Citation: Schmidt BC, Anweiler GG (2014) Taxonomy and biogeography of the Nearctic Raphia Hübner (Lepidoptera, Noctuidae, Raphiinae). In: Schmidt BC, Lafontaine JD (Eds) Contributions to the systematics of New World macro-moths V. ZooKeys 421: 91–113. doi: 10.3897/zookeys.421.7517
The taxonomic status and biogeography of the North American Raphia species is reviewed using adult morphology, larval host plants, geographic phenotypic variation, and variation of mtDNA COI barcode sequences. Lack of diagnostic morphological differences, combined with relatively low mtDNA barcode divergences and clinal phenotypic variation in key geographic regions indicate that the six previously recognized species of North American Raphia are best interpreted as parapatric subspecies. Raphia frater abrupta Grote, stat. n., R. f. coloradensis Putnam-Cramer, stat. r., R. f. piazzi Hill, stat. n., and R. f. elbea Smith, stat. n., are accordingly revised to subspecies of R. frater Grote. Type locality restrictions are provided for Raphia abrupta and Raphia frater and a neotype is designated for Raphia frater var. coloradensis.
DNA barcode, Populus, incipient species, subspecies, parapatry
Raphia Hübner is a small genus of the Holarctic region, with a single African species (
Three Raphia species occur in temperate Asia, one in southern Europe (
Adult genitalia were prepared following the methods of
We examined approximately 4000 specimens during the course of this study, primarily those of the CNC, EME, MEM, USNM, and UASM. Specimen repository abbreviations are as follows:
AMNH American Museum of Natural History, New York;
ANSP Academy of Natural Sciences, Philadelphia, PA;
BIO Biodiversity Institute of Ontario, Guelph, Ontario;
BMNH The Natural History Museum (statutorily British Museum [Natural History]), London;
CNC Canadian National Collection of Insects, Arachnids and Nematodes, Ottawa;
CSU Colorado State University, Fort Collins;
EME Essig Museum of Entomology, University of California, Berkeley, California;
MEM Mississippi Entomological Museum, Mississippi State, MS;
UASM University of Alberta Strickland Museum, Edmonton, Alberta;
USNM National Museum of Natural History (formerly United States National Museum), Washington, D.C.
DNA extraction, PCR amplification, and sequencing of the COI barcode region were performed at the Canadian Centre for DNA Barcoding (CCDB) and followed standard protocols (
Morphology. Comparison of 20 genitalia dissections of each sex, representing all geographic entities, failed to reveal any diagnostic differences. The shape of the male valve apex and clasper varied slightly, but do so even within a single population. The shape of the inflated vesica, the most important diagnostic character in many noctuid species complexes, showed no discernible differences. Female genitalia were similarly conservative in variation. The European Raphia hybris Hübner, which is externally very similar to Raphia frater (and is in fact virtually indistinguishable from some Raphia frater coloradensis phenotypes), differs from Raphia frater in valve shape, vesica structure (including presence of spinules that are absent in Raphia frater) and shape and size of the corpus bursae. This indicates that Raphia genitalic morphology is not unusually homogeneous, where interspecific differences might be lacking.
