Citation: Bang HW, Baguley JG, Moon H (2014) A new genus of Cletopsyllidae (Copepoda, Harpacticoida) from Gulf of Mexico. ZooKeys 391: 37–53. doi: 10.3897/zookeys.391.6903
A new genus and new species of the family Cletopsyllidae Huys & Willems, 1989 from deep-sea sediment in the Gulf of Mexico, are reported and fully described and illustrated. The new genus Pentacletopsyllus (type species: P. montagni sp. n.) can be distinguished from the four known genera of the family by antennule segmentation, length ratio of first and second endopodal segments of P1, and armature pattern on P5 exopod. It also differs from its sister genera by the rostrum being bifid at the tip, third segment of the female antennule having a smooth posterior margin, the baseoendopod of P5 with biarticulate outer setophore bearing basal seta, and female caudal rami without lobate expansion. A revised key to species of the family Cletopsyllidae Huys & Willems, 1989 is provided.
Harpacticoida, Cletopsyllidae, Pentacletopsyllus gen. n. , meiofauna, Gulf of Mexico
The Gulf of Mexico is a large semi-enclosed oceanic basin surrounded by the North American continent and the island of Cuba. The continental slope of the northern Gulf of Mexico is topographically complex and shows a high species diversity of benthic fauna (
The family Cletopsyllidae includes only four genera and 11 species, which are rare in marine benthic habitats (
The subfamily Normanellinae was elevated to family level by
During a recent deep-sea benthic survey to assess impacts of the Deepwater Horizon oil spill in the northern Gulf of Mexico, a new genus and species of Cletopsyllidae was collected. Recent investigations have uncovered significant benthic community impacts from the Deepwater Horizon (
Samples were collected from the northern Gulf of Mexico in May/June 2011 as part of the Deepwater Horizon Natural Resource Damage Assessment (NRDA) follow-up cruise aboard the R/V Sarah Bordelon. Sediments were sampled with an Osil multicorer and were fixed with buffered formalin and stained with Rose Bengal. Meiofauna was extracted from sediments by Ludox isopycnic centrifugation (
Specimens were dissected in lactic acid and the dissected parts were mounted on slides in lactophenol mounting medium. Preparations were sealed with transparent nail varnish. All drawings have been prepared using a camera lucida on a Leica DM 2500 differential interference contrast microscope. Specimens are deposited at the Smithsonian National Museum of Natural History.
The descriptive terminology is adopted from
Cletopsyllidae. Body elongated. Cephalothorax and other somites with numerous spinous processes at margin. Rostrum very prominent, triangular, with bifid tip in its apical portion, with pair of subapical sensilla. Genital and first abdominal somites completely fused forming double-somite. Anal operculum well developed. Caudal rami cylindrical, with 7 setae; setae I-II small, closely set; seta III subapical, setae IV-V bipinnate, seta V longest, seta VI bare, Seta VII tri-articulate at base. Sexual dimorphism in antennule, P2 endopod, P5 and P6.
Antennule 5-segmented in female, 7-segmented and subchirocer in male. Antenna 3-segmented, comprising coxa, allobasis and free endopodal segment; exopod 1-segmented with 2 setae. Mandible with biramous palp; basis with 3 pinnate setae; endopod with 1 lateral and 3 distal setae; exopod 1-segmented with 1 seta. Maxillule with strongly developed praecoxal arthrite, with 2 setae on anterior surface and 9 spines/setae around distal margin; coxal endite with 1 seta and 1 spine; basis with 4 spines/setae; endopod represented by 1 seta; exopod with 2 setae. Maxilla with 3 endites; allobasis drawn out into claw; endopod minute, bearing 3 setae. Maxilliped subchelate, with 3 pinnate setae; basis asetose; endopod drawn out into long, curved, pinnate claw with 1 long bare seta and 1 short accessory seta at base.
P1 basis with outer seta and inner spine; exopod 3-segmented, exp-1 with long outer spine, exp-2 with inner seta, exp-3 with 4 elements; endopod 2-segmented and prehensile, enp-1 as long as enp-2.
P2-P4 with outer spine or seta on basis. Exopods 3-segmented, exp-3 with 3 outer spines; endopod 2-segmented, enp-1 small, enp-2 elongate. Male P2-enp modified; inner apical seta fused to segment forming small apophysis. Armature formula:
| Exopod | Endopod | |
|---|---|---|
| P2 | 0.1.123 | 1.421 |
| P3 | 1.1.223 | 1.321 |
| P4 | 1.1.223 | 1.321 |
P5 of both sexes with separate baseoendopod and exopod. Basal seta on setophore; endopodal lobe triangular with 5 bipinnate setae in female and 3 bipinnate setae in male; exopod elongated with 5 setae in female and 4 setae in male. Male P6 asymmetrical, functional member represented by small plate, opposite member fused to genital somite; each leg with 1 bare seta.
