(C) 2013 Donald R. Davis. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Davis DR, Mendel H (2013) The genus Erechthias Meyrick of Ascension Island, including discovery of a new brachypterous species (Lepidoptera, Tineidae). ZooKeys 341: 1–20. doi: 10.3897/zookeys.341.6146
One previously named and two new species of the tineid genus Erechthias Meyrick are described and illustrated from the small, remote, mid-Atlantic Ascension Island. With these additions the Lepidoptera fauna of Ascension now totals 38 known species. Little is known regarding the biology of the two new species of Erechthias, and none of the species has been reared from larvae from Ascension. Erechthias minuscula (Walsingham) is a widespread, largely pantropical species first described from the West Indies. Larvae of Erechthias minuscula are known to be scavengers on a wide variety of dead plant material. Erechthias ascensionae, new species, is one of two species of Erechthias now known to be endemic to the island. The other endemic species, Erechthias grayi, new species, is further remarkable in having wing reduction occurring in both sexes. It is one of the few species of Lepidoptera known where this extreme of brachyptery involving both sexes has evolved. The larvae of Erechthias grayi are believed to be lichenivorous, and larval cases suspected to represent this species are illustrated.
Brachyptery, distribution, genital morphology, larval case
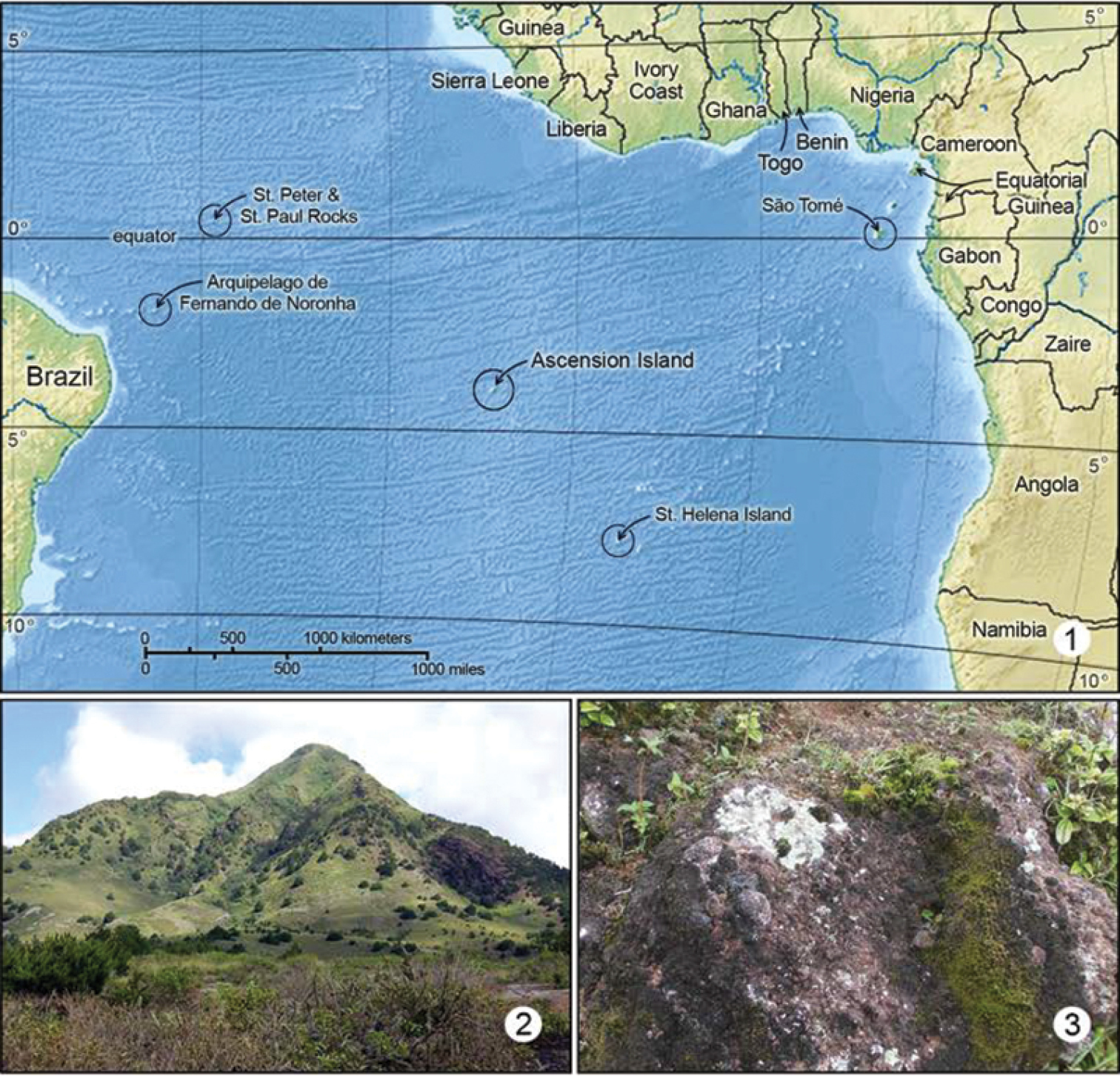
Ascension is a small, remote, tropical, volcanic island located in the middle of the Atlantic Ocean (Fig. 1), on the Mid-Atlantic Ridge. At 7°57'S, 14°22'W, it is some 1500 km from Africa and 2000 km from South America. The nearest land is St. Helena, another small, volcanic island, 1300 km to the south-east. Ascension Island comprises about 98 sq km of volcanic deposit, with a maximum elevation above sea level of 860 m.
