Citation: Taylor CK, Probert A (2014) Two new species of harvestmen (Opiliones, Eupnoi, Neopilionidae) from Waitomo, New Zealand. ZooKeys 434: 37–45. doi: 10.3897/zookeys.434.7486
Two new species of harvestman (Opiliones: Neopilionidae: Enantiobuninae) are described from the Waitomo region of the North Island, New Zealand, Forsteropsalis bona sp. n. and F. photophaga sp. n. Both have been collected within caves in the region, where predation on glow-worms Arachnocampa luminosa has been previously recorded for one or both species (misidentified as ‘Megalopsalis tumida’). However, both are regarded as troglophiles rather than strict troglobites due to the presence of specimens outside the cave systems, and the absence of troglobitic adaptations. Megalopsalis tumida (Forster, 1944) is identified as a junior synonym of Forsteropsalis fabulosa (Phillipps & Grimmett, 1932).
Taxonomy, arachnids, cave biota
The Waitomo region of the North Island, New Zealand, has become internationally renowned as a tourist attraction owing to its extensive cave systems. The primary reason for their fame is their large population of glow-worms Arachnocampa luminosa (Keroplatidae). These luminescent fly larvae construct silken nests on the roof of the cave from which they hang sticky threads to capture flying insects attracted to their light (
While
Specimens were sourced from the collection of Te Papa Tongarewa, Wellington, New Zealand (MONZ) or collected by hand by A. Probert and associates. Specimens collected by A. Probert will be deposited at the New Zealand Arthropod Collection, Landcare Research, Auckland, New Zealand (NZAC). All specimens are assigned to the area code WO (Waikato) by the system established by
WO. Lucky Strike Cave, Te Kuiti, on wall near entrance, 14 Feb 1959, K. A. J. Wise (MONZ).
WO. 2 males, Waitomo Valley, in stream crevice outside cave entrance (shady), specimens intertwined and sluggish, 30 Mar 1959, L. G. Watson (MONZ); 1 male, Weir Cave, Stubbs Farm, Waitomo, ca. 2 m from cave entrance within a 15 cm radius of Arachnocampa luminosa larvae, 23 Aug 2010, A. Probert (NZAC).
From the Latin bonus, good, in contrast to the related Forsteropsalis fabulosa.
Total body length 4.8–6.6; prosoma length 2.5–2.8, width 4.0–4.2. Prosoma (including ocularium) unarmed (Fig. 1A); ground colour in alcohol orange-brown with longitudinal yellow stripes on either side of ocularium (live coloration very dark brown [almost black] with orange-yellow stripes; appendages also black). Ozopores elongate, with small flanking lobes. Opisthosoma grey-yellow. Mouthparts cream-coloured; medial side of pedipalpal coxa with dense array of sharp denticles; cervix unarmed. Coxae yellow. Chelicerae (Fig. 1B): Segment I length 6.2–8.4; segment II 9.4–10.4. Elongate; segment I orange with lighter yellow patch at distal end, segment II dark orange-brown. Segment I denticulate, with denticles concentrated along dorsal, proventral and retroventral margins. Segment II massively inflated, evenly denticulate. Cheliceral fingers elongate, widely bowed apart; setae present on distal half of mobile finger. Pedipalps: Femur length 5.4–5.7; patella 2.2–2.4; tibia 2.8–3.1; tarsus 5.8–6.5. Distinctly elongate, yellow. Femur dorsally denticulate on proximal two-thirds; remainder of pedipalp unarmed. Setae sparse except for small concentration at prodistal end of patella; microtrichia present on tarsus and distal half of tibia; prodorsal end of patella with distinct protrusion but without definite finger-like apophysis (Fig. 1C). Tarsal claw without ventral tooth-row. Legs: Legs I femur length 8.6–9.9, patella 1.9–2.5, tibia 8.3–9.8; leg II femur 14.3–17.2, patella 2.2–2.8, tibia 14.4–17.8; leg III femur 7.5–8.7, patella 1.7–2.3, tibia 5.3–8.2; leg IV femur 8.5–10.7, patella 1.8–2.7, tibia 10.2–10.8. Femora sparsely denticulate, particularly in proximal half; remainder of legs unarmed. Distitarsus I with strong ventral tooth at distal end of each of first five or six pseudosegments (Fig. 1D). Tibia II with nine to fifteen pseudosegments; tibia IV with two pseudosegments. Penis (Fig. 1E–F): Shaft subquadrate; tendon long. Bristle groups relatively long, posterior bristle group with longest bristles reaching dorsal margin in lateral view. Glans short, subtriangular in ventral view, narrowing rapidly in lateral view.
Forsteropsalis bona sp. n. A dorsal view of body, holotype B lateral view of body, pedipalps and chelicerae, holotype C dorsal view of right pedipalpal patella and tibia, holotype D proximal pseudosegments of right distitarsus I (venter upwards), holotype, showing ventrodistal teeth E penis, ventral view, specimen from Waitomo Valley F penis, right lateral view, specimen from Waitomo Valley.
