(C) 2014 Shingo Hosoishi. This is an open access article distributed under the terms of the Creative Commons Attribution License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Hosoishi S, Ogata K (2014) Description and DNA barcoding of Crematogaster fraxatrix Forel, 1911 and two new closely related species from Cambodia and Indonesia (Hymenoptera, Formicidae). ZooKeys 374: 57–68. doi: 10.3897/zookeys.374.5874
Crematogaster fraxatrix Forel, 1911 and two new species, C. chhangi sp. n. and C. simboloni sp. n., are described from Cambodia and Indonesia, respectively. DNA sequences were generated for C. fraxarix and the two newly described species using 3 amplications of two regions of the mitochondrial gene COI with a total of 1129 bp. The mean interspecific divergences are 9.4% and 23.5% for C. fraxatrix vs. C. chhangi, C. simboloni, respectively. DNA sequences reveal that C. simboloni is found to be genetically distinct from the other two species, but C. chhangi is not distinct from C. fraxatrix.
Crematogaster fraxatrix, taxonomy, new species, lectotype designation, DNA barcoding, cytochrome c oxidase I
Crematogaster fraxatrix was described by
Specimens were examined and/or deposited in the collections listed below. Codes for public institutions mainly follow those in
BMNH The Natural History Museum, London, U.K.
CASC California Academy of Sciences, San Francisco, CA, USA.
FRIM Forest Research Institute Malaysia, Kepong, 52109 Kuala Lumpur, Malaysia.
KUM Kyushu University, Fukuoka, Japan.
MCZC Museum of Comparative Zoology, Harvard University, Cambridge, MA, USA.
MHNG Musee d’Histoire Naturelle, Geneva, Switzerland.
MZB Museum Zoologicum Bogoriense, Cibinong, Java, Indonesia.
NHMB Naturhistorisches Museum, Basel, Switzerland.
THNHM Thailand Natural History Museum, Technopolis, Khlong Luang, Pathum Thani, Thailand.
Most observations were made using an Olympus SZX12 microscope. Images were taken using a Canon EOS 50D with a Canon MP-E 65 mm 1-5 × macro lens, then processed using Combine ZM. Measurements were made with an Olympus SZX12 stereomicroscope using micrometers. All measurements are expressed in millimeters, recorded to the second decimal place. The measurements for petiole and postpetiole follow
Head Width (HW): Maximum width of head in full-face view, excluding the eyes.
Head Length (HL): Perpendicular distance from vertex margin to line tangent anteriormost projections of clypeus in full-face view.
Cephalic Index (CI): HW/HL × 100.
Scape Length (SL): Length of the first antennal segment, excluding the neck and basal condyle.
Scape Index (SI): SL/HW × 100.
Eye Length (EL): Maximum length of the compound eye.
Pronotal Width (PW): Maximum width of the pronotum in dorsal view.
Weber’s Length of the mesosoma (WL): Diagonal length, measured in lateral view from the anterior margin of the pronotum (excluding the collar) to the posterior extremity of the propodeal lobe.
Propodeal Spine Length (PSL): measured from tip of propodeal spine to closest point on outer rim of propodeal spiracle.
Petiole Length (PtL): Length of the petiole in lateral view (see
Petiole Width (PtW): Maximum width of petiole in dorsal view.
Petiole Height (PtH): Height of the petiole in lateral view (see
Postpetiole Length (PpL): Length of the postpetiole in lateral view (see
Postpetiole Width (PpW): Maximum width of postpetiole in dorsal view, excluding the helcium.
Petiole Height Index (PtHI): PtH/PtL × 100.
Petiole Width Index (PtWI): PtW/PtL × 100.
Postpetiole Width Index (PpWI): PpW/PpL × 100.
Waist Index (WI): PpW/PtW × 100.
