Citation: Davis SR, Engel MS (2014) A new genus of nemonychid weevil from Burmese amber (Coleoptera, Curculionoidea). ZooKeys 405: 127–138. doi: 10.3897/zookeys.405.6475
The first fossil nemonychid (Nemonychidae) in Burmese amber, belonging to the subfamily Rhinorhynchinae, is described and figured as Burmonyx zigrasi Davis & Engel, gen. n. and sp. n. While this specimen also comprises the first definitive record of the subfamily in the Asian continent, other compression fossils exist at least from the Yixian Formation of China and the Karatau site of Kazakhstan which may also deserve placement within this group. Although several important areas of the body are obscured by the shape and fragmented condition of the amber piece, a sufficient number of features are visible to consider adequate placement within Rhinorhynchinae, including the fairly strongly punctate elytral striae and appendiculate, nearly bifid pretarsal claws.
Coleoptera, amber, Cretaceous, weevils, Nemonychidae, Mesozoic, taxonomy
The origin and evolution of weevils (Curculionoidea) remains one of the more interesting and challenging areas of systematic research among the Coleoptera (
Thorough accounts of the Mesozoic fossil record of Nemonychidae (and of the superfamily Curculionoidea) have been provided and reiterated several times (e.g.,
Photomicrographs (1, 2, 4, 5) and line drawings (3, 6) of holotype of Burmonyx zigrasi Davis and Engel, sp. n. (JZC-Bu228). 1 Dorso-lateral view of specimen inclusion, arrow pointing to antennal scape 2 Slightly more dorsal view of specimen than in figure 1, arrow pointing to scutellary striole 3 Line drawing of specimen (scale bar only applies to this figure) 4 Legs, arrows pointing to appendiculate, nearly bifid pretarsal claws 5 Enlargement of legs, arrows pointing to metatibia and metatarsomeres 6 Illustration of metatarsus.
The amber piece containing the holotype was excavated from the strata in the northern state of Kachin in Myanmar as part of regular and ongoing mining operations and is from the collection of Mr. James S. Zigras, available for study through the American Museum of Natural History (AMNH), New York. The origin, age, and fauna of Burmese amber have been reviewed by
Burmonyx zigrasi Davis & Engel, sp. n. Gender feminine.
The new genus appears similar to several genera in Rhinorhynchinae, particularly to those of Rhinorhynchini, due to the fairly long, narrow rostrum (Figs 1, 3) and moderately wide pronotum. It appears to be differentiated based mainly on its elongate scutellary striole. The striole in extant Nemonychidae always is fairly short and not extending more than 0.25 × the length of the elytra from the base. This genus, however, possesses a striole that extends approximately to the middle of the elytron. The relatively large strial punctures on the elytra may also be a defining feature of this genus, as the punctures typically are quite small in other members of Rhinorhynchinae.
The genus name is a combination of the specimen’s collection locality, Burma (former name of Myanmar), and the Greek nyx, meaning “night”.
http://zoobank.org/15B9C6E0-166E-4D25-A142-5B8E12517468
http://species-id.net/wiki/Burmonyx_zigrasi
Figs 1–7JZC-Bu228, Myanmar: Kachin; Cretaceous: Early Cenomanian; in the private collection of Mr. James S. Zigras, available for study through the Division of Invertebrate Zoology, American Museum of Natural History (AMNH), New York, USA. Material was obtained from ongoing excavations in mines in the Hukawng Valley, northern Myanmar (
As for the genus (vide supra).
Total body length (excluding rostrum): ca. 2.2 mm; maximal width (along middle of elytra): ca. 0.7 mm; elytral length: ca. 1.3 mm. Integument appearing light to dark brown (Figs 1, 2, 7). Scales absent, but dense covering of setae along at least pronotum and elytra (lateral and ventral surfaces not clearly visible). Head and compound eyes not clearly visible (slightly pushed into and obscured by prothorax). Rostrum approximately as long as pronotum along middle (exact length ratio unclear due to obscured head), fairly slender, abruptly widening apically (Fig. 3). Mandibles large, falciform. Antennae orthocerous, inserted dorso-laterally at apical 1/4; clubs composed of 3 loose articles. Pronotum seemingly as wide or nearly as wide as elytral humeri; not constricted anteriorly at collar, slightly rugulose, bearing small, dense, shallow punctures. Mesoscutellum not visible. Elytra with ten shallowly punctate striae (Figs 2, 3, 7); scutellary striole present, extending approximately to mid-length of elytra; interstices lacking punctures; elytral shoulders rounded. Abdomen with pygidium (tergite VII) concealed. Legs approximately equal in length, slender; femora slender; trochanters small, triangular; tibial spur formula 2-2-2; tarsomere 1 rather narrow, elongate (Fig. 6), approximately 2 × as long as tarsomere 2, 2 with rounded apico-lateral margins; 3 strongly bilobed (Figs 5, 6), lobes narrow; 4 short, slightly longer than 0.5 × length of tarsomere 3; 5 slender, approximately 2 × as long as tarsomere 2; pretarsal claws (ungues) divaricate strongly appendiculate, nearly bifid (Figs 4, 6).