Females of this species are currently unknown. Forsteropsalis bona can be distinguished from most other Forsteropsalis species by its unarmed prosoma and enormous, sub-globose cheliceral segment II with widely bowed cheliceral fingers (
An interesting feature of Forsteropsalis bona is the presence of a strong ventrodistal tooth on the end of each of the proximal pseudosegments of the distitarsus. This tooth sits between the two spinose setae generally present on each tarsal pseudosegment in all Enantiobuninae (Fig. 1D). Such a feature has not previously been recorded for this subfamily, though it is also present in Forsteropsalis fabulosa (specimens from MONZ, details given in
The glans of both Forsteropsalis fabulosa (
Forsteropsalis fabulosa and Forsteropsalis tumida were distinguished in
WO. Waitomo, Gardners Gut Cave System, 200 yards from Zweihöllen entrance, 25 June 1977, W. L. Blundell (MONZ).
WO. 1 male, Giants Cavern, Hollow Hill Cave, Te Kuiti, in ‘Crows Nest’, 60–70 ft high, 12 January 1958, coll. R. W. Taylor (MONZ); 1 male, Aussie Cave, Taumatamaire Rd, Waitomo County, 50 ft, 16 May 1966, K. A. J. Wise (MONZ); 2 males, Stubbs Farm, Waitomo, on rocky cave substrate, February 2013, G. Holwell et al. (NZAC); 2 males, Mangapohue Cave, Stubbs Farm, Waitomo, on rocky cave substrate, 21 Oct 2013, A. Probert & D. Townsend (NZAC).
From the Greek phos, light, and phagein, to eat, in reference to this species’ predation of the glow-worm Arachnocampa luminosa.
Total body length 3.5–6.1; prosoma length 1.9–2.1, width 2.5–3.9. Prosoma (including ocularium) unarmed except for few black setae (Fig. 2A); ground colour orange-brown with longitudinal yellow stripes on either side of ocularium (live colouration light to mid-brown with pale yellow stripes). Ozopores elongate, with small flanking lobes. Opisthosoma grey-brown. Mouthparts cream-coloured; medial side of pedipalpal coxa with array of sharp denticles; cervix with single pair of denticles laterally. Coxa I orange; remaining coxae and venter of opisthosoma yellow. Chelicerae (Fig. 2B, D): Segment I length 3.4–6.5, segment II 4.9–9.1. Elongate; orange except for lighter yellow patch at distal end of first segment. First segment dorsally with scattered denticles, becoming more elongate retrolaterally, ventrally with longitudinal prolateral and retrolateral rows of elongate denticles and some scattered median denticles proximally. Second segment mildly to notably inflated, sub-conical, evenly denticulate with longitudinal rows of more elongate denticles dorsally and retrolaterally. Cheliceral fingers elongate, slightly bowed apart; setae present along central third of mobile finger. Pedipalps: Femur length 4.6–6.5, patella 2.8–3.2, tibia 2.2–2.8, tarsus 4.8–5.7. Distinctly elongate; yellow. Median side of coxa with array of sharp denticles. Femur with few denticles dorsally in proximal half; remainder of pedipalp unarmed. Patella, tibia and proximal half of tarsus densely covered with plumose setae; microtrichia present over entirety of patella, tibia and tarsus; patella with small, rounded, prodistal apophysis (Fig. 2C). Tarsal claw without ventral tooth-row. Legs: Leg I femur length 8.1–11.0, patella 1.9–2.2, tibia 8.4–10.7; leg II femur 14.0–17.7, patella 1.9–2.5, tibia 16.0–19.0; leg III femur 7.1–9.4, patella 1.6–1.9, tibia 7.6–9.8; leg IV femur 9.0–12.2, patella 1.8–2.2, tibia 10.3–12.4. Yellow. Proximal half of femur I with few scattered dorsal denticles; remainder of legs unarmed. Tibia II with 12 pseudosegments; tibia IV with three pseudosegments. Penis (Fig. 2E–F): Shaft subquadrate; tendon long. Bristle groups relatively long, posterior bristle group with longest bristles reaching dorsal margin in lateral view. Glans relatively long, subrectangular in ventral view, remaining relatively deep to distal end but with dorsodistal end rounded.
Forsteropsalis photophaga sp. n. A dorsal view of body, holotype, with parasitic mite attached B anterolateral view of body, pedipalps and chelicerae, holotype C dorsal view of right pedipalpal patella and tibia, holotype D anterolateral view of chelicerae of specimen from Aussie Cave, showing more inflated chelicerae E penis, ventral view, holotype F penis, right lateral view, holotype.
Females of this species are currently unknown. The holotype of Forsteropsalis photophaga when first examined had a parasitic mite attached to the opisthosoma (Fig. 2A). This mite is a representative of the Microtrombidiidae, a family that has not previously been recorded as parasitic on Opiliones; a more detailed description is currently being prepared by C. Taylor.
