(C) 2013 Yuchen Ang. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Ang Y, Wong LJ, Meier R (2013) Using seemingly unnecessary illustrations to improve the diagnostic usefulness of descriptions in taxonomy–a case study on Perochaeta orientalis (Diptera, Sepsidae). ZooKeys 355: 9–27. doi: 10.3897/zookeys.355.6013
Many species descriptions, especially older ones, consist mostly of text and have few illustrations. Only the most conspicuous morphological features needed for species diagnosis and delimitation at the time of description are illustrated. Such descriptions can quickly become inadequate when new species or characters are discovered. We propose that descriptions should become more data-rich by presenting a large amount of images and illustrations to cover as much morphology as possible; these descriptions are more likely to remain adequate over time because their large amounts of visual data could capture character systems that may become important in the future. Such an approach can now be quickly and easily achieved given that high-quality digital photography is readily available. Here, we re-describe the sepsid fly Perochaeta orientalis (de Meijere 1913) (Diptera, Sepsidae) which has suffered from inadequate descriptions in the past, and use photomicrography, scanning electron microscopy and videography to document its external morphology and mating behaviour. All images and videos are embedded within the electronic publication. We discuss briefly benefits and problems with our approach.
Taxonomy, species descriptions, illustrations, bioimaging, videography, Sepsidae
Many species descriptions–especially older ones–are very brief: they comprise of discussions and illustrations of diagnostic morphology, geographical distribution, and only occasionally some biology (e.g., see Appendix). The morphology sections are often limited to the most conspicuous features that can be used to differentiate and identify the target species from other species known to the scientific community at the time of description. In the past, this minimalist approach was necessary because journals had tight page restrictions and the cost of including many illustrations was high; this was a particularly serious problem for colour and halftone illustrations. Their high cost contributed to the widespread use of line-drawings in descriptive papers. However, such an exiguous approach towards descriptions is no longer needed given that these restrictions have largely disappeared. While line drawings remain important for clearly illustrating diagnostic features, a description can now afford to include more and different types of data. Electronic journals have fewer limitations on page numbers, and taxonomists now have ready access to high-resolution photography (
Embracing these new opportunities has many advantages. One is that more data makes it less likely that today’s descriptions will be inadequate in the future: a large number of images may serendipitously capture features that will only be revealed to be important in the future. This does not distract from the importance of line drawings, which have the advantage of highlighting important features and can accommodate intraspecific variability (see Discussion). However, line-drawings have the disadvantage that they are unlikely to capture character systems of future importance. For example, 19th and some early 20th century entomologists did not anticipate the importance of genitalia and microtrichosity (pruinosity) patterns in species identification they remained undescribed. Had current-day imaging techniques been available and used by these taxonomists, genitalia [at least “claspers” (hypopygia)] and microtrichosity data would have been captured despite their perceived unimportance at the time of description.
Employing these imaging techniques can also protect against bad taxonomy. For example, Francis Walker (1809–1874), while one of the most prolific taxonomist of his time, was also well known for his poor-quality judgement and descriptions that resulted in numerous synonyms [as his obituary laments; ‘More than twenty years too late for his scientific reputation, and after having done an amount of injury almost inconceivable in its immensity, Francis Walker has passed from among us’ (
The use of modern imaging is slowly beginning to gain traction in taxonomy (
Here, we present a re-description of Perochaeta orientalis (
Perochaeta is a small Oriental genus, with currently six described species (
Collection and rearing of specimens. All new material was acquired from a laboratory culture. This culture was established based on a single female adult specimen collected from a mid-elevation site in Malaysia (Cameron Highlands, 1600m ASL) and reared based on methods described in
Photography & illustrations. Male and female specimens were extracted from the culture for re-description. The habitus for both sexes were imaged using the Visionary DigitalTM Plus Lab System (CF4P3 magnification). Several other structures were also imaged and then digitally transferred into line drawings through tracing with a Wacom® PTZ 630 tablet in Adobe® Photoshop® CS4. Images and illustrations of important diagnostic features are shown in Figs 1 and 2, while images for additional views are shown in Fig. 3. Images of the holotype (Fig. 4A, B) were provided by the Hungarian Natural History Museum, Budapest, Hungary.
