(C) 2010 Sun Xin. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
In this paper, we describe a new species of the genus Allonychiurus Yoshii, 1995, characterized by the presence of an apical swelling on the fourth antennal segment as well as a combination of chaetotaxic and pseudocellar characters. The genus Allonychiurus is redefined. Four of its species are considered as incertae sedis: Allonychiurus michelbacheri (Bagnall, 1948), Allonychiurus spinosus (Bagnall, 1949), Allonychiurus caprariae (Dallai, 1969) and Allonychiurus sensitivus (Handschin, 1928). The three species Allonychiurus borensis (Beruete, Arbea & Jordana, 1994), Allonychiurus sensilatus (Thibaud & Massoud, 1979) and Allonychiurus vandeli (Cassagnau, 1960) are removed from Allonychiurus and placed in Micronychiurus Bagnall, 1949, Thalassaphorura Bagnall, 1949 and Spinonychiurus Weiner, 1996 respectively. The synonymy of Thibaudichiurus Weiner, 1996 with Allonychiurus is rejected and Allonychiurus foliatus (Rusek, 1967) and Allonychiurus mariangeae (Thibaud & Lee, 1994) are re-allocated to Thibaudichiurus. List and identification key to the world species of the genus are given.
Collembola, Allonychiurus , Micronychiurus , Spinonychiurus , Thalassaphorura , Thibaudichiurus , chaetotaxy, taxonomy, China
The genus Allonychiurus Yoshii, 1995 includes 23 species according to
Type-species: Onychiurus flavescens Kinoshita, 1916: p. 458
DiagnosisOnychiurinae Thalassaphorurini.
Postantennal organ oval, with numerous compound vesicles perpendicular
to the long axis; antennal basis rather well delimited. Clubs of
antenna III organ smooth or granulated. No invaginated apical bulb on
Ant. IV. Labral chaetae formula 4/3, 4, 2; labium of AC type (sensu
The genus Allonychiurus is very similar to Onychiurus
Gervais, 1841 differing from it by a furcal area with 4 small
posterior chaetae arranged in two rows versus arranged in one row. It is
also the only difference between Thalassaphorurini and Onychiurini. The attempt to restrict the genus Allonychiurus to species with 11 distal chaetae on tibiotarsus (
The genus Allonychiurus was described by
A further concern is that furcal area chaetotaxy, i.e. the diagnostic character that
The use of the number of chaetae in the distal whorl of tibiotarsus as a diagnostic character to define Allonychiurus deserves further comments. This character is not mentioned in the published descriptions of Allonychiurus by
In this contribution and as a first step, we address
the taxonomic problems raised above in three ways. First, in order to
accommodate several species that would otherwise necessitate the
creation of new poorly defined genera, we extended the diagnosis of Allonychiurus of
In the checklist given below, an asterisk (*) indicates that species assignment requires confirmation.
Allonychiurus flavescens (Kinoshita, 1916) (type species of the genus Allonychiurus by original designation). Originally described in the genus Onychiurus from Japan, later found in Korean caves (
Allonychiurus donjiensis (Lee & Kim, 1994)*. Described in the genus Onychiurus from South Korea, later placed in Allonychiurus by
Allonychiurus hangchowensis (Stach, 1964). Described in the genus Onychiurus from China (Zhejiang: Hangzhou), later placed in Allonychiurus by
Allonychiurus indicus (Choudhuri & Roy, 1965)*. Described in the genus Onychiurus from India (West Bengale), later placed in Allonychiurus by
Allonychiurus jindoensis (Lee & Kim, 1994)*. Described in the genus Onychiurus from South Korea, later placed in Allonychiurus by
Allonychiurus jongaksanensis (Weiner, 1989). Described in the genus Paronychiurus from North Korea, later placed in Allonychiurus by
Allonychiurus kimi (Lee, 1973). Described in the genus Onychiurus from South Korea, reported from North Korea by
Allonychiurus mediasetus (Lee, 1974). Described as Onychiurus mediaseta from South Korea, reported from North Korea by
Allonychiurus megasomus Sun, Yan & Chen, 2009. Described from China (Nanjing).
