(C) 2013 Shanshan Liu. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Liu S, Ren G, Merkl O (2013) Redescription and new records of Ulomimus indicus Bates, 1873 (Coleoptera, Tenebrionidae, Tenebrioninae). ZooKeys 357: 45–51. doi: 10.3897/zookeys.357.6402
Ulomimus indicus Bates, 1873 of the tribe Ulomini is recorded for the first time from China (Guangxi and Hainan), Indonesia (Sumatra) and Thailand (Chiang Dao). A redescription of the male and the first description of the female are also provided.
China, Coleoptera, Indonesia, new country records, Tenebrionidae, Thailand, Ulomimus, Ulomini
Ulomimus indicus and the monotypic genus Ulomimus were described by
Ulomimus is a member of the tribe Ulomini. This tribe is characterized by the presence of placoid sensoria on the antennae, the primitive (“lagrioid”) type of labrum and the exposed posterior part of the 7th abdominal tergite (
Ulomimus is very similar and closely related to the Oriental genus Cneocnemis Gebien, 1914. In Ulomimus the antennomere 6 is transverse and bears one placoid sensorium (antennomere 6 is subequal in length and width and without placoid sensorium in Cneocnemis), the pronotum is widest in anterior 1/3 (widest at base in Cneocnemis), and apicale of aedeagus with small oblique lateral notch (without notch in Cneocnemis).
These two genera are similar to the widely distributed genus Uloma. The antennomeres 5 to 10 of Uloma are strongly transverse, forming a more or less distinct club, and have several placoid sensoria on their distal edge, which are arranged in complete rings on antennomeres 7 to 10. Antennae of Cneocnemis and Ulomimus are more elongate, only the antennomeres 6 or 7 to 10 are transverse (much less than in Uloma), the placoid sensoria are fewer in number, and do not form complete rings. The pronotum of most Uloma species is sexually dimorphic – males have large anterior impression and low protuberances. The pronotum of the males and females are the same in Cneocnemis and Ulomimus.
In the present paper, the distribution range of the species is revised through the inclusion of new records from China, Indonesia and Thailand. A redescription of the male and a description of the female (including diagnostic features) are also provided.
The illustrations were made using a Nikon SMZ800 dissecting microscope (equipped with a camera lucida). The photos were taken with a Leica M205A stereomicroscope equipped with a Leica DFC 450 digital microscope camera. All measurements were made in millimeter. The specimens examined are deposited in the Museum of Hebei University (MHBU), Baoding, China, in the Hungarian Natural History Museum, Budapest, Hungary (HNHM) and in the National Museum of Nature and Science, Tokyo (now in the Masumoto Collection, NMNS).
http://species-id.net/wiki/Ulomimus
Ulomimus indicus Bates, 1873.
Antennae short, antennomeres 6 to 10 gradually widened, 11 large, nearly globose. Pronotum with narrow and complete basal bead. Protarsi dilated with a brush of dense short hairs beneath, and protarsomere 4 much smaller than 2 and 3. Protibia strongly triangularly widened near apex, with large tooth on ventral surface. Aedeagus linearly truncate at apex in dorsal view, parameres with a small notch behind each apical corner.
The original spelling of the generic name is Ulomimimus.
It is unknow for the authors of the present paper who synonymised Pseuduloma with Ulomimus.
http://species-id.net/wiki/Ulomimus_indicus
Figs 1–13“Oblong, sub–parallel, moderately convex; brownish–black, shining, the mentum, antenna, palpi, tarsi, labrum, and margins of epistoma ferruginous, the legs chestnut–red; head coarsely and closely reticulate–punctate; prothorax punctured–sparsely on the disc–the punctures large, deep, rounded, and partly filled in with an apparent exudation of an ashy tint; scutellum smooth; elytra with nine (including the extreme marginal one) fine but deep striae, and a short scutellar one, the striae punctured (the punctures being much wider than the striae, the elytra appear crenulate–striate), the 4th and 5th striae shortest and united at some distance from the apex; intervals convex posteriorly, very minutely and sparsely punctured; pro– and mesosterna, flanks of pro– and mesothorax, and base of epipleural fold, strongly and closely punctured; metasternum, abdomen, and femora sparingly punctured, abdominal joints rugulose at the base. Long. corp. 4 lin.
Hab.: East India; one example.”