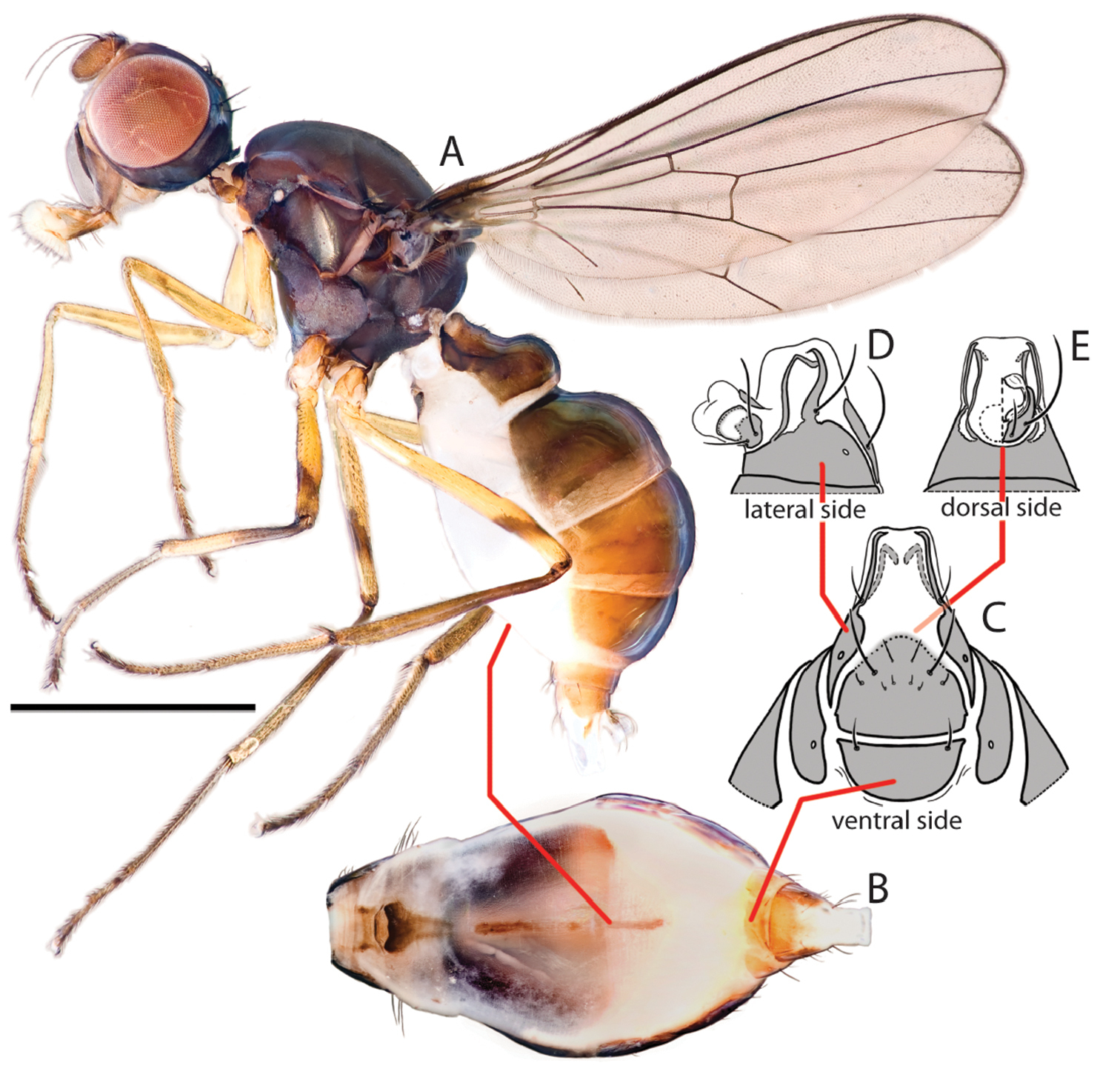
Key views and structures of Perochaeta orientalis, Male. A Habitus, lateral view B Pleural microtomensity pattern; (white = smooth, light grey = lightly microtomentose, dark grey = heavily microtomentose) C Rear tibia, with focus on osomterium D Basal section of wing showing microtrichosity pattern (white=smooth, light grey=with microtrichia) E Whole abdomen, ventral view F Sternite appendage G Hypopygium, dorsal view H Phallus, right, ventral and left views; red arrow indicates basal spiny flap. Scale bars = 0.5mm unless otherwise stated.