1 Map of mid Atlantic Ocean. Ascension Island habitats: 2 Green Mountain 3 Typical habitat of Erechthias grayi; Lichen covered rocks on Green Mountain.
According to
The island of Ascension was discovered on Ascension Day in 1501 by the Portuguese explorer and sea captain João da Nova, but there was no settled population for another three centuries, probably because of the lack of permanent fresh water. Settlement began in 1815 when the British established a garrison to prevent the French navy using the island in any attempt to free Napoleon Bonaparte from St. Helena, where he had been exiled after the Battle of Waterloo. Napoleon never escaped and died on St. Helena in 1821.
The climate is tropical but the oceanic position of the island and the influence of the south-east trade winds are huge modifying influences. Although Green Mountain (Fig. 2) rises to a mere 860 m, it is cool, damp, windy, and frequently shrouded in mist and capped with cloud. It is indeed green compared with most of the island, where the vegetation is sparse and near-desert conditions prevail. However, the vegetation we see today across the island has been highly modified by deliberate and accidental introductions, and the impact of introduced animals such as goats, sheep and donkeys. Surviving areas of near natural vegetation are mostly difficult to access.
For this brief introduction, we have principally relied on information from
Erechthias is the largest, most diverse genus within the tineid subfamily Erechthiinae. Approximately 120 described species of Erechthias have been reported, with almost as many still undescribed (
Specimens examined in this study are deposited in the following institutions.
BMNH Natural History Museum, formerly British Museum (Natural History), London, United Kingdom.
USNM Collections of the former United States National Museum, now deposited in the National Museum of Natural History, Smithsonian Institution, Washington, D.C., USA.
Genitalic dissections were cleared by heating in 10% KOH for ~ 30 minutes, and subsequently cleaned and stained with either 2% chlorazol black E or mercurochrome solutions. The genitalia were then mounted on slides using Canada balsam or euparal mounting media. Genitalic terminology follows
http://species-id.net/wiki/Erechthias
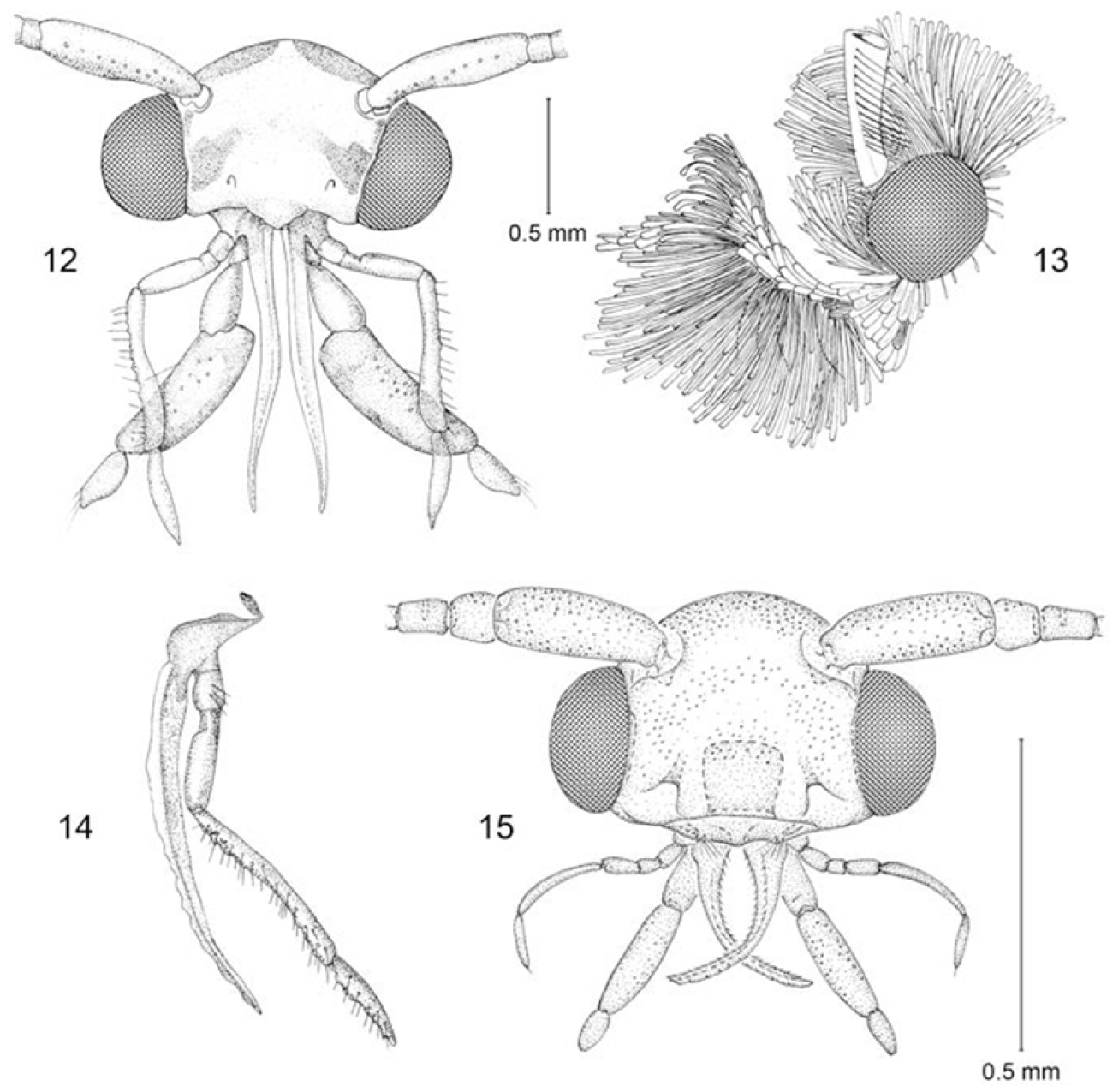
Head (Figs 12–15): Frons with scales moderately broad, either mostly appressed or partially raised, sometimes with weak scale tufts arising from ventro-lateral margin; pilifers present, with numerous, short bristles; vertex with prominent occipital and lateral tufts and scales more slender; Eye of medium size; frons broad; interocular index (
Thorax: Wings (Figs 16–18) relatively narrow; forewing L/W index: 0.23–0.27; hindwing L/W index: 0.24–0.32. Forewing with Sc and R present in all species, Rs usually with 3–4 branches, reduced to one branch in Erechthias grayi; Rs4 and M1 rarely stalked; accessory cell usually absent, sometimes with a trace of chorda; M with 2 branches (M1 and M2+3), with M fused with Cu in Erechthias grayi; base of M usually absent or vestigial in cell; Cu with 2 branches (fused with M in Erechthias grayi); CuP usually weak; A3 vestigial; retinaculum in male on underside of subcoasta; triangular, with a rolled apex. Hindwing with Sc and R fused; Rs usually present but incomplete, unbranched; M usually 3-branched, sometimes with M1and 2 fused; Cu 2-branced; A3 usually present; frenulum with single large spine in male, 1-3 spines in female. Legs unmodified; foretibia with epiphysis arising near distal third of tibia; midtibia with a single pair of spurs of unequal length arising near apex; hindtibia with 2 pairs of spurs of unequal lengths arising near basal third of tibia and near apex.