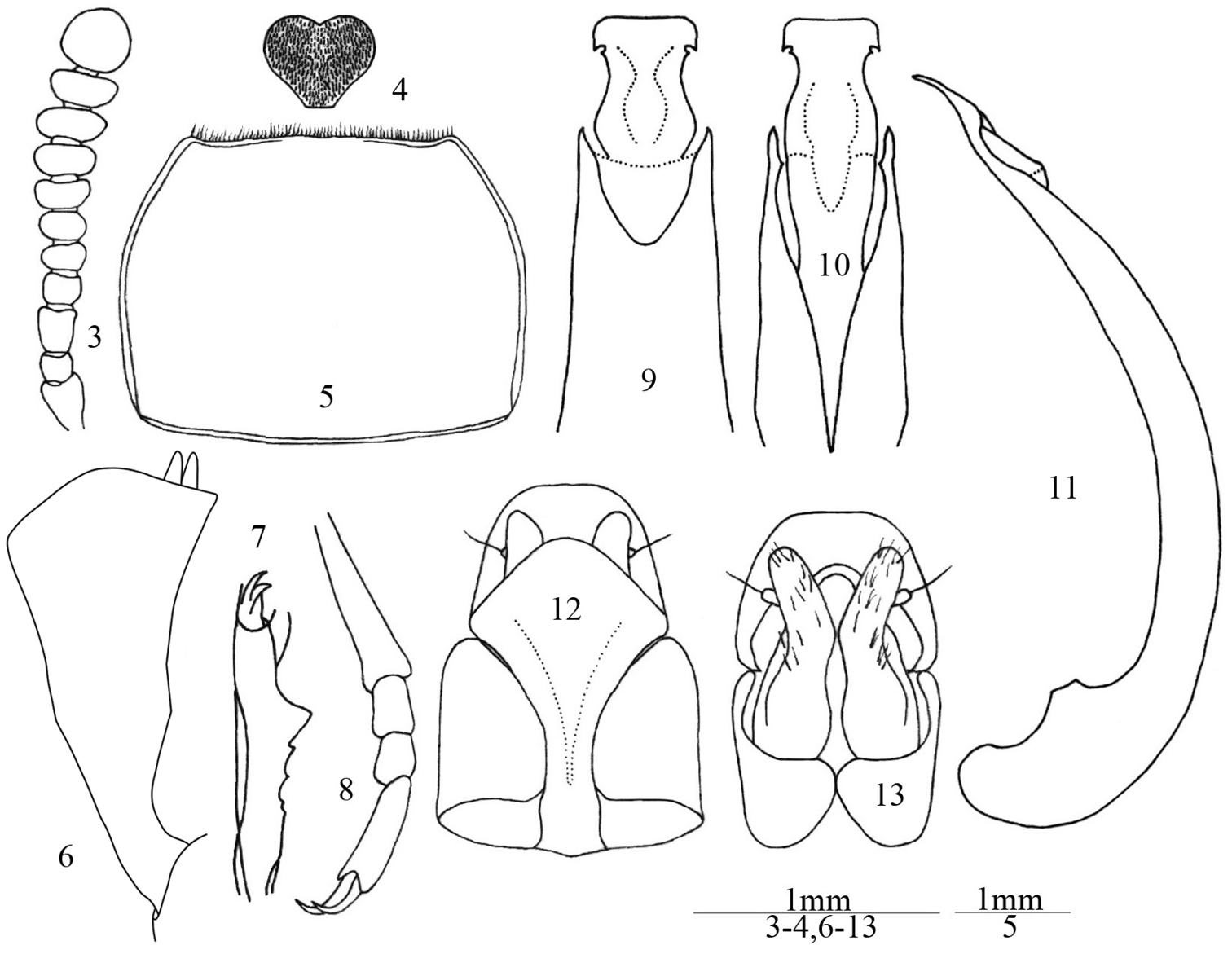
Male. Body length 8.0–9.0 mm; width 3.5–4.0 mm. Body (Fig. 1) elongate, elliptical, black or dark brown. Antennae, mouthparts and legs slightly paler. Head transverse, with small punctures on anterior half, and with sparse large punctures on posterior half; labrum transversely rectangular, densely punctate, scattered with short and yellow hairs; clypeus densely punctate, anterior margin truncate; frontoclypeal suture deeply impressed; genae feebly convex and slightly extended, temples reduced; eyes transverse, with 5–6 facets at narrowest point in lateral view; frons weakly convex, with large punctures; mentum (Fig. 4) cordate, truncate basally, with short and very dense yellow pubescence; ligula deeply emarginate anteriorly, depressed in middle; maxillary palp with narrowly trapezoidal terminal palpomere. Antennae (Fig. 3) short, not reaching half of pronotum; antennomere 1 thick, 2 very short, 3 long and narrow, 4 and 5 short, 6 to 10 gradually widening, 11 nearly globose, ratio of the length (width) of antennomeres 2–11 as follows: 4 (6): 8 (6): 5 (7): 5 (7): 5 (8): 4 (9): 5 (10): 5 (12): 5 (11): 11 (11). Antennomere 6 with one placoid sensorium on inner anterior corner, 7 with two placoid sensoria on inner corner, 8 to 9 with a few on inner and outer corners.