The genera Pantopsalis and Forsteropsalis have hitherto been regarded as well distinguished by the morphology of the cheliceral fingers (crescent-shaped in Pantopsalis vs bowed in Forsteropsalis), pedipalpal patellar apophysis (hypersetose and rounded in Pantopsalis, sparsely setose and triangular in Forsteropsalis) and penile bristle groups (shorter in Pantopsalis than in Forsteropsalis) (
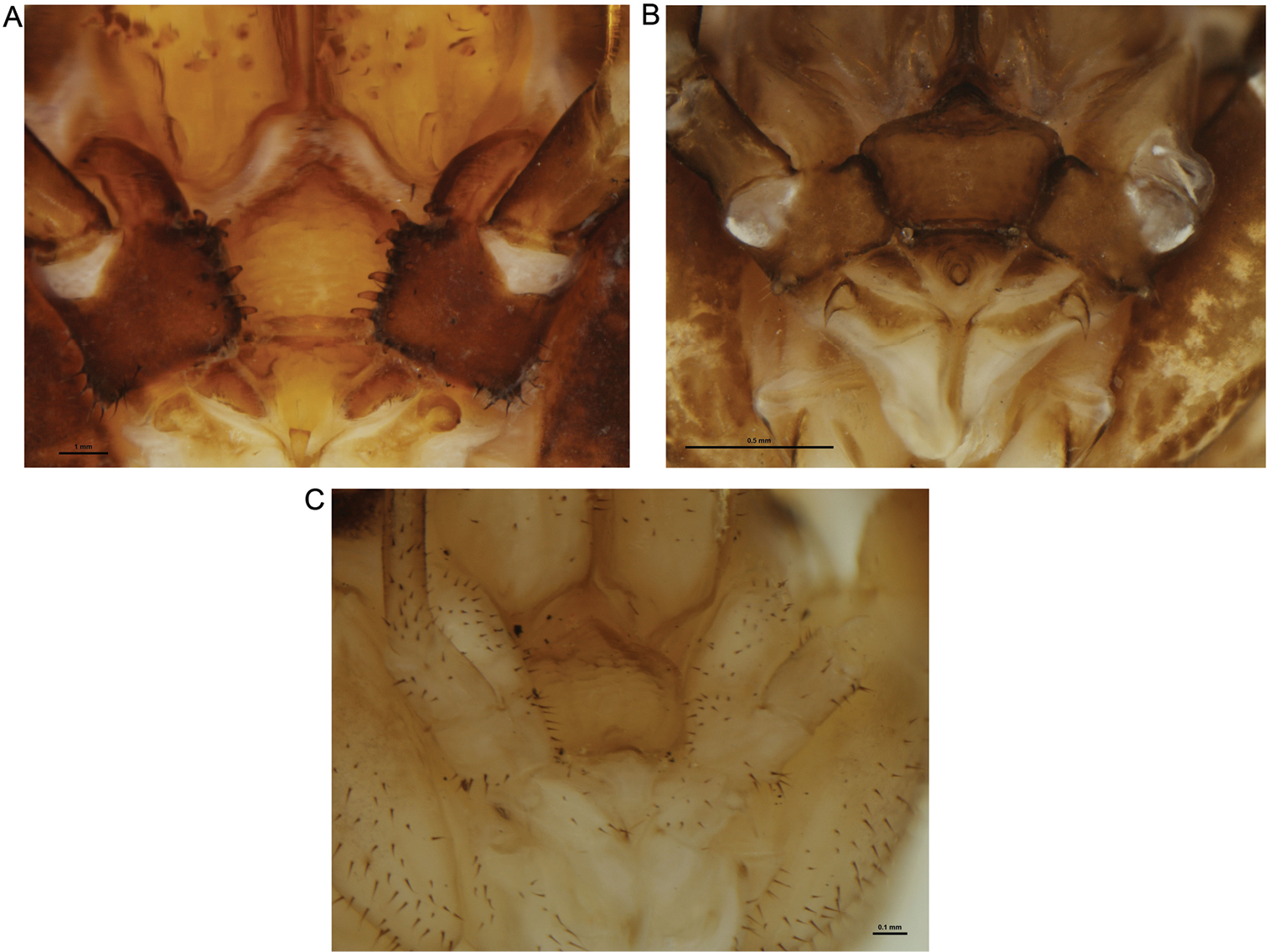
Mouthparts of selected New Zealand Neopilionidae, showing morphology of pedipalpal coxae. A Forsteropsalis chiltoni, with medial denticles B Pantopsalis albipalpis, with sclerotised medial flange overhanging cervix C Mangatangi parvum, with unarmed, simple coxae.
Forsteropsalis photophaga can be readily distinguished from all other Neopilionidae in New Zealand by the hypertrophied denticle rows on the second cheliceral segment. The only other neopilionid with comparable chelicerae is the major male of the Tasmanian species Megalopsalis nigricans (
The authors would like to thank Phil Sirvid, Te Papa Tongarewa, for arranging the loan of specimens. Anna Probert was assisted in collecting specimens by Daniel Townsend, Greg Holwell and Christina Painting. Access to caves on Stubbs Farm was provided by Alister and Ann Stubbs. Christopher Taylor’s position at Curtin University is funded by Chevron Australia.