(C) 2013 Snejana Grozeva. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Grozeva S, Kuznetsova VG, Simov N, Langourov M, Dalakchieva S (2013) Sex chromosome pre-reduction in male meiosis of Lethocerus patruelis (Stål, 1854) (Heteroptera, Belostomatidae) with some notes on the distribution of the species. In: Popov A, Grozeva S, Simov N, Tasheva E (Eds) Advances in Hemipterology. ZooKeys 319: 119–135. doi: 10.3897/zookeys.319.4384
The karyotype and meiosis in males of giant water bug Lethocerus patruelis (Heteroptera: Belostomatidae: Lethocerinae) were studied using standard and fluorochrome (CMA3 and DAPI) staining of chromosomes. The species was shown to have 2n = 22A + 2m + XY where 2m are a pair of microchromosomes. NORs are located in X and Y chromosomes. Within Belostomatidae, Lethocerus patruelis is unique in showing sex chromosome pre-reduction in male meiosis, with the sex chromosomes undergoing reductional division at anaphase I and equational division at anaphase II. Cytogenetic data on the family Belostomatidae are summarized and compared. In addition, the structure of the male internal reproductive organs of Lethocerus patruelis is presented, the contemporary distribution of Lethocerus patruelis in Bulgaria and in the northern Aegean Islands is discussed, and the first information about the breeding and nymphal development of this species in Bulgaria is provided.
Karyotype, NOR, meiosis, sex chromosome pre-reduction, male reproductive organs, distribution, Lethocerus patruelis, Belostomatidae, Heteroptera
The genus Lethocerus Mayr, 1853 is a member of the family Belostomatidae (electric light bugs, toe biters), the subfamily Lethocerinae (Perez Goodwin 2006). The giant water bug Lethocerus patruelis is the largest European true bug and the largest European water insect. The adult bugs reach 80 mm in length. The information on the karyotypesof the genus Lethocerus has been recently summarized by
In the present work, we studied for the first time the structure of the internal reproductive organs, karyotype and meiosis in males of Lethocerus patruelis (Stål, 1854). In addition, we summarize here data on the contemporary distribution of Lethocerus patruelis in Bulgaria and in the northern Aegean Islands, and provide the first information on the reproduction of this species in Bulgaria.
Males of Lethocerus patruelis were collected in 2001–2012 in different regions of southern Bulgaria. Collections were made either in water bodies using plankton net or (predominantly) by light traps. Two adults and three larvae were reared in the laboratory using small fishes (Gambusia affinis, Pseudorasbora parva and Carassius gibelio) as a food. Cytogenetic study was based on three males collected in the area of the border checkpoint Kapitan Andreevo, Bulgaria.