Pentacletopsyllus montagni gen. et sp. n.
The generic name is derived from the Greek penta, meaning five, and refers to the 5-segmented female antennule.
http://zoobank.org/3CD1F4AE-9B8D-422F-A863-4683A3BA1EDC
http://species-id.net/wiki/Pentacletopsyllus_montagni
Figs 1–6Gulf of Mexico; 28°43.20'N, 88°20.68'W; depth 1590 m; mud.
Holotype: 1♀ (USNM No: 1231418) dissected on 8 slides, from the type locality. Paratypes: 2♀♀ and 1♂ (USNM No's listed in order presented in text: 1231419, 1231420, 1231421) each dissected on 8, 4 and 9 slides respectively, and 10♀♀ and 10♂♂ (USNM No's for specimens in vials (female, male): 1231422, 1231423) in 70% ethanol, vial. Additional samples were deposited in the first author’s collection. All from the type locality, collected by J. G. Baguley on May 2011.
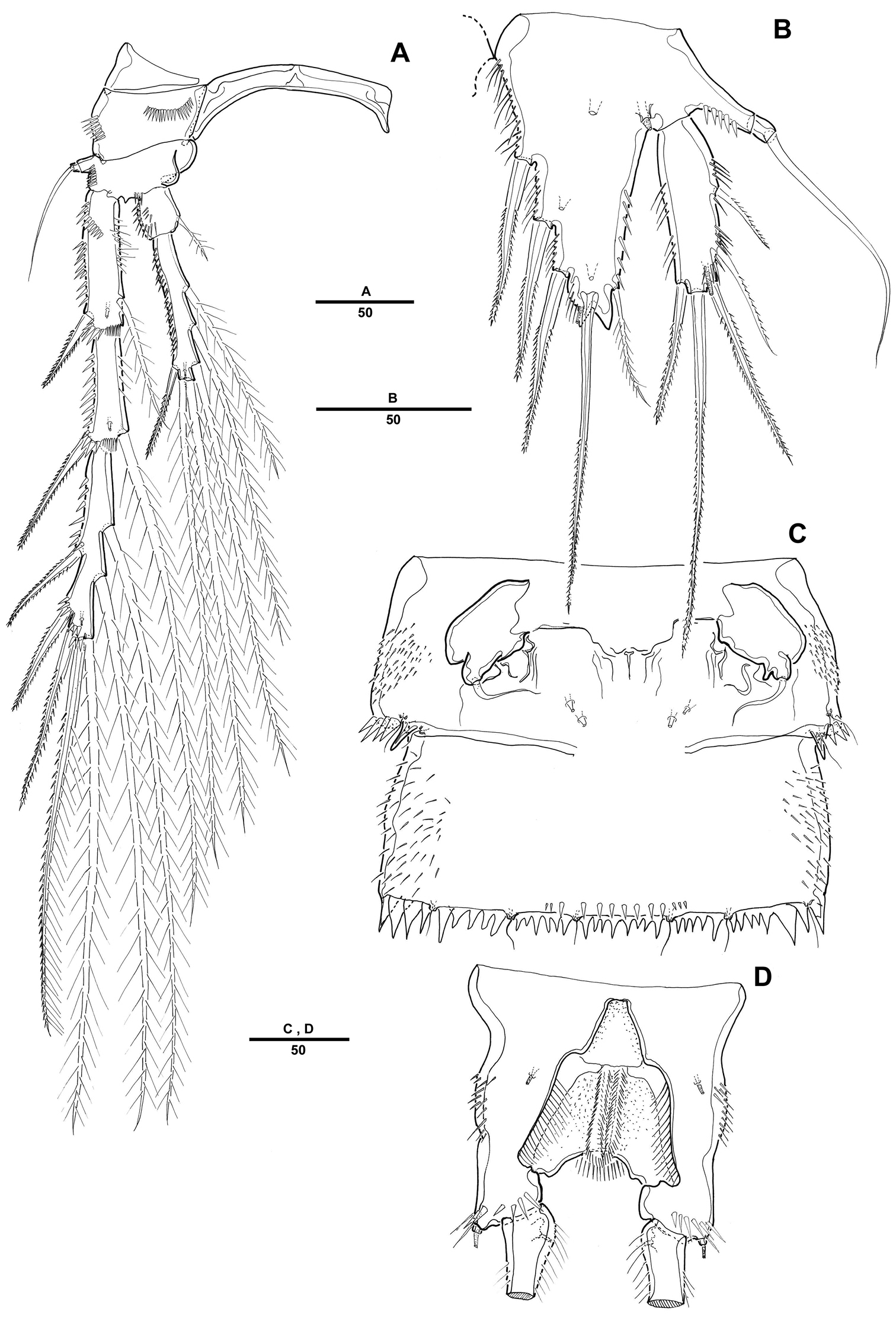
Female. Body (Fig. 1A) elongated. Total body length 1121 µm (n=10; range: 1022–1242 µm, measured from anterior margin of rostrum to posterior margin of caudal rami). Largest width measured at posterior margin of cephalic shield: 234 µm. Urosome narrower than prosome (Fig. 1A). Cephalothorax bell-shaped, with few tegumental sensilla; posterior and lateral margins irregularly serrated (Fig. 1D). Rostrum prominent, triangular; with a pair of sensilla near anterior margin, with bifid tip (Fig. 1E). Pedigerous somites with 1 pair of sensilla on dorsal surface, serrate posteriorly as cephalothorax; pleural areas well developed.
