig. 168]. One specimen simply labeled from “South Carolina” is known (Allen 1972: 156).
USA: AL, AR, FL, IL, IN, KY, LA, MO, MS, OH, TN, TX [SC]
This species is known from a few localities in Maryland, the District of Columbia, northern Ohio, Mississippi (Drew A. Hildebrandt pers. comm. 2007), and Alabama [see Allen 1972: Fig. 159]. The record from “Illinois” (Bousquet and Larochelle 1993: 161) needs confirmation.
USA: AL, DC, MD, MS, OH [IL]
This species ranges along the Coastal Plain from Rhode Island and Connecticut (William L. Krinsky pers. comm. 2008) to central Florida, west to southern Louisiana and north along the Mississippi River drainage to east-central Illinois [see Allen 1972: Fig. 155]. The record from southwestern Ohio (Dury 1902: 113) needs confirmation.
USA: AL, AR, CT, DC, DE, FL, GA, IL, IN, KY, LA, MO, MS, NC, NJ, PA, RI, SC [OH]
This species is known along the Coastal Plain from southern North Carolina to central Florida, west to northeastern Louisiana and southern Arkansas, north along the Mississippi River drainage to southwestern Indiana and southwestern Illinois (Allen 1972: 147-148, Fig. 156).
USA: AL, AR, FL, IL, IN, LA, MO, MS, NC, TN, SC
This species ranges from southern Ontario (Lindroth 1966: 540) south to southeastern Louisiana (Chaudoir 1843b: 766), west to eastern Kansas [see Allen 1972: Fig. 167]. The records from New Jersey (Smith 1910: 206), District of Columbia (Ulke 1902: 7), South Carolina (Kirk 1969: 11; Ciegler 2000: 59), “Georgia” (J.E. LeConte 1849: 26), Florida (Frost 1964: 138), and southwestern Alabama (Löding 1945: 17) need confirmation.
CAN: ON USA: AR, IA, IL, IN, KS, LA, MO [AL, DC, FL, GA, NJ, SC]
This species ranges from northern Indiana to eastern Iowa, south to northern Louisiana and southwestern Alabama [see Allen 1972: Fig. 161). One specimen simply labeled from Florida is known (Allen 1972: 153).
USA: AL, AR, IA, IL, IN, LA, MO, MS, TN [FL]
This species is known from one locality in southwestern Indiana, several localities in Arkansas, and one locality in Noxubee County in eastern Mississippi (CMNH) [see Allen 1972: Fig. 169].
USA: AR, IN, MS
This species is known from a few localities in northwestern Ohio, South Carolina, and southwestern Alabama [see Allen 1972: Fig. 165]. Specimens simply labeled from Virginia and Arkansas are known (Allen 1972: 132).
USA: AL, OH, SC [AR, VA]
This species ranges from Maryland (Erwin 1981b: 160) to southeastern Minnesota (Allen 1972: 137), including north-central Ohio (Lee 1994: 61) and southern Ontario (Lindroth 1966: 539), south to southeastern Louisiana and southern Georgia (Torres and Ruberson 2006: 32) [see Allen 1972: Fig. 158]. The record from “Connecticut” (Bousquet and Larochelle 1993: ) was based on a misidentified specimen of Loxandrus vitiosus (Krinsky and Oliver 2001: 102).
CAN: ON USA: AL, AR, DC, GA, IA, IL, IN, LA, MD, MN, MO, MS, NC, OH, SC, TN, VA, WI, WV
[infimus group]
This species is found from northern Sonora and southeastern Texas south to Brazil (Allen 1972: 34); also known from Haiti [see Allen 1972: Fig. 144].
USA: TX – Brazil, Guatemala, Haiti, Mexico
This species ranges from southeastern Arizona (Allen and Ball 1980: 500) south to Morelos in Mexico [see Allen 1972: Fig. 154].
USA: AZ – Mexico
This species ranges from southern Texas (Allen and Ball 1980: 501) and central Sinaloa south to Panama [see Allen 1972: Fig. 153].
USA: TX – Mexico, Panama
[micans group]
The range of this species extends from central New York (Bousquet 2006a: 152) to east-central Iowa (Johnson County, CMNH), including southern Ontario (CNC), south to southeastern Texas (Cameron County, CMNH), central Louisiana, and northeastern Alabama (DeKalb County, CMNH) [see Allen 1972: Fig. 166].
CAN: ON USA: AL, IA, IL, IN, LA, MO, MS, NY, OH, PA, TX, VA
This species is known from three localities in southwestern Tennessee, eastern Arkansas (Allen 1972: 114, Fig. 165), and southern Louisiana (Chaudoir 1868b: 342).
USA: AR, LA, TN
This species is known only from George and Noxubee Counties in eastern Mississippi (Will and Liebherr 1998: 231).
USA: MS
This species ranges from Connecticut (Krinsky and Oliver 2001: 102) and southeastern Pennsylvania to southwestern Illinois, south to southern Louisiana, southwestern Alabama, and central South Carolina (Ciegler 2000: 60) [see Allen 1972: Fig. 167]. One specimen simply labeled from Florida is known (Allen 1972: 116).
USA: AL, AR, CT, DC, IL, IN, LA, MD, MO, MS, NC, OH, PA, SC, TN, VA [FL]
[rectangulus group]
This species is known from central and southern Florida (Peck and Thomas 1998: 19), the Bahamas (Turnbow and Thomas 2008: 13), Grand Cayman Island, and southern Mexico [see Allen 1972: Fig. 146]. One specimen simply labeled from Texas is known (Allen 1972: 42).
USA: FL [TX] – Bahamas, Cayman Islands, Mexico
[rectus group]
This species is known from a few localities in northwestern Louisiana (Casey 1918: 383, as Loxandrus ludovicianus) and eastern and southern Texas (Casey, 1918: 382; Brazoria, Fort Bend, Lasalle, and Live Oak Counties, CMNH).
USA: LA, TX
This species is found west of the Appalachians from central Kentucky south to eastern Texas and southern Alabama (Allen 1972: 130, Fig. 163). The record from northern Georgia (Fattig 1949: 27) is probably in error.
USA: AL, AR, KY, LA, TN, TX
This species is found along the Coastal Plain from southern North Carolina to the Rio Grande in southeastern Texas [see Allen 1972: Fig. 159].
USA: AL, LA, MS, NC, TX
This species ranges from the District of Columbia to southern Illinois, north to northern Ohio (Lee 1994: 61), south to southeastern Texas and northern Florida including the Panhandle (Peck and Thomas 1998: 19) [see Allen 1972: Fig. 157]. The record from southeastern Pennsylvania (Rathvon 1869: 525) needs confirmation.
USA: AL, AR, DC, FL, GA, IL, IN, KY, LA, MD, MO, MS, NC, OH, SC, TN, TX, VA [PA]
[saphyrinus group]
This species is found from North Carolina to northwestern Arkansas, including south-central Kentucky (Metcalfe County, Peter W. Messer pers. comm. 2012), south to eastern Texas and southern Florida [see Allen 1972: Fig. 164].
USA: AL, AR, FL, GA, KY, LA, MS, NC, SC, TN, TX
Twenty species in the Neotropical Region, one of them extending into southern United States.
Will (2005) provided a key for the identification of the species of this genus.
This genus-group taxon has been ranked as a subgenus of Oxycrepis Reiche by Allen and Ball (1980) but as a distinct genus by Will (2005).
This species ranges from southern Arizona and southern Texas along the Gulf of Mexico coast south to southern Mexico (Allen and Ball 1980: 532). The record from southwestern Ohio (Dury 1906: 257) must be in error or based on a stray.
USA: AZ, TX – Mexico
Worldwide, with about 2, 620 species. The North American fauna is represented by about 255 species (9.7% of the world fauna), of which six are adventive, arrayed in 38 genus-group taxa including the eastern endemic Abacidus (five species), Cyclotrachelus (43 species), Cylindrocharis (three species), Feronina (two species), Gastrellarius (three species), Gastrosticta (ten species), Lophoglossus (six species), Monoferonia (four species), Paraferonia (one species), and Piesmus (one species), the western endemic Anilloferonia (three species), Leptoferonia (26 species), Orsonjohnsonus (one species), and Pseudoferonina (nine species), as well as Hypherpes (60 species) and Lamenius (one species).
Twenty-seven species in the Neotropical Region, one of them reaching southwestern United States. The species are arrayed in two subgenera: Abaris s.str. (13 species) and Abaridius (14 species).
Will (2002b) published a revision of all known species.
A cladistic analysis placed the genus Neotalus Will, with one South American species, as the sister-group to Abaris (Will 2002b).
Fourteen species in the Neotropical Region, one of them reaching southwestern United States.
This species is known from southern Arizona, possibly also southeastern California as suggested by the type locality, Sonora in northwestern Mexico, and the southern part of the Baja California Peninsula [see Will 2002b: Fig. 17].
USA: AZ [CA] – Mexico
Seven species in the Neotropical Region, one of them reaching southwestern United States.
There is no taxonomic revision or available key for the identification of the species.
This species is known from southern Arizona (Schaeffer 1910: 393, as Pterostichus arizonicus; Graham, Pima and Santa Cruz Counties, UASM) south to Chiapas (UASM).
USA: AZ – Mexico
About 140 species (Lorenz 2005: 266-269) in the Nearctic (13 species) and Palaearctic (128 species) Regions; five of the North American species extend into northern Mexico and Cuba. The species of Poecilus are arrayed in seven subgenera: Poecilus s.str. (about 70 species), Angoleus Villa and Villa (23 species), Parapedius Seidlitz (two species in the Mediterranean region), Carenostylus Chaudoir (one Mediterranean species), Derus (23 species), Metapedius Fiori (one species from Italy), and Pseudoderus Seidlitz (18 species in western Asia).
No review or identification key is available for the North American species and a revision of the genus is needed.
Lyropedius Seidlitz (two eastern European species) is treated as a subgenus of Poecilus by some authors, including Kryzhanovskij and Abdurakhmanov (1983). Bousquet (1999: 58) pointed out that the taxon, based on character of the male genitalia, is probably not closely related to Poecilus. The taxon is listed as a subgenus of Pterostichus by Bousquet (2003d: 498).
About 70 species (Lorenz 2005: 266-268) in the Nearctic (12 species, of which five extend into northern Mexico or Cuba) and Palaearctic (about 60 species) Regions.
Chaudoir (1876c) reviewed the species but his work is outdated. Lindroth’s (1966) monograph covers seven of the 12 North American species.
[chalcites group]
This species ranges from southwestern New Brunswick (Majka et al. 2007: 9) to the Black Hills in western South Dakota (Kirk and Balsbaugh 1975: 21), south to northern Chihuahua (Bates 1891a: 249), southern Texas (Johnson 1978: 67), and the Florida Panhandle (Okaloosa County, CNC); also recorded from Cuba (Darlington 1934: 91). The records from Arizona (Snow 1906b: 162, as Poecilus sayi) and “North Dakota” (Bousquet and Larochelle 1993: 165) need confirmation.
CAN: NB, ON, QC USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [AZ, ND] – Cuba, Mexico
This species ranges from “Colorado” (LeConte 1858a: 28) to west-central Nevada (Bechtel et al. 1983: 474), south to the Baja California Peninsula (Horn 1894: 309) and “New Mexico” (Schaupp 1882c: 41). The record from est-central South Dakota (Kirk and Balsbaugh 1975: 21) needs confirmation.
USA: AZ, CA, CO, NM, NV, UT [SD] – Mexico
The range of this species extends from southern Manitoba to the Fraser River in south-central British Columbia (Lindroth 1966: 481), south to southern California (Fall 1901a: 44) and the Mexico City environs on the Mexican Plateau (Ball and Shpeley 1992a: 52).
CAN: AB, BC, MB, SK USA: AZ, CA, CO, ID, KS, MN, MT, ND, NE, NM, NV, OK, OR, SD, TX, UT, WA, WY – Mexico
[lucublandus group]
This species is known for sure only from the syntype. The records from northeastern Kansas (Horn 1872c: 385; Popenoe 1878: 78) are suspect.
USA: [CO, KS]
Adults of this species are structurally similar to, and possibly conspecific with, those of Poecilus lucublandus Say as defined by Lindroth (1966: 482-483).
This species is found from western Ontario (CNC) to southeastern Alberta (Lindroth 1966: 484), south to southeastern Oregon (Harney County, James R. LaBonte pers. comm. 2009; Hatch 1953: 115), central Wyoming (Lavigne 1977: 47), western South Dakota (Kirk and Balsbaugh 1975: 21), and western Minnesota (Tinerella and Rider 2001: 320; Gandhi et al. 2005: 926). The record from northern British Columbia (Jarrett and Scudder 2001: 382) needs confirmation.
CAN: AB, MB, ON, SK USA: MN, MT, ND, OR, SD, WY [BC]
This species is known from western Oregon (Hatch 1953: 115, as Pterostichus occidentalis) to southern California (Fall 1901a: 45, as Pterostichus occidentalis).
USA: CA, OR
This species has been known for a long time under the name Poecilus occidentalis (Dejean, 1828) but Lindroth (1966: 484) showed that Poecilus cursitor and Poecilus occidentalis are different species.
This species ranges from southeastern Washington (Hatch 1953: 115) to southern California (Fall 1901a: 44; Moore 1937: 9) and southern Arizona (Wickham 1898: 300). The records from New Mexico (Wickham 1896a: 157; Fall and Cockerell 1907: 158) and northeastern Kansas (Popenoe 1878: 78) are probably based on misidentified specimens.
USA: AZ, CA (CHI), OR, WA
This widely distributed species ranges from Cape Breton Island (Lindroth 1954c: 303) to Vancouver Island, north at least to Fort Smith in southern Northwest Territories (Lindroth 1966: 483), south to “Oregon” (Hatch 1953: 116), northern New Mexico (Fall and Cockerell 1907: 158), the Texas Panhandle (Michels et al. 2010: 743), southwestern Oklahoma (Kondratieff et al. 2005: 173), northern Georgia (Fattig 1949: 26), and central South Carolina (Ciegler 2000: 61).
CAN: AB, BC (VCI), MB, NB, NS (CBI), NT, ON, PE, QC, SK USA: CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY
The lectotypes of Poecilus lucublandus acomanus Casey and Poecilus lucublandus louisinus Casey are conspecific with members of Poecilus lucublandus as defined by Lindroth (1966) and these names are here placed in synonymy. However, the species exhibits considerable structural variation within its range and it is possible, even likely, that more than one species are included within the actual concept of Poecilus lucublandus.
This species is known from southern California (Fall 1901a: 45).
USA: CA
This species is known yet only from north-central (LeConte 1853a: 254) and southeastern Colorado (Michels et al. 2008), “Kansas” (Horn 1872c: 385), and a few localities in central Mexico (Bates 1882a: 84).
USA: CO, KS – Mexico
This species ranges from southeastern Arizona (Casey 1913: 137, as Poecilus snowi) south at least to the state of Oaxaca in Mexico (Bates 1882a: 84).
USA: AZ – Mexico
This species is known from southeastern Arizona (Snow 1906b: 162) and southern Texas (Snow 1906a: 141; Duval County, CMNH). The records from Kansas (Knaus 1901: 110; Snow 1903: 193) need confirmation.
USA: AZ, TX [KS]
Twenty-three species (Lorenz 2005: 269), mostly in the arctic and subarctic areas, in North America (one species), Asia (21 species), and eastern Europe (three species).
No revision or identification key is available for the species. Lindroth (1966: 476-478) covered the North American species.
This species ranges from the Anderson River delta in northern Northwest Territories (Lindroth 1966: 478) to the Titaluk River (North Slope County) in northern Alaska (CNC). The species is also known from a few localities in eastern Siberia (Alfimov and Berman 2009: Fig. 1). Fossil remnants of this species, from the early Pleistocene and Late Wisconsin, have been found in the lower Kolymian Basin in northeastern Siberia (Matthews 1974a: 208) and western Alaska (Matthews 1974b: 1365) respectively.
CAN: NT, YT USA: AK – Holarctic
Six species in the temperate regions of eastern North America.
Will (1999) reviewed the species and provided a key for their identification.
This species is known from a few localities along the Atlantic Coastal Plain and Piedmont Plateau from central New Jersey (Smith 1910: 205) to central South Carolina (Ceigler 2000: 62). The record from “Pennsylvania” (LeConte 1873a: 316) needs confirmation.
USA: DC, GA, MD, NC, NJ, SC, VA [PA]
This species ranges from southwestern Indiana (Blatchley 1910: 97; Schrock 1985: 354) to southeastern Iowa (Wickham 1911b: 6; Henry County, CNC), south to eastern Texas and central Florida (Will 1999: 272, Fig. 35). The record from North Carolina (Brimley 1938: 121) needs confirmation; that from “New York” (Wickham 1895a: 187) is likely in error.
USA: AL, AR, FL, GA, IA, IL, IN, KS, KY, LA, MO, MS, OK, TN, TX [NC]
This species ranges from southern Quebec (Larochelle 1975: 105) to southeastern Minnesota (Gandhi et al. 2005: 926), south to Missouri (Summers 1873: 145) and West Virginia (Will 1999: 270). The records from North Carolina (Brimley 1938: 121), South Carolina (Kirk 1970: 11), and Georgia (J.E. LeConte 1849: 26; Fattig 1949: 26) need confirmation.
CAN: ON, QC USA: CT, IA, IL, IN, MA, MI, MN, MO, NJ, NY, OH, PA, VT, WI, WV [GA, NC, SC]
This species ranges from Long Island in New York (Schaupp 1883b: 31) and New Jersey to central Missouri, north to northeastern Illinois, south to southeastern Louisiana (Saint Tammany Parish, Igor M. Sokolov pers. comm. 2009) and central Florida [see Will 1999: Fig. 34].
USA: AL, AR, FL, GA, IL, IN, KY, LA, MO, MS, NC, NJ, NY, PA, SC, TN, VA
As far as known, this species ranges along the Coastal Plain from the coast of South Carolina to central Florida, west to east-central Texas, north along the Mississippi Valley to southern Indiana (Will 1999: 271). The records from “Iowa” (Jaques and Redlinger 1946: 297), “Illinois” (Wolcott 1900: 469), “New York” (Wickham 1895a: 187; Casey 1913: 144), New Jersey (Smith 1910: 205), and southeastern Pennsylvania (Rathvon 1869: 526, as Pterostichus complanata) need confirmation.
USA: AL, AR, FL, GA, IN, LA, MS, SC, TX [IA, IL, NJ, NY, PA]
This species is known from a few localities along or near the Atlantic Coast from New Hampshire to Virginia (Will 1999: 268).
USA: CT, MA, MD, NH, RI, VA
One species in the temperate regions of eastern North America.
This species is found from southern Ohio (Purrington and Stanton 1996: 43; Silverman et al. 2008: 733) south to “Texas” (LeConte 1858a: 28), southern Mississippi (Hancock County, CNC), and central Florida (Peck and Thomas 1998: 19). Bates (1882a: 83) recorded it from “Mexico.”
USA: AL, AR, FL, GA, IL, KY, LA, MS, NC, OH, OK, SC, TN, TX, VA, WV – Mexico
Three species in the temperate regions of eastern North America.
Darlington (1932) provided a key for the identification of the species.
This species is found along the Appalachian Mountains from southern West Virginia (Hoffman 1998: 36) to northern Georgia (Leng 1910: 73; Fattig 1949: 21) and northwestern South Carolina (Ciegler 2000: 63).
USA: GA, NC, SC, VA, WV
This species ranges from the Nova Scotia Peninsula (Schaupp 1882c: 41; Chritopher G. Majka pers. comm. 2008) to western Minnesota (Gandhi et al. 2005: 926), south along the Appalachian Mountains to northern Alabama (Löding 1945: 15), northern Georgia (Leng 1910: 73; Fattig 1949: 21), and northwestern South Carolina (Ciegler 2000: 63).
CAN: NB, NS, ON, QC USA: AL, CT, DC, GA, IL, IN, KY, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, SC, TN, VA, VT, WI, WV
This species is known from the Great Smokies along the Tennessee and North Carolina border (Darlington 1932: 155; Barr 1969: 81) and Rabun Bald in northeastern Georgia (Fattig 1949: 22).
USA: GA, NC, TN
About 40 species in the temperate areas of the Nearctic (two species, one of them adventive) and Palaearctic Regions, arrayed in two subgenera, both represented in North America.
One North American species along the Pacific Coast.
Hacker (1968) redescribed the species.
This rarely collected species is known from one specimen collected along the west coast of Vancouver Island (Bousquet 2000: 81) and a few others collected along or near the coast of Oregon (Hacker 1968: 42).
CAN: BC USA: OR
Thirty-six species in Asia (29 species) and Europe (seven species), one of them adventive in eastern North America.
There is no taxonomic revision of the subgenus. The sole species in North America was included in Lindroth’s (1966: 443) monograph.
This European species is adventive in North America where it is known from a few localities in Cape Breton Island (Bousquet 1987a: 124), Prince Edward Island (CNC), and New Brunswick (Kent County, Christopher G. Majka pers. comm. 2011). The first inventoried specimen collected on this continent was found in Cape Breton Highland National Park in 1984 (Bousquet 1987a: 124). The previous North American record from Hemmingford, Quebec (Darlington 1940b: 252) was based on a mislabeled specimen (see Bousquet 1987a: 124).
CAN: NB, NS (CBI), PE – Adventive
Two Holarctic species in the arctic, subarctic, and boreal regions.
Lindroth (1966) redescribed both species and provided a mean for their identification through his key to Canadian species of Pterostichus.
This Holarctic species ranges in the Palaearctic Region from northern European Russia to the Far East, south to Manchuria (Bousquet 2003d: 517), and in the Nearctic Region from Alaska (Lindroth 1966: 533) to Newfoundland (Lindroth 1955a: 88), south to high mountains in New England (Lindroth 1966: 533), the Black Hills in southwestern South Dakota (Robert L. Davidson pers. comm. 2008), and Wyoming (Lindroth 1966: 533). The record from “New York” (Notman 1928: 226) needs confirmation; that from Ohio (Everly 1927: 155) is in error. Fossil remnants of this species, dated between about 10, 400 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96) and Cape Breton Island in Nova Scotia (Miller 1997: 250); others, believed to be 2.0-2.5 million years old, have been found in Greenland (Bennike and Böcher 1990: 336; Böcher 1995: 28).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, SK, YT USA: AK, ME, MT, NH, SD, VT, WY [NY] – Holarctic
This Holarctic species ranges from the northern part of European Russia (Bousquet 2003d: 517) to the Arctic Slope in northern Alaska. The record from “Yukon Territory” (Ball and Currie 1997: 452) could not be confirmed.
USA: AK [YT] – Holarctic
Thirty-two species (Lorenz 2005: 265-266) in the temperate regions of North America (two species), Asia (29 species), and Europe (one species), arrayed in two subgenera (Bousquet 1999: 90): Myas s.str. for the European species and Trigonognatha for the other ones.
Thirty-one species (Lorenz 2005: 266) in eastern North America (two species) and eastern Asia (29 species).
Allen (1980) revised the North American under the generic name Neomyas. Lindroth (1966) redescribed both species.
This species ranges from southwestern Connecticut (Krinsky and Oliver 2001: 104) to northeastern Illinois, north to the northwestern region of the lower peninsula of Michigan (Allen 1980: 20; Hatch 1925: 549), south to northern Mississippi (Union County, Drew A. Hildebrandt pers. comm. 2010), northern Alabama (Löding 1945: 15), and northwestern Georgia (Fattig 1949: 21). The records from southern Wisconsin (Rauterberg 1885: 16) and “Massachusetts” (Bousquet and Larochelle 1993: 168) need confirmation.
CAN: ON USA: AL, CT, DC, DE, GA, DE, IL, IN, KY, MD, MI, MS, NC, NJ, NY, OH, PA, TN, VA, VT, WV [MA, WI]
This species is found from “Nova Scotia” (Larochelle and Larivière 1990a: 29) to northwestern Minnesota (Allen 1980: 24), south to northern Alabama (Löding 1945: 15) and Georgia (Fattig 1949: 21, as Myas foveatus). A specimen simply labeled from Texas is known (Allen 1980: 26).
CAN: NB, NS, ON, QC USA: AL, CT, GA, IA, IL, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WI, WV [TX]
About 1, 055 species (Lorenz 2005: 269-287, 289, as Pterostichus, Lyropedius, and Abacidus) in the Nearctic (about 180 species, of which four are adventive) and Palaearctic (about 875 species) Regions. Percolaus Bates with two Middle American species, Mayaferonia Ball and Roughley with two Middle American species, and Allotriopus Bates with eight Mexican species are also includes in the genus Pterostichus by some authors (e.g., Ball and Roughley 1982).
Pterostichus (Steropus) madidus (Fabricius), a European species, is known in North America from two specimens, one collected in 1913 in Quebec (Lindroth 1966: 476) and the other one in 1984 in Somerset County, Pennsylvania (Cameron and Reeves 1990: 127). Until more specimens are collected on this continent, the species is not considered as a North American resident.
Based on phylogenetic analyses from two nuclear gene sequences (wingless and 28SrDNA), Sasakawa and Kubota (2007) concluded that the genus Pterostichus sensu Bousquet (1999) was monophyletic with the exception of Bothriopterus which forms a clade with Stomis and Myas.
Thirteen species in the boreal and temperate areas of the Nearctic (three species, one of them adventive) and Palaearctic (11 species) Regions.
Bousquet and Webster (2004) reviewed the Nearctic species.
This subgenus was known until the late 1980s under the name Lagarus Chaudoir.
[commutabilis group]
This species ranges from Cape Breton Island and Prince Edward Island to south-central British Columbia, south to the Lake Tahoe region in the Sierra Nevada, southern Colorado along the Rocky Mountains, northeastern Kansas (Popenoe 1877: 23, as Pterostichus erythropus), southern Indiana, and Delaware along the Atlantic Coast [see Bousquet and Webster 2004: Fig. 5].
CAN: BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CA, CO, CT, DE, IA, IL, IN, KS, MA, ME, MI, MN, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VT, WA, WI
This species ranges from Nova Scotia (Christopher G. Majka pers. comm. 2004) to the Missouri River in southeastern South Dakota and eastern Nebraska, south to southwestern Texas (Dajoz 2007: 23, as Pterostichus commutabilis), northern Alabama, and northern Florida [see Bousquet and Webster 2004: Fig. 6].
CAN: NS, ON, QC USA: AL, CT, FL, GA, IA, IL, IN, LA, MA, MD, ME, MI, MN, NE, NH, NJ, NY, OH, PA, RI, SC, SD, TX, VA, VT, WI
[vernalis group]
This European species is adventive in North America where it is known from southern Quebec, south of the Saint Lawrence River, eastern Ontario (near Avonmore, CNC), and northern Vermont (Bousquet and Webster 2004: 658). The first inventoried specimen collected on this continent was found in 1997 (Byers et al. 2001: 84). The previous record from Quebec (Lindroth 1966: 503) was based on a mislabeled specimen.
CAN: ON, QC USA: VT – Adventive
Twenty-eight species (Lorenz 2005: 275) in the boreal and temperate areas of the Nearctic (four species, one of them adventive) and Palaearctic (25 species) Regions.
Lindroth (1966) reviewed the Nearctic species.
This small Pterostichus is known from New Brunswick (Webster and Bousquet 2008: 18) to northern Wisconsin (Marquette County, CMNH; Rauterberg 1885: 16; Messer 2010: 37), south to central Pennsylvania (Clinton County, CMNH) and northeastern New Jersey (Bergen County, CNC).
CAN: NB, ON, QC USA: CT, MA, ME, NH, NJ, NY, PA, RI, WI
The range of this species extends from the Saguenay River in Quebec (Larochelle 1975: 101) to east-central British Columbia (Jarrett and Scudder 2001: 383), south to northeastern New Mexico (Fall and Cockerell 1907: 158; San Miguel County, MCZ), the Texas Panhandle (Robert L. Davidson pers. comm. 2008), and the District of Columbia (Ulke 1902: 6). The records from “Georgia” (J.E. LeConte 1849: 26) and southern Louisiana (Summers 1874a: 80) need confirmation.
CAN: AB, BC, MB, NB, ON, QC, SK USA: AR, CO, CT, DC, IA, IL, IN, KS, MA, MI, MN, MO, MT, ND, NE, NH, NJ, NM, NY, OH, OK, PA, SD, TX, VT, WI [GA, LA]
This species occurs from Newfoundland (Lindroth 1955a: 91) to the Gulf Coast of Alaska (Lindroth 1966: 506), south at least to northeastern Washington (Pend Oreille County, CNC), eastern Kansas (Popenoe 1877: 23; Lindroth 1966: 505), and southwestern Virginia (Hoffman 1998: 39).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), ON, PE, QC, SK, YT USA: AK, CT, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WA, WI, WV
This European species is adventive in North America on both coasts where it is found in southeastern Newfoundland (Lindroth 1955a: 91; Larson and Langor 1982: 593) and the Vancouver area in British Columbia (Spence and Spence 1988: 158, Fig. 8). The first inventoried specimen collected on this continent was found in Newfoundland in 1937 (see Lindroth 1955a: 91).
CAN: BC, NF – Adventive
Eighteen species (Lorenz 2005: 273) in the arctic (marginal), subarctic, boreal, and temperate areas of the Nearctic (six species, one of them circumboreal) and Palaearctic (12 species, including two in the Himalayas) Regions, with one species (Pterostichus tropicalis Bates) in the mountains of northern Mexico.
Tschitschérine (1900d) published a key to the species but his work is outdated. Lindroth (1966: 484-491) reviewed the North American species under his adstrictus and mutus groups.
[adstrictus group]
This circumpolar species ranges from Iceland to the coast of Bering Sea (Bousquet 2003d: 487) and from Alaska, including the Aleutian, Kodiak, and possibly Pribilof Islands (Lindroth 1966: 487), to Newfoundland (Lindroth 1955a: 94), south to north-central Pennsylvania (LeConte 1867a: 346, as Platysma luczotii), northeastern Indiana (Blatchley 1910: 98, as Platysma luczotii; Schrock 1985: 353), the Black Hills in western South Dakota (Kirk and Balsbaugh 1975: 22), the Black Range in west-central New Mexico (Fall and Cockerell 1907: 158, as Platysma luczotii), and the Sierra Nevada in California (Dajoz 2007: 16). The record from Virginia (Goulet 1974a: 19) is apparently based on misidentified specimens of Platysma pensylvanicus (Hoffman 1998: 39); that from “Nebraska” (Schaupp 1882c: 42, as Platysma luczotii) probably refers to Colorado.
FRA: PM CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, IA, ID, IN, MA, ME, MI, MN, MT, ND, NH, NM, NY, OR, PA, RI, SD, UT, VT, WA, WI, WY – Holarctic
This species ranges from southern British Columbia (Lindroth 1966: 489) south to northern Utah (Cache, Davis and Utah Counties, Robert L. Davidson pers. comm. 2012) and eastern Oregon (Hatch 1953: 118).
CAN: BC USA: ID, OR, UT, WA
The range of this species extends from Newfoundland (Lindroth 1955a: 93) to central British Columbia, north to Fort Smith in southern Northwest Territories (Lindroth 1966: 487), south to the Black Hills in western South Dakota (Kirk and Balsbaugh 1975: 22), southeastern Iowa (Wickham 1911b: 6), and west-central Virginia (Hoffman 1998: 39).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: CT, IA, IL, IN, MA, MD, ME, MI, MN, ND, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WI, WV
[mutus group]
This species ranges from Vancouver Island (Lindroth 1966: 491) and northern Idaho (Idaho County, CNC) south to central New Mexico (Bernalillo County, CMNH), southern Arizona (Wickham 1898: 300; Snow 1906b: 162), and southern California (Fall 1901a: 45; Moore 1937: 9).
CAN: BC (VCI) USA: AZ, CA (CHI), CO, ID, NM, NV, OR, UT, WA
This species ranges from western Newfoundland (Lindroth 1955a: 92) to “Montana” (Lindroth 1966: 490), south to northeastern New Mexico (San Miguel County, CNC), northeastern Kansas (Popenoe 1877: 23), and northeastern Georgia (Leng 1910: 73; Fattig 1949: 27); isolated in southwestern British Columbia, including Vancouver Island (Lindroth 1966: 490). The record from “Idaho” (Bousquet and Larochelle 1993: 170) needs confirmation. Lindroth (1966: 490) postulated that the best explanation for the presence of this species in southwestern British Columbia is that it was accidentally introduced into the area.
CAN: BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC USA: CO, CT, DC, DE, GA, IA, IL, IN, KS, MA, MD, ME, MI, MN, MT, NC, ND, NE, NH, NJ, NM, NY, OH, PA, RI, SD, TN, VA, VT, WI, WV [ID]
This species ranges from southeastern South Dakota (Kirk and Balsbaugh 1975: 22) to southwestern New York (Smith 1910: 205), south to Virginia (Nicolay and Weiss 1934: 211; Carrington 2002: 108), central West Virginia (Carrington 2002: 108), and central Missouri (Boone County, CMNH).
USA: IA, IL, IN, KY, MD, MI, MO, NJ, NY, OH, PA, SD, VA, WI, WV
Eight species in the boreal and temperate regions of the Nearctic (three eastern species) and Palaearctic (five species, of which three occur in the Far East) Regions.
Lindroth (1966) reviewed the North American species. One new species was described subsequently by Goulet and Bousquet (1983).
[corvinus group]
The range of this species extends from Nova Scotia (Majka et al. 2007: 9) to south-central British Columbia, as far north as northeastern British Columbia (Lindroth 1966: 499-500) and southern Northwest Territories (CNC), south to eastern Washington (Hatch 1953: 117), northern Colorado (Haubold 1951: 704; Armin 1963: 224), southern Iowa (Union County, Foster F. Purrington pers. comm. 2011), and northeastern South Carolina (Ciegler 2000: 66). The record from southwestern California (Moore 1937: 9) is probably in error.
CAN: AB, BC, MB, NB, NS, NT, ON, PE, QC, SK USA: CO, CT, DC, GA, IA, IL, IN, MA, MD, ME, MI, MN, ND, NE, NH, NJ, NY, OH, PA, RI, SC, SD, VA, VT, WA, WI, WV
[ebeninus group]
This species is restricted to a small area from New Brunswick (Webster and DeMerchant 2012: 6) to western Ontario, south to northern Wisconsin (Messer 2010: 37), northern Michigan (Dunn 1985a: 12), and northeastern Pennsylvania (Pike County, CMNH) [see Goulet and Bousquet 1983: map 1].
CAN: NB, ON, QC USA: MI, NH, PA, VT, WI
This species ranges from “Rhode Island” (Sikes 2003: 7) to “Minnesota” (Gandhi et al. 2005: 926), including southernmost Ontario (Lindroth 1966: 499, possibly only as strays), south to “Texas” (Schaupp 1882c: 42; Nicolay and Weiss 1934: 209), southeastern Louisiana (Lindroth 1966: 498), and southern Florida (Peck and Thomas 1998: 20).
CAN: ON USA: AL, AR, DC, FL, GA, IA, IL, IN, LA, MD, MI, MN, MO, MS, NC, NJ, NY, OH, PA, RI, SC, TN, TX, VA, WI
Eighteen species (Lorenz 2005: 274-275) in the boreal and temperate regions of the Nearctic (two species) and Palaearctic (16 species) Regions.
Bousquet and Pilon (1984) commented on the structural differences between the two North American species.
This species ranges from Newfoundland to Vancouver Island, south to west-central Washington, central Colorado (Jefferson County, Robert L. Davidson pers. comm. 2008), east-central South Dakota, northern Illinois, and eastern Virginia [see Bousquet and Pilon 1984: Fig. 8].
FRA: PM CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CO, CT, DC, IA, ID, IL, IN, MA, MD, ME, MI, MN, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WA, WI, WV
This species ranges from eastern Newfoundland to northeastern Alberta, south to northern Colorado, northern Illinois, and southwestern North Carolina (Macon and Jackson Counties, CNC) [see Bousquet and Pilon 1984: Fig. 7].
CAN: AB, MB, NB, NF, NS (CBI), ON, PE, QC USA: CO, CT, IL, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, VT, WI, WV
Two species in the Appalachian region of eastern North America.
Bousquet (2006b) commented on the structural differences between the two species.
This species is known from northeastern Kentucky, eastern and central West Virginia, and southwestern Virginia (Bousquet 2006b: 159).
USA: KY, VA, WV
This species is found at high elevation in the Appalachian Mountains in North Carolina, Tennessee (Barr 1969: 72), Kentucky (Jackson County, Foster F. Purrington pers. comm. 2009), and northern Georgia (Fattig 1949: 24), including the Black, Bald, Great Balsam, and Great Smoky Mountains. The record from “Virginia” (Bousquet and Larochelle 1993: 172) needs confirmation.
USA: GA, KY, NC, TN [VA]
One species in the Appalachian Mountains of eastern North America.
This species is endemic to the Appalachian Mountains from southwestern Virginia (Hoffman 1998: 38) to northern Georgia (Fattig 1949: 26) and northwestern South Carolina (Ciegler 2000: 67), including the Great Balsam and Great Smoky Mountains (Barr 1969: 80).
USA: GA, NC, SC, TN, VA
Nine species in the Pacific Northwest.
Bousquet (1985a) wrote a key for the identification of the species. Three new species were described subsequently (Bousquet 1992b; Bergdahl and Kavanaugh 2011).
[humidulus group]
This species is known from several localities in western Oregon. Two specimens simply labeled “Was” are also known (Bousquet 1985a: 258).
USA: OR [WA]
This species is known from western Washington (Bousquet 1985a: 259). The record from northwestern Oregon (Hatch 1953: 114) probably refers to Pterostichus campbelli.
USA: WA
This species is known from the type locality in southern Washington and one locality in northwestern Oregon (Westcott et al. 2006: 8).
USA: OR, WA
[lanei group]
This species is known from Latah and Idaho Counties (CNC) in northern Idaho and Mineral County in west-central Montana (James C. Bergdahl pers. comm. 2008).
USA: ID, MT
This species is known from two creeks near their confluences with the South Fork of the Payette River near Lowman in west-central Idaho (Bergdahl and Kavanaugh 2011: 81).
USA: ID
This species is known from a few localities in southeastern Washington and west-central Idaho (Bousquet 1985a: 259).
USA: ID, WA
This species is known only from the type locality.
USA: ID
This species is known only from the type locality in northeastern Idaho.
USA: ID
This species is known only from several specimens collected at the type locality in northeastern Idaho.
USA: ID
Ten species in the temperate regions of North America, east of the Rocky Mountains.
Bousquet (1992b) published a preliminary key to the species.
This species is known only from the holotype.
USA: TX
This species is known only from two specimens collected in “Georgia.”
USA: GA
This species is known only from a few localities in west-central Georgia (Fattig 1949: 25), northern Florida (Bousquet 1992b: 511, 513) including the Panhandle (Wakulla County, CMNH), and “Louisiana” (Bousquet 1992b: 513).
USA: FL, GA, LA
This species is known only from several localities in Mississippi (Bousquet 1992b: 515; Drew A. Hildebrandt pers. comm. 2007) and western Alabama (Greene County, Foster F. Purrington pers. comm. 2010).
USA: AL, MS
This species is known from a few localities from northern Georgia (Fattig 1949: 26, as Gastrosticta amnicola) to eastern Oklahoma (Latimer and Pushmataha Counties, CNC; Cherokee County, CMNH), north to east-central Missouri (Bousquet 1992b: 516), south to “Texas” (Chaudoir 1878: 66) and southwestern Alabama (Löding 1945: 16, as Gastrosticta amnicola).
USA: AL, AR, GA, MO, MS, OK, TX
This species is known only from a few localities in Grayson County (CNC) in Texas.
USA: TX
This species ranges from southwestern Pennsylvania (Lindroth and Freitag 1969: 342) to “Iowa” (Jaques and Redlinger 1946: 297), south to east-central Louisiana (West Feliciana Parish, LSAM), northern Alabama (Löding 1945: 16; Madison County, CMNH), and northern Georgia (Fattig 1949: 25).
USA: AL, AR, GA, IA, IL, IN, KY, LA, MO, NC, OH, PA, TN, VA, WV
This species is known from a few localities in Oklahoma (Latimer and Marshall Counties, CMNH, CNC), eastern, central, and southern Texas (De Witt, Madison, Anderson, Lee, and Houston Counties, CMNH, CNC, MCZ; Riley 2011), and “Louisiana” (CNC).
USA: LA, OK, TX
This species is known from southwestern Florida (Collier County, Foster F. Purrington pers. comm. 2011) and from central and southern Mississippi (Will and Gill 2008: 121; Hancock and Leake Counties, CMNH, CNC) to southeastern Oklahoma (Pushmataha County, CNC), south at least to east-central Texas (Harris County, CNC).
USA: AR, FL, LA, MS, OK, TX
This species is known from eastern Kansas (Lindroth and Freitag 1969: 341; Douglas County, CNC, UASM) to “Iowa” (Jaques and Redlinger 1946: 297) and southwestern Illinois (Saint Clair County, MCZ; Wickham 1895a: 185), south to northwestern Louisiana (Bossier Parish, LSAM; LeConte 1853a: 241) and south-central Texas (Comal County, CMNH). The record from “Florida” (Wickham 1895a: 185) needs confirmation (see Leng 1915: 576).
USA: AR, IA, IL, KS, LA, MO, OK, TX [FL]
Two Palaearctic species (Pterostichus alexeji Zamotajlov and Kryzhanovskij and Pterostichus melanarius with three subspecies), one of them adventive in North America. The status of Pterostichus brevistylis Jeannel, described from Switzerland, remains unclear.
Sasakawa and Kubota (2006) combined members of Petrophilus Chaudoir (type species: Feronia findelii Dejean, 1828), Euryperis Motschulsky (type species: Euryperis uralensis Motschulsky, 1850), Morphnosoma, Euferonia, Feroperis Lafer (type species: Feronia jungens Tschitschérine, 1893), and Moritapterus Berlov (type species: Pterostichus thunbergi Morawitz, 1862) into a single subgeneric taxon, Petrophilus Chaudoir, 1838.
This European species is adventive in North America on both coasts and is now found from Newfoundland (Lindroth 1955a: 87; Larson and Langor 1982: 592) to Vancouver Island (Lindroth 1966: 492), south to northwestern California (Del Norte County, James R. LaBonte pers. comm. 1992), northern Utah (Davis County, CMNH), northern Colorado (Miller and Peairs 2008: 34), South Dakota (Ellsbury et al. 1998: 621; French et al. 2004: 557; Larsen and Purrington 2010: 571), northeastern Iowa (Purrington and Larsen 1997: 50), east-central Ohio (Usis and MacLean 1998: 67), and central Pennsylvania (Capogreco 1989b: 4; Byers et al. 2001: 85). The first inventoried specimen collected on this continent was found in Nova Scotia in 1926 (Lindroth 1957c: 153) and in the west in Seattle in 1927 (Hatch 1933c: 120).
CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CA, CO, CT, IA, ID, IL, IN, MA, ME, MI, MN, MT, ND, NH, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WY – Adventive
Two other subspecies of Pterostichus melanarius are found in Europe, Pterostichus melanarius bulgaricus Lutshnik and Pterostichus melanarius cardioderus Chaudoir (Bousquet 2003d: 499).
Eleven species in the boreal and temperate regions of eastern North America (six species) and Japan (five species: Pterostichus robustistylis Sasakawa and Kubota, Pterostichus sejunctus Bates, Pterostichus thunbergi Morawitz, Pterostichus tschitscherianus Jacobson, and Pterostichus tuberifer Sasakawa).
Nicolay and Weiss (1934) and Lindroth (1966) summarily reviewed the North American species. A thorough revision of this group is needed.
This subgenus is often combined with Morphnosoma Lutshnik (e.g., Sasakawa and Kubota 2005; Lorenz 2005). The Japanese species are phenetically most similar to the North American Pterostichus lachrymosus (see Bousquet 1999: 137).
[lachrymosus group]
This species occurs from New Brunswick (Bousquet 1987a: 124) to the Ontario Peninsula (Lindroth 1966: 495), south along the Appalachian Mountains to the Great Smokies in eastern Tennessee (Stockton 1954: Fig. 19; CNC) and to northern Georgia (Fattig 1949: 24). The record from southwestern Arkansas (Stockton 1954: 16) is probably in error.
CAN: NB, ON, QC USA: CT, DC, GA, KY, MA, MD, ME, NC, NH, NJ, NY, OH, PA, SC, TN, VA, VT, WV
[stygicus group]
This species is found from Newfoundland (Lindroth 1955a: 86) to eastern South Dakota (French et al. 2004: 557), north to the James Bay area in Quebec (Larochelle 1975: 86), south along the Appalachian Mountains to eastern Tennessee (Nicolay and Weiss 1934: 206, as Euferonia coracina roanica) and northern Georgia (Fattig 1949: 24). The record from the Black Hills in southwestern South Dakota (Stockton 1954: 15) is doubtful.
FRA: PM CAN: LB, NB, NF, NS (CBI), ON, PE, QC USA: CT, DC, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, SC, SD, TN, VA, VT, WI, WV
Until the status of this taxon is better defined, its distribution remains inadequately documented. It has been recorded from Indiana, Illinois, Iowa, Missouri (Casey 1918: 367), and South Carolina (Nicolay and Weiss 1934: 200, as Euferonia iripennis).
USA: IA, IL, IN, MO, SC
Lindroth (1966: 493) discussed the status of this taxon and at the time regarded it as an extreme, southern form of Pterostichus stygicus. Lindroth (1975: 124) later listed it as specifically distinct from Pterostichus stygicus and listed Pterostichus iripennis (Nicolay and Weiss) as synonym.
This species is known from scattered localities from southwestern Quebec (Bousquet 1998: 105) to eastern North Dakota (Cass and Ransom Counties, CNC), north to southern Manitoba (Lindroth 1966: 495), south to southern South Dakota including the Black Hills (Kirk and Balsbaugh 1975: 23), northeastern Illinois (Purrington et al. 2002: 201), and northeastern New Jersey (FSCA). The record from Maryland (Erwin 1981b: 162), based on a single old specimen, needs confirmation.
CAN: MB, ON, QC USA: IL, MI, MN, ND, NJ, NY, OH, PA, SD, WI [MD]
The range of this species extends from northeastern New York (Notman 1928: 222) to southern Michigan (Eaton and Washtenaw Counties, UMAA), including southernmost Ontario (Lindroth 1966: 496), south to southwestern Indiana (Blatchley 1910: 93) and along the Appalachian Mountains to northern South Carolina (Ciegler 2000: 67) and central Alabama (Löding 1945: 16; Stockton 1954: Fig. 18). The records from eastern Kansas (Snow 1880: 78, as Pterostichus protensus; Knaus 1907: 233) are likely in error.
CAN: ON USA: AL, GA, IN, KY, MI, NC, NY, OH, PA, SC, TN, VA
This species ranges from Maine (Dearborn and Donahue 1993: 5) to central Iowa (O’Rourke et al. 2008: 126), south to “Louisiana” (Nicolay and Weiss 1934: 201) and southwestern Georgia (Fattig 1949: 24), west to northeastern Kansas (Popenoe 1877: 23). The record from “Minnesota” (Bousquet and Larochelle 1993: 174) needs confirmation. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 26).
CAN: ON, QC USA: CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [MN]
Eleven species in the arctic, subarctic, and boreal regions of North America (four species) and Asia (ten species, one of them extending to European Russia). Three species are Holarctic.
Lindroth (1966) reviewed the North American species. Budarin (1976) reviewed the world fauna, including two species (Pterostichus mirus Tschitschérine and Pterostichus rugosus Gebler) that belong, however, to the subgenus Metallophilus Chaudoir. Recently Sundukov (2005) revised all known species and provided a key for their identification.
This subgenus has been known for sometimes under the name Lyperopherus Motschulsky (e.g., Ball 1960b: 127; Lindroth 1966: 525).
This Holarctic species is found in the arctic regions of eastern Siberia and northeastern China (Bousquet 2003d: 497) and on this continent from the Seward Peninsula in Alaska to northwestern Northwest Territories (Lindroth 1966: 528).
CAN: NT, YT USA: AK – Holarctic
This Holarctic species ranges from the Yenisei River in Russia (Lindroth 1966: 530) to the Melville Peninsula in northern Nunavut, Canada [see Nielsen et al. 1987: Fig. 18c].
CAN: NT, NU, YT USA: AK – Holarctic
This species is found from Newfoundland (Lindroth 1955a: 87) to southeastern Yukon Territory (Lindroth 1966: 527), including northeastern British Columbia (CNC), north to northwestern Northwest Territories, south to central Alberta (Lindroth 1966: 527), northeastern Minnesota (Cook County, CNC), some mountains in New York (Notman 1928: 223), and “Massachusetts” (Wickham 1895a: 186). The record from Cuyahoga County in northern Ohio (Bubna 1902: 193) needs confirmation. Fossil remnants of this species, dated between about 11, 700 and 18, 100 years B.P., have been unearthed in northeastern Wisconsin, central Minnesota (Schwert 1992: 77), and central and southeastern Iowa (Schwert 1992: 77; Baker et al. 1986: 96).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: MA, ME, MI, MN, NH, NY, VT [OH]
Pterostichus punctatissimus Randall. This species is one of the most attractive Pterostichus in North America. The sculpture of the elytra is irregular giving the impression to the naked eye that the striae bear large punctures. For that reason Randall named the species punctatissimus meaning the most punctured. Although not as common as Pterostichus adstrictus, this species is also a characteristic element of boreal forests in the Nearctic Region.
This Holarctic species is found in the arctic regions from European Russia to the Melville Peninsula and Southampton Island in eastern Nunavut, Canada (Lindroth 1966: 528). Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96); others from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 28).
CAN: NT, NU, YT USA: AK – Holarctic
Eight species in North America (one Holarctic species) and Palaearctic Asia (eight species, one extending to European Russia).
According to Bousquet (1999: 145), this group is closely related and possibly paraphyletic in regard to Myosodus Fischer von Waldheim, a subgenus of 22 species centered in the Caucasus Mountains. Brinev and Shilenkov (2001) retained Tundraphilus as a valid subgenus, listing Pterostichus sublaevis Sahlberg, Pterostichus orion Tschitschérine, Pterostichus pfitzenmayeri Poppius, and Pterostichus kamtschaticus Motschulsky in it.
This Holarctic tundra species is found from the northern part of European Russia (Brinev and Shilenkov 2001) to northwestern Northwest Territories (Lindroth 1966: 531). Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96).
CAN: NT, YT USA: AK – Holarctic
Five species in the temperate regions of eastern North America.
Lindroth (1966: 535) provided a key to all species, including also Pterostichus obesulus LeConte, a species that actually belongs to the subgenus Gastrosticta. Sadek (1982) wrote a M.Sc. thesis on the taxonomy of this group.
Lindroth (1966: 534), followed recently by Lorenz (2005: 289), treated this group as a distinct genus. Structural characters of the adults and larvae clearly suggest that members of Abacidus are congeneric with those of Pterostichus. In fact, the group is probably closely related to Metallophilus and Myosodus (see Bousquet 1999: 148).
[fallax group]
This species ranges from east-central Iowa to North Carolina, including southern Michigan (Sadek 1982: 23), south to northern Florida (Peck and Thomas 1998: 20) and “Texas” (Schaupp 1882c: 41).
USA: AL, AR, FL, GA, IA, IL, MI, MO, MS, NC, SC, TN, TX
[hamiltoni group]
This species is known from scattered localities from Ohio (Purrington et al. 1989: 107; Usis and MacLean 1998: 67) and southwestern Pennsylvania (Horn 1880a: 139; Allegheny, Fayette, and Westmoreland Counties, CMNH) south to northern Georgia (Fattig 1949: 25).
USA: GA, MD, OH, PA, WV
In the Zoological Record for the year 1880, this species is registered (page 23) as “Pterostichus (Peristethus) maximiliani.”
[permundus group]
This species is found from eastern Maryland (Queen Annes County, Foster F. Purrington pers. comm. 2009) to southern Wisconsin (Sadek 1982: 34), including Pelee Island in southernmost Ontario (Lindroth 1966: 537), south to southern Louisiana (East Baton Rouge Parish, CNC) and the Florida Panhandle (Peck and Thomas 1998: 19).
CAN: ON USA: AL, FL, GA, IL, IN, KY, LA, MD, MO, MS, NC, OH, PA, SC, TN, VA, WI, WV
This species occurs from central New York (Hajek et al. 2007: 880) to southeastern South Dakota (Kirk and Balsbaugh 1975: 23; Ellsbury et al. 1998: 621), north to southernmost Ontario (Bousquet 1987a: 125) and northeastern Michigan, south to northeastern Texas and northeastern Florida (Sadek 1982: 25, Fig. 16). The records from Quebec (Larochelle 1975: 31) and “New Mexico” (Bousquet and Larochelle 1993: 174) are based on mislabeled specimens or are in error.
CAN: ON USA: AL, AR, FL, GA, IA, IL, IN, KS, LA, MI, MN, MO, MS, NE, NY, OH, OK, PA, SC, SD, TN, TX, WI
The range of this species extends from “New York” (Wickham 1895a: 186) to Iowa (Wickham 1888: 82; Jaques and Redlinger 1946: 297), south to eastern Arkansas (Sadek 1982: 31), Alabama (Löding 1945: 16), Georgia (Fattig 1949: 25; House and All 1981: 195; CMNH, MCZ), and South Carolina (Ciegler 2000: 68).
USA: AL, AR, GA, IA, MD, NC, NJ, NY, OH, PA, SC, TN
One species in the Pacific Northwest.
The species was included in Lindroth’s (1966: 473) monograph.
This species is known from northern Washington to central Oregon (Foltz 2011).
USA: OR, WA
One species in the boreal and temperate regions of North America.
The species was included in Lindroth’s (1966: 500) monograph.
This species has been included for a long time with the species of Melanius Bonelli.
This species is found from Newfoundland (Lindroth 1955a: 89-90) to the Okanagan Valley in south-central British Columbia, north to Fort Smith in southern Northwest Territories (Lindroth 1966: 500), south to northeastern Nevada (Elko County, CNC), central Colorado (Elias 1987: 632; Wickham 1902: 236), Missouri (Summers 1873: 134), and northern Georgia (Fattig 1949: 27).
CAN: AB, BC, MB, NF, NT, ON, QC, SK USA: CO, CT, DC, GA, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NV, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WV, WY
Forty-two species in the arctic, subarctic, boreal, and temperate regions of North America (three species), Palaearctic Asia (34 species), and Europe (five species).
Lindroth (1966: 474-475, 406-497, as circulosus and moestus groups) treated all three North American species in his review of the Canadian Pterostichus.
[circulosus group]
This species is known from a few localities in Yukon Territory (Ball and Currie 1997: 451; CNC) and Alaska (Lindroth 1966: 475). The species is also cited, with a question mark, from the Magadan region in eastern Siberia (Budarin 1985: 15).
CAN: YT USA: AK
[moestus group]
This species occurs from the neighborhood of New York City (Schaupp 1883b: 31; Ulster County, CMNH) to northeastern Ohio (Lee 1994: 60), south along the Appalachian Mountains to northern Georgia (Fattig 1949: 25; CNC) and northwestern South Carolina (Ciegler 2000: 67). The record from “Indiana” (Schrock 1985: 354) needs confirmation. The old records from Ontario and Quebec (see Lindroth 1966: 497) and Lake Superior (LeConte 1853a: 247) were based on misidentified or mislabeled specimens.
USA: DC, DE, GA, KY, MD, NC, NJ, NY, OH, PA, SC, VA, WV [IN]
This species is known from scattered localities from Staten Island, New York (Smith 1910: 205) to northwestern Pennsylvania (Forest County, CMNH), south along the Appalachian Mountains to northeastern Georgia (Fattig 1949: 25). The record from “Michigan” (Wickham 1895a: 185) is in error.
USA: GA, MD, NC, NJ, NY, PA, VA, WV
Four species in the Appalachian region of eastern North America.
Darlington (1932) reviewed the species and provided a key for their identification.
This subspecies is known from the Black Mountains in western North Carolina (Darlington 1932: 162; Barr 1969: 72) and from northeastern Georgia near the border with South Carolina (Fattig 1949: 21). The record from “Tennessee” (Bousquet and Larochelle 1993: 171) needs confirmation.
USA: GA, NC [TN]
This subspecies is endemic to the Great Smoky Mountains along the North Carolina-Tennessee border (Darlington 1932: 163).
USA: NC, TN
This species is found along the Appalachian region from southern Quebec (Larochelle 1975: 101) to Kentucky (Powell County, CNC) and northeastern Georgia (Fattig 1949: 23). The records from northwestern Indiana (Wolcott and Montgomery 1933: 126, as Monoferonia osculans) and “Illinois” (Schaupp 1882c: 41) need confirmation.
CAN: QC USA: CT, DC, GA, KY, MA, MD, ME, NC, NH, NJ, NY, OH, PA, VA, VT, WV [IL, IN]
This species is known from the Roan, Grandfather, and Black Mountains in North Carolina (Barr 1969: 72) and northern Georgia (Fattig 1949: 23).
USA: GA, NC
This species is apparently endemic to the Great Smoky and Unicoi Mountains along the North Carolina - Tennessee border (Barr 1969: 80).
USA: NC, TN
Three species in the Appalachian region.
Barr (1971a) revised the species.
This subspecies occurs in the Appalachians from the Black Mountains in North Carolina to northern Georgia (Barr 1971a: 9). The record from “Kentucky” (Bousquet and Larochelle 1993: 176) refers to Pterostichus acutipes kentuckensis.
USA: GA, NC, SC, TN
This subspecies is known from a few localities in central Kentucky (Barr 1971a: 11) and southwestern Virginia (Hoffman 1998: 37).
USA: KY, VA
This species is known from the Nantahalas to the Snowbird Mountains in Mason and Graham Counties, North Carolina (Barr 1971a: 12).
USA: NC
This species ranges from New Brunswick to southern Ontario (Lindroth 1966: 458), south along the Appalachian Mountains to northeastern Alabama (Löding 1945: 15) and northern Georgia (Fattig 1949: 21), including the higher mountains of Tennessee and North Carolina (Barr 1971a: 7). The records from “Iowa” (Bousquet and Larochelle 1993: 176) and southwestern Alabama (Löding 1945: 15) are likely in error.
CAN: NB, ON, QC USA: AL, CT, DC, GA, KY, MA, MD, ME, NC, NH, NJ, NY, PA, RI, SC, TN, VA, VT, WV
Twenty-six species in western North America.
Hacker (1968) revised the species. Since then, four new species have been described by Will (2007), one species (Pterostichus rothi) has been transferred from Anilloferonia to this subgenus (Will 2007), and one subspecies (Pterostichus stapedius yosemitensis) has been raised to species level by Bousquet (1999: 164).
[fenyesi group]
This species is restricted to the Coast Ranges in southwestern Oregon and northwestern California [see Hacker 1968: Fig. 35].
USA: CA, OR
This subspecies is found from the Eel River in Humboldt County southwards to Mendocino County, California [see Hacker 1968: Fig. 35].
USA: CA
This subspecies is known only from Humboldt County, north of Eel River, in northwestern California [see Hacker 1968: Fig. 35] and from southwestern Oregon (Curry County, James R. LaBonte pers. comm. 2009).
USA: CA, OR
[fuchsi group]
This species is found in western California from near Mount Shasta in Siskiyou County south to the Monterey area in Monterey County [see Hacker 1968: Fig. 38].
USA: CA
This species is known only from the holotype collected in northwestern California.
USA: CA
This species is restricted to the Coast Ranges in Mendocino and Sonoma Counties, California [see Hacker 1968: Fig. 36].
USA: CA
This species ranges along the Coast Ranges from southern Oregon southwards to Humboldt County in northern California [see Hacker 1968: Fig. 37].
USA: CA, OR
This species is found along the Pacific Coast of California south of the Eel River in Humboldt County to the mouth of the Navarro River in Mendocino County [see Hacker 1968: Fig. 37].
USA: CA
This species is, as far as known, restricted to a small area along the Pacific Coast from the Tomales Bay to the north side of the Pacific entrance of the San Francisco Bay [see Hacker 1968: Fig. 36].
USA: CA
This species is restricted to the Coast Ranges in southern Humboldt County and northern Mendocino County, California [see Hacker 1968: Fig. 37].
USA: CA
This species is found in southwestern Humboldt, southwestern Trinity, and northern Mendocino Counties in the Coast Ranges, northwestern California [see Hacker 1968: Fig. 37].
USA: CA
[hatchi group]
This species is known only from the holotype.
USA: CA
This species is restricted to a small area of the Sierra Nevada in Calaveras, El Dorado, and Tuolumne Counties, east-central California [see Hacker 1968: Fig. 39].
USA: CA
This species is restricted to the Sierra Nevada from the Yosemite National Park to near King Canyon National Park, California [see Hacker 1968: Fig. 39].
USA: CA
This species is restricted to a small area of the Sierra Nevada in Tuolumne County, California [see Hacker 1968: Fig. 39].
USA: CA
This taxon has been considered a subspecies of Pterostichus stapedius by Hacker (1968: 34) but regarded as a valid species by Bousquet (1999: 164).
[inanis group]
This species is restricted to the Coast Ranges of California from Mendocino County to southern Sonoma County, east to western Napa County [see Hacker 1968: Fig. 38].
USA: CA
This species is known from two localities in Tehama and Butte Counties in northern California.
USA: CA
This species is found in northern Idaho and western Montana (Hacker 1968: 39).
USA: ID, MT
This species is known from “Washington” to northern California along the Coast Ranges, from the Sierra Nevada, and from Reno in northwestern Nevada (Hacker 1968: 41). Hacker (1968: 41) implied that the species occurs in British Columbia but the record needs confirmation.
USA: CA, NV, OR, WA [BC]
This species is known only from the type locality.
USA: CA
[inopinus group]
This species ranges from northwestern Oregon, as far east as the eastern slopes of the Coast Ranges, southwards to northwestern California [see Hacker 1968: Fig. 34].
USA: CA, OR
This species is restricted to western Oregon, as far north as Mount Hood National Forest, and northwestern California [see Hacker 1968: Fig. 32]. The record from “Washington” in Bousquet and Larochelle (1993: 179) is based on two specimens from Yakima County which Hacker (1968: 11) regarded as mislabeled.
USA: CA, OR
This subspecies ranges from southwestern British Columbia, including Vancouver Island (Lindroth 1966: 471), southwards to the Green Peter Mountain (Hacker 1968: 13) in the Oregon Cascades [see Hacker 1968: Fig. 33].
CAN: BC (VCI) USA: OR, WA
This subspecies is endemic to the Willamette Valley and surrounding foothills in western Oregon [see Hacker 1968: Fig. 33].
USA: OR
[rothi group]
This species is known from four sites in Lincoln and Benton Counties in western Oregon (Brenner 2005).
USA: OR
[sphodrinus group]
This species is known from a few specimens collected in northern Idaho and along the western edge of Montana (Hacker 1968: 36; see Will and Gill 2008: Fig. 15).
USA: ID, MT
This species is found in the Rocky Mountains in southern Alberta, southeastern British Columbia (Lindroth 1966: 470-471), northeastern Washington, northern Idaho (Hatch 1953: 112), and western Montana (Russell 1968: 59; Edwards 1975: 55; Hansen et al. 2009: 353). The record from “Alaska” (Erwin et al. 1977: 4.33; Bousquet and Larochelle 1993: 182) is probably in error.
CAN: AB, BC USA: ID, MT, WA
This species is actually known only from the type locality (Hacker 1968: 37) in southern California.
USA: CA
Three species in the Pacific Northwest are currently recognized.
Hatch (1953) provided a key for the identification of the species.
Pterostichus rothi has been included within this subgenus by most authors but recent molecular data analyses (Will and Gill 2008: 115) suggest that the species is most closely related to members of Leptoferonia Casey.
This taxon is limited to the Coast Ranges in northwestern Oregon (James R. LaBonte pers. comm. 1994).
USA: OR
The name Pterostichus lanei (Hatch, 1935) is a secondary homonym of Pterostichus (Pseudoferonina) lanei Van Dyke, 1926. James R. LaBonte (pers. comm. 2012) is planning to propose a replacement name for Pterostichus lanei Hatch.
This taxon is confined to the Coast Ranges in southwestern Oregon (James R. LaBonte pers. comm. 1994).
USA: OR
This species is confined to the Cascade Range in Washington and Oregon (James R. LaBonte pers. comm. 1994).
USA: OR, WA
Sixty species in eastern (two species) and western (58 species) North America.
Casey (1913) reviewed most of the species then known (under planctus, californicus, menetriesi, congestus, and adoxus groups) but his key is very difficult to use. A taxonomic revision of the species is much needed.
Bousquet (1999) combined members of Leptoferonia, Anilloferonia, and Hypherpes into a single subgeneric taxon. Based on molecular sequence data, Will and Gill (2008) recovered these groups as monophyletic and suggested to recognize them as distinct subgenera.
[adoxus group]
This species ranges from Cape Breton Island (Bousquet 1987d: 105) to northern Minnesota (Petrice et al. 2002: 9), south along the Appalachian Mountains at least to eastern Tennessee (Carter and Morgan Counties, CMNH) and northwestern South Carolina (Ciegler 2000: 66). The records from Georgia (Fattig 1949: 21), northern Alabama (Löding 1945: 15), and Iowa (Wickham 1911b: 6; King 1914: 321) need confirmation since they could refer to Pterostichus tristis; that from “Wyoming” (Wickham 1896c: 132) is in error.
CAN: NB, NS (CBI), ON, QC USA: CT, DC, DE, IN, KY, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, SC, TN, VA, VT, WI, WV [AL, GA, IA]
This species has a similar distribution pattern than the preceding one ranging from Cape Breton Island (Bousquet 1987d: 105) to northern Minnesota (Petrice et al. 2002: 9), south to east-central Iowa (Iowa County, MCZ), Tennessee in the Great Smokies (CNC, MCZ), Georgia (Perrault 1973a: 37), and northwestern South Carolina (Ciegler 2000: 68). One old specimen labeled “La.” (MCZ) is probably mislabeled.
CAN: NB, NS (CBI), ON, PE, QC USA: CT, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV
[amethystinus group]
This species is found along the Pacific Coast from the Yakutat Bay in southeastern Alaska (Lindroth 1966: 465) to Mendocino County in California (Van Dyke 1919b: 5).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is known only from the lectotype collected in coastal California.
USA: CA
The lectotype of Pterostichus obsidianus Casey is structurally similar to and possibly conspecific with members of Pterostichus scutellaris LeConte.
I have seen this species from Marin and Fresno Counties in California (CNC); according to Van Dyke (1919b: 5), it extends south to Monterey County in California. The syntype studied by Lindroth (1966: 462) is not conspecific with the lectotype.
USA: CA
[arcanus group]
This species is known only from the lectotype collected in the Coast Ranges of California.
USA: CA
[castaneus group]
This species ranges from the southeastern coast of Alaska (Lindroth 1966: 466) to “California” (Dejean 1828: 327, as Feronia brunnea; Chaudoir 1868b: 336, as Feronia sejungenda), east to southeastern British Columbia (Lindroth 1966: 466) and “Idaho” (Van Dyke 1924a: 8).
CAN: BC (QCI, VCI) USA: AK, CA, ID, OR, WA
This species is known from a few localities in southern Oregon (Hatch 1936: 702).
USA: OR
[castanipes group]
At present, this species is known only from the Coast Ranges in north and central California. The record from Utah (Knowlton and Wood 1947: 94) is no doubt in error.
USA: CA
[congestus group]
This species is found along the Coast Ranges from Oregon (Lane County, CNC) to southern California (Fall 1901a: 44).
USA: CA, OR
This species is restricted to the Coast Ranges and adjacent areas from the Gulf Coast of Alaska (Lindroth 1966: 460) to northwestern California (Notman 1929b: 222; Will and Gill 2008: 123).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
As far as known, this species is found along the Coast Ranges in northern California.
USA: CA
[craterensis group]
This species is known from a few specimens collected in Klamath County, southern Oregon.
USA: OR
[ecarinatus group]
This species is found along the Rocky Mountains and their foothills in western Alberta, eastern British Columbia (Lindroth 1966: 466), northeastern Washington, northern Idaho (Hatch 1953: 109), and western Montana (Russell 1968: 58; Edwards 1975: 54; Will and Gill 2008: 123).
CAN: AB, BC USA: ID, MT, WA
[gracilior group]
This species is known only from the holotype.
USA: CA
This species is known only from the San Bernardino Mountains in southern California.
USA: CA
This species is known only from two specimens collected in Mendocino County, California.
USA: CA
This species seems to be confined to the mountains of southern California (Fall 1901a: 44). The records from “Oregon, ” “Nevada, ” and “Nebraska” (Schaupp 1882c: 41) are probably in error.
USA: CA
This species is known from a few localities in the southern parts of the Sierra Nevada. The record from Baja California (Horn 1894: 308) needs confirmation.
USA: CA
This species is known only from the lectotype collected in northwestern California.
USA: CA
This species is known from the Coast Ranges and Sierra Nevada in central California.
USA: CA
This species is known only from the holotype.
USA: CA
This species is known only from the lectotype.
USA: CA
This species is known only from the type locality in western California.
USA: CA
This species is known only from Placer (Casey 1913: 124) and Tuolumne Counties (Dajoz 2007: 17) in the Sierra Nevada.
USA: CA
Casey (1913) based the description of this species on two specimens, one from Lake Tahoe, the other from Merced Creek. The specimen from Merced Creek was later (Casey 1918: 335) considered a distinct species which Casey named Hypherpes mercedianus.
This species is known only from the lectotype collected in west-central California.
USA: CA
Besides the lectotype, I have seen one specimen, probably of this species, from Mono County, California (CNC). The species was also reported from Zion National Park, Utah, by Tanner (1928: 270).
USA: CA, NV [UT]
[herculaneus group]
This species ranges from northwestern Montana (Russell 1968: 58) to Vancouver Island (Lindroth 1966: 464), south to the Coast Ranges in southwestern Oregon (Niwa and Peck 2002: 787) and Tuolumne County in the Sierra Nevada (Will and Gill 2008: 123). The record from southeastern Colorado (Wickham 1902: 235) is likely in error.
CAN: BC (VCI) USA: CA, ID, MT, OR, WA
This species is known only from the Lake Tahoe area in the Sierra Nevada, California.
USA: CA
This species is known from the Cascade Range and Coast Ranges of western Oregon [see LaBonte 2006: Fig. 5].
USA: OR
This species occurs west of the Rocky Mountains from southern British Columbia, including Vancouver Island (Lindroth 1966: 463), to northern Oregon (Hatch 1953: 109, as Pterostichus brunneus).
CAN: BC (VCI) USA: OR, WA
Prior to Lindroth (1966), this species was reported in the literature under the name Pterostichus brunneus (Dejean, 1828).
This species is known only from the lectotype collected in northern California.
USA: CA
This species is known from “Oregon” (LeConte 1853a: 239) and a few localities in the Sierra Nevada (Fall 1901a: 45).
USA: CA, OR
This species occurs from the foothills of the Rocky Mountains in south-central Alberta to the Kootenay River drainage in southeastern British Columbia (Lindroth 1966: 463), south to the Sierra Nevada in California (Fall 1901a: 45; Papp 1978: 167), northern Arizona (Apache and Coconino Counties, CNC), and the Sangre de Cristo Mountains in New Mexico (Fall and Cockerell 1907: 158; Casey, 1913: 111, as Pterostichus zunianus). The record from Baja California (Horn 1894: 308) is suspect; that from “Nebraska” (Schaupp 1882c: 40) needs confirmation.
CAN: AB, BC USA: AZ, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY [NE]
This species is known from northern Idaho and adjacent eastern Washington (Hatch 1953: 109).
USA: ID, WA
This species is known only from Siskiyou County in northern California.
USA: CA
[illustris group]
As far as known, this species seems to be restricted to southwestern California.
USA: CA
This name has been listed as a junior synonym of Pterostichus congestus Ménétriés by LeConte (1858a: 28; 1873a: 304) but, as pointed out by Bousquet and Larochelle (1993: 18), it applies to a distinct species.
[inermis group]
This species is known so far only from southwestern California.
USA: CA
This very distinct species is still known only from the lectotype collected at Santa Barbara along the coast of California.
USA: CA
[isabellae group]
This species is known only from the type locality in southwestern California.
USA: CA
This species is known only from the original specimens collected in San Clemente Island, part of the Channel Islands, in the Pacific Ocean along the coast of southern California.
USA: CA (CHI)
According to Fall (1901b: 44), this species is “common and widely diffuse” in southern California.
USA: CA (CHI)
This species is known yet only from San Diego County in southwestern California.
USA: CA
This species is known only from the lectotype collected in southwestern California.
USA: CA
[lama group]
This large species is found from the Queen Charlotte Islands (Kavanaugh 1992: 70) south to southern California (Fall 1901a: 44, as Pterostichus ater; Will and Gill 2008: 124), including Washoe County in northwestern Nevada (La Rivers 1946: 102, as Feronia ater).
CAN: BC (QCI, VCI) USA: CA, NV, OR, WA
[lassulus group]
This species is known only from the lectotype collected in eastern California.
USA: CA
[menetriesii group]
This species is found along the Coast Ranges in central California (Marin, San Mateo, and San Francisco Counties, Will and Gill 2008: 125, CNC). It was also reported from Santa Rosa Island in the Pacific Ocean (Fall 1901a: 44).
USA: CA (CHI)
[morionides group]
This species is found along the northern (Siskiyou County, CNC) and central parts of the Sierra Nevada (Will and Gill 2008: 118, 125).
USA: CA
In analyses using molecular data sequences, this species was consistently positioned as the sister-group to {Pterostichus adoxus + Pterostichus tristis} which occur in eastern North America (Will and Gill 2008: 118).
[nigrocaeruleus group]
This species is known only along the coast in central Oregon [see Kavanaugh and LaBonte 2006: Fig. 14].
USA: OR
This species is found along the Pacific Coast from Vancouver Island to northern California (Lindroth 1966: 467).
CAN: BC (VCI) USA: CA, OR, WA
[restrictus group]
This species is yet known only from the lectotype.
USA: CO
This name has been listed in synonymy with Pterostichus restrictus (Casey) by Erwin et al. (1977: 4.36), based on Armin (1963: 220) unpublished thesis. However, I believe the name probably applies to a distinct species.
This species is found along the Rocky Mountains from southern Montana (Hatch 1933a: 7) to northern New Mexico (Snow 1885: 67; Fall and Cockerell 1907: 157).
USA: CO, MT, NM, WY
[setosus group]
This very distinct species is known only from southwestern Oregon (Hatch 1951: 116; Niwa and Peck 2002: 787).
USA: OR
[tarsalis group]
This species is known so far only from the Yosemite Valley in the Sierra Nevada, California.
USA: CA
This species is known for sure only from the Sierra Nevada, California. The record from “Oregon” (Schaupp 1882c: 40) needs confirmation.
USA: CA [OR]
[vicinus group]
This species ranges along the Pacific Coast from southern Alaska (Lindroth 1966: 462) to northern California (Fall 1901a: 45; Casey, 1913: 114, as Pterostichus humboldti).
CAN: BC (QCI, VCI) USA: AK, CA, OR, WA
This species is known only from the type locality in southwestern California.
USA: CA
As far as known, the range of this species extends along the Coast Ranges of California from Humboldt County (Casey 1913: 114, as Pterostichus cupidus) to Santa Clara County (LeConte 1851: 181, as Pterostichus simplex).
USA: CA
This species is known only from the original specimens collected at the type locality in the Sierra Nevada.
USA: CA
This species seems to be restricted to the Coast Ranges in California. Fall (1901b: 44) statement that the species is “common in the Sierras and throughout the region [i.e., southern California] to the west” needs confirmation.
USA: CA
About 115 species in the arctic, subarctic, and boreal regions of North America (23 species) and the Palaearctic Region (about 100 species). Several species are Holarctic.
Ball (1966a) revised the North American species which he arrayed in three groups. Since then, two new species have been described, one (Pterostichus haftorni) by Lindroth (1969a) which has been subsequently synonymized, the other one (Pterostichus woodi) by Ball and Currie (1997), and several species related to Pterostichus planus have been downgraded to subspecies of Pterostichus bryanti by Ball and Currie (1997). Species identifications are difficult in part because of the important intraspecific variation observed in most species.
Lorenz (2005: 275) listed Parahaptoderus Jeanne as a junior synonym of Cryobius. However, the taxon, which is composed of three European species of which one extends to Turkey, is probably not closely related to Cryobius (see Bousquet 1999: 187).
[brevicornis group]
This Holarctic subspecies ranges from the Kola Peninsula in northern European Russia eastwards to Newfoundland (Ball 1966a: 119); the species is represented by isolated populations in the Gaspé Peninsula, Maine, New Hampshire, Vermont, New York, Michigan (Ball 1966a: 119-121), northeastern Minnesota (Cook County, CNC), and northern Wyoming (Big Horn County, FFPC) [see Ball and Currie 1997: Fig. 4]. The record from northern Colorado (Armin 1963: 217) is likely in error. Fossil remnants, older than 33, 000 years B.P., have been unearthed in southwestern Ontario (Warner et al. 1988: 37); others from a Plio-Pleistocene sequence have been found in northwestern Greenland (Böcher 1995: 28).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, YT USA: AK, ME, MI, MN, NH, NY, VT, WY – Holarctic
The subspecies Pterostichus brevicornis yasudai Morita is endemic to the island of Hokkaidō, Japan.
This Holarctic species ranges from the Kuril and Commander Islands (Eremin 1998: 298) on the east coast of Asia to southern Yukon Territory, including Kodiak and Aleutian Islands, south to west-central British Columbia (Lemieux and Lindgren 2004: 562) [see Ball 1963: Fig. 3].
CAN: BC, YT USA: AK – Holarctic
Lindroth (1966: 524) stated that the taxonomic status of this taxon as a distinct species from Pterostichus brevicornis Kirby is questionable. Ball (1966a: 123) evoked the fact that the taxon may be regarded as a “parthenogenetic race” of Pterostichus brevicornis. Both authors concluded that this taxon is parthenogenetic since, as far as known, no males have been found to date that could be associated with it. This is also the case for the Asian specimens studied by Eremin (1998).
This species is found from the western shore of Hudson Bay in Nunavut westwards to the coast of the Bering Sea in Alaska, southwards to northeastern British Columbia and northwestern Alberta (UASM) [see Nielsen et al. 1987: Fig. 18b].
CAN: AB, BC, NT, NU, YT USA: AK
The range of this Holarctic species extends from eastern Siberia (Ball 1966a: 131) eastwards to east-central Yukon Territory (Dempster Highway Mi 35, CNC), south to the Alaska Peninsula [see Ball 1963: Fig. 3].
CAN: YT USA: AK – Holarctic
[pinguedineus group]
This species ranges from the Labrador coast and Baffin Island to central Alaska, south at least to central British Columbia (Summit Lake, CNC); isolated on the Adirondack Mountains of New York, the White Mountains of New Hampshire, and the Shickshock Mountains in Gaspé Peninsula [see Ball 1963: Fig. 8; Nielsen et al. 1987: Fig. 17d; Ball and Currie 1997: Fig. 2]. Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96).
CAN: BC, LB, MB, NT, NU, QC, YT USA: AK, ME, NH, NY
This species is known from two localities along the west coast of Alaska (Ball 1966a: 82).
USA: AK
This species ranges from northwestern Nunavut and central Northwest Territories westwards to northeastern Alaska (Ball 1962: 19). The record from McMurray in northern Alberta (Ball 1966a: 49), based upon a single specimen, has been rejected by Ball and Currie (1997: 476). Fossil remnants of this species, dated between about 16, 700 and 20, 530 years B.P., have been unearthed in southeastern and northeastern Iowa (Baker et al. 1986: 96; Schwert 1992: 77; Woodman et al. 1996: 17).
CAN: NT, NU, YT USA: AK
This subspecies is known only from the type locality in the arctic tundra and from the “Alpine interior Alaska” (Matthews 1974b: 1365).
USA: AK
This subspecies is known only from a few localities in the arctic from northwestern Northwest Territories (Ball 1966a: 66) to Prudhoe Bay in Alaska, including the Richardson Mountains in Yukon Territory (Ball and Currie 1997: 489).
CAN: NT, YT USA: AK
This subspecies is known for sure only from Kotzebue on the Seward Peninsula (Matthews 1974b: 1365) and the type locality in northeastern Alaska. The record from “Yukon Territory” (Ball and Currie 1997: 452) could not be confirmed.
USA: AK [YT]
This subspecies is known from a few localities between Yukon River and Fairbanks in the Yukon-Tanana Highlands in Alaska (Ball and Currie 1997: 486).
USA: AK
This subspecies is known only from the type locality located at the mouth of the Anderson River along the northwestern coast of Northwest Territories (Ball and Currie 1997: 486).
CAN: NT
This subspecies is known only from a few localities in central Alaska (Lindroth 1966: 515).
USA: AK
This species is found from the Hudson Bay coast in northern Ontario to northeastern British Columbia, north to central Northwest Territories [see Ball 1963: Fig. 9].
CAN: AB, BC, MB, NT, ON
This species is known only from eastern and central Alaska [see Ball 1963: Fig. 8].
USA: AK
The range of this species extends from the Hudson Bay area in Nunavut to Kuskokwim Bay on the western coast of Alaska, south to northeastern British Columbia [see Ball 1963: Fig. 9] and northern Alberta (Birch Mountains, Gerald J. Hilchie pers. comm. 2009, determination of George E. Ball). The records from northern Colorado (Wickham 1902: 236; Armin 1963: 218) are likely in error.
CAN: AB, BC, NT, NU, YT USA: AK
This species is known from the Dempster Highway in Yukon Territory (UASM) to the Seward Peninsula in western Alaska [see Ball 1963: Fig. 8].
CAN: YT USA: AK
This Holarctic species is known from the Kamchatka Peninsula, several islands in the Bering Sea, mainland Alaska, and the Ogilvie Mountains in western Yukon Territory [see Ball 1963: Fig. 4].
CAN: YT USA: AK – Holarctic
This Holarctic species ranges from the Yenisei River in Siberia eastwards to the Mackenzie River delta in western Northwest Territories; isolated on the top of some mountains in New England and on the Shickshock Mountains of the Gaspé Peninsula, Quebec [see Ball 1963: Fig. 5]. The record from Wyoming (Lavigne 1977: 47) is in error. Fossil remnants of this species, dated between about 12, 000 and 18, 100 years B.P., have been unearthed in central and southeastern Iowa (Schwert 1992: 77; Baker et al. 1986: 96) and northeastern Pennsylvania (Barnosky et al. 1988: 178); others, older than 33, 000 years B.P., have been found in southwestern Ontario (Warner et al. 1988: 37).
CAN: NT, QC, YT USA: AK, ME, NH, VT – Holarctic
Platysma poppiusianum Jacobson, 1907 (a replacement name for Pterostichus insulicola Poppius, 1906) was listed as a junior synonym of this species by Ball (1966a: 71), as a junior synonym of Pterostichus subgibbus (Motschulsky, 1860) by Kryzhanovskij et al. (1975: 137), and treated as a valid species by Eremin (1990) and Kryzhanovskijet al. (1995: 102).
This species is known only from a few localities in the Seward Peninsula, western Alaska [see Ball 1963: Fig. 6].
USA: AK
This species is found from eastern Siberia to the Dempster Highway in Yukon Territory (UASM), including several islands of the Bering Sea [see Ball 1963: Fig. 4].
CAN: YT USA: AK – Holarctic
Matthews (1974b: 1365) remarked that some specimens of this species have anomalous male genitalia. He interpreted this condition as either Pterostichus similis “is dimorphic in certain genital characters” or that it “includes two species” as currently defined.
This species is known from the Norton Sound Inlet in western Alaska to the Coronation Gulf in northern Nunavut (Ball 1966a: 28-29).
CAN: NT, NU, YT USA: AK
This species is found along the Rocky Mountains from southern Alberta to Utah and Colorado (Ball 1966a: 26).
CAN: AB USA: CO, MT, UT, WY
This Holarctic species is known in North America along the arctic coast from western Alaska to north-central Nunavut, and from Victoria and Banks Islands; in the Palaearctic Region the species is known only from the northern Bering Sea Coast (Ball 1966a: 47).
CAN: NT, NU, YT USA: AK – Holarctic
This species is known only from the Ogilvie Mountains in Yukon Territory.
CAN: YT
[ventricosus group]
This species ranges from northern Alaska to the western shores of the Hudson Bay in northern Manitoba and Southampton Island in Nunavut [see Ball 1963: Fig. 2; Nielsen et al. 1987: Fig. 18a]; isolated in eastern Ohio (Usis and MacLean 1998: 67). Fossil remnants of this species, dated between about 16, 700 and 21, 500 years B.P., have been unearthed in north-central Illinois (Garry et al. 1990: 394) and east-central Iowa (Baker et al. 1986: 96).
CAN: MB, NT, NU, YT USA: AK, OH
This species ranges from central Alaska to eastern Alberta, including western Northwest Territories, south to west-central Montana, northern Idaho, and northern Oregon [see Ball 1963: Fig. 2]. The records from northern Colorado (Packard 1877: 811; Wickham 1902: 236; Armin 1963: 218; Elias 1987: 632, as Pterostichus fatuus) need confirmation. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 28).
CAN: AB, BC (QCI), NT, YT USA: AK, ID, MT, OR, WA [CO]
This Holarctic species ranges from Novaya Zemlya in the Arctic Ocean off the coast of European Russia east to Franklin Bay on the coast of Northwest Territories, south in the Nearctic Region to central Northwest Territories and Umnak Island in the Aleutian Islands [see Ball 1963: Fig. 2]. Fossil remnants of this species, dated between about 16, 700 and 20, 530 years B.P., have been unearthed in northeastern and southeastern Iowa (Schwert 1992: 77; Baker et al. 1986: 96; Woodman et al. 1996: 17); others, older than 33, 000 years B.P., have been found in southwestern Ontario (Warner et al. 1988: 37).
CAN: NT, YT USA: AK – Holarctic
The subspecies Pterostichus ventricosus nechaevi Lafer and Kuznetsov is endemic to Sakhalin Island and Pterostichus ventricosus paludosus (Sahlberg) (junior synonym: Pterostichus tungusicus Poppius) to southeastern Siberia (Ball 1966a: 101).
Forty-five species in the temperate and subtropical regions of North America with one species (Pterostichus substriatus) extending into northern Mexico.
Freitag (1969) revised the species. Since the publication of his revision, a replacement name has been proposed for Cyclotrachelus obsoleta and two subspecies are raised to species level in this publication.
This genus, certainly one of the most characteristic pterostichine elements in North America, could be closely related to Molops Bonelli (Freitag 1969), whose species inhabit the mountains of Europe and Turkey, or to the Sterocorax complex (Sterocorax Ortuño, Paleocorax Ortuño, Iberopus Ortuño), a clade endemic to the Iberian Peninsula and currently included within the genus Pterostichus Bonelli (see Bousquet 1999: 198). Based on molecular sequence data, Will and Gill (2008: 113) found that the genus Pterostichus was monophyletic if Cyclotrachelus were included in the genus and recommended to treat Cyclotrachelus as a subgenus of Pterostichus.
Eighteen species, most restricted to the Coastal and Gulf Plains.
[approximatus group]
This species is restricted to a small area from “Pennsylvania” (LeConte 1846b: 354) to North Carolina [see Freitag 1969: Fig. 126]. The record from Georgia (Fattig 1949: 24) needs confirmation.
USA: DC, MD, NC, PA, VA [GA]
The range of this species extends from West Sister Island (Ohio) in the Western Basin of Lake Erie (Will et al. 1995: 62) to northeastern Illinois, south to southern Alabama and northern Georgia [see Freitag 1969: Fig. 126]. The record from Missouri (Summers 1873: 134) needs confirmation.
USA: AL, GA, IL, IN, KY, MI, MS, NC, OH, SC, TN, VA [MO]
This species is known from a small area in west-central and southern West Virginia (Roane and Raleigh Counties, CMNH), western Virginia, and North Carolina [see Freitag 1969: Fig. 126].
USA: NC, VA, WV
[faber group]
This species is found in southern Georgia and Florida including the Keys [see Freitag 1969: Fig. 129]. The record from northwestern South Carolina (Kirk 1970: 11) is probably in error.
USA: FL, GA
This species is known only from a few specimens collected in “North Carolina, ” northern South Carolina, and “Georgia” [see Freitag 1969: Fig. 129].
USA: GA, NC, SC
This species is known only from several specimens collected at the type locality in southern Alabama [see Freitag 1969: Fig. 129].
USA: AL
[laevipennis group]
This species ranges from southern Georgia to southwestern Florida [see Freitag 1969: Fig. 125]. The record from Alabama (Löding 1945: 16) needs confirmation.
USA: FL, GA [AL]
This species is known from a few localities in the western parts of the Florida Peninsula [see Freitag 1969: Fig. 125].
USA: FL
This species inhabits the Gulf Plain and southern Piedmont Plateau from South Carolina to the Florida Panhandle and “Louisiana” (LeConte 1853a: 231, as Evarthrus acutus) [see Freitag 1969: Fig. 125].
USA: AL, FL, GA, LA, MS, SC
[ovulum group]
This species is known only from Mobile County in southwestern Alabama (Freitag 1969: 118). The record from “Mississippi” (Bousquet and Larochelle 1993: 186) was based on a misidentified specimen (Drew A. Hildebrandt pers. comm. 2007).
USA: AL
This species is known only from southern Alabama, southern Mississippi (Drew A. Hildebrandt pers. comm. 2007), and east-central (West Feliciana Parish, LSAM) and southern Louisiana [see Freitag 1969: Fig. 128].
USA: AL, LA, MS
This species is known from southern Georgia and northern Florida [see Freitag 1969: Fig. 128].
USA: FL, GA
This species is known from a few specimens collected in eastern Texas [see Freitag 1969: Fig. 128].
USA: TX
This species is found in a small area including eastern Tennessee, southwestern North Carolina, northwestern South Carolina, and northeastern Georgia [see Freitag 1969: Fig. 128].
USA: GA, NC, SC, TN
[spoliatus group]
This species is found along the Coastal Plain from South Carolina to central Mississippi [see Freitag 1969: Fig. 127].
USA: AL, FL, GA, MS, SC
This species ranges from southwestern Pennsylvania to southwestern Illinois (Union County, CNC), south to northern Alabama and northwestern Georgia [see Freitag 1969: Fig. 127].
USA: AL, GA, IL, KY, OH, PA, TN, WV
This species is found in a small area from the District of Columbia south to southern South Carolina [see Freitag 1969: Fig. 127] including northeastern Georgia (LeConte 1853a: 230). The records from “New York” (Leng and Beutenmüller 1893: 138, as Pterostichus rotundatus) and northern Alabama (Löding 1945: 15) need confirmation.
USA: DC, GA, NC, SC, VA [AL, NY]
This species is restricted to the Coastal Plain and southern Piedmont from South Carolina (Ciegler 2000: 71) to northeastern Alabama, south to the Florida Panhandle [see Freitag 1969: Fig. 127]. The record from North Carolina (Brimley 1938: 119) needs confirmation.
USA: AL, FL, GA, SC [NC]
Twenty-seven species are known.
[blatchleyi group]
This species ranges from southern Georgia to southwestern Florida [see Freitag 1969: Fig. 131].
USA: FL, GA
This species is endemic to a small area in the eastern part of central Florida [see Freitag 1969: Fig. 131].
USA: FL
[gigas group]
This species is known from two localities in southeastern Texas [see Freitag 1969: Fig. 136].
USA: TX
This species is known from southeastern Oklahoma south to Comal County, Texas [see Freitag 1969: Fig. 136]. The record from “Arkansas” (Freitag 1969: 167) derives from the locality originally cited by Say (1823b: 145) which possibly refer to the Arkansas Territory. Prior to 1825, this territory included, besides Arkansas, all of the present state of Oklahoma. It could also refer to the Arkansas River which flows through Colorado, southern Kansas, northeastern Oklahoma, and Arkansas. The record from southeastern Kansas (Knaus 1907: 233) needs confirmation.
USA: OK, TX [KS]
This species is known only from eastern Texas [see Freitag 1969: Fig. 136].
USA: TX
[gravesi group]
This species is known from the holotype and four specimens collected in Hinds County in southern Mississippi (Drew A. Hildebrandt pers. comm. 2009).
USA: MS
[hypherpiformis group]
This species is known only from a few specimens collected in western Alabama and northeastern Mississippi [see Freitag 1969: Fig. 132].
USA: AL, MS
[incisus group]
This species is found from southwestern Pennsylvania to southeastern South Dakota, south to southern Colorado (Miller and Peairs 2008: 34; LeConte 1879d: 500; Wickham 1902: 236), Oklahoma, and central Arkansas [see Freitag 1969: Fig. 130]. The records from South Carolina (Kirk 1970: 11; Ciegler 2000: 70) are probably in error; that from “Kentucky” (Bousquet and Larochelle 1993: 188) needs confirmation.
USA: AR, CO, IA, IL, IN, KS, MO, NE, OH, OK, PA, SD, VA [KY]
This species is known from a few localities in eastern Oklahoma, southern Arkansas, and northwestern Louisiana (Natchitoches Parish, LSAM) [see Freitag 1969: Fig. 130].
USA: AR, LA, OK
[seximpressus group]
This species is restricted to the Gulf Coastal Plain from southwestern Alabama to eastern Texas, north to southern Arkansas [see Freitag 1969: Fig. 132]. Freitag (1969: 142) suggested that the specimens labeled from Clay County in northeastern Kansas and Lawrence County in northeastern Arkansas are probably mislabeled.
USA: AL, AR, LA, MS, TX [KS]
This species is found only in eastern Texas (Freitag 1969: 143, Fig. 132).
USA: TX
This species is found along the Gulf Coastal Plain from eastern Texas and southern Arkansas to western Mississippi [see Freitag 1969: Fig. 132]. The record from northwestern South Carolina (Kirk 1970: 11) is in error.
USA: AR, LA, MS, TX
This species is found west of the Appalachians Mountains from southwestern Pennsylvania to eastern Nebraska, north to southern Wisconsin and southeastern Minnesota, south to east-central Texas and southern Mississippi (Walthall County, Paul K. Lago pers. comm. 2009) [see Freitag 1969: Fig. 132].
USA: AR, IA, IL, IN, KS, MI, MN, MO, MS, NE, OH, OK, PA, TX, WI
[sigillatus group]
This species ranges from southwestern Pennsylvania to the Mississippi River in western Illinois, south to southeastern Louisiana and southern Alabama [see Freitag 1969: Fig. 131]. The record from South Carolina (Kirk 1970: 11; Ciegler 2000: 70) is probably in error.
USA: AL, AR, IL, IN, KY, LA, MO, MS, OH, PA, TN, WV
This species is found along the Appalachians, Piedmont Plateau, and Coastal Plain from Massachusetts to the Florida Panhandle and eastern Alabama [see Freitag 1969: Fig. 131]. The records from southeastern Louisiana (Summers 1874a: 80, as Evarthrus americanus), Missouri (Summers 1873: 134, as Evarthrus orbatus), “Iowa” (Wickham 1911b: 6; King 1914: 321; Jaques and Redlinger 1946: 297, as Evarthrus orbatus), and Ohio (Dury 1902: 113; Walker 1957: 270, as Evarthrus americanus) are in error.
USA: AL, DC, DE, FL, GA, MA, MD, NC, NJ, NY, PA, SC, TN, VA, WV
This species is known from a few localities in southern Alabama and southern Mississippi [see Freitag 1969: Fig. 131].