Specimens examined: BULGARIA: Black Sea Coast: Burgas, Lukoil Oil Refinery, 29 m a.s.l., May 2011, 4 specimens, K. Popov leg.; Tundzha River Valley: Kazanlak, 370 m a.s.l., 15.x.2012, 1♀, Z. Antonova leg.; Elhovo, 113m a.s.l., October 2011, at light, 1♀, G. Hristov leg.; Maritsa River Valley: Kapitan Andreevo Checkpoint, 46 m a.s.l.: August–September 2011, at light, more than 60 specimens per night, E. Galabova obs.; 5.ix.2011, at light, 2♂, 4♀, S. Grozeva leg.; 5.x.2011, 4♂, E. Galabova leg.; 12.x.2012, 1♂, at light, E. Galabova leg.; Plovdiv, Plovdiv Thermal Power Station, 170 m a.s.l., October 2011, 1♀ and more than 70 specimens observed in the sewer, Zh. Vlaykov leg; Plovdiv, 168 m a.s.l., autoparking in the northern part of the town, 19.viii.2012, at light, 2 specimens, V. Dimitrov obs.; Peshtera, 435 m a.s.l., October 2011, 1 specimen dead on the road, D. Kajnarov obs.; Eastern Rhodopes Mts.: Madzharovo, Vulture Center, above Arda River, 160 m a.s.l., 16.ix.2009, in light trap, 2♂, 1♀, B. Zlatkov leg.; Kresna Gorge: above Oshtavska River, 315 m a.s.l., 10.x.2004, at light, 3 specimens, S. Beshkov leg.; Tisata reserve, 250 m a.s.l., 13.x.2012, at light, 1♀, B. Zlatkov & O. Sivilov leg.; Struma River Valley: Rupite, 115 m a.s.l., 20.viii.1997, 1 specimen, at light, M. Langourov leg.; Marena artificial pond close to General Todorov Village, 104 m a.s.l.: 28.vi.2009, 1♀, on the vegetation above water surface, V. Gashtarov obs.; 01.viii.2011, 1♀, in the water, M. Langourov leg.; 25.vii.2012, 3 larvae and 2 exuviae, in the water, N. Simov leg.; quarry near General Todorov Village, 113 m a.s.l., 23.x.2010, 1♂, in a puddle near the road, B. Zlatkov & O. Sivilov leg.; MACEDONIA: Dojran Lake, 144 m a.s.l., 1996-1997, many specimens, V. Krpach obs.; GREECE: Thassos Island: Astris Village, Astris Beach, 7.ix.2011, 1♀, dead on the beach, N. Simov & M. Langourov leg.; Astris Village, small beach N of Astris, 9.ix.2010, 1♀, in the sea, N. Simov & T. Stefanov leg.; Pefkari, 7 m a.s.l., 3.ix.2010, 1♂, dead under street lamps, T. Stefanov leg.
To examine the internal reproductive organs, the abdomen of chloroform-anaesthetized males was opened and the entire reproductive system was dissected. For chromosome studies, the gonads were dissected out from the adults and fixed in Carnoy’s fixative consisting of 96% ethanol and glacial acetic acid (3:1) and stored at 4°C. Cytological preparations were made by squashing a piece of the testis in a drop of 45% acetic acid on a slide. The coverslip was removed using a dry-ice technique (
For this staining, the method described in
For revealing the base composition of C-heterochromatin, staining by GC-specific chromomycin A3 (CMA3) and AT- specific 4-6-diamidino-2-phenylindole (DAPI) was used following the method described in
In Lethocerus patruelis males, the internal reproductive organs consisted of a pair of testes united by vasa deferentia (v d) with a median unimpaired tube, ductus ejaculatorius (d e) (Fig. 1). Each vas deferens was dilated to form a large vesicula seminalis (v s). The testes were colorless and spherical in form, and each consisted of a single very long tube (seminal follicle) rolled up into a ball. The follicle decreased in diameter from the apex to the vas deferens and showed synchronized divisions in different parts, with only sperms in its thinner part. There were no bulbus ejaculatorius and accessory glands.
Internal male reproductive system: t testis; v d vas deferens; v s vesicula seminalis d e ductus ejaculatorius.