Abdomen: Apodemes slender, slightly convergent, or short, basally broad and nearly triangular. Segment A8 with male coremata present or absent; female corethrogyne absent.
Male genitalia: Segment A10 mostly membranous, often setose and melanized laterally; apex of uncus variably bilobed. Tegumen a narrow dorsal ring, poorly differentiated from vinculum; vinculum with a well developed, usually broadly rounded, triangular saccus. Valva usually simple, rarely with lobes or processes, usually broad, and often with a dense concentration of thick, costal spines; costal apodemes usually well developed. Gnathos absent. Juxta highly modified, forming sclerotized pouch of variable depth between bases of valvae and articulating with costal apodemes of valvae. Aedeagus typically in the form of a simple cylinder, with a slightly swollen base in some species; vesica sometimes lined with minute spicules, or with 1–2 much larger, spine-like cornuti.
Female genitalia: Ovipositor short to moderately long, 0.05–0.50× length of abdomen; posterior ventral apophyses not developed; posterior (dorsal) apophyses 1.5–2.7× length of anterior apophyses. Eighth tergite often narrowly rectangular, with a few terminal setae; eighth sternum connected to anterior apophyses by ventral rami. Bursa copulatrix 0.5–1.9× length of abdomen. Antrum relatively slender, often narrowly funnel-shaped or sometimes in the form of an incomplete ring; junction with ductus seminalis immediately anterior to antrum. Ductus bursae slender, with or without coarse microtrichia lining interior. Corpus bursae membranous, ovate to pyriform, often with a single small signum with the more slender, rodlike end projecting free from exterior wall of corpus bursae; signum sometimes can be stellate, blade-shaped, in the shape of a small ridged plate, or absent.
| 1 | Adult brachypterous; forewing less than 2 mm long; hindwing nearly absent, less than 0.2 mm long (Fig. 18) | Erechthias grayi |
| – | Adult fully winged; forewing more than 3 mm long; hindwing not reduced | 2 |
| 2 | Apex of forewing not turned sharply upwards; color pale whitish cream heavily irrorated with dark brown scales (Fig. 5) | Erechthias ascensionae |
| – | Apex of forewing sharply upturned; color pale whitish cream, variably irrorated or streaked with medium to dark brown scales (Fig. 4) | Erechthias minuscula |
http://species-id.net/wiki/Erechthias_minuscula
Figs 4, 12–14, 16, 19–23(Fig. 4). Head: Scales of frons shiny whitish cream, moderately broad with 3-4-dentate apices, flatly appressed to frons with apices directed dorsad; a pair of scale tufts consisting of very slender, elongate scales arising from lower corners of frons. Vertex with a prominent pair of lateral, occipital tufts composed of very elongate, piliform, pale cream scales with minutely bidentate scales. Labial palpus with apical segment mostly smoothly scaled; scales of segment 2 flat, strongly appressed dorsally, shiny white to whitish cream; venter of segment 2 with a dense brush of long, slender, erect, whitish (rarely suffused with dark brown) scales with minutely bidentate apices, and a lateral series of ~ 13–16 long, whitish bristles sometimes with dark apices. Antenna ~ 2/3 as long as forewing; scales smoothly appressed, uniformly pale cream except for scales with dark brown apices along anterior edge and sometimes venter of flagellum; scape mostly pale cream irrorated with dark brown scales along anterior edge; pectin well developed, consisting of a longitudinal row of 12–14 long dark setae; scales of scape and pedicel moderately broad; flagellum with a single row of more slender scales completely encircling each segment; short cilia usually not evident but often visible between scales.
