Citation: Cifuentes-Ruiz P, Zaragoza-Caballero S, Ochoterena-Booth H, Morón MA (2014) A preliminary phylogenetic analysis of the New World Helopini (Coleoptera, Tenebrionidae, Tenebrioninae) indicates the need for profound rearrangements of the classification. In: Bouchard P, Smith AD (Eds) Proceedings of the Third International Tenebrionoidea Symposium, Arizona, USA, 2013. ZooKeys 415: 191–216. doi: 10.3897/zookeys.415.6882
Helopini is a diverse tribe in the subfamily Tenebrioninae with a worldwide distribution. The New World helopine species have not been reviewed recently and several doubts emerge regarding their generic assignment as well as the naturalness of the tribe and subordinate taxa. To assess these questions, a preliminary cladistic analysis was conducted with emphasis on sampling the genera distributed in the New World, but including representatives from other regions. The parsimony analysis includes 30 ingroup species from America, Europe and Asia of the subtribes Helopina and Cylindrinotina, plus three outgroups, and 67 morphological characters. Construction of the matrix resulted in the discovery of morphological character states not previously reported for the tribe, particularly from the genitalia of New World species. A consensus of the 12 most parsimonious trees supports the monophyly of the tribe based on a unique combination of characters, including one synapomorphy. None of the subtribes or the genera of the New World represented by more than one species (Helops Fabricius, Nautes Pascoe and Tarpela Bates) were recovered as monophyletic. Helopina was recovered as paraphyletic in relation to Cylindrinotina. One Nearctic species of Helops and one Palearctic species of Tarpela (subtribe Helopina) were more closely related to species of Cylindrinotina. A relatively derived clade, mainly composed by Neotropical species, was found; it includes seven species of Tarpela, seven species of Nautes, and three species of Helops, two Nearctic and one Neotropical. Our results reveal the need to deeply re-evaluate the current classification of the tribe and subordinated taxa, but a broader taxon sampling and further character exploration is needed in order to fully recognize monophyletic groups at different taxonomic levels (from subtribes to genera).
External morphology, Holarctic genera, Neotropical clade, Neotropical genera, male and female genitalia, polyphyly, polytomy, paraphyletic Helopini
The tribe Helopini Latreille, 1802 currently contains two subtribes (Cylindrinotina and Helopina), 42 genera, and 686 species (
Helops, the type genus of the tribe, was described by
In the monumental Biologia Centrali-Americana,
In short, this diverse tribe includes two subtribes and multiple genera with worldwide distributions (
The subtribes Cylindrinotina and Helopina (
Taxon sampling.
| Tribe | Subtribe | Species | Geographic distribution | ||
|---|---|---|---|---|---|
