Citation: Perger R, Wall A (2014) The description of a new species of the Neotropical land crab genus Gecarcinus Leach, 1814 (Crustacea, Decapoda, Brachyura, Gecarcinidae). ZooKeys 435: 93–109. doi: 10.3897/zookeys.435.7271
In this contribution a new species of the land crab genus Gecarcinus Leach, 1814, from the Neotropical Pacific coast of South America is described and illustrated. In addition to its unique body color, Gecarcinus nobilii sp. n. is distinguished from congeners by a distinctly wider carapace front and differences in the shape of the infraorbital margin. The new species is not isolated from Gecarcinus populations from the Pacific coast of Central America by an insurmountable geographic barrier. Considering the closure of the Panamanian Isthmus as a calibration point for morphological divergence between the trans-isthmian mainland populations of Gecarcinus, the virtual lack of morphological differentiation (other than color) between them and the distinctness of G. nobilii sp. n. suggests that G. nobilii sp. n. evolved from a common ancestor before the Isthmus closed.
Brachyura, Colombia, Ecuador, Gecarcinidae, Gecarcinus, Isthmus of Panama, Pacific, new species
Land crabs of the genera Gecarcinus Leach, 1814, Johngarthia Türkay, 1970, and Gecarcoidea H. Milne Edwards, 1837, play an important ecological role on many tropical islands (see
Although both mainland populations of Gecarcinus have been separated by the definite closure of the Isthmus of Panama some three million years ago, the morphology of Pacific and Atlantic forms is so similar that
As in trans-isthmian Gecarcinus, the taxonomical status of the South American Pacific population has been uncertain. Whereas
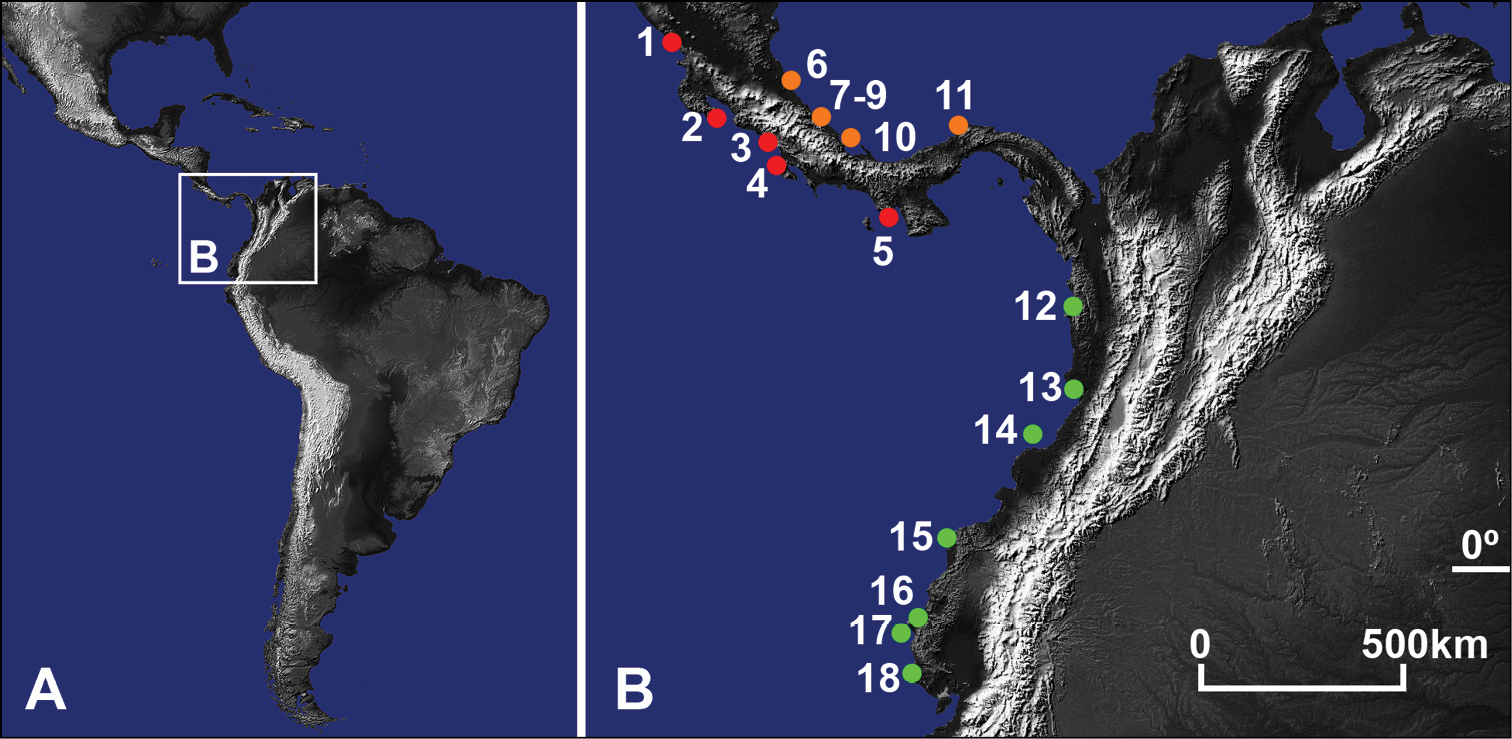
A Central and South America B Study area with locations of examined Pacific (red) and Atlantic (orange) Gecarcinus lateralis (Freminville, 1835) (sensu
Locations, date, coordinates of conducted sampling and number of examined individuals of Gecarcinus lateralis (Freminville, 1835) (sensu
| Location | Date | Coordinates | N |
|---|---|---|---|
| Pacific coast | |||
| Nicaragua, San Juan del Sur | 15–17 July 2011 | 11°15’39’’N, 85°52’52’’W | 54 |
