(C) 2012 Jing Chen. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
The aphid genus Neonipponaphis Takahashi is reviewed and reported in China for the first time, with a description of one new species, Neonipponaphis pustulosis sp. n. on Castanopsis eyrei from Fujian. A key to species, morphological descriptions, features, host plants, and distributions are provided. Holotype and paratypes are deposited in the National Zoological Museum of China, Institute of Zoology, Chinese Academy of Sciences, Beijing, China.
Neonipponaphis, Aphididae, new record genus, new species, China
The aphid genus Neonipponaphis was erected by
Specimens of the new species were collected from Mount Wuyi (Wuyishan City) by J. Chen, Q. H. Liu, and X. T. Li.
Aphid terminology in this paper generally follows
In Table 1, the following abbreviations have been used: Ant.I, Ant.II, and Ant.IIIb, for antennal segments I, II, and the base of antennal segment III, respectively; PT, processus terminalis; Ant.IIIBW, basal width of antennal segment III; URS, ultimate rostral segment; BW URS, basal width of ultimate rostral segment; 2HT, second hind tarsal segment; MW Hind tibia, mid-width of hind tibia; BW Cauda, basal width of cauda; AP, anal plate; GP, genital plate.
Morphometric data of species of Neonipponaphis (in mm).
| Parts (For abbreviations see Materials and methods) |
Neonipponaphis pustulosis sp. n. | Neonipponaphis shiiae Takahashi | |||||
|---|---|---|---|---|---|---|---|
| Apterous vivipara (n=14) | Apterous vivipara (n=10) | ||||||
| Mean | Range | Standard Deviation | Mean | Range | Standard Deviation | ||
| Length (mm) | Body length | 1.532 | 1.406–1.628 | 0.062 | 1.169 | 1.114–1.238 | 0.049 |
| Body width | 1.388 | 1.184–1.628 | 0.126 | 1.058 | 0.960–1.171 | 0.074 | |
| Whole antenna | 0.242 | 0.221–0.259 | 0.011 | 0.211 | 0.187–0.221 | 0.010 | |
| URS | 0.078 | 0.072–0.086 | 0.005 | 0.071 | 0.067–0.077 | 0.004 | |
| Hind trochanter and femur | 0.122 | 0.115–0.134 | 0.009 | 0.092 | 0.077–0.096 | 0.007 | |
| Hind tibia | 0.160 | 0.144–0.173 | 0.013 | 0.112 | 0.106–0.115 | 0.006 | |
| 2HT | 0.057 | 0.053–0.062 | 0.004 | 0.044 | 0.038–0.048 | 0.003 | |
| Cauda | 0.028 | 0.024–0.034 | 0.003 | 0.028 | 0.024–0.029 | 0.002 | |
| BW Cauda | 0.053 | 0.048–0.058 | 0.004 | 0.055 | 0.053–0.058 | 0.002 | |
| Ant.IIIBW | 0.022 | 0.019–0.026 | 0.002 | 0.019 | 0.017–0.022 | 0.001 | |
| MW Hind tibia | 0.034 | 0.029–0.036 | 0.003 | 0.027 | 0.026–0.029 | 0.001 | |
| Cephalic setae | 0.047 | 0.038–0.058 | 0.009 | 0.064 | 0.053–0.074 | 0.008 | |
| Setae on Tergum I | 0.068 | 0.060–0.079 | 0.008 | 0.073 | 0.058–0.091 | 0.010 | |
| Setae on Tergum VIII | 0.055 | 0.046–0.077 | 0.010 | 0.037 | 0.034–0.043 | 0.004 | |
| Setae on Hind tibia | 0.030 | 0.026–0.036 | 0.004 | 0.028 | 0.024–0.034 | 0.004 | |
| No. of setae on | Ant.I | 1 | 1 | ||||
