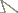(C) 2011 Martin Fikáček. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
The recent hydrophiloid species Helophorus (Gephelophorus) sibiricus (Motschulsky, 1860) is recorded from the early Miocene deposits of Kartashevo assigned to the Ombinsk Formation. A detailed comparison with recent specimens allowed a confident identification of the fossil specimen, which is therefore the oldest record of a recent species for the Hydrophiloidea. The paleodistribution as well as recent distribution of the species is summarized, and the relevance of the fossil is discussed. In addition, the complex geological settings of the Kartashevo area are briefly summarized.
Coleoptera, Hydrophiloidea, Helophorus, Miocene, Kartashevo, long-living species, environmental stability
The understanding of the evolution of beetles in the late
Cenozoic has changed drastically within recent decades. This concerns
especially the Pleistocene beetles preserved as unmineralized subfossil
specimens. Originally, the remains from the Pleistocene peatbog or
asphalt deposits were assigned to extinct species by historical authors
(e.g.
The presence of recent species in Pleistocene deposits
invites the question as to whether the same might not be the case even
in older, Pliocene or Miocene deposits. Pre-Pleistocene beetle fossils
were mostly assigned to extinct species of recent genera (
Representatives of the hydrophiloid genus Helophorus
Fabricius, 1775 are frequently used in the studies of Quaternary
beetle communities. The taxonomy as well as recent distribution of most
species is well known due to the studies by the third author (largely
summarized by
Detailed examination of a well-preserved Miocene Helophorus fossil from the collection of the Paleontological Institute in Moscow revealed that it may be reliably assigned to the living species Helophorus sibiricus and represents therefore the oldest record of recent species for hydrophiloid beetles. The results of the studies of this fossil are summarized within this paper and the relevance of the record is discussed.
We would like to dedicate this contribution to Alexandr P. Rasnitsyn on the occasion of his 75th birthday as our thanks for his outstanding contribution to the paleontology and entomology and his massive support of younger generations of entomologists and paleontologists all over the world. We wish him many more scientifically productive years full of good health and cheerfulness!
Geological settingThe geology of the area around Kartashevo village on the right bank of the Irtysh river (56°06'54"N, 74°41'27"E) is rather complex especially in the eroded parts where two formations of different age are in contact: the older Abrosimovka Formation and the overlying Beshcheul Formation.
The Abrosimovka Formation was dated recently by the
comparison of its palynological spectra with the Upper Baygubek
Subhorizon of Aral and the North Ustyurt by
The overlying Beshcheul Formation was dated as middle Miocene by
When describing the fossil flora of the Kartashevo region,
Only one insect species was previously known from the Kartashevo clay stratum – the caddisfly case described as Terrindusia (s.str.) eugeniae Sukatcheva, 1982 (originally assigned to the Abrosimovka Formation by
The fossil specimen presented in this paper was examined in dry condition. Habitus photographs of both fossil and recent specimens were taken using the Canon D-550 digital camera with attached Canon MP-E65mm f/2.8 1–5X macrolens, drawings were traced from the photographs along with a simultaneous check of the fossil specimen. Scaning electron micrographs of fossil as well as recent specimens were prepared using the Hitachi S-3700N environmental electron microscope in the Department of Entomology, National Museum in Prague. Data on the morphology of recent Helophorus sibiricus are based on the specimens deposited at the Department of Entomology, National Museum in Prague, and the Natural History Museum in London.
The Pleistocene records of Helophorus sibiricus from Europe were taken from the BugsCEP database available on-line (
Superfamily Hydrophiloidea
Family Helophoridae
http://species-id.net/wiki/Helophorus_(Gephelophorus)_sibiricus
PIN 3285/5 (piece and counterpiece): Russia, Omsk region, Western Siberia, right bank of Irtysh river at Kartashevo village [56°6'54.11"N, 74°41'27.20"E], leg. E. K. Sychevskaya 1966. Ombinsk Formation, early Miocene, ca. 23–16 million years ago. Deposited in the collection of the A.A. Borissiak Paleontological Institute of the Russian Academy of Sciences, Moscow, Russia.
