(C) 2011 Stephane Bourassa. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
A study spanning ten years revealed changes in wing-morph ratios corroborating the hypothesis that the wing-dimorphic introduced carabid, Pterostichus melanarius Ill., is spreading through flight, from the city of Edmonton, Canada and establishing populations in natural aspen forest of more rural areas 45-50 km to the East. Comparison of wing-morph ratios between Pterostichus melanarius and the native wing dimorphic species Agonum retractum LeConte suggests that the spatial variation in ratios for Pterostichus melanarius does not reflect underlying environmental variation, but instead the action of selective forces on this wing-dimorphic species. About ten years after its earliest detection in some rural sites the frequency of macropterous individuals in Pterostichus melanarius has decreased c. five-fold, but it is still above the level seen in European populations in which the two wing-morphs are thought to exist in equilibrium. Pterostichus melanarius is expanding its range in native aspen forest much faster than three other introduced species Clivina fossor L.), Carabus granulatus O.F. Müllerand Clivina fossor L also encountered in this study. The two Carabus species are flightless, but Carabus fossor can be dimorphic. Although these four non-native ground beetle species comprise >85% of the carabids collected at sites in urban Edmonton, activity-density of native carabids was similar across the urban-rural gradient, suggesting little direct impact of introduced species on the local abundance of native species. In a second study conducted at a smaller scale near George Lake, Alberta, macropterous individuals of Pterostichus melanarius have penetrated furthest and most rapidly into native aspen forest. Furthermore, the percentage of micropterous individuals has increased markedly in areas first colonized a decade previously. Overall, these studies support the idea that macropterous beetles in wing-d dimorphic species are important vanguards for early colonization of unexploited territory, but that flightless individuals replace the flying morph relatively rapidly once populations are established.
Pterostichus melanarius, wing-dimorphism
Many introduced species of Carabidae (Coleoptera) are thought to have arrived in North America from Europe in ballast of commercial ships (
In some cases, this spread has been impressive and rapid. For example, the first Canadian record of Pterostichus melanarius Illiger is from 1926 in Nova Scotia (
The ubiquitous distribution of Pterostichus melanarius, the size of its populations and its penetration of natural habitats prompts questions about the impact of this species on the structure of native carabid assemblages. Previous studies demonstrated significant impact of Pterostichus melanarius on native carabid assemblages in natural forests in Alberta (
The rapid and successful dispersal of wing-dimorphic species like Pterostichus melanarius is frequently explained in relation to selection promoting the dimorphism. For Pterostichus melanarius, brachyptery (possession of non-functional short hind wings, hereafter designated as ‘SW’) seems to be inherited as a simple Mendelian dominant gene (
Although LW individuals are the exception in Europe (c. 2%) (
In this paper, we consider these propositions with focus on populations of Pterostichus melanarius in central Alberta, Canada. In the first section, we consider variation in the proportion of LW individuals on an urban-rural gradient, starting in the city of Edmonton, the suspected point of initial colonization (
Carabid populations were sampled during the summers of 1998, 1999 and 2007 along an urban-rural gradient using pitfall traps, as described by
Ten pitfall traps were set per site in a transect with traps separated from each other by a minimum of 15 m to ensure independence of samples (
Trap contents were collected every 2 weeks and stored in 70% ethanol until processed. After sorting, all carabids were identified using
All carabids captured at each site along the urban-rural gradient during a particular season were pooled for analysis, i.e., data from each site represents the pooled catch from 10 traps. Percentage data (e.g., %LW) were arcsin transformed prior to analysis in order to meet assumptions of parametric analyses employed. The following statistical analysis were all performed using R version 2.12.2 (R Development Core Team 2010).
A two-way analysis of variance (ANOVA) was performed to evaluate differences in %LW individuals between Pterostichus melanarius and Agonum retractum. Tukey’s HSD post-hoc test was used to evaluate differences revealed by ANOVA. To investigate if %LW varied among sampling years in Pterostichus melanarius, 2- way ANOVA was conducted, using year and position on the gradient (urban, suburban and rural) as model factors. A Tukey-Kramer post hoc test for unbalanced designs was used to identify significant groups in the data. Another two-way ANOVA was performed on the 1999 data (the only year in which sex was recorded) for Pterostichus melanarius to test for significant differences in %LW between sexes along the urban-rural gradient.