| Ant.II | 2 | 2 | |||||
| Ant.IIIb | 0 | 0 | |||||
| PT | 0+3 | 0+3 | |||||
| URS | 6 | 6 | |||||
| Terga II–VII | 17–27 | 14–20 | |||||
| Tergum VIII | 6–8 | 4–6 | |||||
| Cauda | 7–10 | 8–10 | |||||
| Each lobe of AP | 4–6 | 4–6 | |||||
| GP | 14–18 | 14–19 | |||||
| Ratio (times) | Whole antenna / Body | 0.16 | 0.14–0.17 | 0.008 | 0.18 | 0.17–0.19 | 0.008 |
| Hind tibia / Body | 0.11 | 0.10–0.11 | 0.007 | 0.10 | 0.09–0.10 | 0.009 | |
| URS / BW URS | 1.63 | 1.43–1.78 | 0.173 | 1.24 | 1.17–1.45 | 0.102 | |
| URS / 2HT | 1.43 | 1.23–1.64 | 0.203 | 1.57 | 1.47–1.67 | 0.066 | |
| Cauda / BW Cauda | 0.54 | 0.48–0.64 | 0.058 | 0.50 | 0.45–0.55 | 0.040 | |
| Cephalic setae / Ant.IIIBW | 2.21 | 1.60–2.88 | 0.547 | 3.31 | 2.67–3.88 | 0.460 | |
| Setae on Tergum I / Ant.IIIBW | 3.00 | 2.27–3.75 | 0.676 | 3.77 | 3.00–4.75 | 0.572 | |
| Setae on Tergum VIII / Ant.IIIBW | 2.49 | 1.91–3.20 | 0.416 | 1.89 | 1.56–2.25 | 0.253 | |
| Setae on Hind tibia / MW Hind tibia | 0.88 | 0.79–1.00 | 0.102 | 1.00 | 0.91–1.17 | 0.120 | |
Specimen depositories: all specimens studied are deposited in the National Zoological Museum of China, Institute of Zoology, Chinese Academy of Sciences, Beijing, China (NZMCAS).
Taxonomyhttp://species-id.net/wiki/Neonipponaphis
In apterae, body round, flat, and strongly sclerotized. Prosoma consisting of fused head, thorax, and abdominal segment I; abdominal segments II–VII fused and distinctly separated from prosoma; abdominal segment VIII free. Dorsum of prosoma with scattered oval or irregular-shaped pustules and numerous fine setae; abdominal tergites II–VII with scattered shorter setae; each tergite with a pair of submarginal setae, setae on tergites V and VI shorter than setae on the other tergites; tergites II and VII each with a pair of spinal setae; abdominal tergite VIII with 4–8 setae. Eyes with 3 facets in apterae and compound in alatae. Antennae in apterae indistinctly 3-segmented, with primary rhinaria placed wide apart on the terminal segment, in alatae 5-segmented with annular secondary rhinaria. Rostrum short and thick. Ultimate rostral segment blunt wedge-shaped, with 2 pairs of primary setae and a pair of secondary setae. Legs normal, tibial setae long and fine, hind tibiae with several short peg-like setae on distal part; tarsi 2-segmented, claws normal, first tarsal chaetotaxy in apterae: 2, 2, 2. Abdomen with many long dorsal setae and 4 pairs of spiracles in alatae. Siphunculi in apterae small, pore-like, in alatae low but much expanded basally, with distinct minute papillae around the pore. Cauda knobbed and constricted at base. Anal plate bilobed. Wings dusky and reticulated; fore wings with pterostigma dark and broadly rounded at hind margin, media once branched; hind wings with 2 obliques.
Japan and here newly recorded from China (Fujian).
Castanopsis cuspidata and Castanopsis eyrei.