Allonychiurus pamirensis (Martynova, 1975)*. Described in the genus Onychiurus from Tajikistan at high altitude (East Pamir), later placed in Allonychiurus by
Allonychiurus pseudocellitriadis (Lee, 1974). Described in the genus Onychiurus from South Korea, later placed in Allonychiurus by
Allonychiurus shanghaiensis (Rusek, 1971)*. Described in the genus Onychiurus from China (Shanghai), later placed in the genus Allonychiurus by
Allonychiurus shinbugensis (Lee, 1974). Described in the genus Onychiurus from South Korea, reported from North Korea by
Allonychiurus tianshanicus (Martynova, 1971)*. Described in the genus Onychiurus from Kyrgyzstan at high altitude, later placed in the genus Allonychiurus by
Four of the species currently placed in the genus Allonychiurus by
Allonychiurus caprariae (Dallai, 1969). Described in the genus Onychiurus , later placed in Allonychiurus by
Allonychiurus michelbacheri (Bagnall, 1948). Described in the genus Onychiuroides Bagnall, 1948from the USA, later placed in the genus Allonychiurus by
Allonychiurus sensitivus (Handschin, 1928). Described in the genus Onychiurus from Bulgaria , later placed in Allonychiurus by
Allonychiurus spinosus (Bagnall, 1949). Described in the genus Onychiuroides from Ireland , later placed in Allonychiurus by
Thalassaphorura sensilata (Thibaud & Massoud, 1979), comb. n.
This species was originally described from Lesser Antilles (Central America) in the genus Protaphorura and later transferred to Allonychiurus by
Micronychiurus borensis (Beruete, Arbea & Jordana, 1994), comb. n.
Described in the genus Onychiurus from Spanish Pyrenees, later placed in Allonychiurus by
Spinonychiurus vandeli (Cassagnau, 1960), comb. n.
Described in the genus Onychiurus from the French Pyrenees at high altitude, later placed in Allonychiurus by
Thibaudichiurus foliatus (Rusek, 1967). Described in the genus Onychiurus from China (Shanghai), reallocated to Thibaudichiurus by
Thibaudichiurus mariangeae (Thibaud & Lee, 1994). Described in the genus Onychiurus from South Korea, given as type species of the genus Thibaudichiurus Weiner, 1996, later placed in Allonychiurus by
Allonychiurus occurs in a wide range of habitats. Most described species live in soil and litter of lowland areas (
Material. The codes between brackets are field codes of the samples which contained the specimens, for instance (C9581).
Material deposit. Nanjing University (China)—NJU, Museum national d’Histoire naturelle de Paris (France)—MNHN.
Morphology. Labial papillae types are named after
Ant antennal segments, AIIIO Sensory organ of third antennal segment, PAO postantennal organ, Th thoracic segments, Abd abdominal segments, p-chaeta chaeta of row p on head, Sp posterior S-chaeta (on Abd. V or on head), ms S-microchaeta (microsensillum auct.), pso pseudocelli, a-pso postero-internal pso on head, psp pseudopore, AS anal spines, x axial psp of Abd. IV.
The uneven axial chaeta m0 (
Labral chaetae formula is the number of chaetae from prelabrals to distal row of labrum; for instance: 4/342.
Pseudocellar and pseudopore formulae are the number of pseudocelli and pseudopores by half-tergite (dorsally) or half-sternite (ventrally) as follows: head anterior, head posterior/Th. I, Th. II, Th. III/Abd. I, Abd. II, Abd. III, Abd. IV, Abd. V (for instance: 32/022/33343).
S-chaetae formula is the number of S-chaetae by half-tergite from head to Abd. VI (for instance: 11/012/222120).
Formula of tibiotarsal chaetotaxy: total number of chaetae (number of chaetae in the distal whorl (A+T), number of chaetae in the proximal whorl B, number of basal chaetae); for instance: 21 (11, 8, 2).