In order to assess the potential impact of urbanization on the native carabid assemblage, ANOVA was performed to compare whole-season standardized activity density of all native species among the three positions on the gradient. For this analysis, activity density was standardized to the lowest number of undisturbed trapping days recorded at any site.
Local scale movement at George lakeMovement of Pterostichus melanarius has been monitored using a pitfall grid in aspen forest near George Lake, Alberta (52.53°N, 112.10°W) since 1991. The study area and the general approach to trapping has been described in detail by (
In this paper, we present new data from this effort, as collected in 2007 and 2008.
Because our intent was to focus strictly on the population of Pterostichus melanarius, specimens were collected from the traps biweekly during the periods of 6 Jul to 1 Aug in 2007 and 14 Aug to 11 Sept in 2008. Although a few individuals of this species overwinter as adults and are active in the spring, the great majority of the annual adult activity is from overwintered larvae which emerge as teneral adults that are active and breed in Alberta during late summer. All carabids collected were identified to species throughout this study, except in 2007 where only Pterostichus melanarius was identified to species and all other carabids were recorded as “other carabids”. Wing length for all individuals of Pterostichus melanarius was recorded as LW or SW.
Analysis of George lake pitfall trap gridTo compare the earlier data of from the 1990s to the data collected in 2007 and 2008, catches of the two later collections were standardized as described in
The significance of temporal changes in %SW of Pterostichus melanarius was tested using a comparison of two proportions (Zar 1999) using data from 1991 – 1992 and 2007 – 2008. We compared the %SW captured for two sets of traps, those <100 m north of the highway and those >100m north.
Results Landscape scale movement of Pterostichus melanariusPatterns of Wing-Dimorphism in Pterostichus melanarius and Agonum retractum. Two-way ANOVA showed a significant interaction between ‘Gradient’ and ‘Species’ (P < 0.001) for transformed data about %LW in these two species. The %LW individuals was statistically higher for Pterostichus melanarius in rural sites than in either urban or suburban sites (Tukeys HSD, P<0.01) (Figure 1). Differences between urban and suburban populations were not as consistent (P = 0.070), although the average data certainly suggest existence of a gradient. There was no significant difference in the %LW for Agonum retractum across the gradient, suggesting that the clear pattern seen in Pterostichus melanarius did not result from underlying environmental variables that generally affect flight ability in local carabid populations. Also, we note that the %LW for Pterostichus melanarius was significantly higher than for Agonum retractum, but these means differed significantly only in rural sites (P < 0.001).
Figure 1. Percent LW individuals found in Pterostichus melanarius (a non-native species) and Agonum retractum (a native North American carabid) along an urban-rural gradient. Each bar represents the mean of 4 sites ± 1 S.E.
Spatio-temporal changes in pattern of wing-dimorphism in Pterostichus melanarius. There was significant interaction between the model factors ‘Year’ and ‘Gradient’ (P < 0.01) on %LW in Pterostichus melanarius between 1998 and 2007. A Tukey-Kramer multiple comparison test for unbalanced design confirmed that %LW was significantly lower in 2007 than in 1998 and 1999 in the rural sites (Figure 2). Thus, during this period, the %LW in Pterostichus melanarius decreased in rural sites but remained rather constant in both rural and suburban areas.
Figure 2. Comparison of %LW in Pterostichus melanarius along an urban-rural gradient over the years 1998, 1999 and 2007. Error bars are ± 1 S.E of the mean.
Sex-associated differences in %LW in Pterostichus melanarius. There was a significant effect of ‘Gradient’ (P < 0.001) on the overall %LW in Pterostichus melanarius but neither ‘Sex’ nor the (‘Sex’ × ‘Gradient’) interaction was significant. Thus, given the small numbers of Pterostichus melanarius caught in the rural sites in 2007 (2 males and 3 females), we could not detect any significant differences in %LW between males and females.
Distribution of introduced and native species over the gradient. Figure 3 shows a clear pattern of decreasing proportion of non-native of species with increasing distance from the urban center. We detected the presence of only Pterostichus melanarius, among non-native species, in the rural sites in two of the three years sampled. At the urban sites, however, 85.3% of the carabid fauna was attributable to the four introduced species. Populations of all four were detected in the suburban zone where they constituted 49.1% of the carabid fauna. Thus, among introduced carabids in central Alberta, the wing-dimorphic species, Pterostichus melanarius, is the superior colonizer.
