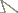(C) 2010 A. Yassin, J.R. David. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
A new classification of the subgenus Zaprionus is proposed in light of recent phylogenetic findings. The boundaries of the armatus and inermis species groups are redefined. The vittiger subgroup is upgraded to the level of a species group. The tuberculatus subgroup is transferred from the armatus to the inermis group. A new monotypic group, neglectus, is erected. Full morphological descriptions of four species belonging to the vittiger group are given: Zaprionus lachaisei sp. n. from Tanzania and Zaprionus santomensis sp. n. from São Tomé and Principé, and two cryptic species of the indianus complex, Zaprionus africanus Yassin & David and Zaprionus gabonicus Yassin & David. Three nominal species are synonymised: Zaprionus beninensis Chassagnard & Tsacas, syn. n. with Zaprionus koroleu Burla, Zaprionus simplex Chassagnard & McEvey, syn. n. with Zaprionus neglectus Collart, and Zaprionus megalorchis Chassagnard & Tsacas, syn. n. with Zaprionus ornatus Séguy. Half of the 46 species of the subgenus are available as laboratory strains and this has allowed full descriptions of the internal structure of their reproductive systems and their immature stages.
classification, reproductive system, immature stages, taxonomy, cryptic species, Tropical Africa
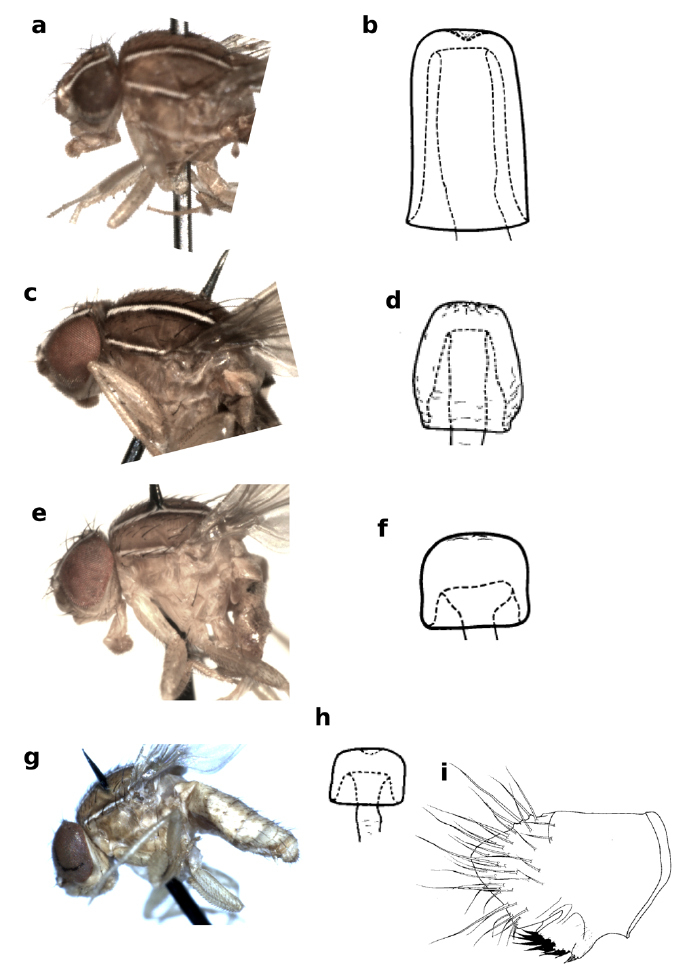
The drosophilid genus Zaprionus Coquillett, 1902 is characterized by the presence of longitudinal white stripes on the frons and the mesonotum (Fig. 1). It is a Paleotropical genus whose species are classified under two subgenera: Zaprionus sensu stricto in the Afrotropical region (48 species), and Anaprionus in the Oriental and Australasian regions (11 species) (
In this paper, we propose a new classification based on recent
phylogenetic findings, describe two new species, and provide a
taxonomic key to all African Zaprionus species. In the early 1990s, several taxonomic keys were published for African Zaprionus (
Frons and mesonotum of Zaprionus (Anaprionus) bogoriensis Mainx, 1954 a, Zaprionus (Zaprionus) ghesquierei Collart, 1937a b, Zaprionus (Zaprionus) litos Chassagnard & McEvey, 1992 c, Zaprionus (Zaprionus) sexstriatus Chassagnard, 1996 d, Zaprionus (Zaprionus) cercus Chassagnard & McEvey, 1992 e, Zaprionus (Zaprionus) kolodkinae Chassagnard & Tsacas, 1987 f, Zaprionus (Zaprionus) verruca Chassagnard & McEvey, 1992 g, Zaprionus (Zaprionus) multivittiger Chassagnard, 1996 h, and Zaprionus (Zaprionus) davidi Chassagnard & Tsacas, 1993 i.
Examined specimens were museum-preserved material or laboratory strains. Laboratory strains in the Laboratoire Evolution, Génomes et Spéciation (LEGS) belonged to 23 species (Table 1), and they were used in describing internal structures of the male and female reproductive systems and immature stages. As shown in Table 1, a congeneric Oriental species, Zaprionus (Anaprionus) bogoriensis Mainx, was added to the analysis.
List of laboratory strains used in studying internal reproductive structures and immature stages.
| Species Founder | females collection data |
|---|---|
| Subgenus Anaprionus | |
| Zaprionus bogoriensis Mainx | India: Bangalore; 2004, J. R. David |
| Subgenus Zaprionus s.s. | |
| Zaprionus africanus Yassin & David | Uganda: Kibale (1100 m); vii.2003, D. Lachaise |
| Zaprionus burlai Yassin | Tanzania: East Usambara Mt, Amani (900 m); 25-ix-2002, D. Lachaise |
| Zaprionus camerounensis Chassagnard & Tsacas | Tanzania: East Usambara Mt, Amani (900 m); 25-ix-2002, D. Lachaise |
| Zaprionus capensis Chassagnard & Tsacas | South Africa: Cape Town; ii.1984, J. R. David |
| Zaprionus cercus Chassagnard & McEvey | Madagascar: Maroantsetra; 18-26.x.1987, S. F. McEvey, J. R. David & S. Aulard |
| Zaprionus davidi Chassagnard & Tsacas | Congo: Brazzaville; iii.2006, J. Vouidibio |
| Zaprionus gabonicus Yassin & David | Gabon: Ogoué-Ivindo, Makoukou (500 m); i.2004, F. Mavoungou |
| Zaprionus ghesquierei Collart | Congo: Brazzaville; iii.2006, J. Vouidibio |
| Zaprionus indianus Gupta | Brazil: Rio de Janeiro, Tijuca; 2001, J. R. David |
| Zaprionus inermis Collart | Uganda: Kibale (1100 m); vii.2003, D. Lachaise |
| Zaprionus kolodkinae Chassagnard & Tsacas | Madagascar: Antananarivo, Tsimbazaza (1200 m); ii.2008, A. Yassin & J. R. David |
| Zaprionus lachaisei sp. n. | Tanzania: East Usambara Mt, Amani (900 m); 25-ix-2002, D. Lachaise |
| Zaprionus mascariensis Tsacas & David | La Reunion (France): 2004, P. Capy |
| Zaprionus neglectus Collart | Madagascar: Andasibe; ii.2008, A. Yassin & J. R. David |
| Zaprionus ornatus Séguy | Congo: Brazzaville; iii.2006, J. Vouidibiou |
| Zaprionus proximus Collart | Kenya: S. Dupas |
| Zaprionus santomensis sp. n. | Sao Tomé & Príncipe: Pico de São Tomé Park (1500 m); iii.2001, D. Lachaise |
| Zaprionus sepsoides Duda | Congo: Brazzaville; iii.2006, J. Vouidibiou |
| Zaprionus taronus Chassagnard & Tsacas | Kenya: S. Dupas |
| Zaprionus tsacasi Yassin | Sao Tomé & Príncipe: Pico de São Tomé Park (1500 m); iii.2001, D. Lachaise |
| Zaprionus tuberculatus Malloch | Congo: Brazzaville; iii.2006, J. Vouidibiou |
| Zaprionus verruca Chassagnard & McEvey | Madagascar: Antananarivo, Tsimbazaza (1200 m); ii.2008, A. Yassin & J. R. David |
| Zaprionus vittiger Coquillett | South Africa: Cape Province, Stellarbush; xii.2006, M. Debiais-Thibaud |
Formal morphological description of the new species followed standard Drosophila terminology and index formulae as in
Morphological structures are abbreviated as: fw = front width; fl = front length; hw = head width; o = maximum diameter of the eye; j = width of gena in line with o; ch = maximum width of gena; or1 = proclinate orbital seta; or2 = anterior reclinate orbital seta; or3 = posterior reclinate orbital seta; oc = ocellar seta; poc = post-ocellar seta; iv = inner vertical seta; ov = outer vertical seta; acs = acrostichal setulae; adc = anterior dorsocentral; pdc = posterior dorsocentral; psc = prescutullar seta; bsc = basal scutellar seta; asc = apical scutellar seta; F1 = forefemur; WL = wing length; Wl = wing width; TL = thorax length; WV = width of white vittae at adc; BV = width of black vittae surrounding WV at adc; A = number of abdominal bristles summed over successive sternites. Measurements on immature stages were taken from uncrowded cultures grown under the same conditions (at 21°C). Measurements are abbreviated as: EL = egg length; El = egg width; PF = length of egg posterior filament; PL = puparium length; Pl = puparium width; H (horn index) = the ratio of the length of the anterior spiracles to the total length of the puparium × 100.