| Costa Rica, Pochote Beach | 5–6 June 2011 | 9°44’51’’N, 85°00’01’’W | 75 |
| Costa Rica, Hermosa Beach | 6–8 Nov. 2011 | 9°10’07’’N, 83°45’39’’W | 69 |
| Costa Rica, Drake Bay | 16–18 Mar. 2011 | 8°41’33’’N, 83°39’42’’W | 57 |
| Panama, Santa Katalina | 25–27 Oct. 2011 | 7°37’39’’N, 81°14’50’’W | 60 |
| Atlantic coast | |||
| Costa Rica, Parismina | 12–14 Nov. 2011 | 10°18’34’’N, 83°20’85’’W | 56 |
| Costa Rica, Puerto Viejo | 26–28 May 2011 | 9°39’20’’N, 82°44’28’’W | 68 |
| Costa Rica, Manzanillo | 20–22 Nov. 2011 | 9°37’53’’N, 82°39’47’’W | 73 |
| Costa Rica, Punta Mona | 16–18 Nov. 2011 | 9°37’24’’N, 82°37’11’’W | 123 |
| Panama, Bocas del Toro, Bluff beach | 10–12 Oct. 2011 | 9°23’25’’N, 82°14’14’’W | 23 |
| Panama, Maria Chiquita | 20–22 Oct. 2011 | 9°26’42’’N, 79°45’44’’W | 19 |
An Internet search revealed additional photographs of Gecarcinus individuals from the Pacific coast of South America with a body color different from the specimens that were collected on the Central American coasts. Further examination of museum specimens and re-examination of the freshly collected specimens from Central America has revealed that the color differences are accompanied by differences in morphological structures as well. In accordance with these differences, a new species of Gecarcinus is herein proposed.
Specimens from the following institutions were examined: Academy of Natural Sciences of Drexel University, Philadelphia, USA (ANSP); Natural History Museum of Los Angeles County, Los Angeles, USA (LACM); Muséum National d’Histoire Naturelle, Paris, France (MNHN); Museo de Zoología, Universidad de Costa Rica, San José, Costa Rica (MZUCR); Naturhistorisches Museum, Basel, Switzerland (NHMB) and National Museum of Natural History, Smithsonian Institution, Washington, D.C., USA (USNM).
Cancer ruricola Linnaeus, 1758 (original combination).
Exopod not projecting beyond third maxilliped ischium-merus articulation, without flagellum; palp concealed by third maxilliped merus margin. Mesial segment of first male gonopod distally reduced and terminal segment basally exposed, terminal segment projecting beyond apical setae.
