(C) 2012 Paul E. Marek. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
With up to 750 legs, the millipede Illacme plenipes Cook and Loomis, 1928 is the leggiest animal known on Earth. It is endemic to the northwestern foothills of the Gabilan Range in San Benito County, California, where it is the only known species of the family Siphonorhinidae in the Western Hemisphere. Illacme plenipes is only known from 3 localities in a 4.5 km2 area; the 1926 holotype locality is uncertain. Individuals of the species are strictly associated with large arkose sandstone boulders, and are extremely rare, with only 17 specimens known to exist in natural history collections. In contrast with its small size and unassuming outward appearance, the microanatomy of the species is strikingly complex. Here we provide a detailed redescription of the species, natural history notes, DNA barcodes for Illacme plenipes and similar-looking species, and a predictive occurrence map of the species inferred using niche based distribution modeling. Based on functional morphology of related species, the extreme number of legs is hypothesized to be associated with a life spent burrowing deep underground, and clinging to the surface of sandstone boulders.
California Floristic Province, paleoendemic, endemic, silk, San Benito County, Silicon Valley, Salinas Valley, sandstone, burrowing, conservation, Gabilan Range
The millipede Illacme plenipes has more legs than any other known organism, with one female individual possessing 750 legs on 192 body segments. The Siphonophorida, the order in which Illacme plenipes is placed, comprises a diversity of taxa that have fascinating anatomical features, biogeographical patterns, and very intriguing biology. Siphonophoridan species are mainly Pantropical in distribution with a few outlying taxa in the Himalayas, New Zealand, South Africa and California (
Like many other colobognath millipedes, the Siphonophorida often occur in cryptic subterranean habitats, shun light, are infrequently encountered, and therefore are rare in natural history collections. All known taxa are eyeless and have relatively large antennae. Species of the family Siphonophoridae have the front of the head drawn out into a long, narrow extension that is paralleled by a similar extension of the gnathochilarium, forming a tube that encloses reduced, stylet-like mandibles. Fungivory, the consumption of soft fungal tissues and spores, may be linked to this suite of adaptations. Siphonorhinids, in contrast, do not have this “beak” and the head is not strongly modified. The siphonorhinid gnathochilarium has all of its elements indistinguishably fused and is tightly appressed to the ventral surface of the head, leaving only a small opening anteriorly, which may be homologous to the labral indentation in eugnathan millipedes.
The cuticle of Illacme plenipes isadorned with a surprising diversity of peculiarly shaped spines, teeth, setae, sensilla, and other phaneres. Numerous setae clothing the dorsum of the millipede appear to secrete a viscous silk-like substance. The posterior one-third of its gut (the metenteron) is spiraled and visible through its translucent exoskeleton.
Illacme plenipes was described by O.F. Cook and H.F. Loomis in 1928 from seven individuals collected from a site located “a short distance after crossing the divide between Salinas and San Juan Bautista…in a small valley of a northern slope wooded with oaks, under a rather large stone” (
In 2005 and 2007, new specimens were collected from near the type locality (
Following the locality description of
As an approach to understanding species ecology and geography, a niche-based distribution model (DM) was constructed for Illacme plenipes. Niche-based DMs provide estimates for the probability of finding a species at a particular location and general area on a landscape given a known set of coincident ecological and climatic parameters for the species. Locality coordinates for each species were imported into ArcMap (ESRI, Redlands, CA) and converted into shape files. Following the procedure outlined in
Specimens from which DNA was not extracted (typically longer females possessing more than 170 segments) were directly preserved in 80% ethanol. The posterior seven segments of two specimens (# SPC000924 and SPC001187) were dissected from live individuals with flame-sterilized forceps and stored in RNAlater (Qiagen Inc., Valencia, CA) at 10°C for 24h, and subsequently at -80°C for long-term preservation and archival storage of DNA and RNA. The enteron was removed from the segments to prevent contamination due to the DNA or RNA of the millipede’s gut contents. Specimens from which DNA was extracted were subsequently preserved in 70% isopropanol.
DNA barcodingGenomic DNA was extracted from frozen tissue preserved in RNAlater using standard DNeasy tissue extraction protocol (Qiagen Inc., California). Extracted DNA was purified from a fragment of the millipede (specimen #SPC001187) approximately four segments in length, with remaining tissue archived at -80°C in RNAlater. Genomic DNA is archived in Qiagen AE buffer at -20°C and stored in the cryo-collections at the University of Arizona and Auburn University. A region of DNA from the cytochrome c oxidase I gene (COI), was amplified using polymerase chain reaction (PCR) with the universal DNA barcoding primers of
Illacme plenipes is represented in natural history museum collections by 17 known specimens, which includes type and non-type material. These specimens were borrowed from the following repositories: Florida State Collection of Arthropods (FSCA), Smithsonian Institution (USNM), and Virginia Museum of Natural History (VMNH). Newly collected material, compared with historical type specimens to confirm species identity, was subsequently georeferenced and databased. The precise locations of recently collected specimens are not plotted on the distribution map; instead, a circle around the coordinates is shown to preserve the confidentiality of sensitive habitat (Fig. 1). Type specimens collected by Cook in 1926 are from an imprecise location on “San Juan grade above Salinas, San Juan Bautista, Calif. Nov. 27, 1926”. However, based on the description, it pro-bably lies on the north side of the Gabilan Range on San Juan Grade Road or Old Stage Road in a radius of 4 km around the coordinates 36.831371°N, -121.562808°W. Due to sensitivity of the habitat and extreme rarity of individuals, locality coordinates from georeferenced material is available upon request from the corresponding author. All of the material (including types and non-type material) was measured, examined in detail and is listed in the “Material examined” section. Specimens were measured at 18 locations on the exoskeleton to summarize continuous morphological variation: (1) body length from anterior margin of labrum to posterior margin of paraprocts, BL; (2) head width, HW; (3) head length, HL; (4) interantennal socket width, ISW; (5) antennomere 6 width, AW; (6) collum width, CW; (7) metazonite width at 1/4 length of body, W1; (8) metazonite width at mid-length of body, W2; (9) metazonite width at 3/4 length of body, W3; (10) metazonite length at 1/4 length of body, L1; (11) metazonite length at mid-length of body, L2; (12) metazonite length at 3/4 length of body, L3; (13) metazonite height at 1/4 length of body, H1; (14) metazonite height at mid-length of body, H2; (15) metazonite height at 3/4 length of body, H3; (16) first apodous metazonite width, AS1; (17) anterior gonopod article 5 width, A5W; and (18) posterior gonopod article 5 width, P5W. Body length was measured from digital photographs of specimens captured through the eyepieces of a Leica M125 stereomicroscope (Wetzlar, Germany) with an iPhone 4 (Apple, Cupertino, CA) using the segmented line measurement tool in ImageJ64 (
The data underpinning the analysis reported in this paper are deposited in the Dryad Data Repository at https://doi.org/10.5061/dryad.3b3h8 and in the National Center for Biotechnology Information’s genetic sequence database GenBank under the accession numbers: JX962721 – JX962725 (http://www.ncbi.nlm.nih.gov ).
Results FieldworkIndividuals of Illacme plenipes were found at three localities, geographically separated by a maximum of 4.5 aerial km. The first collecting event was on 29 November 2005, the second on 8 December 2005, and the third 16 December 2007. One survey, at which time specimens were found but not collected, occurred 27 January 2006. Each locality is in the northwestern Gabilan foothills no more than 4.5 aerial km from the mission at San Juan Bautista and 3.2 aerial km southwest of the San Andreas Fault. Illacme plenipes were not found in any of the other sites investigated. Individuals were found in moist oak-wooded valleys beneath large arkose sandstone boulders (approximate mean mass = 40 kg), clinging to the surface usually about 10 – 15 cm below the top of the soil. Illacme plenipes specimens were always found on these boulders and underground, either on the stone surface, in the lacuna between the stone and the soil, or partially imbedded in the soil horizon. Specimens were never found directly on the normally dry bottom of the stones, or on fallen logs or any other decaying organic matter or detritus. Illacme plenipes were consistently discovered by closely examining the stone surface (approximately 10–15 cm below soil) and the edge of the crater after removing the stone. Nine additional specimens, comprising 4 males, 4 females, and a juvenile were found throughout 2005–2007 in three localities (increasing the total number of specimens for Illacme plenipes, which includes the type series, to 17 total: 6 males, 10 females, and a juvenile). Illacme plenipes were uncommon at every locality and individuals were only found after one hour of two persons surveying a suitable-appearing site. Individuals were typically encountered beneath the stones singularly; no more than two individuals were ever found simultaneously.
