(C) 2013 Donald M. Windsor. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
Citation: Windsor DM, Dury GJ, Frieiro-Costa FA, Lanckowsky S, Pasteels JM (2013) Subsocial Neotropical Doryphorini (Chrysomelidae, Chrysomelinae): new observations on behavior, host plants and systematics. In: Jolivet P, Santiago-Blay J, Schmitt M (Eds) Research on Chrysomelidae 4. ZooKeys 332: 71–93. doi: 10.3897/zookeys.332.5199
A summary of literature, documented observations and field studies finds evidence that mothers actively defend offspring in at least eight species and three genera of Neotropical Chrysomelinae associated with two host plant families. Reports on three Doryphora species reveal that all are oviparous and feed on vines in the Apocyanaceae. Mothers in the two subsocial species defend eggs and larvae by straddling, blocking access at the petiole and greeting potential predators with leaf-shaking and jerky advances. A less aggressive form of maternal care is found in two Platyphora and four Proseicela species associated with Solanaceae, shrubs and small trees. For these and other morphologically similar taxa associated with Solanaceae, genetic distances support morphology-based taxonomy at the species level, reveal one new species, but raise questions regarding boundaries separating genera. We urge continued study of these magnificent insects, their enemies and their defenses, both behavioral and chemical, especially in forests along the eastern versant of the Central and South American cordillera.
Maternal care, Subsociality, Coleoptera, Chrysomelidae, Chrysomelinae, Neotropical
Arthropod parents influence the survival prospects of offspring in a multitude of ways. When parents bring resources to larvae, guide larvae to resources or actively shield offspring from predators and parasitoids, they are engaging in subsocial behavior (
The Coleoptera include numerous examples of independently evolved subsocial behavior where the importance of competition, resource provisioning and defensive parental behavior can be examined (
Below we review evidence of restricted host plant use and the presence of defensive maternal behavior in eight species of Neotropical Chrysomelinae. Detailed observations from one of these species suggests that mothers modify leaf resources in advance of bearing live offspring, and later block and herd movement of larvae among leaves on the same food plant. To clarify species relationships among Solanaceae-feeding species, some varying only in minor aspects of elytra color, we present genetic distance estimates obtained from mitochondrial sequence data for eleven described and one undescribed species, including both maternal care and non-care species.
Tissue samples are preserved in ethanol at -80° C, and pinned adult voucher specimens are stored in the working collections of D.W., both at the Smithsonian Tropical Research Institute, Tupper Research and Conference Center, Panama City, Panama. Field observations reported below come from field notes and photographic records made at diverse occasions in Central and South America over the past 20 years.
Adult Chrysomelinae were stored in 95% ethanol at -80° C, flight muscle removed and ground in 180 µl ATL tissue lysis buffer (Qiagen Inc., Valencia, CA USA) and 20 µl proteinase K with a sterile pestle, vortexed for 10 s and incubated overnight at 55° C. Following incubation, 200 µl AL lysis buffer (Qiagen Inc.) was added and the sample was heated at 70° C for 10 min, then 200 µl molecular grade ethanol was added to each sample. This mixture was then pipetted into a DNeasy mini spin column and centrifuged at 8000 rpm (~6000 g) for 1 min, then the flow-through and collection tube were discarded. The DNeasy mini spin column was placed in a new 2-ml collection tube and 500 µl wash buffer AW1 (Qiagen, Inc.) was added, the sample was centrifuged for 1 min at 8000 rpm, then the flow-through and collection tube were discarded. Again a new collection tube was used, 500 µl wash buffer AW2 (Qiagen, Inc.) was added and the sample centrifuged for 3 min at 14, 000 rpm (20, 000 g); the collection tube was then discarded. The mini column was placed in a 1.5-ml tube and 200 µl AE elution buffer (Qiagen, Inc.) was added, the sample was incubated for 2.5 min at room temperature, and the sample centrifuged for 1 min at 8000 rpm (~6000 g). Extractions were held at -20° C between use, and at -80° C for long-term storage. The primers: C1-J-1718F (26-mer; 5’-GGA GGA TTT GGA AAT TGA TTA GTT CC-3’) and C1-N-2191 (26-mer; CCC GGT AAA ATT AAA ATA TAA ACT TC-3’) (
Forward and Reverse sequences were combined and reconciled in Sequencher v5 (Gene Codes Corporation, Ann Arbor, MI, USA) and trimmed, leaving a single 472 bp fragment, which was then translated to amino acids and found free of stop codons. Sequences from ten species were combined with three sequences from GenBank creating an ingroup of 12 species (20 individuals) and a single outgroup species. Where possible we included two separate individuals from the same population. New sequences were deposited in GenBank under accession numbers in Table 1. Evolutionary relationships of the samples were inferred by Bayesian analysis, with 2 million generations, and Maximum Likelihood analysis, with 100 bootstrap pseudo-replications. The ideal partitioning strategy and models of nucleotide substitution were determined using PartitionFinder v.1.0.1 (
Apocyanaceae and Solanaceae feeding taxa mentioned in the text, collection and host plant information, life history characteristics, accession numbers and references.