Because further evidence challenging
| 1 | Mesial lobe of infraorbital margin curved around ventrolateral edge of carapace front. Third maxilliped merus covers epistome and can reach carapace front. Spines on lateral carina of dactylus and carpus of ambulatory legs prominently developed in adults | Gecarcinus ruricola (Linnaeus) |
| – | Contact between carapace front and mesial lobe of infraorbital margin straight (Fig. 2B, C). Third maxilliped merus at the farthest reaching epistome. Spines on lateral carina of dactylus and carpus of ambulatory legs weakly developed to absent | 2 |
| 2 | Carapace front wider than distance between mesial ends of suborbital cristae (Figs 2C; 3B). Width of mesial lobe of infraorbital margin at point of contact with carapace front longer than shortest distance between carapace front and mesial end of suborbital crista (Figs 2C; 3B). Light lateral margin on dorsal carapace without lighter anterolateral and posterior patches (Figs 4C; 5D) | Gecarcinus nobilii sp. n. |
| – | Carapace front about as wide as distance between mesial ends of suborbital cristae (Figs 2B; 6B, E). Width of mesial lobe of infraorbital margin at point of contact with carapace front shorter than shortest distance between carapace front and mesial end of suborbital crista (Figs 2B; 6B, E). Without light lateral margin on dorsal carapace (Pacific, Fig. 5A; Atlantic, Fig. 5C) or with light lateral margin with anterior and posterior yellow to orange patches (Atlantic, Fig. 5B) | Gecarcinus lateralis (Fréminville) |
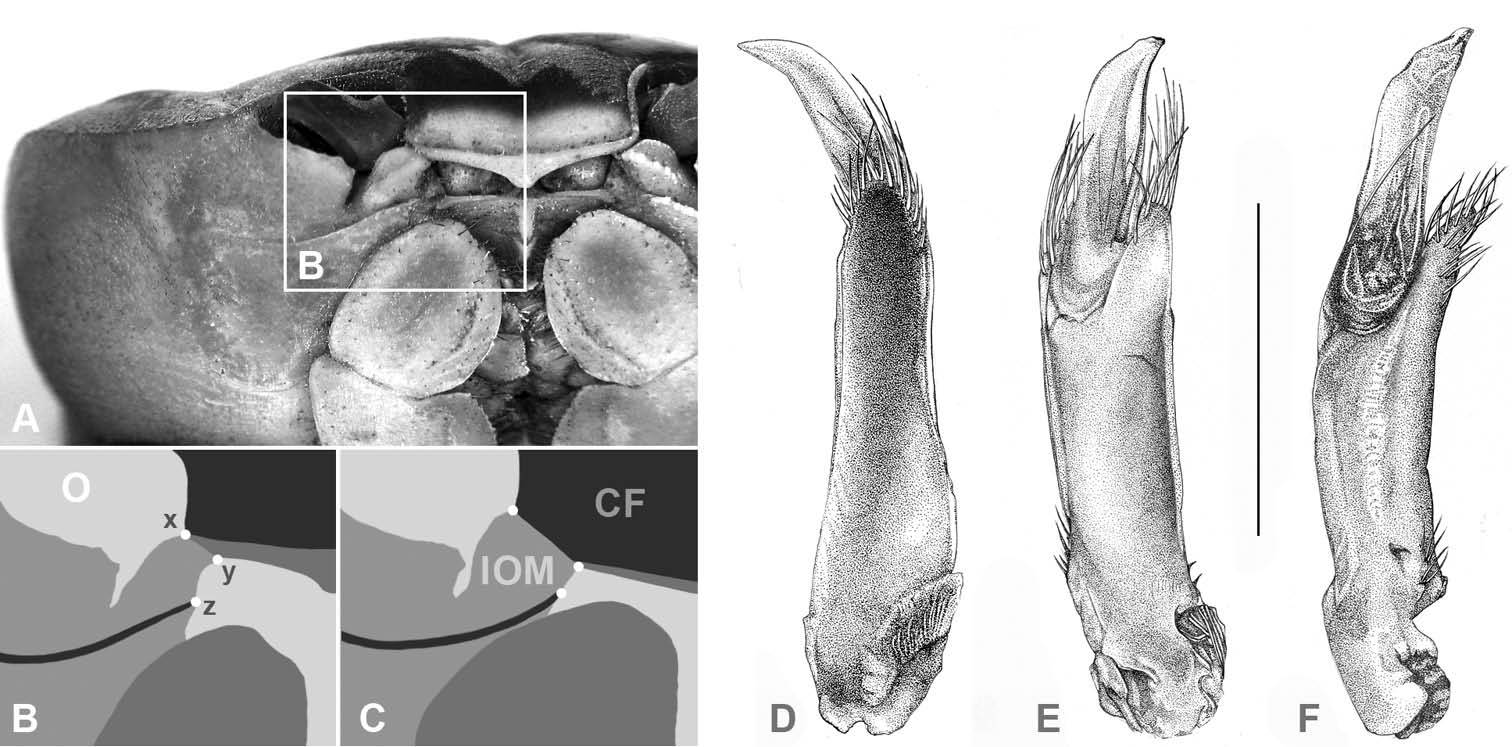
(CF) Carapace front; (O) orbit; (IOM) mesial lobe of infraorbital margin; (x) widest width of CF; (z) mesial end of suborbital crista; (x–y) width of IOM at point of contact with CF; (y–z) shortest distance between CF and mesial end of suborbital crista; A, B Atlantic Gecarcinus lateralis (
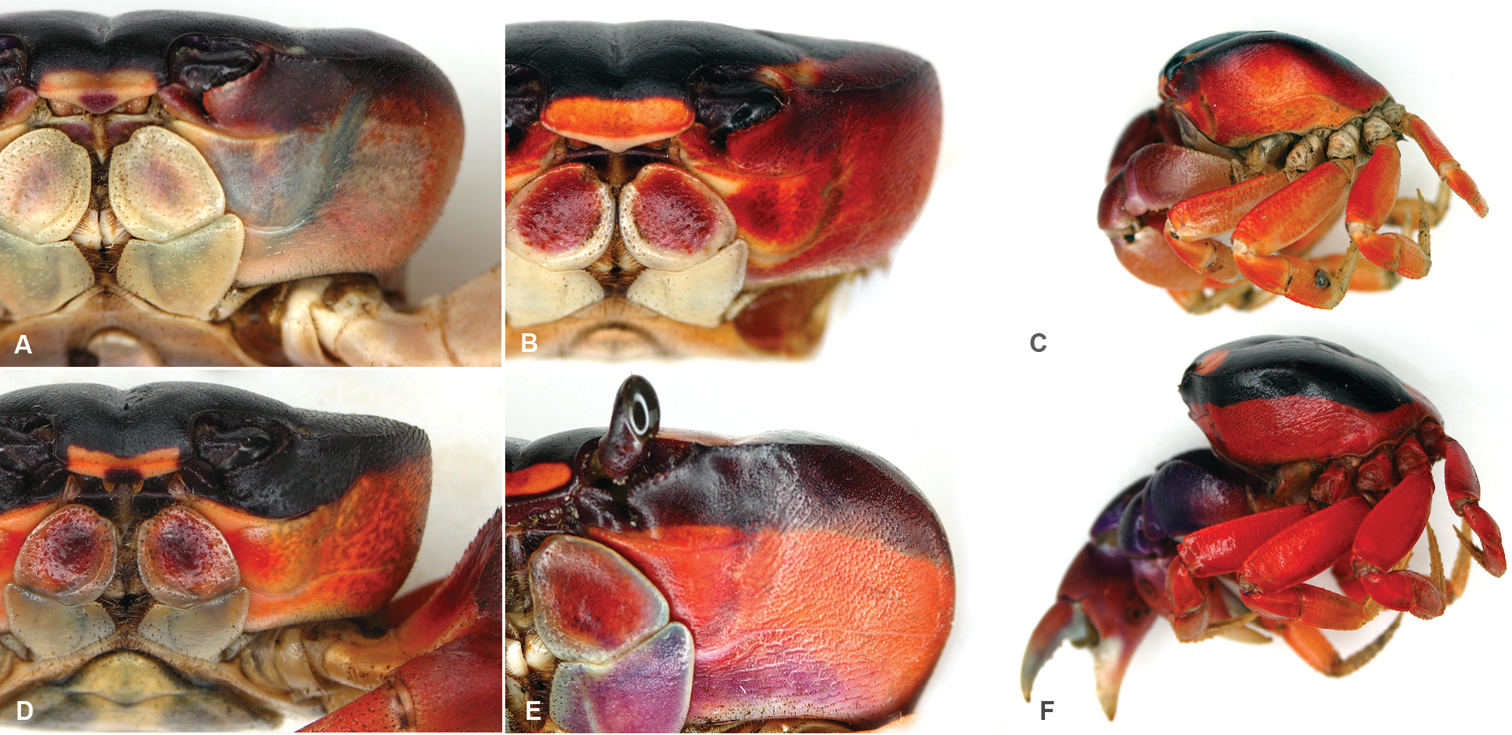
Gecarcinus nobilii sp. n., holotype, male, carapace width 31 mm, Ecuador, Punta Galera (LACM CR 1968-477), preserved in alcohol (color faded): A dorsal view B frontal view C ventral view. Paratype, female, Ecuador, St. Elena (MNHN, B12314), preserved in alcohol (color faded): D carapace front E frontal view F ventral view.