| Ingroup | Helopini | Cylindrinotina | Nalassus plebejus | Küster, 1850 | Europe, Asia |
| Odocnemis californicus | (Mannerheim, 1843) | Mexico, U.S.A. | |||
| Stenomax aeneus | Scopoli, 1763 | Europe | |||
| Helopina | Entomogonus peryronis | Reiche, 1861 | Asia | ||
| Helops aereus | Germar, 1824 | U.S.A. | |||
| Helops cisteloides | Germar, 1824 | U.S.A. | |||
| Helops farctus | LeConte, 1858 | U.S.A. | |||
| Helops inanis | Allard, 1877 | Mexico | |||
| Helops insignis | Lucas, 1846 | North of Africa | |||
| Helops perforatus | Horn, 1880 | Mexico, U.S.A. | |||
| Helops punctipennis | LeConte, 1870 | U.S.A. | |||
| Helops rossii | Germar, 1817 | Europe | |||
| Helops rufipes | (LeConte, 1851) | Mexico, U.S.A. | |||
| Probaticus tentyrioides | Küster, 1851 | Asia, Europe | |||
| Raiboscelis corvinus | Küster, 1850 | Asia, Europe | |||
| Tarpela aerifera | Allard, 1876 | Mexico, Central America | |||
| Tarpela browni | Bates, 1870 | Nicaragua | |||
| Tarpela contigua | Champion, 1887 | Mexico | |||
| Tarpela cordicollis | Marseul, 1876 | Japan | |||
| Tarpela costata | Champion, 1887 | Mexico, Nicaragua | |||
| Tarpela depressa | Champion, 1887 | Mexico | |||
| Tarpela reticulata | Champion, 1887 | Honduras | |||
| Tarpela torrida | Champion, 1887 | Mexico | |||
| unassigned | Nautes belti | Allard, 1877 | Central America | ||
| Nautes enoplopoides | Champion, 1887 | Guatemala | |||
| Nautes fervidus | Pascoe, 1866 | Mexico, Central America | |||
| Nautes magnificus | Champion, 1887 | Guatemala | |||
| Nautes splendens | Champion, 1887 | Panama | |||
| Nautes striatipennis | Champion, 1887 | Mexico | |||
| Nautes varians | Champion, 1887 | Mexico | |||
| Outgroup | Ulomini | Uloma mexicana | Champion, 1886 | Mexico, Central America | |
| Hypogena biimpressa | Champion, 1886 | Mexico, Central America, South America | |||
| Tenebrionini | Tenebrio molitor | Linnaeus, 1758 | global |
Specimens were kindly loaned by curators at the following national and international institutions:
AMNH American Museum of Natural History, New York, NY, USA (Lee Herman)
BNHM The Natural History Museum, London, U. K. (Max Barclay)
CASC California Academy of Sciences, San Francisco, CA, USA (Dave Kavanaugh)
CNIN Colección Nacional de Insectos, Intituto de Biología, UNAM, Mexico City, Mexico (Santiago Zaragoza Caballero)
EMEC Essig Museum of Entomology, University of California, Berkeley, CA, USA (Peter T. Oboyski)
FMNH Field Museum of Natural History, Chicago, IL, USA (James Boone)
HNHM Hungarian Natural History Museum, Budapest, Hungary (Otto Merkl)
IEXA Colección entomológica, Instituto de Ecología, A. C., Xalapa, Veracruz, Mexico (Miguel Ángel Morón & Delfino Hernández)
MNHN Museum National d’Histoire Naturelle, Paris, France (Antoine Mantilleri)
LACM Natural History Museum of Los Angeles County, Los Angeles, CA, USA (Weiping Xie)
NMNH National Museum of Natural History, Smithsonian Institution, Washington, DC, USA (Warren Steiner & David Furth)
