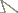(C) 2010 K. D. Prathapan. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
A new species of subaquatic Longitarsus pupating inside the stem aerenchyma of its hydrophyte host plant is described. Eggs are laid on tender leaves and buds and the larvae are open feeders. This is the first report of an Oriental flea beetle pupating inside the stem of its hydrophyte host. A key to the species of southern Indian Longitarsus is provided.
Chrysomelidae, subaquatic, Longitarsus, new species, key, stem aerenchyma pupation, Limnophila
Larvae of flea beetles, in general, are subterranean
root feeders. A few of them mine the leaves or feed exposed on it,
while fruit borers and stem borers are extremely rare (
The cosmopolitan Longitarsus
isthe most speciose genus of flea beetles with about 700 species.
Though about 100 species are known from the Oriental Region, only eight
named species of Longitarsus occur in south India (
Natural populations of the beetle under field conditions were observed during 2009–2011 at Vembayam, Trivandrum, Kerala, India during several visits. The host plant, Limnophila aquatica (Roxb.) Alston was grown in a concrete tank partially filled with soil and water at Vellayani and live beetles were released on to these plants to confirm the biology observed in the field.
Descriptive terminology follows
urn:lsid:zoobank.org:act:A24AE7BC-6B6D-4880-994E-BB8D05667079
Figs 1–8, 12–15♂, with labels as follows: “INDIA Kerala / Vembayam / 12. ix. 2009 Prathapan Coll.” “Longitarsus limnophilae sp. nov. / Prathapan & Viraktamath” “HOLOTYPE [red printed label]” (BMNH).
(30 specimens): 7 ♂, 3 ♀. The same labels as holotype; 5 ♀. same data as for holotype except dating 3.x.2009; 5 ♂. same data except dating 24.x.2009; 9 ♂, 1 ♀. same data except dating 16.i.2010 (5 BMNH, 5 USNM, 5 UASB, 12 NPC, 3 PKDC).
Longitarsus limnophilae sp. n., dorsal habitus
This unique species is named after its host plant. The name is a noun in the genitive case.
Length 1.89 – 2.15 mm; width 0.91 – 1.08 mm; female (2.09 – 2.15 mm) slightly larger than male (1.89 – 2.12 mm). General color brown (Fig. 1). Fore- and middle legs, hind tibia and tarsi light brown. Antenna piceous with proximal three to five antennomeres gradually turning brown. Labrum dark brown to piceous, suture narrowly piceous. Ventrites lighter than dorsum.
Longitarsus limnophilae sp. n. 2 median lobe of aedeagus, ventral view; 3 median lobe of aedeagus, dorsal view; 4 median lobe of aedeagus, lateral view; 5 last abdominal ventrite of male (macerated specimen); 6 spermatheca; 7 tignum; 8 vaginal palpi.
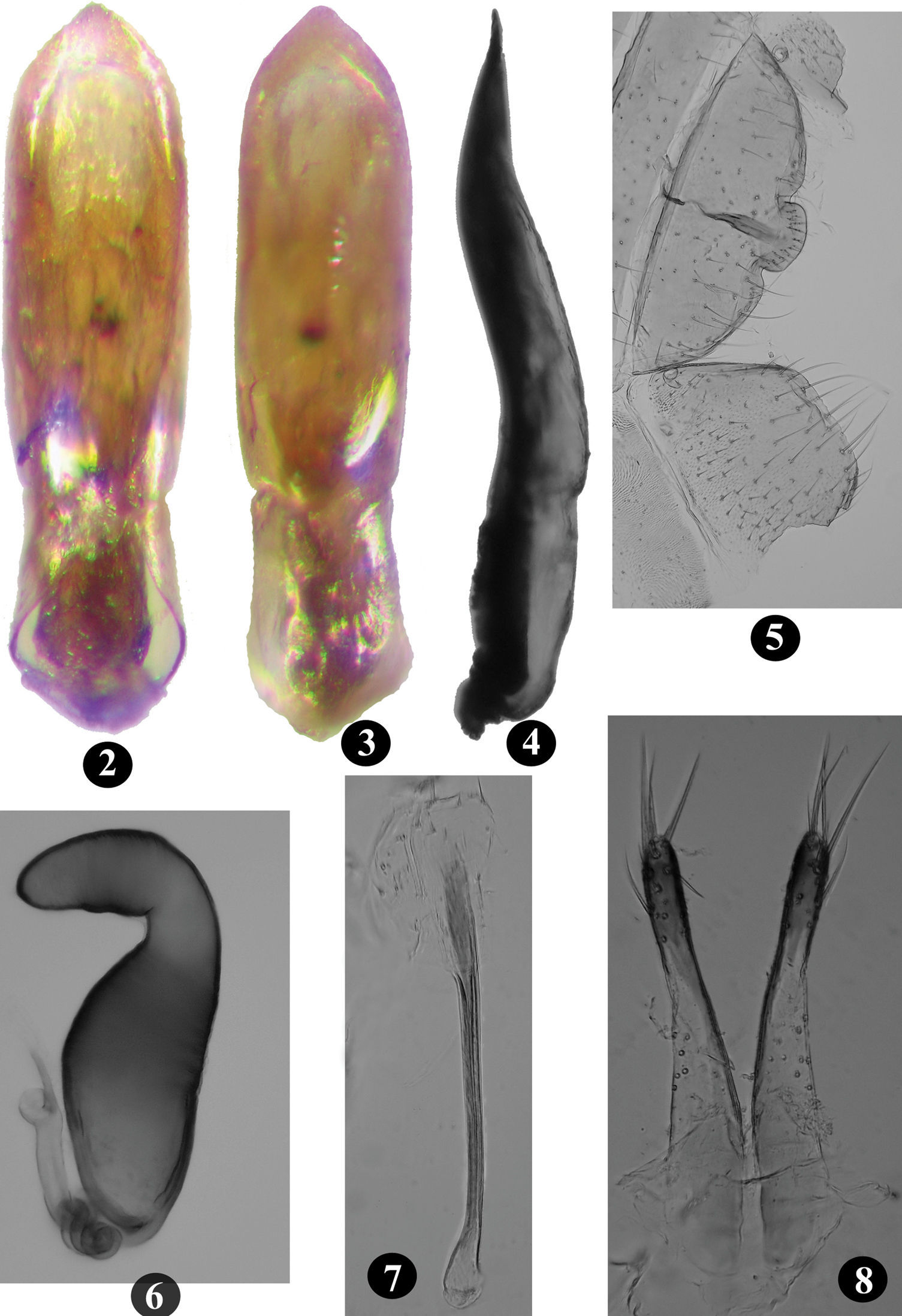