SystematicsOnychiuridae Börner, 1913
Allonychiurus Yoshii, 1995
urn:lsid:zoobank.org:act:3F37A29C-FD87-45DD-94AC-C73FCFCF6A7C
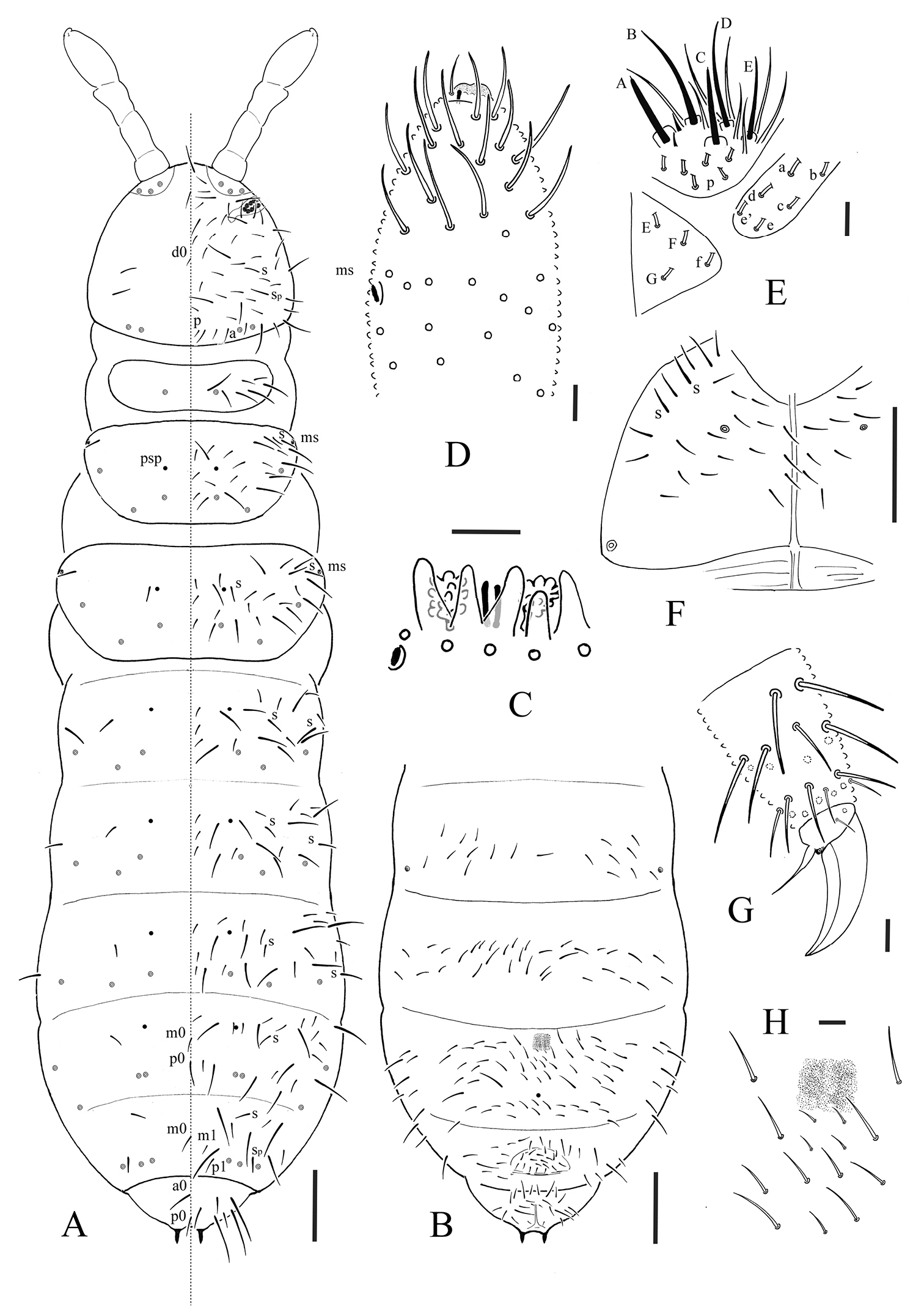
Figs 1–2. Table 1.Holotype female, 3 female paratypes. China: Jiangsu Province: Nanjing: Zijinshan: 10.iv.2009, litter, Berlese extraction, Zhang Feng et al. leg. (C9581). –ibid: Nanjing: Baima Park: 14 paratypes (2 males, 3 females and 9 juveniles) on slides, 13.v. 2007, litter, Berlese extraction, Chen Jian-xiu et al. leg. (C9544).