OSUC C. A. Triplehorn Insect Collection, Ohio State University, Columbus, OH, USA (Charles A. Triplehorn & Luciana Musetti)
SBMNH Santa Barbara Museum of Natural History, Santa Barbara, CA, USA (Michael Caterino)
TAMU Texas A & M University Insect Collection, College Station, TX, USA (Edward Riley)
UCDC Bohart Museum, University of California, Davis, CA, USA (Steve Heydon)
ZMHB Museum für Naturkunde der Humboldt-Universitat, Berlin, Germany (Bernd Jaeger)
Two hundred eighty-one specimens were examined with an Olympus SZH10 stereomicroscope (magnification: 17.5–350×) equipped with an ocular graticule for length measurements, and a drawing tube. Morphological characters were measured as follows: width of the head was measured across the vertex, length of the last antennomere in the female was measured along its longest edge; width was measured across its widest point; length of pronotum was measured along the midline from its anterior edge to its posterior edge; width was measured across its widest point. Puncture density follows modified conventions used by
Thirty-two characters correspond to external morphology; characters used in generic descriptions (
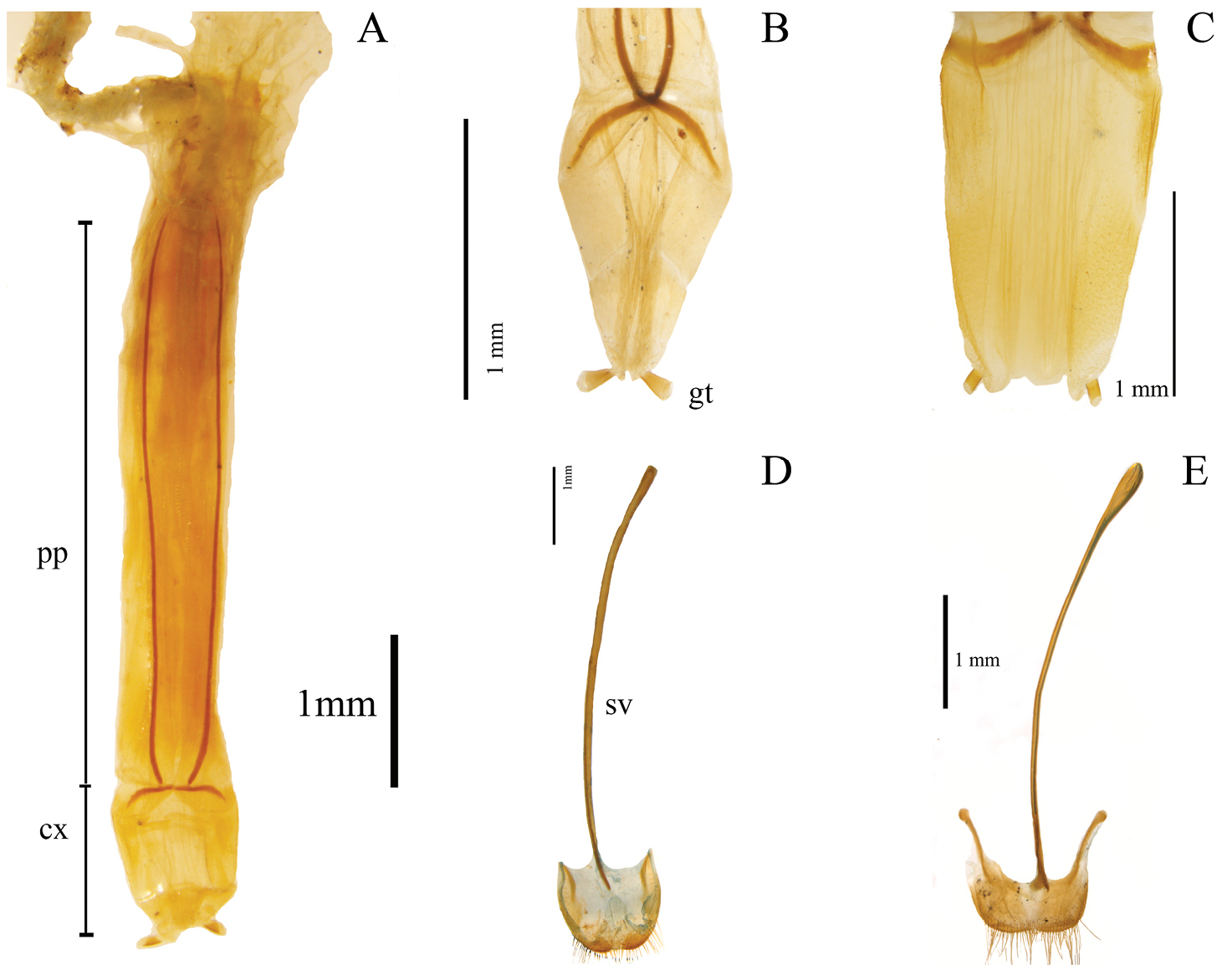
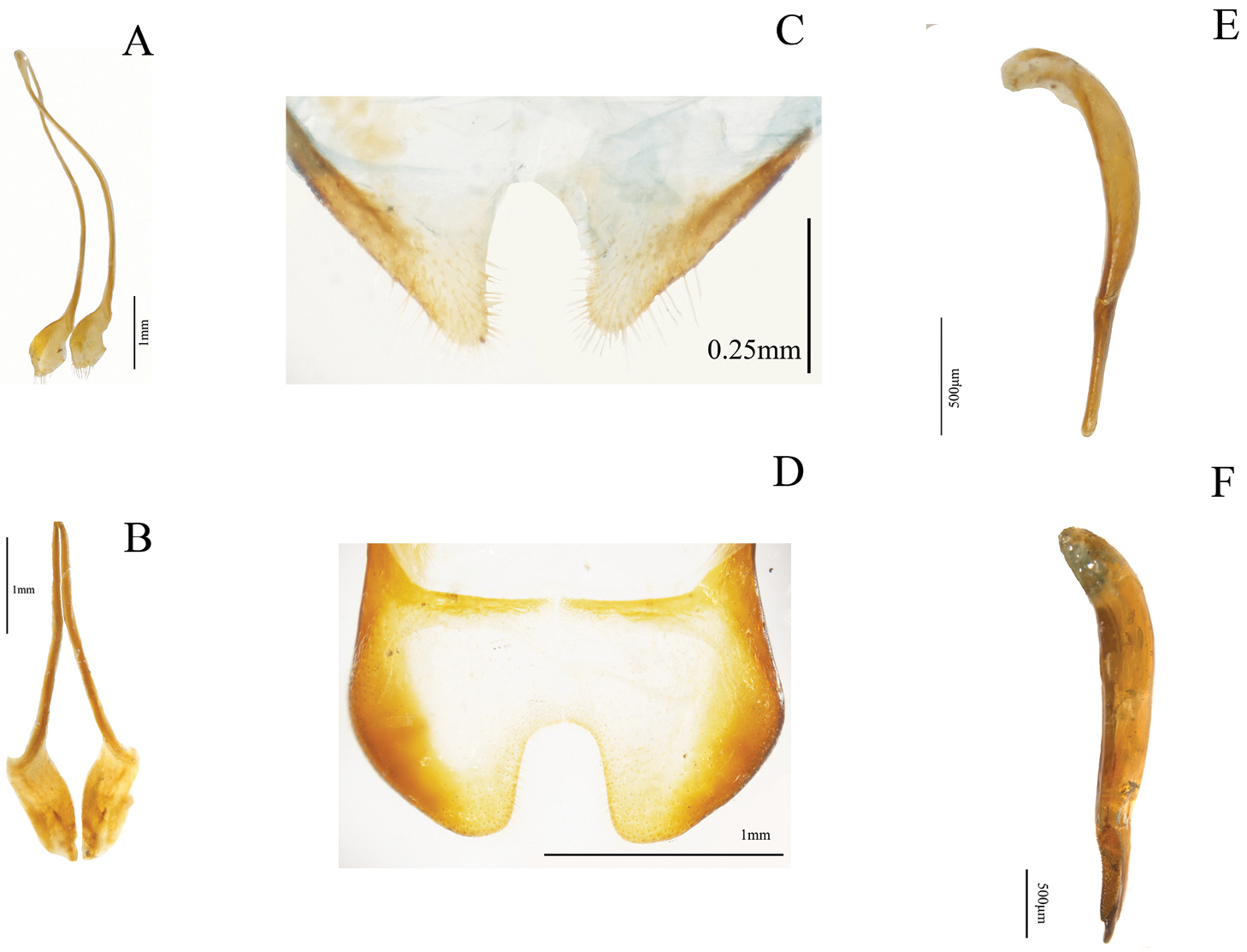
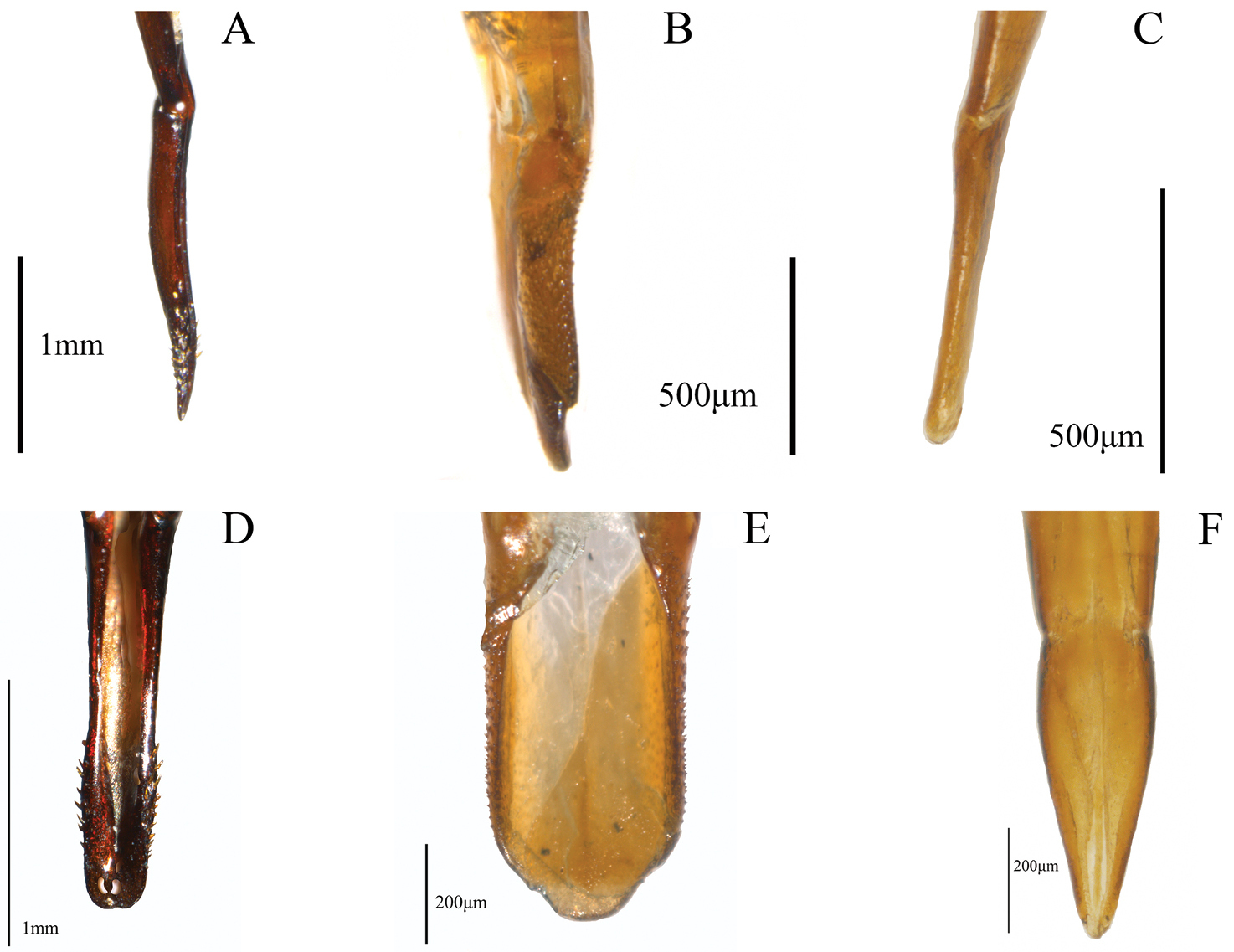
Examples of external characters (mainly diagnostic), traditionally used in Helops, Tarpela and Nautes, (number of character: character state): A broad apical antennomere (5:0), shorter than the third antennomere (4:0) illustrated from Helops aereus Germar B filiform apical antennomeres (5:2), as long as the third antennomere (4:1) illustrated from Nautes fervidus Pascoe C male maxillary palps with length of inner edge 2.6–2.9 times the length of posterior edge (7:2) illustrated from Nautes chrysomeloides Champion D third lobate segment of male tarsi (30:0) and short fourth tarsomere (31:0) illustrated from Nautes fervidus E not prominent prosternum (27:1) in Helops cisteloides Germar F prominent-acute prosternum (27:0) in Nautes fervidus.
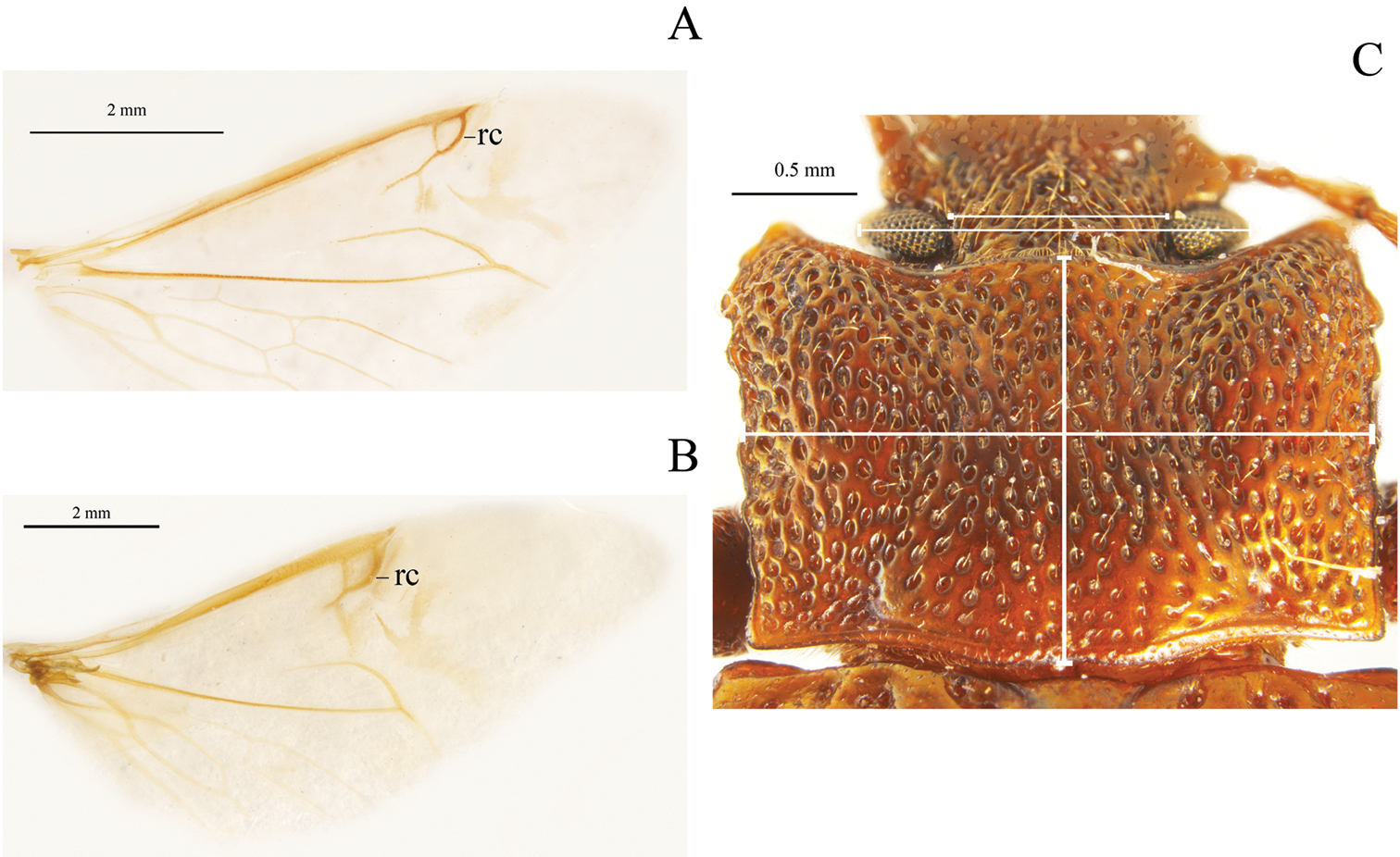
Examples of non-traditional external characters in Helopini: A reduced recurrent cell (rc) (26:0) in fully developed wing (25:1) of Helops californicus Mannerheim B wide recurrent cell (rc) (26:1) in fully developed wing (25:1) of Tarpela aerifera Allard C head width and interocular width (6:0) and pronotum width and length (18:0) in Tarpela costata
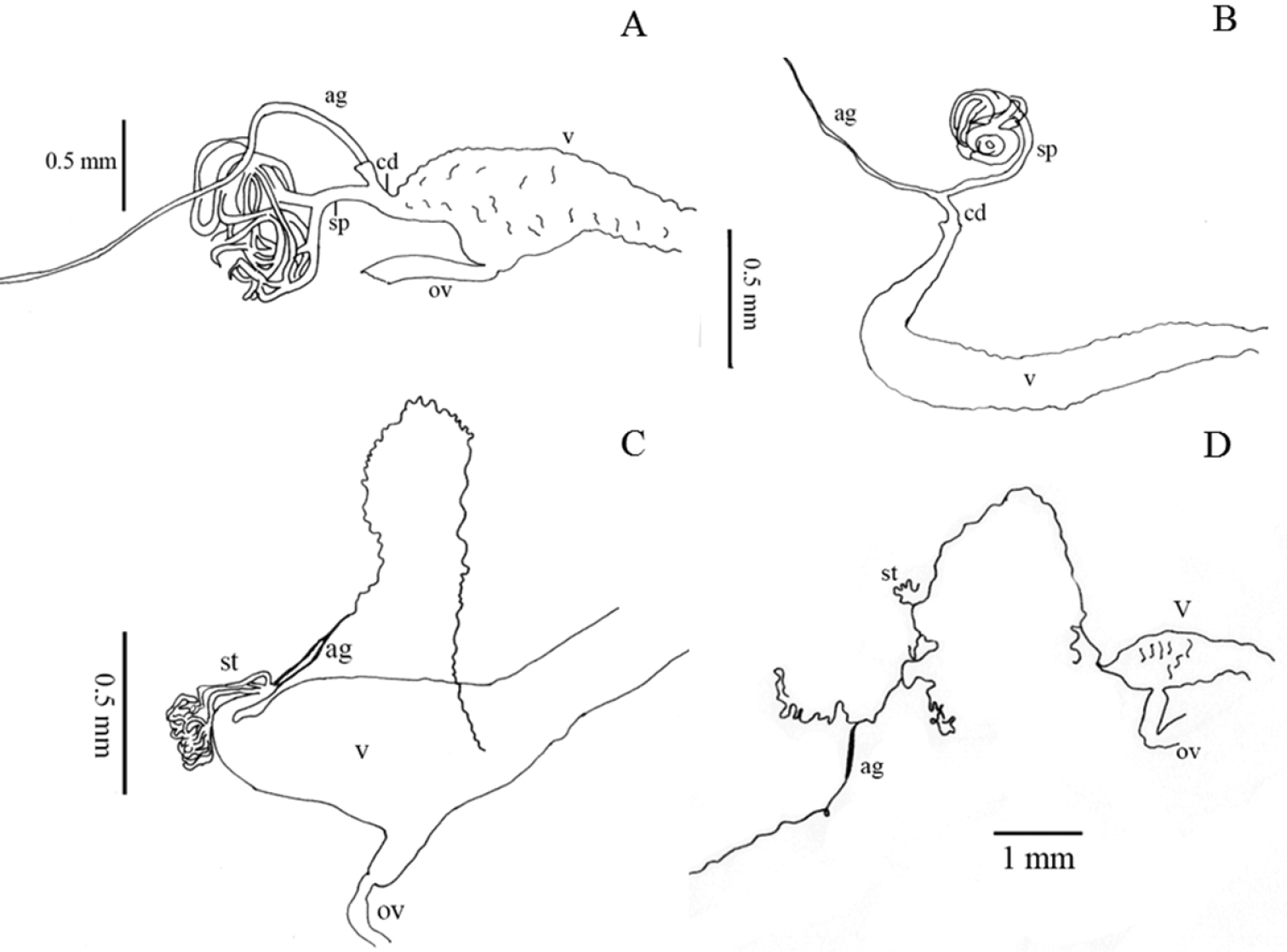
Internal morphological characters (female genitalia) in Helopini: A length of paraproct (pp) three or more times length of coxite (cx) (39:0) illustrated from the ovipositor of Helops cisteloides Germar B long gonostyles (gt) (37:1) with wide apex (38:1), represented by the ovipositor of Odocnemis exaratus Germar, not included in the analysis C reduced gonostyles (37:0), with base as wide as apex (38:0) represented by the ovipositor of Tarpela micans (Fabricius), not included in the analysis D blunt, narrow apex of eighth sternite (33:0), not evident arms (34:1) and not dilated distal end of the spiculum ventrale (sv) (35:1) illustrated from Helops cisteloides E trapeziform apex of eighth sternite (33:1), evident arms (34:0) and dilated distal end of spiculum ventrale (35:0), represented by sclerite of Odocnemis exaratus.