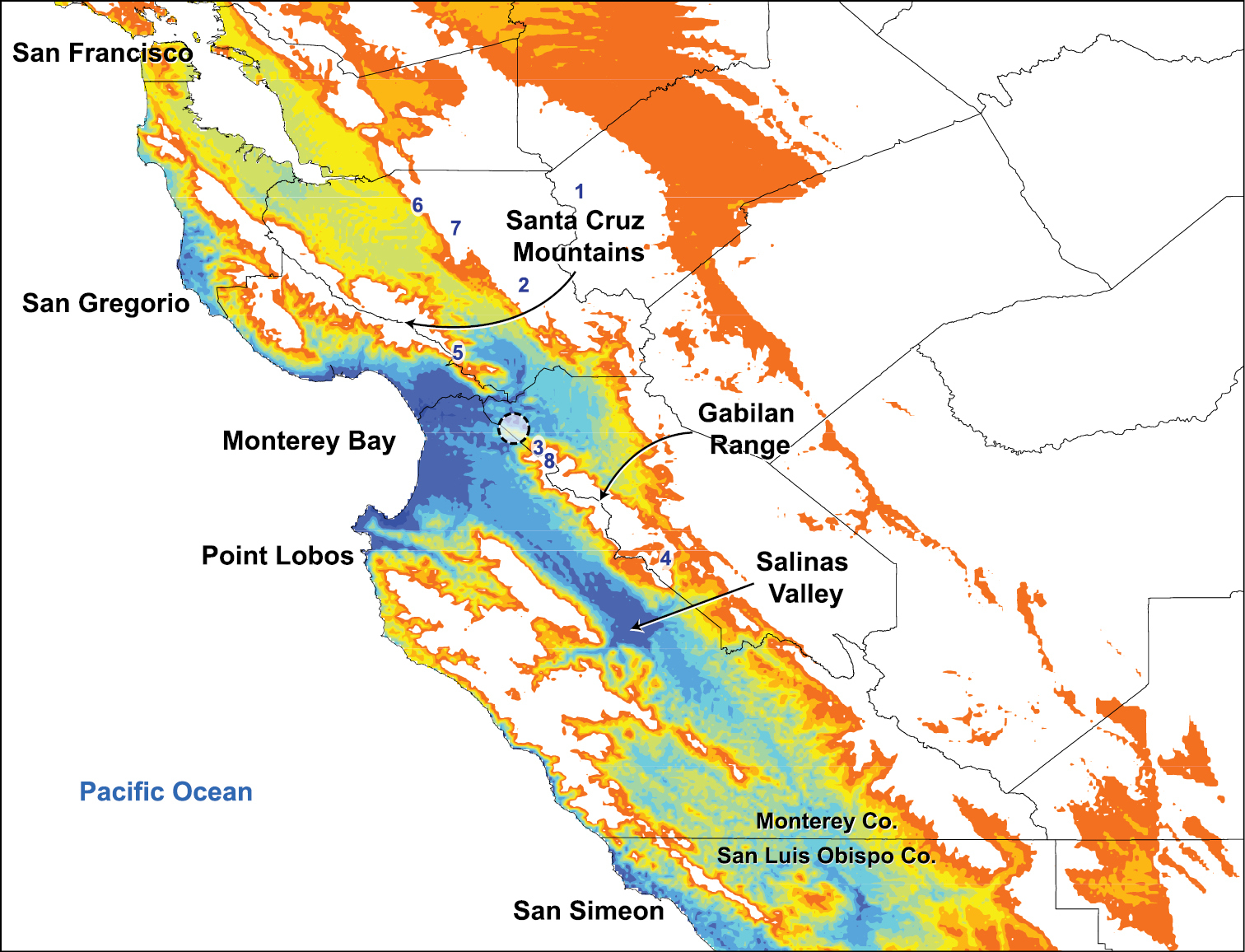
Ecological niche modelingThe niche-based distribution model for Illacme plenipes indicates the highest probability of occurrence, representing ecological suitability for the species, in the terrestrial areas on the periphery of Monterey Bay extending just past the gap between the Santa Cruz Mountains and Gabilan Range and throughout the Salinas Valley (Fig. 1). Areas of medium to high probability extend from Monterey Bay along a thin region on the coast northward to San Gregorio and southward to Point Lobos. There are other areas of medium to high probability, also restricted to the coast, between San Simeon in the north and the western boundary between Monterey and San Luis Obispo counties.
Niche-based distribution model inferred in Maxent. The model indicates predicted habitat suitability for Illacme plenipes based on climatic variables extracted from known geographical coordinates of the species. High levels of habitat suitability are denoted in blue and low levels in red (reverse heat map). Coordinates of recently collected specimens are indicated by a circle around the locations (northwest of the Gabilan Range) to preserve the confidentiality of sensitive habitat. Localities surveyed for additional populations of Illacme plenipes: 1 Frank Raines Park 2 Henry Coe State Park 3 Fremont Peak State Park 4 Pinnacles National Monument 5 Mount Madonna County Park 6 Alum Rock 7 Joseph D. Grant County Park 8 El Rancho Cienega del Gabilan.
Polymerase chain reaction of the COI barcoding region, when electrophoresed and visualized on a 12% agarose gel, recovered single bands of uniform lengths in all species. Sanger sequencing resulted in sense/antisense chromatograms reads of ~600 bp in length when contiguous fragments were assembled in Mesquite. Mean Phred quality scores of individual contigs are between 73–80. When aligned and ragged ends trimmed, sequence length is invariant between species. Mean nucleotide percent sequence difference between species is 25% and between amino acid sequences (total difference), 17%. The NCBI GenBank accession numbers are as follows: Illacme plenipes (JX962724), Gosodesmus claremontus (JX962723), Brachycybe producta (JX962721), Brachycybe rosea (JX962722), and Siphonacme lyttoni (JX962725). The COI barcodes of the Siphonophorida species (Illacme plenipes and Siphonacme lyttoni) and the Platydesmida species (Gosodesmus claremontus, Brachycybe producta, and Brachycybe rosea) are hitherto the only that exist for these two orders; there is only one other DNA barcode for the entire subterclass Colobognatha. The following species are listed in order of increasing percent nucleotide difference from Illacme plenipes, indicated in parentheses (mean percent difference of amino acids proceeds after the “/”): Gosodesmus claremontus (28.7% / 23.8%), Brachycybe producta (29.7% / 24.4%), Siphonacme lyttoni (29.9% / 22.3%), and Brachycybe rosea (30.6% / 24.4%).
Taxonomy Class Diplopoda de Blainville in Gervais, 1844 Subclass Chilognatha Latreille, 1802/1803 Infraclass Helminthomorpha Pocock, 1887 Subterclass Colobognatha Brandt, 1834 Order Siphonophorida Hoffman, 1980 Family Siphonorhinidae Cook, 1895http://species-id.net/wiki/Illacme
Illacme plenipes
Illacme is placed with other taxa in the family Siphonorhinidae based on the following characters: Head pear-shaped (♂) or triangular (♀), not elongate or bird beak-shaped, as in the Siphonophoridae (Fig. 2, Morphbank 805574, Appendix I). Antennae elbowed between antennomeres 3, 4 (Fig. 3, Mb-805578). Antennomeres 5, 6 with apical dorsal cluster of 7 or 8 basiconic sensilla (Bs2) in slight depression, not deep-set into circular pits, as in the Siphonophoridae (Fig. 4, Mb-805575). Posterior gonopods with distal podomere divided into 2 or 3 branches (Fig. 5, Mb-805576, Fig. 6c). See also diagnoses of Illacme in Shelley (1996b, p. 23) and of Siphonorhinidae in Shelley and Hoffman (2004, p. 218).
2 Lateral (right) view of head and segments 1–5 (♂). a Lateral opening apparent between gnathochilarium and head capsule; gnathochilarium, mandible and head capsule noticeably separate at base, 1/3 head length distally from mandibular joint b Collum not covering head, with straight cephalic edge, gradually tapering laterally. Scale bar 0.5 mm. 3 Ventral view of head, antennae and segments 1 – 5 (♂).Scale bar 0.3 mm. 4 Lateral (right) view of antennomeres 5, 6 (♂).Arrow, small basiconic sensilla (Bs2) in cluster of 7 or 8 oriented apical dorsally (retrolaterally) in slight depression on antennomeres 5, 6. Scale bar 0.05 mm. 5 Oblique (right) view of right posterior gonopod (♂).Posterior gonopodal podomere 6 divided, comprising a bundle of 3 stylus-shaped articles. Scale bar 0.05 mm.