USA: AL, MS
[sodalis group]
This species ranges from northwestern Wisconsin to northeastern South Dakota, south to central Missouri [see Freitag 1969: Fig. 134]. The record from “North Dakota” (Bousquet and Larochelle 1993: 187) needs confirmation.
USA: IA, IL, MN, MO, SD, WI [ND]
The range of this species extends from southern Pennsylvania and New Jersey to central Virginia [see Freitag 1969: Fig. 133]. The records from Ohio (Everly 1927: 155; Everly 1938: 141), “Michigan” (Leng 1920: 57), Indiana (Blatchley 1910: 101), North Carolina (Brimley 1938: 199), and Georgia (Fattig 1949: 22) are probably in error.
USA: DC, MD, NJ, PA, VA, WV
This species is known from a few localities in southeastern Minnesota, Iowa, and eastern South Dakota [see Freitag 1969: Fig. 134].
USA: IA, MN, SD
This species is restricted to Tennessee and northern Alabama [see Freitag 1969: Fig. 133].
USA: AL, TN
This form has been treated as a subspecies of Cyclotrachelus sodalis (LeConte) by Freitag (1969) but I believe it should be regarded as a distinct species based on the character states listed by Freitag (1969: 148).
This species is known only from Arkansas [see Freitag 1969: Fig. 133].
USA: AR
This subspecies is found west of the Mississippi River from southeastern Minnesota (Gandhi et al. 2005: 926), western Iowa and eastern Nebraska south to southern Kansas and central Missouri [see Freitag 1969: Fig. 133]. At least one specimen simply labeled from Arkansas is known (Freitag 1969: 150). The records from southern Wisconsin (Rauterberg 1885: 15) and northern Colorado (LeConte 1853a: 229, as Feronia corax) need confirmation; that from southeastern Louisiana (Summers 1874a: 80) is probably in error.
USA: IA, KS, MN, MO, NE [AR, CO, WI]
This subspecies ranges from southwestern Vermont (Bell and Nielsen 1978: 8) to northeastern Minnesota, including southernmost Ontario, south to northeastern Mississippi and eastern Tennessee [see Freitag 1969: Fig. 133]. The record from northwestern Missouri (Freitag 1969: 149) probably refers to Cyclotrachelus sodalis colossus.
CAN: ON USA: IA, IL, IN, KY, MI, MN, MS, NJ, NY, OH, PA, TN, VA, VT, WI
[substriatus group]
This species ranges from southeastern South Dakota and western Iowa south to central Texas, west to central Arizona [see Freitag 1969: Fig. 134].
USA: AZ, CO, IA, KS, NE, NM, OK, SD, TX
This species ranges from southern Minnesota to southern Wyoming, south to the state of Durango and southeastern Texas [see Freitag 1969: Fig. 134]. The record from Missouri (Summers 1873: 134) needs confirmation.
USA: AZ, CO, KS, MN, NE, NM, OK, SD, TX, WY [MO] – Mexico
[torvus group]
This species is known from Texas, as far south as DeWitt County and west to El Paso County [see Freitag 1969: Fig. 135]. The record from “Oklahoma” (Bousquet and Larochelle 1993: 189) needs confirmation.
USA: TX [OK]
This taxon has been regarded as a subspecies of Cyclotrachelus torvus (LeConte) by Freitag (1969) but I believe it should be treated as a distinct species.
This species is known from southern Oklahoma (Hatch and Ortenburger 1930: 11, as Eumolops ampla) and Texas, as far south as Comal County and west to El Paso County [see Freitag 1969: Fig. 135].
USA: OK, TX
This species inhabits the Great Plains from southwestern Minnesota (Gandhi et al. 2005: 926) to western South Dakota, south to southern New Mexico and southern Arkansas [see Freitag 1969: Fig. 135]. The record from “Arizona” (Bousquet and Larochelle 1993: 189) is probably in error; one specimen labeled from Hidalgo County in southeastern Texas is probably mislabeled (Freitag 1969: 163).
USA: AR, CO, IA, KS, MO, MN, NE, NM, OK, SD, WY
Eighteen species in Europe, with one species extending into Turkey, arrayed in two subgenera: Abax s.str. (16 species) and Abacopercus Ganglbauer (two species). One species is adventive in eastern North America.
Sixteen species in Europe and Turkey, one of them adventive in the Nearctic Region.
The species in North America was covered in Lindroth’s (1969a: 1116) monograph.
This European species is adventive in North America where it is known from Sydney, Nova Scotia (Brown 1967: 87) and western Newfoundland (David W. Langor pers. comm. 1990). The first inventoried specimen collected on this continent was found in 1965 in Nova Scotia.
CAN: NF, NS – Adventive
About 700 species arrayed in two subtribes: Zabrina (107 species of Zabrus in Europe and western Asia, the vast majority restricted to the Mediterranean region) and Amarina. More than 98% of the species inhabit the Northern Hemisphere.
About 595 species placed in one genus.
About 595 species (Hieke 2007) in the Nearctic (105 species, including ten adventive species), Neotropical (13 species in Middle America, only two of them endemic), Oriental (five species), Palaearctic (about 505 species, 16 of them Holarctic), and Afrotropical (about 12 species, nine of them endemic) Regions. These species are arrayed in 47 subgenera.
Almost all new state and province records listed here are based on specimens determined by Fritz Hieke.
Eighty-five species (Hieke 2007) in the arctic, subarctic, boreal, and temperate regions of North America (15 species of which three extend into Mexico), Asia (about 60 species), and Europe (13 species, all but possibly one shared with Asia). Six species are Holarctic and one is adventive in eastern North America.
There is no modern taxonomic revision of the species and such study would be useful. Lindroth (1968, as aulica group) covered ten of the 15 species found in North America.
This Holarctic species is found from Great Britain to the Pacific Coast of Siberia (Hieke 2003a: 558), and from the west coast of Alaska, including the Saint Lawrence Island, to northwestern Newfoundland, south to the James Bay area and to northern New Mexico along the Rocky Mountains [see Lindroth 1963b: Fig. 58]; isolated on the Shickshock Mountains in Gaspé Peninsula (Larochelle 1975: 42) and some mountains in New England (Lindroth 1966: 676). The record from “New York” (Notman 1928: 226) needs confirmation. According to Lindroth (1968: 676), this is the carabid occurring farthest north in North America. Fossil remnants of this species, dated between about 14, 000 and 21, 500 years B.P., have been unearthed in southeastern and central Iowa (Baker et al. 1986: 96; Schwert 1992: 78) and north-central Illinois (Garry et al. 1990: 394); others from interglacial deposits have been found in northwestern Greenland (Bennike 2000: 31).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, YT USA: AK, CO, ME, NH, NM, UT, VT, WY [NY] – Holarctic
This European species is adventive in North America where it is known from Newfoundland (Larson and Langor 1982: 593) and Saint Pierre and Miquelon (Lindroth 1955a: 95) to the Saint Lawrence Valley in Quebec (Larochelle 1975: 42), south to Boston Harbor, Massachusetts (Davidson et al. 2011: 512). The first inventoried specimen collected on this continent was found in Cape Breton Island, Nova Scotia in 1929 (Fall 1934: 171). The record from the Similkameen Valley in British Columbia (Smith et al. 2004: 96) was based on a misidentified specimen of Amara carinata LeConte (personal observation).
FRA: PM CAN: NB, NF, NS (CBI), PE, QC USA: MA, ME, NH – Adventive
This western species occurs from southern Saskatchewan (MCZ) to south-central British Columbia (Lindroth 1968: 666), south to east-central California (Inyo County, CAS) and northwestern New Mexico (San Juan County, CNC).
CAN: AB, BC, SK USA: AZ, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY
This species ranges from eastern Siberia to the Hudson Bay coast in northern Manitoba (Lindroth 1968: 677).
CAN: MB, NT, NU, YT USA: AK – Holarctic
This species ranges from the Gaspé Peninsula (Larochelle 1975: 43) to south-central British Columbia (Lindroth 1968: 669), south to the Sierra Nevada in California (Dajoz 2007: 16), central New Mexico (Bernalillo County, CMNH; Lindroth 1968: 668), the Texas Panhandle (Michels et al. 2010: 743), northwestern Arkansas (Carroll County, MCZ), and northeastern Ohio (Lake County, CMNH). The record from “West Virginia” (Bousquet and Larochelle 1993: 190) is in error; that from “Yukon Territory” (Ball and Currie 1997: 452) could not be confirmed.
CAN: AB, BC, MB, ON, QC, SK USA: AR, AZ, CA, CO, IA, ID, IL, IN, KS, MA, MN, MO, MT, ND, NE, NH, NM, OH, OR, SD, TX, UT, VT, WI, WY [YT]
This species is found in Kazakhstan, Mongolia, northern China, and across Siberia (Hieke 2003a: 559). In North America, it is known only from the Anderson River Delta area in northwestern Northwest Territories (Hieke 1994: 306).
CAN: NT – Holarctic
The North American specimens differ from the Siberian ones in structural details and, according to Hieke (1994: 307), it is possible that they constitute a distinct subspecies.
This species ranges from Baja California Norte (CAS), southeastern California (Inyo and San Bernardino Counties, CAS, CMNH), and southern Nevada (Clark County, MCZ) east to northwestern Oklahoma (Texas County, CMNH), north to northern Colorado and “Utah” (Hayward 1908: 25, as Amara bowditchi), south at least to the state of México (Bates 1882a: 76).
USA: AZ, CA, CO, NM, NV, OK, TX, UT – Mexico
This Holarctic species ranges from northern Finland to the Pacific Coast, south to Mongolia and northeastern China (Hieke 2003a: 560) and from Alaska (Lindroth 1968: 679) to Newfoundland (Lindroth 1955a: 99), south to the upper peninsula of Michigan (Lindroth 1968: 678). The record from the White Mountains in New Hampshire (Wickham 1896b: 37) needs confirmation; those from “Vermont” (Hamilton 1894a: 10) and Colorado (Snow 1877: 17; Wickham 1902: 236; Armin 1963: 208, as Amara elongatus) are probably in error.
CAN: AB, BC, LB, MB, NF, NT, ON, QC, SK, YT USA: AK, MI [NH] – Holarctic
The range of this species extends from “Washington” (Hayward 1908: 23) to the Baja California Peninsula (Horn 1894: 309) and Arizona (Griffith 1900: 565; Hayward 1908: 23). The records from north-central Utah (Horn 1894: 309), “Colorado” (Wickham 1902: 236), and New Mexico (Fall and Cockerell 1907: 159) need confirmation.
USA: AZ, CA, OR, WA [CO, NM, UT] – Mexico
This Holarctic species is known from northeastern Siberia and the Anderson River Delta in northwestern Northwest Territories (Hieke 1994: 311).
CAN: NT – Holarctic
This species occurs from Nova Scotia (Larochelle and Larivière 1990a: 30, 34) to southwestern British Columbia, north to the coast of Nunavut and northern Alaska (Lindroth 1968: 671), south to northwestern Washington (Hatch 1953: 121), south-central Utah (Wayne County, CNC), southwestern Colorado (La Plata County, CNC), the Black Hills in southwestern South Dakota (Kirk and Balsbaugh 1975: 25), and “Michigan” (Garry A. Dunn pers. comm. 1986). The record from “Nebraska” (Bousquet and Larochelle 1993: 191) needs confirmation.
CAN: AB, BC, MB, NB, NS, NT, NU, ON, SK, YT USA: AK, CO, ID, MI, MN, MT, ND, SD, UT, WA, WI, WY [NE]
This species ranges from Nova Scotia (Lindroth 1968: 678) to “Wyoming” (Hayward 1908: 34), south to northeastern Colorado (Sedgwick County, Ken Karns pers. comm. 2009), northeastern Texas (Dallas County, MCZ), southwestern Alabama (Mobile County, USNM), and northwestern South Carolina (Ciegler 2000: 75). An old specimen simply labeled from “New Mexico” (MCZ) is known. The record from western Montana (Hatch 1933a: 8) needs confirmation.
CAN: NS, ON, QC USA: AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, MI, MN, MO, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV, WY [MT, NM]
This species ranges from southern Arizona (Cochise County, CNC) to western Texas (Dajoz 2007: 23; Brewster and Jeff Davis Counties, CNC, MCZ), south to the Federal District and central Veracruz in Mexico (Bates 1882a: 76).
USA: AZ, NM, TX – Mexico
The range of this species extends from southern Saskatchewan (CNC) to the Okanagan Valley in south-central British Columbia (Lindroth 1968: 668), south to northern Arizona (Coconino County, CMNH) and central New Mexico (Torrance County, CMNH; Milford et al. 2000: 21).
CAN: AB, BC, SK USA: AZ, CO, MT, NM, NV, WY
This circumpolar species is found from northern Finland to the Far East (Hieke 2003a: 561) and from the west coast of Alaska (Lindroth 1968: 673) to Newfoundland (Lindroth 1955a: 96-97), south to Nova Scotia (Lindroth 1968: 673), the Magdalen Islands, Gaspé Peninsula (Larochelle 1975: 48), southern Minnesota (Gandhi et al. 2005: 929), northern Colorado (LeConte, 1878a: 450, as Amara cylindrica; Hayward 1908: 28, as Amara rufimana; Armin 1963: 209, as Amara melanogastrica) along the Rocky Mountains, and southeastern British Columbia (Lindroth 1968: 673); isolated on Mount Washington in New Hampshire (CNC). The records from Prince Edward Island (Bousquet and Larochelle 1993: 192, see Majka et al. 2008: 131), “Wisconsin” (Wickham 1896b: 36, as Amara rufimana), northwestern Iowa (Wickham 1911b: 6, as Amara rufimana), and “South Dakota” (Hayward 1908: 28, as Amara rufimana) are probably in error; those from “New Mexico” (Hamilton 1894a: 10, as Amara eschscholtzii) and Seboomook in Maine (Dearborn and Donahue 1993: 6) need confirmation. Fossil remnants of this species, dated between about 16, 700 and 18, 100 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS, NT, ON, QC, SK, YT USA: AK, CO, MN, MT, NH, WY [ME, NM] – Holarctic
Thirty-five species (Hieke 2007) in the arctic, subarctic, boreal, and temperate regions of North America (12 species, of which two are adventive), Asia (26 species), and Europe (seven species, all shared with Asia). One species (Amara glacialis) is Holarctic.
There is no modern taxonomic revision of the species. Lindroth (1968, as apricaria and insignis groups) covered all but two (Amara neomexicana and Amara lindrothi) of the North American species.
[fulva group]
This Palaearctic species is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 100-101) and southern Labrador (Lindroth 1954d: 368) to the Kenai Peninsula in Alaska (Derek S. Sikes pers. comm. 2008), south to northern California (Trinity and Lassen Counties, CAS), west-central Nevada (Bechtel et al. 1983: 474), southern Colorado (Huerfano and Pueblo Counties, CMNH), Kansas (Trego County, CNC), and Virginia (Hoffman et al. 2006: 23; Clarke County, USNM). The first inventoried specimen collected on this continent was found in Quebec prior to 1865 (Couper 1865: 60, as Amara pygmea). Several records (e.g., AL, AR, GA, NT, SC, YT) listed in Bousquet and Larochelle (1993: 192) are in error or need confirmation.
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, CA, CO, CT, DC, DE, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MT, ND, NE, NH, NJ, NV, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WY – Adventive
This species ranges from Newfoundland (Lindroth 1955a: 100) to southeastern British Columbia (Lindroth 1968: 690), south to southern Colorado (Wickham 1902: 236; Armin 1963: 200), southeastern Kansas (Knaus 1907: 233), Missouri (Summers 1873: 145), and southwestern North Carolina (Macon County, MCZ). The record from southeastern Louisiana (Summers 1874a: 80) is likely in error.
FRA: PM CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CO, CT, DC, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WI
This species is known from a few localities in Yukon Territory and the Anderson River delta in northern Northwest Territories (Lindroth 1968: 687).
CAN: NT, YT
The range of this species extends from southern New Hampshire (Sullivan County, MCZ) to eastern South Dakota (Kirk and Balsbaugh 1975: 26; French et al. 2004: 557), south to northeastern Texas (Dallas County, MCZ), northeastern Georgia (Fattig 1949: 28; Clayton County, USNM), and northwestern South Carolina (Ciegler 2000: 74). The record from “Florida” (Bousquet and Larochelle 1993: 192) is probably in error.
CAN: ON USA: AL, CT, DC, GA, IA, IL, IN, KS, MA, MD, MI, MN, MO, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV
This European species is adventive in North America where it is known only in eastern Canada from Newfoundland (Lindroth 1955a: 101) to the Gaspé Peninsula (Larochelle 1975: 44) and the north shore of the Saint Lawrence in Quebec (Brown 1932b: 200), including southern Labrador (Lindroth 1955a: 101). The first inventoried specimen collected on this continent was found in Newfoundland in 1905 (Lindroth 1955a: 101).
CAN: LB, NB, NF, NS (CBI), PE, QC – Adventive
This species is found from the Yenisei River in western Siberia to the coast of Labrador [see Alfimov and Berman 2009: Fig. 1].
CAN: BC, LB, MB, NT, NU, ON, QC, SK, YT USA: AK – Holarctic
The range of this species extends from western Newfoundland (Lindroth 1955a: 102) to Vancouver Island (Lindroth 1968: 682), south to northwestern California (Humboldt County, USNM; Hayward 1908: 44), Arizona (Casey, 1918: 240, as Bradytus humphreysi; Chen et al. 2006: 171; Coconino County, USNM), New Mexico (Fall and Cockerell 1907: 158; Casey, 1918: 241, as Bradytus deceptus; Taos County, CMNH), “Texas” (Lindroth 1955a: 102), and northern Virginia (Page County, USNM).
CAN: AB, BC (VCI), MB, NB, NF, NS, ON, PE, QC, SK USA: AR, AZ, CA, CO, CT, DC, IA, ID, IL, IN, KS, KY, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NM, NY, OH, OR, PA, SD, TX, UT, VA, VT, WA, WI, WY
This species is known from a few localities in Labrador (CNC), northern Manitoba (CNC), northeastern Alberta (Andrew Lake, UASM), and Yukon Territory as far north as Swim Lakes (CNC), south to east-central Nevada (White Pine County, CMNH) and northern New Mexico (Hieke 1990: 234).
CAN: AB, LB, MB, YT USA: CO, NM, NV, UT, WY
This species is known from Arizona (Coconino National Forest, CNC), northern New Mexico (Casey 1924: 48; Stehr 1949: 207, as Bradytus vegasensis), and western Texas (Stehr 1949: 207, as Bradytus novellus).
USA: AZ, NM, TX
This species inhabits the Laurentian Highlands from the north shore of the Saint Lawrence Gulf (Larochelle 1975: 48) to the Great Slave Lake in the Northwest Territories (Lindroth 1968: 684), south to central Washington (Kittitas County, CMNH), southwestern Alberta (Indian Creek at 49º48’N - 114º07’W, CNC), northeastern Minnesota, and the upper peninsula of Michigan (Lindroth 1968: 684). The record from South Dakota (Kirk and Balsbaugh 1975: 26) was based on a small specimen of Amara latior in MCZ; those from Labrador (Larochelle 1975: 48) and Yukon Territory (Lindroth 1968: 684), and probably also from Colorado (Wickham 1902: 237), refer to Amara lindrothi (CNC). The records from Ohio (Everly 1927: 155), New Jersey (Smith 1910: 206), and “New York” (Leng and Beutenmüller 1893: 139; Wickham 1896b: 38) are likely in error; that from northern Wisconsin along Lake Superior (Wickham 1896c: 133, as Amara septentrionalis) needs confirmation.
CAN: AB, MB, NT, ON, QC USA: MI, MN, WA [WI]
[insignis group]
This species is found along the Pacific Coast from central California (Lindroth 1968: 692) south to Guadalupe Island off the coast of Baja California (Casey 1918: 295, as Amara guadalupensis). The record from “Oregon [Territory]” (LeConte 1857c: 9) is probably in error.
USA: CA (CHI) – Mexico
This species is known from “all the islands” (Fall 1901a: 45) off the coast of southern California.
USA: CA (CHI)
One species in southern North America.
The species was redescribed in Hieke’s (1978: 290-292) revision of the subgenus Percosia.
This species is known from McLennan (Horn 1892b: 27) and Dallas (Hieke 1978: 292) Counties in Texas and from southwestern Oklahoma (Kondratieff et al. 2005: 171).
USA: OK, TX
Six species in the boreal and temperate regions of North America (one species), Asia (two species), Europe (three species, two shared with Asia), and northern Africa (two endemic species).
Hieke (1978) revised the species of this subgenus.
This species ranges from Newfoundland (Lindroth 1955a: 102) to southern Alaska (CNC), south to central California (Merced County, CAS), northern Arizona (Chen et al. 2006: 171; Apache and Coconino Counties, CMNH, CNC, UASM), northeastern Texas (Hieke 1978: 289), and northern Georgia (Fattig 1949: 28).
CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AR, AZ, CA, CO, CT, DC, GA, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY
Thirty-two species (Hieke 2007) in the arctic (marginal), subarctic, boreal, and temperate regions of North America (11 species, two extending into northern Mexico), Asia (17 species), and Europe (ten species, many shared with Asia and one extending into northern Africa). One species (Amara hicksi) is Holarctic.
Hieke (2001) revised the species. Two Asian species have been described subsequently.
This species ranges from northern Oregon to southern California and Sonora, east to western Texas and Chihuahua in Mexico (Hieke 2001: 128). The record from southwestern Utah (Tanner 1928: 270) needs confirmation.
USA: AZ, CA, NM, OR, TX [UT] – Mexico
This species is known from two localities, Manhattan in northeastern Kansas and Gatesville in central Texas (Hieke 2001: 136).
USA: KS, TX
This species is found from western Maine (Oxford County, Ross T. Bell pers. comm. 2008) to southeastern Wyoming (Hieke 2001: 116), including southernmost Ontario (Lindroth 1968: 700), south to the Taos Mountains in northern New Mexico (LeConte 1876: 299; McKinley County, USNM), “Texas” (Horn 1892b: 32), southwestern Alabama (Hieke 2001: 116), northern Georgia (Fattig 1949: 29), and central South Carolina (Ciegler 2000: 74).
CAN: ON USA: AL, AR, CO, CT, DC, GA, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, NC, NE, NH, NJ, NM, NY, OH, PA, RI, SC, TX, VT, WI, WY
This western species ranges from southern Manitoba to southern British Columbia, south to southeastern California (San Bernardino County, David H. Kavanaugh pers. comm. 2008), southern Utah (Garfield County, CNC), south-central New Mexico, and northwestern Nebraska (Hieke 2001: 118).
CAN: AB, BC, MB, SK USA: CA, CO, ID, MT, NE, NM, NV, OR, SD, UT, WA, WY
This species ranges from Newfoundland to Alaska including Kodiak Island; it is also known from the Lake Superior region in northern Michigan (Hieke 2001: 113) and from northeastern New York (Essex County, CNC).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, QC, YT USA: AK, MI, NY
This form, listed as a synonym of Amara discors Kirby by Lindroth (1968: 697), was regarded as a valid species by Hieke (1990: 226).
This species is known from a few localities in eastern Washington, northern Oregon, and west-central Idaho (Hieke 2001: 119).
USA: ID, OR, WA
This Holarctic species is found from western Northwest Territories to Alaska and from Kamtschatka to northern European Russia, south to Mongolia and northern China (Hieke 2001: 61).
CAN: NT USA: AK – Holarctic
This species ranges from northern California to north-central Colorado, south to southwestern New Mexico and southern Arizona (Hieke 2001: 126-127).
USA: AZ, CA, CO, NM, NV, UT
This form was considered a synonym of Amara discors Kirby by Lindroth (1968: 697) but treated as a valid species by Hieke (2001: 124).
This species ranges from the Kodiak Island and Kenai Peninsula in Alaska south to the Sierra Nevada in east-central California, northern Arizona, and northern New Mexico along the Rocky Mountains (Hieke 2001: 122-124).
CAN: AB, BC USA: AK, AZ, CA, CO, ID, MT, NM, NV, OR, UT, WA, WY
This form was regarded as a synonym of Amara discors Kirby by Lindroth (1968: 697) but treated as a valid species by Hieke (1993: 128).
This subspecies ranges from northern California to northwestern Oklahoma (Cimarron County, CMNH), north to northern Colorado, south to Durango in Mexico (Hieke 2001: 135-136), including western Texas (Jeff Davis County, CMNH), and south-central California (Hieke 2001: 135).
USA: AZ, CA, CO, NM, OK, TX, UT – Mexico
Lindroth (1968: 697) listed Amara obsolescens Casey as a junior synonym of Amara discors Kirby but Hieke (1993: 132) considered it a synonym of Amara nupera Horn.
This subspecies ranges from northern Idaho and eastern Washington south at least to Monterey County in western California (Hieke 2001: 134).
USA: CA, ID, OR, WA
This species is known from a few localities in Northwest Territories, Yukon Territory (CNC), Alaska, and from Manning Provincial Park in southern British Columbia (Hieke 2001: 110).
CAN: BC, NT, YT USA: AK
Twenty species (Hieke 2007) in northern North America (one Holarctic species) and eastern Asia.
Hieke (1999a) revised the species of this subgenus. Subsequently, ten new Chinese species were described.
This Holarctic species is found in Siberia east of the Lena River and in North America from Alaska to the Hudson Bay coast in Manitoba and Nunavut (Hieke 1999a: 347).
CAN: MB, NU, YT USA: AK – Holarctic
Forty species (Hieke 2007) in North America (12 species), Middle America (two species, one of them endemic, Amara chihuahuae Casey), Asia (22 species), and Europe and northern Africa (14 species, several shared with Asia). One species found in the Nearctic Region is adventive (Amara bifrons), another one is Holarctic (Amara brunnea).
There is no modern revision of the species of this subgenus and such work is much needed.
This European species is adventive in North America where it is known from Newfoundland (Lindroth 1955a: 105) to western Quebec (Larochelle 1975: 43), south to Massachusetts (Davidson et al. 2011: 513). The first inventoried specimen collected on this continent was found in Cape Breton Island in 1929 (Lindroth 1963a: Fig. 39).
FRA: PM CAN: LB, NB, NF, NS (CBI), PE, QC USA: MA, ME, NH – Adventive
This Holarctic species is found from northern Manitoba (CNC) to the Kenai Peninsula in Alaska (Lindroth 1968: 706), and in the Palaearctic Region from the Far East to Norway, south to Spain, Italy, Kazakhstan, and Mongolia (Hieke 2003a: 556). The records from British Columbia (Hamilton 1894a: 11; Jarrett and Scudder 2001: 379) need confirmation; those from “Washington, ” “Colorado” (Hamilton 1894a: 11; Wickham 1902: 238), and Idaho (Horning and Barr 1970: 24, as Amara amplicollis) are in error.
CAN: AB, MB, NT, YT USA: AK [BC] – Holarctic
This subspecies ranges from Vancouver Island (Lindroth 1968: 694) to eastern South Dakota (Brookings County, USNM), south to Costa Rica (Hieke 1990: 221) and the Baja California Peninsula (Horn 1894: 309).
CAN: BC (VCI) USA: AZ, CA (CHI), CO, ID, MT, NM, NV, OR, SD, TX, UT, WA, WY – Costa Rica, Guatemala, Mexico
The subspecies Amara californica costaricensis (Bates) is found in Costa Rica (Hieke 1990: 221).
This species is known from southwestern Alberta (Waterton Lakes National Park, UASM), southern British Columbia (Jarrett and Scudder 2001: 379), Washington (Minsk and Hatch 1939: 215; Pierce County, UASM), eastern Oregon (Baker County, CNC), and northwestern Montana (Flathead County, UASM). There is one specimen, identified by Hieke, from Mayo in central Yukon Territory (CNC).
CAN: AB, BC USA: MT, OR, WA [YT]
This form was considered a synonym of Amara idahoana Casey by Lindroth (1968: 703) but regarded as a valid species by Hieke (1993: 123).
This species is known from eastern Oregon (Baker County, USNM) to western South Dakota (Lawrence County, USNM), north to southeastern Montana (Powder River County, USNM), south to western Texas (Culberson County, CMNH) and central Arizona (Gila County, MCZ).
USA: AZ, CO, ID, MT, NM, OR, TX, UT, WY
This species ranges from southern Alaska (Anchorage area, UASM) to southern Northwest Territories (CNC), south to northern Oregon (Hood River County, MCZ) and southern Colorado (Elias 1987: 633).
CAN: AB, BC (VCI), NT USA: AK, CO, ID, MT, OR, WA, WY
This widely distributed species ranges from Nova Scotia (Sable Island, CNC) to south-central British Columbia (Lindroth 1968: 708), south to southern California (San Diego County, CNC), southern New Mexico (Grant County, USNM; Wickham 1896c: 133; Fall and Cockerell 1907: 158), northern Oklahoma (French et al. 2001: 228; Alfalfa County, CMNH), central Mississippi (Casey 1918: 287, as Celia fluminea), and the Florida Panhandle (Okaloosa County, CNC). One old specimen labeled from “Texas” (USNM) is known.
CAN: AB, BC, MB, NB, NS, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, UT, VA, VT, WA, WI, WV, WY [TX]
This northern species ranges from the coast of Labrador to central Alaska (Lindroth 1968: 707), south in the west to northern Idaho (Bonner County, CNC) and northwestern Montana (Edwards 1975: 56).
CAN: AB, BC, LB, MB, NU, QC, SK, YT USA: AK, ID, MT
This species ranges from Nova Scotia (Majka et al. 2007: 9) to southwestern North Dakota (Tinerella 2003: 636), south to central Colorado (Summit County, UASM; Horn 1892b: 37; Wickham 1902: 238), northeastern Texas (Lamar County, Brian Raber pers. comm. 2010), northern Alabama (Madison County, CMNH, USNM), and northwestern South Carolina (Ciegler 2000: 75).
CAN: NB, NS, ON, QC USA: AL, CO, CT, DC, DE, GA, IA, IN, KS, MA, MD, ME, MI, MN, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species is found from Newfoundland (Lindroth 1955a: 104-105, as Amara subaenescens) to the Queen Charlotte Islands (Kavanaugh 1992: 75) and Vancouver Island, north to the Alaskan coast (Lindroth 1968: 703), south at least to east-central Utah (Grand County, CMNH, CNC), southern Colorado (Costilla and Mineral Counties, UASM), the Black Hills in western South Dakota (USNM), northwestern Indiana (Blatchley 1910: 109, as Amara subaenea), and northern New York (Casey 1918: 278, as Celia nupta). The records from southeastern New Jersey (Smith 1910: 206, as Amara subaenea), “Nebraska” (Bousquet and Larochelle 1993: 199), and Kansas (Knaus 1898: 19 and Snow 1903: 193, as Amara subaenea) need confirmation.
CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NT, ON, PE, QC, SK, YT USA: AK, CO, IA, ID, IL, IN, ME, MI, MN, MT, NH, NY, SD, UT, WI, WY [KS, NE, NJ]
This species is known from central and southern Texas (Horn 1892b: 38; Riley 2011; Bexar, Harris, and Lee Counties, MCZ) and central and southwestern New Mexico (Bernalillo and Luna Counties, CMNH). The record from southeastern Kansas (Knaus 1885: 58) needs confirmation.
USA: NM, TX [KS]
This species is found along the Atlantic Coast from “New York” to northern Florida (Hieke 1993: 143).
USA: FL, NC, NJ, NY, SC, VA
Thirteen species in North America, of which two are also found in Mexico and three are Holarctic.
Lindroth (1968) covered all but four (Amara nexa, Amara sodalicia, Amara lugens, and Amara tenebrionella) species known at the time. Two species were described subsequently by Hieke (2002). A revision of the subgenus is needed.
This species ranges from the Magdalen Islands in the Gulf of Saint Lawrence (CNC) to the Queen Charlotte Islands (Kavanaugh 1992: 75), south to southern California (San Bernardino County, CAS, MCZ), southern Arizona (Greenlee County, UASM), southern New Mexico (Otero County, MCZ), “Kansas” (Casey 1918: 252), central Michigan (Dunn 1982b: 37), and eastern New Brunswick (Webster and Bousquet 2008: 18).
CAN: AB, BC (QCI), MB, NB, ON, QC, SK USA: AZ, CA, CO, IA, ID, KS, MI, MN, MT, ND, NE, NM, NV, OR, SD, UT, WA, WI, WY
This circumpolar species is found over most of the boreal-alpine region in the Palaearctic Region and in North America from Alaska (Lindroth 1968: 717) to Newfoundland (Lindroth 1955a: 107), south to New England (Lindroth 1968: 716), northern Wisconsin (Messer 2010: 38), southern Colorado (Mineral County, UASM; Lindroth 1968: 716), northern Utah (Salt Lake County, CMNH), and Mount Rainier in western Washington (Lindroth 1968: 717). Several state records (e.g., CA, IA, MI, MN, ND, NM, OR) listed in Bousquet and Larochelle (1993: 196) and taken from earlier authors probably refer to other species or need confirmation.
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NT, NU, ON, QC, SK, YT USA: AK, CO, ID, ME, MT, NH, UT, VT, WA, WI, WY – Holarctic
This species ranges from eastern Manitoba to southwestern British Columbia, north at least to the Magunday River in southern Yukon Territory (Lindroth 1968: 714), south to southeastern California (San Bernardino County, CAS), southern Arizona (Maricopa County, UASM), southern New Mexico (Grant County, USNM; LeConte 1855: 353), and Durango in Mexico (UASM). The record from Wisconsin (Rauterberg 1885: 17) is probably in error; that from “Ontario” (Bousquet and Larochelle 1993: 196), based on a specimen from Nipigon Bay (CMNH) determined by Hieke, needs confirmation as the specimen could be mislabeled or could be a stray.
CAN: AB, BC, MB, SK, YT USA: AZ, CA, CO, ID, MT, ND, NE, NM, NV, OR, SD, UT, WA, WY [ON] – Mexico
This Holarctic species ranges from Finland to the Kamtschatka Peninsula (Hieke 2003a: 556) and from Alaska to northwestern Northwest Territories (Lindroth 1968: 716). The records from British Columbia (Jarrett and Scudder 2001: 380) and west-central Montana (Hansen et al. 2009: 353) need confirmation.
CAN: NT, YT USA: AK [BC, MT] – Holarctic
This boreal species is found from Newfoundland to southwestern British Columbia, north to east-central Alaska (Lindroth 1968: 717), south to southwestern California (Ventura County, MCZ), northeastern New Mexico (Snow 1885: 67; Las Vegas, CMNH) along the Rocky Mountains, the Black Hills in western South Dakota (Lawrence County, USNM), northeastern Minnesota along Lake Superior (Casey 1918: 312, as Amara aeneolucens), northwestern Pennsylvania (Warren County, CMNH), and “Massachusetts” (LeConte 1855: 353; MCZ).
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, CA, CO, ID, MA, ME, MI, MN, MT, NH, NM, NY, OR, PA, SD, UT, VT, WA, WI, WY
This species ranges from northern Arizona (Coconino County, USNM) and New Mexico (San Miguel County, USNM) south at least to the Isthmus of Tehuantepec (Bates 1882a: 77).
USA: AZ, NM – Mexico
This species is known only from the Sierra Nevada in California (Casey 1918: 253; Alpine, Amador, Eldorado, Placer, Plumas, and Shasta Counties, CAS, MCZ, USNM).
USA: CA
This species ranges from Newfoundland (Lindroth 1955a: 107) to the Seward Peninsula in western Alaska (Lindroth 1968: 715), south to “California” (Lindroth 1968: 715), central Colorado (Elias 1987: 633; Casey 1918: 252, as Celia reducta), eastern South Dakota (Brookings County, USNM), and southwestern North Carolina (Jackson County, USNM) along the Appalachian Mountains. The record from Siberia (Lindroth 1968: 715) refers to Amara transberingiensis Hieke (Hieke 2002: 671).
CAN: AB, BC, MB, NB, NF, NS, NT, ON, PE, QC, SK, YT USA: AK, CA, CO, CT, DC, IA, ID, IL, IN, MA, ME, MI, MN, MT, NC, ND, NH, NJ, NV, NY, OR, PA, RI, SD, UT, VA, VT, WA, WI, WY
This species is known from Andrew Lake in northeastern Alberta to south-central British Columbia, south to southeastern California, southeastern Arizona, and northeastern New Mexico (Hieke 2002: 660).
CAN: AB, BC USA: AZ, CA, CO, MT, NM, UT, WY
This species is known only from a few specimens collected in Colfax and Catron Counties, New Mexico (Hieke 1997: 250).
USA: NM
This species is known from the Rocky Mountains in northern Arizona (Hieke 1990: 223) and from the states of Durango (Bates 1891a: 248, as Celia tenebrionella var. aeneicolor) and Veracruz (Bates 1882a: 78) in Mexico.
USA: AZ – Mexico
This species is known from the Hudson Bay region in northeastern Manitoba, from northern British Columbia (CNC), and from the Northwest Territories to the Seward Peninsula in Alaska. In the Palaearctic Region, the species ranges from eastern Siberia to the Taimyr Peninsula, south at least to the Yakutsk area (Hieke 2002: 668).
CAN: BC, MB, NT, YT USA: AK – Holarctic
Eighty-seven species (Hieke 2007) in North America (29 species, of which six are adventive), Middle America (four species of which one, Amara dolosa Say, is endemic), Asia (about 55 species), Europe and northern Africa (about 25 species, most shared with western Asia), and the Afrotropical Region (nine species). Two species are Holarctic (Amara littoralis and Amara lunicollis).
No taxonomic revision has been published for the species of this group and such study is much needed. Hieke (2000) revised the six species of the impuncticollis group found in North America. Lindroth (1968) covered all but three (Amara haywardi, Amara pomona, and Amara sera) of the currently valid North American species known at the time. Since his publication, four taxa listed by Lindroth as synonyms (Amara neoscotica, Amara otiosa, Amara tenax, and Amara turbata) have been revalidated (Hieke 1994, 2000, 2003b), two new species described (Hieke 2002), and three adventive species (Amara communis, Amara eurynota, and Amara ovata) discovered on this continent. Identifications of some species are difficult and require examination of the male genitalia for confirmation.
[aurata group]
The range of this western species extends from western Montana (Russell 1968: 61; Hansen et al. 2009: 353) to Vancouver Island, north to the Prince Rupert area in British Columbia (Lindroth 1968: 711), south to Baja California Norte (CAS). The records from northern Colorado (Armin 1963: 201) and South Dakota (Kirk and Balsbaugh 1975: 26) need confirmation.
CAN: BC (VCI) USA: CA (CHI), ID, MT, NV, OR, WA [CO, SD] – Mexico
[cupreolata group]
This species ranges from “Maine” to “Montana” (Hieke 2003b: 207), south to southern Colorado (Elias 1987: 633), southern Texas (Bastrop and Kerr Counties, CNC), and the Florida Panhandle (Okaloosa County, CNC); also recorded from “Oregon” (Hieke 2003b: 207).
USA: AL, AR, CO, CT, DC, FL, GA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, VA, WI, WV
This species is found from Prince Edward Island (King County, CNC) and Nova Scotia (NSNH) to southwestern British Columbia (Creston area, UASM), south to northern Idaho (Hatten et al. 2007: 359), northern Colorado (Miller and Peairs 2008: 34; Larimer County, UASM), southern Oklahoma (Elliott et al. 2006: 126), southwestern Alabama (Mobile County, USNM), and North Carolina (Hieke 2003b: 205). Several state records (e.g., AR, DC, DE, GA, MS, NE, SC, UT) reported in Bousquet and Larochelle (1993: 195) refer to other species of the group or need confirmation.
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: AL, CO, CT, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, ND, NH, NJ, NY, OH, OK, PA, RI, SD, TN, TX, VA, VT, WI, WV
This species is yet known only from two specimens, both holotypes.
USA: IL
This form was considered a synonym of Amara cupreolata Putzeys by Lindroth (1968: 733) but regarded as a valid species by Hieke (2003b: 207).
This species ranges from Nova Scotia (Hieke 2003b: 206) to the Rocky Mountains in Alberta (CNC), south to “Colorado, ” “Illinois, ” and “New Jersey” (Hieke 2003b: 206).
CAN: AB, MB, NB, NS, ON, QC, SK USA: CO, IL, MA, ME, MI, NJ, VT, WI
This form was considered a synonym of Amara cupreolata Putzeys by Lindroth (1954b: 136) but regarded as a valid species by Hieke (2003b: 205).
This species is found from southern Manitoba (Casey 1924: 63, as Amara inflaticollis) to southeastern British Columbia (Robson, CNC), north to the Great Slave Lake area in Northwest Territories (CNC), south to “Nevada” (Hieke 2003b: 207), western Colorado (Montrose County, MCZ), “Illinois” (Hieke 2003b: 207), and Massachusetts (Middlesex County, MCZ).
CAN: AB, BC, MB, NT, SK USA: CO, IA, IL, KS, MA, MI, MN, ND, NE, NV, NY, SD, UT, VT, WI, WY
This form was considered a synonym of Amara cupreolata Putzeys by Lindroth (1968: 733) but regarded as a valid species by Hieke (2003b: 206).
[impuncticollis group]
This species is found east of the Rocky Mountains from Maine to at least southern Wisconsin, including southern Quebec and the Ontario Peninsula, south to southern Texas (Hieke 2000: 69), southeastern Louisiana (East Baton Rouge Parish, Igor M. Sokolov pers. comm. 2009), and southern Georgia (Torres and Ruberson 2006: 31). One specimen is known also from Nuevo León in Mexico and four from Costa Rica (Hieke 2000: 69). Several old records listed by Bousquet and Larochelle (1993: 196) (e.g., CO, CT, DE, FL, MB, MN, MS, NB, NF, NM, NS, PE, RI, SD, SK, VT) refer to Amara otiosa Casey or need confirmation. The record from Montana listed by Hieke (2000: 69) refers to Missouri; that from British Columbia (Jarrett and Scudder 2001: 380) is likely in error.
CAN: ON, QC USA: AL, AR, DC, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, NC, NE, NH, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI, WV – Costa Rica, Mexico
This Holarctic species ranges from Newfoundland to the Gulf Coast of Alaska, including Kodiak Island (Lindroth 1968: 731), south to southern California (Dajoz 2007: 20; San Diego County, CNC), northern New Mexico (Casey 1918: 297, as Amara acuminata; Rio Arriba County, CMNH), western Texas (Dajoz 2007: 23; Brewster County, CMNH), east-central Alabama (Lee County, CNC; LeConte 1855: 352), and eastern South Carolina (Ciegler 2000: 74). In the Palaearctic Region, the species is known only from northwestern Siberia and the Kamchatka Peninsula (Hieke 2000: 67).
CAN: AB, BC (QCI), MB, NB, NF, NS, NT, ON, PE, QC, SK, YT USA: AK, AL, AR, AZ, CA, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Holarctic
This species ranges from Newfoundland to southern Manitoba, south to northeastern Colorado, central Missouri, and North Carolina (Hieke 2000: 72-74). One specimen simply labeled from Texas is known (Hieke 2000: 69).
CAN: MB, NB, NF, NS (CBI), ON, PE, QC USA: CO, IL, MA, ME, MI, MN, MO, NC, NH, NJ, NY, OH, PA, RI, VT, WI, WV [TX]
Lindroth (1968: 728) regarded this form as a synonym of Amara impuncticollis Say but Hieke (2000: 71) considered it as a valid species.
This European species is adventive in North America where it is known in the east from Nova Scotia (Majka et al. 2006: 605) to southern Wisconsin (Messer 2010: 38), south to Virginia (Hieke 2000: 82). The first inventoried specimen collected on the east coast was found in Massachusetts in 1925. The species is also adventive in the western part of North America where it is known from southeastern British Columbia and Alberta (Hieke 2000: 82). The first inventoried specimen collected on the west coast was found in southeastern British Columbia in 1936 (see Hieke 2000: 82). One specimen simply labeled from Iowa is known (Hieke 2000: 82). The record from “Cleveland, Or.” (Hieke 2000: 82) obviously refers to Ohio.
CAN: AB, BC, NB, NS, ON, PE, QC USA: CT, MA, NH, NY, OH, PA, RI, VA, WI [IA] – Adventive
The range of this species extends from southern Arizona and western Texas south to Guatemala (Hieke 1993: 110).
USA: AZ, TX – Guatemala, Mexico
This species ranges from southern Quebec to southeastern Alberta, south to southern Arizona, southern Texas, west-central Alabama, and North Carolina (Hieke 2000: 76-79).
CAN: AB, ON, QC, SK USA: AL, AR, AZ, CO, IA, ID, IL, IN, KS, KY, MA, MD, MI, MT, NC, NE, NH, NJ, NM, NV, NY, PA, RI, SD, TN, TX, UT, WI, WV, WY
Lindroth (1968: 730) regarded this form as a synonym of Amara littoralis Mannerheim but Hieke (1994: 337) considered it as a valid species.
This European species is adventive in North America where it is known in the east from Newfoundland (Lindroth 1955a: 111) to southwestern Manitoba (Stjernberg 2011: 71), south to northeastern Oklahoma (Hieke 1990: 203), northern Louisiana (Morehouse and West Carroll Parishes, Igor M. Sokolov pers. comm. 2009), and the Florida Panhandle (Okaloosa County, CNC) and in the west from southern British Columbia (Jarrett and Scudder 2001: 378; CNC) to southeastern Alberta (CNC), south to northern Colorado, north-central Arizona, and the San Francisco Bay area in west-central California [see Hieke 1990: Fig. 23]. The first inventoried specimen collected in the eastern part of this continent was found in Brooklyn, New York, in 1904 and in the western part in San Francisco, California, in 1941 (see Hieke 1990: 202, 204). This species was recorded from “Amérique septentrionale” by Dejean (1828: 466), under the name Amara trivialis Gyllenhal, a synonym of Amara aenea, suggesting that it was already present in North America by that time. The identification of the specimen(s) was confirmed by Lindroth (1968: 732). However, considering that the next documented specimen from this continent was collected only at the beginning of the XX Century, there is doubt about the provenance of Dejean’s specimen(s).
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, NE, NH, NJ, NV, NY, OH, OK, OR, PA, RI, SC, TN, VA, VT, WA, WI, WV, WY – Adventive
This species ranges from Newfoundland (Lindroth 1955a: 108) to west-central Northwest Territories, south to northern Alberta (Lindroth 1968: 723), east-central South Dakota (Kirk and Balsbaugh 1975: 27), central Iowa (O’Rourke et al. 2008: 126; Larsen et al. 2003: 292), and northeastern New York (Essex and Hamilton Counties, USNM); apparently isolated in central Colorado (Elias 1987: 633). The record from “Yukon Territory” (Ball and Currie 1997: 453) could not be confirmed.
CAN: AB, LB, MB, NB, NF, NT, ON, QC, SK USA: CO, IA, ME, MI, ND, NH, NY, SD, VT [YT]
This European species is adventive in North America where it is known from southern British Columbia (Jarrett and Scudder 2001: 379) to southern California (Los Angeles County, CMNH). The first inventoried specimen collected in that area was found in southwestern Washington in 1929 (Hatch and Kincaid 1958: 5). The species has been recorded also from the Washington D.C. area since 1964 (Hieke 1990: 205) and has been recently collected in Westchester County in southeastern New York (Peter W. Messer pers. comm. 2011).
CAN: BC USA: CA, MD, NY, OR, VA, WA – Adventive
This species ranges from “New Hampshire” (Hayward 1908: 54) to southeastern South Dakota (Kirk and Balsbaugh 1975: 27), south to Oklahoma (Alfalfa and Logan Counties, CMNH, USNM), northeastern Georgia (Clarke County, USNM), and central South Carolina (Ciegler 2000: 74). One old specimen labeled “Tex” (USNM) is known.
USA: CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, MN, NC, NE, NH, NJ, NY, OK, PA, RI, SC, SD, VA, WV [TX]
This species ranges from southern Manitoba to south-central British Columbia (Lindroth 1968: 724-725), south to east-central Washington (Casey 1924: 60, as Amara leviceps), northwestern New Mexico (Sandoval County, UASM, USNM), Kansas (Cheyenne, Douglas and Riley Counties, CNC, UASM, USNM), and central Illinois (Macon County, USNM).
CAN: AB, BC, MB, SK USA: CO, IA, ID, IL, KS, MN, ND, NE, NM, SD, WA, WI, WY
This European species is adventive in North America where it is known from the Maritime Provinces (Bousquet 1992a: 504; Majka 2005: 534) and Connecticut (Majka 2005: 534) [see Majka 2005: Fig. 1]. The record from the Gaspé Peninsula, Quebec (Majka 2005: 534), is based on a misidentified specimen of Amara aenea (CNC). The first inventoried specimen collected on this continent was found in New Brunswick in 1988 (Bousquet 1992a: 504).
CAN: NB, NS, PE USA: CT – Adventive
This species ranges from northwestern Montana (Russell 1968: 60) to Vancouver Island (Lindroth 1968: 728), south to southern California (San Diego County, CAS, CNC) and northern Utah (Davis, Salt Lake, and Utah Counties, CMNH).
CAN: BC (VCI) USA: CA (CHI), ID, MT, OR, UT, WA
This species is found from southern Manitoba to south-central British Columbia, north to central Alaska (Lindroth 1968: 726), south to the Sierra Nevada in east-central California (Tuolumne County, MCZ), central Arizona (Gila County, MCZ), central New Mexico (Fall and Cockerell 1907: 158), southern Kansas (Knaus 1905a: 218; Casey 1918: 311), and northern Indiana (Blatchley 1910: 107, as Amara protensa).
CAN: AB, BC, MB, SK, YT USA: AK, AZ, CA, CO, ID, IL, IN, KS, MT, ND, NE, NM, NV, OR, SD, UT, WA, WY
The range of this species extends from Nova Scotia (Majka et al. 2007: 9) to central British Columbia (Lindroth 1968: 735), south to southeastern Arizona (Greenlee County, MCZ), southern New Mexico (Grant County, USNM), western Texas (Jeff Davis County, Ken Karns pers. comm. 2009), and southern New Jersey (Cape May County, MCZ). The record from North Carolina (Brimley 1938: 122, as Amara polita) is probably in error. Three old specimens labeled from Brownsville in southeastern Texas (USNM) are known.
CAN: AB, BC, MB, NS, ON, QC, SK USA: AZ, CO, CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NM, NY, OH, OK, PA, SD, TX, UT, VT, WI, WY
This species is found along and west of the Rocky Mountains from southwestern Alberta to south-central British Columbia, south to “Utah” and northern Colorado (Hieke 2002: 642).
CAN: AB, BC USA: CO, ID, MT, UT, WA, WY
This European species is adventive in North America where it is known only from a few localities in eastern Newfoundland (Bousquet 1987a: 128-129). The first inventoried specimen collected on this continent was found in 1971.
CAN: NF – Adventive
This species is known only from two specimens collected in Maryland [not Pennsylvania as stated by Hieke (2002: 643)] and North Carolina (Hieke 2002: 643).
USA: MD, NC
This European species is adventive in North America where it is known in the east from Newfoundland (Lindroth 1955a: 111) to southern Saskatchewan (Ronald R. Hooper pers. comm. 2007), south to central Oklahoma (Grady County, Robert L. Davidson pers. comm. 2012), northwestern Mississippi (Panola County, CMNH) and the Florida Panhandle (Okaloosa County, CNC) [see Hieke 1990: Fig. 25]. The first inventoried specimen collected on the east coast was found in Rhode Island in 1901 (Hieke 1990: 207). The species is also adventive in the western parts of North America where it ranges from southeastern Alaska to central California, east to western Montana [see Hieke 1990: Fig. 25]. The first inventoried specimen collected on the west coast was found in western Washington in 1913 (Hatch 1953: 26).
FRA: PM CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AK, AL, AR, CA, CT, DC, FL, IA, ID, IL, IN, KY, MA, MD, ME, MI, MO, MS, MT, NC, NH, NJ, NY, OH, OK, OR, PA, RI, SC, TN, VA, VT, WA, WI, WV – Adventive
This circumpolar species ranges from eastern Siberia to Ireland, south to Spain, Italy, Mongolia, and northern China (Hieke 2003a: 550) and from Alaska (Lindroth 1968: 721) to Newfoundland (Lindroth 1955a: 109), south to northeastern West Virginia (Randolph County, CMNH), east-central Ohio (Usis and MacLean 1998: 67), southern New Mexico (Otero County, CNC), and southwestern Oregon (Niwa and Peck 2002: 787).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, CT, MA, ME, MI, MN, MT, NH, NM, NY, OH, OR, PA, RI, VT, WI, WV – Holarctic
This species is known from a few specimens collected in Baker and Klamath Counties in Oregon and Marin and El Dorado Counties in California (Hieke 2002: 654).
USA: CA, OR
This species ranges from northern Washington south to the Mexican border in southern California, east to eastern Nevada (Hieke 1993: 114). One specimen simply labeled from Mexico is known (Hieke 1993: 114).
USA: CA, NV, WA – Mexico
This species is, as far as known, restricted to southern British Columbia, including Vancouver Island (Lindroth 1968: 722), northern Washington, northern Idaho (Hatch 1953: 124), and northwestern Montana (Russell 1968: 60).
CAN: BC (VCI) USA: ID, MT, WA
Twenty-one species (Hieke 2007) in the Palaearctic Region (including the Himalayas and northern Africa), one of them (Amara quenseli) extending into the Nearctic Region and one (Amara simplex Dejean) extending into the Afrotropical Region.
Hieke (2006) revised the species of this subgenus.
This Holarctic species is widely distributed in the Nearctic Region from Newfoundland (Lindroth 1955a: 103) to Alaska, including the Kodiak, Aleutian and Pribilof Islands (Lindroth 1968: 696), south to southern California (Fall 1901a: 45, as Amara remotestriata), northern Arizona (Villa-Castillo and Wagner 2002: 246; Apache County, UASM), northern New Mexico (Fall and Cockerell 1907: 158, as Amara remotestriata and Amara femoralis; Milford et al. 2000: 21; Rio Arriba and Sandoval Counties, CMNH, UASM), Oklahoma (Hamilton 1894b: 353; Hatch and Ortenburger 1930: 8, as Amara remotestriata), and southwestern North Carolina (Jackson County, USNM).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, DE, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SD, UT, VA, VT, WA, WI, WY – Holarctic
The subspecies Amara quenseli silvicola Zimmermann is endemic to Europe (Hieke 2003a: 565).
Twenty-five species (Hieke 2007) in North America (nine species, all endemic), Asia (six species, only one endemic), Europe (13 species, six endemic), and northern Africa (four species, two endemic).
Lindroth (1968: 735-741, as pallipes group) covered all the North American species except Amara belfragei. Three new species have been described subsequently (Hieke 1990, 2000) and one of Lindroth’s species (Amara angustata) was found to consist of two species (Hieke 2000). Hieke (2000) revised the species of the angustata group.
[angustata group]
This species ranges from southern Quebec to southeastern British Columbia, south to southeastern Wyoming, southeastern Texas (Fort Bend and Harris Counties, Brian Raber pers. comm. 2010), east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), southern Mississippi (Hinds County, Drew A. Hildebrandt pers. comm. 2010), northern Alabama (Lawrence County, UASM), and South Carolina (Hieke 2000: 49-51). The records from “St. Louis, Montana” and “Cleveland, Oregon” listed by Hieke (2000: 51) refers to Missouri and Ohio respectively. The records from “New Brunswick” and “Prince Edward Island” (Bousquet and Larochelle (1993: 199) refer to other species; that from west-central Colorado (Wickham 1902: 237) needs confirmation.
CAN: BC, MB, ON, QC, SK USA: AL, CT, DC, DE, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV, WY [CO]
This species ranges from New Brunswick to South Dakota, south to southern Pennsylvania (Hieke 2000: 55-57) and northeastern West Virginia (Randolph County, CMNH).
CAN: NB, ON, QC USA: CT, IA, IL, IN, MA, ME, MD, MI, MN, NH, NY, OH, PA, SD, VT, WI, WV
This species is known from a few localities in central and southern Texas (Hieke 2000: 58).
USA: TX
This species ranges from Cape Breton Island to southern Manitoba, south to northern Arizona, northeastern Colorado, northern Illinois, and northeastern Virginia (Hieke 2000: 53-55). The record “Cleveland, Oregon” (Hieke 2000: 55) refers to Ohio.
CAN: MB, NB, NS (CBI), ON, QC USA: AZ, CO, CT, DC, IA, IL, IN, MA, MD, ME, MI, MN, ND, NH, NJ, NY, OH, PA, VA, VT, WI, WV
This form has been treated as a synonym of Amara angustata (Say) by Lindroth (1968: 736) but considered a valid species by Hieke (2000: 53).
[scitula group]
This species occurs along the Pacific Coast from central Oregon (Lincoln County, CNC) to Monterey County in California (Hieke 1990: 196).
USA: CA, OR
This species is known from southern Oregon (Lake County, CNC) and several localities in northern and central California (Hieke 1990: 198).
USA: CA, OR
This species is found from southwestern British Columbia, including Vancouver Island (Lindroth 1968: 740), to southern California (Hayward 1908: 46; Moore 1937: 10; CAS) and “Arizona” (Casey 1918: 318, as Triaena profuga), including eastern Nevada (White Pine County, UASM). The records from “Idaho” and “Utah” (Bousquet and Larochelle 1993: 200) are probably in error.
CAN: BC (VCI) USA: CA, NV, OR, WA
The range of this species extends from Cape Breton Island (Lindroth 1954c: 305) to Vancouver Island (Lindroth 1968: 739), north to Fort Smith in southern Northwest Territories (Bousquet 1987a: 130), south to western Washington (Hatch 1953: 129), southwestern Colorado (Montezuma County, CNC), Iowa (Hayward 1908: 46; Wickham 1911b: 6; Dickinson County, USNM), and Virginia (Casey 1918: 314). The records from “Nevada, ” “Tennessee” (Bousquet and Larochelle 1993: 200), and Georgia (J.E. LeConte 1849: 26; Fattig 1949: 30) need confirmation.
CAN: AB, BC (VCI), MB, NB, NS (CBI), NT, ON, QC, SK USA: CO, CT, DC, IA, ID, IL, IN, MA, ME, MI, MN, MT, ND, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WA, WI, WV [GA, NV, TN]
This species ranges from southern Manitoba to Vancouver Island (Lindroth 1968: 740), south to southern California (Fall 1901a: 45; Moore 1937: 10) and southwestern Colorado (Wickham 1902: 237), east to northwestern South Dakota (Kirk and Balsbaugh 1975: 27).
CAN: AB, BC (VCI), MB, SK USA: CA, CO, ID, MT, NV, OR, SD, UT, WA, WY
Worldwide, with about 300 species in 32 genera (Lorenz 2005: 324-327). The Northern Hemisphere is represented by about 45 species (only 14.5% of the world fauna) and the Western Hemisphere by 55 species (about 18.5%). Seventeen species occur in North America.
Bousquet (1996a) revised and provided a key to the Nearctic, Mexican, and West Indian species.
One species in temperate North America.
This species is known from a few scattered localities in North Carolina (Brimley 1938: 127), Rabun County in northeastern Georgia (Fattig 1949: 45), southwestern Alabama (Washington County, Paul K. Lago pers. comm. 2009), northeastern Mississippi (Tishomingo County, Drew A. Hildebrandt pers. comm. 2008), southern Louisiana (Bousquet 1996a: 453), central Arkansas (Pulaski County, CMNH), eastern Missouri (Anonymous 2007), east-central Illinois (Coles County, Robert L. Davidson pers. comm. 2012), eastern Oklahoma (Bousquet 1996a: 453), and eastern Texas (Sabine County, Brian Raber pers. comm. 2010; Riley 2011).
USA: AL, AR, GA, IL, LA, MO, MS, NC, OK, TX
One species in temperate North America.
This species is restricted to the Coastal Plain ranging from the District of Columbia to the Florida Panhandle, west to southeastern Mississippi (Drew A. Hildebrandt pers. comm. 2007) [see Bousquet 1996a: map 1].
USA: AL, DC, FL, GA, MS, NC, SC
Six species (Bousquet 1996a: 456), possibly eight (Lorenz 2005: 324), in the temperate and tropical areas of the Nearctic (two species), Neotropical, Australian, and Oriental Regions.
This species ranges from Massachusetts to central Kansas, south to southeastern Texas and southern Florida [see Bousquet 1996a: map 2]; also recorded from the Bahamas (Turnbow and Thomas 2008: 10).
USA: AL, AR, CT, DC, FL, GA, IL, IN, KS, KY, LA, MA, MD, MO, MS, NC, NH, OH, OK, RI, SC, TN, TX, VA, WV – Bahamas
This species ranges from eastern Texas south to Honduras; it also occurs in the Bahamas and Cuba [see Bousquet 1996a: 459, map 3].
USA: TX – Bahamas, Belize, Cuba, Guatemala, Honduras, Mexico
Ten species in the temperate and tropical areas of the Nearctic (two species), Neotropical, and Australian (one species in the Malay Archipelago) Regions.
This species ranges from southeastern Texas (Bousquet 1996a: 465; map 5) south to Columbia (Martínez 2003: 17).
USA: TX – Colombia, Costa Rica, Honduras, Mexico
This species is known from the Florida Peninsula and several islands of the West Indies [see Bousquet 1996a: map 4].
USA: FL – Bahamas, Cuba, Dominican Republic
Three species in temperate regions of North America (one species) and eastern Asia (two species).
This species has a disjunct distribution over eastern North America. In the north, it ranges from Nova Scotia (Majka et al. 2007: 11) to southeastern Manitoba, south to Nebraska, northwestern Tennessee (Obion County, CMNH), and Maryland; in the south, it occurs along the Coastal Plain from Georgia (Fattig 1949: 45) to southern Florida, west to southern Louisiana [see Bousquet 1996a: map 6]. The species could reach the Rio Grande since Summers (1874b: 135) reported seeing one specimen “from Mexico, near the Rio Grande.”
CAN: MB, NB, NS, ON, QC USA: AL, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SD, TN, VA, VT, WI, WV
Lachnocrepis parallela (Say). This species is found in marshes and swamps in temperate eastern North America and is closely related to two species living under similar conditions in eastern Asia. The genus Lachnocrepis is another example of taxa exhibiting disjunct eastern Asia-eastern North America distributional patterns. Most current biogeographers agree that such disjunct patterns result from range restrictions of taxa associated with the mixed mesophytic forest during the mid-Tertiary.
About 50 species in temperate and tropical areas of the Nearctic (four species), Palaearctic (11 species), Oriental, Afrotropical (one species), and Australian Regions.
This species ranges from southern Maine to east-central Minnesota (Kamal J.K. Gandhi pers. comm. 2008), south to southeastern Texas and the Florida Peninsula; also known from one locality in Cuba [see Bousquet 1996a: map 7]. The record from southeastern Wyoming (Lavigne 1977: 47) is probably based on a mislabeled specimen or a stray.
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI, WV – Cuba
This species is restricted to the Coastal Plain ranging from Maryland to central Florida, west to central Louisiana [see Bousquet 1996a: map 9].
USA: AL, FL, GA, LA, MD, MS, NC, SC, VA
This species ranges from southern New Hampshire to northwestern Arkansas, north to southernmost Ontario and the central part of the lower peninsula of Michigan, south to eastern Texas and the Florida Panhandle [see Bousquet 1996a: map 10]. The record from “Iowa” (Bousquet and Larochelle 1993: 200) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, FL, GA, IL, IN, LA, MA, MD, MI, MO, MS, NC, NH, NJ, NY, OH, RI, PA, SC, TN, TX, VA [IA]
This species occurs from southern Maine and southern Quebec to western Minnesota, south to east-central Texas (Riley 2011), southwestern Louisiana and southern Florida [see Bousquet 1996a: map 8].
CAN: ON, QC USA: AL, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, PA, RI, SC, TN, TX, VA, VT, WI
About 30 species in temperate and tropical areas of the Nearctic (six species) and Neotropical Regions, including the West Indies. The species are currently arrayed in two subgenera: Stenocrepis s.str. and Stenous Chaudoir.
Ten species, ranging from southern Texas to central Argentina and including the West Indies, belong to this subgenus.
This species ranges from southern Texas to southern Mexico [see Bousquet 1996a: map 11]; it is also known from the Bahamas, Cuba, Cayman Islands, Trinidad and Tobago, and French Guiana (Bousquet 1996a: 482-483). The record from southeastern Louisiana (Summers 1874a: 80, as Oodes texanus) is suspect; that from northeastern Kansas (Knaus 1901: 110) refers to Stenocrepis cuprea (Knaus 1903: 187).
USA: TX [LA] – Bahamas, Cuba, Cayman Islands, French Guiana, Trinidad and Tobago
Twenty species in temperate and tropical areas of the Nearctic (five species) and Neotropical (19 species) Regions, including the West Indies.
This species ranges from southeastern Wisconsin (Messer 2010: 39) and southernmost Ontario south to southern Florida and the Rio Grande in southern Texas, west along the Rio Grande to central New Mexico [see Bousquet 1996a: map 15]. The species is not yet know from Mexico but it certainly occurs at least along the states bordering Texas. The record from “Pennsylvania” (Bousquet and Larochelle 1993: 201) needs confirmation.
CAN: ON USA: AL, AR, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NE, NM, OH, OK, SC, TN, TX, VA, WI, WV [PA]
This species ranges from Long Island, New York, to south-central Oklahoma, south to Nicaragua (Bates 1882a: 46) and southern Florida, west to the Mexican state of Nayarit; also known from several islands in the Bahamas and the Greater Antilles [see Bousquet 1996a: map 12].
USA: AL, AR, FL, GA, IL, KY, LA, MD, MS, NC, NJ, NY, OH, OK, SC, VA, TX, WV – Bahamas, Cuba, Dominican Republic, Guatemala, Haiti, Jamaica, Mexico, Nicaragua
This species ranges from southwestern California to eastern Arizona, south along the Gulf of California to Nayarit [see Bousquet 1996a: map 17]. A few specimens simply labeled from New Mexico and Texas are known.
USA: AZ, CA [NM, TX] – Mexico
This species ranges from New Jersey to southern Wisconsin, south through southeastern Kansas (Knaus 1903: 188) and eastern and central Texas to southern Mexico, and to the Florida Keys, west to the Pacific Coast in the Mexican state of Colima; also recorded from the Bahamas (Turnbow and Thomas 2008: 15) and Cuba [see Bousquet 1996a: map 13]. The record from “New Mexico” (Bousquet and Larochelle 1993: 201, as Stenocrepis quatuordecimstriata) is likely in error.
USA: AL, AR, FL, GA, IL, IN, KS, KY, LA, MS, NC, NJ, OH, OK, SC, TN, TX, VA, WI, WV – Bahamas, Cuba, Mexico
This species ranges from southern Texas south to Argentina (Brullé 1837: 32, as Oodes pallipes) and along the West Indies from Cuba to at least Puerto Rico [see Bousquet 1996a: map 16].
USA: TX – Argentina, Brazil, Cayman Islands, Colombia, Costa Rica, Cuba, Dominican Republic, Ecuador, Guatemala, Haiti, Jamaica, Mexico, Peru, Puerto Rico.
Worldwide, with about 270 species in 19 genera (Lorenz 2005: 319-323, as Panagaeina). The Northern Hemisphere is represented by only about 40 species (15.5 % of the world fauna). Four species in two genera are found in North America.
Fourteen species in temperate and tropical areas of the Nearctic (three species), Neotropical (four species), and Palaearctic (eight species) Regions arrayed in two subgenera: Panagaeus (Palaearctic Region) and Hologaeus (Western Hemisphere).
Ogueta (1966a) reviewed the Western Hemisphere species and provided a key for their identification.
Western Hemisphere, with six species in the Nearctic (three species) and Neotropical (four species) Regions.
This species ranges from Long Island, New York (Notman 1928: 213; MCZ), to southern Florida (Peck and Thomas 1998: 20), west to eastern Texas along the Gulf Coast (Snow 1906a: 141; Galveston County, USNM); also recorded from northern Indiana (Blatchley 1910: 65; Schrock 1985: 352) and along the Ohio River in southwestern Ohio (Dury 1902: 111). The record from “Pennsylvania” (Bousquet and Larochelle 1993: 202) needs confirmation.
USA: AL, DE, FL, IN, LA, MD, MS, NC, NJ, NY, OH, TX, VA [PA]
This species is found from Massachusetts (Harris in Scudder 1869: 264) and Connecticut (Krinsky and Oliver 2001: 136) to western South Dakota (Larsen and Purrington 2010: 571), including southern Ontario (Lindroth 1969a: 969), south to southeastern Texas (San Patricio County, CNC) and southern Florida (Peck and Thomas 1998: 20); also recorded from northeastern New Mexico (Fall and Cockerell 1907: 157).
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, VA, WI, WV
This species ranges from central and southern Arizona (Cochise, Santa Cruz, and Yavapai Counties, CMNH, CNC; LeConte 1879c: 60) and western and south-central Texas (Presidio and Bexar Counties, CMNH, MCZ; LeConte 1879c: 60), south at least to central Veracruz and the Federal District in Mexico (Ogueta 1966a: 12); also found in the Baja California Peninsula (Horn 1894: 307).
USA: AZ, TX – Mexico
Two species in temperate and tropical areas of the Nearctic (one species) and Neotropical (two species in Mexico) Regions.
Van Dyke (1927b: 93) discussed the structural differences between the two species.
This species ranges from southwestern Tennessee (Hardeman County, CMNH) to southwestern Arizona (Maricopa County, USNM), north to southeastern Colorado (Michels et al. 2008; Las Animas County, Robert L. Davidson pers. comm. 2008) and northeastern Kansas (Riley County, CNC), south to the state of Guanajuato on the Mexican Plateau (Ball and Shpeley 1992a: 60). The record from southern Wisconsin (Rauterberg 1885: 12) is probably in error.
USA: AZ, CO, KS, NM, OK, TN, TX – Mexico
Worldwide, with about 980 species (Lorenz 2005: 328-342) arrayed in two subtribes: Callistina (77 afro-oriental and one Palaearctic species) and Chlaeniina (about 900 species).
Worldwide, with about 900 species (Lorenz 2005: 328-341). The vast majority of species (about 81 % of the world fauna) are found in the Oriental and Afrotropical Regions.
Worldwide, with about 855 species (Lorenz 2005: 328-341) arrayed in 62 subgenera. The genus is mainly represented in the Afrotropical and Oriental Regions with about 650 species (approximately 76% of the world fauna). The North American fauna has 52 species (6% of the world fauna) in the boreal, temperate, and subtropical regions.
Bell (1960) revised the North American species. Since the publication of this work, one Neotropical species was discovered in southern Texas (Chlaenius azurescens), the name of one species (Chlaenius perplexus) was replaced by an older one (Chlaenius circumcinctus), and two of Bell’s species (Chlaenius cursor and Chlaenius herbaceus) have been shown to be a complex of two species each and the species found in North America to be Chlaenius sparsus and Chlaenius patruelis respectively. A number of subspecies recognized by Bell (1960) are placed in synonymy in this catalogue.
One species along the Coastal Plain of eastern North America.
This species is found from southeastern Georgia (Fattig 1949: 44) to southern Florida (Bell 1960: 102), west to southeastern Texas (Aransas County, USNM); also recorded from South Bimini Island in the Bahamas (Darlington 1953: 8).
USA: AL, FL, GA, MS, TX – Bahamas
Seven species in the Nearctic (two species) and Neotropical (five species) Regions.
To date this species has been recorded only from southeastern Arizona (Bell 1960: 103).
USA: AZ
This species ranges from Maine to southern Alberta, south to southeastern Arizona, central Texas, and central Florida (Bell 1960: 104-105).
CAN: AB, MB, ON, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
Three species in eastern North America.
The generic name Agreuter Lepeletier and Audinet-Serville (in Latreille et al. 1828: 633) was proposed for two species, Chlaenius chlorodius Dejean, 1826 and Chlaenius elegantulus Dejean, 1826 (= Chlaenius pusillus Say, 1823). Bousquet (2002b: 6) designated Chlaenius elegantulus as type species and so the name is listed as a synonym of Anomoglossus Chaudoir (e.g., Lorenz 2005: 333). However, Duponchel (1840a: 196) had selected Chlaenius chlorodius as the type species and so Agreuter is a senior synonym of Amblygenius LaFerté-Sénectère, 1851.
This species ranges from Virginia (Robert L. Davidson pers. comm. 1992) to northeastern Iowa (Purrington et al. 2002: 201), south to eastern Texas (Sabine County, CMNH), southeastern Louisiana (Colby 2002: 37), and northern Florida (Bell 1960: 106).
USA: AL, AR, FL, GA, IA, IL, LA, MO, MS, NC, SC, TN, TX, VA
This species ranges from Nova Scotia (Lindroth 1969a: 975) to eastern South Dakota (Kirk and Balsbaugh 1975: 36; French et al. 2004: 557), south to central Oklahoma and central Florida (Bell 1960: 107). Old specimens labeled “Tex” are also known (Bell 1960: 107).
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, VA, VT, WI, WV [TX]
This species ranges from southern Quebec (Saint-Clet, CNC) to southeastern Minnesota (Gandhi et al. 2005: 929), south to southern Texas and central Florida (Bell 1960: 108). At least one specimen labeled “Mass” is known (Bell 1960: 108).
CAN: ON QC USA: AL, AR, CT, DC, DE, GA, FL, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NC, NE, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI [MA]
About 140 species in all zoogeographical regions except the Australian Region. The Nearctic Region is represented by 14 species.
[aestivus group]
This species ranges from Massachusetts (Bell 1960: 120) to east-central Iowa (Wickham 1888: 82), including southernmost Ontario (Lindroth 1969a: 981), south to southeastern Louisiana (Allen 1965: 75) and central Florida (Bell 1960: 120). The record from eastern Kansas (Popenoe 1877: 23) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KY, LA, MA, MD, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, SC, TN, VA, WI [KS]
This species ranges from New Jersey (Bell 1960: 121) to east-central South Carolina (Ciegler 2000: 80) and southwestern Alabama (Löding 1945: 23; Bell 1960: 121). The record from “Mississippi” (Bousquet and Larochelle 1993: 204) needs confirmation.
USA: AL, DE, GA, NC, NJ, SC, TN, VA [MS]
This species ranges from southern Texas (Johnson 1978: 68) to Veracruz (Davidson 1980: 88) along the east coast of Mexico.
USA: TX – Mexico
This species ranges from “Massachusetts” to central North Dakota (Bell 1960: 122), north to southeastern Manitoba (Ryan and Holliday 2006: 414), south to northeastern New Mexico, central Texas, northwestern Georgia (Fattig 1949: 44) along the Appalachians, and Long Island, New York, along the east coast (Bell 1960: 122).
CAN: MB USA: AR, CO, CT, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NJ, NM, NY, OH, OK, PA, RI, SD, TN, TX, VA, WI, WV
This rarely collected species is known from Arkansas, southern Missouri (Bell 1960: 121), and eastern Oklahoma (Latimer County, UASM). Specimens labeled «S[outh] Ill[inois]» are known (Bell 1960: 121).
USA: AR, MO, OK [IL]
[cursor group]
This species is known from southeastern Arizona to central Texas (Bell 1960: 118), south to Durango and San Luis Potosí in Mexico (Davidson 1980: 133).
USA: AZ, NM, TX – Mexico
This species is known from Sonora (Davidson 1980: 129), southern Arizona, southern California (Bell 1960: 117), and the Baja California Peninsula (Horn 1894: 311).
USA: AZ, CA (CHI) – Mexico
This species is known from southern Alabama, southern Georgia, and throughout Florida (Bell 1960: 118). A specimen labeled “N. Car.” is known (Bell 1960: 118).
USA: AL, FL, GA [NC]
This species was known for a long time under the name Chlaenius herbaceus Chevrolat, 1834 (see Bell 1960: 117) until Bell (1966a) showed that Chlaenius patruelis and Chlaenius herbaceus were two distinct species. Chlaenius herbaceus is known only from Veracruz and Campeche in Mexico (Davidson 1980: 125).
This species is known from the Baja California Peninsula and from southern Arizona south, through central and western Mexico, to Central America (Robert L. Davidson pers. comm. 2007).
USA: AZ – Mexico
Horn (1876d: 275) listed this name as a junior synonym of Chlaenius cursor Chevrolat, 1835 described from “Orixaba” in Veracruz. However, both names apply to distinct species (Robert L. Davidson pers. comm. 2007).
[laticollis group]
This species ranges from New Jersey (Bell 1960: 131) to eastern South Dakota (Kirk and Balsbaugh 1975: 36), including southernmost Ontario (Lindroth 1969a: 982), south to northeastern New Mexico (Union County, CNC), southeastern Texas, and southern Florida (Bell 1960: 131).
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MI, MN, MO, MS, NC, NE, NJ, NM, OH, OK, PA, SC, SD, TN, TX, VA, WI
This species is found in southeastern United States from South Carolina (Kirk 1970: 16; Ciegler 2000: 80) to northern Arkansas, south to southeastern Texas and southeastern Georgia (Bell 1960: 130). The records from Illinois (Leng and Beutenmüller 1893: 143; Blatchley 1910: 166), “Iowa” (Jaques and Redlinger 1946: 296), Kansas (Popenoe 1877: 23; Snow 1903: 193), “Oklahoma” (Arnold 2008), and Missouri (Summers 1873: 145) need confirmation; that from southeastern Pennsylvania (Rathvon 1869: 525) must be in error.
USA: AL, AR, GA, LA, MS, SC, TN, TX [IA, IL, KS, MO, OK]
This species ranges from southern Maine (Majka et al. 2011: 47) to southeastern Nebraska (Nemaha County, Foster F. Purrington pers. comm. 2012), including southernmost Ontario (Bousquet 1987a: 132), south to Durango and Nuevo León (Davidson 1980: 92) in Mexico and southern Florida (Bell 1960: 129), west along southern United States to southeastern Arizona (Dajoz 2004: 116). The record from Tabasco in Mexico (Davidson 1980: 92) is suspect; that from southern Wisconsin (Rauterberg 1885: 18) needs confirmation.
CAN: ON USA: AL, AR, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NM, NY, OH, OK, PA, SC, TN, TX, VA, WV [WI] – Mexico
This species ranges from southeastern Arizona to southeastern Texas, south to Oaxaca in Mexico (Bell 1960: 128); also recorded from San Bernardino County in southeastern California (Dajoz 2007: 20).
USA: AZ, CA, NM, TX – Mexico
[sericeus group]
This transamerican species ranges from Newfoundland (Lindroth 1955a: 135) to southwestern British Columbia (Lindroth 1969a: 983), south to southern California (Bell 1960: 125, 127), Chihuahua (Davidson 1980: 84) and northern Sonora (Bates 1884: 267, as Chlaenius regularis) in Mexico, and central Florida (Peck and Thomas 1998: 20).
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
Bell (1960: 123-128) recognized four subspecies within Chlaenius sericeus: sericeus s.str., regularis, viridifrons, and sierricola, all separated by color and relatively minor differences in the pronotum shape.
Nineteen species can be assigned to this subgenus: five in North America (Bell 1960: 109, as solitarius group), six in the Neotropical Region (Chlaenius argentinicus Jedlička, Chlaenius chlorochrous Chaudoir, Chlaenius ecuadoricus Jedlička, Chlaenius leucoscelis Chevrolat, Chlaenius peruanus Erichson, and Chlaenius purpureus Chaudoir), and ten in the Palaearctic Region (see Liu et al. 2011), of which one extends into the Oriental Region.
Liu et al. (2011: 22) argued that it is premature to place any of the Western Hemisphere species of Chlaenius in the subgenus Lithochlaenius because they differ from the Palaearctic species of Lithochlaenius in having the lateral edge of elytron rounded at level of the humerus, instead of being angulate. However, I agree with Robert L. Davidson (pers. comm.) that the Western Hemisphere species related to Chlaenius prasinus are closely related to the Palaearctic species of Lithochlaenius and at this time I prefer to include these species in the subgenus Lithochlaenius.
This species ranges from New Brunswick (Restigouche and Queens Counties, CNC) to southern Manitoba (Lindroth 1969a: 981), south to northwestern Arkansas (Bell 1960: 112), northwestern Mississippi (Coahoma County, Drew A. Hildebrandt pers. comm. 2008), and North Carolina (Robert L. Davidson pers. comm. 1992). The record from southwestern Colorado (Elias 1987: 634) needs confirmation.
CAN: MB, NB, ON, QC USA: AR, CT, DC, DE, IA, IL, IN, KY, MA, MD, ME, MI, MN, MO, MS, NC, ND, NH, NJ, NY, OH, PA, SD, TN, VA, VT, WI, WV [CO]
This subspecies is found in the drainage basin of the Colorado River in southwestern Colorado, southern Utah, Arizona, and southeastern California (Bell 1960: 114).
USA: AZ, CA, CO, UT
This subspecies ranges from central Colorado to southernmost New Mexico and central Texas (Bell 1960: 115), including northwestern Oklahoma (Cimarron County, CMNH, CNC).
USA: CO, NM, OK, TX
This subspecies is known from southwestern Oregon (Hatch 1953: 163, as Chlaenius leucoscelis) and through much of California as far south as Kern County (Bell 1960: 114).
USA: CA, OR
This species ranges from Massachusetts to “Colorado” (Horn 1876d: 267), including southern Michigan and “Minnesota” (Gandhi et al. 2005: 929), south at least to central Texas and southern Florida (Bell 1960: 110-111).
USA: AL, AR, CO, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WI, WV
This species ranges from central Missouri (Morgan County, CMNH) to Arizona (Bell 1960: 110), south to Nicaragua (Bates 1882a: 45); also found along the Baja California Peninsula (Davidson 1980: 69).
USA: AZ, KS, MO, NM, TX – Guatemala, Mexico, Nicaragua
This species inhabits the Great Plains and the Great Central Plains from southern Alberta to southwestern Pennsylvania (Allegheny and Westmoreland Counties, Robert L. Davidson pers. comm. 2008), south to northern Mississippi (Drew A. Hildebrandt pers. comm. 2007), east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), central Texas, and central New Mexico (Bell 1960: 111). The records from southwestern Ohio (Dury 1906: 257) and “Utah” (Bousquet and Larochelle 1993: 206) need confirmation. The record from “Georgia” (J.E. LeConte 1849: 26) is probably in error; that from Beaver Islands in Michigan (Hatch 1925: 553) was based on misidentified specimens of Chlaenius cordicollis (Dunn 1985a: 10). The specimen labeled from northeastern Florida (see Bell 1960: 111) is probably mislabeled (Robert L. Davidson pers. comm. 1992). Old specimens simply labeled from Michigan and Minnesota are known (Bell 1960: 111).
CAN: AB USA: AR, CO, IA, IL, IN, KS, KY, LA, MO, MS, MT, ND, NE, NM, OK, PA, SD, TN, TX, WI, WY [MI, MN, OH, UT]
About 80 species in all zoogeographical regions except the Australian Region. The subgenus is best represented in the Northern Hemisphere with 58 species (about 73.5% of the world fauna) of which 17 are found in North America.
[circumcinctus group]
This species ranges from southern Georgia (Fattig 1949: 43) and throughout Florida west along the Gulf Coast to southeastern Texas, north to north-central Oklahoma (Alfalfa County, CMNH) and central Arkansas (Pulaski County, Robert L. Davidson pers. comm. 2012), south at least to Veracruz along the Gulf Coast (Bell 1960: 146, as Chlaenius perplexus Dejean); also known from Cuba and Puerto Rico (Bell 1960: 146, as Chlaenius perplexus Dejean) and the Dominican Republic (CMNH).
USA: AL, AR, FL, GA, LA, MS, OK, TX – Cuba, Dominica Republic, Mexico, Puerto Rico
This species has been known for a long time under the name Chlaenius perplexus Dejean, 1831 but Say’s name has priority (see Bousquet 1993: 6).
[glaucus group]
This species is found from central California (Bell 1960: 144) to western Texas (Jeff Davis, Ken Karns pers. comm. 2009), south to northern Sonora (Bates 1884: 267).
USA: AZ, CA, NM, TX – Mexico
This species is known only from a few specimens collected in the lower Sacramento River Valley of northwestern California (Bell 1960: 145).
USA: CA
[impunctifrons group]
This species ranges from New Brunswick (Webster and Bousquet 2008: 21) to North Dakota (Tinerella 2003: 635), north to southern Manitoba (Lindroth 1969a: 984), south to eastern Texas (Houston County, CNC; Riley 2011) and southern Florida (Bell 1960: 137). The records from “Wyoming” and “Colorado” (Bousquet and Larochelle 1993: 207) are likely in error.
CAN: MB, NB, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
The name Chlaenius indutus LaFerté-Sénectère, 1851 is often listed as a junior synonym of this species (e.g., Bell 1960: 136; Lindroth 1969a: 983). However LaFerté-Sénectère never provided a description of the taxon and the name is a nomen nudum.
[nemoralis group]
This species ranges from south-central New Hampshire (Merrimack County, Ross T. Bell pers. comm. 2008) to eastern South Dakota (Kirk and Balsbaugh 1975: 35), including southern Ontario (Lindroth 1969a: 987), south to southern Texas (Bell 1960: 143) and southwestern South Carolina (Hampton County, CMNH). The records from “Utah” (Bousquet and Larochelle 1993: 207) and New Mexico (Fall and Cockerell 1907: 160) need confirmation.
CAN: ON USA: AL, AR, CT, DC, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NE, NH, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WI, WV [NM, UT]
This species ranges from Massachusetts (Easton 1909: 37) to northeastern South Dakota (Kirk and Balsbaugh 1975: 36), including southern Ontario (Lindroth 1969a: 984), south to central Texas and southern Florida (Bell 1960: 140). The record from “New Hampshire” (Bousquet and Larochelle 1993: 208) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NJ, NY, OH, OK, ON, PA, RI, SC, SD, TN, TX, VA, WI, WV [NH]
This species occurs from northwestern Georgia and southwestern South Carolina (Hampton County, CMNH) to central Florida, west to eastern Texas, north to central Oklahoma (Grady County, CMNH) and east-central Arkansas (Bell 1960: 141).
USA: AL, AR, FL, GA, LA, MS, OK, SC, TX
This subspecies ranges from Newfoundland (Lindroth 1955a: 134) to northwestern Montana (Russell 1968: 66), south to central New Mexico (Fall and Cockerell 1907: 160), central Texas, northwestern Georgia, and northern South Carolina (Bell 1960: 142).
CAN: AB, NB, NF, NS, ON, QC, SK USA: AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
Bell (1960: 142) noted the presence of intergrade specimens between the two forms of Chlaenius tricolor in the area immediately west of the Rocky Mountains.
This subspecies ranges from southern British Columbia, including Vancouver Island (Lindroth 1969a: 986), to southern California (Bell 1960: 142) and Guatemala (Bates 1882a: 44).
CAN: BC (VCI) USA: AZ, CA, ID, NV, OR, UT, WA – Guatemala, Mexico
[pennsylvanicus group]
This species is known only from a few localities in central and southern Texas (Bell 1960: 149). The record from Georgia (Fattig 1949: 43) is likely in error (Bell 1960: 149).
USA: TX
This species is known from northern Georgia (Fattig 1949: 43) to southern Florida (Peck and Thomas 1998: 20) and from Inagua in the Bahamas (Darlington 1953: 8).
USA: FL, GA – Bahamas
This species is found from southern Saskatchewan and southern Alberta (Lindroth 1969a: 988) south to southern Arizona and southern Texas (Bell 1960: 148).
CAN: AB, SK USA: AZ, CO, KS, MT, ND, NE, NM, NV, OK, SD, TX, UT
This subspecies ranges from northern Utah to southern Arizona, east to western Texas (Bell 1960: 147).
USA: AZ, NM, TX, UT
This subspecies ranges from Newfoundland (Lindroth 1955a: 134) to Vancouver Island (Lindroth 1969a: 987), south at least to southwestern Oregon, southern Colorado along the Rocky Mountains (Bell 1960: 147), southeastern Louisiana (Saint Tammany Parish, Igor M. Sokolov pers. comm. 2009; Summers 1874a: 80), and southern Florida (Peck and Thomas 1998: 20). The record from southwestern California (Moore 1937: 12) is probably in error.
FRA: PM CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AL, AR, CO, CT, DC, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, TN, VA, VT, WA, WI, WV, WY
[vafer group]
This species is endemic to the Florida Peninsula (Bell 1960: 165; Peck and Thomas 1998: 20).
USA: FL
This species is known from southern Mississippi (Hancock County, Drew A. Hildebrandt pers. comm. 2008), southern Texas, and northern Mexico (Bell 1960: 151).
USA: MS, TX – Mexico
This species ranges from southeastern Nebraska (Lancaster County, Foster F. Purrington pers. comm. 2010) and central Missouri (Boone County, CMNH) south to Mississippi (CMNH), southwestern Louisiana (Hine 1906: 77), and southern Texas (Bell 1960: 151). The record from eastern South Dakota (Kirk and Balsbaugh 1975: 36) needs confirmation.
USA: AR, KS, LA, MO, MS, NE, OK, TX [SD]
[variabilipes group]
This species ranges from central California (Bell 1960: 150) south to “Nicaragua” (Bates 1882a: 44) and the Baja California Peninsula (Horn 1894: 311), including western New Mexico (Catron County, UASM; Snow 1885: 67; Fall and Cockerell 1907: 160) and western Texas (Jeff Davis County, UASM). The record from “Utah” (Bousquet and Larochelle 1993: 208) needs confirmation.
USA: AZ, CA (CHI), NM, TX [UT] – Guatemala, Mexico, Nicaragua
The range of this species extends from southeastern Oregon (Harney County, James R. LaBonte pers. comm. 1992) and west-central Nevada (Bell 1960: 150) south to the Baja California Peninsula (Horn 1894: 311) and at least Durango in Mexico (Ball and Shpeley 1992a: 61). The specimens labeled from Texas (Bell 1960: 150) are probably mislabeled or represent strays.
USA: AZ, CA, NV, OR, UT [TX] – Mexico
Three species in southwestern North America (one species) and Mexico (three species).
This species ranges from southern California to southwestern New Mexico, south to Oaxaca in Mexico (Bell 1960: 116).
USA: AZ, CA, NM – Mexico
The name Chlaenius dimidiatus Motschulsky, 1858 is often listed as a junior synonym of this species (e.g., Csiki 1931: 971; Bell 1960: 115). However, Motschulsky never provided a description of the taxon and the name is a nomen nudum.
Two species in the boreal and temperate regions of North America.
This species is known only from the type locality in north-central California (Bell 1960: 139).
USA: CA
Bell (1960: 139) stated that the holotype and still only known specimen may simply be an aberrant individual of Chlaenius lithophilus.
This species ranges from Newfoundland (Lindroth 1955a: 135) to central British Columbia, north to Fort Smith in southern Northwest Territories (Lindroth 1969a: 989), south to northeastern Washington (Hatch 1953: 163), “New Mexico” (Robert L. Davidson pers. comm. 1992), northern Texas (Cooke County, CNC), and northeastern Georgia (Bell 1960: 138). The record from “Oregon” (Wickham 1896c: 135) needs confirmation.
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AL, AR, CO, CT, DC, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY [OR]
Nine species in the Nearctic (five species), Neotropical (one Mexican species also found in Arizona), and Palaearctic (four species) Regions.
This species ranges from Newfoundland (Larson and Langor 1982: 594) to central Alaska (Lindroth 1969a: 993), south to northeastern Washington (Hatch 1953: 162), central Colorado along the Rocky Mountains, western South Dakota (Meade County, Foster F. Purrington pers. comm. 2010), and eastern Minnesota (Bell 1960: 134). The species is unknown east of the Mississippi south of the Canadian border. Fossil remnants of this species, dated between about 16, 700 and 25, 200 years B.P., have been unearthed in southeastern Iowa (Baker et al. 1986: 96) and southeastern Illinois (Schwert 1992: 78).
CAN: AB, BC, MB, NB, NF, NS, NT, ON, QC, SK USA: AK, CO, MN, MT, ND, SD, WA, WY
This species is known from a few specimens simply labeled from Arizona and from Mexico as far south as Oaxaca and central Veracruz (Bell 1960: 132; Davidson 1980: 31).
USA: AZ – Mexico
This species ranges from southeastern Alberta to Vancouver Island (Lindroth 1969a: 990-991), south at least to central California and northern Utah (Bell 1960: 133). One old specimen labeled from “Arizona” is also known (Bell 1960: 133).
CAN: AB, BC (VCI) USA: CA, ID, MT, NV, OR, UT, WA [AZ]
This species is restricted to the Pacific Coast ranging from southern British Columbia, including Vancouver Island (Lindroth 1969a: 993), south through the Coast Range to northern California (Bell 1960: 135). The record from Inyo County, California (Dajoz 2007: 19) needs confirmation. Fossil remnants from a Plio-Pleistocene sequence have been unearthed in northwestern Greenland (Böcher 1995: 25).
CAN: BC (VCI) USA: CA, OR, WA
This species ranges from Newfoundland (Lindroth 1955a: 133) to south-central British Columbia (Lindroth 1969a: 992), south to northeastern Washington (Hatch 1953: 162), “Wyoming” (Robert L. Davidson pers. comm. 1992), east-central Texas, southern Florida, and Cuba (Bell 1960: 133-134). The record from “Yukon Territory” (Ball and Currie 1997: 453) could not be confirmed.
CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, MT, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WA, WI, WY, TX [YT] – Cuba
The name Chlaenius exaratus LaFerté-Sénectère, 1851 is often listed as a junior synonym of this species (e.g., Bell 1960: 133; Lindroth 1969a: 991). However, LaFerté-Sénectère never provided a description of the taxon and the name is a nomen nudum.
One North American species.
In my opinion, Chlaenius purpuricollis is taxonomically isolated and within the actual structure of the genus Chlaenius deserves to be placed in its own subgenus. See Bell (1960: 135) and Lindroth (1969a: 994) as “purpuricollis group” for the descriptive character states of this new taxon.
This species is found from southern Quebec (Lindroth 1969a: 994) to southeastern British Columbia (CNC), north to Fort Smith in southern Northwest Territories (Bousquet 1987a: 132), south to Oregon (Bell 1960: 136), New Mexico (Perrault 1970: 56), Kansas (Popenoe 1878: 78; Bell 1960: 136), and “New Jersey” (Smith 1890: 92; Smith 1910: 213).
CAN: AB, BC, MB, NT, ON, QC, SK USA: CO, CT, IA, ID, IL, IN, KS, MA, MI, MN, ND, NE, NH, NJ, NM, NY, OR, PA, RI, UT, WA, WI, WY
Chlaenius purpuricollis Randall. This relatively small chlaeniine had been rarely collected until pitfall trapping showed that it is common in alvars, which are flat, relatively open grassy areas with shallow or sporadic soil cover over calcareous bedrock. Alvars are known from Sweden, Estonia, New York, Michigan, Ohio, and particularly central Canada. The chlaeniine is also found in similar habitats in other parts of North America.
Worldwide, with about 13, 200 species placed in the following 32 tribes: Anthiini (about 130 species), Atranini (two species), Calophaenini (about 50 species), Catapieseini (nine species), Corsyrini (six species), Ctenodactylini (about 65 species), Cyclosomini (about 120 species), Dryptini (about 85 species), Enoicini (five South African species), Galeritini (about 135 species), Geobaenini (four species), Ginemini (one South American species), Graphipterini (about 155 species), Harpalini (about 2, 725 species), Helluonini (about 180 species), Hexagoniini (about 65 species), Lachnophorini (about 115 species), Lebiini (about 4, 260 species), Licinini (about 235 species), Masoreini (about 50 species), Odacanthini (about 325 species), Omphreini (14 species), Pentagonicini (about 165 species), Perigonini (about 120 species), Physocrotaphini (about 40 species), Platynini (about 2, 670 species), Pseudomorphini (about 320 species), Sarothrocrepidini (about 25 species), Somoplatini (13 species), Sphodrini (about 825 species), Xenaroswellianini (one species), and Zuphiini (about 290 species).