Internal morphological characters (female genitalia) representing the different morphological types found in our sample of Helopini ag = accessory gland, sp = spermatheca, st = spermathecal tube(s), cd = common duct of accessory gland and spermatheca, v = vagina, ov = oviduct: A infundibular vagina (40:0), single spermatheca branched near its base (41:0, 42:0) and accessory gland in the common duct (49:1) illustrated from Helops insignis Germar representing the helopiod type (
Internal morphological characters (male genitalia) representing the different morphological types found in our sample of Helopini: A pleural rods of gastral spicula close only at the end (50:2), representing the nalassoid type (
Aedeagal characters (male genitalia) representing the different morphological types found in or sample of Helopini: A evident setae (57: 0) representing the helopiod type (
In total, 44 binary and 23 multistate characters were coded and treated as non-additive. Individual consistency and retention indices (ci, ri) are provided for all characters from the consensus tree (synapomorphies have a value of 1 for both indices). Four additional characters (listed and explained at the end of the character list) were initially explored but removed from the final analysis due to their high homoplasy, assessed by a character removal methodology (see below).
-
Shape of union between clypeus and frons: (0) clypeus slightly depressed, weak fronto-clypeal suture (Fig. 1C); (1) clypeus strongly depressed, evident fronto-clypeal suture (ci = 0.25; ri = 0.80).
-
Length of antennae (male): (0) short, slightly over posterior margin of pronotum; (1) long, clearly surpassing posterior margin of pronotum (ci = 0.33; ri = 0.50).
-
Shape of antennae: (0) filiform (Fig. 1B); (1) submoniliform (synapomorphy).
-
Length of third antennomere: (0) longer than apical antennomeres (Fig. 1A); (1) shorter than or as long as apical antennomeres (Fig. 1B) (ci = 0.14; ri = 0.57).
-
Size of last antennomere (female): (0) almost as long as wide or wider than long (Fig. 1A); (1) 2.5 or more times as long as wide; (2) 1.5-2 times as long as wide (Fig. 1B) (ci = 0.30; ri = 0.30).
-
Head width (Fig. 2C): (0) 1.5 times width of intraocular space; (1) less than 1.5 times width of intraocular space (ci = 0.25; ri = 0).
-
Length of inner edge of maxillary palp (male): (0) 1-1.5 times length of posterior edge; (1) 1.6-2.5 times length of posterior edge; (2) 2.6-2.9 times length of posterior edge (Fig. 1C) (ci = 0.18; ri = 0.25).