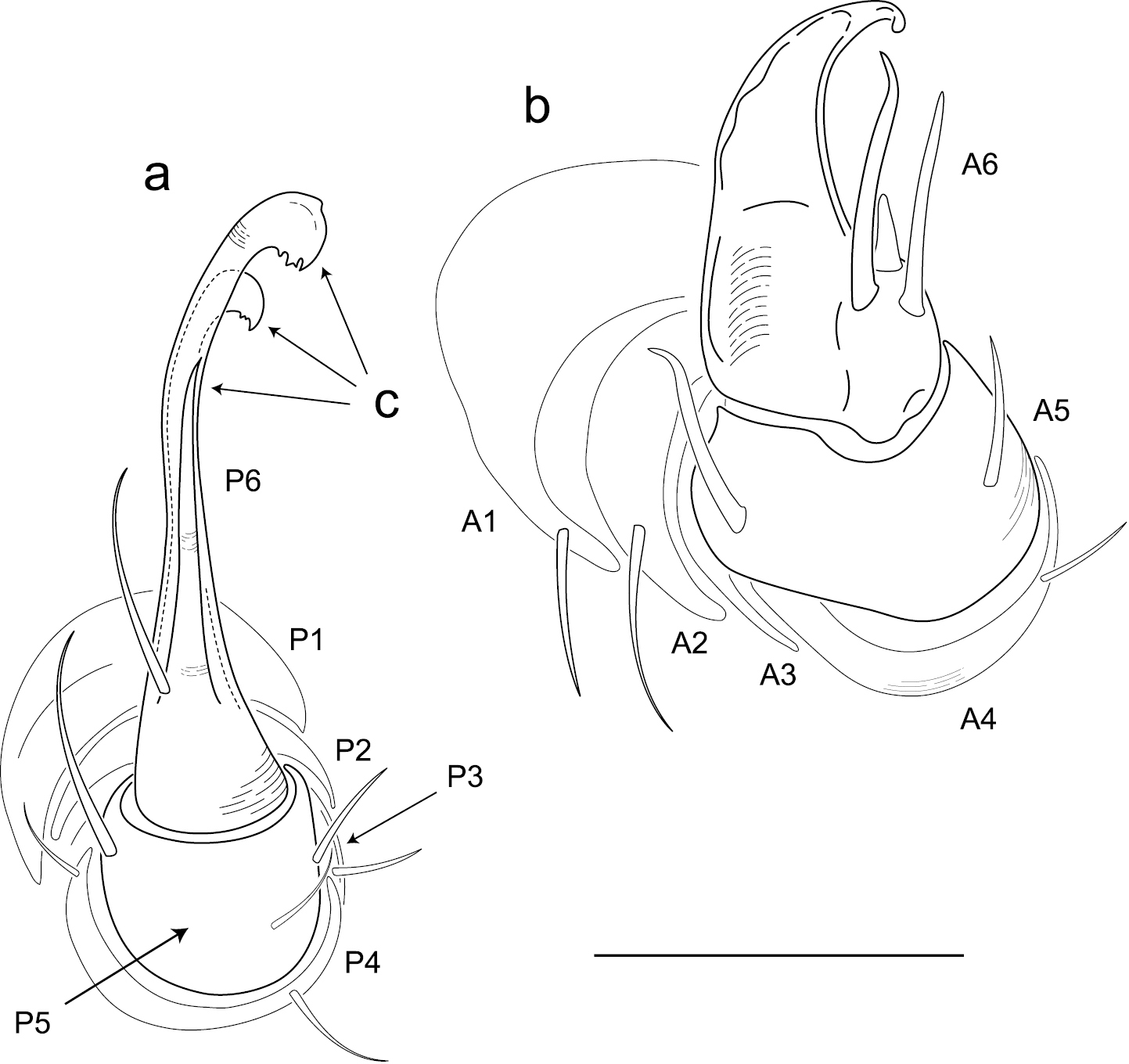
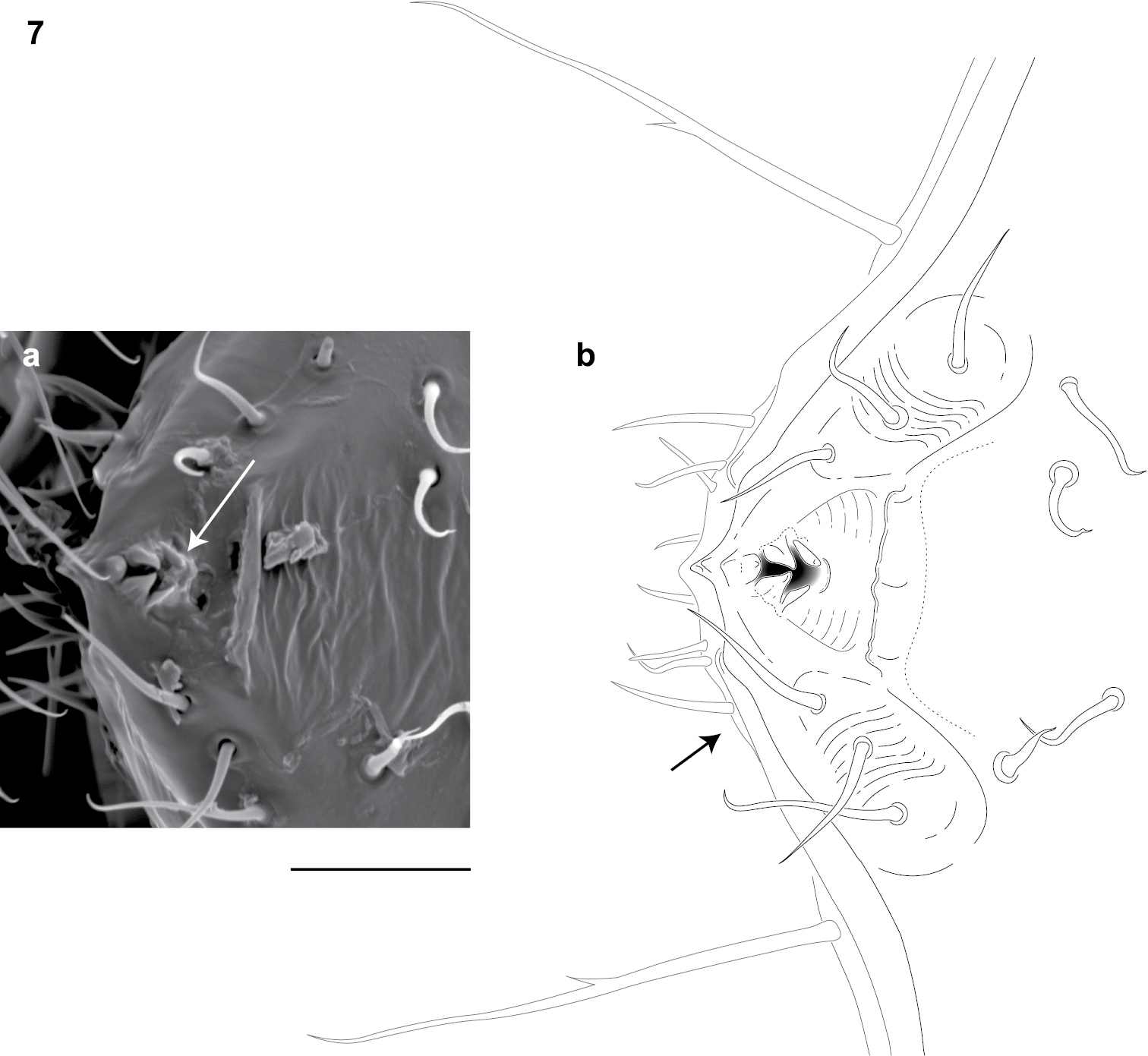
Adults of Illacme are distinct from other siphonorhinid genera (and commonly-encountered millipedes co-occurring with Illacme plenipes) based on the combination of – Exoskeleton: Body light cream-colored, thread-like, extremely narrow and long (max. width: ♂ 0.55, ♀ 0.64; max. length: ♂ 28.16, ♀ 40.40). Adult individuals with 84 – 192 segments, and with 318 – 750 legs (VMNH paratype ♀ with 192 segments and 750 legs, more than any other organism known on Earth). Body with hirsute vestiture, appearing velvety (Fig. 2, Mb-805577). Antennae elbowed between antennomeres 3, 4 (Figs 2, 3, Mb-805578). Antennomeres 5, 6 enlarged, appearing oversized relative to other millipedes (Figs 2, 3, Mb-805579). Head pear-shaped (♂) or triangular/chevron-shaped (♀), eyeless (Figs 2, 3, Mb-805574, Appendix I). Mouthparts (gnathochilarium, mandibles) and labrum tightly appressed, tapered anteriorly to rounded apex, not bird beak-shaped, as in the Siphonophoridae (Fig. 3, Mb-805586). Labrum with triangular tooth-lined orifice (Fig. 7a, b; Mb-805580). Denticulate shelf-like carina, projecting dorsally from labrum-epistome margin (Fig. 8a, b; Mb-805588). Internal anatomy. Posterior one-quarter length of enteron loosely spiraled; when alive, visible through translucent cuticle (Fig. 9, Mb-805582). Male gonopods. 9th and 10th leg pairs modified into gonopods, each comprising 6 podomeres (Fig. 6a, b). Anterior gonopod thick, more robust than posterior gonopod (Fig. 10, Mb-805583, Fig. 6b). Anterior gonopodal apex (podomere 6, Fig. 6a, A6) shovel-shaped; in repose, cupped sheath-like around flagelliform posterior gonopodal apex (podomere 6, Fig. 11, Mb-805584, Fig. 6b, P6). Posterior gonopodal podomere 6 divided, comprising a bundle of 3 stylus-shaped articles (Fig. 5, Mb-805627, Fig. 6a, P6); remaining siphonorhinid taxa have 2 stylus-shaped articles with a small spine (Nematozonium filum) or 2 articles without a spine (Siphonorhinus Pocock, 1894 species and Kleruchus olivaceus Attems, 1938). 2 dorsal-most, longest articles of P6 laminate distally and recurved laterally, with denticulate posterior margins appearing claw-like (Fig. 12, Mb-805585, Fig. 6a, P6). Ventral-most, shortest article of P6 acuminate distally, spike-like. Habit in life. Movement very slow, nearly imperceptible (Appendix II, III). Antennae movement rapid, independent. Terminal antennomeres held flat and rapidly tap substrate and surroundings (Appendix IV).