Pentacletopsyllus montagni gen. et sp. n. female: A habitus, dorsal B habitus, lateral C cephalothorax, lateral D tooth-like process of cephalothorax lateral anterior margin E rostrum, dorsal F caudal ramus, dorsal.
Urosome 5-segmented, comprising P5-bearing somite, genital double-somite and 3 free abdominal somites. All urosomites covered with small spinules dorsally and laterally. Urosomite with serrate posterior and posterolateral margin.
Genital double-somite (Fig. 4C), completely fused ventrally with original segmentation indicated by a transverse surface ridge dorsally and laterally. Genital field located near anterior margin with gonopore and copulatory pore located in median depression. P6 with 1 bare seta on a small protuberance. Anal somite with well-developed rounded operculum bearing row of setules (Fig. 4D).
Caudal rami cylindrical, about 7 times as long as wide, each ramus with 7 setae: setae I-II small, closely set, seta III subapical, setae IV-V bipinnate, seta V longest, seta VI bare and small, seta VII tri-articulate at base; tube pore presented dorsally near proximal inner margin (Fig. 1F).
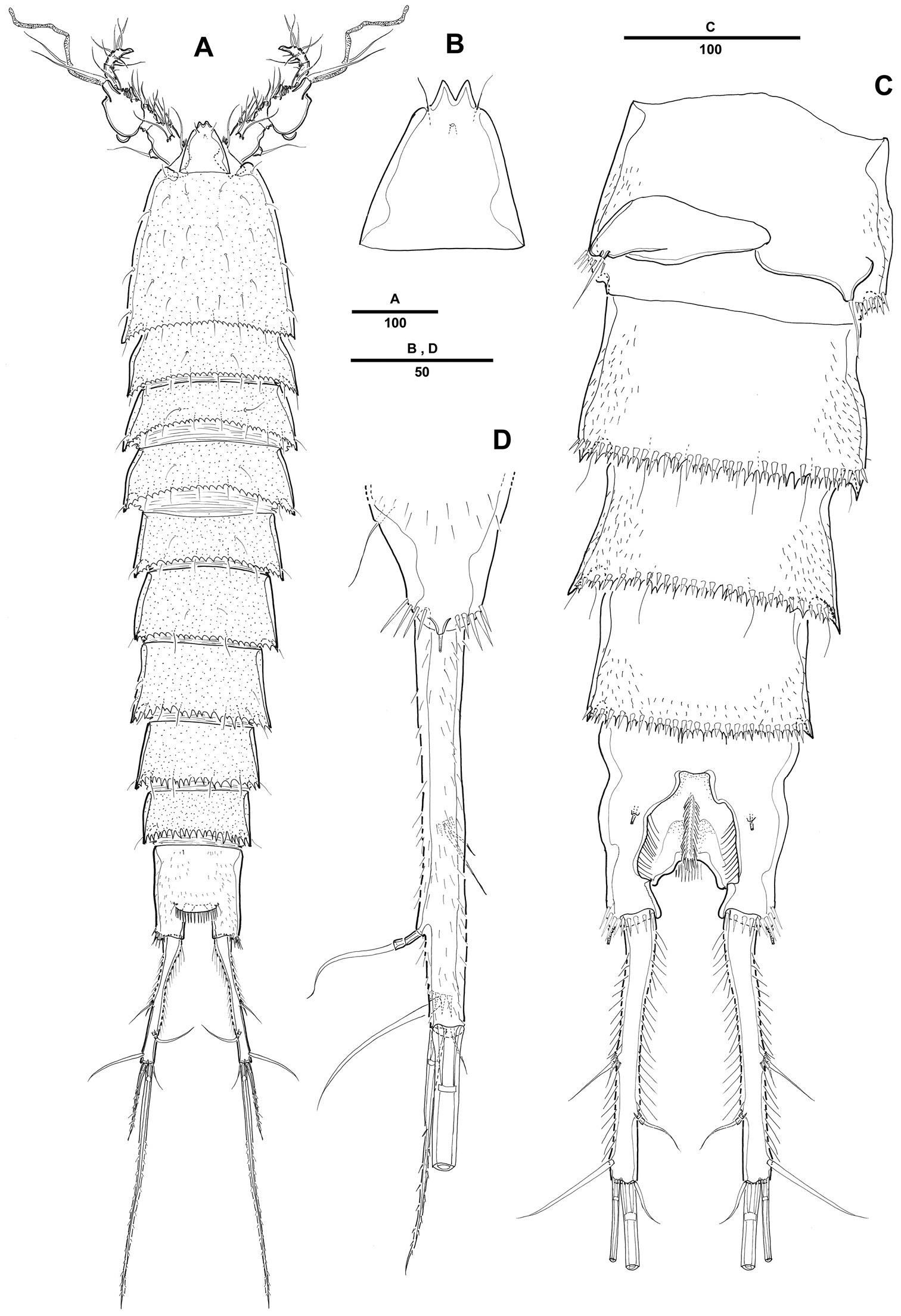
Antennule (Fig. 2A) 5-segmented, with well-developed sclerite around base of segment 1. Segment 1 short, with 2 long spinule rows and 1 seta. Segment 2 with 2 outer processes, distal one longer than proximal one, each one bearing one seta distally. Segment 3 longest, with smooth posterior margin. Armature formula: 1-[1], 2-[9], 3-[8 + (1 + ae)], 4-[3], 5-[7 + acrothek]. Apical acrothek consisting of a small aesthetasc fused basally to 2 bare setae.
Pentacletopsyllus montagni gen. et sp. n. female: A antennule, dorsal B antenna, dorsal C labrum, posterior D mandible E maxillule (inset showing armature on coxa) F maxilla (inset showing armature on middle endite) G maxilliped.