This genus is related to Nipponaphis Pergande, sharing several characters such as body of apterae aleyrodiform, flattened dorsoventrally, consisting of three parts - prosoma, fused abdominal segments II–VII, and separate abdominal segment VIII; dorsum of prosoma with scattered pustules; abdominal tergites II–VII with 6 pairs of submarginal setae and a pair of posteromesial setae on abdominal tergite VII; siphunculi pore-like; tarsi normal, 2-segmented, with normal claws; abdomen of alatae with 4 pairs of spiracles, and median vein of fore wings once branched. Neonipponaphis is distinguished by abdominal tergites II–VII distinctly separated from prosoma and the presence of numerous fine setae on the dorsum of prosoma and abdominal tergites II-VII in apterae.
(Apterous viviparous females)
| 1 | Body large, 1.41–1.63 mm long, with longer hind legs, the hind tibiae being 0.10–0.11 times as long as body. Pustules on prosoma small. Spinal and submarginal setae on dorsum of prosoma distinctly longer, thicker, and stiffer than the scattered dorsal setae. Abdominal tergite VIII with 6–8 dorsal setae | Neonipponaphis pustulosis sp. n. |
| – | Body relatively small, 1.11–1.24 mm long, with shorter hind legs, the hind tibiae being 0.09–0.10 times as long as body. Pustules on prosoma large. Spinal and submarginal setae on dorsum of prosoma longer than the scattered dorsal setae but similar to the latter in thickness and hardness. Abdominal tergite VIII with 4–6 dorsal setae | Neonipponaphis shiiae Takahashi |
urn:lsid:zoobank.org:act:ED5004C9-48A1-4503-B6C3-60B8746CFC6C
http://species-id.net/wiki/Neonipponaphis_pustulosis
Figures 1–19China (Fujian, 27.73279°N, 117.64512°E, altitude 1080 m).