-
Pronotum disk surface: (0) gibbous (Fig. 2C); (1) not gibbous (ci = 0.33; ri = 0.50).
-
Density of pronotum punctures: (0) very dense or confluent; (1) moderately dense; (2) sparse (ci = 0.25; ri = 0.40).
-
Depth of pronotum punctation: (0) deep (more than 20 μm) (Fig. 2C); (1) medium (10-20 μm); (2) shallow (less than 10 μm) (ci = 0.25; ri = 0.64).
-
Setae of head and pronotum (observed at a magnification up to 140X): (0) present; (1) not evident (ci = 0.50; ri = 0.60).
-
Width of lateral carinae of pronotum: (0) lateral carinae 2-5 times width of anterior carinae; (1) lateral carinae less than 2 times width of anterior carina (ci = 0.33; ri = 0.50).
-
Shape of anterior angles of pronotum: (0) acute; (1) blunt or slightly acute; (2) straight (ci = 0.25; ri = 0.40).
-
Lateral sides of pronotum: (0) with crenate carinae; (1) with smooth carinae; (2) without conspicuous carinae (ci = 0.40; ri = 0.40).
-
Shape of posterior angles of pronotum: (0) straight; (1) acute; (2) blunt; (3) obtuse (ci = 0.40; ri = 0.40).
-
Projection of posterior angles of pronotum: (0) strong; (1) weak (Fig. 2C) (ci = 0.50; ri = 0.50).
-
Posterior margin of pronotum: (0) convex; (1) straight; (2) bisinuate (Fig. 2C) (ci = 0.40; ri = 0.66).
-
Pronotum shape: (0) rectangular (its width 1.5 times or more its length) (Fig. 2C); (1) almost square (its width less than 1.5 its length) (ci = 0.50; ri = 0.92).
-
Propleura texture: (0) strongly rugose or punctated; (1) smooth or slightly rugose or punctated (ci = 0.20; ri = 0.33).
-
Elytra shape in lateral view: (0) strongly arcuate; (1) slightly arcuate; (2) more evident towards the middle and posteriorly (ci = 0.33; ri = 0.42).
-
Pronotum tegument: (0) smooth; (1) chagrined (ci = 0.16; ri = 0.28).
-
Elytra punctures: (0) in grooves; (1) in rows (ci = 0.12; ri = 0.50).
-
Shape of elytral interstriae: (0) convex; (1) flat; (2) acute (ci = 0.16; ri = 0.09).
-
Elytral tegument: (0) lustrous; (1) dull (ci = 0.14; ri = 0.33).
-
Metathoracic wings: (0) brachypterous or not evident; (1) fully developed (Figs 2A–B) (ci = 0.11; ri = 0.38).
-
Size of recurrent cell: (0) reduced (due to the approximation of the radial cross-vein to the recurrent radius) (Fig. 2A); (1) wide (due to the separation of the radial cross-vein to the recurrent radius) (Fig. 2B) (ci = 0.50; ri = 0.75).
-
Shape of prosternal process apex in ventral view: (0) strongly projected (Fig. 1F); (1) weakly, or not projected (Fig. 1E) (ci = 0.25; ri = 0.40).
-
Shape of prosternal process apex in lateral view: (0) straight; (1) declivous (ci = 0.14; ri = 0.25).
-
Density of leg punctures: (0) femur punctures sparser than tibia punctures; (1) density of femur and tibia punctures similar (ci = 0.14; ri = 0.53).
-
Shape of third tarsomere: (0) lobate (Fig. 1D); (1) not lobate (ci = 0.16; ri = 0.54).
-
Size of fourth tarsomere: (0) shorter than third tarsomere (Fig. 1D); (1) as long as third tarsomere (ci = 0.20; ri = 0.66).
-
Density of punctures and pubescence of abdominal ventrites (male): (0) high towards middle of ventrites 1-3; (1) homogeneous on ventrites 1-5; (2) high towards middle of ventrites 1-5 (ci = 0.15; ri = 0.42).
-
Shape of inner sternite VIII (female): (0) blunt and narrow (Fig. 3D); (1) trapeziform or blunt and wide (Fig. 3E) (ci = 0.33; ri = 0).
-
Arms of spiculum ventrale (female): (0) evident (Fig. 3E); (1) not evident (Fig. 3D) (ci = 0.20; ri = 0.33).
-
Shape of distal end of stalk of spiculum ventrale (female): (0) round or oval and dilated (Fig. 3E); (1) round but not dilated (Fig. 3D) (ci = 0.50; ri = 0.80).
-
Length/width ratio of gonostyles (female): (0) length twice or more its width; (1) length less than twice its width (ci = 0.25; ri = 0.25).
-
Relative length of coxites (female): (0) 8 or more times gonostyle length (Fig. 3C); (1) less than 8 times gonostyle length (Fig. 3B) (ci = 0.33; ri = 0.33).
-
Shape of gonostyles (female): (0) apex as wide as base (Fig. 3C); (1) with apex wider than base (Fig. 3B) (ci = 0.25; ri = 0.62).
-
Relative length of paraproct (female): (0) three or more times coxite length (Fig. 3A); (1) two times coxite length; (2) as long as coxites; (3) less than coxite length (ci = 0.37; ri = 0.50).
-
Shape of vagina (female): (0) infundibular or sacciform, curved or not at the apex (i.e., at the connection with the spermatheca or common duct) (Fig. 4A–C); (1) sacciform and strongly narrowed and curved before the apex (Fig. 4B) (synapomorphy).
-
Number of spermathecal tubes: (0) one (Fig. 4A-B); (1) more than one (Fig. 4C–D) (synapomorphy).
-
Spermathecal tubes structure: (0) branched near the base (Fig. 4A); (1) not branched, branched at the base (looking like a fascicule of tubes), or branched far from the base (Fig. 4B–D) (ci = 0.33; ri = 0.77).
-
Spermathecal tubes arrangement: (0) near to each other (Fig. 4C); (1) distant from each other (Fig. 4D) (ci = 0.50; ri = 0.50).
-
Common duct: (0) present; (1) absent (ci = 0.16; ri = 0).
-
Length of common duct of spermatheca and accessory gland: (0) long (Fig. 4D); (1) short (Fig. 4C); (2) intermediate (Fig. 4B) (ci = 0.20; ri = 0.27).
-
Position of common duct (female): (0) apical to vagina (Fig. 4A-D); (1) anterior to vagina apex (ci = 0.33; ri = 0).
-
Width of spermathecal tube(s) (female): (0) increases distally; (1) homogeneous width or gradually decreasing (ci = 0.50; ri = 0.66).
-
Texture of spermathecal tubes: (0) smooth; (1) annulate (synapomorphy).
-
Position of accessory gland: (0) emerging directly from the vagina, far from the spermatheca; (1) in the common duct (Fig. 4A-B); (2) terminal to the spermathecal tubes and common duct (Fig. 4C-D) (synapomorphy).
-
Arrangement of pleural rods of gastral spicula (male): (0) close towards the middle of their length (Fig. 5B); (1) close towards the proximal third; (2) close only at the end (Fig. 5A) (ci = 0.50; ri = 0.66).
-
Shape of pleural rods of gastral spicula (male): (0) straight of slightly curved (Fig. 5B); (1) strongly curved (Fig. 5A) (ci = 0.33; ri = 0.33).
-
Depth of notch of eighth sternite (male) measured as the ratio of sternite length (SL) and notch length (NL): (0) deep (SL/NL <3) (Fig. 5C); (1) shallow (SL/NL >3) (Fig. 5D); (2) without notch (ci = 0.50; ri = 0.81).
-
Width of notch of eighth sternite (male): (0) wide; (1) narrow (ci = 0.33; ri = 0).