Adults and larval cases 4 ♂, Erechthias minuscula, (forewing length 4 mm), Florida, USA 5 ♂, Erechthias ascensionae, (forewing length 6.8 mm), Ascension Island 6 ♂, Erechthias dracaenura (forewing length 8 mm), São Tomé 7, 8 ♂, Erechthias grayi (forewing length 1.7 mm), Ascension Island 9 (dorsal) 10 (lateral) Larval cases Erechthias sp. grayi ? (length 4.2 mm), Ascension Island 11 ♂, Erechthias darwini (forewing length 6 mm), St. Paul’s Rocks.
Adult head structure. 12–14 Erechthias minuscula 12 (anterior view) 13 (lateral view) 14 (maxilla) 15 Erechthias grayi (anterior view).
Wing venation 16 Erechthias minuscula 17 Erechthias ascensionae 18 Erechthias grayi.
Thorax: Forewing length 3.5–4.0 mm (wing apex not extended) – 5.0 mm (wing apex fully extended). Dorsum and tegula pale cream, usually with suffusion of light brown medially; apices of light brown scales often with dark brown apices giving a mottle appearance to dorsum; thorax whitish cream ventrally. Forewing predominantly pale whitish cream, variably irrorated or streaked with medium to dark brown scales; a medium to dark brown, variously interrupted streak usually extending from wing base almost to tornus (completely lacking in some specimens); costal margin often with small concentrations of dark brown scales present near base of forewing, near distal 2/3, distal 4/5 and at apex; those markings near apex sometimes forming short striae; a pale whitish cream patch of scales along margin between spot at distal 4/5 and apex; apex of forewing bent abruptly dorsad; fringe white to pale cream, variably banded at apex with 1–2 slender bands of dark brown scales; most of ventral surfaces of fore and hindwings (except for whitish cream fringes) medium to dark brown. Hindwing uniformly pale grayish brown; fringe mostly concolorous except for 2-3 narrow bands of dark brown scales around apex. Frenulum a single long spine in male and female. Fore and midlegs mostly pale to dark brown dorsally and pale cream ventrally with pale to dark brown banding on tibia and tarsomeres; hind leg generally paler, almost entirely pale cream in color.
Abdomen: Mostly pale golden brown dorsally, whitish cream ventrally. Eighth segment without coremata.
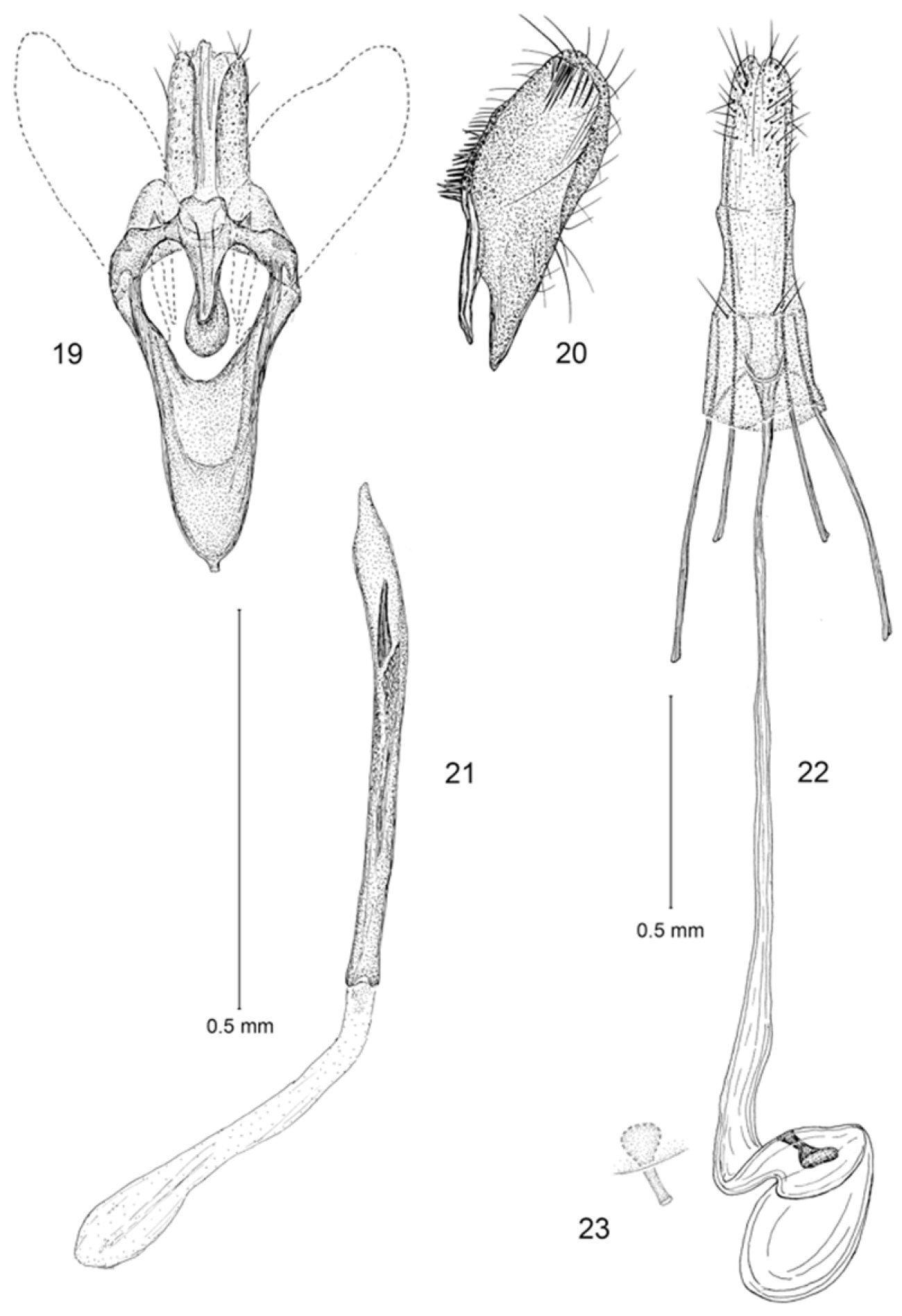
Male genitalia (Figs 19–21): Segment 10 mostly membranous, moderately sclerotized to form 2 elongate lateral lobes, with caudal margin superficially bifid. Tegumen a relatively narrow dorsal ring. Vinculum slender, narrowly V-shaped, elongate, ~ 0.8× the length of valva. Valva simple; cucullus broad with broadly rounded apex; costal margin densely setose, with setae concentrated near base of costa and less so at apex of valva. Juxta well developed as an elongate U-shaped pouch. Aedeagus a slender, simple cylinder, ~ 1.6× length of valva; vesica lined with numerous, minute spicules and with a single large, apical cornutus, sometimes closely accompanied with a shorter cornutus ~ half the length of the larger one.