Worldwide, with about 235 species arrayed in four subtribes: Dicaelina (45 species), Dicrochilina (30 species), Lestignathina (about 60 species), and Licinina (about 100 species). The Northern Hemisphere is represented by about 150 species (roughly 64% of the world fauna) and North America alone by 62 species (approximately 26.5 %).
Forty-five species arrayed in two genera, both represented in the Nearctic Region.
Twenty-nine species in the boreal, temperate, and tropical areas of the Nearctic (nine species), Oriental (12 species), Palaearctic (nine species), and Afrotropical (two species) Regions. The species are placed in three subgenera: Diplocheila (nine Oriental and one east Palaearctic species), Isorembus (18 species), and Neorembus Ball (one Asian species).
Ball (1959) revised the species of this genus. Since this publication, one new North American species has been described by Will (1998) and a few nomenclatural changes affecting the Nearctic species have been introduced by Lindroth (1968-1969a): Ball’s Diplocheila modesta corresponds to Diplocheila assimilis and his Diplocheila assimilis to Diplocheila impressicollis. Lindroth’s (1968: 940-941) key encompasses seven North American species.
Eighteen species in the Nearctic (nine species), Oriental (three species), Palaearctic (five species), and Afrotropical (two species, one shared with northern Africa and Spain) Regions. Several species listed in this subgenus by Lorenz (2005: 342-343) belong to the subgenus Diplocheila s.str.
This species is found from New Brunswick (Webster and Bousquet 2008: 20) to east-central South Dakota (Kirk 1971: 239), south to “Georgia” (LeConte 1844: 51).
CAN: NB, ON, QC USA: CT, GA, IA, IL, IN, MA, MD, ME, MI, MN, NH, NJ, NY, OH, PA, SC, SD, VT, WI
This species is known from a few localities in the Florida Panhandle and along the Mississippi Basin in northwestern Mississippi, western Tennessee, and southern Illinois [see Will 1998: Fig. 10].
USA: FL, IL, MS, TN
This species ranges from southern Quebec (Larochelle 1975: 80) to west-central South Dakota (Kirk and Balsbaugh 1975: 34), south to central New Mexico (Ball 1959: 71, as Diplocheila assimilis), the Rio Grande in southeastern Texas (Ball 1959: 72, as Diplocheila assimilis planulata), and southern Virginia (Hoffman 2010: 22). The records from “North Dakota” (Bousquet and Larochelle 1993: 209) and Connecticut (Britton 1920: 214, as Diplocheila laticollis; see Krinsky and Oliver 2001: 4) need confirmation.
CAN: ON, QC USA: DC, IA, IL, IN, KS, MA, MD, MI, MN, MO, MS, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SD, TX, VA, VT, WI [CT, ND]
This subspecies ranges from “Rhode Island” to east-central South Dakota (Ball 1959: 78), south to east-central Kansas (Snow 1880: 78; Ball 1959: 78), “Missouri” (Ball 1960b: 78), and New Jersey (Smith 1890: 82; Smith 1910: 206). The records from Louisiana (Summers 1874a: 80; Casey 1913: 148) and Alabama (Löding 1945: 18) refer to subspecies melissisa; those from “North Dakota” (Bousquet and Larochelle 1993: 209) and north-central Colorado (Wickham 1902: 238) need confirmation.
CAN: ON USA: CT, IA, IL, IN, KS, MI, MN, MO, NE, NJ, NY, OH, PA, RI, SD, WI [CO, ND]
This subspecies ranges along the Coastal Plain from eastern Virginia (Hoffman et al. 2006: 24) to southern Florida (Peck and Thomas 1998: 21), west to southeastern Texas (Ball 1959: 80); also recorded from Cuba (Ball 1959: 80).
USA: AL, FL, LA, MS, NC, SC, TX, VA – Cuba
As far as known, this species is endemic to the southern half of the Florida Peninsula (Ball 1959: 73).
USA: FL
This species occurs from Cape Breton Island (Bousquet 1987d: 107) to south-central British Columbia, north to central Northwest Territories along the Mackenzie River (Lindroth 1969a: 948), south to eastern Washington, “Nevada” (Ball 1959: 82), the Sierra Blanca in south-central New Mexico (Fall and Cockerell 1907: 159), northwestern Texas (Oldham County, Foster F. Purrington pers. comm. 2010), “Missouri” (Ball 1959: 82), northwestern Tennessee (Stewart County, Foster F. Purrington pers. comm. 2010), and western Virginia (Hoffman et al. 2006: 24). The record from southern Louisiana (Summers 1874a: 80) is probably in error; that from “Arkansas” (Bousquet and Larochelle 1993: 209) needs confirmation.
CAN: AB, BC, MB, NB, NS (CBI), NT, ON, PE, QC, SK USA: CO, CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, MT, ND, NE, NH, NM, NV, NY, OH, PA, SD, TN, TX, VA, VT, WA, WI, WY [AR]
This species ranges from southeastern Manitoba to the Okanagan Valley in southern British Columbia (Lindroth 1968: 943), south to northwestern Oregon (Hatch 1951: 119), “Utah” (Ball 1959: 64), northwestern Wyoming (Sikes 1994), and “North Dakota” (Donald P. Schwert pers. comm. 1989).
CAN: AB, BC, MB, SK USA: MT, ND, NV, OR, UT, WY
This species ranges from central Nova Scotia (Majka et al. 2007: 10) to the Okanagan Valley in British Columbia, north to Fort Smith in southern Northwest Territories (Lindroth 1968: 941), south to Inyo County in California (Dajoz 2007: 19), southeastern Arizona (Dajoz 2007: 21, as Diplochaetus striatopunctata), “Kansas, ” and northeastern New Jersey (Ball 1959: 63). Old specimens simply labeled from Texas and Georgia are known (Ball 1959: 63). The record from “Virginia” (Bousquet and Larochelle 1993: 209) needs confirmation.
CAN: AB, BC, MB, NB, NS, NT, ON, QC, SK USA: AZ, CA, IA, ID, IL, IN, KS, MI, MN, MO, MT, ND, NE, NH, NJ, NY, OH, OR, PA, SD, UT, VT, WA, WI [GA, TX, VA]
This species is known from Alberta, as far north as Edmonton, southern Manitoba (Lindroth 1969a: 946), Minnesota (Tinerella and Rider 2001: 321; Kamal J.K. Gandhi pers. comm. 2008), Wisconsin (Messer 2010: 39), “Illinois” (Ball 1959: 83), and southeastern Nebraska (Hall County, UASM).
CAN: AB, MB USA: IL, MN, NE, WI
Sixteen species (24 species-group taxa) restricted to the temperate and tropical zones of North America and Mexico (south to the vicinity of Mexico City). Three subgenera are recognized: Dicaelus s.str., Paradicaelus, and Liodicaelus.
Ball (1959) revised the species of this genus. Since the publication of this work, Ball (1992a) described one new Mexican species (Diplocheila franclemonti), one Mexican subspecies was raised to specific level (Diplocheila abbreviatus Bates), and another one (Diplocheila laevipennis dicaeloides) was placed in synonymy. A few changes are introduced here: Diplocheila subtropicus and Diplocheila quadratus treated as subspecies are raised to specific level, and Diplocheila purpuratus darlingtoni is placed in synonymy with Diplocheila quadratus.
Seven North American species arrayed in three species groups.
[elongatus group]
This species ranges from Maine (Foss 2001: 14) to eastern South Dakota (Kirk and Balsbaugh 1975: 35), including southern Ontario (Lindroth 1969a: 950), south to east-central Texas (Riley 2011) and southern Florida, including the Keys (Peck and Thomas 1998: 21). The record from Montreal, Quebec (Perrault 1977: 153) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI [QC]
[furvus group]
This subspecies ranges from “Maine” (Larochelle and Larivière 1990a: 32) to west-central New York, south to northern Florida (Ball 1959: 131-132), west to eastern Texas (Sabine County, Brian Raber pers. comm. 2010).
USA: AL, CT, DC, DE, FL, GA, MA, MD, ME, MS, NC, NH, NJ, NY, PA, RI, SC, TX, VA, VT, WV
Ball (1959: 132) reported the presence of intergrade populations of this and the sinuatus forms in Mississippi, Alabama, northern Florida, Georgia, South Carolina, and North Carolina. For practical reasons, these populations are considered to belong to the nominotypical subspecies.
This subspecies ranges from western Pennsylvania to southeastern Iowa (Ball 1959: 134) and west-central Illinois (Willand et al. 2011: 273 as Dicaelus dilatatus), south to southern Tennessee and southwestern North Carolina (Ball 1959: 134).
USA: IA, IL, IN, KY, NC, OH, PA, TN, VA, WV
This subspecies ranges from eastern Minnesota (Gandhi et al. 2005: 929) south to eastern Texas (Sabine County, Brian Raber pers. comm. 2010) and southern Mississippi, east to northwestern Georgia (Ball 1959: 123).
USA: AL, AR, GA, IA, KS, MN, MO, MS, NE, OK, TX
This subspecies ranges from eastern Pennsylvania to west-central Illinois (McCravy and Willand 2008: 157), south to Tennessee (Ball 1959: 122) and South Carolina (Kirk 1969: 14; Kirk 1970: 15; Ciegler 2000: 84). The records from New York (Notman 1928: 229, as Dicaelus ovalis) and “Georgia” (J.E. LeConte 1849: 26) need confirmation; that from southern Louisiana (Summers 1874a: 80) is probably in error.
USA: DC, DE, IL, IN, KY, MD, NC, NJ, OH, PA, SC, TN, VA, WV [GA, NY]
Ball (1959: 122) reported the presence of intergrade populations between this and the carinatus forms in Ohio, Indiana, Illinois, Tennessee, and North Carolina. For practical reasons, these populations are considered here to belong to the nominotypical subspecies.
This subspecies is found from southernmost Ontario and southeastern Michigan south to southern Indiana and central Maryland (Ball 1959: 143-144).
CAN: ON USA: IN, MD, MI, OH, PA, VA, WV
This subspecies is found in the southern part of the Great Plains from northeastern Kansas to central Iowa, south to central Arkansas and eastern Oklahoma (Ball 1959: 142) including southwestern Illinois (Union County, CNC). The record from western Pennsylvania by LeConte (1859d: 53) is probably in error.
USA: AR, IA, IL, KS, MO, OK
Ball (1959: 142) reported the presence of intergrade populations between all three subspecies of Dicaelus sculptilis in Iowa.
This subspecies ranges from western Ontario to Saskatoon in Saskatchewan (Lindroth 1969a: 953), south to southern New Mexico (Ball 1959: 145), southeastern Nebraska (Lancaster County, Foster F. Purrington pers. comm. 2009), and southern Illinois (Ball 1959: 145).
CAN: MB, ON, SK USA: CO, IA, IL, MN, ND, NE, NM, SD, WI, WY
[politus group]
This species is known from northern New Jersey (Smith 1910: 207) to southeastern Iowa, south to southeastern Texas (Ball 1959: 113) and the Florida Panhandle (Peck and Thomas 1998: 21).
USA: AL, AR, DC, FL, GA, IA, IL, IN, KY, LA, MD, MO, MS, NC, NJ, OH, OK, PA, SC, TN, TX, VA
This species ranges from southern Quebec (Larochelle 1975: 80) to northern Iowa, south to northern Alabama (Ball 1959: 111) and South Carolina (Ciegler 2000: 84). The record from “Florida” (Horn 1880c: 52) needs confirmation.
CAN: ON, QC USA: AL, CT, DC, DE, GA, IA, IL, IN, KY, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [FL]
This species ranges from southern Quebec (Larochelle 1975: 80) to southwestern Indiana, south to northern Alabama (Ball 1959: 115), northern Georgia (Fattig 1949: 31), and northwestern South Carolina (Ciegler 2000: 85). The record from South Dakota (Kirk and Balsbaugh 1975: 35) is likely in error.
CAN: ON, QC USA: AL, CT, DC, GA, IN, KY, MA, MD, MI, NC, NJ, NY, OH, PA, SC, TN, VA, VT, WV
Six North American species, one of them extending into northern Mexico, arrayed in three species groups.
[costatus group]
This species is known from eastern Texas south to southern Tamaulipas and Durango (Ball 1959: 167) in Mexico. Two specimens are known from northern Florida and “Virginia” (Ball 1959: 167).
USA: TX [FL, VA] – Mexico
[crenatus group]
This species is known from southeastern coastal Georgia south to southern Florida (Ball 1959: 164).
USA: FL, GA
This species inhabits the southern part of the Great Plains from “Kansas” to southeastern Missouri (Ball 1959: 162), south to northwestern Mississippi (Coahoma County, Drew A. Hildebrandt pers. comm. 2008), “Louisiana” (LeConte 1853c: 389), and southern Texas (Ball 1959: 162). The records from Alabama (Löding 1945: 18), the Florida Panhandle (Harris and Whitcomb 1974: 99), Georgia (Fattig 1949: 31), and North Carolina (Brimley 1938: 122) need confirmation.
USA: AR, KS, LA, MO, MS, OK, TX [AL, FL, GA, NC]
This species is known only from a few localities in the southern third of Florida, including the Keys (Ball 1959: 165).
USA: FL
This form has been considered as a subspecies of Dicaelus alternans Dejean by Ball (1959: 164) but I believe it is more appropriately treated as a distinct species.
[purpuratus group]
The range of this subspecies extends from Massachusetts to western Wisconsin (Ball 1959: 156), south to southeastern Texas (Cameron County, Brian Raber pers. comm. 2010), southern Louisiana (Summers 1874a: 80; Allen 1965: 73), southwestern Alabama (Ball 1959: 156), and central Georgia (Fattig 1949: 30).
CAN: ON USA: AL, CT, DC, GA, IA, IL, IN, KY, LA, MA, MD, MI, MO, MS, NC, NJ, NY, OH, PA, SC, TN, TX, VA, WI, WV
The range of this subspecies extends from “Minnesota” (Ball 1959: 158) to southwestern North Dakota (Tinerella 2003: 636), south to southeastern Arizona, southeastern Texas (Ball 1959: 158), and southeastern Louisiana (Allen 1965: 73), including west-central Illinois (Willand et al. 2011: 273).
USA: AR, AZ, CO, IA, IL, KS, LA, MN, MO, ND, NE, NM, OK, SD, TX
This species has been recorded from “Georgia” (LeConte 1847: 422), the Florida Panhandle and Peninsula including the Keys (Peck and Thomas 1998: 21), and southwestern Alabama (Löding 1945: 18; Ball 1959: 159).
USA: AL, FL, GA
Following Lindroth (1969a: 954), I believe this taxon should be regarded as a valid species, not as a subspecies of Dicaelus purpuratus Bonelli.
Five species in North America (three species) and Mexico (four species, two of them endemic).
This species is confined to the Huachuca and Chiricahua Mountains of southeastern Arizona [see Ball 1992a: Fig. 17].
USA: AZ
This subspecies ranges from southeastern Alberta to southwestern South Dakota, south to southeastern Texas, Chihuahua in Mexico, and southeastern Arizona [see Ball 1992a: Fig. 18].
CAN: AB USA: AZ, CO, KS, MT, NE, NM, OK, SD, TX, UT – Mexico
The subspecies Dicaelus laevipennis flohri Bates is endemic to Mexico ranging from the state of Durango to the Mexico City area (Ball 1992a: 366).
This species is known only from the Chiricahua Mountains in southern Arizona and the Sierra Madre Occidental in Chihuahua and Sonora [see Ball 1992a: Fig. 17].
USA: AZ – Mexico
Worldwide, with about 100 species in the Nearctic (14 species of Badister), Neotropical (five species, one of them belonging to the Chilean genus Eutogeneius Solier), Australian (11 species), Oriental (four species), Palaearctic (68 species), and Afrotropical (three species of Badister) Regions. Approximately 82% of the species are found in the Northern Hemisphere.
Several specimens of Licinus silphoides (Rossi) were taken alive in Massachusetts as noted by LeConte (1873b: 324). It is likely that a population persisted for a while but since no specimen has been caught in North America in the XX Century, the species is certainly not established on this continent.
Forty-eight species in the boreal, temperate, and tropical areas in the Nearctic (14 species), Neotropical (five species in Mexico, the West Indies, and Peru), Australian (two species), Oriental (one species), Palaearctic (27 species), and Afrotropical (three species) Regions. The species are arrayed in three subgenera: Badister (23 species), Baudia (22 species), and Trimorphus Stephens (two Palaearctic species).
Ball (1959) revised the North American species. No new North American species have been described since. Lindroth’s (1969a) key encompasses all North American species but Badister submarinus.
Twenty-four species in North America (eight species, one of them extending into the Bahamas), Mexico (three species, one of them endemic), Peru (one species), and the Palaearctic Region (14 species).
Erwin and Ball (2011: 407-409) published a key to the Western Hemisphere species.
[elegans group]
This rarely collected species ranges from “Illinois” to eastern Kansas (Ball 1959: 206), south to Coahuila and Tamaulipas (Ball 1992a: 375) in northern Mexico, southeastern Louisiana (East Baton Rouge Parish, Igor M. Sokolov pers. comm. 2009), and western Alabama (Tuscaloosa County, UASM). The records from southwestern New Jersey (Smith 1910: 207), “Florida” (Leng 1915: 580), “Delaware” (Houghton 1905: 211), and “Colorado” (Csiki 1931: 903) need confirmation.
USA: AL, AR, IL, KS, LA, MO, OK, TX [CO, DE, FL, NJ] – Mexico
This subspecies ranges from south-central New York (Ball 1959: 213) to southeastern Nebraska (Lancaster County, UASM), including southern Wisconsin (Rauterberg 1885: 17; Purrington et al. 2002: 201), south to southern Texas (Gonzales County, CMNH; Brazoria County, Robert L. Davidson pers. comm. 2008) and southern Florida (Peck and Thomas 1998: 21); also recorded from the Bahamas (Darlington 1953: 8).
USA: AR, DC, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NE, NY, OH, PA, TN, TX, WI – Bahamas.
Ball (1959) recognized three subspecies within this taxon, two of them occurring north of Mexico. Because the differences between the two forms are largely based on color and that intermediate specimens are known from Louisiana (Ball 1959: 210), I prefer not to recognize the color forms as distinct subspecies. Badister flavipes mexicanus Van Dyke is endemic to Mexico.
This rarely collected species ranges from southwestern New Jersey (Smith 1910: 207) to central Kansas (Knaus 1898: 19), south to east-central Texas (Riley 2011), central Louisiana (Concordia Parish, CNC), and northern Florida (Peck and Thomas 1998: 21).
USA: AL, AR, DC, FL, GA, IA, IN, KS, KY, LA, MO, MS, NJ, OH, OK, PA, SC, VA, TX
[notatus group]
This species is found from southwestern New Brunswick (Webster and Bousquet 2008: 20) to eastern South Dakota (Kirk and Balsbaugh 1975: 35; French et al. 2004: 557), south to east-central Texas (Brazos County, CNC; Riley 2011), northern Georgia (Ball 1959: 216-217), and northwestern South Carolina (Ciegler 2000: 85).
CAN: NB, ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
[pulchellus group]
This species ranges from southwestern British Columbia, including Vancouver Island (Lindroth 1969a: 962), south to southern California (Ball 1959: 200-201). One old specimen labeled “Nev.” is known (Ball 1959: 200).
CAN: BC (VCI) USA: CA, OR, WA [NV]
This species ranges from Cape Breton Island to southwestern British Columbia, north to the Great Slave Lake in Northwest Territories (Lindroth 1969a: 959), south to south-central California (Ball 1992a: 375), northeastern Kansas (Popenoe 1878: 78, as Badister pulchellus), and northeastern Georgia (Fattig 1949: 31, as Badister pulchellus). Two specimens simply labeled “Tex” (MCZ) are known (Ball 1959: 196).
CAN: AB, BC, MB, NB, NS (CBI), NT, ON, PE, QC, SK USA: CA, CO, CT, DC, GA, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WV, WY [TX]
Badister neopulchellus Lindroth. This nicely colored licinine was first recognized by Carl Lindroth when he realized that the Badister pulchellus of authors included two species, this one which is transcontinental and relatively commonly collected and Badister pulchellus, a much rarer species restricted to the eastern parts of the continent. With the advent of molecular tools, there is little doubt that several of our carabid species will eventually be showed to be complexes of species that cannot be separately with confidence based on morphology alone.
This species is found from Cape Breton Island (Bousquet 1987a: 132) to south-central British Columbia (Jarrett and Scudder 2001: 380), north to central Northwest Territories (Ball 1992a: 375), south to northeastern New Mexico (Ball 1992a: 375) along the Rocky Mountains, the Guadalupe Mountains in western Texas (Ball 1992a: 375), the upper peninsula of Michigan (LeConte 1878c: 594), southern Ontario (Lindroth 1969a: 961), and southern Maine (Majka et al. 2011: 46). The record from the Pacific Northwest (Hatch 1953: 961) refers to Badister neopulchellus (Lindroth 1969a: 959).
CAN: AB, BC, MB, NB, NS (CBI), NT, ON, QC, SK USA: CO, ME, MI, MN, MT, ND, NM, SD, TX, WI
This rarely collected species is known from southern Ontario (Lindroth 1969a: 959), northern Ohio (Ashtabula County, Harry J. Lee, Jr. pers. comm. 2008), southern Michigan (Clinton County, CMNH), northern and southwestern Indiana (LeConte 1847: 418; Ball 1959: 205), western Illinois, “Tennessee” (Ball 1959: 205), southwestern Alabama (Lindroth 1954b: 153), and Connecticut (New London County, Foster F. Purrington pers. comm. 2009). Most old (pre-1950s) records of this species refer to Badister neopulchellus.
CAN: ON USA: AL, CT, IL, IN, MI, OH, TN
Twenty-two species in the Nearctic (six species), Neotropical (one species shared with North America), Australian (two species, one of them endemic to New Caledonia), Oriental (one species shared with the Australian Region), Palaearctic (11 species), and Afrotropical (three endemic species) Regions.
[micans group]
This transamerican species ranges from Cape Breton Island to Vancouver Island, south to “Oregon” (Lindroth 1969a: 964-965), northern Utah (Ball 1959: 225), east-central South Dakota (Kirk and Balsbaugh 1975: 35), northeastern Illinois (Purrington et al. 2002: 200), and the District of Columbia (Ball 1959: 225). The record from “Nebraska” (Bousquet and Larochelle 1993: 213) needs confirmation.
CAN: AB, BC (VCI), MB, NB, NS (CBI), ON, PE, QC USA: CT, DC, IA, IL, IN, KY, MA, ME, MI, MN, MT, NH, NJ, NY, OH, OR, PA, SD, UT, VT, WA, WI [NE]
This species occurs from Nova Scotia (Lindroth 1954c: 305) to western Minnesota (Gandhi et al. 2005: 929, as Badister ocularis), south to northwestern Tennessee (Lake County, UASM) and northern Georgia (Leng 1910: 73; Fattig 1949: 32). The records from “Florida” (Schaupp 1882b: 7), northeastern Kansas (Popenoe 1878: 78), and southern Texas (Wickham 1897: 105) need confirmation.
CAN: NB, NS, ON, PE, QC USA: CT, GA, IL, IN, MA, ME, MI, MN, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI [FL, KS, TX]
Casey (1920: 206-207), followed by Ball (1959: 227) and Lindroth (1969a: 966), stated that the species described by LeConte in 1844 as Badister micans was “very different” from that described under the same name in 1847 (page 418). However the only difference I can see between the 1844 and 1847 descriptions is the size: 4½ lines (= 9 mm) in the 1844 description and .24 [inch] (= 6 mm) in the 1847 description. In 1844, Georgia is listed as the provenance of the specimen(s) and in 1847 he reported he had a specimen from Georgia and one from Long’s Peak. LeConte (1844: 52) added that Badister micans “differs very much in its general appearance from all the species of this genus” which also led Casey (1920: 206) to believe that the specimen was “probably not a Badister.” However, the other two species of Badister described by LeConte in 1844 were Badister terminalis (= Badister notatus Haldeman) and Badister testaceus (= Philodes alternans LeConte). Both species are indeed quite different from Badister ocularis. My interpretation is that LeConte made a simple mistake in the size of the species in his 1844 description. Blatchley (1928a: 47) also commented on this species.
This species ranges from southwestern Quebec (Bousquet 1987a: 132) to southeastern Minnesota (Ball 1959: 226), north to southern Manitoba (Lindroth 1969a: 967), south to southeastern Texas (Ball 1992a: 377) and northwestern Alabama (Colbert County, CMNH). One specimen labeled from Creston, British Columbia in UBC seen by Lindroth (1969a: 967) is probably mislabeled.
CAN: MB, ON, QC USA: AL, AR, CT, DC, IA, IL, IN, KS, MA, MD, MI, MN, MO, MS, NY, OH, PA, TN, TX, VA, WI, WV
This species is known from Rhode Island (Sikes and Webster 2005: 315) to southern Wisconsin (Messer 2010: 39), including southernmost Ontario (Lindroth 1969a: 966, probably only as strays), south to the Rio Grande in southeastern Texas (Ball 1992a: 376), the Florida Keys, and the Greater Antilles (Ball 1992a: 376-377); it is also known from a single specimen, possibly a stray, collected in southwestern New Brunswick (Webster and Bousquet 2008: 21) and from Quintana Roo in the Yucatán Peninsula (Ball 1992a: 376). The record from “Quebec” (Bousquet and Larochelle 1993: 213) is in error.
CAN: NB, ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, MI, MS, NC, NE, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI, WV – Bahamas, Cayman Islands, Cuba, Dominican Republic, Haiti, Jamaica, Mexico
In the Zoological Record for the year 1880 this species is registered (page 22) under the name “Badister albescens.”
This species is known from three females collected in southern Oregon (Ball 1992a: 377) and north-central California (Motschulsky 1859a: 158).
USA: CA, OR
[transversus group]
This species ranges from New Brunswick (Webster and Bousquet 2008: 20) to southern Manitoba (Lindroth 1969a: 964; Roughley et al. 2010: 230), south to central Nebraska (Loup County, Foster F. Purrington pers. comm. 2010), northern Indiana, and New Jersey (Ball 1959: 219).
CAN: MB, NB, ON, QC USA: CT, IL, IN, MA, ME, MI, MN, NE, NH, NJ, NY, OH, PA, SD, VT, WI
This species has been referred to the subgenus Trimorphus Stephens (type species: Trimorphus scapularis Stephens, 1828 (= Carabus sodalis Duftschmid, 1812)) by Ball (1959) but Lindroth (1969a: 962) concluded that it is probably more closely related to members of Baudia than to those of Trimorphus.
Worldwide, with about 2, 725 species arrayed in four subtribes: Anisodactylina (about 345 species), Harpalina (about 1, 665 species), Pelmatellina (about 90 species), and Stenolophina (about 625 species).
Worldwide, with about 345 species arrayed in 30 genera (Lorenz 2005: 348-353). The North American fauna is represented by 48 species (about 14% of the world fauna).
Ball and Bousquet (2000: 90), following Noonan (1973: 388), recognized two genus-groups among the North American taxa, the Notiobii (an unavailable name; referred to as the “Notiobioid main branch” by Noonan) for the genus Notiobia and Anisodactyli (“Anisodactyloid main branch” of Noonan) for all remaining genera.
About 95 species in the Nearctic (eight species), Neotropical (47 species), Australian (28 species), and Afrotropical (ten species) Regions arrayed in three subgenera: Anisotarsus (53 species), Diatypus Murray (ten Ethiopian species), and Notiobia s.str. (26 Neotropical species).
Fifty-three species in the Nearctic (eight species, four of them endemic), Neotropical (21 species), and Australian (28 species) Regions.
Noonan (1973) revised the North and Middle American species and provided a key for their identification.
[brevicollis group]
This species ranges from southeastern Arizona to southeastern Texas, south to southern Mexico; it is also known from southern Baja California [see Noonan 1973: Fig. 139].
USA: AZ, NM, TX – Mexico
[terminata group]
This species is endemic to the Florida Peninsula (Shpeley 2001: 118).
USA: FL
This form has been listed as a synonym of Notiobia nitidipennis (LeConte) by Noonan (1973: 305) but treated as a distinct species by Shpeley (2001).
This species is known from Connecticut (Krinsky and Oliver 2001: 212), Long Island, New York (Noonan 1973: 307), the coast of southern North Carolina (Brunswick County, Ken Karns pers. comm. 2009), and from “Kansas” and northern Arkansas south to southern Mississippi and eastern Texas [see Noonan 1973: Fig. 134]. The records from New Mexico (Snow 1885: 67; Fall and Cockerell 1907: 162, as Anisodactylus maculicornis) are likely in error.
USA: AR, CT, KS, LA, MS, NC, NY, OK, TX
This species ranges from the mountains in Arizona and northern New Mexico south to northern Panama; also known from the Laguna Mountains in southern Baja California [see Noonan (1973: Fig. 137)].
USA: AZ, NM, TX – Costa Rica, Guatemala, Mexico, Panama
This species ranges from Maine and southern Quebec to southwestern South Dakota (Larsen and Purrington 2010: 571), south to eastern Texas and northeastern Georgia (Shpeley 2001: 119), including “Colorado” (van Emden 1953b: 544) [see Noonan 1973: Fig. 135]. The previous records from Florida (Noonan 1973) refers to Notiobia cephala (Casey) (see Shpeley 2001).
CAN: ON, QC USA: AL, AR, CO, CT, DC, GA, IA, IL, IN, MA, MD, ME, MI, MO, MS, NC, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species ranges from the San Francisco Bay area to southwestern Alabama, including central Missouri (Shockley and Cline 2004: 281), south to central Mexico and southern California [see Noonan 1973: Fig. 145]. The species is adventive on several islands of Hawaii (Liebherr 2009: 403).
USA: AL, AZ, CA, LA, MO, MS, NM, TX – Mexico
This species ranges from southern Quebec (Larochelle 1975: 51, as Anisotarsus piceus) to northeastern South Dakota (Kirk and Balsbaugh 1975: 32, as Anisotarsus piceus), south to eastern Texas and northeastern Florida [see Noonan 1973: Fig. 133].
CAN: ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VT, WI
This widely distributed species ranges from western Newfoundland (CNC) to South Dakota (Kirk and Balsbaugh 1975: 32), south to Panama and southern Florida, west to southeastern Arizona [see Noonan 1973: Fig. 144]; also known from single specimens collected in southeastern Saskatchewan (Ronald R. Hooper pers. comm. 2007) and northern Colorado (Larimer County, William L. Krinsky pers. comm. 2008). The species is also recorded from Bermuda (Hilburn and Gordon 1989: 677).
CAN: NB, NF, NS, ON, PE, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Bermuda, Costa Rica, Guatemala, Honduras, Mexico, Panama
One North American species in the temperate regions.
The species is included in Lindroth’s (1968: 864) monograph.
Lindroth (1968: 864) regarded this taxon as a subgenus of Anisodactylus while Noonan (1973: 347) treated it as a valid genus. As discussed by Bousquet and Tchang (1992: 760), larval characters support Noonan’s view.
This species ranges from Nova Scotia (Lindroth 1954c: 308) and Prince Edward Island (Majka et al. 2008: 132) to southeastern South Dakota (Kirk and Balsbaugh 1975: 31), south to southern Kansas (Snow 1903: 194), southern Arkansas (Kraim 1983: 229), and northwestern North Carolina (Watauga County, USNM). The records from “Colorado” (Snow 1877: 17; Wickham 1902: 243) need confirmation.
CAN: NB, NS, ON, PE, QC USA: AR, CT, DC, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, NC, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WI, WV [CO]
About 50 species in North America (33 species, one of them adventive), Mexico (seven species, two of them endemic), and the Palaearctic Region (17 species) arrayed in nine subgenera: Anadaptus (seven species), Anisodactylus s.str. (22 species), Aplocentrus (two species), Gynandrotarsus (10 species), Hexatrichus Tschitschérine (four west Palaearctic species), Pseudanisodactylus Noonan (three Palaearctic species), Pseudaplocentrus (one species), Pseudodichirus Lutshnik (one west Palaearctic species), and Spongopus (one species). One Palaearctic species (Anisodactylus binotatus) is adventive in New Zealand (Larochelle and Larivière 2005: 34).
Twenty-two species in the Nearctic (14 species, of which two extends into the Baja California Peninsula and one is adventive) and Palaearctic (nine species, including the Himalayas) Regions.
Noonan (1996) revised all the species. One Palaearctic species, Anisodactylus emarginatus Ito, has been described subsequently. Lindroth’s key (1968: 835-838) included all North American species except Anisodactylus pseudagricola described subsequently.
[binotatus group]
This Palaearctic species is adventive in North America where it is known from the Skeena River valley in central British Columbia (Spence and Spence 1988: 158) to northeastern Oregon, east to west-central Montana (Hansen et al. 2009: 353) [see Noonan 1996: Fig. 230]; the species has been recorded also from Sacramento County in California (Clark 1999: 202). The specimen simply labeled from Nevada and the one labeled from Dickenson County in Iowa (Noonan 1996: 113) are likely mislabeled. The first inventoried specimen collected on this continent was found in 1911 at Portland, Oregon (Noonan 1996: 38). The species is also adventive in New Zealand since 1938 (Larochelle and Larivière 2005: 35).
CAN: BC (VCI) USA: CA, MT, OR, WA – Adventive
This species ranges from northern Idaho and northern Washington south to northern Baja California Peninsula. A few specimens are known also from central Arizona and southern New Mexico [see Noonan 1996: Fig. 225].
USA: AZ, CA (CHI), ID, NM, NV, OR, WA – Mexico
[californicus group]
This western species ranges from central Alberta to Vancouver Island, south to the Baja California Peninsula, southern Arizona, and northern Colorado [see Noonan 1996: Fig. 240]. Specimens labeled from “New Mexico, ” Dallas (Texas), Jalisco in west-central Mexico, “Nebraska, ” Lake County in Illinois, and “New Jersey” are known.
CAN: AB, BC (VCI) USA: AZ, CA (CHI), CO, ID, MT, NV, OR, UT, WA, WY [IL, NE, NM, TX] – Mexico
This eastern species ranges from New Jersey to northwestern Missouri, north to southernmost Ontario, south to east-central Texas and the Florida Panhandle [see Noonan 1996: Fig. 244]. Three specimens labeled from “Colorado, ” Santa Cruz County in Arizona, and Lane County in Oregon are known.
CAN: ON USA: AL, AR, DE, FL, GA, IL, IN, KY, LA, MA, MI, MO, MS, NC, NJ, OH, OK, PA, SC, TN, TX, VA [AZ, CO]
This species occurs from central British Columbia south to the Mexican border in California and the eastern edge of the Rocky Mountains in northern Colorado [see Noonan 1996: Fig. 244]. Two old specimens labeled from “Arizona” and “Texas” are known.
CAN: BC (VCI) USA: CA (CHI), CO, ID, MT, NV, OR, UT, WA [AZ, TX]
[carbonarius group]
This species is found from southern Quebec to northeastern South Dakota (Kirk and Balsbaugh 1975: 31), south to northern Colorado (Miller and Peairs 2008: 34), northeastern Texas, and central Florida [see Noonan 1996: Fig. 247]; the species has been collected at three sites in Washington which suggest that it has been introduced into the Pacific northwest.
CAN: ON, QC USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, MA, MD, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV
This species was included in the subgenus Gynandrotarsus by Lindroth (1968: 848) but in the nominotypical subgenus by Noonan (1973: 351).
This species occurs from Nova Scotia to southwestern British Columbia, south to southern California, southern Arizona, the Texas Panhandle (Michels et al. 2010: 743), east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009), central Mississippi, and west-central South Carolina (Ciegler 2000: 90) [see Noonan 1996: Fig. 248].
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: AL, AR, AZ, CA, CT, CO, DE, GA, IA, ID, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SC, SD, TX, UT, VA, VT, WA, WI, WV
This species ranges from Long Island, New York, south to northern Florida and southeastern Mississippi [see Noonan 1996: Fig. 246]. One specimen labeled from “Massachusetts” is known.
USA: AL, FL, GA, MS, NY, NC [MA]
This species is found from Newfoundland to western Ontario, south to northwestern Arkansas, southern Mississippi, and northern Georgia, west to eastern Kansas and Nebraska [see Noonan 1996: Fig. 245]. Seven specimens labeled from “Texas, ” “South Dakota, ” “California, ” southwestern Saskatchewan, and southeastern Alberta are also known (Noonan 1996: 135-138). Several specimens were recently caught in the southern Similkameen Valley in British Columbia (Smith et al. 2004: 96) suggesting that the range of the species extends farther west or that the species was introduced in the area; a misidentification is also possible.
CAN: NB, NF, NS (CBI), ON, PE, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV [AB, BC, CA, SD, SK, TX]
[melanopus group]
This species ranges from Maine and southern Quebec to southeastern Minnesota, south to northern Arkansas (Kraim 1983: 176), central Alabama, and northwestern South Carolina (Ciegler 2000: 89) [see Noonan 1996: Fig. 235]. The record from “Colorado” (Leng 1920: 72) is probably in error.
CAN: ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, NE, NH, NJ, NY, OH, PA, RI, SC, TN, VA, WI, WV
This species ranges from Massachusetts to southeastern Minnesota (Gandhi et al. 2005: 930), south to southern Missouri, “Mississippi” (Drew A. Hildebrandt pers. comm. 2007), and central South Carolina (Ciegler 2000: 90) [see Noonan 1996: Fig. 237]. There are two specimens from more western sites: one from western Nebraska, the other one from Utah. Whether these specimens are strays or represent elements of permanent populations remain to be seen. The record from South Dakota (Kirk and Balsbaugh 1975: 31) needs confirmation.
CAN: ON USA: CT, DC, DE, GA, IA, IL, IN, MA, MD, MI, MN, MO, MS, NC, NJ, NY, OH, PA, RI, SC, VA, VT, WI, WV [NE, SD, UT]
This species, known only from relatively few specimens, ranges from New Hampshire to eastern Michigan, south to northern Ohio (Ashtabula and Huron Counties, Harry J. Lee, Jr. pers. comm. 2008) and southern Pennsylvania (Somerset County, Robert L. Davidson pers. comm. 2012) [see Noonan 1996: Fig. 236]. One specimen collected in central Missouri (Boone County, CMNH) and identified by Gerald R. Noonan in 1994 is known (Robert L. Davidson pers. comm. 2012).
CAN: ON USA: CT, MA, MI, MO, NH, NY, OH, PA
[nigrita group]
This species has a disjunct distribution. In the east, the species is found from Cape Breton Island to southeastern Manitoba south to southern Indiana, southern Pennsylvania, and New Jersey; in the west, the species ranges from southern British Columbia south to southwestern California, east to northern Idaho [see Noonan 1996: Fig. 238].
CAN: BC (VCI), MB, NB, NS (CBI), ON, PE, QC USA: CA, CT, ID, IL, IN, MA, MD, ME, MI, MN, ND, NH, NJ, NY, OH, OR, PA, RI, SD, VT, WA, WI
This species ranges from Nova Scotia to southern British Columbia, south to southern Washington, Colorado along the Rocky Mountains, Nebraska, and eastern Tennessee and western North Carolina along the Appalachian Mountains [see Noonan 1996: Fig. 239]. A few specimens simply labeled from California, Kansas, and Mississippi are known. The record from southwestern Alabama (Löding 1945: 25) is probably in error.
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: CO, CT, DC, IA, IL, IN, KY, MA, MD, ME, MI, MN, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SD, TN, VA, VT, WA, WI, WV [CA, KS, MS]
Ten species in North America (nine species) and Mexico (four species, one of them endemic, Anisodactylus darlingtoni Noonan).
Noonan (1973) revised all species.
[dulcicollis group]
This species ranges from extreme southern Ontario (Bousquet 1987a: 130) to eastern Nebraska, south to southeastern Texas and west-central Georgia [see Noonan 1973: Fig. 165]. Three specimens labeled from “Colorado, ” Arizona (Huachuca Mountains), and California (Los Angeles County) are known. The record from New Jersey (Smith 1890: 98) needs confirmation.
CAN: ON USA: AL, AR, GA, IA, IL, IN, KS, KY, LA, MO, MS, NC, NE, OH, OK, SC, TN, TX, VA [AZ, CA, CO, NJ]
This species ranges from northeastern Nebraska to eastern Virginia (Hoffman et al. 2006: 26), south to northern Mississippi (Lafayette and Panola Counties, Paul K. Lago pers. comm. 2009), northwestern Louisiana, and northeastern Texas (Noonan 1973: 361, Fig. 159). The records from “North Carolina” (Leng 1920: 72), South Carolina (Kirk 1969: 13; Kirk 1970: 14; Ciegler 2000: 90), Georgia (Fattig 1949: 50), “Florida” (Noonan 1973: 361), and southwestern Louisiana (Hine 1906: 77) need confirmation.
USA: AR, IL, KS, LA, MO, MS, NE, OK, TN, TX, VA [FL, GA, NC, SC]
This species ranges from northern Kansas to northeastern Kentucky (Fayette County, Robert L. Davidson pers. comm. 2012) south to western Alabama and Nuevo León in northeastern Mexico [see Noonan 1973: Fig. 158]. The western limit of the species range is along the Rio Grande in western Texas, although it has been recorded from one locality in southwestern Arizona (Snow 1907: 142). The species is also known from south-central Wisconsin (Sauk County, Robert L. Davidson pers. comm. 2012). The records from South Carolina (Kirk 1969: 13; Kirk 1970: 14; Ciegler 2000: 90) need confirmation.
USA: AL, AR, KS, KY, LA, MO, MS, NM, OK, TN, TX, WI [AZ, SC] – Mexico
This species ranges from southeastern Louisiana to southeastern Arizona, south to northern Mexico [see Noonan 1973: Fig. 164].
USA: AZ, LA, TX – Mexico
[rusticus group]
This species ranges from Tulare County in east-central California to southwestern Texas, south to the Federal District area of Mexico [see Noonan 1973: Fig. 160].
USA: AZ, CA, NM, TX – Mexico
This species ranges from Long Island, New York, to northeastern Oklahoma (Foster F. Purrington pers. comm. 2010), south to east-central Texas (Riley 2011) and southern Florida [see Noonan 1973: Fig. 166].
USA: AL, AR, DC, FL, GA, IL, KY, LA, MO, MS, NC, NY, OH, OK, SC, TN, TX, VA
This eastern species ranges from southern Quebec to west-central Minnesota, north to southwestern Manitoba (Lindroth 1968: 846), south to southeastern Texas and the Florida Keys [see Noonan 1973: Fig. 161]. One specimen labeled from Anaheim, California is known. The records from “Arizona” and “North Dakota” (Bousquet and Larochelle 1993: 217) need confirmation.
CAN: MB, ON, QC USA: AL, AR, CO, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI [AZ, CA, ND]
This species ranges from Long Island, New York, to central South Dakota (Kirk and Balsbaugh 1975: 31), including southern Ontario, south to northeastern Texas, northeastern Georgia (House and All 1981: 195; Morrill 1992: 181), and west-central South Carolina (Ciegler 2000: 90) [see Noonan 1973: Fig. 163].
CAN: ON USA: AR, GA, IA, IL, IN, KS, LA, MD, MI, MN, MO, MS, NE, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV
This species ranges from Prince Edward Island to Yellowstone National Park in Wyoming, north to southern Manitoba (Lindroth 1968: 845), south to northern Colorado (Miller and Peairs 2008: 34), eastern Texas, and northern Florida [see Noonan 1973: Fig. 162]. The records from “Arizona” (Leng 1920: 72) and New Mexico (Fall and Cockerell 1907: 162) need confirmation.
CAN: MB, NB, NS, ON, PE, QC USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV, WY [AZ, NM]
Seven species in North America (six species) and Mexico (Anadaptus rotundangulus Bates).
Noonan (2001) revised the species and provided a key for their identification.
This western species is found from southeastern British Columbia south to the Mexican border in California, southwestern Utah, and central Colorado [see Noonan 2001: Fig. 51].
CAN: BC USA: CA, CO, ID, MT, NV, OR, UT, WA
This species ranges from southern Nova Scotia to eastern Montana, north to southern Manitoba (Lindroth 1968: 839) and west-central Saskatchewan, south to southeastern Texas and northern Florida [see Noonan 2001: Fig. 49]. The species is also, quite unexpectedly, found in western Oregon, some 1200 km from the western edge of the species main distribution area (Noonan 2001: 313).
CAN: MB, NB, NS, ON, QC, SK USA: CT, DC, FL, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species ranges from southeastern Manitoba (CNC) to northwestern Washington, south to the Los Angeles area in California and, along the Rocky Mountains, to Arizona and northern New Mexico [see Noonan 2001: Fig. 44].
CAN: AB, MB, SK USA: AZ, CA, CO, ID, NM, NV, OR, UT, WA
This species occurs from northern Idaho and eastern Washington, south to central California [see Noonan 2001: Fig. 49]. The records from New Mexico (LeConte, 1859c: 2; Fall and Cockerell 1907: 162) and “Arizona” (Leng 1920: 73) need confirmation. One specimen labeled “V[ancouver] I[sland]” is known (Lindroth 1968: 842, as Anisodactylus viridescens).
USA: CA, ID, OR, WA [AZ, BC, NM]
This species ranges from southwestern British Columbia to northwestern Wyoming (Park County, Ken Karns pers. comm. 2009), south to northeastern Nevada and the Mexican border in California [see Noonan 2001: Fig. 56].
CAN: BC USA: CA, ID, NV, OR, WA, WY
This species ranges from the Sept-Iles region in easternmost Quebec to Vancouver Island, south to the Sierra Nevada in east-central California, southern Colorado in the Rocky Mountains, Oklahoma, southern Louisiana, and northern Florida; seemingly isolated in northwestern British Columbia [see Noonan 2001: Fig. 50].
CAN: AB, BC (VCI), MB, NB, NS, ON, PE, QC, SK USA: AL, AR, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NY, OH, OK, OR, PA, RI, SC, SD, TN, UT, VA, VT, WA, WI, WV, WY
One North American species in the temperate regions.
Lindroth (1968: 863-864) covered the species.
Lindroth (1968: 862) and Noonan (1973: 374) treated this taxon as a subgenus of Anisodactylus. As discussed by Bousquet and Tchang (1992: 762), larval characters suggest that Spongopus is probably not closely related to Anisodactylus and thus may be more appropriately treated as a distinct genus as done by Ball (1960b: 144).
This species ranges from New Brunswick (Webster and Bousquet 2008: 19) to southern Saskatchewan (Roche Percee, CNC), south to east-central Texas (Riley 2011), northwestern Louisiana (Natchitoches Parish, Igor M. Sokolov pers. comm. 2009), northern Georgia (Fattig 1949: 52), and northwestern South Carolina (Ciegler 2000: 91).
CAN: MB, NB, ON, QC, SK USA: AR, CT, DC, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WI
Two North American species in the temperate regions.
Both species are included in Lindroth’s (1968: 857, 860) monograph.
This species is found west of the Rocky Mountains, from south-central British Columbia (Lindroth 1968: 857) south to southern California (Fall 1901a: 51; Moore 1937: 13).
CAN: BC USA: CA, OR, WA
This species ranges from “Massachusetts” (LeConte, 1847: 386, as Anisodactylus obscurus) to central Kansas (Casey 1924: 125, as Anisodactylus viridans), including central Iowa (Larsen and Purrington 2010: 570), south to northern Texas (Tarrant and Dallas Counties, CNC, UASM), northeastern Louisiana (Allen 1965: 70), southwestern Alabama (Löding 1945: 25), and northwestern South Carolina (Ciegler 2000: 89).
USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MO, MS, NC, NJ, NY, OH, OK, PA, SC, TN, TX, VA
One eastern North American species in the temperate and subtropical regions.
The species is covered in Lindroth’s (1968: 860) monograph.
This species occurs from southeastern New York (Notman 1928: 248) to eastern South Dakota (Kirk and Balsbaugh 1975: 31), including southernmost Ontario (Bousquet 1987a: 131), south to east-central Texas (Riley 2011; Horn 1880d: 175) and southern Florida (Peck and Thomas 1998: 21).
CAN: ON USA: AL, AR, DC, FL, GA, IA, IL, KS, KY, LA, MI, MN, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, SD, TX, VA, WI
One North American species in the temperate regions.
The species is covered in Lindroth’s (1968: 832-833) monograph.
This taxon has always been treated as a valid genus. In a phylogenetic analysis based on characters of the adult conducted by Noonan (1973: Fig. 241), this taxon nested within the genus Anisodactylus. Bousquet and Tchang’s (1992: 769) phylogenetic analysis based on larval characters suggested that Geopinus may be the sister-group to Gynandrotarsus of the genus Anisodactylus. Based on these two studies, Anisodactylus as presently recognized is probably paraphyletic. However, I prefer to leave Geopinus, with its highly distinctive species, as a distinct genus until more conclusive evidence is found.
This widely distributed species ranges from Maine (Robert E. Nelson pers. comm. 1989) and southern Quebec (Larochelle 1975: 85) to southwestern Idaho and northwestern Nevada, north to the southern parts of the Prairie Provinces, south to northern Arizona (Johnson and Clark 1989: 443; Fig. 1), southern Texas (Johnson 1978: 67), and southern Georgia (Fattig 1949: 46).
CAN: AB, MB, ON, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WY
Geopinus incrassatus (Dejean). This species is a good illustration of the fact that overall morphological similarity is not always a good indicator of close relationship. It was described by Dejean in the genus Daptus which currently includes five species in the Palaearctic Region. John Lawrence LeConte recognized its distinctness and proposed a new genus for it but still most taxonomists interested in harpalines thought it was closely related to daptines. However, several small structural details of the adults and larvae suggest that the species is simply an aberrant member of the genus Anisodactylus and is not closely related to daptines.
Two North American species arrayed in two subgenera.
Lindroth (1968: 860-861) covered both species in his treatment of the genus Anisodactylus.
Lindroth (1968: 860) regarded this taxon as a subgenus of Anisodactylus while Noonan (1973: 347) treated it as a valid genus. Larval characters support Noonan’s view (Bousquet and Tchang 1992: Fig. 42).
One North American species in the temperate regions.
This species ranges from New Brunswick (Webster and Bousquet 2008: 19) to western South Dakota (Kirk and Balsbaugh 1975: 32), north to southern Saskatchewan (Ronald R. Hooper pers. comm. 1990), south to east-central Texas (Riley 2011), south-central Louisiana (Evangeline Parish, MCZ) and the Florida Panhandle (Liberty County, CNC).
CAN: MB, NB, ON, QC, SK USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
One North American species in the temperate regions.
This species ranges from southern Quebec (Larochelle 1975: 49) to southeastern South Dakota (Kirk and Balsbaugh 1975: 32), south to east-central Texas (Riley 2011), northern Arkansas (Kraim 1983: 221), and west-central South Carolina (Kirk 1970: 14; Ciegler 2000: 92).
CAN: ON, QC USA: AR, CT, DC, DE, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
Five North American species, of which two extend into northern Baja California and Guadalupe Island.
Noonan (1968) revised the species and provided a key for their identification.
This species is known from east-central Washington (Casey 1924: 134, as Dicheirus gracilis) south to Eldorado County in eastern California [see Noonan 1968: Fig. 16].
USA: CA, OR, WA
This subspecies is known from a few localities in southernmost California and northern Baja California [see Noonan 1968: Fig. 19].
USA: CA – Mexico
This subspecies is found over most of California, south to the Los Angeles area and San Clemente Island [see Noonan 1968: Fig. 19].
USA: CA (CHI)
This species is known from northeastern Oregon south to Los Angeles County in southern California [see Noonan 1968: Fig. 15].
USA: CA, OR
This species ranges from Vancouver Island to northwestern Montana (Russell 1968: 67), south to northwestern Utah and the Mexican border in California [see Noonan 1968: Fig. 18]; also recorded from Guadalupe Island in the Pacific (Noonan 1968: 289).
CAN: BC (VCI) USA: AZ, CA (CHI), ID, MT, OR, UT, WA – Mexico
This species is known only from a few localities in Kern and Tulare Counties in southern California [see Noonan 1968: Fig. 17].
USA: CA
New World, with about 90 species arrayed in eight genera: Hakaharpalus Larochelle and Larivière (five New Zealand species), Kupeharpalus Larochelle and Larivière (three New Zealand species), Lecanomerus Chaudoir (35 Australian species), Nemaglossa Solier (one Chilean species), Notospeophonus Moore (three species in Australia), Pelmatellus (28 species), Syllectus Bates (three New Zealand species), and Trachysarus Reed (eight species from the Juan Fernández).
In the phylogenetic analysis of the tribe Harpalini using molecular sequence data conducted by Martínez-Navarro et al. (2005) members of Pelmatellina clustered within those of Stenolophina.
Twenty-eight species in the Nearctic (two species) and Neotropical (28 species) Regions arrayed in three subgenera: Pelmatellopsis Perrault (17 South American species), Pelmatellus s.str. (10 species), and Thenarellus Bates (one Middle American species).
Ten North and Middle American species.
Goulet (1974b) revised the species and provided a key for their identification.
The range of this species extends from southeastern Arizona and southwestern New Mexico south to Guatemala [see Goulet 1974b: Fig. 32].
USA: AZ, NM – Guatemala, Mexico
This subspecies is known only from the holotype collected in southern Arizona.
USA: AZ
The subspecies Pelmatellus stenolophoides stenolophoides Bates occurs from central Mexico to Guatemala. The record from “Arizona” (Bousquet and Larochelle 1993: 213) for the nominotypical subspecies is an error and refers to the subspecies Pelmatellus stenolophoides parallelus.
Worldwide, with about 625 species (Lorenz 2005: 353-362) arrayed in approximately 35 genera. The Northern Hemisphere is represented by about 325 species (roughly 52% of the world fauna) and North America alone by 102 species (16.3%).
Ball and Bousquet (2000: 90) recognized two genus-groups among the North American taxa, the Polpochili for the genus Polpochila and Stenolophi for all remaining genera.
About 170 species (Lorenz 2005: 353-355, excluding Agonoleptus) arrayed in four subgenera: Agonoderus (seven species), Astenolophus Habu (11 Palaearctic species in Asia, two of them extending into the Oriental Region), Egadroma Motschulsky (about 90 species in Africa, Asia, and the Australian Region), and Stenolophus s.str. (about 60 species). The North American fauna has 22 species (roughly 13% of the world fauna). Egadroma is often listed as a separate genus.
About 60 species in the Nearctic (15 species), Neotropical (ten species), Oriental (11 species), and Palaearctic (about 25 species) Regions.
Lindroth (1968: 905-907) treated all currently recognized valid species in his key except for Stenolophus splendidulus. Bousquet (1997b: 335) commented on Stenolophus splendidulus and illustrated the median lobe.
This species ranges from Vancouver Island (Lindroth 1968: 911) to western Montana (Russell 1968: 67), south to southern Colorado (Elias 1987: 634) and southern California (Fall 1901a: 50; Andrews et al. 1979: 28).
CAN: BC (VCI) USA: CA, CO, ID, MT, NV, OR, UT, WA
The range of this species extends from northern New Hampshire (Reeves et al. 1983: 459) to eastern South Dakota (Kirk and Balsbaugh 1975: 33), including southernmost Ontario (Lindroth 1968: 908), south to southern Texas (Johnson 1978: 67) and southern Florida (Peck and Thomas 1998: 22). The record from Nova Scotia (Lindroth 1954c: 309) is considered doubtful by Majka et al. (2007: 10).
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KY, LA, MA, MD, MI, MO, MS, NC, NH, NJ, NY, OK, RI, SC, SD, TN, TX, VA, WI [NS]
This species ranges from southeastern California (Imperial County, MCZ, USNM) to westernmost Texas (Casey 1914: 279, as Stenolophus semitinctus), south at least to the state of Durango in Mexico. The record from “Colorado” (Csiki 1932a: 1260) needs confirmation.
USA: AZ, CA, NM, TX [CO] – Mexico
This species ranges from northeastern Ohio (Purrington et al. 1999: 47) to southern Wisconsin (Rauterberg 1885: 21; Messer 2010: 40), north to Charity Island in the Michigan waters of Lake Huron (Andrews 1916: 79), south to southern Texas (Johnson 1978: 67), northern Georgia (Fattig 1949: 54), and northwestern South Carolina (Ciegler 2000: 94). The record from New Jersey (Smith 1910: 216) needs confirmation.
USA: AL, AR, GA, IL, IN, KS, KY, LA, MI, MO, MS, NC, OH, OK, SC, TN, TX, WI [NJ]
This species is known from northern California (Shasta County, USNM) to southern California (Fall 1901a: 50; Dajoz 2007: 20) and “Arizona” (Leng 1920: 74). The record from “Oregon” (Schaupp 1883a: 14) is probably in error (Hatch 1953: 184).
USA: AZ, CA
The range of this species extends from Newfoundland (Lindroth 1955a: 149) to Vancouver Island (Lindroth 1968: 910), south to the Sierra Nevada in California (Dajoz 2007: 16), northern Colorado (Haubold 1951: 706; Armin 1963: 122), and eastern Georgia (Fattig 1949: 54). The record from southern Louisiana (Summers 1874a: 81) needs confirmation.
CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AR, CA, CO, CT, DC, GA, IA, ID, IL, IN, MA, MD, ME, MI, MN, MT, NC, ND, NH, NJ, NY, OH, OR, PA, RI, SC, SD, VA, VT, WA, WI, WY [LA]
This species occurs from southwestern New Brunswick (Webster and Bousquet 2008: 19) to eastern South Dakota (Kirk and Balsbaugh 1975: 33), south to northeastern Kansas (Popenoe 1878: 79) and south-central Maryland (Prince Georges County, USNM). The record from southern Colorado (Elias 1987: 634) needs confirmation.
CAN: NB, ON, QC USA: CT, IL, IN, KS, MA, MD, ME, MI, NE, NH, NJ, NY, OH, PA, RI, SD, VT, WV [CO]
This species ranges from southern Nova Scotia (Christopher G. Majka pers. comm. 2007) to southwestern Michigan (Allegan County, CMNH), south to southeastern Mississippi (Stone County, Drew A. Hildebrandt pers. comm. 2008), northern Georgia (Fattig 1949: 54), and eastern South Carolina (Ciegler 2000: 94).
CAN: NS, ON, QC USA: CT, DC, GA, KY, MA, MD, ME, MI, MS, NC, NH, NJ, NY, OH, PA, RI, SC, VA, VT, WV
This species is found from Vancouver Island (Lindroth 1968: 911) to northern Idaho (Hatch 1953: 183), south to “Arizona” (Casey 1914: 273, as Stenolophus abstinens) and southern California (Casey 1914: 274, as Stenolophus remissus). The record from “Montana” (Bousquet and Larochelle 1993: 220) needs confirmation.
CAN: BC (VCI) USA: AZ, CA, ID, OR, UT, WA [MT]
This species ranges from Vancouver Island (Lindroth 1968: 909) to northwestern Montana (Russell 1968: 67), south to central Utah (Casey 1914: 276) and southern California (Fall 1901a: 50; Moore 1937: 14). The species is adventive on Oahu, Hawaii (Liebherr 2009: 403).
CAN: BC (VCI) USA: CA (CHI), ID, MT, NV, OR, UT, WA
This species is restricted to a small area from southern Quebec and the Ontario Peninsula (Bousquet 1987a: 131) south to central Pennsylvania (Clinton County, CMNH) and “Rhode Island” (Sikes 2003: 8).
CAN: ON, QC USA: CT, KY, MA, NH, NY, PA, RI
This species ranges from Nova Scotia (Lindroth 1954c: 309) to southeastern Saskatchewan (Ronald R. Hooper pers. comm. 2007), south to southern Texas (Johnson 1978: 67) and southern Florida (Peck and Thomas 1998: 22), west along the southwest to southern California (Horn 1894: 312; Andrews et al. 1979: 28); also found in southern Baja California (Horn 1894: 312), the Bahamas (Darlington 1953: 10), Cuba (Darlington 1934: 112), Jamaica (Darlington 1941a: 14), Dominican Republic (Robert L. Davidson pers. comm. 2012) and Puerto Rico (Wolcott 1936: 191).
CAN: MB, NB, NS (CBI), ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV – Bahamas, Cuba, Dominican Republic, Jamaica, Mexico, Puerto Rico
The range of this species extends from southern Quebec (Larochelle 1975: 255) to eastern Iowa (Johnson County, USNM), south to southeastern Texas (Aransas County, USNM) and southern Florida (Peck and Thomas 1998: 22); also recorded from Bermuda (Hilburn and Gordon 1989: 677). The records from central Colorado (Wickham 1902: 242) and Tabasco in Mexico (Bates 1891a: 246) are probably in error.
CAN: ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WV – Bermuda
Besides the three original specimens, I assign to this species a series of specimens collected in southernmost Ontario (Rondeau Provincial Park, CNC).
CAN: ON
This species is known from New Jersey (Smith 1910: 215; Lindroth 1968: 905) to the Florida Keys (Peck and Thomas 1998: 22), west along the Gulf Plain to southern Texas (Johnson 1978: 67), north to western Kansas (Scott County, CMNH).
USA: AL, AR, DC, FL, GA, KS, LA, MD, MS, NJ, OK, SC, TX, VA
Seven species in North America, of which two extend into northern Mexico, Bermuda, the Bahamas, and Cuba.
The subgenus has been reviewed by Heading (1964) but his work remains unpublished. Lindroth (1968: 916-920, as comma group) covered four species. A revision of the group would be useful.
This species is known from southern Oklahoma (Marshall County, CMNH) to eastern Coahuila in Mexico (UASM), east to southeastern Louisiana (Heading 1964: 34).
USA: LA, OK, TX – Mexico
This species ranges from Newfoundland (Lindroth 1955a: 150) to Vancouver Island (Lindroth 1968: 919), south to southeastern California (Dajoz 2007: 20), southern Arizona, central Texas, northern Alabama, and east-central South Carolina (Ciegler 2000: 93) [see Heading 1964: Fig. 19]. The record from “Mississippi” (Bousquet and Larochelle 1993: 221) needs confirmation.
FRA: PM CAN: AB, BC (VCI), MB, NB, NF, NS, ON, PE, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY [MS]
This species ranges from “New York” (LeConte 1847: 373) and New Jersey to southern Florida including the Keys (Peck and Thomas 1998: 22), west to northern Oklahoma (Alfalfa County, Robert L. Davidson pers. comm. 2008) and southeastern Texas [see Heading 1964: Fig. 18]; also recorded from the Bahamas and Cuba (Darlington 1953: 11). The records from southeastern Minnesota (Gandhi et al. 2005: 931) and southwestern Ohio (Blatchley 1910: 177) need confirmation.
USA: AL, DE, FL, GA, LA, MD, MS, NC, NJ, NY, OK, SC, TX, VA [MN, OH] – Bahamas, Cuba.
The range of this species extends from western Maine (Kennebec and Somerset Counties, Ross T. Bell pers. comm. 2008) to eastern South Dakota (Ellsbury et al. 1998: 621), south to east-central Texas (Riley 2011) and the Florida Panhandle (Peck and Thomas 1998: 22). Also known from northern Idaho (Hatten et al. 2007: 359) and from a single, old (1926) specimen (possibly mislabeled) from western Oregon (Westcott et al. 2006: 9). The records from northern Colorado (Armin 1963: 115) and New Mexico (Fall and Cockerell 1907: 161, Agonoderus pallipes) need confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [CO, NM, OR]
This species is found from Prince Edward Island to southern Alberta (Lindroth 1968: 918), south to “Baja California” (Leng 1920: 74), southeastern California (Andrews et al. 1979: 28), southern Arizona (Heading 1964: Fig. 18), north-central Mexico (Heading 1964: 30), and central Florida (Peck and Thomas 1998: 22); also known from one specimen collected in northern Oregon (Westcott et al. 2006: 9) and reported as “common in light traps” in Bermuda (Hilburn and Gordon 1989: 677).
CAN: AB, MB, ON, PE, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV, WY – Bermuda, Mexico
This species is found throughout California, in southeastern Arizona [see Heading 1964: Fig. 18], and in “Nevada” (LeConte 1869a: 374); one specimen labeled from south-central Washington is also known (Heading 1964: 32).
USA: AZ, CA, NV [WA]
This species is known from central Washington to southwestern California [see Heading 1964: Fig. 19].
USA: CA, OR, WA
Seven North American species in the temperate regions.
There is no key for the identification of all species. Lindroth (1968: 921-924) covered four species, leaving Agonoleptus dolosus and Agonoleptus parviceps.
Adults and particularly larvae (personal observation) are structurally quite different from those of Stenolophus s.str. and Agonoderus and I am unable to find any character states that would conclusively suggest that Agonoleptus is closely related to the other two taxa. For that reason, I consider Agonoleptus as a distinct genus.
[conjunctus group]
This species ranges from Cape Breton Island (Lindroth 1954c: 309) to Vancouver Island (Lindroth 1968: 922), south to the Sierra Nevada in California (Dajoz 2007: 16), at least San Luis Potosí in Mexico (CNC), and central Florida (Peck and Thomas 1998: 22). The record from Guatemala (Bates 1882a: 71) needs confirmation.
CAN: AB, BC (VCI), MB, NB, NS (CBI), ON, PE, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
This species ranges from east-central Ohio (Usis and MacLean 1998: 67) to southeastern Wyoming (Lavigne 1977: 47) and northeastern Colorado (Lavigne 1978: 102), including southernmost Ontario (Lindroth 1968: 924; Bousquet 1987a: 131), south to southeastern Texas (Casey 1914: 282) and northwestern South Carolina (Kirk 1970: 15). The record from “New York” (Bousquet and Larochelle 1993: 222) needs confirmation.
CAN: ON USA: AL, AR, CO, IN, KS, KY, LA, MD, MO, MS, OH, OK, PA, NC, SC, TN, TX, VA, WV, WY [NY]
The range of this species extends from southern Quebec (Larochelle 1975: 109) to eastern Minnesota (Gandhi et al. 2005: 931), south to west-central Mississippi (Washington County, CMNH) and northwestern South Carolina (Kirk 1970: 15). The record from Colorado (Elias 1987: 634) is probably in error.
CAN: ON, QC USA: CT, MA, MD, ME, MI, MN, MS, IA, IL, NC, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV
This species ranges from east-central Vermont to southeastern North Dakota, south to northeastern Kansas, Tennessee, and northeastern Virginia [see Bousquet and Messer 2010: Fig. 3].
USA: DC, IA, IL, IN, KS, KY, MA, MD, MI, MO, ND, NJ, NY, OH, PA, SD, TN, VA, VT, WI
This taxon has been listed in synonymy with Agonoleptus conjunctus (Say) by Lindroth (1968: 921) but considered a valid species by Bousquet and Messer (2010).
[unicolor group]
This species is known only from the type series collected in southwestern California.
USA: CA
I have studied both syntypes, including the male genitalia of one of them, and I have little doubt that they belong to a distinct species, though probably closely related to Agonoleptus unicolor (Dejean) based on Lindroth’s (1968: 924) description of the holotype of Agonoleptus unicolor. The name Stenolophus dolosus is listed as a junior synonym of Stenolophus unicolor (Dejean, 1829) in Lorenz (2005: 354).
This species is known from two localities in eastern Colorado and central New Mexico (Bousquet 1990: 203).
USA: CO, NM
This species is known for sure only from southern California, as far north as the San Francisco Bay (Casey 1914: 280). The records from Colorado (Wickham 1902: 242; Elias 1987: 634) need confirmation; that from the Similkameen Valley in British Columbia (Smith et al. 2004: 96) was based on misidentified specimens of Agonoleptus conjuctus (Say).
USA: CA [CO]
About 130 species (Lorenz 2005: 356-358) arrayed in ten subgenera: Atlantocellus Wrase and Jaeger (five species from the Canary and Madeira Islands), Bradycelloides Habu (one east Palaearctic species), Bradycellus s.str. (45 species), Catharellus (one species), Desbordesius Maindron (one east Palaearctic species), Liocellus (eight species), Lipalocellus (two species), Stenocellus (34 species), Tachycellus Morawitz (27 Palaearctic species from Asia with one species extending into the Oriental Region), and Triliarthrus (six species). The North American fauna is represented by 51 species (roughly 39% of the world fauna).
Noonan (1976: 21) also included Psychristus Andrewes (six species) and Nipponobradycellus Habu (four species) in this genus. In Lorenz (2005: 358), Nipponobradycellus is listed as a subgenus of the genus Psychristus.
Eight species in North America (seven species) and Mexico (one species).
Fall (1905: 175, as Glycerius) published a key to the four North American species then known. Casey (1924) subsequently added three new species. The subgenus is in need of a taxonomic revision.
This species is known only from the type locality in west-central California.
USA: CA
This species is known from the Sierra Nevada and San Bernardino Mountains (Fall 1905: 176) and from San Diego County (Moore 1937: 14) in California.
USA: CA
This species is known only from the type locality in northwestern Nevada.
USA: NV
The range of this species extends from southwestern Washington (Hatch and Kincaid 1958: 6; Pacific County, MCZ) southwards to the Baja California Peninsula (Horn 1894: 312) and Guatemala (Ball and Shpeley 1992a: 58). The record from “British Columbia” (Horn 1894: 312) is likely in error (Lindroth 1968: 883).
USA: AZ, CA (CHI), NM, OR, UT, WA – Guatemala, Mexico
This species is known so far only from southern California (Fall 1905: 176).
USA: CA
This species ranges from the Okanagan Valley in south-central British Columbia (Lindroth 1968: 883) to southern California (Fall 1905: 176; Dajoz 2007: 20).
CAN: BC USA: CA, OR, WA
This species is known only from the type locality in the Sierra Nevada.
USA: CA
About 45 species in the Nearctic (two species, one of them adventive), Neotropical (25 species), and Palaearctic (17 species) Regions.
The two species found in North America have been covered by Lindroth (1968: 883-884, as harpalinus group).
This species is found along the Pacific Coast from northwestern Washington (Nelson 1988b: 56) to southwestern Oregon (Coos County, CNC).
USA: OR, WA
This Palaearctic species is adventive in North America where it is known from the Vancouver area in southwestern British Columbia (Lindroth 1968: 883) south to Oregon (Westcott et al. 2006: 7; Lincoln County, Foster F. Purrington pers. comm. 2009). The first inventoried specimen collected on this continent was found in Vancouver in 1951 (Lindroth 1968: 883).
CAN: BC USA: OR, WA – Adventive
One North American species.
The species was covered in Lindroth’s (1968: 885) treatment of the Canadian Bradycellus.
This species ranges from Newfoundland (Lindroth 1955a: 147) to eastern Alaska (Lindroth 1968: 885), south to northern Oregon (Hatch 1953: 182, as Bradycellus cordicollis), northern Colorado (Larimer County, UASM; Lindroth 1955a: 147), eastern South Dakota (Kirk and Balsbaugh 1975: 32), and northeastern West Virginia (Tucker County, CMNH).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, ID, IL, MA, ME, MI, MN, MT, ND, NH, NY, OR, PA, SD, VT, WA, WI, WV, WY
Thirty-four species in North America (33 species), Mexico (three species, one endemic to Guadalupe Island), and the Bahamas (one species).
Casey (1914: 243-257) published a key to the species then known except Stenocellus nebulosus, Stenocellus nigriceps, and Stenocellus subcordatus. He subsequently described three new species (Casey 1924). Lindroth (1968, as rupestris, nigriceps, and tantillus groups) covered nine species. The subgenus is in need of a revision.
According to Kataev and Matalin (in Kryzhanovskij et al. 1995: 135), the sole syntype (a badly damage ♂) of Stenolophus elongatus Motschulsky, 1860 in ZMMU, reported from the Kuril Islands, is a specimen of this subgenus. Since no species of Stenocellus are known from the Far East, they concluded that the specimen is mislabeled and probably originated from northwestern North America.
[nigriceps group]
This species, as far as known, ranges from southeastern New Hampshire (Rockingham County, Ross T. Bell pers. comm. 2008) to northwestern Minnesota (Gandhi et al. 2005: 930), including southernmost Ontario (Lindroth 1968: 892), south to South Carolina (Charleston County, CNC).
CAN: ON USA: CT, MD, MI, MN, NH, NJ, NY, OH, PA, SC, VA, VT, WV
This species ranges from Newfoundland (Lindroth 1955a: 147-148) to northeastern British Columbia (Lindroth 1968: 892), south to southwestern Oklahoma (Kondratieff et al. 2005: 171), southern Arkansas (Kraim 1983: 277), northeastern Georgia (Fattig 1949: 53), and southeastern South Carolina (Ciegler 2000: 96). The record from southwestern Alabama (Löding 1945: 26) needs confirmation; that from “Utah” (Bousquet and Larochelle 1993: 225) is probably in error.
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AR, CT, GA, IL, MA, ME, MI, MN, NC, ND, NH, NY, OH, OK, PA, SC, SD, TN, VT, WI [AL]
This species ranges from New Brunswick (Lindroth 1968: 891) to North Dakota (Tinerella 2003: 636), north to southeastern Manitoba (Lindroth 1968: 891), south to east-central Arkansas (Kraim 1983: 280), northeastern Mississippi (Snodgrass and Cross 1983: 16), and southeastern South Carolina (Ciegler 2000: 96). The record from “New Mexico” (Fall and Cockerell 1907: 162) is probably in error.
CAN: MB, NB, ON, QC USA: AR, CT, DC, IA, IL, IN, MA, MD, ME, MI, MN, MS, ND, NH, NJ, NY, OH, PA, SC, VA, VT, WI, WV
This species is known only from the type locality.
USA: NJ
The two syntypes are morphologically similar to adults of Bradycellus insulsus Casey and may be conspecific with them.
[rupestris group]
This species ranges from Nova Scotia (Majka et al. 2007: 10) to Vancouver Island (Lindroth 1968: 888), south at least to central California (Clark 1999: 202; Dajoz 2007: 17), northern Sonora in Mexico (Bates 1884: 277), southern Texas (Johnson 1978: 67), southeastern Louisiana (East Baton Rouge Parish, Igor M. Sokolov pers. comm. 2009), Tennessee (Cumberland County, CNC), and southern West Virginia (McDowell County, Ken Karns pers. comm. 2009).
CAN: AB, BC (VCI), MB, NS, ON, QC, SK USA: AZ, CA, CO, IA, ID, IL, KS, LA, MA, ME, MI, MN, MT, ND, NH, NM, NV, OH, OR, SD, TN, TX, UT, VT, WA, WI, WV, WY – Mexico
This species is known only from the type locality along the Pacific Coast of California.
USA: CA
This species is known from northwestern Georgia (Fattig 1949: 53) and “Texas” (Horn 1883a: 52). The record from Baja California (Horn 1894: 312) needs confirmation.
USA: GA, TX
Fall (1905: 171) stated that Bradycellus nebulosus is “closely resembling and probably not distinct from rupestris.”
This species is known from a small area including southern Saskatchewan, southern Alberta (Lindroth 1968: 889), and southwestern North Dakota (Tinerella 2003: 636). The records from “Montana” and “Wyoming” (Bousquet and Larochelle 1993: 225) need confirmation.
CAN: AB, SK USA: ND [MT, WY]
The range of this western species extends from central Alberta to Vancouver Island (Lindroth 1968: 887), south to southern California and northern Sonora (LeConte 1858b: 60). The record from southern Texas (Johnson 1978: 67) needs confirmation.
CAN: AB, BC (VCI) USA: AZ, CA, ID, NV, OR, WA [TX] – Mexico
This species ranges from Cape Breton Island (Lindroth 1954c: 308) to southeastern Wyoming (Lavigne 1977: 44), south to southern New Mexico (Fall and Cockerell 1907: 162), southern Texas (Wickham 1897: 113), southern Louisiana (Summers 1874a: 81; Allen 1965: 72, as Bradycellus debilipes), northern Georgia (Fattig 1949: 53), and eastern South Carolina (Ciegler 2000: 96). One specimen simply labeled from Florida is known (Leng 1915: 599).
CAN: NB, NS (CBI), ON, QC USA: AL, AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV, WY [FL]
[subcordatus group]
This species is known only from the holotype. Casey (1914: 258) assumed that the species was “possibly a native of California” because the holotype was sent to Chaudoir by Motschulsky. Although Motschulsky described several species from the west coast, he never set foot in western United States.
USA: ?CA
[tantillus group]
This species is found from Vancouver Island (Lindroth 1968: 894) to western Montana (Russell 1968: 68), south to west-central Nevada (Lyon County, CNC) and southern California (Fall 1901a: 50).
CAN: BC (VCI) USA: CA (CHI), ID, MT, NV, OR, WA
This species is known from “Texas” (Horn 1894: 312), Arizona (Griffith 1900: 566; Casey 1914: 249), southeastern California (Fall 1901a: 50), and the Baja California Peninsula (Horn 1894: 312).
USA: AZ, CA, TX – Mexico
This species ranges from southwestern Maine (Majka et al. 2011: 46) to eastern South Dakota (Kirk and Balsbaugh 1975: 33), including southern Ontario (Lindroth 1968: 893; Bousquet 1987a: 131), south to southeastern Texas (Fort Bend, Brian Raber pers. comm. 2010), northern Louisiana (Allen 1965: 72), southern Florida (Peck and Thomas 1998: 21), and the Bahamas (Darlington 1953: 10). The record from “Quebec” (Bousquet and Larochelle 1993: 226) was based on a misidentified specimen of Bradycellus nigriceps (CNC); those from northern Colorado (Haubold 1951: 706; Armin 1963: 118) need confirmation.
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WV [CO] – Bahamas
This species is known only from the type locality in northern California.
USA: CA
This species is known only from the type locality in southwestern California.
USA: CA
This species is known only from the type locality.
USA: NC
This species is known only from the type locality in southern Arizona.
USA: AZ
This species is known only from the type locality in northwestern California.
USA: CA
This species is known only from the type locality in north-central California.
USA: CA
This species ranges from southern Montana (Hatch 1933a: 10) and South Dakota (Kirk and Balsbaugh 1975: 33) south to “Texas” (Casey 1914: 257) and southern Arkansas (Kraim 1983: 283). The record from Cuba (Darlington 1934: 110) is doubtful.
USA: AR, KS, MT, SD, TX, WY
This species is known only from the type locality in northwestern California.
USA: CA
This species is known only from the type locality in westernmost Texas.
USA: TX
This species is known only from the type locality in western California.
USA: CA
This species is known only from the type locality in the Sierra Nevada.
USA: CA
This species is known only from the type locality in the Sierra Nevada.
USA: CA
This species is known only from the type locality in north-central Utah.
USA: UT
This species is known from the type series and San Diego County (Moore 1937: 14) in California.
USA: CA
This species is known only from the type locality in southwestern California.
USA: CA
This species is known for sure only from the type series collected in western California; it was also recorded from Humboldt County (Notman 1929b: 222).
USA: CA
This species is known for sure only from the type locality in east-central Texas. The record from South Dakota (Kirk and Balsbaugh 1975: 33) needs confirmation.
USA: TX [SD]
This species is known for sure only from the type series collected on the west coast in central California. Casey (1914: 254) reported it from Saint Helena, Sonoma County, California.
USA: CA
This species is known yet only from the type locality.
USA: FL
Two North American species.
Lindroth (1968: 894-898, as nigrinus group) reviewed the species.
This species extends from Newfoundland (Lindroth 1955a: 146) to southeastern Alaska, including Kodiak Island (Lindroth 1968: 896), south to mountains in southeastern California (Lindroth 1968: 896; San Bernardino County, CNC) and New Mexico (Fall and Cockerell 1907: 162), northwestern Indiana (Blatchley 1910: 193), and southeastern West Virginia (Greenbrier County, CNC). The record from “North Carolina” (Bousquet and Larochelle 1993: 226) needs confirmation.
FRA: PM CAN: AB, BC (QCI, VCI), MB, NB, NF, NS (CBI), ON, PE, QC, SK, YT USA: AK, CA, CO, CT, ID, IL, IN, MA, ME, MI, MN, MT, NH, NM, NY, OH, OR, PA, UT, VA, VT, WA, WI, WV [NC]
This species ranges from Nova Scotia (Majka et al. 2007: 10) to central Minnesota (Gandhi et al. 2005: 930), north to southeastern Manitoba (Roughley et al. 2010: 230), south at least to east-central Missouri (Saint Louis, MCZ), southwestern Illinois (Saint Clair County, MCZ), and southeastern Pennsylvania (Dauphin County, MCZ). The single specimen from McMurray, Alberta (Lindroth 1968: 898) is possibly mislabeled or represents a stray.
CAN: MB, NB, NS, ON, QC USA: IA, IL, MA, ME, MI, MN, MO, NH, NY, PA, VT, WI [AB]
Six North American species in the boreal and temperate regions.
Lindroth (1968: 898-903, as badipennis group) reviewed all species.
The range of this species extends from New Brunswick (Lindroth 1968: 899) to eastern South Dakota (Kirk and Balsbaugh 1975: 32), north to southeastern Manitoba (Lindroth 1968: 899), south to “Texas” (Fall 1905: 172), Tennessee (Sevier County, CNC), and northeastern Georgia (Fattig 1949: 53).
CAN: MB, NB, ON, QC USA: DC, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, NE, NH, NJ, NY, OH, PA, RI, SD, TN, TX, VA, VT, WI, WV
This species is known from southwestern New Brunswick (Webster and Bousquet 2008: 19) to northern Minnesota (Gandhi et al. 2005: 930), including southern Quebec and the Ontario Peninsula (Lindroth 1968: 899), south to northeastern Kansas (Popenoe 1877: 24), Tennessee (Knox County, CNC), northern Georgia (Fattig 1949: 53), and eastern South Carolina (Ciegler 2000: 96). The records from Colorado (Wickham 1902: 242; Armin 1963: 119) are probably in error.
CAN: NB, ON, QC USA: CT, DC, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV
This species is restricted to the Pacific Coast from southern British Columbia, including Vancouver Island (Lindroth 1968: 903), south at least to the San Francisco Bay in California (Fall 1905: 173). The record from northern Colorado (Armin 1963: 119) is likely in error.
CAN: BC (VCI) USA: CA, OR, WA
This species is yet known only from a few localities in Alberta.
CAN: AB
This species ranges from southwestern New Brunswick (Webster and Bousquet 2008: 19) to central Minnesota (Sherburne County, CNC), including southern Quebec and the Ontario Peninsula (Lindroth 1968: 901), south to northeastern West Virginia (Randolph County, CMNH).
CAN: NB, ON, QC USA: CT, MA, ME, MI, MN, NH, NJ, NY, OH, PA, RI, VA, VT, WI, WV
The range of this species extends from northeastern Newfoundland (Lindroth 1955a: 146, as Triliarthrus protractus) to the Rocky Mountains in Alberta (Lindroth 1968: 902), south to Minnesota (Lindroth 1955a: 146; Epstein and Kulman 1990: 214), Tennessee (Lindroth 1968: 901), and southwestern North Carolina (Jackson County, CNC).
CAN: AB, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CT, DC, IL, MA, ME, MI, MN, NC, NH, NJ, NY, OH, PA, TN, VA, VT, WI, WV
One North American species in the temperate regions.
The species was treated by Lindroth (1968: 877-878).
This species ranges from Massachusetts (Lindroth 1968: 878) to southeastern Wisconsin (Rauterberg 1885: 20), including southernmost Ontario (Bousquet 1987a: 131), south to southeastern Texas (Harris County, Brian Raber pers. comm. 2010), northern Louisiana (Allen 1965: 71, as Amerinus longipennis; Claiborne Parish, CNC), west-central Mississippi (Casey 1914, as Amerinus longipennis), and west-central South Carolina (Ulyshen et al. 2005: 252).
CAN: ON USA: AR, DC, IA, IL, IN, LA, MA, MD, MO, MS, NC, NJ, NY, OH, PA, SC, TX, VA, WI
Forty-six species (Lorenz 2005: 358) in the Nearctic (two Holarctic species) and Palaearctic (46 species) Regions arrayed in five subgenera: Cardiostenus Tschitschérine (four Palaearctic species), Dicheirotrichus s.str. (nine Palaearctic species), Oreoxenus (one Holarctic species), Pelagophilus Tschitschérine (one west Palaearctic species), and Trichocellus (31 species).
Lindroth (1968: 874-877) covered both species found in North America under the generic name Trichocellus.
One Holarctic species.
This Holarctic subspecies ranges from eastern Siberia (Jaeger and Kataev 2003: 402) to Labrador (Lindroth 1954d: 370); isolated at high altitude in Colorado (Lindroth 1968: 877). Fossil remnants from the Early Wisconsinan have been unearthed in southern Ontario (Morgan and Morgan 1981: 1108).
CAN: AB, BC, LB, MB, NT, QC, SK, YT USA: AK, CO – Holarctic
Two other subspecies, Dicheirotrichus mannerheimii oreophilus K. Daniel and J. Daniel and Dicheirotrichus mannerheimii ponojensis Sahlberg (synonym: Dicheirotrichus setiporus Reitter) are found in the Palaearctic Region.
Thirty-one species in the Nearctic (one Holarctic species) and Palaearctic (31 species) Regions.
This circumpolar species ranges from Iceland to eastern Siberia (Jaeger and Kataev 2003: 403) and from Alaska (Lindroth 1968: 876) to Greenland (Böcher 1988: 14), south to southeastern Massachusetts (Cape Cod, CNC), central Iowa (Wickham 1911b: 8), central South Dakota (Kirk and Balsbaugh 1975: 32), the Sacramento Mountains in New Mexico (Fall and Cockerell 1907: 162), and the Baja California Peninsula (Horn 1894: 312). The records from southeastern New York (Notman 1928: 249) and “Pennsylvania” (Bousquet and Larochelle 1993: 227) need confirmation.
DEN: GL FRA: PM CAN: AB, BC (QCI), LB, MB, NB, NF, NS (CBI), NT, NU, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, IA, ID, IN, MA, ME, MI, MN, MT, ND, NH, NM, NV, OR, SD, UT, VT, WA, WI, WY [NY, PA] – Mexico – Holarctic
Worldwide, with about 125 species in the Nearctic (12 species), Neotropical (ten species), Australian (about ten species), Oriental (about 15 species), Palaearctic (about 50 species), and Afrotropical (30 species) Regions arrayed in eight subgenera: Acupalpus s.str. (about 75 species), Ancylostria Schauberger (four species), Anthracus (32 species), Palcuapus Habu (one east Palaearctic species), Pseudanthracus Habu (three species), Setacupalpus Habu (two east Palaearctic species), Subacupalpus Habu (two Asian species), and Tachistodes (four species). The North American fauna is represented by 12 species (roughly 9.6% of the world fauna), one of them is adventive.
Noonan (1976: 23) treated Hemiaulax Bates (two species), with Idiomelas Tschitchérine (two species) as synonym, as a subgenus of Acupalpus. In Lorenz (2005: 361), Hemiaulax and Idiomelas are listed as valid genera with Egaploa Alluaud (two species) considered a subgenus of Idiomelas.
Worldwide with about 75 species in the Nearctic (six species, one of them adventive), Neotropical (ten species), Australian (three species), Oriental (six species), Palaearctic (28 species), and Afrotropical (22 species) Regions.
Lindroth (1968: 929-934, as meridianus group) covered all species found in North America.
This species ranges from Newfoundland (Lindroth 1955a: 149) to southern Saskatchewan (Lindroth 1968: 934), south to “South Dakota” (Lindroth 1955a: 149), northern Indiana (La Porte County, CMNH), and central Pennsylvania (Lycoming and Clinton Counties, CMNH). The record from “Delaware” (Bousquet and Larochelle 1993: 228) was based on a misidentified specimen of Acupalpus carus in MCZ.
CAN: MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: IN, MA, ME, MI, MN, ND, NH, NY, OH, PA, SD, VT, WI
The range of this species extends from Saint Pierre and Miquelon and southwestern Newfoundland (Lindroth 1955a: 148, as Acupalpus expertus) to southern Saskatchewan (Ronald R. Hooper pers. comm. 2007), south to northern Louisiana (Allen 1965: 72) and southern North Carolina (Macon County, CNC); also known from northwestern Oregon (Westcott et al. 2006: 6), Washington (Spokane County, CMNH), northern Idaho (Hatch 1953: 185), and south-central British Columbia (Lindroth 1968: 931). The record from northern Colorado (Armin 1963: 114) needs confirmation.
FRA: PM CAN: BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AR, CT, DC, DE, IA, ID, IL, IN, LA, MA, ME, MI, MN, MO, NC, NH, NJ, NY, OH, OR, PA, RI, SD, VT, WA, WI, WV [CO]
This species ranges from southeastern New Hampshire (Rockingham County, Ross T. Bell pers. comm. 2008) to at least northern Illinois (Cook County, CMNH), south to northeastern Louisiana (Allen 1965: 72) and northern Florida (Alachua County, CNC).
USA: CT, DC, DE, FL, IL, IN, KY, LA, MA, MD, MI, MO, NH, NJ, NY, OH, PA, RI, VA, VT, WV
This Palaearctic species is adventive in North America where it is known from the Quebec City area (Chantal 1971: 202) in the east and from south-central Saskatchewan (Ronald R. Hooper pers. comm. 2007) to Vancouver Island (Lindroth 1968: 930), including central Alberta (Pollock 1991a: 705), south to “Oregon” (Hatch 1953: 185) and northern Idaho (Hatten et al. 2007: 359) in the west. The first inventoried specimen collected on this continent was found in Seattle in 1931 (Hatch 1946: 77).
CAN: AB, BC (VCI), QC, SK USA: ID, OR, WA – Adventive
This species ranges from Nova Scotia (Lindroth 1968: 933) to northwestern Minnesota (Polk County, CMNH), north to southeastern Manitoba (Roughley et al. 2010: 230; CMNH), south at least to southwestern Pennsylvania (Allegheny County, CMNH) and Rhode Island (Casey 1914: 268). One old specimen simply labeled from Illinois is known (CMNH). The record from “Connecticut” (Bousquet and Larochelle 1993: 228) needs confirmation (see Krinsky and Oliver 2001: 4).
CAN: MB, NB, NS, ON, QC USA: MA, ME, MI, MN, NH, NY, OH, PA, RI, VT, WI [CT, IL]
The range of this species extends from Nova Scotia (Lindroth 1968: 932) to northwestern Minnesota (Polk County, CMNH), south to west-central West Virginia (Mason County, CMNH) and Delaware (Lindroth 1968: 932). The record from eastern South Dakota (Kirk and Balsbaugh 1975: 33) needs confirmation.
CAN: NS, ON, PE, QC USA: CT, DE, IL, MA, ME, MI, MN, NH, NY, OH, PA, VA, WI, WV [SD]
Four North American species, one of them extending into the Bahamas.
Lindroth (1968: 934-938, as pauperculus group) covered all species.
This species ranges from Massachusetts (Suffolk County, MCZ) to central Iowa (Wickham 1911b: 8), including southernmost Ontario (Lindroth 1968: 935; Bousquet 1987a: 132), south to southern Texas (Johnson 1978: 67) and central Florida (Peck and Thomas 1998: 21); also recorded from north-central Maine (Majka et al. 2011: 46). The records from Colorado (Elias 1987: 634) and southeastern Arizona (Dajoz 2004: 116) need confirmation.
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MS, NC, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI, WV [AZ, CO]
This species is found from Maine (Procter 1946: 111) to South Dakota (Kirk and Balsbaugh 1975: 33), including southern Quebec (Larochelle 1975: 31) and southern Ontario (Lindroth 1968: 937), south to southeastern Colorado (Miller and Peairs 2008: 34), southeastern Texas (Casey 1914: 288), and northeastern Florida (Saint Johns County, MCZ).
CAN: ON, QC USA: AR, CO, CT, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI
This species occurs from Nova Scotia (Lindroth 1954c: 309) to eastern South Dakota (Kirk and Balsbaugh 1975: 33; French et al. 2004: 557), south to southeastern Colorado (Miller and Peairs 2008: 34), southern Texas (Wickham 1897: 113; Harris County, Foster F. Purrington pers. comm. 2009), and southern Florida (Monroe County, Drew A. Hildebrandt pers. comm. 2007).
CAN: NB, NS (CBI), ON, PE, QC USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species ranges from southern Quebec (Larochelle 1975: 32) to southeastern South Dakota (Kirk and Balsbaugh 1975: 33), south to eastern Texas (Snow 1906a: 141, as Agonoderus micros; Nacogdoches, Orange, and San Augustine Counties, CNC, CMNH), northeastern Louisiana (Allen 1965: 72), southern Florida, and the Bahamas (Peck and Thomas 1998: 21).
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV – Bahamas
Thirty-two species in the Nearctic (two western species), Australian (five species), Oriental (five species), Palaearctic (15 species), and Afrotropical (eight species) Regions.
The two North American species have never been compared and the specific independence of the two not tested.
This taxon is considered a distinct genus by several authors, including Jaeger and Kataev (2003: 399). However, the main character state separating members of Anthracus from those of Acupalpus is the shape of the pronotum which is cordiform with the sides sinuate in basal half and the posterior angles well distinct, right (see Jeannel 1948a: 714; Basilewsky 1951: 232; Habu 1973a: 301-302). However this character is not constant. Antoine (1959: 453) reported that adults of Acupalpus (Acupalpus) cantabricus cantabricus Piochard de la Brûlerie in Morocco have the sides of the pronotum arcuate and the posterior angles rounded while those of Acupalpus cantabricus zaerensis Antoine (these two taxa are now considered separate species) have the sides sinuate and the posterior angles right and that some specimens greatly resemble adults of Anthracus. Basilewsky (1951: 237) separated Acupalpus (Acupalpus) angulatus Jeannel from most other species in his key to Afrotropical Acupalpus by the clearly sinuate sides of pronotum with the posterior angles markedly acute. Other character states usually reported, such as the mandibles and antennae relatively longer, the prosternum with small setae, and the median line of the pronotum deeper, more or less sulciform in Anthracus are also variable and not at all diagnostic of Anthracus. Until the relationships between members of the acupalpine complex are better known, I prefer to follow Ball (1960b: 147), Lindroth (1968: 925), Noonan (1976: 24), and Ball and Bousquet (2000: 94) and treat Anthracus as a subgenus of Acupalpus.
This species is known from a few localities from northwestern Oregon (Hatch 1953: 185) to southeastern Oregon (Harney County, CNC).
USA: OR
This species is known from California, from Humboldt County (CAS) to Santa Clara County (LeConte, 1857c: 29).
USA: CA
Four North American species, one of them extending into the Bahamas, placed in two subgenera.
Lindroth (1968: 925-926) treated all currently recognized species in his key to Canadian Acupalpus.
This taxon has been treated as a subgenus of Acupalpus by North American students since Ball (1960b: 147). However, members of the group are characteristic in having three or more discal setae on the third elytral interval and the endophallus with four large, curved spines and a basal pillow of pigmented hairs. To my knowledge, all other acupalpines have one or no discal seta on the third elytral interval. The situation regarding the sclerites in the endophallus is more complex. While most species of Acupalpus (including Anthracus) are described as having the internal sac unarmed, there are exceptions. Jeannel (1948a: 720) reported that the endophallus of the Madagascan Anthracus madecassus Jeannel has some large spines (quelques grosses dents visibles par transparence), Habu (1981: 44) described the endophallus of Acupalpus hilaris Tschitschérine as having “four large thorn-like copulatory pieces, ” and Antoine (1959: 451) stated that the internal sac of Acupalpus elegans Dejean has 18 large hooks arranged in two rows (18 grands crochets à base ovale régulièrement disposés sur deux rangs, un droit et un gauche) reminiscent of the condition found in members of Stenolophus. However, in members of Philodes the four endophallus structures are proportionally bigger than in any of these species. At this time, I prefer to regard Philodes as generically distinct from Acupalpus even though Acupalpus may eventually be shown to be paraphyletic in regard to Philodes.
