ZooKeys 130: 153–165, doi: 10.3897/zookeys.130.1430
The oldest psyllipsocid booklice, in Lower
Cretaceous amber from Lebanon (Psocodea, Trogiomorpha, Psocathropetae,
Psyllipsocidae)
Dany Azar1,†, André Nel2,‡
1
Lebanese University, Faculty of Sciences II,
Department of Natural Sciences, Fanar, Fanar – Matn – P.O. Box
26110217, Lebanon
2
CNRS UMR 7205, CP 50, Entomologie, Muséum National
d’Histoire Naturelle, 45 rue Buffon, F-75005, Paris, France
Corresponding author: Dany Azar (azar@mnhn.fr).
Academic editor: D. Shcherbakov
Abstract
Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., of Psyllipsocidae
is described and figured from the Lower Cretaceous amber of Lebanon.
The position of the new taxon is discussed and the fossil is compared to
other psyllipsocids. The species represents the earliest record of the
family Psyllipsocidae.
KeywordsNew genus, new species, Psyllipsocidae, Lower Cretaceous, Lebanese amber
Introduction
The Psocodea (Psocoptera + Phthiraptera)
is a rather small order with about 10, 000 valid extant species.
Earlier, fossils from the Permian were assigned to the Psocodea but the
attribution of these is uncertain. The first unquestionable records of
psocodeans are from the Middle Jurassic of China (Huang et al. 2008). The Psyllipsocidae
are a relatively small family of trogiomorphan psocathropetan Psocodea,
including about 50 extant species in five genera distributed
worldwide. Psyllipsocids live in caves, on rock surfaces, and among
dead leaves. To date four fossils have been assigned to Psyllipsocidae: Psyllipsocus eocenicus Nel et al., 2005 (lowermost Eocene French Oise amber), ?Psyllipsocus banksi Cockerell, 1916 (mid-Cretaceous Burmese amber), a ”Psyllipsocus sp. (nymph)“ from Lower Miocene Mexican amber (Mockford 1969), and Khatangia inclusa Vishniakova, 1975 (Late Cretaceous Taimyr amber) but the last three attributions to Psyllipsocidae are dubious and require revision.
The Lower Cretaceous Lebanese amber is so far the oldest (Neocomian) with complete macroscopic biological inclusions (Azar 2007).
Its formation corresponds to the epoch of the origin and rise of
flowering plants, a critical period for the study of the evolution,
with extinction of many ancient insect groups and emergence of
effectively modern lineages. Up to now nine species of psocids have been
described from Lebanese amber (Perrichot et al. 2003; Azar and Nel 2004; Grimaldi and Engel 2006; Azar et al. 2008, 2010).
Herein a new psyllipsocid genus and species is described
from the Lower Cretaceous amber of Lebanon, representing the earliest
record of this family.
Material and methods
The amber piece containing the inclusion was cut,
shaped, and polished, before preparations between two coverslips and
in a Canada balsam medium, as described in Azar et al. (2003).
The specimen was examined with a Nikon SZ10 stereomicroscope and a
Leitz Laborlux-12 compound microscope, both equipped with camera lucida
for line drawings. Photographs were made with an Olympus FE-5000
digital camera.
The works of (Smithers (1972, 1990), Lienhard (1998) and Mockford (1993) are followed herein for the systematics of Psocodea. The wing venation nomenclature and terminology of body structures of Smithers (1972) and Lienhard (1998) were adopted.
Systematic Paleontology
Suborder Trogiomorpha Roesler, 1944.
Infraorder Psocathropetae Pearman, 1936.
Family Psyllipsocidae Enderlein, 1911
Type species.
Libanopsyllipsocus alexanderasnitsyni sp. n.
Etymology.
After “Libano” (Lebanese in Latin), and “Psyllipsocus”, type genus of the family Psyllipsocidae; gender masculine.
Diagnosis.
Hypopharynx with two distinct filaments.
Macropterous; forewing without pterostigma; M two-branched; areola
postica elongate and free; Cu2 and A ending together forming a nodulus.
Hindwing with R1 absent; Rs two-branched; M simple; basal cell present.
Distal end of tibia with two inner spines; tarsi three-segmented; distal
pretarsi with claw with preapical tooth, and without pulvillus and
microtrichia.
Holotype.
Specimen n° 30 (male, coll. Azar); provisionally deposited in the Muséum National d’Histoire Naturelle, Paris, France.
Locality and horizon.
Lower Cretaceous, Upper Barremian – Lower
Aptian, amber of Hammana-Mdeyrij, Caza, Baabda, Mohafazat Jabal
Loubnan, central Lebanon.
Etymology.
In honor of our friend Prof. Alexandr P. Rasnitsyn, a world authority in entomology and palaeoentomology.
Diagnosis.
As for the genus (vide supra).
Description.
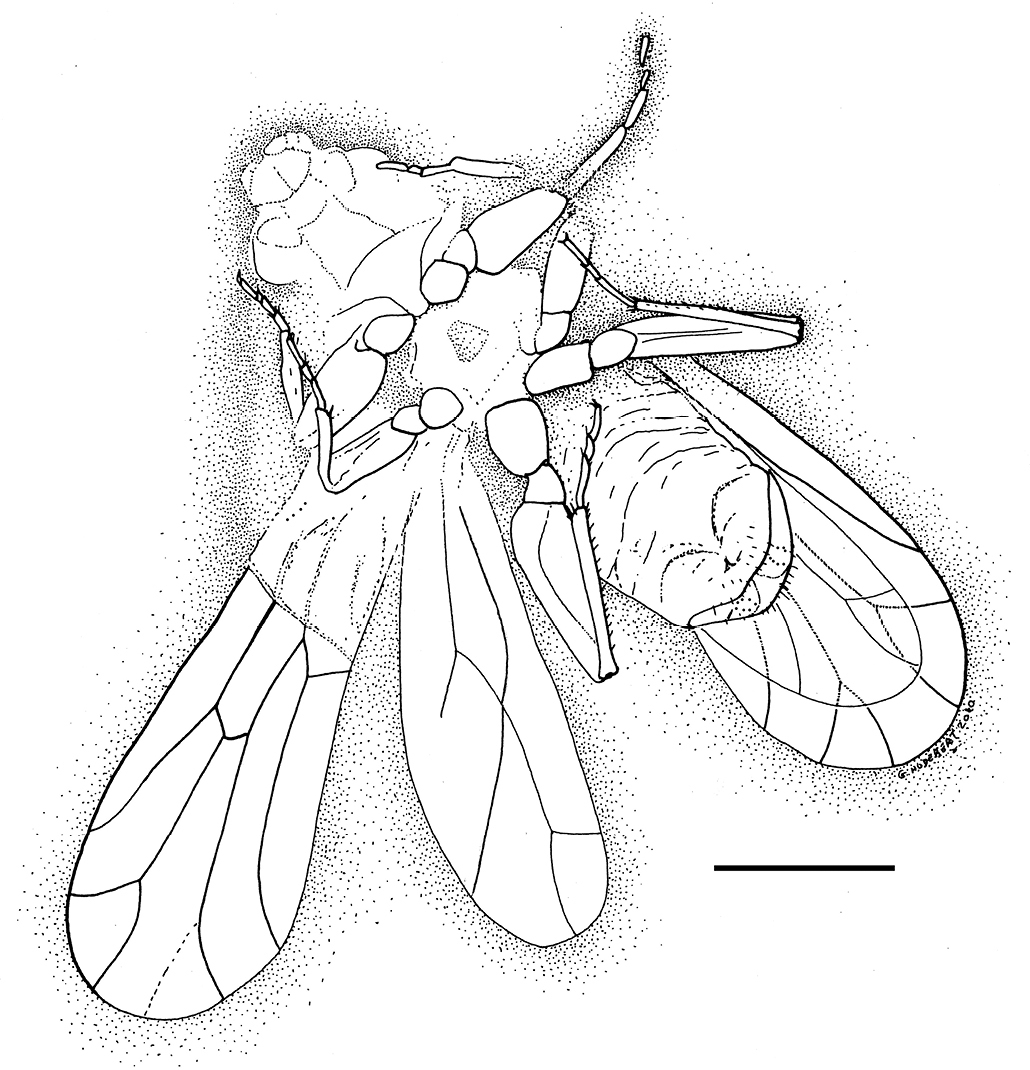
Dorsal parts of head and body not preserved. Body and wings without scales (Figs 1–2).