Illustration of anterior and posterior gonopods (♂). a Posterior gonopod with podomeres labeled P1-6 b Anterior gonopod with podomeres labeled A1-6 c 3 stylus-shaped articles. Scale bar 0.05 mm.
Dorsal view of anterior region of head and labrum (♂). a Scanning electron micrograph: arrow, labrum with triangular tooth-lined orifice b Line drawing: shaded area, triangular tooth-line orifice; arrow, gnathochilarium. Scale bar 0.02 mm.
http://species-id.net/wiki/Illacme_plenipes
Type specimens: ♂ holotype (USNM), 1♂, 3♀ paratypes (FSCA) and 3♀ paratypes (VMNH)—from United States, California, San Benito County, from “near divide between Salinas and San Juan Bautista” [an imprecise location probably on the north side of the Gabilan Range on San Juan Grade Road or Old Stage Road in a radius of 4 km around the coordinates 36.831371°N, -121.562808°W], 27.xi.1926 (Coll. O.F. Cook). Non-type specimens: California, San Benito County: 1♂ (SPC000924), 2♀ (SPC000930, -931), Gabilan Range, San Juan Bautista, 29.xi.2005 (Colls: P. and R. Marek); 3♂ (SPC000932, -933, -934), 1 juvenile (SPC000935), loc. ibid., 8.xii.2005, (Coll: J. Bond). 2♀ (SPC001187, MIL0020), Gabilan Range, San Juan Bautista, 16.xii.2007, 13:00 (Colls: P. and R. Marek).
(See generic diagnosis.)
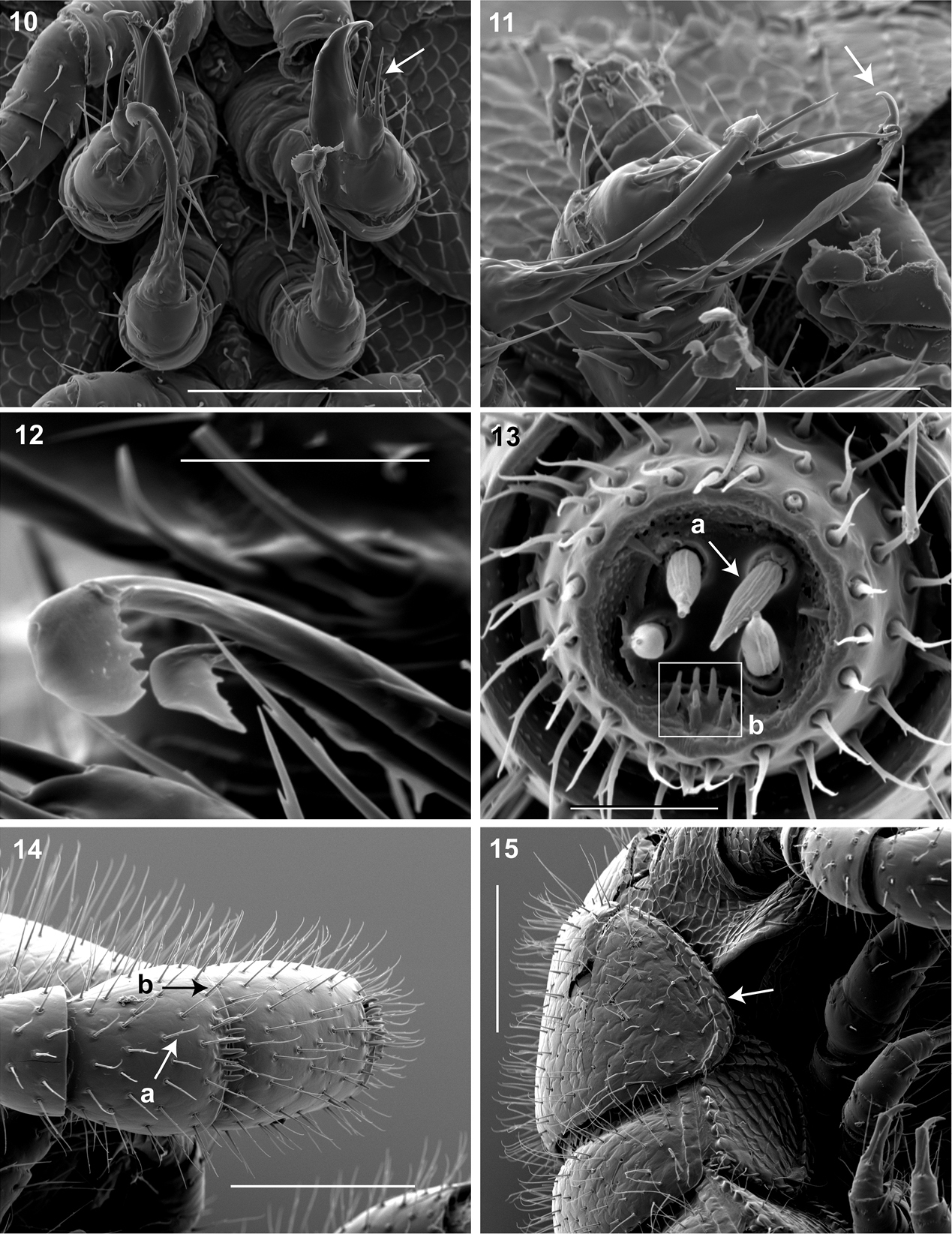
(♂) USNM TYPE NO. 976 – Counts and measurements: p = 143. a = 2. l = 562. (143 + 2 + T). HW = 0.30. HL = 0.34. ISW = 0.20. AW = [antennae missing]. CW = 0.42. W1 = 0.53. W2 = 0.55. W3 = 0.55. L1 = 0.20. L2 = 0.20. L3 = 0.18. H1 = 0.31. H2 = 0.30. H3 = 0.33. AS1 = 0.45. A5W = 0.05. P5W = 0.04. BL = 28.16. Head pear-shaped, tapered anteriorly to round point at a 160° angle anterior from antennal sockets; occipital area posterior from antennal sockets gradually curved medially towards neck (Figs 2, 3, Mb-805574—note: all SEMs herein are images of specimen #SPC000932, not the holotype). Head pilose, covered with long, slender setae (Fig. 2, Mb-805577). Mouthparts (gnathochilarium, mandibles) and labrum tightly appressed, tapered anteriorly to round point (Fig. 3, Mb-805586). Gnathochilarium elements (stipes, promentum, etc.) indistinguishably fused, tightly appressed to the ventral surface of the head, leaving a small opening anteriorly. Lateral opening apparent between gnathochilarium and head capsule (Fig. 2a, Mb-805587). Mandibles thin, stylet-like, with heavily calcified apices (viewed dorsally through translucent head capsule at 400× with a compound microscope). Labrum with triangular tooth-lined orifice (Fig. 7a, b; Mb-805580). Denticulate shelf-like carina, projecting dorsally from labrum-epistome margin (Fig. 8a, b; Mb-805588). Gnathochilarium, mandible and head capsule noticeably separate at base, 1/3 head length distally from mandibular joint (Fig. 2a, Mb-805589). Antennae sub-geniculate, elbowed between antennomeres 3, 4, comprising 7 antennomeres (Fig. 3, Mb-805578). Antennae massive distally; antennomeres 5, 6 enlarged (Fig. 3, Mb-805579). Five sensillum types: 4 apical cones (AS) oriented in a trapezoidal cluster on 7th antennomere, with longitudinally grooved outer surface and apical circular invagination (Fig. 13, Mb-805590). Chaetiform sensilla (CS) widely spaced on antennomeres 1-7, each sensillum with 2 or 3 barbules (Fig. 14a, Mb-805591). Trichoid sensilla (TS) oriented apically encircling antennomeres 1–7, lacking barbules (Fig. 14b, Mb-805592). Small basiconic sensilla (Bs2) in clusters of 7 or 8; in slight depressions oriented apical dorsally (retrolaterally) on antennomeres 5 and 6; smooth, finger-shaped, 1/2 length of chaetiform sensillum (Fig. 4, Mb-805593). Spiniform basiconic sensilla (Bs3) in cluster of 5, oriented apical dorsally on 7th antennomere; tips facing apical cones (on longitudinal axis with Bs2 on antennomeres 5, 6); each sensillum with 2 barbules (Fig. 13b, Mb-805594). Antennae extend posteriorly to middle of 3rd tergite. Relative antennomere lengths 6>2>5>3>4>1>7. Segments:Collum not covering head, with straight cephalic edge, gradually tapering laterally (Fig. 2b, Mb-805595). Collum with carina present on anterolateral margin, appearing scaly (Fig. 15, Mb-805596). Carina repeated serially on lateral tergal and pleural margins (absent from telson). Lateral tergal and pleural carinae jagged, pronounced on midbody segments (Fig. 16a, Mb-805597). Lateral margin of collum round. Tergites: Metazonites rectangular, 3× wider than long, slightly convex (Fig. 17, Mb-805598). Paranota absent. Metazonite dorsal surface pilose, covered with long, slender setae (Fig. 2, Mb-805599). Tergal setae hollow, cavity diameter 1/8 that of setae diameter; tipped with silk-like exudate, tangled, appearing adhered to neighboring setae (Fig. 18, Mb-805600). (NB: Tergal silk-like exudate observed in scanning electron micrographs, and by the observation of fine strands issuing from the metaterga of live individuals, viewed while magnified at 80× with a stereomicroscope. Silk stickiness was indicated by increased adherence of soil particles after handling and live observation of the millipede’s coiled body becoming stuck together.) Metazonite posterior margin (limbus) lined with posteriorly projecting anchor-shaped spikes and a row of conical spikes just dorsal to anchor-shaped spikes (Fig. 17a, Mb-805601). Anchor-shaped spikes alternating in size (large, small) along row. Ozopores oriented dorsally, located near limbus, absent from tergites 1 – 3 and telson. Ozopores elevated slightly (porosteles absent), with 2 stout posteriorly projecting spines and encircled by 13 – 15 robust setae (Fig. 19, Mb-805602). 