Anatomy of the internal reproductive systemMature, about 10 days old adults were dissected in a Drosophila Ringer solution. For the male reproductive system (see drawings in
| 1 | F1 without a row of spines (Fig. 2a, b) | 2 |
| – | F1 with a row of spines (Fig. 2c–f) | 19 |
| 2(1) | F1 with a protruding tubercule bearing a bristle (Fig. 2b) | 3 |
| – | F1 without a protruding tubercule (Fig. 2a) | 7 |
| 3(2) | Frons without a median white stripe; >A = 46–57; aedeagus subterminally concave (Fig. 3a); spermatheca smooth (Fig. 3c) | Zaprionus mascariensis [Madagascar; Mauritius; Mayotte (France) (loc. n.); La Reunion (France)] |
| – | Frons with a median white stripe; >A = 22–37; aedeagus subterminally convex (Fig. 3e, i); spermatheca rough (Fig. 3g, k) | 4 |
| 4(3) | TST = 1–2 mm; spermatheca very papillate (Fig. 3g); posterior egg filament spatulate (Fig. 3h) | 5 |
| – | TST = 3–5 mm; spermatheca somewhat papillate (Fig. 3k); posterior egg filament not spatulate (Fig. 3l) | 6 |
| 5(4) | >WV = 1.5–1.8 ?m; TST = 2.0 mm | Zaprionus sepsoides [Benin; Cameroon; Cote d’Ivoire; Gabon; Congo; Madagascar; Malawi; South Africa; Uganda] |
| – | >WV = 1.9–2.5 ?m; TST = 1.2 mm | Zaprionus tsacasi [Sao Tome and Principe] |
| 6(4) | TST = 3.2 mm | Zaprionus tuberculatus [Cameroon; Canary Islands (Spain); Cabo Verde; Central African Republic; Chad; Congo; Cote d’Ivoire; Cyprus; Democratic Republic of Congo; Egypt; Gabon; Greece; Kenya; Israel; Madagascar; Malawi; Malta; Mauritius; Mayotte (France) (loc. n.); Mozambique; Niger; Nigeria; La Reunion (France); Zambia; Seychelles; South Africa; St. Helena; Tanzania; Uganda; Zimbabwe] |
| – | TST = 4.4 mm | Zaprionus burlai [Tanzania] |
| 7(2) | Frons without a median stripe | 8 |
| – | Frons with a median stripe | 14 |
| 8(7) | Scutum velvety black, especially posteriorly; scutellum with a white spot at tip (Fig. 1b) | Zaprionus ghesquierei [Benin; Cameroon; Congo; Cote d’Ivoire; Democratic Republic of Congo; Gabon; Kenya; Madagascar; Malawi; Niger; Nigeria; Sao Tome and Principe; Swaziland; Tanzania; Turkey; Uganda; Hawaii Islands (United States of America); Zimbabwe] |
| – | Scutum and scutellum not as above | 9 |
| 9(8) | Scutellum entirely and scutum posteromedially black (Fig. 1c) | Zaprionus litos [Madagascar] |
| – | Scutellum and scutum not as above | 10 |
| 10(9) | Wing darkened anteriorly | 11 |
| – | Wing uniformally hyaline | 12 |
| 11(10) | Thorax and abdomen entirely dark brown (Fig. 4a) | Zaprionus momorticus [Cote d’Ivoire; Democratic Republic of Congo] |
| – | Thorax and abdomen yellow (Fig. 4b) | Zaprionus badyi [Cote d’Ivoire] |
| 12(10) | > basitarsus without a hairy brush (Fig. 5a) | Zaprionus neglectus [Cote d’Ivoire; Democratic Republic of Congo; Madagascar] |
| – | > basitarsus with a hairy brush (Fig. 5b-d) | 13 |
| 13(12) | Thorax yellow; the last 3 abdominal segments shining dark brown (Fig. 4c) | Zaprionus niabu [Cote d’Ivoire] |
| – | Thorax reddish yellow; abdomen shining yellow (Fig. 4d) | Zaprionus arduus [Cote d’Ivoire; Democratic Republic of Congo] |
| 14(7) | Scutum with 6 longitudinal white stripes (Fig. 1d) | 15 |
| – | Scutum with 4 longitudinal white stripes (Fig. 1e) | 16 |
| 15(14) | Aedeagal flap smooth and pointed basally (Fig. 6a) | Zaprionus sexvittatus [Democratic Republic of Congo; Kenya] |
| – | Aedeagal flap finely serrated and truncated basally (Fig. 6b) | Zaprionus sexstriatus [South Africa] |
| 16(14) | Cercus with elongate, ventromedial expansion (Fig. 7a, b) | 17 |
| – | Cercus without ventromedial expansion (Figs 7c, d) | 18 |
| 17(16) | Thorax with a faint median white stripe (Fig. 1e); >WL:TL = 2.02–2.15; abdomen with dark spots at the base of tergal bristles; cercal prominence long and basomedially setulate (Fig. 7b) | Zaprionus cercus [Madagascar] |
| – | Thorax without a faint median white stripe; >WL:TL = 2.25–2.35; abdomen without dark spots at the base of tergal bristles; cercal prominence short and almost entirely setulate along median edge (Fig. 7a) | Zaprionus inermis [Cameroon; Central African Republic; Congo; Cote d’Ivoire; Democratic Republic of Congo; Gabon; Kenya; Uganda] |
| 18(16) | BV = 9–11 ?m (Fig. 1f); testis short; epandrial phragma with a broad hump at the middle of the anterior margin (Fig. 7c); spermatheca smooth | Zaprionus kolodkinae [Madagascar] |
| – | BV = 6–8 ?m (Fig. 1g); testis long; epandrial phragma with a narrow hump at the dorsal quarter of the anterior margin (Fig. 7d); spermatheca papillate; F1 sometimes with a minute tubercule | Zaprionus verruca [Madagascar] |
| 19(1) | F1 with spines not fused with long bristles at their bases (Figs 2c, d, 8) | 20 |
| – | F1 with spines fused with long bristles at their bases (Figs 2e, f) | 33 |
| 20(19) | F1 with 2 spines pointed in opposite orientation (Fig. 2c) | 21 |
| – | F1 with more than 2 spines usually pointed to the same direction (Fig. 2d) | 22 |
| 21(20) | F1 small (Figs 2c, 8a); abdomen with dark spots at base of bristles | Zaprionus campestris [Cameroon; Cote d’Ivoire; Madagascar, Sao Tome and Principe] |
| – | F1 large (Fig. 8b); abdomen without dark spots at base of bristles | Zaprionus montanus [Burundi; Cote d’Ivoire; Democratic Republic of Congo; Kenya; Rwanda; South Africa] |
| 22(20) | > basitarsus without a hairy brush | 23 |
| – | > basitarsus with a hairy brush | 24 |
| 23(22) | F1 with 3–4 spines; basalmost spine strong (Fig. 8c) | Zaprionus spinosus [Cameroon; Cote d’Ivoire; Democratic Republic of Congo] |
| – | F1 with 5 spines internally and sometimes 2 spines externally (Fig. 8d) | Zaprionus spineus [Democratic Republic of Congo] |
| 24(22) | F1 spines differentiated; basalmost spine strong (Figs 2d, 8e) | Zaprionus serratus [Cameroon; Democratic Republic of Congo; Uganda] |
| – | F1 spines undifferentiated (Fig. 8f–n) | 25 |
| 25(24) | Wing anterior margin black or darkened (Fig. 9a–c) | 26 |
| – | Wing hyaline (Fig. 9d) | 29 |
| 26(25) | Wing anterior margin black (Fig. 9a, b); F1 spines fine (Fig. 8f, g) | 27 |
| – | Wing anterior margin darkened (Fig. 9c); F1 spines robust (Fig. 8h, i) | 28 |
| 27(26) | F1 with 2–3 spines (Fig. 8g) | Zaprionus fumipennis [Congo; Cote d’Ivoire; Democratic Republic of Congo; Kenya] |
| – | F1 with 5–6 spines (Fig. 8f) | Zaprionus vrydaghi [Congo; Cote d’Ivoire; Democratic Republic of Congo; Gabon; Tanzania; Uganda] |
| 28(26) | F1 middle bristle borne on a tubercule (Fig. 8h) | Zaprionus tuberarmatus [Cameroon; Democratic Republic of Congo] |
| – | F1 middle bristle not borne on a tubercule (Fig. 8i) | Zaprionus hoplophorus [Cameroon; Congo] |