Head elongate (but cannot be measured accurately due to state of
preservation), sublingual sclerite and hypopharynx with two distinct
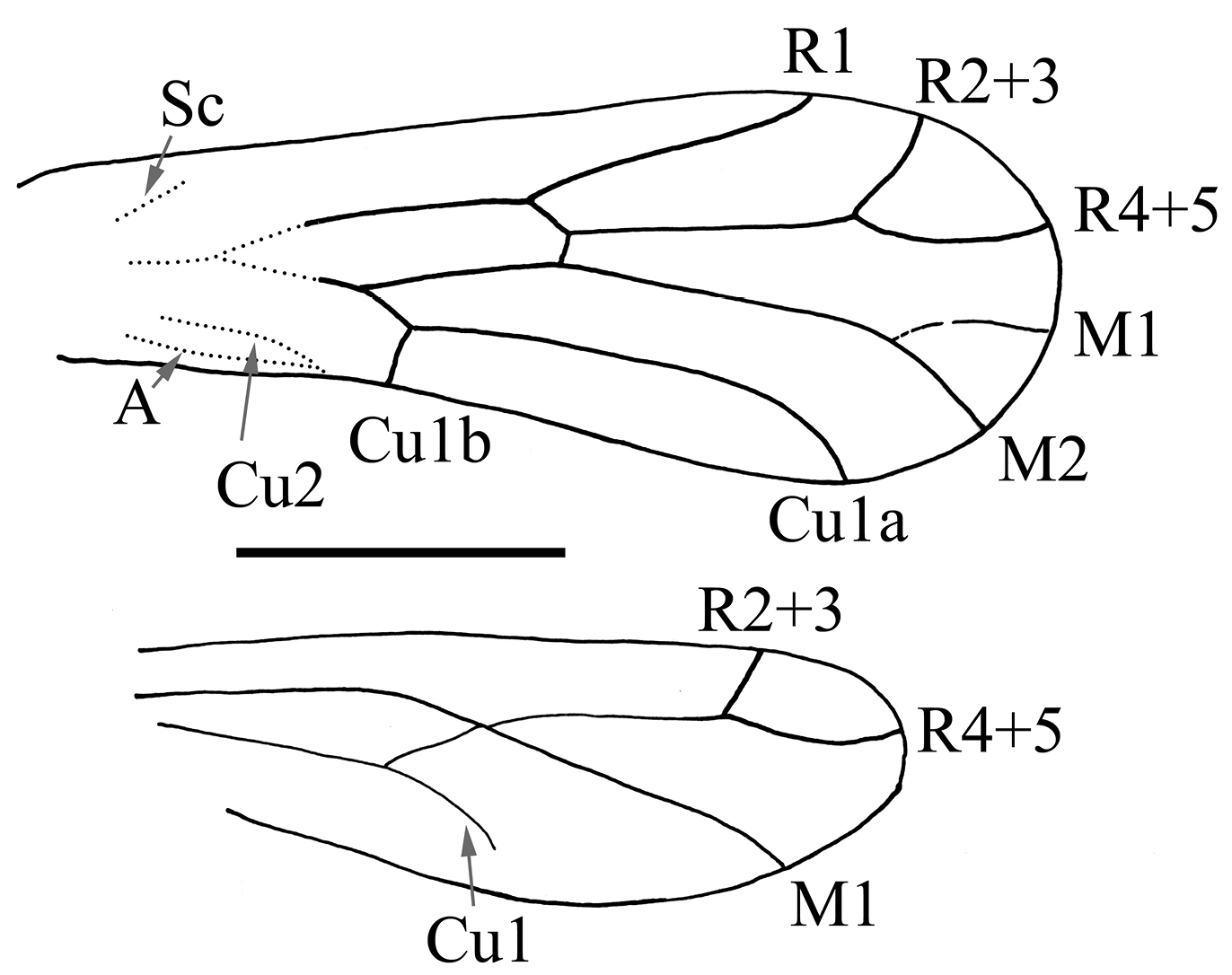
filaments (Fig. 3). Forewing transparent (Fig. 4),
with infrequent small setae, 0.96 mm long, 0.35 mm broad; apex
rounded; Sc badly preserved, not reaching C nor R; R1 reaching costal
margin 0.73 mm from wing base; pterostigma absent; Rs separating from R
0.46 mm from wing base; Rs bifurcating into R2+3 and R4+5 at 0.76 mm;
R2+3 curved, reaching wing margin 0.83 mm distally; R4+5 curved,
reaching wing margin at 0.95 mm distally; a transverse crossvein between