Erechthias minuscula, genitalia 19 Male, ventral view 20 Valva, mesal view 21 Aedeagus, ventral view 22 Female, ventral view 23 Detail of signum in Fig. 22.
Female genitalia (Fig. 22, 23): Eighth sternite weakly sclerotized; ostium opening near anterior margin, with a pair of moderately short setae on either side of ostium. Antrum reduced, triangular; length ~ equal to maximum width. Ductus bursae very slender and elongate; length ~ 1.7× that of posterior apophyses, ductus gradually enlarging to relatively small, ovate corpus bursae; walls of corpus bursae membranous except for a very small, elongate, triangular signum; distal, more slender half of signum projecting beyond wall of corpus bursae.
♂ (present designation), WEST INDIES: Type H.T.; St. Thomas, Danish West Indies, 17 March 1894, Hedemann 7084; BM genitalia slide No. 4177; Walsingham Collection 1910-427; Ereunetis minuscula Wlsm., P.Z.S. p. 155 (1897), Type ♂; (BMNH).
ASCENSION ISLAND: Green Mountain: 4 ♂, 17–26 June 1988, C.M.StG Kirke, BM 1988-311, ♂ slide 29708, (BMNH).
(Fig. 1). Erechthias minuscula is probably pantropical in distribution, and occurs widely in South America, the West Indies into southern Florida, USA.
Larvae of Erechthias minuscula are scavengers on a wide variety of dead plant material and have been reported feeding on or within dead tree trunks, stems, seed pods, fruits, flowers, and leaves (
http://zoobank.org/2FEB95B4-F5DA-4C6D-B10A-A602725E862F
http://species-id.net/wiki/Erechthias_ascensionae
Figs 5, 17, 24, 25Female unknown.
Adult (Fig. 5). Head: Scales very slender with bidentate apices; scales of frons partially raised and directed forward, light brown strongly irrorated with dull white; scales of vertex erect or mostly so, especially over occipital tufts, and mostly directed forward; color similar to frons with most scales with dull white apices, . Labial palpus with scales slightly appressed dorsally, mostly dull white with light brown bases to scales; venter of labial palpus with a dense brush of long, slender, erect, brown scales usually with white apices, and a lateral series of ~ 5-7 long, dark bristles; 2–3 bristles also arising laterally from basal segment. Antenna nearly as long as forewing; scales smoothly appressed, dark brown dorsally, paler, more white ventrally; scape dark brown irrorated with white scales and with a row of white scales bordering distal margin; scales of scape and pedicel moderately broad; flagellum without cilia and with a single row of more slender scales completely encircling each segment.
Thorax: Forewing (Fig. 17) length 5–6 mm. Dorsum and tegula similar to head in color but with broader scales; thorax mostly pale cream ventrally. Forewing predominantly pale whitish cream, irrorated with scattered dark brown scales; 2–3 small patches of dark brown scales usually present on basal half of forewing along costal and dorsal margins and 2 patches within discal cell; fringe mostly light brown irrorated with dull white. Hindwing and fringe uniformly pale grayish brown; frenulum a single stout spine in male, not examined in female; M1 and 2 stalked ~ 0.6 their length. Fore and midlegs mostly light grayish brown dorsally and whitish cream ventrally with prominent dark brown banding on tibia and tarsomeres; hindleg generally paler in color.
Abdomen: Pale grayish brown dorsally, whitish cream ventrally. Eighth segment without coremata.