| 29(25) | Aedeagal flap absent (Fig. 6c, d) | 30 |
| – | Aedeagal flap present | 31 |
| 30(29) | F1 with a hairy tuft proximally (Fig. 8j); aedeagus short and robust | Zaprionus armatus [Democratic Republic of Congo] |
| – | F1 without a hairy tuft proximally (Fig. 8k); aedeagus very long and slender | Zaprionus enoplomerus [Cameroon; Cote d’Ivoire] |
| 31(29) | F1 middle bristle borne on a minute tubercule (Fig. 8l); spermatheca voluminous, sclerified at apex and with deep apical introvert (Fig. 6e) | Zaprionus spinipes [Cameroon] |
| – | F1 middle bristle not borne on a tubercule (Fig. 8m, n); spermatheca not as above | 32 |
| 32(31) | F1 not broadened, with a series of short bristles (Fig. 8m); spermatheca sclerified (Fig. 6f) | Zaprionus seguyi [Cameroon; Congo; Democratic Republic of Congo] |
| – | F1 broadened, with a few long bristles (Fig. 8n); spermatheca smooth (Fig. 6g) | Zaprionus spinormatus [Cameroon; Cote d’Ivoire; Nigeria] |
| 33(19) | WV < 15 ?m; thorax and abdomen blackish brown | Zaprionus camerounensis [Cameroon; Malawi; Tanzania (loc. n.); Uganda] |
| – | WV > 15 ?m; thorax and abdomen not black | 34 |
| 34(33) | Abdominal tergal bristles with dark spots basally | 35 |
| – | Abdominal tergal bristles without dark spots basally | 43 |
| 35(34) | Thorax with two incomplete submedian white stripes between two complete dorsocentral stripes (Fig. 1h) | Zaprionus multivittiger [Kenya; Rwanda] |
| – | Thorax without submedian stripes | 36 |
| 36(35) | F1 setiferous spines differentiated; basalmost borne on a protruding tubercule (Fig. 2e) | Zaprionus proximus [Kenya; Uganda] |
| – | F1 setiferous spines undifferentiated | 37 |
| 37(36) | BV enlarged posteriorly; abdomen dark brown (Fig. 10b, d, e) | 38 |
| – | BV not enlarged posteriorly; abdomen light yellow | 40 |
| 38(37) | Abdomen darker than thorax (Fig. 10b) | Zaprionus koroleu [Benin; Cote d’Ivoire] |
| – | Abdomen and thorax concolorous (Fig. 10d) | 39 |
| 39(38) | First and second tarsomeres of the foreleg with strong black spines (Fig. 5c); >TL = 1.62–1.68 mm (Fig. 10e); H = 5.2 (Fig. 11d) | Zaprionus lachaisei sp. n. [Tanzania] |
| – | First and second tarsomeres of the foreleg without strong black spines; >TL = 1.44–1.56 mm (Fig. 10d); H = 9.6 (Fig. 11e) | Zaprionus vittiger [Cameroon; Ethiopia; Madagascar; Malawi; South Africa] |
| 40(37) | Head orange tan lighter than thorax (Fig. 10f); hairy brush 1/3 > basitarsus (Fig. 5e); spermatheca without introvert (Fig. 13d) | Zaprionus santomensis sp. n. [Sao Tome and Principe] |
| – | Head and thorax concolorous reddish brown; hairy brush 2/3 > basitarsus; spermatheca with an introvert (Fig. 12) | 41 |
| 41(40) | > aedeagal flap highly serrated apically (Fig. 12a) ; oviscape constricted basally with 8 (rarely 7) peg-like ovisensilla (Fig. 12b); spermatheca length:width = 0.62–0.84 (Fig. 12c) | Zaprionus africanus [Gabon ; Uganda] |
| – | > aedeagal flap highly smooth apically (Fig. 12d, g); oviscape with 6 peg-like ovisensilla (Fig. 12e, h); spermatheca length:width = 0.95–1.16 (Fig. 12f, i) | 42 |
| 42(41) | > aedeagal flap smooth basally (Fig. 12d) | Zaprionus gabonicus [Gabon] |
| – | > aedeagal flap serrated basally (Fig. 12g) | Zaprionus indianus [Argentina; Austria; Benin; Brazil; Cabo Verde; Canary Islands (Spain); Congo; Cote d’Ivoire; Egypt; India; Iran; Israel; Italy; Kenya; Madagascar; Madeira (Portugal); Malawi; Mauritius; Morocco (loc. n.); Mozambique; Niger; Nigeria; Panama; La Reunion (France); Sao Tome and Principe; Saudi Arabia; Seychelles; South Africa; Tanzania; United States of America; Uruguay] |
| 43(34) | Abdomen yellow with brown posterior fine stripes on tergites II to IV; TST > 12.0 mm; spermatheca elongated (Fig. 14a) | Zaprionus ornatus [Cameroon; Cote d’Ivoire; Congo; Gabon; Madagascar; South Africa] |
| – | Abdomen uniformally yellow; TST < 6.0 mm; spermatheca globulous (Figs 14b, c, e) | 44 |
| 44(43) | TST = 2.6 mm; spermatheca chitinized at base and apex (Fig. 14b); egg with 2 filaments | Zaprionus davidi [Congo; Sao Tome and Principe (loc. n.)] |
| – | TST = 4.0–5.2 mm; egg with 4 filaments | 45 |
| 45(44) | >TL = 1.56–1.70 mm; > epandrium not expanded dorsally (Fig. 14d); spermatheca (Fig. 14c) | Zaprionus taronus [Congo (loc. n.); Gabon; Kenya; Malawi; Sao Tome and Principe (loc. n.)] |
| – | >TL = 1.44–1.50 mm; > epandrium expanded dorsally (Fig. 14e); spermatheca (Fig. 14f) | Zaprionus capensis [South Africa] |
Table 2 also shows the breeding niche and the possibility to rear in the
laboratory for some species. These two attributes are interrelated, as
generalist fruit-breeding species are usually those that can be reared
with ease on standard Drosophila medium.
Forefemur of Zaprionus cercus Chassagnard & McEvey, 1992 a, Zaprionus mascariensis Tsacas & David, 1975 b, Zaprionus campestris Chassagnard, 1989 c, Zaprionus serratus Chassagnard, 1989 d, Zaprionus proximus Collart, 1937 e, and Zaprionus indianus Gupta, 1970 f.
Classification and ecology of the subgenus Zaprionus. Breeding niches are abbreviated as: FL = flowers; FR = fruits; and TR = decaying tree trunk. Ability to be reared in the laboratory (L) is indicated as (+) for species that are reared and (-) for species that are not.
| Group | Subgroup | Complex | Species | Authorship | L | Breeding niche | Reference |
|---|---|---|---|---|---|---|---|
| armatus | armatus | armatus | armatus | Collart, 1937a | - | TR: Ficus sp. (Moraceae) | C37a |
| FR: Myrianthus sp. (Cercopiaceae) | TC90 | ||||||
| enoplomerus | Chassagnard, 1989 | + | FR: Ficus macrosperma (Moraceae); Ficus sur (Moraceae); Ficus capensis (Moraceae) | C89 | |||
| seguyi | Tsacas & Chassagnard, 1990 | - | FR | TC90 | |||
| spinipes | Tsacas & Chassagnard, 1990 | - | ? | TC90 | |||
| spinoarmatus | Tsacas & Chassagnard, 1990 | - | FR: Dacryodes sp. (Burseraceae) | TC90 | |||
| TR: Raphia sp. (Arecaceae) | B54 | ||||||
| hoplophorus | hoplophorus | Tsacas & Chassagnard, 1990 | - | ? | |||
| tuberarmatus | Tsacas & Chassagnard, 1990 | - | ? | ||||
| vrydaghi | fumipennis | Séguy, 1938 | - | FL: Costus afer (Costaceae) | TC90 | ||
| vrydaghi | Collart, 1937a | - | FL: Costus afer (Costaceae) | B76, C86 | |||
| montanus | campestris | Chassagnard, 1989 | - | ? | C89 | ||
| montanus | Collart, 1937b | - | TR: Andropoganeae (Poaceae) | G57 | |||
| TR: Bambuseae (Poaceae) | C89 | ||||||
| spinosus | serratus | Chassagnard, 1989 | - | FL: Bignoniaceae | C89 | ||
| spineus | Tsacas & Chassagnard | - | ? | TC90 | |||
| spinosus | Collart, 1937a | - | ? | ||||
| inermis | arduus | Collart, 1937b | - | FR: Musa sp. (Musaceae) | C37b | ||