Genomic DNA was extracted from tissues rich in mitochondria (e.g. legs) using a DNeasy Blood & Tissue kit (Qiagen, Maryland, USA). A 497 bp region of the mitochondrial genome, including barcoding regions of the cytochrome oxidase I (COI) was amplified via the polymerase chain reaction (PCR) using the following combinations of the primers, “LepF1” 5’-ATTCAACCAATCATAAAGATATTGG-3’ and “C_ANTMR1D-RonIIdeg_R” 5’-GGRGGRTARAYAGTTCATCCWGTWCC-3’ (used only for PCR), and “MLepF1” 5’-GCTTTCCCACGAATAAATAATA-3’ and “LepR1” 5’-TAAACTTCTGGATGTCCAAAAAATCA-3’ (
PCR products were visualized on a 1% agarose E-Gel 96-well system (Invitrogen), and then purified with 1.0 µl of ExoSAP-IT (GE Healthcare Life Sciences). All products were sequenced in both directions (except for C_ANTMR1D-RonIIdeg_R) using BigDye Terminator v3.1 (Applied Biosystems) on an ABI 3100 Avant DNA Sequencer (Applied Biosystems) at the Faculty of Science, Kyushu University, Fukuoka. Some fragments were removed prior to alignment, due to low quality. After trimming, the 5’ DNA barcoding region sequenced in this study were 497bp, therefore these sequences were not strictly DNA barcodes. Using the three primer sets, non-overlapping fragments of 244, 253 and 632 bp were sequenced respectively. DNA sequence data for eight individuals of three Crematogaster species were thus generated, and deposited at DNA Data Base of Japan, DDBJ (with accession numbers shown in Table 1). All data were registered in the project called: “Crematogaster ants in Asia” (CREAA) on Barcode of Life Database (BOLD). Among the three species, Crematogaster chhangi was sequenced from one nest series (SH12-Cam-70) from Cambodia, and Crematogaster simboloni was successfully sequenced only from one Krakatau specimen. Contigs were assembled using Vector NTI Advance TM ver. 11 (Invitrogen Corp.) and subsequently aligned by eye. Genetic distances were estimated using the Kimura-2-parameter (
Specimen data and DDBJ accessions.
| Species | Voucher specimen | Locality | DDBJ accession numbers | |
|---|---|---|---|---|
| first half of COI | second half of COI | |||
| Crematogaster chhangi | KUMANT001 | Cambodia, Koh Kong | AB828274, AB828377 | AB828264 |
| Crematogaster fraxatrix | KUMANT002 | Malaysia, Peninsula | AB828275, AB828381 | AB828265 |
| Crematogaster fraxatrix | KUMANT003 | Malaysia, Borneo | AB828276, AB828382 | AB828266 |
| Crematogaster fraxatrix | KUMANT004 | Malaysia, Borneo | AB828277, AB828383 | AB828267 |
| Crematogaster fraxatrix | KUMANT005 | Malaysia, Borneo | AB828278, AB828384 | AB828268 |
| Crematogaster fraxatrix | KUMANT006 | S. Thailand | AB828279, AB828385 | AB828269 |
| Crematogaster fraxatrix | KUMANT007 | Malaysia, Peninsula | AB828280, AB828378 | AB828270 |
| Crematogaster simboloni | KUMANT008 | Indonesia, Krakatau | AB828281, AB828386 | AB828271 |
| Crematogaster osakensis | KUMANT009 | Japan | AB828282, AB828379 | AB828272 |
| Crematogaster modiglianii | KUMANT010 | Malaysia, Peninsula | AB828283, AB828380 | AB828273 |
Intraspecific variation for Crematogaster fraxatrix was 4.19% on average, with a range of 0.4–6.9%. Relatively large divergence in Crematogaster fraxatrix (6.2–6.9%) was recorded when comparing Peninsular to Bornean specimens. By contrast, the divergence within Peninsular (0.7–1.2%) or Bornean specimens (0.4–0.9%) was low. Interspecific sequence divergence was 17.1% on average, and ranged from 8.1–24.6% (Table 2). The lowest interspecific genetic distance occurred between specimens of Crematogaster chhangi and Bornean Crematogaster fraxatrix (8.1%).