The North American Raphia species have previously been delineated based on wing colour and pattern (
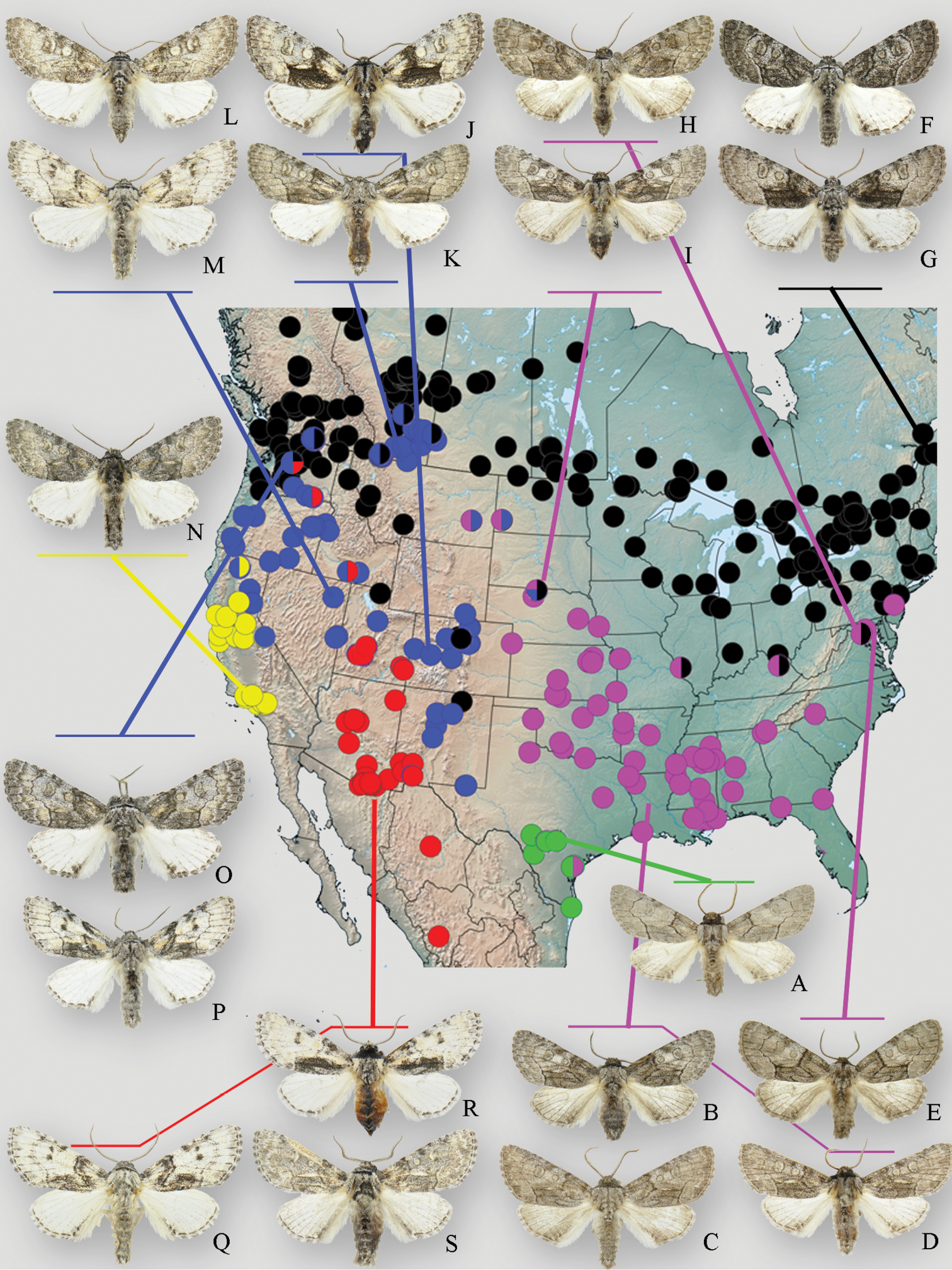
Geographic distribution and phenotypic variation of Raphia frater subspecies. Circles indicate specimens examined during this study: green – subsp. piazzi; pink – subsp. abrupta; black – subsp. frater; blue – subsp. coloradensis; yellow – subsp. cinderella. Multi-coloured circles indicate transitional populations and/or phenotypically intermediate specimens between respective subspecies. a Raphia frater piazzi (Zavallo Co., TX) b Raphia frater abrupta (Oktibeha Co., MS) c Raphia frater abrupta (Cottle Co., TX) d Raphia frater abrupta (Cottle Co., TX) e Raphia frater abrupta (Montgomery Co., MD) f, g Raphia frater frater (Edmunston, NB) h Raphia frater abrupta – frater intermediate (Anne Arundel Co., MD) i Raphia frater abrupta – frater – coloradensis intermediate from highly variable population in Cherry Co., NE j Raphia frater coloradensis (Alamosa Co., CO) k Raphia frater coloradensis (Milk River valley, AB) l Raphia frater coloradensis (Sanpete Co., UT) m Raphia frater coloradensis (Elko Co., NV) n Raphia frater cinderella (Ventura Co., CA) o, p Raphia frater coloradensis – frater intermediates (Chelan Co., WA) q Raphia frater elbea (Cochise Co., AZ) r Raphia frater elbea (San Juan Co., UT) s Raphia frater elbea (Santa Cruz Co., AZ). All specimens are males.
Raphia has an extensive North American distribution, occupying virtually all biomes. Phenotypes are generally quite consistent regionally, but can appear drastically different in geographically disparate areas, which led early authors such as
In the central Great Plains, a large series of over 60 specimens from northern Nebraska (Cherry Co.) is so variable that scarcely two individuals are alike, varying from the granular, dark grey forewing and white hindwing of ssp. frater, to the even, light grey forewing and slightly fuscous hindwing of Raphia frater abrupta; an intermediate specimen is shown in Fig. 1i. Some individuals show the blotchy black and grey pattern (with a contrasting black medio-anal shade) characteristic of Raphia frater coloradensis. A shorter series from Kansas (Riley Co.) falls within the variation of the Nebraska population. Interestingly,
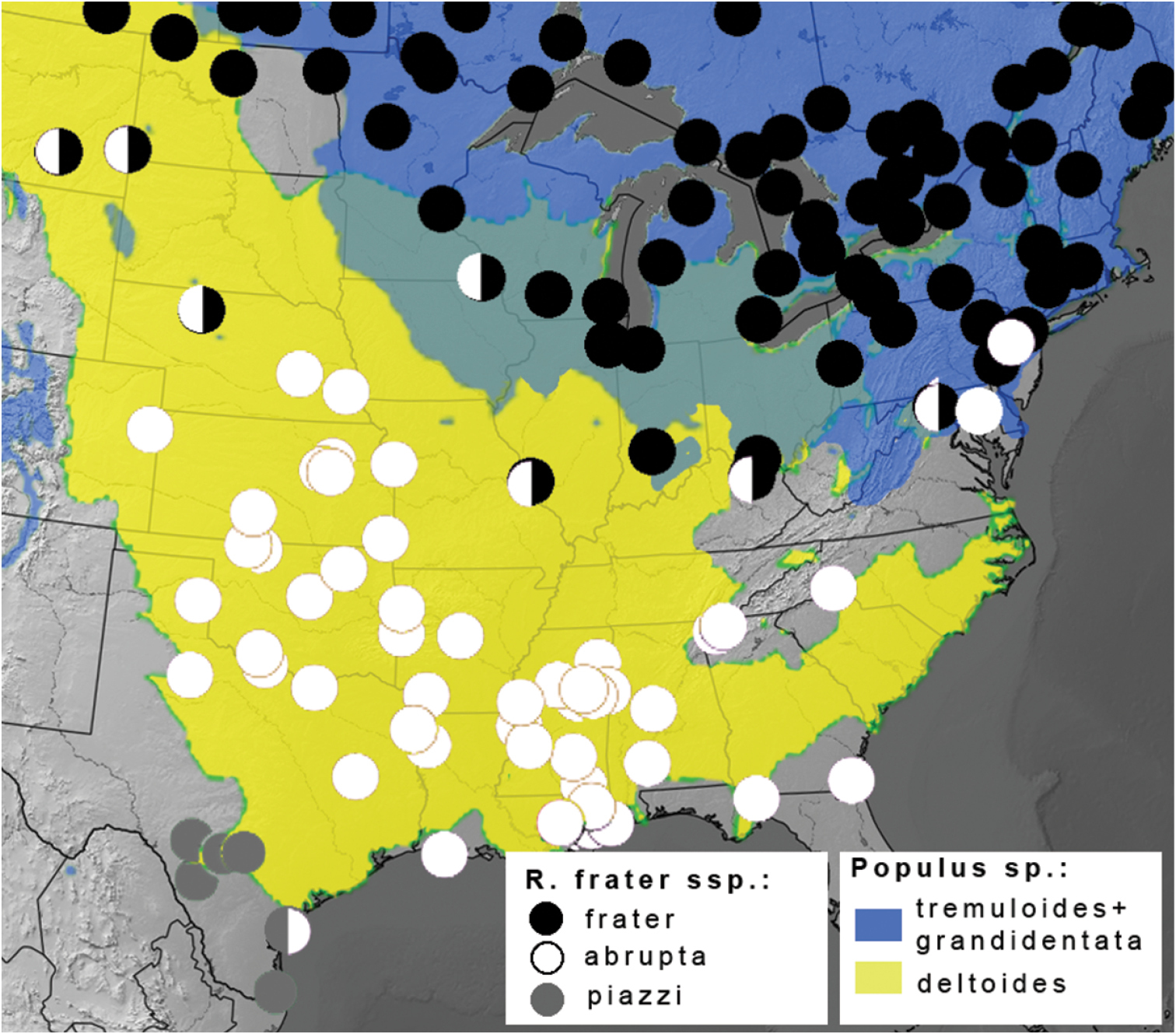
The nature of the ssp. abrupta – frater interface is somewhat different in the Northeast, and is seemingly more influenced by topography and host plant distribution (Fig. 2); at least three Populus species occur regionally among topography ranging from coastal floodplains to the Appalachian Mountains. Specimens from the Pocono Mtns. of Pennsylvania are Raphia frater frater, whereas nearby central Maryland (Ann Arundel Co.) specimens (Fig. 1h) show transitional features in having a forewing pattern much like ssp. frater, but with a fuscous hindwing and a darker prothoracic