-
Shape of lobes of eighth sternite (male): (0) notably and anteriorly wide (Fig. 5D); (1) narrow and acute or slightly blunt (Fig. 5C) (ci = 0.20; ri = 0.66).
-
Relative length of basal piece (male): (0) three or more times the length of parameres; (1) less than three times the length of parameres (Fig. 5E–F) (ci = 0.16; ri = 0).
-
Shape of parameres in lateral view (male): (0) sinuate (Fig. 6A); (1) straight or slightly curved (Fig. 6B-C) (ci = 0.25; ri = 0.57).
-
Setae on parameres (male): (0) present (Fig. 6D-E); (1) not evident (observed at a magnification up to 140X) (Fig. 6F) (ci = 0.25; ri = 0.66).
-
Distribution of evident setae on parameres (male): (0) covering apical half of parameres (Fig. 6D); (1) covering more than two thirds of parameres (Fig. 6E) (synapomorphy).
-
Sides of parameres in ventral view (male): (0) convergent to the apex, with a fusiform space in between (Fig. 6F); (1) not convergent (Fig. 6D–E) (ci = 0.50; ri = 0.92).
-
Constriction of the apex of parameres (male): (0) present (Fig. 6E); (1) absent (ci = 0.25; ri = 0.70).
-
Constriction of the apex of parameres (male): (0) strong; (1) weak (Fig. 6E) (ci = 0.33; ri = 0).
-
Shape of the apex of parameres (male): (0) blunt or straight (Fig. 6D); (1) acute (Fig. 6F); (2) fan shaped (ci = 0.50; ri = 0.80).
-
Apical projection of parameres in ventral direction (lateral and ventral view): (0) present; (1) absent (ci = 0.33; ri = 0.33).
-
Apical compression of parameres view laterally as a dorsal or dorso-ventral projection or keel: (0) present; (1) not evident (observed at a magnification up to 140X) (synapomorphy).
-
Width of parameres (male) at the middle: (0) narrower than basal piece (Fig. 6D); (1) as wide as basal piece (Fig. 6E–F) (ci = 0.25; ri = 0.57).
-
Shape of apex of median lobe (male): (0) blunt or with an inconspicuous notch; (1) lobate; (2) constricted (ci = 0.33; ri = 0.73).
-
Shape of anterior part of basal piece (male): (0) projected, basal piece “J” shaped (Fig. 5E); (1) not projected (Fig. 5F); (2) projected, basal piece “C” shaped (ci = 0.66; ri = 0.92).
Removed characters:
-
Width of pronotum: (0) widest towards the middle; (1) widest before middle; (2) widest at posterior margin or from middle to posterior margin.
-
Length of pronotum setae: (0) long (more than 100 μm); (1) short (less than 50 μm).
-
Projection of anterior angles of pronotum: (0) strong; (1) weak or absent.
-
Diameter of elytra punctures: (0) reduced (less than 200 μm); (1) large (more than 200 μm).
The matrix was compiled using WinClada (
A simple sequential character removal analysis (modified after
To evaluate statistical branch support, a bootstrap analysis was conducted with NONA (
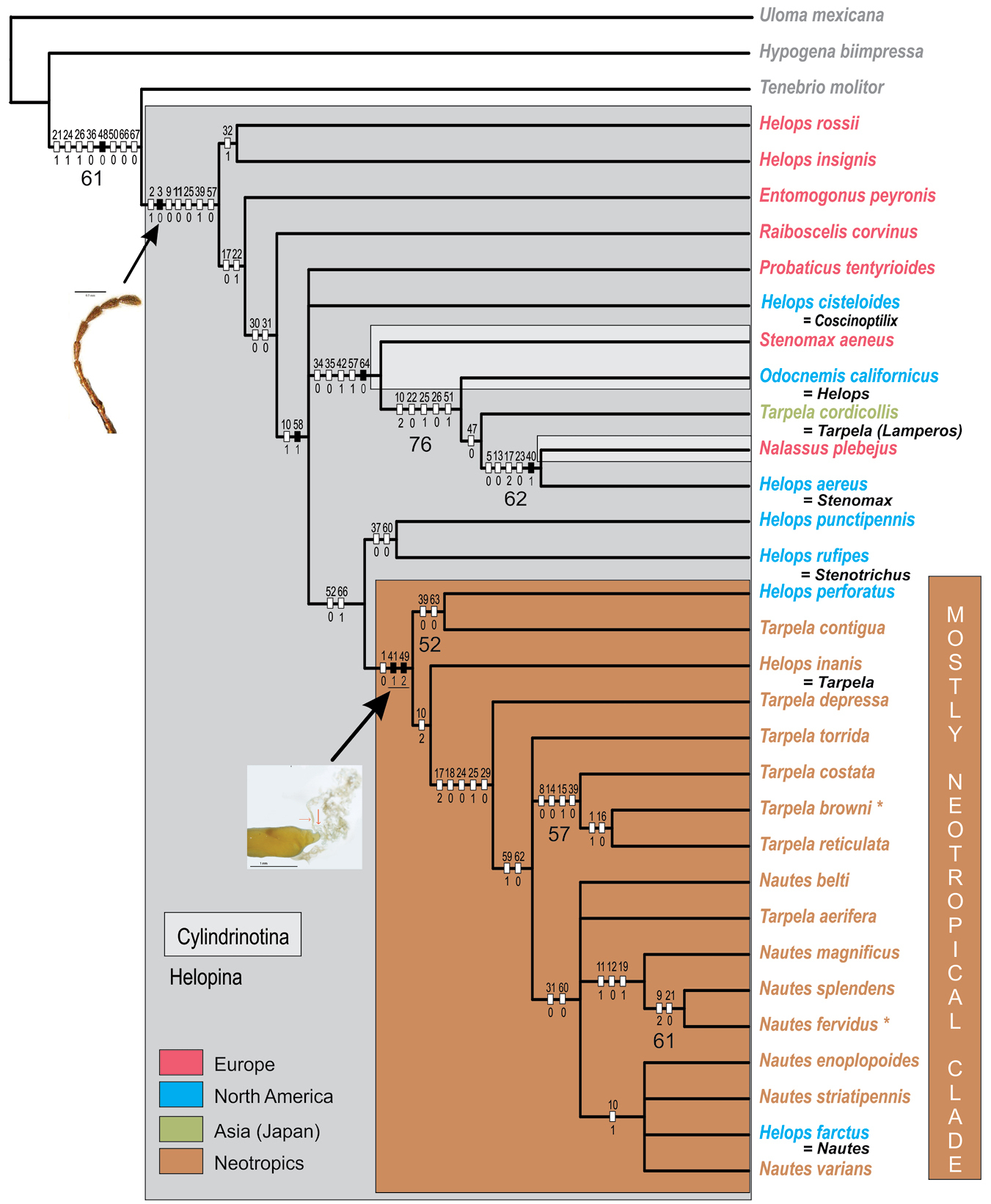
The 67-character matrix (Table 2) yielded 12 most parsimonious trees with 301 steps (length = L), a consistency index (ci) of 0.29, and a retention index (ri) of 0.59. The strict consensus (L = 314; ci = 0.28; ri = 0.56) is presented in Figure 7. Six out of seven characters retrieved as synapomorphies are from internal morphology. Four synapomorphies correspond to the female genitalia: vagina strongly curved in the apex (character 40: state 1), more than one spermathecal tube (41:1), smooth texture of spermathecal tube (48:0), and terminal position of the accessory gland (49:2). Two synapomorphies correspond to the male genitalia: distribution of evident setae on the parameres (58:1), and presence of a dorsal projection or keel on the parameres (64:0). One synapomorphy corresponds to external morphology: the filiform shape of the antennae: (3:0). Although only six clades had bootstrap values over 50%, most clades are supported by a unique combination of at least two characters.