| Chrysomelinae species | Location | Host Plant Family |
Host Plant Species | Larval Group Defense | Maternal Care | Reproduction | Genbank Accession Numbers | References |
|---|---|---|---|---|---|---|---|---|
| Doryphora paykulli (Stål, 1859) | Gamboa, Panama Province, Panama | Apo | Prestonia seemannii Miers (subfamily Apocynoideae) | aggregated | yes | oviparous | - | new observation |
| Doryphora reticulata (Fabricius, 1787) | Boqueirão Reserve, Minas Gerais State, Brazil | Apo | Prestonia tomentosa R. Br. (subfamily Apocynoideae) | aggregated | yes | oviparous | - | new observation |
| Doryphora sp. near Doryphora punctatissima (Olivier, 1790) | El Porvenir, Meta Province, Colombia | Apo | Prestonia isthmica Woodson (subfamily Apocynoideae) | aggregated | no | oviparous | - | |
| Eugonycha melanostoma (Stål, 1859) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum sp. | aggregated | no | larviparous | - | |
| Platyphora amabilis (Baly, 1859) | Yasuní, Orellana Province, Ecuador | Sol | Solanum sp. | ? | ? | ? | AY055517 | new observation |
| Platyphora anastomozans (Perty, 1832) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum bullatum Vell., Solanum muritianum (Scopoli), Solanum sancta-catarine Dunal, Solanum megalochiton Mart., Solanum rufescens Sendt. | aggregated | no | larviparous | KF251110, KF251111 | |
| Platyphora aulica (Olivier, 1807) | Montagne de Kaw, Roura Commune, French Guiana | Sol | Solanum rugosum Dunal, Solanum torvum Sw. | solitary | no | larviparous | KF251112, KF251113 | new observation |
| Platyphora conviva (Stål, 1858) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum spp. | aggregated |
no | larviparous | - | |
| Platyphora fasciatomaculata (Stål, 1857) | Ijuí, Rio Grande do Sul State, Brazil | Sol | Solanum mauritianum | aggregated | no | larviparous | - | |
| Platyphora microspina Bechyné, 1954 | Cerro Campana, Panama Province | Sol | Markea megalandra (Dunal) | aggregated | yes | larviparous | KF251120 | new observation |
| Platyphora nigronotata (Stål, 1857) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum bullatum, Solanum muritianum, Solanum sancta-catarine, Solanum megalochiton | aggregated | no | larviparous | KF251121 | |
| Platyphora nitidissima (Stål, 1857) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum bullatum | aggregated | no | larviparous | - | |
| Platyphora quadrisignata (Germar, 1824) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum variabile Mart., Solanum fastigatum Willd | aggregated |
no | larviparous | - | |
| Platyphora selva Daccordi, 1994 | La Selva Biological Station, Heredia Province, Costa Rica | Sol | Solanum (Witheringia) heteroclita. | aggregated | yes | larviparous | - | |