collar characteristic of ssp. abrupta. Coastal Maryland (Montgomery Co.) specimens are typical Raphia frater abrupta (Fig. 1e). As discussed below, the transition zone between ssp. frater and ssp. abrupta seems to be mediated by habitat and host plant differences, with frater largely associated with aspen (Populus tremuloides Michx. and Populus grandidentata Michx.; Fig. 2) and ssp. abrupta with cottonwood (Populus deltoides; Fig. 2). Study of the populations on either side of the Ohio River is needed because frater occurs throughout Ohio (
Distribution of Raphia frater subspecies (circles) relative to range of Populus larval host plants (coloured shading) in eastern North America. Black circles - subsp. frater; white circles – subsp. abrupta; grey circles – subsp. piazzi; half-circles represent transitional populations and/or phenotypically intermediate specimens. Blue shading – combined ranges of Populus tremuloides and Populus grandidentata; yellow shading – Populus deltoides; range overlap depicted in green. Populus ranges adapted from
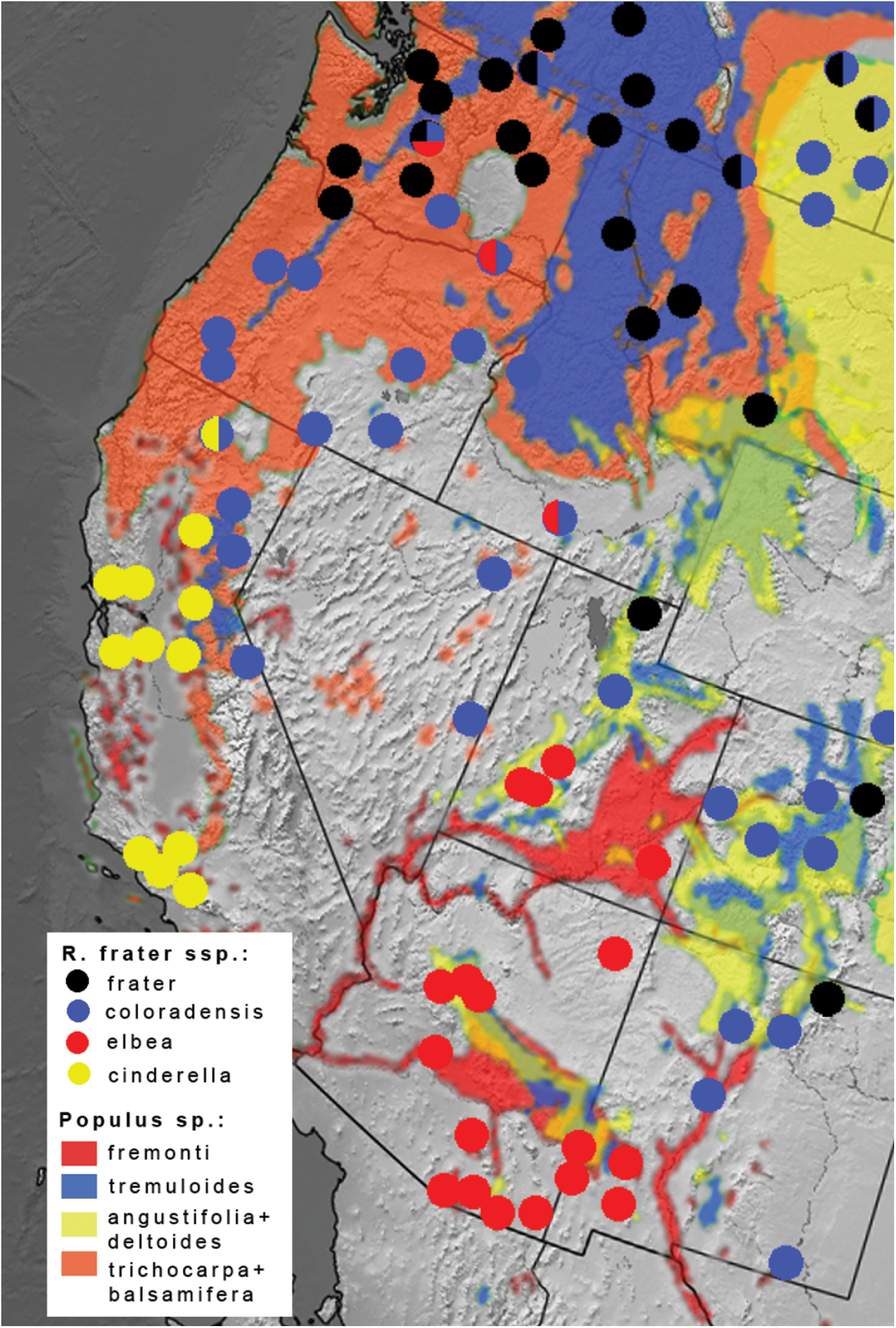
In southern New Mexico where elbea and coloradensis meet, elbea is known from the Mimbres Mountains (Grant Co.) in the southwest, with the nearest documented coloradensis locations 180 km to the northeast in the Rio Grande valley, and 300 km to the east in Eddy County (Fig. 1). The phenotypic transition between the two taxa is more abrupt in southern New Mexico than it is in the Great Basin where elbea imperceptibly transitions to the very pale Great Basin forms of coloradensis. A series from Twin Falls, Idaho, and some individuals from Leavenworth, Washington (Fig. 1p), are indistinguishable from Arizona elbea (Fig. 1q). Raphia frater elbea therefore grades into Raphia frater coloradensis in low-elevation habitats of the northern Great Basin. Specimens from the Eddy Co., New Mexico population are most like coloradensis, but some individuals are again indistinguishable from elbea. The mtDNA haplotypes associate this population with coloradensis (Fig. 3), and the available larval hosts are Populus angustifolia James and Populus deltoides (
Distribution of Raphia frater subspecies (circles) relative to range of Populus species (coloured shading) in western North America. Half-circles represent transitional populations and/or phenotypically intermediate specimens. Ranges for Populus trichocarpa + Populus balsamifera and Populus angustifolia + Populus deltoides are combined, with both Populus balsamifera and Populus deltoides occuring in Alberta – Montana (upper right). Populus ranges adapted from
Populations at the edges of the Great Basin can be extremely variable, much like the situation between abrupta and frater in the north-central Great Plains. Series of specimens from Waterton Lakes, Alberta; Okanagan Valley, British Columbia; Baker County, Oregon; and Leavenworth, Washington range from the typical dark grey frater to pale yellowish coloradensis (Fig. 1). The Leavenworth population is remarkable in that it exhibits phenotypes ranging from frater to coloradensis (Fig. 1o) and elbea (Fig. 1p).