3 or 4 stout flat tubercles opposite ozopore near anterior margin, lunate arrangement encircling ozopore (Fig. 17b, Mb-805603). Posterior tergites more convex, covered with a greater density of long, slender “silk”-exuding setae (Fig. 20, Mb-805604). Lunate-arranged tubercles opposite ozopores on posterior metazonites: conical and spiked, not flat. Apodous segments lacking sterna, pleurites contiguous in midline. Apodous tergites densely setose, covered with unevenly distributed spikes (Fig. 21, Mb-805605). Telson densely covered with irregularly oriented and unevenly distributed stout spines; posterior margin lined with variably-shaped posterodorsally oriented anchor-shaped spikes. Tergal tubercles and spikes: consistently projecting posteriorly, occasionally posterodorsally. Prozonite highly sculptured, with 5 rows of discoidal flat tubercles; anterior 3 rows staggered and posterior 2 rows aligned (Fig. 22, Mb-805606). Pleurites quadrate, flat, with jagged scaly lateral, posterior and medial margins (Fig. 16, Mb-805609). Pleurite medial margin broad, with scaly carina (Fig. 16b, Mb-805610). Left and right pleurites plate-like, comprising 4/5’s of ventral segment space. Left and right pleurites broadly overlapping sternite, covering spiracles (Fig. 23, Mb-805612). Sternites free, separate from pleurites; heart-shaped, wider anteriorly. Sternal surface with broad, jagged scales. Medial sternal ridge projecting ventrally, with spiracles and legs oriented ventrally (Fig. 24, Mb-805614). Spiracles circular, orifice open; oriented dorsally above legs (Fig. 25, Mb-805615). Anterior and posterior sternites separate. Tergites, pleurites and sternites separated by arthrodial membrane (Fig. 20, Mb-805616). Arthrodial membrane between tergites and pleurites wider posteriorly. Telson pilose, covered with long, slender posteriorly recurved setae (Fig. 20, Mb-805628). Paraprocts semihemispherical, anterior margins slightly scaly. Epiproct absent. Hypoproct small, one-eighth area of paraproct, with row of posterior projecting setae. Legs: six subequally shaped podomeres, with coxa slightly shorter and tarsus slightly longer. Legs with sparse setae, appearing similar to trichoid sensilla, with 2 or 3 barbules. Coxae nearly contiguous medially, separated by thin sternal ridge. Large posteroventral D-shaped opening for eversible sac (Fig. 26, Mb-805618). Eversible sacs membranous, bulging slightly from opening (Fig. 24b, Mb-805620). Pregonopodal tarsus with stout bifurcate claw; dorsal subdivision thicker, more arcuate (Fig. 27, Mb-805621). Postgonopodal tarsus with two separate claws, co-terminal on tarsal apex; dorsal claw thick and arcuate, ventral claw thin and setiform (Fig. 16c, Mb-805623). 2nd leg pair with posteriorly oriented coxal gonapophyses; rounded, protuberant, one-third length of prefemur. Gonopods: 9th, 10th leg pairs modified into gonopods, each comprising 6 podomeres (Fig 6a, b). Anterior gonopod thick, more robust than posterior gonopod (Fig. 10, Mb-805583, Fig. 6b). Anterior gonopodal apex (podomere 6) shovel-shaped; in repose cupped sheath-like around flagelliform posterior gonopodal apex (podomere 6, Fig. 11, Mb-805584). Posterior gonopodal podomere 6 divided, comprising a bundle of 3 stylus-shaped articles (Fig. 5, Mb-805627, Fig 6a). 2 dorsal-most, longest articles of P6 laminate distally, recurved laterally, denticulate posterior margins, appearance similar to a chicken foot in rigor mortis (Fig. 12, Mb-805585, Fig 6a). Ventral-most, shortest article of P6 acuminate distally, spike-like. Thin ridge-shaped sterna present between left and right gonopods, thicker between anterior gonopods.
Lateral (right) view of antennal and cephalic apices (♂). a Scanning electron micrograph: arrow, denticulate shelf-like carina, projecting dorsally from labrum-epistome margin. Scale bar 0.1 mm b Line drawing: top arrow, shelf-like carina; middle arrow, triangular tooth-lined orifice; bottom arrow, gnathochilarium. Scale bar 0.01 mm.
Illacme plenipes ♀ with 170 segments and 662 legs (specimen # SPC000931). Top inset, 2× magnified view of posterior segments with corkscrew-shaped metenteron visible through cuticle; bottom inset, 3× magnified illustration of corkscrew-shaped metenteron. Scale bar 1 mm.