The new species is named for the small and crowded pustules on the dorsum of prosoma. “Pustulosis” (Latin) means “blister, bubble”.
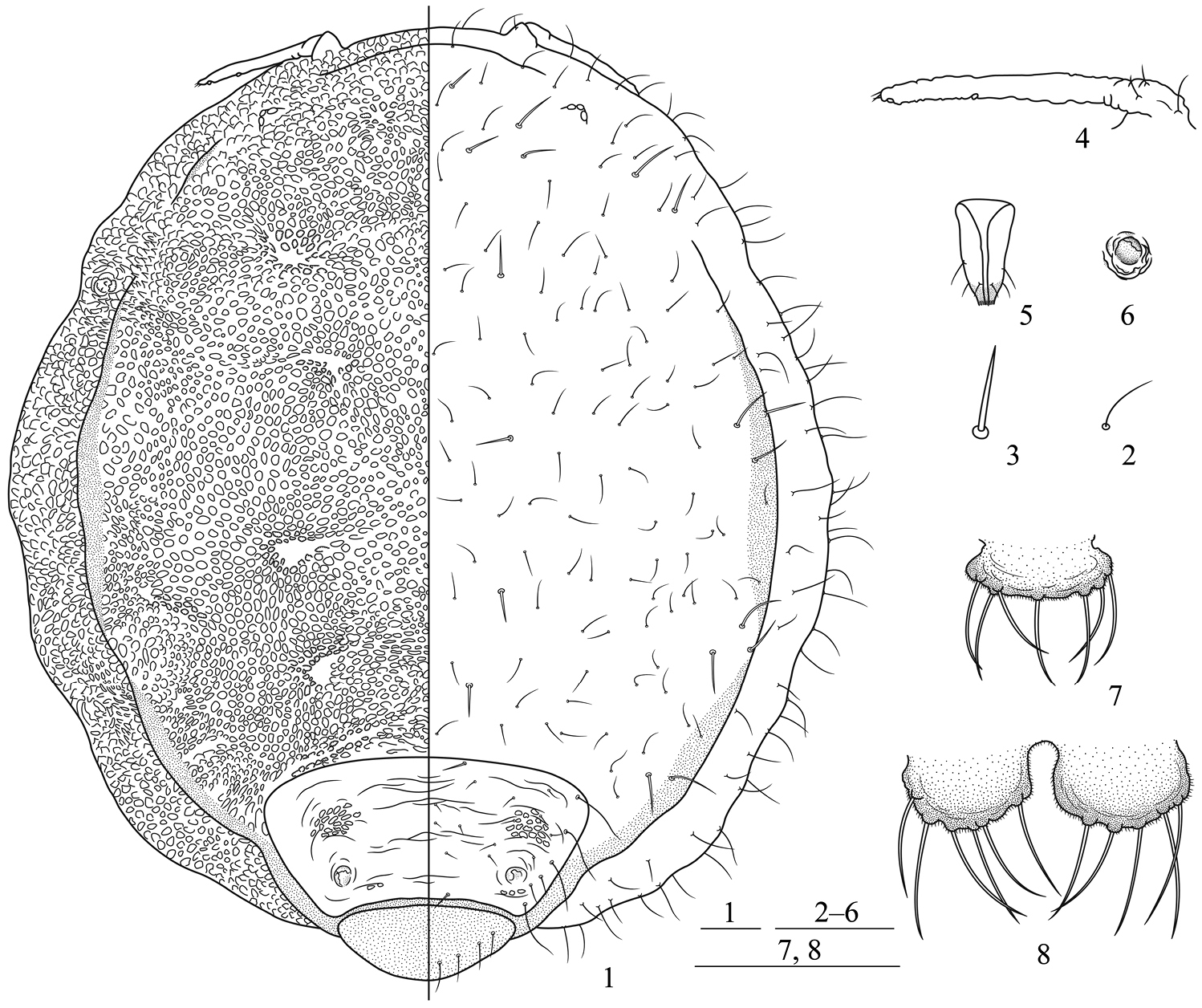
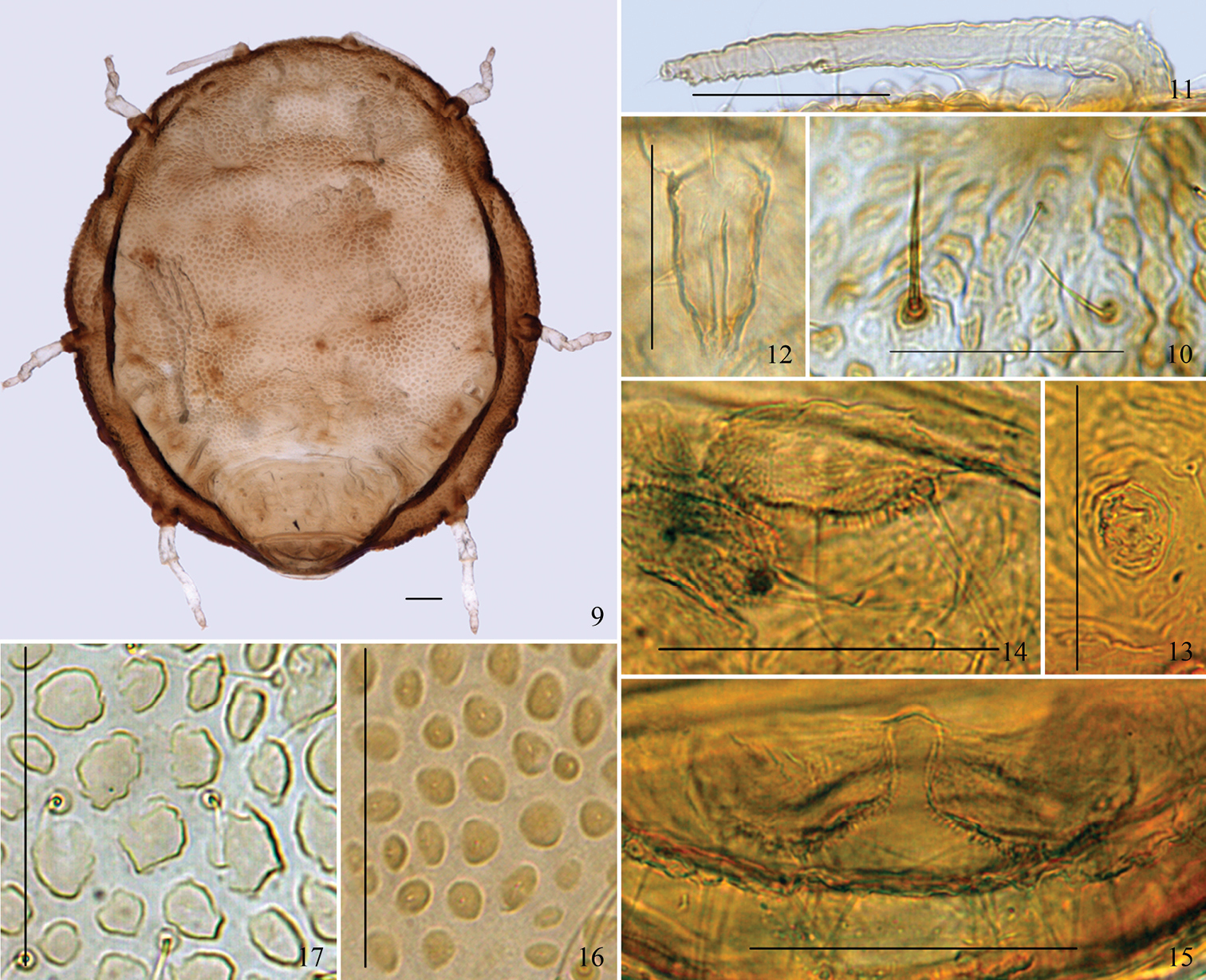
Apterous viviparous females: Body round, flat, thickened, and strongly sclerotized (Figs 1, 9, 19). Reddish brown or blackish brown in life (Figs 18, 19). For morphometric data see Table 1.
Body brown; antennae and legs light brown. Prosoma consisting of fused head, thorax, and abdominal segment I; abdominal segments II–VII fused and distinctly separated from prosoma; abdominal segment VIII free (Figs 1, 9). Dorsum of prosoma with many oval or irregular-shaped pustules, small and crowded (Figs 1, 9, 16); pustules on vertical area of body similar, but those around the thoracic spiracles much smaller, protuberant, and conical in shape. Muscle attachment plates distinct, forming radial pattern with dorsal pustules (Figs 1, 9). Abdominal tergites II–VII wrinkled and with irregular oval markings (Fig. 1). Cauda (Figs 7, 14), anal plate (Figs 8, 15), and genital plate with spinulose sculptures. Dorsum of prosoma and marginal vertical area of body with numerous fine and pointed setae; head with a pair of cephalic setae, thick, stiff, and pointed; dorsum of prosoma with 13 pairs of submarginal setae, long, thick, and stiff, head dorsum with 3 pairs, pronotum with 2 pairs, mesonotum with 3 pairs, metanotum with 3 pairs, abdominal tergite I with 2 pairs; pro-, meso-, metanotum, and abdominal tergite I each with a pair of spinal setae, long, thick, and stiff; abdominal tergites II–VII with 17–27 scattered fine and pointed setae, shorter than dorsal setae on prosoma; tergites II–VII