Rs and M; fork of M into M1 and M2 0.8 mm from wing base, but one wing
is aberrant in having M simple [a frequent teratology in Psyllipsocidae as noted by Lienhard (1998)];
Cu1 bifurcating into Cu1a and Cu1b 0.36 mm from wing base forming a
free elongate and narrow areola postica; Cu1a much longer than Cu1b; Cu2
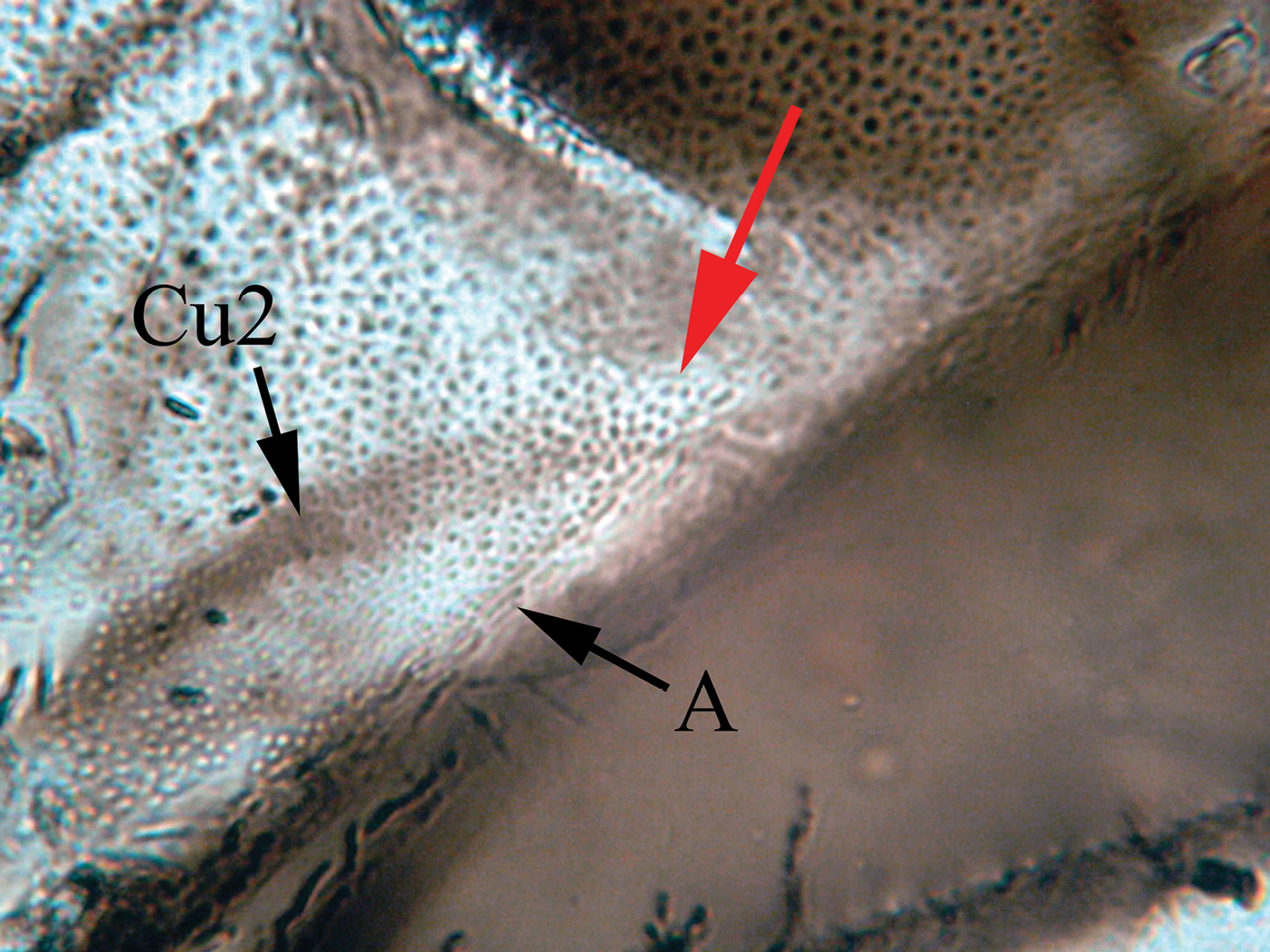
(badly preserved) joining anal vein at posterior wing margin in a
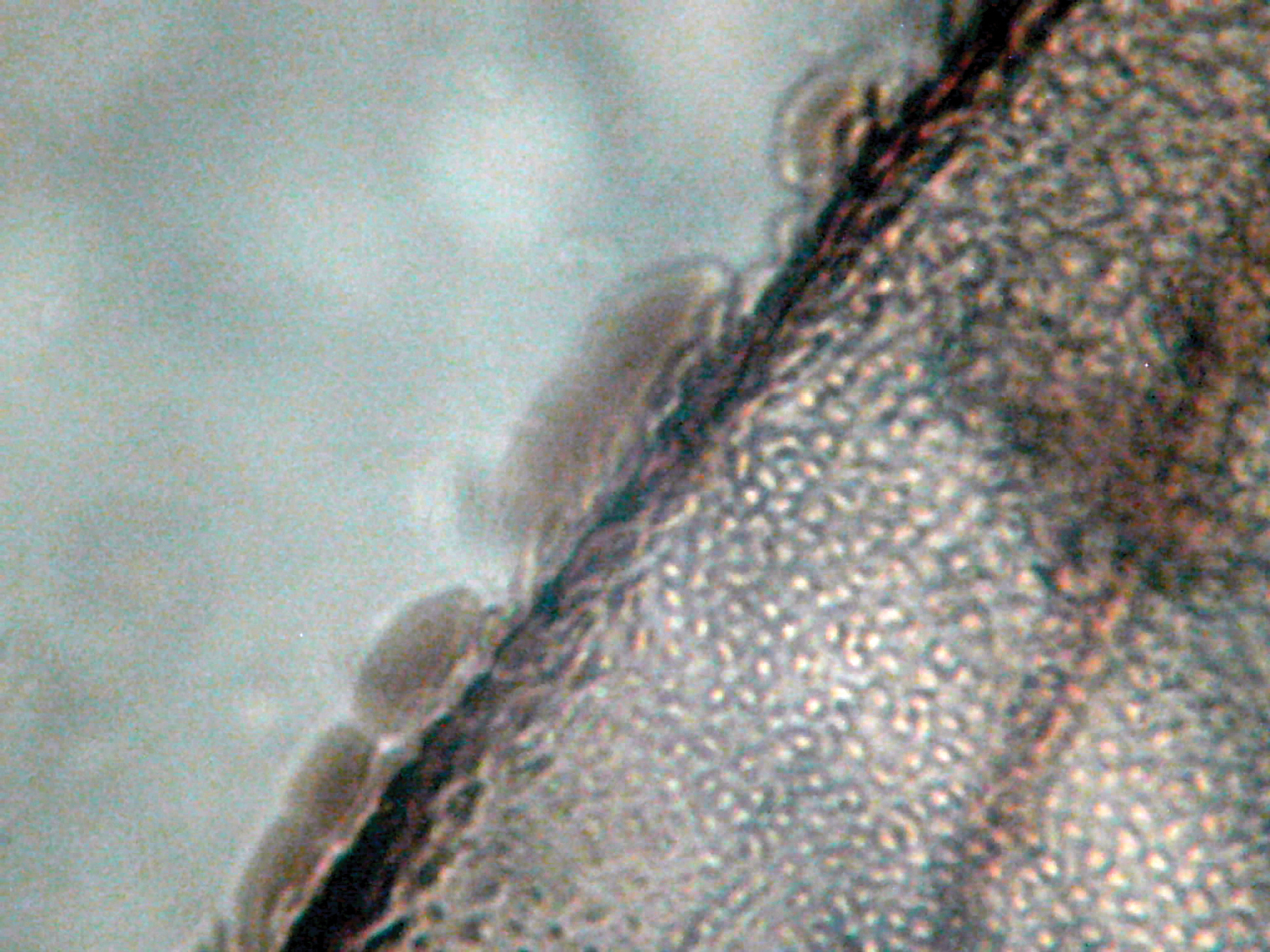
nodulus 0.28 mm from wing base (Fig. 5); A strongly convex. Wing margin glabrous (Fig. 6), similar in structure to that illustrated by Lienhard (1998: 111, fig. 31b). Hind wing transparent (Fig. 4),