10 Ventral in situ view of gonopods (♂). Arrow, anterior gonopod thick, more robust than posterior gonopod. Scale bar 0.1 mm 11 Medial view of right gonopods (♂). Arrow, Anterior gonopodal apex (podomere 6) shovel-shaped; in repose cupped sheath-like around flagelliform posterior gonopodal apex (podomere 6). Scale bar 0.05 mm. 12 Oblique (right) view of right posterior gonopodal apex (♂). 2 dorsal-most, longest articles laminate distally and recurved laterally, with denticulate posterior margins appearing claw-like. Scale bar 0.02 mm. 13 Antennomere 7 apex (♂). a Four apical cones (AS) oriented in a trapezoidal cluster on 7th antennomere, with longitudinally grooved outer surface and apical circular invagination b Spiniform basiconic sensilla (Bs3) in cluster of 5, oriented apical dorsally on 7th antennomere; tips facing apical cones (on longitudinal axis with Bs2 on antennomeres 5, 6); each sensillum with 2 barbules. Scale bar 0.02 mm.14 Lateral (right) view of right antenna (♂). a Chaetiform sensilla (CS) widely spaced on antennomeres 1-7, each sensillum with 2 or 3 barbules b Trichoid sensilla (TS) oriented apically encircling antennomeres 1–7, lacking barbules. Scale bar 0.1 mm. 15 Lateral (right) view of head, collum and segments 2, 3 (♂).Arrow, collum with carina present on anterolateral margin, appearing scaly. Carina repeated serially on lateral tergal and pleural margins (absent from telson). Scale bar 0.1 mm.
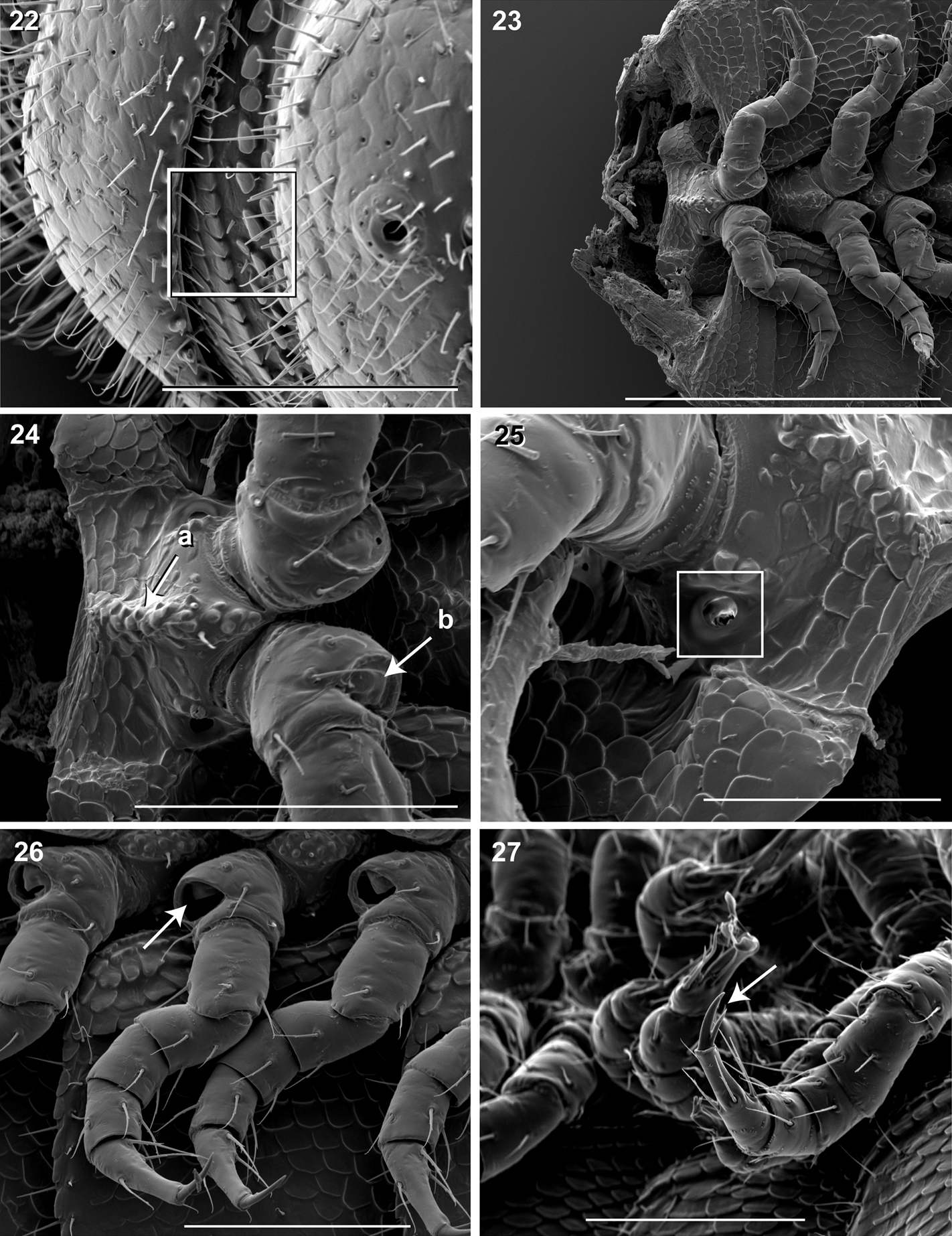
16 Ventral view of segments (♂). a Lateral tergal and pleural carinae jagged, pronounced on midbody segments b Pleurite medial margin broad, with scaly carina c Postgonopodal tarsus with thinner claw and without bifurcation, but with stout seta. Scale bar 0.4 mm. 17 Dorsal view of segments (♂). a Metazonite posterior margin (limbus) lined with posteriorly projecting anchor-shaped spikes and a row of conical spikes just dorsal to anchor-shaped spikes b 3 or 4 stout flat tubercles opposite ozopore near anterior margin, lunate arrangement encircling ozopore. Scale bar 0.4 mm. 18 Dorsal view of tergites (♂). Square, tergal setae tipped with silk-like exudate, tangled, appearing adhered to neighboring setae. Scale bar 0.05 mm. 19 Dorsal view of left ozopore (♂).Square, ozopores elevated slightly, with 2 stout posteriorly projecting spines and encircled by 13 – 15 robust setae. Scale bar 0.05 mm. 20 Right lateral view of posterior segments and telson (♂). Arrow, tergites, pleurites and sternites separated by arthrodial membrane. Scale bar 0.4 mm. 21 Oblique (right) ventrolateral view of 2 apodous segments, telson, hypoproct and paraprocts (♂).Apodous segments lacking sterna, pleurites contiguous in midline. Apodous tergites densely setose, covered with unevenly distributed spikes. Scale bar 0.2 mm.
22 Lateral view of fifth metatergite and prozonite (♂).Square, prozonite highly sculptured, with 5 rows of discoidal flat tubercles; anterior 3 rows staggered and posterior 2 rows aligned. Scale bar 0.1 mm. 23 Ventral view of mid-length sternites, pleurites and legs (♂).Left and right pleurites broadly overlapping sternite, covering spiracles. Scale bar 0.3 mm. 24 Ventral view of mid-length sternites and leg bases (♂). a Medial sternal ridge projecting ventrally, with spiracles and legs oriented ventrally b Eversible sacs membranous, bulging slightly from opening. Scale bar 0.1 mm. 25 Oblique (right) lateral view of sterna and spiracle (♂). Square, spiracles circular, orifice open; oriented dorsally above legs. Scale bar 0.05 mm. 26Ventral (right) view of legs, with posteroventral eversible sac opening (♂). Arrow, large posteroventral D-shaped opening for eversible sac. Scale bar 0.1 mm. 27 Oblique (right) lateral view of pregonopodal legs (♂).Arrow, pregonopodal tarsus with stout bifurcate claw. Scale bar 0.1 mm.
(♀) VMNH – Counts and measurements: p = 190. a = 2. l = 750. (190 + 2 + T). HW = 0.37. HL = 0.44. ISW = 0.30. AW = antennae missing. CW = 0.44. W1 = 0.58. W2 = 0.58. W3 = 0.57. L1 = 0.23. L2 = 0.21. L3 = 0.23. H1 = 0.46. H2 = 0.44. H3 = 0.48. AS1 = 0.44. BL = 40.40. Anatomical description similar to male holotype. In combination with its measurements, the following structures differ from male holotype. Head triangular, chevron-shaped, tapered anteriorly to round point at a 135° angle anterior from antennal sockets; occipital area posterior from antennal sockets straight, not curved medially towards neck. Cyphopods large, area 1/6 the segmental area in widest cross-section; almond-shaped, bivalvular, narrow apex oriented ventrolaterally. Valves transparent, glassy. Ventral valve thickened and clam-like, with 4 or 5 thick setae; dorsolateral valve thin and flat, with 2 or 3 spines. Oviduct connected posteriorly to cyphopod, opening oriented ventromedially and located between valves. Oviduct tube wrinkled, appearing highly expandable in width, cross-section 1/8 area of cyphopod. Receptacle, suture and operculum absent.