Key views and structures of Perochaeta orientalis, Female. A Habitus, lateral view B Whole abdomen, ventral view C Abdominal posterior, ventral view D Same, lateral view E Same, dorsal view. Scale bar = 0.5mm.
Additional views for Perochaeta orientalis, Male (MA-MF) and Female (FA-FE). M and F prefixes refer to male and female specimen respectively. A Habitus, dorsal view (sans wings) B head and thorax, ventral view C Head capsule, anterior view D Head capsule, posterior view E Thorax, posterior view F (male only)–Rear tibia, dorsal view showing osmeterium. Scale bars = 0.5mm unless otherwise stated.
Images of holotype (A, B) and drawing (C) from description for Perochaeta orientalis, male. A Image of habitus, lateral view B Image of hypopygium, dorsal view; red arrow pointing to the median protrusion on the surstylus C Drawing of abdominal posterior (lateral view) as reproduced from
Scanning electron microscopy (SEM). A phallus was dissected and dehydrated in an alcohol series, then critical-point dried with CO2 (Balzers® CPD-030) and mounted on a metal stub and platinum sputter-coated (JEOL® JFC 1600 Pt Fine Coater). SEM was performed at 100× with the JEOL JSM 6510 SEM. The image was then cleaned up with Adobe® Photoshop® CS4, and incorporated into Fig. 1.
Mating experiments. Each mating trial involved two male-female pairs because this species has very low mating success rates. The flies were introduced simultaneously into a small petri-dish and placed under a Leica MZ16A microscope. The mating behaviour was then recorded with an analogue video recorder (36 trials). Recording of behaviour began immediately upon the introduction of specimens into the petri-dish, and ended after 45 minutes if no mounting attempts made, or if they were not successful. The recordings were afterwards digitised and the non-linear editing software Final Cut Pro was used to study the behaviour ‘frame by frame’ (25 f.p.s.) in order to create a detailed mating profile. This profile was then compared with that of Perochaeta dikowi (
Online curation of specimens. All images, videos and the appendix in their original resolution are deposited in the species entry for Sepsidnet, an online digital reference collection dedicated to the Sepsidae of the world. These materials are also deposited as a project in Morphobank (Project 1062).
Taxonomic terminology. We adopt the terminology as described by
Holotype ♂ (Figs 4A, B). Type locality: “Chip Chip” (Jiji, = 集集) Township, Nantou County (南投), Taiwan ROC [likely, approximate coordinates 23°50'7"N, 120°46'4"E] (type label info: “Formosa Sauter. Chip-Chip 909. III. Nemopoda orientalis det de Meijere. Type.”). ♂ in the Hungarian Natural History Museum, Budapest, Hungary.
(Figs 1–3). Locality: Brinchang Jungle Trail, Cameron Highlands, Pahang, Peninsular Malaysia [4°30'9.55"N, 101°23'20.85"E. 1600m ASL]. Isoline culture based on ♀ collected 4.I.2011 (R. Meier). ♂♂♀♀ in the Raffles Museum of Biodiversity Research.