One species occurring over eastern North America.
This species ranges from southern Quebec (Larochelle 1975: 31) south to eastern Oklahoma (Latimer County, UASM), northwestern Arkansas (Newton County, Peter W. Messer pers. comm. 2008), southeastern Mississippi (George and Greene Counties, Drew A. Hildebrandt pers. comm. 2008), east-central Kentucky (Jessamine County, MCZ), and northern Virginia (Hoffman and Roble 2000: 38).
CAN: ON, QC USA: AR, DC, DE, IL, IN, KY, MS, NH, NJ, NY, OH, OK, PA, VA, VT
Three eastern North American species of which one extends into the Bahamas.
This subgenus is in need of a taxonomic revision. I have seen at least four species in eastern North America, one of them (from Baker and Ware Counties in Georgia and Highlands County in Florida) being undescribed.
This species ranges from Georgia (LeConte 1869a: 378; Fattig 1949: 53) to eastern Texas along the Gulf Coast (Casey 1914: 264, as Goniolophus lucens), south to central Florida (Peck and Thomas 1998: 21), north to north-central Tennessee (Smith County, Robert L. Davidson pers. comm. 2012) and central Arkansas (Montgomery and Arkansas Counties, Foster F. Purrington pers. comm. 2009).
USA: AL, AR, FL, GA, LA, MS, TN, TX
This species ranges from southern Virginia (Hoffman and Roble 2000: 38) and Tennessee (Bradley, Crockett, Loudon, Maury, and Smith Counties, CMNH) to southern Florida (Peck and Thomas 1998: 21) and the Bahamas (Darlington 1953: 11), west at least to east-central Texas (Riley 2011), including southeastern Louisiana (Saint Tammany and Tangipahoa Parishes, CNC). The records from Rhode Island (Davis 1904: 14), Delaware (Houghton 1905: 212), and “Arkansas” (Bousquet and Larochelle 1993: 228) need confirmation.
USA: AL, FL, GA, KY, LA, MS, NC, SC, TN, TX, VA [AR, DE, RI] – Bahamas
The range of this species extends from southern Quebec (Larochelle 1975: 32) to eastern South Dakota (Kirk and Balsbaugh 1975: 33), south to eastern Texas (San Augustine and Hardin Counties, CMNH, UASM) and southern Florida (Peck and Thomas 1998: 21).
CAN: ON, QC USA: AL, AR, DC, DE, FL, GA, IA, IL, IN, LA, MA, MD, MI, MO, MS, NC, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI
Two species in southern United States and Middle America (one species) and the West Indies (one species in Haiti).
Darlington (1936c: 205) discussed the structural differences between the two species.
This species is known from central (Grady County, Robert L. Davidson pers. comm. 2008) and southwestern Oklahoma (Kondratieff et al. 2005: 173), Arkansas (Garland and Ouachita Counties, Robert L. Davidson pers. comm. 2012), western Louisiana (Sabine Parish, CMNH), and southern Texas (Wickham 1897: 113) south at least to Nicaragua (Blackwelder 1944: 48).
USA: AR, LA, OK, TX – Guatemala, Mexico, Nicaragua
About 25 species in the Western Hemisphere arrayed in two subgenera: Phymatocephalus (two species) and Polpochila s.str. (23 species).
Nègre (1963) revised the species, including all three found in North America, known at the time.
Two species in southern North America and Mexico.
This species ranges from southern Arizona southwards through the Sonoran and Chihuahuan deserts to Oaxaca in Mexico (Ball and Shpeley 1992a: 55). The records from southwestern New Mexico (Fall and Cockerell 1907: 161) and “California” (Csiki 1932a: 1061) need confirmation.
USA: AZ [CA, NM] – Mexico
This species is known from southern Arizona (Nègre 1963: 215) to western Texas (Culberson, Jeff Davis, and Presidio Counties, CMNH), south to Durango and Sinaloa (Nègre 1963: 215).
USA: AZ, NM, TX – Mexico
Twenty-three species in the Nearctic (one species) and Neotropical (23 species) Regions.
This species is known from southern Arizona to the state of Guerrero in western Mexico (Nègre 1963: 218).
USA: AZ – Mexico
Worldwide, with about 1, 665 species (Lorenz 2005: 362-387, as Harpalina, Amblystomina, and Ditomina). The Northern Hemisphere is represented by about 895 species (53.5% of the world fauna) and North America alone by 138 species (about 8%), of which five are adventive.
Noonan (1976) arrayed the genera of this subtribe into the following genus-groups: Acinopi (no North American representatives), Amblystomi (no North American representatives), Bleusei (no North American representatives), Bradybaeni (no North American representatives), Dapti (including Cratacanthus), Ditomi (no North American representatives), Harpali (including Euryderus, Harpalobrachys, Harpalus, Hartonymus, Ophonus, and Piosoma), and Selenophori (including Amblygnathus, Athrostictus, Aztecarpalus, Discoderus, Selenophorus, Stenomorphus, and Trichotichnus). Ball and Bousquet (2000: 91) followed Noonan’s classification with the exception that the Selenophori were divided into the Selenophori proper and the Trichotichni (including Aztecarpalus and Trichotichnus).
One North American species in the temperate regions.
Lindroth (1968: 745-746) treated the species.
This species ranges from Saskatchewan to central British Columbia (Lindroth 1968: 745-746), south to southern Arizona (Snow 1907: 142; Pima County, CMNH), central New Mexico (Fall and Cockerell 1907: 161; Bernalillo and Socorro Counties, CMNH, UASM), and southeastern Kansas (Wilson County, MCZ). One old specimen labeled “Tex” is known (MCZ).
CAN: AB, BC, SK USA: AZ, CO, ID, KS, MT, ND, NE, NM, NV, SD, UT, WY [TX]
Piosoma setosum LeConte. Adults of this species are easily recognized by the presence of many, long and erect setae over the body, hence the specific name setosum. Prairie inhabitants, the first documented adults were found by John Lawrence LeConte during his expedition of 1845 which brought him along the Platte River up to Fort Laramie in Wyoming, thence to the foot of the Rocky Mountains.
One North American species in the temperate regions.
Ball (1960b) and Lindroth (1968: 747-748) treated the species.
This species ranges from western New York (Ball 1960a: 59, Fig. 3) to the Okanagan Valley in British Columbia (Lindroth 1968: 747), south to northeastern Oregon (Hatch 1953: 165), southern Arizona, southern Texas (Ball 1960a: Fig. 3), southern Louisiana (Allen 1965: 69), and northwestern Mississippi (Bolivar County, Drew A. Hildebrandt pers. comm. 2008), east to southern Georgia (Torres and Ruberson 2006: 32). The records from southeastern Pennsylvania (Rathvon 1869: 526, as Nothopus zabroides) and “California” (Leng 1920: 70, as Nothopus valens) are probably in error.
CAN: AB, BC, MB, ON, SK USA: AR, AZ, CO, GA, IA, ID, IL, IN, KS, LA, MI, MN, MO, MS, MT, ND, NE, NM, NY, OH, OK, OR, SD, TX, UT, WA, WI, WY
About 70 Palaearctic species arrayed in six subgenera: Brachyophonus Sciaky (two species), Hesperophonus Antoine (16 species), Incisophonus Sciaky (one species), Macrophonus Tschitschérine (three species), Metophonus (about 40 species), and Ophonus s.str. (ten species). A single species is found in the Far East and two species are adventive in the Nearctic Region.
Lindroth (1968: 756-758, as rufibarbis group) reviewed both species found in North America and provided a mean for their identification through his key to Harpalus. Both species are also included in Noonan’s (1991: 20-45) key to the North American species of Harpalus excluding the subgenera Pseudoophonus and Glanodes.
About 40 Palaearctic species, of which a single one, Ophonus stricticollis Tschitschérine, is found in eastern Asia. Two species are adventive in eastern North America.
This Palaearctic species is adventive in North America where it is known from Prince Edward Island and Nova Scotia (Majka et al. 2006: 606) to eastern Iowa (Linn County, Doug A. Veal pers. comm. 2009), south to southern Pennsylvania [see Larochelle and Larivière 1989b: Fig. 1] and New Jersey (Steffens and Davidson 1979: 64). The first inventoried specimen collected on this continent was found in Long Island, New York, in 1954 (Dietrich 1958: 46).
CAN: NB, NS, ON, PE, QC USA: CT, IA, IL, MA, ME, MI, NH, NJ, NY, OH, PA, RI, VT, WI – Adventive
This Palaearctic species is adventive in North America where it is known from Montreal (Lindroth 1968: 758) and the Quebec City region (Landry and Rancourt 1976: 53; CNC) in Quebec. The first inventoried specimen collected on this continent was found in Montreal in 1953.
CAN: QC – Adventive
About 415 species (Lorenz 2005: 363-372) in the Nearctic, Neotropical (Mexico only), Australian (one adventive species in New Zealand), Oriental, Palaearctic, and Afrotropical Regions. The Northern Hemisphere is represented by about 360 species (roughly 87% of the world fauna) and North America alone by 59 species (approximately 14%). Five species are Holarctic and three North American species are adventive.
About 60 species in North America (13 species), Mexico (four species, one of them, Harpalus alienus Bates, endemic), Asia (about 45 species), and Europe and northern Africa (three species, none endemic).
Ball and Anderson (1962) revised the North American species and provided a key for their identification. Lindroth (1968: 758-765) covered ten species. Subsequently one new species was described by Will (2002a).
[compar group]
This species ranges from New Jersey and Virginia to northeastern North Dakota, south to the Gulf Coast of Texas and northwestern Mississippi (Bolivar County, Drew A. Hildebrandt pers. comm. 2007); also known from north-central Colorado [see Ball and Anderson 1962: Fig. 39].
USA: AR, CO, IA, IL, IN, KS, MD, MO, MS, ND, NE, NJ, OH, OK, PA, SD, TX, VA, WI
This species ranges from Nova Scotia to southern Manitoba, south to southeastern Arizona, southeastern Texas, and central Florida [see Ball and Anderson 1962: Fig. 39].
CAN: MB, NS, ON, QC USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV, WY
This species is widely distributed east of the Rocky Mountains from Nova Scotia (Lindroth 1968: 764) to southern Manitoba, south to north-central Colorado, southern Oklahoma, and central Florida [see Ball and Anderson 1962: Fig. 39]. The record from south-central Montana (Hatch 1933a: 10) is probably in error.
CAN: MB, NS, ON, QC USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, VA, VT, WI, WV, WY
This primarily prairie species ranges from southeastern Michigan to southern Alberta, south to central Arizona and the Rio Grande Basin in south-central Texas [see Ball and Anderson 1962: Fig. 39]. According to Ball and Anderson (1962: 76), the species is also found in “Mexico.” The records from “Arkansas” and “Pennsylvania” (Bousquet and Larochelle 1993: 232) need confirmation.
CAN: AB, SK USA: AZ, CO, IA, ID, IN, KS, MI, MN, ND, NE, NM, OH, OK, SD, TX, UT, WI [AR, PA] – Mexico
This eastern species occurs from the Saguenay River in southern Quebec to eastern South Dakota, south to central Kansas, southern Mississippi, and southwestern Georgia [see Ball and Anderson 1962: Fig. 39]. The record from “Nova Scotia” (Bousquet and Larochelle 1993: 231) was based on a misidentified specimen of Harpalus erythropus (Majka and Bousquet 2008: 474).
CAN: ON, QC USA: AL, AR, CT, DC, GA, IA, IL, IN, KS, MA, MD, MI, MO, MS, NC, NJ, NY, OH, OK, PA, RI, SC, SD, TN, VA, VT, WI, WV
[pensylvanicus group]
This species is known only from “Arizona” (Erwin et al. 1977: 4.48) and “Mexico” (Bates 1882a: 57). The record from “near Philadelphia, ” Pennsylvania (Casey 1914: 82) is likely in error.
USA: AZ – Mexico
Ball and Anderson (1962: 45) listed Harpalus liobasis Chaudoir as a junior synonym of Harpalus pensylvanicus but Lindroth (1968: 761) treated it as a distinct species.
This common species occurs over a large area in North America from Prince Edward Island (Lindroth 1968: 761) and Nova Scotia to south-central British Columbia, south to southern California, northwestern Mexico, southeastern Texas, southern Florida, and the Bahamas [see Ball and Anderson 1962: Fig. 38].
CAN: BC, MB, NB, NS, ON, PE, QC, SK USA: AK, AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Bahamas, Mexico
This species is found from Maryland to southern Nebraska (Nuckolls County, Foster F. Purrington pers. comm. 2011), south to south-central Oklahoma, northern Louisiana, and east-central Alabama [see Ball and Anderson 1962: Fig. 38].
USA: AL, GA, IL, IN, KS, KY, LA, MD, MO, MS, NC, NE, OH, OK, SC, TN, VA, WV
This species is restricted to the Coastal Plain ranging from south-central North Carolina to southern Florida, west to eastern Texas [see Ball and Anderson 1962: Fig. 38].
USA: FL, GA, MS, NC, TX
[rufipes group]
This species ranges from southwestern Maine (Majka et al. 2011: 46) and southern Quebec (Larochelle 1975: 87) to western North Dakota, south to western (Dajoz 2007: 23) and northeastern Texas, southeastern Louisiana, and the Florida Panhandle (Peck and Thomas 1998: 21); also known from northern (Villa-Castillo and Wagner 2002: 246) and east-central Arizona [see Ball and Anderson 1962: Fig. 37].
CAN: MB, ON, QC USA: AR, AZ, CO, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species is known only from three specimens collected in Kansas (Ball and Anderson 1962: 43) and five specimens from central Oklahoma (Seminole County, Robert L. Davidson pers. comm. 2008).
USA: KS, OK
This adventive species is found from Newfoundland (Larson and Langor 1982: 593) to near Montreal, Quebec (Mercado Cárdenas and Buddle 2007: 140), south to Connecticut (Krinsky and Oliver 2001: 210) and Rhode Island (Zhang et al. 1994: 69). The record from Wayne County, New York (Hajek et al. 2007) is in error (see Hajek et al. 2009: 913). The first inventoried specimen collected on this continent was found on Prince Edward Island in 1937 (Brown 1950b: 199).
FRA: PM CAN: NB, NF, NS (CBI), PE, QC USA: CT, MA, ME, NH, RI, VT – Adventive
This species is known only from two specimens collected in 1963 in two localities in Florida, along the coast of the Gulf of Mexico [see Will 2002a: Fig. 5].
USA: FL
Two North American species, both extending into northern Mexico.
Will (1997) reviewed the species of this subgenus and provided a key for their identification.
This taxon is listed as a synonym of Pseudoophonus Motschulsky by Kataev et al. (2003: 384).
This species ranges from Nova Scotia (King County, CNC) to Washington, north to southern Manitoba (Lindroth 1968: 765) and southern Saskatchewan (CNC), south to the northern part of the Baja California Peninsula, northeastern Mexico, and northern Florida [see Will 1997: Fig. 1A].
CAN: MB, NS, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
This species ranges from northern Colorado (Will 1997: 47) to southern Virginia (Hoffman and Roble 2000: 37), north to Wisconsin (Purrington et al. 2002: 201), south to northern Florida, northeastern Mexico (Will 1997: 47, 49), southeastern New Mexico (Dunn 1986: 2), and southeastern Arizona (Cochise County, Ken Karns pers. comm. 2009) [see Will 1997: Fig. 1B].
USA: AL, AR, AZ, CO, GA, FL, IL, LA, MO, MS, NC, NE, NM, OK, SC, TN, TX, VA, WI – Mexico
Two North American species.
El-Moursy (1959) and Noonan (1991: 131-135) revised the species. Both species were also treated by Lindroth (1968: 766-767).
This species occurs east of the Rocky Mountains ranging from southern Quebec to southeastern Alberta, south to central New Mexico, southern Kansas, central Alabama [see Noonan 1991: Fig. 285], and southeastern South Carolina (Ciegler 2000: 100).
CAN: AB, MB, ON, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, GA, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, MS, MT, ND, NE, NH, NJ, NM, NY, OH, RI, SC, SD, TN, VT, WI
This species is restricted to mountains in “Utah” (Noonan 1991: 135), Arizona, New Mexico, and southern Colorado [see Noonan 1991: Fig. 285]. The record from “Mexico” (Csiki 1932a: 1186) is probably in error.
USA: AZ, CO, NM, UT
This group is represented in the Nearctic and Palaearctic Regions but the number of species cannot be assessed at this time. The North American fauna has 19 species.
The species of this subgenus have been revised by Noonan (1991: 45-127). Lindroth (1968) covered all the species except Harpalus gravis, Harpalus cordatus, and Harpalus apache.
This taxon corresponds to the fraternus stock of Noonan (1991: 45). Although I have doubt about monophyly of this complex, I have followed Noonan’s (1991) conclusion since it is the last major study on these species. Kataev et al. (2003: 371) listed Opadius Casey and its synonyms as synonyms of the subgenus Harpalus Latreille. In 2010, Kataev included Harpalus apache and Harpalus cordatus (as cordatus group) in the subgenus Glanodes Casey.
[cordatus group]
This species is known only from the type locality in east-central New Mexico.
USA: NM
This species is restricted to arid grasslands in eastern Arizona, southern Colorado, and New Mexico [see Noonan 1991: Fig. 276].
USA: AZ, CO, NM
Ball (1972: 185) proposed Harpalus tadorcus to replace Harpalus cordatus (LeConte, 1853), a junior secondary homonym of Harpalus cordatus (Duftschmid, 1812), both species being then placed in the genus Harpalus. Since Duftschmid’s species is currently included in the genus Ophonus and the replacement name was proposed after 1960, the original species-group name is to be reinstated (ICZN 1999: Article 59.4).
This species ranges from the Alexander Archipelago and northwestern British Columbia south to southwestern Oregon, east to eastern Washington [see Noonan 1991: Fig. 276].
CAN: BC (VCI) USA: AK, OR, WA
[desertus group]
This western species occurs from south-central British Columbia to western Minnesota (Gandhi et al. 2005: 930), south to east-central and westernmost Texas, southern Arizona, and northern California [see Noonan 1991: Figs 279-282].
CAN: AB, BC, SK USA: AZ, CA, CO, ID, KS, MN, MT, ND, NE, NM, NV, OK, OR, SD, TX, UT, WA, WY
Noonan (1991: 119-124) recognized two morphs within this species, the desertus and furtivus morphs, that Lindroth (1968: 809) regarded as distinct species. Noonan (1991: 124) indicated that intergradation between the two morphs occurs throughout the areas in which the furtivus morph occurs.
This species is found along the Coastal Plain and Piedmont Plateau from southeastern New York and New Jersey to northern Florida, west to southeastern Oklahoma and southeastern Texas [see Noonan 1991: Fig. 270]. The record from southwestern Ohio (Blatchley 1910: 186) is probably in error.
USA: AL, AR, FL, GA, LA, MO, MS, NC, NJ, NY, OK, SC, TX, VA
This species ranges from southernmost Ontario (Bousquet 1987a: 130) to southeastern South Dakota (Kirk and Balsbaugh 1975: 28), south to southeastern Kansas (Noonan 1991: 115) and northern Mississippi (Drew A. Hildebrandt pers. comm. 2007). Two old specimens simply labeled from Louisiana and Florida are known (Noonan 1991: 115).
CAN: ON USA: AR, IA, IL, IN, KS, MI, MO, MS, NE, OH, SD, TN, WI [FL, LA]
[laticeps group]
This western species occurs from northwestern British Columbia to southwestern Saskatchewan, south to north-central New Mexico and northern Arizona in the Rocky Mountains and to northern Oregon along the coast [see Noonan 1991: Fig. 267]. The record from south-central South Dakota (Kirk and Balsbaugh 1975: 29, as Harpalus montuosus) needs confirmation.
CAN: AB, BC (VCI), SK USA: AZ, CO, ID, MT, NM, NV, OR, UT, WA, WY [SD]
This species is transcontinental in the north ranging from Newfoundland (Lindroth 1955a: 139; probably only as strays) to western Yukon Territory, south to northern Oregon along the west coast, southeastern Arizona and central New Mexico in the Rocky Mountains, the Black Hills in western South Dakota, northern Illinois, and southwestern Pennsylvania (Noonan 1991: 56) in the east [see Noonan 1991: Fig. 268]. The records from Connecticut (Britton 1920: 218; see Krinsky and Oliver 2001: 5), Indiana (Blatchley 1910: 186), and southwestern Oklahoma (Kondratieff et al. 2005: 172) need confirmation.
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CO, ID, IL, MA, ME, MI, MN, MT, ND, NH, NM, NV, NY, OR, PA, SD, VT, WA, WI, WY [CT, IN, OK]
This eastern species ranges from southern Quebec to northern Minnesota, south to central Missouri and southwestern Virginia (Hoffman and Roble 2000: 38) [see Noonan 1991: Fig. 267]; also known from southwestern South Dakota (Larsen and Purrington 2010: 571). An old specimen simply labeled from Kansas and one labeled from Alabama are known (Noonan 1991: 51).
CAN: ON, QC USA: CT, DC, IA, IL, IN, KY, MA, MD, ME, MI, MN, MO, NE, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WI, WV [AL, KS]
[nigritarsis group]
This species ranges from Maine (Piscataquis County, CNC) and northern New Brunswick (Webster and Bousquet 2008: 20) west to Manitoba and Minnesota (Lindroth 1968: 785), south to the Black Hills in western South Dakota (CMNH). The record from the Ungava Bay in Labrador (Sherman 1910: 181) is probably in error.
CAN: MB, NB, ON, QC USA: ME, MI, MN, SD, WI
This species is externally very similar to Harpalus fulvilabris and Lindroth (1968: 783) included these two species in one species group. However, Noonan (1991) listed the two species in different species groups suggesting that the similarities are probably due to convergent evolution.
This Holarctic species ranges in the Nearctic Region from Newfoundland to the Alaskan coast, south to the Sierra Nevada in California, southern Arizona, southeastern Nebraska, and New Hampshire and New York [see Noonan 1991: Figs 277 and 278]. The species is known only from a few scattered localities in the prairies. The record from southern Indiana (Wiedenmann et al. 1992: 286) needs confirmation. In the Palaearctic Region, the species is known from Sweden to the Far East and the Altai in Kazakhstan (Kataev et al. 2003: 379).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, YT USA: AK, AZ, CA, CO, ID, MI, MN, MT, NE, NH, NM, NV, NY, OR, SD, UT, WA, WI, WY [IN] – Holarctic
Noonan (1991: 102-104) recognized two morphs in this species, the nigritarsis and seclusus morphs, which Lindroth (1968) regarded as distinct species. Noonan stated that a broad zone of intergradation between the two morphs occurs in the northern part of the Rocky Mountains and adjacent mountains and in lowland areas of northwestern United States, western Canada, and Alaska.
[spadiceus group]
This western species occurs from Vancouver Island to southeastern Manitoba (Roughley et al. 2010: 230), north to northeastern British Columbia, south to western Texas, southern Arizona, and the White Mountains in California, east to eastern Nebraska [see Noonan 1991: Fig. 271].
CAN: AB, BC (VCI), MB, SK USA: AZ, CA, CO, ID, KS, MT, NE, NM, NV, OR, SD, TX, UT, WA, WY
Noonan (1991: 75) recognized two forms within this species, a southern one, known as Harpalus lecontei, that Lindroth (1968) treated as a distinct species, and a more northern one which corresponds to Harpalus fraternus LeConte sensu Lindroth.
This species is transcontinental in the north, ranging from Newfoundland to the Aleutian Islands in Alaska, south to the Lillooet Land District in British Columbia, north-central New Mexico along the Rocky Mountains, the Black Hills in western South Dakota, and New England in the east [see Noonan 1991: Fig. 276].
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, ME, MI, MN, NH, NM, NY, SD, VT, WI
This species ranges from the Nova Scotia Peninsula (Christopher G. Majka pers. comm. 2008) to southern South Dakota (Kirk and Balsbaugh 1975: 30), north to southeastern Manitoba (Roughley et al. 2010: 230), south to Nebraska and southern Pennsylvania (Noonan 1991: 85; Fig. 272).
CAN: MB, NB, NS, ON, QC USA: CT, IA, IL, MA, ME, MI, NE, NH, NJ, NY, PA, SD, VT, WI
This Holarctic species ranges in North America from Newfoundland to southeastern Alaska (Lindroth 1968: 777), south to west-central Oregon, southern Nevada, southeastern Arizona and south-central New Mexico along the Rocky Mountains, the lower peninsula of Michigan, and southern New York [see Noonan 1991: Fig. 269]. The records from southern Indiana (Wiedenmann et al. 1992: 282, as Harpalus egregius) and “Iowa” (Jaques and Redlinger 1946: 297, as Harpalus rufimanus) need confirmation.
CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CO, ME, MI, MN, MT, NH, NM, NV, NY, OR, UT, WA, WI [IA, IN] – Holarctic
This species ranges from Nova Scotia to central Alberta and southeastern Northwest Territories, south to southern Wisconsin and Connecticut [see Noonan 1991: Fig. 270]. All known sites in the United States are east of the Mississippi.
CAN: AB, MB, NB, NS, NT, ON, QC, SK USA: CT, IN, MA, ME, MI, MN, ND, NH, NY, VT, WI
With the exception of two specimens labeled from “British Columbia” and “Victoria” on Vancouver Island (Noonan 1991: 70), this species occurs east of the Rocky Mountains from central Alberta to the Quebec City area (Larochelle 1975: 87, map 302), south to Massachusetts, northwestern Pennsylvania (Erie County, Robert L. Davidson pers. comm. 2012), southern Kansas, central New Mexico, and southeastern Arizona [see Noonan 1991: Fig. 271]. This species is much more abundant in the prairies of central North America and both Lindroth (1968: 779; 1971: 1456, 1457) and Noonan (1991: 69) concluded that the species was originally an inhabitant of the prairies that expanded its range eastwards after humans cut down forests in the east.
CAN: AB, MB, ON, QC, SK USA: AZ, CO, IA, KS, MA, ME, MI, MN, MT, ND, NE, NH, NM, NY, PA, SD, VT, WI, WY [BC]
This eastern species ranges from southern Quebec, southern Ontario, and the New England states south through the Appalachian mountains to northern Georgia [see Noonan 1991: Figs 273-275]. The record from southwestern Manitoba (Stjernberg 2011: 70) is probably in error.
CAN: ON, QC USA: CT, DC, DE, GA, KY, MA, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WV
Noonan (1991: 87-88) recognized two morphs for this species, the spadiceus and carolinae morphs, that Lindroth (1968: 780-781) treated as distinct species. According to Noonan (1991: 88), the two morphs intergrade over an area extending southwards from Virginia to Georgia.
This species ranges from central Alberta to southeastern Manitoba (Roughley et al. 2010: 230), south to western Kansas, northern New Mexico, and northeastern Utah [see Noonan 1991: Fig. 272].
CAN: AB, MB, SK USA: CO, KS, MN, MT, ND, NE, NM, SD, UT, WY
This subgenus includes native species in North America, Mexico, and the Palaearctic and Afrotropical Regions. The total number of species cannot be determined at this time. The North American fauna is represented by 17 species, of which two are adventive.
Noonan (1991) revised all the North American species except the two adventive ones but his key (pages 20-45) included all 17 species. Lindroth (1968) covered all but three (Harpalus balli, Harpalus martini, and Harpalus rubripes) species in his monograph of the Canadian Carabidae.
[affinis group]
This Palaearctic species is adventive in North America where it is known in the east from Newfoundland (Lindroth 1955a: 137) to eastern Minnesota (Ramsey County, CMNH), south to “Kansas” (Lindroth 1955a: 137) and northern (Tucker and Preston Counties, CMNH) and eastern West Virginia (Pendleton County, Foster F. Purrington pers. comm. 2009). The record from southern Florida (Peck and Thomas 1998: 21) may represent a separate introduction. In the west, the species ranges from Alberta and British Columbia, including Vancouver Island (Lindroth 1968: 769), south to western Oregon (Clackamas, Linn, and Yamhill Counties, CMNH) and southwestern Idaho (Owyhee County, Ken Karns pers. comm. 2009); the species is also found in southeastern Arizona (Cochise County, CNC). The first inventoried specimen caught in the east was collected prior to 1798 in Pennsylvania (Palisot de Beauvois, 1811: 108, as Harpalus viridiaeneus). The species is also adventive in New Zealand since 1975 (Larochelle and Larivière 2005: 52).
FRA: PM CAN: AB, BC (VCI), LB, NB, NF, NS (CBI), ON, PE, QC USA: AZ, CT, DE, FL, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MO, NH, NJ, NY, OH, OR, PA, RI, VA, VT, WA, WI, WV – Adventive
This Palaearctic species is adventive in North America where it is known from Nova Scotia (Majka et al. 2006: 606) and Prince Edward Island (Majka et al. 2008: 132) to eastern Ontario (near Reids Mills, CNC), including southern Quebec (Chantal 1994: 29), south to eastern Pennsylvania (Davidson et al. 2011: 517). The first inventoried specimen collected on this continent was found in New Hampshire in 1981 (Bell and Davidson 1987: 56).
CAN: NB, NS, ON, PE, QC USA: CT, MA, ME, NH, NY, PA, RI, VT – Adventive
[amputatus group]
This species is found west of the Mississippi River from northeastern Alaska to northwestern Minnesota, south to the highlands of central Mexico (Noonan 1991: 179) and southern California [see Noonan 1991: Fig. 289].
CAN: AB, BC, MB, NT, SK, YT USA: AK, AZ, CA, CO, ID, KS, MN, MT, ND, NE, NM, NV, OK, OR, SD, TX, UT, WA, WY – Mexico
Three subspecies of Harpalus amputatus are known from Asia (see Kataev et al. 2003: 372).
[atrichatus group]
This species is known only from a few scattered localities from southwestern British Columbia, including Vancouver Island, south to west-central Oregon, east to northern Idaho [see Noonan 1991: Fig. 291].
CAN: BC (VCI) USA: ID, OR, WA
[cautus group]
This species is known from only six specimens collected near the coast in Massachusetts and New Jersey [see Noonan 1991: Fig. 283]. The collection date of the last known specimen is 1932 and Noonan (1991: 153) believes that it “might be extinct because of destruction of ocean side habitats.”
USA: MA, NJ
This western species occurs from central Alaska south to southern California, southeastern Arizona, and south-central Colorado, east to western Nebraska [see Noonan 1991: Fig. 286]. The records from northeastern Kansas (Popenoe 1877: 24) and Beaver Island in Michigan (Hatch 1925: 554) are probably in error. According to Noonan (1991: 143), this species is one of the most abundant Harpalus along the west coast.
CAN: AB, BC (VCI) USA: AK, AZ, CA, CO, ID, MT, NE, NV, OR, UT, WA, WY
The range of this species extends from central Alberta and southern Saskatchewan south to southwestern Texas and southeastern Arizona; seemingly isolated in Minnesota (Gandhi et al. 2005: 930) and northwestern Oregon [see Noonan 1991: Fig. 287]; also recorded from northern Sonora in Mexico (Bates 1884: 271).
CAN: AB SK USA: AZ, CO, ID, KS, MN, MT, NE, NM, OR, SD, TX, UT, WY – Mexico
This species ranges from western Quebec to Vancouver Island, north to southern Northwest Territories, south to the Sierra Nevada in California, southern Colorado in the Rocky Mountains, southwestern South Dakota (Custer County, Peter W. Messer pers. comm. 2011), the upper peninsula of Michigan, and southwestern Pennsylvania [see Noonan 1991: Fig. 288]. Several specimens simply labeled from “New Mexico” are known (Noonan 1991: 159). East of the Rocky Mountains, the species is known only from a few scattered localities.
CAN: AB, BC (VCI), NT, ON, QC, SK USA: CA, CO, ID, MI, MN, MT, OR, PA, SD, WA, WI, WY [NM]
The range of this species extends from Alaska (Lindroth 1968: 808) east to Anticosti Island in Quebec, south to northeastern Minnesota (Purrington and Maxey 2007: 219) and the southern part of the Prairie Provinces [see Noonan 1991: Fig. 287]. The records from Colorado (Wickham 1902: 242) and New Mexico (Fall and Cockerell 1907: 161) are likely in error.
CAN: AB, BC, MB, ON, QC, SK, YT USA: AK, MN
This Holarctic subspecies ranges from eastern Siberia (Kataev et al. 2003: 384) to the Alaska-Yukon border along the Porcupine River. On this continent, the subspecies is known from extant specimens only from the type locality. It was cited as a dominant taxon in a late Pleistocene site in the Klondike region of Yukon Territory (Zazula et al. 2006: 265).
USA: AK – Holarctic
This taxon was originally described as a distinct species but Kataev (1990: 396) listed it as a subspecies of Harpalus vittatus Gebler. The nominotypical subspecies and Harpalus vittatus kiselevi Kataev and Shilenkov are found in Asia (Kataevet al. 2003: 384).
[obnixus group]
This species ranges from southwestern Alberta and British Columbia (Noonan 1991: 162-163) south to west-central Oregon, northeastern Nevada, and northwestern New Mexico (Casey 1924: 106, as Harpalus antiphon) [see Noonan 1991: Fig. 290]. One old specimen simply labeled from Nebraska is known (Noonan 1991: 163).
CAN: AB, BC USA: CO, ID, MT, NM, NV, OR, UT, WA, WY [NE]
This species is known from scattered localities from Newfoundland (Lindroth 1955a: 143) to southern British Columbia, north to southwestern Northwest Territories, south to central Arizona, north-central New Mexico, eastern Texas, and southwestern North Carolina [see Noonan 1991: Fig. 292].
CAN: BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AR, AZ, CO, ID, IL, KS, MA, ME, MI, NC, NH, NJ, NM, NY, TX, VA, VT, WI
[opacipennis group]
This species ranges from southern New Brunswick (Webster and DeMerchant 2012: 6) to south-central Alaska (Lindroth 1968: 806), south to the Sierra Nevada in California, central Arizona and central New Mexico along the Rocky Mountains, southern Indiana, and Maryland (Erwin 1981b: 175) [see Noonan 1991: Fig. 296].
CAN: AB, BC (VCI), MB, NB, NT, ON, QC, SK, YT USA: AK, AZ, CA, CO, ID, IN, MA, MD, ME, MI, MN, MT, ND, NM, NV, NY, OH, OR, PA, SD, UT, VT, WA, WI, WY
[somnulentus group]
The range of this species extends from Nova Scotia to south-central British Columbia, north to central Alberta, south to southern Oregon, east-central Arizona and central New Mexico along the Rocky Mountains, the Texas Panhandle (Michels et al. 2010: 743), northern Missouri, and southern Georgia [see Noonan 1991: Fig. 293]. The record from “Florida” (Leng 1920: 71) needs confirmation.
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY [FL]
This species is known from only 16 specimens (including 15 ♂) from mountains in southern California [see Noonan 1991: Fig. 287] and could be either endangered or extinct according to Noonan (1991: 189). The last known specimen found was collected in 1919.
USA: CA
The range of this Holarctic species extends in northern North America from Newfoundland to south-central Alaska (Lindroth 1968: 799), south to east-central British Columbia, “Minnesota” (Lindroth 1968: 799), northeastern Wisconsin (Messer 2010: 41), “Michigan” (Lindroth 1968: 799), and Maine (Noonan 1991: 187) [see Noonan 1991: Fig. 294]. The records from the Sierra Nevada and Colorado (Elias 1987: 633) are probably in error; that from Prince Edward Island (Bousquet and Larochelle 1993: 237) needs confirmation (see Majka et al. 2008: 132).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS, NT, ON, QC, SK, YT USA: AK, ME, MI, MN, NH, WI [PE] – Holarctic
This species is transcontinental in the north ranging from Newfoundland to the Kenai Peninsula in Alaska, south to southern California, central Arizona and New Mexico in the Rocky Mountains, Nebraska, and northern Virginia [see Noonan 1991: Figs 297-309]. The records from central Alabama (see Noonan 1991: Figs 302 and 304), “Arkansas” (Bousquet and Larochelle 1993: 237) and southern Kansas (Snow 1903: 194, as Harpalus fallax) need confirmation.
FRA: PM CAN: AB, BC (QCI, VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, DE, IA, ID, IL, IN, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NM, NV, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WV, WY [AL, AR, KS]
Noonan (1991: 196-207) recognized six morphs within this species, the pleuriticus, fallax, “dark, ” uteanus, somnulentus, and carbonatus morphs, which Lindroth (1968) treated as distinct species, except for the “dark” one. Noonan went on to report numerous intergradation zones between all morphs.
Six species in southwestern North America.
Ball (1972) revised the species and provided a key for the identification of the males.
This species is known only from western Texas (Ball 1972: Fig. 13) and southeastern New Mexico (Perrault 1974: 120).
USA: NM, TX
This species is known for sure only from Summit and Washington Counties in Utah (Ball 1972: 192). The record from “Arizona” (Bousquet and Larochelle 1993: 237) needs confirmation.
USA: UT [AZ]
This species is known only from the holotype collected in southeastern Arizona [see Ball 1972: Fig. 13].
USA: AZ
This species is found in southern (Perrault 1974: 120) and western New Mexico and southeastern Arizona [see Ball 1972: Fig. 13].
USA: AZ, NM
This species is known only from northwestern Arizona [see Ball 1972: Fig. 13; Perrault 1982b: 269].
USA: AZ
Ball (1973: 74) proposed Harpalus cunctipeps to replace Harpalus puncticeps (Casey, 1914), a junior secondary homonym of Harpalus puncticeps (Stephens, 1828), both species being then placed in the genus Harpalus. Since Stephens’ species is currently included in the genus Ophonus and the replacement name was proposed after 1960, the original species-group name is to be reinstated (ICZN 1999: Article 59.4).
This species is known only from Culberson County in western Texas and Pima County in southern Arizona [Ball 1972: 194, Fig. 13].
USA: AZ, TX
Four Palaearctic species, of which one is Holarctic.
The North American species was covered in Lindroth (1968: 815-817) monograph under the name Harpalellus basilaris.
This Holarctic species ranges from Germany to eastern Siberia (Kataev et al. 2003: 376) and from Alaska to Nova Scotia (Lindroth 1968: 817, as Harpalellus basilaris), south to northwestern Ohio (Lucas County, Harry J. Lee, Jr. pers. comm. 2008), southeastern Wisconsin (Messer 2010: 41), northern Iowa (Wickham 1911b: 8, as Harpalus basilaris), northwestern New Mexico (Lindroth 1968: 816), and northern California (Siskiyou County, CNC).
CAN: AB, BC (VCI), MB, NS, NT, ON, QC, SK, YT USA: AK, AZ, CA, CO, IA, ID, MI, MN, MT, ND, NH, NM, NV, OH, OR, SD, UT, WA, WI, WY – Holarctic
One Holarctic species.
The species was treated by Lindroth (1968: 748-749) and Noonan (1991: 212-213).
Noonan (1991: 211) treated this taxon as a group within the genus Harpalus. Lindroth (1968: 748), Kryzhanovskij et al. (1995: 140), and Kataev et al. (2003: 370) regarded Harpalobrachys as a distinct genus.
This Holarctic species ranges from the European part of Russia to eastern Siberia (Kataev et al. 2003: 370) and in the Nearctic Region from east-central Alaska to southern Northwest Territories (Lindroth 1968: 749), south to central Alberta (Bousquet 1987a: 130). The species is rarely collected on this continent.
CAN: AB, NT, YT USA: AK – Holarctic
Two allopatric species in temperate areas of North America.
Ball (1976b) revised the species and provided a key for their recognition.
This species is known from “Arkansas” to eastern New Mexico [see Ball 1976b: Fig. 7], including southeastern Colorado (Michels et al. 2008).
USA: AR, CO, NM, OK, TX
This species in found along the Mississippi Basin from west-central Wisconsin (Messer 2010: 41) to eastern Nebraska (Colfax County, Foster F. Purrington pers. comm. 2010; Greeley County, R. Michael Brattain collection), south to Oklahoma (Ball and Bousquet 2000: 99) and Missouri [see Ball 1976b: Fig. 7].
USA: IA, IL, MO, NE, OK, WI
Twenty-four species in temperate, subtropical, and tropical areas of the Nearctic (four species, only one endemic) and Neotropical (23 species) Regions, including the West Indies (three species, one of them endemic).
Ball and Maddison (1987) revised the species and provided a key for their identification.
This species ranges from southeastern California and southwestern Arizona, south along the Pacific Coast to El Salvador [see Ball and Maddison 1987: Fig. 29].
USA: AZ, CA – El Salvador, Guatemala, Mexico
This species is found along and near the Atlantic Coastal Plain from New Jersey to the Florida Keys [see Ball and Maddison 1987: Fig. 28] and along the Gulf of Mexico in southwestern Alabama (Löding 1945: 25; Robert L. Davidson pers. comm. 2012). The record from “Pennsylvania” (LeConte 1847: 392), “Illinois, ” and “Texas” (Horn 1880e: 182) are probably in error.
USA: AL, FL, GA, MD, NC, NJ, SC, VA
This species ranges along the Coastal Plain from New Jersey to southern Florida, west to southeastern Texas, north along the Mississippi Basin to southern Arkansas (Ouachita County, CMNH) and northeastern Mississippi; south of the Rio Grande it occurs through eastern Mexico and Central America to the Canal Zone in Panama [see Ball and Maddison 1987: Fig. 29]. The record from “Pennsylvania” (Ball and Maddison 1987: 210) needs confirmation.
USA: AL, AR, FL, GA, LA, MS, NC, NJ, SC, TX, VA [PA] – Guatemala, Honduras, Mexico, Nicaragua, Panama
The range of this species extends along the Gulf Coast from the Florida Panhandle (Jefferson County, CMNH) to central Veracruz in Mexico [see Ball and Maddison 1987: Fig. 31].
USA: FL, LA, MS, TX – Mexico
Eighteen species in the Neotropical Region, of which one extends into southern Texas.
There is no modern key for the identification of the species. The genus is in need of a revision.
This species is known from northeastern Arkansas (Kraim 1983: 137, as Selenophorus perpolitus), Amite County in southwestern Mississippi (Peter W. Messer pers. comm. 2012), East Baton Rouge Parish in southern Louisiana (Igor Sokolov pers. comm. 2009), and from southern Texas (Johnson 1978: 68; Zapata, Live Oak, and Uvalde Counties, CMNH) to Yucatán (Putzeys 1878: 65).
USA: AR, LA, MS, TX – Mexico
The systematic position of this species is not quite settled. It was transferred to the genus Athrostictus by Erwin [in Reichardt 1977: 428, under the spelling Athrostictus punculatus (Putzeys, 1878)]. It was retained in the genus Selenophorus by Noonan (1985: 41) although he did not study specimens. Bates (1891a: 243) wrote “the species [i.e., Selenophorus punctatulus] belongs to a group of the genus in which the thorax (except on the disk) and the elytra are closely punctured, a group which connects Selenophorus with Arthrostictus [sic].”
Western Hemisphere, with about 190 species arrayed in two subgenera: Celiamorphus and Selenophorus s.str., both represented in North America.
Putzeys (1878) reviewed the genus but his work is outdated. About one-third of the currently recognized valid North American species were described by Casey and there is little doubt that several of them will turn out to be synonyms. A revision of the genus is much needed.
Fourteen species in temperate, subtropical, and tropical areas of the Nearctic (nine species) and Neotropical (eight species) Regions.
This species is known only from the type locality in southeastern Texas.
USA: TX
This species is known from North Carolina (Casey 1914: 144) and northeastern Georgia (Fattig 1949: 48).
USA: GA, NC
This species is found in Mississippi (Harrison, Jackson, and Stone Counties, Paul K. Lago pers. comm. 2009), throughout the Florida Peninsula (Peck and Thomas 1998: 22), on several islands of the West Indies (Peck 2005: 32; Peck 2011: 13), and in Venezuela (Reiche 1843b: 142, as Selenophorus harpaloides) and Colombia (Reiche 1843b: 142, as Selenophorus aeratus).
USA: FL, MS – Antigua, Bahamas, British Virgin Islands, Cayman Islands, Colombia, Cuba, Dominica, Guana Island, Hispaniola, Jamaica, Montserrat, Navassa, Puerto Rico, Venezuela
The range of this species extends from west-central Maine (Majka et al. 2011: 46) to eastern Minnesota (Gandhi et al. 2005: 931), south to “Texas” (Horn 1880e: 183) and southern Florida (Peck and Thomas 1998: 22); also cited from “northern Mexico” (Casey 1914: 143).
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI – Mexico
This species is known from North Carolina (Brimley 1938: 128), Georgia (Horn 1880e: 182; Fattig 1949: 48), throughout Florida (Peck and Thomas 1998: 22), Alabama (Löding 1945: 25), and southeastern coastal Mississippi (Drew A. Hildebrandt pers. comm. 2007).
USA: AL, FL, GA, MS, NC
This species ranges from “Massachusetts” (Lindroth 1968: 830) to southern Florida (Vince Golia pers. comm. 2007), west to southern Texas (Johnson 1978: 67), and north along the Mississippi Basin to eastern Iowa (Linn County, Doug A. Veal pers. comm. 2009), “Illinois” (Bousquet and Larochelle 1993: 239), and “Indiana” (Schrock 1985: 355).
USA: AL, CT, FL, GA, IA, IL, IN, MA, MD, MS, NC, NJ, NY, RI, TX, VA
This species is known only from the type locality in central North Carolina.
USA: NC
This species is known only from the type locality in central North Carolina.
USA: NC
This species is known only from the type series.
USA: TX – Mexico
About 175 species in temperate, subtropical, and tropical areas of the Nearctic (29 species) and Neotropical (about 150 species) Regions.
This species is known from northern Colorado (Denver and Boulder Counties, UASM) and southern Utah (Kane and Garfield Counties, CMNH) south to the Isthmus of Tehuantepec in Mexico (UASM).
USA: AZ, CO, NM, TX, UT – Mexico
This species is known from North Carolina (Manee 1915: 175), northeastern Georgia (Fattig 1949: 49), and southern South Carolina (Ciegler 2000: 103).
USA: GA, NC, SC
This species is known from a few localities on the lower Rio Grande of southern Texas (Purrington 2000: 9).
USA: TX
This species is known yet only from the Huachuca Mountains in southeastern Arizona.
USA: AZ
This species is known only from the type series.
USA: TX
This species is known only from southeastern Texas.
USA: TX
This species ranges from “Georgia” (LeConte 1847: 408) to central Florida (Peck and Thomas 1998: 22), west to southwestern Alabama (Löding 1945: 25).
USA: AL, FL, GA
This species is known from southern Arizona (Cochise, Graham, Maricopa, Pima and Santa Cruz Counties, UASM), northern Sonora (UASM), Baja California Norte (UASM), and “California” (CMNH).
USA: AZ, CA – Mexico
This species ranges from North Carolina (Brimley 1938: 128) and northern Tennessee (Montgomery County, Foster F. Purrington pers. comm. 2011) to southern Florida (Peck and Thomas 1998: 22), west to eastern Texas (Johnson 1978: 68) and south to eastern Mexico (George E. Ball pers. comm. 2008).
USA: AL, FL, GA, LA, MS, NC, SC, TN, TX – Mexico
The range of this species extends from Nova Scotia (Kings County, NSNH) to central Wisconsin (Purrington et al. 2002: 201), south to central Texas (Travis County, UASM) and southern Florida (Peck and Thomas 1998: 22). The records from southern Arizona (Horn 1880e: 181; Wickham 1898: 301) need confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, FL, GA, IL, IN, MA, MD, ME, MI, MO, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TX, VA, VT, WI, WV [AZ]
This species is known only from the type locality in central Texas.
USA: TX
This species is found from southwestern Maine (Majka et al. 2011: 46) to eastern Minnesota (Gandhi et al. 2005: 931), including southernmost Ontario (possibly only as strays) (Lindroth 1968: 821), south to eastern Texas (Nacogdoches County, CMNH) and central Florida (Manatee County, CNC).
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV
This species is known from central Louisiana (Casey 1914: 151) and Texas (Leng 1920: 72; McCulloch County, USNM). One specimen determined by Casey (USNM) from Kansas is known.
USA: LA, TX [KS]
This species is known from southern Florida and several islands of the West Indies (Peck and Thomas 1998: 22).
USA: FL – Bahamas, Cuba, Hispaniola, Jamaica, Puerto Rico
This species ranges from central Texas south at least to southern Tamaulipas in Mexico (LeConte 1858b: 59). The record from “Arizona” (Bousquet and Larochelle 1993: 241) needs confirmation.
USA: TX [AZ] – Mexico
Horn (1880e: 180) regarded Selenophorus palliatus (Fabricius) as synonym of this species.
This species is known from central and southern Florida (Vince Golia pers. comm. 2007; Highland and Palm Beach Counties, CMNH, UASM), southern Mississippi (Hancock and Jackson Counties, Drew A. Hildebrandt pers. comm. 2008; Lago et al. 2002: 202), and southern Texas (Casey 1914: 148; Zapata and San Patricio Counties, CMNH, UASM).
USA: FL, MS, TX
The range of this species extends from western New Brunswick (Webster and Bousquet 2008: 19) to Minnesota (Epstein and Kulman 1990: 215) and South Dakota (Kirk and Balsbaugh 1975: 30), including southern Quebec and Ontario (Lindroth 1968: 824), north to southeastern Manitoba (Roughley et al. 2010: 230), south to southern Texas (Johnson 1978: 67), the Florida Keys (Peck and Thomas 1998: 22), and South Bimini Island in the Bahamas (Ball and Shpeley 1992b: 96).
CAN: MB, NB, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Bahamas
This species is known only from the type series.
USA: AZ
This species is found throughout much of southern United States from North Carolina (Brimley 1938: 128) to the Florida Keys and the Bahamas (Peck and Thomas 1998: 22), west to southeastern California (Andrews et al. 1979: 28) and the Baja California Peninsula (Horn 1894: 312), including southern Illinois (Union and Pope Counties, UASM), south to southeastern Texas (Wickham 1897: 113; Johnson 1978: 68; Cameron and San Patricio Counties, UASM) and San Luis Potosí (UASM). The record from “Virginia” (Bousquet and Larochelle 1993: 241) needs confirmation.
USA: AL, AR, AZ, CA, FL, GA, IL, LA, MS, NC, NM, SC, TX [VA] – Bahamas, Mexico
This species is known from southern Florida (Peck and Thomas 1998: 22, as Selenophorus sinuatus) and several islands of the West Indies (Peck 2011: 13). The record from South Carolina (Ciegler 2000: 103, as Selenophorus mustus) is probably in error.
USA: FL – Antigua, Bahamas, Barbados, Cayman Islands, Cuba, Dominica, Guadeloupe, Guana Island, Hispaniola, Jamaica, Montserrat, Navassa, Puerto Rico, Virgin Islands.
This species is found from Connecticut (Krinsky and Oliver 2001: 218) to southern Montana (Hatch 1933a: 10), south to southern New Mexico (Fall and Cockerell 1907: 162), southern Texas (Johnson 1978: 67), and central Florida (Peck and Thomas 1998: 22), west along the south to southern Arizona (Griffith 1900: 566; Snow 1906b: 163; Snow 1907: 142; Greenlee and Pima Counties, CMNH) and Baja California (Horn 1894: 311).
CAN: ON USA: AL, AR, AZ, CO, CT, DC, FL, GA, IA, IL, IN, KS, LA, MD, MO, MS, MT, NC, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, VA, WI, WV, WY – Mexico
This species ranges from southernmost Ontario (possibly only as strays) to south-central British Columbia (Lindroth 1968: 828), south to northeastern Oregon (Westcott et al. 2006: 9), Durango in Mexico (Ball and Shpeley 1992a: 59), northwestern Mississippi (Bolivar County, Drew A. Hildebrandt pers. comm. 2008), and northwestern Ohio (Lucas County, Harry J. Lee, Jr. pers. comm. 2008).
CAN: AB, BC, MB, ON, SK USA: AZ, CO, IA, IL, KS, MI, MN, MS, MT, ND, NE, NM, OH, OK, OR, SD, TX, UT, WI, WY – Mexico
This species is known only from the type locality in western Mississippi.
USA: MS
This species is known from the Huachuca Mountains and Yuma County in southern Arizona (Schaeffer 1910: 403) and from Riverside County in southeastern California (Andrews et al. 1979: 28, as Selenophorus semirufus). The record from “Texas” (Csiki 1932a: 1201) is probably in error.
USA: AZ, CA
This species is known only from the type series.
USA: CO
This species is known from the type locality in southern Arizona, from southern New Mexico (Dona Ana County, CMNH), and from southern Sonora in Mexico (UASM).
USA: AZ, NM – Mexico
This species is known so far from Florida and several islands of the West Indies (Peck and Thomas 1998: 22; Peck 2009a: 13), from south-central Louisiana (Saint Landry Parish, Igor M. Sokolov pers. comm. 2009), as well as from southern Texas (Johnson 1978: 67; San Patricio and Cameron Counties, UASM), southern Mexico (Putzeys 1878: 33), and Colombia (Martínez 2003: 9). The record from Georgia (Fattig 1949: 48, as Selenophorus depressulus) needs confirmation. The species is adventive on several islands of Hawaii (Liebherr 2009: 403).
USA: FL, LA, TX [GA] – Bahamas, Barbados, Cayman Islands, Cuba, Colombia, Dominican Republic, Jamaica, Leeward Islands, Mexico, Puerto Rico, Windward Islands
This species is known only from the type locality in southern Florida.
USA: FL
This peculiar species is known from southern Kansas (Barber County, Robert L. Davidson pers. comm. 2012), west-central Oklahoma (Horn 1880e: 181), and northeastern Texas (Hunt County, CNC).
USA: KS, OK, TX
The systematic position of this species is not settled. It was not listed by Noonan (1985) in his classification and names of the Selenophori group. The species is markedly distinctive in its structural characters.
Twenty-eight species in North America (19 species) and Middle America (11 species).
This genus has never been revised and such study is much needed. More than 60% of the currently valid North American species have been described by Casey and some of them will certainly fall in synonymy.
This species is known from western Missouri (Vernon County, UASM), Texas (several Counties, UASM), southeastern New Mexico (Chaves County, UASM), and southeastern Arizona (Cochise County, UASM).
USA: AZ, MO, NM, TX
This species ranges from eastern Wyoming (Lavigne 1977: 45) to southeastern Oregon (Westcott et al. 2006: 8), south to southern California (Fall 1901a: 50) and south-central New Mexico (Fall and Cockerell 1907: 161).
USA: AZ, CA, CO, ID, NM, NV, OR, UT, WY
This species is known from southeastern Colorado (Bent County, CNC), central New Mexico (Bernalillo County, CNC), and southern Arizona (Cochise and Santa Cruz Counties, UASM).
USA: AZ, CO, NM
This species is known from southern Arizona (Horn 1891: 34; Griffith 1900: 565; Cochise and Maricopa Counties, UASM), New Mexico (Hidalgo and Quay Counties, CMNH; Luna County, UASM; Fall and Cockerell 1907: 161), western Texas (Brewster County, UASM), and Coahuila, Sonora, and Baja California Norte in Mexico.
USA: AZ, NM, TX – Mexico
This species is known, besides the type series, from a few specimens collected in Cochise County in southeastern Arizona (Robert L. Davidson, pers. comm. 2012).
USA: AZ
This species is known from northern Texas only (Casey 1924: 120).
USA: TX
This species ranges from Kansas (Knaus 1907: 233; Trego, Scott, and Barber Counties, CMNH, CNC) to southeastern Utah (San Juan County, UASM), south to northern Sonora (Bates 1884: 276), Coahuila (UASM), and southeastern Texas (San Patricio County, UASM).
USA: AZ, CO, KS, NM, OK, TX, UT – Mexico
This species is known only from the type locality in central Texas.
USA: TX
This species is known only from Cochise County in southeastern Arizona (UASM).
USA: AZ
This species is known from southeastern Arizona (Cochise County, UASM) and southern New Mexico (Casey 1924: 119).
USA: AZ, NM
This species ranges from southeastern New Hampshire (Rockingham County, Ross T. Bell pers. comm. 1992) to the Okanagan Valley in south-central British Columbia (Lindroth 1968: 831), south to northern Oregon (Union, Wallowa, and Wasco Counties, James R. LaBonte pers. comm. 2009), northern Utah (Salt Lake County, Foster F. Purrington pers. comm. 2009), New Mexico (Fall and Cockerell 1907: 161), western and central Texas (Lee and Brewster Counties, MCZ; Lindroth 1968: 831), southwestern Alabama (Clarke County, CMNH), central Georgia (Fattig 1949: 49), and eastern South Carolina (Ciegler 2000: 104).
CAN: BC, ON USA: AL, AR, AZ, CO, DC, GA, IA, ID, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, MT, NC, NE, NH, NJ, NM, OH, OK, OR, PA, SC, SD, TN, TX, UT, VA, WA, WI, WY
This species is known only from the type locality in western Texas.
USA: TX
This species is known only from the type locality in western Texas.
USA: TX
This species is known from southern Arizona (Cochise County, UASM).
USA: AZ
This subspecies is known only from the type locality in southeastern Arizona.
USA: AZ
This subspecies ranges from southern Arizona (Wickham 1898: 301; Griffith 1900: 565; Snow 1906b: 162; Snow 1907: 142) and northern Sonora in Mexico (Bates 1884: 276, as Discoderus ?, see Horn 1886a: ix) to western Texas (Brewster and Jeff Davis Counties, CMNH, UASM).
USA: AZ, NM, TX – Mexico
This species is known from Arizona (Cochise, Gila, Graham, Pima, Santa Cruz, and Yavapai Counties, UASM; Casey 1914: 160) and western Texas (Terrell and Jeff Davis Counties, UASM).
USA: AZ, TX
This species is known only from the type series.
USA: AZ
This species has been recorded only from Santa Fe in New Mexico (Fall and Cockerell 1907: 161).
USA: NM
This species is known only from the type locality in northeastern Texas.
USA: TX
Six species in North America (three species), Middle and South America (five species), and the West Indies (two species, one of them endemic).
Ball et al. (1991) revised the species.
Ball et al. (1991: 942) postulated that the Neotropical genus Trichopselaphus Chaudoir (eight species) is the sister-group to this genus and that Anisocnemus Chaudoir (two Neotropical species) is the sister-group to {Trichopselaphus + Stenomorphus}.
[angustatus group]
This subspecies is known from a small area in southeastern Arizona and southwestern New Mexico [see Ball et al. 1991: Fig. 24].
USA: AZ, NM
Besides the two subspecies found in North America, two other subspecies are known, one (Stenomorphus californicus manni Darlington) from Haiti and the other (Stenomorphus californicus darlingtoni Ball and Shpeley) from Central America.
This subspecies ranges from northern Missouri to southern Nebraska (Adams County, Foster F. Purrington pers. comm. 2009), south to the Yucatán Peninsula, west to the southern portion of the Baja California Peninsula, east to central Alabama [see Ball et al. 1991: Fig. 24]. One specimen is known from Clemson, South Carolina (Ciegler 2000: 104).
USA: AL, AR, AZ, CA, KS, LA, MO, MS, NE, OK, TX [SC] – Mexico
[convexior group]
This species ranges from southern Arizona southwards along the Pacific Versant of Mexico to central Jalisco [see Ball et al. 1991: Fig. 21].
USA: AZ – Mexico
[sinaloae group]
This species ranges from southeastern Arizona and southwestern New Mexico south along the Pacific Versant of Mexico to Guerrero; it is also known from the southern parts of the Baja California Peninsula [see Ball et al. 1991: Fig. 23]. As pointed out by Ball et al. (1991: 951) the record from central Colorado based on a male is questionable but not impossible.
USA: AZ, NM [CO] – Mexico
About 250 species (Lorenz 2005: 380-382), more than 85% of them inhabiting Asia, arrayed in six subgenera: Amaroschesis Tschitschérine (about 55 species), Bellogenus Clarke (about 45 species), Harpaloxenus Schauberger (12 species), Iridessus (six species), Lampetes Andrewes (nine species), and Trichotichnus s.str. (about 120 species). The Western Hemisphere has only four species, all endemic to eastern North America.
Lindroth (1968) reviewed all four North American species, one of them (Trichotichnus fulgens) under the genus Harpalus.
About 120 species in the Nearctic (two eastern species), Australian (six species), Oriental (about 25 species), and Palaearctic (about 90 species, the vast majority in its eastern part) Regions.
The range of this species extends from southern Quebec (Serge Laplante pers. comm. 2002) to southeastern South Dakota (Kirk and Balsbaugh 1975: 30), south to “Texas” (Lindroth 1968: 819) and central South Carolina (Ciegler 2000: 105).
CAN: ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, VT, WI, WV
Say (1830c: 19) noted that “H[arpalus] iricolor, Say, has been recently described by Dejean under the name of dichrous.” I have not found any species described by Say under the name Harpalus iricolor. Say’s name has been listed as a junior synonym of Trichotichnus dichrous (Dejean) in several checklists but it is a nomen nudum as pointed out by Lindroth and Freitag (1969: 353).
Trichotichnus dichrous (Dejean). This eastern species is color-dimorphic, a phenomenon rarely seen in North American carabids. While most adults have the head and pronotum reddish-yellow and the elytra much darker, a small number of adults are known to be uniformly piceous. When he described the species, Dejean had a single specimen of the common morph. The strong color contrast between the forebody and elytra prompt him to propose the name dichrous, from the Greek prefix di- (two) and chroma (color of the skin).
The range of this species extends from western New Brunswick (Webster and Bousquet 2008: 19) to southwestern Wisconsin (Grant County, Peter W. Messer pers. comm. 2008), south to northern Arkansas (Boone County, UASM) and eastern Georgia (Fattig 1949: 47). The records from “Florida” and “Minnesota” (Bousquet and Larochelle 1993: 244) need confirmation.
CAN: NB, ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KY, MA, MD, ME, MI, MO, NC, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WI, WV [FL, MN]
Six species in the Nearctic (two species) and east Palaearctic (four species) Regions.
Kataev (in Ball and Bousquet 2000: 96) revalidated Iridessus Bates as a subgenus, considered by most authors as a synonym of Trichotichnus.
This species ranges from west-central Maine (Majka et al. 2011: 46) and southern Quebec (CNC) to east-central Minnesota (Gandhi et al. 2011: 673), north to Kapuskasing in central Ontario (CNC), south at least to northeastern Kansas (Popenoe 1877: 24), central Arkansas (Pulaski County, Robert L. Davidson pers. comm. 2008), southwestern Mississippi (Hinds County, CMNH), northern Georgia (Fattig 1949: 52), and eastern South Carolina (Ciegler 2000: 105).
CAN: ON, QC USA: AR, CT, DC, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, PA, RI, SC, VA, VT, WI, WV
This eastern species extends from Massachusetts (Middlesex County, Peter W. Messer pers. comm. 2008) to northeastern Kansas, including southernmost Ontario (Bousquet 1987a: 130) and southeastern Iowa, south to southeastern Texas and southern Florida, west to western Texas [see Noonan 1991: Fig. 286]. The record from southern Wisconsin (Rauterberg 1885: 21, as Harpalus nitidulus) needs confirmation.
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MO, MS, NC, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WV [WI]
This species was included in the genus Harpalus Latreille by Lindroth (1968: 811) and Noonan (1991: 136) but placed in the genus Trichotichnus Morawitz by Ball and Bousquet (2000: 96) on information provided in litteris by Boris Kataev.
Nine species in eastern Mexico, one of them extends into southeastern Texas.
Ball (1970, 1976a) revised the species and provided a key (Ball 1976a: 62-63) for their identification.
This species is known only from southeastern Texas and northeastern Mexico (Ball 1970: 120).
USA: TX – Mexico
One North American species in the temperate regions.
The species is covered in Lindroth’s (1968: 744-745) monograph.
The range of this species extends from Long Island, New York (Notman 1928: 244), to southeastern Alberta (Lindroth 1968: 745), south to southern Arizona (Cochise, Greenlee and Pima Counties, CNC, UASM), Durango (Bates 1891a: 241) and southern Coahuila (UASM) in Mexico, western Alabama (Pickens and Tuscaloosa Counties, UASM), and “Florida” (Leng 1920: 70, as Cratacanthus subovalis).
CAN: AB, SK USA: AL, AR, AZ, CO, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, MT, NC, ND, NE, NJ, NM, NY, OH, OK, PA, SC, SD, TN, TX, UT, VA, WI, WY – Mexico
Cratacanthus dubius (Palisot de Beauvois). This widely distributed and morphologically variable species was described by the French naturalist traveler Ambroise Marie Joseph Palisot, Baron de Beauvois, one of the first entomologists to collect and describe American insects. On North American soil, Palisot de Beauvois collected from the Ohio River in the west to Savannah in Georgia. Unfortunately most of his collection was lost in a shipwreck off Nova Scotia in 1798 and his subsequent published descriptions were based on his notes and drawings.
About 825 species in the Nearctic (13 species), Neotropical (16 species in Mexico), Oriental (about eight species), Palaearctic (about 775 species), and Afrotropical (17 species in Ethiopia) Regions, arrayed in six subtribes: Atranopsina (about 100 species), Calathina (about 185 species), Dolichina (17 species), Pristosiina (about 65 Asian species), Synuchina (about 100 species), and Sphodrina (about 360 species).
Several authors, including Lindroth (1966) and Ball and Bousquet (2000), placed the sphodrines within the tribe Platynini.
About 100 species in Europe (about 60 species), northern Africa (about 20 species), western Asia, including India (about 25 species), and eastern North America (one species); most species occur in the Mediterranean region.
Lindroth (1968: 653) listed the genus Pseudamara in the tribe Amarini (= Zabrini) and pointed out that the sole species of the genus was “not very closely related to Amara”. Recently Hieke (2010) concluded that Pseudamara belongs to the subtribe Atranopsina, of the Sphodrini, and was probably closely related to the genus Amaroschema Jeannel, which includes a single species in the Canary Islands.
One species in the boreal and temperate regions of eastern North America.
Lindroth (1968: 653-654) treated the species in his monograph.
This species is found from Cape Breton Island (Bousquet 1987a: 128) to northern Minnesota (Gandhi et al. 2005: 929), south to southwestern North Carolina (Macon County, MCZ).
CAN: NB, NS (CBI), ON, PE, QC USA: IL, MA, ME, MI, MN, NC, NH, NY, OH, PA, RI, VA, VT, WI, WV
About 185 species (Lorenz 2005: 396-399) placed in the genus Calathus. Based on recent molecular sequence analyses (Ruiz et al. 2009), Synuchidius Apfelbeck and Thermoscelis Putzeys, often ranked as distinct genera, are considered subgenera of Calathus.
About 185 species (Lorenz 2005: 396-399) in North America (eight species, one of them adventive), Mexico (14 species), Ethiopia (17 species), and the Palaearctic Region (about 145 species) arrayed in 11 subgenera: Calathus s.str. (50 species), Lauricalathus Machado (19 species endemic to the Canary Islands), Trichocalathus Bolívar y Pieltain (three species endemic to the Canary Islands), Neocalathus (45 species), Amphyginus Haliday (two European species), Tachalus Ball and Nègre (one Mexican species), Bedelinus Ragusa (one species in Europe and northern Africa), Lindrothius Kurnakov (13 species restricted to Caucasia), Acalathus (11 species), Synuchidius Apfelbeck (one southeast European species), and Thermoscelis Putzeys (one Caucasian species). About 40 species, including all those from Ethiopia, are currently unplaced (Lorenz 2005: 398-399).
Ball and Nègre (1972) revised the Western Hemisphere species and provided a key for their identification.
Fifty species (Lorenz 2005: 396-397) in Europe, the Middle East, and Nepal. Most of the species are endemic to the Mediterranean region. One species is adventive in western North America.
Fuscocalathus Nègre (1969: 7) is usually cited as a junior synonym of this subgenus following Ball and Nègre (1972: 510). However, the name is a nomen nudum since Nègre (1969) failed to designate a type species for his new taxon.
This Palaearctic species is adventive in North America where it is known from southwestern British Columbia (Lindroth 1966: 543) to northwestern Oregon (Westcott et al. 2006: 7). The first inventoried specimen collected on this continent was found in the vicinity of Vancouver, British Columbia, in 1928 (Hatch 1949c: 151).
CAN: BC USA: OR, WA – Adventive
Forty-five species in North America (six species), Mexico (13 species), and the Palaearctic Region (27 species, no endemic species in eastern Asia).
Gañán and Novoa (2006) considered Neocalathus as a synonym of Amphyginus Haliday, 1841 (type species: Carabus piceus Marsham, 1802 (= Calathus rotundicollis Dejean, 1828)). If this approach is followed, then Amphyginus is the valid name for this subgenus (see Alonso-Zarazaga 2006).
The record of Calathus ambigens Bates from Huachuca Mountains in southeastern Arizona (Schaeffer 1910: 394) is probably in error since the species is recorded only from Durango and Chihuahua in Mexico by Ball and Nègre (1972: 461-462).
This species ranges from the southern Gulf Islands of southwestern British Columbia (James C. Bergdahl pers. comm. 1993) to southern Idaho, south to northern Utah and Mono County in the Sierra Nevada of California [see Ball and Nègre 1972: Fig. 53].
CAN: BC USA: CA, ID, NV, OR, UT, WA
This species is found from Cape Breton Island in Nova Scotia (Lindroth 1966: 544) to western North Dakota (Tinerella 2003: 636), south to Nebraska, northern Alabama, and northeastern Georgia (Fattig 1949: 32) [see Ball and Nègre 1972: Fig. 51]. The records from Prince Edward Island (Bousquet and Larochelle 1993: 245, see Majka et al. 2008: 133) and eastern Kansas (Popenoe 1877: 23) need confirmation; those from “Florida, ” Texas (Wickham 1896c: 133; Knaus 1905b: 348), and New Mexico (Fall and Cockerell 1907: 159) are likely in error (see Ball and Nègre 1972: 486).
CAN: NB, NS (CBI), ON, QC USA: AL, CT, DC, DE, GA, IA, IL, IN, KY, MA, MD, ME, MI, MN, MO, NC, ND, NE, NH, NJ, NY, OH, PA, RI, SC, SD, TN, VA, VT, WI, WV [KS, PE]
The range of this transamerican species extends from Newfoundland (Lindroth 1955a: 112-113) to Alaska, including the Aleutian Islands (Lindroth 1966: 545), south to Washington (Hatch 1953: 131), central Arizona, southern New Mexico, the Black Hills in southwestern South Dakota (Kirk and Balsbaugh 1975: 23), and the Adirondack Mountains in northeastern New York (Notman 1928: 230) [see Ball and Nègre 1972: Fig. 53].
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CO, ID, ME, MI, MN, MT, ND, NH, NM, NY, SD, UT, VT, WA, WI, WY
The range of this species extends from southern Quebec (Larochelle 1975: 69) to southeastern Utah, south to southeastern Arizona, southern Texas, and northern Florida [see Ball and Nègre 1972: Fig. 52]. The record from New Brunswick (see Majka et al. 2007: 11) is in error (Christopher G. Majka pers. comm. 2009).
CAN: ON, QC USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, TN, TX, UT, VA, VT, WI, WV, WY
This species is confined, as far as known, to mountains in southeastern Arizona (Ball and Nègre 1972: 505).
USA: AZ
The range of this subspecies extends from southwestern British Columbia, including Vancouver Island, south to Humboldt County in northern California [see Ball and Nègre 1972: Fig. 40].
CAN: BC (VCI) USA: CA, OR, WA
This subspecies occurs from eastern Washington and western Idaho south to central California [see Ball and Nègre 1972: Fig. 40].
USA: CA, ID, OR, WA
This subspecies ranges from Mendocino County in California south to the northern parts of the Baja California Peninsula and southern Arizona (Ball and Nègre 1972: 482, Fig. 40); it also occurs on the Channel Islands. The record from “Nevada” (Ball and Nègre 1972: 482) is doubtful. The subspecies is adventive in Hawaii (Liebherr and Zimmerman 2000: 465).
USA: AZ, CA (CHI) [NV] – Mexico
According to Ball and Nègre (1972: 482), this form intergrades with the ignicollis form along the Great Central Valley of California.
Eleven species in North America (one species) and the Chinese provinces of Kansu, Szechwan, and Tsinghai, and the Autonomous Region of Tibet (ten species).
This taxon has been listed as a distinct genus in the subtribe Dolichina by Hovorka and Sciaky (2003: 529). However, Ruiz et al. (2009) concluded from molecular sequence analyses that the taxon is closely related to Calathus and should be ranked as a subgenus of Calathus.
This species ranges in the east from eastern Newfoundland to Lake Superior, south to southwestern New York, and in the west from Kodiak Island in Alaska south to northern Oregon, east-central Nevada, and southern New Mexico along the Rocky Mountains, east to the Black Hills in southwestern South Dakota (Pennington County, CNC) [see Ball and Nègre 1972: Fig. 1]. The records from “California” (Lindroth 1955a: 114) and “Vermont” (Hamilton 1894a: 11) need confirmation.
FRA: PM CAN: AB, BC, LB, NB, NF, ON, QC, SK USA: AK, AZ, CO, ID, ME, MI, MN, MT, ND, NH, NM, NV, NY, OR, SD, UT, WA, WY [CA, VT]
About 100 species (Lorenz 2005: 400-401) in North America and Mexico (three species) and the Palaearctic Region (about 95 species), arrayed in four genera: Parabroscus Lindroth (two species from Japan and Taiwan), Nipponosynuchus Morita (one Japanese species), Synuchus (about 80 species), and Trephionus Bates (14 Japanese species).
About 80 species in the Nearctic (two species), Neotropical (one species from northwestern Mexico), and Palaearctic (77 species, of which one is Eurasian, nine are from the Himalayas, and the remaining from eastern Asia) Regions.
Lindroth (1966: 550-552) treated both North American species.
This species is found in the southern part of the Rocky Mountains in western Utah (Casey 1924: 87, as Pristodactyla juabitica), Colorado (Wickham 1902: 238; Lindroth 1956b: 523), New Mexico (LeConte 1854b: 38; Fall and Cockerell 1907: 159; Lindroth 1956b: 523), and Arizona (Wickham 1898: 300; Casey 1913: 160, as Pristodactyla arizonica; UASM).
USA: AZ, CO, NM, UT
This species ranges from Newfoundland (Lindroth 1955a: 115-116) to southwestern British Columbia (Lindroth 1966: 551), south to western Washington (Hatch 1953: 132), northern Idaho (Hatten et al. 2011: 325), eastern Kansas (Cook and Holt 2006: 2313), and northern Georgia (Fattig 1949: 32). The record from southwestern Colorado (Wickham 1902: 238) needs confirmation.
FRA: PM CAN: AB, BC, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VA, VT, WA, WI, WV [CO]
About 360 species (Lorenz 2005: 401-407), all but one (Miquihuana rhadiniformis Barr from Mexico) native to the Palaearctic Region. Two species are adventive in several regions of the world.
Casale (1988) revised all species then known.
About 195 species (Lorenz 2005: 403-406) in Europe, northern Africa, western and central Palaearctic Asia, and the Himalayas arrayed in 12 subgenera. Two species are adventive in several parts of the world.
Lindroth (1966: 549-550) treated both species found in North America.
Twenty species in Europe (eight species), northern Africa (four species), and Asia (13 species), one of them has been dispersed by trade to several continents and islands.
This species is now subcosmopolitan (Casale 1988: 460). It is adventive in North America and known from southern British Columbia, including Vancouver Island (Lindroth 1966: 550), to southwestern California, east across the Central Valley to the western foothills of the Sierra Nevada (David H. Kavanaugh pers. comm. 2008). The first inventoried specimen collected on this continent was found prior to 1874 since the species is reported by Crotch (1874: 12).
CAN: BC (VCI) USA: CA, OR, WA – Adventive
About 55 species in Europe, northern Africa, western Asia, and the Himalayas. One European species is adventive in North America and India (Casale 1988: 786-787).
This European subspecies is adventive in North America where it is known from Newfoundland (Lindroth 1966: 550; Larson and Langor 1982: 593) and the Maritimes (Lindroth 1966: 550) to the Saint Lawrence Estuary (Larochelle 1975: 98), from three specimens collected in 2005 and 2008 in Boston Harbor, Massachusetts (Davidson et al. 2011: 518), and from one specimen collected in 1980 in southeastern British Columbia (Bousquet 1987a: 125). The first inventoried specimen collected on this continent was found in Nova Scotia prior to 1894 (Bousquet 1992a: 507).
FRA: PM CAN: BC, NB, NF, NS, PE, QC USA: MA – Adventive
The subspecies Laemostenus terricola punctatus Dejean is restricted to eastern Europe.
Worldwide, with about 2, 670 species arrayed in 170 genera (Lorenz 2005: 407-437, excluding Enoicina and Atranus). The Northern Hemisphere has about 750 species (28% of the world fauna) and North America alone about 160 (6%).
Twenty-one species in temperate areas of the Nearctic (seven species) and Palaearctic (14 species, only one extending to eastern Asia) Regions.
Casey (1913: 169-171) published a key to all North American species except Olisthopus micans but the genus is in need of a taxonomic revision.
[micans group]
This species has been recorded from Rhode Island (Casey 1913: 171) and from Richland County in South Carolina (Ciegler 2000: 106).
USA: RI, SC
The range of this species extends from southern Quebec (Larochelle 1975: 96) to “Iowa” (Jaques and Redlinger 1946: 295), south to southern Louisiana (Calcasieu Parish, CNC) and northern Florida (Baker County, CMNH). The record from “Wisconsin” (Bousquet and Larochelle 1993: 248) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, MI, MO, MS, NH, NJ, NY, OH, PA, RI, SC, VA, VT [WI]
[parmatus group]
This species is known only from the type series.
USA: IL
This species is known only from the holotype.
USA: RI
This species is known only from the original two specimens.
USA: IN
Lindroth (1966: 553) listed this form in synonymy with Olisthopus parmatus Say but Bousquet and Larochelle (1993: 18) treated it as a distinct species.
This species ranges from “Nova Scotia” (Larochelle and Larivière 1990a: 30) to eastern Minnesota (Epstein and Kulman 1990: 215; Wickham 1896c: 134), south to northern Oklahoma (French et al. 2001: 229), Arkansas (Logan County, CNC), the Florida Panhandle (Leng 1915: 582), and southwestern Georgia (Fattig 1949: 35). The record from northern Colorado (Armin 1963: 158) needs confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, VA, VT, WI, WV [CO]
This species is known only from the holotype collected in central Louisiana.
USA: LA
Eleven Mexican species, of which one extends into southern United States.
Liebherr (1991b) revised the species.
This species extends from east-central Arizona and southwestern Texas south to the state of San Luis Potosí in central Mexico [see Liebherr 1991b: Fig. 269]. Only two localities have been reported for the United States.
USA: AZ, TX – Mexico
Eight species in North America (four species, only one of them endemic), Middle America (two species, no endemism), the West Indies (one endemic Cuban species), South America (one species, no endemism), Asia (four species, three of them endemic), and Europe (two species, no endemism).
Liebherr (1991b) revised the species. One species, Sericoda balli Schmidt, was subsequently describedfrom Pakistan.
[bogemannii group]
This species is widely distributed in the Western Hemisphere ranging from southeastern Manitoba to central Alaska, south to northern Oregon and through the Rocky Mountains, Transverse Volcanic, Central America, and the Andes to southern Ecuador, east in South America to western Venezuela [see Liebherr 1991b: Fig. 240].
CAN: AB, BC (VCI), MB, NT, SK, YT USA: AK, AZ, CO, ID, KS, MT, NM, OR, WA – Colombia, Costa Rica, Ecuador, El Salvador, Guatemala, Mexico, Panama, Venezuela
This Holarctic species is found in the New World from west-central Alaska south to central California and to southern New Mexico along the Rocky Mountains. Disjunct populations are also known from Chiapas in Mexico and Guatemala [see Liebherr 1991b: Fig. 230]. In the Old World, the species is known from disjunct populations in Lapland, the Austrian Alps, and Burma [see Liebherr 1991b: Fig. 231].
CAN: AB, BC (VCI), YT USA: AK, CA, CO, ID, MT, NM, OR, WA – Guatemala, Mexico – Holarctic
This Nearctic species ranges from Newfoundland to Vancouver Island (Casey 1920: 97, as Sericoda insulina), north to southern Northwest Territories, south to central Oregon, central Colorado (Wickham 1902: 239), eastern Texas, and coastal Georgia [see Liebherr 1991b: Fig. 229]. One specimen simply labeled from California is known (Liebherr 1991b: 74). There is also one specimen recorded from near Mexico City (Liebherr 1991b: 72) which in my opinion is probably mislabeled.
CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK USA: AL, AR, CO, CT, DC, GA, IA, ID, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NH, NJ, NY, OH, OR, PA, RI, SC, TN, TX, VA, VT, WA, WI, WV [CA]
[quadripunctata group]
This Holarctic species ranges in North America from central Alaska to Newfoundland (Lindroth 1955a: 117), south to eastern North Carolina and western Tennessee along the Appalachian Mountains, to northern New Mexico along the Rocky Mountains, and to central Washington [see Liebherr 1991b: Fig. 221]. In the Palaearctic Region, the range of the species extends from Great Britain to the Kamchatka Peninsula, south to the Philippines Islands, the Himalayan Mountains, and Italy in Europe [see Liebherr 1991b: Fig. 222].
CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: AK, ID, IL, IN, MA, ME, MI, MN, MT, NC, NH, NJ, NM, NY, PA, SD, TN, VT, WA, WI – Holarctic
Sericoda quadripunctata (DeGeer). This widely distributed Holarctic species is attracted to fire. The adults are often found in great numbers just after forest fires when the ashes are still hot. This behavior is shared by the other species in the genus and a few other carabids, such as Harpalus laticeps and Harpalus laevipes. The presence of charcoal is probably a stimulus for oviposition. Henri Goulet was unsuccessful in obtaining eggs from Sericoda obsoleta in the laboratory until he added charcoal to his rearing containers.
One eastern North American species in the temperate regions.
Liebherr (1991b) redescribed the species and illustrated some of its structural character states.
This species is known from scattered localities in eastern North America ranging from western Vermont to “Minnesota, ” south to east-central Texas (Riley 2011), southern Louisiana and central Florida [see Liebherr 1991b: Fig. 187]. The record from Sainte-Rose (= Laval) in Quebec (Hausen 1891b: 159) needs confirmation.
CAN: ON USA: AL, AR, DC, FL, IA, IL, IN, KY, LA, MD, MN, MS, NC, NY, OH, PA, SC, TX, VA, VT, WI, WV [QC]
Fourteen species in the Nearctic (three species) and Palaearctic (ten species) Regions and one species (Anchomenus capensis Liebherr) endemic to Baja California. These species are arrayed in two subgenera: Anchodemus Motschulsky (three Palaearctic species) and Anchomenus s.str. (11 species).
Liebherr (1991b) revised the species except for three Palaearctic species of the subgenus Anchomenus s.str.
Eleven species in North America (three endemic species), Baja California (one endemic species), Asia (seven species, four of them endemic), Europe (three species), and northern Africa (one species).
This taxon is found from the Rocky Mountains in southwestern Alberta to south-central British Columbia, south to central Oregon and south-central Idaho [see Liebherr 1991b: Fig. 200].
CAN: AB, BC USA: ID, MT, OR, WA
This western species ranges from central Oregon to Baja California Norte, including west-central Nevada [see Liebherr 1991b: Fig. 200].
USA: CA (CHI), NV, OR – Mexico
This western species is found from west-central British Columbia south to northern California [see Liebherr 1991b: Fig. 187].
CAN: BC USA: CA, OR, WA
Forty-nine species in North America (40 species) and Mexico (11 species) arrayed in six species groups.
Barr (1960b) revised the cavernicolous species and provided a key for their identification, but several new species have been described since. Barr (1974b) published a key to the species groups and a revision of the species of the subterranea group. Five new species in this group have been described subsequently by Reddell and Cokendolpher (2001, 2004) and Reddell and Dupérré (2009). Barr (1982) also revised the Mexican species of Rhadine. A taxonomic revision of the other five species groups is needed.
[dissecta group]
This species is known only from the type series collected in northwestern New Mexico.
USA: NM
This species is known from a few localities in southeastern Oregon (Harney County, James R. LaBonte pers. comm. 1992), southeastern Washington, and southern Idaho (Hatch 1953: 146; Stafford et al. 1986: 288). The record from northern Colorado (Armin 1963: 147) needs confirmation.
USA: ID, OR, WA [CO]
This species is known only from the type series collected in southeastern Arizona.
USA: AZ
This species is known from “Nebraska” (LeConte 1863c: 8) to southwestern Colorado (LeConte 1879b: 54; Wickham 1902: 238), south to northern Arizona (Coconino County, CMNH; Snow 1906b: 162), the Sacramento Mountains in south-central New Mexico (Fall and Cockerell 1907: 159), and “Texas” (LeConte 1863c: 8).
USA: AZ, CO, NE, NM, TX
This species is found from southern Saskatchewan and southern Alberta (Lindroth 1966: 647) south to Utah (Barr 1965b: 141), northeastern Colorado (Bell 1971: 52), and South Dakota (Kirk and Balsbaugh 1975: 25). The record from “Nebraska” (Bousquet and Larochelle 1993: 251) needs confirmation.
CAN: AB, SK USA: CO, ID, ND, SD, UT, WY [NE]
Rhadine lindrothi Barr. This species, named after Carl H. Lindroth, is the only one of the genus Rhadine that reaches as far north as southern Canada. Ecologically, members of the genus can be divided into two groups, one that includes cavernicolous species and the other that includes surface dwelling species like Rhadine lindrothi. Most species are more or less depigmented and with relatively long legs.
This species is known only from the type locality in northeastern New Mexico.
USA: NM
This species is known from many specimens collected in south-central Texas.
USA: TX
This species is known from a few specimens collected in southeastern Colorado and northwestern Texas (Barr 1960b: 50).
USA: CO, TX
This species is known only from the syntype collected in central Colorado.
USA: CO
[jejuna group]
This species ranges from eastern Washington to southwestern Wyoming (Parmenter and MacMahon 1984: 26), south to northern Arizona (Wichkam 1896a: 157) and the Sierra Nevada in California (Casey 1913: 166). The records from Colorado (Cockerell 1893: 72; Wickham 1902: 238) need confirmation.
USA: AZ, CA, ID, NV, OR, WA, WY [CO]
A preliminary study of the type specimens of Rhadine gracilenta Casey, Rhadine plumasensis Casey, and Rhadine tenuipes Casey did not show any significant external differences from members of Rhadine jejuna (LeConte).
This species is known only from the type locality in southeastern Washington (Hatch 1953: 146).
USA: WA
[larvalis group]
This species ranges from southwestern Pennsylvania (Hamilton 1895: 350, 379; Ehrman 1900: 499) to southeastern Wisconsin (Rauterberg 1885: 14), south to northern Alabama (Jeannel 1949b: 95; Barr 1960b: 47) and northern Georgia (Fattig 1949: 32). The record from southwestern Colorado (Wickham 1902: 238) must be in error; that from “Arkansas” (Bousquet and Larochelle 1993: 250) needs confirmation.
USA: AL, DC, GA, IL, IN, KY, MD, NC, OH, PA, TN, VA, WI, WV [AR]
This species is found from the Florida Panhandle (Choate and Rogers 1976: 364) to central Arkansas (Barr 1982a: 178), north to east-central Missouri (LeConte 1846b: 219). The records from northern Colorado (Wickham 1902: 238), northern Arizona (Wickham 1896a: 157), southern Montana (Hatch 1933a: 9), and southwestern Pennsylvania (Ehrman 1900: 500) are probably in error.
USA: AL, AR, FL, IL, MO, MS, TN
This species is known only from the original specimens collected in Fincher’s Cave, near Fayetteville, Arkansas (Barr 1960b: 49).
USA: AR
[nivalis group]
This species has been recorded from several localities in New Mexico (Fall and Cockerell 1907: 159; Casey 1913: 163).
USA: NM
This species is known from Greenlee (Casey 1913: 164) and Coconino Counties (CMNH) in southeastern and northern Arizona.
USA: AZ
This species is known only from the type series collected in northwestern New Mexico.
USA: NM
[perlevis group]
This species is known only from the original specimens collected in the White Mountains, eastern California.
USA: CA
This species is known from a number of caves in Sutton, Edwards, and Pecos Counties, western Texas (Barr 1960b: 52).
USA: TX
This taxon has been considered a subspecies of Rhadine araizi by Bolívar y Pieltain and Hendricks (1964: 6) but treated as a distinct species by Barr (1982a: 183-184).
This species is known from a number of caves in Kerr, Edwards, Uvalde, and Bexar Counties, southern Texas (Barr and Lawrence 1960: 143; Reddell and Cokendolpher, 2004: 157).
USA: TX
This species is known only from the type locality in western Texas.
USA: TX
This species is known from numerous caves in Eddy County, southeastern New Mexico, and Culberson County, western Texas (Cokendolpher and Polyak 2004: 192).
USA: NM, TX
This species has been recorded from southwestern Utah (Tanner 1928: 270), west-central (Bechtel et al. 1983: 474) and southern (Tanner and Tanner 1974: 218) Nevada, “Arizona” (Horn 1892c: 42), and New Mexico as far south as the Sierra Blanca Range (Fall and Cockerell 1907: 159).
USA: AZ, NM, NV, UT
This species is known from southern Arizona and western Chihuahua in northern Mexico (Barr 1982a: 182).
USA: AZ – Mexico
[subterranea group]
This species is found in several caves in central Travis County, Texas (Barr 1974b: 12).
USA: TX
This species is known from several caves on Camp Bullis, a U.S. Army installation in Bexar and Comal Counties, south-central Texas (Reddell and Cokendolpher, 2004: 154-155).
USA: TX
This species is known only from caves near Helotes and Camp Bullis in Bexar County, south-central Texas (Barr 1974b: 16; Reddell and Cokendolpher, 2004: 156).
USA: TX
This species has been listed as endangered by the U.S. Fish and Wildlife Service in December 2000.
This species is known from three caves in or near San Marcos, Hays County, in south-central Texas (Reddell and Dupérré 2009: 112).
USA: TX
This subspecies is known from three caves in southern Bexar County, south-central Texas (Reddell and Cokendolpher, 2004: 157).
USA: TX
This subspecies is known from a few small caves, near Helotes, in the highlands north of San Antonio, southern Texas (Barr 1974b: 23).
USA: TX
A series of eight specimens collected at Bat Cave in Government Canyon, Bexar County, Texas, are reported as hybrids infernalis x ewersi by Barr (1974b: 24). The species Rhadine infernalis has been listed as endangered by the U.S. Fish and Wildlife Service in December 2000.
This species is known only from the type-locality cave in south-central Texas.
USA: TX
This species is known only from the type-locality cave in south-central Texas (Reddell and Cokendolpher, 2004: 161).
USA: TX
This subspecies is known from two caves in south-central Texas (Barr 1974b: 24).
USA: TX
This subspecies is known from a few caves in south-central Texas (Barr 1974b: 25).
USA: TX
This species is known only from Cobb Cavern and Cricket Cave in northern Williamson County, central Texas (Barr 1974b: 11).
USA: TX
This species has been collected in caves in northern Travis and southern Williamson Counties, central Texas (Reddell and Cokendolpher 2001: 110).
USA: TX
This species, also known under the vernacular name “Tooth Cave Ground Beetle”, is listed as an endangered species by the World Wildlife Fund.
This species is known from a number of caves on the Fort Hood Military Reservation in Bell and Coryell Counties, central Texas (Reddell and Cokendolpher 2001: 110).
USA: TX
This species is known only from the type-locality cave on the Jollyville plateau in central Texas.
USA: TX
This subspecies has been found yet only in two caves along Cibolo Creek in southern Texas (Barr 1974b: 16).
USA: TX
This subspecies is known only from two caves in south-central Texas (Barr 1974b: 15).
USA: TX
This subspecies is known from a few caves in Kendall and Comal Counties, southern Texas (Barr 1974b: 13; Reddell and Cokendolpher, 2004: 158).
USA: TX
This species is known yet only from the type-locality cave in south-central Texas (Reddell and Cokendolpher, 2004: 159).
USA: TX
This subspecies is known from several caves in central Texas (Barr 1974b: 8).
USA: TX
This subspecies is known from several caves in southern Williamson and northern Travis Counties, central Texas (Barr 1974b: 7).
USA: TX
This subspecies is found in a few caves in Real and Uvalde Counties, southwestern Texas (Barr 1974b: 20).
USA: TX
This subspecies is known from a few caves in Kerr, Real, and Bandera Counties, southwestern Texas (Barr 1974b: 19).
USA: TX
This species is known for sure only from the type locality in southwestern Nevada. The record from “California” (Bousquet and Larochelle 1993: 251) needs confirmation.
USA: NV [CA]
Eleven species in Texas (one species) and Mexico (ten species).
Barr (1982a) revised the species and provided a key for their identification.
Valentine (1987) believed this genus is closely related to the genera Bryanites Valentine of Western Samoa and Prosphodrus Britton of New Zealand. He proposed the family-group name Prosphodrini to include the three genera.
This species is known from two nearby caves in southwestern Texas.
USA: TX
This species belongs to the boneti species group which includes only the polymorphic Mexisphodrus boneti (Bolívar y Pieltain and Hendrichs) from Nuevo León and Mexisphodrus valverdensis Barr.
Five species in western North America (four species) and the Baja California Peninsula and Guadalupe Island in Mexico (two species, one of them endemic).
Liebherr (1985) revised the species. Subsequently he described a new species, the endemic Baja Californian Tanystoma diabolicum, and provided a key to all species (Liebherr 1989).
According to Liebherr (1989: 184), the genus Rhadine is the sister-group of Tanystoma.
This species ranges through the Coast Ranges from central California south to the Mexican border [see Liebherr 1985: Fig. 27].
USA: CA
This species, also known under the vernacular name “tule Beetle”, ranges from southwestern Oregon south through California, including the Channel Islands, to Baja California Norte and Guadalupe Island [see Liebherr 1985: Fig. 25]. The species is apparently adventive and possibly established at Seattle, Washington (Liebherr 1985: 1194). The record from “Utah” (Bousquet and Larochelle 1993: 252) is likely in error.
USA: CA (CHI), OR, WA – Mexico
This species occurs along the Coast Ranges of California from Siskiyou County to Santa Cruz and Alameda Counties and in the Sierra Nevada between Tulare and Shasta Counties [see Liebherr 1985: Fig. 27].
USA: CA
This species ranges from northwestern Oregon to northern California [see Liebherr 1985: Fig. 27]. An old specimen labeled from western Washington is known.
USA: CA, OR [WA]
Two species endemic to the Canary Islands and one European species, Paranchus albipes, which is adventive in northeastern North America.
The North American species is covered in Lindroth’s (1966: 630-631, as Agonum albipes) monograph.
This Palaearctic species is adventive in North America where it is known from southern Newfoundland, Nova Scotia, New Brunswick, and Maine (Lindroth 1966: 631). The first inventoried specimen collected on this continent was found in Newfoundland before 1835. The Canadian Museum of Nature in Gatineau holds color drawings made by Philip Henry Gosse executed prior to 1834 of insects from Newfoundland and one drawing represents this species; the plates are bound together under the title Entomologia Terrae Novae - P.H. Gosse - 1833.