The geographic structure of California populations is not well documented; typical cinderella occurs from the San Francisco Bay area southward through the Central Valley to Los Angeles Co., but Raphia frater is apparently absent from southeastern California and the southern Sierra Nevada. Northern California (including the Sierra Nevada) specimens are most like Great Basin coloradensis but the transition from cinderella to this paler form is subtle, with Siskiyou Mountains material appearing intermediate. The Siskiyou Mountains are part of a northern California – southern Oregon suture zone also identified for other flora and fauna (see
Extreme phenotypic variation is therefore the modal geographic pattern at suture zones. In all cases where we examined suture zones between putative taxa, phenotype variation was moderate to extreme, and specimens could not consistently be assigned to existing taxonomic categories. Similarly, mtDNA variation patterns show no evidence of sympatric, reproductively isolated taxa, as discussed below.
Host plants. 72% of the 132 larval collections of Raphia frater from across Canada summarized by
Although it is reasonably certain that Raphia frater larvae are Salicaceae specialists, the geographic variation in host use and extent of specialization is not well understood. Nonetheless, it is possible to extrapolate broader host use patterns based on larval collections, host plant distributions and habitat associations. Below, we outline some potential scenarios of host use among Raphia frater subspecies.
Host plant records for Raphia frater frater (
Throughout most of the range of Raphia frater abrupta, Populus deltoides is the only Populus species present. Swamp cottonwood (Populus heterophylla) has a small eastern North American range, occurring primarily along the Mississippi and Ohio River valleys and along the Atlantic seaboard (see e.g.,
In the Pacific Northwest, Raphia frater frater is associated with Populus tremuloides in northern Washington and British Columbia, with Raphia frater coloradensis of dry, low-elevation habitats associated with Populus trichocarpa (L. Crabo, pers. comm.).
Arizona populations of Raphia frater elbea feed on Populus fremonti (
In summary, larval host plant associations of Raphia frater populations shows some broad congruencies between subspecies and Populus species distributions, but with limited evidence for high host fidelity: range edges of Raphia frater subspecies generally do not closely follow those of the various Populus hosts, suggesting that Populus availability rather than high host fidelity may be the limiting factor to Raphia distribution, and that climatic and topographic effects have a greater selective influence that does host plant specialization. To what extent these congruencies reflect common co-evolutionary trajectories, and what factors drive intraspecific divergence, would be a fascinating and fruitful area of study.
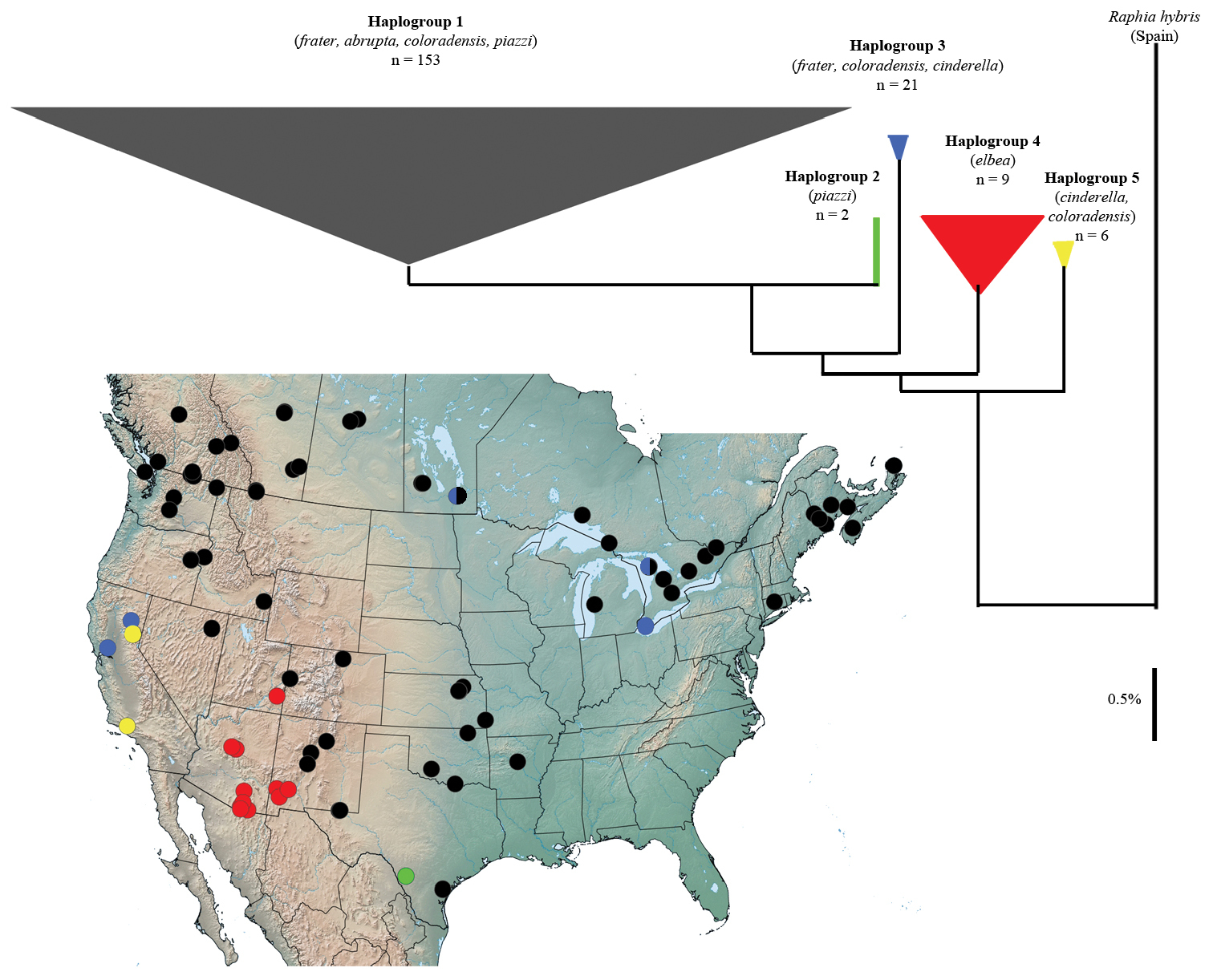
Molecular variation. The deepest splits in mtDNA barcode variation (alluded to previously in
Neighbour-joining tree and associated sampling sites of mtDNA barcode haplotypes in Raphia frater. Haplogroup colour corresponds to that of sampling sites. Subspecies assignment based on morphology and sample size is indicated for each haplogroup. Width of triangles is proportional to number of haplotypes, height represents maximum divergence within haplogroup.