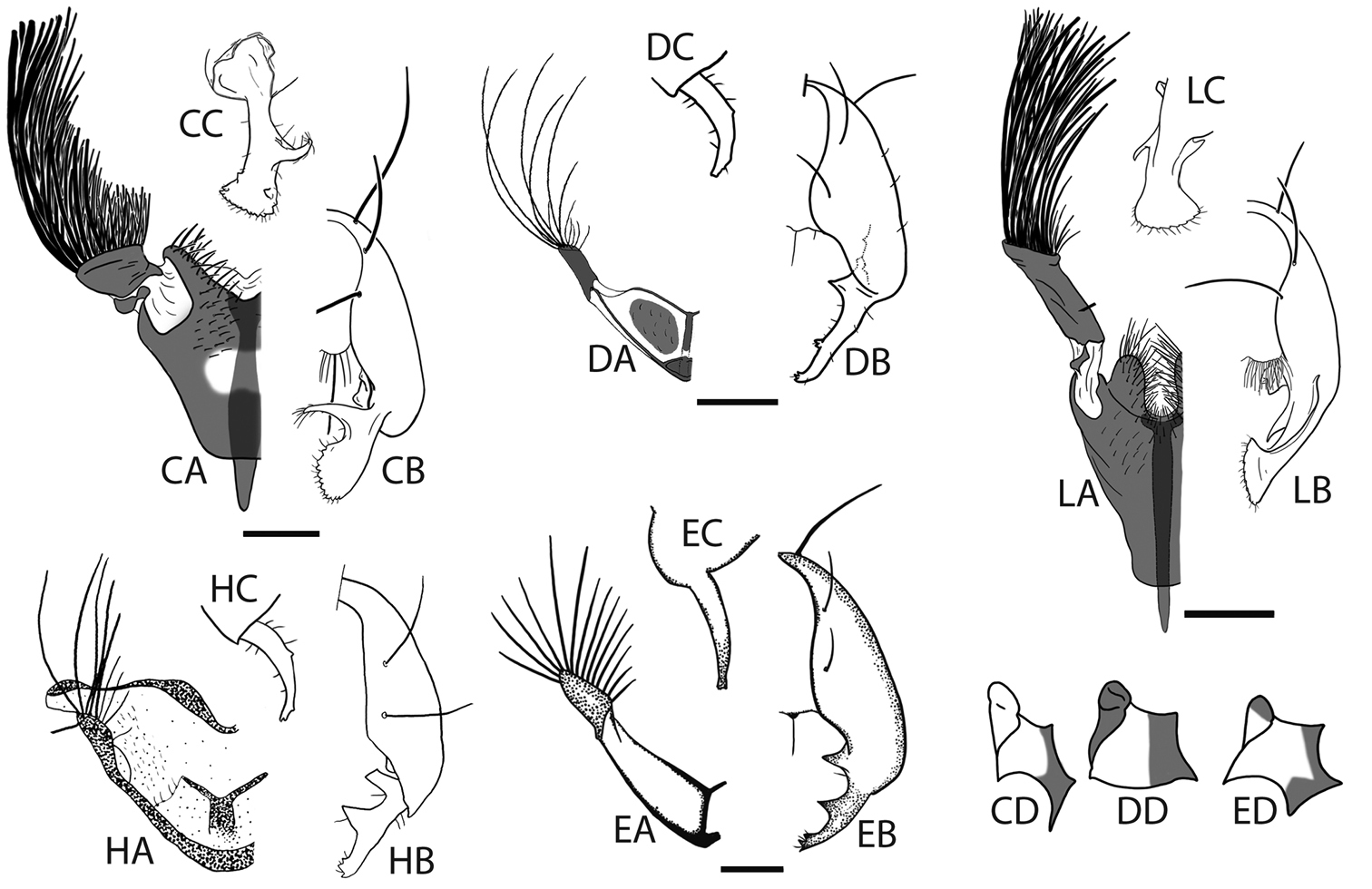
Male Perochaeta orientalis are most easily differentiated from other described Perochaeta species based on two large, flattened bristles of the main tuft on the sternite appendage, of which one has a triangular, submedial protrusion (red arrows on Fig. 1F) while all other described Perochaeta species have unmodified bristles (Figs 5 with suffix ‘A’). The surstylus in Perochaeta orientalis (Fig. 1G) is also unique in that the median inward protrusion consists of a large, broad-based triangle that spans a third of the surstylus (see Figs 5 with suffixes ‘B’ and ‘C’). The hind tibia of Perochaeta orientalis also has a distinct, raised osmeterium (Fig. 1C) which is barely visible or missing in other Perochaeta. Adult female Perochaeta orientalis can be distinguished from the females of Perochaeta dikowi (the only other species with a female description) based on the presence of sternites 3 and 4 (Fig. 2B), which are missing in the latter. For both sexes, the pleural, thoracical microtrichosity for Perochaeta orientalis (red arrow on Fig. 1B) is most similar to that of Perochaeta exilis (Fig. 5ED) because it is tomentose on the posterior third of the anepimeron and the dorsal tip of the greater ampulla. In contrast, Perochaeta cuirassa and Perochaeta lobo (Fig. 5CD) have a glossy greater ampulla, while Perochaeta dikowi is pruinose wholly on the greater ampulla and on slightly less than the posterior half of the anepimeron (Fig. 5DD).