| badyi | Burla, 1954 | - | FR | B54 | |||
| ghesquierei | Collart, 1937a | - | FR: Citrus sinensis (Rutaceae); Coffea sp. (Rubiaceae); Cola acuminata (Malvaceae); Rollinia sieberi (Annonaceae); Sarcocephalus sp. (Rubiaceae); Psidium sp. (Myrtaceae); Terminalia sp. (Combredaceae); Murraya exotica (Rutaceae); Pseudospondia sp. (Anacridaceae); Myrianthus sp. (Cercopiaceae); Dorstenia sp. (Moraceae); Uapaca sp. (Phyllanthaceae) | C37a | |||
| FR: Musa sp. (Musaceae), Averrhoa carambola (Oxalidaceae); Turraeanthus africana (Meliaceae); Conopharyngia dusissima (Apocynaceae), | B54 | ||||||
| FR: Mangifera indica (Anacardiaceae); Carica papaya (Caricaceae); Persea americana (Lauraceae); Ficus ovata (Moraceae); Musa sp. (Musaceae); Averrhoa carambola (Oxalidaceae); Cyphonandra betacea (Solanaceae); Solanum gilo (Solanaceae) | B76 | ||||||
| FR: Polyalthia sauveolens (Annonaceae); Detarium senegalense (Caesalpiniaceae) | LT83 | ||||||
| FR: Dacryodes sp. (Burseraceae); Hugonia sp. (Linaceae); Parinari sp. (Rosaceae); Gambeya perpulchra (Sapotaceae) | L79 | ||||||
| FR: Cocos romanzoffiana (Palmaceae) | L47 | ||||||
| FR: Pancovia bijuga (Sapindaceae) | L74 | ||||||
| TR: Elaeis guinensis (Palmaceae) | L47 | ||||||
| FR: Ficus thonningii (Moraceae) | C97 | ||||||
| momorticus | Graber, 1957 | - | FL: Momordica pterocarpa (syn. M. runsorrica) (Cucurbitaceae) | G57 | |||
| FL: Crinum sanderianum (Amaryllidaceae); Crinum jagus (Amaryllidaceae) | L79 | ||||||
| FL: Rothmania whitfieldi (Rubiacea) | L74 | ||||||
| niabu | Burla, 1954 | - | FR: Carica papaya (Caricaceae) | B54 | |||
| inermis | cercus | Chassagnard & McEvey | + | FR: ex-banana trap | CM92 | ||
| inermis | Collart, 1937a | + | FR: Eugenia malaccensis (Myrtaceae) | C37a | |||
| FR: Citrus sp. (Rutaceae); Carica papaya (Caricaceae); | B54 | ||||||
| TR: Raphia sp. (Arecaceae) | B54 | ||||||
| FR: Musa sapientum (Musaceae) | LT83 | ||||||
| TR: Elaeis guineensis (Arecaceae) | LT83 | ||||||
| tuberculatus | kolodkinae | Chassagnard & Tsacas, 1987 | + | FR: ex-banana trap | CT87 | ||
| mascariensis | Tsacas & David, 1975 | + | FR: ex-banana trap | TD75 | |||
| sepsoides | sepsoides | Duda, 1939 | + | FR: Dacryodes sp. (Burseraceae); Hugonia sp. (Linaceae); Guarea cedrata (Meliaceae); Turraeanthus africanus (Meliaceae); Parinari sp. (Chrysobalanaceae) | L79 | ||
| FR: Ficus sur (Moraceae); Ficus lyrata (Moraceae); Ficus macrosperma (Moraceae); Ficus elasticoides (Moraceae); Ficus ovata (Moraceae); Ficus sp. (Moraceae) | L82 | ||||||
| FR: Pandanus candelabrum (Pandanaceae) | R83 | ||||||
| FR: Spondias mombin (Anacardiaceae); Detarium senegalense (Cesalpinaceae); Pentadesma butyraceae (Guttifereae); Treculia africana (Moraceae); Hirtella sp. (Rosaceae); Parinari excelsa (Rosaceae); Nauclea poheguinii (Rubiaceae); Gambeya taiensis (Sapotaceae); Tieghemella heckelii (Sapotaceae) | C86 | ||||||
| tsacasi | Yassin, 2008 | + | FR: ex-banana trap | Y08 | |||
| tuberculatus | burlai | Yassin, 2008 | + | FR: ex-banana trap | Y08 | ||
| tuberculatus | Malloch, 1932 | + | FR: Santiria trimera (Burseraceae); Dacryodes sp. (Burseraceae); Guarea cedrata (Meliaceae); Parinari sp. (Rosaceae); Parinari excelsa (Rosaceae); Tieghemella heckelii (Sapotaceae) | L79 | |||
| FR: Ficus sur (Moraceae); Ficus saussureana (Moraceae); Ficus mucuso (Moraceae); Ficus lutea (Moraceae); Ficus natalensis (Moraceae); | L82, LT83 | ||||||
| FR: Spondias mombin (Anacardiaceae); Detarium senegalense (Cesalpinaceae); Artocarpus sp. (Moraceae); Hirtella sp. (Rosaceae); Uncaria sp. (Rubiaceae); Gambeya taiensis (Sapotaceae) | C86 | ||||||
| verruca | Chassagnard & McEvey, 1992 | + | FR: ex-banana trap | CM92 | |||
| neglectus | neglectus | Collart, 1937 | + | FR | B54 | ||
| FL: Ipomoea digitata (Convolvulaceae) | B54 | ||||||
| FL: Crinum jagus (Amaryllidaceae); Pentadesma butyracea (Guttiferae); Rothmania whifieldi (Rubiaceae) | C86 | ||||||
| FR: Ficus ovata (Moraceae) | L79 | ||||||
| FR: Treculia africana (Moraceae) | C86 | ||||||
| vittiger | davidi | davidi | Chassagnard & Tsacas, 1993 | + | FR: ex-banana trap | CT93 | |
| taronus | Chassagnard & Tsacas, 1993 | + | FR: Polyalthia sauveolens (Annonaceae); Staudtia gabonensis (Myristicaceae); Cissus dinklagei (Vitaceae) | LT83 | |||
| indianus | africanus | Yassin & David, 2008 | + | FR: ex-banana trap | Y08b | ||
| gabonicus | Yassin & David, 2008 | + | FR: ex-banana trap | Y08b | |||
| indianus | Gupta, 1970 | + | FR: ex-banana trap | Y08b | |||
| FR: date palm, guava and citrus | Y09 | ||||||
| ornatus | litos | Chassagnard & McEvey, 1992 | - | ? | CM92 | ||
| ornatus | Séguy, 1933 | + | FR: Averrhoa carambola (Oxalidaceae) | B54 | |||
| FR: Spondias mombin (Anacardiaceae); Gambeya taiensis (Sapotaceae) | C86 | ||||||
| FR: Ficus sur (Moraceae) | L76 | ||||||
| FR: Ficus macrosperma (Moraceae); Ficus saussureana (Moraceae); Ficus elasticoides (Moraceae); Ficus vogeliana (Moraceae); Ficus mucuso (Moraceae); Ficus ovata (Moraceae); Ficus lutea (Moraceae); Ficus thoningii (Moraceae) | L82, LT83 | ||||||
| FL: Rothmania whitfieldii (Rubiaceae) | L74 | ||||||
| proximus | capensis | Chassagnard & Tsacas, 1993 | + | FR: ex-banana trap | CT93 | ||
| proximus | Collart, 1937b | + | FR: Cussonia sp. (Araliaceae) | C37b | |||
| sexvittatus | multivittiger | Chassagnard, 1996 | + | FR: Rhamnus prinoides (Rhamnaceae) | C96 | ||
| sexstriatus | Chassagnard, 1996 | + | FR? | ||||
| sexvittatus | Collart, 1937c | - | FR: Acokanthera sp. (Apocynaceae) | C37c | |||
| FR: Juniperus procera (Cupressaceae) | C96 | ||||||
| vittiger | camerounensis | Chassagnard & Tsacas, 1993 | + | FR: ex-banana trap | CT93 | ||
| koroleu | Burla, 1954 | + | FR: ex-banana trap | ||||
| TR: Raphia sp. (Arecaceae) | B54 | ||||||
| lachaisei | sp. n. | + | FR: ex-banana trap | ||||
| santomensis | sp. n. | + | FR: ex-banana trap | ||||
| vittiger | Coquillett, 1902 | + | FR: ex-banana trap | YP | |||
It is still difficult to estimate with certainty the niches for some of the problematic species in
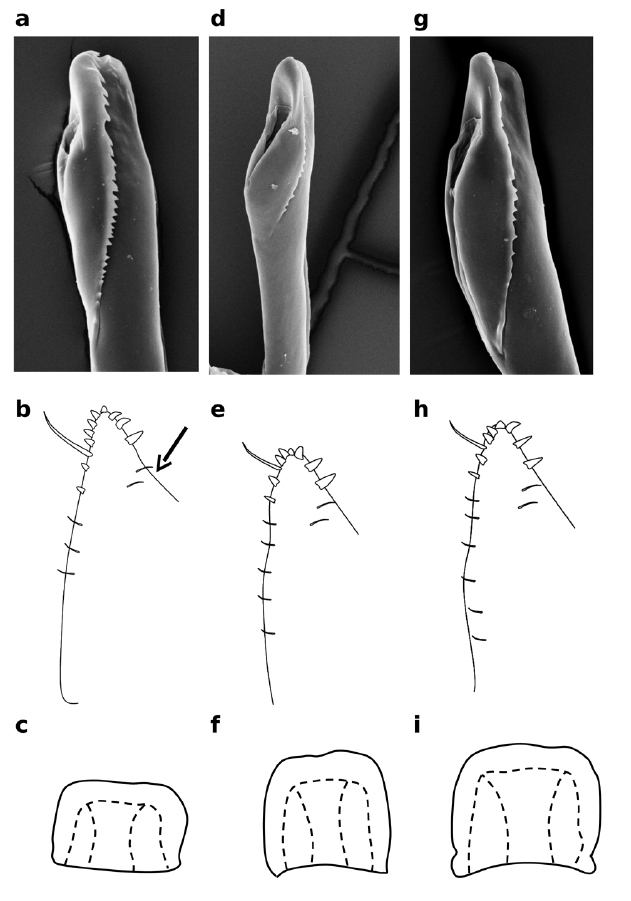
Distiphallus, testis and accessory gland, spermatheca and egg of Zaprionus mascariensis Tsacas & David a–d, Zaprionus sepsoides Duda, 1939 e–h, and Zaprionus tuberculatus Malloch, 1932 i–l [From
The armatus group was initially erected to include three subgroups: armatus, tuberculatus and vittiger (
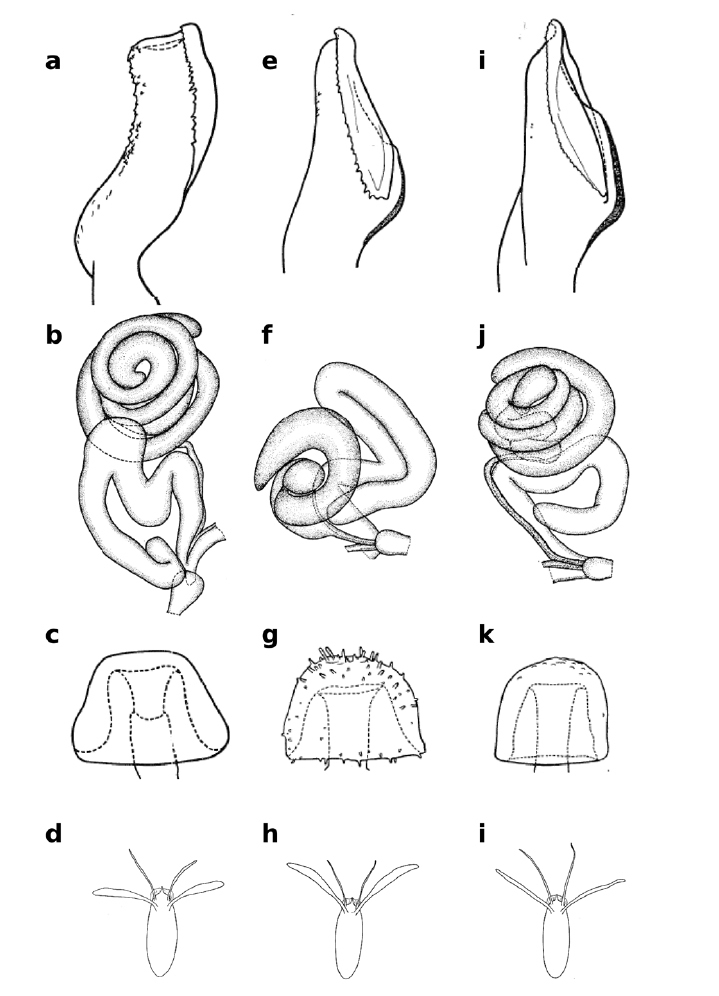
Dorsal views of Zaprionus momorticus Graber, 1957 a, Zaprionus badyi Burla, 1954 b, abdomen of Zaprionus niabu Burla, 1954 c, and lateral view of Zaprionus arduus Collart, 1937 d.