The combination of California and eastern Canada samples in haplogroup 3 to the exclusion of all others was quite unexpected, given the geographic structuring of other haplogroups. Two haplogroup-3 populations (Bird Hill, Manitoba; Bruce Peninsula, Ontario) also exhibited group 1 haplotypes (Fig. 4), the only sampled populations to yield more than one haplogroup. Representative specimens from these sites were of the same phenotype and from the same sampling event. This haplogroup could therefore be a retained ancestral mtDNA polymorphism, or indicative of Wolbachia-induced mtDNA lineage sorting similar to that documented by
Considering the general lack of taxonomic resolution of North American Raphia in the barcode sequence, and comparing divergences among Palaearctic Raphia as a metric of mean species divergences within the genus, mtDNA variation is most parsimonious with a geographically structured, single-species interpretation. The contrast between often considerably different adult phenotypes and lack of significant mtDNA and morphological differentiation may reflect strong regional selection on bark-cryptic wing patterns, which in turn is dependent on dominant host trees that vary according to regional host preferences.
Noctua hybris Hübner, [1813] by subsequent designation by
Despite variation in adult facies and lack of a particular diagnostic trait, Raphia frater is recognizable by the combination of a broad, rounded forewing, often conspicuous antemedial and postmedial band, obsolete medial band (rarely faint), an orbicular, reniform and usually also a claviform stigma that are clearly outlined, black shading in the anal angle of the hindwing, and the conspicuously bipectinate male antennae. Pseudopanthea palata (Grote) and Colocasia Ochsenheimer species share some superficial similarities with Raphia frater, but attention to the above-stated characters relative to those in Pseudopanthea McDunnough and Colocasia Ochsenheimer will provide an easy diagnosis of this unique species.
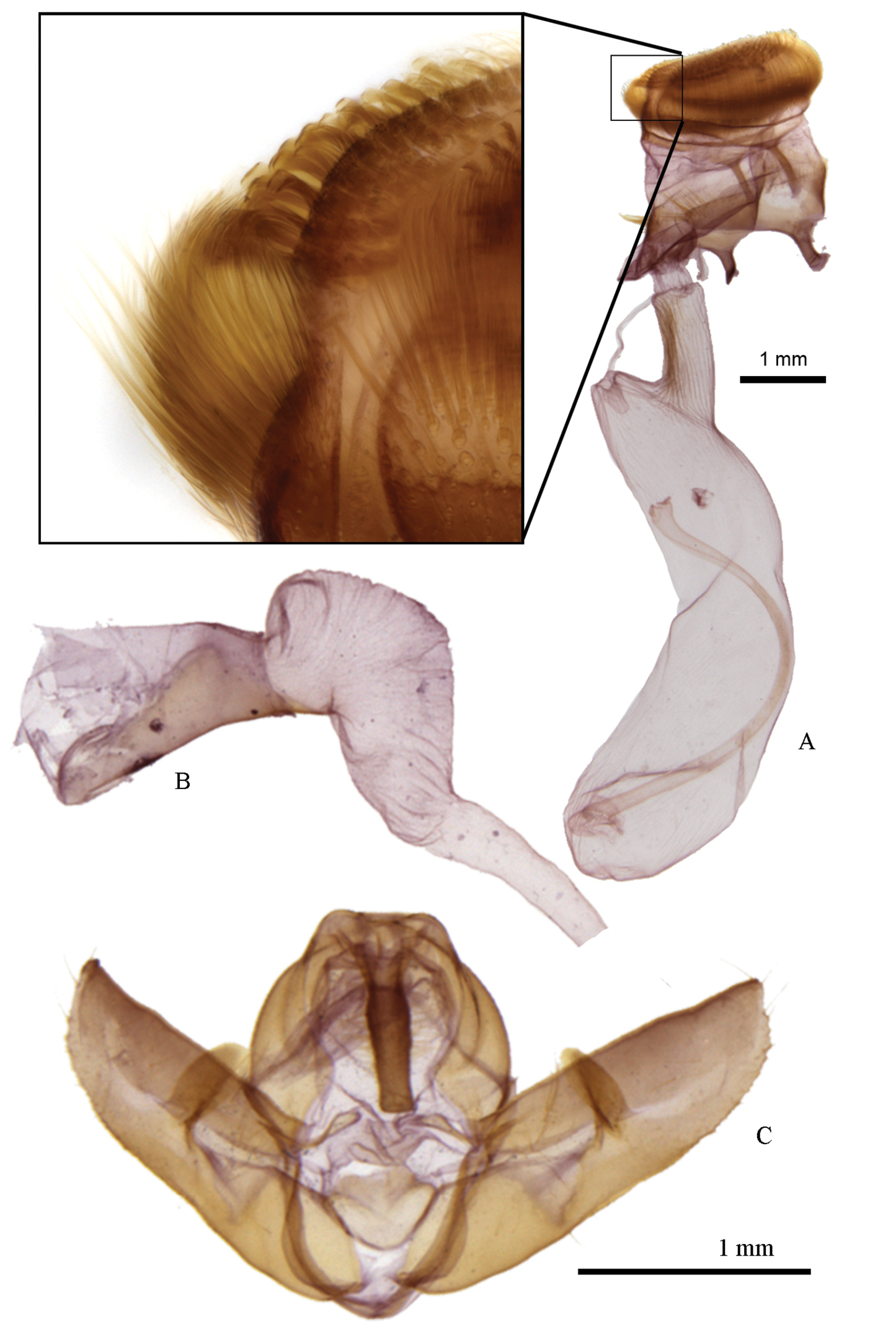
Head – Male antennae bipectinate, anterior rami 3× longer than segment length, posterior rami 3.3× longer; female antennae simple; eyes round, with short, sparse interfacetal setae, visible only at high magnification; labial palpus with second segment clothed in long strap-like scales ventrally; third segment 0.6 × length of second segment (when denuded) and smoothly scaled; occiput and frons with mix of grey and black scales, frons with transverse line of black scales; frons rounded and moderately protuberant when denuded. Thorax – vestiture dark grey to yellowish grey, thoracic collar sometimes contrastingly darker than dorsum; tarsi smoothly scaled, with transverse bands of black and light to dark grey; tibia with similar scaling but with faint or indistinct banding; femur with long, shaggy hair-like scales. Abdomen – lacking specialized secondary sexual structures such as coremata; vestiture of smooth, short grey scales; small, rounded dorsal tufts on segments A3, A4 and A5, consisting of densely set spatulate scales. Forewing – ground colour varies from a dark charcoal grey to pale yellowish ochre; antemedial band a parallel-sided, double black line, varying from slightly irregular and rounded to nearly linear, acute, and angled at the cubital vein; medial band obsolete, usually reduced to a black bar or two diffuse lines at costa adjacent to reniform stigma, but band sometimes visible as a faint, diffuse black line extending from bottom of reniform stigma to anal margin; postmedial band a single black line, sinuate and slightly sagittate