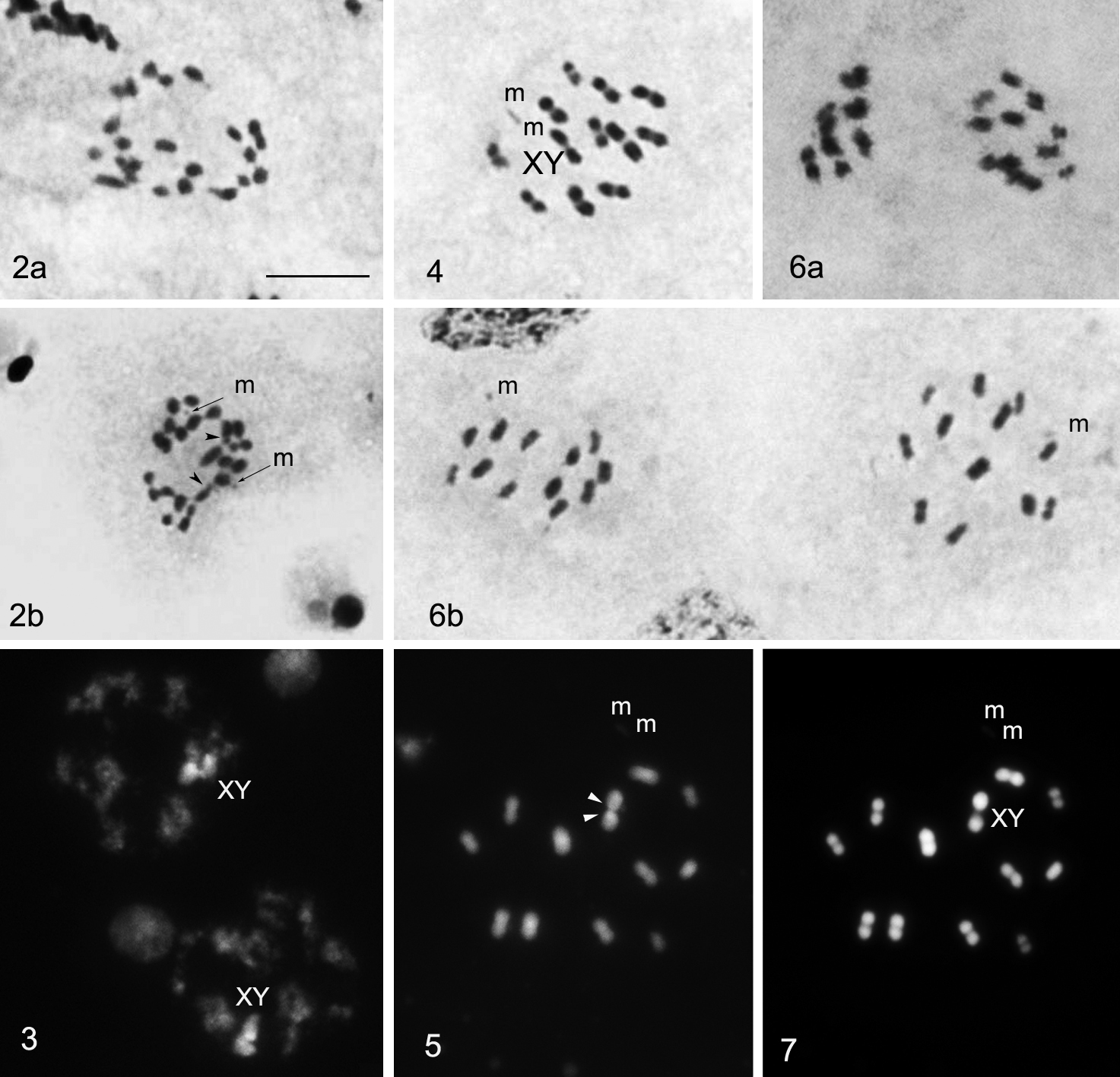
All three studied Lethocerus patruelis males presented the same chromosome complement. Spermatogonial metaphases showed 26 chromosomes including four larger and two very small ones, and the rest of the chromosomes formed a gradual size row. There was also a pair of very small m-chromosomes (= microchromosomes) (Fig. 2b) but these were not apparent in many nuclei (Fig. 2a). The chromosomes had no primary constrictions, i.e. centromeres. Two of larger chromosomes showed each a subtelomeric unstained gap, or secondary constriction, representing the nucleolus organizing region (NOR). These chromosomes are X and Y sex chromosomes as was revealed by the observation of meiotic stages (see below).