Vertex shiny, impunctate, minutely wrinkled. Ommatidia fully developed. Postcallinal sulcus weak but distinct. Frontal ridge unusual, broad and not sharply raised, anteriorly widening towards frontoclypeal suture, anteriorly forming ill-defined denticle in middle of flat, poorly developed anterofrontal ridge. Maxillary palpus with last palpomere longest. Antenna extends well beyond apex of elytra over pronotum. Second antennomere longer than half of third; second and third together longer than first, subequl to fourth; fifth longer than fourth; fifth to seventh subequal, eighth to tenth progressively shorter than previous antennomere. Pronotum anteriorly wider than posteriorly; 1.27 – 1.34 times wider than long; anterolateral callosity posteriorly lower than anteriorly, not forming denticle at pore; posterolateral callosity protrudes beyond lateral margin; lateral margin weakly curved, anteriorly broader than posteriorly; disc shiny with minute punctures more evident posteriorly. Elytra with well developed humeral calli, punctures distinct, width of interstices smaller than diameter of one puncture in middle of elytron. Elytral apex convex, with one long seta. Hind wings well developed. Scutellum triangular. First male protarsomere 1.60 – 1.67 times longer than wide; first female protarsomere 2.00 – 2.43 times longer than wide. Metatibia strongly curved in dorsal view, slightly curved in lateral view. Number of spinules on dorsolateral margin of metatibia, proximal to row of stiff bristles, vary from seven to ten. In lateral view, first metatarsomere 0.55 – 0.57 times as long as metatibia. Proximal end of first metatarsomere ventrally with thick characteristic patch of short pointed and capitate setae in both sexes. Last male ventrite internally with longitudinal ridge along middle (Fig. 5); posterior margin bisinuate.
Aedeagus in lateral view gently curved, apex acutely pointed and slightly recurved (Fig. 4); ventral side depressed with transparent window, lateral edges raised (Fig. 2); dorsal opening covered with lamina not extending to apex (Fig. 3). Arms of tegmen subequal to or slightly shorter than stem.
Spermatheca with receptacle widest in middle, internal side strongly convex, external side weakly concave; pump with horizontal part longer than vertical; spermathecal duct curved towards receptacle, coiled thrice proximally, not reaching half of receptacle (Fig. 6). Vaginal palpus narrow at distal 1/3, widest at proximal 1/4; distal sclerotization shorter than proximal sclerotization or lateral membranous area (Fig. 8). Tignum nearly straight, anterior sclerotization slightly wider than posterior (Fig. 7).