Antenna (Fig. 2B) 3-segmented, comprising coxa, allobasis and free 1-segmented endopod. Coxa small with spinule row. Allobasis elongated; original segmentation marked by incomplete surface sutures; 2 groups of strong spinules on abexopodal margin; with a long distal abexopodal seta. Exopod small, 1-segmented; with 1 apical and 1 lateral seta. Endopod elongated, with spinules along inner margin; lateral armature consisting of 2 pinnate spines and a minute seta; distal armature consisting of 1 apically curved pinnate spine, 1 bipinnate seta and 3 geniculate setae, the outer-most basally fused to an additional short seta.
Labrum with spinular ornamentation and covered with densely packed setules as in Fig. 2C.
Mandible (Fig. 2D) with large coxa bearing well-developed gnathobase, with 2 strong teeth, several multicuspidate teeth around distal margin and 1 pinnate spine at distal corner; spinules near base of palp. Palp biramous, basis with 3 pinnate setae; endopod with 1 lateral and 3 distal setae; exopod 1-segmented, with 1 apical seta.
Maxillule (Fig. 2E). Precoxa with few spinules near outer margin; arthrite strongly developed, with 2 surface setae and 9 apical spines and setae. Coxa with cylindrical endite bearing 1 naked seta, and 1 curved, pinnate spine. Basis with 2 setae and 1 bipinnate spine apically, and 1pinnate seta along outer margin; with several spinules around inner distal margin and base of endopod. Endopod incorporated in basis and presented by 1 seta. Exopod 1-segmented, with 1 pinnate and 1 naked setae.
Maxilla (Fig. 2F). Syncoxa with 3 endites, each with a short row of spinules. Proximal endite small and with 1 strong pinnate spine. Middle endite produced into pectinate spine and with 2 setae. Distal endite with 3 pinnate setae. Allobasis drawn out into strong, slightly curved, distally pinnate claw, accessory armature consisting of 2 bare and 1 pinnate seta. Endopod small, with 3 naked setae.
Maxilliped (Fig. 2G) comprising syncoxa, basis, and 1-segmented endopod. Syncoxa with 3 plumose setae and several short rows of spinules. Basis with 1 longitudinal row of spinules along palmar margin. Endopodal segment produced into strong and distally pinnate curved claw; accessory armature consisting of 1 long naked seta and 1 small seta at base.