Percent mitochondrial cytochrome c oxidase I (COI) sequence divergence among species of Crematogaster chhangi, Crematogaster fraxatrix and Crematogaster simboloni.
| Species | Crematogaster chhangi [Cambodia] | Crematogaster fraxatrix [M. Peninsula] | Crematogaster fraxatrix [Borneo] | Crematogaster fraxatrix [Borneo] | Crematogaster fraxatrix [Borneo] | Crematogaster fraxatrix [S. Thailand] | Crematogaster fraxatrix [M. Peninsula] |
|---|---|---|---|---|---|---|---|
| Crematogaster chhangi [Cambodia] | |||||||
| Crematogaster fraxatrix [M. Peninsula] | 0.105 | ||||||
| Crematogaster fraxatrix [Borneo] | 0.082 | 0.069 | |||||
| Crematogaster fraxatrix [Borneo] | 0.082 | 0.067 | 0.009 | ||||
| Crematogaster fraxatrix [Borneo] | 0.081 | 0.067 | 0.004 | 0.007 | |||
| Crematogaster fraxatrix [S. Thailand] | 0.108 | 0.007 | 0.065 | 0.063 | 0.063 | ||
| Crematogaster fraxatrix [M. Peninsula] | 0.107 | 0.013 | 0.063 | 0.062 | 0.062 | 0.008 | |
| Crematogaster simboloni [Krakatau] | 0.243 | 0.241 | 0.227 | 0.23 | 0.226 | 0.246 | 0.241 |
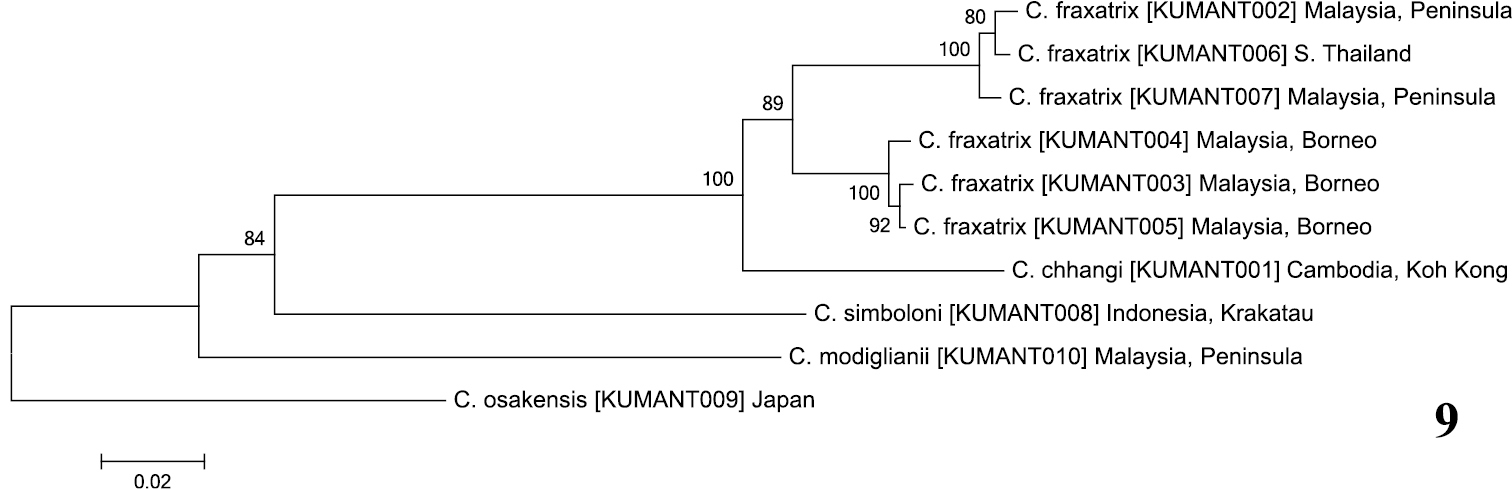
The neighbor-joining tree (Fig. 9) shows Crematogaster chhangi sister to Crematogaster fraxatrix, with high bootstrap support (100%). Among the three species examined, Crematogaster simboloni was distinctly separated from the Crematogaster chhangi and Crematogaster simboloni with higher genetic divergence: 24.3% to Crematogaster chhangi and 22.6% to 24.6% to Crematogaster fraxatrix. Crematogaster chhangi is distinguished from Crematogaster fraxatrix only in having an acutely developed subpetiolar process, whereas Crematogaster simboloni is quite different from Crematogaster chhangi and Crematogaster fraxatrix in having a densely sculptured promesonotum.
| 1 | Promesonotum sculptured | Crematogaster simboloni |
| – | Promesonotum not sculptured, but feebly striated with longitudinal rugulae | 2 |
| 2 | Propodeal spiracles strongly flattened dorsoventrally. Subpetiolar process acutely developed | Crematogaster chhangi |
| – | Propodeal spiracles oval in shape. Subpetiolar process weakly developed | Crematogaster fraxatrix |
http://zoobank.org/DDB2C024-BAE9-4A7B-9006-1C4BA647F716
http://species-id.net/wiki/Crematogaster_chhangi
Figs 1–2CAMBODIA: Koh Kong, 11°31'N, 103°09'E, 19.v.2012, S. Hosoishi. (SH12-Cam-70).