There is negligible variation in coloration among live specimens. (FSCA paratype specimens that have been stored in alcohol for 86 years are dark mahogany brown, which is likely an unnatural color and a result of alcohol preservative, vial stopper and age.) The predominant source of variation between specimens is segment and leg counts (Tables 1–3). Females have between 486-750 legs with a standard deviation of 78, and males between 318–562 legs with a standard deviation of 107. The segments of Illacme plenipes (males and females) are uniform in length, width and height along the trunk, and are slightly taller, and more convex, in posterior segments—potentially to accommodate the spiraled metenteron.
Segment and leg count, head measurements.
| p | l | HW | HL | ISW | AW | CW | |
|---|---|---|---|---|---|---|---|
| ♂ | 84–145 (107/27) | 318–562 (410/107) | 0.295–0.308 (0.301/0.006) | 0.344–0.406 (0.382/0.024) | 0.172–0.202 (0.189/0.011) | 0.098–0.103 (0.101/0.002) | 0.374–0.422 (0.393/0.019) |
| ♀ | 126–192 (159/20) | 486–750 (619/78) | 0.308–0.369 (0.335/0.020) | 0.408–0.556 (0.446/0.045) | 0.185–0.295 (0.217/0.033) | 0.098–0.113 (0.103/0.006) | 0.407–0.472 (0.431/0.021) |
Width and length measurements.
| W1 | W2 | W3 | WM | L1 | L2 | L3 | LM | |
|---|---|---|---|---|---|---|---|---|
| ♂ | 0.437–0.526 (0.485/0.033) | 0.467–0.554 (0.500/0.036) | 0.455–0.545 (0.488/0.034) | 0.491/ 0.032 | 0.148–0.203 (0.173/0.021) | 0.150–0.197 (0.162/0.020) | 0.140–0.183 (0.159/0.017) | 0.165/0.019 |
| ♀ | 0.520–0.620 (0.564/0.035) | 0.531–0.640 (0.569/0.037) | 0.517–0.610 (0.559–0.032) | 0.564/0.034 | 0.172–0.228 (0.195/0.018) | 0.176–0.209 (0.194/0.012) | 0.157–0.234 (0.194/0.021) | 0.194/0.017 |
Height, apodous segment/gonopodal width, body length measurements.
| H1 | H2 | H3 | HM | AS1 | A5W | P5W | BL | |
|---|---|---|---|---|---|---|---|---|
| ♂ | 0.273–0.400 (0.350/0.057) | 0.277–0.418 (0.337/0.055) | 0.295–0.381 (0.336/0.036) | 0.341/0.047 | 0.394–0.445 (0.423/0.022) | 0.047–0.055 (0.051/0.003) | 0.036–0.043 (0.040/0.003) | 13.368–28.156 (19.251/6.305) |
| ♀ | 0.220–0.486 (0.365/0.077) | 0.289–0.488 (0.384/0.064) | 0.295–0.504 (0.370/0.079) | 0.373/0.071 | 0.412–0.482 (0.451/0.024) | - | - | 24.541–40.399 (31.055/5.474) |
Illacme plenipes specimens were collected during the day in a small valley adjacent to cattle pasture. The woodland habitat was primarily composed of California live-oak, Quercus agrifolia (Fig. 28). Understory flora included ferns (bracken, Pteridium aquilinum; California polypody, Polypodium californicum; and California maiden-hair, Adiantum jordanii), California blackberry (Rubus ursinus), and poison oak (Toxicodendron diversilobum) (Fig. 29). Specimens were found beneath large moss-covered boulders, typically with a mass > 30 kg (Fig. 30). The millipede Tylobolus uncigerus (Wood, 1864) (order Spirobolida) was found co-occurring with Illacme plenipes at this locality. Other arthropods encountered include: Aptostichus and Calisoga trapdoor spiders (Mygalomorphae), Evalljapyx (Diplura), and Promecognathus ground beetles (Carabidae). Edaphic setting: Specimens collected in 2007 were found beneath a large stone (Fig 30, about 30 kg). When the stone was removed, individuals were seen corkscrewing outward into the cavity from the soil (Fig. 31). The soil, consisting of moist small-grained substrate, was dark chocolate brown in coloration and somewhat sandy (Fig. 31). The soil did not contain clay particles and seemed to drain water quickly. During the 16 December 2007 collections, soil moisture extended 15 cm below the surface.
Habitat of Illacme plenipes. Top left, view of oak forest where Illacme plenipes were encountered. Top right, close up of oak forest and sandstone pinnacle where Illacme plenipes occur. Bottom, landscape view of oak forest, cattle trails evident (composite stitched image of three photos, image sides slightly distorted).
Oak forest understory habitat of Illacme plenipes. Top, base of sandstone pinnacle (from Fig. 28), where specimens were found. Bottom, mossy oak forest—close-up of habitat where Illacme plenipes individuals were encountered.
Sandstone microhabitat of Illacme plenipes.Top left, 50 kg sandstone from 29.xi.2005 rediscovery locality of Illacme plenipes; one ♀ with 666 legs was discovered from beneath the stone (scale bar = 5 cm, hand shovel shown for scale). Bottom left, 30 kg sandstone from the 16.xii.2007 locality, two ♀ (specimen #s: SPC001187, MIL0020) were discovered below the stone (scale bar = 5 cm, 15 cm ruler shown for scale). Top right, surface close up of sandstone from 16.xii.2007 locality with ♂ Illacme plenipes, not collected (scale bar = 5 mm). Bottom right, surface close up of sandstone from 29.xi.2005 locality with ♂ Illacme plenipes (specimen #: SPC000924, scale bar = 5 mm). Millipedes shown in right two pictures were found clinging to the surface of the stone.
Subterranean soil microhabitat of Illacme plenipes. Left, sandstone crater; dotted line indicates crater’s edges, arrows indicate two ♀ Illacme plenipes shown in situ upon removal of stone (specimen #s: SPC001187, MIL0020—lower individual with anterior trunk segments embedded in soil, upper individual with middle segments embedded in soil). Bottom middle, close up of lower individual from left image. Top right, dark sandy soil from microhabitat. Bottom right, close up of soil showing sandy grain structure.
Illacme plenipes is only known from a small area, ca. 4.5 km in diameter, in the northwestern foothills of the Gabilan Range in San Benito County, California.