Gecarcinus nobilii sp. n., color in life; Colombia: Gorgona Island A male, photo by Karla Garcia Burneo (Peru) B, C male, photos by Rhett A. Butler (USA) D female, Buenaventura dept., Chucheros Beach, photo by Elena Gómez E sex unknown, Chocó dept., Nuquí prov., Canangucho Forest Reserve. Ecuador: F sex unknown, Manabí prov., Ayampe, photo by David Liebman (USA). Captive individuals from the pet trade, origin unknown: G sex unknown, photo by Oliver Mengedoht (Germany) H, I females, photos by John Beatty (USA) (the individual shown above in Fig. I belongs to the Pacific population of Gecarcinus lateralis (sensu
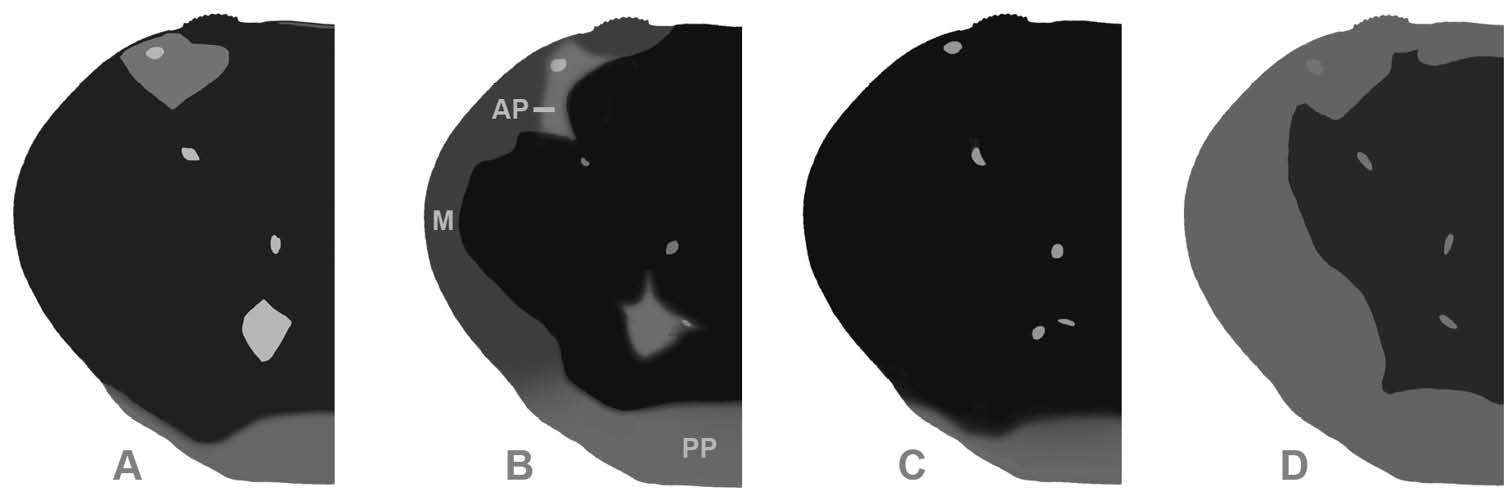
Dorsal carapace pattern (shape of light patches could vary slightly): Gecarcinus lateralis (
A–C lectotype of Gecarcinus lateralis (Freminville, 1835), male, carapace width 47.2 mm, Guadeloupe (MNHN-3758) (dried specimen, color faded) D–F syntype of Gecarcinus quadratus Saussure, 1853, male, carapace width 39.7 mm, Mexico, Mazatlan (ANSP-CA3741) (dried specimen, color faded) (photos by Paul Callomon, Academy of Natural Sciences of Drexel University, Philadelphia).
http://zoobank.org/F3BA2617-49DF-4C26-ACB5-5407122301FF
Holotype: male, carapace width (CW) 31 mm, Ecuador, Punta Galera, 0°50'N, 80°6'W, shore, collected near a pile of fairly fresh cow manure, Te Vega Expeditions, Sta. NO. XVIII-6, 22 April 1968. Coll. E. Ball (LACM CR 1968-477). Paratypes: 1 male, CW 26 mm, same location data as holotype (LACM CR 1968-478); 1 female, Ecuador, St. Elena (MNHN- B12314); 3 females, CW 28, 23, 23.5 mm, Ecuador, Esmeraldas (NHMB-NMB1010b). (Note: The female paratype (MNHN-B12314) (Fig. 3D–F) of Gecarcinus nobilii sp. n. was labeled as "Gecarcinus festae Nobili/(co-type)/St. Helena/Festa/Museum Paris/Ecuador/Nobili 1901” without information about the label author. Nobili never published a description of a species by this name. In 1901 he described Sesarma festae and Uca festae and in the same contribution referred to specimens of Gecarcinus collected in Ecuador as Gecarcinus ruricola, which is restricted to West Atlantic Islands (
Additionally, high resolution photographs of 14 captive individuals with unknown origin and 17 individuals taken at the following locations (confirmed by the photographers): Gorgona Island (Colombia) (Fig. 4A–C), Chucheros Beach (Buenaventura, Colombia) (Fig. 4D), Canangucho Forest Reserve (Nuquí, Chocó, Colombia) (Fig. 4E), Ayampe (Manabí, Ecuador) (Fig. 4F), and Isla de la Plata (Ecuador), were examined.