Photomicrographs of nemonychid elytra (dorsal aspect). 7 Burmonyx zigrasi Davis and Engel, sp. n. 8 Nemonyx lepturoides (Fabricius, 1801) 9 Cimberis elongata (LeConte, 1876) 10 Doydirhynchus austriacus (Olivier, 1807) 11 Basiliorhinus araucariae Kuschel, 1994 12 Nannomacer germaini (Voss, 1952) 13 Rhinorhynchus rufulus (Broun, 1880).
The specific epithet is dedicated to the collector, Mr. James S. Zigras, who permitted study of the material and has generously supported amber research.
Presence of scutellary strioles excludes a placement of Burmonyx in Caridae, Brentidae, and Curculionidae. However, as the dorsal area encompassing the elytral suture is somewhat difficult to observe, if the presence of strioles represents a misinterpretation of complete striae, a superficial resemblance to the group Mesophyletinae
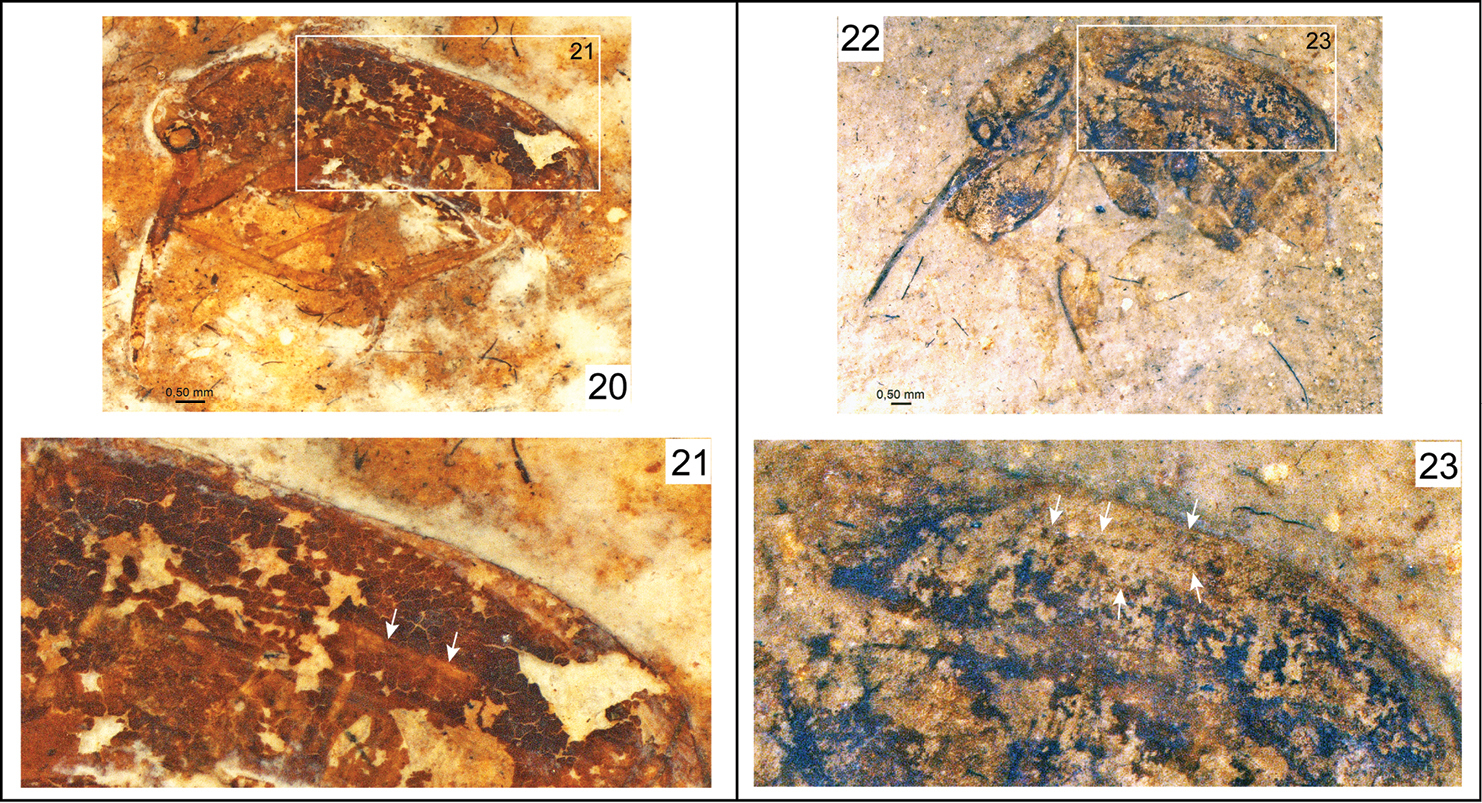
In Nemonychidae, as members of Nemonychinae do not possess distinctly punctate striae (Fig. 8), those of Cimberidinae lack striae (Figs 9–10), and both groups lack elytral strioles, Burmonyx does not belong within those subfamilies and a possible placing within Rhinorhynchinae or Eobelinae remains. Eobelinae are a difficult assemblage of taxa to comprehend in the least, and it is yet rather unclear how these different groups relate to the extant nemonychid fauna. It is clear that many of the taxa, which appear to have simple, divaricate pretarsal claws and a rostrum emerging from the ventral part of the head capsule, also bear distinctly punctate elytral striae, and may also bear scutellary strioles (Table 1; Figs 20–29). It also is interesting to note that there are some undescribed taxa from the Jurassic deposits of Karatau (Kazakhstan) and Daohugou (Inner Mongolia, China) that have a dense scattering of elytral punctures and lack elytral striae (appearing to also lack scutellary strioles), similar to extant Cimberidinae. Nonetheless, Burmonyx can be excluded from Eobelinae based on its appendiculate, almost bifid pretarsal claws.