Longitarsus limnophilae sp. n. 9 habitat; 10 Limnophila aquatica; 11 Longitarsus repens; 12 adult feeding scars on leaf.
Longitarsus limnophilae sp. n. 13 egg; 14 larva; 15, 16 pupa inside stem aerenchyma.
Longitarsus limnophilae can easily be separated from all other south Indian species of Longitarsus by the anteriorly widening, flat frontal ridge (in the other species, the frontal ridge is more or less narrowly raised). Longitarsus belgaumensis Jacoby closely resembles Longitarsus limnophilae in having narrowly piceous elytral suture and dark distal antennomeres. But Longitarsus limnophilae can be separated from Longitarsus belgaumensis based on the antenna extending slightly beyond the apex of elytra over pronotum (in Longitarsus belgaumensis, antenna does not extend beyond the apex of elytra over pronotum), pronotum anteriorly wider than posteriorly (in Longitarsus belgaumensis, pronotum is anteriorly as wide as posteriorly with the maximum width in the middle), structure of frontal ridge (frontal ridge sharply raised along middle in Longitarsus belgaumensis) and genitalia.
Limnophila aquatica (Roxb.) Alston (Scrophulariaceae) (Fig. 10) is a rooted emergent hydrophyte growing in shallow streams, marshes and rice fields (Fig. 9). Species of Limnophila
R. Br., 1810 are widely distributed in the tropics and subtropics of
the Old World and also occur as weeds. Stem aerenchyma in Longitarsus aquatica resembles the same in Longitarsus sessiliflora (Vahl) Blume termed as “wheel-type” by
Eggs (Fig. 13) are laid on tender leaves and buds and the larvae are open feeders (Fig. 14). The closely oriented tender leaves provide sufficient cover for the larva. Mature larva enters the stem aerenchyma in the internode by boring a tiny hole and pupation occurs in it, a little above the entry hole (Figs 15, 16). Adult emerges through an exit hole nearly circular in shape with irregular margin. Adult feeds on both adaxial and abaxial surface of, mostly, tender leaves by scraping, often resulting in holes on the lamina (Fig. 12). Adults when thrown in water floated initially and then swam with raised antennae held back over the sides of the pronotum. After swimming for a while, some performed a short jump on to the shore.
The types were collected from a single locality only. India (Kerala, Vembayam, 8°38'28"N, 76°56'39"E).
This is the first report of an Oriental flea beetle pupating inside the stem of its hydrophyte host.
| 1 | Vertex with conspicuous deep punctures | Longitarsus rangoonensis Jacoby |
| – | Vertex impunctate to minutely punctuate | 2 |
| 2 | Elytra laterally with prominent longitudinal ridge extending backwards from the humerus | Longitarsus liratus Maulik |
| – | Elytra laterally without longitudinal ridge extending from humerus | 3 |
| 3 | Metatibial spur minutely serrulated on either side dorsally | Longitarsus serrulatus Prathapan, Faizal & Anith |
| – | Metatibial spur not serrulated | 4 |
| 4 | Elytral suture narrowly piceous compared to rest of elytra | 5 |
| – | Elytral suture not distinctly darker than rest of elytra | 6 |
| 5 | Frontal ridge broad, widening towards frontoclypeal suture, not sharply raised; antenna extends well beyond apex of elytra over pronotum | Longitarsus limnophilae Prathapan & Viraktamath, sp. n. |
| – | Frontal ridge sharply raised, narrow; antenna hardly reaches apex of elytra over pronotum | Longitarsus belgaumensis Jacoby |
| 6 | Dorsum red | Longitarsus rufipennis Jacoby |
| – | Dorsum yellow brown or dark brown | 7 |
| 7 | Three basal and four apical antennomeres light brown, four intermediate antennomeres dark brown to piceous | Longitarsus gilli Gruev & Askevold |
| – | Middle antennomeres not distinctly darker than distal or basal antennomeres | 8 |
| 8 | Dorsum uniform light brown; hind wings well developed; elytral punctures tend to be regular | Longitarsus sari Maulik |
| – | Elytra dark brown with lighter margins; hind wings absent; punctures confused | Longitarsus fumidus Maulik |
Dr A. K. Pradeep, Calicut University identified the host plants. Drs A. S. Konstantinov, M. Dőberl and P. Jolivet critically reviewed the manuscript. PKD’s work on flea beetles is supported by the Kerala State Council for Science, Technology and Environment, Trivandrum.