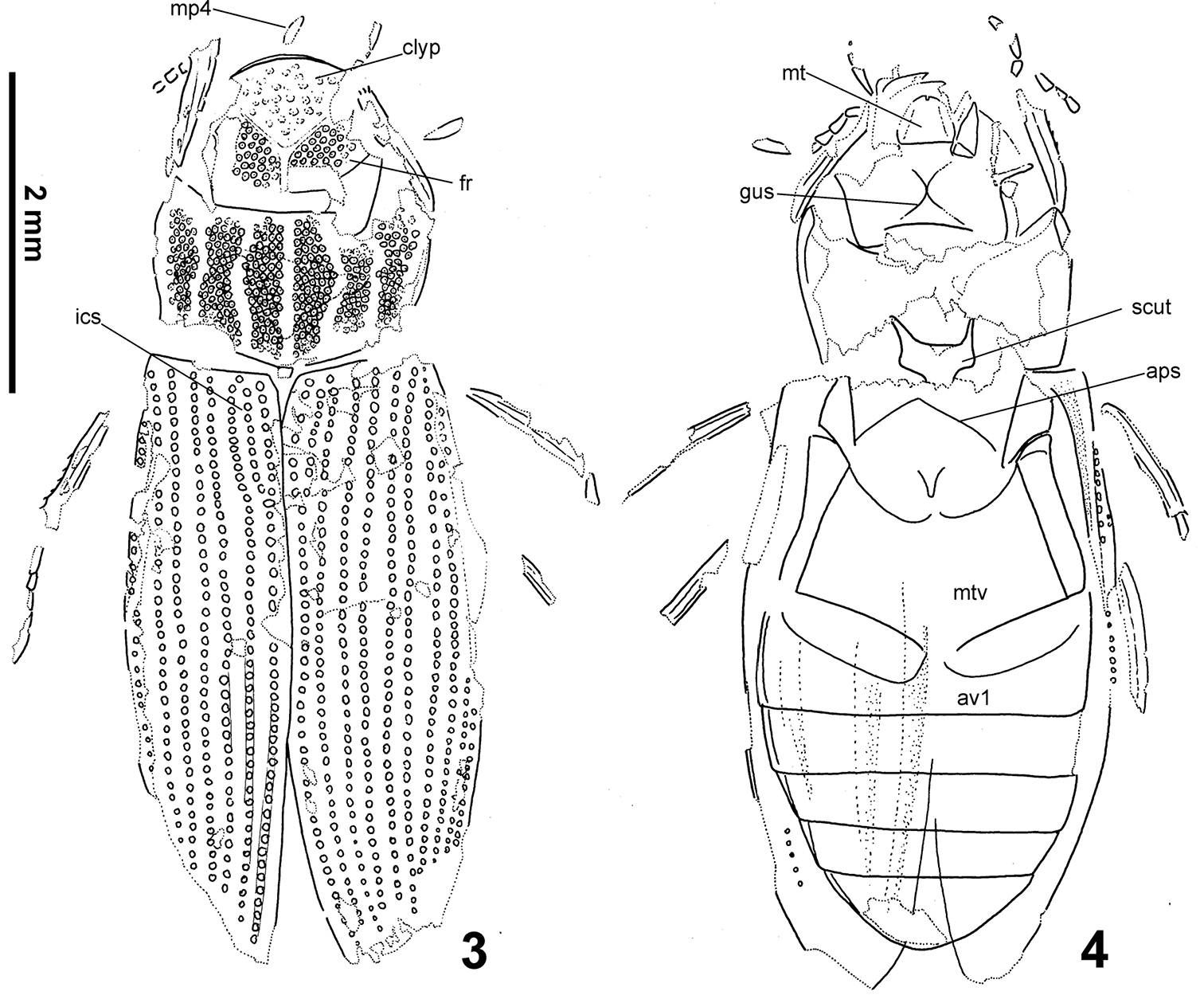
(Figs 1–4, 6, 9). Body length 5.76 mm. Head black, with a deeply impressed Y-shaped frontoclypeal suture, basal portion of the groove wide, slightly widened anteriorly. Clypeus with weak remnants of granules only, frons bearing very distinct large setiferous granules isolated by ca. a half of their diameters. Gula strongly constricted behing tentorial pits, gular sutures meeting at one point. Mentum 1.3× wider than long, bearing a deep median emargination on anterior margin (Fig. 6, see the arrow). Apical segment of maxillary palpi asymmetrical. Pronotum1.85× as wide as long, widest at anterior third bearing five wide longitudinal furrows. Bottom of the grooves without granulation. All intervals bearing a uniform, rather dense granulation, granules rather weakly delimited, nearly contacting each other; granulation becoming sparser sublaterally, consisting of several isolated granules along pronotal margins (Fig. 9). Lateral margin regularly convex, not excised subbasally, lacking any apparent tooth-like projections. Pronotal flanks moderately wide anteriorly, slightly narrowing posteriad. Elytrawith 8 preserved series of large rounded to subquadratic punctures (lateral series not preserved due to deformation of elytra during fossilization). Intervals bearing fine and small irregular series of punctures. Scutellar stria present, very long, consisting of 9 punctures on both elytra. Alternate elytral intervals elevate into low ridges (preserved as elongate ridges and furrows on the ventral imprint of the counter-piece). Epipleuron with rather narrow inner pubescent portion, ca. as wide or slightly narrower than elytral flanks. Mesoventritesubtriangular, anapleural sutures nearly straight. Metaventritewider than long, metanepisternum ca. 5.2× as long as wide, with a transverse ridge anteriorly. Abdomenwith five ventrites, ventrite 5 without median emargination, finelly denticulate on whole posterior margin. Legs rather long and slender, protarsi with five tarsomeres.
Helophorus sibiricus (Motschulsky, 1860), photo of the early Miocene fossil No. PIN 3285/5 from Kartashevo 1 piece 2 counterpiece.