2 Spermatogonial metaphases: two of larger chromosomes, X and Y, each show a subtelomeric unstained gap, representing the nucleolus organizing region (NOR) (arrow head) (routine staining) 3 Meiotic prophase:sex chromosomes are visible as a large, positively heteropycnotic and brightly fluorescent body (CMA3 staining) 4 Metaphase I (n = 13) (routine staining) 5 Metaphase I: GC-rich NORs located on both X and Y chromosomes (CMA3 staining) 6 After the first meiotic division all the chromosomes segregate to opposite poles (6a) resulting in two daughter MII cells (6b) with 13 elements each, 11A + m + X and 11A + m + Y, respectively (routine staining) 7 Metaphase I: DAPI staining did not reveal any differentiation along the length of the chromosomes. Bar = 10μm.
During meiotic prophase, the sex chromosomes were united and visible as a large, positively heteropycnotic body brightly fluorescent after CMA3 staining (Fig. 3). Cells at metaphase I (MI) showed 13 bivalents, including a small and negatively heteropycnotic pair of m-chromosomes (n = 13). At this stage, all bivalents were distributed randomly relative to each other. Distinguishing between bivalents of autosomes and XY pseudobivalent involved difficulties since the latter was only slightly heteromorphic in form due to the size resemblance of X and Y chromosomes (Fig. 4). However CMA3-staining appeared a foolproof method for the identification of sex-pseudobivalent as one of the largest pairs with GC-rich NORs located in X and Y chromosomes (Fig. 5). At anaphase I (AI), all the chromosomes segregated to opposite poles resulting in two daughter metaphase II (MII) cells with 11A + m + X and 11A + m + Y, respectively (Fig. 6a). In the studied MII plates, X and Y- chromosomes were distributed randomly among other chromosomes (Fig. 6b). DAPI staining did not reveal any differentiation along the length of the chromosomes (Fig. 7).
In 2008 and 2011, we collected adult specimens of Lethocerus patruelis in water bodies in Struma River Valley near Rupite (Bulgaria). Several water bodies in Struma River Valley, close to these localities were checked by plankton net, and in July 2012, four larvae and five exuviae were found in the Marena artificial pond near General Todorov Village representing thus the first evidence of breeding of Lethocerus patruelis in Bulgaria. Marena would be classified as semi-natural mesotrophic to eutrophic lake with macrophytic vegetation (