Figure 3. Representation of four introduced carabid species in the carabid fauna along the urban-rural gradient (1998-99).
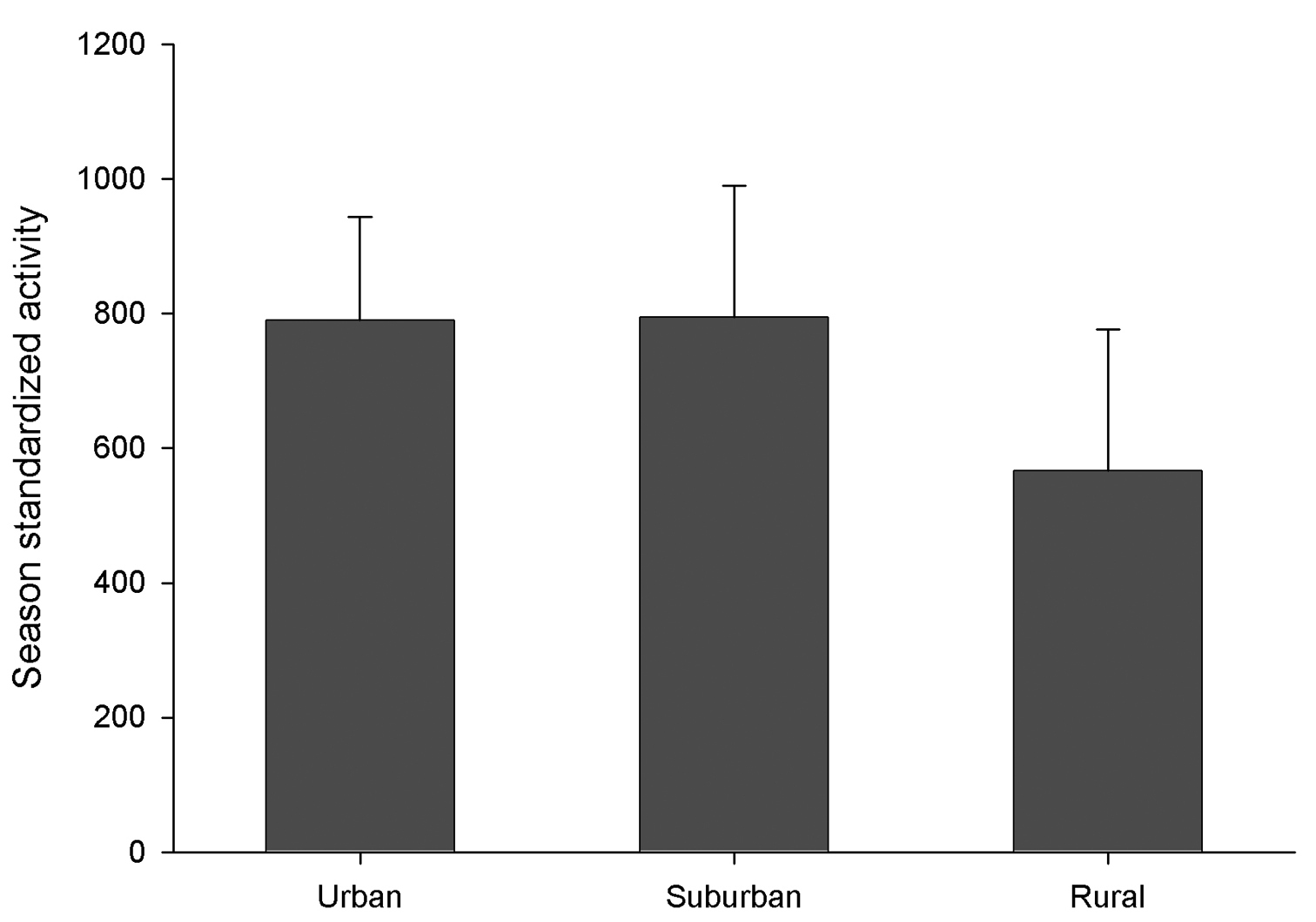
Despite the numerical dominance of introduced species at urban sites there is little evidence for overall negative impact on the native fauna. There was no change in overall activity density of native species along the urban-rural gradient (p > 0.05) (Figure 4), despite the huge abundance of introduced species at the urban sites.