Hypopygia, sternite appendages and anepimeral + greater ampullal microtrichosity of the five other Perochaeta: Perochaeta cuirassa (CA-CC), Perochaeta dikowi (DA-DC), Perochaeta exilis (EA-EC), Perochaeta hennigi (HA-HC) and Perochaeta lobo (LA-LC); adapted from Ang and Meier (2008; Perochaeta cuirassa and Perochaeta lobo), Ang et al. (2008; Perochaeta dikowi), Iwasa (2011; Perochaeta exilis) and Ozerov (1992; Perochaeta hennigi). Suffixes refer to: A sternite appendage, left side ventral view B hypopygium, right side dorsal view C Surstylus, lateral view D Anepimeron + greater ampulla [image not available for Perochaeta hennigi (prefix H)]. Perochaeta lobo (prefix L) has a similar anepimeral microtrichosity as Perochaeta cuirassa (CD). Scale bars = 0.5mm.
Colour. Similar in males (Fig. 1A) and females (Fig. 2A). Head capsule black except for face and a connecting thin strip below the eye, which is light-brown. Antennal pedicel dark brown, first flagellomere paler. Proboscis dark-brown with yellow labellum. Thorax wholly black, abdomen with glossy dark-brown tergites and sternites. All femora largely yellow with diffuse obfuscate rings post medially (faint on fore femur). Fore tibia wholly yellow; mid tibia darkened on the basal half; rear tibia entirely dark. All tarsi with first two segments yellow and last three dark-brown. Wing cells clear except for darkened basicostal cell and basal third of costal cell. Veins mostly dark brown. Calypter creamy; haltere whitish with brown base.
Head. Similar in males and females (Figs 1A, 2A). Roundish; facial carina short and shallow, facial area receding. Gena and parafacial region narrow. Ocellar prominence and occipital region lightly microtomentose. Chaetotaxy: ocellar longer than divergent postocellar; 1 outer vertical; inner vertical absent; orbital very reduced; 2 vibrissae; 2–3 weak postoculars; Lower fascial margin lined with setulae.
Thorax. Similar in males and females. Scutum, scutellum and subscutellum lightly microtomentose. Mediotergite microtomentose but glossy in the medial region (Figs 3ME, 3FE). Scutellum twice as wide as long (Figs 3MA, 3FA). Pleural pruinosity pattern (Fig. 1B): Protonotopleural lobe glossy on pleural region but microtomentose on dorsal region. Proepisternum fully microtomentose. Anepisternum largely glossy with anterioventral region densely microtomentose. Katepisternum with dense tomentosity except for glossy anterioventral region. Greater ampulla lightly microtomentose on the dorsal tip. Anepimeron glossy with lightly microtomentose strip on posterioventral region. Katatergite, katepimeron, metakatepisterum, meron and metepimeron lightly-dusted. Chaetotaxy: 1 apical scutellar, 1 reduced, setulae-like basal scutellar, 1 dorsocentral, 1 postalar, 1 supraalar, 2 notopleural, 1 postpronotal, 1 anepisternal and 1 posterior spiracular. Postpronotoum, prescutum and anepisternum with few, sporadic setulae.
