(C) 2011 Lixia Xie. This is an open access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC-BY), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
For reference, use of the paginated PDF or printed version of this article is recommended.
A new species Damaeus (Paradamaeus) yushuensis sp. n. is described from Three Rivers’ Headwaters National Natural Reserve, Qinghai province, China. This is the first record of this subgenus in China. A key is given to distinguish all species of the genus.
Oribatida, Damaeus, Paradamaeus, new species, new record
We firstly recorded the species of Paradamaeus from Three Rivers’ Headwaters National Natural Reserve, Qinghai, China, which represented as new species, Damaeus (Paradamaeus) yushuensis. A key to the identification of all known species of Damaeus (Paradamaeus) is given.
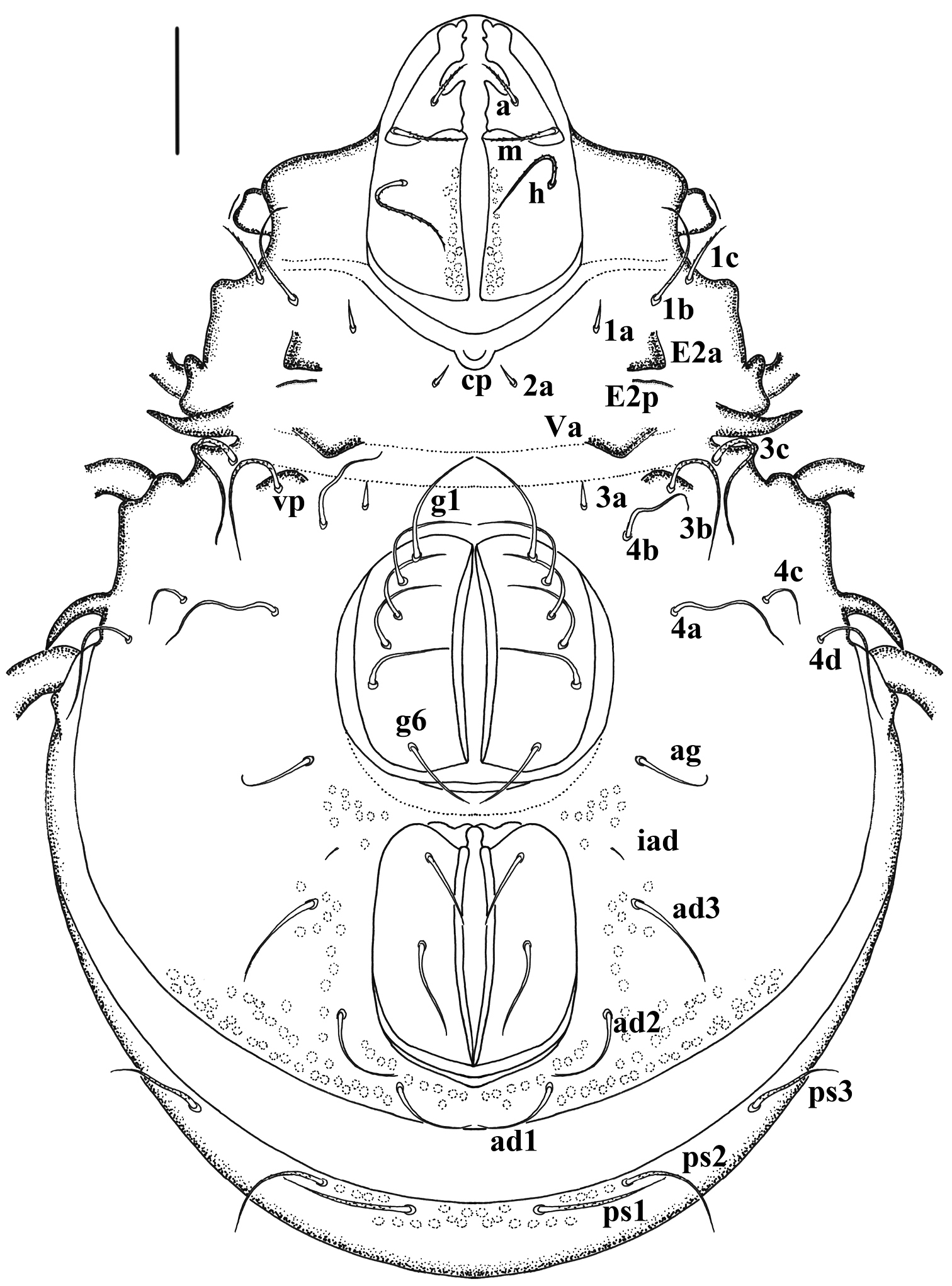
Material and methodsMeasurements and descriptions are based on specimens mounted in temporary cavity slides that were studied using a standard light microscope equipped with a drawing tube. In figures we used the following abbreviations: prodorsal and sejugal apophyses (Ba, Bp), lateral apophyses (Sa, Sp) and coxisternal apophyses (E2a, E2p, Va, Vp), spina adnatum (s.a.), discidium (di), prodorsal setae (ro, le, in, ex), bothridium (bo), sensillus (ss), notogastral or gastronotal setae (c-, l-, h-, p- series), adanal and anal setae (ad-, an- series), aggenital setae (ag), coxisternal setae (1a, 1b, 1c, 2a, 3a, 3b, 3c, 4a, 4b, 4c, 4d), opisthonotal gland opening (gla), lyrifissures or cupules (ia, im, ip, ian, iad, ips, ih).
Terminology generally follows
China, Germany, Ireland, Southern Mediterranean, Faroe Islands, Norway, Latvia, Caucasus, Crimea, Ukraina, Czechoslovakia, Czech Republic, Belgium, Sweden, England, America, Finland, Azores Islands, France, Austria, Netherlands.