The inermis group comprises species with spineless F1 (Figs 2a, b). The F1 spinelessness is also found in the Oriental subgenus Anaprionus, suggesting a plesiomorphy, and the monophyly of this group was questionable (
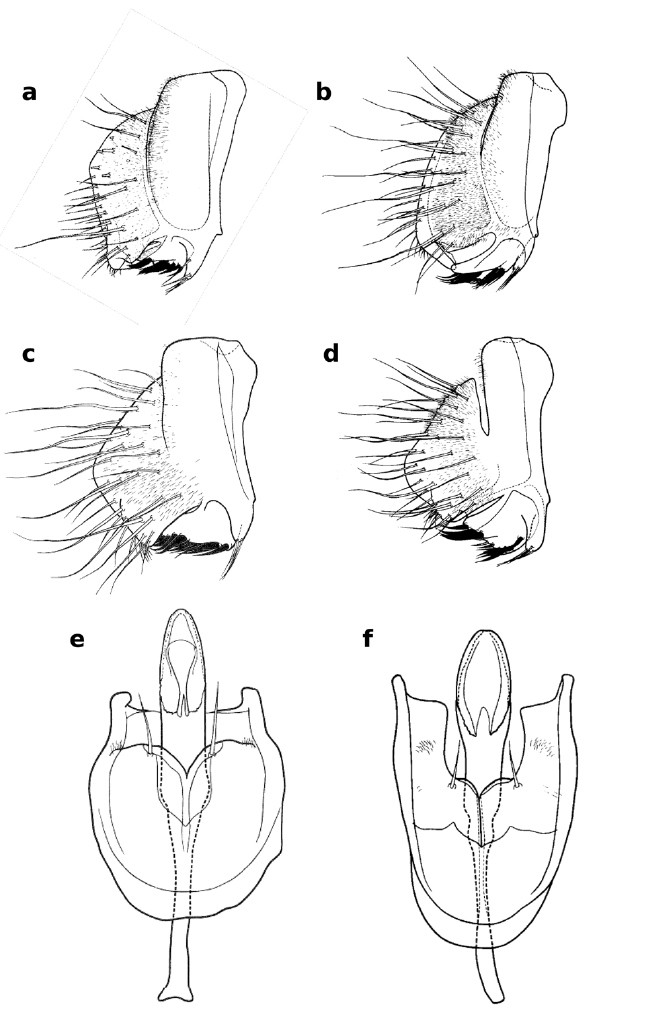
Tarsomeres of male foreleg of Zaprionus neglectus Collart, 1937 a, Zaprionus kololdkinae Chassagnard & Tsacas, 1987 b, Zaprionus lachaisei Yassin & David, sp. n. c, Zaprionus taronus Chassagnard & Tsacas, 1993 d, and Zaprionus santomensis Yassin & David, sp. n. e.
Ventral views of distiphallus of Zaprionus sexvittatus Collart, 1937 a, Zaprionus sexstriatus Chassagnard, 1996 b, Zaprionus armatus Collart, 1937 c, and Zaprionus enoplomerus Chassagnard, 1989 d; spermatheca of Zaprionus spinipes Tsacas & Chassagnard, 1990 e, Zaprionus seguyi Tsacas & Chassagnard, 1990 f, and Zaprionus serratus Chassagnard, 1989 g, [From
Zaprionus neglectus is a continental species lacking F1 ornamentation and the hairy brush on F1 basitarsus in males (
We have recently collected a strain of Zaprionus simplex from Crinum sp. in Madagascar and reared it in the laboratory.
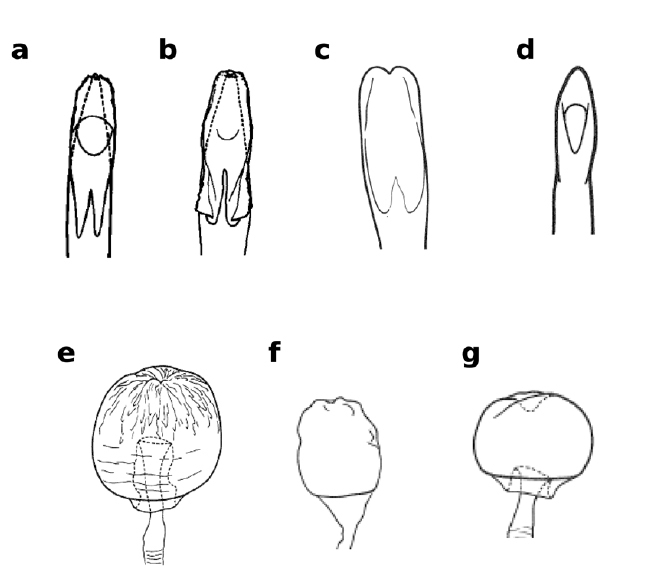
Lateral views of male epandrium and cercus and ventral views of aedeagus and hypandrium of Zaprionus inermis Collart, 1937 a, Zaprionus cercus Chassagnard & McEvey, 1992 b, Zaprionus kolodkinae Chassagnard & Tsacas, 1987 c, e, and Zaprionus verruca Chassagnard & McEvey, 1992 d, f [From Chassagnard and Tsacas 1987;
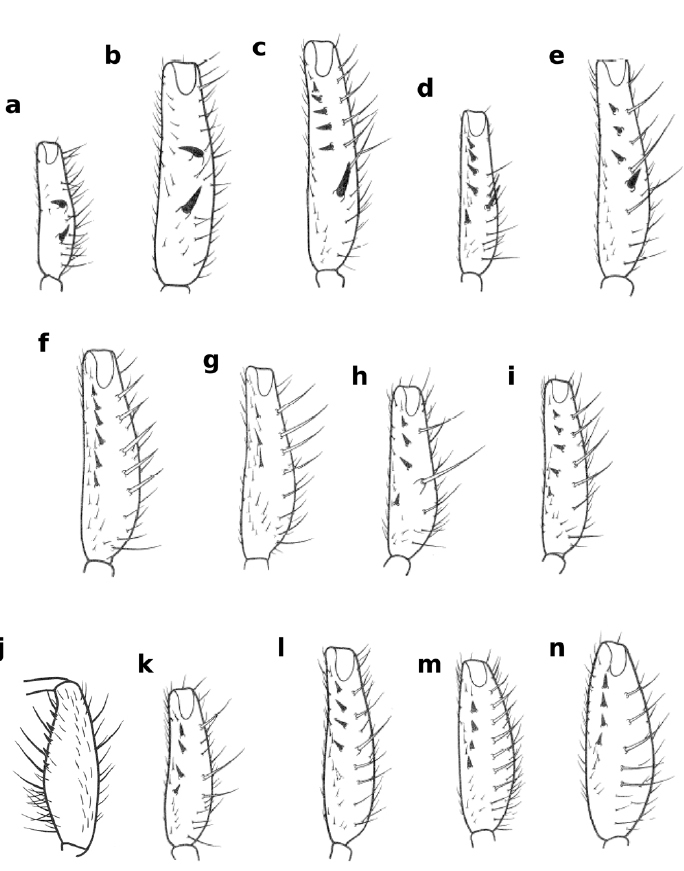
Ventral views of forefemur of Zaprionus campestris Chassagnard, 1989 a, Zaprionus montanus Collart, 1937 b, Zaprionus spinosus Collart, 1937 c, Zaprionus spineus Tsacas & Chassagnard, 1990 d, Zaprionus serratus Chassagnard, 1989 e, Zaprionus fumipennis Seguy, 1938 f, Zaprionus vrydaghi Collart, 1937 g, Zaprionus tuberarmatus Tsacas & Chassagnard, 1990 h, Zaprionus hoplophorus Tsacas & Chassagnard, 1990 i, Zaprionus armatus Collart, 1937 j, Zaprionus enoplomerus Chassagnard, 1989 k, Zaprionus spinipes Tsacas & Chassagnard, 1990 l, Zaprionus seguyi Tsacas & Chassagnard, 1990 m, and Zaprionus spinoarmatus Tsacas & Chassagnard, 1990 n [From
The vittiger group comprises 17 species with usually hairy epandrium carrying more than 2 posterior bristles (Fig. 14d, f).