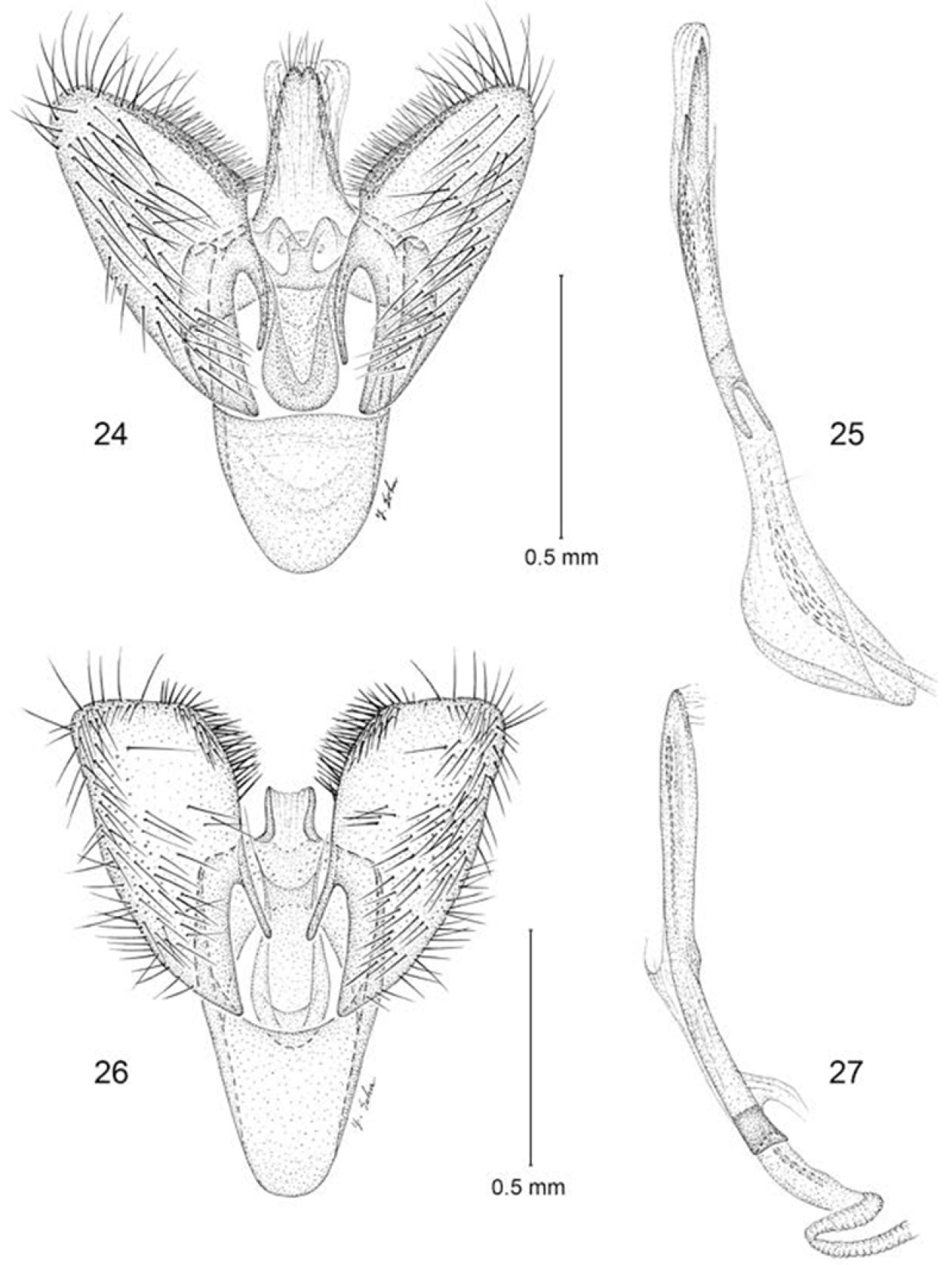
Male genitalia (Figs 24, 25): Segment 10 mostly sclerotized, fused to tegumen; uncus lobes minute, with caudal margin bifid. Tegumen a relatively narrow dorsal ring, with extended medium lobe fused to uncus. Vinculum broad, U-shaped, anterior margin broadly rounded, ~ 0.7× the length of valva. Valva simple; cucullus broad with rounded apex; costal margin densely setose. Juxta well developed as an elongate U-shaped pouch. Aedeagus slender, nearly as long as valva; vesica with numerous, minute spiculiform cornuti; base of aedeagus relatively deeply divided.
Male genitalia 24–25 Erechthias ascensionae, ventral view 25 Aedeagus, lateral view 26–27 Erechthias dracaenura, ventral view 27 Aedeagus, lateral view.
Female unknown.
The species name is derived from the genitive case of the type locality (Ascension).
♂, ASCENSION ISLAND: [Specific locality unknown] 4 Sept. 1958, E.A.G. Duffey, B.M. 1958-760, digital image captured, (BMNH).
ASCENSION ISLAND: Same data as holotype: 6 ♂, BMNH genitalia slide ♂, 5854, BMNH wing slide 30835, (BMNH, NMNH).
(Fig. 1). Ascension Island.
Unknown; larvae are most likely plant detritvores or lichenivorous.
The species nearest to Erechthias ascensionae, both morphologically and geographically, is Erechthias dracaenura (Meyrick), which is known only from São Tomé, an island located off the western coast of central Africa in the Gulf of Guinea (Fig. 1). The forewing patterns of both species are similar (Figs 5, 6) in possessing a whitish background color irrorated with isolated brown scales and marked with 6–8 scattered, moderately large, darker brown to black spots. The forewings of Erechthias dracaenura generally appear more whitish and less heavily marked than those of ascensionae. The male genitalia of Erechthias ascensionae (Figs 24–25) differ from that of Erechthias dracaenura (Figs 26–27) in the apex of cucullus being more slender and in possessing a more elongate, tapered uncus.
http://zoobank.org/06C8414F-FC41-4DD7-BAB9-CCA4F5B3BC83
http://species-id.net/wiki/Erechthias_grayi
Figs 7–10, 18, 28–31Adult (Figs 7, 8). Head: Scales generally slender with bidentate apices; scales of frons smooth, appressed, directed dorsad, pale brown to gray on lower frons becoming dull white to pale gray at top of frons; scales of vertex erect or mostly so, especially over occipital tufts, fuscous, some with grayish white apices. Labial palpus with scales flattened and appressed dorsally, mostly dark grayish brown with scattered paler scales; venter of second segment with a dark brush of long, slender, erect scales and a lateral series of ~ 6-8 long, dark bristles; 1–2 bristles also arising laterally from basal segment. Maxillary palpus elongate, 5-segmented, approximately as long as labial palpus. Antenna ~ 1.6× the length of forewing; scales smoothly appressed, dark fuscous; scales of scape and pedicel moderately broad; flagellum without cilia and with a single row of more slender scales completely encircling each segment.