Gecarcinus lateralis (Freminville, 1835): lectotype male, CW 47.2 mm, Guadeloupe, M. Beaupertius leg. (MNHN-3758). Paralectotypes: 1 male, CW 50 mm, Guadeloupe, M. Beaupertius leg. (MNHN-3757); 1 female, CW 28 mm, same data as preceding specimen. 1 male, CW 32 mm, Martinique, M. Bellanger leg. (MNHN-3756); 1 female, CW 37 mm, Martinique, Bellanger leg., 24.09.1964 (MNHN-3755). Gecarcinus quadratus Saussure, 1853: syntype male, CW 39.7 mm, Mexico, Mazatlan (ANSP-CA3741). Pacific Gecarcinus lateralis (sensu
The species is named in honor of Giuseppe Nobili, who provided important contributions on the knowledge of crustaceans and built the crustacean collection in the Turin Museum (Italy). The species name is a noun in the genitive case.
Frontal width distinctly wider than the distance between the mesial ends of the suborbital cristae (Figs 2C; 3B, E). Width of mesial lobe of infraorbital margin at point of contact with carapace front longer than shortest distance between carapace front and mesial end of suborbital crista (Figs 2C; 3B, E). Light lateral margin on dorsal carapace without lighter anterolateral and posterior patches (Figs 4C; 5D); cheliped carpus and palm homogeneously red or white (Fig. 4).
Carapace transversely ovate, widest in anterior half, dorsal surface smooth. Cardiac, gastric and branchial chambers moderately swollen (Fig. 3B, E). Median groove distinct, cervical and urogastric grooves weakly developed; three relatively small pits anterior (close to orbit), median and posterior of cervical groove, one in urogastric groove (Fig. 3A). Supraorbital margin gently sinuous, with small granules, confluent with anterolateral margin; exorbital tooth weakly developed, tip not over-reaching orbit (Fig. 3B, D, E); granules along anterolateral and orbital margins weakly developed. Eyes well developed, filling orbital cavity; eyestalks short (Fig. 3B, E). Carapace front distinctly wider than the distance between mesial ends of the suborbital cristae (Figs 2C; 3B, E), deflexed downwards, concealing basal segments of antennules. Width of mesial lobe of infraorbital margin at point of contact with carapace front longer than shortest distance between carapace front and mesial end of suborbital crista (Figs 2C; 3B, D; 4H). Suborbital, pterygostomial regions sparsely granular laterally. Subhepatic region with rounded postero-lateral margins, with rows of small granules. Epistom linear, sunken.
Third maxilliped merus and outer ventral orbital border furnished with long setae (Fig. 3D); third maxilliped merus enlarged, reaching mesial border of suborbital crista, triangular, apex straight or moderately convex (Figs 3B, 4I); exopodit short, not protruding beyond third maxilliped ischium-merus joint, without flagellum; palpus concealed by external border of third maxilliped merus.
Chelipeds subequal; in large males larger with respect to the carapace width, surfaces relatively smooth, weakly granulate. Merus with transversal rows of small tubercles; dorsal margin rugose or with moderately developed, obtuse granules; ventral margin lined with weakly developed granules, otherwise smooth. Carpus with 2–5 well developed triangular inner subdistal tooth (Fig. 3A, B, E). Merus and carpus margins smooth in large individuals. Chela large, length not exceeding carapace width, surfaces smooth; lower margin gently sinuous. Fingers slightly shorter or as long as palm, tapering, gently curved, drop-shaped in cross-section, proximal half with irregular arranged pores and low, pectinated teeth; teeth on distal portion of finger arranged on well defined, subparallel longitudinal ridges, longitudinally separated by grooves with pores. Cutting margins with distinct triangular teeth along length; fingers without or with small gap between them when closed, ending in sharp, pectinated tips.