bare and smaller than forewing, about 0.75 mm long, 0.24 mm broad;
free or individualized and distinct arm of R1 missing; Rs forked into
R2+3 and R4+5 0.16 mm from wing apex; basal cell present; simple M
reaching wing margin apically; basal part of wing badly preserved. Hind
leg with tibia + tarsi slightly longer than abdomen. Coxal rasp of
Pearman’s organ present on hind leg (Fig. 7),
but without tympanum (the coxal rasp is preserved on one leg as the
coxal area is missing on the other leg). Tibiae with two apical spurs on
inner sides; all tarsi three-segmented with first tarsomere longer than
others; pretarsal claws with a preapical tooth, without microtrichia
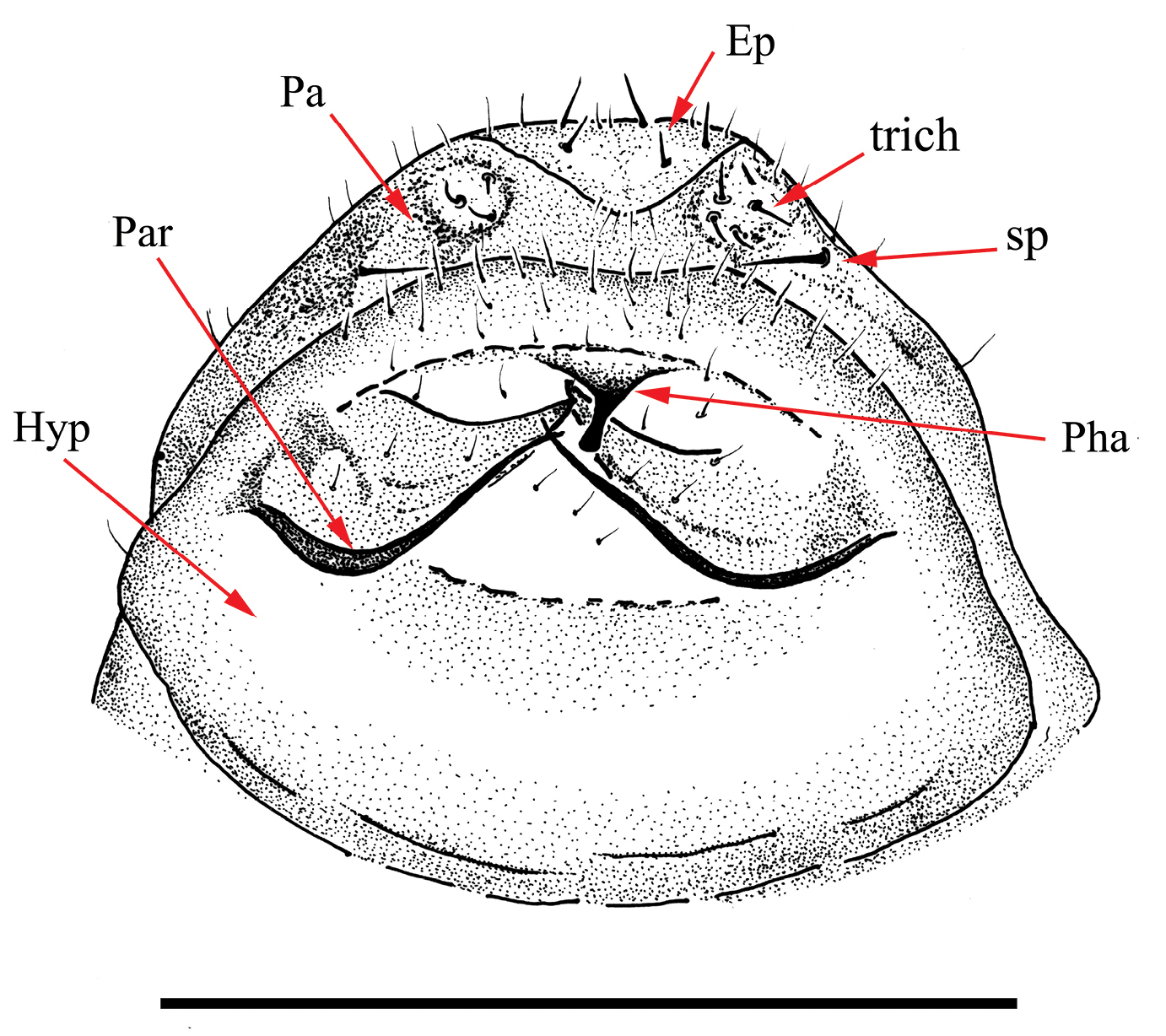
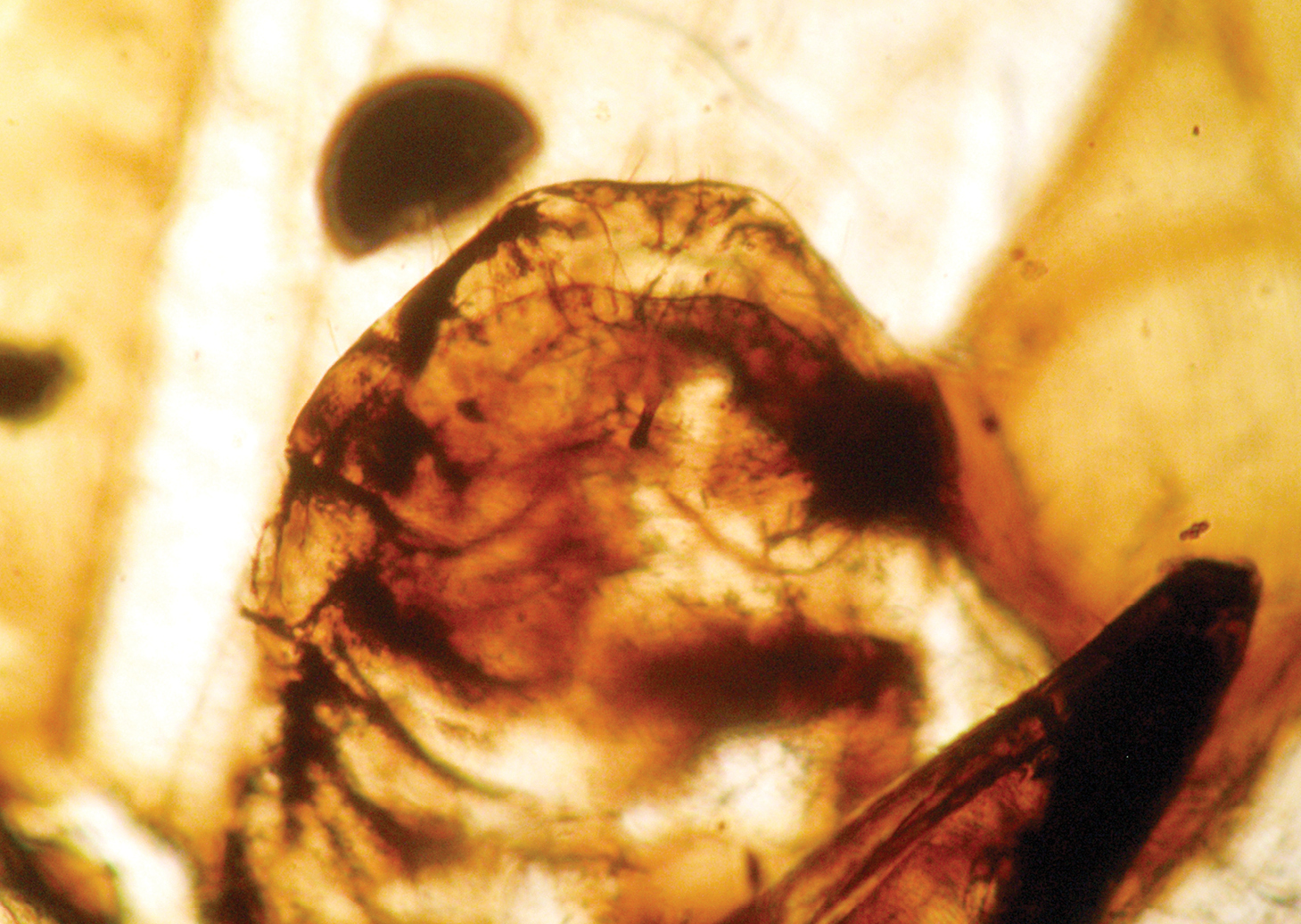
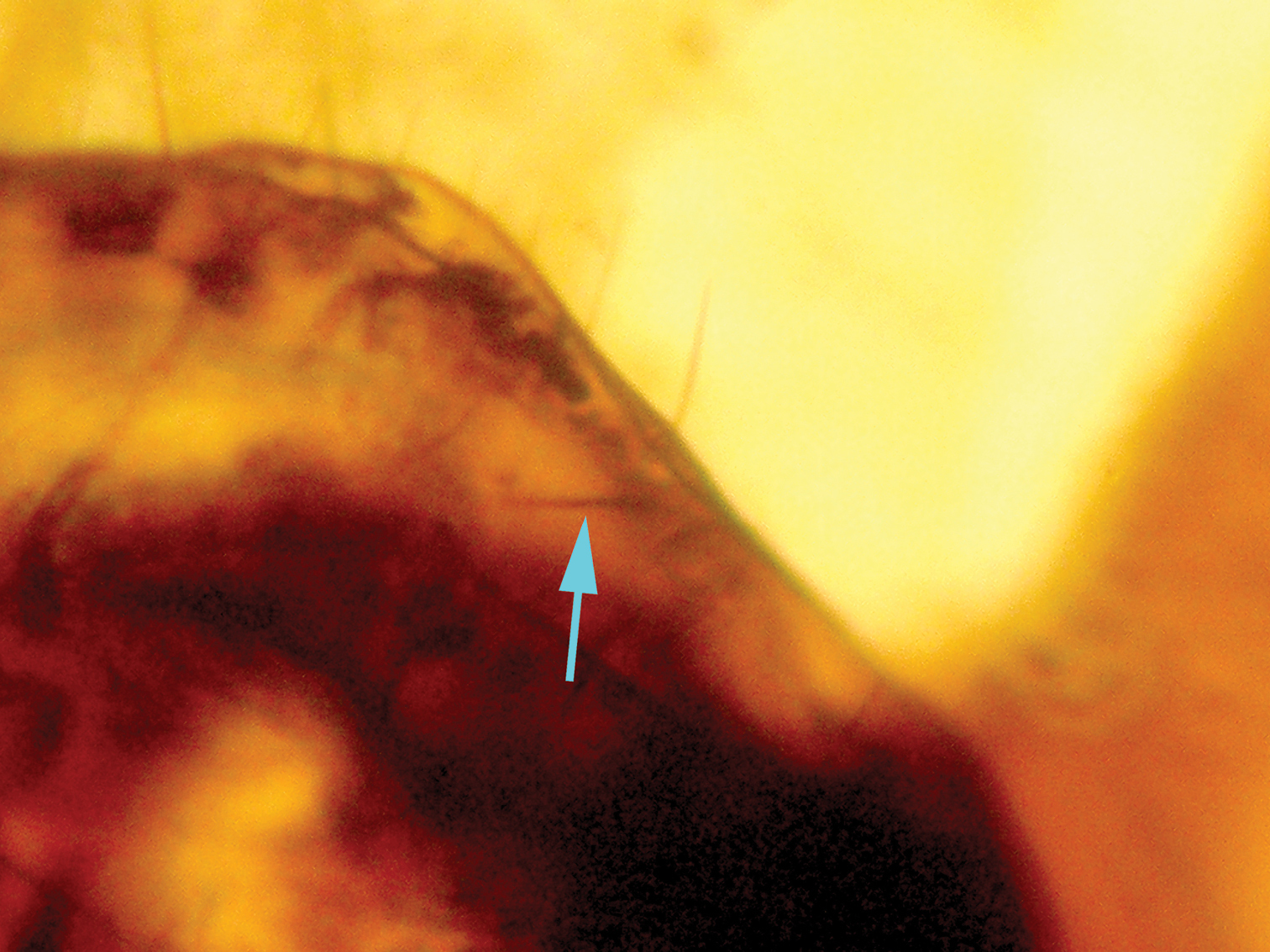
nor pulvillus (Fig. 8). Abdomen 0.52 mm long, 0.32 mm broad; telson with epiproct and paraproct as represented in Figs 9–12,
paraproct with a rounded hemispherical sensory area of trichobotria,
and a spine in its ventral inner side; male genitalia with simple
hypandrium, aedeagus with phallosome presenting parameres with curved
arms not fused anteriorly as represented in Fig. 9 (it can be seen owing to transparency of sclerites and is comparable in structure to what occurs in other Psyllipsocidae (cf. Lienhard 1998: 112, fig. 32b).