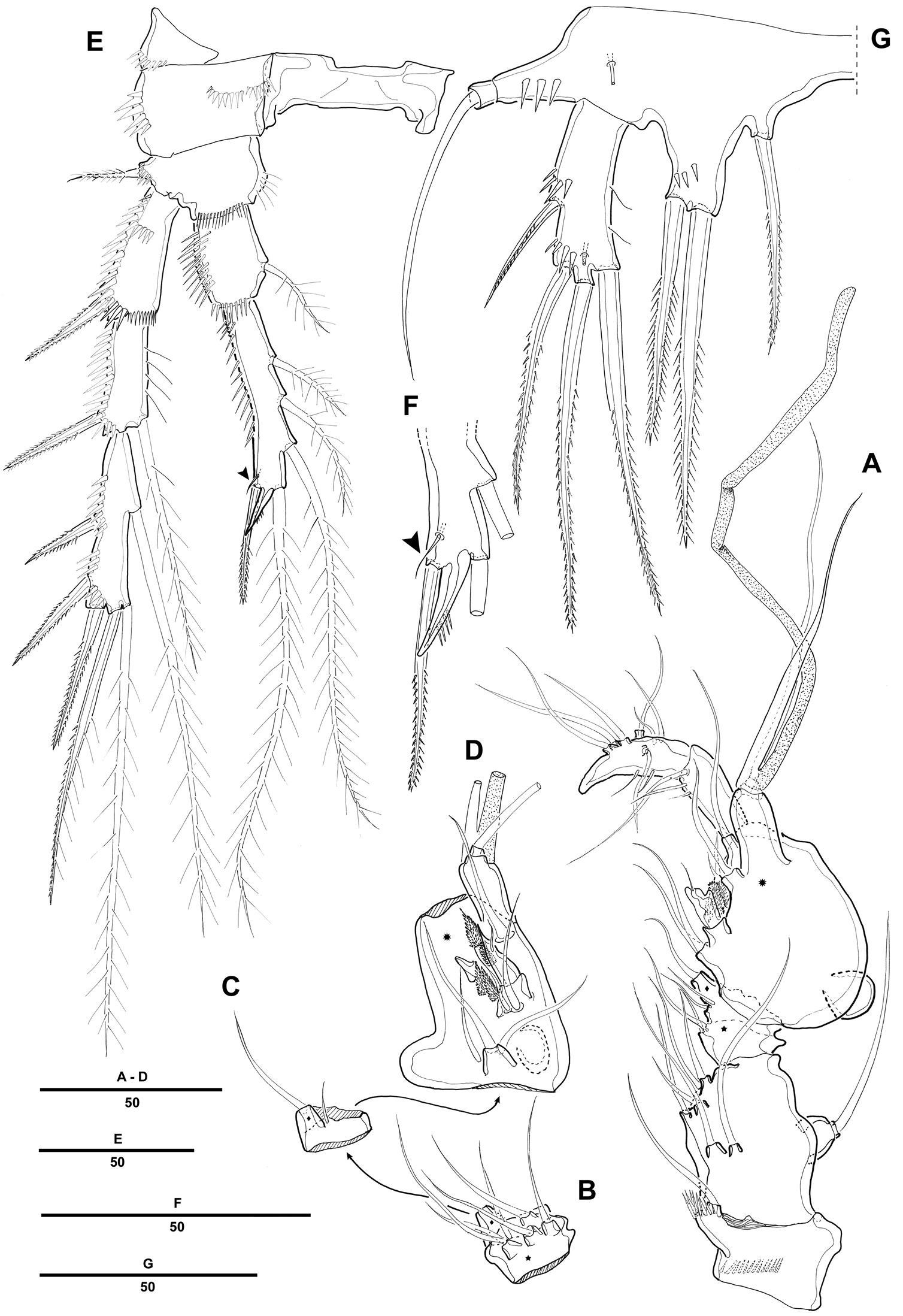
Swimming legs 1–4 (Fig. 3A, 3B, 3C, 4A) with wide intercoxal sclerite, biramous, endopods 2-segmented, exopods 3-segmented. Coxae and bases with row of spinules along outer margins as illustrated.
Pentacletopsyllus montagni gen. et sp. n. female: A P1, anterior B P2, anterior C P3, anterior.
Pentacletopsyllus montagni gen. et sp. n. female: A P4, anterior B P5, anterior C genital field, ventral D anal somite, ventral.
P1 (Fig. 3A). Coxa large, with inner and outer spinular rows. Basis with strong bipinnate seta on outer margin and bipinnate spine on inner distal surface; inner portion produced as a cylindrical pedestal for endopod. Endopod 2-segmented, prehensile; enp-1 as long as enp-2; enp-1 with one small seta on middle third of inner margin; enp-2 with 2 pinnate inner setae, and 1 pinnate spine and 1 geniculate seta distally. Exopod 3-segmented. Exp-1 and exp-2 with 1 pinnate spine; exp-2 with 1 inner seta; exp-3 with 2 geniculate distal setae and 2 strong spinulose outer spines.
P2-P4 (Figs 3B, 3C, 4A). Coxae and bases with spinular rows along outer margin and anterior surface. Basis wider than long, with pinnate spine (P2-P3) or bare seta (P4), each seta arising from a setophore. Each ramus consisting of 3-segmented exopod and 2-segmented endopod. Armature formula as in generic diagnosis.