| 1 | With dorsosejugal tubercles Da, Dp; setae in short and thin; notogastral setae similar to one another | Damaeus (Paradamaeus) clavipes |
| – | Without dorsosejugal tubercles Da, Dp; setae in long and thick; notogastral setae not similar to one another | Damaeus (Paradamaeus) yushuensis sp. n. (Figs 1–6) |
urn:lsid:zoobank.org:act:9CA2E39F-5E98-43B2-87D5-DDD3F4119690
http://species-id.net/wiki/Damaeus_yushuensis
Figs 1 –6Holotype: male (in alcohol, QHYS-XLX-8-5), China, Three Rivers' Headwaters Natural Reserve Area of Yushu Tibetan Autonomous Prefecture, Qinghai province (32°33'48.65"N , 97°39'55.66"E), from soil under the Picea crassifolia, 3464M, 5 Aug., 2009, col. Lixia Xie. Paratypes: Three females (in alcohol, QHYS-XLX-8-5), same data as holotype; Two males (in alcohol, QHYS-XLX-8-6), same data as QHYS-XLX-8-5, from soil under the Kobresia pygmaea.
Propodolateral apophysis P distinct, with broader base and arched tip; setae ro slightly barbed, setae le heavily barbed, thick. Sensillus short, thick, heavily barbed and rod-like. Interlamellar setae long, rather thick and conspicuously barbed. Prodorsal tubercles Da, Dp absent, Ba well developed, Bp weakly developed. Spinae adnatae beak like, short, distinct, strongly curved inwards (about 30 µm in total), with broader base and quite sharp tip. Notogastral setae smooth, slender except c- series and p- series. Setae of c- series rather thick, with conspicuously barbed and frizzled tip, oriented forwards and the rest backwards.
Comparative length of notogastral setae: lp< lm= ps3< ps2< la= h3= h2= h1< ps1< c1<c2. Epimeral setae mostly smooth except 1b, 1c, 3b, 3c, 4d and hypostomal setae a, m, h. Seta 1a, 2a and 3a rather short. Epimeral setal formula: 3-1-3-4. Enantiophyses E2 and V present, E2p and Vp weakly developed, E2a and Va well developed. Parastigmatic tubercle Sa long, acuminate, with sharp tip; Sp small, triangular. Hypostomal setae a, h and m thin, slightly barbed. Legs rod-like and longer than body.