Ambulatory legs with second pair longest, last pair shortest; surfaces smooth to slightly rugose. Merus dorso-laterally flattened, cross-section triangular, stout; with transversal rows of small tubercles, dorsal margin distinct, granulated, with separate, short setae. Carpus stout, subtriangular in cross-section; dorsal surface with three carinae, median carina distinct, serrated or granular; dorso-lateral carinae weakly developed or absent; margins and carinae lined with separate, short setae. Propodus subrectangular in cross-section; lateral margins subparallel, lined with low, obtuse spines and separate, short setae (Fig. 3A–C). Dactylus elongate, styliform, gently curving, subquadrate in cross-section, margins lined with distinct spines and separate, short setae; apical half of spines and dactylus tip corneous; lateral carina of dactylus weakly developed or absent (Fig. 3A–C).
Male abdomen relatively broad (Fig. 3C), all abdominal somites and telson distinct, freely articulating. Somite 1 filling space between last pair of ambulatory legs, longitudinally very narrow. Shape of somite 2 similar to somite 1 but narrower. Somites 3–5 increasingly trapezoidal in shape, lateral margins relatively straight. Somite 6 longest, longer than telson, distinctly wider than long, with lateral margin strongly convex. Telson sub-triangular, narrowest abdominal segment; as long as wide, lateral margins gently concave to almost straight, tip rounded (Fig. 3C).
First male gonopod with basal and terminal segment (Fig. 2E). Basal segment stout, straight, with digiform projection on distomesial end, projection directed in same manner with distal segment, fringed with long setae. Terminal segment about one-third of first gonopod (when seen from lateral view, Fig. 2E), folded longitudinally, compressed dorsoventrally, tapering and curved distally, slightly projecting over distal setae, laterally with narrow, longitudinal furrow.
Sex independent color dimorphism: red and white males and females (Fig. 4). Both forms with dark median pattern and contrasting light lateral margin on dorsal carapace without lighter anterior and posterior patches (Fig. 5D), margin of same color as lateral carapace; dark dorsal carapace pattern with pointed anterolateral edges anteriorly reaching the orbits (Fig. 5D). Mesial lobe of infraorbital margin mostly grey (Fig. 4). Red form with red lateral margin on dorsal carapace. Carapace pits white to orange. Legs and chelipeds uniformly red, inner sides of fingers cream to white, margin of the third maxilliped merus, coxa, basis and ischium of chelipeds and ambulatory legs whitish. In white form, lateral margin on dorsal carapace orange/white or completely white. Ventral carapace and chelipeds white, legs and carapace pits light orange to white (Fig. 4).
Gecarcinus nobilii sp. n. is currently known to occur from Punta Galera and St. Elena (Ecuador). It is also documented in photographs taken at Gorgona Island (Colombia) (Fig. 4A–C), Chucheros Beach (Buenaventura, Colombia) (Fig. 4D), Canangucho Forest Reserve (Nuquí, Chocó, Colombia) (Fig. 4E), Ayampe (Manabí, Ecuador) (Fig. 4F), and Isla de la Plata (Ecuador). Individuals of Gecarcinus previously reported from Peru (
Available data and the photographs found during the Internet search suggest that Gecarcinus nobilii sp. n. replaces Pacific Gecarcinus lateralis between the Darien province (Panama) and the Choco dept. (Colombia). In addition to the individuals of Pacific Gecarcinus lateralis found during fieldwork (Table 1), the Internet search revealed numerous photographs of Pacific Gecarcinus lateralis from Central America. However, there are no photographs of Pacific Gecarcinus lateralis from within the distributional area of Gecarcinus nobilii sp. n.
The resemblance of the general habitus, the shape and the surface structure of carapace, chelipeds, ambulatory legs (Figs 3; 6–8) and the first male gonopod (Fig. 2E, F) indicate a close relationship between all mainland Gecarcinus populations. However, the trans-isthmian populations of Gecarcinus lateralis differ from Gecarcinus nobilii sp. n. by having a carapace front approximately as wide as the distance between the mesial ends of the suborbital cristae, and the width of the mesial lobe of the infraorbital margin at the point of contact with the carapace front is shorter than the shortest distance between the carapace front and the mesial end of the suborbital crista (Figs 2B; 6B, E; 7A, B).
Gecarcinus lateralis (Freminville, 1835) (sensu
Gecarcinus lateralis (Freminville, 1835) (sensu
A character commonly used to distinguish between species of the Gecarcinidae is the shape of the third maxilliped merus margin (e.g.