| Platyphora sphaerica (Jacoby, 1903) | Serra de Baturite, Fortaleza, Brazil | Sol | Solanaceae, 2 spp. | solitary | no | larviparous | AY055529 | |
| Platyphora vinula (Stål, 1858) | Ijuí, Rio Grande do Sul State, Brazil | Sol | Solanum sp. aff. Solanum megalochiton, Solanum sancta-catarinae | aggregated | no | larviparous | - | |
| Proseicela antennalis (Kirsch, 1883) | Reventador, Napo Province, Ecuador | Sol | Solanum sp. | ? | ? | ? | KF251114, KF251115 | new observation |
| Proseicela bicruciata Jacoby, 1880 | Yanayacu Biological Station, Napo Province, Ecuador | Sol | Solanum abitaguense S. Knapp | aggregated | yes | larviparous | KF251116, KF251117 | new observation |
| Proseicela crucigera (Sahlberg, 1823) | Serra do Japi, Jundiaí, São Paulo State, Brazil | Sol | Solanum decompositiflorum Sendtn. | aggregated | no | larviparous | - | |
| Proseicela flavipennis (Erichson, 1847) | Reventador, Napo Province, Ecuador | Sol | Solanum abitaguense S. Knapp | ? | ? | ? | KF251118, KF251119 | new observation |
| Proseicela sp. n. “Yasuni” | Yasuní, Orellana Province, Ecuador | Sol | Cuatresia sp. | aggregated | yes | larviparous | KF251126, KF251127 | new observation |
| Proseicela spectabilis (Baly, 1858) | Río Malo & Reventador, Napo Province, Ecuador | Sol | Solanum abitaguense S. Knapp, Solanum sp. (sect. Dulcamara) | aggregated | yes | larviparous | KF251122, KF251123 | new observation |
| Proseicela vittata (Fabricius, 1781) | Montagne de Kaw, Roura Commune, French Guiana | Sol | Solanum morii S. Knapp | aggregated | yes | larviparous | KF251124, KF251125 | new observation |
| Stilodes modesta Jacoby, 1882 | Cerro Campana, Panama Province, Panama | Mal | Banisteriopsis sp. | aggregated | no | oviparous | AY055522 | new observation |
1 Apo=Apocynaceae, Ast=Asteraceae, Sol=Solanaceae, Mal=Malpighiaceae
2=nocturnally active
3=larvae cut and cover themselves with trichomes
According to
Maternal care providing Doryphora species, a Doryphora paykulli female with eggs and first instar larvae under an apical leaf of Prestonia seemanii (photo by S.L.) b female straddling a mix of first and second instar larvae (photo by S.L.) c Doryphora paykulli larvae moving to a new leaf followed by their mother (photo by S. Van Bael) d Doryphora paykulli larvae stripping the cortex of their host while descending in pairs to pupate, (photo by D.W.) e Doryphora reticulata ovipositing under apical leaf of Prestonia tomentosa in Central Brazil (photo by F.F.) f Doryphora reticulata larvae on the natal leaf (photo by F.F.) g female Doryphora reticulata stradding first instar larvae (photo by F.F.) h Doryphora reticulata female tending fully-developed larvae at the base of the food plant just prior to pupating underground (photo by F.F.).