Exemplar taxa sampled from the four recognized subfamilies of Nemonychidae (
| Subfamily: Tribe | Species | Elytron puncture type |
|---|---|---|
| Nemonychinae | Nemonyx lepturoides (Fabricius, 1801) | Punctures scattered, striae present, faint; striole absent |
| Cimberidinae: Cimberidini | Cimberis elongata (LeConte, 1876) | Punctures scattered, striae absent; striole absent |
| Doydirhynchini | Doydirhynchus austriacus (Olivier, 1807) | Punctures scattered, striae absent; striole absent |
| Rhinorhynchinae: Rhinorhynchini | Basiliorhinus araucariae Kuschel, 1994 | Punctures aligned into striae; striole present |
| Nannomacer germaini (Voss, 1952) | Punctures aligned into striae; striole present | |
| Rhinorhynchus rufulus (Broun, 1880) | Punctures aligned into striae; striole present | |
| Rhynchitomacer flavus Voss, 1937 | Punctures aligned into striae; striole present | |
| Rhinorhynchini? | Burmonyx zigrasi Davis & Engel, sp. n. | Punctures aligned into striae; striole present |
| Mecomacerini | Aragomacer leai Kuschel, 1994 | Punctures aligned into striae; striole present |
| Mecomacer scambus Kuschel, 1954 | Punctures aligned into striae; striole present | |
| Brarus mystes Kuschel, 1997 | Punctures aligned into striae; striole present | |
| Rhynchitomacerinus kuscheli (Voss, 1952) | Punctures aligned into striae; striole present | |
| Rhynchitoplesius eximius (Voss, 1952) | Punctures aligned into striae; striole present | |
| Eobelinae: Eobelini | Eobelus longipes Arnol’di, 1977 | Punctures aligned into striae; striole present |
| Archaeorrhynchus paradoxopus Arnol’di, 1977 | Punctures aligned into striae; striole present | |
| Oxycorynoidini | Oxycorynoides similis Arnol’di, 1977 | Punctures aligned into striae; striole present |
| Brenthorrhinoidini | Brenthorrhinoides mandibulatus Gratshev & Zherikhin, 1996 | Punctures aligned into striae; striole present |
| Distenorrhinini | Distenorrhinus spp. | Punctures aligned into striae; striole present |
Photomicrographs of nemonychid elytra (dorsal aspect). 14 Rhynchitomacer flavus Voss, 1937 15 Aragomacer leai Kuschel, 1994 16 Mecomacer scambus Kuschel, 1954 17 Brarus mystes Kuschel, 1997 18 Rhynchitomacerinus kuscheli (Voss, 1952) 19 Rhynchitoplesius eximius (Voss, 1952).
Photomicrographs of fossil nemonychid taxa and their elytra. 20 Eobelus longipes Arnol’di, 1977 (holotype), PIN 2452-275 21 Enlargement of outlined elytron in Figure 20 22 Archaeorrhynchus paradoxopus Arnol’di, 1977 (holotype), PIN 2335-42 23 Enlargement of outlined elytron in Figure 22.
Photomicrographs of fossil nemonychid taxa and their elytra. 24 Oxycorynoides similis Arnol’di, 1977 (holotype), PIN 2554-713 25 Enlargement of outlined elytron in Figure 24 26 Brenthorrhinoides mandibulatus Gratshev and Zherikhin, 1996 (holotype), PIN 2239-1508 27 Enlargement of outlined elytron in Figure 26 28 Distenorrhinus sp., PIN 2239-1547 29 Enlargement of outlined elytron in Figure 28.
The only nemonychids described from amber inclusions thus far include Kuschelomacer kerneggeri
Much appreciation is given to James S. Zigras who provided the opportunity to study this specimen and has generally supported amber research at the AMNH. Partial support for this work was provided by the Byers Opportunity Fund, Division of Entomology, University of Kansas Natural History Museum (to SRD), and US National Science Foundation grants DEB-0542909 (to MSE) and DEB-1110590 (to MSE, P. Cartwright, and SRD). This is a contribution of the Division of Entomology, University of Kansas Natural History Museum.