CAN: NB, NF, NS (CBI) USA: ME – Adventive
Carabus ruficornis Goeze, 1777 is often listed as a synonym of this species (e.g., Lindroth 1966: 630; Bousquet 2003c: 462). However, Goeze (1777: 663) did not propose a new species under such name since he referred to Carabus ruficornis DeGeer, 1774 which is considered a doubtful synonym of Amara aulica (Panzer, 1796) following Schönherr (1806: 181). Goeze (1777: 663) apparently misidentified DeGeer’s Carabus ruficornis.
Four species, two endemic to northern Africa, one to Europe and western Asia, and one to North America.
The North American species is covered in Lindroth’s (1966: 633-634, as Agonum puncticeps) monograph.
Jeanne (1988: 77-78) pointed out the morphological differences between the type species of Oxypselaphus and Anchus and suggested that the two should be treated as generically distinct.
This species ranges from Cape Breton Island (Bousquet 1987a: 126) to the Okanagan river in south-central British Columbia (Lindroth 1966: 634), south to western Oregon (Hatch 1953: 138), northeastern Kansas (Popenoe 1878: 78), and northwestern South Carolina (Anderson County, CNC). The record from “Georgia” (Bousquet and Larochelle 1993: 253) needs confirmation.
CAN: AB, BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CT, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, VA, VT, WA, WI [GA]
About 130 species (Liebherr and Schmidt 2004: 202-203) in the Nearctic (72 species, one adventive), Neotropical (15 species in Middle America, all but two shared with North America), Palaearctic (63 species, of which seven are Holarctic), and Afrotropical (three endemic species) Regions. The species are arrayed in four subgenera, following Liebherr and Schmidt (2004): Europhilus, Platynomicrus, Agonum s.str., and Olisares, all represented in North America. All other subgenera of Agonum listed in Lorenz (2005: 411-412) have been excluded by Liebherr and Schmidt (2004: 152) as most of them “are most likely evolutionary affiliates of the highly diverse and taxonomically confused Platynus-“Colpodes” group of genera.” About 50 species are left unplaced in Lorenz (2005: 412) and some of them may belong to Agonum.
Liebherr (1994) published a key to all species found in the Western Hemisphere as well as a review of the Middle American species. Lindroth (1966, 1969a) covered all North American species except Agonum anthracinum Dejean, Agonum cyanopis Bates, Agonum elongatulum Dejean, Agonum extimum Liebherr, Agonum muiri Liebherr, Agonum pacificum Casey, and Agonum parextimum Liebherr.
Two species in North America, one of them is Holarctic.
The range of this species extends from Saskatchewan to Vancouver Island (Lindroth 1966: 589), north to southeastern Alaska (Lindroth 1966: 589), south to southeastern California (San Bernardino County, CNC; Casey 1920: 65) and southwestern Colorado (Casey 1920: 65, as Anchomenus marcidus).
CAN: AB, BC (QCI, VCI), SK USA: AK, CA, CO, ID, MT, NV, OR, UT, WA, WY
This Holarctic species is known from the Kamchatka Peninsula (Lindroth 1966: 584) and from Alaska (Lindroth 1966: 584) to Newfoundland (Lindroth 1955a: 129), south to northern New Hampshire (Coos County, Donald S. Chandler pers. comm. 2008), southeastern Michigan (Oakland County, UMAA), “Iowa” (Lindroth 1966: 584), east-central South Dakota (Brookings County, MCZ), east-central Colorado (Kiowa County, Foster F. Purrington pers. comm. 2010), “Idaho” (Lindroth 1966: 584), and northern Washington (Okanogan County, UASM). The record from “Pennsylvania” (Bousquet and Larochelle 1993: 255) needs confirmation.
CAN: AB, BC, MB, NB, NF, NT, ON, PE, QC, SK, YT USA: AK, CO, IA, ID, IL, ME, MI, MN, MT, ND, NH, NY, SD, WA, WI, WY [PA] – Holarctic
Twenty-nine species in the Nearctic (16 species) and Palaearctic (17 species, six of them endemic to eastern Asia) Regions. Four species are Holarctic.
[limbatum group]
This species ranges along the west coast from southern British Columbia, including Vancouver Island (Lindroth 1966: 589, as Agonum variolatum), to southern California (Fall 1901a: 46, as Agonum variolatum).
CAN: BC (VCI) USA: CA (CHI), OR, WA
[sordens group]
This species ranges from western Newfoundland (Lindroth 1955a: 125) to eastern Alaska (Lindroth 1966: 590), south to northeastern Washington (Gray 1937: 310, as Agonum lanellum), western Montana (Russell 1968: 63), east-central South Dakota (Kirk and Balsbaugh 1975: 24), and southern Pennsylvania (Hamilton 1895: 322; Allegheny, Lancaster, and Westmoreland Counties, Robert L. Davidson pers. comm. 2008). The records from “Wyoming, ” “Colorado, ” “Iowa, ” “Nebraska, ” and “Kansas” (Bousquet and Larochelle 1993: 253) are probably in error.
CAN: AB, BC, MB, NB, NF, NS, NT, ON, PE, QC, SK, YT USA: AK, CT, IL, IN, MA, ME, MI, MN, MT, ND, NH, NJ, NY, OH, PA, SD, VT, WA, WI
This species is found from Cape Breton Island (Bousquet 1987d: 106) to southern Manitoba (Goulet 1969: 280), south to east-central South Dakota (Larsen and Purrington 2010: 570), northern Illinois (Goulet 1969: 280), and northern West Virginia (Tucker and Preston Counties, CMNH). The record from “Nebraska” (Bousquet and Larochelle 1993: 253) needs confirmation.
CAN: MB, NB, NS (CBI), ON, PE, QC USA: CT, IL, IN, MA, ME, MI, NH, NJ, NY, PA, SD, VT, WI, WV [NE]
A Holarctic species occurring in the Palaearctic Region from Norway to the Far East (Bousquet 2003c: 452) and in North America from Alaska (Lindroth 1966: 573) to Newfoundland (Lindroth 1955a: 126), south to Cape Breton Island (Lindroth 1954c: 307), Vancouver Island (Lindroth 1966: 573), and northern Washington (Glesne et al. 2000: 89). Fossil remnants, dated between 14, 000 and 15, 400 years B.P., have been unearthed in central Iowa (Schwert 1992: 78).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, YT USA: AK, WA – Holarctic
This species is restricted to a small area from Nova Scotia to the Ontario Peninsula (Lindroth 1966: 583), south to central Pennsylvania (Clinton County, CMNH) and New Jersey (Lindroth 1966: 583).
CAN: NB, NS, ON, QC USA: CT, MA, ME, NH, NJ, NY, PA, RI, VT
A Holarctic species ranging from the Kola Peninsula to eastern Siberia and in North America from Alaska to the Hudson Bay in Nunavut and northern Manitoba (Lindroth 1966: 574). Fossil remnants, dated between 14, 000 and 20, 700 years B.P., have been unearthed in east-central Iowa and central Illinois (Schwert 1992: 78).
CAN: BC, MB, NT, NU, YT USA: AK – Holarctic
This rarely collected species is known from Pennsylvania (Carbon, Chester and Greene Counties, Robert L. Davidson pers. comm. 2012), southernmost Ontario (Lindroth 1966: 583) and southern Michigan (Ingham County, CMNH) south to southeastern Texas (Casey 1920: 126) and eastern South Carolina (Kirk 1969: 12; Ciegler 2000: 112).
CAN: ON USA: MI, MO, MS, OH, PA, SC, TN, TX, VA, WV
This widely distributed species ranges from western Newfoundland (Lindroth 1955a: 128) to Kodiak Island, north to the Anderson River delta in northern Northwest Territories (Lindroth 1966: 578), south to the Sierra Nevada in California (El Dorado County, CAS), San Miguel County in New Mexico (Fall and Cockerell 1907: 159, as Platynus ruficornis), northeastern Kansas (Popenoe 1878: 78, as Platynus ruficornis), northern Illinois (Lake County, CNC), and northern West Virginia (Tucker and Randolph Counties, CMNH). The species is also known from the Far East (Bousquet 2003c: 453).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CA, CO, CT, IA, IL, IN, KS, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WV, WY – Holarctic
The range of this species extends from Cape Breton Island (Lindroth 1954c: 307) to Vancouver Island (Lindroth 1966: 581), north to southern Northwest Territories (Bousquet 1987a: 125), south to eastern Washington (Hatch 1953: 145), central Kansas (Trego and Scott Counties, CNC, CMNH), and northwestern Maryland (Garrett County, CMNH).
CAN: AB, BC (VCI), MB, NB, NS (CBI), NT, ON, PE, QC, SK USA: CT, DC, DE, IA, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NY, OH, PA, RI, SD, VT, WA, WI, WV
This species is found from southwestern New Brunswick (Webster and Bousquet 2008: 22) to Minnesota (Epstein and Kulman 1990: 214; Sherburne County, CNC), south to southwestern Nebraska (Lincoln County, Foster F. Purrington pers. comm. 2010) and along the Appalachian Mountains to central Alabama (Lee County, CNC).
CAN: NB, ON, QC USA: AL, CT, IA, IL, IN, KY, MA, MD, ME, MI, MN, NC, NE, NH, NJ, NY, OH, PA, RI, TN, VA, VT, WI, WV
This species ranges from the Nova Scotia Peninsula to south-central British Columbia, north to northern Alberta and Anticosti Island in Quebec, south to southeastern Wisconsin (Messer 2010: 42), the Saginaw Bay area in northern Michigan, and southern Vermont (Lindroth 1966: 574). Fossil remnants, dated about 10, 800 years B.P., have been unearthed in central North Dakota (Ashworth and Schwert 1992: 260).
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: ME, MI, NH, VT, WI
This species ranges from Newfoundland (Lindroth 1955a: 127) to western British Columbia (Lindroth 1966: 576), north to southern Northwest Territories (CNC), south to “Montana” (Casey 1920: 129, as Europhilus collusor), South Dakota (Kirk and Balsbaugh 1975: 25), central West Virginia (Pocahontas County, CMNH), and northern Virginia (Madison and Rappahannock Counties, CNC). The record from New Mexico (type locality of Agonum dilutipenne Motschulsky) is probably in error; those from “Illinois” and “Indiana” (Bousquet and Larochelle 1993: 254) need confirmation.
FRA: PM CAN: AB, BC, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: CT, MA, MD, ME, MI, MN, MT, NH, NY, PA, RI, SD, VA, VT, WI, WV [IL, IN]
This rarely collected species is known from scattered localities from north-central Manitoba to the Gulf Coast of Alaska (Lindroth 1966: 571).
CAN: AB, BC, MB, SK, YT USA: AK
This species ranges from Newfoundland (Lindroth 1955a: 127) to central Alaska (Lindroth 1966: 576), south to “Oregon” (Hatch 1953: 144), southern Colorado (Wickham 1902: 239; LeConte 1879d: 500), northern Illinois (Lake County, CNC), and Maryland (Erwin 1981b: 154). The records from “Iowa” (Jaques and Redlinger 1946: 295), northeastern Kansas (Popenoe 1878: 78), and “North Carolina” (Bousquet and Larochelle 1993: 254) need confirmation.
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, ID, IL, MA, MD, ME, MI, MN, MT, ND, NH, NJ, NY, OR, PA, SD, UT, VT, WA, WI, WV [IA, KS, NC]
This species ranges from New Brunswick (Bousquet 1987a: 125) to the Rocky Mountains in Alberta and eastern Alaska (Lindroth 1966: 579), south to northwestern Wisconsin (Messer 2010: 42), Beaver Islands, Lake Michigan, in northern Michigan (Dunn 1987: 11), and New England (Lindroth 1966: 579).
CAN: AB, MB, NB, NT, ON, PE, QC, SK, YT USA: AK, ME, MI, NH, VT, WI
A Holarctic species widely distributed in the Palaearctic Region from Ireland to the Far East, south to Spain and Italy (Bousquet 2003c: 453). In North America, the taxon ranges from Alaska (Lindroth 1966: 580) to Newfoundland (Lindroth 1955a: 129), south to the District of Columbia (Ulke 1902: 7, as Platynus picipennis), central Kansas (Trego County, CNC), east-central Colorado (Kiowa County, Foster F. Purrington pers. comm. 2010), and “California” (Lindroth 1955a: 129).
CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CA, CO, CT, DC, IA, ID, IL, IN, KS, MA, ME, MI, MN, MT, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SD, VT, WA, WI – Holarctic
Twenty-nine species in the Nearctic (four species, of which one extends into southern Mexico and one is adventive), Palaearctic (24 species, of which one is Holarctic), and Afrotropical (three species in eastern Africa including Madagascar) Regions.
[bicolor group]
A Holarctic species ranging in the Palaearctic Region across Siberia up to the Ob River (Lindroth 1966: 586), south to Kazakhstan and Mongolia (Bousquet 2003c: 454). In North America, the species ranges from Alaska and northern British Columbia to northeastern Alberta (Lindroth 1966: 586). The records from Colorado (Wickham 1902: 238) and New Mexico (Fall and Cockerell 1907: 159) probably refer to Agonum piceolum.
CAN: AB, BC, NT, YT USA: AK – Holarctic
This species ranges from Newfoundland (Lindroth 1955a: 125) to Vancouver Island (Lindroth 1966: 588), north to southern Yukon Territory (Hyland River, UASM), south to west-central California (Lindroth 1966: 587), southwestern Arizona (Maricopa County, UASM), New Mexico (Fall and Cockerell 1907: 159; UASM), and mountains of New England (Lindroth 1966: 587). The record from “Connecticut” (Bousquet and Larochelle 1993: 255) was based on a misidentified specimen of Agonum extensicolle (Krinsky and Oliver 2001: 5); that from “Massachusetts” (Bousquet and Larochelle 1993: 255) needs confirmation.
CAN: AB, BC (VCI), MB, NB, NF, ON, QC, SK, YT USA: AZ, CA, CO, ID, ME, MI, MT, NH, NM, NV, OR, UT, WA, WI, WY [MA]
[muelleri group]
This Palaearctic species is adventive in North America where it is known from west-central Alberta (Emerald Lake, Gerald J. Hilchie pers. comm. 2009) and southern British Columbia, including Vancouver Island (Lindroth 1966: 593), to northwestern California (Humboldt County, James R. LaBonte pers. comm. 1992) in the west and from Newfoundland (Lindroth 1955a: 121) to northeastern Minnesota (Kamal J.K. Gandhi pers. comm. 2008), south to northeastern Iowa (Larsen and Purrington 2010: 570) and northeastern West Virginia (Tucker County, CMNH) in the east. The first inventoried specimen collected in the east was found in Newfoundland around 1840 (Lindroth 1955a: 122) and in the west in 1933 (Lindroth 1966: 593; Leech 1935: 122, as Platynus hardyi). The species is also adventive in Hawaii (Liebherr et al. 2009: 98).
FRA: PM CAN: AB, BC (VCI), NB, NF, NS (CBI), ON, PE, QC USA: CA, CT, IA, MA, ME, MI, MN, NH, NY, OH, OR, PA, RI, VA, VT, WA, WI, WV – Adventive
This widely distributed species ranges from Newfoundland (Lindroth 1955a: 120; Larson and Langor 1982: 594) to western British Columbia (Lindroth 1966: 614), south to Inyo County in California, the Sierra de Miahuatlán in Oaxaca (Liebherr 1994: 32), and northern Georgia (Fattig 1949: 34).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), ON, PE, QC, SK USA: AR, AZ, CA, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Mexico
Seventy-two species in the Nearctic (50 species), Neotropical (14 species in Middle America but only two endemic), and Palaearctic (21 species, one Holarctic) Regions.
[albicrus group]
The range of this species extends from southeastern New Hampshire (Rockingham County, Ross T. Bell pers. comm. 2008) to eastern Kansas (Douglas County, Robert L. Davidson pers. comm. 2009), including southernmost Ontario (Lindroth 1966: 615), south to eastern Oklahoma (Latimer County, UASM), northwestern Louisiana (Bossier Parish, CMNH) and the Florida Panhandle (Jackson County, CNC). The record from “Iowa” (Bousquet and Larochelle 1993: 255) needs confirmation.
CAN: ON USA: AL, AR, CT, FL, GA, IL, IN, KS, LA, MA, MD, MS, NC, NH, NJ, OH, OK, PA, RI, SC, TN, VA [IA]
[cupripenne group]
This species is known from a few localities in the Pacific Northwest from the Queen Charlotte Islands (Kavanaugh 1992: 74) to northwestern Oregon (Clackamas County, James R. LaBonte pers. comm. 1992).
CAN: BC (QCI) USA: OR, WA
This species ranges from central Prince Edward Island (CNC) and New Brunswick (Kouchibouguac National Park, CNC) to central Alaska (Lindroth 1966: 598), south at least to east-central California (Mono County, CAS), southeastern Arizona (Dajoz 2007: 21; UASM), northeastern New Mexico (San Miguel County, UASM), South Dakota including the Black Hills (Kirk and Balsbaugh 1975: 25), and northern Michigan (Dunn 1985a: 8; Casey 1920: 108, as Agonum marquettense). The record from “Kansas” (Horn 1872c: 385, as Platynus chalceus) needs confirmation.
CAN: AB, BC, MB, NB, NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, ID, MI, MN, MT, ND, NM, NV, OR, SD, UT, WA, WI, WY [KS]
Lindroth (1966: 597) treated Agonum uintanum Casey as a subspecies of Agonum cupreum Dejean.
The range of this species extends from Cape Breton Island (Lindroth 1954c: 305) to Vancouver Island (LeConte 1869c: 370), north to west-central Northwest Territories (Bousquet 1987a: 125), south to the Sierra Nevada in California (Casey, 1920: 106, as Agonum tahoense; Dajoz 2007: 16), southeastern Arizona (Greenlee County, UASM), New Mexico (Wickham 1896c: 133; Fall and Cockerell 1907: 159; Cibola County, CMNH), “Kansas” (Lindroth 1966: 591), Tennessee (Sevier County, CNC), and northern Georgia (Fattig 1949: 34). The record from “Louisiana” (Bousquet and Larochelle 1993: 256) needs confirmation.
CAN: AB, BC (VCI), MB, NB, NS (CBI), NT, ON, PE, QC, SK USA: AZ, CA, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, MA, ME, MD, MI, MN, MO, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OR, PA, RI, SD, TN, VA, VT, WA, WI, WV, WY [LA]
This species is known from southwestern Oregon (Jackson County, CNC) to southern California (Fall 1901a: 46), including the Sierra Nevada (Papp 1978: 163). The record from “Idaho” (Horn 1872c: 385) is probably in error.
USA: CA, OR
Agonum fallianum Leng has been retained as the valid name for this taxon by most authors since 1919 on the account that Agonum deplanatum Ménétriés was a junior homonym of Agonum deplanatum (Chaudoir). Leng (1919b: 203) believed the date of publication for Ménétriés’ name was 1844 and Chaudoir’s name 1843. However, both names were published in 1843. Ménétriés’ publication was issued on 29 July 1843 as indicated on page 64 of the journal and Chaudoir’s publication was issued after 7 October 1843, the date of permission for publication as indicated on the recto of the title page. The reversal of precedence (ICZN 1999: Article 23.9) cannot be applied in this case because Agonum deplanatum Ménétriés has been used as a valid name after 1899 (e.g., Csiki 1931: 841).
This species is found west of the Rocky Mountains from “Washington” (Hatch 1953: 143) and western Idaho (Nez Perce County, CNC) to southern California (Fall 1901a: 46). The record from Baja California (Horn 1894: 309) needs confirmation since the species is not listed in Liebherr’s (1994) review of the Mexican Agonum; those from southwestern British Columbia (Hatch 1953: 143) and Colorado (LeConte 1858a: 28) are probably in error.
USA: CA, ID, NV, OR, WA
This species is endemic to the Sierra Nevada [see Liebherr 1984b: Fig. 6], ranging from Lassen County to Tulare County.
USA: CA
This species ranges from west-central California south through the Coast Ranges to Baja California Norte [see Liebherr 1984b: Fig. 6].
USA: CA – Mexico
This Holarctic species is found in eastern Siberia (Bousquet 2003c: 451) and in North America from Alaska (Lindroth 1966: 595) to Labrador (Goose Bay, CNC). The record from “Newfoundland” (Bousquet and Larochelle 1993: 258) refers to Labrador. Fossil remnants of this species, dated between about 14, 000 and 18, 100 years B.P., have been unearthed in central and southeastern Iowa (Baker et al. 1986: 96; Schwert 1992: 78).
CAN: AB, BC, LB, MB, NT, ON, QC, SK, YT USA: AK – Holarctic
The range of this species extends from southern Saskatchewan to Vancouver Island (Lindroth 1966: 592, as Agonum subsericeum), south to southern California (Casey 1920: 107, as Agonum sybariticum) and Tlaxcala in Mexico (Liebherr 1994: 34).
CAN: AB, BC (VCI), SK USA: AZ, CA, CO, ID, KS, MT, NM, NV, OR, UT, WA, WY – Mexico
[cyclifer group]
This species ranges from southeastern Arizona to the Isthmus of Tehuantepec [see Liebherr 1994: Fig. 29].
USA: AZ – Mexico
The range of this species extends from southeastern Arizona to the Rio Grande drainage in south-central Texas, north to southeastern Colorado (Michels et al. 2008; Las Animas County, Robert L. Davidson pers. comm. 2008), south to the Federal District of Mexico [see Liebherr 1994: Fig. 28].
USA: AZ, CO, NM, TX – Mexico
[errans group]
This species ranges from the Saguenay River in Quebec (Larochelle 1975: 35) to southeastern British Columbia (Lindroth 1966: 616), south to “Oregon” (Hatch 1953: 143), New Mexico (LeConte 1879b: 56; UASM), “Texas” (LeConte 1858a: 28, as Platynus subcordatus), and southeastern South Carolina (Ciegler 2000: 111).
CAN: AB, BC, MB, ON, QC, SK USA: AL, AR, CO, GA, IA, ID, IN, KS, MA, MI, MN, MS, MT, NC, ND, NE, NH, NM, NY, OR, PA, SC, SD, TX, UT, VA, VT, WA, WI, WY
This species ranges from western New Hampshire (Grafton County, Ross T. Bell pers. comm. 2008) to eastern Oklahoma (Foster F. Purrington pers. comm. 2012), including southernmost Ontario (Lindroth 1966: 624), south to eastern Mississippi (Noxubee County, CMNH), southern Alabama (Löding 1945: 19), and central South Carolina (Ciegler 2000: 112). The record from northern Wisconsin along Lake Superior (Wickham 1896c: 134) is likely in error.
CAN: ON USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, MA, MD, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, VA, VT, WV
This species is known from a few localities along the Coastal Plain from southern Georgia (Fattig 1949: 33) and the Florida Panhandle (Peck and Thomas 1998: 23) west to northeastern Mississippi (Snodgrass and Cross 1983: 13), north along the Mississippi Basin to southern Illinois (Gallatin, Alexander, and Jackson Counties, CMNH, CNC). The record from “North Carolina” (Bousquet and Larochelle 1993: 260) needs confirmation.
USA: AL, FL, GA, IL, MS, TN [NC]
[excavatum group]
This species ranges from New Brunswick (Lindroth 1966: 623) to southern Wisconsin (Messer 2010: 42), south to eastern Oklahoma (Latimer County, UASM), west-central Arkansas (Logan County, CNC), northern Georgia (Fattig 1949: 34), and eastern South Carolina (Ciegler 2000: 111). The records from “Minnesota” and “Louisiana” (Bousquet and Larochelle 1993: 256) need confirmation.
USA: NB, ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KY, MA, MD, ME, MI, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, VA, VT, WI, WV [LA, MN]
[extensicolle group]
This species is found from eastern Arizona and western New Mexico south through the Sierra Madre Occidental to the Mexican states of Guerrero, Puebla, and Vera Cruz [see Liebherr 1986: Fig. 37].
USA: AZ, NM – Mexico
This widely distributed species ranges from southern Quebec to Vancouver Island, south to Baja California Norte, the Isthmus of Tehuantepec, and central Florida; also present on several islands of the West Indies [see Liebherr 1986: Figs 51-54].
CAN: BC (VCI), MB, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WY – Cayman Islands, Cuba, Hispaniola, Jamaica, Mexico
This species is found only in southern Georgia, throughout Florida including the Keys, and on Bimini [see Liebherr 1986: Fig. 58].
USA: FL, GA – Bahamas
Liebherr (1986: 140) reported the presence of a series of putative hybrids elongatulum x decorum from Okracoke Island, North Carolina.
This species ranges from Cape Breton Island (Bousquet 1987d: 106) to northwestern Montana, including southern Manitoba and southern Saskatchewan (Ronald R. Hooper pers. comm. 2007), south to south-central Arizona, Jalisco in Mexico, and northern Florida [see Liebherr 1986: Fig. 46].
CAN: MB, NB, NS (CBI), ON, QC, SK USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, UT, VA, VT, WI, WV, WY – Mexico
This species ranges from southern Arizona to southeastern Texas, south to southern Coahuila in Mexico [see Liebherr 1986: Fig. 41].
USA: AZ, NM, TX – Mexico
This species ranges from south-central Arizona south to the Mexican states of Sonora and southern Sinaloa along the Gulf of California Coast [see Liebherr 1986: Fig. 41].
USA: AZ – Mexico
This taxon ranges from central Kansas south to Chiapas in Mexico, west to central Arizona [see Liebherr 1986: Fig. 43].
USA: AZ, KS, NM, OK, TX – Mexico
[melanarium group]
This species ranges from Newfoundland (Lindroth 1955a: 123, as Agonum carbo) to central Alaska (Lindroth 1966: 605), south to southern Oregon (Westcott et al. 2006: 6), the Sangre de Cristo Mountains in New Mexico (Fall and Cockerell 1907: 159), northern Indiana (Blatchley 1910: 130; Wolcott and Montgomery 1933: 127), southwestern Pennsylvania (Westmoreland County, William L. Krinsky pers. comm. 2012) and the District of Columbia (Ulke 1902: 7, as Platynus carbo).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CO, CT, DC, IA, ID, IL, IN, MA, ME, MI, MN, MT, NH, NJ, NM, NY, OH, OR, PA, RI, VT, WA, WI, WY
This western species ranges from western British Columbia (Lindroth 1966: 609), as far north as the Skeena river area and Queen Charlotte Islands (Kavanaugh 1992: 75), south to southern California (Fall 1901a: 46, as Platynus frater), including eastern Nevada (Liebherr 1986: 130).
CAN: BC (QCI, VCI) USA: CA, NV, OR, WA
This species ranges from Rhode Island (Sikes 2003: 8; CMNH) to northeastern Illinois (Cook County, CMNH), south to eastern Texas along the Gulf Coast (Snow 1906a: 141) and central Florida (Brevard and Highlands Counties, CNC). The records from “Wisconsin” and “Pennsylvania” (Bousquet and Larochelle 1993: 258) need confirmation.
USA: AL, CT, FL, GA, IL, IN, KY, LA, MD, MI, MS, NC, NJ, NY, OH, PA, RI, SC, VA, TX [PA, WI]
This species ranges from the Saint Lawrence Valley in southern Quebec (Larochelle 1975: 33) to the Fraser Valley in British Columbia (Lindroth 1966: 603), south to northern California (Siskiyou County, CNC), east-central California (Mono County, Foster F. Purrington pers. comm. 2012), northern Arizona (Coconino and Apache Counties, CNC), southern Colorado (Alamosa County, CNC), and southern Nebraska (Chase County, Foster F. Purrington pers. comm. 2010). The record from “Michigan” (Bousquet and Larochelle 1993: 256) needs confirmation.
CAN: AB, BC, MB, ON, QC, SK USA: AZ, CA, CO, ID, MN, MT, ND, NE, OR, SD, UT, VT, WA, WV, WY [MI]
This rarely collected species is found along the provinces and states bordering the Atlantic Coast from Cape Breton Island (Lindroth 1966: 611) to Connecticut (New London County, William L. Krinsky pers. comm. 2008).
CAN: NS (CBI) USA: CT, MA, ME, NH
This species ranges from Cape Breton Island (Lindroth 1954c: 306) to western Wisconsin (Kleintjes et al. 2003: 81; Messer 2010: 42), south to northeastern Mississippi (Snodgrass and Cross 1983: 13) and west-central South Carolina (Ciegler 2000: 112). The record from “Prince Edward Island” (Bousquet and Larochelle 1993: 256) is based on misidentified small specimens of Agonum melanarium Dejean (CNC).
CAN: NB, NS (CBI), ON, QC USA: CT, DC, IA, IL, KY, MA, MD, ME, MI, MS, NC, NH, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI, WV
This species ranges from Cape Breton Island (Lindroth 1954c: 306) to Vancouver Island (Lindroth 1966: 609), south to “Oregon” (CAS), north-central Idaho (Idaho County, CNC), southeastern Wyoming (Lavigne 1977: 43), southern Minnesota (Hennepin County, CNC), northeastern Illinois (Purrington et al. 2002: 200), and southeastern Virginia (Surry County, CMNH). The record from “Nebraska” (Bousquet and Larochelle 1993: 257) is probably in error.
CAN: AB, BC (VCI), MB, NB, NS (CBI), ON, PE, QC, SK USA: CT, ID, IL, IN, MA, ME, MI, MN, MT, NH, NJ, NY, OH, OR, PA, RI, VA, VT, WA, WI, WV, WY
This species is known only from the holotype which has no locality or state label (see Liebherr 1991a: 121-122).
None.
The range of this species extends from Cape Breton Island (Lindroth 1954c: 306) to southwestern British Columbia (Lindroth 1966: 600), south to northwestern California (Trinity County, CNC), northern Colorado (Haubold 1951: 705; Armin 1963: 150), west-central Kansas (Trego County, CNC), and North Carolina (Haywood County, CNC).
CAN: AB, BC, MB, NB, NS (CBI), ON, PE, QC, SK USA: CA, CO, CT, DC, DE, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SD, UT, VA, VT, WA, WI, WV, WY
This species ranges from Newfoundland (Lindroth 1955a: 122) to the Queen Charlotte Islands (Kavanaugh 1992: 74), north to southern Northwest Territories (Bousquet 1987a: 125), south to northern Idaho (Peter W. Messer pers. comm. 2008), northwestern Montana (Glacier County, CNC), northern Wisconsin (Wickham 1896c: 133; Casey 1920: 114, as Agonum lacustre), the upper peninsula of Michigan along Lake Superior (Casey 1920: 114, as Agonum lacustre), and New Jersey (Smith 1890: 85; Smith 1910: 208).
FRA: PM CAN: AB, BC (QCI, VCI), MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: ID, MA, ME, MI, MN, MT, ND, NH, NJ, NY, VT, WI
This species is known from western Maine (Franklin, Kennebec, and York Counties, Ross T. Bell pers. comm. 2008), southwestern Quebec, the Ontario Peninsula, and southern Manitoba (Lindroth 1966: 611) south to southern Louisiana (Assumption and East Baton Rouge Parishes, LSAM) and northern Florida (Alachua County, CNC).
CAN: MB, ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, LA, MA, MD, ME, MI, MN, MS, NC, NJ, NY, OH, PA, RI, SC, TN, VA, VT, WI
This species is found from Newfoundland (Lindroth 1955a: 124) to British Columbia, north to east-central Alaska (Lindroth 1966: 601), south to west-central Washington (Lindroth 1966: 601), northwestern Montana (Russell 1968: 64), central Indiana (Downie 1957: 116; Schrock 1985: 343), and eastern South Carolina (Kirk 1969: 11; Ciegler 2000: 112).
CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, CT, DE, IA, IN, MA, ME, MI, MN, MT, NC, NH, NJ, NY, OH, PA, RI, SC, VT, WA, WI, WV
This widely distributed species extends from Newfoundland (Lindroth 1955a: 124) to Alaska (Lindroth 1966: 612), south to the Sierra Nevada in northeastern California (Liebherr 1991a: 119), southwestern Colorado (Wickham 1902: 239; Ouray County, CNC), northwestern Nebraska (Sheridan County, CNC), northwestern Pennsylvania (Erie County, CMNH), and “Maryland” (Liebherr 1994: 30). One specimen is also known from the city of Mexico (Liebherr 1994: 30) but in my opinion it is likely mislabeled.
CAN: AB, BC (VCI), MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CA, CO, CT, ID, IN, MA, MD, ME, MI, MN, MT, ND, NE, NH, NJ, NY, OR, PA, SD, VT, WA, WI, WY
This species ranges from Cape Breton Island (Lindroth 1954c: 306) to northeastern Minnesota (Kamal J.K. Gandhi pers. comm. 2008), south to southern Georgia (Fattig 1949: 34). The record from “Nebraska” (Bousquet and Larochelle 1993: 258) needs confirmation.
CAN: NB, NS (CBI), ON, PE, QC USA: CT, DC, DE, GA, IA, IL, IN, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, SC, VA, VT, WI, WV [NE]
This species is known from Cape Breton Island (CNC) to southern Manitoba (Lindroth 1966: 601), south to southwestern North Carolina (Macon County, CNC).
CAN: MB, NB, NS (CBI), ON, PE, QC USA: CT, MA, MD, ME, MI, MN, NC, NH, NJ, NY, OH, PA, RI, VT, WI, WV
[nutans group]
This species ranges from Nova Scotia (Lindroth 1954c: 306) to southeastern South Dakota (Kirk and Balsbaugh 1975: 25), south at least to northeastern Kansas (Popenoe 1877: 23), central Louisiana (East Feliciana Parish, CMNH, CNC), and the Florida Panhandle (Peck and Thomas 1998: 22).
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, PA, RI, SC, SD, TN, VA, VT, WI, WV
This species ranges from “New York” (LeConte 1854b: 52, as Platynus vagans) and New Jersey (Smith 1890: 85; Smith 1910: 208) to northeastern Colorado (LeConte 1854b: 52), south to northwestern South Carolina (Kirk 1970: 12; Ciegler 2000: 110). The record from “Florida” (Leng and Beutenmüller 1893: 140) is probably in error; that from “Wisconsin” (Bousquet and Larochelle 1993: 258) needs confirmation.
USA: CO, IA, IL, IN, KS, NE, NJ, NY, PA, SC [WI]
This species ranges from central North Carolina (Casey 1920: 86, as Micragonum ovalipenne and Micragonum pinorum) and South Carolina (Ciegler 2000: 111) west to southeastern Texas (Casey 1920: 85, 87, as Micragonum maritimum and Micragonum houstoni). The records from Missouri (Summers 1873: 134) and “Florida” (Bousquet and Larochelle 1993: 258) need confirmation.
USA: AL, GA, LA, MS, NC, SC, TN, TX [FL, MO]
This eastern species is found from southern Quebec (Larochelle 1975: 38) to eastern South Dakota (Kirk and Balsbaugh 1975: 24), south to Oklahoma (French et al. 2001: 228; Latimer County, UASM) and the District of Columbia (Ulke 1902: 7). The records from central Arizona (Griffith 1900: 565), northern Colorado (Wickham 1902: 239; Armin 1963: 149), southeastern Texas (Snow 1906a: 141), “Mississippi, ” “Alabama” (Bousquet and Larochelle 1993: 259), and “Florida” (LeConte 1879b: 56) are probably in error; those from “Maryland, ” “Delaware” (Bousquet and Larochelle 1993: 259), and South Carolina (Kirk 1970: 12, as Agonum possibly nutans) need confirmation.
CAN: ON, QC USA: AR, CT, DC, IA, IL, IN, KS, MA, ME, MI, MN, MO, NE, NH, NJ, NY, OH, OK, PA, RI, SD, VT, WI [DE, MD, SC]
This species ranges from New Jersey (Essex County, CMNH) to east-central Iowa (Muscatine County, Doug A. Veal pers. comm. 2010), including southernmost Ontario (Lindroth 1966: 619), south to southeastern Texas (Casey 1920: 88) and northern Florida (Peck and Thomas 1998: 23). The record from “Wisconsin” (Bousquet and Larochelle 1993: 259) needs confirmation.
CAN: ON USA: AL, AR, DC, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NC, NJ, OH, OK, PA, SC, TN, TX, VA [WI]
[octopunctatum group]
This species ranges from Nova Scotia (Lindroth 1966: 594) to southeastern Manitoba (UASM), south to eastern Texas (Orange and Sabine Counties, CNC; Dajoz 2007: 23) and central Florida (Peck and Thomas 1998: 23).
CAN: MB, NB, NS, ON, PE, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV
[pallipes group]
The range of this species extends from Cape Breton Island (Bousquet 1987a: 125) to southeastern South Dakota (Kirk and Balsbaugh 1975: 25), south to southeastern Nebraska (Hall County, Foster F. Purrington pers. comm. 2009), east-central Missouri (Saint Louis, CNC), and northwestern South Carolina (Kirk 1970: 12; Ciegler 2000: 111). The records from southern Louisiana (Summers 1874a: 80), “Kansas, ” and “Arkansas” (Bousquet and Larochelle 1993: 259) need confirmation.
CAN: NB, NS (CBI), ON, PE, QC USA: CT, DC, IA, IL, IN, KY, MA, MD, ME, MI, MN, MO, NE, NH, NJ, NY, OH, PA, RI, SC, SD, VA, VT, WI, WV [AR, KS, LA]
This species ranges from “Maryland” (CMNH) to north-central Oklahoma (Kay County, CNC), south at least to central Texas (Travis and Blanco Counties, CMNH; Casey 1920: 73), eastern Louisiana (Washington Parish, LSAM), and the Florida Panhandle (Peck and Thomas 1998: 23). The record from “Kansas” (Bousquet and Larochelle 1993: 259) needs confirmation.
USA: AL, AR, DC, FL, GA, IL, IN, KY, LA, MD, MO, MS, NC, OK, SC, TN, TX, VA [KS]
This species ranges from southeastern New Hampshire (Rockingham County, Ross T. Bell pers. comm. 2008) to southeastern Colorado (Michels et al. 2008), including southern Ontario (CNC), south to southern Mexico (Liebherr 1994: 20) and southern Florida (Peck and Thomas 1998: 23), west along the southwest to southern California (Liebherr 1994: 19-20); also recorded from the Bahamas and Bermuda where it is probably adventive (Hilburn and Gordon 1989: 677; Liebherr 1994: 19). The record from Quebec (Lindroth 1966: 622) refers to Agonum rufipes (Lindroth 1969a: 1120); that from northern Wisconsin along Lake Superior (Wickham 1896c: 134) is probably in error.
CAN: ON USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WV – Bahamas, Bermuda, Mexico
This species ranges from New Jersey (Ocean County, CMNH) south to northwestern Louisiana (Bossier Parish, LSAM), southwestern Mississippi (Hancock and Hinds Counties, CMNH), southwestern Alabama (Casey 1920: 75, as Circinalia ventricula), and southwestern Georgia (Fattig 1949: 33, as Platynus ventricula).
USA: AL, GA, LA, MD, MS, NC, NJ, TN, VA
The range of this eastern species extends from southeastern New Hampshire (Rockingham County, Donald S. Chandler pers. comm. 2009) to eastern South Dakota (Kirk and Balsbaugh 1975: 25), including southern Ontario (Lindroth 1969a: 1120), south to southeastern Texas (Casey 1920: 74) and southeastern Georgia (Fattig 1949: 32, as Platynus rubripes); also recorded from southern Arizona (Casey 1920: 74). The record from Connecticut (Britton 1920: 215, as Platynus rubripes) needs confirmation (see Krinsky and Oliver 2001: 4); that from Quebec (Lindroth 1966: 622, as Agonum punctiforme) based on specimens collected by Beaulne are very likely mislabeled.
CAN: ON USA: AL, AR, AZ, CA, DC, GA, IL, IN, KS, KY, MD, MI, MO, NC, NE, NH, NJ, NY, OH, OK, PA, SD, TN, TX, VA, WI, WV [CT]
[quadrimaculatum group]
This species is known from scattered localities from southwestern Indiana (Blatchley 1910: 123) south to southwestern Georgia (Fattig 1949: 34), southwestern Alabama (Casey 1920: 71; Mobile County, MCZ), southeastern Mississippi (George County, MCZ), north-central Louisiana (Winn Parish, MCZ), and northeastern Texas (Wood County, UASM). The record from Alberta (see Lindroth 1966: 631) must be in error after all; those from northwestern New York (Notman 1928: 235) and “Michigan” (Bousquet and Larochelle 1993: 261) need confirmation.
USA: AL, AR, GA, IN, KY, LA, MO, MS, TN, TX [MI, NY]
The number of species of Platynus is difficult to assess. Lorenz (2005: 429-434) assigned about 550 species to the genus arrayed in 14 subgenera, with about 95 species left unplaced. However, Liebherr (1998: 997) restricted the concept of Colpodes Macleay to three species from Java and suggested assigning the remaining species to “a broader Platynus.” Lorenz (2005: 416-418) listed about 270 species in Colpodes and if Liebherr’s advice is followed, this brings the total number of Platynus species to about 820. The Northern Hemisphere has about 155 species (19% of the world fauna) of the Platynus-“Colpodes-like” complex and 23 of them, placed in five subgenera, occur in North America.
Liebherr and Will (1996: 303-307) published a key for the identification of all species found in North America. Bousquet (2012: 77-78) described a new species and issued a key for the species found east of the Mississippi River.
Two species in southwestern North America.
This species is restricted to southern California from the Santa Ynez Mountains to San Diego County (Liebherr and Will 1996: 318).
USA: CA
This species is known only from the Sierra Blanca (Barr 1982b: 98), Capitan, and Sacramento Mountains (Liebherr and Will 1996: 318) in New Mexico.
USA: NM
Sixteen species in the Nearctic (nine species, of which one extends into Mexico) and Palaearctic (seven species) Regions.
This species ranges from southern British Columbia, including Vancouver Island (Lindroth 1966: 639), south to the northern parts of the Baja California Peninsula (Horn 1894: 309), east to western New Mexico (Liebherr and Will 1996: 316) and northern Sonora (Bates 1884: 280).
CAN: BC (VCI) USA: AZ, CA (CHI), ID, NM, NV, OR, UT, WA – Mexico
This species occurs along the Appalachian Mountains from Connecticut to southern Pennsylvania, south to northwestern Alabama [see Bousquet 2012: Fig. 4]
USA: AL, CT, KY, MD, NC, NY, PA, TN, VA, WV
This species ranges from Newfoundland (Lindroth 1955a: 118, as Agonum sinuatum) to south-central British Columbia (Lindroth 1966: 637), north to the coast of southern Alaska (Lindroth 1969a: 1120), south to western Oregon (Hatch 1953: 135), the Sangre de Cristo Mountains in New Mexico (Fall and Cockerell 1907: 159; Lindroth 1955a: 118), northeastern Kansas (Knaus 1901: 110; Ottawa County, CNC), east-central Alabama (Lee County, CNC), and southern Georgia (Torres and Ruberson 2006: 32). The records from “Louisiana” and “Texas” (Bousquet and Larochelle 1993: 262) need confirmation.
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK USA: AK, AL, AR, CO, CT, DC, DE, GA, IA, ID, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SC, SD, TN, UT, VA, VT, WA, WI, WV, WY [LA, TX]
This species ranges from Nova Scotia (Majka et al. 2007: 11) to eastern Ohio, south to West Virginia and Maryland [see Liebherr and Will 1996: Fig. 18].
CAN: NB, NS, ON, QC USA: CT, MD, ME, NH, NY, OH, PA, RI, VT, WV
This species ranges from southwestern New Brunswick (Webster and Bousquet 2008: 22) to northeastern Iowa (Allamakee County, Doug A. Veal pers. comm. 2009), including the upper peninsula of Michigan (Casey 1920: 30, as Anchomenus inquisitor), south to west-central Illinois (Fulton County, Ken Karns pers. comm. 2009), southwestern Indiana (Blatchley 1910: 125), and west-central South Carolina (Ciegler 2000: 115).
CAN: NB, ON, QC USA: CT, IA, IL, IN, MA, MI, NH, NJ, NY, OH, PA, RI, SC, VT, WI
This species is found along the west coast from Vancouver Island (Lindroth 1966: 645) to at least west-central California (Marin County, CNC). The record from northern Colorado (Wickham 1902: 238; Armin 1963: 153) is in error.
CAN: BC (VCI) USA: CA, OR, WA
This species ranges from southwestern Pennsylvania southwestwards to eastern Oklahoma [see Bousquet 2012: Fig. 4].
USA: AR, IL, IN, MO, OK, PA
The range of this species extends from Cape Breton Island (Lindroth 1966: 643) to the Black Hills in southwestern South Dakota (Kirk and Balsbaugh 1975: 24), south to northern Colorado (Armin 1963: 154; UASM), northwestern Texas (Wheeler County, CMNH), northeastern Oklahoma (Delaware County, CMNH), and southern South Carolina (Ciegler 2000: 115). The record from “Florida” (Bousquet and Larochelle 1993: 262) needs confirmation.
CAN: NB, NS (CBI), ON, PE, QC USA: AL, AR, CO, CT, DC, GA, IA, IL, IN, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [FL]
This species is known from high mountains along the Tennessee - North Carolina border (Barr 1969: 81); it was also reported from northeastern Georgia (Fattig 1949: 33).
USA: GA, NC, TN
Forty-four species are recorded by Lorenz (2005: 429-430), five in the Nearctic Region, with one of them Holarctic, and 40 in the Palaearctic Region. A number of Palaearctic species considered as incertae sedis by Lorenz (2005: 433-434) have been listed in this subgenus by Bousquet (2003c: 462-464).
This species is found from the Adirondack Mountains in New York (Casey 1920: 9 as Platynidius aesopus; Liebherr and Song 2002: 135) to at least south-central Ohio (Will and Androw 1993), south to southwestern Alabama (Löding 1945: 19) and southeastern South Carolina (Ciegler 2000: 114). The records from “Indiana” (Schrock 1985: 353), “Illinois” (Thomas 1861: 635), and Mount Washington in New Hampshire (Bowditch 1896: 2) need confirmation.
USA: AL, GA, KY, MD, NC, NJ, NY, OH, PA, SC, TN, VA, WV [IL, IN, NH]
This species ranges from Sable Island (CNC) off the Nova Scotia coast to southeastern Minnesota (Gandhi et al. 2005: 932), south to southern Texas (Casey 1920: 40, as Platynus cincticollis deplanatus; Dajoz 2007: 23) and southern Florida (Peck and Thomas 1998: 23); also recorded from Bermuda (Hilburn and Gordon 1989: 677).
CAN: NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KY, LA, MA, MD, MI, MN, MO, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV – Bermuda
This species is found from Vermont (Rutland County, CNC) to the lower peninsula of Michigan (Hubbard and Schwarz 1878: 644), including southern Ontario (Lindroth 1966: 646), south to east-central Kentucky (Powell County, CNC). The record from “Iowa” (Bousquet and Larochelle 1993: 263) needs confirmation; that from “Virginia” (Bousquet and Larochelle 1993: 263) is probably in error (Richard L. Hoffman pers. comm. 2009).
CAN: ON USA: IN, KY, MA, MD, MI, NY, OH, PA, VT, WV [IA]
Morvan (2000: 76) treated this taxon as a subspecies of Platynus angustatus Dejean.
This Holarctic species ranges from Newfoundland (Lindroth 1955a: 119) to central British Columbia and Alaska (Lindroth 1966: 636), south to northeastern Minnesota (Casey 1920: 36, as Platynus stygicus), central Wisconsin (Messer 2010: 43), and northern West Virginia (Preston County, CMNH). In the Palaearctic Region, the species is found from the Far East to Finland (Bousquet 2003c: 463).
FRA: PM CAN: AB, BC, LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, CT, ME, MI, MN, NH, NY, VT, WI, WV – Holarctic
This characteristic species is known for sure only from the holotype. However, Fattig (1949) recorded it also from Dalton, Georgia but the specimen(s) was apparently destroyed (Liebherr 1990: 432).
USA: GA
Thirty-six species in Middle America (28 species, of which one extends into southern North America and two into South America) and the West Indies (eight species).
Liebherr (1992) revised the species.
This species is known from southeastern Arizona and the northern Sierra Madre Occidental in Sonora and Chihuahua [see Liebherr 1992: Fig. 160].
USA: AZ – Mexico
Four species are assigned to this subgenus, one from southwestern United States and three from Mexico (Liebherr and Will 1996: 316).
This species is known from several localities in southeastern Arizona [see Liebherr and Will 1996: Fig. 24].
USA: AZ
About 320 species are listed in this subgenus by Lorenz (2005: 430-433). Of these only five unrelated species are found in southern United States, two of them being endemic (Platynus falli and Platynus rufiventris). The other species of the subgenus occur in the Neotropical Region, including the West Indies and the Galapagos Islands.
[cazieri group]
This species is known only from Pima and Santa Cruz Counties in southeastern Arizona and from one specimen without precise locality collected in Mexico [see Liebherr and Will 1996: Fig. 24].
USA: AZ – Mexico
[falli group]
This species is known from Pima, Cochise, and Santa Cruz Counties in southern Arizona (Liebherr and Will 1996: 318).
USA: AZ
Whitehead (1973: 196) stated that adults of this form are possibly conspecific with those of Platynus segregatus (Bates), a species known from the states of Michoacán, Colima, Morelos, Guerrero, and Oaxaca.
[lyratus group]
This species ranges from southern Utah, eastern Arizona, and western New Mexico south to Oaxaca in Mexico; it also occurs throughout the Baja California Peninsula (Liebherr and Will 1996: 318).
USA: AZ, NM, UT – Mexico
[megalops group]
The range of this species extends from southern Arizona and western Texas (Jeff Davis County, CMNH) south to the state of Oaxaca (Whitehead 1973: 201).
USA: AZ, TX – Mexico
[rufiventris group]
This species is yet known only from Cochise, Pima, and Santa Cruz Counties in southern Arizona (Liebherr and Will 1996: 318). The record from southwestern Utah (Tanner 1928: 270) is probably in error.
USA: AZ
Twenty-three species in the Palaearctic (11 Asian species) and Oriental (15 species) Regions are listed in this genus by Lorenz (2005: 423). Liebherr (2005), on the other hand, redefined cladistically the genus to include seven Pacific and Asian species. One of these species is adventive in western North America.
The sole species present in North America is included in Hatch’s (1953: 132) monograph on the beetles of the Pacific Northwest. It is also described in length by Habu (1978: 126-128).
This adventive species is known in North America from a few specimens collected in southern British Columbia (Jarrett and Scudder 2001: 382), southwestern Washington (Pacific County, Peter W. Messer pers. comm. 2011), western Oregon (Hatch 1953: 132), and northern Idaho (Liebherr and Will 1996: 303). The first specimen collected on this continent was found in 1931 in Portland, Oregon (Hatch 1953: 132). The species is also adventive in Hawaii (Liebherr and Zimmerman 2000: 466).
CAN: BC USA: ID, OR, WA – Adventive
About 120 species (Lorenz 2005: 438-439), mainly in the tropical regions, arrayed in five genera. Only one endemic species (Perigona pallipennis) is found in North America and one in Europe (Galiciotyphlotes weberi Assmann from northwestern Spain).
Excluding the subcosmopolitan Perigona nigriceps, about 105 species (Lorenz 2005: 438-439) in the temperate, subtropical, and tropical areas of the Nearctic (one species), Neotropical (twelve species), Australian (about 17 species), Oriental (about 30 species), eastern Palaearctic (five species), and Afrotropical (about 40 species) Regions. These species are arrayed in 12 subgenera: Cryptoperigona Perrault (one Neotropical species), Euripogena Basilewsky (four Afrotropical species), Euryperigona Jeannel (three species in the Philippines, Indonesia, and New Guinea), Hirtoperigona Perrault
Eighteen species in the Nearctic (one endemic species), Australian (seven endemic species), and Afrotropical (ten endemic species) Regions and one (Perigona nigriceps) subcosmopolitan species.
Bousquet (1987a: 128) commented on the structural differences between the two species found in North America.
Hirtoperigona Perrault is placed in synonymy with Trechicus in Lorenz (2005: 438). However, the position of the median group of setae of the umbilical series in the type species (Perigona tronqueti Perrault from Sri Lanka) and in Perigona pubescens Jeannel (from Vietnam) suggests that members of Hirtoperigona are more closely related to Perigona s.str. than to Trechicus. Perrault (1988) placed a third species (Perigona hirtella Basilewsky from Africa) in Hirtoperigona on the basis of the original description alone; according to Basilewsky (1989: 447), the species belongs to Trechicus.
The relationship of the endemic North American species (Perigona pallipennis LeConte) has not been studied and, in my opinion, it may be more closely related to members of Cryptoperigona than to those of Trechicus.
This species is subcosmopolitan. It is adventive in North America where it is known from Nova Scotia (Pictou County, CNC) to eastern Iowa (Muscatine County, Doug A. Veal pers. comm. 2009), south to east-central Texas (Riley 2011), central Louisiana (Grant Parish, CNC) and southern Florida (Peck and Thomas 1998: 23) in the east, and from Oregon (Hatch 1953: 147) to southern California (Fall 1901a: 47) in the west. The first inventoried specimen collected on this continent was found prior to 1853 (LeConte 1853c: 386, as Trechicus umbripennis). Basilewsky (1953a: 108) believed the species originated along the coast of the Indian Ocean where it is very abundant in rubbish and groundnut.
CAN: NB, NS, ON, QC USA: AL, AR, CA, CT, DC, FL, GA, IA, IL, IN, KY, LA, MA, MD, ME, MI, MS, NC, NE, NH, NJ, NY, OH, OR, PA, RI, SC, TX, VA, VT, WI, WV – Adventive
Perigona nigriceps (Dejean). While most adventive carabid species have been introduced from Europe, this species, which is now subcosmopolitan, is probably native from southern Asia where it is common around the Indian Ocean. Contrary to European adventive species which have been introduced to North America through ballast and nursery stocks, this species was probably transported around the world among stored products on ships.
The range of this species extends from southwestern Maine (Nelson 1991: 284) to eastern Iowa (Wickham 1911b: 7), including southernmost Ontario, south to “Missouri, ” “Alabama” (Bousquet 1987a: 127-128), and northern Florida (Hamilton, Okaloosa, and Taylor Counties, CNC).
CAN: ON USA: AL, DC, FL, GA, IA, IL, IN, MD, ME, MI, MO, MS, NC, NJ, NY, OH, PA, SC, VA
One genus with two species in the Nearctic and Palaearctic Regions.
Two species in the temperate regions of eastern North America (Atranus pubescens) and Europe (Atranus ruficollis Gautier des Cottes).
Lindroth (1968: 649) commented on the structural differences between the two species.
The range of this species extends from central Nova Scotia (Hants County, Christopher G. Majka pers. comm. 2008) to southwestern Nebraska (Foster F. Purrington pers. comm. 2010), south to “Texas” (Barr 1964: 3; Lindroth 1968: 649) and west-central South Carolina (Ciegler 2000: 108).
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, SC, TN, TX, VA, VT, WI, WV
Atranus pubescens (Dejean). This species is found over eastern North America and the only other species in the genus, Atranus ruficollis, occurs in southern Europe and the Middle East. The only other carabid genus with a similar vicariant distribution is Olisthopus although one European species, Olisthopus sturmii, extends to the Far East. Most amphiatlantic vicariant carabid groups, such as Pseudanophthalmus-Duvaliopsis, are at least generically distinct.
About 115 species (Lorenz 2005: 440-441, as Lachnophorina) in the Nearctic and Neotropical Regions, arrayed in 11 genera, and one species (Selina westermanni Motschulsky) in the Oriental and Afrotropical Regions.
Ball and Bousquet (2000: 108) recognized two groups of genera within this tribe. The eucaerine complex comprising Eucaerus, Aporesthus Bates, Amphitasus Bates, and Asklepia Liebke and the lachnophorine complex encompassing Lachnophorus, Euphorticus, Calybe, Anchonoderus, and Selina. Subsequently to their investigation, three new genera have been proposed: Pseudophorticus Erwin (four species), Guatemalteca Erwin (one species), and Quammenis Erwin (one species).
Western Hemisphere, with 26 species (Lorenz 2005: 440) in the Nearctic (two species) and Neotropical (24 species) Regions.
Schaeffer (1910: 395) commented on the structural differences between the two species found in North America.
This species is known only from Texas (Val Verde County, CNC).
USA: TX
This species is known from southeastern Arizona (Graham County, CNC) and southern Texas (Schaeffer 1910: 395).
USA: AZ, TX
Western Hemisphere, with about 35 species in temperate, subtropical, and tropical areas of the Nearctic (one species only) and Neotropical Regions.
According to Liebherr (1988: 34), Stigmaphorus Motschulsky, 1855 (type species: Stigmaphorus tessellatus Motschulsky, 1855), considered a junior synonym of Lachnophorus by several authors, “should not be considered the same as Lachnophorus.”
This species ranges from Kansas (LeConte 1858a: 28; Horn 1872c: 385) to southwestern Oregon (Hatch 1953: 146; Jackson County, MCZ, UASM), south to southern California (Moore 1937: 11; San Diego County, CNC, MCZ) and Costa Rica (Liebke 1936: 463).
USA: AZ, CA, KS, NM, OK, OR, TX, UT – Costa Rica, Guatemala, Mexico, Nicaragua
Four species in temperate, subtropical, and tropical areas of the Nearctic (two species) and Neotropical (three species) Regions.
Horn (1891: 38) commented on the structural differences between the two species found in North America.
This species is known so far only from southwestern California (Horn 1891: 38; Fall 1901a: 47; Moore 1937: 11).
USA: CA
This species is found along the Coastal Plain from southeastern North Carolina to central Florida, west to northeastern Texas (Dallas County, MCZ) and central Oklahoma (Grady County, Robert L. Davidson pers. comm. 2012), including west-central Louisiana (Allen 1965: 67), along the Mississippi Basin in southwestern Ohio (Blatchley 1910: 137) and central Indiana (Downie and White 1967: 308), in the West Indies from the Bahamas (Turnbow and Thomas 2008: 12) to Trinidad, in tropical mainland from central Mexico to Santa Catarina in Brazil [see Liebherr 1988: Fig. 17], and along southwestern United States from New Mexico (Snow 1885: 66; Fall and Cockerell 1907: 159) to southern Arizona (Pima County, MCZ); also recorded from the southern part of the Baja California Peninsula (Horn 1895: 226). The record from southeastern Pennsylvania (Rathvon 1869: 523) must be in error.
USA: AL, AZ, FL, GA, IN, LA, MS, NC, NM, OH, OK, SC, TX – Bahamas, Brazil, Colombia, Cuba, French Guiana, Guatemala, Honduras, Jamaica, Mexico, Venezuela
Twenty-five species (Lorenz 2005: 440-441) in the Western Hemisphere arrayed in two subgenera: Calybe s.str. (eight species) represented in the Neotropical Region only and Ega (17 species).
Seventeen species in the temperate, subtropical, and tropical areas of the Nearctic (two species) and Neotropical (17 species) Regions.
Horn (1881: 153) commented on the structural differences between the two species found in North America.
This species ranges from southernmost Nevada (Clark County, CNC) south to Guatemala (Bates 1883a: 157), including southeastern California (Riverside County, MCZ, UASM) and southern Arizona (Fall 1901a: 48; Maricopa and Pima Counties, MCZ, UASM). The record from “Colorado” (LeConte 1858a: 28) probably refers to the Colorado River.
USA: AZ, CA, NV – Guatemala, Mexico
This species is known along the Coastal Plain from South Carolina (Kirk 1969: 12; Ciegler 2000: 116; Clarendon County, MCZ) to southern Florida (Peck and Thomas 1998: 23), west to central and southeastern Texas (Lee and Cameron Counties, MCZ; Wickham 1897: 107), and south to Nicaragua (Bates 1883a: 157). The record from “Arizona” (Ball and Bousquet 2000: 109) refers to Calybe laetula. Old specimens simply labeled “Ill.” and “Ks” are known (MCZ).
USA: AL, AR, FL, GA, LA, MS, SC, TX [IL, KS] – Guatemala, Mexico, Nicaragua
Fourteen species in the Western Hemisphere arrayed in two subgenera: Eucarus s.str. (11 species) and Lachnaces Bates (three Amazonian species).
Eleven species in temperate, subtropical, and tropical areas of the Nearctic (one species) and Neotropical (ten species) Regions arrayed in two species groups (Ball and Hilchie 1983: 107-108).
This species inhabits the Coastal Plain from central Maryland (Hoffman et al. 2006: 28) to southern Florida (Peck and Thomas 1998: 23), west to eastern Texas (Hardin County, UASM), including southeastern Mississippi (Jackson County, Drew A. Hildebrandt pers. comm. 2008).
USA: AL, FL, GA, LA, MD, MS, NC, SC, TX, VA
Worldwide but excluding Europe and northern Africa, with about 165 species (Lorenz 2005: 445-447) arrayed in five genera: Aeolodermus Andrewes (one Oriental species), Homethes Newman (ten australo-oriental species), Parascopodes Darlington (one Australian species), Pentagonica (about 85 species), and Scopodes Erichson (68 australo-oriental species). The Northern Hemisphere is underrepresented with only 17 species (about 10% of the world fauna).
About 85 species in the temperate, subtropical, and tropical areas of the Nearctic (six species, only one endemic), Neotropical (about 25 species), Australian (10 species), Oriental (about 20 species), Palaearctic (11 Asian species), and Afrotropical (25 species) Regions.
Lindroth’s (1969a: 1013) key covered all North American known at the time. Two new species have been described subsequently by Bell (1987) and Mateu (1995).
This species ranges from Colorado County in southeastern Texas to Guatemala (Reichardt 1968: 150).
USA: TX – Guatemala, Mexico
This species is known from a few localities from southeastern Arizona and southwestern New Mexico (Bell 1987: 373) south to Chiapas in southern Mexico (Bell 1989a: 156).
USA: AZ, NM – Mexico
This subspecies ranges from southwestern New Jersey (Smith 1910: 212) and northern Virginia (Hoffman and Roble 2000: 39) to northeastern Kansas (Popenoe 1877: 23, as Rhombodera pallipes), south to eastern Texas (Sabine County, CNC) and the Florida Keys (Peck and Thomas 1998: 22). It is also known from Central America, Colombia, Brazil, and the West Indies (Reichardt 1968: 153; Bell 1985a: 326). The record from “Arizona” (Leng 1915: 589) needs confirmation.
USA: AL, AR, FL, GA, IA, IN, KS, LA, MS, NC, NJ, OH, OK, SC, TX, VA [AZ] – Antigua, Bahamas, Belize, Brazil, Cayman Islands, Colombia, Costa Rica, Cuba, Dominica, Dominican Republic, Guadeloupe, Guana Island, Guatemala, Monserrat, Panama, Trinidad and Tobago
The subspecies Pentagonica flavipes picipes Darlington is restricted to Jamaica, Hispaniola, Puerto Rico, and the Virgin Islands (Reichardt 1968: 154; Bell 1985a: 326).
This species is known from southeastern Georgia (Camden and Glynn Counties, CMNH), central Florida (Pinellas County, CMNH), north-central Mississippi (Grenada County, Drew A. Hildebrandt pers. comm. 2010), and east-central (Riley 2011) and south-central Texas (Bastrop County, Peter W. Messer pers. comm. 2010).
USA: FL, GA, MS, TX
This species is known only from the Florida Peninsula as far north as Alachua County (Peck and Thomas 1998: 22), the Bahamas (Turnbow and Thomas 2008: 14), Cuba (Bell 1985a: 323), and the Cayman Islands (Darlington 1947: 211).
USA: FL – Bahamas, Cuba, Cayman Islands
This species ranges from southern Quebec (Larochelle 1975: 98) to western Wisconsin (Messer 2010: 42), south to Guatemala (Bates 1883a: 217), southeastern Louisiana (Saint Tammany Parish, Igor M. Sokolov pers. comm. 2009), and central Florida (Vince Golia pers. comm. 2007), west to southern Arizona and the Baja California Peninsula (Horn 1894: 311).
CAN: ON, QC USA: AR, AZ, FL, GA, IA, IL, IN, KS, LA, MD, MI, MO, MS, NE, NH, NJ, NM, OH, OK, PA, TN, TX, VA, VT, WI, WV – Guatemala, Mexico
Worldwide, with about 325 species (Lorenz 2005: 441-445, as Odacanthina) in the Nearctic (five species), Neotropical, Australian, Oriental, Palaearctic (21 species, several shared with the Oriental Region), and Afrotropical Regions.
Western Hemisphere, with about 100 species (Lorenz 2005: 442-444, as Cosnania and Colliuris) in temperate, subtropical, and tropical areas of the Nearctic (five species) and Neotropical Regions. The species are arrayed in 20 subgenera.
Bousquet (2010b) reviewed the Nearctic, Mexican and West Indian (Greater Antilles) species and provided a key for their identification.
Liebke (1930, 1938) recognized an array of subgenera and several appear to be polyphyletic assemblages. A taxonomic revision of the genus is much needed. Lorenz (2005: 442) recognized Cosnania as generically distinct from Colliuris. I see no reason to support this approach particularly since Cosnania, as currently recognized, is likely polyphyletic (see below).
Three species in the Neotropical Region (Liebke 1938: 57), one of them extending into southern Arizona.
This species ranges from southern Arizona (Bousquet 2010b: 7) to Panama (FSCA).
USA: AZ – Costa Rica, Guatemala, Honduras, Mexico, Panama
Fourteen species are listed in this subgenus by Lorenz (2005: 442) but some (e.g., Cosnania lioptera, Cosnania tetrastigma, and Cosnania emdeni Liebke) are not closely related to the type species, Cosnania pensylvanica. Two North American species belong to this subgenus.
This species has a very restricted distribution; it is known from southeastern Arizona and adjacent northern Mexico [see Bousquet 2010b: Fig. 19].
USA: AZ – Mexico
This species ranges from Maine (Dearborn and Donahue 1993: 8; Foss 2001: 14) to eastern South Dakota (Kirk and Balsbaugh 1975: 37), including southern Quebec (Larochelle 1975: 78) and the Ontario Peninsula (Lindroth 1969a: 1008), south to the Yucatán Peninsula and southern Florida, west to southern Arizona and Baja California Sur [see Bousquet 2010b: Fig. 18]; also known from Cuba and Jamaica (Bousquet 2010b: 10).
CAN: ON, QC USA: AL, AR, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Cuba, Jamaica, Mexico
Five Nearctic and Neotropical species were included in this subgenus by Liebke (1938: 55-56). However, a number of species (e.g., Colliuris liptera Bates, Colliuris tetrastigma Chaudoir, Colliuris caymanensis Darlington, Colliuris ellipticeps Liebke, and Colliuris gundlachi Darlington), placed in other subgenera by Liebke (1938), are closely related to members of Calocolliuris (personal observation).
This species is known from southern Florida, Cuba, the Cayman Islands, Jamaica, and Haiti (Bousquet 2010b: 23).
USA: FL – Cayman Islands, Cuba, Haiti, Jamaica
This species ranges from southeastern Arizona (Bousquet and Larochelle 1993: 334) south at least to Honduras (USNM).
USA: AZ – Honduras, Mexico
This species inhabits the Coastal Plain and ranges from southeastern New York to southern Florida, west to eastern Louisiana [see Bousquet 2010b: Fig. 20]. The species was also recorded from “N. Yucatan” by Liebke (1938: 56) and from “Pennsylvania” by Leng (1920: 65); both records are suspect.
USA: AL, DC, FL, LA, MD, NC, NJ, NY, VA [PA]
Western Hemisphere, with about 65 species (Lorenz 2005: 393) arrayed in 16 genera.
Thirty-four species (Lorenz 2005: 393) in the temperate, subtropical, and tropical areas of the Nearctic (three species) and Neotropical (32 species) Regions.
Bousquet (1997b: 336) commented on the structural differences between Leptotrachelus dorsalis and Leptotrachelus pallidulus. The third species (Leptotrachelus depressus) is known only from the holotype which is very similar to members of Leptotrachelus pallidulus and may eventually prove to be conspecific with them.
This species is known only from the Florida Peninsula (Peck and Thomas 1998: 23).
USA: FL
This species ranges from Rhode Island (William L. Krinsky pers. comm. 2009) and Connecticut (Krinsky and Oliver 2001: 232) to southeastern South Dakota (Kirk and Balsbaugh 1975: 36), including the Ontario Peninsula (Lindroth 1969a: 1006), south to southeastern Texas (Wharton County, CNC) and southern Florida (Peck and Thomas 1998: 23); also found in Cuba (Gundlach 1891: 19), Barbados, and Hispaniola (Peck and Thomas 1998: 23).
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NC, NE, NJ, NY, OH, PA, RI, SC, SD, TN, TX, VA, WI, WV – Barbados, Cuba, Hispaniola
This species is known from a few scattered localities in Tennessee, Louisiana (Bousquet 1997b: 338), and Texas (Chaudoir 1872d: 412).
USA: LA, TN, TX
About 120 species in the Nearctic (four species), Neotropical (about 40 species of Tetragonoderus), Australian (one adventive Tetragonoderus), Oriental, Palaearctic (20 species), and Afrotropical Regions. These species are arrayed in four genera: Cyclosomus Latreille (13 species), Cyclicus Jeannel (22 afro-oriental species), Mnuphorus Chaudoir (11 species) and Tetragonoderus (about 75 species). The Northern Hemisphere is represented by about 25 species (approximately 21% of the world fauna).
This tribe is treated here in a restricted sense following Ball and Bousquet (2000: 109) and excluded the graphipterines, corsyrines, masoreines, somoplatines, and nemotarsines.
About 75 species (Lorenz 2005: 453) arrayed in two subgenera, Tetragonoderus s.str. with about 35 species in the Old World and Crossonychus for the Western Hemisphere species.
Lindroth (1969a: 1010) included a key to four of the five species found in North America.
Western Hemisphere, with about 40 species in the Nearctic (five species, one of them adventive) and Neotropical (about 40 species) Regions. One South American species (Tetragonoderus undatus Dejean) is adventive in Australia (Moore 1976). The species have been arrayed in two species groups by Ball (2000: 190), the figuratus and intersectus groups; only the last one is represented in North America.
This species ranges from southwestern Maine (Majka et al. 2011: 47) and southern Quebec (CNC) to southwestern Minnesota (Gandhi et al. 2005: 932), south to southern Texas (Wickham 1897: 107; Zapata County, CMNH) and central Florida (Peck and Thomas 1998: 23), west along the south to Riverside County in California (Dajoz 2007: 19) and the Baja California Peninsula (Horn 1894: 310; Horn 1895: 226).
CAN: ON, QC USA: AL, AR, AZ, CA, CT, DC, DE, FL, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, SC, TN, TX, VA, VT, WI – Mexico
This species ranges from North Carolina (Moore County, CNC) to southern Arizona (Pima County, CMNH), south to Jalisco in Mexico (CMNH), southern Texas (Johnson 1978: 68), and southern Florida (Peck and Thomas 1998: 23); also known from the Bahamas (Darlington 1953: 16). The record from “Virginia” (Bousquet and Larochelle 1993: 266) needs confirmation; that from southern Ontario (Belaoussoff et al. 2003: 878) probably refers to Tetragonoderus fasciatus.
USA: AL, AR, AZ, FL, GA, KY, LA, MS, NC, SC, TN, TX [VA] – Bahamas, Mexico
This South American species is adventive in southeastern Florida (Shpeley and Ball 2008: 9). The first inventoried specimen collected on this continent was found in March 2007 (Halbert 2007: 7; Shpeley and Ball 2008: 9). In South America, the species ranges from the Baía de Marajó along the Atlantic Coast in northern Brazil to southern Uruguay and central Argentina (Shpeley and Ball 2008: Fig. 6).
USA: FL – Adventive
This species occurs from southwestern Alabama (Baldwin and Clarke Counties, CMNH) to central Arizona (Yavapai County, CNC), south to southern Texas (Wickham 1897: 107; Johnson 1978: 68); also recorded from Mexico (Blackwelder 1944: 52).
USA: AL, AR, AZ, LA, MS, TX – Mexico
This species is known from southwestern California (Fall 1901a: 48; Moore 1937: 11) to the western tip of Texas (El Paso County, CMNH), including western Nevada (Bechtel et al. 1983: 474); also recorded from the Baja California Peninsula (Horn 1895: 226).
USA: AZ, CA, NV, TX – Mexico
Worldwide, with about 4, 260 species arrayed in 237 genera (Lorenz 2005: 453-502, as Nemotarsina and Lebiini). The North American fauna is underrepresented both in term of species (about 155 species or 3.6% of the world fauna) and genera (25 genera or 10.5%). The lebiine genera are classified into the following 17 subtribes: Actenonycina (one New Zealand species), Agrina (about 585 species), Apenina (about 115 species), Calleidina (about 660 species), Celaenephina (two species), Cymindidina (about 315 species), Demetriadina (14 species), Dromiusina (about 735 species), Gallerucidiina (ten species), Lebiina (about 800 species), Metallicina (about 70 species), Nemotarsina (nine species), Peliocypadina (about 70 species), Pericalina (about 825 species), Physoderina (45 species), Sugimotoina (one species), and Trichina (five species).
Worldwide, with about 825 species arrayed in 77 genera (Lorenz 2005: 454-464). The subtribe is underrepresented in the Northern Hemisphere with about 70 species (8.5% of the world fauna), of which nine (about 1%) only occur in North America.
Shpeley and Ball (2001) reviewed all 111 native species-group taxa found in the Western Hemisphere and provided keys for their identification. Their work is to be used in conjunction with a previous study (Ball and Shpeley 1983).
In a cladistic analysis based on characters of the adults performed by Ball et al. (1995: Fig. 7), this subtribe is positioned as the sister-group to {Sugimotoina + Actenonycina}. In Casale’s (1998: Fig. 91) cladistic analysis, the pericalines showed up as the sister-group to the remaining Lebiini.
Nine species (Lorenz 2005: 460) in the Palaearctic (eastern Asia only), Oriental, and Australian Regions, with one of them adventive in Florida.
The character states of the sole species found in North America are described in detail by Habu (1967: 104-105) under the name Dolichoctis tetraspilotus.
Habu (1967: 100) treated this taxon as a subgenus of Dolichoctis Schmidt-Göbel.
This species is widely distributed in Asia from India to Japan, south through the Malay Archipelago to New Guinea; it is also known from Christmas Island and Samoa. The species is adventive in southeastern United States and currently known from Florida, where the first specimens were detected in 1992 in Palm Beach County (Choate 2001), southern Mississippi (Pearl River County, Peter W. Messer pers. comm. 2010), and east-central Louisiana (West Feliciana Parish, Igor M. Sokolov pers. comm. 2009).
USA: FL, LA, MS – Adventive
Thirty-four species in the temperate, subtropical, and tropical areas of the Nearctic (one eastern species) and Neotropical (34 species) Regions. These species are arrayed in five subgenera: Oenaphelox Ball (nine species), Tacana Ball (one species), Phloeoxena s.str. (15 species), Oxephloena Shpeley and Ball (one species), and Ochropisus Bates (eight species).
Shpeley and Ball (2001: 78-80) published a key for the identification of all known species.
Nine species in North America and Middle America.
This species extends from Maryland (Steiner et al. 2007: 224) to central Florida (Peck and Thomas 1998: 24) and southeastern Louisiana, and from the states of Tamaulipas and Jalisco in Mexico to Panama [see Ball 1975: Fig. 112].
USA: AL, FL, GA, LA, MD, MS, NC, SC, TN, VA – Costa Rica, Guatemala, Honduras, Mexico, Nicaragua, Panama
Twenty-four species (Lorenz 2005: 463-464) in the Neotropical Region, one of them reaching southeastern Texas. The species are arrayed in five subgenera: Inna Putzeys (ten species), Eucheila s.str. (six species), Pseudoinna Mateu (four species), Bordoniella Mateu (two species), and Hansus Ball and Shpeley (two species).
Ten species in the Neotropical Region, one of them reaching southeastern Texas.
This species ranges from southern Texas (Shpeley and Ball 2001: 162) south to northeastern Brazil [see Ball and Shpeley 1983: Fig. 62].
USA: TX – Brazil, Colombia, Costa Rica, Guatemala, Honduras, Mexico, Netherlands Antilles, Nicaragua, Panama, Venezuela
Two species from the Afrotropical Region, one restricted to Madagascar, the other subcosmopolitan, having been dispersed through commerce.
The character states of the species found in North America are described in detail by Habu (1967: 75-77).
This genus was listed in the tribe Singilini (currently considered a synonym of Dromiusina) by Jeannel (1949a: 915-916) along with the genera Singilis Rambur (eight species in the Mediterranean region and Middle East), Phloeozetus Peyron (about 55 species in Africa, Mediterranean region, and Middle East), Velindopsis Burgeon (three Afrotropical species), Pephrica Alluaud (six Afrotropical species), and Paulianites Jeannel (one Madagascan species).
This species is adventive in North America where it is known from Randolph County in North Carolina (Peter W. Messer pers. comm. 2012), Aiken County in South Carolina (Peter W. Messer pers. comm. 2011), Telfair and Jeff Davis Counties in southern Georgia (Harry J. Lee, Jr. pers. comm. 2010), Broward County in Florida (Peck and Thomas 1998: 25), Polk and Garland Counties in Arkansas (CMNH), and Santa Cruz County in southern Arizona (Ober and Maddison 2008: 30, as Somotrichus elevatus). The record from Seattle, Washington (Hatch 1953: 153) is based on specimens intercepted in nuts from Brazil; that from “Texas” (Bousquet and Larochelle 1993: 270) needs confirmation.
USA: AR, AZ, FL, GA, NC, SC [TX] – Adventive
About 105 species (Lorenz 2005: 457-458) in the Nearctic (five species), Neotropical (43 species), Australian, Afrotropical, Oriental, and Palaearctic (13 species in eastern Asia only) Regions. These species are arrayed in four subgenera: Coptodera s.str. (about 45 species), Haplocrepis Jeannel (six Afrotropical species), Coptoderina Jeannel (53 species), and Coptoderinella Hansen (one Afrotropical species).
About 45 species in temperate, subtropical, and tropical areas of the Western Hemisphere arrayed in 11 species groups. Five species are found in North America.
Shpeley and Ball (1994) revised the species and provided a key for their identification.
[aerata group]
The range of this species extends from Connecticut (New London County, William L. Krinsky pers. comm. 2012) to southeastern Nebraska, south to eastern Texas and west-central Florida [see Shpeley and Ball 1994: map 19]. According to Shpeley and Ball (1994: 119), the specimens labeled from Gallatin County in Montana (CAS) and Santa Cruz County in Arizona (AMNH) are probably mislabeled.
USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NC, NE, NJ, NY, OH, OK, PA, SC, TN, TX, VA, WV
This species is found from southeastern Arizona and southeastern New Mexico south to Honduras and Belize [see Shpeley and Ball 1994: map 18].
USA: AZ, NM – Belize, Honduras, Mexico
The range of this species extends from southern Arizona and southwestern New Mexico south to southeastern Brazil and northern Argentina [see Shpeley and Ball 1994: map 17].
USA: AZ, NM – Argentina, Bolivia, Brazil, Colombia, Costa Rica, Ecuador, Guatemala, Guyana, Mexico, Nicaragua, Panama, Paraguay, Peru, Surinam, Trinidad and Tobago, Venezuela
[festiva group]
This species occurs in southern Florida, the West Indies, and from Mexico (as far north as San Luis Potosí) to Brazil [see Shpeley and Ball 1994: map 12].
USA: FL – Brazil, Colombia, Costa Rica, Cuba, Ecuador, Guatemala, Haiti, Jamaica, Mexico, Panama, Puerto Rico, Venezuela
[picea group]
This species is known from the tip of Florida, the West Indies, including the Bahamas (Turnbow and Thomas 2008: 12), Cuba, and the Dominican Republic (Shpeley and Ball 2001: 148), and from the state of San Luis Potosí in Mexico to southeastern Brazil [see Shpeley and Ball 1994: map 16].
USA: FL – Bahamas, Belize, Bolivia, Brazil, Colombia, Cuba, Dominican Republic, Ecuador, French Guiana, Guatemala, Guyana, Honduras, Mexico, Nicaragua, Panama, Paraguay, Peru, Surinam, Trinidad and Tobago, Venezuela
About 315 species (Lorenz 2005: 465-470, excluding Metaxymorphus) in the Nearctic (27 species), Neotropical (12 species), Oriental (six species), Palaearctic (about 170 species), and Afrotropical (about 115 species) Regions. These species are arrayed in nine genera: Taridius Chaudoir (four Oriental species), Afrotarus Jeannel (seven species in Africa, Arabia, and India), Cymindis (about 200 species), Petrimagnia Kryzhanovskij and Mikhailov (one species from Tadzhikistan), Leptosarcus Péringuey (two Afrotropical species), Pseudomasoreus Desbrochers des Loges (20 species in Africa, one of them extending into southwestern Europe), Assadecma Basilewsky (one Madagascan species), Hystrichopus Boheman (about 70 species in Africa and one in Yemen), and Plagiopyga Boheman (13 Afrotropical species). According to Ball and Hilchie (1983: 197), the Afrotropical genus Metaxymorphus Chaudoir belongs to the subtribe Dromiusina. On the other hand, Basilewsky (1984: 551), followed by Lorenz (2005: 470), listed it, but with strong doubt, with the Cymindidina.
About 200 species (Lorenz 2005: 465-469, as Pinacodera and Cymindis) in the Nearctic (27 species), Neotropical (13 species), and Palaearctic (167 species) Regions arrayed in 15 subgenera.
Ball and Hilchie (1983) regarded Afrotarus, Pinacodera, and Taridius as subgenera of Cymindis, implicitly considering all 14 subgenera of Cymindis currently recognized in Lorenz (2005) as synonyms of Cymindis s.str. For practical reasons this approach is not followed here except that Pinacodera is retained as a subgenus of Cymindis.
Twenty-one species in North America (16 species, one of them Holarctic), Mexico (two species shared with North America), Eurasia (two species, one endemic to eastern Asia), and North Africa (four endemic species).
Lindroth (1969a) reviewed all North American species. Since then, one Palaearctic species (Coptodera vaporariorum) has been found to occur naturally also in North America. Shpeley and Ball (1999: 420-421) published an emendation to Lindroth’s (1969a: 1072-1073) key of Cymindis to incorporate Coptodera vaporariorum and commented on the structural differences to separate that species from Coptodera unicolor which is most similar externally.
Lorenz (2005) listed all Nearctic species in the subgenus Cymindis s.str. However, Lindroth (1969a: 1070) noted that the North American species belong to the subgenus Tarulus, to which Coptodera vaporariorum is included.
This species ranges from southern Quebec (Larochelle 1975: 78) to eastern South Dakota (Kirk and Balsbaugh 1975: 39), south to northeastern Texas (Lamar County, Brian Raber pers. comm. 2010), southern Louisiana (Chaudoir 1873c: 103), northern Alabama (Madison County, CMNH), and southeastern South Carolina (Ciegler 2000: 119). The record from “Florida” (Bousquet and Larochelle 1993: 267) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, GA, KS, IA, IL, IN, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [FL]
This species ranges from southwestern California to “New Mexico” (Lindroth 1969a: 1075).
USA: AZ, CA, NM
The range of this species extends from Newfoundland (Lindroth 1955a: 132) to southern British Columbia, south to “New Mexico” (Lindroth 1969a: 1082), northern Wisconsin along Lake Superior (Wickham 1896c: 135; MCZ), northwestern Pennsylvania (Erie County, CMNH), and Connecticut (Krinsky and Oliver 2001: 272). The record from “South Dakota” (Bousquet and Larochelle 1993: 267) is probably in error.
CAN: AB, BC, MB, NB, NF, NS (CBI), ON, QC, SK USA: CO, CT, ME, MI, MN, ND, NH, NM, NY, PA, UT, VT, WI, WY
This species is known so far only from the holotype (Lindroth 1969a: 1086) collected in southwestern California.
USA: CA
This widely distributed species ranges from Newfoundland (Lindroth 1955a: 131) to Vancouver Island, north to Yukon Territory (Lindroth 1969a: 1076), south to “Oregon” (Horn 1882: 152), southeastern Arizona (Snow 1906b: 162), southern New Mexico (Otero County, CMNH; Lindroth 1969a: 1076), northeastern South Dakota (Kirk and Balsbaugh 1975: 38), and western Maryland (Bailey et al. 1994: 320). The records from “Prince Edward Island” (Bousquet and Larochelle 1993: 267, see Majka et al. 2008: 133), “Kansas” (Horn 1872c: 385), and “Nebraska” (Wickham 1896c: 135) need confirmation; that from southern California (Moore 1937: 12) is probably in error though one old specimen labeled “Cal.” is known (MCZ).
FRA: PM CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, QC, SK, YT USA: AZ, CO, CT, MA, MD, ME, MI, MN, MT, ND, NH, NJ, NM, NY, OH, OR, PA, RI, SD, UT, VT, WA, WI, WY [CA, KS, NE, PE]
This species is found from Massachusetts (Darlington 1936b: 148; Hampden County, MCZ) south to the Florida Panhandle (Peck and Thomas 1998: 24) and southwestern Alabama (Casey 1920: 288).
USA: AL, DC, FL, GA, MA, NC, NJ, NY, RI, SC
This species is known from a few localities in southern Oregon (Westcott et al. 2006: 8; Harney County, CMNH; Lake County, Foster F. Purrington pers. comm. 2009), west-central Idaho (Valley County, USNM), southwestern Wyoming (Parmenter and MacMahon 1984: 26), south-central Utah (Casey 1913: 179; Knowlton 1939: 2; Piute County, MCZ), “Nevada” (CMNH, MCZ), and east-central California (Lindroth 1969a: 1079). The record from Seattle, Washington (Hatch 1953: 158) needs confirmation.
USA: CA, ID, NV, OR, UT, WY [WA]
This species is found from the southern part of the Prairie Provinces south to southeastern Arizona (Graham County, UASM), central New Mexico (Lindroth 1969a: 1074), western Texas (Ward County, Ken Karns pers. comm. 2009), and northwestern Oklahoma (Woods County, CMNH).
CAN: AB, MB, SK USA: AZ, CO, KS, MN, ND, NE, NM, OK, SD, TX, WY
This species ranges from northwestern South Dakota (Kirk and Balsbaugh 1975: 38) to western Montana (Hatch 1933a: 9), south to northern Sonora (Bates 1884: 296) and southern Texas (Sutton, Val Verde, Edwards, and Colorado Counties, CMNH). The record from “Arkansas” (Bousquet and Larochelle 1993: 268) needs confirmation; those from southern Wisconsin (Rauterberg 1885: 14) and southwestern California (Moore 1937: 12) are probably in error.
USA: AZ, CO, KS, MT, NM, OK, SD, TX, WY [AR] – Mexico
The range of this species extends from Sable Island (Christopher G. Majka pers. comm. 2007) off the coast of Nova Scotia to southern Alberta (Lindroth 1969a: 1086), south to northwestern South Dakota (Kirk and Balsbaugh 1975: 39) and southern South Carolina (Ciegler 2000: 119).
CAN: AB, MB, NB, NS, ON, QC, SK USA: CT, DC, DE, IA, IL, IN, MA, ME, MI, MN, NC, ND, NH, NJ, NY, OH, PA, SC, SD, VA, VT, WI, WV
This species ranges from southern Quebec (Larochelle 1975: 79) to southeastern North Dakota (Ransom County, CNC), north to southwestern Manitoba (Stjernberg 2011: 71), south to “Texas” (Lindroth 1969a: 1083) and northeastern Florida (Duval County, USNM). The record from the state of Puebla in Mexico (Bates 1891a: 270) needs confirmation.
CAN: MB, ON, QC USA: AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
The range of this species extends from southern Manitoba to the Okanagan Valley in south-central British Columbia, north to southern Northwest Territories (Lindroth 1969a: 1078), south to northern Oregon (Baker and Wasco Counties, CMNH, USNM; Hatch 1953: 158), northern Arizona (Coconino County, USNM; Snow 1906b: 162), northwestern Texas (Potter County, USNM), and Wisconsin (Purrington and Maxwell 1998: 190); also known from one specimen collected in southern Quebec (Lindroth 1969a: 1078).
CAN: AB, BC, MB, NT, QC, SK USA: AZ, CO, IA, ID, KS, MN, MT, ND, NE, NM, NV, OR, SD, TX, UT, WA, WI, WY
This species is known from a few localities in Washington (Hatch 1953: 158) and western Oregon (Westcott et al. 2006: 8).
USA: OR, WA
This species occurs from Newfoundland (Lindroth 1955a: 131) to southwestern Yukon Territory (Shpeley and Ball 1999: 422), south to the Sierra Nevada in east-central California (Papp 1978: 165; Nelson 1988a: Fig. 1), Santa Catalina Mountains in southern Arizona (Nelson 1988a: Fig. 1), and southern Colorado (Wickham 1902: 240; Lindroth 1969a: 1085); isolated on some mountains of northern New England. The records from “Vermont, ” “Massachusetts, ” “Connecticut, ” and “Rhode Island” (Erwin et al. 1977: 4.58) are likely in error; that from Alaska (Lindroth 1969a: 1085) probably refers to Cymindis vaporariorum Linnaeus (Shpeley and Ball 1999: 423). Fossil remnants of this species, dated between 11, 200 and 20, 530 years old, have been found in northeastern Wisconsin (Schwert 1992: 78), south-central Minnesota (Ashworth et al. 1981: 77), and southeastern and northeastern Iowa (Baker et al. 1986: 96; Schwert 1992: 78; Woodman et al. 1996: 17).
CAN: AB, BC, LB, MB, NF, NT, NU, ON, QC, SK, YT USA: AZ, CA, CO, ME, MT, NH, OR, UT, WA, WY
This species is known from southeastern Arizona (Bousquet and Larochelle 1993: 334) and central New Mexico (Torrance County, Ken Karns pers. comm. 2009) south to the state of Durango along the Sierra Madre Occidental (Ball and Shpeley 1992a: 61).
USA: AZ, NM – Mexico
This Holarctic species ranges from Ireland to the Far East, south to Spain, Portugal, Greece, Kazakhstan, Mongolia, northern China (Kabak 2003: 418), and in the Nearctic Region from Alaska to northwestern Northwest Territories [see Shpeley and Ball 1999: Fig. 6].
CAN: NT, YT USA: AK – Holarctic
Nineteen species in the temperate, subtropical, and tropical regions of North America (11 species) and Middle America (11 species).
Casey (1920) published a key to all but three (Pinacodera complanata, Pinacodera punctigera, and Pinacodera punctifera) of the currently valid North American species. As usual his key is difficult to use. A taxonomic revision of the subgenus is needed.
Lorenz (2005: 465) listed Pinacodera as a valid genus. However, Ball and Hilchie (1983: 149) presented arguments to treat this group as a subgenus of Cymindis.
This species is known only from the type series.
USA: CO
This species is known only from the type series.
USA: CO
This species ranges from east-central Georgia (Emanuel County, UASM) to southern Florida (Collier County, CMNH). The record from “South Carolina” (Bousquet and Larochelle 1993: 268) needs confirmation.
USA: FL, GA [SC]
This species is known only from the type locality in southeastern Arizona.
USA: AZ
This species ranges from Maryland (Prince Georges County, UASM) and northeastern Virginia (UASM) south to central Florida (Peck and Thomas 1998: 24) and southwestern Alabama (Mobile and Washington Counties, UASM). All other state records listed in Bousquet and Larochelle (1993: 269) as well as that from southeastern Pennsylvania (Rathvon 1869: 524, as Lebia russata) need confirmation.
USA: AL, FL, GA, MD, NC, SC, VA [LA, NJ, PA, TX]
This species ranges from Nova Scotia (Lindroth 1954c: 307) to northwestern Minnesota (Becker County, UASM), south to southeastern Texas (Brazoria County, CMNH) and central Florida (Peck and Thomas 1998: 24). The record from “North Dakota” (Bousquet and Larochelle 1993: 269) needs confirmation.
CAN: NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV [ND]
This species is known only from the type locality in central North Carolina and one locality in northern Georgia (Fattig 1949: 40).
USA: GA, NC
This species ranges from southwestern Maine (Majka et al. 2011: 47) and southern Quebec (Bousquet 1987a: 133) to eastern North Dakota (Tinerella 2003: 636), south to western Texas, Nuevo León in Mexico, and southern South Carolina [see Hunting 2009: Fig. 4.12]. The record from northern Sonora (Bates 1884: 296) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MN, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, TN, TX, VA, VT, WI, WV – Mexico
Cymindis platicollis (Say). This Cymindis is a typical element of the temperate forest of eastern North America. Unlike most carabids inhabiting the temperate and boreal regions, this species lives above the surface of the ground in trees and is usually collected by beating branches. An arboreal way of life is a common feature among tropical carabids.
This species is known from western Arizona to western Texas, south to Nuevo León, Coahuila, Durango, and Sonora in Mexico (Ball and Shpeley 1992a: 62).
USA: AZ, NM, TX – Mexico
This species ranges from northeastern California to north-central Colorado, south to eastern Michoacán in Mexico and southern California [see Hunting 2009: Fig. 4.23].
USA: AZ, CA, CO, NM, NV, TX, UT – Mexico
This species is known only from the type locality in southern Arizona.
USA: AZ
About 115 species (Lorenz 2005: 464-465) in the Nearctic (eight species of Apenes), Neotropical (72 species of Apenes), Australian (one species in New Guinea), Oriental (nine species), Palaearctic (22 species), and Afrotropical (14 species) Regions. These species are arrayed in five genera: Apenes (75 species), Habutarus Ball and Hilchie (one species from New Guinea), Cymindoidea Laporte (15 species), Platytarus Fairmaire (12 species), and Trymosternus Chaudoir (13 species from the Iberian Peninsula and northern Africa).
1. The family-group names Platytarini and Trymosternini were proposed by Zaballos and Jeanne (1994: 116) and recorded as synonyms of Apenina by Lorenz (2005: 464). However, both names are unavailable since they were not accompanied by a description or a reference to a published description (ICZN 1999: Article 13.1). 2. Apenina came out as the sister-group to the remaining lebiine subtribes, with the exclusion of sugimotoinines, actenocynines, and pericalines, in Ball et al. (1995) and Casale (1998: Fig. 91) cladistic analyses.
Seventy-five species in the Western Hemisphere arrayed in two subgenera: Apenes s.str. (67 species) and Didymochaeta Chaudoir (eight Neotropical species).
Sixty-seven species in the temperate, subtropical, and tropical areas of the Nearctic (nine species) and Neotropical (64 species) Regions.
Horn (1882: 156) wrote a key to four of the species found in North America. A taxonomic revision of the subgenus is needed.
This species is known from Alabama (Löding 1945: 22) and northwestern Georgia (Fattig 1949: 41) south to southern Florida (Peck and Thomas 1998: 23).
USA: AL, FL, GA
This species is known from Monroe and Dade Counties in southern Florida (Thomas 2011: 308), Cuba, Jamaica, and the Grand Cayman Island [see Ball and Shpeley 2009: Fig. 9].
USA: FL – Cayman Islands, Cuba, Jamaica
This species is known from the Mexican states of Morelos, Colima (Bates 1891a: 271) and Baja California Norte (Notman 1919b: 234) and from southern Arizona (Ober and Maddison 2008: 30).
USA: AZ – Mexico
This subspecies ranges from Massachusetts (Davidson et al. 2011: 514) to southeastern Minnesota (Gandhi et al. 2005: 932), south to southern Texas (Live Oak County, Brian Raber pers. comm. 2010), northwestern Louisiana (Natchitoches Parish, USNM), and central Florida (Highlands and Pasco Counties, CMNH; Horn 1882: 157); also known from southern Arizona (Pima and Santa Cruz Counties, UASM, CMNH) and the Baja California Peninsula (Horn 1895: 226).