Following the keys of Smithers (1972), Mockford (1993), and Lienhard (1998), Libanopsyllipsocus alexanderasnitsyni gen. et sp. n. would fall into Trogiomorpha and can be placed in Psyllipsocidae owing to the following combination of characters, considered diagnostic of the family (Smithers 1972, 1990; Mockford 1993; Lienhard 1998),
and present in the current fossil: head elongate; hypopharynx with two
distinct sclerotized filaments; presence of nodulus; posterior legs
with tibia + tarsi longer than abdomen; tarsi three-segmented; distal
pretarsi with claw bearing preapical tooth, without pulvillus;
paraproct with anal spine; phallosome with two curved arms not fused
anteriorly. Other diagnostic characters of the family Psyllipsocidae
could be present but cannot be seen in the specimen owing to the state
of preservation, i.e., some parts of the animal are missing.
Our fossil presents a striking and astonishing similarity
in some of the wing features (like elongate areola postica and M
2-branched in forewing) with the psocids belonging to the troctomorphan
family Pachytroctidae, to the point that any psocidologist at first glance to the specimen would assign it to this family, and as in Pachytroctidae
only females could be winged, he would tell directly that this is a
female! A minute examination of the specimen shows that the fossil is a
male with genitalia structure comparable to those of Psyllipsocidae
as figured by Lienhard (1998: 112, fig. 32b) with the very particular
conical phallosome directed forwards. Moreover, our fossil could not
belong to the Pachytroctidae
for several reasons: (1) in the forewing Cu2 and A are meeting together
on the wing margin thus forming a nodulus, the absence of this feature
being characteristic for Pachytroctidae; (2) presence of Pearman’s coxal organ in hind legs, here too the absence of this feature is characteristic of the Pachytroctidae (Yoshizawa and Lienhard 2010),
and even for the whole Nanopsocetae that include this family; (3)
presence of distinct separated sclerotized filaments of hypopharynx
characteristic of all Trogiomorpha, whereas in Troctomorpha (that
enclose Pachytroctidae) these filaments are fused.
Our fossil shows a derived character for its hind wing
with M simple, though the majority of the fully-winged extant species
of Psyllipsocidae,
like most of the trogiomorphan psocids, have hind wings with M forked
(a feature considered plesiomorphic in general for the Psocodea).
Libanopsyllipsocus gen. n. differs from all modern genera by several features of its wings; i.e., from the genera Dorypteryx Aaron, 1899 and Psocathropos Ribaga, 1899 by the glabrous anterior margin of the forewing; from Pseudodorypteryx García-Aldrete, 1984 and Dorypteryx Aaron, 1899 by the broad forewing. Libanopsyllipsocus gen. n. resembles mostly the genus Psyllipsocus
Sélys-Longchamps, 1872 but differs from it in its forewing without a
pterostigma and M two-branched, and in its hind wing with R1 absent and
M simple. Libanopsyllipsocus gen. n. differs from the genus Khatangia
Vishniakova, 1975 in the absence of a pterostigma and an elongate
areola postica in the forewing. All these differences and many others
lead us to the proposal of a new genus.
The monophyly of the family Psyllipsocidae
is supported by one character of the female genitalia: the spermathecal
sac with complex scleritizations at the origin and usually with an
accessory vesicle. Molecular analysis also supports the monophyly of the
family (Yoshizawa et al. 2006). Libanopsyllipsocus alexanderasnitsyni gen. et sp. n. represents the oldest record of the Psyllipsocidae
and demonstrates that this family is at least as old as the lowermost
Cretaceous, if not older. Unexpectedly some features of this very old
fossil are probably derived, viz., the absence of a pterostigma in the
forewing and M simple in the hind wing. The discovery of new fossils
belonging to this family could bring more information on the polarity
and potential homoplasy of some characters in use for the
classification, and increase as such our knowledge of the evolutionary
history of this lineage.
Acknowledgments
The authors are grateful to Michael S. Engel for his
important comments on an earlier version of the manuscript. This paper
is a contribution to the team project “Biodiversity: Origin, Structure,
Evolution and Geology” granted to DA by Lebanese University.
ReferencesAzar
D (2007) Preservation and accumulation of biological inclusions in
Lebanese amber and their significance. Comptes rendus – Palevol 6(1/2):
151–156.
doi:10.1016/j.crpv.2006.10.004
Azar D, Hajar L, Indary C, Nel A (2008)
Paramesopsocidae, a new Mesozoic psocid family (Insecta: Psocodea
“Psocoptera”: Psocomorpha). Annales de la Société Entomologique de
France (NS) 44: 459-470.
Azar D, Engel MS, Grimaldi DA (2010) A new
genus of sphaeropsocid bark lice from the Early Cretaceous amber of
Lebanon (Psocodea: Sphaeropsocidae). Annales de la Société Entomologique
de France (NS) 46: 103-107.
Azar D, Nel A (2004) Four new Psocoptera from
Lebanese amber (Insecta: Psocomorpha: Trogiomorpha). Annales de la
Société Entomologique de France (NS) 40: 185-192.
Azar D, Perrichot V, Néraudeau D, Nel A
(2003) New psychodid flies from the Cretaceous ambers of Lebanon and
France, with a discussion about
Eophlebotomus connectens Cockerell, 1920 (Diptera, Psychodidae). Annals of the American Society of America 96: 117-127.
doi:10.1603/0013-8746(2003)096[0117:NPFTCA]2.0.CO;2
Cockerell TDA (1916) Insects in Burmese amber. American Journal of Science, Series 4, 42: 135–138.
Enderlein G (1911) Die Fossilen Copeognathen und ihre Phylogenie. Palaeontographica 58: 279-360.
Huang D-Y, Nel A, Azar D, Nel P (2008)
Phylogenetic relationships of the Mesozoic paraneopteran family
Archipsyllidae (Insecta: Psocodea). Geobios 41: 461-464.
doi:10.1016/j.geobios.2007.11.003
Lienhard C (1998) Psocoptères Euro-Méditerranéens. Faune de France 83, 1-533.
Mockford EL (1969) Fossil insects of the order
Psocoptera from Tertiary amber of Chiapas, Mexico. Journal of
Paleontology 43: 1267-1273.
Mockford EL (1993) North American Psocoptera (Insecta). Flora & Fauna Handbook 10: 1-455.
Nel A, Prokop J, De Ploëg G, Millet J
(2005) New Psocoptera (Insecta) from the lowermost Eocene amber of Oise,
France. Journal of Systematic Palaeontology 3: 371-391.
doi:10.1017/S1477201905001598
Perrichot V, Azar D, Néraudeau D, Nel A
(2003) New Psocoptera in the Lower Cretaceous ambers of southwestern
France and Lebanon (Insecta: Psocoptera: Trogiomorpha). Geological
Magazine 140 (6): 669-683.
doi:10.1017/S0016756803008355
Roesler R (1944) Die Gatungen der Copeognatha. Stettiner Entomologisches Zeitung 105: 117-66.