The range of Lethocerus patruelis includes Balkan Peninsula, Anatolia, Israel, Syria, Saudi Arabia, Yemen, the United Arab Emirates, Kuwait, Iraq, Iran, Afghanistan, Oriental Region (Pakistan, India, Nepal, Burma), and recently this species was recorded from Italy (
In Bulgaria, only few records of Lethocerus patruelis specimens migrating from southern parts of the Balkan Peninsula, attracted to light, were published up to 2000 year (
Distribution of Lethocerus patruelis (Stål, 1854) on Balkan Peninsula: ● published records; ♦ new records with data of breeding; ✹ new records of specimens attracted to light.
A number of facts (records of the breeding population in Marena; the existence of similar habitats in other regions with records of Lethocerus patruelis at light; the last years’ tendency to milder winters) led us to suppose that this species would breed successfully also in other regions in southern Bulgaria (Maritsa River Valley, Burgas lakes). If such is the case, it would be a further evidence of the recent changes of European bug fauna caused by climate changes and global warming (
We have studied Lethocerus patruelis in respect of male reproductive organs, karyotype and meiosis. The internal reproductive system in this species appeared to be quite similar to that in Diplonychus rusticus (Fabricius, 1871) (Belostomatinae), the only belostomatid species studied so far on this point (
We found that Lethocerus patruelis had 2n = 26 (22 + 2m + XY). The eight Lethocerus species studied so far with respect to karyotypes (Table 1) represent a large proportion of the 22 species currently known in this genus (
Belostomatidae are composed of 3 subfamilies (Belostomatinae, Horvathiniinae, Lethocerinae) with 10 genera and approximately 150 species (
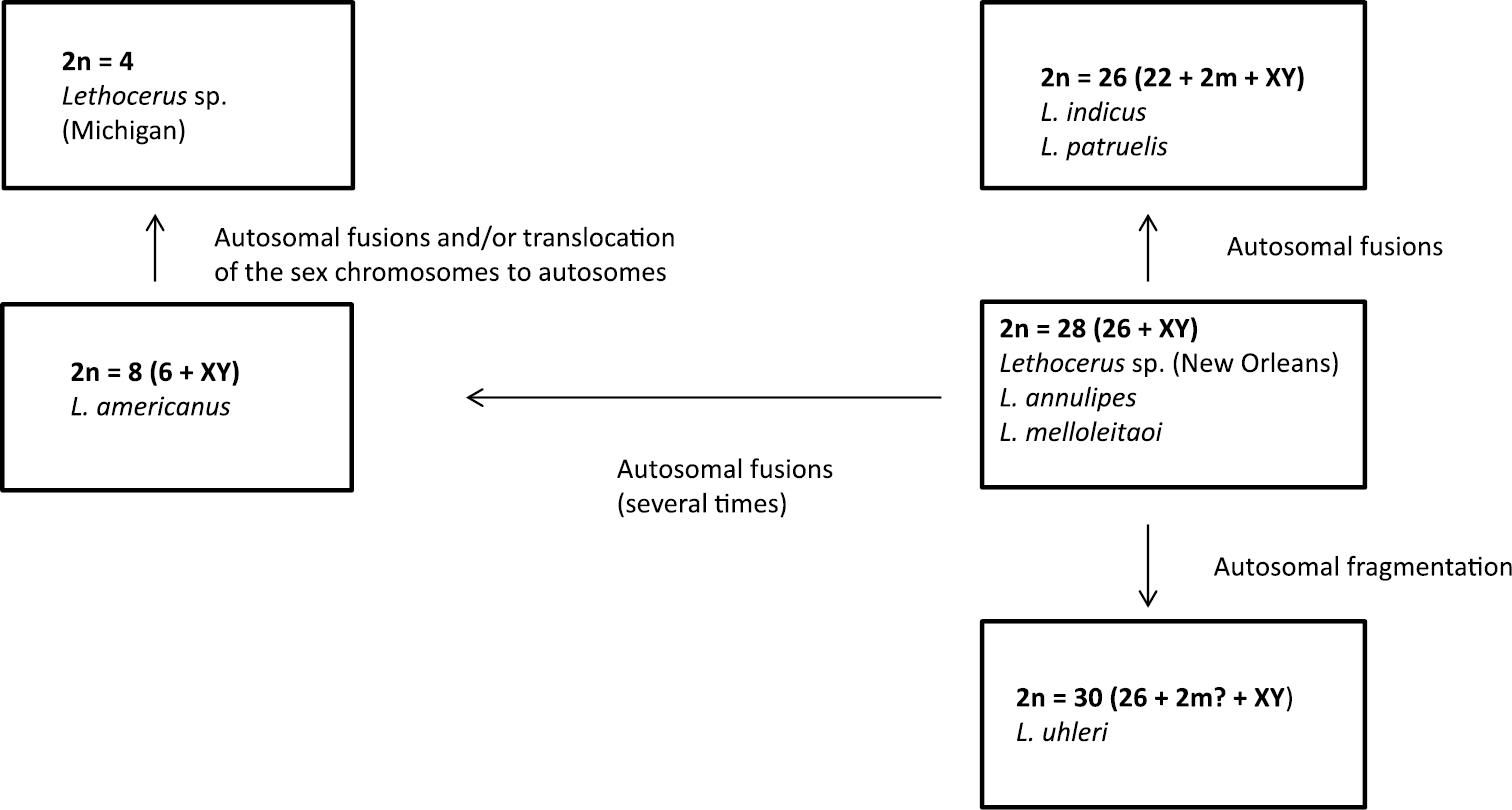
The genus Lethocerus shows a fairly wide range of chromosome numbers, with both extreme for Belostomatidae 2n = 4 and 2n = ca. 30, and three intermediate ones of 2n = 8, 26 and 28 (Table 1). The species studied share the conventional cytological features of Heteroptera, such as holokinetic chromosomes (lacking centromeres, that facilitates karyotype evolution via occasional fusion/fission events;
Cytogenetical data for the family Belostomatidae.