It is mainly characterized by the relatively deep serration of the
aedeagal flap. The F1 of most of its species carry composite spines that
have bristles fused at their bases and usually are borne on protruding
tubercules (Fig. 2e, f). Three species (Zaprionus sexstriatus, Zaprionus sexvittatus and Zaprionus litos) have the unarmed F1 and have been classified in the inermis group (
Wing of Zaprionus fumipennis Seguy, 1938 a, and dorsal views of Zaprionus vrydaghi Collart, 1937 b, Zaprionus hoplophorus Tsacas & Chassagnard, 1990 c, and Zaprionus tuberarmatus Tsacas & Chassagnard, 1990 d.
Lateral and dorsal views of Zaprionus koroleu Burla, 1954 a, b, Zaprionus vittiger Coquillett, 1902 c, d, Zaprionus lachaisei Yassin & David, sp. n. e, Zaprionus santomensis Yassin & David, sp. n. f, and Zaprionus camerounensis Chassagnard & Tsacas, 1993 g, h.
Puparium of Zaprionus neglectus Collart, 1937 a, Zaprionus inermis Collart, 1937 b, Zaprionus cercus Chassagnard & McEvey, 1992 c, Zaprionus santomensis Yassin & David, sp. n. d, Zaprionus lachaisei Yassin & David, sp. n. e and Zaprionus vittiger Coquillett, 1902 f.
This species resembles Zaprionus indianus and Zaprionus gabonicus, but can be distinguished from them by the deep serration of the apical margin of the aedeagal flap, the shape of the spermatheca being wider than long and the presence of 8 (rarely 7) peg-like ovisensilla on the oviscape, which is constricted ventrally (Fig. 12).
♂. TL = 1.38 mm.
Head. Arista with 3 dorsal and 2 ventral rays plus terminal fork; pedicel white, flagellomere I dark brown. Frons orange, without a median stripe but with orbital stripes inwardly bordered with black; ocellar triangle concolorous with frons; hw:fw = 2.42, fw:fl = 0.96. Orbital setae in straight line; or1:or2:or3 = 3:2:3, orbito-index = 1.1, oc:or1 = 1.45, poc:oc = 0.63, iv:ov = 0.88. Face whitish yellow; carina broad and bulbous. Gena broad, o:j = 9.3, o:ch = 6.2. Eye red.
Thorax. Scutum brown, darker than frons, with 2 silvery white stripes. acs in 6 rows in front of adc; adc:pdc = 0.8. Scutellum darker than scutum, with black borders of the stripes expanded posteriorly; bsc:asc = 0.7. Pleura yellow; sterno-index = 0.38. Forefemur with 4–5 spines borne on warts on the anteroventral margin. Male basitarsus with a hairy brush.
Wing. Yellowish. C-index = 2.5, 4v-index = 1.3, 4c-index = 0.9, 5x-index = 1.0, M-index = 0.4, ac-index = 2.5, b/c = 0.7, C3 fringe = 47%, and WL = 2.90 mm.
Abdomen. Entirely yellow with deep dark spots at the bases of tergal setae.
Terminalia. Epandrium densely pubescent throughout its entire length; posterior margin pubescent at dorsal portion with 4 long setae; epandrial ventral lobe with 3 long setae. Surstylus quadrate with two rows of prensisetae. Cercus triangular laterally. Hypandrium densely pubescent at the lateral portion of the paraphyses. Aedeagus expanded apically with a hook-like appendix; aedeagal flap expanded and deeply serrated. Apodeme subequal in length to aedeagus.
♀. TL = 1.39 mm, resembling male.
Terminalia. Oviscape constricted ventrally, with 8 peg-like and 6 short, marginal setae plus 4 supernumeraries. Spermatheca wide, campaniform and smooth.
Egg. Elliptical with 4, equally long and fine filaments.
Larva. Escaping the culture medium when crowded.
Puparium. Horn-index 9.8.
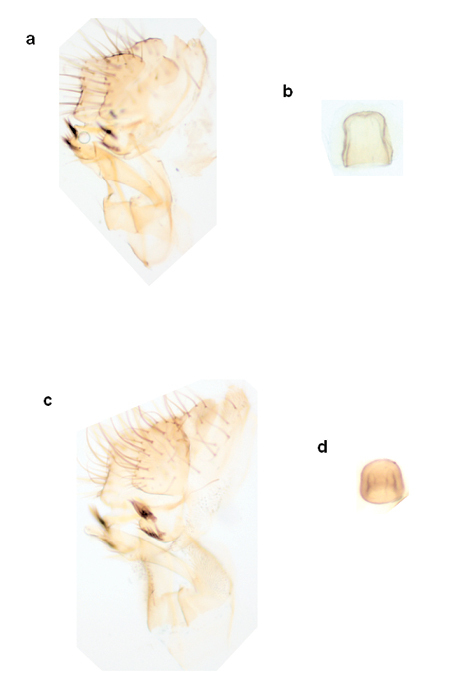
Distiphallus, oviscape and spermatheca of Zaprionus africanus Yassin & David in
This species resembles Zaprionus indianus, but it can be distinguished from it by the small body size and the total lack of serration on the aedeagal flap (Fig. 12)
♂. TL = 1.40 mm.
Head. Arista with 3 dorsal and 2 ventral rays plus terminal fork; pedicel white, flagellomere I dark brown. Frons orange, sometimes with highly vestigial median stripe plus orbital stripes inwardly bordered with black; ocellar triangle concolorous with frons; hw:fw = 2.45, fw:fl = 0.85. Orbital setae in straight line; or1:or2:or3 = 1.1:1.0:1.2, orbito-index = 1.1, oc:or1 = 1.4, poc:oc = 0.7, iv:ov = 0.7. Face whitish yellow; carina broad and bulbous. Gena narrow; o:j = 10, o:ch = 4.9. Eye red.
Thorax. Scutum brown, darker than frons, with 2 silvery white stripes. acs in 6 rows in front of adc; adc:pdc = 0.75. Scutellum darker than scutum, with black borders of the stripes expanded posteriorly; bsc:asc = 0.9. Pleura yellow; sterno-index = 0.44. Forefemur with 4–5 spines borne on warts on the anteroventral margin. Male basitarsus with a hairy brush.
Wing. Yellowish. C-index = 2.3, 4v-index = 1.4, 4c-index = 0.8, 5x-index = 1.0, M-index = 0.4, ac-index = 2.2, b/c = 0.6, C3 fringe = 52%, and WL = 2.7 mm.
Abdomen. Entirely yellow with deep dark spots at the bases of tergal setae.
Terminalia. Epandrium densely pubescent throughout its entire length; posterior margin pubescent at dorsal portion with 4 long setae; epandrial ventral lobe with 3 long setae. Surstylus quadrate with two rows of prensisetae. Cercus triangular laterally. Hypandrium densely pubescent at the lateral portion of the paraphyses. Aedeagus slender expanded apically without a hook-like appendix; aedeagal flap expanded and not serrated. Apodeme subequal in length to aedeagus.
♀. TL = 1.34 mm, resembling male.
Terminalia. Oviscape not constricted ventrally, with 6 (rarely 7) peg-like and 6 short, marginal setae plus 4 supernumeraries. Spermatheca globulous and smooth, not wider than longer.
Egg. Elliptical with 4 equally long and fine filaments.
Larva. Escaping the culture medium when crowded.
Puparium. Horn-index 10.4.
Male genitalia and spermatheca of Zaprionus lachaisei Yassin & David, sp. n. a, b, and Zaprionus santomensis Yassin & David, sp. n. c, d.
The identity of the dark species Zaprionus koroleu has long been problematic since its description by
Spermatheca and male epandrium of Zaprionus ornatus Seguy, 1933 a, Zaprionus davidi Chassagnard & Tsacas, 1993 b, Zaprionus taronus Chassagnard & Tsacas, 1993 c, d, and Zaprionus capensis Chassagnard & Tsacas, 1993 e, f [Illustrations from
Larval cephalopharyngeal skeleton of Zaprionus sepsoides Duda, 1939.
urn:lsid:zoobank.org:act:842BCF21-9ACF-48C1-9B53-9DAC95C49554
This species resembles Zaprionus vittiger, but has the bigger body size (TL > 1.60 mm), spiniform spines enlarged and blackened on the first two tarsomeres of the foreleg (Fig. 5), and shorter puparial anterior spiracles (H = 5) (Fig. 11). It is also distinguishable by a peculiar behavior of the larvae which do not leave the culture bottle when disturbed or crowded.
♂. TL = 1.68 mm.