Holotype worker: pinned. Original label: CAMBODIA, Koh Kong, 11°31'N, 103°09'E, 19.v.2012, S. Hosoishi leg., SH12-Cam-70, arboreal; deposited at THNHM.
Eight paratype workers: pinned, same data as holotype; deposited at BMNH, CASC, FRIM, KUM, MCZC, MHNG, MZB, NHMB.
HW 0.64–0.83; HL 0.64–0.75; CI 100–111; SL 0.57–0.61; SI 73–86; EL 0.13–0.16; PW 0.37–0.42; WL 0.7–0.78; PSL 0.13–0.18; PtL 0.17–0.21; PtW 0.16–0.19; PtH 0.13–0.15; PpL 0.11–0.12; PpW 0.14–0.18; PtHI 65–82; PtWI 89–100; PpWI 127–150; WI 82–95 (holotype and eight paratype workers measured).
This species is similar to Crematogaster fraxatrix, but can be distinguished by the dorso-ventrally flattened propodeal spiracles and acutely developed subpetiolar process in the worker caste. The COI divergence between Crematogaster chhangi and Crematogaster fraxatrix did not seem relatively high (8.1–10.8% K2P distances) (cf.
Workers presumably monomorphic. Posterior corners of head rounded. Anterior clypeal margin slightly concave in the median portion. Compound eyes not projecting beyond lateral margins of head in full face view. Scape reaching posterior corner of head. Antennal club 3-segmented. Pronotal dorsum with distinct ridges laterally. Mesonotal dorsum with lateral ridges. Mesonotum not higher than pronotum in lateral view; forming same dorsal outline with pronotum in lateral view. Metanotal groove straight in dorsal view, deep and forming a concave region between mesonotum and propodeum. Propodeal spiracles oval, flattened dorso-ventrally, located on the lateral sides of propodeum; the horizontal diameter more than two times larger than the vertical diameter. Propodeal spines developed long, directed upward and straight. Petiole broader in the middle portion. Subpetiolar process acutely developed. Postpetiole weakly bilobed, but without longitudinal median sulcus. Petiole slightly wider than postpetiole in dorsal view. Erect pilosity sparse. Scape with abundant erect to suberect setae. Dorsal face of head with suberect setae. Clypeus with suberect setae; one pair of longer setae directed medially on anteriormost portion. Anterior clypeal margin with one single setae and one pair of longer setae, mixed with some shorter setae on the sides. Mesosoma with sparse erect setae. Fourth abdominal tergite with erect to suberect sparse setae. Dorsal surface of head generally smooth and shining, but feeble rugulae between frontal carinae; longitudinal rugulae surrounding antennal sockets and on gena. Clypeus weakly striated with longitudinal rugulae. Promesonotum striated with feeble rugulae. Lateral surface of pronotum smooth and shining. Mesopleuron sculptured. Lateral surface of propodeum generally smooth, but with feeble rugulae on the lower portion. Body color brown.
Crematogaster chhangi. 1 lateral view 2 dorsal view of mesosoma.
This species is known only from the type locality of Cambodia.
This species is dedicated to Mr. Phourin Chhang, Forestry Administration of Cambodia, who helped with field surveys in Cambodia.
http://species-id.net/wiki/Crematogaster_fraxatrix
Figs 3–6MALAYSIA: Sarawak, Borneo (Haviland). Lectotype worker by present designation: top specimen of three specimens of one pin.
THAILAND: 5 workers, Khlong Klai Stn., Khao Nan N. P., Nakhon S. Thamarat, 13.iii.2007 (TH07-SKY-22) (Sk. Yamane); MALAYSIA: 2 workers, Ulu Gombak, Selangor, 09.iii.2009 (SH09-Mal-51) (S. Hosoishi); 2 workers, Mt. Ophir, Gunung Ledan, Johor, 11.x.2011 (SH11-Mal-47) (S. Hosoishi); 3 workers, Lambir Hill’s National Park, Borneo, 21-27.ii.2009 (Y. Hashimoto).