Subsequently, individual Doryphora paykulli adults were observed (D.W., S.L.) during late April and early May of 2005 moving slowly and feeding among low vegetation. Pairs of individuals were observed interacting aggressively on the small leaf fragments remaining on the host plant, Prestonia seemannii Miers (Apocynaceae) under late dry season conditions along trails in the Parque Metropolitano (elevation 30 m, 8°59.24'N, 79°32.797'W), Panama City. Whether these were contests over resources or precopulatory courtship is unclear, however, analysis of video taken of one of these interactions shows the use of the mesosternal horn in dislodging a competitor, much as described by
Larval emergence began on days 6 and 7 following first oviposition with clutches (n=3) at that time containing from 40–50 eggs. Within 24 hours after larvae began to emerge many of the original eggs were missing, apparently consumed by early-emerging larvae such that clutches were reduced to 8, 15 and 20 surviving larvae accompanied by some intact, opaque, less-developed eggs and opened eggs with red-colored residues of once-developing larvae visible under close inspection (Fig. 1). One day later an adult was observed (S.L.) returning to a natal group after having fallen from the plant on a detached leaf, then deposit a single egg which was immediately consumed by a nearby larva. First instar larvae (1.5–2.0 mm in length) had small black heads at the time of emergence, these easily distinguished them from second instar larvae appearing, 1 to 2 days later with red head capsules and a larger and more rotund appearance. Larvae expanded rapidly in size following their first meals but did not feed on leaf tissue until after the first molt, 2 to 3 days after emerging from the egg. Mothers at times tightly straddled their aggregated first and second instar larvae on the natal leaf, preventing their advance down the leaf petiole (Fig. 1b). While guarding, mothers reacted aggressively by charging to the edge of the leaf when a thin stick was introduced to the area by an observer. Charges, stamping and shaking continued for at least two minutes after the stimulus was presented and removed. The strongest reaction was given to a camera held approximately 10 cm under and to the side of the natal leaf. The mother seemed to be reacting to the camera lens–suggesting that a mirror held near guarding mothers might provide a non-invasive means of assessing defensiveness. On one occasion an Ectatomma tuberculatum (Olivier) worker was observed to pass by the base of the petiole, eliciting aggressive shaking of the natal leaf and short charges, after which the ant reversed course and departed that portion of the plant. As larvae became larger and began moving between leaves the intensity of the mothers’ reactions to foreign stimuli appeared to subside. Increasingly, mothers were seen feeding on leaves and leaf petioles, rather than guarding, as larval development proceeded.
Once the natal leaf was consumed, larvae began moving down the petiole to the stem, where they then moved either up or down in smaller groups to other leaves, sometimes moving as solitary individuals. Mothers, often fed from the pedicel of the leaf just consumed, occasionally accompanied by one or two larvae. Mothers actively trampled upon the backs of larvae still located on the pedicel, in effect pushing them away from the leaf and toward the stem. Mothers on other occasions stepped on and over larvae, rapidly tapping larvae with antennae and tarsi until they reversed direction. After leaving the natal leaf, mothers resumed guarding one of the several larval groups that reassembled. However, some groups continued to split into ever smaller units and moved to adjoining leaves and stems, leaving mothers guarding smaller sets of offspring and spending more time travelling among groups in what seemed to the observer as an effort to herd offspring back together (Fig. 1c). Mothers also increasingly divided their time between guarding and feeding on the cortex of the stem, girdling the vine over distances from a few cm to nearly 1 m. Seven days following eclosion larvae were arranged largely in doublets, girdling stems as intact leaves had disappeared in substantial sections of the plant. Larvae then descended in unguarded pairs to pupation sites by backing down the stem from which all cortex was stripped (Fig. 1d), effectively killing that section of the plant. By day eleven, most larvae had descended the host plant and moved along small above-ground roots into the leaf litter. One mother was last seen guarding two slow-developing larvae high (2 m) on the plant thirteen days after oviposition. Development from first oviposition to larvae wandering on the ground took approximately 20 days. Several larvae collected and placed in a plastic container with moist leaves, molted at day 4 and eclosed as teneral adults on days 18 and 19, a metamorphosis period slightly shorter than the 24 days estimated for Doryphora sp. near punctatissima by
Recent observations by F.F. reveal clearly that maternal care is expressed by Doryphora (Megistomela) reticulata (Fabr.) in the cerrado of south-central Brazil (Fig. 5) (see also photo in
Within New World Chrysomelinae, reports of subsociality until recently were limited to a single species studied at the La Selva Field station in the Atlantic lowlands of Costa Rica (
Platyphora microspina was initially observed on Cerro Campana (Parque Nacional Altos de Campana), along a ridgeline approximately 50 m west and up-slope from the Podocarpus trail in July 1999 (elevation 900 m; 8°41.07'N, 79°55.82'W). Large numbers of adult and immature beetles were observed feeding on Markea megalandra (Dunal), a woody hemiepiphyte which grows within the canopy of forests at elevations of 1000–2000 m in Western and Central Panama (
Maternal care providing Platyphora microspina in Panama, a female with recently deposited larvae (photo by D.W.) b female guarding mid-sized larvae (photo by D.W.) c female and young larval brood moving among leaves (photo by D.W.) d female tending overlapping cohorts of larvae (photo by D.W.).