Legs. Fore legs unmodified in males and females; all femora and tibiae without robust setae except for a longitudinal row of short spines on the anterior basal half of mid femur. Male rear tibia with a small but distinct osmeterium with raised hairs at the posteriodorsal region, and with three enlarged ventral setae on basitarsus (Fig. 1C). Females similar but lacking in osmeterium.
Wings. Similar in males and females. Slender. Without apical pterostigma. Veins bare. Wing microtrichia pattern (basal half; Fig. 1D): cells covered with microtrichiae except for subcostal, basal-medial, posterior-cubital cells and alula. Costal, radial 1, radial 2+3, radial 4+5, basal-radial, disco-medial, anterior cubital cells and anal lobe with portions lacking microtrichia. Radial-medial cross-vein divides discal-medial cell by ratio of 2: 1. Length: 4.4–4.8 mm.
Male abdomen. Ventral view (Figs 1E, F). Syntergite 1+2 to tergite 5 normal, tergite 6 missing, syntergite 7+8 present and extending ventrad as a narrow sclerite. Spiracles 1–4 on intersegmental membrane, spiracle 5 on ventral margin of tergite 5, spiracle 7 and 8 adjacent on margin of syntergite 7+8. Sternite 1 as a thin lateral band with tapering ends while sternite 2 is triangular, tapering posteriorly; sternite 3 is longitudinally oblong. Sternite 4 heavily modified into paired moveable appendages [Fig. 1F; see
Hypopygium (Fig. 1G). Cercal plate with two very weak lobes, each with one setae. Hypopygium triangular with a large tooth-like projection originating from the inner base of the surstylus. Surstylus itself fused to hypopygium and branches off dorsally. Each surstylus is curved ventrally, with a large, flattened, inward-facing posteriomedial triangular process; terminus with “teeth” and setulae.
Phallus (Fig. 1H). Basal region with scales on left side and relatively smooth on right side (crinkles and cracks on the surface are artifacts due to drying process). Basal region with large flap adorned with numerous long spines. Distal portion short (ca. 1/3 of basal portion) and membranous. We refrain from assigning terminology, for reasons explained in Discussion.
Female abdomen (Fig. 2B–E). Syntergite 1+2–tergite 5 similar to male, tergites 6 and 7 well defined and sclerotized. Spiracles 1–5 in intersegmental membrane while spiracles 6 and 7 are within the tergites. Sternites 1 and 2 similar to male, sternite 3 as a very thin longitudinal strip. Sternite 4 also a thin strip with barely visible sclerotization and a diffuse margin, sternite 5 missing. Sternites 6 as a lateral rectangle and sternite 7 tapering posteriorly. Postabdominal segments 6 and 7 with the tergites and sternites separated laterally, the sternites (like the tergites) thus very broad and short; segment 8, when not invaginated, long, extended posteriorly and ventrally, with a ventral element (sternite 8) on each side that remains separated at tip and a dorsal element (tergite 8) that forms the usual pair of ring-like bars that do not quite touch apically. Cercus small and round, with hypoproct present, bare.