Damaeus (Paradamaeus) yushuensis sp. n.– dorsal view (100μm)
Damaeus (Paradamaeus) yushuensis sp. n. – Ventral view (100μm)
Damaeus (Paradamaeus) yushuensis sp. n. – lateral view (100μm)
Dimensions. Holotype: Body length 980; length of prodorsum: 420, width 280, height 370, setae: ss 175, in 100, le 150, ro 125, ex 75, distance between setae: ro-ro 110, in-in 125, le-le 125, in-le 105, le-ro 40; length of notogaster: 700, width 670, height 780; setae: c1 135, c2 175, la 110, lm 95, lp 90, h3 110, h2 110, h1 110, ps1 125, ps2 105, ps3 95; c1-c2 75, c2-c2 275, la-la 375, lm-lm 435; ventral region: genito-aggenital plate 225×220, ano-adanal plate 190×175. Paratypes: length of prodorsum: 415-425, width 275-285, height 365-375; length of notogaster: 695-705, width 665-670, height 775-780; ventral region: genito-aggenital plate 220×215, ano-adanal plate 185×170.
Integument. Surface of body and leg segments with filamentous cerotegument. Conspicuous microtubercles present on prodorsum and around leg acetabula, legs with dense fungal mycelic.
Prodorsum (Fig. 1). Triangular, propodolateral apophysis P distinct, with broader base and arched tip. Lamellar setae (le) and rostral setae (ro) both in dorsalaterad position, long, arched and tapered. Lamellar setae with obvious barbs, thick, being longer than slender, weakly barbed rostral pair. Bothridia well developed, funnel-like, with broad margin and pair of thick, heavily barbed sensillus. Interlamellar setae (in) long, thick and conspicuously barbed (specially in holotype). Exobothridial setae (ex) thick, with obvious barbs, frizzled. Comparative length of prodorsal setae: ex< in< ro< le< ss. Weakly developed transverseridge connected to the base of bothridium and directed to median end of prodorsum. Prodorsal tubercles Da absent, Ba distinct; Bp weakly developed, usually as tuberculate sclerotised ridge, in light microscope sometimes discernible only in lateral view.
Notogaster (Fig. 1). Circular viewed perpendicular to circumgastric scissure, length almost equivalent to wider. Spinae adnatae beak like, short, distinct, strongly curved inwards (about 30 µm in length), with broader base and quite sharp tip. Notogastral setae short, smooth, slender except c- series and p- series. Setae of c- series rather thick and long, with conspicuous barbed and frizzled tip, oriented forwards and the rest backwards. Comparative length of notogastral setae: lp< lm= ps3< ps2< la= h3= h2= h1< ps1< c1< c2. Pseudanal setae comparatively long, with obvious barbs, attenuate.
Ventral region. (Fig. 2). Epimere I with medial pit (cp). Epimeral setae mostly smooth except 1b, 1c, 3b, 3c, 4d; setae 1c, 3b, 3c, 4d long, with obvious barbs; Seta 1a, 2a and 3a rather short, lanciform. Epimeral setal formula: 3–1–3–4. Ano-genital setal formula: 6–1–2–3. Enantiophyses E2 and V present, E2a triangular with pointed tip; E2p weakly developed, usually as tuberculate sclerotised ridge. Ventrosejugal tubercle Va large, strong, represented by broad ridge; Vp represented by low, broadly curved ridge, with setae 3b Parastigmatic tubercle Sa long, acuminate, with broader base and heavily pointed tip; Sp small, triangular; Discidium (di) long, acuminate, with broader base and heavily pointed tip, directed posterolaterad.