USA: AL, AZ, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, NC, NJ, NY, OH, PA, RI, SC, TN, TX, VA, WV – Mexico
The subspecies Apenes lucidulus dulculia Ball and Shpeley and Apenes lucidulus michelii Ball and Shpeley are found in the West Indies.
This species ranges from southern and western Texas (Duval, Val Verde, and Jeff Davis Counties, CMNH) to southeastern California (Dajoz 2007: 20) and the Baja California Peninsula (LeConte 1867b: 364).
USA: AZ, CA, NM, TX – Mexico
This species is found along the Coastal Plain from South Carolina (Ciegler 2000: 120) to southern Florida (Peck and Thomas 1998: 23), west to southern Alabama (Löding 1945: 22); also recorded from the Bahamas (Turnbow and Thomas 2008: 11).
USA: AL, FL, GA, SC – Bahamas
This species ranges from southern Arizona (Bousquet and Larochelle 1993: 334) to Panama (Bates 1883a: 189).
USA: AZ – Guatemala, Mexico, Panama
This subspecies is known from Dade County in southeastern Florida (Thomas 2011: 307), the Bahamas, Cuba, and the Cayman Islands [see Ball and Shpeley 2009: Fig. 13].
USA: FL – Bahamas, Cayman Islands, Cuba
Two other subspecies, Apenes parallela inaguae Darlington and Apenes parallela sublaevis Darlington, are known from the West Indies.
The range of this species extends from Rhode Island (USNM) to northeastern Kansas (Popenoe 1877: 23), including southernmost Ontario (Lindroth 1969a: 1088), south to southern Texas (Wickham 1897: 112; Galveston and San Patricio Counties, UASM) and the Florida Keys (Peck and Thomas 1998: 23).
CAN: ON USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MO, MS, NC, NE, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA, WV
Worldwide, with about 735 species arrayed in 48 genera (Lorenz 2005: 470-478, 480, as Lionychina, Pseudotrechina, Dromiina, Lichnasthenina, Singilina, plus Metaxymorphus). The North American fauna is represented by 33 species (about 4.5 % of the world fauna), of which two are adventive.
Jeannel (1949a: 991) grouped this subtribe and the demetriadines under the subfamily Dromiitae and stated that this complex is characterized by having the basal bulb of the median lobe reduced. The peliocypidines were included in this subtribe by Ball and Bousquet (2000: 112).
About 105 species (Lorenz 2005: 475-477) in the Nearctic (two species), Neotropical (20 species), Australian (one adventive species in New Zealand), Oriental, Palaearctic (53 species), and Afrotropical Regions. These species are placed in three subgenera: Dromius s.str. (about 50 species), Obodromius Jedlička (one species), and Klepterus Péringuey (47 species), with six species unplaced.
About 50 species in the Nearctic (two species, one of them adventive), Neotropical (20 species), Australian (one adventive species in New Zealand), Oriental (four species), and Palaearctic (28 species) Regions.
Larson (1998: 126) discussed the structural differences between the two species found in North America.
This European species is adventive in North America where it is known only from three specimens collected in Newfoundland (Larson 1998: 126) and Nova Scotia (Majka and Klimaszewski 2004: 9; Halifax County, UASM). The first inventoried specimen collected on this continent was found in 1952 in Armdale, near Halifax, Nova Scotia (UASM).
CAN: NF, NS – Adventive
The range of this species extends from Cape Breton Island (Bousquet 1987d: 107) to Vancouver Island (Lindroth 1969a: 1043), south to southern California (Fall 1901a: 48; MCZ), the states of Sonora (CNC) and Durango (CNC) in Mexico, and the Florida Panhandle (Peck and Thomas 1998: 24). Fossil remnants, undated but believed to be younger than 3 million years B.P., have been unearthed in northwestern Yukon Territory (Elias and Matthews 2002: 914).
CAN: AB, BC (VCI), MB, NB, NS (CBI), ON, PE, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, FL, GA, IA, IL, IN, KY, LA, MA, MD, ME, MI, MN, MS, MT, NC, NE, NH, NJ, NM, NY, OH, OR, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV, WY – Mexico
Lindroth (1969a: 1042-1043) studied superficially the structural variation in this species and concluded there was a single species but that the specimens west of the Rockies “may very well be treated” as a subspecies for which the name Dromius quadricollis LeConte, 1859 is available. Until a detailed study of the variation is done, I prefer not to recognize subspecies for this taxon.
About 50 species (Lorenz 2005: 477) in the Nearctic (two species, one of them adventive), Oriental (two species), Palaearctic (about 35 species, 15 of them endemic to the Canary and Madeira Islands), and Afrotropical (11 species) Regions.
Bousquet (2004b: 49-51) commented on the structural differences between the two species found in North America.
This Coastal Plain species is found from “Massachusetts” (Lindroth 1969a: 1043) to southern Florida (Peck and Thomas 1998: 24), west to Louisiana (LeConte 1880b: 163) and north to western Tennessee (Tipton County, CMNH).
USA: AL, CT, FL, GA, LA, MA, MS, NJ, NY, RI, SC, TN, VA
This Palaearctic species is adventive in North America where it known from four specimens (CNC) collected in 1996 on Vancouver Island, at Saanich Peninsula, Island View Beach (Bousquet 2004b: 49).
CAN: BC (VCI) – Adventive
About 130 species (Lorenz 2005: 473-474) in the Nearctic (eight species), Neotropical (one species endemic to Cuba and one species in northern Mexico), Oriental, Palaearctic (about 55 species), and Afrotropical Regions.
Lindroth (1969a: 1047-1054) reviewed the Nearctic species. Since then, one new North American species (Microlestes lindrothi) has been described by Mateu (1995).
This subspecies is known from scattered localities from southernmost Ontario (Lindroth 1969a: 1054) to southern Arizona (Pima County, CMNH), southwestern Texas (Val Verde County, CNC), northwestern Alabama (Walker County, CNC), and southwestern North Carolina (Macon County, CMNH). One specimen simply labeled from California is also known (Lindroth 1969a: 1054).
CAN: ON USA: AL, AR, AZ, CO, IA, KS, MO, MS, NC, OH, OK, TX, WV [CA]
The subspecies Microlestes brevilobus mexicanus Mateu is known from Michoacán in Mexico.
This species is known from scattered localities from central Saskatchewan (Lindroth 1969a: 1053) to the Okanagan Valley in south-central British Columbia (Blades and Maier 1996: 66), south to the San Francisco area (Casey 1920: 270), northern Utah (Lindroth 1969a: 1053), northeastern Colorado (Bell 1971: 47), and southwestern South Dakota (Kirk and Balsbaugh 1975: 38).
CAN: AB, BC, SK USA: CA, CO, OR, SD, UT, WY
This species is known only from the original two specimens collected at Berkeley, on the San Francisco Bay in western California.
USA: CA
The range of this species extends from Nova Scotia to southwestern British Columbia, though not quite reaching the coast (Lindroth 1969a: 1051), south to northern California (Lindroth 1969a: 1051; Casey 1920: 269, as Blechrus prominulus), northern Arizona (Lindroth 1969a: 1051), southern Oklahoma (Elliott et al. 2006: 126), and “Virginia” (CMNH, collection Ulke). The record from “Mississippi” (Bousquet and Larochelle 1993: 274) needs confirmation.
CAN: AB, BC, MB, NS, ON, QC, SK USA: AR, AZ, CA, CO, CT, DC, IA, ID, IL, IN, KS, MA, MD, ME, MI, MN, MO, MT, ND, NE, NH, NJ, NV, NY, OH, OK, OR, PA, UT, VA, VT, WA, WI, WY [MS]
This species has passed under the name of Blechrus glabratus (Duftschmid, 1812) until the 1960s.
This subspecies is known from southern California (Fall 1901a: 48; Mateu 1974: 264), “Nevada” (Horn 1882: 134), southeastern Colorado (Bent County, CNC), and central Texas (Casey 1920: 270). The records from northeastern Kansas (Popenoe 1877: 23) and “Oregon” (Horn 1882: 134) need confirmation. The previous records from “Colorado” (Wickham 1902: 239; Mateu 1974: 264) were probably based on a misinterpretation of the type locality.
USA: CA, CO, NV, TX [KS, OR]
The subspecies Microlestes lucidus subdeserticus Mateu is known from the states of Durango and Querétaro in Mexico.
This species is known from a few specimens collected along the coast in southwestern British Columbia, west-central Washington (Lindroth 1969a: 1049), and Oregon (Lane County, CNC).
CAN: BC USA: OR, WA
The range of this species extends from Vancouver Island to eastern South Dakota (Kirk and Balsbaugh 1975: 38; French et al. 2004: 557), south to southern Colorado (Wickham 1902: 239; Boulder County, CMNH), northern Arizona (Coconino County, CMNH), and southern California (Lindroth 1969a: 1053). The record from “New Mexico” (Wickham 1896c: 135) needs confirmation.
CAN: BC (VCI) USA: AZ, CA, CO, ID, OR, SD, UT, WA, WY [NM]
This species is found from Long Island, New York (Notman 1928: 238) to eastern South Dakota (Kirk and Balsbaugh 1975: 38), including southernmost Ontario (Lindroth 1969a: 1054), south to southeastern Texas (Horn 1882: 135; Snow 1906a: 141; Casey 1920: 270), east-central Mississippi (Snodgrass and Cross 1983: 17), and northeastern Tennessee (Hylton 1980: 25). The record from the District of Columbia (Ulke 1902: 7) needs confirmation.
CAN: ON USA: IL, IN, KS, LA, MO, MS, NY, OH, OK, PA, SD, TN, TX, WI [DC]
About 60 species (Lorenz 2005: 472) in the Nearctic (13 species), Neotropical (five species in Middle America and the West Indies), Australian, Oriental, Palaearctic (27 species), and Afrotropical Regions.
Casey (1920: 272-276) wrote a key to all but one (Apristus latens) North American species. As usual his key is difficult to use. Ten of the current 13 species in North America have been described by Casey (1920) and only two (Apristus constrictus and Apristus pugetanus) have been study subsequently (Lindroth 1969a). A taxonomic revision of the genus is needed.
This species is known only from the type locality in central North Carolina and two localities in northern Georgia (Fattig 1949: 39).
USA: GA, NC
This species is known only from the holotype collected in southwestern California.
USA: CA
This species is known only from the holotype collected along the coast of California.
USA: CA
The range of this species extends from southwestern Alberta to Vancouver Island, south to west-central California (Lindroth 1969a: 1046) and southwestern Colorado (Elias 1987: 634).
CAN: AB, BC (VCI) USA: CA, CO, MT, OR, WA
The range of this species extends from Cape Breton Island (Lindroth 1969a: 1046) to southern Saskatchewan (Ronald R. Hooper pers. comm. 2007), south to southern Arizona (Pima County, CMNH), central Texas (Casey 1920: 274, as Apristus fuscipennis; Terrell County, CMNH), and southern Georgia (Torres and Ruberson 2006: 31). The record from “Montana” (Bousquet and Larochelle 1993: 273) needs confirmation.
CAN: MB, NB, NS (CBI), ON, QC, SK USA: AL, AR, AZ, CO, GA, IA, IL, MA, MD, ME, MN, MS, NE, NH, NJ, NM, OH, OK, PA, SD, TN, TX, VA, VT, WV [MT]
This species has passed under the name Apristus subsulcatus (Dejean, 1826) in the literature until Lindroth (1969a: 1045-1046).
This species is known from east-central Oregon (Grant County, UASM; Horn 1882: 134; Lindroth 1969a: 1044) to northern Colorado (Armin 1963: 179), including southern Idaho (Horning and Barr 1970: 25), south to central New Mexico (Bernalillo County, CMNH), southern Arizona (Pima County, CMNH; Horn 1882: 134), and the Baja California Peninsula (Horn 1894: 310).
USA: AZ, CA, CO, ID, NM, NV, OR, UT – Mexico
This species is known only from the two syntypes collected along coastal California.
USA: CA
This species is known only from the holotype collected in northwestern Nevada.
USA: NV
This species ranges from Vancouver Island (Lindroth 1969a: 1047) to northwestern Montana (Flathead County, Ken Karns pers. comm. 2009), south to southwestern New Mexico (Hidalgo County, CMNH), southern Arizona (Graham, Pinal, Cochise, Pima, and Greenlee Counties, CMNH), and northern California (Shasta and Yolo Counties, CMNH).
CAN: BC (VCI) USA: AZ, CA, CO, ID, MT, NM, OR, UT, WA
This species is known only from the holotype collected in western California.
USA: CA
The range of this species extends from Cape Breton Island (Lindroth 1954c: 307, as Apristus cordicollis) to Minnesota (Gandhi et al. 2005: 932), south to Mississippi (Casey 1920: 275, as Apristus cordicollis), southeastern Alabama (Kharboutli and Mack 1991: 1017), and southern South Carolina (Ciegler 2000: 122). The records from Colorado (LeConte 1879d: 500; Armin 1963: 179), “New Mexico, ” and Texas (Horn 1882: 133; Wickham 1897: 109) need confirmation since they could refer to Apristus latens (LeConte).
CAN: NB, NS (CBI), ON, QC USA: AL, AR, CT, DC, GA, IA, IL, IN, MA, MD, ME, MI, MN, MO, MS, NH, NJ, NY, OH, PA, RI, SC, VA, VT, WI, WV [CO, NM, TX]
This species is known only from the syntypes collected in northwestern New Mexico.
USA: NM
This species is known only from the type series collected in southern Arizona.
USA: AZ
About 50 species (Lorenz 2005: 470-471) in the boreal, temperate, and tropical areas of the Nearctic (one species), Australian (one widely distributed Old World species), Oriental, Palaearctic (about 35 species), and Afrotropical Regions.
The North American species is included in Lindroth’s (1969a: 1056) monograph on the Canadian and Alaskan Carabidae.
The range of this species extends from Newfoundland (Lindroth 1955a: 130) to east-central Alaska (Lindroth 1969a: 1056), south to the Sierra Nevada in California (Dajoz 2007: 16), southeastern Arizona (Graham and Greenlee Counties, UASM), central New Mexico (Fall and Cockerell 1907: 160), south-central South Dakota (Kirk and Balsbaugh 1975: 38), central Illinois (Wolcott 1895: 309), and western North Carolina (Yancey County, USNM) along the Appalachians. The records from “Nebraska” and “Texas” (Bousquet and Larochelle 1993: 274) need confirmation.
CAN: AB, BC (VCI), LB, MB, NB, NF, NS (CBI), NT, ON, PE, QC, SK, YT USA: AK, AZ, CA, CO, CT, DC, IA, ID, IL, IN, MA, MD, ME, MI, MN, MT, NC, ND, NH, NJ, NM, NY, OH, OR, PA, RI, SD, TN, UT, VA, VT, WA, WI, WV, WY [NE, TX]
Fifteen species in the temperate, subtropical, and tropical areas of the Nearctic (seven species) and Neotropical (nine species) Regions.
There is no modern taxonomic revision of the species of Axinopalpus and such study is much needed. A preliminary study of the type material of the species described by Casey and Hatch led to the three new synonyms proposed herein.
This species ranges from “Maine” (Larochelle and Larivière 1990a: 32) to “Washington” (Hatch 1953: 156), north to the southern part of the Prairie Provinces (Bousquet 1987a: 133), south to southwestern California (Casey 1920: 266, as Axinopalpus demissus; Moore 1937: 12), southern Arizona (Wickham 1898: 300; Ober and Maddison 2008: 31), southern Texas (Wickham 1897: 109; Kerr and Hidalgo Counties, CNC, USNM), and west-central Georgia (Fattig 1949: 39). The records from “Pennsylvania, ” “Florida” (Bousquet and Larochelle 1993: 272), Vancouver Island (see Lindroth 1969a: 1058), and Mexico (Blackwelder 1944: 58) need confirmation.
CAN: AB, MB, ON, QC, SK USA: AL, AZ, CA, CO, CT, DE, GA, IA, ID, IL, IN, KS, MA, ME, MI, MN, MO, ND, NE, NH, NJ, NM, NY, OK, OR, RI, SD, TX, UT, VT, WA, WI, WY [BC, FL, PA]
This species is known only from the holotype collected in central Washington.
USA: WA
This species ranges from western Idaho (Washington County, CNC) to northern Oklahoma (Alfalfa County, CMNH), south to Guatemala (Bates 1883a: 193) and southwestern California (LeConte 1851: 175; Fall 1901a: 48).
USA: AZ, CA, CO, ID, NM, OK, TX – Guatemala, Mexico
This species is known only from the type series collected in west-central California.
USA: CA
This species is known only from the holotype collected in northwestern Washington.
USA: WA
This species is known only from Utah and Washington Counties in Utah (Tanner 1928: 270).
USA: UT
This species is known from the type locality in west-central Washington (Hatch 1953: 156) and from southeastern Oregon (Westcott et al. 2006: 6).
USA: OR, WA
Worldwide, with about 800 species arrayed in 25 genera (Lorenz 2005: 481-489). The North American fauna has 49 species (about 6 % of the world fauna) placed in two genera.
Casale’s (1998: Fig. 91) cladistic analysis suggests that this subtribe is possibly the sister-group to {cymindidines + dromiusines + physoderines + agrines + metallicines + calleidines + demetriadines + peliocypadines}.
Worldwide, with about 740 species (Lorenz 2005: 481-488) arrayed in 17 subgenera. The North American fauna includes 48 species (about 6.5 % of the world fauna) in the boreal, temperate, and subtropical regions. These species are currently placed in four subgenera.
Madge (1967) revised the North American species. Since the publication of his work, one of his species (Lebia viridis) has been shown to be a complex of two species (Lebia viridis and Lebia moesta) by Lindroth (1969a).
Twenty-five species in the Nearctic (eight species) and Neotropical (18 species) Regions.
[atriventris group]
This species ranges from southern Manitoba to eastern Oregon (Baker County, James R. LaBonte pers. comm. 1992), as far north as central Alberta, south to northern Sonora (Bates 1884: 298) and southwestern Texas [see Madge 1967: Fig. 141]. On specimen is known from Strafford County, northeastern Virginia (Hoffman 2010: 23), probably resulting from anthropogenic transport.
CAN: AB, MB, SK USA: AZ, CO, ID, KS, MT, ND, NE, NM, NV, OK, OR, SD, TX, UT, WY – Mexico
This species ranges from “Maine” (Larochelle and Larivière 1990a: 32) to southwestern Montana, north to northern Alberta (Fort McMurray area, Gerald J. Hilchie pers. comm. 2009), Saskatchewan and southern Manitoba, south to central New Mexico (Fall and Cockerell 1907: 159), central Texas, and northeastern Florida [see Madge 1967: Fig. 117]. The records from “California” (Erwin et al. 1977: 4.61; Bousquet and Larochelle 1993: 274) and Mexico (Chaudoir 1871a: 143) are likely in error.
CAN: AB, MB, ON, QC, SK USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
[tricolor group]
This species is known from southern Arizona and western Texas (Madge 1967: 159).
USA: AZ, TX
This species is found east of the Rocky Mountains from southern Quebec (Larochelle 1975: 91) to northeastern North Dakota (Tinerella 2003: 637), south to southeastern Texas along the Rio Grande and southern South Carolina, west to western Texas and western Oklahoma [see Madge 1967: Fig. 137]. The records from Baja California (Horn 1894: 310), Arizona (Wickham 1898: 300; Snow 1907: 142, as Lebia majuscula), New Mexico (Fall and Cockerell 1907: 159, 160 as Lebia majuscula), and “Colorado” (Snow 1877: 17; Wickham 1902: 239) need confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [AZ, CO, NM]
This species is known so far only from southern Arizona (Madge 1967: 159).
USA: AZ
This species is known from southern Arizona and western Texas (Madge 1967: 158), including southwestern New Mexico (Luna County, Ken Karns pers. comm. 2009).
USA: AZ, NM, TX
This species ranges from southern Arizona to western Texas (Madge 1967: 161).
USA: AZ, NM, TX
This species occurs from Nova Scotia (Halifax and Colchester Counties, CNC) to eastern Minnesota, north to southern Manitoba (CNC), south to northern Oklahoma (Alfalfa County, Foster F. Purrington pers. comm. 2010), southern Louisiana, and central Florida [see Madge 1967: Fig. 126]. Old specimens simply labeled from “Texas” (Madge 1967: 157) are known.
CAN: MB, NB, NS, ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, VA, VT, WI, WV [TX]
One species in southern United States and northeastern Mexico.
This species is known from a few specimens collected in western Texas (Madge 1967: 164), southeastern Arizona (Cochise County, CNC, UASM), and “Tamaulipas” (MCZ, collection LeConte) and Nuevo León (UASM) in Mexico. The record from Baja California (Horn 1894: 310, as Lebia testacea) needs confirmation.
USA: AZ, TX – Mexico
Fourteen species in the Nearctic (one species) and Palaearctic (13 species) Regions.
This species ranges from the Prairie Provinces, as far north as central Alberta, south to northern Colorado (Wickham 1902: 239) and eastern Kansas (Popenoe 1877: 23), east to west-central Indiana (Downie 1957: 116; Schrock 1985: 351), west to northwestern Idaho [see Madge 1967: Fig. 120]. The record from northeastern New Mexico (Fall and Cockerell 1907: 159) needs confirmation.
CAN: AB, MB, SK USA: CO, ID, IL, IN, KS, MN, MT, SD, WI, WY [NM]
Worldwide, with about 525 species (Lorenz 2005: 481-488) of which 38 (about 7 % of the world fauna) occur in North America.
[analis group]
This species ranges from northwestern Vermont to eastern South Dakota (Kirk and Balsbaugh 1975: 37), including southernmost Ontario (Bousquet 1987a: 133), south to Guatemala (Bates 1883a: 229, as Lebia anchora) and the Florida Keys, west along the southwest to southeastern Arizona [see Madge 1967: Fig. 130] and the Baja California Peninsula (Blackwelder 1944: 52); also recorded from the Bahamas (Turnbow and Thomas 2008: 13). The record from “Maine” (Bousquet and Larochelle 1993: 276) needs confirmation.
CAN: ON USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, NE, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [ME] – Bahamas, Guatemala, Mexico
[bitaeniata group]
This species ranges from southeastern Texas (Madge 1967: 172) to Colombia (Martínez 2003: 14); also recorded from the Bahamas (Turnbow and Thomas 2008: 13), Cuba (Gundlach 1891: 16), Jamaica (Darlington 1941a: 14), Puerto Rico (Wolcott 1936: 188), and Dominica (Peck 2006: 176).
USA: TX – Bahamas, Colombia, Costa Rica, Cuba, Dominica, Guatemala, Jamaica, Mexico, Nicaragua, Puerto Rico
[bivittata group]
This species ranges from New Jersey to northeastern Kansas (Popenoe 1877: 23), south to Nicaragua (Bates 1883a: 240) and southern Florida, west to northwestern Texas [see Madge 1967: Fig. 119]; also recorded from Cuba (Gundlach 1891: 16; Darlington 1934: 113) and Jamaica (Darlington 1941a: 14). The record from “Wisconsin” (Bousquet and Larochelle 1993: 276) needs confirmation.
USA: AL, AR, DC, DE, FL, GA, IL, IN, KS, LA, MD, MO, NC, NJ, OH, PA, SC, TN, TX, VA, WV [WI] – Belize, Cuba, Guatemala, Jamaica, Mexico, Nicaragua
This species ranges from central Idaho and northern Oregon south to southern California along the Mexican border [see Madge 1967: Fig. 133].
USA: CA, ID, NV, OR
The range of this species extends from northern New Jersey to north-central Colorado, north to southeastern Michigan, south at least to southern Arizona, Guanajuato and the Federal District in Mexico (Bates 1883a: 241), and central Georgia [see Madge 1967: Fig. 124]. At least one old specimen simply labeled from Wisconsin is known (Madge 1967: Fig. 124). The record from “Massachusetts” (Leng and Beutenmüller 1893: 142) needs confirmation.
USA: AL, AR, AZ, CO, DC, DE, GA, IA, IL, IN, KS, KY, LA, MD, MI, MO, MS, NE, NJ, NM, NY, OH, PA, TX, VA [MA, WI] – Mexico
[collaris group]
This rarely collected species is known from southern Ohio (Purrington et al. 1999: 48) and southern Indiana, from North Carolina to southern Florida [see Madge 1967: Fig. 136], from southeastern Louisiana (Saint Tammany Parish, Igor M. Sokolov pers. comm. 2009), from east-central Texas (Riley 2011), and from western Arkansas (Polk and Garland Counties, Robert L. Davidson pers. comm. 2012). The records from New York (LeConte 1846b: 195), Alabama (Löding 1945: 21), and Cuba (Darlington 1934: 113) need confirmation.
USA: AR, FL, GA, IN, LA, NC, OH, TX [AL, NY]
[fuscata group]
This species is known from southern Arizona and the Baja California Peninsula (Madge 1967: 202).
USA: AZ – Mexico
This species ranges from southeastern Texas (Madge 1967: 212) along the Rio Grande south at least to Guatemala (Bates 1883a: 231).
USA: TX – Guatemala, Mexico
This species is known from northwestern and south-central Louisiana (Caddo and Saint Landry Parishes, Igor M. Sokolov pers. comm. 2009), eastern Texas (Madge 1967: 212; Riley 2011), and “Mexico” (Erwin et al. 1977: 4.62); also recorded from the Bahamas (Turnbow and Thomas 2008: 13) and Cuba (Mateu 1977: 378). The record from South Dakota (Kirk and Balsbaugh 1975: 38) needs confirmation.
USA: LA, TX [SD] – Bahamas, Cuba, Mexico
This species is found from Nova Scotia to western Washington, as far north as southern Manitoba and southwestern British Columbia (Lindroth 1969a: 1036), south to west-central California, east-central Texas (Riley 2011), and southern Florida [see Madge 1967: Fig. 127]. The species has been rarely collected in the Great Plains.
CAN: BC (VCI), MB, NB, NS, ON, PE, QC USA: AL, AR, CA, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NY, OH, OR, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV
The range of this species extends from southern Alberta (Drumheller, CNC) and southern British Columbia south to southern California, southern New Mexico [see Madge 1967: Fig. 134], and western Texas (Jeff Davis County, Ken Karns pers. comm. 2009). One old specimen simply labeled from Kansas is known (Madge 1967: 201).
CAN: AB, BC USA: AZ, CA, CO, ID, MT, ND, NM, NV, OR, TX, UT, WA, WY [KS]
This species is known only from southeastern Texas (Madge 1967: 203).
USA: TX
This species ranges from southeastern Maine (Majka et al. 2011: 47) to eastern North Dakota (Tinerella 2003: 637), south to eastern Texas and central Florida [see Madge 1967: Fig. 135].
CAN: ON USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, LA, MD, ME, MO, MS, NC, ND, NJ, NY, OH, OK, PA, SC, SD, TN, TX, VA, WI, WV
This species ranges from Cape Breton Island, Nova Scotia (Christopher G. Majka pers. comm. 2007) to northwestern South Dakota (Kirk and Balsbaugh 1975: 37), including northern Minnesota, south to eastern Texas and central Florida (Peck and Thomas 1998: 24) [see Madge 1967: Fig. 128].
CAN: NB, NS (CBI), ON, QC USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV
This species is known only from a few specimens collected in southern Arizona (Madge 1967: 207).
USA: AZ
This species is known from southern Arizona to western Texas (Madge 1967: 206), south to Guatemala (Bates 1883a: 230).
USA: AZ, NM, TX – Guatemala, Mexico
[lecta group]
This species is known only from a few specimens collected in southeastern Texas (Madge 1967: 213).
USA: TX
This species is known only from Dade, Monroe, and Walton Counties in Florida (Peck and Thomas 1998: 24).
USA: FL
[pulchella group]
This species is found from northern New Hampshire to central Alberta, south to central California, the state of Hidalgo in Mexico (Ball and Shpeley 1992a: 64), and southern Florida [see Madge 1967: Fig. 123].
CAN: AB, MB, ON, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IL, IN, KS, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, PA, RI, SC, TN, TX, VA, WI, WV, WY – Mexico
[pumila group]
This species ranges from Nova Scotia (Lindroth 1954c: 307) and Prince Edward Island (Majka et al. 2008: 133) to southeastern Alberta, south to southeastern Texas along the Rio Grande and southern Florida. The species is also known from one locality in southwestern British Columbia and one in western Washington [see Madge 1967: Fig. 132].
CAN: AB, BC, MB, NB, NS, ON, PE, QC, SK USA: AL, AR, CT, DC, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WA, WI, WV
Lebia floricola, credited to Harris, has been cited as a synonym of this species, first by LeConte (1846b: 195), on the basis that the name was published in the “N[ew] E[ngland] Farmer.” I have not found any publication of Harris citing such a name. The name is a nomen nudum as mentioned by Madge (1967: 216).
[scalpta group]
This species ranges from southern Arizona and southern Texas (Madge 1967: 186) south at least to the Yucatán Peninsula (Bates 1883a: 230) in Mexico.
USA: AZ, TX – Mexico
[scapula group]
This species is found from Arizona (Madge 1967: 184) to western Texas (Graves and Suter 1979: 6).
USA: AZ, NM, TX
[viridipennis group]
The range of this species extends from southwestern Maine (Majka et al. 2011: 47) and southwestern Quebec (LeSage 1996: 22) to southeastern South Dakota (Kirk and Balsbaugh 1975: 37), including southernmost Ontario (Pettit 1869: 107; CNC), south to southeastern Texas along the Rio Grande and southern Florida [see Madge 1967: Fig. 122]. The record from “North Dakota” (Bousquet and Larochelle 1993: 278) needs confirmation.
CAN: ON, QC USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, LA, MA, MD, ME, MI, MN, MO, MS, NC, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [ND]
[viridis group]
The range of this species extends from southern Arizona to western Texas (Madge 1967: 175). The record from “Nevada” (Bousquet and Larochelle 1993: 276) needs confirmation.
USA: AZ, NM, TX [NV]
This species ranges from southern Saskatchewan to south-central British Columbia, south to southern California along the Mexican border and northern New Mexico [see Madge 1967: Fig. 129]. At least one specimen is known from “Texas” (Madge 1967: 177). The record from “North Dakota” (Bousquet and Larochelle 1993: 276) needs confirmation.
CAN: AB, BC, SK USA: AZ, CA, CO, ID, MT, NM, OR, UT, WA, WY [ND, TX]
The range of this species extends from New Jersey (Smith 1890: 87; Smith 1910: 210) to Arizona, north to northwestern Ohio (Hancock County, Foster F. Purrington pers. comm. 2009), southern Michigan, and northern Illinois, south to Brazil (Chaudoir 1871a: 185) and southern Florida [see Madge 1967: Fig. 125]; also found “on all the Greater Antilles” (Darlington 1953: 11, as Lebia cyanea) and Dominica (Peck 2006: 176). The record from Montana (Hatch 1933a: 9) is probably in error.
USA: AL, AR, AZ, DC, FL, GA, IL, IN, LA, MI, MO, MS, NC, NJ, NM, OH, OK, SC, TN, TX, VA – Bahamas, Brazil, Cuba, Dominica, French Guiana, Guatemala, Jamaica, Mexico, Nicaragua, Puerto Rico
This species ranges from Newfoundland (Larson and Langor 1982: 594) and southern Labrador to Vancouver Island (Lindroth 1969a: 1028-1029), south at least to northern Oregon (Tillamook and Umatilla Counties, CNC), southern Wyoming (Lavigne 1977: 46; Lincoln County, CNC), eastern Nebraska (Cuming County, Foster F. Purrington pers. comm. 2009), east-central Ohio (Usis and MacLean 1998: 67), and the District of Columbia (Ulke 1902: 7). The record from east-central Missouri (Summers 1873: 134) needs confirmation.
CAN: AB, BC (VCI), LB, MB, NB, NF, NS, ON, PE, QC, SK USA: DC, ID, IL, MA, ME, MI, MN, MT, NE, NH, NJ, NY, OH, OR, RI, SD, VT, WA, WI, WY [MO]
Madge (1967: 177) listed this form as synonym of Lebia viridis Say.
This species occurs from southern British Columbia, including Vancouver Island, south to southern California along the Mexican border, east to southwestern Idaho [see Madge 1967: Fig. 140].
CAN: BC (VCI) USA: CA, ID, OR, WA
This species extends from southwestern Quebec (LeSage 1996: 22) to south-central Saskatchewan (Hooper 1977: 51), south to northeastern Kansas (Madge 1967: 174), central Missouri (Cooper County, CMNH), and northwestern North Carolina (Watauga County, CMNH). The records from the Organ Mountains in southern New Mexico (Fall and Cockerell 1907: 160) and “Mexico” (Blackwelder 1944: 55) are probably in error.
CAN: ON, QC, SK USA: DC, IA, IL, IN, KS, MA, MI, MN, MO, NC, NE, NJ, NY, PA, SD, VT, WI
This species is known only from a few localities in southeastern Texas (Madge 1967: 173).
USA: TX
This species is found from southern California to western Texas, north to Mesa County in Colorado (Madge 1967: 175).
USA: AZ, CA, CO, NM, TX
This widely distributed species ranges from Nova Scotia to northwestern Yukon Territory, south to southern California along the Mexican border, Guatemala (Chaudoir 1871a: 192), and southern Florida [see Madge 1967: Fig. 139]; also reported from some islands of the Greater Antilles (Blackwelder 1944: 56). According to Shimonoya (2004), this species has been recently found in Fukui Prefecture, Japan.
CAN: AB, BC (VCI), MB, NB, NS (CBI), NT, ON, PE, QC, SK, YT USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WV, WY – Cuba, Dominican Republic, Guatemala, Mexico, Puerto Rico
[vittata group]
This species ranges from southern Arizona (Madge 1967: 194) to Guatemala (Bates 1883a: 240).
USA: AZ – Guatemala, Mexico
This species is found in southern Arizona (Madge 1967: 189). At least one specimen labeled from “Texas” is known.
USA: AZ [TX]
Madge (1967: 189) retained this name only for the Arizona (and possibly Texas) populations but pointed out that populations, probably representing the same species but with different color patterns, were seen from Mexico and Colombia.
This species is known only from a few localities in Arizona (Madge 1967: 196).
USA: AZ
This species is found from New Hampshire to northeastern Kansas (Knaus 1903: 188), south to eastern Texas and northern Florida [see Madge 1967: Fig. 121]. The state record of “New Mexico” (Bousquet and Larochelle 1993: 277), based on two specimens in Horn collection (MCZ) labeled “N.M., ” needs confirmation.
USA: AL, AR, CT, DC, FL, GA, IL, IN, KS, KY, LA, MA, MD, MI, MS, NC, NH, NJ, NY, OH, OK, PA, RI, SC, TN, TX, VA [NM]
This species is found east of the Rocky Mountains from southern Nova Scotia to southwestern Saskatchewan, south to southeastern Texas along the Rio Grande and southern Florida [see Madge 1967: Fig. 131]; also recorded from Cuba (Darlington 1934: 113) and “Mexico” (Lindroth 1969a: 1031). The record from southwestern New Mexico (Fall and Cockerell 1907: 160, as Lebia scapularis) needs confirmation.
CAN: MB, NS, ON, QC, SK USA: AL, AR, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV [NM] – Cuba, Mexico
This species is found from Nova Scotia to southeastern British Columbia (Lindroth 1969a: 1033), south to southern California, southwestern New Mexico (Fall and Cockerell 1907: 160), southeastern Texas, and southern Florida [see Madge 1967: Fig. 118]; also recorded from “Mexico” (Bates 1883a: 240) and the state of Tabasco (Bates 1891a: 274, as Lebia furcata).
CAN: AB, BC, MB, NS, ON, QC, SK USA: AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, VT, WA, WI, WY – Mexico
Seven species are known (Erwin 2004: 33) in the Neotropical Region, one of which extends into southern Texas.
Reichardt (1973) reviewed the species then known and published a key for their identification. Subsequently, two new species were described by Erwin (2004).
This species ranges from southern Texas to Costa Rica (Erwin 2004: 37).
USA: TX – Costa Rica, Mexico
Worldwide, with about 660 species arrayed in 46 genera (Lorenz 2005: 491-498). The North American fauna is represented by 24 species (about 3.5 % of the world fauna).
Erwin (2004: 6) advocated combining calleidines with agrines into one subtribe (Agrina) but did not provide any justification. In a cladistic analysis of lebiine exemplars performed by Casale (1998: Fig. 91), calleidines turned out as the sister-group to metallicines and agrines as the sister-group to physoderines. In this work, agrines are retained as a distinct subtribe. In the analysis of Ball et al. (1995), calleidines showed up as the sister-group to gallerucidiines. Jeannel (1949a: 946) listed the gallerucidiines and thysanotines (currently considered as pericalines) with the calleidines (including trichines).
Seventeen species (Lorenz 2005: 495) in the Nearctic (four species), Neotropical (14 species), and Australian (one species from New Caledonia) Regions. One species, Plochionus pallens, has become subcosmopolitan through commerce.
Horn (1882: 145) provided a key for the identification of the North American species. Since then, one new species (Plochionus bicolor) has been described by Notman (1919b: 234). A taxonomic revision of the species would be useful.
Fourteen species in the temperate, subtropical, and tropical areas of the Nearctic (four species, all of them extending into the West Indies or Mexico) and Neotropical Regions.
This species ranges from southeastern Georgia (Fattig 1949: 40; McIntosh County, CNC) to southern Florida (Peck and Thomas 1998: 24), west to southwestern Alabama (Leng 1915: 588; Mobile County, MCZ, USNM); also recorded from the Bahamas (Turnbow and Thomas 2008: 13). The record from Guana Island (Valentine and Ivie 2005: 275) in the British Virgin Islands needs confirmation.
USA: AL, FL, GA – Bahamas
This species is known only from southern Florida (Peck and Thomas 1998: 24), Cuba (Darlington 1934: 117), and Navassa (Steiner 2008: 132).
USA: FL – Cuba, Navassa
According to Ball (in Peck 2005: 39), Plochionus rubrofasciatus Zayas, 1988, described from Cuba, is probably a synonym of this species.
This species is known from northern Georgia (Fattig 1949: 40), the Florida Peninsula, as far south as Dade County (Peck and Thomas 1998: 24), and the Bahamas (Darlington 1953: 13).
USA: FL, GA – Bahamas
The range of this species extends from southeastern New Hampshire (Strafford County, Ross T. Bell pers. comm. 2008) to southern Wisconsin (Messer 2010: 44) and northern Iowa (Wickham 1911b: 7), including southern Ontario (Pettit 1869: 107), south to central Texas (Haldeman 1852: 373; Blanco County, CMNH), southern Louisiana (East Baton Rouge, Iberville, Saint Martin, and Saint Tammany Parishes, Igor M. Sokolov pers. comm. 2009), and central Florida (Pinellas County, CMNH; Schwarz 1878: 435), west along southwestern United States to southern California (Moore 1937: 12; Kern and Fresno Counties, CNC) and the Baja California Peninsula (Horn 1894: 310).
CAN: ON USA: AL, AR, AZ, CA, CT, DC, DE, FL, GA, IA, IL, IN, KS, LA, MA, MD, MI, MO, MS, NC, NH, NJ, NM, NV, NY, OH, PA, RI, SC, TN, TX, VA, WI, WV – Mexico
Two Neotropical species, one of them subcosmopolitan, and one New Caledonian species (Plochionus niger Fauvel).
Lindroth (1969a: 1066) stated that this species is almost cosmopolitan and probably of South American origin. In North America, it is known from “Massachusetts” (Harris 1833: 566, as Plochionus bonfilsii?), “Pennsylvania” (LeConte 1846b: 192; Lindroth 1969a: 1066), Maryland (Charles County, USNM), District of Columbia (USNM), northern Georgia (Rabun County, USNM), eastern Florida (Leng 1915: 588), Missouri (Summers 1873: 133, as Plochionus bonfilsii), Kansas (Douglas County, USNM), and Indiana (Alien County, USNM). The record from “California” (Csiki 1932b: 1451) needs confirmation.
USA: DC, FL, GA, IN, KS, MA, MD, MO, PA [CA] – Adventive
Two Nearctic species, one of them extending into northern Mexico.
Larson (1969) revised the species.
This subspecies occurs from northern California to eastern Colorado, south to southeastern Texas, Durango and Sinaloa in Mexico, and southern California [see Larson 1969: Fig. 63]. The record from “Oregon” (Bousquet and Larochelle 1993: 280) needs confirmation.
USA: AZ, CA, CO, NM, NV, TX, UT [OR] – Mexico
The range of this subspecies extends from southern Saskatchewan (several localities, CNC) and southern Alberta south to southwestern Idaho, northern Utah, and “Colorado” [Larson 1969: 62, Fig. 63].
CAN: AB, SK USA: CO, ID, MT, UT, WY
Tecnophilus croceicollis peigani Larson. Adults of this handsome subspecies are usually found during the day on clay-sand soil on the ground or climbing on very low vegetation along the bases of south facing coulees. The subspecific name derives from the name of an Indian tribe of the Blackfoot Confederation, Peigan, which inhabited the prairies of southern Alberta where the taxon is found in Canada.
This species is restricted to the Gulf Coast of Texas [see Larson 1969: Fig. 62].
USA: TX
About 275 species primarily in tropical areas of the Neotropical, Oriental, and Afrotropical Regions (over 95 % of the world fauna) with a few elements found in the Nearctic (11 species) and Palaearctic (ten species in Asia) Regions. The species are arrayed in three subgenera: Calleida s.str. (about 175 species), Callidiola Jeannel (60 species), and Stenocallida Jeannel (40 species), though some authors (e.g., Casale 2008) treat Callidiola and Stenocallida as distinct genera.
About 175 species confined to the Western Hemisphere. Only 11 species are endemic (five species) or extends (six species) into the Nearctic Region.
There is no key for the identification of the North American species.
This species is known from southeastern Texas (Schaeffer 1905: 142; MCZ) south at least to Veracruz, Mexico (Bates 1883a: 212); it may occur much further south, as far as Venezuela (Schaeffer 1910: 397).
USA: TX – Mexico
This species ranges from south-central North Dakota (Burleigh County, Donald P. Schwert pers. comm. 1989) to southeastern Virginia (Hoffman and Roble 2000: 40), south to southern Florida (Peck and Thomas 1998: 24), the Bahamas (Darlington 1953: 11), and Nicaragua (CMNH), west to southern Arizona (Santa Cruz and Pima Counties, CMNH, UASM).
USA: AL, AR, AZ, FL, GA, IA, IL, IN, KS, LA, MS, NC, ND, NE, OK, SC, SD, TN, TX, VA – Bahamas, Belize, Mexico, Nicaragua
This species is found in southern Texas (Bousquet and Larochelle 1993: 334) south at least to Veracruz (Bates 1883a: 212) in Mexico.
USA: TX – Mexico
This species ranges from southern South Carolina (Ciegler 2000: 126) to southern Florida (Peck and Thomas 1998: 24), west to “Texas” (Leng 1915: 587) and central Oklahoma (Hatch and Ortenburger 1930: 11). The records from Colorado (Wickham 1902: 240) and northeastern Kansas (Knaus 1901: 110) need confirmation.
USA: AL, FL, GA, OK, SC, TX [CO, KS]
This species is known only from the holotype collected in central Florida.
USA: FL
This species ranges from the Rio Grande Valley in southern Texas (Wickham 1897: 111) south to Guatemala and Belize (Bates 1883a: 208, as Calleida metallescens).
USA: TX – Belize, Guatemala, Mexico
This species ranges from southwestern California (Moore 1937: 12) to southeastern Texas (Cameron County, CNC), including northern Sonora (Bates 1884: 298); also known from Baja California Sur (Santa Rosa, MCZ).
USA: AZ, CA, NM, NV, TX, UT – Mexico
The range of this species extends from Maine (Dearborn and Donahue 1993: 8; Foss 2001: 14) to southern Manitoba (Bousquet 1987a: 133), south to “Kansas” (Horn 1882: 141) and northwestern South Carolina (Anderson County, Robert L. Davidson pers. comm. 2012). The records from “Texas, ” “Florida” (Erwin et al. 1977: 4.59), “Louisiana” (Chaudoir 1873b: 153), “Arkansas” (Bousquet and Larochelle 1993: 281), northern Alabama (Löding 1945: 22), and southwestern Georgia (Fattig 1949: 40) need confirmation.
CAN: MB, ON, QC USA: CT, IA, IL, IN, KS, KY, MA, MD, ME, MI, MN, MO, NC, ND, NE, NH, NJ, NY, OH, PA, SC, SD, TN, VA, VT, WI, WV [AL, AR, FL, GA, LA, TX]
This species is known from the Baja California Peninsula (Horn 1894: 361, as Calleida rugicollis) and southern Texas (Wickham 1897: 111; Cameron County, CNC), south at least to Nicaragua (CMNH). The record from “California” (Bousquet and Larochelle 1993: 281) is probably in error.
USA: TX – Guatemala, Mexico, Nicaragua
This species ranges from “New Hampshire” (Lindroth 1969a: 1062) to northeastern South Dakota (Kirk and Balsbaugh 1975: 38), north to southern Manitoba (Lindroth 1969a: 1062; Bousquet 1987a: 133), south to eastern Kansas (Popenoe 1877: 23), central Louisiana (Rapides Parish, Igor M. Sokolov pers. comm. 2009), and northern Florida (Peck and Thomas 1998: 24). The records from Colorado (LeConte 1846b: 188; Snow 1877: 17) need confirmation.
CAN: MB USA: AL, CT, FL, GA, IA, IL, KS, LA, MA, MI, MO, MN, NC, NE, NH, NJ, NY, OH, PA, RI, SC, SD, WI [CO]
This species ranges from southwestern New York (Notman 1928: 239) to eastern New Mexico (Chaves and De Baca Counties, CMNH), including southwestern Iowa (Fremont County, Doug A. Veal pers. comm. 2009), south to southeastern Texas (Wickham 1897: 111), southern Louisiana (East Baton Rouge, Iberville, Pointe Coupee, Saint Martin, and Saint Tammany Parishes, Igor M. Sokolov pers. comm. 2009), and southern Florida (Peck and Thomas 1998: 24). The records from “Utah, ” “Arizona, ” and “California” (Csiki 1932b: 1449) are likely in error.
USA: AL, AR, DC, DE, FL, GA, IA, IL, KY, LA, MD, MO, MS, NM, NY, OH, OK, PA, SC, TN, TX, VA, WV
Four species comprised this genus: one (Philophuga brachinoides Bates) is found in Mexico only, another one (Anchomenus viridis) in Canada and United States, and the other two in United States and Mexico.
Larson (1969) revised the species.
This taxon is ranked as a subgenus of Calleida by some authors.
This species ranges from Arizona south to the Isthmus of Tehuantepec [see Larson 1969: Fig. 58].
USA: AZ – Mexico
This species ranges from eastern Colorado and Kansas south to northeastern Mexico and southwestern Texas [see Larson 1969: Fig. 58]. The record from South Dakota (Kirk and Balsbaugh 1975: 38) needs confirmation. One specimen labeled from “Alabama” and two specimens from California seen by Larson (1969: 32) are probably mislabeled.
USA: CO, KS, NM, OK, TX [SD] – Mexico
The range of this subspecies extends from southern Manitoba to south-central British Columbia, south to northern Oregon, Colorado (Larson 1969: 42-43), and east-central Kansas (Knaus 1907: 233). The records from “New Mexico” and “Utah” (Bousquet and Larochelle 1993: 281), based on misplaced dots on Larson’s (1969) figure 59, are in error; those from “Oklahoma” (Arnold 2008) and “Texas” (Casey 1913: 174, as Philophuga canora) need confirmation.
CAN: AB, BC, MB, SK USA: CO, ID, KS, MT, ND, NE, OR, WA, WY [OK, TX]
This subspecies ranges from northern Washington to eastern Wyoming (Lavigne 1977: 45, as Calleida viridis viridis), south to northeastern New Mexico, south-central Arizona, and northeastern California (Larson 1969: 41, Fig. 59).
USA: AZ, CA, CO, ID, NM, NV, OR, UT, WA, WY
This subspecies is known only from south-central Oregon and northern California [see Larson 1969: Fig. 59].
USA: CA, OR
This subspecies is known only from the San Francisco Bay area in California [see Larson 1969: Fig. 59].
USA: CA
One Nearctic species extending into northern Mexico.
The character states of the sole species are described in detail by Larson (1969: 43-44).
This species is known from “Nevada” and the Lake Tahoe region in the Sierra Nevada south to the Mexican border in California [see Larson 1969: Fig. 61]. According to Ball and Bousquet (2000: 114), the species is also found in northwestern Mexico.
USA: CA, NV – Mexico
Ten species in Central America and South America, of which two reach southern United States.
This genus has not been revised in modern times and such work is needed.
This species ranges from southeastern Texas (Cameron County, Edward G. Riley pers. comm. 2009) south to the Amazonia (Chaudoir 1873b: 166). This is a new record for America north of Mexico.
USA: TX – Brazil, Colombia, Costa Rica, Guatemala, Nicaragua, Panama, Peru, Surinam.
This species is known only from Brevard County in central Florida (Peck and Thomas 1998: 24).
USA: FL
Seven Neotropical species, of which one reaches southeastern Texas.
This genus has not been revised in modern times and such work would be useful.
This species is found from southeastern Texas (Wickham 1897: 108; Snow 1906a: 141 as “Micragra brunnea Putzeys”) south to Panama and French Guiana (Bates 1883a: 199).
USA: TX – Belize, French Guiana, Guatemala, Mexico, Nicaragua, Panama.
About 585 species (Lorenz 2005: 498-502) belonging to the Western Hemisphere genus Agra.
About 585 species in the Neotropical Region, one of them extending into southeastern Texas.
1. Erwin (1982a: 45) postulated that Agra is the putative sister-group of Callidiola Jeannel of Africa and Asia, currently recognized as a subgenus of Calleida. Casale (1998: 401) challenged this hypothesis and postulated that Agra could be the sister-group to physoderines which are found in Australia, some Pacific islands, eastern Asia, and central and south Africa. 2. According to Erwin (in Ball and Bousquet 2000: 115), two species, both undescribed, reach southeastern Texas. Therefore, the species currently recorded from United States is probably not a resident of North America.
This subspecies ranges from southeastern Texas (Wickham 1897: 106) to Guatemala (Bates 1883a: 247). As mentioned previously, the Texas specimens actually belong to an undescribed species.
USA: TX – Guatemala, Mexico
The subspecies Agra oblongopunctata hypsophila Straneo is found in Costa Rica.
About 70 species (Lorenz 2005: 490) in the Nearctic (three species), Neotropical (16 species of Euproctinus), Oriental, Palaearctic (31 species), and Afrotropical Regions. These species are arrayed in four genera: Parena Motschulsky (47 species), Metallica Chaudoir (six species), Pachycallida Jeannel (three species), and Euproctinus (16 species).
This subtribe came out as the sister-group to {Calleidina + Gallerucidiina} in the phylogenetic analysis of Ball et al. (1995).
Sixteen species restricted to the Western Hemisphere, currently arrayed in two subgenera: Euproctinus s.str. (one species, Euproctinus fasciatus Solier from Chile and Argentina) and Neoeuproctus (15 species).
Shpeley (1986) revised the species and provided a key for their identification.
Fifteen species in the Neotropical Region, of which three extend into southern United States.
[abjectus group]
This species is found from southern California and southeastern Texas south to Guatemala and southern Baja California [see Shpeley 1986: map 4].
USA: CA, TX – Guatemala, Mexico
This species ranges from southeastern Arizona south to southern Mexico [see Shpeley 1986: map 3].
USA: AZ – Mexico
[trivittatus group]
This species is found along the east coast of Florida, including the Keys, and in Cuba [see Shpeley 1986: map 6].
USA: FL – Cuba
One genus endemic to the Western Hemisphere.
Jeannel (1949a: 860) and Basilewsky (1984: 527) considered this taxon as a masoreimorph (i.e., masoreines and cyclosomines). Ball and Bousquet (2000: 113-114) retained it as a lebiines, as previously done by Ball (1960b: 158) and Lindroth (1969a: 1014), but recognized that its position remains to be determined.
Nine species in the temperate and tropical areas of the Nearctic (two species) and Neotropical (eight species) Regions.
This genus has not been revised in modern times and such work is needed.
The range of this species extends from the District of Columbia (CMNH) to “Illinois” (LeConte 1853c: 378), including southwestern Pennsylvania (Westmoreland County, Robert L. Davidson pers. comm. 2008), south to southern Texas (Cameron, Colorado, and Sabine Counties, USNM, Brian Raber pers. comm. 2010) and northern Florida (Peck and Thomas 1998: 24). The record from “Iowa” (Bousquet and Larochelle 1993: 279) needs confirmation.
USA: AL, AR, DC, FL, GA, IL, LA, MD, MS, NC, OK, PA, SC, TX, VA [IA]
This species ranges from southern Texas (Cameron and Hidalgo Counties, Edward G. Riley pers. comm. 2009; Brian Raber pers. comm. 2010) south to Guatemala (Bates 1883a: 173). This is a new record for America north of Mexico.
USA: TX – Guatemala, Mexico
Worldwide, with about 290 species (Lorenz 2005: 504-507, excluding Planetina) arrayed in 20 genera. The tribe is represented in the Northern Hemisphere by about 65 species (22% of the world fauna). Three subtribes are recognized: Leleupidiina (about 90 species in the Old World and the Australian Region), Zuphiina (about 200 species), and Dicrodontina (three species endemic to the Canary Islands).
The planetines are included in this tribe by Lorenz (2005: 507). However Basilewsky (1963b), Reichardt (1967), Ball (1985), and Baehr (1986) presented arguments suggesting that planetines are probably more closely related to galeritines than to zuphiines.
Worldwide, with about 200 species placed in 14 genera. The North American fauna is represented by 18 species (about 9% of the world fauna).
Worldwide, with about 75 species of which six are found in the temperate and subtropical areas of North America.
Mateu (1981) reviewed the North American species and provided a key for their identification.
This species ranges from New Jersey (Morris County, USNM) to southern South Dakota (Kirk and Balsbaugh 1975: 39), including southernmost Ontario (Lindroth 1969a: 1090), south to southeastern Texas (Snow 1906a: 141; Cameron, Kleberg, and Gonzales Counties, CMNH, MCZ) and the Florida Keys (Peck and Thomas 1998: 25), west to southern Arizona (Mateu 1981: 118); seemingly isolated in western Oregon (Malkin 1943: 52).
CAN: ON USA: AL, AR, AZ, FL, GA, IA, IL, IN, KS, LA, MD, MI, MO, MS, NC, NE, NJ, NM, OH, OK, OR, PA, SC, SD, TN, TX, VA, WV
This species is known only from the original two specimens.
USA: MA
This species is known from western Arkansas (Polk and Hempstead Counties, CMNH, MCZ), Texas, as far south as the Rio Grande in the southeast (Mateu 1981: 125; Dajoz 2007: 18-19), southern Arizona (Pima County, MCZ), and central California (LeConte 1879c: 62). The record from “Oklahoma” (Arnold 2008) needs confirmation.
USA: AR, AZ, CA, TX [OK]
This species is known from “Louisiana” (USNM) and from Cameron (Schaeffer 1910: 396) and Zapata Counties (CMNH) in southern Texas.
USA: LA, TX
This species ranges from southwestern Arizona to southeastern Texas (Mateu 1981: 127). It was also reported from “Mexico” (Chaudoir 1863: 314; Liebke 1933: 471).
USA: AZ, NM, TX – Mexico
This species is known from a few scattered localities from southern California to southeastern Texas (Mateu 1981: 119).
USA: AZ, CA, TX
About 55 species in temperate, subtropical, and tropical areas of the Nearctic (12 species but only four endemic), Neotropical (about 40 species), and Australian (nine species in Australia) Regions.
Eighteen species (Lorenz 2005: 505, as Pseudaptinus s.str.) in the Nearctic (three species) and Neotropical (16 species) Regions.
Two of the North American species are included in Liebke’s (1934: 372-373) key to the species of this genus. A taxonomic revision is needed.
This Coastal Plain species ranges from southeastern Virginia (Davidson 1995: 18) to central Florida (Peck and Thomas 1998: 25), west to southeastern Texas (Cameron County, USNM).
USA: AL, AR, FL, GA, LA, MS, NC, SC, TX, VA
This species is known only from the type locality in southwestern California.
USA: CA
The range of this species extends from southern Washington (Zack et al. 2003) to southern California (Fall 1901a: 47), east along the southwest to western Arkansas (Polk County, Robert L. Davidson pers. comm. 2008) and southern Texas (Zapata County, CMNH; Liebke 1934: 373), north to central Kansas (Knaus 1905a: 218), south at least to Oaxaca (Bates 1883a: 166).
USA: AR, AZ, CA, KS, NM, OK, OR, TX, WA – Mexico
Thirty-four species (Lorenz 2005: 505) in the Nearctic (nine species but only two endemic), Neotropical (23 species), and Australian (nine species in Australia) Regions.
Messer (2011: 422-423) published a key to all species found in the United States and Mexico.
This taxon is considered as a distinct genus by some authors (e.g., Ball and Bousquet 2000: 115) but Baehr (1985: 36) found out that the character states between Thalpius and Pseudaptinus were “very few and rather weak.” I am following him and treat Thalpius as a subgenus of Pseudaptinus.
This species is found in central and southern Florida (Darlington 1935a: 161; Peck and Thomas 1998: 25), the Bahamas (Turnbow and Thomas 2008: 15), Cuba (Chaudoir 1877: 252; Peck 2005: 39), and the Dominican Republic (CMNH).
USA: FL – Bahamas, Cuba, Dominican Republic
This species is known from Hidalgo and Cameron Counties in southern Texas (Peter W. Messer pers. comm. 2011), from southern Florida (Bousquet and Larochelle 1993: 334), and from Cuba (Darlington 1934: 128), the Cayman Islands, and Dominican Republic (Messer 2011: 422).
USA: FL, TX – Cayman Islands, Cuba, Dominican Republic
The range of this species extends from the District of Columbia (Ulke 1902: 7) to western South Dakota (Kirk and Balsbaugh 1975: 39), south to southern Texas (Johnson 1978: 68) and southern Florida (Peck and Thomas 1998: 25); also recorded from the Bahamas (Turnbow and Thomas 2008: 15), Cuba (Darlington 1934: 128), Jamaica (Darlington 1941a: 14), Guana Island in the British Virgin Islands (Valentine and Ivie 2005: 275), Dominican Republic, and Mexico (Messer 2011: 423). The record from Arizona (Snow 1906b: 162) is probably in error.
USA: AL, AR, DC, FL, GA, KY, LA, MS, OH, OK, SC, SD, TN, TX, VA – Bahamas, British Virgin Islands, Cuba, Dominican Republic, Jamaica, Mexico
This species is known from Horn Island in southeastern Mississippi (Drew A. Hildebrandt pers. comm. 2010), “Texas” (Liebke 1934: 374), and Veracruz in Mexico (Bates 1883a: 166).
USA: MS, TX – Mexico
This species ranges from southeastern California (Fall 1901a: 47; Andrews et al. 1979: 28) and west-central Nevada (Storey County, David H. Kavanaugh pers. comm. 2008) east to southeastern Texas (Cameron County, CMNH, MCZ; LeConte 1880a: 85; Snow 1906a: 141), south to the states of Veracruz and Nayarit in Mexico (Peter W. Messer pers. comm. 2011); also recorded from “Oklahoma” (Messer 2011: 423).
USA: AZ, CA, NM, NV, OK, TX – Mexico
This species is known only from the type locality in southwestern California.
USA: CA
This species is known from Veracruz in Mexico and from two specimens collected in Live Oak County in southern Texas (Messer 2011: 420).
USA: TX – Mexico
This species ranges from southeastern Virginia (Davidson 1995: 18) to eastern Missouri (Anonymous 2007), south to southern Texas (Johnson 1978: 68) and central Florida (Peck and Thomas 1998: 25); also recorded from Cuba (Liebke 1934: 373; Peck 2005: 39).
USA: AL, AR, FL, GA, LA, MO, MS, NC, OK, SC, TN, TX, VA – Cuba
This species ranges from southeastern Oregon (LaBonte 1996: 357) to the Baja California Peninsula (Horn 1897: 367) and southern Arizona (Maricopa and Pima Counties, MCZ); its is also known from western Texas (El Paso and Brewster Counties, Robert L. Davidson pers. comm. 2012).
USA: AZ, CA, OR, TX – Mexico
Worldwide (though not represented in Europe), with about 130 species (Lorenz 2005: 507-509, as Planetina and Galeritini). The group is underrepresented in the Northern Hemisphere with only 17 species (about 12.5% of the world fauna). Two subtribes are recognized: Galeritina (about 105 species) and Planetina (27 species in the Eastern Hemisphere).
About 105 species in the temperate, subtropical, and tropical areas of the Nearctic (eight species of Galerita), Neotropical (64 species), Oriental (seven species of Galerita), Palaearctic (five species of Galerita), and Afrotropical (31 species) Regions. The species are placed in four genera: Ancystroglossus Chaudoir (six Neotropical species), Eunostus Laporte (14 Afrotropical species), Galerita (82 species), and Trichognathus Latreille (one Neotropical species).
Eighty-two species in the Western Hemisphere (58 species) and the Old World (24 species) currently arrayed in two subgenera, both represented in the Nearctic Region. Only eight species are found in North America and a single one (Galerita bicolor) is endemic.
Reichardt (1967) revised the Western Hemisphere species and provided keys for their identification.
Seven species in North America (seven species), Middle America (six species), and the West Indies (one species).
Subsequent to Reichardt’s (1967) revision, Ball and Nimmo (1983) published a synopsis of the species of Progaleritina leading to two new species-group taxa (Galerita reichardti and Galerita lecontei veracrucis) and subspecific status for two of Reichardt’s species (Galerita tenebricosa Klug and Galerita bicoloripes Reichardt).
This species is known from southeastern Louisiana (Saint Tammany Parish, Igor M. Sokolov pers. comm. 2009), Missouri, southeastern Nebraska (Nemaha County, Foster F. Purrington pers. comm. 2011), eastern Kansas (Popenoe 1877: 23; Horn 1872c: 385), eastern Texas, several mountain ranges in southern Arizona, and from the Sierra Huachinera of eastern Sonora and western Chihuahua in northern Mexico [see Ball and Nimmo 1983: Fig. 10]; it is also listed from “Oklahoma” by Arnold (2008). The record from eastern Iowa (Wickham 1911b: 7) needs confirmation.
USA: AZ, KS, LA, MO, NE, OK, TX [IA] – Mexico
The range of this eastern species extends from Rhode Island (Sikes and Webster 2005: 317) to southeastern South Dakota (Kirk and Balsbaugh 1975: 39), south to southeastern Texas and southern Florida [see Ball and Nimmo 1983: Fig. 14].
USA: AL, AR, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MD, MI, MN, MO, MS, NC, NJ, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, WI, WV
This species ranges from southern Arizona southwards along the Pacific Coast to Guerrero [see Ball and Nimmo 1983: Fig. 7].
USA: AZ – Mexico
This species is found from southern Quebec to southeastern South Dakota, north to southern Manitoba (Bousquet 1987a: 134), south to east-central Texas and central Florida; isolated populations are known from southwestern New Mexico (Grant County, Robert L. Davidson pers. comm. 2008), southeastern Arizona, and Chihuahua in Mexico [see Ball and Nimmo 1983: Fig. 10].
CAN: MB, ON, QC USA: AL, AR, AZ, CO, CT, DC, DE, FL, GA, IA, IL, IN, KS, KY, LA, MA, MD, MI, MN, MO, MS, NC, ND, NE, NH, NJ, NM, NY, OH, OK, PA, RI, SC, SD, TN, TX, VA, VT, WI, WV – Mexico
The range of this subspecies is disjunct: there is an eastern component ranging from Virginia to southwestern Kentucky (Trigg County, Foster F. Purrington pers. comm. 2011) southwards to southern Florida and South Bimini Island in the Bahamas, westwards to eastern Texas; the western population ranges from central California southeastwards to the state of Durango in Mexico [see Ball and Nimmo 1983: Fig. 17]. The record from east-central Missouri (Summers 1873: 133) needs confirmation.
USA: AL, AR, AZ, CA, FL, GA, KY, LA, MS, NC, NM, SC, TN, TX, VA [MO] – Bahamas, Mexico
The subspecies Galerita lecontei bicoloripes Reichardt is found in central Mexico, that of Galerita lecontei veracrucis Ball and Nimmo along the Gulf states of Mexico, and that of Galerita lecontei tenebricosa Klug in several islands of the Greater Antilles.
This species ranges from southeastern Nebraska (Lancaster County, Foster F. Purrington pers. comm. 2011) to south-central Arizona, south to Costa Rica [see Ball and Nimmo 1983: Fig. 12].
USA: AZ, KS, NE, NM, OK, TX – Costa Rica, El Salvador, Guatemala, Honduras, Mexico
This species is known from a few localities from eastern Texas southwards along the Gulf and Caribbean Coasts to Honduras [see Ball and Nimmo 1983: Fig. 7].
USA: TX – Honduras, Mexico
Seventy-five species in the Western Hemisphere (51 species) and the Old World (24 species). One Neotropical species extends into southern Texas.
This species ranges from the Rio Grande in southeastern Texas south to Colombia (Martínez 2003: 7) [see Reichardt 1967: Fig. 76]. The record from “Arkansas” (Bousquet and Larochelle 1993: 285) is in error.
USA: TX – Colombia, Mexico, Panama
Worldwide (though absent from Europe) with about 180 species (Lorenz 2005: 510-512) arrayed in 26 genera: five of these genera (about 60 species) are represented in the Western Hemisphere and 21 (about 120 species) in the Eastern Hemisphere. The group is underrepresented in the Northern Hemisphere with 14 species (about 8% of the world fauna), only five of them being endemic (about 3%). Two subtribes are recognized: Helluonina, with 40 species in the Australian Region, and Omphrina with about 140 species.
About 140 species in the Nearctic (seven species of Helluomorphoides), Neotropical (about 55 species), Australian (two species of Creagris Nietner, one of them endemic), Oriental (27 species), Palaearctic (seven Asian species but only two endemic, both in the Middle East), and Afrotropical (about 50 species) Regions.
Twenty-three species in temperate, subtropical, and tropical areas of the Nearctic (seven species) and Neotropical (20 species) Regions.
Ball (1956a) revised the North American species. The taxonomy and nomenclature of these species have not changed since.
This species is found along the Coastal Plain and Piedmont from southern New Jersey to southern Florida (Peck and Thomas 1998: 25), west to southeastern Mississippi (Ball 1956a: 85, Fig. 34). The record from south-central Kansas (Knaus 1901: 110) is probably in error.
USA: AL, FL, GA, MS, NC, NJ, SC
This species ranges from New Jersey (Smith 1890: 90; Smith 1910: 212) to northwestern South Carolina (Ciegler 2000: 130), west to the Baboquivari Mountains in southern Arizona, north to southwestern Utah (Washington County, CMNH) and northeastern Kansas, south to Sinaloa in Mexico (Ball 1956a: 78). The records from New York City and vicinity (Leng and Beutenmüller 1893: 143) and “Florida” (Leng 1920: 67) need confirmation.
USA: AL, AR, AZ, GA, KS, NC, NJ, NM, OK, SC, TX, UT, VA [FL, NY] – Mexico
This species is found from western Texas to south-central Arizona, south at least to Durango in Mexico (Ball 1956a: 84, Fig. 34). The record from “Oklahoma” (Arnold 2008) needs confirmation.
USA: AZ, NM, TX [OK] – Mexico
This species is found from Massachusetts to west-central Georgia (Fattig 1949: 41), west to eastern Texas (Ball 1956a: 75), and north to central Illinois (Purrington et al. 2002: 200) and western Michigan (Newaygo County, CMNH).
USA: AL, AR, DC, GA, IL, LA, MA, MD, MI, MS, NC, NJ, NY, OK, SC, TX, VA
This species ranges from the mountains of southern Arizona to southwestern Texas, south at least to Durango (Ball 1956a: 86, Fig. 34).
USA: AZ, TX – Mexico
This subspecies occurs from southeastern New Hampshire (Cooper 1976: 165) to southeastern Wyoming (Platte County, CMNH) and northern Colorado (Ball 1956a: Fig. 33), south to Oklahoma (French et al. 2001: 228; Grady County, CMNH), west-central Arkansas (Garland County, Robert L. Davidson pers. comm. 2012), and northeastern Georgia (Fattig 1949: 42). The record from southwestern Alabama (Löding 1945: 23) needs confirmation.
USA: AR, CO, CT, DC, DE, GA, IA, IL, IN, KS, MA, MD, MI, MN, MO, ND, NE, NH, NJ, NY, OH, OK, PA, SD, TN, VA, WI, WV, WY [AL]
Helluomorphoides praeustus bicolor (Harris). Adults of most carabids are opportunist predators or scavengers but those of Helluomorphoides seem to have adopted a different, more dangerous life-style. Adults of two North American species have been observed to plunder foraging and migrating columns of army ant species of the genus Neivamyrmex and run away with their prey and broods. The beetles do not inhabit the ant nest or bivouac but can detect and follow the chemical trails of the ants at night. Whether this habit is common to all species of Helluomorphoides remains to be investigated.
This subspecies is endemic to the Florida Peninsula (Peck and Thomas 1998: 25).
USA: FL
This subspecies is known for sure only from Mississippi (Harrison and Lafayette Counties, Drew A. Hildebrandt pers. comm. 2008), “New Jersey, ” and “Alabama” (Ball 1956a: 79) where Löding (1945: 23) recorded it from Colbert and Mobile Counties. The record from southwestern Ohio (Blatchley 1910: 155) is probably in error; those from Sand Point and Tampa in Florida (Leng 1915: 589) need confirmation.
USA: AL, NJ, MS [FL]
This species ranges from southwestern Kentucky (Trigg County, Foster F. Purrington pers. comm. 2010) and central Tennessee to southeastern New Mexico (Lea and Chaves Counties, CMNH), north to southwestern Iowa (Fremont County, Doug A. Veal pers. comm. 2009) and southeastern Nebraska (Jefferson County, Foster F. Purrington pers. comm. 2010), south to northeastern Mexico (Ball 1956a: 82-83, Fig. 34) and southwestern Georgia (Fattig 1949: 42). The records from southern Arizona (Snow 1907: 142) and Indiana (Blatchley 1910: 156) need confirmation.
USA: AL, AR, CO, GA, IA, KS, KY, MO, MS, NE, NM, OK, TN, TX [AZ, IN] – Mexico
About 325 species (Lorenz 2005: 388-390) in the Western Hemisphere (35 species) and the Australian (285 species, close to 90% of the world fauna), Oriental (four species of Cryptocephalomorpha), and Afrotropical (one species of Cryptocephalomorpha in South Africa) Regions. These species are arrayed in 12 genera: Adelotopus Hope (123 Australian species), Cainogenion Notman (11 Australian species), Cryptocephalomorpha Ritsema (seven species in the Eastern Hemisphere), Manumorpha Erwin and Geraci (one South American species), Notopseudomorpha Baehr (two species in South America), Paussotropus Waterhouse (one Australian species), Procainogenion Baehr (one Australian species), Pseudomorpha (30 species), Samiriamorpha Erwin and Geraci (one species in Peru), Sphallomorpha Westwood (145 Australian species), Tuxtlamorpha Erwin and Geraci (one species in Middle America), and Yasunimorpha Erwin and Geraci (one species from Ecuador).
Thirty species in temperate, subtropical, and tropical areas of the Western Hemisphere (30 species) and southern Australia (three species) arrayed in two subgenera: Pseudomorpha (27 species) and Austropseudomorpha Baehr (three Australian species). The North American fauna has 19 known species (57.5% of the world fauna).
According to Baehr (1994: Fig. 1), Pseudomorpha is the sister-group to a clade including Adelotopus Hope, Cainogenion Notman, Paussotropus Waterhouse, and Cryptocephalomorpha Ritsema.
Pseudomorpha sp. Members of Pseudomorpha are among the most unusual North American carabids. The body shape of the adults is very characteristic. In addition, contrary to other Nearctic carabids that lay eggs, they are ovoviviparous. The females carry hatched larvae in their bursa copulatrix, vagina, and lateral oviducts. Such behavior probably greatly reduces egg mortality.
Twenty-seven species in the Western Hemisphere.
Notman (1925) published a key to all species known at the time except Pseudomorpha ruficollis described the previous year. Since then, two new North American species have been described by Van Dyke (1943, 1953a). Species identification of the Nearctic fauna is almost impossible without recourse to type material. A thorough revision is needed.
This species is known only from the type locality in northern Arizona.
USA: AZ
This species is known only from the type locality in southern New Mexico.
USA: NM
This species is known from central and southern Arizona (Griffith 1900: 566; Notman 1925: 22) and southwestern New Mexico (Fall and Cockerell 1907: 162; Notman 1925: 22).
USA: AZ, NM
This species has been cited from Butte County in southeastern Idaho (Stafford et al. 1986: 289), Churchill County in western Nevada (Bechtel et al. 1983: 474), Contra Costa County (Notman 1925: 21) and the San Joaquin Valley (Fall 1901a: 51) in California, and the state of Durango (Bates 1883a: 255) in Mexico.
USA: CA, ID, NV – Mexico
This species is known from southeastern Oregon (Hatch 1953: 187), Sacramento County in California, and northwestern Utah (Notman 1925: 22).
USA: CA, OR, UT
This species is known from southeastern Arizona (Notman 1925: 20). The record from “California” (Csiki 1933a: 1638) needs confirmation.
USA: AZ [CA]
This species is known from San Diego County in southwestern California and Yavapai County in central Arizona (Notman 1925: 19).
USA: AZ, CA
This species is known from Inyo (Horn 1867b: 151) and Tulare (Notman 1925: 21) Counties in California.
USA: CA
This species is known only from the type locality in westernmost Texas.
USA: TX
This Coastal Plain species is found from northern South Carolina (Ciegler 2000: 130) to Louisiana (Notman 1925: 22; Erwin and Geraci 2008: 85).
USA: AL, GA, LA, MS, SC
This species is known only from the holotype collected in southwestern California.
USA: CA
This species is known from Pima, Pinal (Notman 1925: 15), and Cochise (MCZ) Counties in southern Arizona.
USA: AZ
This species is known from southern California (Van Dyke 1943: 30; Andrews et al. 1979: 28).
USA: CA
This species is known only from the holotype collected in southern Arizona.
USA: AZ
This species is known only from the holotype collected in southern Arizona.
USA: AZ
This species is known only from the holotype collected in southern Arizona.
USA: AZ
This species has been reported from San Diego County in southwestern California and Lyon County in west-central Nevada (Notman 1925: 18).
USA: CA, NV
This species is known only from the holotype collected in northwestern Utah.
USA: UT
Several persons have contributed to this catalogue. Thomas C. Barr, Jr., Ross T. and Joyce Bell, James C. Bergdahl, Eric van den Berghe, Sydney G. Cannings, Donald S. Chandler, Robert L. Davidson, Garry A. Dunn, Kamal J.K. Gandhi, Vince Golia, Gerald J. Hilchie, Drew A. Hildebrandt, Richard L. Hoffman, Ronald R. Hooper, Ken Karns, David H. Kavanaugh, William L. Krinsky, James R. LaBonte, Paul K. Lago, David W. Langor, Serge Laplante, André Larochelle, Kirk J. Larsen, Harry J. Lee, Jr., David R. Maddison, Christopher G. Majka, Peter W. Messer, Steve Nanz, Robert E. Nelson, Darren A. Pollock, Foster F. Purrington, Brian Raber, Edward G. Riley, Donald P. Schwert, Derek S. Sikes, Igor M. Sokolov, Doug A. Veal, and Reginald P. Webster provided many new state or province records. George E. Ball, Robert L. Davidson, Serge Laplante, David R. Maddison, and Gerald R. Noonan granted permission to include unpublished nomenclatural findings (new synonymies) from their research. Philip D. Perkins and Hans Silfverberg provided information on T.W. Harris’ and Mannerheim’s collections respectively, Robert E. Acciavatti, George E. Ball, Shawn M. Clark, Lee H. Herman, Fritz Hieke, Stuart Hine, and Bernd Jaeger information on type specimens, and Shun-Ichi Uéno information about K. Shirahata. Nadine Dupérré provided label data of a holotype housed at AMNH and information from books and journals available at the AMNH. Paul Skelley and Boris Kataev sent photocopies of pages of books housed in the Division of Plant Industry Library (FSCA) and the Zoological Museum of Moscow University Library respectively. Warren E. Steiner, Jr. and Terry L. Erwin, Robert L. Davidson, and Philip D. Perkins assisted by arranging visits I made in 2008 to collections at the USNM, CMNH, and MCZ respectively. George E. Ball reviewed the manuscript section on the genus Selenophorus. Vasily V. Grebennikov and Aleš Smetana translated texts in Russian and German respectively. Anthony Davies, Patrice Bouchard, and Serge Laplante reviewed sections of the manuscript. I extend my sincere appreciation to all these individuals.
I wish to thank also Steve Gamman and Patricia Madaire of the Canadian Agricultural Library (Entomology) in Ottawa, Eileen Mathias of The Ewell Sale Stewart Library at The Academy of Natural Sciences in Philadelphia, Dana Fischer of the Ernst Mayr Library at the Museum of Comparative Zoology in Cambridge, Massachusetts, and Marty Schlabach at the Comstock Memorial Library of Entomology at Cornell University in Ithaca, New York, for their assistance in locating obscure and difficult-to-find works. I am indebted to my colleague Aleš Smetana for access to his impressive personal library.
Finally, I acknowledge Henri Goulet for the permission to use some of his nice color illustrations.
Nebria abstracta Scudder, 1900b: 70. Type locality: «Reservoir Park, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. According to Kavanaugh (1978: 276), the holotype probably belongs to the genus Agonum.
Nebria occlusa Scudder, 1900a: 17. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1755]. Note. 1. Although noted originally as from the Miocene period, most authors agree that the shales of the Florissant Basin are from the Early Oligocene (Wilson 1978: 17). 2. According to Kavanaugh (1978: 275), the holotype probably belongs to the genus Amara.
Nebria paleomelas Scudder, 1879: 178B. Type locality: «Nicola River, below main coal seam [British Columbia]» (original citation), from the Miocene period. Holotype [by monotypy] location unknown. Note. According to Kavanaugh (1978: 275), the holotype probably belongs to the genus Amara.
CychriniCychrus minor Horn, 1876b: 243. Type locality: «Port Kennedy, Penn[sylvani]a» (original citation), from the Pleistocene period. Holotype location unknown. Note. According to Horn (1876b: 243), this species is “almost exactly that of andrewsii” but Roeschke (1907a: 270) believed it belongs to the elevatus group rather than to be related to Scaphinotus andrewsii.
Cychrus wheatleyi Horn, 1876b: 242. Type locality: «Port Kennedy, Penn[sylvani]a» (original citation), from the Pleistocene period. Holotype location unknown. Etymology. The specific name was proposed for Charles M. Wheatley [1822-1882], mine operator, mineralogist, palaeontologist, naturalist, conchologist, and copper metallurgist. Born in England, Wheatley emigrated to the United States with his parents as a young boy. Note. Horn (1876b: 242) believed this species is related to Scaphinotus viduus.
Nomaretus serus Scudder, 1900a: 13. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1762]. Note. Roeschke (1907a: 271) stated that this species is not a member of the tribe Cychrini.
CarabiniCalosoma calvini Wickham, 1909: 126. Type locality: «Florissant [Teller County, Colorado]» (original citation), from the early Oligocene period. Holotype in PMNH [# 4]. Note. According to Jeannel (1940a: 34), the only difference between this species and Calosoma emmonsii is the slightly longer size of the elytra and the specimen could be conspecific with those of Calosoma scrutator.
Calosoma cockerelli Wickham, 1910: 47. Type locality: Florissant [Teller County], Colorado (inferred from title of the book), from the early Oligocene period. Holotype in PMNH [# 10]. Note. Jeannel (1940a: 34) had no opinion on the specimen.
Calosoma emmonsii Scudder, 1900a: 16. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1756, 1757]. Etymology. The specific name honors the American geologist, educator, and physician Ebenezer Emmons [1799-1863] who is credited for having named the Adirondack Mountains in 1838 and the Taconic Mountains in 1844. Note. Lapouge (1930: 159) stated that this species does not differ from Calosoma externum and Jeannel (1940a: 34) believed it could be identical to Calosoma scrutator.
Calosoma fernquisti Cockerell, 1924: 14. Type locality: «Deep Creek Canon, near Spokane, Washington» (original citation), from the Upper Eocene period. Holotype in USNM [# 69218]. Note. 1. Jeannel (1940a: 34) believed the species is very likely Calosoma externum. 2. According to Nel (1989: 265), fossil specimens described as Calosoma by Wickham, Scudder, and Cockerell could all be conspecific.
Carabus jeffersoni Scudder, 1900a: 15. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Two syntypes [2 specimens originally cited] in MCZ [# 1758, 1759]. Etymology. The specific name honors Thomas Jefferson [1743-1826], third president of the United States and one of the earliest writers on American palaeontology. Jefferson is also famous for writing the Declaration of Independence in 1776. Note. According to Lapouge (1930: 159), this species does not belong to the subfamily Carabinae.
Carabus maeander sangamon Wickham, 1917b: 139. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Holotype [by monotypy] in UICU.
Neothanes testeus (Scudder, 1878: 758) (as Cychrus testeus). Type locality: «immediate vicinity of Green River Station on the Union Pacific Railroad in Wyoming» (original citation), from the Eocene period. Holotype [by monotypy] location unknown. Note. The genus Neothanes was proposed by Scudder (1890: 535) for Cychrus testeus Scudder, 1878 and, according to him, is “allied to Carabus.”
CicindeliniCicindelopsis eophilus Cockerell, 1920: 254. Type locality: «White River, Colorado» (original citation), from the “Eocene rock of Green River Age.” Holotype in USNM [# 66576]. Note. 1. According to Wilson (1978: 14), the “White River” collections made by Scudder, some of which were described by Cockerell, came from the Green River Formation localities along the White River near the Colorado-Utah border. 2. According to Cockerell (1920: 254), the single elytron representing this species “suggests a cicindelid” but is narrower, without any distinct inner apical angle, and the surface is neither punctured nor striate. He also stated that “the Collyrinae have narrow elytra shaped essentially as in Cicindelopsis.” The species was subsequently transferred to the Melandryidae (Cockerell 1924: 12) based on Herbert S. Barber’s opinion. The holotype was also studied by Walther Horn who believed it did not belong to the Cicindelinae (Cockerell 1924: 12).
ElaphriniDiacheila matthewsi Böcher, 1995: 21. Type locality: «Peary Land, Greenland» (original citation), from the Plio-Pleistocene period (2-2.5 million years B.P.). Holotype in ZMUC. Note. Böcher (1995: 21, 22) noted that fossil remnants of Diacheila reported from Alaska (about 2.9 million B.P.), Yukon Territory, Banks, Prince Patrick and Meighen Islands (Pliocene), and Ellesmere Island probably belong to this species.
Elaphrus clairvillei lynni Pierce, 1948a: 52. Type locality: «Lynn Creek, British Columbia» (original citation), from the Pleistocene period. Holotype in LACM. Note. Goulet (1983: 271) listed this name in synonymy with Elaphrus clairvillei Kirby, 1837.
Elaphrus irregularis Scudder, 1890: 534. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] in MCZ. Note. Goulet (1983: 321) concluded from a study of the fossil elytron that several character states present in the holotype suggest “a match with Elaphrus parviceps [= Elaphrus trossulus Semenov].”
Elaphrus ruscarius foveatus Pierce, 1948b: 54. Type locality: «McKittrick [Kern County, California]» (original citation), from the Pleistocene period. Holotype in LACM. Note. Goulet (1983: 303) listed this name in synonymy with Elaphrus finitimus Casey, 1920.
LoriceriniLoricera exita Scudder, 1900b: 70. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. Ball and Erwin (1969: 894) stated that the single fragment of an elytron representing this species probably belongs “to a carabid, possibly an agonine.”
Loricera glacialis Scudder, 1877: 763. Type locality: Scarborough, Ontario (inferred from title of the paper), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. Ball and Erwin (1969: 894) indicated that the elytra of two specimens assigned to this species “are probably those of a weevil.”
Loricera ? lutosa Scudder, 1890: 533. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. Ball and Erwin (1969: 894) stated that the single elytron representing this species has characteristics “of a brachyrhinine weevil, not of a carabid, and certainly not of Loricera.” Morgan and Morgan (1980: 23) stated that the species is “definitely a member of the Scarabaeidae, almost certainly of the genus Aegialia and probably Aegialia lacustris LeC.”
TrechiniTrechus fractus Wickham, 1912: 6. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in UCM [# 8272]. Note. Based on the illustration provided, this species, if really of a carabid, could belong to any groups and not necessary to the genus Trechus.
BembidiiniBembidium damnosum Scudder, 1900b: 73. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Bembidion davidae Pierce, 1944: 7. Type locality: «Cuyama River outcrop section on the west bank of the river, near its junction with Huasna Creek, in San Luis Obispo County near center of section 27, Township 11 N, R. 33 W. San Bernardino Base Line and Meridian [California]» (original citation), from the “Upper Lower Miocene.” Holotype in LACM. Etymology. The specific name was proposed for the American palaeontologist Lore Rose David [1905-1985]. Note. This species is represented by a single elytron and was assigned to the subgenus Ochthedromus by Pierce (1944: 7).
Bembidion everestae Pierce, 1944: 11. Type locality: «Bellevue District, near town of Rosedale, 6 miles due west of Bakersfield, Kern Co[unty], California» (original citation), from the “Upper Lower Miocene, Saucesian Age.” Holotype in LACM. Note. This species, represented by a fragment of an elytron and its impression in oil shale core, was tentatively assigned to the subgenus Odontium LeConte by Pierce (1944: 12).
Bembidium exoletum Scudder, 1876: 77. Type locality: «Green River group, near White River, in one of the two localities called ... Chagrin Valley and Fossil Cañon» (original citation), from the Eocene period. Holotype [by monotypy] location unknown.
Bembidium expletum Scudder, 1900b: 72. Type locality: «Reservoir Park, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Bembidium florissantensis Wickham, 1913: 6. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 90451].
Bembidion fragmentum Scudder, 1890: 531. Type locality: «shores of Lake Erie, near Cleveland, Ohio» (Scudder 1895: 54), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. The type locality was originally cited as “Scarboro[ugh], Ontario” but corrected to the present one subsequently. Scudder (1890: 531) indicated that this species “is represented by a single elytron with the tip broken off” and agrees “better among modern types with Bembidion constrictum.”
Bembidion glaciatum Scudder, 1890: 531. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [2 originally cited] location unknown. Note. According to Scudder (1890: 531), this species is represented by a couple of elytra and is “nearly allied to the scarcely smaller Bembidion longulum LeC. [= Bembidion concolor (Kirby)].”
Bembidium haywardi Scudder, 1900b: 70. Type locality: «Logan’s brickyard, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Bembidium obductum Scudder, 1900a: 18. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype in MCZ [# 1768].
Bembidium praeteritum Scudder, 1900b: 72. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Bembidium tumulorum Scudder, 1900a: 19. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype in USNM.
Bembidium vanum Scudder, 1900b: 71. Type locality: «Scarborough; Reservoir Park, Toronto [Ontario]» (original citation), from the Pleistocene period. Syntypes [2 originally cited] location unknown.
Bembidium vestigium Scudder, 1900b: 71. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Tachys haywardi Wickham, 1913: 7. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 90582B].
PatrobiniPatrobus decessus Scudder, 1900b: 73. Type locality: «Reservoir Park, Toronto; Scarborough [Ontario]» (original citation), from the Pleistocene period. Lectotype [as type], designated by Darlington (1938: 155), in MCZ. Note. Darlington (1938: 155) listed this name in synonymy with Patrobus stygicus Chaudoir, 1871.
Patrobus frigidus Scudder, 1900b: 74. Type locality: «Reservoir Park, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] in MCZ. Note. Darlington (1938: 155) listed this name in synonymy with Patrobus stygicus Chaudoir, 1871.
Patrobus gelatus Scudder, 1890: 530. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] in MCZ. Note. Darlington (1938: 155) listed this name in synonymy with Patrobus stygicus Chaudoir, 1871.
Patrobus henshawi Wickham, 1917b: 140. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Holotype [by monotypy] in UICU.
BrachininiBrachinus newberryi Scudder, 1900a: 32 (as Brachynus newberryi). Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Four syntypes [4 originally cited] in MCZ [# 1850-1853]. Etymology. The specific name was proposed in honor of the American geologist, palaeontologist, physician, explorer, and author John Strong Newberry [1822-1892]. Note. Erwin (1970a: 164) noted that the specimens assigned to this species “should be placed in the genus Lebia.”
Brachinus repressus Scudder, 1900a: 33 (as Brachynus repressus). Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype in MCZ [# 1848]. Note. According to Erwin (1970a: 164), the single specimen of this species “is too fragmentary to make definitive comparisons, but is similar in some respects to members of Cymindis.”
PaussiniPaussopsis nearctica Cockerell, 1911: 71. Type locality: «Florissant [Teller County, Colorado]» (original citation), from the early Oligocene period. Holotype in UCM [# 4508]. Note. The genus Paussopsis Cockerell, 1911 was proposed for this species.
Paussopsis secunda Wickham, 1912: 10. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype in UCM [# 8200]. Note. According to Wasmann (1926: 25), members of Paussopsis do not belong to paussines.
PterostichiniCyclotrachelus tenebricus (Scudder, 1900a: 24) (as Evarthrus tenebricus). Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1786]. Note. This species is represented only by a head. Based on the original description, Freitag (1969: 168) stated that the specimen “may or may not be a member of the genus Evarthrus [= Cyclotrachelus].”
Loxandrus gelidus Scudder, 1877: 763. Type locality: Scarborough, Ontario (inferred from title of the paper), from the Pleistocene period. Syntype(s) [5 originally cited] location unknown. Note. According to Scudder (1890: 528), this species closely resembles Pterostichus hudsonicus LeConte.
Myas rigefactus Scudder, 1900a: 20. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1766].
Myas umbrarum Scudder, 1900a: 21. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [3 originally cited] in MCZ [# 1763, 1764, 1767].
Pterostichus abrogatus Scudder, 1890: 525. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. According to Scudder (1890: 525), this species is represented by a “fragment of an elytron” and its features suggest “a species closely allied to Pterostichus herculaneus Mann.”
Pterostichus depletus Scudder, 1900b: 75. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Pterostichus destitutus Scudder, 1890: 526. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. According to Scudder (1890: 526), this species is “represented by a single elytron of a mahogany color, which seems to be nearly related to Pterostichus sayi Brullé [= Poecilus chalcites (Say)].”
Pterostichus destructus Scudder, 1890: 527. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [2 originally cited] location unknown. Note. According to Scudder (1890: 527), this species, represented by a couple of elytra, “closely resembles Pterostichus patruelis Dej.”
Pterostichus dormitans Scudder, 1890: 526. Type locality: «shores of Lake Erie, near Cleveland, Ohio» (Scudder 1895: 51), from the Pleistocene period. Syntype(s) [2 originally cited] location unknown. Note. The type locality was originally listed as “Scarboro[ugh], Ontario” but corrected to the present one subsequently. According to Scudder (1890: 526), this species, “represented by two opposite ends of elytra, is very close indeed in size and general character to Pterostichus laetulus LeC. [= Poecilus laetulus (LeConte)].”
Pterostichus fernquisti Wickham, 1931: 318. Type locality: «Deep Creek Canyon [near Spokane, Washington]» (original citation), from the Miocene period. Holotype in MCZ. Note. According to Wickham (1931: 318), this species, represented by three elytra, is perhaps “most reminiscent of Pterostichus gracilior.”
Pterostichus fractus Scudder, 1890: 527. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. This species is represented by a single elytron with the extreme apex broken. According to Scudder (1890: 527), the species is “closely allied to Pterostichus destitutus.”
Pterostichs laevigatus Scudder, 1890: 528. Type locality: «Bone caves of Port Kennedy, Pennsylvania» (original citation), from the Pleistocene period. Syntype(s) location unknown.
Pterostichus pumpellyi Scudder, 1900a: 23. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Two syntypes [2 originally cited] in MCZ [# 1775, 1776]. Etymology. The specific name was proposed for the American geologist and explorer Raphael Pumpelly [1837-1923].
Pterostichus walcotti Scudder, 1900a: 23. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [6 originally cited] in MCZ [# 1769-1772, 1774] and USNM. Etymology. The specific name honors the American geologist and palaeontologist Charles Doolittle Walcott [1850-1927] who for some time was director of the United States Geological Survey.
ZabriniAmara cockerelli Wickham, 1912: 7. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in UCM [# 8236].
Amara danae Scudder, 1900a: 27. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [15 originally cited] in MCZ [# 1787, 1788, 1801, 1802, 1804-1806, 1808, 1809, 1811, 1812, 4167] and USNM. Etymology. The specific name honors the American geologist, mineralogist, and zoologist James Dwight Dana [1813-1895].
Amara powellii Scudder, 1900a: 26. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [12 originally cited] in MCZ [# 1777, 1790-1794, 1796-1800] and USNM [# 59646]. Etymology. This species was named after the American geologist and explorer Major John Wesley Powell [1834-1902] who led a three-month expedition in 1867 down the Green and Colorado rivers that included the first passage through the Grand Canyon. Powell served as the second director of the United States Geological Survey from 1881 to 1894.
Amara revocata Scudder, 1900a: 25. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1785].
Amara sterilis Scudder, 1900a: 25. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [4 originally cited] in MCZ [# 1782-1784] and USNM.
Amara veterata Scudder, 1900a: 26. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [5 originally cited] in MCZ [# 1778-1781, 1813].
ChlaeniiniChlaenius plicatipennis Wickham, 1917b: 143. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Holotype [by monotypy] in UICU.
Chlaenius punctulatus Horn, 1876b: 244 [homonym of Chlaenius punctulatus Chaudoir, 1876]. Type locality: «Port Kennedy, Penn[sylvani]a» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. This species is represented by one elytron “of the size and very similar to that of Chlaenius laticollis” (Horn 1876b: 244).
LicininiBadister antecursor Scudder, 1900b: 75. Type locality: «Logan’s brickyard, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Dicaelus alutaceus Horn, 1876b: 244. Type locality: «Port Kennedy, Penn[sylvani]a» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Diplocheila ? henshawi Scudder, 1890: 523. Type locality: «Florissant [Teller County, Colorado]» (original citation), from the early Oligocene period. Holotype [by monotypy] location unknown.
HarpaliniCratacanthus florissantensis Wickham, 1917a: 464. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 63442]. Note. Based on the illustration provided by Wickham (1917a: plate 37, figure 2), the specimen is not necessarily a Cratacanthus, not even a carabid.
Euryderus kingii (Scudder, 1900a: 34) (as Nothopus kingii). Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1847]. Etymology. The species was named for the American geologist and mountaineer Clarence King [1842-1901] who served as first director of the United States Geological Survey from 1879 to 1881.
Harpalus conditus Scudder, 1900b: 77. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Harpalus maceratus Wickham, 1911a: 54. Type locality: «Florissant [Teller County, Colorado]» (original citation), from the early Oligocene period. Holotype in UCM [# 8246].