The pattern by which Illacme plenipes add segments and subsequently legs post-embryonically between developmental stadia is referred to as anamorphosis (
The diet of Illacme plenipes is unknown. Given the shape of its mouthparts, the typical millipede diet in which decaying organic matter is mechanically fragmented is unlikely for the species. Illacme plenipes possesses a comb-like structure on the posterior margin of the labrum and an open triangular tooth-lined “mouth” formed by an orifice though the labrum (Fig. 7a, b; Fig. 8a, b; Mb-805580; Mb-805588). The mouthparts are composed of the stylet-like mandibles and the gnathochilarium (structures observed between 500-2000× with a scanning electron microscope and the mandibles through the translucent head capsule at 400× with a compound microscope). These mouthparts are tightly appressed and tapered anteriorly to a rounded point. Given that the mandibles appear stylet-like, and assuming the mouthparts are moveable, a functional hypothesis for feeding is that the gnathochilarium hinges open, the mandibles are protruded to pierce plant and/or fungal tissue, and then the tooth-lined mouth is used to suck out the fluid contents. The teeth and labral comb could serve to filter particulates exceeding a certain size. Other Colobognath millipedes with somewhat reduced mouthparts, for example species of the family Andrognathidae, feed on fungus or other live plant or soft organic matter (
Based on natural history observations of Illacme plenipes in the field, individuals are always found approximately 10 – 15 cm beneath the soil, or clinging to the surface of large sandstones. The great number of legs may benefit a deep subterranean lifestyle clinging to sandstone. Illacme plenipes has bifurcate claws on anterior legs and two separate claws, coterminal on the tarsal apex (in lieu of the abifurcation), on posterior legs. In several millipede species, e.g. Cylindroiulus fimbriatus Enghoff, 1982 and Dolistenus savii Fanzago, 1874, the additional claws serve a stone-clinging function for surface adherence and an epilithic lifestyle (
In contrast with the smooth exoskeleton of the bulldozer millipedes, Illacme plenipes’ has a multiplicity of projections and cuticular ornaments including anchor-shaped spikes, discoidal tubercles, long silk-secreting setae and jagged body plates. Several of these projections (e.g., the peculiar anchor-shaped spikes — Fig. 17a, Mb-805601) have been documented in other taxa in the Siphonophorida and Julida (
The widely scattered distribution of modern Siphonorhinidae, predominately in the Southern Hemisphere except with Illacme plenipes in North America, indicates that their most recent common ancestor likely predates the breakup of Pangaea more than 200 million years ago. A phylogeny for Siphonorhinidae, or any taxa in the four orders of Colobognatha, does not exist, except for a recent species phylogeny of the genus Brachycybe in the order Platydesmida (
The paleoendemic species Illacme plenipes is the sole representative of the family in the Western Hemisphere. Remaining genera in the family occur primarily in the Old World tropics in Wallacea, Sundaland, Himalayas (Siphonorhinus species), Indo-Burma (Kleruchus olivaceus and Siphonorhinus species), and Maputaland-Pondoland-Albany (Nematozonium filum). The closest relative of Illacme plenipes is uncertain. The present day range of Siphonorhinidae may be the remnant of an ancient and widespread tropical distribution across Pangaea. The most likely sister taxon to Illacme plenipes is Nematozonium filum from South Africa, as they share a number of anatomical similarities. Among the known species of Siphonorhinidae, a South African species is a probable candidate for closest relative based on other close relationships between co-distributed taxa, for example the flightless Californian beetle genus Promecognathus and its close relatives in the tribe Axinidiini in South Africa (
The influence of the marine layer and thick inland fog, which creates a unique climate for the area, may have contributed to a stable environment for Illacme plenipes. Areas with high probability of occurrence (Fig. 1) also receive a frequent layer of fog (
Illacme plenipes is threatened by extinction as a result of its restricted geographical distribution, narrow microhabitat requirements, seasonal rarity, and low observed population numbers. Natural populations are threatened by habitat loss due to rampant development and intense land use in the area (agricultural, industrial, transit and housing), climate change, invasive species, and potential for over-collecting. The restricted location of Illacme plenipes, limited to the gap between the Santa Cruz Mountains and Gabilan Range at the eastern fog limit, may be due to edaphic requirements (soils composed of sandstone or other native formations in the area: San Lorenzo Formation or Dacitic volcanic rocks), or extirpation due to the heavy agricultural influence around Monterey Basin and the Salinas Valley since the 1800s. In contrast with habitat degradation from development and farming, the presence of cattle does not appear to negatively affect Illacme plenipes. At each locality where Illacme plenipes was discovered, there was noticeable influence of cattle on the habitat. Boulders under which Illacme plenipes occurred were sometimes a meter away from deep cattle hoof prints. The most serious impacts that Illacme plenipes faces are human-induced habitat loss and climate change. As suggested by the distribution model and Illacme plenipes’ apparent dependence on marine layer fog (likely influencing moisture and stability of its habitat), the documented 33% reduction in coastal California fog due to higher atmospheric and ocean temperature since the early 1900s (
(Published at www.morphbank.net ):
http://www.morphbank.net/?id=805574
http://www.morphbank.net/?id=805575
http://www.morphbank.net/?id=805576
http://www.morphbank.net/?id=805577
http://www.morphbank.net/?id=805578
http://www.morphbank.net/?id=805579
http://www.morphbank.net/?id=805580
http://www.morphbank.net/?id=805582
http://www.morphbank.net/?id=805583
http://www.morphbank.net/?id=805584
http://www.morphbank.net/?id=805585
http://www.morphbank.net/?id=805586
http://www.morphbank.net/?id=805587
http://www.morphbank.net/?id=805588
http://www.morphbank.net/?id=805589
http://www.morphbank.net/?id=805590
http://www.morphbank.net/?id=805591
http://www.morphbank.net/?id=805592
http://www.morphbank.net/?id=805593
http://www.morphbank.net/?id=805594
http://www.morphbank.net/?id=805595
http://www.morphbank.net/?id=805596
http://www.morphbank.net/?id=805597
http://www.morphbank.net/?id=805598
http://www.morphbank.net/?id=805599
http://www.morphbank.net/?id=805600
http://www.morphbank.net/?id=805601
http://www.morphbank.net/?id=805602
http://www.morphbank.net/?id=805603
http://www.morphbank.net/?id=805604
http://www.morphbank.net/?id=805605
http://www.morphbank.net/?id=805606
http://www.morphbank.net/?id=805609
http://www.morphbank.net/?id=805610
http://www.morphbank.net/?id=805612
http://www.morphbank.net/?id=805614
http://www.morphbank.net/?id=805615
http://www.morphbank.net/?id=805616
http://www.morphbank.net/?id=805618
http://www.morphbank.net/?id=805620
http://www.morphbank.net/?id=805621
http://www.morphbank.net/?id=805623
Thanks to Rob Marek for assistance in the field to collect Illacme plenipes. Peter Raven recommended Illacme plenipes localities in San Benito County and shared helpful knowledge of the area and co-occurring native plants. Avery Lane, David Beamer, Amy Stockman and Matthew Walker assisted in the field and laboratory. Many thanks to Patrick, Peter and Tom Breen, the Reeves Family, and Bart O’Brien who shared information and details about California mountain ranges and potential localities. The Nature Conservancy kindly allowed access to sites in the Gabilan Range. Dotti Marek and Katy Murphy provided local support during fieldtrips to California. Thanks to anonymous reviewers and Charity Hall for reading earlier versions of the manuscript. California State Parks and the National Park Service supported research in the parks and permits for collections. Jonathan Coddington, Dana DeRoche, G.B. Edwards, Charles Whitehill, Judith Winston, and Joe Keiper provided essential type specimens and access to natural history collections in support of the project. This research was supported by a U.S. National Science Foundation Partnerships for Enhancing Expertise in Taxonomy Grant to P. Sierwald, J.E.B., and W.A.S. (DEB-0529715); and by a NSF Phylogenetic Systematics grant to P.E.M. (DEB-1119179). Wendy Moore and the Entomology Department at the University of Arizona is acknowledged for their support of P.E.M. and systematic entomology. This article is in memoriam of Richard Hoffman (1927 – 2012), whose support for the first author’s study of millipedes will always be appreciated. Dr. Hoffman loaned the Virginia Museum of Natural History’s Illacme plenipes specimens shortly before he passed away. His inquisitive naturalist spirit and scientific legacy lives on in the 50+ organisms named in his honor, numerous scientific contributions, and love of the natural history of Virginia.
Movie of ♀ Illacme plenipes (specimen # SPC000931) with 662 legs showing live movement and head shape. Individual filmed in a glass petri dish with a Nikon Coolpix 995 digital camera mounted to a Leica 12.5 stereomicroscope. (doi: 10.3897/zookeys.241.3831.app1). File format: Apple QuickTime Movie (MOV).
Movie of ♀ Illacme plenipes (specimen # SPC000930) with 666 legs showing very slow, nearly imperceptible locomotion. Individual filmed on an oak leaf with a Nikon Coolpix 995 digital camera. (doi: 10.3897/zookeys.241.3831.app2). File format: Apple QuickTime Movie (MOV).
Movie of ♀ Illacme plenipes (specimen # SPC000930) with 666 legs showing very slow, nearly imperceptible locomotion. Individual filmed on a cardboard sheet with the same method described in Appendix II. (doi: 10.3897/zookeys.241.3831.app3). File format: Apple QuickTime Movie (MOV).
Movie of ♀ Illacme plenipes (specimen # SPC000931) with 662 legs showing live motion and rapid, independent antennal movement. The species is blind and presumably relies on the antennae to sense its environment. Individual filmed in a glass petri dish with the same method described in Appendix I. (doi: 10.3897/zookeys.241.3831.app4). File format: Apple QuickTime Movie (MOV).
Times lapse series of visible satellite images of Monterey Bay, California, showing the occurrence of fog extending into the Monterey Basin and Salinas Valley. (doi: 10.3897/zookeys.241.3831.app5). File format: Apple QuickTime Movie (MOV).
Explanation note: Times lapse series of 330 visible satellite images of Monterey Bay, California, recorded every 15 mins by the GOES-15, Geostationary Operational Environmental Satellite (U.S. National Environmental Satellite, Data, and Information Service) from 10-18 September 2012. Contour lines = 61 m (200 ft). Images provided by the U.S. Naval Research Laboratory, Monterey, California http://www.nrlmry.navy.mil/NEXSAT.html
Images of ♀ Illacme plenipes (specimen # MIL0020) with 618 legs. Individual photographed with a Nikon D40 dSLR and a 60 mm 1:2.8 AF-S macro lens. (doi: 10.3897/zookeys.241.3831.app6). File format: JPEG Interchange Format (JPG).