each with a pair of long submarginal setae, setae on tergites V and VI shorter; tergites II and VII each with a pair of spinal setae, stiff and pointed; tergite VIII with 6–8 dorsal setae (Fig. 1). Cephalic setae, marginal setae on abdominal tergite I, and dorsal setae on tergite VIII 1.60–2.88 times, 2.27–3.75 times, and 1.91–3.20 times as long as basal width of antennal segment III, respectively. Medial frons not protuberant (Figs 1, 9). Eyes 3-faceted (Fig. 1). Antennae short, indistinctly 3-segmented, 0.14–0.17 times as long as body (Figs 4, 11). Setae on antennae sparse; segments I–III each with 1, 2, 0+0 setae, respectively; processus terminalis with 3 apical setae. Primary rhinaria small, round, protuberant, and placed wide apart at the apex of terminal segment. Rostrum short and thick, not reading mid-coxae. Ultimate rostral segment blunt wedge-shaped, 1.43–1.78 times as long as its basal width, 1.23–1.64 times as long as second hind tarsal segment, with 2 pairs of primary setae and a pair of secondary setae (Figs 5, 12). Legs short, smooth, trochanter and femur fused (Fig. 9). Hind tibia 0.10–0.11 times as long as body. Setae on legs sparse, tibiae setae long and fine, hind tibiae with several short peg-like setae on distal part. Setae on hind tibia 0.79–1.00 times as long as its mid-width. First tarsal chaetotaxy: 2, 2, 2. Claws normal. Siphunculi small, pore-like, on abdominal tergite VI (Figs 6, 13). Cauda knobbed, constricted at base, 0.48–0.64 times as long as its basal width, with 7–10 setae (Figs 7, 14). Anal plate bilobed, each lobe with 4–6 setae (Figs 8, 15). Genital plate transversely oval, with two anterior setae and 12–16 setae along the posterior margin.