P5 (Fig. 4B) with separate exopod and baseoendopod. Baseoendopod longer than wide, forming long biarticulate (not triarticulate) outer setophore bearing the basal seta. Endopodal lobe triangular, with 5 bipinnate setae. Exopod about three times as long as wide, with 1 inner, 1 distal and 3 outer pinnate setae.
Male. Body (Fig. 5A). Male slightly smaller and more slender than in female. Body length 1075 µm (N=10; range: 1007–1132 µm, measured from anterior margin of rostrum to posterior margin of caudal rami). Largest width measured at P2-bearing thoracic somite: 196 µm. Sexual dimorphism in antennule, P2 endopod, P5 and P6.
Pentacletopsyllus montagni gen. et sp. n. male: A habitus, dorsal B rostrum, dorsal C Urosome (excluding P5-bearing somite), ventral D anal somite and left caudal rami, lateral.
Prosome (Fig. 5A) 4-segmented, comprising cephalothorax (bearing first pedigerous somite) and 3 free pedigerous somites. Posterior margin of cephalothorax and pedigerous somites with serrated process, with integumental sensilla. Rostrum as in female (Fig. 5B).
Urosome (Fig. 5C) 6-segmented, comprised of P5-bearing somite, genital somite, and 4 free abdominal somites. Urosomite with crenulate posterior margin dorsally and ventrally.
Antennule (Fig. 6A) 7-segmented; subchirocer with geniculation between segments 5 and 6. Segment 1 with 1 row of long spinules along outer distal margin. Segment 2 with 1 cylindrical process along posterior margin, with 1 seta apically. Segment 4 represented by a small sclerite along anterior margin. Segment 5 swollen with large bump along posterior margin. Segment 7 with triangular distal half. Armature formula: 1-[1], 2-[7], 3-[6], 4-[2], 5-[7 + 4 pinnate + 1 modified + (1 + ae)], 6-[3], 7-[7 + acrothek]. Apical acrothek consisting of 2 small naked setae.
Pentacletopsyllus montagni gen. et sp. n. male: A antennule, ventral B third segment of antennule, anterior C fourth segment of antennule, anterior D fifth segment of antennule, anterior E P4, anterior F P4 endopod 3 (arrow indicating reduced outer seta), anterior G P5, anterior.
P2 (Fig. 6E). Exopod as in female. Endopod modified, 2-segmented; enp-2 with 4 plumose inner setae; inner apical seta fused to segment forming short apophysis; outer apical seta shorter than female; outer seta reduced and presented by minute naked seta (Fig. 6F).
Fifth pair of legs (P5) (Fig. 6G) fused medially. Baseoendopod with outer setophore bearing the basal seta. Endopodal lobe with 1 inner and 2 distal bipinnate setae. Exopod shorter than in female, about three times as long as wide, with 1 inner, 1 distal and 2 outer pinnate setae.
P6 (Fig. 5C) asymmetrical, bearing 1 naked seta on a cylindrical process.
The species is named in honor of Dr. Paul Montagna, Endowed Chair for Ecosystem Studies and Modeling at the Harte Research Institute, Texas A&M University Corpus Christi. Dr. Montagna has had a long and distinguished career studying meiofauna, hapacticoid systematics, and marine ecosystem dynamics, particularly in the Gulf of Mexico.
The subfamily Cletopsyllinae of the family Normanellidae was raised to familial rank by
Pentacletopsyllus gen. n. is placed in the family Cletopsyllidae with the character sets of the presence of a crenulated posterior margin of body somite, posterior margin of female antennule second segment with 2 distinct conical processes, 1-segmented antenna exopod with 2 setae, armature formula of swimming legs, and P5 with separate exopod and baseoendopod in both sexes and baseoendopod with elongated basal seta. The new genus can be readily identified on the basis of the following three characters:
-
Antennule: The genus Bathycletopsyllus Huys & Lee, 1999 has a 6-segmented female antennule and the other genera – Cletopsyllus Willey, 1935, Isocletopsyllus Huys & Lee, 1999, and Retrocalcar Huys & Lee, 1999 – have a 4-segmented antennule. However, the new genus displays a 5-segmented female antennule. Generally the third segment of the antennule has a smooth posterior margin, but a crenulated posterior margin is presented in the genus Cletopsyllus. The new genus does not show any modifications as in the genus Cletopsyllus.