Female Proseicela vittata (Fig. 3a) were observed by D.W. tending offspring during each of three visits to Montagne de Kaw, French Guiana (elevation 150 m; 4°32.686'N, 52°09.151'W), 11–18 April 2010, 25–31 January 2011 and 19 June–2 July 2012. All individuals and family groups were found on a single species of host plant, Solanum morii S. Knapp, a 1-3 m shrub with glabrous leaves and pendulous green and white fruit and a colonist of disturbed areas (Plate 121a,
Maternal care providing Proseicela species, a Proseicela vittata adult (Photo by D.W.) b Proseicela vittata female and larvae from two cohorts. Insert shows detail of vein pinching along approximately 1cm of the primary vein (Photo by D.W.) c Proseicela vittata female with late stage larvae (Photo by D.W.) d Proseicela bicruciata adult female, (photo by G.D.) e Proseicela bicruciata female tending larvae (photo by G.D.) f Proseicela bicruciata food plant, Solanum abitaguense (photo by G.D.) g Proseicela spectabilis adult (photo by G.D.) h Proseicela spectabilis with nearly full-grown larval brood and tachinid parasitoid (photo by G.D.) i. Proseicela spectabilis host plant, Solanum sp. (photo by G.D.) j Proseicela sp. n. adult female (photo by G.D.) k the same female tending three feeding larvae feeding on Cuatresia sp. (Solanaceae) (photo by G.D.) l wider view of the host plant (photo by G.D.).
The 17 larvae belonging to another female were observed to take approximately 30 hours to consume the entire lamina of the natal leaf. While the last of the leaf was being consumed some larvae began to molt while still on the remnants of the natal leaf. The mother maintained a tight grip on the leaf petiole (blocking behavior), but eventually larvae pushed by and began traversing nearby stem and petioles solitarily or in small groups. Commonly families split into two or more separated feeding groups at this stage, with the mother usually remaining with a larger group. Groups often reunited but others remained separated until pupation. The transition from the natal to second leaf appears to be a crucial and dynamic time for larvae and events proceeded differently for most groups. During this period mothers moved actively among different leaves and branches in what seemed to be attempts to herd and reconstitute a single larval group. While we observed what we interpret as herding behavior in most species in this report, its possible importance to group safety and success remains open and in need of experimental study.
Strong indications of maternal care in Proseicela bicruciata (Fig. 3d) were gathered by G.D. while walking trails in the montane cloud forest of the Yanayacu Biological Station and Center for Creative Studies, Napo Province, Ecuador (2150 m elevation, 0°36.27'S, 77°53.25'W) during the first week of July 2011. A total of five females were found, each tending small groups of uniform larvae (Fig. 3e), on the undersides of large leaves of Solanum abitaguense S. Knapp growing in streamside habitats (Fig. 3f). The group containing the smallest larvae was composed of nine individuals, while groups with larger larvae contained five and six individuals. The group with five larvae had two individuals separated on leaves 20–30 cm in different directions from the central three larvae arranged in a small rosette with heads to the inside and the mother to the side.
Observations of Proseicela spectabilis were taken by G.D. while walking the main trail leading to the Cascadas de San Rafael, Reventador, Napo province, Ecuador (1300 m elevation; 0°6.07'S, 77°35.18'W) on July 17, 2011. A single female (Fig. 3g), was perched half above its larvae and half on the branch of its food plant. Six of the seven larvae in the formation were tightly aggregated around the thin stem of the plant, while the seventh larva was on a leaf a few centimeters away. A tachinid fly was present on the dorsal surface of a larva located on the side opposite the mother (Fig. 3h). The fly departed when the observer approached but promptly returned to land on the larva opposite the mother. The host plant (Fig. 3i) was later identified as a nightshade, Solanum sp. section Dulcamara.