Head. Arista with 3 dorsal and 2 ventral rays plus a terminal fork, pedicel tan. Frons orange-tan with lateral white stripes; median white stripe absent; ocellar triangle raised and darker; hw:fw = 2.04, fw:fl = 1.05. Face pale; carina large; palpus yellow. Gena broad, o:j = 10.2, o:ch = 5.2. Orbital bristles in straight line; or2 very minute, or1:or2:or3 = 7:2:5, orbito-index = 1.4. Ocellar setae long, divergent; oc:or1 = 1.3, poc:oc = 0.5, iv:ov = 0.6. Eye red and densely pilose.
Thorax. Scutum tan, darker than frons, with four white longitudinal stripes continuing on scutellum; white stripes narrow, bordered with large black stripes, especially on the inner side; acs in 6 regular rows anterior to adc and 4 irregular rows between them; psc enlarged, adc:psc = 1.5; adc:pdc = 0.6. Scutellum slightly pointed at the apex, where white spot absent; bsc:asc = 1.3. Sterno-index = 0.6. F1 with 4 setiferous spines not borne on tubercules on the anteroventral margin. Basitarsus of the foreleg with a hairy brush on the ventral margin. Spiniform spines of the first and second tarsomeres of the foreleg enlaged and blackened.
Wing. Dusky; WL:WW = 2.3, C-index = 3.0, 4v-index = 1.5, 4c-index = 0.8, 5x-index = 0.7, M-index = 0.3, ac-index = 2.5, b/c = 0.6, C3 fringe 0.45, WL = 3.8 mm.
Abdomen. Uniformly tan, with dark spots at the bases of tergal bristles.
Terminalia (Fig. 13a). Epandrium densely pubescent at ventral portion; posterior margin pubescent at dorsal portion with 5 long bristles; anterior phragma narrow; epandrial ventral lobe with 3 long bristles. Surstylus quadrate with two rows of prensisetae. Cercus triangular laterally. Hypandrium with a small pubescent patch at the lateral portion of the paraphyses. Aedeagus expanded apically; aedeagal flap expanded and deeply serrate. Apodeme subequal in length to aedeagus.
♀. TL = 1.76 mm, resembling male.
Terminalia. Oviscape with 8 peg-like and 7 short, marginal setae plus 4 supernumary. Spermatheca large, globulous and smooth (Fig. 13b).
Egg. Elliptical with 4 equally long and fine filaments.
Larva. Not escaping the culture medium when disturbed or crowded.
Puparium. H = 5.0 (Fig. 11d).
Tanzania.
Holotype (male) and allotype (female), Tanzania: East-Usambara Mountains, Amani (870 m), ex type strain ZMI.12, 11-VIII-2008, founder female coll. 25-IX-2002, D. Lachaise. Paratypes: 10 males and 10 females with the same label. Types deposited in MNHN.
Attempts to hybridize this strain with others belonging to the vittiger complex have all failed. The species is very prolific and easy to breed in the laboratory.
Patronym, in honor of the French Drosophila systematist Dr. Daniel Lachaise (1948–2006), collector of the types of two new species described here.
urn:lsid:zoobank.org:act:4DE262CC-1AD9-4D00-827B-FC62FC28BACD
This species resembles those of the indianus complex in having abdominal tergal spots and F1 spines not borne on protruding tubercule. It can be distinguished from them by the bigger body size, the darker body color mainly in contrast with the frons which is bright orange (Fig. 10f), the wings being dusky rather than hyaline, the smaller hairy brush of the male basitarsus (1/3 of basitarsus) (Fig. 5e), and the lack of an apical introvert in the spermatheca (Fig. 13d).
♂. TL = 1.40 mm.
Head. Arista with 2 dorsal and 3 ventral rays plus terminal fork; pedicel dark brown. Frons orange tan, with vestigial median stripe plus orbital stripes inwardly bordered with black; ocellar triangle blackened; hw:fw = 2.16, fw:fl = 0.8. Orbital setae in straight line; or1:or2:or3 = 3:2:3, orbito-index = 1.8, oc:or1 = 1.5, poc:oc=0.6, iv:ov = 0.4. Face tan. Gena narrow, o:j = 7.6, o:ch = 5.1. Eye red.
Thorax. Scutum brown, darker than frons, with 2 silvery white stripes. acs in 6 rows in front of adc; adc:pdc = 0.9. Scutellum darker than scutum, with black borders of the stripes expanded posteriorly; bsc:asc = 1.2. Pleura with white pilosity; sterno-index = 0.4. Forefemur with 4 spines not borne on warts on the anteroventral margin. Male basitarsus with a hairy brush.
Wing. Dusky; WL:WW = 2.3, C-index = 2.8, 4v-index = 1.4, 4c-index = 0.8, 5x-index = 0.9, M-index = 0.3, ac-index = 2.7, b/c = 0.6, C3 fringe 0.40, and WL = 3.2 mm.
Abdomen. Entirely yellowish, lighter than thorax, with faint dark spots at the bases of tergal setae.
Terminalia (Fig. 13c). Epandrium densely pubescent at ventral portion; posterior margin pubescent at dorsal portion with 3 long setae; anterior phragma slightly humped dorsally; epandrial ventral lobe with 4 long setae. Surstylus quadrate with two rows of prensisetae. Cercus triangular laterally. Hypandrium densely pubescent at the lateral portion of the paraphyses. Aedeagus expanded apically; aedeagal flap expanded and deeply serrate. Apodeme subequal in length to aedeagus.
♀. TL = 1.50 mm, resembles male.
Terminalia. Oviscape with 8 peg-like and 6 short, marginal setae plus 4 supernumeraries. Spermatheca globulous and smooth (Fig. 13d).
Egg. Elliptical with 4 equally long and fine filaments.
Larva. Escaping the culture medium when crowded.
Puparium. Horn-index 10.6.
Sao Tomé and Príncipe.
Holotype (male) and allotype (female), Sao Tomé and Príncipe: Pico de São Tomé Park (1, 500 m), ex type strain ZNG, 11-VIII-2008, founder female coll. III-2001, D. Lachaise. Paratypes: 10 males and 10 females with the same label. Types deposited in MNHN.
This species resembles Zaprionus proximus,
from which it can be distinguished on the basis of F1 ornamentation.
An important physiological difference also exists between these species,
as Zaprionus santomensis
is a very heat-sensitive species since a growth temperature of 25°C is
lethal for both sexes and males are sterile at 23 and 24°C (cf.
The species epithet is in reference to the type locality.
Many authors described the internal anatomy of some Zaprionus species that can be grown in laboratory (
Comparative morphometry of internal structures of male and female reproductive systems in Zaprionus.
| Male | Female | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| TST | SV | VD | PAR | EC | EB | CAE | SR | SP | |
| Subgenus Anaprionus | |||||||||
| Zaprionus (Anaprionus) bogoriensis | 4.4 | 2.0 | 0.80 | 2.6 | 2.2 | 0.30 | 0.6 | 3.8 | 0.07 |
| Subgenus Zaprionus | |||||||||
| neglectus group | |||||||||
| Zaprionus (Zaprionus) neglectus | 2.8 | 1.0 | 0.60 | 0.7 | 2.4 | 0.20 | 2.0 | 3.2 | 0.06 |
| inermis group | |||||||||
| Zaprionus (Zaprionus) ghesquierei | 1.2 | 0.6 | 0.04 | 2.0 | 1.1 | 0.22 | 1.0 | 1.5 | 0.04 |
| Zaprionus (Zaprionus) inermis | 1.5 | 1.1 | 0.20 | 2.6 | 2.1 | 0.32 | 0.4 | 1.0 | 0.09 |
| Zaprionus (Zaprionus) cercus | 1.4 | 0.9 | 0.16 | 2.2 | 2.0 | 0.22 | 1.6 | 0.9 | 0.08 |
| Zaprionus (Zaprionus) mascariensis | 4.4 | 0.9 | 0.40 | 3.2 | 1.1 | 0.22 | 0.5 | 7.2 | 0.12 |
| Zaprionus (Zaprionus) kolodkinae | 1.0 | 0.7 | 0.20 | 1.6 | 2.1 | 0.20 | 0.8 | 0.8 | 0.06 |
| Zaprionus (Zaprionus) sepsoides | 2.0 | 0.6 | 0.20 | 3.2 | 1.6 | 0.20 | 0.1 | 1.0 | 0.04 |
| Zaprionus (Zaprionus) tsacasi | 1.3 | 0.8 | 0.40 | 3.6 | 1.2 | 0.20 | 0.4 | 1.2 | 0.06 |
| Zaprionus (Zaprionus) tuberculatus | 3.2 | 1.2 | 0.70 | 2.2 | 0.9 | 0.20 | 0.3 | 3.6 | 0.06 |
| Zaprionus (Zaprionus) burlai | 4.4 | 1.0 | 1.10 | 2.0 | 1.3 | 0.12 | 0.3 | 6.3 | 0.06 |
| Zaprionus (Zaprionus) verruca | 3.8 | 1.6 | 0.80 | 2.0 | 2.0 | 0.20 | 1.2 | 4.0 | 0.06 |
| vittiger group | |||||||||
| Zaprionus (Zaprionus) ornatus | 12.4 | 7.2 | 2.20 | 3.6 | 0.9 | 0.30 | 0.7 | 12.0 | 0.18 |
| Zaprionus (Zaprionus) indianus | 5.3 | 2.2 | 1.30 | 2.2 | 1.5 | 0.30 | 0.7 | 4.8 | 0.16 |
| Zaprionus (Zaprionus) africanus | 5.4 | 1.0 | 0.70 | 1.6 | 1.3 | 0.30 | 0.8 | 3.8 | 0.07 |
| Zaprionus (Zaprionus) gabonicus | 2.5 | 0.7 | 0.40 | 0.7 | 0.7 | 0.16 | 0.4 | 3.5 | 0.06 |
| Zaprionus (Zaprionus) davidi | 2.6 | 1.4 | 0.80 | 2.0 | 1.6 | 0.30 | 0.6 | 3.0 | 0.06 |
| Zaprionus (Zaprionus) taronus | 5.2 | 1.4 | 1.40 | 3.2 | 2.2 | 0.30 | 0.8 | 4.6 | 0.06 |
| Zaprionus (Zaprionus) capensis | 4.0 | 2.0 | 0.80 | 2.6 | 1.2 | 0.30 | 0.6 | 4.6 | 0.07 |
| Zaprionus (Zaprionus) proximus | 3.6 | 2.4 | 2.00 | 1.4 | 2.0 | 0.28 | 0.3 | 4.2 | 0.06 |
| Zaprionus (Zaprionus) santomensis sp. n. | 3.6 | 1.6 | 1.20 | 2.0 | 1.6 | 0.34 | 0.7 | 3.2 | 0.10 |
| Zaprionus (Zaprionus) lachaisei sp. n. | 4.4 | 2.4 | 1.30 | 2.0 | 2.1 | 0.30 | 0.7 | 4.6 | 0.10 |
| Zaprionus (Zaprionus) vittiger | 4.4 | 2.4 | 1.30 | 2.0 | 2.4 | 0.30 | 0.8 | 4.2 | 0.12 |
| Zaprionus (Zaprionus) camerounensis | 4.2 | 2.0 | 0.70 | 3.2 | 1.2 | 0.20 | 0.6 | 4.5 | 0.09 |
Measurements of immature stages in Zaprionus species grown under the same laboratory conditions.