| Taxon | 2n♂ | Karyotype formula♂ | Sex chromosome division in male meiosis | NOR location | Remarks | Reference |
|---|---|---|---|---|---|---|
| Belostomatinae Leach, 1815 | ||||||
| Abedus indentatus (Haideman, 1854) | 29 | 24 + 2m + X1X2Y | Post-reduction | data absent | ||
| Belostoma bergi (Montandon, 1899) | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma bifoveolatum Spinola, 1852 | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma candidulum Montandon 1903 | 16 | 14 + XY | Post-reduction | sex chromosomes | Technique: CMA3 - |
|
| Belostoma cummingsi De Carlo, 1935 | 29 | 26 + X1X2Y | Post-reduction | sex chromosomes | Technique: Ag-staining, acridine orange, Hoechst 33258 - Papeschi, Bidau 1985. The authors did not specify whether NORs are located on every sex chromosome or only on some of them. | |
| Belostoma dentatum (Mayr, 1863) | 29 | 26 + X1X2Y | Post-reduction | a pair of autosomes | Technique: Ag-staining, acridine orange, Hoechst 33258 - Papeschi, Bidau 1985 | |
| Belostoma dilatatum (Dufour, 1863) | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| 30 | 26 + X1X2X3Y | Post-reduction | the terminal region of a medium-sized autosome pair | Technique: DAPI dull/CMA3-bright band on terminal position on one of the medium-sized autosomal bivalents - Chirino, Bressa 2011; CMA3 - |
||
| Belostoma discretum Montandon, 1903 | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma elegans (Mayr, 1871) | 29 | 26 + X1X2Y | Post-reduction | a pair of autosomes | Technique: CMA3 - Papeschi, Bidau 1985; FISH - Papeschi, Bressa 2006 |
|
| Belostoma elongatum Montandon, 1908 | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma flumineum Say, 1832 | 24 | 20 + 2m + XY | Post-reduction | data absent | ||
| Belostoma gestroi Montandon, 1900 | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma martini (Montandon, 1899) | 29 | 26 + X1X2Y | Post-reduction | data absent | ||
| Belostoma micantulum Stål, 1860 | 16 | 14 + XY | Post-reduction | sex chromosomes | Technique: CMA3, FISH - |
|
| Belostoma orbiculatum Estévez & Polhemus, 2001 | 16 | 14 + XY | Post-reduction | data absent | Sex chromosome polymorphism (XY/X1X2Y) | |
| Belostoma oxyurum (Dufour, 1863) | 8 | 6 + XY | Post-reduction | sex chromosomes | Technique: AgNO3, acridine orange, Hoechst 33258 - |
|
| Belostoma plebejum (Stål, 1860) | 16 | 14 + XY | Post-reduction | data absent | polymorphism for sex chromosomes (XY/X1X2Y) and number of autosomes (13/14) | |
| Belostoma sp. | 24 | 20 + 2m + XY | Post-reduction | data absent | ||
| Diplonychus annulatus (Fabricius, 1781) | 28 | 24 + 2m + XY | Post-reduction | data absent | ||
| Diplonychus rusticus (Fabricius, 1781) | 28 | 26 + XY | Post-reduction | data absent | ||
| Diplonychus molestus (Dufour, 1863) | 28 | 26 + XY | Post-reduction | data absent | ||
| Lethocerinae Lauck & Menke, 1961 | ||||||
| Benacus griseus (Say, 1832) | 28 | 24 + 2m + XY | Post-reduction | data absent | ||
| Kirkaldyia deyrolli (Vuillefroy, 1864) | 26 | 24 + XY | Post-reduction | data absent | ||
| Lethocerus americanus (Leidy, 1847) | 8 | 6 + XY | Post-reduction | data absent | ||
| Lethocerus annulipes (Herrich-Schaeffer, 1845) | 28 | 26 + XY | Post-reduction | data absent | ||
| Lethocerus indicus (Lepeletier & Serville, 1825) | 26 | 22 + 2m + XY | Post-reduction | data absent | ||
| Lethocerus melloleitaoi De Carlo, 1933 | 28 | 26 + XY | Post-reduction | data absent | ||
| Lethocerus patruelis (Stål, 1854) | 26 | 22 + 2m + XY | Pre-reduction | sex chromosomes | Technique: CMA3 | present paper |
| Lethocerus uhleri (Montandon, 1896) | ca. 30 | ? | Post-reduction | data absent | ||
| Lethocerus sp. 1 (from New Orleans) | 28 | ? | Post-reduction | data absent | ||
| Lethocerus sp. 2 (from Michigan) | 4 | ? | Post-reduction | data absent | ||
Suggested pathways of karyotype evolution in the genus Lethocerus.