at veins (often faint or absent in ssp. coloradensis and elbea), expanding to diffuse black patch at costa; orbicular stigma paler than ground colour, with black border and often with a diffuse dark pupil (orbicular often absent entirely in ssp. coloradensis and elbea); reniform stigma paler than ground colour, with a black border (border often lacking in coloradensis and elbea) and a diffuse black central crescent; subterminal band absent, faint, or diffusely sagittate with paler distal edging; terminal area often darker grey than subterminal area. Average size is greatest in subspecies frater, while abrupta and piazzi are smallest; forewing length varies from 16.2 mm and 18.5 mm in male and female Raphia frater frater to 13.7 mm and 15.2 mm in male and female Raphia frater abrupta, respectively. Hindwing – ground colour varying from white, white and dusted with fuscous grey (ssp. frater, coloradensis, elbea, cinderella, piazzi), or entirely pale fuscous grey (ssp. abrupta), females with more fuscous than males; crescentic discal spot diffuse or absent; postmedial band faint or absent, although nearly always with a contiguous diffuse black patch at anal angle. Male genitalia (Fig. 5) – uncus slightly compressed dorsoventrally, with slight medial bulge, apex blunt; valva tapering more or less evenly to a rounded point, sacculus poorly differentiated from remaining valva; ampulla long and flattened, 0.7 × length of valva width, projecting mesially; aedeagus stout and sausage shaped, 2.1 × longer than wide; vesica a simple kidney-shaped, unarmed chamber equal in length to aedeagus, tapering gradually into ductus. Female genitalia (Fig. 5) – bursa copulatrix membranous, lacking apparent differential sclerotization, including ostium, ante- and postvaginal plate; ductus bursae a simple rugose tube, 3.3 × longer than diameter, connecting subbasally to corpus bursae; corpus bursae a simple kidney-shaped chamber, slighter wider over apical two-thirds; ductus seminalis arising from basal end of corpus bursae, caudad of ductus bursae; papillae anales short, bluntly rounded, with a broadly joined base, with two types of unusual, modified setae: 1) a dense band of thin, evenly curved setae arising from base of papillae and curving up to caudal margin, outer surface of lobe virtually encircled by a dense setal crown; and 2) highly modified thick, spatulate setae densely set along caudal margin of lobe. Immature stages – larva described by
Genitalia of Raphia frater. a female (Konza Prairie Biol. Stn., Riley Co., KS; leg. Metlevski), with inset showing apical spatulate setae and subapical dense setal crown of papillae anales b male vesica c male genital capsule (Pitchfork Ranch, Grant Co., NM; leg. C. Ferris); note magnification difference between sexes.
Biology and distribution. Raphia frater occurs in virtually all wooded or shrubby habitats of the boreal region since the larval hosts form a dominant part of most non-coniferous forest types. It can be one of the most common late spring noctuids in aspen-dominated boreal forests of central Canada. In the West it becomes increasingly more restricted to riparian areas, particularly major river systems in drier regions of the Pacific Northwest and the desert of the Southwest. Raphia frater has a nearly transcontinental distribution, absent only from the arctic and most of the subarctic. The records from northern subarctic Labrador are surprising, but are based on three CNC specimens from two localities, so the data appear to be authentic.