Strict consensus of 12 most parsimonious trees (L = 314; ci = 0.28; ri = 0.56). Characters are mapped onto the consensus only if their optimization is not ambiguous and if they are present among all the MPTs. The consensus is used to map homoplasy at the level of characters. Black rectangles represent single, non-homoplasious character state transformations, and white rectangles represent multiple, homoplasious character state transformations. The number depicted above each rectangle represents the character and the number below the rectangle represents the character state. The bigger number below the branches corresponds to Bootstrap values over 50%. The combination of characters for each terminal is not shown. Three important synapomorphies are illustrated in the cladogram: the filiform antennae (3:0), as the single synapomorphy of the tribe, and the number of spermathecal tubes (41:1) plus the terminal position of the accessory gland (49:2), as the synapomorphies supporting a mostly Neotropical clade. These character states are reported for the first time for the tribe. Two shades of gray in the cladogram indicate the subtribe to which the terminals belong (except Nautes). Colors in the terminals indicate their geographic distribution. Below seven terminals the former classification (genus or subgenus) is shown. An asterisk indicates the type species included in the analysis: Tarpela browni and Nautes fervidus.
Matrix for the cladistics analysis of the tribe Helopini (Coleoptera, Tenebrioninae, Tenebrionini); “-” represents inapplicable character states, “?” represents not observed data.
| Taxon/character | 5 | 10 | 15 | 20 | 25 | 30 | 35 | 40 | 45 | 50 | 55 | 60 | 65 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uloma mexicana | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | ? | 1 | 0 | 0 | 3 | 0 | 0 | 0 | - | 0 | 1 | 1 | 0 | 1 | 1 | 2 | 0 | 2 | - | - | 0 | 0 | 1 | - | 0 | 0 | 1 | 2 | 1 | 1 | 1 | 2 | 2 |
| Helops punctipennis | 1 | 1 | 0 | 1 | 3 | 0 | 1 | 1 | 0 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 2 | 0 | 0 | 0 | - | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 1 |
| Entomogonus peyronis | 1 | 1 | 0 | 0 | 0 | 0 | ? | 1 | 0 | 0 | 0 | 1 | 1 | 2 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | - | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | - | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | ? | ? | ? | 1 | 0 | 0 | 0 | 0 | 1 | - | 0 | 1 | 1 | 0 | ? | 0 |
| Raiboscelis corvinus | 0 | 1 | 0 | 0 | 3 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 3 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | - | 1 | 1 | 1 | 0 | 0 | 2 | ? | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | - | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | - | 0 | 1 | 1 | 0 | 0 | 0 |
| Probaticus tentyrioides | 1 | 1 | 0 | 0 | 2 | 0 | 2 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 3 | 1 | 0 | 1 | 0 | 0 | 1 | - | - | 1 | 0 | - | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | - | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | - | 0 | 1 | 1 | 0 | 0 | 0 |
| Helops rossii | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | - | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | - | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | - | 0 | 1 | 1 | 0 | 0 | 0 |
| Helops insignis | 1 | 1 | 0 | 0 | 3 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | - | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | - | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | - | 0 | 1 | 1 | 0 | 0 | 0 |
| Helops cisteloides | 1 | 1 | 0 | 0 | 2 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | - | 1 | 1 | 0 | 0 | 0 | 2 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | - | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | - | 0 | 1 | 1 | 0 | 0 | 0 |
| Nautes enoplopoides | 0 | 1 | 0 | 1 | ? | 0 | 1 | 1 | 2 | - | 0 | 1 | 2 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | ? | 0 | 1 | 1 | 0 | - | 1 | 0 | 1 | 0 | 0 | 2 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | - | - | 1 | 0 | 2 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 1 | 1 |
| Helops perforatus | 0 | 1 | 0 | 0 | 2 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 2 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | - | 1 | 0 | 1 | 1 | 1 | 1 |
| Nautes striatipennis | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 2 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| Helops farctus | 0 | 1 | 0 | 1 | 2 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | - | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 |
| Nautes splendens | 0 | 1 | 0 | 1 | 2 | 0 | 2 | 1 | 2 | 2 | 1 | 0 | 0 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | ? | ? | ? | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| Tarpela browni | 1 | 1 | 0 | 1 | ? | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 0 | 2 | 0 | 0 | 1 | ? | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 0 | 1 |
| Tarpela torrida | 0 | 1 | 0 | 1 | 2 | 0 | 2 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 1 | 1 | ? | 1 | 2 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 1 | 1 |
| Nautes magnificus | 0 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 1 | 1 | 1 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| Tarpela depressa | 0 | 1 | 0 | 0 | ? | 0 | 1 | 1 | 1 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 2 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 2 | 0 | - | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | - | 1 | 0 | 1 | 1 | 1 | 1 |
| Nautes varians | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | - | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 |
| Tarpela contigua | 0 | 0 | 0 | 1 | 2 | 0 | 2 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 0 | 1 | ? | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | - | 1 | 0 | 1 | 1 | 1 | 1 |
| Nautes belti | 0 | 1 | 0 | 1 | 2 | 0 | 2 | 1 | 1 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 2 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 2 | 1 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| Nautes fervidus | 0 | 1 | 0 | 1 | 2 | 0 | ? | 1 | 2 | 2 | 1 | 0 | 0 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | - | - | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| Tarpela aerifera | 0 | 1 | 0 | 0 | 2 | 0 | 2 | 1 | 1 | 2 | 0 | 1 | 1 | 0 | 0 | 0 | 2 | 0 | 0 | 1 | 1 | 1 | 2 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | - | 0 | 1 | 0 | 2 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 |
| Tarpela reticulata | 1 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 1 | ? | 1 | 2 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | - | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 1 | 1 |
| Helops rufipes | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 2 | 2 | 3 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | - | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | - | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 0 |
| Helops inanis | 0 | 1 | 0 | 0 | 2 | 0 | 0 | 1 | 0 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | - | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | - | 1 | 1 | 1 | 1 | 1 | 1 |
| Tarpela costata | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 2 | 0 | 0 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 0 | 1 |
| Nalassus plebejus | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 2 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 2 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | - | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | - | 0 | 1 | - | 1 | 1 | 0 | 1 | 0 | 0 |
| Helops aereus | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | - | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | - | 0 | 2 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | - | 0 | 1 | - | 1 | 1 | 0 | 1 | 0 | 0 |
| Odocnemis californicus | 1 | 1 | 0 | 0 | 2 | 0 | 1 | 1 | 0 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | - | 0 | 2 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | - | 0 | 1 | - | 1 | 1 | 0 | 1 | 0 | 0 |
| Stenomax aeneus | 1 | 1 | 0 | 0 | 2 | 0 | 2 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | - | 0 | 0 | 1 | 1 | 0 | 1 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | - | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| Tarpela cordicollis | 1 | 1 | 0 | 0 | 2 | 0 | 2 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 2 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | - | 0 | 2 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | - | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 |
| Hypogena biimpressa | 1 | 0 | 1 | 0 | 2 | 1 | 2 | 1 | 1 | 0 | 2 | 0 | 0 | 1 | 0 | 1 | 2 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 1 | 0 | 2 | 0 | 0 | 1 | - | 1 | - | - | 1 | 1 | - | 2 | 1 | 1 | 0 | 0 | 1 | 0 | 1 | - | 0 | 1 | - | 0 | 1 | 1 | 0 | 2 | 2 |
| Tenebrio molitor | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 2 | 1 | 1 | 1 | 2 | 0 | 1 | 2 | 1 | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 2 | 0 | 0 | 1 | 0 | 0 | 1 | 3 | 0 | 0 | 0 | - | 1 | - | - | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | - | 0 | 1 | - | 0 | 1 | 1 | 1 | 0 | 0 |
The consensus shows that the monophyly of the tribe Helopini is supported by one synapomorphy: the filiform antennae (3:0). In contrast, none of the subordinated taxa within Helopini is supported as monophyletic: neither the subtribes (Cylindrinotina and Helopina) nor the genera represented by more than one species: Helops, Nautes, or Tarpela. Cylindrinotina is nested within Helopina and Tarpela cordicollis (Marseul, 1824) plus Helops aereus Germar, 1824 (Helopina) are in turn nested within Cylindrinotina. Helops and Tarpela are polyphyletic, while Nautes is paraphyletic (Helops farctus LeConte, 1858, at some point transferred to Nautes, and Tarpela aerifera Allard, 1876 share a common ancestor with it).