Holotype and 13 paratypes on slides are deposited in the Department of Biological Science and Technology of NJU, 4 paratypes on slides in MNHN.
pso formula as 32/133/33343 dorsally, 11/000/01000 ventrally; subcoxa 1 of legs I, II and III with 1, 1 and 1 pso respectively; parapseudocelli (psx) absent; presence of small, finely granulated, apical swelling at the apex of Ant. IV; Th. II and III each with 3 dorsal chaetae on both side of axial line; tibiotarsus with 11 chaetae in the distal whorl, no clavate tenent hair; ventral tube with 6+6 distal chaetae, without anterior or basal chaetae.
Body length: 1.3–1.7 mm (females), 1.0 mm (males). Body shape cylindrical, Abd. III–IV more or less broadened. Body colour white in alcohol.
Pseudocellar formulae as 32/133/33343 dorsally, 11/000/01000 ventrally (Figs 1A, B), subcoxa 1 of legs I, II and III with 1, 1 and 1 pso respectively. Parapseudocelli absent. Pseudopore formulae as 00/011/11110 dorsally, 00/111/000x0 ventrally (Figs 1A, B).
S-chaetae formula as 11/012/222120 dorsally. Sp present on head. S-microchaetae tiny and blunt, present on Th. II and III dorsally (Fig. 1A).
Head. Antennae short and distinctly segmented, as long as head (Fig. 1A). Length ratio of antennal segments I: II: III: IV = 1: 1.8–2: 1.8–2: 3.8–4.0. Ant. I with 9–10 chaetae. Ant. II with 14–15 chaetae. Ant. III sensory organ composed of 5 papillae, 5 guard chaetae, 2 small rods and 2 weakly granulated sensory clubs, both morel-like; lateral ms just posterior to sensory organ (Fig. 1C). Ant. IV subapical organite rod-like; basolateral ms at about 2/5 length from base; presence of a small, finely granulated (probably only primary granulation), flat apical swelling at the apex of antenna (possibly remnant of apical bulb fused to the apex) (Fig. 1D); invaginated apical bulb absent. Antennal base with distinct granulation. PAO composed of 18–22 compound vesicles arranged in 2 rows along axis of organ (Fig. 1A). Dorsal cephalic chaeta d0 present. 4+4 p-chaetae between posterior a-pso on head (Fig. 1A). Mandible with strong molar plate and 4 apical teeth. Maxilla bearing 3 teeth and 6 lamellae. Maxillary palp simple with 1 basal chaeta and 2 sublobal hairs. Labral chaetae formula 4/342. Labium with 6 proximal, 4 basomedian (E, F, G, and f) and 6 basolateral (a, b, c, d, e, e’) chaetae; labial papillae of AC type, papillae A–E respectively with 1, 3, 0, 3 and 3 guard chaetae (Fig. 1E). Postlabial chaetae 4+4 along ventral groove (Fig. 1F).
Body chaetotaxy. Ordinary chaetae differentiated in meso- and macro-chaetae, ratio Sp: m1: p1 on Abd. V = 1: 0.6: 1.1 (Fig. 1A). Th. I with 7+7 chaetae dorsally. Three chaetae on both side of axial line and no uneven axial chaetae from Th. II to Abd. III tergites. Abd. IV tergite with two uneven axial chaetae (m0 and p0 ), Abd. V tergite with one uneven axial chaeta (m0), Abd. VI with two uneven axial chaetae (a0 and p0) (Fig. 1A). Th. I, II and III sternites with 0+0, 1+1 and 1+1 chaetae respectively.