On the other hand, the ancestrality of a XY sex determination in Lethocerus is beyond question, since neo-XY and XnY systems occurring in Belostomatidae (both), including Lethocerus (at least neo-XY), are clearly derivative being originated by X-autosome fusions or X-chromosome fissions, respectively. It cannot be doubted also that low chromosome numbers such as 2n = 8 in Lethocerus americanus and 2n = 4 in Lethocerus sp. from Michigan, are the derived characters brought about a series of autosome fusions during the course of evolution in this genus.
It seems likely that the ancestral karyotype in Lethocerus includes 26 autosomes and XY mechanism as found in many representatives of this genus and Belostomatidae as a whole (Table 1). It is not possible even to suggest whether this karyotype includes a pair of m-chromosomes as was speculated by
CMA3 staining of Lethocerus patruelis C-banded chromosomes revealed GC-rich sites corresponding to NORs in the X and Y chromosomes. This is the first case of NOR detection in Lethocerus. On the other hand, ribosomal genes have been already located in Belostoma chromosomes using various techniques such as fluorochrome staining, silver staining and FISH (Table 1). In Belostoma, five species were shown to have NORs also in sex chromosomes while three other species in a pair of autosomes. Noteworthy, the species with the same chromosome complement sometimes differ in rDNA location (for example, in sex chromosomes in Belostoma cummingsi while in autosomes in Belostoma dentatum, both with 2n = 26 + X1X2Y).
In the greatest majority of living organisms, during the first division of meiosis all chromosomes reduce in number (reductional division), whereas during the second division the chromatids separate (equational division), and this pattern is named “pre-reduction” (
Male meiosis in Heteroptera can further be characterized by radial configuration of one or sometimes both MI and MII plates. In this case, autosomal bivalents at MI and autosomes at MII form a ring on the periphery of the spindle, while the sex chromosomes are located in the center of this ring (
The study was supported for Snejana Grozeva and Nikolay Simov by DO-02-259 NSF Sofia, Bulgarian Academy of Sciences, and for Valentina Kuznetsova by the RFBR (grant 11-04-00734) and programs of the Presidium of the Russian Academy of Sciences “Gene Pools and Genetic Diversity” and “Origin of the Biosphere and Evolution of Geo-biological Systems”. We thank S. Nokkala for making a valuable contribution to the interactive discussion of the meiotic images. We thank Z. Andreeva, S. Beshkov, V. Dimitrov, G. Hristov, E. Galabova, V. Gashtarov, D. Kajnarov, K. Popov, O. Sivilov, T. Stefanov, Zh. Vlaykov, B. Zlatkov for the help in collecting material and data.
Electronic supplementary material video S1. (doi: 10.3897/zookeys.319.4384.app) File format: MPEG Video File (mpg).
Explanation note: A stalking/ambush attack of Lethocerus patruelis larva against small topmouth gudgeon (Pseudorasbora parva).