From the sampled Palearctic Helopina, only Helops rossii Germar, 1817 and Helops insignis Lucas, 1846 constitute a clade that is sister to the rest of the tribe, and is supported by the pubescent ventrites with homogeneous punctures (32:1).
An heterogeneous clade formed by three genera of Cylindrinotina, Stenomax aeneus, Scopoli, 1763, Odocnemis californicus (Mannerheim, 1843) and Nalassus plebejus Küster, 1850 plus two species of Helopina: Tarpela cordicollis and Helops aereus is supported by the following internal characters: evident arms of the spiculum ventrale (34:0), dilated distal end of stalk of the spiculum ventrale (35:0), parameres without evident setae (57:1), and parameres with a keel (64:0), the last recovered as a synapomorphy. Helops aeneus was placed in Stenomax by
A large clade of mostly Neotropical species from the genera Helops, Nautes and Tarpela, plus two Nearctic species of Helops, was recovered with support from three characters: clypeus slightly depressed (1:0), more than one spermathecal tube (41:1) (retrieved as synapomorphy), and an accessory gland terminal to the spermathecal tubes (49:2) (retrieved as synapomorphy). Helops punctipennis LeConte, 1870 and Helops rufipes (LeConte, 1851), both Neartic, are supported as sister to this mostly Neotropical clade by two internal characters: the deep notch of the eighth sternite (52:0) and the lobate shape of the median lobe (66:1). The earlier divergent lineage within this mostly Neotropical species is a clade formed by Tarpela contigua Champion, 1887 and Helops perforatus Horn, 1880, supported by two internal characters: paraproct three or more times longer than the coxite (39:0) and the presence of a ventral projection at the parameres apex (63:0). Helops inanis Allard, 1877 and Tarpela depressa Champion, 1887 form a grade with respect to the remaining mostly Neotropical species. There is then a polytomy that includes Tarpela torrida
Although supported by our results, the monophyly of the tribe still requires a more rigorous test including a wider sample of species from more tribes including species from other closely related tribes (e.g. Triboliini, Blaptini). The only synapomorphy supporting the tribe, the filiform shape of the antennae, could be an artifact of our sampling, as the antenna have also been reported as moniliform or gradually clavate within the tribe (
The fact that Cylindrinotina is nested within Helopina implies that there is no justification for the recognition of two subtribes: either no subtribes should be recognized or more subtribes should be recognized. A denser sampling of Palearctic species could help reveal which of these alternatives is better supported. According to the current sampling, it is possible that the Palearctic Helops remain as an independent earlier divergent lineage within the tribe, including the type species (Helops caeruleus), which is morphologically similar to the sampled Paleartic species. If this was the case, Helops would have to be re-circumscribed to include only the Palearctic species and new generic names would be necessary for the New World lineages.
Further earlier divergent lineages may be revealed as sampling of Entomogonus, Raiboscelis and Probaticus is improved, as well as other genera not included in our sampling (e.g. Catomus Allard, 1876, Hedyphanes Fischer von Waldheim, 1820, and Nesotes Allard, 1876). The unresolved position of Helops cisteloides Germar, 1824 indicates the possibility that other New World lineages could be identified as sampling is increased. If subtribes are to be recognized, Cylindrinotina would need to be expanded to include Asian species of Tarpela (as Tarpela cordicollis) and Neartic Helops (as Helops aereus). This subtribe would also have to include several Holartic genera (besides Odocnemis). The Holarctic region has an intricate history (
The polyphyletic nature of Helops and Tarpela render Champion’s classification (
The paraphyletic nature of Tarpela with respect to Nautes, could imply different outcomes as a wider taxon sampling (including more Nearctic species of Helops, Nautes and mainly Tarpela) and character (e.g. from DNA or fine structures revealed using SEM) is considered. Either several lineages could be recognized as different genera or all the species could be lumped in a single larger genus (Nautes due to nomenclatural priority, or if applicable, a conserved name Tarpela). Even if Nautes was supported as a different genus, taxonomic rearrangements seem to be likely. According to the current sampling, Helops farctus and Tarpela aerifera would need to be reassigned to Nautes.
Female genitalia have been used as a source of characters to study the relationships among suprageneric taxa in Tenebrionidae (
Due to its high variation, male genitalia have also been used to explore the relationships among species and higher taxonomic groups (e.g.
The recognition of the female and male genitalia types is translated into several homology hypotheses reflected in the matrix as characters 33 to 67 and their corresponding character states (see the list of phylogenetic data: characters above).
Although widely used as a taxonomic character, the keel on the parameres (64) has been reported as not always present through the subtribe (
Other diagnostic or traditionally used characters of the clypeus, antennae, prosternum, wings and tarsi were homoplastic but generally informative, contributing to the overall resolution of the tree. Only four characters from the original matrix introduced high levels of conflict, resulting in a lack of resolution in the consensus. These characters were all continuous and without a more refine codification, e.g., using statistical or morphometric tools, they only obscured the relationships posed by the remaining characters. On the other extreme, the shape of the antenna, generally considered to be a homoplastic character, was recovered as synapomorphic for the tribe. However, this synapomorphy needs to be tested with a broader taxon sampling.
Although supported by our results, the monophyly of the tribe still requires a more rigorous test in terms of the taxon sampling from related tribes.
None of the subtribes or the analyzed subordinate genera of Helopini sampled by more than one species was corroborated as monophyletic. A wider taxon sampling is required to circumscribe them in a natural way.
Helops and Tarpela are polyphyletic, while Nautes is paraphyletic, and hence it is expected that further taxon and character sampling in a cladistic context will provide evidence for further splitting of Helops and Tarpela and a re-circumscription of Nautes including some Helops and Tarpela.
Our results show that in order to achieve a natural classification of Helopini, sampling of taxa should not be based on geographic distribution, although there might be some geographically correlated lineages. This approach has shown that there is a derived New World clade that is mainly composed by Neotropical species. Future efforts should also concentrate on increased sampling within this clade, to reveal other lineages or to corroborate the current ones, so that taxonomic changes can be concordantly proposed.
The authors would like to thank the steering committee of the Third International Tenebrionoidea Symposium for the invitation to present our results, especially to Dr Aaron Smith, for his help and useful comments. We are also grateful to the curators of the collections mentioned in the materials and methods section for loaning the specimens used in this work. We greatly appreciate the revision and valuable comments by Dr E. Nearns and Dr M. Zurita-García, as well as those by two anonymous reviewers and most particularly to the associate editor, Dr Patrice Bouchard, who not only provided relevant comments, but was also very understanding and helpful during the publication procedure. We would like to thank S. Guzmán (IBUNAM) for her help with the use of the Leica equipment. The first author thanks the Posgrado en Ciencias Biológicas, UNAM, for its support and the fund of the “Programa de Apoyo para Estudios de Posgrado” provided to visit the entomology collections at the Natural History Museum (BMNH) and the Muséum National d’Histoire Naturelle (MNHN). This study was funded with a doctoral fellowship from the Consejo Nacional de Ciencia y Tecnología (CONACYT 202666) to the first author.