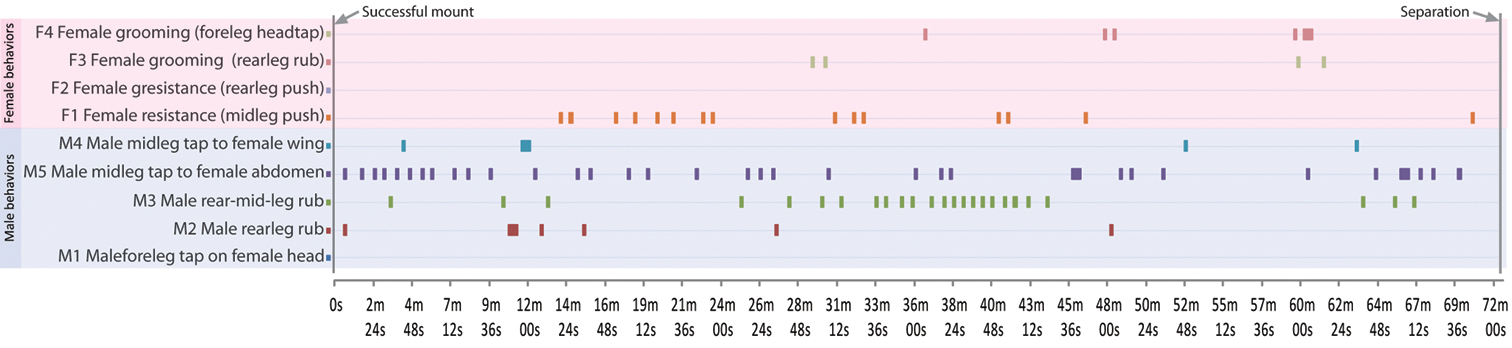
Here, we conducted 36 mating trials with virgin males and females. Only two of these trials were successful (=5.6% mating success rate), and the copulation time for these two were ca. 75 and 72 minutes. Virgin mating behaviour can be divided into four phases: (1) courtship, (2) approach and mount, (3) copulation and (4) separation. The copulatory profile (section 3) for Perochaeta orientalis is shown in Fig. 6, based on a frame-by-frame analysis of one of the trials and documented in Video 1 (time in video given as mm:ss). Where available, we will compare and differentiate the behaviour of Perochaeta orientalis with Perochaeta dikowi (
Video montage for the various behaviours described. Section 1, Courtship: Male wing-flutter dance (00:07). Section 2, Approach and Mount: Failed attempt with female resistance, lateral view (00:15), Successful mount, dorsal view (00:29). Section 3, Copulation: M1 Male fore leg tap to female head (00:41), M2 Male rear leg rub (01:03), M3 Male rear- to mid-leg rub (01:10), M4 Male mid legs tap to female wing (01:18), M5 Male mid legs tap to female abdomen (01:29), F1 Female resistance (mid legs push) (01:39), F2 Female resistance (rear leg push) (01:51), F3 Female grooming (rear leg rub) (02:00), F4 Female grooming (fore leg-head rub) (02:06). Section 4, Separation (02:15). Video available for download in full resolution from https://www.pensoft.net/J_FILES/video/Ang_Wong_Meier_Video_1.avi
Courtship. When the male detects and shows interest in a female, he courts the female by using a “wing flutter dance”; i.e., he rapidly circles the female from his side while fluttering the wing facing the female (00:07). This behaviour is not observed in Perochaeta dikowi.
Copulatory profile for Perochaeta orientalis, as described in Section 2 (Copulation). Horizontal bars in graph indicate point in time (X-axis) where then the particular behaviour (Y-axis) is performed. The profile begins from when the male mounts the female, and ends when they begin to separate (total time = 72m 30s).
Approach and mount. The male will approach the female from the rear and attempt to mount her. Unlike most sepsid species, Perochaeta orientalis males lack modified fore legs, and do not clasp the female wing or perform pre-copulatory behaviours when mounted like other sepsids (
Copulation (Fig. 6). Once the male locks its genitalia with the female, they copulate for a long time (73.7±1.2 min; based on the two successful trials), which is over 3 times longer than that in Perochaeta dikowi (22.6±2.48 min). There are periods of rest and activity during copulation. During rest, males place their fore tarsi on the female pronotal callus while mid- and rear legs are splayed out. During active periods, the male displays five types of behaviours: “M1: fore leg head tap”–males using fore tarsi to tap repeatedly on female head (00:41), “M2: rear leg rub”–males rubbing rear legs together (01:03), “M3: rear-mid-leg rub”–males rubbing rear legs with mid legs (01:10), “M4: mid legs wing tap”–males using mid legs to tap repeatedly on female wing (01:18) and “M5: mid legs abdomen tap”–males use mid legs to tap repeatedly on female abdomen (01:29). Behaviours M3 and M4 mostly occur after M1 and M2, suggesting a transfer of substance from the rear tibial osmoteria to the mid legs and then onto the female wing and/or abdomen. Female resistance was recorded even after copulation commenced; the female mostly used her mid legs (F1; 01:39) and only occasionally her hindlegs to push against the male (F2; 01:51). The female also indulged in grooming herself at times, either performing a rear leg rub (F3; 02:00) or a fore leg-head rub (F4; 02:06)
Separation. Just prior to separation, the male performs the “fore leg head tap” as well as the consecutive “rear-mid-leg rub” and “mid legs abdomen tap”. The separation event itself is initiated by the male, where he turns 180° and pulls away from the female (02:15). Both males and females will also use their rear legs to push against each other during this time. This is similar in Perochaeta dikowi.