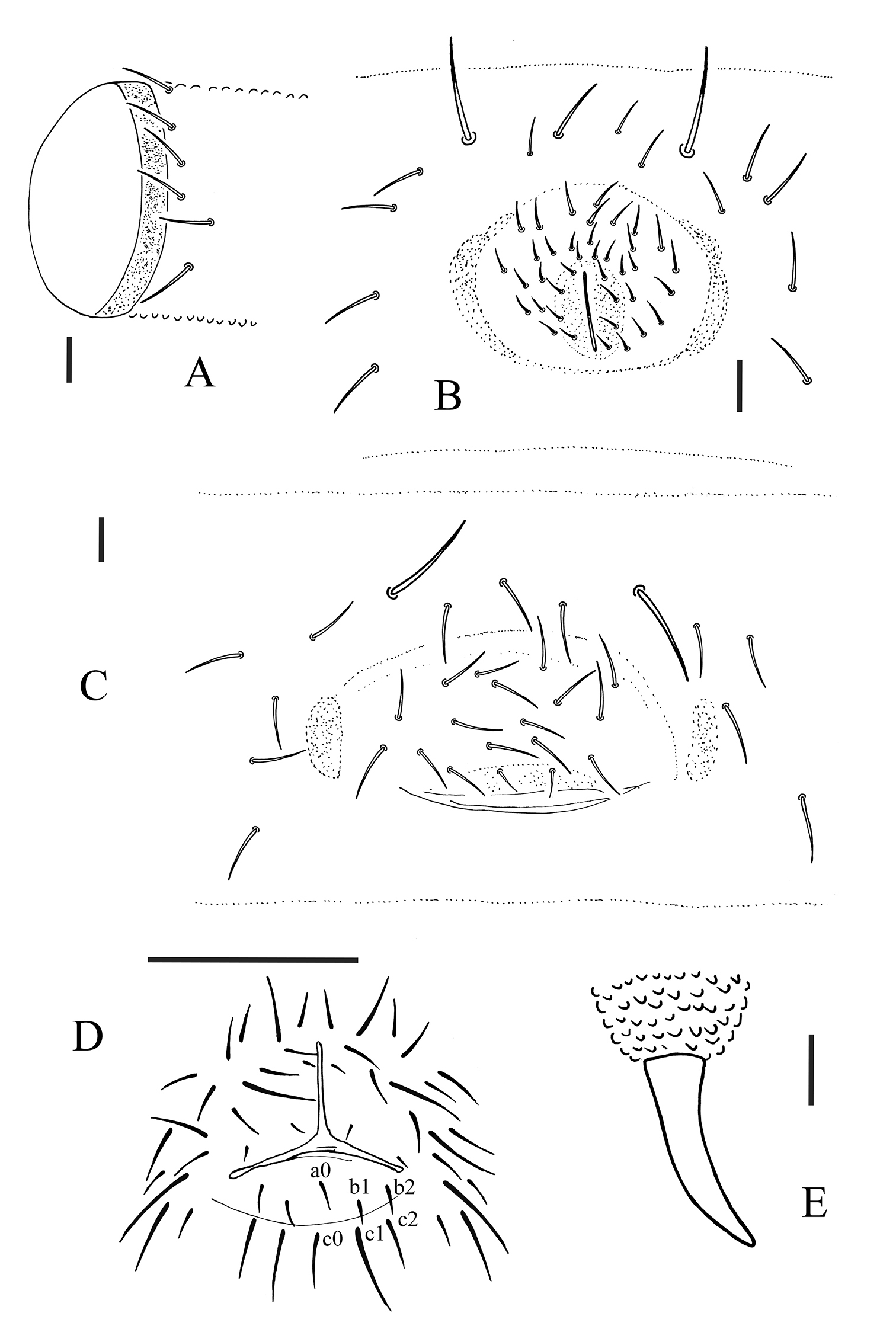
Appendages. Subcoxa 1 of legs I, II and III with 4, 4 and 4 chaetae, subcoxa 2 with 1, 4 and 4 chaetae, respectively. Tibiotarsi of legs I, II and III with 22 (11, 8, 3), 21 (11, 8, 2) and 21 (11, 8, 2) chaetae. Unguis without tooth. Unguiculus slender and pointed, 0.6 times as long as inner edge of unguis, with narrow inner basal lamella (Fig. 1G). Ventral tube with 6+6 distal chaetae, anterior and basal chaetae absent (Fig. 2A). Furca reduced to a finely granulated area, with 4 short chaetae in two rows posterior to furcal rudiment (Fig. 1H).