Smithers CN (1990) Keys to the family and
genera of Psocoptera (Arthropoda: Insecta). Technical reports of the
Australian Museum, 1–82.
Vishniakova VN (1975) Psocoptera in Late Cretaceous insect-bearing resins from the Taimyr. Entomological Review 54: 63-75.
Yoshizawa K, Lienhard C (2010) In search of
the sister group of the true lice: A systematic review of booklice and
their relatives, with an updated checklist of Liposcelididae (Insecta:
Psocodea). Arthropod Systematics & Phylogeny 68 (2): 181-195.
Yoshizawa K, Lienhard C, Johnson K (2006)
Molecular systematics of the suborder Trogiomorpha (Insecta: Psocodea:
‘Psocoptera’). Zoological Journal of the Linnean Society 146: 287-299.
doi:10.1111/j.1096-3642.2006.00207.x
Plates
Figure 1.
Photograph of habitus of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male, specimen number 30.
Figure 2.
Drawing of habitus of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male, scale bar = 0.3 mm.
Figure 3.
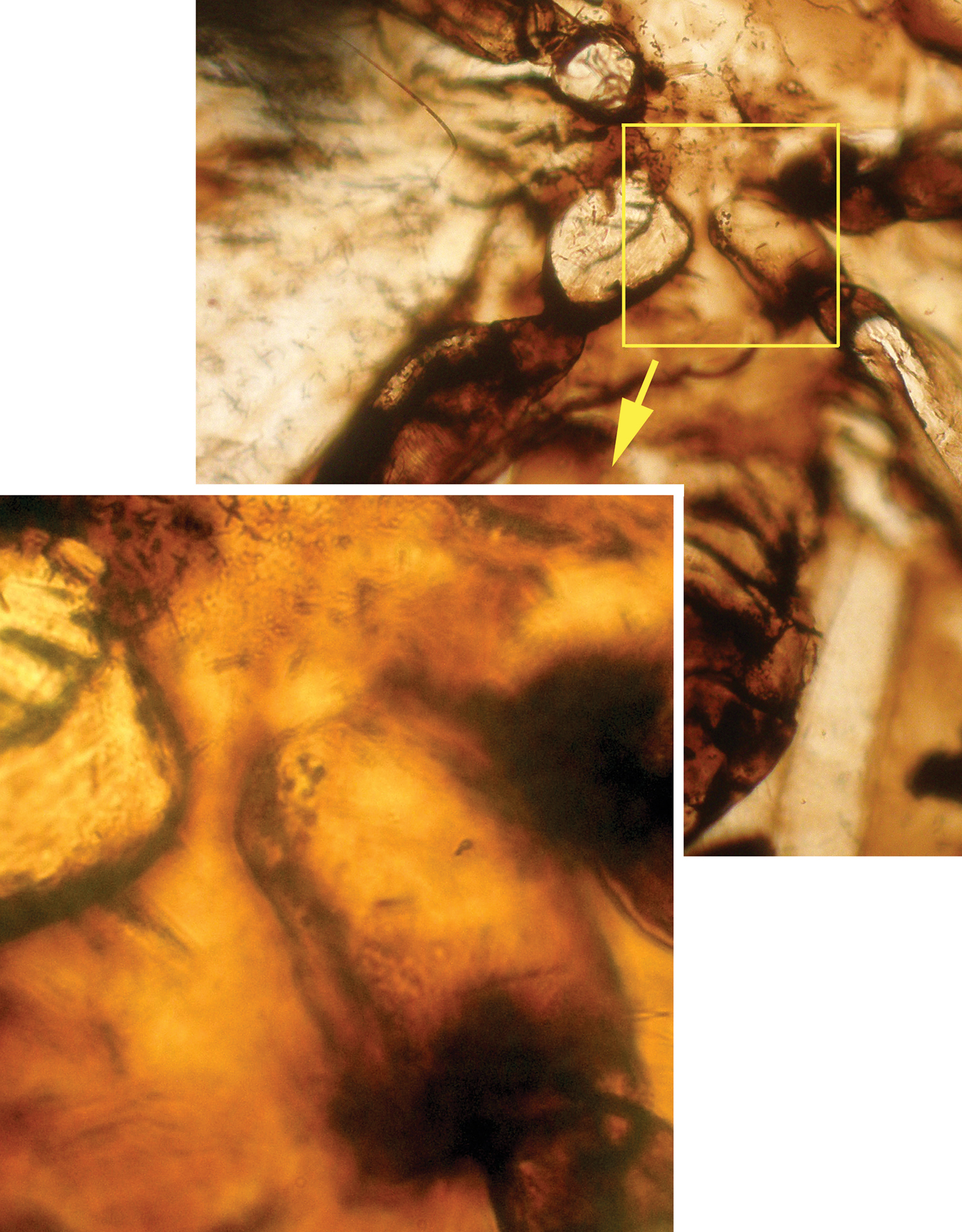
Photograph of hypopharynx filaments (arrows) of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male.
Figure 4.