Huys and Lee (1999) mentioned that the second segment of the male antennule has 2 processes along the posterior margin, however the genus Pentacletopsyllus has only one process on the posterior margin of antennule, moreover the fifth segment has an additional swelling at the posterior margin. -
Structure of P1: Normally in the family Cletopsyllidae, P1 exopod is 3-segmented, shorter than endopod; P1 endopod 2-segmented, prehensile, enp-1 at least twice as long as enp-2, and consists of an elongate proximal segment with 1 inner seta. The setation on exopod and endopod of P1 in the genus Pentacletopsyllus is the same as above, but the proximal and distal segment of P1 endopod are nearly equal in length.
-
P5:
Huys and Lee (1999) mentioned that the baseoendopod of both sexes in the family Cletopsyllidae is characterized by an extremely long extension bearing the outer basal seta, and this setophore is typically tri-articulate. The P5 exopod has six setae in the female and 4-5 setae in the male except for Cletopsyllus papillifer Willey, 1935 with seven setae in the female. The new genus has long setophore but not extremely long, and the P5 baseoendopod has a bi-articulate outer setophore in both sexes, and the female P5 has only five setae on the exopod.
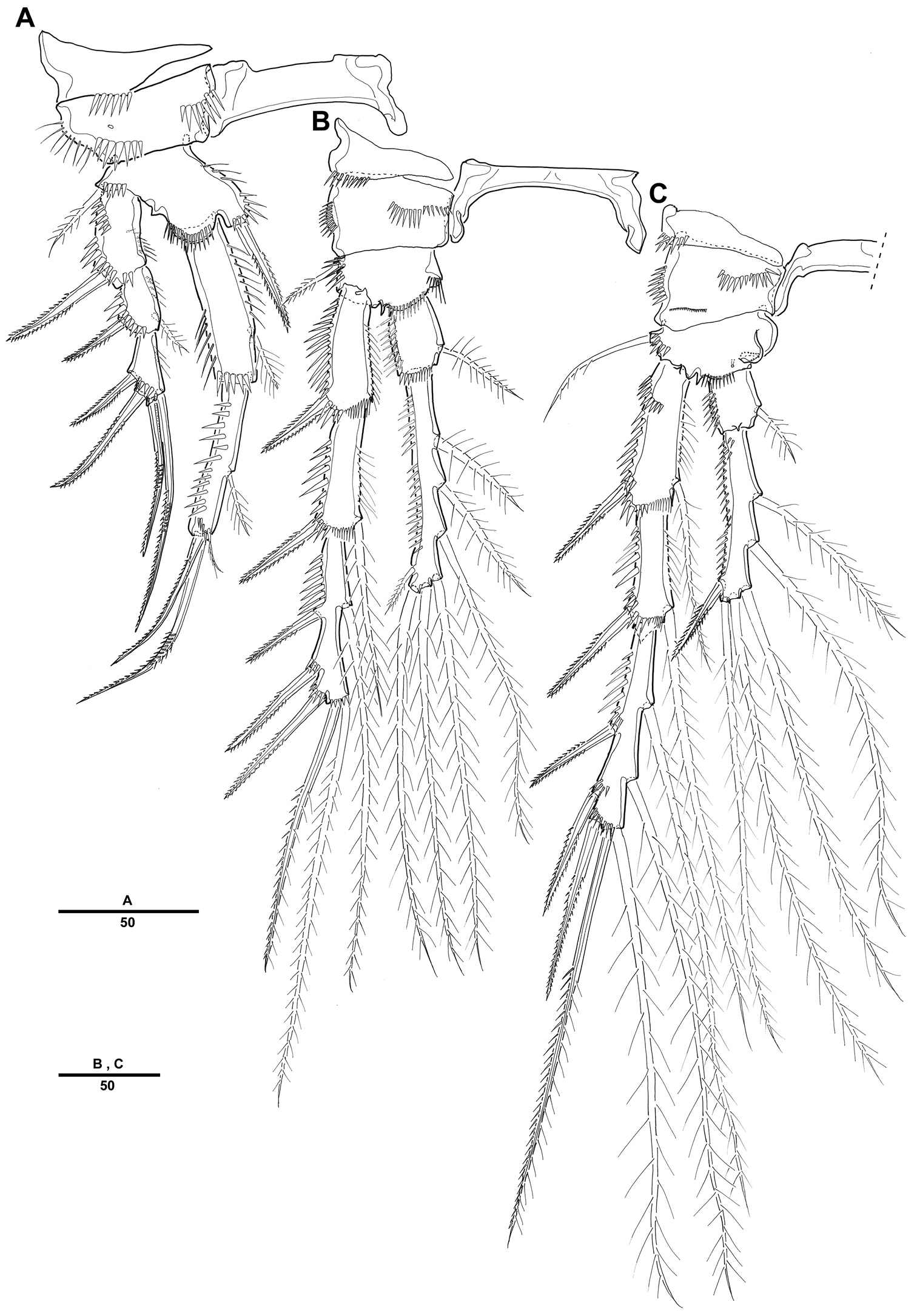
Currently, ten species of Cletopsyllidae are known from shallow and sublittoral marine habitats in India (Retrocalcar secundus (Nicholls, 1945); Cletopsyllus bacescui Marcus, 1976), in Far East Asia (Retrocalcar sagamiensis (Itô, 1971); Isocletopsyllus maximus Song, Kim & Hwang, 2010), in northeastern America and in Caribbean (Cletopsyllus papillifer Willey, 1935; Retrocalcar brattstroemi (Geddes, 1981); Cletopsyllus rotundifera Fiers, 1986), and in the Mediterranean (Isocletopsyllus tertius (Por, 1964); Isocletopsyllus quartus (Soyer, 1966); Isocletopsyllus sardus Addis, Floris & Carcupino, 2011). Additionally, Bathycletopsyllus hexarthra was reported from deep-sea (depth of 460 m) in the Indian Ocean (
Together with newly described Pentacletopsyllus montagni, the 5 genera and 12 species currently recognized as valid in the family Cletopsyllidae can be identified with the specific key given below. It is amended from
| 1 | Female antennule 6-segmented | Bathycletopsyllus hexarthra |
| – | Female antennule 5-segmented | Pentacletopsyllus montagni sp. n. |
| – | Female antennule 4 segmented | 2 |
| 2 | Third segment of female antennule with crenulate posterior margin; male fifth leg with 4 setae/spines on exopod | 3 |
| – | Third segment of female antennule with smooth posterior margin; male fifth leg with 5 setae/spines on exopod | 5 |
| 3 | P5 endopodal lobe without (male) or with (female) short rounded terminal process; second endopodal segment of first leg with 1 inner seta; rostrum with rounded apex | Cletopsyllus rotundifera |
| – | P5 endopodal lobe of both sexes with long curved terminal process; second endopodal segment of first leg with 2 inner setae; rostrum trifid or bifid at tip | 4 |