Harpalus nuperus Scudder, 1900a: 34. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype in MCZ [# 1846]
Harpalus redivivus Wickham, 1917a: 465. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 63444].
Harpalus ulomaeformis Wickham, 1917a: 464. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 63443].
Harpalus veterum Cockerell, 1920: 256. Type locality: «Roan Mountain, Colorado» (original citation), from the Eocene period. Holotype in USNM [# 66578].
Harpalus whitfieldi Scudder, 1900a: 35. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. One syntype [3 originally cited] in MCZ [# 1845] and two in USNM. Etymology. The species was named after the American palaeontologist, teacher, and curator Robert Parr Whitfield [1828-1910].
Stenolophus religatus Scudder, 1900a: 35. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1858].
PlatyniniPlatynus caesus Scudder, 1890: 522. Type locality: «Green River, Wyoming» (original citation), from the Eocene period. Syntype(s) [2 originally cited] location unknown.
Platynus calvini Wickham, 1917b: 142. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Syntypes [2 originally cited] in UICU. Etymology. The specific name was proposed for Samuel Calvin [1840-1911], professor of geology at the State University of Iowa.
Platynus casus Scudder, 1890: 519. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. This species is represented by a single elytron. According to Scudder (1890: 519), it is best comparable to Platynus rubripes Zimmermann (= Agonum rufipes Dejean) among extant species.
Platynus desuetus Scudder, 1890: 521. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [6 originally cited] location unknown. Note. According to Scudder (1890: 521), this species “is more nearly allied to Platynus crenistriatus LeC. than to any other living species.”
Platynus dilapidatus Scudder, 1895: 49. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown. Note. According to Scudder (1895: 49), this species, represented by a fragment of a single elytron, “belongs rather in the near vicinity of Platynus maculicollis Dej. [= Tanystoma maculicolle (Dejean)].”
Platynus dissipatus Scudder, 1890: 521. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [2 originally cited] location unknown. Note. According to Scudder (1890: 521), this species agrees with Platynus halli “in its general features and in the minute texture of the surface.”
Platynus exterminatus Scudder, 1900b: 75. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Platynus florissantensis Wickham, 1913: 7. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 63441]. Note. Wickham (1913: 7) noted that this species “compared with recent species ... remind one of Platynus placidus by the fine striae and of Platynus melanarius by the size and form.”
Platynus halli Scudder, 1890: 520. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [3 originally cited] location unknown. Etymology. The specific name honors the American geologist and palaeontologist James Hall [1811-1898]. Note. According to Scudder (1890: 520), this species is “allied to Platynus crenistriatum LeC. ... [but] more nearly related to Platynus hindei.”
Platynus harttii Scudder, 1890: 522. Type locality: «Scarboro[ugh], Ontario» (original citation), from the Pleistocene period. Syntype(s) [2 originally cited] location unknown. Etymology. The specific name was proposed in honor of the American geologist and teacher Charles Frederic Hartt [1840-1878] who led, in his last years, the Brazilian Geological Commission. Note. According to Scudder (1890: 522), this small species “is most nearly allied to the largest [= Platynus desuetus Scudder].”
Platynus hindei Scudder, 1890: 520. Type locality: «Scarboro[ugh], Ontario» (original citation), from the “clay beds of interglacial deposits” [Pleistocene period]. Syntype(s) [9 originally cited] location unknown. Etymology. This species was named for the geologist and palaeontologist George Jennings Hinde [1839-1918]. Note. The species is represented by “a number of fragments” and, according to Scudder (1890: 520), “seems to be allied to Platynus rubripes Zimm. [= Agonum rufipes Dejean].”
Platynus interglacialis Scudder, 1900b: 76. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Platynus interitus Scudder, 1900b: 76. Type locality: «Reservoir Park, Toronto [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Platynus insculptipennis Wickham, 1917a: 463. Type locality: Florissant [Teller County], Colorado (inferred from title of the paper), from the early Oligocene period. Holotype in USNM [# 63441].
Platynus longaevus Scudder, 1900b: 77. Type locality: «Scarborough [Ontario]» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Platynus pleistocenicus Wickham, 1917b: 141. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Holotype [by monotypy] in UICU.
Platynus senex Scudder, 1878: 759. Type locality: «immediate vicinity of Green River Station on the Union Pacific Railroad in Wyoming» (original citation), from Eocene period. Holotype [by monotypy] location unknown.
Platynus subgelidus Wickham, 1917b: 142. Type locality: «north bank of the Sangamon River near Mahomet, Champaign County, Illinois» (original citation), from the Pleistocene period. Holotype [by monotypy] in UICU.
Platynus tartareus Scudder, 1900a: 30. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Syntype(s) [4 originally cited] in MCZ [# 1838, 1840, 1841, 1857].
LebiiniCymindis aurora Horn, 1876b: 243. Type locality: «Port Kennedy, Penn[sylvani]a» (original citation), from the Pleistocene period. Syntype(s) location unknown. Note. According to Horn (1876b: 243), the species “is closely related to Cymindis americana, but somewhat larger.”
Cymindis extorpescens Scudder, 1898: 743. Type locality: «Fort River in Hadley, Massachusetts, near its entrance into the Connecticut» (original citation), from the Pleistocene period. Holotype [by monotypy] location unknown.
Lebia protospiloptera Cockerell, 1921: 35. Type locality: «back of house at Smith’s ranch, shale of Green River formation, Colorado» (original citation), from the Eocene period. Holotype in USNM [# 66928].
Plochionus lesquereuxi Scudder, 1900a: 31. Type locality: «Florissant [Teller County], Colorado» (original citation), from the early Oligocene period. Holotype [by monotypy] in MCZ [# 1855]. Etymology. The species was named after the bryologist and paleobotanist Leo Lesquereux [1806-1889]. Born in Switzerland, Lesquereux progressively lost his hearing, consequent to a near-fatal fall from the top of a cliff at the age of seven, which had considerable ramifications in his professional life. He moved with his wife and five children to the United States in 1847. Note. According to Scudder (1900a: 31), this species could rather belong to the genus Cymindis of the subgenus Pinacodera.
GaleritiniGalerita marshii Scudder, 1900a: 31. Type locality: «Green River, Wyoming» (original citation), from the Eocene period. Holotype [by monotypy] location unknown. Etymology. The specific name was proposed for the American palaeontologist Othniel Charles Marsh [1831-1899] who uncovered the first pterosaur fossil in America. Marsh is also known for his heated rivalry with Edward Drinker Cope [1840-1897] which led the two palaeontologists to use underhanded methods to out-compete the other in the field, resorting to bribery, theft, and destruction of bones (the Bone Wars). They attack each other in scientific publications, attempting to ruin the other’s credibility. Marsh is credited to have persuaded his uncle, George Peabody [1795-1869], to establish the Peabody Museum of Natural History at Yale.
Incertae SedisCarabites (?) arapahoensis Cockerell, 1918: 307. Type locality: «one mile west of Spicer, Arapahoe Pass Road, North Park, Colorado, 2½ miles south of fork of road» (original citation), from the Eocene period. Holotype [by monotypy] in UCM [# 5822].
Carabites eocenicus Cockerell, 1920: 255. Type locality: «White River, Colorado» (original citation), from the Eocene period. Holotype in USNM [# 66577].
Carabites exanimus Scudder, 1892: 17. Type locality: «White River, Utah, about 5 miles from the Colorado line» (original citation), from the Eocene period. Holotype [by monotypy] location unknown. Note. Based on Scudder’s description, this species, which is represented only by the elytra, is that of a large species with the elytral epipleura crossed. Scudder (1892: 17) stated that the species “seems to agree best with the Pterostichini.”
Carabites feildenianus Heer, 1878: 38. Type locality: Grinnell-Landes [= central section of Ellesmere Island, Nunavut] (inferred from title of the paper), from the Miocene period. Syntype(s) location unknown. Etymology. The specific name was proposed for Henry Wemyss Feilden [1838-1921], naturalist of the British north-polar expedition of Captain George Nares in 1875-76.
Carabites kincaidi Cockerell, 1908: 51. Type locality: «Green River, Wyoming» (original citation), from the Eocene period. Holotype [by monotypy] in PMNH. Note. This species was described from a larva. Cockerell (1908: 52) noted that the larva may be that of “the genus Neothanes Scudder, described from the Green River beds; but as this cannot be demonstrated, I leave it in the blanket genus Carabites.”
Carabites russelli Cockerell, 1928: 37. Type locality: Fox Hills, South Dakota (inferred from title of the paper), from the Cretaceous period. Holotype probably in PMNH.
Along with the reference, the following information is provided: pages where the character states of the species are indicated, the figures illustrating character states, the stage described, and the provenance of the material, either reared (RR) or field collected (FC). A question mark in front of a reference indicates that the identification of the taxon is uncertain according to the author(s). A dagger after the name of a species indicates that the taxon is adventive in North America. The abbreviations used are:
FC field collected
LIT Literature
L1 first instar
L2 second instar
L3 third instar
L1-3 instar not determined
L2-3 second and/or third instar
RR reared
TrachypachiniTrachypachus gibbsii LeConte
Trachypachus inermis Motschulsky: Bousquet and Goulet (1984: Figs 25, 37, 42, 51, as Trachypachus holmbergi); Beutel (1993: Figs 2, 8, 13, 16, 21, 29, as Trachypachus holmbergi); Arndt and Beutel (1995: 442; Figs 4, 6, 12, as Trachypachus holmbergi) [L1; RR].
ClinidiiniClinidium (Arctoclinidium) sculptile (Newman): Böving and Craighead (1931: Pl. 3); Hůrka (1978: Fig. H-10); Thompson (1979a: Figs 10a-c).
PelophiliniPelophila borealis (Paykull): Johnson and Carpenter (1898: 136-139; Figs 1-12) [L3; RR]; Sharova (1958: 37; Figs 31, 81, 82) [L1-3; ?]; Sharova (1964: 143, 144; Figs 99-4, 128, 129-1) [L1-3; ?]; Haberman (1968: Fig. 57-1); Andersen (1970: 92, 93; Figs 2a-i, 3a-c) [L1-3; FC]; Luff (1972: 176, 177; Figs 51-62) [L1-3; RR+FC]; Thompson (1979a: Figs 34a-c); Luff (1993: 58, 59; Figs 160-163) [L1, L2-3; ?]; Makarov (1994: Fig. 84); Bousquet (2010a: Figs 373-376).
OpisthiiniOpisthius richardsoni Kirby: Garner (1954: 160, 161; Figs a-c [Pl. 9]) [L1, L2-3; RR+FC]; Lindroth (1960a: 38, 40, 41; Figs 3, 6a-g) [L1, L2, L3; FC]; Thompson (1979a: Figs 37a-c); Bousquet and Smetana (1991: 110; Figs 3, 9a) [L1, L2, L3; RR].
NebriiniLeistus (Leistus) ferrugineus (Linnaeus)†: Larsson (1941: 280) [L1-3; ?]; Verhoeff (1921: Fig. 22); Larsson (1968: 324) [L1-3; ?]; Luff (1972: 162, 165; Fig. 12) [L1-3; FC]; Arndt (1991b: 72) [L1-3; LIT]; Luff (1993: 48; Figs 122, 123) [L1-3; ?]; Bousquet (2010a: Figs 378, 379).
Nebria (Nebria) brevicollis (Fabricius)†: Blisson (1848: 73-76; Figs 1-6 [Pl. 1]) [L2-3; FC]; Schiødte (1867: 461-465; Figs 1-7 [Pl. 14]) [L1-3; ?]; Znojko (1929: Fig. 4b); Raynaud (1935: 12, 13, 16) [L3?; FC]; Larsson (1941: 278; Figs 5, 6, 17a, 18a) [L1-3; ?]; Jeannel (1941b: Figs 74a-h); Sharova (1958: 38; Figs 6, 18, 85a) [L1-3; ?]; Habu (1958: Figs 12, 14); Sharova (1964: 145; Figs 88, 93-2, 130-1) [L1-3; ?]; Larsson (1968: 321, 322; Figs 5, 6, 17a, 18a) [L1-3; ?]; Haberman (1968: Fis 57-2, 57-4, 58-2); Andersen (1970: 91) [L3; LIT]; Luff (1972: 171, 173; Figs 32-41) [L1, L2-3; FC]; Arndt (1991b: 71, 72; Figs 2, 62) [L1, L2-3; ?]; Luff (1993: 49, 54, 57; Figs 2, 3, 7, 8, 143-155) [L1, L2-3; ?]; Sustek (1993: 522) [L3; LIT].
Nebria (Boreonebria) gyllenhali Schönherr: Larsson and Gígja (1959: 15, as Nebria gyllenhali munsteri) [L1-3; FC]; Andersen (1970: 91-93; Figs 4a-i, 5a-c) [L1-3; FC]; Luff (1972: 171, 174, 176, as Nebria rufescens; Figs 45-49) [L1, L2-3; RR+FC]; Böcher (1988: Fig. 1, as Nebria rufescens); Arndt (1991b: 71, 72) [L1, L2-3; ?]; Luff (1993: 50-52, as Nebria rufescens; Figs 131-136) [L1, L2-3; ?]; Sustek (1993: 522) [L3; LIT]; Makarov (1994: Figs 85, 86).
Nebria (Boreonebria) lacustris Casey: Spence (1974: 13, 17; Fig. 4) [L1, L2, L3; RR]; Spence et al. (1976: 81; Figs 2a-c, 3a) [L1, L2, L3; RR]; Spence and Sutcliffe (1982: Figs 1, 2, 6-19).
Nebria (Boreonebria) nivalis (Paykull): Andersen (1970: 91-93; Figs 6a-d) [L1-3; FC]; Luff (1993: 50, 52; Figs 137-142) [L1-3; FC].
Nebria (Reductonebria) pallipes Say: Spence (1974: 13, 17; Fig. 4) [L1, L2, L3; RR]; Spence et al. (1976: 81; Figs 1, 3b) [L1, L2, L3; RR].
Nebria prob. schwarzi (Alberta): Garner (1954: 186, 202, 203) [L2-3; FC].
Nebria prob. trifaria (Oregon): Garner (1954: 189, 204, 205) [L2-3; FC].
Nebria sp. A (Washington, D.C.): Garner (1954: 189-191) [L2-3; FC].
Nebria sp. B (California): Garner (1954: 189, 191, 192) [L2-3; FC].
Nebria sp. C (Utah): Garner (1954: 189, 192-194) [L2-3; FC].
Nebria sp. D (Oregon): Garner (1954: 189, 194, 195) [L2-3; FC].
Nebria sp. E (Oregon): Garner (1954: 189, 196, 197) [L2-3; FC].
Nebria sp. F (Tennessee): Garner (1954: 189, 197-199) [L2-3; FC].
Nebria sp. G (Montana): Garner (1954: 189, 199, 200) [L2-3; FC].
Nebria sp. H (Illinois): Garner (1954: 189, 200-202) [L2-3; FC].
Nebria sp. (Colorado): Thompson (1979a: Figs 36a-c).
Nebria sp.: Bousquet and Goulet (1984: Figs 27, 48); Bousquet (1991a: Figs 34.77, 34.101); Bousquet (2010a: Figs 377, 380, 382-385).
NotiophiliniNotiophilus aquaticus (Linnaeus): Schiødte (1867: 456; Fig. 19 [Pl. 13]) [L1-3; ?]; Larsson (1941: 283) [L1-3; ?]; Garner (1954: 174, 175) [L2-3; FC]; Sharova (1958: 38) [L1-3; ?]; Davies (1964: 208) [L1-3; LIT]; Sharova (1964: 145) [L1-3; ?]; Larsson (1968: 326-328) [L1-3; ?]; Haberman (1968: 135) [L2-3; ?]; Luff (1976: 52, 59; Figs 17-19) [L1, L3; FC]; Arndt (1991b: 74) [L1-3; ?]; Luff (1993: 61, 62; Figs 168-170) [L1, L2-3; ?].
Notiophilus biguttatus (Fabricius)†: Schiødte (1867: 456; Figs 12-17 [Pl. 13]) [L1-3; ?]; Saalas (1923: 667) [L1-3; ?]; Larsson (1941: 283, 284; Figs 20, 22a) [L1-3; ?]; Jeannel (1941b: Figs 75a-d); van Emden (1942: Fig. 4); Garner (1954: 176, 177) [L2-3; FC]; Sharova (1958: 39; Fig. 86a) [L1-3; ?]; Davies (1964: 207, 208; Fig. 1) [L1, L2-3; FC]; Sharova (1964: 146; Fig. 130-3) [L1-3; ?]; Larsson (1968: 327, 328; Figs 20, 22a) [L1-3; ?]; Haberman (1968: 135; Fig. 68-3) [L2-3; ?]; Luff (1976: 52, 53, 55; Figs 1-10) [L1, L2-3; FC]; Arndt (1991b: 74) [L1-3; ?]; Luff (1993: 61, 64; Figs 178-183) [L1, L2-3; ?].
Notiophilus novemstriatus LeConte: Thompson and Allen (1974: 186-189; Figs 1a-c) [L2-3; FC]; Thompson (1979a: Figs 38a-b).
Notiophilus palustris (Duftschmid)†: Larsson (1941: 283, 284) [L1-3; ?]; Sharova (1958: 39; Fig. 80) [L1-3; ?]; Davies (1964: 207, 208) [L1, L2-3; FC]; Sharova (1964: 146; Fig. 127) [L1-3; ?]; Larsson (1968: 327-329) [L1-3; ?]; Haberman (1968: 136) [L2-3; ?]; Luff (1976: 52, 56; Figs 12, 13) [L2; FC]; Arndt (1991b: 74; Fig. 64) [L1-3; ?]; Luff (1993: 61-63) [L1-3; ?].
Notiophilus sp. A (California): Garner (1954: 169, 170; Figs a-c [Pl. 10]) [L2-3; FC].
Notiophilus sp. B (Illinois): Garner (1954: 170-172) [L2-3; FC].
Notiophilus sp. C (Illinois): Garner (1954: 172-174) [L1, L2-3; FC].
Notiophilus sp. D (Oregon): Garner (1954: 177, 178) [L2-3; FC].
Notiophilus sp. E (Texas): Garner (1954: 179, 180) [L2-3; FC].
Notiophilus sp.: Bousquet (2010a: Fig. 381).
CychriniScaphinotus (Nomaretus) bilobus (Say): Bousquet (1977: 4.9; Figs 12, 13) [L3; FC]; Bousquet (2010a: 396; Fig. 396) [L3; FC].
Scaphinotus (Scaphinotus) elevatus (Fabricius): Wickham (1895b: 763; Fig. 1) [L3; FC].
Scaphinotus (Maronetus) hubbardi (Schwarz): Lapouge (1929a: 60) [L2-3; ?].
Scaphinotus (Pseudonomaretus) mannii Wickham: Greene (1975: Figs 2-3).
Scaphinotus (Brennus) marginatus (Fischer von Waldheim): Garner (1954: 58, 67, 68) [L2-3; FC].
Scaphinotus (Irichroa) viduus (Dejean): Bousquet (1977: 4.4-4.8; Figs 1-11) [L2-3; FC]; Bousquet (2010a: 396; Figs 390, 394) [L2-3; FC].
Scaphinotus (Brennus) sp. A (California): Garner (1954: 58, 59) [L2-3; FC].
Scaphinotus (Brennus) sp. B (California): Garner (1954: 55, 60, 61) [L2-3; FC].
Scaphinotus (Brennus) sp. C (California): Garner (1954: 58, 61, 62; Figs a-c [Pl. 2]) [L2-3; FC].
Scaphinotus (Brennus) sp. D (California): Garner (1954: 55, 63, 64) [L2-3; FC].
Scaphinotus (Nomaretus?) sp. E (Pennsylvania, North Carolina): Garner (1954: 55, 64, 65) [L2-3; FC].
Scaphinotus sp. F (Massachusetts): Garner (1954: 58, 66, 67) [L2-3; FC].
Scaphinotus (Nomaretus ?) sp. (Pennsylvania): van Emden (1942: 56) [L2-3; FC].
Scaphinotus sp. (Arkansas): Thompson (1979a: Figs 32a-b, as Sphaeroderus sp.).
Sphaeroderus canadensis Chaudoir: Bousquet (1977: 1.9; Figs 2, 3, 8, 16) [L1, L2, L3; RR+FC]; Bousquet (2010a: 398) [L2-3; RR+FC].
Sphaeroderus nitidicollis Guérin-Méneville: Lindroth (1955a: Fig. 7d); Bousquet (1977: 1.9, 1.10; Figs 5, 7, 17, as Sphaeroderus nitidicollis brevoorti) [L1, L2, L3; RR+FC]; Bousquet (2010a: 398) [L2-3; RR+FC].
Sphaeroderus stenostomus lecontei Dejean: Lindroth (1955a: Fig. 7c); Bousquet (1977: 1.9, 1.10; Figs 4, 6, 9-15, 18-20) [L1, L2, L3; RR+FC]; Bousquet (1991a: Figs 34.75, 34.79, 34.87, 34.93, 34.96, 34.99, 34.102); Bousquet (2010a: 398; Figs 391-393, 395, 397) [L2-3; RR+FC].
CarabiniCalosoma (Chrysostigma) affine Chaudoir: Garner (1954: 91, 108, 109) [L2-3; FC].
Calosoma (Chrysostigma) calidum (Fabricius): Schaupp (1882d: 33-34) [L3; FC]; Burgess (1897: 426-428; Figs 10 [Pl. 3], 3, 5-6 [Pl. 4]) [L1, L2, L3; RR]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1908b: 164) [L1-3; LIT]; Burgess and Collins (1917: 19, 101; Figs a-d [Pl. 18], a-b [Pl. 19]) [L1, L2, L3; RR]; Garner (1954: 91, 100-102) [L2-3; FC]; Kirk (1972: 1355; Figs 7 [Pl. 1], 3-7) [L1-3; FC]; Bousquet (2010a: 399; Fig. 403) [L2-3; RR+FC].
Calosoma (Chrysostigma) cancellatum Eschscholtz: Burgess and Collins (1917: 19, 112, 113; Fig. g [Pl. 17]) [L1, L2, L3; RR]; Garner (1954: 91, 99, 100) [L2-3; FC].
Calosoma (Callitropa) externum (Say): Burgess and Collins (1917: 20, 23, 24) [L1, L2, L3; RR]; Lapouge (1927: 438) [L2-3; ?]; Lapouge (1929a: 56) [L2-3; ?]; Garner (1954: 90, 113, 114) [L2-3; FC].
Calosoma (Calosoma) frigidum Kirby: Burgess (1897: 420-425; Figs 3-9 [Pl. 3], 1, 4, 7 [PL. 4]) [L1, L2, L3; RR]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1908b: 163) [L1-3; LIT]; Burgess and Collins (1917: 19, 48-50) [L1, L2, L3; RR]; Lapouge (1927: 439) [L2-3; ?]; Lapouge (1929a: 57) [L2-3; ?]; Garner (1954: 90, 93, 94) [L2-3; FC]; Bousquet and Goulet (1984: Figs 40, 45, 47); Bousquet (1991a: Fig. 34.86); Bousquet (2010a: 399; Figs 398-402, 404, 405) [L2-3; RR+FC].
Calosoma (Blaptosoma) haydeni Horn: Burgess and Collins (1917: 20, 96; Fig. e [Pl. 17]) [L1, L2, L3; RR]; Garner (1954: 91, 106, 107) [L2-3; FC].
Calosoma (Chrysostigma) lepidum LeConte: Lapouge (1927: 439) [L2-3; ?]; Lapouge (1929a: 58) [L2-3; ?].
Calosoma (Camegonia) marginale Casey: Burgess and Collins (1917: 20, 83, as Calosoma lugubre) [L1, L2, L3; RR]; Lapouge (1927: 439, as Calosoma lugubris) [L2-3; ?]; Lapouge (1929a: 57, as Calosoma lugubris) [L2-3; ?]; Garner (1954: 90, 109-111, as Calosoma lugubre) [L2-3; FC].
Calosoma (Chrysostigma) obsoletum Say: Burgess and Collins (1917: 19, 89; Fig. d [Pl. 17]) [L1, L2, L3; RR]; Garner (1954: 91, 104-106) [L2-3; FC].
Calosoma (Carabosoma) peregrinator Guérin-Méneville: Burgess and Collins (1917: 20, 77) [L1, L3; RR]; Lapouge (1927: 438) [L2-3; ?]; Lapouge (1929a: 56) [L2-3; ?]; Garner (1954: 90, 111, 112) [L2-3; FC].
Calosoma (Castrida) sayi Dejean: Lapouge (1927: 437, 438) [L2-3; ?]; Lapouge (1929a: 56) [L2-3; ?]; Thompson (1979a: Figs a-d).
Calosoma (Calodrepa) scrutator (Fabricius): Chapuis and Candèze (1853: 372, 373; Fig. 4 [Pl. 1]) [L1-3; FC]; Schaupp (1879: 14) [L2-3; LIT]; Burgess and Collins (1917: 19, 31; Figs a-d [Pl. 8], a, b [Pl. 9]) [L1, L2, L3; RR]; Lapouge (1927: 440) [L2-3; ?]; Lapouge (1929a: 59) [L2-3; ?]; Garner (1954: 90, 95, 96; Figs a-c [Pl. 4]) [L2-3; FC].
Calosoma (Chrysostigma) semilaeve LeConte: Burgess and Collins (1917: 19, 92; Fig. f [Pl. 17]) [L1, L2, L3; RR]; Garner (1954: 91, 102-104) [L2-3; FC].
Calosoma (Calosoma) sycophanta (Linnaeus)†: Burmeister (1837: 235, 236; Figs 1-5 [Pl. 13]) [L1-3; FC]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1908b: 159, 160) [L1-3; FC]; Burgess (1911: 26, 27) [L1, L2, L3; RR]; Burgess and Collins (1915: 7, 8) [L1-3; RR]; Burgess and Collins (1917: 18, 19, 64, 65; Figs a-d [Pl. 10], Figs a, b [Pl. 11]) [L1, L2, L3; RR]; Lapouge (1927: 440) [L2-3; ?]; Lapouge (1929a: 59) [L2-3; ?]; Larsson (1941: 263-265; Figs 9b, 10b) [L1-3; ?]; Jeannel (1941b: Fig. 61k); Garner (1954: 90, 97, 98) [L2-3; FC]; Sharova (1957: 882; Figs 1a, 2a, 3a) [L1-3; LIT]; Sharova (1958: 28; Figs 69a, 71a) [L1-3; ?]; Sturani (1962: Fig. 38); Sharova (1964: 135; Figs 118-1, 120-1) [L1-3; ?]; Larsson (1968: 306, 308; Figs 9b, 10b) [L1-3; ?]; Haberman (1968: 114, 115; Figs 55-1, 55-6) [L2-3; ?]; Thompson (1979a: Figs 13a-c); Casale et al. (1982: 82-83; Fig. 26b) [L1-3; ?]; Arndt (1991b: 62; Fig. 29) [L1-3; LIT]; Luff (1993: 33, 34; Figs 58-62) [L1, L2-3; ?].
Calosoma (Calodrepa) wilcoxi LeConte: Burgess and Collins (1917: 19, 41, 42) [L1, L2, L3; RR]; Lapouge (1927: 439, 440) [L2-3; ?]; Lapouge (1929a: 58) [L2-3; ?]; Garner (1954: 90-93) [L2-3; FC].
Carabus (Tachypus) auratus Linnaeus†: Laboulbène (1862: 562, 563, as Calathus gallicus, see Raynaud 1940a; Figs 8-15 [Pl. 13]) [L3; FC]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1907: 52, 53) [L1; RR]; Lengerken (1921: 82-111; Figs 1-3, 7-21, 23-31, 33-51, 57-70) [L1, L2, L3; RR]; Verhoeff (1921: 185, 188) [L2-3; ?]; van Emden (1925; Fig. 1); Bengtsson (1927: 37, 43-46; Figs 14, 15, 22-26) [L1, L3; ?FC]; Lapouge (1927: 447) [L2-3; ?]; Lapouge (1929a: 53) [L2-3; ?]; Sturani (1962: Figs 37, 38); Hůrka (1971: 23, 24, 44, 46, 47; Figs 13a-o) [L1, L2, L3; FC]; Raynaud (1976a: 38; Pl. 71) [L1-3; ?]; Arndt (1985: 50; Figs 13, 64, 66, 70, 104) [L1-3; ?]; Arndt (1991b: 64) [L1-3; LIT]; Luff (1993: 38) [L1-3; ?].
Carabus (Diocarabus) chamissonis Fischer von Waldheim: ?Garner (1954: 75, 81, 82) [L2-3; FC]; Lindroth (1955a: Figs 8b, e); Bousquet (2010a: 401; Figs 406, 413) [L2-3; FC].
Carabus (Carabus) goryi Dejean: Garner (1954: 75, 79, 80, as Carabus limbatus) [L2-3; FC].
Carabus (Carabus) granulatus Linnaeus†: Schiødte (1867: 493-494; Figs 13-15 [Pl. 17]) [L1-3; ?]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1908b: 152, 153) [L1-3; RR]; Verhoeff (1917: 42) [L1-3; ?]; Verhoeff (1921: 175, 176, 184, 187; Fig. 7) [L2, L3; RR]; Oertel (1924: 326-356; Figs h, j-z, a1-h1, j1) [L1, L2, L3; RR]; Bengtsson (1927: 38, 57-59; Figs 46-50) [L1, L2-3; FC]; Lapouge (1927: 448) [L2-3; ?]; Lapouge (1929a: 54) [L2-3; ?]; Larsson (1941: 267, 269, 270; Fig. 12a) [L1-3; ?]; Garner (1954: 75, 85, 86) [L2-3; FC]; Sharova (1958: 32; Figs 7, 72h, 73d, 76g, 79e) [L1-3; ?]; Sturani (1962: Fig. 37); Sharova (1964: 139; Figs 89-1, 121-8, 122-4, 124-7, 126-5) [L1-3; ?]; Larsson (1968: 310, 313; Fig. 12a) [L1-3; ?]; Haberman (1968: 95; Figs 35-5, 37-a, 38-1, 41-4) [L2-3; ?]; Luff (1969: 251, 255; Figs 40-46) [L1-3; FC]; Hůrka (1971: 23, 24, 54-56; Figs 16a-u) [L1, L2, L3; FC]; Raynaud (1976a: 107; Pls 90, 91, 107) [L1-3; ?]; Casale et al. (1982: 285; Fig. 103) [L1-3; ?]; Arndt (1985: 52; Figs 14, 68b, 105) [L1-3; ?]; Arndt (1991b: 66) [L1-3; LIT]; Luff (1993: 38, 42, 43; Figs 101-103) [L1-3; ?]; Turin et al. (1993; Figs 4a, c, e); Huk and Kühne (2000: Fig. 1); Bousquet (2010a: 401; Fig. 407) [L2-3; FC].
Carabus (Homoeocarabus) maeander Fischer von Waldheim: Lindroth (1955a: Figs 8a, d); Raynaud (1976a: 107) [L1-3; ?]; Bousquet (2010a: 401; Figs 408-410) [L2-3; FC].
Carabus (Archicarabus) nemoralis Müller†: Schiødte (1867: 490-491; Figs 9-11 [Pl. 16]) [L1-3; ?]; Zang (1901: 274-275; Figs 2-5) [L1; RR]; Lapouge (1905: 172) [L3; ?RR]; Lapouge (1908b: 150, 151) [L1-3; RR]; Verhoeff (1917: 42, 43) [L1-3; ?]; Verhoeff (1921: 186-188; Figs j-1, q [Pl. 9]) [L2-3; ?]; van Emden (1925; Fig. 2); Bengtsson (1927: 38, 51-54; Figs 1, 2, 37-41) [L1, L2-3; RR]; Lapouge (1927: 448) [L2-3; ?]; Lapouge (1929a: 53) [L2-3; ?]; Delkeskamp (1930: 30-54; Figs 12-40) [L1, L2, L3; RR]; Larsson (1941: 267, 269; Figs 7, 11c) [L1-3; ?]; Garner (1954: 75-77) [L2-3; FC]; Sharova (1958: 31; Figs 72a, 74a, 76a, 79a) [L1-3; ?]; Sturani (1962: Fig. 44b); Sharova (1964: 137, 138; Figs 121-1, 123-1, 124-1, 126-1) [L1-3; ?]; Larsson (1968: 310, 312; Figs 7, 11c) [L1-3; ?]; Haberman (1968: 93, 95; Figs 35-1, 38-7, 39-1, 41-1) [L2-3; ?]; Luff (1969: 251, 255, 257; Figs 47-52) [L1-3; FC]; Raynaud (1969: 24) [L3; ?]; Sturani (1971: Fig. 2); Hůrka (1971: 22, 25, 62, 64, 65; Figs 19a-o) [L1, L2, L3; FC]; Raynaud (1976a: 66; Pl. 76) [L1-3; ?]; Hůrka (1978: Fig. H-2); Casale et al. (1982: 177; Fig. 71b) [L1; FC]; Arndt (1985: 52; Figs 1, 18, 68a, 107, 110) [L1-3; ?]; Arndt (1991b: 66; Figs 26, 39, 51) [L1-3; LIT]; Makarov (1992: Fig. 9e); Luff (1993: 38, 42; Figs 6, 98-100) [L1-3; ?]; Deuve (1994a: Fig. 36); Bousquet (2010a: 401) [L2-3; FC].
Carabus (Hemicarabus) serratus Say: Lapouge (1927: 446) [L2-3; FC]; Lapouge (1929a: 50) [L2-3; FC]; Lindroth (1955a: Figs 8c, f); Raynaud (1975: 308) [L1-3; ?]; Bousquet (2010a: 401; Fig. 411) [L2-3; FC].
Carabus (Tanaocarabus) sylvosus Say: Lapouge (1927: 446) [L2-3; FC]; Lapouge (1929a: 50) [L2-3; FC]; Garner (1954: 75, 77-79; Figs a-c [Pl. 3]) [L2-3; FC]; Raynaud (1975: 264; Pl. 14) [L1-3; ?]; Thompson (1979a: Figs 29a-b); Bousquet (2010a: 402; Fig. 412) [L2-3; FC].
Carabus (Tanaocarabus) taedatus Fabricius: ?Lapouge (1927: 446) [L2-3; FC]; Lapouge (1929a: 50) [L2-3; FC]; ?Garner (1954: 75, 83, 84) [L2-3; FC]; Sturani (1971: Fig. 3t); Raynaud (1975: 222; Pl. 5) [L1-3; ?].
Carabus (Aulonocarabus) truncaticollis Eschscholtz: Kincaid (1900: 368, 369; Figs 1-6 [Pl. 22]) [L2-3; FC]; Lapouge (1907: 34) [L3; LIT]; Lapouge (1927: 445) [L2-3; ?]; Lapouge (1929a: 50) [L2-3; ?]; Garner (1954: 75) [L2-3; LIT]; Raynaud (1975: 260; Pl. 12) [L1-3; ?]; Makarov (1992: Figs 10b, g).
Carabus (Megodontus) vietinghoffii Adams: Lapouge (1927: 443) [L2-3; ?]; Raynaud (1976a: 14; Pl. 57) [L1-3; ?].
Carabus sp. No 19 (Alabama): Lapouge (1908b: 164) [L1; FC].
AmblycheiliniAmblycheila baroni Rivers: Knisley and Pearson (1984: 502; Figs 25-31) (L3; FC].
Amblycheila cylindriformis (Say): Horn (1878a: 29-31; Figs 1a-g [Pl. II]) [L2-3; FC]; Riley et al. (1878: Figs 53a-h); Schaupp (1879: 3) [L2-3; LIT]; Hamilton (1925: 70; Figs 4, 19, 11, 16, 20, 26, 31, 36, 41, 81, 123, 148) [L2-3; RR]; Böving and Craighead (1931: Figs b, d [Pl. 4]).
Amblycheila schwarzi Horn?: Horn (1908a: 285, 286; Figs 1-5) [L2-3; FC]; Putchkov and Arndt (1994: Figs 6, 13, 23, 44, 46, 50).
Omus audouini Reiche: Hamilton (1925: 66-68, as Omus ambiguus [see Leffler 1986b: 54]); Figs 47, 86, 121) [L2-3; RR]; Leffler (1979a: 70-71; Fig. 7a) [L3; FC]; Leffler (1986b: 55, 62; Fig. 2) [L3; FC].
Omus californicus Eschscholtz: Hamilton (1925: 66-67; Figs 3, 12, 13, 19, 25, 30, 35, 44, 46, 82-84, 118, 119) [L2-3; RR]; Hamilton (1925: 69, as Omus edwardsii [see Leffler 1986b: 54]) [L2-3; FC]; Hamilton (1925: 66, 68, 69, as Omus sequoiarum [see Leffler 1986b: 54]; Figs 45, 85, 120) [L2-3; RR]; Böving and Craighead (1931: Fig. e [Pl. 4]); van Emden (1935: Fig. 1); Leffler (1986b: 55-56, 62) [L3; FC]; Leffler (1979a: Fig. 4a).
Omus dejeanii Reiche: Horn (1878a: 31-33; Figs 2a-e [Pl. II]) [L2-3; FC]; Schaupp (1879: 3) [L2-3; LIT]; Leffler (1979a: 68-69) [L1, L2, L3; FC]; Leffler (1986b: 56-57, 62; Fig. 1) [L3; FC].
Omus submetallicus Horn: Leffler (1986b: 57-58, 62) [L3; FC].
Omus sp.: Putchkov and Arndt (1994: Fig. 24).
MegacephaliniTetracha (Tetracha) carolina (Linnaeus): Horn (1878a: 34-35; Figs 3a-e [Pl. II]) [L2-3; FC]; Schaupp (1879: 3) [L2-3; LIT]; Hamilton (1925: 59-60; Figs 2, 9, 15, 18, 24, 29, 34, 43, 80, 116) [L2-3; RR]; Putchkov and Arndt (1997: 61) [L3; LIT]; Leonard and Bell (1999: 27, 153; Figs 35-37, 148, 149) [L3; LIT].
Tetracha (Tetracha) virginica (Linnaeus): Hamilton (1925: 60-61; Figs 79, 117) [L2-3; RR]; Putchkov and Arndt (1997: 61) [L3; LIT].
CicindeliniCicindela (Cicindelidia) abdominalis Fabricius: Hamilton (1925: 19, 44-45; Figs 72, 109, 139) [L2-3; FC]; Spangler (1956: 84) [L2-3; LIT]; Willis (1967: 173) [L2-3; LIT].
Cicindela (Cicindelidia) amargosae Dahl: Leffler (1979a: 86-87) [L2-3; FC].
Cicindela (Cicindela) ancocisconensis Harris: Leonard and Bell (1999: 33, 63; Figs 76-78) [L3; FC]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindela) arenicola Rumpp: Rumpp (1967: 133; Figs 1[a-b]) [L3; FC]; Shook and Clark (1988: Fig. 2).
Cicindela (Cicindela) bellissima Leng: Leffler (1979a: 76-77; Fig. 5e) [L1-2, L3; FC].
Cicindela (Cicindela) decemnotata Say: Leffler (1979a: 80-81) [L3; FC].
Cicindela (Cicindela) denikei Brown: Kaulbars and Freitag (1993b: 45, 46, 48; Figs 1-7) [L3; FC].
Cicindela (Cicindela) depressula Casey: Leffler (1979a: 73-74) [L1-2, L3; FC].
Cicindela (Cicindela) duodecimguttata Dejean: Shelford (1908: 170, 172, 173; Figs 37-39) [L1, L2, L3; RR]; Hamilton (1925: 19, 39-40; Figs 68, 105, 135) [L2-3; RR]; Leonard and Bell (1999: 33, 97; Figs 51, 104, 105) [L3; LIT]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindela) formosa Say: Shelford (1908: 170, 172, 173; Figs 40-42) [L1, L2, L3; RR]; Hamilton (1925: 19, 46-48; Figs 49-51, 74, 88-90, 111, 141, 142) [L2-3; FC]; Gaumer
Cicindela (Cicindela) fulgida Say: Willis (1967: 165-168, 173; Figs 41-46) [L3; FC].
Cicindela (Cicindelidia) hemorrhagica LeConte: Leffler (1979a: 88-89) [L3; FC]; Knisley and Pearson (1984: 497-499, 533-534; Figs 86-91) [L1, L2, L3; RR+FC].
Cicindela (Cicindela) hirticollis Say: Shelford (1908: 172, 173) [L1, L2, L3; RR]; Hamilton (1925: 20, 55; Figs 55-57, 92, 93, 146) [L2-3; RR]; Leffler (1979a: 75; Fig. 5f) [L2, L3; FC]; Thompson (1979a: Figs 12a-e); Leonard and Bell (1999: 36, 80, 81; Figs 57, 92, 93) [L3; LIT]; Bousquet (2010a: 404; Fig. 418) [L2-3; LIT].
Cicindela (Cicindelidia) hornii Schaupp: Knisley and Pearson (1984: 496-498, 511-514; Figs 44-49) [L1, L2, L3; RR+FC].
Cicindela (Cicindela) latesignata LeConte: Hamilton (1925: 18, 29; Figs 61, 98, 128) [L2-3; FC].
Cicindela (Cicindela) lengi Horn: Leffler (1979a: 89-90) [L3; FC].
Cicindela (Cicindela) limbalis Klug: Shelford (1908: 172, 173; Figs 49-51) [L1, L2, L3; RR]; Hamilton (1925: 18, 25-26; Figs 1, 5-8, 14, 17, 21-23, 27, 28, 32, 33, 37-40, 42, 52-54, 96, 126) [L2-3; RR]; Böving and Craighead (1931: Fig. c [Pl. 4]); ?Bousquet and Goulet (1984: Fig. 28); Leonard and Bell (1999: 30, 119; Figs 44, 122-124) [L3; LIT]; Bousquet (2010a: 405; Figs 421, 423) [L2-3; LIT].
Cicindela (Cicindela) limbata Say: Hamilton (1925: 20, 56-57; Figs 77, 114, 145) [L2-3; FC]; Leonard and Bell (1999: 36, 89; Figs 101, 102) [L3; LIT].
Cicindela (Cicindela) longilabris Say: Leffler (1979a: 81-82) [L2; FC]; Spanton (1988: 99; Figs 28-32) [L1-3; RR]; Leonard and Bell (1999: 32, 107, 108; Figs 113, 114) [L2, L3; LIT]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindela) nebraskana Casey: Leffler (1979a: 83-84) [L3; FC]; Brust and Hoback (2010: 345; Figs 3-8) [L1, L2, L3; RR].
Cicindela (Cicindelidia) nigrocoerulea LeConte: Knisley and Pearson (1984: 497-499, 528-530; Figs 11, 74-79) [L1, L2, L3; RR].
Cicindela (Cicindelidia) obsoleta Say: Hamilton (1925: 18, 34-35, as Cicindela species D [see Knisley and Pearson (1984: 507)]; Figs 152, 164, 178) [L2-3; FC]; Knisley and Pearson (1984: 496-498, 505, 507; Figs 32-37) [L1, L2, L3; RR+FC].
Cicindela (Cicindelidia) ocellata Klug: Hamilton (1925: 19, 42-43, as Cicindela flavopunctata rectilatera; Figs 70, 107, 137) [L2-3; RR]; Knisley and Pearson (1984: 497, 499, 536-537; Figs 18, 21, 92-97) [L1, L2, L3; RR+FC].
Cicindela (Cicindela) oregona LeConte: Hamilton (1925: 18, 38; Figs 67, 104, 134) [L2-3; FC]; Leffler (1979a: 72; Figs 5b, 7b-c) [L2, L3; FC].
Cicindela (Cicindela) parowana Wickham: Leffler (1979a: 84-85) [L3; FC].
Cicindela (Cicindela) patruela Dejean: Willis (1982: 49, 56; Figs 1-6) [L3; RR]; Leonard and Bell (1999: 33, 46, 47; Figs 63-65) [L3; LIT].
Cicindela (Cicindela) pimeriana LeConte: Knisley and Pearson (1984: 496, 498, 519-520; Figs 13, 56-61) [L1, L2, L3; RR].
Cicindela (Cicindela) pugetana Casey: Leffler (1979a: 78-79) [L3; FC].
Cicindela (Cicindela) pulchra Say: Hamilton (1925: 19, 51; Figs 76, 113, 144) [L2-3; FC]; Hamilton (1925: 19, 51-52, as Cicindela obsoleta [see Knisley and Pearson (1984: 507)]; Figs 158, 169, 179) [L2-3; FC]; Knisley and Pearson (1984: 496-498, 508-509; Figs 1-9, 22-24, 38-43) [L1, L2, L3; RR+FC].
Cicindela (Cicindelidia) punctulata Olivier: Shelford (1908: 172, 173) [L1, L2, L3; RR]; Hamilton (1925: 19, 41; Figs 69, 106, 136) [L2-3; RR]; Knisley and Pearson (1984: 497-499, 525-526; Figs 68-73) [L1, L2, L3; RR]; Leonard and Bell (1999: 34, 112; Figs 52, 117, 118) [L3; LIT]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindela) purpurea Olivier: Shelford (1908: 170, 172-173; Figs 7-11, 25-27) [L1, L2, L3; RR]; Hamilton (1925: 18, 27; Figs 60, 97, 127) [L2-3; RR]; Leffler (1979a: 77-78; Fig. 5c) [L3; FC]; Leonard and Bell (1999: 31, 124; Figs 47, 128, 129) [L3; LIT]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindela) repanda Dejean: Horn (1878a: 35-37; Figs a-d [Pl. II]) [L2-3; FC]; Schaupp (1879: 13) [L2-3; LIT]; Schaupp (1884b: 123; Fig. 124) [L2-3; FC]; Shelford (1908: 170, 172, 173; Figs 34-36) [L1, L2, L3; RR]; Hamilton (1925: 18, 31; Figs 63, 100, 130) [L2-3; RR]; Leffler (1979a: 71; Fig. 5a) [L2, L3; FC]; Leonard and Bell (1999: 32, 53, 54; Figs 49, 68, 69) [L3; LIT]; Bousquet (2010a: 405; Figs 420, 422) [L2-3; LIT].
Cicindela (Cicindelidia) rufiventris Dejean: Beatty and Knisley (1984: 4-8; Figs 1-11) [L1, L2, L3; RR]; Leonard and Bell (1999: 32, 103, 104; Figs 46, 48, 109, 110) [L3; LIT].
Cicindela (Cicindela) scutellaris Say: Shelford (1908: 170-173; Figs 46-48) [L1, L2, L3; RR]; Hamilton (1925: 19, 48-49; Figs 75, 112, 143) [L2-3; RR]; Leonard and Bell (1999: 35, 131, 132; Figs 55, 133, 134) [L3; LIT]; Bousquet (2010a: 404; Fig. 417) [L2-3; LIT].
Cicindela (Cicindelidia) sedecimpunctata Klug: Knisley and Pearson (1984: 497, 539; Figs 116-119) [L3; RR].
Cicindela (Cicindela) sexguttata Fabricius: Shelford (1908: 170, 172, 173; Figs 28-30) [L1, L2, L3; RR]; Hamilton (1925: 17, 23-24; Figs 58, 94, 124) [L2-3; RR]; Willis (1967: 172) [L2-3; LIT]; Kaulbars and Freitag (1993b: 48) [L3; LIT]; Leonard and Bell (1999: 29, 41, 42; Figs 59, 60) [L3; LIT]; Bousquet (2010a: 405) [L2-3; LIT].
Cicindela (Cicindelidia) tenuisignata LeConte: Knisley and Pearson (1984: 497-499, 530-531; Figs 80-85) [L1, L2, L3; RR].
Cicindela (Cicindela) theatina Rotger: Pineda and Kondratieff (2002: 166, 170, 171; Figs 1-8) [L1, L2, L3; RR+FC].
Cicindela (Cicindela) tranquebarica Herbst: Shelford (1908: 172-173; Figs 43-45) [L1, L2, L3; RR]; Hamilton (1925: 18, 36; Figs 66, 103, 133) [L2-3; RR]; Leffler (1979a: 91) [L3; FC]; Leonard and Bell (1999: 33, 69; Figs 82, 83) [L3; LIT]; Bousquet (2010a: 405; Fig. 424) [L2-3; LIT].
Cicindela (Cicindelidia) willistoni LeConte: Willis (1967: 172, 173; Figs 59-64) [L3; FC]; Leffler (1979a: 87) [L3; FC]; Knisley and Pearson (1984: 497, 498, 514, 515; Figs 120-121) [L1, L2; FC].
Cicindela sp. A (California; probably Cicindela trifasciatus sigmoidea): Hamilton (1925: 17, 22-23; Figs 59, 95, 125) [L2-3; FC].
Cicindela sp. B (Colorado; possibly Cicindela cimarrona or Cicindela obsoleta): Hamilton (1925: 18, 28; Figs 62, 99, 129; according to Leffler (1979a: 78) and Knisley and Pearson (1984: 507) Hamilton’s species is neither Cicindela cimarrona or Cicindela obsoleta respectively) [L2-3; FC].
Cicindela sp. C (Indiana): Hamilton (1925: 18, 30; Figs 151, 163, 175) [L2-3; FC].
Cicindela sp. D (Colorado): Hamilton (1925: 18, 34-35; Figs 152, 164, 178) [L2-3; FC].
Cicindela sp.: Bousquet (1991a: Figs 34.76, 34.78, 34.92); Bousquet (2010a: Figs 414-416, 425).
Cylindera (Cylindera) cursitans (LeConte): Brust et al. (2005: 383-385; Figs 2-7) [L1, L2, L3; RR].
Cylindera (Cylindera) debilis (Bates): Knisley and Pearson (1984: 540, 542; Figs 16-17, 98-103) [L1, L2, L3; RR+FC].
Cylindera (Cylindera) lemniscata (LeConte): Knisley and Pearson (1984: 496, 498, 499, 542-545; Figs 104-109) [L1, L2, L3; RR+FC].
Cylindera (Cylindera) terricola Say: Leffler (1979a: 91-92, as Cylindera cinctipennis) [L3; FC].
Cylindera (Cylindera) unipunctata (Fabricius): Hamilton (1925: 19, 43-44; Figs 71, 108, 138) [L2-3; FC].
Cylindera (Cylindera) viridisticta (Bates): Knisley and Pearson (1984: 496, 498, 499, 545-546; Figs 110-115) [L1, L2, L3; RR+FC].
Dromochorus pruininus Casey: Spomer et al. (2008b: 38-40; Figs 1-6) [L1, L2, L3; RR].
Ellipsoptera gratiosa (Guérin-Méneville): Hamilton (1925: 18, 34; Figs 65, 102, 132) [L2-3; RR].
Ellipsoptera lepida (Dejean): Shelford (1908: 171-173; Figs 49-51) [L1, L2, L3; RR]; Hamilton (1925: 18, 32-33; Figs 64, 101, 131) [L2-3; RR]; Leonard and Bell (1999: 32, 146, 147; Figs 50, 144, 145) [L3; LIT]; Bousquet (2010a: 405; Fig. 419) [L2-3; LIT].
Ellipsoptera macra (LeConte): Shelford (1908: 171-173, as Ellipsoptera cuprascens; Figs 55, 56) [L1, L2; RR]; Hamilton (1925: 19, 49-50; Figs 153, 165, 172) [L2-3; RR+FC].
Ellipsoptera marginata (Fabricius): Hamilton (1925: 19, 45-46; Figs 73, 110, 140) [L2-3; RR]; Spangler (1956: 84) [L2-3; LIT]; Willis (1967: 173) [L2-3; LIT]; Leonard and Bell (1999: 29, 86; Figs 40, 98, 99) [L3; LIT]; Bousquet (2010a: 404) [L2-3; LIT].
Ellipsoptera nevadica (LeConte): Willis (1967: 168, 173; Figs 47-52) [L3; RR].
Ellipsoptera puritana (Horn): Leonard and Bell (1999: 33, 74, 75; Figs 87-90) [L3; LIT (unpublished)]; Bousquet (2010a: 405) [L2-3; LIT].
Ellipsoptera rubicunda (Harris): Knisley and Pearson (1984: 496-498, 516-517, as Ellipsoptera marutha; Figs 12, 50-55) [L1, L2, L3; RR+FC].
Eunota togata (LaFerté-Sénectère): Willis (1967: 170, 173; Figs 53-58) [L3; RR].
Habroscelimorpha circumpicta (LaFerté-Sénectère): Spangler (1956: 82, 84; 14 figs) [L2-3; FC]; Willis (1967: 163-165, 173; Figs 35-40) [L3; RR].
Habroscelimorpha dorsalis (Say): Hamilton (1925: 17, 20-21; Figs 78, 115, 147) [L2-3; RR]; Leonard and Bell (1999: 29, 142; Figs 141, 142) [L3; LIT].
Habroscelimorpha fulgoris (Casey): Knisley and Pearson (1984: 496-498, 522-523; Figs 14-15, 19-20, 62-67) [L1, L2, L3; RR+FC].
LoriceriniLoricera (Loricera) decempunctata Eschscholtz: Ball and Erwin (1969: 897; Fig. 49) [L3; FC].
Loricera (Loricera) pilicornis (Fabricius): Schiødte (1867: 465-469; Figs 8-16 [Pl. 14]) [L1-3; ?]; Gernet (1867: 10-12; Figs 2a-f [Pl. 1]) [L1-3; FC]; Larsson (1941: 290, as Loricera coerulescens; Figs 27, 28a-c) [L1-3; ?]; Jeannel (1941b: Figs 92a-f); van Emden (1942: Fig. 48); Garner (1954: 149, 150) [L2-3; FC]; Sharova (1958: 40; Figs 25a-e) [L1-3; ?]; Sharova (1964: 146; Figs 97-1, 2, 3, 4, 5, 6) [L1-3; ?]; Larsson (1968: 335; Figs 27, 28a-c) [L1-3; ?]; Haberman (1968: 155, 156; Figs 85.1-6) [L2-3; ?]; Ball and Erwin (1969: 894, 896; Figs 50, 53, 56, 57) [L1, L3; RR+FC]; Raynaud (1976d: 167, 168; Figs 1-7 [Pl. 16B]) [L3; FC]; Luff (1978: 269, 270; Figs 9-17) [L1, L2-3; FC]; Thompson (1979a: Figs 39a-c, as Loricera sp.); Bousquet and Goulet (1984: Figs 30, 49); Bousquet (1991a: Figs 34.85, 34.91); Arndt (1991b: Figs 67, 68); Luff (1993: 69, 70; Figs 1, 206-213) [L1, L2-3; ?]; Makarov (1994: Fig. 102); Makarov (1996; Fig. 39); Bousquet (2010a: Figs 426-430).
Loricera sp. A (California): Garner (1954: 149, 151, 152; Figs a-c [Pl. 8]) [L2-3; FC].
Loricera sp. B (California): Garner (1954: 149, 153, 154) [L2-3; FC].
ElaphriniBlethisa julii LeConte: Lindroth (1954a: 23, 24; Figs 9b, 10b, 11b, 12b, 13c, 15b) [L2-3; FC]; Sharova (1958: Fig. 90b); Sharova (1964: Figs 133-1); Thompson (1979a: Figs 41a-e).
Blethisa multipunctata (Linnaeus): Bøving (1910: 357-363; Figs 8, 12, 15) [L2-3; FC]; Znojko (1929: Fig. 4e); Larsson (1941: 287; Fig. 25) [L1-3; ?]; Jeannel (1941b: Figs 87a-e); van Emden (1942: Fig. 74); Lindroth (1954a: 24, Figs 11c, 13b, 14b, 15a) [L2-3; FC]; Garner (1954: 121, 122; Figs a-c [Pl. 6]) [L2-3; FC]; Sharova (1958: Figs 89a-e); Sharova (1964: Figs 132-1, 2, 3, 4, 5); Larsson (1968: 332; Fig. 25) [L1-3; ?]; Haberman (1968: Figs 83.1-5); Luff (1976: 60, 65, 67; Figs 39-47) [L3; FC]; Goulet (1983: Figs 96a-b); Arndt (1991b: 75; Fig. 66) [L1-3; ?]; Luff (1993: 65; Figs 184-189) [L2-3; ?]; Makarov (1994: Fig. 51); Makarov (1996: Fig. 26); Bousquet (2010a: 411; Figs 432, 435) [L2-3; RR+FC].
Blethisa quadricollis Haldeman: Lindroth (1954a: 23; Figs 9c, 11d, 13d, 14c) [L2-3; FC]; Goulet (1983: Figs 88a-b, 95); Bousquet (2010a: 411; Fig. 436) [L2-3; FC].
Diacheila arctica (Gyllenhal): Lindroth (1954a: Figs 9a, 10a, 11a, 12a, 13a, 14a); Sharova (1958: 41; Figs 27b, 90a, 92a) [L1-3; ?]; Sharova (1964: 149; Figs 98-2, 3, 133-1, 135-1) [L1-3; ?]; Goulet (1983: Figs 78, 84, 94); Luff (1993: 66; Figs 190-192) [L1-3; ?]; Makarov (1994: Fig. 50).
Diacheila polita (Faldermann): Sharova (1958: 41; Figs 26, 27a, 88, 92b) [L1-3; ?]; Sharova (1964: 149; Figs 98-1, 131-2, 135-2) [L1-3; ?]; Goulet (1983: Figs 79, 83a-c, 87a-b, 93a-b); Luff (1993: 66) [L1-3; LIT]; Bousquet (2010a: Figs 431, 434).
Elaphrus (Elaphrus) americanus americanus Dejean: Goulet (1983: 287, 288, 309; Figs 100a-b) [L1, L2, L3; RR]; Bousquet (2010a: 413; Figs 433, 437) [L2-3; RR].
Elaphrus (Elaphrus) americanus sylvanus Goulet: Goulet (1983: 311) [L1, L3; FC].
Elaphrus (Elaphroterus) angusticollis Sahlberg: Goulet (1983: 325, 326, 335-336; Figs 151, 152, 156) [L1, L2, L3; RR].
Elaphrus (Elaphrus) californicus Mannerheim: Goulet (1983: 286-288, 302; Figs 154, 155) [L1, L2, L3; RR]; Bousquet (2010a: 413) [L2-3; RR].
Elaphrus (Neoelaphrus) cicatricosus LeConte: Goulet (1983: 250, 251, 253, 263-264; Figs 99a-b) [L1, L2, L3; RR]; Bousquet (2010a: 412; Fig. 439) [L2-3; RR].
Elaphrus (Neoelaphrus) clairvillei Kirby: Lindroth (1954a: 26; Figs 12c, 14d, 16c, 17c) [L2-3; FC]; Thompson (1979a: Fig. 40b); Goulet (1983: 251-253, 275; Figs 76a-g, 80, 82a-b, 85a-c, 90a-b, 98a-c) [L1, L2, L3; RR]; Beutel (1993: Figs 17, 25); Bousquet (2010a: 412; Fig. 438) [L2-3; RR].
Elaphrus (Elaphrus) finitimus Casey: Goulet (1983: 288, 305-306) [L3; FC].
Elaphrus (Neoelaphrus) fuliginosus Say: Goulet (1983: 250, 251, 253, 261-262) [L1, L2, L3; RR]; Bousquet (2010a: 412) [L2-3; RR].
Elaphrus (Neoelaphrus) laevigatus LeConte: Goulet (1983: 251-253, 281-282) [L1, L2, L3; RR].
Elaphrus (Arctelaphrus) lapponicus Gyllenhal: Lindroth (1954a: 25; Figs 13e, 16a, 17a) [L2-3; FC]; Sharova (1958: Fig. 91); Sharova (1964: Fig. 134); Luff (1976: 62, 63, 65; Figs 34-38) [L1, L2-3; FC]; Thompson (1979a: Fig. 40a); Luff (1993: 67, 68; Figs 193-194) [L1, L2-3; ?]; Goulet (1983: 243; Figs 89a-b, 97) [L3; FC]; Bousquet (2010a: 412) [L2-3; FC].
Elaphrus (Elaphrus) lecontei Crotch: Garner (1954: 127, 128) [L3; FC]; Goulet (1983: 286-288, 297-298; Figs 81, 91a-b) [L1, L2, L3; RR].
Elaphrus (Neoelaphrus) lindrothi Goulet: Goulet (1983: 250-252, 265-266) [L1, L2, L3; RR].
Elaphrus (Neoelaphrus) olivaceus LeConte: Lindroth (1954a: 25; Figs 16b, 17b) [L2-3; FC]; Goulet (1983: 251-253, 279-280) [L1, L2, L3; RR]; Bousquet (2010a: 413) [L2-3; RR].
Elaphrus (Elaphroterus) purpurans Hausen: Goulet (1983: 324-326, 332-333; Figs 86, 92a-b, 102a-b) [L1, L2, L3; RR].
Elaphrus (Elaphrus) ruscarius Say: Goulet (1983: 287, 288, 295) [L1, L2, L3; RR].
Elaphrus (Elaphrus) tuberculatus Mäklin: Goulet (1983: 287, 288, 318) [L1, L2, L3; RR].
Elaphrus (Elaphrus) viridis Horn: Goulet and Smetana (1997: 215) [L3; FC].
OmophroniniOmophron (Omophron) americanum Dejean: Silvey (1936: 691-692; Figs 17-19) [L3; FC].
Omophron (Omophron) nitidum LeConte: ?Böving and Craighead (1931: Figs a-e [Pl. 5]).
Omophron (Omophron) tessellatum Say: Silvey (1936: 688-690; Figs 13, 15, 16) [L3; FC]; Landry and Bousquet (1984: Figs 1-21); Bousquet and Goulet (1984: Figs 29, 50, 52, 56); Bousquet (1991a: Figs 34.73, 34.88, 34.94, 34.97); Bousquet (2010a: Figs 440-447).
Omophron sp. A (California): Garner (1954: 134-136) [L2-3; FC].
Omophron sp. B (California): Garner (1954: 134, 136, 137) [L2-3; FC].
Omophron sp. C (Nebraska): Garner (1954: 134, 138, 139) [L2-3; FC].
Omophron sp. D (Nebraska): Garner (1954: 134, 140, 141; Pl. 7, Figs a-c) [L2-3; FC].
Omophron sp. (Ontario): Thompson (1979a: Figs 33a-e).
PasimachiniPasimachus (Pasimachus) elongatus LeConte: Garner (1954: 234, 237, 238) [L2-3; FC]; Kirk (1972: 1355; Figs 22 [Pl. 2], 3-22) [L1-3; FC]; Thompson and Allen (1974: 189-192; Figs 2a-c) [L3; FC]; Thompson (1979a: Figs 45a-d).
Pasimachus sp. A (North Carolina): Garner (1954: 234, 238, 239; Pl. 16, Figs a-c) [L2-3; FC].
Pasimachus sp. B (Texas): Garner (1954: 234, 240, 241) [L2-3; FC].
Pasimachus sp. C (Florida): Garner (1954: 234, 241, 242) [L2-3; FC].
Pasimachus sp. D (Montana): Garner (1954: 234, 242, 243) [L2-3; FC].
ScaritiniScarites (Scarites) quadriceps Chaudoir: van Emden (1942: Fig. 51); Garner (1954: 227, 231, 232, as Scarites prob. substriatus) [L2-3; FC]; Kirk (1972: 1355; Figs 23 [Pl. 2], 3-23, as Scarites substriatus) [L2-3; FC]; Thompson (1979a: Figs 46a-c, as Scarites substriatus).
Scarites (Scarites) subterraneus Fabricius: Wickham (1893b: 332, 333; Figs 3-4 [Pl. 9]) [L3; FC]; Garner (1954: 227, 230; Pl. 15, Figs a-c) [L2-3; FC]; Bousquet (2010a: Figs 448-450).
ClivininiClivina (Clivina) collaris (Herbst)†: Luff (1978: 272, 281; Fig. 57) [L1, L2; RR+FC]; Vaněk (1984: 100-103, 111; Figs 1-47) [L1, L2, L3; RR]; Arndt (1991b: 78, Figs 78, 80) [L1-3; LIT]; Luff (1993: 71, 72; Fig. 222) [L1, L2-3; ?]; Makarov (1994: Fig. 24).
Clivina (Semiclivina) dentipes Dejean: Thompson (1979a: Figs 47a-c).
Clivina (Clivina) fossor (Linnaeus)†: Bøving (1911: 148-153, 172-178; Pl. 6; Figs 4, 10, 15, 20, 23) [L2-3; FC]; Larsson (1941: 296) [L1-3; ?]; Jeannel (1941b: Figs 100a-g); van Emden (1942: Fig. 76); Garner (1954: 258, 262, 263) [L2-3; FC]; Sharova (1958: 44; Figs 32, 34, 97, 99a) [L1-3; ?]; Sharova (1964: 153; Figs 100-1, 101-1, 138, 139-2) [L1-3; ?]; Larsson (1968: 342; Fig. 32) [L1-3; ?]; Haberman (1968: Figs 87.1, 88.1, 3, 4); Luff (1978: 272, 280, 281; Figs 49-56) [L1, L2-3; RR+FC]; Vaněk (1984: 108, 109, 111; Figs 48-53) [L1, L2, L3; RR]; Arndt (1991b: 78) [L1-3; LIT]; Luff (1993: 71, 72; Figs 214-221) [L1, L2-3; ?]; Bousquet (2010a: Figs 451, 458).
Clivina (Clivina) impressefrons LeConte: Phillips (1909: 14-16; Figs 8b, e, f) [L3; FC]; Kirk (1972: 1355; Figs 16 [Pl. 2], 3-16) [L1-3; FC].
Clivina sp. A (Nebraska): Garner (1954: 258, 261, 262; Pl. 18, Figs a-c) [L2-3; FC].
Clivina sp. B (Louisiana): Garner (1954: 258, 264, 265) [L2-3; FC].
Clivina sp. C (Illinois): Garner (1954: 258, 265, 266) [L2-3; FC].
Schizogenius (Schizogenius) lineolatus (Say): Bousquet (1996b: 348-351; Figs 1-11) [L1, L2-3; RR+FC]; Bousquet (2010a: Figs 452, 454, 456, 459, 461).
Semiardistomis viridis (Say): Bousquet (2006c: 5; Figs 36-39) [L1; RR].
Genus A sp. A (Florida): Garner (1954: 270, 271; Pl. 19, Figs a-c) [L2-3; FC].
DyschiriiniAkephorus marinus LeConte: Garner (1954: 248, 251; Pl. 17, Figs a-c) [L2-3; FC].
Dyschirius dejeanii Putzeys: Kirk (1972: 1355; Figs 18 [Pl. 2], 3-18, as Dyschirius integer) [L1-3; FC].
Dyschirius globosus (Herbst)†: Larsson (1941: 292, 295) [L1-3; ?]; Sharova (1958: 44) [L1-3; ?]; Sharova (1964: 154) [L1-3; ?]; Larsson (1968: 338, 341) [L1-3; ?]; Haberman (1968: 167) [L1-3; ?]; Raynaud (1976e: 276, 277; Figs 4-8) [F3; FC]; Luff (1978: 271, 277; Figs 41-47) [L3; FC]; Arndt (1991b: 78) [L1-3; ?]; Luff (1993: 73, 76; Figs 243-245) [L1-3; ?].
Dyschirius globulosus (Say): Garner (1954: 248, 254) [L3; FC].
Dyschirius politus (Dejean): Larsson (1941: 293, 294; Figs 30d, 31e) [L1-3; ?]; Sharova (1958: 45; Fig. 101e) [L1-3; ?]; Sharova (1964: 154; Fig. 139-7) [L1-3; ?]; Larsson (1968: 338 340; Figs 30d, 31e) [L1-3; ?]; Haberman (1968: 168; Fig. 95.7) [L1-3; ?]; Luff (1978: 271, 277; Figs 38-40) [L3; FC]; Arndt (1991b: 78) [L1-3; ?]; Luff (1993: 73, 75, 76; Figs 231, 232) [L1-3; ?].
Dyschirius sphaericollis (Say): Silvey (1936: 685-687; Figs 9, 11, 12) [L3; FC].
Dyschirius sp. A (Louisiana): Garner (1954: 248, 252, 253) [L2-3; FC].
Dyschirius sp. (Quebec): Bousquet (1988a: Figs 31-34); Bousquet (2010a: Figs 453, 455, 457, 460).
PromecognathiniPromecognathus laevissimus (Dejean)
Broscus cephalotes (Linnaeus)†: Schiødte (1867: 504-507; Figs 1-8 [Pl. 19]) [L1-3; ?]; Znojko (1929: Fig. 8a); Larsson (1941: 297; Figs 32a-b) [L1-3; ?]; Jeannel (1941b: Figs 110a-e); van Emden (1942: Fig. 39); Sharova (1958: 45; Figs 36, 37) [L1-3; ?]; Sharova (1964: 154, 155; Figs 102-1, 2) [L1-3; ?]; Larsson (1968: 343; Figs 33a-b) [L1-3; ?]; Andersen (1968: 74) [L3; ?]; Haberman (1968: Figs 112d1-d2); Luff (1978: 282-285; Figs 58-65) [L1, L2-3; FC]; Sharova and Makarov (1984: 742, 744, 746; Figs 1-2) [L1, L2, L3; FC]; Arndt (1991b: Figs 69-71); Luff (1993: 77, 78; Figs 246-251) [L1, L2-3; ?]; Beutel (1993: Figs 4, 10, 18); Makarov (1994: Fig. 26); Bousquet (2010a: Figs 464, 468).
Miscodera arctica (Paykull): Andersen (1968: 71-74; Fig. 1, 2a-c, 3a-b) [L3; FC]; Larsson (1968: 343) [L1-3; ?]; Luff (1978: 282, 285, 286; Figs 66-75) [L1, L2-3; FC]; Thompson (1979a: Figs 43a-f); Luff (1993: 79; Figs 252, 253) [L1, L2-3; FC]; Makarov (1994: Fig. 25); Bousquet (2010a: Figs 462, 463, 465-467).
GehringiiniGehringia olympica Darlington: Lindroth (1960a: 37; Figs 5a-f) [L1; RR]; Thompson (1979a: Figs 27a-f); Luna de Carvalho (1989: 55; Fig. 27b) [L1-3; LIT].
TrechiniBlemus discus (Fabricius)†: ?Bousquet (2010a: Figs 469-473).
Neaphaenops tellkampfii (Erichson): Packard (1874: 563, as Pseudanophthalmus sp.) [L3; FC]; Hubbard (1888: 77-78; Figs 18a-b, as Pseudanophthalmus sp.) [L3; FC]; Jeannel (1920a: 538, 539; Fig. 62) [L3; FC]; Jeannel (1926: 396) [L3; FC].
Pseudanophthalmus sp. A (Higginbotham’s Cave, West Virginia): Boldori (1951: 143, 144; Figs 39, 42) [L2-3; FC].
Pseudanophthalmus sp. B (Higginbotham’s Cave, West Virginia): Boldori (1951: 145) [L2-3; FC].
Pseudanophthalmus sp. C (Coffman’s Cave, West Virginia): Boldori (1951: 145; Fig. 40) [L2-3; FC].
Pseudanophthalmus sp. D (Shelta Cave, Alabama): Boldori (1951: 145; Fig. 41) [L2-3; FC].
Pseudanophthalmus sp. E (Johnson’s Cave, Tennessee): Boldori (1951: 145) [L2-3; FC].
Trechus (Trechus) apicalis Motschulsky: Thompson (1979a: Figs a-d); ?Bousquet (2010a: Fig. 474).
Trechus (Trechus) obtusus Erichson†
Trechus (Trechus) quadristriatus (Schrank)†: Bøving (1911: 141-147, 172-178; Figs 3, 9, 14, 19, 22 [Pl. 5]) [L2-3; FC]; Jeannel (1920a: 523, 539; Figs 13-17) [L1-3; FC]; Jeannel (1926: 387) [L2-3; FC]; Beier and Strouhal (1928: 1-4; Figs 1a-b, 2a-e, 3 [Pl. 1]) [L3; FC]; Boldori (1931a: 6) [L3; FC]; Boldori (1931b: 158; Figs 1-2, 2-2) [L3; FC]; Larsson (1941: 303; Fig. 34b) [L1-3; ?]; Jeannel (1941b: Figs 113a-f); Boldori (1951: Fig. 2); Sharova (1958: 47; Fig. 105d) [L1-3; ?]; Sharova (1964: 157; Fig. 143-4) [L1-3; ?]; Larsson (1968: 352, 353; Fig. 36b) [L1-3; ?]; Haberman (1968: 273; Fig. 122.3) [L1-3; ?]; Luff (1985: 302, 304; Figs 1-10) [L1, L2-3; FC]; Arndt (1991b: 82; Figs 72, 73) [L1-3; ?]; Luff (1993: 84-86; Figs 281-283) [L1, L2-3; ?]; Makarov (1994: Fig. 32).
Trechus (Trechus) rubens (Fabricius)†: Larsson (1941: 303, 304; Fig. 34a) [L1-3; ?]; Sharova (1958: 47; Fig. 105b) [L1-3; ?]; Larsson and Gígja (1959: 27) [L1-3; FC]; Sharova (1964: 157; Fig. 143-2) [L1-3; ?]; Larsson (1968: 352, 353; Fig. 36a) [L1-3; ?]; Haberman (1968: 273; Fig. 122.2) [L2-3; ?]; Luff (1985: 302, 304, 306; Figs 12-16) [L3; FC]; Bousquet (1991a: Figs 34.95, 34.98); Arndt (1991b: 82) [L1-3; ?]; Luff (1993: 85; Figs 278-280) [L1-3; ?].
BembidiiniAsaphidion alaskanum Wickham: Maddison (1993: 153, 271; Figs 195, 225) [L1; RR].
Asaphidion yukonense Wickham: Thompson (1979a: Figs a-c).
Bembidion (Odontium) aenulum Hayward: Maddison (1993: 172; Fig. 223) [L1; RR].
Bembidion (Bracteon) alaskense Lindroth: Maddison (1993: 172, 178; Figs 212, 227, 233, 236) [L1, L2; RR].
Bembidion (Pseudoperyphus) antiquum Dejean: Maddison (1993: Figs 222, 247).
Bembidion (Bracteon) balli Lindroth: Maddison (1993: 173-174; Figs 191, 196, 210, 240, 242, 245, 248, 252, 255, 257, 258) [L1, L2; RR].
Bembidion (Odontium) bowditchii LeConte: Maddison (1993: 173) [L1; RR].
Bembidion (Peryphus) bruxellense Wesmael†: Larsson (1941: 299, 300, as Bembidion rupestre; Fig. 33g) [L1-3; ?]; Sharova (1958: 49, as Bembidion rupestre; Fig. 108f) [L1-3; ?]; Sharova (1964: 159, as Bembidion rupestre; Fig. 143-10) [L1-3; ?]; Larsson (1968: 347, 349; Fig. 35g) [L1-3; ?]; Luff (1993: 91, 98, 99; Figs 327, 328) [L1-3; ?].
Bembidion (Bracteon) carinula Chaudoir: Maddison (1993: 172, 181; Figs 197, 198, 213, 228, 234, 243, 246, 250, 253, 256, 260) [L1, L2; RR].
Bembidion (Pseudoperyphus) chalceum Dejean: Maddison (1993: Fig. 221).
Bembidion (Ochthedromus) cheyennense Casey: Maddison (1993: 172; Fig. 220) [L1; RR].
Bembidion (Odontium) confusum Hayward: Silvey (1936: 682-684; Figs 5, 7, 8) [L3; FC]; Maddison (1993: 173) [L1; RR].
Bembidion (Notaphus) contractum Say: Lindroth (1955a: Fig. 15b).
Bembidion (Odontium) coxendix Say: Maddison (1993: 173; Figs 192, 200, 219) [L1; RR].
Bembidion (Peryphus) femoratum Sturm†: Netolitzky (1926: 118; Fig. 3) [L1; RR]; Arndt (1991b: Fig. 75).
Bembidion (Bracteon) foveum Motschulsky: Maddison (1993: 173, 175; Figs 199, 201-203, 205, 211, 226, 232, 238, 249) [L1, RR].
Bembidion (Peryphanes) grapii Gyllenhal: Larsson and Gígja (1959: 23) [L1-3; FC]; Böcher (1988: Fig. 4); Luff (1993: 92, 99; Figs 329, 330) [L1-3; ?].
Bembidion (Bracteon) hesperium Fall: Maddison (1993: 173, 186) [L1, L2; RR].
Bembidion (Bracteon) inaequale Say: Maddison (1993: 192; Fig. 193) [L1, L2; RR].
Bembidion (Pseudoperyphus) integrum Casey: Maddison (1993: Fig. 244).
Bembidion (Leja) lampros (Herbst)†: Larsson (1941: 299; Fig. 33b) [L1-3; ?]; Larsson (1968: 346, 348; Fig. 35b) [L1-3; ?]; Desender and Crappé (1983: 39, 46; Figs 4a-i, 5a-h, 6a-h) [L1-3; RR+FC]; Luff (1993: 91, 93; Figs 304, 305) [L1, L2-3; ?].
Bembidion (Bracteon) lapponicum Zetterstedt: Andersen (1966: 446, 448, 449; Figs 1, 17-26) [L1, L2, L3; RR+FC]; Maddison (1993: 172, 184; Figs 206-209, 214, 229, 235, 259) [L1, L2; RR]; Luff (1993: 91-93; Figs 299-301) [L1, L2-3; LIT].
Bembidion (Bracteon) levettei Casey: Maddison (1993: 190; Figs 218, 231) [L1, L2; RR].
Bembidion (Bracteon) lorquinii Chaudoir: Maddison (1993: 173, 187-188; Fig. 216) [L1, L2; RR].
Bembidion (Eurytrachelus) nitidum (Kirby): Kirk (1972: 1355; Figs 26 [Pl. 3], 3-26) [L2-3; FC].
Bembidion (Phyla) obtusum Audinet-Serville†: Maddison (1993: Fig. 224).
Bembidion (Peryphus) petrosum Gebler: Andersen (1966: Fig. 43); Luff (1993: 91, 98; Figs 325, 326) [L1-3; ?].
Bembidion (Leja) properans (Stephens)†: Desender and Crappé (1983: 39, 46; Figs 1a-h, 2a-h, 3a-h) [L1-3; RR+FC].
Bembidion (Bracteon) punctatostriatum Say: Maddison (1993: 172, 185; Figs 194, 204, 215, 230, 237, 239, 241, 251, 254) [L1, L2; RR].
Bembidion (Bembidion) quadrimaculatum (Linnaeus): Kirk (1972: 1355; Figs 25 [Pl. 3], 3-25, as Bembidion quadrimaculatum) [L1-3; FC].
Bembidion (Notaphus) semipunctatum (Donovan): Makarov (1994: Fig. 37).
Bembidion (Plataphus) sulcipenne prasinoides Lindroth: Lindroth (1955a: Fig. 15a, as Bembidion lenense).
Bembidion (Peryphus) tetracolum Say†; Larsson (1941: 299, 300, as Bembidion ustulatum; Fig. 33f) [L1-3; ?]; Sharova (1958: 49, as Bembidion ustulatum; Fig. 108e) [L1-3; ?]; Sharova (1964: 159, as Bembidion ustulatum; Fig. 143-9) [L1-3; ?]; Larsson (1968: 347, 349; Fig. 35f) [L1-3; ?]; Kirk (1972: 1355; Figs 27 [Pl. 3], 3-27) [L1-3; FC]; Smrž (1979: 247, 249; Figs 1, 4, 6, 7, 10, 12, 14, 15, 17, 19, 21, 24, 26, 27, 29, 31, 33, 35, 38, 39, 42, 44, 46, 47, 50, 52, 54, 56) [L1, L2, L3; RR]; Luff (1993: 91, 97; Figs 320-324) [L1, L2-3; ?].
Bembidion (Bracteon) zephyrum Fall: Maddison (1993: 189; Fig. 217) [L1, L2; RR].
Bembidion sp. (Arkansas): Thompson (1979a: Figs a-b).
Bembidion sp.: Bousquet (2010a: Figs 475, 478, 480, 482).
Elaphropus (Barytachys) anceps (LeConte): Kirk (1972: 1355; Figs 24 [Pl. 2], 3-24) [L1-3; FC].
Elaphropus (Tachyura) parvulus (Dejean)†: Cerruti (1939: 121-123; Figs 1-3) [L1; RR]; van Emden (1942 : 31) [L1; LIT]; Luff (1993: 102; Figs 337-339) [L1; FC].
Elaphropus (Barytachys) tripunctatus (Say): Grebennikov and Maddison (2000: 237; Figs 8, 33) [L1, L2; RR].
Elaphropus (Barytachys) sp.: Bousquet (2010a: Figs 477, 484).
Mioptachys flavicauda (Say): Thompson (1979a: Figs 48); Grebennikov and Maddison (2000: 230; Figs 3, 12, 22, 40) [L1; RR]; Bousquet (2010a: Figs 476, 479, 481, 483).
Phrypeus rickseckeri (Hayward): Grebennikov and Maddison (2005: 43, 44; Figs 5-10) [L1; RR].
Polyderis laeva (Say): Grebennikov and Maddison (2000: 236; Figs 20, 30, 52) [L2; RR].
Polyderis rufotestacea (Hayward): ?Grebennikov and Maddison (2000: 236; Figs 6, 29) [L1; RR].
Porotachys bisulcatus (Nicolai)†: Grebennikov and Maddison (2000: 238; Figs 11, 17, 38, 39, 48, 56, 60) [L1, L2, L3; RR].
Tachys (Tachys) halophilus Lindroth: Grebennikov and Maddison (2000: 235; Fig. 19) [L1; RR].
Tachyta (Tachyta) inornata (Say): Erwin (1975: Figs 158, 160, 162, 164, 169, 171-173); Thompson (1979a: Figs 49a-d).
PogoniniThalassotrechus barbarae (Horn): Moore (1956: 215; Figs 1-7) [L2-3; FC]; Thompson (1979a: Figs a-g); Grebennikov and Bousquet (1999: 438; Figs 6, 13, 16, 20) [L1; RR].
PatrobiniDiplous (Platidius) aterrimus (Dejean): Thompson (1979a: Figs a-c).
Diplous (Platidius) rugicollis (Randall): Bousquet (2010a: Figs 487, 490, 492).
Patrobus foveocollis (Eschscholtz): Bousquet and Goulet (1984: Figs 31, 53, 57).
Patrobus longicornis (Say): Schaupp (1882a: 56) [L2-3; FC]; Thompson (1979a: Figs 55a-d); Bousquet (2010a: Figs 485, 488, 489).
Patrobus septentrionis Dejean: Larsson (1941: 305, 306; Fig. 35b) [L1-3; ?]; Larsson and Gígja (1959: 30) [L1-3; FC]; Larsson (1968: 356, 357; Fig. 38b) [L1-3; ?]; Houston and Luff (1975: 60, 64; Fig. 16) [L1, L2-3; FC]; Böcher (1988: Fig. 2); Arndt (1991b: 84, 85) [L1, L2-3; LIT]; Luff (1993: 79, 80; Figs 254-257) [L1, L2-3; ?].
Platypatrobus lacustris Darlington: Bousquet and Grebennikov (1999: 6, 8, 10; Figs 7-15) [L1, L2, L3; RR+FC].
PsydriniNomius pygmaeus (Dejean): Jeannel (1948b: 74, 75; Figs 1a-c) [L3; FC]; Hůrka (1978: 54) [L1-3; LIT].
Psydrus piceus LeConte: Dajoz (1997b: 133-134; Figs 1-11) [L3; FC].
MetriiniMetrius contractus Eschscholtz
Goniotropis kuntzeni Bänninger: Moore and Di Giulio (2006: 10-14, 16; Figs 9-36) [L1, L2; RR].
BrachininiBrachinus (Neobrachinus) americanus (LeConte): Thompson (1979a: Figs 86a-b).
Brachinus (Neobrachinus) cyanipennis Say: Clausen (1940: Figs 226a-c).
Brachinus (Neobrachinus) janthinipennis (Dejean): Wickham (1893b: 330-332; Figs 1-2 [Pl. 9]) [L1-3; FC]; Dimmock and Knab (1904: 31-34; Fig. 4; Figs 1-2 [Pl. 3]) [L1, L2-5; FC].
Brachinus (Neobrachinus) pallidus Erwin: Erwin (1967: 44-47; Figs 1-3 [Pl. 1]; Figs 1-6 [Pl. 2]; Figs 1-6 [Pl. 3]) [L1, L2, L3; L4-5; FC]; Thompson (1979a: Figs 85a-b).
Brachinus (Neobrachinus) sp.: Bousquet and Goulet (1984: Figs 26, 38, 43, 55); Bousquet (1991a: Figs 34.90, 34.100); Bousquet (2010a: Figs 493-497).
MorioniniMorion monilicornis (Latreille): van Emden (1953a: 51, as Morion georgiae) [L2-3; FC]; Thompson (1979b: 85, 86; Figs 48a-g) [L2-3; FC].
AbacetiniLoxandrus velocipes Casey: Thompson (1979a: Figs a-d); Thompson (1979b: 53, 54; Figs 28a-d) [L1, L2-3; RR]; Bousquet (1985c: 200, 203, 207-208; Figs 14, 26) [L1, L2, L3; RR]; Bousquet (1989: 30, 33) [L1, L2, L3; RR].
PterostichiniAbax (Abax) parallelepipedus (Piller and Mitterpacher)†: Schiødte (1872: 179-180, as Pterostichus striola; Figs 3-8 [Pl. 1]) [L1-3; ?]; Larsson (1941: 330, as Abax ater; Fig. 49g) [L1-3; ?]; van Emden (1942: Fig. 99, as Abax ater); Jeannel (1942: Figs 251a-f); Sharova (1958: Figs 50a-e, Abax ater); Sharova (1964: 168, as Abax ater; Figs 108-1, 2, 3, 4, 5) [L1-3; ?]; Larsson (1968: 390, Abax ater; Fig. 53g) [L1-3; ?]; Raynaud (1976d: 171, 172, 174, as Abax ater; Figs 1-7 [Pl. 17B]) [L3; FC]; Bousquet (1985c: 200, 203, 221; Figs 17, 58, 65, 73, 84, 96) [L1, L2, L3; RR]; Bousquet (1989: 30, 33; Figs 33, 38) [L1, L2, L3; RR]; Arndt (1989: 264-268; Figs 16-21) [L1, L2, L3; RR+FC]; Bousquet (1991a: Fig. 34.89); Arndt (1991b: 94; Fig. 14) [L1, L2-3; LIT]; Luff (1993: 104, 115; Figs 379-384) [L1, L2-3; ?]; Makarov (1994: Fig. 72); Bousquet (1999: Fig. 126); Bousquet (2010a: Figs 500, 526, 531).
Cyclotrachelus (Evarthrus) alternans (Casey): Kirk (1972: 1355; Figs 20 [Pl. 2], 3-20) [L1-3; FC]; Bousquet (2010a: Fig. 499).
Cyclotrachelus (Evarthrus) parasodalis (Freitag): Thompson (1979a: Figs 62a-c); Thompson (1979b: 52; Figs 26a-h) [L3; FC].
Cyclotrachelus (Evarthrus) seximpressus (LeConte): Thompson (1979b: 52; Figs 27a-g) [L1, L2-3; RR]; Bousquet (1985c: 200, 202, 222; Figs 18, 32, 66, 69, 74) [L1, L2; RR]; Bousquet (1989: 30, 33) [L1, L2; RR]; Bousquet (1999: Fig. 122).
Cyclotrachelus (Evarthrus) torvus (LeConte): Kirk (1972: 1355; Figs 21 [Pl. 2], 3-21) [L1; FC].
Cyclotrachelus sp.: Bousquet (2010a: Figs 509, 542, 546).
Gastrellarius blanchardi (Horn): Bousquet (1989: 29, 32, 33; Figs 5-7) [L1, L2, L3; RR].
Gastrellarius honestus (Say): Bousquet (1985c: 200, 203, 224-225; Figs 19, 33, 34, 59, 75, 97) [L1, L2, L3; RR]; Bousquet (1989: 30, 33; Figs 15, 34) [L1, L2, L3; RR]; Bousquet (2010a: Figs 510, 533, 549).
Lophoglossus haldemanni (LeConte): Thompson (1979b: 57, 68, 69; Figs 37a-e) [L1, L2; RR].
Lophoglossus scrutator (LeConte): Bousquet (1985c: 200, 204, 211; Figs 15, 27, 28, 63, 71, 88) [L1, L2, L3; RR]; Bousquet (1989: 30, 34; Figs 8, 13, 32) [L1, L2, L3; RR]; Bousquet (1999: Fig. 120); Bousquet (2010a: Figs 505, 506, 539).
Myas (Trigonognatha) cyanescens Dejean: Thompson (1979a: Figs a-b); Thompson (1979b: 54, 55; Figs 29a-g) [L3; FC]; Bousquet (1985c: 203, 219-220; Figs 31, 77, 95) [L3; FC]; Bousquet (1989: 33; Figs 14, 36) [L3; FC]; Makarov (1994: Figs 73, 76); Bousquet (1999: Fig. 125); Bousquet (2010a: Figs 501, 508, 550).
Piesmus submarginatus (Say): Bousquet (1999: 47, 48, 71, 72) [L1, L2, L3; RR].
Poecilus (Poecilus) chalcites (Say): Kirk (1972: 1355; Figs 29 [Pl. 3], 3-29) [L1-3; FC]; Thompson (1979b: 55, 62, 63; Figs 33a-f) [L1, L2-3; RR]; Bousquet (1985c: 201, 204, 213-214; Figs 16, 57, 76, 93, 100) [L1, L2, L3; RR]; Bousquet (1989: 31, 34; Figs 9-10, 30) [L1, L2, L3; RR]; Bousquet (1999: Figs 119, 123); Bousquet (2010a: 441; Figs 532, 547) [L2, L3; RR].
Poecilus (Poecilus) corvus (LeConte): Thompson (1979b: 55, 65, 66; Figs 35a-h) [L1, L2; RR].
Poecilus (Poecilus) lucublandus (Say): Schaupp (1881a: 88, 89; Figs Di-v) [L3; FC]; van Emden (1942: Fig. 23); Larsson (1968: Fig. 54c); Kirk (1972: 1355; Figs 28 [Pl. 3], 3-28) [L1-3; FC]; Thompson (1979b: 55, 74, 75; Figs 41a-g) [L1, L2-3; RR]; Bousquet (1985c: 201, 204, 212-213; Fig. 29) [L1, L2, L3; RR]; Bousquet (1989: 31, 34) [L1, L2, L3; RR]; Bousquet (2010a: 441; Fig. 498) [L2, L3; RR].
Poecilus (Derus) nearcticus (Lindroth): Bousquet (1999: 65) [L2, L3; FC].
Poecilus (Poecilus) sp.: Dogger and Olson (1966: 93, 94, as Micromaseus femoralis; Figs 1-2) [L3; FC].
Pterostichus (Hypherpes) adoxus (Say): Bousquet (1985c: 200, 203, 246; Figs 55, 99) [L1, L2, L3; RR]; Bousquet (1989: 30, 34) [L1, L2, L3; RR]; Bousquet (2010a: 444; Fig. 523) [L2, L3; RR].
Pterostichus (Bothriopterus) adstrictus Eschscholtz: Larsson and Gígja (1959: 37) [L1-3; FC]; Goulet (1974a: 21, 22) [L1, L2, L3; RR]; Thompson (1979b: 56, 59; Figs 30a-c) [L1, L2-3; RR]; Bousquet (1985c: 202, 205, 234) [L1, L2, L3; RR]; Bousquet (1989: 32, 35) [L1, L2, L3; RR]; Bousquet (1991a: Fig. 34.103); Luff (1993: 107, 109) [L1-3; ?]; Bousquet (2010a: 445) [L2, L3; RR].
Pterostichus (Hypherpes) algidus LeConte: Thompson (1979b: 56, 59, 60; Figs 31a-h) [L1, L2-3; RR].
Pterostichus (Hypherpes) amethystinus Mannerheim: Thompson (1979b: 57, 61, 62; Figs 32a-e) [L1, L2-3; RR].
Pterostichus (Cryobius) arcticola (Chaudoir): Bousquet (1985c: 202, 206, 249) [L1, L2, L3; RR]; Bousquet (1989: 32, 37) [L1, L2, L3; RR]; Bousquet (2010a: 447) [L2, L3; RR].
Pterostichus (Cryobius) brevicornis (Kirby): Bousquet (1985c: 202, 203, 248; Fig. 98) [L1, L2, L3; RR]; Bousquet (1989: 32, 33) [L1, L2, L3; RR]; Bousquet (2010a: 444; Fig. 553) [L2, L3; RR].
Pterostichus (Melanius) castor Goulet and Bousquet: Bousquet (1985c: 202, 206, 237) [L1, L2, L3; RR]; Bousquet (1989: 33, 36) [L1, L2, L3; RR]; Bousquet (2010a: 446) [L2, L3; RR].
Pterostichus (Lamenius) caudicalis (Say): Bousquet (1985c: 201, 206, 235; Figs 24, 42, 91) [L1, L2, L3; RR]; Bousquet (1989: 31, 36; Fig. 12) [L1, L2, L3; RR]; Bousquet (2010a: 446; Figs 515, 529) [L2, L3; RR].
Pterostichus (Argutor) commutabilis (Motschulsky): Bousquet (1985c: 202, 206, 225-226, as Pterostichus leconteianus; Fig. 35) [L1, L2, L3; RR]; Bousquet (1989: 32, 36, as Pterostichus leconteianus; Fig. 16) [L1, L2, L3; RR]; Bousquet (2010a: 446, 447; Fig. 511) [L2, L3; RR].
Pterostichus (Euferonia) coracinus (Newman): Bousquet (1985c: 201, 205, 242-243; Figs 62, 81) [L1, L2, L3; RR]; Bousquet (1989: 31, 35; Fig. 20) [L1, L2, L3; RR]; Bousquet (2010a: 446; Fig. 534) [L2, L3; RR].
Pterostichus (Melanius) corvinus (Dejean): Thompson (1979b: 56, 65; Figs 34a-g) [L1, L2-3; RR+FC]; Bousquet (1985c: 202, 206, 236; Figs 43, 44, 86, 92) [L1, L2, L3; RR]; Bousquet (1989: 33, 36; Fig. 28) [L1, L2, L3; RR]; Bousquet (2010a: 446; Figs 516, 530, 538) [L2, L3; RR].
Pterostichus (Monoferonia) diligendus (Chaudoir): Bousquet (1985c: 202, 206, 239; Figs 1-13, 48, 70, 82, 87) [L1, L2, L3; RR]; Bousquet (1989: 32, 36; Figs 21, 29, 31) [L1, L2, L3; RR]; Bousquet (1991a: Figs 34.74, 34.81); Bousquet (2010a: 446; Figs 502, 503, 537, 545, 551) [L2, L3; RR].
Pterostichus (Phonias) femoralis (Kirby): Bousquet (1985c: 201, 204, 227-228) [L1, L2, L3; RR]; Bousquet (1989: 31, 34) [L1, L2, L3; RR]; Bousquet (2010a: 444, 445) [L2, L3; RR].
Pterostichus (Euferonia) lachrymosus (Newman): Thompson (1979b: 56, 71; Figs 38a-g) [L1, L3; RR]; Bousquet (1985c: 201, 205, 241-242; Figs 51, 61) [L1, L2, L3; RR]; Bousquet (1989: 31, 35; Fig. 19) [L1, L2, L3; RR]; Bousquet (2010a: 446; Figs 520, 535) [L2, L3; RR].
Pterostichus (Hypherpes) lama (Ménétriés): Thompson (1979b: 56, 71, 72; Figs 39a-f) [L3; FC].