Gnathosoma. Infracapitular mentum without noticeable microtubercles. Hypostomal setae a, m, h slender, weakly barbed. Chelicera rather strong, fixed and movable digits with four blunt teeth; setae cha with obvious barbsand chb smooth. Palpal setation: 0–2–1–3–8, including solenidion ω (Fig. 4M).
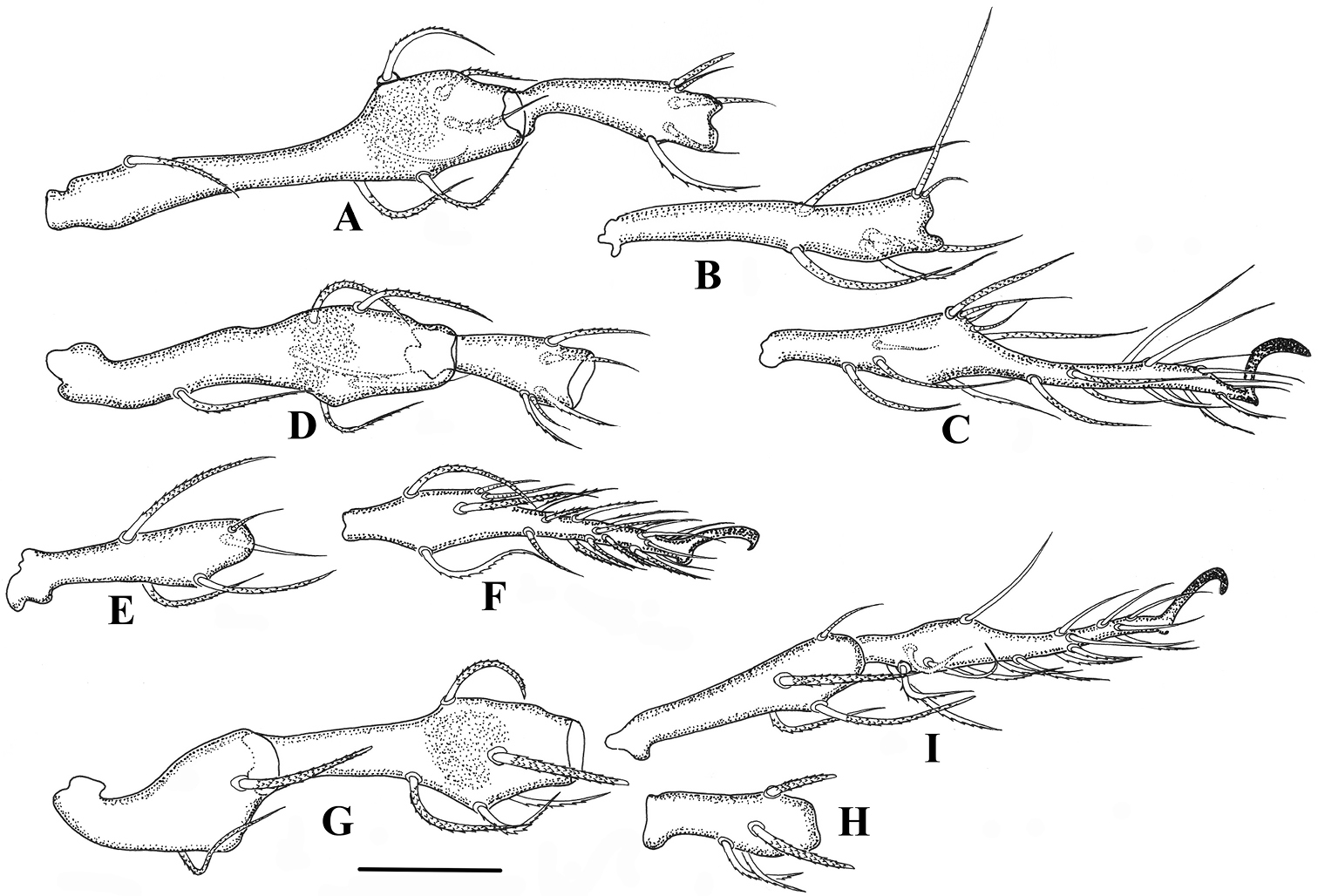
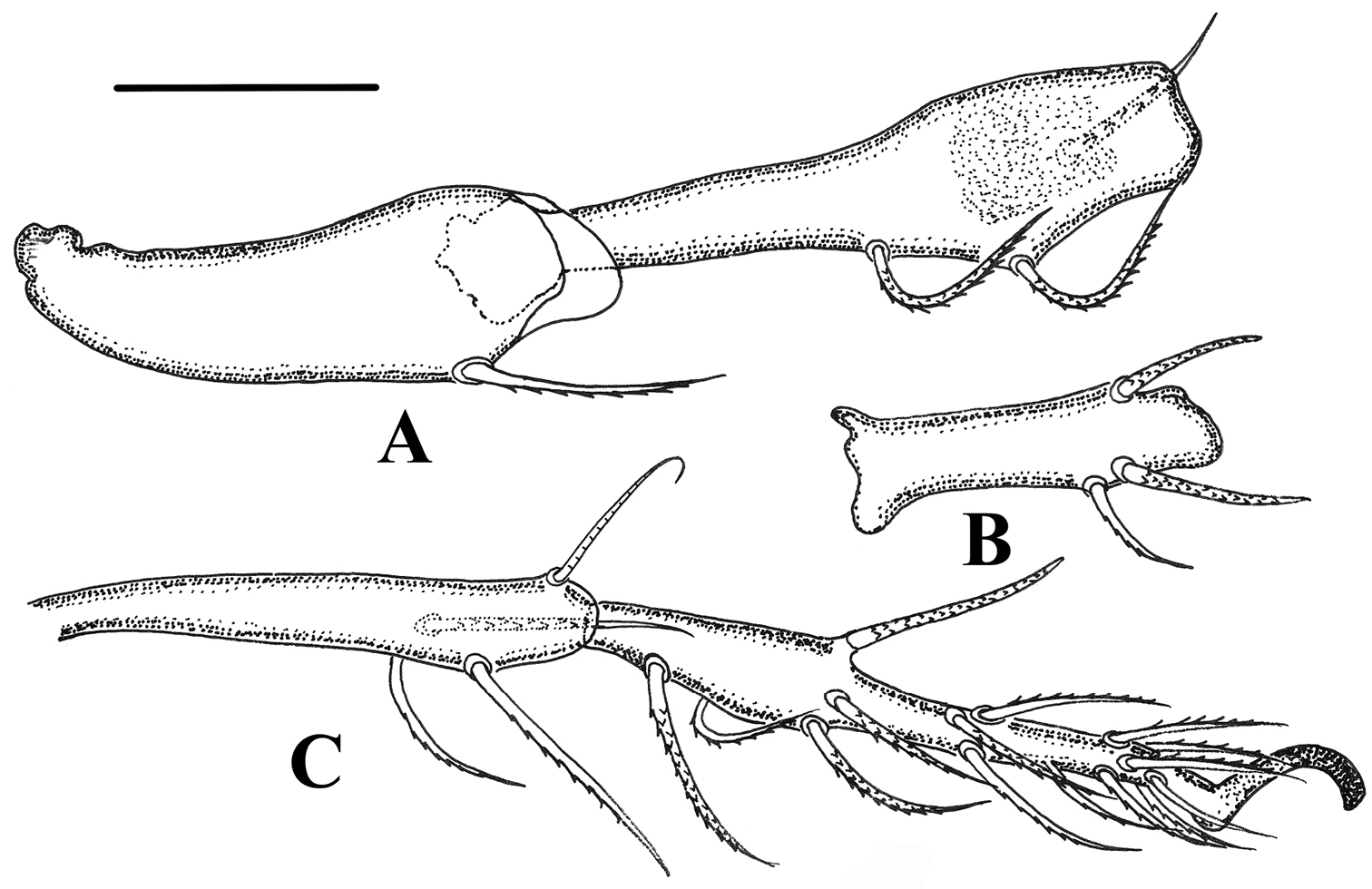
Legs. (Figs 5–6). Monodactylous, moderately long, leg I, III, IV longer than body, leg II shorter than body. Relative length of femur to tarsus of legs I to IV 1: 0.88: 1.01: 1.2. Leg IV 1.3 times ventral body length. Femur IV 1.4 times length of trochanter IV, proximal stalk 1.4 times length of bulb. Leg setae medium in length and thick, mostly with distinct short barbs on outer curvature. Setal formulas of legs as follows (from trochanter to tarsus, famulus and solenidia included): I: 1–7–4 (1)–4 (2)–20 (2); II: 1–6-4 (1)–4 (1)–18 (2); III: 2–4-3 (1)–3 (1)–16 (0); IV: 1–4-3 (1)–3 (1)–15 (0). Solenidia of genua I-II with companion seta d. Solenidia δ equivalent to seta d on genuaI, Solenidia δ shorter and thinner than seta d on genuaII. Solenidion φ1 on tibia I 3 times longer than φ2. Seta d absent from all tibiae, solenidia on all tibiae free, as usual for genus.
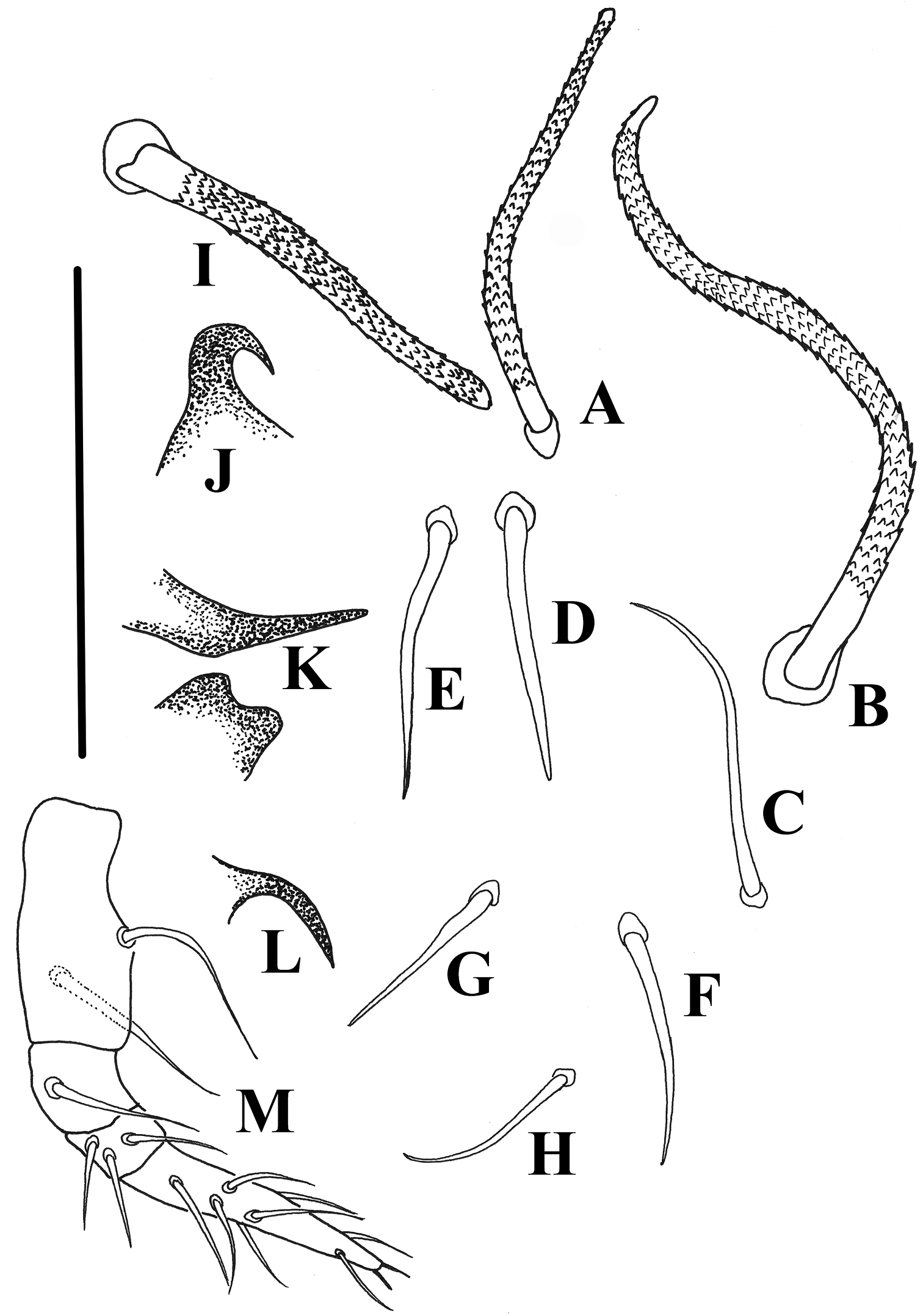
Damaeus (Paradamaeus) yushuensis sp. n. A–H notogastral setae (100μm) I interlamellar seta(100μm) J s.a.–spina adnata(100μm) K Sa –anterior sejugal apophysis(100μm), Sp –posterior sejugal apophysis(100μm) L di –discidium(100μm) M Pe. –pedipalp(100μm).
Damaeus (Paradamaeus) yushuensis sp. n. A femur, genu I (100μm) B tibia I (100μm) C tarsus I (100μm) D femur, genu II (100μm) E Tibia II (100μm) F tarsus II (100μm) G trochanter, femur III (100μm) H genu III (100μm) I tibia, tarsus III (100μm).
Damaeus (Paradamaeus) yushuensis sp. n. A trochanter, femur IV (100μm) B genu IV (100μm) C tibia, tarsus IV (100μm).