Raphia frater Grote, 1864 – # 7675 [ANSP]. Type locality: Middle States [eastern USA]; here restricted to Mount Pocono, Monroe Co., Pennsylvania.
The nominal subspecies of Raphia frater typically has an even, powdery, dark grey forewing ground colour with all of the markings complete, consisting of the antemedial and postmedial band, and the orbicular, reniform and usually the claviform stigmas. Average forewing length is 16.3 mm (n = 9) in males, 18.6 mm in females (n = 9). The male hindwing is white with little or no dusting of black scales in the subterminal area, and with a pronounced, diffusely-edged black patch in the anal angle, this often with an adjacent black line formed by the terminus of the postmedial band; females usually have some fuscous scales on the hindwing, especially on a slight postmedial band. This subspecies generally lacks the form with contrastingly darker medio-anal and costal black patches that is prevalent in Raphia frater coloradensis, but it does occur rarely even in Atlantic Canada (Fig. 1g). The yellowish-ochre forewing scales typical of Raphia frater coloradensis are absent. Raphia frater abrupta differs in having a more angulate and linear antemedial band, a paler grey and less powdery-appearing forewing, duskier hindwing, and smaller size. As discussed in the section on Raphia frater, geographically intermediate populations are extremely variable with respect to these traits, and are considered to be transitional between subspecies frater and coloradensis/abrupta, the only two subspecies abutting the range of Raphia frater frater.
Raphia frater frater is primarily a boreal taxon, especially common in aspen (Populus tremuloides and Populus grandidentata) dominated forests and the Aspen Parkland ecoregion of the Prairie Provinces. In the East, it extends south of the Great Lakes region into Pennsylvania, Ohio and Indiana, but apparently not southward into the southern Appalachians, which are essentially devoid of Raphia records. The transition zone between Raphia frater frater and Raphia frater abrupta extends from Maryland westward roughly along the Ohio River Valley to east-central Missouri, then northwestward through the northern Great Plains. The southeastern range edge of Raphia frater frater is virtually identical to that of both trembling and bigtooth aspens (Fig. 2). In the West, Raphia frater frater occurs south along mid-elevation mountain ranges of the Pacific Northwest into Washington, and southward along the Rocky Mountains. Specimens from high elevations in Colorado (Gilpin Co., 9500’) and New Mexico (Sangre de Cristo Mtns., 7900’) are of the typical frater phenotype, the coloradensis phenotypes occurring at lower elevations.
Raphia abrupta - female holotype # 7675 [ANSP]. Type locality: not given; here restricted to Sycamore Landing, Seneca, Montgomery Co., Maryland. The female type bears no locality or collector label data, and since this is a widespread, geographically variable taxon, we restrict the type locality to Sycamore Landing, Seneca, Montgomery Co., Maryland; a series in USNM from this locality, collected by D. C. Ferguson, is phenotypically more similar to the female type than specimens from the Great Plains; it is also more likley that the holotype originated from the eastern US rather than the Great Plains, which were not well collected in the mid 1800’s.
Certila flexuosa Walker - [BMNH; not examined]. Type locality: North America.
Raphia frater abrupta replaces Raphia frater frater from the central Great Plains eastward to the mid-Atlantic seaboard, and southward to eastern Texas and Florida. It is on average smaller with a more evenly-coloured forewing, a more linear, angulate antemedial band and a fuscous hindwing. Average forewing length is 13.7 mm (n = 9) in males, 15.2 mm in females (n = 9). The thoracic collar is often darker than the dorsal thorax, not concolorous as in Raphia frater frater. The wing facies of subspecies abrupta is in many ways intermediate between Raphia frater piazzi of central and southern Texas and Raphia frater frater to the north, but the exact nature of the interface between abrupta and piazzi in Texas remains unstudied.
Subspecies abrupta occurs south of the range of the aspen species favoured by Raphia frater frater larvae, and its riparian haunts suggest it feeds on eastern cottonwood (Populus deltoides), the only Populus species in much of its range. Swamp cottonwood (Populus heterophylla) and willows (Salix spp.) may also be suitable hosts. This subspecies is apparently rare on the Atlantic seaboard and absent altogether in the Appalachians. We examined only a single historical specimen from New Jersey (Trenton), with records north of there assignable to Raphia frater frater. All Ohio records were attributed to Raphia frater frater by
Holotype male [USNM]. Type locality: Brownsville, Texas [USA].
Raphia frater piazzi is the least-known member of the group with a restricted distribution in central and southern Texas. Most similar in size and facies to Raphia frater abrupta, it is distinguished from that subspecies by the paler, more evenly grey forewing with sharper transverse lines than in Raphia frater abrupta. The biology and biogeographic relationship to Raphia frater abrupta, which occurs to the northeast of piazzi’s range, is not known, and very few specimens of this taxon are present in collections. An additional enigma is whether or not Rio Grande piazzi populations interact with the vastly-different looking Sonoran Raphia frater elbea.
Described from southernmost Texas, this subspecies is otherwise known only from the Edwards Plateau region; a single specimen from Sinton County to the southeast is phenotypically intermediate between abrupta and piazzi, but clearly more field work is needed to establish the limits of both subspecies. mtDNA barcode data of three piazzi specimens (Sinton Co. and Zavalla Co.) are very similar to the haplotypes of Raphia frater abrupta, Raphia frater frater, and Raphia frater coloradensis.
We were unable to obtain DNA sequence from topotypical specimens of piazzi from the lower Rio Grande near Brownsville, Texas. The unique haplotype of the Edwards Plateau specimens (Fig. 4) may represent nominal piazzi, but could equally represent a unique genetic lineage from the Edwards Plateau, with its unique fauna much of which is not shared with the Rio Grande fauna.