| 4 | Inner seta on first endopodal segment of leg 1 inserted at 55% of segment length; endopodal lobe of female fifth leg elongate, rectangular | Cletopsyllus bacescui |
| – | Inner seta on first endopodal segment of leg 1 inserted at 66% of segment length; endopodal lobe of female fifth leg short, triangular | Cletopsyllus papillifer |
| 5 | Female caudal rami with outer proximal margin produced as a lobate expansion bearing a spur-like process posteriorly and secondary process dorsally | 6 |
| – | Female caudal rami without lobate expansion; ramus markedly longer in male | 8 |
| 6 | Second endopodal segment of first leg with 1 inner seta | Retrocalcar secundus |
| – | Second endopodal segment of first leg with 2 inner setae | 7 |
| 7 | Caudal ramus 3.6 (male) and 6.5 (female) times as long as greatest width; outer spines on third exopodal segment of leg 3 modified in male | Retrocalcar brattstroemi |
| – | Caudal ramus 5.3 (male) and 9.0 (female) times as long as greatest width; outer spines on third exopodal segment of leg 3 not modified in male | Retrocalcar sagamiensis |
| 8 | Caudal ramus less than 6 times as long as wide; rostrum trifid at tip | 9 |
| – | Caudal ramus more than 7 times as long as wide; rostrum bifid at tip | 10 |
| 9 | First antennular segment without outer process; caudal ramus without expanded inner border | Isocletopsyllus tertius |
| – | First antennular segment with outer process; caudal ramus with expanded inner border | Isocletopsyllus maximus |
| 10 | Antennary exopod with 2 setae of equal length; distal margin of basis of leg 1 without spinous process between exopod and endopod | Isocletopsyllus quartus |
| – | Antennary exopod with 2 setae of different length; distal margin of basis of leg 1 with spinous process between exopod and endopod | Isocletopsyllus sardus |
Sample analysis and production of this manuscript was funded in part by contract AB133C-11-CQ-0050 from the National Oceanic and Atmospheric Administration (NOAA) via subcontract 5700-UNR from Industrial Economics as part of the Deepwater Horizon Oil Spill Natural Resource Damage Assessment (NRDA). Christopher Lewis (Industrial Economics, Inc.) and Rob Ricker (NOAA) reviewed earlier versions of the manuscript. The views expressed herein are those of the authors and do not necessarily reflect the views of NOAA or any of its personnel. Neither the DWH NRDA Trustees nor representatives of BP had any role in sample processing, data analysis, decision to publish, or preparation of the manuscript. Pre-approval to submit the manuscript for publication was provided by representatives of the NRDA Trustees. We thank professor Wonchoel Lee, who reviewed our illustrations and preliminary descriptions prior to drafting this manuscript. We thank the two anonymous reviewers and the editor Danielle Defaye, whose comments greatly improved the quality of this manuscript.