Figure 4. Mean activity density of all native carabid species along the urban-rural gradient (1998- 1999). Activity density is expressed as standardized whole-season catch (see text for details). Means exclude 2 rural sites that were not sampled in 1999 and are therefore an average of 2 sites. There were no statistically significant differences among activity in these three zones (ANOVA, p > 0.05). Error bars show standard error.
Small scale movement of Pterostichus melanarius
A total of 236 adult Pterostichus melanarius were collected in the George Lake pitfall grids, 134 in 2007 and 102 in 2008. In contrast to the earlier years (1991-92 and 1997), the vast majority of Pterostichus melanarius individuals were collected in the 5 m forest edge (46.6%) while only 11.4% of the individuals were collected in the grassy road verge (Figure 5a). A total of 10.5% of the individuals were captured 50 m into the forest, leaving only 31.5% of the catch to be distributed over trap sites ≥ 100 m into the forest interior.
A total of 21 carabid species was collected at George Lake in 2008, with the highest species richness (12 species) found 450m from the highway. Pterostichus melanarius was the most abundantly collected species on the grid, followed by Scaphinotus marginatus Fischer (85 individuals)and Pterostichus adstrictus (45 individuals). Activity density of Pterostichus melanarius was similar to the earlier data up to 200 m into the forest; however, there was a clear increase in activity density at each sampling location beyond 200 m (Figure 5a), suggesting that the species is still colonizing the deciduous forest at George Lake.
In contrast to data from the previous decade (
Figure 5. Distributions of Pterostichus melanarius pitfall trapped at George Lake, starting in the road verge and extending 950 m into the forest for three sampling periods: 1991-92, 1997 and 2007-08 (see text for details). (a) Activity density standardized by the number of traps and number of days traps were in operation. In 2007 and 2008, activity density was adjusted for shorter sampling period as explained in the text. (b) Percentage of Pterostichus melanarius that was macropterous. Captures from some transects are pooled for presentation.
We found a clear pattern of increasing frequency of LW Pterostichus melanarius with increasing distance from the centre of Edmonton, using data from 1998 and 1999, collected forty years after the first report of this species in the city. Because a similar pattern was not reflected for the native wing-dimorphic species Agonum retractum, we suggest that the pattern in Pterostichus melanarius did not result from general response to some underlying habitat gradient. Instead, we propose this is evidence that Pterostichus melanarius is spreading outward onto the rural landscape from a local centre of high population in Edmonton through flight of LW individuals, as suggested in Lindroth’s general hypothesis (
In 1998-99 populations of Pterostichus melanarius appeared to be more stable in the urban area but were still expanding in the rural area. Data collected in 2007, however, suggest that the %LW individuals in the rural area had dropped significantly. Because the frequency of the recessive LW trait (
Our data are in agreement with the hypothesis that LW Pterostichus melanarius are responsible for movement associated with the early vanguard of first colonization and that over a relatively short time (in this case, about one decade), the SW morph becomes the dominant phenotype. Our data also support the hypothesis that population age can be roughly estimated using the frequency of LW in a population. After about 50 years, the frequencies of the SW phenotype in Pterostichus melanarius of urban and suburban areas have risen considerably, although they are still short of levels characteristic of established European populations, e.g., around 98% (
Our study also reinforces the view that given sufficient time, introduced species like Pterostichus melanarius can become established in at least some natural habitats in North America. All the non-native species reported here and several others have previously been recorded far from urban areas in agricultural land and disturbed habitats (
It is not presently clear how to best interpret the high captures of non-native carabids in urban areas (Figure 3), where they accounted for 85.3 % of the total catch in this study. It is presently impossible to separate two possible explanations: 1) human disturbances in the city landscape favour these non-native, synanthropic species, either by direct habitat optimization effects that open up ‘niche space’ accessible mainly to the introduced species, or through broader landscape effects that increase the local pool of colonists every season, and 2) their high populations, even in urban aspen patches, simply reflect a longer history of these local populations. However, the lack of change in activity density of native carabids along the urban-rural gradient (see Figure 4) suggests that 1) urbanization around sufficiently large urban forest patches does not have a strong impact on the native fauna, and/or 2) the impact of introduced carabids on the native species is low. Defining the potential roles of inter-specific competition and intraguild predation in these systems awaits further research.
Small scale dispersal at George lakeThis study and previous work (
In contrast to the high and relatively stable activity density of Pterostichus melanarius in the road verge, there was a general increase ofactivity density in the forest, especially at sites >200 m north of the highway. As suggested by