| Egg | Puparium | |||
|---|---|---|---|---|
| EL:El | PF:EL | PL:Pl | H | |
| Subgenus Anaprionus | ||||
| Zaprionus (Anaprionus) bogoriensis | 3.45 | 1.13 | 2.54 | 9.3 |
| Subgenus Zaprionus | ||||
| neglectus group | ||||
| Zaprionus (Zaprionus) neglectus | 2.90 | 0.83 | 2.31 | 15.3 |
| inermis group | ||||
| Zaprionus (Zaprionus) ghesquierei | 3.00 | 0.54 | 2.54 | 9.4 |
| Zaprionus (Zaprionus) inermis | 3.26 | 1.13 | 2.62 | 13.1 |
| Zaprionus (Zaprionus) cercus | 2.90 | 0.97 | 2.40 | 10.3 |
| Zaprionus (Zaprionus) mascariensis | 2.91 | 0.73 | 2.47 | 6.8 |
| Zaprionus (Zaprionus) kolodkinae | 2.75 | 0.97 | 2.43 | 9.0 |
| Zaprionus (Zaprionus) sepsoides | 3.10 | 0.90 | 2.57 | 8.6 |
| Zaprionus (Zaprionus) tsacasi | 2.73 | 0.90 | 2.53 | 8.4 |
| Zaprionus (Zaprionus) tuberculatus | 2.86 | 0.90 | 2.59 | 7.0 |
| Zaprionus (Zaprionus) burlai | 3.00 | 0.91 | 2.29 | 7.2 |
| Zaprionus (Zaprionus) verruca | 3.40 | 0.88 | 2.31 | 10.6 |
| vittiger group | ||||
| Zaprionus (Zaprionus) ornatus | 3.18 | 1.14 | 2.52 | 10.0 |
| Zaprionus (Zaprionus) indianus | 3.44 | 0.81 | 2.49 | 8.3 |
| Zaprionus (Zaprionus) africanus | 3.26 | 0.90 | 2.46 | 9.8 |
| Zaprionus (Zaprionus) gabonicus | 3.33 | 0.83 | 2.43 | 10.4 |
| Zaprionus (Zaprionus) davidi | 3.05 | 1.16 | 2.54 | 10.5 |
| Zaprionus (Zaprionus) taronus | 2.87 | 0.91 | 2.29 | 12.0 |
| Zaprionus (Zaprionus) capensis | 2.43 | 1.00 | 2.45 | 9.8 |
| Zaprionus (Zaprionus) proximus | 3.67 | 1.06 | 2.44 | 10.6 |
| Zaprionus (Zaprionus) santomensis sp. n. | 2.86 | 0.60 | 2.24 | 10.6 |
| Zaprionus (Zaprionus) lachaisei sp. n. | 3.28 | 0.78 | 2.64 | 5.0 |
| Zaprionus (Zaprionus) vittiger | 3.20 | 1.06 | 2.65 | 9.3 |
| Zaprionus (Zaprionus) camerounensis | 3.00 | 0.93 | 2.56 | 11.0 |
Testis length (TST) ranges from 1.0 mm in Zaprionus kolodkinae to 12.4 mm in Zaprionus ornatus. The Oriental species, Zaprionus (Anaprionus) bogoriensis, has TST of 4.4 mm which approaches that of the mean of the African species (3.7 ± 0.5 mm). Species of the inermis group can be classidfied under two categories: those with small testis ranging from 1.0 to 2.0 mm (Zaprionus inermis, Zaprionus cercus, Zaprionus kolodkinae, Zaprionus sepsoides and Zaprionus tsacasi), and those with large testis ranging from 3.2 to 4.4 mm (Zaprionus mascariensis, Zaprionus tuberculatus, Zaprionus burlai and Zaprionus verruca). Species of the last category are all members of the tuberculatus subgroup which also include some species of the first category, and TST presents a very informative taxonomic clue (Fig. 3;
The seminal vesicle (SV) is the part of the vas deferens that has undergone a differentiation for sperm storage. It ranges from 0.6 mm in Zaprionus ghesquierei and Zaprionus sepsoides to 7.2 mm in Zaprionus ornatus, with the mean of 1.6 ± 0.3 mm in African Zaprionus. Species of the inermis group tend to have small SV, ranging from 0.6 to 1.6 mm, whereas species of the vittiger group have larger SV, ranging from 0.7 to 2.4 mm (excluding Zaprionus ornatus).
The vas deferens (VD) ranges from 0.04 mm in Zaprionus ghesquierei to 2.20 mm in Zaprionus ornatus. The quasi-absence of VD in Zaprionus ghesquierei is exceptional as the next value to it is 0.20 mm in a number of species of the inermis group (Zaprionus inermis, Zaprionus kolodkinae and Zaprionus sepsoides). Indeed,
The ejaculatory bulb of Zaprionus species is moderately large, rounded and bearing long posterior caecae (
The seminal receptacle (SR) ranges from 0.8 mm in Zaprionus kolodkinae to 12.0 mm in Zaprionus ornatus. As with TST, species of the vittiger group tend to have larger SR than those of the inermis group. The correlation between TST and SR is a well-established fact in the Drosophilidae, although the correlation is thought to be functional rather than genetic (
The eggs of species of the Oriental subgenus Anaprionus have two filaments (
The length of the filaments varies between species (Table 3). In Zaprionus momorticus, the four filaments are very short (
Larvae of the genus Zaprionus are all of the amphipneustic type as in other drosophilid flies (
Puparia of the two subgenera are reddish brown in color (Fig. 11). The puparial length (PL) ranges from 2.82 mm in Zaprionus gabonicus to 4.58 mm in Zaprionus inermis, in complete concordance with the differences of body size in the adults (
The horn-index (H) is a classical taxonomic measurement in drosophilid systematics. H ranges from 5.0 in Zaprionus lachaisei sp. n. (Fig. 11d) to 15.3 in Zaprionus neglectus (Fig. 11A) with the mean of 9.7 ± 0.4 in African Zaprionus (9.3 in the Oriental species Zaprionus bogoriensis). With the exception of the two extremes, H ranges from 6.8 to 13.1. In the tuberculatus species complex, H discriminates Zaprionus verruca (H = 10.6) from its two sibling species, Zaprionus tuberculatus (H = 7.0) and Zaprionus burlai (H = 7.2).
Another important taxonomic character of the puparium is the branches of the anterior spiracle. In all Zaprionus species, these branches are of the clubbed type (
The two new species described here were collected by the late Dr. Daniel Lachaise (LEGS, CNRS) to which the authors would like to acknowledge their debt. The authors are also much grateful to two anonymous reviewers for constructive suggestions, Mrs. Marie-Thérèse Chassagnard and Dr. Léonidas Tsacas (MNHN) for allowing the reproduction of their drawings, Dr. Pierre Capy (LEGS) for providing the opportunity to travel to Madagascar from which several strains important to this study were collected, Dr. Shane F. McEvey (Australian Museum, Sydney) for his constructive critiques on a previous form of this manuscript, Dr. Brian Fischer (California Academy of Science), Dr. Lala Ravaomanarivo (University of Antananarivo) and workers in the Bibikely Biodiversity Institute for providing help during the collection of the flies, and to Drs. Vincent Debat (MNHN), Gerhard Baechli (Zoological Museum, Zurich), and Patrick Grootaert (Royal Belgian Institute of Natural Sciences, Brussels) for allowing the junior author to visit the collections and photograph the types. This work was funded by the Agence Universitaire de la Francophonie (AUF), Fondation des Treilles and Fondation Bettencourt-Schueller.