Raphia frater var. coloradensis - Neotype female, here designated [CNC]. Type locality: Deer Creek Cyn. Park, 39°33.18'N, 105°08.49'W, 5950’, SW Littleton, Jefferson Co., Colorado. None of the original types, three males and four females “taken in Colorado by D. Bruce, ” could be located and are presumed lost. The primary type of Xylena thoracica Putnam-Cramer, the only other noctuid named by Putnam-Cramer, is housed at USNM. Prior to 1886, D. Bruce collected in the mountains and foothills near Denver (
Raphia pallula - Holotype female [AMNH]. Type locality: Siskiyou Co., California [USA]. Published several months after coloradensis Putnam-Cramer, Edwards was apparently not aware of Putnam-Cramer’s name as it is not mentioned in his description.
Within the range of coloradensis, specimens identical to the typical boreal Raphia frater frater are often present; in the most arid parts of the range of coloradensis in the southern Great Basin, coloradensis is more consistently pale ochre yellow with obsolete transverse lines and diffuse black costal/reniform blotches, overall very similar to elbea, but with less pronounced costal and reniform dark patches. Average forewing length is 14.9 mm (n = 9) in males, 16.8 mm in females (n = 6).
This subspecies occurs from southernmost British Columbia / Alberta to New Mexico, Utah, and California. It is most commonly associated with riparian, low-elevation habitats. Northern populations fly from late May to July in a single generation. Flight dates spanning from May into August in the Great Basin and Southern Rocky mountain region indicate a second or partial second generation.
Raphia frater coloradensis is the most weakly-differentiated subspecies, and may simply be an ecologically induced phenotype of Raphia frater frater that occurs in the warmer, drier regions of the West. Several populations, spanning a large geographical area, have been identified that exhibit a large range of phenotypic variation, as discussed above in the ‘Morphology’ section. Specimens from Siskiyou Co., California and the east slope of the northern Sierra Nevada (Sierra Co.) are phenotypically very similar to Great Basin coloradensis, and we therefore treat pallula as a junior subjective synonym. DNA barcodes of two specimens from the northern Sierra Nevada (Sierra Co.) belonged to the frater-coloradensis-abrupta haplogroup (Fig. 4).
A male lectotype was designated by
Raphia frater cinderella is a Californian subspecies that is similar in size and colour to Raphia frater coloradensis, but with a more diffuse, poorly contrasting forewing pattern that usually lacks the pronouncedly darker reniform and costal dark patches. The forewing ground colour is also pale powdery grey, not pale ochre as it often is in coloradensis. The two taxa appear to intergrade in the Siskiyous and northern Sierra Nevada.
The range of this subspecies is restricted to central and southern California west of the Sierra Nevada. Fremont Cottonwood and willows are the most likely larval hosts, although records specific to this subspecies are lacking. Most collection dates are from June; Records from Stanislaus Co. for April - May and July may indicate a second generation.
A male lectotype was designated by
Raphia frater elbea is most similar to the pale yellowish-ochre forms of Raphia frater coloradensis, but differ from that subspecies in having both the costal and reniform dark patches more prominent; when present, the black medio-anal patch is also darker and more elongate; additionally, Raphia frater elbea appears to exhibit a unique, divergent mtDNA haplotype group.
This subspecies occurs from southeastern Utah and western New Mexico southward through Arizona into northern Mexico. In southeastern Arizona it occurs in riparian areas in association with the larval host, Populus fremonti. Flight records are from February to October, with most being from March to May and August to September, indicating at least two generations annually.
The North American Raphia populations exhibit considerable geographic variation in phenotype, previously segregated into six species. Despite these geographically structured phenotypic differences, diagnostic morphological differences in genitalia and larvae are not evident. Scrutiny of geographic contact zones between putative taxa revealed populations with extensive phenotypic and conservative molecular variation, rather than bimodal phenotypic variation coupled with deep molecular divergences that would be expected for sympatric, reproductively isolated taxa. Raphia frater larvae are not highly restricted to a host species or genus, but do specialize on Populus and Salix, with a pattern of regional host availability and possibly also preference. Differences in host plant suitability among the various species of Salicaceae remain unstudied. Assessment of morphology, mtDNA variation, and biogeography therefore leads us to conclude that the geographic segregates of North American Raphia are best treated as subspecies of a single species. The regional adaptation to habitats representing nearly all North American biomes, combined with relatively discrete geographic ranges of unique adult phenotypes, suggest a pattern of young or incipient species in the Raphia frater group.
The taxonomy and biogeography of the North American Raphia populations is a complex interplay between topography, host plant use, phenotypic variation and evolutionary history. This study is only the first attempt at a better understanding of this interesting group. Many questions remain unanswered: what are the exact geospatial and host plant patterns of the contact zone between Raphia frater abrupta and Raphia frater frater? Is there geographic overlap with altitudinal segregation in the West between aspen-feeding frater and cottonwood feeding elbea? Does the mtDNA haplogroup 3 represent Wolbachia infection? Do the lower Rio Grande / Edwards Plateau piazzi populations grade into abrupta? Raphia would provide a fertile area of study in understanding large-scale patterns of host plant use and biogeography of a widely distributed continental Lepidopteran.
We thank James Adams, Charles Bordelon, Vernon A. Brou Jr., Richard Brown, Lars Crabo, Terhune Dickel, Cliff Ferris, Ed Knudson, Paul Opler, Jan Metlevski, Eric Metzler, Hugo Kons, Bruce Walsh and Chuck Harp for generously providing specimens and/or data used in this study, and Jocelyn Gill for preparing the specimen images. Discussions with Lars Crabo and Don Lafontaine on Raphia biogeography and species limits provided insight and guidance during this project. Evgeny Zakharov, Paul Hebert and other members of the Barcode of Life Project at the University of Guelph, Ontario, Canada, provided DNA data. Molecular analyses were carried out through grants from the National Science and Engineering Research Council of Canada and Genome Canada through the Ontario Genomics Institute.
Table S1. Specimen data for mtDNA barcode vouchers.
Authors: B. Christian Schmidt, Gary G. Anweiler
Data type: data spreadsheet
Explanation note: Haplogroup numbers refer to those given in Fig. 4; Abbreviations for specimen depositories are as given in Methods and materials section.
Copyright notice: This dataset is made available under the Open Database License (http://opendatacommons.org/licenses/odbl/1.0/). The Open Database License (ODbL) is a license agreement intended to allow users to freely share, modify, and use this Dataset while maintaining this same freedom for others, provided that the original source and author(s) are credited.