Remarks. A single Proseicela adult tending a group of three larvae (Fig. 3j, k) was observed and photographed by G.D. while walking a trail leading to the 50 ha forest dynamics plot, within the Estación Científica Yasuní (ECY), Orellana province, Ecuador (220 m elevation; 0°40.83'S, 76°23.89'W) on 15 July 2011. Following discovery the larvae formed a small rosette with heads to the inside. The female and larvae were attached to the underside of a leaf of Cuatresia sp. (Solanaceae) (Fig. 3l). Two other adults of the same species were found nearby, one on a different branch of the same host and the other on an unidentified plant. According to M. Daccordi, this is an undescribed species.
Two additional Proseicela species are known from understory Solanaceae at Cascadas de San Rafael, Ecuador. Several Proseicela antennalis adults (Fig. 4a) were collected by D.W. and J.P. from unidentified Solanaceae, 12 August 2001. Additionally, one Proseicela flavipennis adult (Fig. 4b) was collected by G.D. at the same site on leaves of Lycianthes glandulosa (Ruiz & Pav.) Bitter, 17 July 2011. As none of the individuals in these two species were reproducing, their interactions with offspring remain unknown, however high morphological similarity to adults of other Proseicela species in the area suggest they are good candidates to be subsocial. Platyphora amabilis (Fig. 4c) adults were observed and collected from a well-armed solanaceous food plant growing in open, roadside habitats at the Estación Científica Yasuní (Fig. 4d) by D.W. during August 2001. Larvae of this species were not observed. Platyphora aulica (Fig. 4e) was observed on numerous occasions and collected from Solanum rugosum Dunal and Solanum torvum Sw. in the same roadside habitats and the same dates in French Guiana as Proseicela vittata. An adult female Platyphora aulica placed in a container with abundant food, deposited approximately one larva per day. Mothers of this species walk away from their live born larvae, leaving all to feed and develop as solitary individuals. Three additional Solanaceae-feeding species from Brazil are included in the analysis that follows. Platyphora anastomozans (Perty) and Platyphora nigronotata (Stal) specimens were collected from Serra do Japi, Brazil where aspects of their biology has been studied (
Other Solanaceae associated Chrysomelinae of unknown habits (a, b, c, g), known not to provide maternal care (d, e, f) and outgroup taxon (h), a Proseicela antennalis (Photo by D.W.) b Proseicela flavipennis (Photo by G.D.) c Platyphora amabilis (Photo by D.W.) d Platyphora aulica (Photo by D.W.) e Platyphora nigronotata (Photo by D.W.) f Platyphora anastomozans (Photo by D.W.) g Platyphora sphaerica (Photo by J.P.) h Stilodes modesta (Photo by D.W.).