Male genital plate with 30 circumgenital and 8 genital chaetae (Fig. 2B); female genital plate with 16–18 anterior and 2 genital chaetae (Fig. 2C). No modified chaetae ventrally in males. Anal valves with numerous acuminate chaetae; each lateral valve with chaetae a0 and 2 a1; upper (posterior) valve with chaetae a0, 2 b1, 2 b2, c0, 2 c1, 2 c2 (Fig. 2D). Anal spines set on distinct papillae, 0.6 times as long as inner edge of leg III unguis (Fig. 2E).
Allonychiurus antennalis sp. n. A dorsal side of body with chaetotaxy, S-chaetae, pso and psp B ventral side of Abd. II–VI C organ of Ant. III D dorsal side of left Ant. IV E labium (p, proximal group of chaetae of labial palp) F ventral side of head G distal part of leg III H furcal area. Scales: 0.1 mm (A, B & F), 0.01 mm (C–E & G–H).
Allonychiurus antennalis sp. n. A ventral tube, lateral side B male genital plate C female genital plate D anal valves E anal spine. Scales: 0.1 mm (D), 0.01 mm (A–C & E).
Named for its peculiar antennal morphology.
In broadleaved litter, in a recreational park in town, and at the forested foot of a hill close to the town, altitude 10 to 50 m a.s.l.
Bisexual species. Allonychiurus antennalis sp. n. can be easily recognized by the presence of its apical swelling on Ant. IV, not reported in other species of the genus. It has the same dorsal pseudocellar formula (32/133/33343) as Allonychiurus shinbugensis, Allonychiurus megasomus and Allonychiurus mediasetus. Diagnostic characters are summarized in Table 1.
Comparison of the four species of Allonychiurus with a dorsal pseudocellar formula of 32/133/33343.
| Allonychiurus antennalis sp. n. | Allonychiurus mediasetus (Lee, 1974) | Allonychiurus megasomus Sun et al., 2009 | Allonychiurus shinbugensis (Lee, 1974) | |
|---|---|---|---|---|
| Ant. IV apical swelling | present | not mentioned | absent | not mentioned |
| Ventral pso formula | 11/000/01000 | 11/000/01110 | 11/000/01110 | 10/000/01010 |
| Inner basal lamella of unguiculus | present | present | absent | present |
| Number of chaetae on ventral tube | 6+6 | 8+8 | 8+8 | 8+8 |
| Number of chaetae on Th. I tergite | 7+7 | 9+9 | 7-8+7-8 | 7+7 |
| Number of p-chaetae between posterior a-pso on head | 4+4 | 4+4 | 4+4 | 3+3 |
| Number of axial chaetae on Th. II and III tergites | 3+3 | 4+4 | 4+4 | 4+4 |
| Uneven axial chaetae on Abd. IV | m0 and p0 | p0 | m0 and p0 | m0 and p0 |
| Maximum length (mm) | 1.7 | 1.6 | 2.1 | 1.5 |
Note. Some forms of Allonychiurus flavescens from USA may lack pso on Th. I or are polymorphic (Christiansen and Bellinger, 1998). In the absence of more detailed information regarding other characters, they are not included in this key.