HW 0.7–0.98; HL 0.64–0.93; CI 105–114; SL 0.58–0.68; SI 69–91; EL 0.13–0.18; PW 0.37–0.62; WL 0.69–0.95; PSL 0.13–0.21; PtL 0.18–0.25; PtW 0.18–0.25; PtH 0.13–0.17; PpL 0.11–0.15; PpW 0.16–0.23; PtHI 65–74; PtWI 94–111; PpWI 123–155; WI 83–92 (thirteen workers measured).
This species is similar to Crematogaster chhangi, but can be distinguished by the oval-shaped propodeal spiracles and weakly developed subpetiolar process in the worker caste. Based on COI divergence, the specimens from Peninsular Malaysia were separated from the Bornean specimens with a high support value (Fig. 9). This is presumably due to lack of gene flow between the populations, but they showed no distinct morphological differences between each other. The COI divergence of 0-9.3% (K2P distances) was recorded within Crematogaster ranavalonae clade in Madagascar (
Workers with weak polymorphism in size. Posterior corners of head rounded in smaller worker, but squared in larger workers. Anterior clypeal margin slightly concave in the median portion. Compound eyes not projecting beyond lateral margins of head in full face view. Scape reaching posterior corner of head. Antennal club 3-segmented. Pronotal dorsum with distinct ridges laterally. Mesonotal dorsum with lateral ridges. Mesonotum slightly higher than pronotum in larger workers in lateral view. Metanotal groove straight in dorsal view, deep and forming a concave region between mesonotum and propodeum. Propodeal spiracles dorso-ventrally oval, located on lateral sides of propodeum; the horizontal diameter slightly larger than the vertical diameter even in smaller workers. Propodeal spines long, directed upward and straight. Petiole broader in the middle portion. Subpetiolar process developed as small, blunt denticle. Postpetiole weakly bilobed, but without longitudinal median sulcus. Petiole slightly wider than postpetiole in dorsal view.
Sparsely hirsute with erect setae. Scape with abundant erect to suberect setae. Dorsal face of head with suberect setae. Clypeus with suberect setae; one pair of longer setae directed medially on anteriormost portion. Anterior clypeal margin with one single setae and one pair of longer setae, mixed with some shorter setae on the sides. Mesosoma with sparse erect setae. Fourth abdominal tergite with sparse erect to suberect setae.
Dorsal surface of head generally smooth and shining, but feeble rugulae between frontal carinae; longitudinal rugulae surrounding antennal sockets and on gena. Clypeus weakly striated with longitudinal rugulae. Pronotum striated with feeble rugulae. Mesonum weakly striated with feeble rugulae. Lateral surface of pronotum smooth and shining. Mesopleuron sculptured, but the central portion relatively smooth. Lateral surface of propodeum with feeble rugulae.
Body color reddish-brown to black.
Crematogaster fraxatrix. 3 lateral view 4 dorsal view of mesosoma 5 full face view 6 dorsal view of petiole and postpetiole.
This species is known from southern Thailand and Malaysia (Peninsular and Borneo).
http://zoobank.org/FFEDDB4D-EE5D-47D6-90A3-473F11F91056
http://species-id.net/wiki/Crematogaster_simboloni
Figs 7–8INDONESIA: Rakata Island, Krakatau Islands, 06°09'S, 105°28'E, 11.x.2000, H. Simbolon.
Holotype worker: pinned. Original label: INDONESIA, Rakata Island, Krakatau Islands, 06°09'S, 105°28'E, 11.x.2000, H. Simbolon leg., deposited at MZB.
Eight paratype workers: pinned, same data as holotype; deposited at BMNH, CASC, FRIM, KUM, MCZC, MHNG, NHMB, THNHM.
INDONESIA: 11 workers, Rakata Island, Krakatau Islands, 11.x.2000 (K. Ogata); 3 workers, Rakata Island, Krakatau Islands, 10.x.2000 (K. Ogata); 4 workers, Rakata Island, Krakatau Islands, 11.x.2000 (S. Matsui); 1 worker, Rakata Island, Krakatau Islands, 10.x.2000 (S. Matsui); 1 worker, Rakata Island, Krakatau Islands, 30.xii.2006 (Sk. Yamane); 1 worker, Rakata Island, Krakatau Islands, 31.xii.2006 (Sk. Yamane).