Helophorus sibiricus (Motschulsky, 1860), drawings of the early Miocene fossil No. PIN 3285/5 from Kartashevo 3 piece 4 counterpiece. Abbreviations: mp4 maxillary palpomere 4, clyp clypeus, fr frons, ics intercalary stria, mt mentum, scut mesoscutellum, aps anapleural sutures of mesothorax, mtv metaventrite, av1 abdominal ventrite 1.
Helophorus sibiricus (Motschulsky, 1860) 5 habitus of a recent specimen (Mongolia, Baga-Tenger, coll. National Museum, Prague) 6–7 detail of mentum, the arrow indicates a characteristic emargination on the anterior margin of mentum (6 fossil specimen, SEM micrograph 7 recent specimen, view from inside) 8–9 detailgranulation of head and pronotum, SEM micrographs (8 recent specimen 9 fossil specimen).
The subgenus Gephelophorus Sharp, 1915 to which we assign the fossil is easily recognizable from other Helophorus subgenera by the combination of large body size (4.6–7.0 mm), asymmetrical apical segment of maxillary palpi, elevated alternate elytral intervals, pronotal flanks moderately wide anteriorly and narrowing posteriorly, elytral flanks slightly wider than epipleura. Helophorus sibiricus to which we assign the fossil may be recognized from the only other species of Gephelophorus, Helophorus (Gephelophorus) auriculatus Sharp, 1884, by the continuously curved sides of pronotum not excised behind the anterolateral corners (deeply excised anteriorly and projecting into lateral lobes in Helophorus auriculatus), alternate elytral intervals evenly elevated throughout (bearing only isolated elevate tubercles in Helophorus auriculatus), and completely and densely granulate pronotal intervals (internal and median interval nearly lacking granules in Helophorus auriculatus). Besides, the fossil corresponds with the recent Helophorus sibiricus also in other preserved characters: (1) scutellar stria present and very long [absent in several subgenera, extremelly long especially in Helophorus sibiricus]; (2) mentum 1.3× wider than long [more than 1.5× as wide as long in Rhopalohelophorus Kuwert, 1886, Helophorus s.str., and Lihelophorus Zaitzev, 1908]. The shape of gular sutures is sexually dimorphic in Helophorus sibiricus: the gular sutures are separated throughout in males but meeting in one point in females. Based on this character, we can conclude that the fossil specimen is a female.
(Fig. 10).Helophorus sibiricus
is at present widely distributed throughout the Holarctic region, from
the northern parts of Scandinavia and European Russia through the whole
of Siberia and the Russian Far East to Alaska (
Fossil and recent distribution of Helophorus sibiricus (Motschulsky, 1860). Subfossil Holocene records omitted.
Aquatic species; it is recorded from various
kinds of water bodies predominantly with sandy bottom in southern Yamal
Penninsula (northeastern Siberia) (Prokin et al. 2008);
The genus Helophorus
is currently divided into 11 subgenera whose monophyly and phylogenetic
relationships have not been tested so far. Regardless, its subgroups
differ sharply by their morphological and taxonomic diversity. Most Helophorus
subgenera consist of few species only, each rather distinct
morphologically from the others, and are traditionally considered as
the remnants of ancient divergences (
The presented early Miocene fossil of Helophorus sibiricus is still too young to be taken as a proof of the relic character expected for Gephelophorus
due to its high morphological difference but low species diversity. In
spite of that, it is the first fossil definitely indicating that at
least some small subgenera of Helophorus
contain species of rather ancient origin rather than recently diverged
ones. This seems to contradict the evolutionary scenario proposed for
another small and morphologically distinct subgroup within Helophorus, subgenus Kyphohelophorus Kuwert, 1886, by
The Miocene record of Helophorus sibiricus presented in this paper is not the only pre-Pleistocene record of recent hydrophiloid beetles.
The habitat as well as climatic requirements of the beetle species are usually considered stable over the time (
We are indebted to several colleagues who provided us with their unpublished data and comments relevant to the present paper: S. K. Ryndevich (Baranovichi State University, Belarus) for the notes on the recent distribution of Helophorus sibiricus, E. K. Sychevskaya (Paleontological Institute of the Russian Academy of Sciences, Moscow, Russia) for the details of the collecting circumstances and comments on the recent understanding of the geology of the Kartashevo area, A. G. Ponomarenko, D. E. Shcherbakov and A. P. Rasnitsyn (Paleontological Institute of the Russian Academy of Sciences, Moscow, Russia) for their continuous support of the work by MF and AP on the hydrophiloid fossil record and valuable help with the literature and understanding the geology of the Kartashevo area, and D.V. Vasilenko (Paleontological Institute of the Russian Academy of Sciences, Moscow, Russia) for his help with the literature and valuable discussions on the geology and stratigraphy of Western Siberia.
The study was supported by grant KJB301110901 from the Czech Academy of Sciences (GAAV), grant MK 00002327201 from the Ministry of Culture of the Czech Republic, grant MSM 0021620828 from the Ministry of Education of the Czech Republic, and grant 11-04-90798 from the Russian Foundation for Basic Research.