In addition to observed differences in morphological structures, the body color of Pacific Gecarcinus lateralis (sensu
Apart from the consistent color differences between Gecarcinus nobilii sp. n. and the other mainland populations of Gecarcinus, which provided the starting point for this contribution, the color differences between the trans-isthmian populations of Gecarcinus (Table 2; Figs 5A–C; 7; 8) also drew our attention. According to
Comparison of color in life for mainland Gecarcinus spp. (carapace width of Gecarcinus lateralis (Freminville, 1835) (sensu
| Atlantic Gecarcinus lateralis (n = 362) | Pacific Gecarcinus lateralis (n = 316) | Gecarcinus nobilii sp. n. (n = 19) | |
|---|---|---|---|
| Color polymorphism | Morphs with transitional forms | - | Red and white forms |
| Light lateral margin on dorsal carapace | Present or absent, when absent, dark median pattern laterally extending over ACB with smooth transition to color of ventrolateral carapace | Absent, dark median pattern laterally extending over ACB, anteriorly mostly reaching SUB, with sharp contrast to color of ventrolateral carapace | Present, contrasting to dark median pattern, same color as lateral carapace, in some white forms with orange tinge |
| Orange patch at anterolateral carapace border | In forms with lateral margin on dorsal carapace, patch with smooth transition to bordering color, only posteriorly bordered by dark carapace pattern; patch absent in forms without light lateral margin on carapace | Always present, encircled by dark, sharply contrasting dorsal carapace color | Absent |
| Patch at posterior lateral urogastric groove | In forms with lateral margin on dorsal carapace heterogeneously yellowish, grayish, cream, sometimes with smooth transition to lateral margin; weakly developed to absent in forms without lateral margin |
Homogeneously whitish, subtriangular to rhomboid, well defined, sharply contrasting with surrounding dark carapace color | Absent |
| Ventro-lateral carapace color | Heterogeneous, transition between dorsal and ventral carapace color | Uniformly bright deep orange to red, sharply contrasting to dorsolateral carapace color | Homogeneously red or white |
| Cheliped palm color | Orange to red, often with purple tinge anteriorly, or orange-purplish-whitish | Deep violet, sometimes with purplish tinge | Uniformly red or white |
| Cheliped dactylus color | Light orange, orange to light red, cutting edges cream | Completely white or base light violet, purplish, becoming white distally | Red or white, cutting edges cream or white |
| IOM color | As suborbital area | Mostly grey | |
While the color of examined Gecarcinus lateralis from the Atlantic coast (n= 362) varied within the range described by
Studies of genetic divergence and reproductive isolation are needed to evaluate whether Gecarcinus quadratus should be retained as a synonym of Gecarcinus lateralis, or alternatively, the trans-isthmian populations of Gecarcinus lateralis represent allopatric sister species.
When we consider the closure of the Panamanian Isthmus as a calibration point for morphological divergence between the trans-isthmian populations of Gecarcinus lateralis, the virtual lack of morphological differentiation (other than color) between them and the distinctness of Gecarcinus nobilii sp. n. suggests that Gecarcinus nobilii sp. n. evolved from a common ancestor before the Isthmus closed. The common ancestor of the trans-isthmian Gecarcinus lateralis may have been restricted to North America and/or the emerging Isthmus, which is assumed to have been a peninsula of North America (
We wish to extend a hearty thanks to Oliver Mengedoht (Germany), Elena Gómez, David Liebman, John Beatty, Rhett A. Butler (USA) and Karla Garcia Burneo (Peru) for granting us permission to publish their photographs. We are very grateful to Paula Martin-Lefevre (Muséum National d’Histoire Naturelle, Paris), Edgar Stöckli (Naturhistorisches Museum, Basel, Switzerland), Paul Callomon (Academy of Natural Sciences of Drexel University, Philadelphia), Karen Reed and Katie Ahlfeld (Smithsonian Institution, Washington, D.C.) for providing photographs and data of museum specimens. Martha Iserman (bigredsharks.com) is thanked for their drawings of the gonopods. RP thanks to Jorge Cortés, Cristian Pacheco, and Rita Vargas (Universidad de Costa Rica, San José, Costa Rica) for supporting this project and for providing an abundance of insight and information, and to David Wilcox (USA), Joanne Taylor (Museum Victoria, Melbourne, Australia) and the anonymous reviewers for providing comments and suggestions on the manuscript.