Thorax: Forewing brachypterous (Fig. 18), length 1.4–1.8 mm. Dorsum of thorax dark fuscous dorsally, with a few dull white scales at caudal margins of tegula and mesonotum; mostly grayish white ventrally. Forewing similar to dorsum in color, dark fuscous with an irregular scattering of dull white scales at base of wing and mostly crossing wing beyond middle; a slightly larger concentration of dull white scales at apex and extending a short distance along costa; fringe almost completely lacking, restricted to apex. Hindwing minute (Fig. 18), slightly variable in size, without scales; length ~ 0.15 mm; a single stout frenulum present in male ~ equal to length of hindwing (frenulum not examined in female); fringe absent. Fore and midlegs fuscous, lightly irrorated with pale grayish white scales; apices of tibia and tarsomeres ringed with grayish white; hindleg generally paler in color.
Abdomen: Dark fuscous dorsally, mostly grayish white ventrally. Eighth segment without coremata.
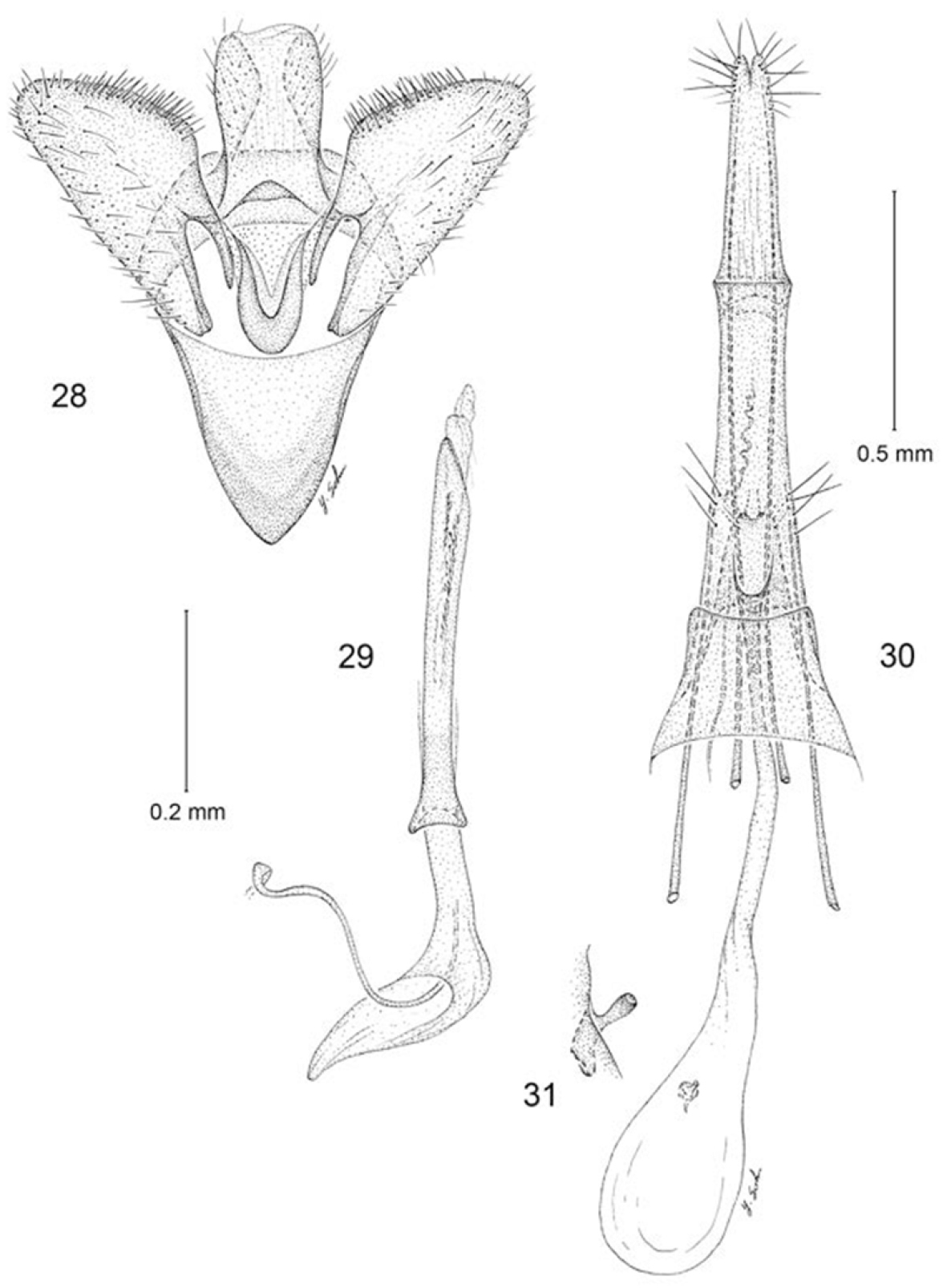
Male genitalia (Figs 28, 29): Segment 10 mostly membranous; uncus lobes indistinct, broadly rounded. Tegumen consisting of a relatively narrow dorsal ring. Vinculum broad, V-shaped, gradually tapering anteriorly with an acute anterior apex; vinculum ~ 0.7× the length of valva. Valva simple; cucullus broadly triangular with narrowly rounded apex; costal margin densely setose. Juxta well developed as an elongate U-shaped pouch. Aedeagus slender, ~ 1.3× length of valva; vesica with numerous, minute, spicular cornuti; base of aedeagus moderately flared, not divided.