HW 0.59–0.72; HL 0.57–0.67; CI 102–108; SL 0.49–0.55; SI 75–85; EL 0.11–0.15; PW 0.34–0.39; WL 0.62–0.74; PSL 0.1–0.12; PtL 0.16–0.2; PtW 0.16–0.19; PtH 0.12–0.15; PpL 0.11–0.12; PpW 0.15–0.17; PtHI 72–82; PtWI 95–113; PpWI 125–155; WI 83–100 (fourteen workers measured).
This species is similar to Crematogaster chhangi and Crematogaster fraxatrix, but can be easily distinguished from these by the sculptured promesonotum in the worker caste. The COI divergence between Crematogaster simboloni and Crematogaster chhangi (24.3% K2P distances), as well as Crematogaster simboloni and Crematogaster fraxatrix (22.6 to 24.6% K2P distances) were also high.
Workers monomorphic. Posterior corners of head rounded. Anterior clypeal margin slightly concave in the median portion. Compound eyes projecting slightly beyond lateral margins of head in full face view. Scape reaching posterior corner of head. Antennal club 3-segmented. Pronotal dorsum with distinct ridges laterally. Mesonotal dorsum with lateral ridges. Mesonotum slightly higher than pronotum in lateral view. Metanotal groove straight in dorsal view, deep and forming a concave region between mesonotum and propodeum. Propodeal spines long, directed upward and straight. Propodeal spiracles oval, flattened dorso-ventrally, located on the lateral sides of propodeum, or the postero-lateral corners; the horizontal diameter slightly larger than the vertical diameter. Petiole broader in the middle portion. Subpetiolar process undeveloped. Postpetiole weakly bilobed with feeble median sulcus. Petiole as wide as postpetiole in dorsal view. Sparsely hirsute with erect setae. Scape with abundant erect to suberect setae. Dorsal face of head with suberect setae. Clypeus with suberect setae; one pair of longer setae medially on anteriormost portion. Anterior clypeal margin with one single setae and one pair of longer setae, mixed with some shorter ones on the side. Mesosoma with short and sparse erect setae. Fourth abdominal tergite with few erect to suberect setae. Dorsal surface of head generally smooth and shining, but feeble rugulae between frontal carinae; longitudinal rugulae surrounding antennal sockets and on gena. Clypeus striated with longitudinal rugulae. Pronotum striated with longitudinal rugulae with the sculptured space; the longitudinal rugulae separated from anterior mesonotal margin. Mesonotum sculptured. Lateral surface of pronotum smooth and shining. Mesopleuron sculptured, but the central portion relatively smooth. Propodeal dorsum sculptured anteriorly. Lateral surface of propodeum weakly sculptured and striated with feeble rugulae. Body color brown.
Crematogaster simboloni. 7 lateral view 8 dorsal view of mesosoma.
This species is known only from Indonesia (Krakatau).
This species is dedicated to Dr. Herwint Simbolon, Research Centre for Biology, Lembaga Ilmu Pengetahuan Indonesia (The Indonesian Institute of Sciences), who collected the type material.
Neighbor-joining (
We are grateful to Mr Phourin Chhang (Forestry Administration, Phnom Penh, Cambodia), Dr Rosli Hashim (Universiti Malaya), Dr Tsuyoshi Kajisa and Dr Nobuya Mizoue (Faculty of Agriculture, Kyushu University, Japan) and Dr Tetsukazu Yahara (Faculty of Science, Kyushu University, Japan) for assistance during field surveys for this study. We also thank the following persons for loan of material and access to museum collections: Dr Daniel Burckhardt (NHMB); Dr Bernhard Merz (MHNG). Dr Munetoshi Maruyama provided assistance with DNA sequencing and phylogenetic analyses. Additional material was received from Dr Seiki Yamane and Dr. Yoshiaki Hashimoto. We would like to thank Dr. Jusup Marko for his critical reading the manuscript. We would like to thank ANeT members for their encouragement. This work was supported in part by the Global COE program (Center of Excellence for Asian conservation ecology as a basis of human-nature mutualism), MEXT (The Ministry of Education, Culture, Sports, Science and Technology), Japan, and the Sumitomo Foundation (No. 113391).