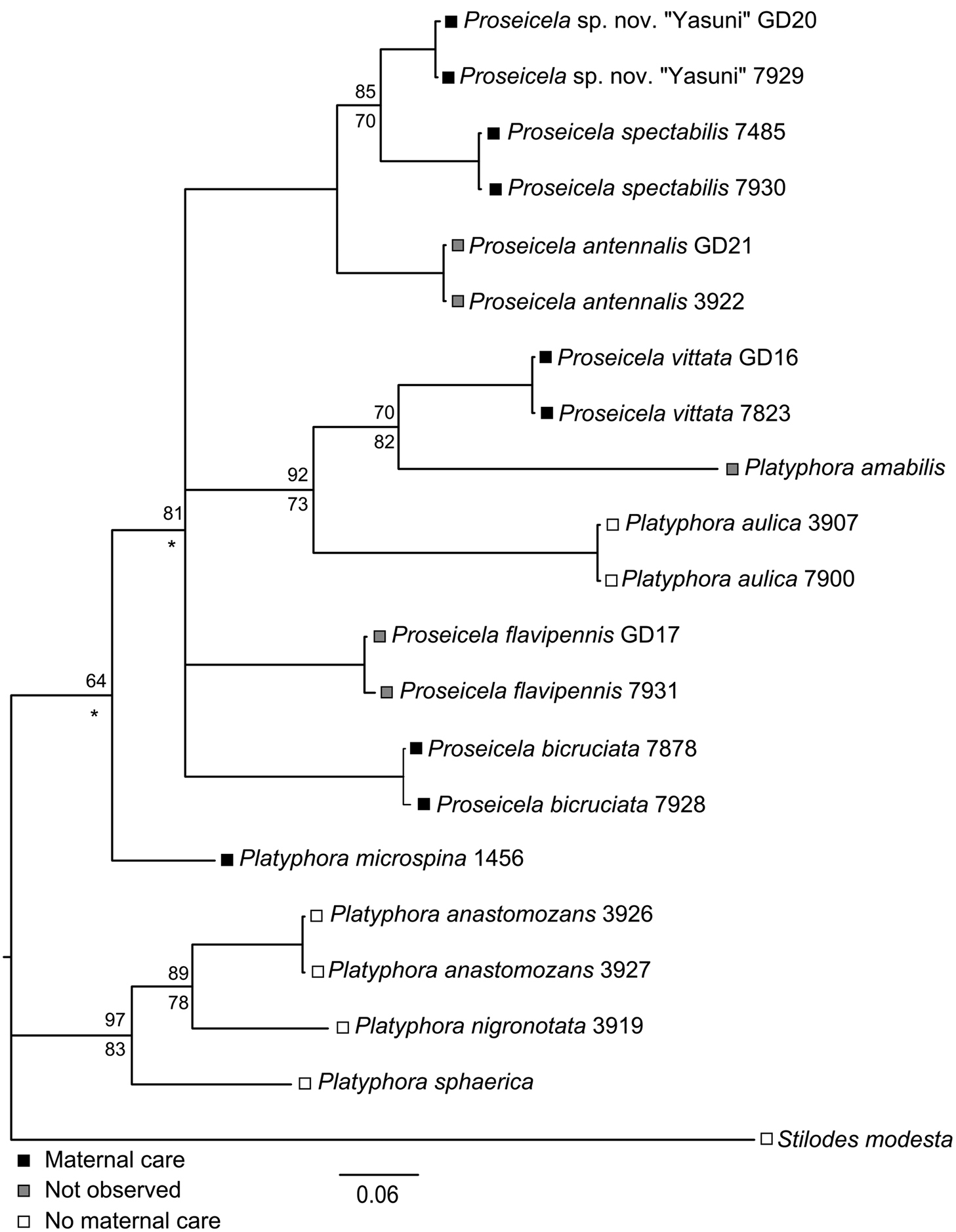
Two lines of evidence support the validity of the Solanum-feeding species recognized above. The first is the expert opinion of M. Daccordi who has reviewed each of the species in this report including voucher specimens and finds only one unidentified species, Proseicela sp. n. “Yasuni”. Nevertheless, while an experienced taxonomist can separate species, the criteria can be subtle and based on few characters. The five Ecuadorean Proseicela species differ only in subtle aspects of elytral and pronotal color pattern and for this reason we sought genetic evidence of species limits. The topology of the resulting consensus tree generated by Bayesian Inference (Fig. 5) resolved all taxa as separate entities, with Bayesian support values ranging from 64 to 100%, while Maximum Likelihood bootstrap estimates for nodes common to both trees ranged from 70 to 100%. The smallest pair-wise genetic distance estimates occurred between the pairs, Proseicela sp. n. “Yasuni”, Proseicela spectabilis (6.4%) and Proseicela antennalis (7.5%), and between Proseicela spectabilis and Proseicela antennalis (9.0%) with all remaining pair-wise distances ranging between 10.2 and 18.6%. Thus all mean distances fall above the 2 to 6% range considered a threshold for distinct species, depending on COI evolution rates within particular clades (
Bayesian Consensus tree of 472 bp COI sequences obtained for 12 species of Central and South American Solanaceae-feeding Doryphorini and one outgroup. For nodes with less than 100% support, Bayesian values are placed above node, Maximum Likelihood bootstrap values below the node, while asterisks (*) indicate nodes with different taxon placement under ML analysis and thus are not strictly comparable.
Aspects of the biology of 20 Neotropical species of Solanaceae-feeding Doryphorini (i.e. excluding Leptinotarsa and similar genera) are now known and where the mode of reproduction has been recorded, all (17) are live-bearing or “larviparous” (Table 1). Further, 15 species are reported to have aggregated larvae, while only two species (Platyphora aulica and Platyphora sphaerica) have solitary larvae. Maternal care is now known to occur in six species and known not to occur in another eleven species. While five Proseicela species are maternal care providers, Proseicela crucigera in south-central Brazil has gregarious larvae but mothers do not extend care after larviposition (João Vasconcellos-Neto, personal communication, 2013).