Neonipponaphis pustulosis sp. n. Apterous viviparous female: 1 dorsal view of body, with pustules in left and chaetotaxy in right 2 fine and pointed scattered dorsal seta 3 long, thick, and stiff dorsal seta 4 antenna 5 ultimate rostral segment 6 siphunculus 7 cauda 8 anal plate. Scale bars = 0.10 mm.
(9–15) Neonipponaphis pustulosis sp. n. Apterous viviparous female: 9 dorsal view of body 10 dorsal setae (long, thick, and stiff seta in left, fine and pointed seta in right) 11 antenna 12 ultimate rostral segment 13 siphunculus 14 cauda 15 anal plate. (16–17) Dorsal pustules on the same scale: 16 Neonipponaphis pustulosis sp. n. 17 Neonipponaphis shiiae Takahashi. Scale bars = 0.10 mm.
Holotype: apterous viviparous female, CHINA: Fujian (Wuyishan City, Xingcun Town, Mount Wuyi, 27.73279°N, 117.64512°E, altitude 1080 m), 11 Jun. 2011, No. 26868-1-3, on Castanopsis eyrei, coll. J. Chen, Q. H. Liu, and X. T. Li (NZMCAS). Paratypes: 13 apterous viviparous females, with the same collection data as holotype.
The new species is similar to the type species Neonipponaphis shiiae Takahashi, but differs in morphology by the characters given in the key.
Castanopsis eyrei.
Apterous exules live on the twigs of the host plants and are attended by ants (Figs 18, 19). Other morphs and life cycle are unknown. Typical life cycle of nipponaphidines is host-alternating and holocyclic, with gall formation on Distylium. Thus, this species is either anholocyclic on Castanopsis eyrei or has gall-inhabiting generations still unknown or known under another name on Distylium. Field observations, transfer experiments, and molecular study are needed to elucidate its life cycle.
Neonipponaphis pustulosis sp. n. 18 a colony on the twig of Castanopsis eyrei, attended by an ant 19 apterous viviparous females in life.
http://species-id.net/wiki/Neonipponaphis_shiiae
Figure 2010 apterous viviparous females, JAPAN: Gifu Prefecture, 20 Jul. 1968, No. E534, on Castanopsis sp., coll. M. Sorin (NZMCAS).
Japan.
Castanopsis cuspidata.
We are grateful to Prof. M. Sorin for presenting specimens of Neonipponaphis shiiae Takahashi. We also thank Q. H. Liu and X. T. Li for collections and F. D. Yang for making slides. The work was supported by the National Natural Sciences Foundation of China (Nos. 30830017, 31093430), National Science Funds for Distinguished Young Scientists (No. 31025024), National Science Fund for Fostering Talents in Basic Research (No. J0930004), and CAS Innovation Program (No. KSCX2-EW-Z-8) to G. X. Qiao.