Pterostichus (Pseudomaseus) luctuosus (Dejean): Thompson (1979b: 56, 74; Figs 40a-g) [L1, L2-3; RR]; Bousquet (1985c: 201, 204, 238; Figs 45-47, 68) [L1, L2, L3; RR]; Bousquet (1989: 31, 34; Fig. 27) [L1, L2, L3; RR]; Bousquet (2010a: 444; Figs 517, 518, 544) [L2, L3; RR].
Pterostichus (Morphnosoma) melanarius (Illiger)†: Schiødte (1867: 511) [L1-3; ?]; Rupertsberger (1872: 573-575) [L3; FC]; van Emden (1936b: Fig. 1, as Pterostichus vulgaris); Larsson (1941: 326-328, as Pterostichus vulgaris; Fig. 49d) [L1-3; ?]; Sharova (1958: 64, as Pterostichus vulgaris; Figs 120aa, 123f) [L1-3; ?]; Sharova (1964: 175, as Pterostichus vulgaris; Figs 150-24, 154-6) [L1-3; ?]; Larsson (1968: 384, 387, as Pterostichus vulgaris; Fig. 53d) [L1-3; ?]; Haberman (1968: 461; Fig. 275.11, 276.9) [L2-3; ?]; Thompson (1979b: 56, 77; Figs 42a-f) [L2-3; FC]; Bousquet (1985c: 201, 205, 241; Figs 50, 80, 85) [L1, L2, L3; RR]; Bousquet (1989: 31, 35) [L1, L2, L3; RR]; Arndt (1991b: 89, 93) [L1, L2-3; ?]; Arndt and Hůrka (1992a: 106; Figs 7, 10) [L1, L2, L3; RR]; Luff (1993: 108, 111; Figs 364-366) [L1, L2-3; ?]; Arndt and Hůrka (1993: 45, 48) [L1, L2-3; ?]; Bousquet (2010a: 445; Figs 519, 525) [L2, L3; RR].
Pterostichus (Eosteropus) moestus (Say): Bousquet (1984b: 1616; Figs 7-12) [L1, L2, L3; RR]; Bousquet (1989: 31, 35, 36; Figs 22, 35) [L1, L2, L3; RR].
Pterostichus (Bothriopterus) mutus (Say): Schaupp (1881a: 89; Figs Ei-v) [L3; FC]; Bousquet (1985c: 201, 204, 232; Figs 22, 40, 78) [L1, L2, L3; RR]; Bousquet (1989: 32, 35) [L1, L2, L3; RR]; Bousquet (2010a: 445) [L2, L3; RR].
Pterostichus (Phonias) patruelis (Dejean): Thompson (1979b: 56, 77, 78; Figs 43a-f) [L1, L2-3; RR]; Bousquet (1985c: 201, 204, 227; Figs 21, 38, 39, 67, 89) [L1, L2, L3; RR]; Bousquet (1989: 31, 34; Figs 11, 25-26) [L1, L2, L3; RR]; Bousquet (1999: Fig. 121); Bousquet (2010a: 444, 445; Figs 513, 514, 528, 543) [L2, L3; RR].
Pterostichus (Bothriopterus) pensylvanicus LeConte: Goulet (1974a: 19) [L1, L2, L3; RR]; Thompson (1979b: 55, 80; Figs 44a-d) [L1, L2-3; RR+FC]; Bousquet (1985c: 202, 205, 233; Figs 23, 41, 79, 90) [L1, L2, L3; RR]; Bousquet (1989: 32, 35; Fig. 24) [L1, L2, L3; RR]; Bousquet (2010a: 445; Fig. 527) [L2, L3; RR].
Pterostichus (Abacidus) permundus (Say): Kirk (1972: 1355; Figs 17 [Pl. 2], 3-17) [L1; FC]; Thompson (1979a: Fig. 61); Thompson (1979b: 49, 50; Figs 25a-g) [L1, L3; RR]; Bousquet (1985c: 200, 240; Figs 25, 49, 60) [L1; RR]; Bousquet (1989: 30; Fig. 18) [L1; RR]; Bousquet (2010a: Fig. 524).
Pterostichus (Cryobius) pinguedineus (Eschscholtz): Bousquet (1985c: 202, 205, 206, 248-249; Figs 56, 83) [L1, L2, L3; RR]; Bousquet (1989: 32, 37) [L1, L2, L3; RR]; Bousquet (2010a: 446, 447; Fig. 504) [L2, L3; RR].
Pterostichus (Lenapterus) punctatissimus (Randall): Thompson (1979b: 56, 80, 81; Figs 45a-i) [L1, L2-3; RR]; Bousquet (1985c: 202, 205, 244; Figs 52, 53) [L1, L2, L3; RR]; Bousquet (1989: 33, 35) [L1, L2, L3; RR]; Bousquet (2010a: 445; Fig. 521) [L2, L3; RR].
Pterostichus (Cylindrocharis) rostratus (Newman): Bousquet (1985c: 202, 206, 245; Fig. 54) [L1, L2, L3; RR]; Bousquet (1989: 32, 36) [L1, L2, L3; RR]; Bousquet (2010a: 446; Fig. 522) [L2, L3; RR].
Pterostichus (Leptoferonia) sphodrinus LeConte: Thompson (1979b: 57, 83, 84; Figs 47a-h) [L1, L2-3; RR].
Pterostichus (Phonias) strenuus (Panzer)†: Larsson (1941: 325, 329; Fig. 48b) [L1-3; ?]; Sharova (1958: 62; Fig. 120s) [L1-3; ?]; Sharova (1964: 173; Fig. 150-16) [L1-3; ?]; Larsson (1968: 383, 388, 389) [L1-3; ?]; Haberman (1968: 460; Fig. 276.2) [L2-3; ?]; Desender and Panné (1983: 139, 141-143; Figs 1, 2a-g, 3a-g, 4a-h) [L1, L2-3; RR]; Bousquet (1985c: 202, 206, 226-227; Figs 20, 36, 37) [L1, L2, L3; RR]; Bousquet (1989: 32, 36; Fig. 17) [L1, L2, L3; RR]; Arndt (1991b: 90, 92; Fig. 85) [L1, L2-3; ?]; Arndt and Hůrka (1992b: 264; Figs 26, 27) [L1, L2-3; RR]; Luff (1993: 108, 113; Figs 373, 374) [L1-3; ?]; Arndt and Hůrka (1993: 46, 48) [L1, L2-3; RR]; Makarov (1996: Figs 27, 53); Bousquet (2010a: 446, 447; Fig. 512) [L2, L3; RR].
Pterostichus (Euferonia) stygicus (Say): Dimmock and Knab (1904: 41-43; Fig. 3; Figs 1, 4 [Pl. 4]); Thompson (1979a: Fig. 60); Bousquet (1985c: 201, 205, 243) [L1, L2, L3; RR]; Bousquet (1989: 31, 35) [L1, L2, L3; RR]; Bousquet (2010a: 446) [L2, L3; RR].
Pterostichus (Eosteropus) superciliosus (Say): Bousquet (1984b: 1616) [L1, L2, L3; RR+FC]; Bousquet (1989: 31, 36; Fig. 23) [L1, L2, L3; RR+FC].
Pterostichus (Pseudomaseus) tenuis (Casey): Bousquet (1985c: 201, 204, 238) [L1, L2, L3; RR]; Bousquet (1989: 31, 34) [L1, L2, L3; RR]; Bousquet (2010a: 444) [L2, L3; RR].
Pterostichus (Bothriopterus) trinarius (Casey, 1918): Bousquet (1989: 27-28, 32, 35, as Pterostichus ohionis; Figs 1-4) [L1, L2, L3; RR].
Pterostichus (Hypherpes) tristis (Dejean): Dimmock and Knab (1904: 46-47, as Pterostichus adoxus; Fig. 6 [Pl.4]); Bousquet (1985c: 200, 203, 246-247) [L1, L2, L3; RR]; Bousquet (1989: 30, 34) [L1, L2, L3; RR]; Bousquet (2010a: 444) [L2, L3; RR].
Pterostichus (Argutor) vernalis (Panzer)†: Larsson (1941: 326, 327; Figs 48d, 49c) [L1-3; ?]; Sharova (1958: 64; Fig. 120ab) [L1-3; ?]; Sharova (1964: 175; Fig. 150-25) [L1-3; ?]; Larsson (1968: 383, 386; Figs 52b, 53c) [L1-3; ?]; Haberman (1968: 460; Fig. 276.10) [L2-3; ?]; Desender and Panné (1983: 139, 141-143; Figs 5a-g, 6a-g, 7a-h, 8) [L1, L2-3; RR]; Arndt (1991b: 90, 93) [L1, L2-3; ?]; Arndt and Hůrka (1992b: 263) [L1, L2-3; RR]; Luff (1993: 108, 109; Figs 357, 358) [L1, L2-3; ?]; Arndt and Hůrka (1993: 46, 48) [L1, L2-3; RR].
Stereocerus haematopus (Dejean): Thompson (1979b: 56, 68; Figs 36a-h) [L1, L2-3; RR+FC]; Bousquet and Goulet (1984: Fig. 58); Bousquet (1985c: 201, 203, 214-219; Figs 30, 64, 72, 94) [L1, L2, L3; RR]; Bousquet (1989: 31, 33; Fig. 37) [L1, L2, L3; RR]; Bousquet (1999: Fig. 124); Bousquet (2010a: Figs 507, 540, 548).
Stomis (Stomis) pumicatus (Panzer)†: Luff (1993: 106; Figs 350-352) [L2-3; RR+FC]; Zetto Brandmayr and Marano (1994: 30-32; Figs 1-6 [Pl. 4], Figs 1-10 [Pl. 5]) [L1, L2, L3; FC].
ZabriniAmara (Amara) aenea (DeGeer)†: Boldori (1935: 150-151; Figs 1, 6) [L1; RR]; Scatizzi Branchini (1938: 216-220; Fig. 1) [L1; RR]; Larsson (1941: 333, 335) [L1-3; ?]; Sharova (1958: 68) [L1-3; ?]; Sharova (1964: 180) [L1-3; ?]; Larsson (1968: 395, 397) [L1-3; ?]; Haberman (1968: 394) [L2-3; ?]; Raynaud (1976b: 24; Figs 17-20 [Pl. 8]) [L3; FC]; Desender (1988: 154, 158; Figs 1-7 [Pl. 1], 1-7 [Pl. 2], 1-7 [Pl. 3]) [L1, L2, L3; RR+FC]; Arndt (1991b: 109) [L1, L2-3; ?]; Luff (1993: 133, 134, 136; Fig. 440) [L1, L2-3; ?]; Saska (2004: 198, 200, 232, 233; Figs 1, 11, 12, 15, 16, 26, 33, 39, 40, 42, 44, 46, 49-51, 57-61, 71, 73, 78, 83-84, 89, 93, 102, 111, 113, 115-117, 124-126, 128) [L1, L2, L3; RR].
Amara (Curtonotus) alpina (Paykull): Forsskåhl (1966: 29, 31; Figs 1-7) [L2-3; FC]; Luff (1993: 133, 143) [L1-3; LIT].
Amara (Amara) anthobia Villa and Villa†: Saska (2004: 200, 202, 232, 233; Figs 18, 27, 52, 62, 91) [L1, L2, L3; RR].
Amara (Bradytus) apricaria (Paykull)†: Schiødte (1867: 530) [L1-3; ?]; Xambeu (1892: 13, 14) [L1-3; FC]; Larsson (1941: 333, 336; Fig. 52i) [L1-3; ?]; Sharova (1958: 69; Figs 54, 58, 126b) [L2-3; ?]; Sharova (1964: 181; Figs 110-2, 112, 158-9) [L1-3; ?]; Larsson (1968: 393, 400; Fig. 57i) [L1-3; ?]; Haberman (1968: 426; Figs 114.4, 114.6, 240.2, 240.10, 240.16) [L2-3; ?]; Hůrka and Ducháč (1980a: 167, 169, 181; Figs 1-19) [L1, L2, L3; RR]; Arndt (1991b: 106, 107; Fig. 103) [L1, L2-3; LIT]; Luff (1993: 132, 141; Figs 448-451) [L1, L2-3; ?].
Amara (Curtonotus) aulica (Panzer)†: Larsson (1941: 333, 334; Fig. 52a) [L1-3; ?]; Sharova (1958: 66; Figs 125, 127c) [L1-3; ?]; Sharova (1964: 178; Figs 157, 158-3) [L1-3; ?]; Larsson (1968: 394, 395; Fig. 57a) [L1-3; ?]; Haberman (1968: 432; Figs 240.1, 240.6) [L2-3; ?]; Bílý (1975: Fig. 12-4); Hůrka and Ducháč (1980b: 259-261, 263, 269, 352; Figs 1-18, 29) [L1, L2, L3; RR]; Arndt (1991b: 108, 109) [L1, L2-3; LIT]; Luff (1993: 133, 142, 143; Figs 456-461) [L1, L2-3; ?]; Zetto Brandmayr et al. (1995: 196) [L1-3; ?]; Makarov and Brinev (2001: Fig. 75).
Amara (Bradytus) avida (Say): Bousquet (2010a: Figs 555, 558, 561, 563).
Amara (Celia) bifrons (Gyllenhal)†: Schiødte (1867: 530, as Amara livida) [L1-3; ?]; Larsson (1941: 333, 336; Fig. 52h) [L1-3; ?]; Sharova (1958: 67; Figs 127e, 129b) [L1-3; ?]; Sharova (1964: 179; Figs 158-5, 159-2) [L1-3; ?]; Larsson (1968: 393, 399; Fig. 57h) [L1-3; ?]; Haberman (1968: 414; Figs 240.8, 240.11) [L2-3; ?]; Bílý (1975: 35-36, 62; Fig. 5) [L2, L3; RR]; Raynaud (1976b: 23, 24; Figs 14-16 [Pl. 8]) [L3; FC]; Thompson (1979b: 87-89; Figs 49a-f) [L2; FC]; Arndt (1991b: 107, 108) [L1, L2-3; LIT]; Luff (1993: 133, 140; Fig. 445) [L1, L2-3; ?]; Makarov (1994: Fig. 54).
Amara (Celia) brunnea (Gyllenhal): Larsson (1941: 332, 336) [L1-3; ?]; Sharova (1958: 67) [L1-3; ?]; Sharova (1964: 179) [L1-3; ?]; Larsson (1968: 394, 399) [L1-3; ?]; Bílý (1975: Fig. 12-3); Arndt (1991b: 106) [L1-3; ?]; Luff (1993: 132, 141; Figs 446, 447) [L1-3; ?]; Makarov (1996: Fig. 30); Makarov and Brinev (2001: Fig. 68).
Amara (Curtonotus) carinata (LeConte): Kirk (1972: 1355; Figs 14 [Pl. 2], 3-14) [L1-3; FC]; Thompson (1979b: 87, 89; Figs 50a-h) [L1, L2-3; RR].
Amara (Amara) communis (Panzer)†: Larsson (1941: 333, 335; Fig. 52c) [L1-3; ?]; Sharova (1958: 68) [L1-3; ?]; Sharova (1964: 180) [L1-3; ?]; Larsson (1968: 394, 396; Fig. 57c) [L1-3; ?]; Haberman (1968: 394) [L2-3; ?]; Arndt (1991b: 109, 110) [L1, L2-3; ?]; Luff (1993: 134, 135; Figs 435, 436) [L1, L2-3; ?]; Hůrka and Růžičková (1999: 452, 457, 461; Figs 9-19) [L1, L2, L3; RR].
Amara (Amara) cupreolata Putzeys: Dogger and Olson (1966: 94, 95; Figs 3-4) [L3; FC].
Amara (Amarocelia) erratica (Duftschmid): Xambeu (1903: 8-9) [L1-3; FC]; Sharova (1958: 68) [L1-3; ?]; Sharova (1964: 180) [L1-3; ?]; Haberman (1968: 414) [L2-3; ?]; Bílý (1971: 89-93; Figs 1-15) [L1, L2, L3; RR]; Bílý (1975: 48-49, 60, 62; Figs 11, 12-1) [L1, L2, L3; RR]; Arndt (1991b: 107, 108, 110) [L1, L2-3; LIT]; Luff (1993: 134, 140) [L1-3; LIT]; Zetto Brandmayr et al. (1995: 196) [L1-3; ?].
Amara (Amara) eurynota (Panzer)†: Larsson (1941: 334; Fig. 52b) [L1-3; ?]; Sharova (1958: 68; Fig. 129d) [L1-3; ?]; Sharova (1964: 179; Fig. 159-4) [L1-3; ?]; Larsson (1968: 393, 394, 396; Fig. 57b) [L1-3; ?]; Haberman (1968: 394; Fig. 240.12) [L2-3; ?]; Bílý (1972: 324-328; Figs 1-19) [L1, L2, L3; RR]; Raynaud (1976b: 22, 23; Figs 7-13 [Pl. 8]) [L3; FC]; Arndt (1991b: 109, 110; Figs 102, 144) [L1, L2-3; ?]; Luff (1993: 133, 136) [L1, L2-3; LIT].
Amara (Bradytus) exarata Dejean: Kirk (1972: 1355; Figs 12 [Pl. 1], 3-12) [L1-3; FC]; Thompson (1979b: 87, 91; Figs 51a-f) [L1, L2-3; RR+FC].
Amara (Amara) familiaris (Duftschmid)†: Schiødte (1867: 531) [L1-3; ?]; Beling (1877: 45-47) [L1-3; ?]; Larsson (1941: 333, 335; Fig. 52e) [L1-3; ?]; Sharova (1958: 67; Fig. 129a) [L1-3; ?]; Sharova (1964: 178; Fig. 159-1) [L1-3; ?]; Larsson (1968: 393, 397; Fig. 57e) [L1-3; ?]; Haberman (1968: 394; Fig. 240.14) [L2-3; ?]; Desender (1988: 159; Figs 1-7 [Pl. 4], 1-7 [Pl. 5], 1-7 [Pl. 6]) [L1, L2, L3; RR+FC]; Arndt (1991b: 109; Fig. 104) [L1, L2-3; ?]; Luff (1993: 132, 137; Fig. 441) [L1, L2-3; ?]; Saska (2004: 202, 204, 232, 233; Figs 5, 17, 35, 63, 72, 90, 103, 122, 129, 137) [L1, L2, L3; RR].
Amara (Amarocelia) farcta LeConte: Kirk (1972: 1355; Figs 13 [Pl. 2], 3-13) [L2-3; FC]; Thompson (1979b: 87, 91, 92; Figs 52a-f) [L1, L2-3; RR].
Amara (Bradytus) fulva (Müller)†: Boldori (1935: 151; Figs 3, 5) [L1; RR]; Larsson (1941: 333, 336) [L1-3; ?]; van Emden (1942: Fig. 10); Sharova (1958: 70; Fig. 130) [L1-3; ?]; Sharova (1964: 180; Fig. 160) [L2-3; ?]; Larsson (1968: 393, 400, 401) [L2-3; ?]; Haberman (1968: 426; Fig. 240.3) [L2-3; ?]; Raynaud (1976b: 20, 22; Figs 1-6 [Pl. 8]) [L3; FC]; Hůrka and Ducháč (1980a: 173, 174, 176, 181; Figs 20-33, 670) [L1, L2, L3; RR]; Arndt (1991b: 106, 107) [L1, L2-3; LIT]; Luff (1993: 132, 141, 142; Figs 452, 453) [L1, L2-3; ?].
Amara (Amara) impuncticollis (Say): Thompson (1979a: Figs 66a-c); Thompson (1979b: 87, 93, 94; Figs 53a-c) [L3; RR].
Amara (Amara) littoralis Dejean: Thompson (1979b: 87, 94; Figs 54a-f) [L1, L2; RR].
Amara (Amara) lunicollis Schiødte: Larsson (1941: 333, 335; Figs 52d, 53) [L1-3; ?]; Sharova (1958: 66; Fig. 128) [L1-3; ?]; Sharova (1964: 178; Fig. 158-7) [L1-3; ?]; Larsson (1968: 394, 396, 397; Figs 57d, 58) [L1-3; ?]; Haberman (1968: 394; Fig. 240.9) [L2-3; ?]; Luff (1993: 132, 135; Figs 437-439) [L1-3; ?]; Saska (2004: 206, 208, 232, 233; Figs 2, 19, 32, 34, 48, 55, 64, 76, 79, 104, 114, 119, 130, 135) [L1, L2, L3; RR].
Amara (Celia) musculis (Say): Bousquet (2010a: Figs 557, 560, 565).
Amara (Percosia) obesa (Say): Riley et al. (1878: 291, 292; Figs 25a, d-g) [L2-3; FC]; Lugger (1899: Figs 20a, d-g); Kirk (1972: 1355; Figs 11 [Pl. 1], 3-11) [L1-3; FC]; ?Bousquet (2010a: Figs 556, 559, 564).
Amara (Amara) ovata (Fabricius)†: Arndt (1991b: 110) [L2-3; ?]; Makarov and Brinev (2001: Fig. 74).
Amara (Paracelia) quenseli (Schönherr): Sharova (1958: 67; Fig. 127d) [L1-3; ?]; Larsson and Gígja (1959: 42, 43) [L1-3; FC]; Sharova (1964: 179; Fig. 158-4) [L1-3; ?]; Larsson (1968: 393, 399) [L1-3; ?]; Haberman (1968: 414; Fig. 240.7) [L2-3; ?]; Bílý (1975: 51-54, 60, 61; Figs 8-10) [L1, L2, L3; RR]; Arndt (1991b: 106) [L1-3; ?]; Luff (1993: 132, 139; Figs 46, 443, 444) [L1, L2-3; ?]; Zetto Brandmayr et al. (1995: 193) [L1-3; ?].
Amara (Curtonotus) sp.: Bousquet (2010a: Figs 554, 562).
OodiniLachnocrepis parallela (Say): Bousquet (2010a: Figs 566, 569).
Oodes americanus Dejean: Thompson (1979a: Figs 75a-c).
Oodes fluvialis LeConte: Bousquet (2010a: Figs 567, 568).
Oodes sp.: Chu (1945: 29, 30; Figs 6, 75, 79) [L2-3; FC].
ChlaeniiniChlaenius (Lithochlaenius) cordicollis Kirby: Schaupp (1880b: 26; Figs Bi-viii, as Chlaenius leucoscelis) [L3; FC]; Bousquet and Goulet (1984: Figs 34, 41); Bousquet (2010a: Fig. 574).
Chlaenius (Chlaeniellus) impunctifrons Say: Claassen (1919: 97-99, Figs 3, 4, 7 [Pl. 6]) [L1, L2, L3; RR].
Chlaenius (Chlaenius) laticollis Say: Schaupp (1880a: 17-18) [L2-3; FC].
Chlaenius (Agostenus) niger Randall: Makarova (2001: 302-305) [L2; RR].
Chlaenius (Chlaeniellus) pennsylvanicus Say: Chu (1945: 21, 24, 25; Figs 5, 8, 14, 17, 18) [L3; FC].
Chlaenius (Chlaenius) platyderus Chaudoir: Kirk (1972: 1355; Figs 8 [Pl. 1], 3-8) [L2-3; FC].
Chlaenius (Lithochlaenius) prasinus Dejean: Thompson and Allen (1974: 196-199; Figs 4a-c) [L3; FC]; Thompson (1979a: Fig. 74).
Chlaenius (Chlaenius) sericeus (Forster): Wickham (1895b: 763, 764) [L3; FC]; Boldori (1940: 285) [L1-3; LIT]; ?van Emden (1942: Figs 62, 79); Chu (1945: 22, 28, 29; Figs 2, 15, 16, 47) [L2-3; FC]; Kirk (1972: 1355; Figs 9 [Pl. 1], 3-9) [L2-3; FC]; Bousquet (2010a: Fig. 571).
Chlaenius (Eurydactylus) tomentosus (Say): Kirk (1972: 1355; Figs 10 [Pl. 1], 3-10) [L1-3; FC]; Thompson (1979a: Figs 73a-d); Bousquet (2010a: Figs 570, 575).
Chlaenius (Chlaeniellus) tricolor Dejean: Chu (1945: 21, 25, 26; Figs 4, 11, 13) [L3; FC]; Bousquet (2010a: Figs 572, 573, 576).
LicininiBadister sp.: Bousquet (2010a: Figs 579-581, 583, 585).
Dicaelus (Paradicaelus) dilatatus Say: Schaupp (1878a: 3; Fig. 1) [L3; FC]; Schaupp (1879: 14) [L3; LIT].
Dicaelus (Paradicaelus) elongatus Bonelli: Schaupp (1878b: 43) [L3; FC]; Schaupp (1879: 14) [L3; LIT].
Dicaelus (Paradicaelus) politus Dejean: Schaupp (1878c: 44) [L3; FC]; Bousquet (2010a: Figs 577, 582, 584).
Dicaelus (Dicaelus) purpuratus Bonelli: Dimmock and Knab (1904: 21-26; Figs 1-2 [Pl. 1], Figs 1-10 [Pl. 2]) [L3; FC].
Dicaelus (Dicaelus) purpuratus splendidus Say: Wickham (1893a: 195-197; Figs 1, 1c-e) [L3; FC].
Dicaelus (Paradicaelus) sculptilis Say: Kirk (1972: 1355; Figs 19 [Pl. 2], 3-19) [L2-3; FC]; Thompson (1979a: Figs a-c).
Dicaelus sp.: Horn (1878a: 37-39; Figs 5a-e [Pl. II]) [L2-3; FC]; Schaupp (1879: 14) [L3; LIT]; Beutel (1992c: Figs 9, 10).
Diplocheila (Isorembus) impressicollis (Dejean): van Emden (1942: Fig. 15, as Rembus laticollis); ?Bousquet (2010a: 465) [L2, L3; FC].
Diplocheila (Isorembus) obtusa (LeConte): Bousquet (2010a: 465; Figs 578, 586) [L3; FC].
Diplocheila (Isorembus) striatopunctata (LeConte): Bousquet and Goulet (1984: Fig. 33); Bousquet (1991a: Fig. 34.83); Makarov (1994: Fig. 62); Bousquet (2010a: 465) [L2, L3; FC].
HarpaliniAcupalpus (Acupalpus) meridianus (Linnaeus)†: Larsson (1941: 350; Figs 60d, 62c) [L1-3; ?]; Sharova (1958: Figs 133f, 134b); Sharova (1964; Figs 161-6, 162-1); Larsson (1968: 420; Figs 66d, 68c) [L1-3; ?]; Haberman (1968: Figs 206.15, 207.2); Arndt (1991a: 50; Fig. 5.9) [L2; RR]; Arndt (1991b: 121; Fig. 120) [L1-3; ?]; Luff (1993: 159; Fig. 510) [L1-3; RR].
Agonoleptus conjunctus (Say): Chu (1945: 22, 49, 50; Figs 54, 63, 64) [L3; FC]; Bousquet (2010a: Figs 596, 621).
Amphasia (Amphasia) interstitialis (Say): van Emden (1942: Fig. 12); Chu (1945: 46-47; Figs 36, 67, 68) [L2-3; FC]; Bousquet and Tchang (1992: Figs 4, 13, 21); Bousquet (2010a: Fig. 599).
Anisodactylus (Anisodactylus) binotatus (Fabricius)†: Rupertsberger (1872: 576) [L3; FC]; Xambeu (1911: 70, 71) [L1-3; FC]; Bøving (1911: 163-169, 172-178; Figs 6, 12, 17, 21 [Pl. 6]) [L2-3; FC]; Larsson (1941: 342; Fig. 57a) [L1-3; ?]; Jeannel (1942: Figs 217a-h); Sharova (1958: 76; Fig. 133n) [L1-3; ?]; Sharova (1964: 187; Fig. 161-12) [L1-3; ?]; Larsson (1968: 414, 415; Fig. 65a) [L1-3; ?]; Haberman (1968: Fig. 206.19); Arndt (1991b: 112) [L1, L2-3; ?]; Luff (1993: 152, 153; Figs 493-496) [L1, L2-3; ?].
Anisodactylus (Anisodactylus) californicus Dejean: Chu (1945: 22, 44; Figs 39, 65, 66) [L2-3; FC].
Anadaptus (Anadaptus) discoideus Dejean: Dogger and Olson (1966: 95, 96; Figs 5-6) [L3; FC].
Anisodactylus (Anisodactylus) harrisii LeConte: Bousquet and Tchang (1992: Figs 23, 30).
Anisodactylus (Anisodactylus) kirbyi Lindroth: Bousquet and Tchang (1992: Figs 16, 40); Bousquet (2010a: Figs 602, 619).
Anisodactylus (Gynandrotarsus) merula (Germar): Bousquet and Tchang (1992: Figs 9, 24); Bousquet (2010a: Figs 592, 612, 618).
Anisodactylus (Anisodactylus) nigrita Dejean: Bousquet and Goulet (1984: Fig. 32); Bousquet (1991a: Fig. 34.82); Bousquet and Tchang (1992: Fig. 7).
Anisodactylus (Gynandrotarsus) rusticus (Say): Bousquet and Tchang (1992: Figs 18, 27, 39); Bousquet (2010a: Figs 604, 606).
Anisodactylus (Anadaptus) sanctaecrucis (Fabricius): Kirk (1976: 354; Fig. 1) [L1-3; RR]; Thompson (1979a: Figs 69a-c); Bousquet and Tchang (1992: Figs 8, 17, 33, 35); Bousquet (2010a: Figs 593, 603, 622).
Anisodactylus (Spongopus) verticalis (LeConte): Bousquet and Tchang (1992: Figs 6, 15, 34); Bousquet (2010a: Figs 601, 624).
Anisodactylus (or close) sp.: Chu (1945: 22, 45, 46; Figs 40, 69, 70) [L2-3; FC].
Bradycellus (Bradycellus) harpalinus (Audinet-Serville)†: van Emden (1945: 27) [L1-3; ?]; ?Larsson (1968: 418) [L1-3; ?]; Luff (1993: 156, 157; Figs 507, 508) [L1, L2-3; FC].
Bradycellus (Stenocellus) rupestris (Say): Chu (1945: 48, 49; Figs 56, 58, 60) [L3; FC]; Matalin (1996: 290) [L1-3; LIT]; Hůrka (1997: 197) [L1-3; LIT]; Matalin (2001: 317) [L1-3; LIT].
Cratacanthus dubius (Palisot de Beauvois): van Emden (1942: Fig. 77); Chu (1945: 22, 32, 33; Figs 37, 48, 50) [L3; FC]; Chu (1945: 22, 33, 34, as Cratacanthus sp.; Figs 38, 43, 45, 46) [L2-3; FC]; Thompson (1979a: Figs a-f).
Dicheirotrichus (Trichocellus) cognatus (Gyllenhal): Larsson (1941: 347, 348; Fig. 61b) [L1-3; ?]; Larsson and Gígja (1959: 46) [L1-3; FC]; Larsson (1968: 417; Fig. 67b) [L1-3; ?]; Böcher (1988: Fig. 5); Luff (1993: 154, 155; Figs 503, 504) [L1, L2-3; ?]; Matalin (2001: 314, 320; Figs 15, 22, 30, 31, 51, 59, 60) [L2; RR].
Euryderus grossus (Say): van Emden (1942: 73, as Euryderus zabroides) [L3; FC]; Chu (1945: 30, 31, as Nothopus zabroides; Figs 35, 41, 42) [L3; FC].
Geopinus incrassatus (Dejean): Kirk (1972: 1354, 1355; Figs 6 [Pl. 1], 3-6) [L1-3; FC]; Bousquet and Tchang (1992: Figs 10, 11, 19, 26, 36, 41); Bousquet (2010a: Figs 594, 605, 610, 620, 623).
Harpalus (Harpalus) affinis (Schrank)†: Schiødte (1867: 531-535, as Harpalus aeneus; Figs 1-3 [Pl. 22]) [L1-3; ?]; Larsson (1941: 344, 345, as Harpalus aeneus; Figs 58b, 59b) [L1-3; ?]; Chu (1945: 23, 36, 37, as Harpalus viridiaeneus; Figs 25, 80, 83) [L3; FC]; Sharova (1958: Figs 133q, 134a, as Harpalus aeneus); Sharova (1964: 187, as Harpalus aeneus; Figs 163-1, 164-1) [L1-3; ?]; Sharova (1967: Figs 8.6, as Harpalus aeneus); Larsson (1968: 408, 410, 411, as Harpalus aeneus; Figs 63b, 64b) [L1-3; ?]; Haberman (1968: Fig. 206.4, 207.5, as Harpalus aeneus); Arndt (1991b: 115, as Harpalus aeneus; Figs 107, 145) [L1-3; ?]; Putchkov (1992: 73) [L1; ?]; Luff (1993: 145, 149; Figs 481-483) [L1, L2-3; ?].
Harpalus (Megapangus) caliginosus (Fabricius): ?Chu (1945: 23, 34, 35; Figs 26, 89, 90) [L2-3; FC]; Kirk (1972: 1354; Figs 1 [Pl. 1], 3-1) [L2-3; FC]; Thompson (1979a: Figs 71a-e).
Harpalus (Pseudoophonus) compar LeConte: Chu (1945: 24, 40, 41; Figs 22, 73, 74) [F3; FC].
Harpalus (Plectralidus) erraticus Say: Kirk (1972: 1354; Figs 4 [Pl. 1], 1, 3-4) [L1-3; FC].
Harpalus (Pseudoophonus) erythropus Dejean: Chu (1945: 23, 39, 40; Figs 23, 27, 77, 78) [L3; FC].
Harpalus (Harpalus) herbivagus Say: Lugger (1899: Figs 21a-c); Kirk (1972: 1354; Figs 2 [Pl. 1], 2, 3-2) [L1-3; FC].
Harpalus (Opadius) laevipes Zetterstedt: Sharova (1958: Fig. 51, as Harpalus quadripunctatus); Sharova (1964: 188, 189, as Harpalus quadripunctatus; Figs 109, 163-7, 164-7) [L1-3; ?]; Sharova (1967: Fig. 8.3, as Harpalus quadripunctatus); Larsson (1968: 407, 412, as Harpalus quadripunctatus) [L1-3; ?]; Haberman (1968: Figs 203, 206.8, 207.10, as Harpalus quadripunctatus); Arndt (1991b: 116, as Harpalus quadripunctatus) [L1-3; ?]; Luff (1993: 145, 150, 151, as Harpalus quadripunctatus) [L1-3; ?].
Harpalus (Pseudoophonus) pensylvanicus (DeGeer): Quaintance and Jenne (1912: Figs 32b-d); Chu (1945: 23, 41, 42; Figs 21, 29-31, 33, 84) [L3; FC]; Kirk (1972: 1354; Figs 3 [Pl. 1], 3-3) [L1-3; FC]; Bousquet (2010a: Figs 588, 611).
Harpalus (Harpalus) rubripes (Duftschmid)†: Xambeu (1894: 119-121) [L1-3; FC]; Larsson (1941: 344-346; Figs 58d, 59d) [L1-3; ?]; Sharova (1958: Figs 133s, 134g); Sharova (1964: 189; Figs 163-8, 164-8) [L1-3; ?]; Sharova (1967: Figs 1.4, 2.4); Larsson (1968: 407, 412; Fig. 64d) [L1-3; ?]; Haberman (1968: Figs 206.9, 207.11); Arndt (1991b: 116; Figs 108, 113) [L1-3; ?]; Luff (1993: 145, 151; Figs 490, 491) [L1, L2-3; ?].
Harpalus (Pseudoophonus) rufipes (DeGeer)†: Schiødte (1867: 535, as Harpalus ruficornis; Figs 4-11 [Pl. 22]) [L1-3; ?]; Znojko (1929: 351, 352, as Harpalus pubescens; Figs 19, 20a-b, 21a-e, 22, 23, 24) [L1-3; ?]; Larsson (1941: 343, as Pseudophonus pubescens; Figs 58a, 59a) [L1-3; ?]; van Emden (1942: Fig. 78); van Emden (1945: 27; Figs 19, 26) [L1-3; ?]; Sharova (1958: 74; Figs 52, 133p) [L1-3; ?]; Sharova (1964: 186; Figs 110-1, 161-14) [L1-3; ?]; Sharova (1967: 892; Figs 1.2, 2.3, 8.1) [L1-3; ?]; Larsson (1968: 406, 409, 410, as Harpalus pubescens; Figs 60, 63a, 64a) [L1-3; ?]; Brandmayr et al. (1980; Fig. 1d); Brandmayr and Zetto Brandmayr (1982: Fig. 2); Arndt (1991b: Figs 115, 116); Luff (1993: 145, 147; Figs 476-480) [L1, L2-3; ?]; Makarov (1994: Fig. 97).
Harpalus (Harpalus) solitaris Dejean: Hůrka and Papoušek (2002: 112; Figs 54-61) [L3; RR].
Harpalus (Pseudoophonus) vagans LeConte: Chu (1945: 24, 37, 38; Figs 20, 49, 85, 86) [L3; FC].
Harpalus sp. [probably Harpalus herbivagus]: Riley et al. (1878: 290; Figs 24a-c) [L2-3; FC].
Harpalus sp. [might be a young instar of Harpalus caliginosus]: Chu (1945: 23, 35, 36; Figs 19, 28, 76) [L1-3; FC].
Notiobia (Anisotarsus) nitidipennis (LeConte): Bousquet and Tchang (1992: 756; Figs 2, 12, 20, 25, 29, 31) [L1, L2-3; RR]; Bousquet (2010a: 469; Figs 590, 598, 608, 613) [L2, L3; RR].
Notiobia (Anisotarsus) sayi (Blatchley): van Emden (1942: Fig. 11); Chu (1945: 47-48; Figs 32, 44, 51, 71, 72) [L2-3; FC].
Notiobia (Anisotarsus) terminata (Say): Thompson (1979a: 68a-b); Bousquet and Tchang (1992: 756; Figs 1, 3, 28) [L1, L2-3; RR]; Bousquet (2010a: 469; Fig. 607) [L2, L3; RR].
Ophonus (Metaphonus) puncticeps Stephens†: Larsson (1941: 340, 341, as Ophonus angusticollis; Figs 55c, 56c) [L1-3; ?]; Larsson (1968: 406, 409, as Harpalus angusticollis; Figs 61c, 62c) [L1-3; ?]; Brandmayr and Zetto Brandmayr (1982: 87, 89, 98; Figs 69-75, 102, 103, 115) [L1, L3; RR]; Arndt (1991b: 117) [L1-3; LIT]; Luff (1993: 145, 146; Figs 472, 473) [L1, L2-3; ?]; Bousquet (2010a: Figs 587, 597, 609, 616).
Ophonus (Metaphonus) rufibarbis (Fabricius)†: Larsson (1941: 340, 341, as Ophonus seladon; Figs 55d, 56d) [L1-3; ?]; Larsson (1968: 406, 409, as Harpalus seladon; Figs 61d, 62d) [L1-3; ?]; ?Brandmayr and Zetto Brandmayr (1982: 95, 98; Figs 91-95, 106) [L3; FC]; Arndt (1991b: 118) [L1-3; LIT].
Polpochila (Phymatocephalus) capitata (Chaudoir): Chu (1945: 53, 54; Figs 34, 57, 59) [L2-3; FC].
Stenolophus (Agonoderus) comma (Fabricius): ?Chu (1945: 24, 52, 53, as Agonoderus pallipes; Figs 53, 87, 88) [L2-3; FC]; Kirk (1972: 1354; Figs 5 [Pl. 1], 3-5) [L1-3; FC]; Bousquet (2010a: Figs 595, 615).
Stenolophus (Stenolophus) dissimilis Dejean: Thompson (1979a: a-b).
Stenolophus (Agonoderus) lecontei (Chaudoir): Bryson and Dillon (1941: 50; Fig. 2, as Agonoderus pallipes Fabricius) [L3; FC].
Stenolophus (Agonoderus) lineola (Fabricius): van Emden (1942: Fig. 61); Chu (1945: 24, 51, 52; Figs 52, 91, 92) [L2-3; FC].
Stenolophus (Stenolophus) sp.: Chu (1945: 22, 50, 51) [L2-3; FC]; Sharova (1958: Fig. 133e); Sharova (1964; Fig. 161-5).
Trichotichnus (Trichotichnus) dichrous (Dejean): Chu (1945: 21, 43; Figs 24, 81, 82) [L3; FC].
Trichotichnus (Trichotichnus) vulpeculus (Say): Bousquet (2010a: Figs 589, 614).
Xestonotus lugubris (Dejean): Bousquet and Tchang (1992: Figs 5, 14, 22, 32, 37, 38); Bousquet (2010a: Figs 591, 600, 617).
SphodriniCalathus (Acalathus) advena (LeConte): Lindroth (1956b: Fig. 35d, 36-Id, 36-IId, 36-IIId, 36-Ivd). Bousquet (2010a: Figs 627, 639).
Calathus (Calathus) fuscipes (Goeze)†: Bøving (1910: 335-341; Figs 5, 9, 13) [L2-3; FC]; Raynaud (1940b: 83, 84, 86, 87) [L1, L2, L3; FC]; Larsson (1941: 317; Fig. 41b) [L1-3; ?]; van Emden (1942: Fig. 47); Raynaud (1944: 89) [L3; FC]; van Emden (1945: Fig. 24); Sharova (1958: 59, 60; Fig. 120b) [L1-3; ?]; Sharova (1964: 171; Fig. 152-1) [L1-3; ?]; Larsson (1968: 370; Fig. 44b) [L1-3; ?]; Haberman (1968: 481; Fig. 297.1) [L2-3; ?]; Kůrka (1971: 247, 248, 260, 261; Figs 1-4, 6-7, 8a, 9, 11a-e, 13a-c, 14a) [L1, L2, L3; RR]; Arndt (1991b: 104; Fig. 97) [L1, L2-3; LIT]; Hovorka (1991: Fig. 18); Luff (1993: 117, 118; Figs 385-388) [L1, L2-3; ?].
Calathus (Neocalathus) gregarius (Say): Thompson (1979b: 35-37; Figs 17a-d) [L1, L2-3; RR+FC]; Bousquet (2010a: 482) [L2; RR].
Calathus (Neocalathus) ingratus Dejean: Thompson (1979a: Figs 65a-b); Thompson (1979b: 35, 39; Figs 18a-i) [L1, L2-3; RR+FC]; Bousquet (2010a: 482) [L2; RR].
Calathus (Neocalathus) opaculus LeConte: Thompson (1979b: 35, 40, 41; Figs 19a-c) [L3; FC]; Bousquet (2010a: 482) [L3; LIT].
Laemostenus (Laemostenus) complanatus (Dejean)†: van Emden (1945: 24) [L1-3; ?]; Thompson (1979b: 46, 47) [L2-3; FC]; Vereshchagina and Makarov (1986: 372) [L1-3; ?]; Casale (1988: 90) [L1-3; ?]; Arndt (1991b: 103) [L1-3; ?]; Luff (1993: 121) [L1-3; ?].
Laemostenus (Pristonychus) terricola terricola (Herbst)†: Chapuis and Candèze (1853: 376-378; Fig. 3 [Pl. 1]) [L1-3; FC]; Böving and Craighead (1931; Figs f-h [Pl. 4]); Larsson (1941: 315; Fig. 41a) [L1-3; ?]; van Emden (1945: 24; Figs 17, 18) [L1-3; ?]; Sharova (1958: 57; Fig. 120a) [L1-3; ?]; Sharova (1964: 167; Fig. 150-1) [L1-3; ?]; Larsson (1968: 368; Fig. 44a) [L1-3; ?]; Haberman (1968: 445; Fig. 306.1) [L2-3; ?]; Raynaud (1976c: 256, 257, as Pristonichus terricola subcyanescens; Figs 1-4) [L3; RR]; Vereshchagina and Makarov (1986: 372) [L1-3; ?]; Casale (1988: 90; Fig. 114) [L1-3; ?]; Arndt (1991b: 103) [L1-3; ?]; Hovorka (1991: 102, 103; Figs 8-12) [L1, L2, L3; RR]; Luff (1993: 120; Figs 399-401) [L1, L2-3; ?]; Bousquet (2010a: Figs 629, 638).
Synuchus impunctatus (Say): Lindroth (1956b: 564; Figs 35b, 36-Ib, 36-IIbg, 36-IIIb, 36-IVb) [L1; RR]; Thompson (1979a: Figs 64a-d); Thompson (1979b: 48; Figs 24a-f) [L1, L2-3; RR+FC]; Bousquet (2010a: Figs 626, 637).
PlatyniniAgonum (Europhilus) anchomenoides Randall: Thompson (1979b: 7, 10; Figs 1a-e) [L3; FC].
Agonum (Europhilus) consimile Gyllenhal: Lindroth (1955c: Fig. 1b).
Agonum (Olisares) cupripenne (Say): Bousquet (2010a: Fig. 634).
Agonum (Olisares) errans (Say): Bousquet (2010a: Figs 635, 640).
Agonum (Europhilus) exaratum (Mannerheim): Makarov (1996: Fig. 29).
Agonum (Olisares) extensicolle (Say): Schaupp (1881b: 91-92; Figs Ai-v) [L3; FC]; Thompson (1979b: 9, 11, 12; Figs 2a-e) [L3; RR].
Agonum (Europhilus) gratiosum (Mannerheim): Thompson (1979b: 7, 16; Figs 4a-g) [L1, L3; RR+FC]; Bousquet (2010a: Fig. 631).
Agonum (Olisares) melanarium Dejean: Thompson (1979b: 8, 18; Figs 5a-d) [L3; FC]; Bousquet (2010a: Fig. 633).
Agonum (Agonum) muelleri (Herbst)†: Raynaud (1940b: 77-79) [L3; FC]; Larsson (1941: 321; Figs 43b, 44a) [L1-3; ?]; van Emden (1942: Fig. 52); Sharova (1958: 61; Fig. 120k) [L1-3; ?]; Sharova (1964: 172; Fig. 150-8) [L1-3; ?]; Larsson (1968: 376, 378, 379; Figs 47f, 48a-b) [L1-3; ?]; Haberman (1968: 501; Fig. 306.8) [L2-3; ?]; Arndt (1991b: 98, 100; Figs 1, 6-11, 16, 92, 96) [L1; ?]; Luff (1993: 124, 128, 130; Figs 417-419) [L1, L2-3; ?]; Makarov (1996: Fig. 23).
Agonum (Platynomicrus) nigriceps LeConte: Bousquet (2010a: Fig. 630).
Agonum (Olisares) octopunctatum (Fabricius): Thompson (1979b: 8, 21; Figs 7a-e) [L2-3; RR].
Agonum (Olisares) pallipes (Fabricius): Thompson (1979b: 8, 21, 22; Figs 8a-b) [L3; FC].
Agonum (Agonum) placidum (Say): Kirk (1972: 1355; Figs 15 [Pl. 2], 3-15) [L1-3; FC].
Agonum (Olisares) propinquum (Gemminger and Harold): Thompson (1979b: 8, 24, 25; Figs 10a-g) [L1, L2-3; RR+FC].
Agonum (Olisares) punctiforme (Say): Thompson and Allen (1974: 192-195; Figs 3a-d) [L3; FC]; Thompson (1979a: Figs a-b); Thompson (1979b: 8, 26, 27; Figs 11a-e) [L3; FC].
Agonum (Europhilus) retractum LeConte: Thompson (1979b: 7, 29; Figs 12a-f) [L1, L2-3; RR+FC].
Agonum (Olisares) striatopunctatum Dejean: Thompson (1979a: Figs a-d); Thompson (1979b: 8, 31; Figs 13a-e) [L2; RR].
Agonum (Olisares) sulcipenne (Horn): Thompson (1979b: 8, 31, 32; Figs 14a-f) [L2-3; FC].
Agonum (Olisares) tenue (LeConte): Thompson (1979b: 8, 34; Figs 15a-h) [L1, L2-3; RR].
Agonum (Europhilus) thoreyi Dejean: Larsson (1941: 323; Figs 45c, 46d-e) [L1-3; ?]; Lindroth (1955c: Fig. 1a); Sharova (1958: 60; Fig. 120g) [L1-3; ?]; Sharova (1964: 172; Fig. 150-4) [L1-3; ?]; Larsson (1968: 376, 380; Figs 49c, 50d-e) [L1-3; ?]; Haberman (1968: 525; Fig. 306.4) [L2-3; ?]; Thompson (1979b: 7, 34, 35; Figs 16a-f) [L1, L2; RR+FC]; Arndt (1991b: 100; Fig. 94) [L2-3; ?]; Luff (1993: 123, 126; Figs 411, 412) [L1, L2-3; ?].
Anchomenus (Anchomenus) funebris (LeConte): Thompson (1979b: 9, 13, 14; Figs 3a-f) [L1, L2; RR]; Liebherr (1991b: 128-130; Figs 317-327) [L1; RR].
Metacolpodes buchanani (Hope): Liebherr (2000: Fig. 3g).
Olisthopus parmatus (Say): Bousquet (2010a: 483; Fig. 625) [L1; RR].
Oxypselaphus pusillus (LeConte): Bousquet (2010a: Fig. 628).
Paranchus albipes (Fabricius)†: Xambeu (1898: 55-57, as Anchomenus pallipes) [L1-3; ?FC]; Kemner (1913: 21, 22, as Platynus ruficornis; Figs 12, 3-4 [Pl. 2]) [L3; FC]; Raynaud (1940b: 76-78, as Platynus ruficornis) [L3; FC]; Larsson (1941: 319, 320, as Platynus ruficornis; Fig. 42b) [L1-3; ?]; Sharova (1958: 61, as Agonum ruficorne; Fig. 120o) [L1-3; ?]; Sharova (1964: 172, 173, as Agonum ruficorne; Fig. 150-12) [L1-3; ?]; Larsson (1968: 376, 377, as Agonum ruficorne; Fig. 47b) [L1-3; ?]; Haberman (1968: 517, as Agonum ruficorne; Fig. 306.12) [L2-3; ?]; Arndt (1991b: 98, 99, as Platynus ruficornis) [L1, L2-3; ?]; Luff (1993: 125, 130, 131; Figs 422-424) [L1, L2-3; ?].
Platynus (Platynus) decentis (Say): Thompson (1979a: Figs a-d); Thompson (1979b: 41, 42; Figs 20a-j) [L1, L2-3; RR+FC]; Bousquet (2010a: Figs 632, 636, 641).
Platynus (Batenus) hypolithos (Say): Thompson (1979b: 41, 44; Figs 21a-i) [L3; FC].
Platynus (Batenus) mannerheimii (Dejean): ?Saalas (1917: 286; Figs 1-2) [L2-3; FC]; ?Saalas (1923: 667) [L2-3; FC]; Thompson (1979b: 41, 45, 46; Figs 22a-f) [L2-3; FC].
Sericoda bembidioides Kirby: Liebherr (1991b: 130, 132; Figs 328-338) [L1; RR].
Sericoda obsoleta (Say): Thompson (1979b: 8, 18, 19; Figs 6a-f) [L1, L2-3; RR].
Tanystoma maculicolle (Dejean): van Emden (1945: 24) [L1-3; ?]; Liebherr (1984a: 531, 532, 534, 536; Figs 1, 2a-g, 3a-i) [L1, L2, L3; RR].
Tetraleucus picticornis (Newman): Thompson (1979b: 8, 24; Figs 9a-f) [L1, L2-3; RR]; Liebherr (1991b: 125; Figs 293-304) [L1; RR].
AtraniniAtranus pubescens (Dejean): Bousquet (1985b: 330-331; Figs 1-6) [L1, L2, L3; FC]; Makarov (1994: Fig. 81); Bousquet (2010a: Figs 642-646).
LachnophoriniCalybe (Ega) sallei (Chevrolat): Liebherr (1983: 255; Figs 1-7) [L2, L3; FC].
OdacanthiniColliuris (Cosnania) pensylvanica (Linnaeus): Thompson (1979a: Figs 77a-c); Bousquet (2010a: 491; Figs 647-650) [L1, L2-3; FC].
CtenodactyliniLeptotrachelus dorsalis (Fabricius): Erwin and White (2012: 22, 24, 25, 27, 28 ; Figs 3-12) [L1, 3; RR].
LebiiniApristus subsulcatus (Dejean): Bousquet (2010a: 496-498; Figs 660, 669, 671, 673) [L1, L2, L3; RR+FC].
Axinopalpus biplagiatus (Dejean): Bousquet (2010a: 496; Fig. 659) [L2-3; FC].
Calleida (Calleida) decora (Fabricius): Thompson (1979a: Figs 78a-d); Zhou and Goyer (1993: Fig. 3a).
Calleida (Calleida) punctata LeConte: Bousquet and Goulet (1984: Fig. 36); Bousquet (1991a: Fig. 34.84); Zhou and Goyer (1993: Fig. 3b); Bousquet (2010a: Figs 663, 664, 676).
Calleida (Calleida) viridipennis (Say): Zhou and Goyer (1993: 234-236; Figs 1a-d) [L1, L2, L3; RR].
Cymindis (Tarulus) vaporariorum (Linnaeus): Hůrka (1969: 105-107; Figs 3a-m) [L1, L2, L3; FC]; Hůrka (1986: 47; Figs 85-99) [L1, L2, L3; RR]; Hůrka (1986: 48, as Cymindis vaporariorum immaculata; Figs 100-103) [L1, L2; RR]; Arndt (1991b: 133) [L1, L1, L2-3; LIT]; Luff (1993: 176, 177; Figs 581, 582) [L1, L2-3; ?]; Makarov (1996: Fig. 31).
Cymindis sp.: Bousquet and Goulet (1984: Figs 35, 46, 54, 59); Bousquet (1991a: Fig. 34.104); Bousquet (2010a: Figs 665, 666, 677, 678).
Dromius (Dromius) piceus Dejean: Mahar et al. (1983: 24-26; Figs 1-7) [L1, L2, L3; FC].
Lebia (Lebia) analis Dejean: Thompson (1979a: Figs 80a-b).
Lebia (Loxopeza) grandis Hentz: Chaboussou (1939: 414-419; Figs 18-20, 22-34) [L1, L2; RR].
Lebia (Lebia) viridis Say: Capogreco (1989a: 187-191; Figs 2-5, 7-16) [L1, L2; RR]; Makarov (1994: Figs 123, 125); Bousquet (2010a: Figs 656-658).
Microlestes sp.: Bousquet (2010a: 499; Figs 662, 670, 672, 674) [L2-3; FC].
Mochtherus tetraspilotus (Macleay)†: Gardner (1936: 193, 194; Figs 47-50) [L3; FC]; van Emden (1942: 78) [L3; FC].
Onota floridana Horn: van Emden (1942: 79) [L2; FC].
Philophuga viridicollis (LeConte): Larson (1969: 26; Figs 9, 11) [L3; FC].
Philophuga viridis amoena (LeConte): Mahar et al. (1983: Fig. 9).
Philorhizus melanocephalus (Dejean)†: Xambeu (1904: 88, 89) [L1-3; FC]; Larsson (1941: 357, 358; Fig. 67f) [L1-3; ?]; Larsson (1968: 429, 431; Fig. 73f) [L1-3; ?]; Luff (1993: 169, 172, 173; Figs 564-568) [L1, L2-3; FC].
Plochionus (Plochionus) pallens (Fabricius): van Emden (1945: 25) [L1-3; ?]; Arndt (1991b: Fig. 143).
Plochionus (Menidius) timidus Haldeman: Duffey (1892: 538; Figs a-e [Pl. 10]) [L1, L3; RR]; Larson (1969: 25; Figs 7, 8) [L2-3; FC]; Thompson (1979a: Figs 79a-b); Zhou and Goyer (1993: 237-240; Figs 2a-d) [L1, L2, L3; RR].
Somotrichus unifasciatus (Dejean): van Emden (1945: 27) [L1-3; ?].
Syntomus americanus (Dejean): Bousquet (2010a: Figs 661, 667, 668, 675).
Tecnophilus croceicollis croceicollis (Ménétriés): Larson (1969: 24; Figs 6, 10, 12) [L1-3; RR]; Mahar et al. (1983: Fig. 8).
Tecnophilus croceicollis peigani Larson: Larson (1969: 24; Figs 13, 14) [L1-3; RR].
GaleritiniGalerita (Progaleritina) janus (Fabricius): Packard (1871: 29, 30; Figs 13a-b) [L3; FC]; Hubbard (1875: 49-51) [L3; FC]; Schaupp (1879: 14) [L2-3; LIT]; Kirk (1981: 368; Fig. 1) [L2-3; FC]; Arndt and Drechsel (1998: Fig. 6); Bousquet (2010a: Figs 679-681).
Galerita (Progaleritina) lecontei Dejean: Sallé (1849: 298-299; Figs 2c-d [Pl. 8]) [L3; FC]; Chapuis and Candèze (1853: 367-369) [L3; FC]; Schaupp (1879: 14) [L2-3; LIT].
Galerita sp. (Arkansas): Thompson (1979a: Figs 82a-c).
HelluoniniHelluomorphoides praeustus bicolor (Harris): Bousquet (1987c: 922, 925-927; Figs 1-7) [L1, L2; FC]; Bousquet (2010a: Figs 682-686).
Helluomorphoides sp. (Georgia): Thompson (1979a: Figs 83a-c).
PseudomorphiniPseudomorpha (Pseudomorpha) augustata Horn: Liebherr and Kavanaugh (1985: 1081) [L1; RR].
Pseudomorpha (Pseudomorpha) hubbardi Notman: Liebherr and Kavanaugh (1985: 1081; Figs 1a-b) [L1; RR].
Pseudomorpha sp. [Arizona]: Erwin (1981a: 56, 58; Fig. 24) [?L2; FC].
Pseudomorpha sp. [Nevada]: Thompson (1979a: Figs 84a-f); Erwin (1981a: 54-56; Figs 1-23) [L1, L2-3; FC].
A number of available species were listed originally as from North America and were subsequently found not to belong to the region. The following is an annotated alphabetical list of species described from specimens mislabeled or incorrectly assumed to be from North America.
Agonum splendidulum Motschulsky, 1844: 138. Type locality listed as “Sitka et du Kamtchatka.” Chaudoir (1850b: 123) stated that the specimens he identified as Agonum splendidulum from Kamchatka seemed to represent a very minor variety of Agonum impressum (Panzer). Agonum splendidulum is currently considered a junior synonym of Agonum impressum, a Palaearctic species (Bousquet 2003c: 450). The original description is very short and uninterpretable (see Lindroth 1966: 594). According to Keleinikova (1976: 218), three syntypes from Kamchatka are present in ZMMU.
Aretharea helluonis Say, 1830a: 68. Type locality listed as “Pennsylv[ania]” although Say (1830a: 68) added “I am not sure whether I obtained it on our journey to the Rocky Mountains, to St. Peter’s river, or in Pennsylvania, but I think the latter.” LeConte (1859e: 524) wrote the following about this species: “No insect has yet been seen by other entomologists, which at all approaches the description here given ... In fact, there is nothing in the description of the head, thorax and anterior legs, which forbids a reference to Cryptobium bicolor or Cryptobium melanocephalum [Staphylinidae]. With regard to the elytra and posterior legs, I must suppose them to have been derived from some foreign Carabide, and that the union was effected unintentionally, before the specimen came under Say’s notice.”
Bembidium glabriusculum Motschulsky, 1844: 272. Type locality given as “Sitka [Baranof Island, Alaska].” Maddison (1993: 176) remarked that the holotype of this species is conspecific with members of Bembidion argenteolum Ahrens, a species found only west of the Lena River and Ussuri River areas of Russia. The original locality given by Motschulsky (1844: 272) is in error.
Calleida rubricollis Dejean, 1825: 225. Type locality given as “Amérique septentrionale.” Dejean (1831: 328) acknowledged receiving specimens of this species from Cuba, including some from Klug under the name elegans, which did not differ from his specimens of Calleida rubricollis. Chaudoir (1844: 469) described Calleida elegans (which he credited to Klug) by pointing out structural differences between his sole specimen and those of Calleida rubricollis. Later Chaudoir (1873b: 144) concluded that the differences reported in 1844 between the specimens from the United States and Cuba were only “accidental” and that all the specimens belonged to the same species. I have seen a specimen from Chaudoir’s collection in MHNP labeled “rubricollis mihi elegans [illegible letters] in Ins. Cuba D. Klug [handwritten] / Lectotype ♂ Calleida rubricollis Dej. by Erwin ‘76 [partly handwritten].” The specimen is probably one received from Klug and noticed in 1831 by Dejean and therefore is likely not a syntype. Based on the original description by Dejean (1825: 225) and this specimen, I believe that Calleida rubricollis is structurally most similar to Calleida decora (Fabricius) among the North American species but specifically distinct. I have not seen a conspecific specimen from North America and believe the type locality listed by Dejean (i.e., Amérique septentrionale) is in error. The statement by Chaudoir (1873b: 144) that the specimens from United States were identical with those from Cuba may be based on a comparison of his Cuban specimens with those of Dejean, which he believed came from the United States. At the time, Chaudoir owned Dejean’s carabid collection. Already in his first catalogue, LeConte (1863b: 6) stated that “Calleida rubricollis Dej. is not found within the present limits of the United States, but is confined to Cuba.”
Carabus beauvoisi Dejean, 1826: 67. Type locality listed as “Amérique septentrionale.” Motschulsky (1850a: 81) listed this name as a “var.?” of Carabus californicus, a species he described only in 1866 [see next entry]. LeConte (1863b: 3) reported that this species is identical to “Carabus catenulatus Fabricius.” Motschulsky (1866: 287, as Carabus bauvoisii Dej.) stated that Haldeman gave him specimen(s) of this species during his trip to America. Kraatz (1878: 158) agreed with LeConte and stated that Dejean’s specimen(s) was conspecific with Carabus catenulatus and that the locality given was in error. Lapouge (1931: 570) considered this name to be a questionable junior synonym of Carabus taedatus agassii LeConte. Csiki (1927: 81) listed it as a variety of Carabus problematicus Herbst, and Breuning (1933b: 810) as a junior synonym of Carabus problematicus problematicus natio gallicus Géhin. There is no indication that either Csiki or Breuning saw the name-bearing type specimen(s) and therefore, if their conclusions are correct, the type locality was in error. Jeannel (1941b: 132) listed also the name as a junior synonym of Carabus problematicus, adding that the type locality of “Amér. du Nord” is in error. In the latest catalogue of the genus Carabus (Deuve 2004: 240), this name is listed as a junior synonym of Carabus problematicus harcyniae Sturm. Dejean’s original specimens came from Palisot de Beauvois’ collection which, according to Dejean (1825: 101), was not in order.
Carabus bifasciatus Olivier, 1790b: 347. Type locality listed as “Amérique septentrionale.” This species was transferred to the genus Calophaena by Klug (1821: 297) which is confirmed by the illustration provided by Olivier (1795: Fig. 80 [plate 7]). The genus is not found in North America, north of Mexico, and the type locality is obviously in error.
Carabus californicus Motschulsky, 1866: 288. This name was listed as a variety of the Palaearctic Carabus problematicus Herbst by Csiki (1927: 81), as a junior synonym of Carabus problematicus problematicus natio gallicus Géhin by Breuning (1933b: 810), and as a junior synonym of Carabus problematicus gallicus by Kryzhanovskij (1968: 175). In the latest catalogue of the genus Carabus (Deuve 2004: 240), this name is listed as a questionable junior synonym of Carabus problematicus solutus Oberthür. Obviously the provenance, implied by the specific name, is incorrect.
Carabus carolinus Fabricius, 1792: 126. Type locality given as “Carolina meridionali.” LeConte (1863b: 3) reported that Fabricius’ taxon was identical to Carabus splendens Olivier. The name is still listed in synonymy with Carabus splendens in Deuve’s (2004) latest catalogue of the genus Carabus. Since this species is found only in southwestern France, the provenance given by Fabricius is obviously incorrect.
Carabus latus Linnaeus, 1758: 415. Type locality listed as “Europa; America septentrionali.” Lindroth (1957b: 332) discussed the status of this species and came to the conclusion that the name should be retained for Harpalus latus auctorum, one of the commonest species in Sweden. However, none of the six “Linnean” specimens in LSL belong to this species. Motschulsky (1855a: 44) interpreted Linnaeus’ species as the North American Harpalus herbivagus Say.
Casnonia rufipes Dejean, 1825: 172. Type locality listed as “Amérique septentrionale.” The holotype came from Palisot de Beauvois’ collection. Chaudoir (1863: 278) indicated that the species is not found in North America but occurs from Cayenne (in French Guiana) to Brazil.
Chlaenius nimrod Horn, 1897: 372. Type locality given as “Custer, South Dakota.” Bell (1960: 116) stated that the original specimens were obviously mislabeled and probably belonged to an Old World species.
Cicindela baltimorensis Herbst, 1806: 181. Type locality given as “Baltimore.” Schaum (see LeConte 1863b: 1) reported that this taxon is the East Indian Cylindera minuta (Olivier) with an erroneous locality. Acciavatti and Pearson (1989: 291) found syntypes of Cicindela baltimorensis in ZMHB and confirmed Schaum’s identification.
Cicindela californica californica Ménétriés, 1843: 52. Type locality listed as “Californie.” The nominotypical subspecies of Habroscelimorpha californica is known from the coast of central and southern Baja California (Pearson et al. 2006: 138) suggesting that the type locality is in error. However, all the other species described from California by Ménétriés (1843), except maybe Agaosoma californicum, are currently found in California. Two other possibilities are conceivable. The subspecies had a wider range in the xix Century and did occur in California or our concept of Habroscelimorpha californica californica is in error and the name actually applies to one of the two subspecies of Habroscelimorpha californica found in California. As far as I know, nobody has reported studying syntype(s) of the nominotypical subspecies.
Cicindela coerulea Herbst, 1806: 182. Type locality given as “Nordamerica.” LeConte (1863b: 2) listed this name as a synonym of the Palaearctic Cylindera germanica (Linnaeus). The type locality is obviously in error.
Cicindela magdalenae LeConte, 1873b: 321. Type locality listed as “found in turpentine barrels brought to London, supposed to be from North Carolina.” Horn (1880f: xix) noted the similarity between the elytral markings of LeConte’s species and those of some South African species and later (1883b: 269-270) reported that the species was identical to Lophyra senegalensis (Dejean).
Cicindela tortuosa Dejean, 1825: 87. Type locality: “Amérique septentrionale et dans les Antilles.” Gistel (1837: 49) listed this name in synonymy with Cicindela trifasciata Fabricius. LeConte’s (1851: 172) record of this species in “Georgia et Louisiana, in oryzaceis” refers to Cicindela trifasciata ascendens LeConte (Horn 1926: 294). Currently Cicindela tortuosa is listed in catalogues (e.g., Wiesner 1992: 146; Lorenz 2005: 49) as a junior synonym of Cicindela trifasciata trifasciata Fabricius, which occurs commonly over the West Indies. However, Dejean’s type series likely includes specimens of Cicindela trifasciata trifasciata and Cicindela trifasciata ascendens LeConte. As far as I know, a lectotype has not been designated for Cicindela tortuosa Dejean.
Cicindela triguttata Herbst, 1806: 182. Type locality given as “Nordamerica.” LeConte (1863b: 2) stated that Herbst’s species was “not North American.” Acciavatti and Pearson (1989: 253), upon an examination of the syntypes in ZMHB, concluded that Cicindela triguttata Herbst is a synonym of Cylindera viduata (Fabricius), a species found in Asia. The type locality originally reported is in error.
Clivina biguttata Putzeys, 1867b: 157. Type locality given as “Cuba. Louisiane.” Nichols (1988a: 129) reported this species from Cuba, Isla de Pinos, and Jamaica. As far as I know, the species has not been reported subsequently from North America.
Clivina picipes Bonelli, 1813: 481. Bonelli originally stated that the country of origin was unknown. Dejean (1825: 416) reported that his specimen(s) of this species originated from “Amérique” and Putzeys (1846: 105) redescribed the sole specimen in Dejean’s collection and listed “Amérique boréale” as the provenance. Subsequently Putzeys (1867b: 110) listed this name as a junior synonym of Clivina attenuata (Herbst) and pointed out that the species was from India.
Curtonotus transversicollis Putzeys, 1866b: 236. Type locality given as “Amér[ique] Russe. (Akina).” According to Lindroth (1968: 665), the lectotype is conspecific with members of the Palaearctic Amara brevicollis (Chaudoir) and probably originated from Akima in Transbaikalia.
Cymindis morio Dejean, 1825: 219. Type locality listed as “Amérique septentrionale.” The specimen(s) was given to Dejean by Palisot de Beauvois. Schaum (1857a: 293) stated that this species belongs to the genus Apenes LeConte and occurs in Haiti. LeConte (1863b: 6) said that it “does not occur in the United States.” According to Ball (1992b: Fig. 5.6), the species is found in the Bahamas and on Hispaniola.
Dinodes rotundicollis Dejean, 1826: 373. Type locality given as “Amérique septentrionale.” The specimen(s) was given to Dejean by Palisot de Beauvois. According to LeConte (1863b: 11), this species “is probably not North American.” Chaudoir (1876b: 58), who had access to Dejean’s types, stated that Dinodes rotundicollis “n’est qu’un individu de l’azureus [= Chlaenius decipiens (Dufour)]” and that the locality originally given was in error.
Dromius geminatus Haldeman, 1843b: 298. Type locality indicated as “south-eastern Pennsylvania” by Haldeman (1843a: 296). LeConte (1863b: 5) stated that this species is identical to Dromius quadrinotatus (Panzer) [= Calodromius spilotus (Illiger)] of Europe, suggesting that the type locality given was in error.
Feronia quadricollis LeConte, 1846b: 343. Type locality given as “Pennsylvania.” LeConte (1853a: 235) stated that his species is identical to Abax parallelus Duftschmid of Europe, suggesting that the type locality was in error.
Lebia angulata Boheman, 1858: 7. Type locality given as “California.” As pointed out by Horn (1872a: 141), the California record is unreliable and the specimen(s) probably originated from South America (see also LeConte 1869b: 248). Chaudoir (1871a: 113) transferred the species to the genus Rhombodera (= Pentagonica). Reichardt (1968: 154) listed Lebia angulata as a questionable synonym of Pentagonica picea Chaudoir. Lebia goniodera Gemminger and Harold, 1868 is a replacement name for Lebia angulata Boheman, 1858.
Lebia cupripennis Boheman, 1858: 7. Type locality given as “California.” LeConte (1869b: 248) stated that Lebia cupripennis Boheman is identical to Lebia cupripennis Chaudoir and that Boheman’s specimens did not originate from California but from Peru and Chile. Lebia chalcoptera Gemminger and Harold, 1868 is a replacement name for Lebia cupripennis Boheman, 1858.
Myosodus femoratus Motschulsky, 1845a: 22. Type locality given as “Sitka.” Motschulsky (1850a: 47) listed this species from “Sitka? Irkutsk?” Tschitschérine (1902b: 105) suspected this taxon to be a form of Pterostichus kamtschaticus (Motschulsky). Csiki (1930: 681) listed the name as a valid species in section Lyperopherus Motschulsky and reported it from Siberia and Alaska. Lindroth (1966: 526) believed the type locality was “almost certainly false.” Finally, Kryzhanovskij et al. (1995: 105), followed by Bousquet (2003d: 497), listed the name as a questionable junior synonym of Pterostichus costatus (Ménétriés). According to Keleinikova (1976: 197), there is one “corruptum” syntype in Motschulsky’s collection (ZMMU). Pterostichus cruralis (Tschitschérine, 1902) is a replacement name for Pterostichus femoratus (Motschulsky, 1845).
Nebria macrocephala Motschulsky, 1844: 128. Type locality listed as “probablement de Sitka ou d’Ounalachka [Alaska].” Mannerheim (1853: 111), who apparently saw syntypes of this taxon, noted that they were conspecific with specimens of Nebria stigmula Dejean, 1826 (= Nebria hellwigii Panzer, 1803). Farkač and Janata (2003: 95) listed this form as a valid species of the subgenus Tetungonebria Shilenkov from “Siberia.” The provenance given by Motschulsky is likely incorrect.
Oodes schaumi Chaudoir, 1882: 346. Type locality given as “trouvée probablement dans la Louisiane.” The holotype in ZMHB belongs to the genus Coptocarpus Chaudoir (Bousquet 1996a: 471) which is endemic to the Australian Region. The provenance originally given is obviously in error.
Pogonus minutus Dejean, 1828: 15. Type locality listed as “Amérique septentrionale.” The sole specimen came from Palisot de Beauvois’ collection. Chaudoir (1872a: 27), who owned Dejean’s collection at the time, stated that the type of this species was conspecific with that of Pogonus senegalensis Dejean [= Pogonus minutus Dejean] from western Africa. The locality as originally given is in error.
Scarites attenuatus Herbst, 1806: 255. Type locality given as “Nordamerica.” Putzeys (1867b: 110) stated that this species, which belongs to the genus Clivina Latreille, occurs in India and it is by error that Herbst listed it from North America.
Scarites emarginatus Herbst, 1806: 255. Type locality listed as “Nordamerica.” Bänninger (1933: 116) studied the “type” of Herbst’s species in ZMHB and concluded that it was conspecific with Scarites dentatus Andrewes of India. The locality as originally given is in error.
Scarites gagates Bonelli, 1813: 475. Type locality given as “Amérique septentrionale.” Bänninger (1938: 112) stated that Bonelli’s specimen(s) is conspecific with those of Scarites indus Olivier of the Oriental Region and that the provenance originally given was obviously incorrect.
Scarites polyphemus Herbst, 1806: 254. Type locality given as “Nordamerica.” Dejean (1825: 372) listed Herbst’s name in synonymy with a new species, Scarites herbstii Dejean, from Cape of Good Hope although Dejean’s description indicates that he considered the two as distinct species. Bänninger (1938: 176), who saw Herbst’s type specimen(s), listed Scarites polyphemus as a synonym of Scarites herbstii Dejean and noted that the species does not occur in North America but in South Africa.
Selenophorus beauvoisii Dejean, 1829: 98. Type locality listed as “Amérique septentrionale.” Dejean’s specimen(s) came from Palisot de Beauvois’ collection. LeConte (1870: 403) saw the name-bearing type(s) in Paris and reported that the species was “abundant in the Antilles, but does not occur in the United States.” In Blackwelder (1944: 49) the species is listed as valid and from Jamaica, with Selenophorus aeneocupreus Dejean as a junior synonym.
Steropus rugulosus Motschulsky, 1845b: 342. Type locality given as “Ounalaschka.” Motschulsky’s original description was based on a single specimen in ZMMU. Csiki (1930: 658) listed the taxon as a valid species in the subgenus Cryobius and reported it from Alaska. Based on a study of the holotype, Bousquet and Larochelle (1993: 333) concluded that it belongs to an undetermined species of Abacetus Dejean. The provenance originally given by Motschulsky is obviously incorrect. Pterostichus rugatinus Csiki, 1930 is a replacement name for Pterostichus rugulosus (Motschulsky, 1845).
Tachypus elongatus Motschulsky, 1850a: 16. Type locality given as “Sitka?” According to Lindroth (1963b: 204), this Asaphidion is “almost certainly not from N. America.” A syntype of Motschulsky’s species is present in ZMMU (Keleinikova 1976: 196) and a study of the specimen is needed to determine its identity.
Trechus ruficollis Van Dyke, 1926a: 66. Type locality given as “Lawrence, Massachusetts.” Barr (1962a: 76-77) stated that the four original specimens of this species were likely mislabeled and Donabauer (2010b: 45) pointed out that they are conspecific with members of Trechus striatulus Putzeys, a species endemic to the western Carpathians. Because of homonymy, Van Dyke’s name was first changed to Trechus vandykei Jeannel, 1928 (also a homonym) and subsequently to Trechus dietrichi Barr, 1962. The holotype (♂) is in CUIC [# 1635].
Many species- or genus-group names have been reported from North America but are unavailable (nomina nuda) because they were either not described [DES], or proposed as infrasubspecific entities [INF], or first proposed as junior synonyms and not treated as valid names or senior homonyms before 1961 [SYN], or proposed as generic entities after 1930 without type species designations [TYP], or not published [PUB]. All these names have no taxonomic status and are not listed in the catalogue itself. This list does not pretend to be complete.
Acupalpus minuatus Rathvon, 1869: 527. DES
Agonothorax anchomenoides Motschulsky, 1850a: 68. DES
Agonum aeneicolle Sturm, 1843: 23. DES
Agonum coracinum Sturm, 1843: 22. DES
Agonum formosum Sturm, 1826: 89. DES
Agonum foveolatum Sturm, 1826: 89. DES
Agonum rotundicolle Sturm, 1843: 23. DES
Agonum saltuum Sturm, 1826: 89. DES
Agonum sulcatum T.W. Harris [in Scudder], 1869: 230. SYN
Amara aerea Sturm, 1843: 29. DES
Amara cylindrica Motschulsky, 1850a: 60. DES
Amara elongatula Motschulsky, 1850a: 60. DES
Amara fulvipes Sturm, 1843: 29. DES
Amara misera Dejean, 1828: 462. SYN
Amara ovalis Sturm, 1826: 91. DES
Amara quadrifoveolata Motschulsky, 1850a: 61. DES
Anchomenus aeruginosus Sturm, 1826: 91. DES
Anchomenus decorus ab. syracusensis Hatch, 1926: 247. INF
Anchomenus laticollis Sturm, 1826: 91. DES
Anchomenus proximus T.W. Harris, 1828d: 132. SYN
Anchomenus viridanus Dejean, 1821: 10. DES
Angoleus picipes Sturm, 1843: 24. DES
Anisodactylus brevis Sturm, 1843: 32. DES
Argutor ornatus Sturm, 1826: 97. DES
Argutor rufipes Dejean, 1821: 11. DES
Argutor rufitarsis Sturm, 1843: 24. DES
Bembidium apicale Sturm, 1843: 36. DES
Bembidium axillare Sturm, 1843: 37. DES
Bembidium bisbiguttatum Sturm, 1843: 38. DES
Bembidion browni Lindroth, 1954b: 158. DES
Bembidium elegans Sturm, 1843: 36. DES
Bembidium maeklini LeConte, 1863b: 14. DES
Bembidion planiusculum ab. pallidum Hatch, 1950: 99. INF
Bembidium unistriatum Sturm, 1843: 36. DES
Brachinus glabripennis LeConte, 1858a: 28. DES
Brachinus puncticollis LeConte, 1858a: 28. DES
Brachinus similis Sturm, 1843: 2. DES
Brachystylus megas LeConte, 1853a: 250. DES
Bradytus planiusculus Sturm, 1843: 29. DES
Calathus flavipes Sturm, 1843: 21. DES
Calosoma luxatum zimmermanni ab. striata Breuning, 1928b: 86. INF + DES
Carabus catena Gosse, 1840: 107, 150, 185, 307. DES
Carabus fimbriatus Sturm, 1826: 110. DES
Carabus maeander monstr. excatenatus Kraatz, 1880: 337. INF
Celia compacta Motschulsky, 1850a: 59. DES
Chlaenius aeratus Dejean, 1821: 8. DES
Chlaenius angusticollis Dejean, 1821: 8. DES
Chlaenius aureliensis LaFerté-Sénectère, 1851: 248. DES
Chlaenius dimidiatus Motschulsky, 1858: 157. DES
Chlaenius discolor Strum, 1826: 115. DES
Chlaenius exaratus LaFerté-Sénectère, 1851: 249. DES
Chlaenius feisthamelii LaFerté-Sénectère, 1851: 248. DES
Chlaenius indutus LaFerté-Sénectère, 1851: 247. DES
Chlaenius lugubris Motschulsky, 1850a: 64. DES
Chlaenius nigrocoeruleus Sturm, 1843: 18. DES
Chlaenius nigrovirens Sturm, 1843: 18. DES
Chlaenius opacus Sturm, 1843: 18. DES
Chlaenius thalassinus Sturm, 1843: 18. DES
Chlaenius viridiceps LaFerté-Sénectère, 1851: 248. DES
Cicindela carthagena haemorrhagica var. nigroides Hatch, 1938: 236. INF
Cicindela collaris LaFerté-Sénectère, 1841a: 40, 41, 42. DES
Cicindela continua Sturm, 1843: 2. DES
Cicindela deflexa Sturm, 1843: 2. DES
Cicindela deflua Sturm, 1843: 2. DES
Cicindela dejeanii Dejean, 1831: 215. SYN
Cicindela deleta Sturm, 1843: 2. DES
Cicindela erythrogaster T.W. Harris [in Scudder], 1869: 20. DES
Cicindela excisa Sturm, 1843: 2. DES
Cicindela formosa mescalero Gaumer, 1977: 225. PUB
Cicindela formosa yampa Gaumer, 1977: 219. PUB
Cicindela hamata Sturm, 1843: 2. DES
Cicindela humeralis Dejean, 1831: 215. SYN
Cicindela hydropica Dejean, 1836: 3. SYN
Cicindela latecincta Gould, 1834: 42. SYN
Cicindela lemnisticta Smyth, 1908: 188. SYN
Cicindela maculosa Sturm, 1843: 2. DES
Cicindela purpurea ab. nigerrimoides Hatch, 1938: 233. INF
Cicindela raiana Frost, 1920: 229. DES
Cicindela scripta Sturm, 1843: 2. DES
Cicindela thalassina Dejean, 1821: 1. DES
Clivina brunnipes Sturm, 1843: 2. DES
Clivina trisulcata Sturm, 1843: 2. DES
Clivina unicolor Sturm, 1843: 2. DES
Cratacanthus pallidus Sturm, 1843: 30. DES
Dicaelus ambiguus Dejean, 1836: 31. DES
Dicaelus lecontei Dejean, 1836: 30. DES
Dicaelus quadratus Dejean, 1836: 31. DES
Dicaelus tristis Sturm, 1843: 19. DES
Dromius auratus Sturm, 1826: 134. DES
Dromius collaris Sturm, 1826: 134. DES
Dromius festinans Dejean, 1821: 3. DES
Dromius picipes Sturm, 1826: 134. DES
Dyschirius californicus Motschulsky, 1850a: 18. DES
Emphanes flavopictus Motschulsky, 1850a: 13. DES
Galerita elegans Chaudoir, 1861b: 553. SYN
Harpalus brevis Sturm, 1826: 148. DES
Harpalus brunnipennis Dejean, 1821: 15. DES
Harpalus cuprescens Sturm, 1843: 33. DES
Harpalus laevigatus Sturm, 1843: 33. DES
Harpalus moestus Dejean, 1821: 14. DES
Hirmoplataphus Netolitzky, 1942: 46. TYP
Lebia albomarginata Sturm, 1826: 161. DES
Lebia marginella Sturm, 1826: 161. DES
Limodromus parallelus Motschulsky, 1850a: 70. DES
Lorocera flavipes Motschulsky, 1850a: 66. DES
Loxopeza testacea LeConte, 1880b: 164. DES
Nebria fusiformis Van Dyke, 1926b: 11. DES
Nebria strawberriensis Kavanaugh, 1978: 413. PUB
Neonebria Hatch, 1939a: 117. TYP
Notiophilus intermedius Lindroth, 1954b: 157. DES
Notiophilus plumbeus Sturm, 1843: 17. DES
Ochthedromus compar LeConte, 1857a: 5. DES
Olisthopus dorsalis Sturm, 1843: 23. DES
Omaseus cephalotes Sturm, 1843: 24. DES
Omaseus punctatostriatus Sturm, 1843: 24. DES
Omaseus sulcatus Sturm, 1843: 24. DES
Pangus melanarius Sturm, 1843: 31. DES
Pasimachus laterisulcatus Sturm, 1826: 182. DES
Pasimachus obtusus Sturm, 1826: 182. DES
Pasimachus sulcatus Dejean, 1821: 4. DES
Platynus dixianus Zimmermann [in LeConte], 1869b: 246. DES
Platysma alternata Motschulsky, 1850a: 53. DES
Platysma breviuscula Motschulsky, 1850a: 54. DES
Platysma punctatosulcata Sturm, 1826: 185. DES
Poecilus albionicus Motschulsky, 1850a: 53. DES
Poecilus cupreonitens Sturm, 1843: 23. DES
Poecilus similis Dejean, 1821: 11. DES
Pogonus californicus Motschulsky, 1850a: 6. DES
Pseudomaseus dilutus Motschulsky, 1850a: 51. DES
Pterostichus maeklini LeConte, 1863b: 9. DES
Pterostichus muticus LeConte, 1863b: 8. DES
Rhombodera pallipes LeConte, 1863b: 6. DES
Scarites laevistriatus Sturm, 1843: 2. DES
Scarites picicornis Sturm, 1843: 2. DES
Selenophorus pedicularius var. semiopacus Snow, 1903: 194. DES
Stenolophus americanus Dejean, 1821: 15. DES
Stenolophus marginellus Dejean, 1821: 15. DES
Stenolophus palliatus Motschulsky, 1850a: 21. DES
Steropus picipes Sturm, 1843: 25. DES
Steropus politus Sturm, 1843: 25. DES
Tachys carolinus Schwarz, 1878: 438. DES
Tachys columbiensis Schwarz, 1878: 438. DES
Tachys truncorum Crotch, 1874a 21. DES
Tennessarius Valentine, 1952: 15. TYP
Trechus laevigatus LeConte, 1863b: 14. DES
New taxon
Randallius Bousquet, new subgenus. Type species: Chlaenius purpuricollis Randall, 1838.
New replacement name
Pterostichus amadeus Bousquet for Pterostichus vexatus Bousquet, 1985.
New synonymies
Agonum canadense Goulet, 1969 = Agonum elongatulum Haldeman, 1843.
Anisodactylus lecontei Chaudoir, 1868 = Anisodactylus nigrita Dejean, 1829.
Axinopalpus coloradensis Casey, 1920 = Axinopalpus biplagiatus (Dejean, 1825).
Axinopalpus demissus Casey, 1920 = Axinopalpus biplagiatus (Dejean, 1825).
Axinopalpus habilis Casey, 1920 = Axinopalpus biplagiatus (Dejean, 1825).
Badister laticeps Blatchley, 1910 = Badister flavipes flavipes LeConte, 1853.
Bembidion arizonae Lindroth, 1963 = Bembidion durangoense Bates, 1891.
Bembidium lapponicum C.G. Thomson, 1857 = Bembidion lapponicum Zetterstedt, 1828.
Brachylobus lithophilus indigaceus Casey, 1914 = Chlaenius lithophilus Say, 1823.
Calleida striata Casey, 1913 = Calleida fulgida Dejean, 1831.
Carabus franciscanus Casey, 1913 = Carabus taedatus agassii LeConte, 1850.
Celia impunctata Putzeys, 1867 = Amara chalcea Dejean, 1828.
Chlaenius frostii Carr, 1920 = Chlaenius purpuricollis Randall, 1838.
Chlaenius purpuricollis Randall, 1838 = Chlaenius purpuratus Harris, 1836.
Chlaenius sierricola Casey, 1914 = Chlaenius sericeus (Forster, 1771).
Chlaenius texanellus Casey, 1914 = Chlaenius brevilabris LeConte, 1847.
Chlaenius viridifrons Eschscholtz, 1833 = Chlaenius sericeus (Forster, 1771).
Clivina cordata Putzeys, 1846 = Clivina acuducta Haldeman, 1843.
Cryobius delicatus Casey, 1918 = Pterostichus brevicornis brevicornis (Kirby, 1837).
Cymindis elegans mobilensis Casey, 1920 = Cymindis elegans LeConte, 1846.
Darlingtonea kentuckensis lexingtoni Valentine, 1952 = Darlingtonea kentuckensis Valentine, 1952.
Dicaelus darlingtoni Fall, 1932 = Dicaelus quadratus LeConte, 1847.
Euharpalops Casey, 1924 = Opadius Casey, 1914.
Glanodes regressus Casey, 1914 = Harpalus obliquus Horn, 1880.
Goniolophus lucens Casey, 1914 = Philodes flavilimbus (LeConte, 1869).
Haploharpalus Schauberger, 1926 = Opadius Casey, 1914.
Harpalus cyrtonotoides Notman, 1919 = Harpalus desertus LeConte, 1859.
Harpalus subaeneus Mannerheim, 1853 = Harpalus fulvilabris Mannerheim, 1853.
Hypherpes alamedae Casey, 1918 = Pterostichus vicinus Mannerheim, 1843.
Hypherpes intectus Casey, 1918 = Pterostichus protractus LeConte, 1860.
Hypherpes kansanus Casey, 1918 = Pterostichus algidus LeConte, 1853.
Hypherpes placerensis Casey, 1918 = Pterostichus laborans Casey, 1913.
Hypherpes protensipennis Casey, 1918 = Pterostichus mercedianus (Casey, 1918).
Lebia scapularis var. limbigera Chaudoir, 1871 = Lebia solea Hentz, 1830.
Micranillodes Jeannel, 1963 = Anillinus Casey, 1918.
Moritapterus O. Berlov, 2000 = Euferonia Casey, 1918.
Neomyas lindrothi Allen, 1980 = Myas cyanescens Dejean, 1828.
Pharalus Casey, 1914 = Opadius Casey, 1914.
Platynus hornii Hausen, 1891 = Agonum muelleri (Herbst, 1784).
Poecilus lucublandus acomanus Casey, 1924 = Poecilus lucublandus (Say, 1823).
Poecilus lucublandus louisinus Casey, 1924 = Poecilus lucublandus (Say, 1823).
Pterostichus breviusculus Casey, 1913 = Pterostichus congestus (Ménétriés, 1843).
Pterostichus breviusculus mimus Casey, 1913 = Pterostichus congestus (Ménétriés, 1843).
Pterostichus bucolicus Casey, 1913 = Pterostichus algidus LeConte, 1853.
Pterostichus cuneatulus Casey, 1913 = Pterostichus suffusus Casey, 1913.
Pterostichus diabolus Casey, 1913 = Pterostichus californicus (Dejean, 1828).
Pterostichus gregalis Casey, 1913 = Pterostichus castanipes (Ménétriés, 1843).
Pterostichus humboldti Casey, 1913 = Pterostichus algidus LeConte, 1853.
Pterostichus plutonicus Casey, 1913 = Pterostichus congestus (Ménétriés, 1843).
Pterostichus tahoensis Casey, 1913 = Pterostichus laborans Casey, 1913.
Pterostichus zunianus Casey, 1913 = Pterostichus protractus LeConte, 1860.
Rhadine gracilenta Casey, 1913 = Rhadine jejuna (LeConte, 1878).
Rhadine plumasensis Casey, 1920 = Rhadine jejuna (LeConte, 1878).
Rhadine tenuipes Casey, 1920 = Rhadine jejuna (LeConte, 1878).
Selenophorus excisus LeConte, 1878 = Selenophorus parumpunctatus (Dejean, 1829).
Selenophorus parumpunctatus Dejean, 1829 = Selenophorus sinuatus (Gyllenhal, 1806).
Selenophorus perpolitus Casey, 1884 = Athrostictus punctatulus Putzeys, 1878.
Steniridia andrewsi barksdalei Valentine, 1936 = Scaphinotus andrewsi darlingtoni (Valentine, 1935).
Steniridia andrewsi montana Valentine, 1935 = Scaphinotus andrewsii amplicollis (Casey, 1920).
Steniridia andrewsi nantahalae Valentine, 1936 = Scaphinotus andrewsi darlingtoni (Valentine, 1935).
Steniridia andrewsi saludae Valentine, 1936 = Scaphinotus andrewsi darlingtoni (Valentine, 1935).
Steniridia ridingsi intermedia Valentine, 1935 = Scaphinotus ridingsii ridingsii (Bland, 1863).
Steniridia violacea carolinae Valentine, 1935 = Scaphinotus violaceus (LeConte, 1863).
Stenolophus abstinens Casey, 1914 = Stenolophus incultus Casey, 1914.
Stenolophus extensicollis Casey, 1924 = Stenolophus cincticollis LeConte, 1858.
Stenolophus lamprotus Bates, 1891 = Stenolophus cincticollis LeConte, 1858.
Stenolophus peregrinus Casey, 1914 = Stenolophus anceps LeConte, 1857.
Stenolophus quadripunctatus Mannerheim, 1853 = Dicheirotrichus cognatus (Gyllenhal, 1827).
Stenolophus remissus Casey, 1914 = Stenolophus incultus Casey, 1914.
Stenolophus semitinctus Casey, 1914 = Stenolophus cincticollis LeConte, 1858.
New status
Agonoleptus dolosus Casey, 1914 from Agonoleptus unicolor dolosus.
New combinations
Agonoleptus conjunctus (Say, 1823) from Stenolophus.
Agonoleptus dolosus Casey, 1914 from Stenolophus.
Agonoleptus rotundatus (LeConte, 1863) from Stenolophus.
Agonoleptus rotundicollis (Haldeman, 1843) from Stenolophus.
Agonoleptus thoracicus (Casey, 1914) from Stenolophus.
Agonoleptus unicolor (Dejean, 1829) from Stenolophus.
Anillinus affabilis (Brues, 1902) from Anillodes.
Anillinus depressus (Jeannel, 1963) from Micranillodes.
Anillinus sinuatus (Jeannel, 1963) from Anillodes.
Calosoma dawsoni (Dajoz, 1997) from Callisthenes.
Scaphinotus hoffmani (Barr, 2009) from Maronetus.
Scaphinotus reichlei (Barr, 2009) from Maronetus.
Changes in precedence
Agonum deplanatum Ménétriés, 1843 for Agonum fallianum (Leng, 1919).
Badister micans LeConte, 1844 for Badister ocularis Casey, 1920.
Ellipsoptera rubicunda Harris, 1911 for Ellipsoptera marutha Dow, 1911.
1 These asterisks were dropped in the publication of Say’s entomological works by LeConte.
2 These names have been credited to Eschscholtz by almost all authors I have seen although there are proper citations of Fischer von Waldheim’s plates in Eschscholtz’s work. This suggests that the plates were available before the publication of Eschscholtz’s work. However because Fischer von Waldheim’s validation of the names is through illustrations, it is always possible that Fischer von Waldheim simply gave Eschscholtz the position of these species on his forthcoming plates.
3 By 1825, Major LeConte had sent Dejean more than 600 species of beetles (Dejean 1825: xxv). He also visited Paris in 1828 and gave Dejean a huge [“une immense”] collection of insects from the United States (Dejean 1828: vi). All species named “lecontei” by Dejean were in the honour of the Major and not his son.
4 A copy of the letter was published in The Coleopterists Bulletin in December 1961.
5 Following Maddison et al. (1999: 104), the expression ‘basal grade carabids’ is restricted to lineages branching off early along the evolutionary path of the family, ‘middle grade carabids’ to the lineages placed by Jeannel (1941b) in his ‘Stylifera’ and ‘higher carabids’ to the numerous lineages currently included in the subfamily Harpalinae.
6 † = adventive species in North America; ‡ = Holarctic species. The species list is alphabetic within the subgenera and does not necessary follow the catalogue listing.
7 All vernacular names of tiger beetles are taken from Pearson et al. (2006) and Erwin and Pearson (2008).
8 Several new synonyms are proposed in this subgenus. All are based on a manuscript written by Serge Laplante in the 1990s originating from a study of the type specimens of all species then considered valid and the specimens in the CNC. The new synonymies should be credited to him.
9 This subgenus is herein removed from synonymy with Trechicus, see “Taxonomic Note” under Trechicus.
10 The field-collected larva described as Trachypachus sp. by Garner (1954: 49, 50; Figs a-c [Pl. 1]) is misidentified.
11 Gaumer (1977) described larvae of five subspecies of Cicindela formosa.
12 The field-collected and reared larvae described by Garner (1954: 220, 221; Pl. 14, Figs a-c) as Promecognathus laevissimus are misidentified.
13 As pointed out by Jeannel (1920a: 510), the field-collected larva described by Xambeu (1901: 58, 59) as Trechus obtusus has been incorrectly associated.
14 The field-collected larvae described by Garner (1954: 211, 212; Pl. 13, Figs a-c) as Metrius contractus are obviously misidentified.