Because of the caveats to which mitochondrial data are subject (
New records of maternal care in New World Chrysomelinae are beginning to reveal patterns. The first is that maternal care behavior is primarily defensive in nature. Doryphora mothers straddle larvae and take measures to physically confront pedestrian predators. There is no convincing evidence that mothers act to supply or direct offspring to resources, although there are indications that Proseicela vittata mothers are modifying resources through vein-pinching. Proseicela and Platyphora mothers closely tend, straddle and herd offspring much as observed in Doryphora, but aggressive challenges of nearby insects or artificial stimuli have not been observed. For the most part, mothers remain immobile and close to their larvae (e.g. the tachinid parasitoid on the Proseicela spectabilis larva, Fig. 3h). But, elaborate vein-pinching by Proseicela vittata mothers on the natal leaf just prior to and after larvaposition brings a dimension to maternal care which is apparently absent in Doryphora species. Presently, we do not know whether vein-pinching behavior simply deactivates plant defensive canals (
As Doryphora species are specialist feeders on Apocynaceae it is entirely possible that adults have a defense system based on the dual sequestration of plant amyrins and lycopsamine-type alkaloids (
The second pattern beginning to emerge concerns choice of food plants. New World Chrysomelinae are specialist feeders on one of approximately nine different host plant families, yet all maternal care species to date are restricted to only two, Apocynaceae and Solanaceae. Clearly more documentation is desirable, but if this pattern were to continue then it will be important to look for attributes possessed by these two plant families which promote evolution of maternal defenses but which are absent in other families, such as the Malpighiaceae, a family which hosts many poorly known Chrysomelinae species in South America.
We note that all eight maternal care species in the New World occur in distinctly tropical latitudes whereas subsocial Gonioctena species occur from Europe (
Maternal care behavior appears limited to three genera of Neotropical Chrysomelinae and is not present in all species of these genera. Doryphora species are exclusively associated with lianas in the family Apocynaceae, possess a long mesosternal horn, are oviparous and reproduce at the transition from dry to wet seasons. Proseicela and some species currently placed within Platyphora are associated with Solanaceae host plants, and are live-bearing throughout the year. While extreme rareness remains an impediment to the study of most Doryphora species, Proseicela and allied species on the eastern slope of the Andes in Ecuador can be found more predictably on moderately common host plants. Large voids remain in our understanding of the natural history of both groups, including the identity and importance of predators and parasitoids and the diverse ways in which mothers may be influencing the survival of offspring.
We thank A. Valderama and E. Medianero (Universidad de Panamá) who provided key observations leading to the discovery of Platyphora microspina; S. Van Bael (STRI), G. Keller (STRI) and M. Cuignet (U. Leuven la Neuf, Belgium) who provided important observations on the location and behavior of Doryphora paykulli; L. Sekerka (National Museum, Prague, Czech Republic) for location and behavior of Proseicela in Ecuador and for suggestions on taxonomy, to C. Galdames (STRI, Panama) who identified host plants, and to M. Daccordi (Museo Civico di Storia Naturale, Verona, Italy) who kindly identified beetles. We also thank two anonymous reviewers for their generous and helpful comments. The Insect Diversity component of the International Cooperative Biodiversity Group (ICBG) project in Panama, principal investigators; P. Coley and T. Kursar (U. of Utah) provided funding for invertebrate inventories, while the Smithsonian Tropical Research Institute provided crucial logistical support. Specimen collections in Panama were made under permits issued by the Panamanian Autoridad Nacional del Ambiente (ANAM) over 20 years—the latest of which was permit No. SE/A-89-10. Ecuadorean collections were authorized by the Ministerio del Ambiente under permit N° 004-IC-FL-PNY-RSO and exported under permit N° 012-EXP-IC-FA-RSO-MA. We are grateful to these ministries and their continued support of basic research.
1 Contribution to the 8th International Symposium on the Chrysomelidae, held August 23, 2012, in Daegu, South Korea