Biogeography. Perochaeta has been consistently found only in mid- to high-elevation areas [see
Laboratory records. Under laboratory conditions, Perochaeta orientalis has been bred successfully from bovine (cow and gaur) dung. They are also attracted to this substrate in the wild, which makes sampling an area for Perochaeta a “bait-and-wait” strategy.
DNA sequence information. Molecular data from our new Perochaeta orientalis material are presented as part of the updated sepsid phylogeny (
A summary of the pairwise distances between the COI of Perochaeta orientalis with that of Perochaeta cuirassa (KF199839), Perochaeta dikowi (KF199840) and Perochaeta lobo (KF199841). Perochaeta orientalis has the most similar sequence to Perochaeta dikowi’s (3.82%), and all pairwise distances are relatively high.
| Perochaeta orientalis | Perochaeta cuirassa | Perochaeta dikowi | Perochaeta lobo | |
|---|---|---|---|---|
| Perochaeta orientalis | 0.00% | 11.44% | 3.82% | 13.15% |
| Perochaeta cuirassa | 8.70% | 0.00% | 12.95% | 11.89% |
| Perochaeta dikowi | 11.89% | 12.95% | 0.00% | 8.70% |
| Perochaeta lobo | 13.15% | 3.82% | 11.44% | 0.00% |
The decision to re-describe Perochaeta orientalis was based on the quality and accessibility of the two precedent descriptions by de Meijere and Duda (Appendix). de Meijere’s description (
In this paper we include images of the unlabelled phallus (Fig. 1H). There is still a dearth of information on this structure in Sepsidae, but we anticipate that it will gain in importance in the future. It is well-recognised that insect genitalia evolve rapidly and divergently, and are often the most reliable source of characters to delimit and describe species (
Illustration of Archisepsis phallus, as reproduced from
We here richly illustrate the morphology of Perochaeta orientalis with line drawings, photography and SEM images. This may raise the question whether too much effort was invested into a single species. However, all these visual data were acquired within a day, while much more time was needed for getting access to the original type material, literature, and confirming species identity. Of course, one obvious question raised by our proposal is where to stop. While we have covered the external morphology with images, our treatment is far from exhaustive. For example, we did not investigate internal morphology, nor the cuticular hydrocarbon profile (
One may argue that this will add to the taxonomic impediment, because future descriptions would require more images. This is a legitimate concern, given that taxonomists are already overwhelmed with the amount of undescribed species (
The authors would like to thank G.D. Lengyel (Hungarian Natural History Museum) for providing the excellent images of the holotype specimen, F. Friedrich (Hamburg University) for access to the
Scan of precedent descriptions of Perochaeta orientalis (doi: 10.3897/zookeys.355.6013.app). File format: Adobe PDF file (pdf).
Explanation note: Scanned original and subsequent description of Perochaeta orientalis by de Meijere (1913) and Duda (1926). All media have been archived at:
1) Sepsidnet (http://sepsidnet-rmbr.nus.edu.sg/Perochaeta_orientalis.html)
2) Morphobank (http://morphobank.org/permalink/?P1062)