Ulomimus indicus Bates, 1873. 1 Male 2 female.
Ulomimus indicus Bates, 1873. 3 Antenna, male, dorsal view 4 mentum, male, ventral view 5 pronotum, male, dorsal view 6 protibia, male, dorsal view 7 protibia, male, lateral view 8 metatarsus, male, dorsal view 9 apical aedeagus, dorsal view 10 apical aedeagus, ventral view 11 aedeagus, lateral view 12 ovipositor, dorsal view 13 ovipositor, ventral view.
Pronotum (Fig. 5) transverse, about 1.35 times as wide as long, widest at anterior 1/3, with large punctures widely spaced in middle but becoming denser toward sides; anterior margin emarginate with narrow bead interrupted in middle, and with dense short hair fringes; lateral margins arcuate, strongly narrowing forward and less so from widest point to base, with narrow bead; basal margin slightly convex, with narrow bead; anterior angles nearly rectangular, posterior angles obtuse. Prosternum with sparse and large punctures, prosternal process rounded in lateral view, with small subapical tubercle. Mesoventrite with deep triangular impression; mesepisternum, metepimeron and metepisternum coarsely and sparsely punctate.
Scutellum triangular, impunctate. Elytra distinctly punctato–striate, intervals weakly convex, very finely and sparsely punctate, lateral margins visible only at humeri in dorsal view. Epipleura sparsely and coarsely punctate in basal 1/3.
Protibia (Figs 6–7) with two equal apical spurs; narrow at base, then explanate on both inner and outer edges, outer edge forming blunt subapical angulation, inner edge slightly concave at middle; outer edge without denticulation; inner edge fringed with yellow hairs becoming denser and longer toward apex; dorsal surface with low and blunt longitudinal keel and with fine and sparse punctures; ventral surface with sharp longitudinal keel and sharp tooth at middle (Fig. 7), ventral surface with a few coarse punctures and short, sparse, thick hairs. Protarsomeres 2 to 4 dilated, with long, sparse yellow dorsal hairs and dense, yellow ventral hair pads. Mesotibia and metatibia gradually dilated toward apex, outer edge with small denticles and sparse, long hairs. Length ratio of metatarsomeres 1 to 4 (Fig. 8) as follows: 10: 3: 3: 7.
Abdominal ventrites finely and sparsely punctate, punctuation denser and subcontiguous toward lateral portions; last ventrite with deep apical groove.
Aedeagus (Figs 9–11) with basale parallel–sided; apicale broad at base, constricted at middle, widening and truncate at apex in dorsal view, with longitudinal depression in ventral view, slightly curved in lateral view; with small oblique notch at posterior corners of widened apical part.
Female (Fig. 2). Mentum cordate, without dense pad of pubescence, but with sparse hairs and coarse wrinkles. Protibia with shape similar to or narrower than that of male, ventral surface concave, without keel and large tooth. Protarsomeres 2 to 4 not dilated and without ventral hair pads. Ovipositor (Figs 12–13) with coxites relatively smooth, bearing long sensorial hairs and a few short hairs at base.
1♀ (MHBU): China, Guangxi, Tian’e County, 14 September 2002, M. Bai leg; 1♂, 1♀ (HNHM), 2♂♂, 2♀♀ (MHBU): China, Hainan, Baisha County, Nankai Town, Shenbo Village, 1 June 2007, Y. B. Ba leg; 1♀ (NMNS): Thailand, Chiang Dao Hill Resort, 10–11 November 2012, K. Masumoto & K. Takahashi leg; 1♀ (HNHM): Indonesia, Sumatra, Dolok Merangir, 25 June 1970, collector unknown; 1♂ (HNHM): Indonesia, Sumatra, Palembang, date unknown, W. Knappert leg.
“East India” (
We are grateful to Dr. Wolfgang Schawaller (Staatliches Museum für Naturkunde, Stuttgart, Germany), Dr. Kiyoshi Ando (Osaka, Japan) and two unknown referees for their valuable advice, and to Dr. Kimio Masumoto (Osaka, Japan) for the loan of the Thai specimen. This study was supported by the Ministry of Science and Technology of the People’s Republic of China (No. 2012FY111100).