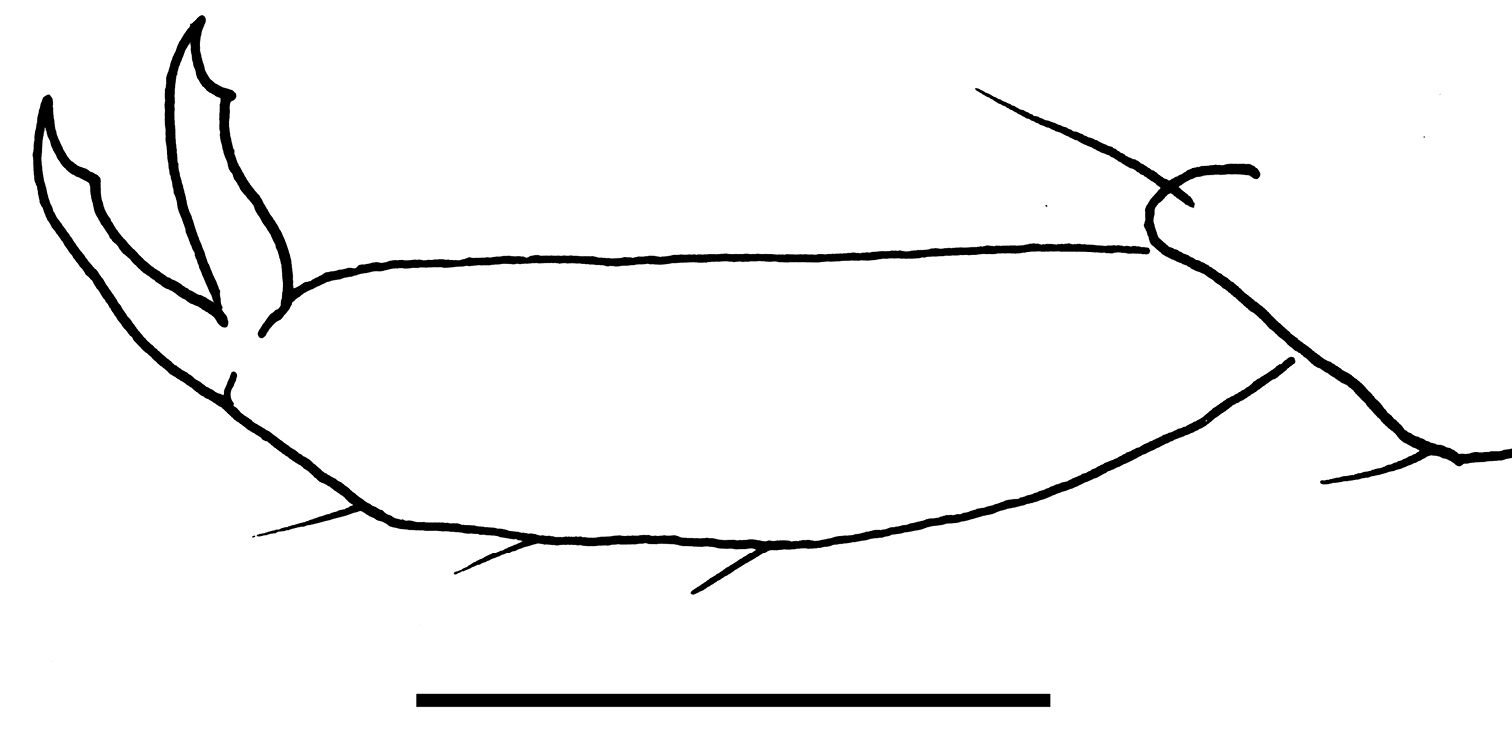
Drawing of wings of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male, scale bar = 0.3 mm.
Figure 5.
Microphotograph of nodulus, arrow showing the meeting area of Cu2 and A.
Figure 6.
Microphotograph of structure of forewing margin.
Figure 7.
Microphotograph of hind leg coxal rasp (Pearman’s organ).
Figure 8.
Drawing of pretarsal claw of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male, scale bar = 0.03 mm.
Figure 9.
Drawing of aedeagus of Libanopsyllipsocus alexanderasnitsyni
gen. et sp. n., holotype, male; Ep = epiproct, Hyp = hypandrium,
Par = paraproct, par = paramers, Pha = phallosome, sp = anal spine,
trich = trichobothria; scale bar = 0.3 mm.
Figure 10.
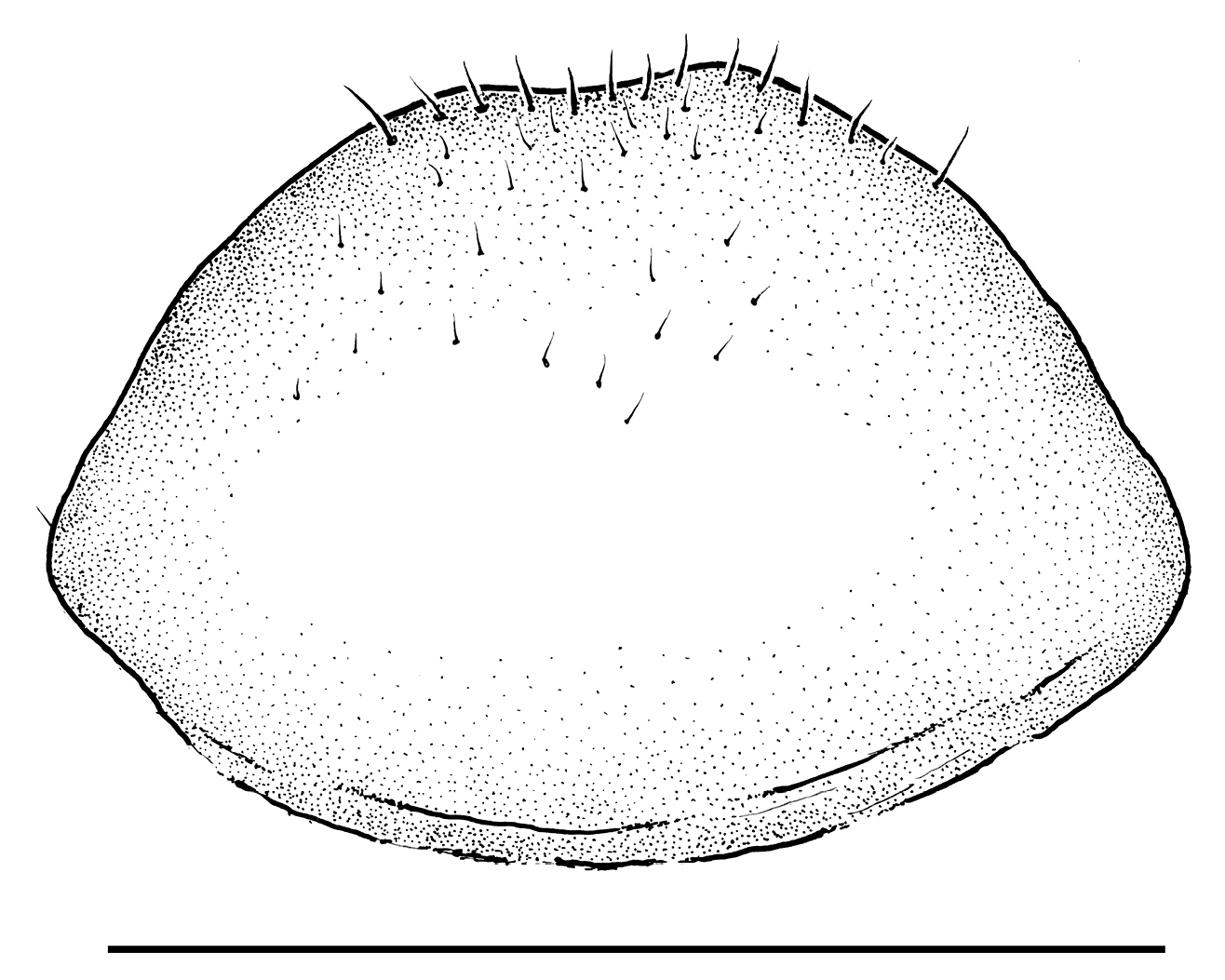
Drawing of hypandrium of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male; par = paraproct, ep = epiproct, trich = trichobothria; scale bar = 0.3 mm.
Figure 11.
Photograph of hypandrium and aedeagus of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male.
Figure 12.
Photograph of paraproct of Libanopsyllipsocus alexanderasnitsyni gen. et sp. n., holotype, male; arrow shows the anal spine.