| 1 | AIIIO with 4 papillae, 2+2 or 3+3 pso on Abd IV | 2 |
| – | AIIIO with 5 papillae, more than 3+3 pso on Abd IV | 3 |
| 2(1) | Dorsal pso formula 22/222/22222 after original description | Allonychiurus donjiensis(Lee & Kim)(South Korea) |
| – | Dorsal pso formula 32/233/33333 | Allonychiurus jindoensis (Lee & Kim) (South Korea) |
| 3(1) | Th. I tergite with 2+2 pso | 4 |
| – | Th. I tergite with 1+1 pso or without pso | 5 |
| 4(3) | Th. I tergite with 12+12 chaetae, Abd. I tergite with 4+4 pso, dorsal pso formula 33/233/4444-54 | Allonychiurus pamirensis(Martynova) (Russia) |
| – | Th. I tergite with 8+8 chaetae, Abd. I tergite with 3+3 pso, dorsal pso formula 32/233/34454 after original figure (given as 32/233/34445 in original description), ventral pso formula 1/000/00010, ventral tube with 8+8 distal chaetae | Allonychiurus tianshanicus (Martynova) (Russia) |
| 5(3) | Th. I tergite without pso, dorsal pso formula 32/022/33343, ventral pso formula 11/000/00000, 4+4 p-chaetae between posterior a-pso on head, Th. I tergite with 8+8 chaetae, subcoxa 1 of legs I, II and III with 2, 2 and 2 pso respectively, ventral tube with 8+8 distal chaetae | Allonychiurus pseudocellitriadis (Lee) (South Korea) |
| – | Th. I tergite with 1+1 pso | 6 |
| 6(5) | Th. II and III each with 3+3 pso dorsally; dorsal pso formula 32/133/33343 | 7 |
| – | Number of pso on Th. II and III not as above | 10 |
| 7(6) | Ventral pso formula 11/000/01110, 4+4 p-chaetae between posterior a-pso on head | 8 |
| – | Ventral pso formula not as above | 9 |
| 8(7) | Distal whorl of tibiotarsi with 9 chaetae (interpretation of original drawing), Th. I tergite with 9+9 chaetae, unguiculus with basal lamella, Abd. IV tergite with one uneven axial chaeta (p0) dorsally | Allonychiurus mediasetus (Lee) (South and North Korea) |
| – | Distal whorl of tibiotarsi with 11 chaetae, Th. I tergite with 7-8+7-8 chaetae, unguiculus without basal lamella, Abd. IV tergite with two uneven axial chaetae (m0 and p0) dorsally | Allonychiurus megasomus Sun, Yan & Chen (China) |
| 9(7) | Ventral pso formula 10/000/01010, Ant. IV without apical swelling, 3+3 p-chaetae between posterior a-pso on head, 4+4 axial chaetae on Th. II and Th. III tergites, ventral tube with 8+8 distal chaetae | Allonychiurus shinbugensis (Lee) (South and North Korea) |
| – | Ventral pso formula 11/000/01000, Ant. IV with a flat apical swelling, 4+4 p-chaetae between posterior a-pso on head, 3+3 axial chaetae on Th. II and Th. III tergites, ventral tube with 6+6 distal chaetae | Allonychiurus antennalis sp. n. (China) |
| 10(6) | Th. II and III with 2+2 and 2+2 pso dorsally; dorsal pso formula 32/122/33343 | 11 |
| – | Number of pso on Th. II and III not as above | 13 |
| 11(10) | Unguiculus with basal lamella, Th. I tergite with 9+9 chaetae, Abd. V tergite without uneven axial chaeta dorsally after original drawing | Allonychiurus shanghaiensis (Rusek) (China) |
| – | Unguiculus without basal lamella, Th. I tergite with 8+8 chaetae, Abd. V tergite with one uneven axial chaeta (m0) dorsally | 12 |
| 12(11) | Ventral pso formula 10/000/01000, Abd. IV tergite with two axial chaetae (m0 and p0) dorsally | Allonychiurus kimi (Lee) (South and North Korea) |
| – | Ventral pso formula 10/000/01110, Abd. IV tergite with one axial chaeta (p0) dorsally | A. flavescens (Kinoshita) (Japan, South Korea, USA) |
| 13(10) | Dorsal pso formula 32/123/33343, distal whorl of tibiotarsi with less than 11 chaetae (interpretation of original figure) | Allonychiurus hangchowensis (Stach) (China) |
| – | Dorsal pso formula 32/132/33343, distal whorl of tibiotarsi with 11 chaetae, 4+4 p-chaetae between posterior a-pso on head, 3+3 axial chaetae on Th. II and Th. III tergites | Allonychiurus jongaksanensis (Weiner) (North Korea) |
To the China Scholarship Council (CSC) who gave the grant to the first author, to the Muséum national d’Histoire naturelle of Paris who supported the laboratory work, to the Ministry of Science and Technology of the People’s Republic of China (2006FY120100) who supported the study, to Wanda M. Weiner for invaluable help about the status of Thibaudichiurus.