The specific name is derived from the type locality, Yushu Tibetan Autonomous Prefecture, Qinghai province.
This new species is characterised by following characters: interlamellar setae (in) long, rather thick and conspicuously barbed; spinae adnatae beak like, short, distinct, strongly curved inwards; c-series rather thick, long with conspicuous barbed and frizzled tip, oriented forwards and the rest backwards, other notogastral setae smooth, slender and short, except p- series (see Table 1).
Comparison of two species belonging to the subgenus Paradamaeus
| Morphological character | Damaeus (Paradamaeus) clavipes | Damaeus (Paradamaeus) yushuensis sp. n. |
|---|---|---|
| propodolateral apophysis P | with angular tip | with arched tip |
| Interlamellar setae (in) | short, thin | long, thick |
| Spinae adnatae (s.a.) | slender, medium long, triangular with sharp tip | beak like, short, quite sharp tip |
| Notogastral setae c1, c2 | similar in length, slender | unequal length, thick |
| Number of setae on Femora III–IV | 5 | 4 |
| Prodorsal tubercles Da, Dp | present | absent |
| Parastigmatic tubercle Sa | small, indistinct | large, acuminate, with broader base and heavily pointed tip |
The authors are very grateful to Prof. Roy A. Norton (College of Environmental Science and Forestry, State University of New York, Syracuse) for his encouragement to our research and kind help. The authors are also grateful to Dr. Owen Seeman (Queensland Museum, South Brisbane, Australia) and Dr. Sergey G. Ermilov (Laboratory of Entomology, Center of Independent Examinations, Nizhniy Novgorod, Russia) for their help. We are very grateful to Qiongzhang Song, Fangling Xu, Zaihua Yang, Bin Li (Institute of Entomology, Guizhou University, Guiyang, Guizhou province, China) for making it possible to work by their collections.