Erechthias grayi, genitalia 28 Male, ventral view(1 mm) 29 Aedeagus, ventral view 30 Female, ventral view 31 Detail of signum in Fig. 30.
Female genitalia (Figs 30, 31): Eighth sternite weakly sclerotized; ostium opening near anterior margin; an irregular cluster of ~ 5 pairs of long setae encircling caudal margin of eighth segment. Antrum slender, length ~ 3× maximum width. Ductus bursae slender, elongate, slightly longer than anterior apophysis, gradually enlarging to moderately large, ovate corpus bursae; walls of corpus bursae membranous except for very small, elongate signum; distal, more slender half of signum projecting beyond wall of corpus bursae.
The species name is a patronym for Alan Gray, a botanist who assisted Howard Mendel with the collection of this species on Ascension Island.
♂, ASCENSION ISLAND: Green Mountain, 743 m, Elliot’s Path, (Windy Corner), GPS 7.57S, 14.21W: 6 Aug. 2003, H. Mendel, BMNH(E) 2003-137, digital image captured (BMNH).
ASCENSION ISLAND: same locality as holotype: 11 ♂, 1 ♀, 13 Dec. 2005, H. Mendel, BMNH slide 33642 ♀, BMNH(E) 2006-13; 4 ♂, 21 Nov. 2012, H. Mendel and A. Gray, USNM slide 34532 ♂. ASCENSION ISLAND: White Horse Hill [Little White Hill], S. E. Bay: 2 ♂, 23 Aug. 2012; [pitfall trap]; Ms L. White, USNM slide 34533 ♂. White Horse Rock [Little White Hill, S. E. Bay]: 2 ♂, 29 May 2013; running over lichen covered rock; pooter; leg. A. Wakeham-Dawson; digital image captured. (BMNH, USNM).
(Fig. 1). Ascension Island. Erechthias grayi was at first thought to be confined to the higher elevations of Green Mountain where it was found on several occasions at altitudes around 743 m, in a very moist area frequently shrouded in cloud. Recent captures at Little White Hill, an extremely arid area at altitudes below 200 m show that Erechthias grayi is tolerant of a wide range of conditions. What the two areas do have in common is that the vegetation and habitat are comparatively undisturbed. Probably, Erechthias grayi would have been more widespread on Ascension prior to human habitation and the disturbances accompanying human settlement.
Becauseadults have been collected in close association with lichen covered rocks, it is likely the larvae are lichenivorous, a frequently used food source in this group of moths. Several (13) small (3.5–4.2 mm long) mature larval cases, some with pupal exuviae attached (Figs 9, 10), were collected under lichen covered rocks at White Horse Hill, on the same day that an adult was collected there by A. Wakeham-Dawson. It is likely that these are the larval cases of Erechthias grayi, but larval rearings will need to be conducted to confirm this. The cases are mostly white, speckled with small grains of sand and minute, dark fragments from the rocky substrate.
The behavior of the moths in the field was unusual. They would cling firmly to the bare rock, lichens (Fig. 3), and small plants in exposed situations and were only seen to move when disturbed, and then reluctantly. They would hop a few inches in a very bug-like (Heteroptera) manner and were most easily collected using an aspirator (pooter). Wakeham-Dawson (see paratype data) observed adults running over a lichen covered rock at White Horse Hill.
Erechthias grayi is the only species within this large genus known to possess brachypterous adults, and thus is the most distinctive moth within Erechthias. The male and female genitalia of Erechthias grayi are most similar to those of Erechthias darwini Robinson (1983), one of the few species of Lepidoptera known to inhabit St. Paul’s Rocks (Pedro e Săo Paulo, Fig. 1), located slightly over 1000 miles northeast of Ascension Island. The male saccus of Erechthias grayi is more triangular and the female signum is more slender than those of Erechthias darwini. Most significantly, the adults of Erechthias darwini (Fig. 11) are fully winged and capable of flight. However, considering their genitalic similarities and geographical proximity, it is possible that grayi may have shared relatively recent common ancestrywith darwini.
Reportedly, some form of wing reduction has occurred in either 25 (
HM had the good fortune to visit Ascension on three occasions, and the new Erechthias was collected each time. The first trip (August 2003) was greatly assisted by Dr Alan Gray, who was commissioned to carry out an environmental impact assessment on a proposed development at the US airbase there. The second trip (December 2005) consisted of a few days in transit en route to St. Helena on a project (
We thank Young Sohn, Karolyn Darrow, and Donald Harvey of the Department of Entomology, Smithsonian Institution for the line drawings and graphics support. Kevin Tuck of the Department of Life Sciences (Entomology), Natural History Museum, London, was very helpful in providing images of Erechthias darwini and Erechthias dracaenura, and for arranging loans of specimens.